Abstract
A new methodology was developed to study the cell-surface glycoproteins of cultured human skin fibroblasts. This was based on the binding of a variety of biotinyl-lectins to nitrocellulose electrophoretic transfers of total fibroblast lysates after separation in sodium dodecyl sulphate/polyacrylamide gels, followed by reaction with avidin-biotinyl-peroxidase complexes and detection with 3,3'-diaminobenzidine. The technique proved to be very sensitive and a large number of glycoproteins were detected by binding of concanavalin A and wheat-germ agglutinin. Binding of peanut agglutinin and to a lesser extent of Ricinus communis agglutinin I were found to be dependent on prior removal of sialic acid residues from the glycoproteins. Since by treatment of intact viable cells with neuraminidase only external sialic acid residues were removed, peanut agglutinin and Ricinus communis agglutinin I could thus be utilized for selective detection of cell-surface glycoproteins. Also, because peanut agglutinin was known to bind preferentially to oligosaccharides of the O-glycosidic type, and Ricinus communis agglutinin I to those of the N-glycosidic type, the two lectins were complementary in displaying the surface glycoproteins and in providing information about their oligosaccharide composition.
Full text
PDF



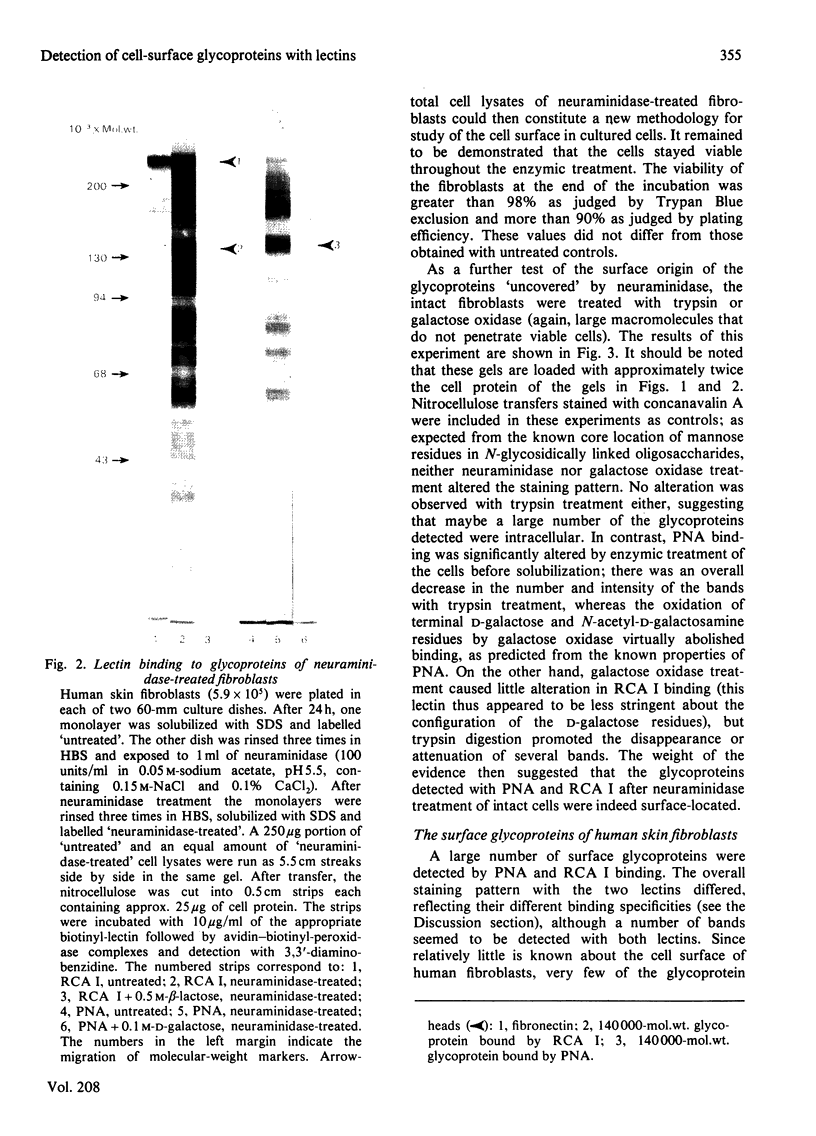
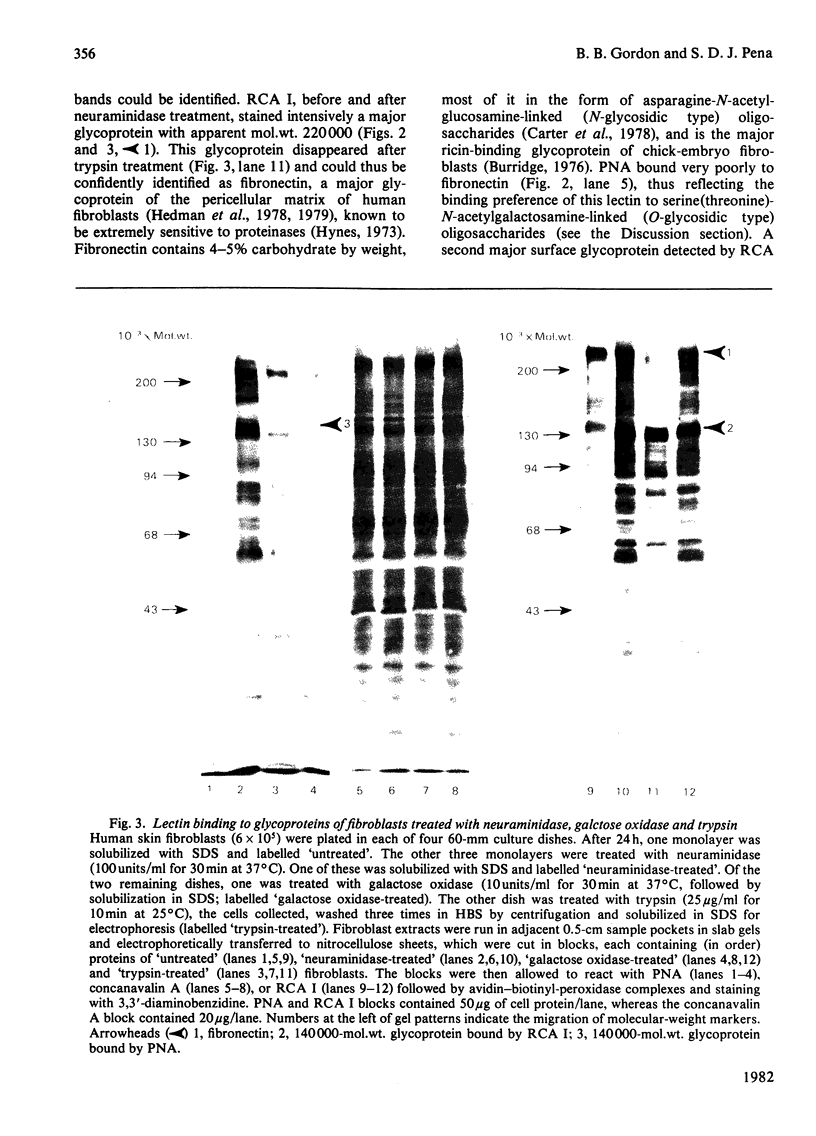


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Burger M. M., Martin G. S. Agglutination of cells transformed by Rous sarcoma virus by wheat germ agglutinin and concanavalin A. Nat New Biol. 1972 May 3;237(70):9–12. doi: 10.1038/newbio237009a0. [DOI] [PubMed] [Google Scholar]
- Burridge K. Changes in cellular glycoproteins after transformation: identification of specific glycoproteins and antigens in sodium dodecyl sulfate gels. Proc Natl Acad Sci U S A. 1976 Dec;73(12):4457–4461. doi: 10.1073/pnas.73.12.4457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butters T. D., Hughes R. C. Lectin binding to mosquito Aedes aegyptii and human KB cells: structural comparisons of membrane oligosaccharides. Carbohydr Res. 1978 Mar;61:159–168. doi: 10.1016/s0008-6215(00)84476-6. [DOI] [PubMed] [Google Scholar]
- Carraway K. L. Covalent labeling of membranes. Biochim Biophys Acta. 1975 Dec 29;415(4):379–410. doi: 10.1016/0304-4157(75)90005-2. [DOI] [PubMed] [Google Scholar]
- Carter W. G., Fukuda M., Lingwood C., Hakomori S. Chemical composition, gross structure, and organization of transformation-sensitive glycoproteins. Ann N Y Acad Sci. 1978 Jun 20;312:160–177. doi: 10.1111/j.1749-6632.1978.tb16801.x. [DOI] [PubMed] [Google Scholar]
- Carter W. G., Hakomori S. A new cell surface, detergent-insoluble glycoprotein matrix of human and hamster fibroblasts. The role of disulfide bonds in stabilization of the matrix. J Biol Chem. 1981 Jul 10;256(13):6953–6960. [PubMed] [Google Scholar]
- Critchley D. R. Cell surface proteins of NIL1 hamster fibroblasts labeled by a galactose oxidase, tritiated borohydride method. Cell. 1974 Oct;3(2):121–125. doi: 10.1016/0092-8674(74)90115-9. [DOI] [PubMed] [Google Scholar]
- Gahmberg C. G., Kiehn D., Hakomori S. Changes in a surface-labelled galactoprotein and in glycolipid concentrations in cells transformed by a temperature-sensitive polyoma virus mutant. Nature. 1974 Mar 29;248(447):413–415. doi: 10.1038/248413a0. [DOI] [PubMed] [Google Scholar]
- Goldstein I. J., Hayes C. E. The lectins: carbohydrate-binding proteins of plants and animals. Adv Carbohydr Chem Biochem. 1978;35:127–340. doi: 10.1016/s0065-2318(08)60220-6. [DOI] [PubMed] [Google Scholar]
- Gonatas N. K., Avrameas S. Detection of plasma membrane carbohydrates with lectin peroxidase conjugates. J Cell Biol. 1973 Nov;59(2 Pt 1):436–443. doi: 10.1083/jcb.59.2.436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham J. M., Hynes R. O., Davidson E. A., Bainton D. F. The location of proteins labeled by the 125I-lactoperoxidase system in the NIL 8 hamster fibroblast. Cell. 1975 Apr;4(4):353–365. doi: 10.1016/0092-8674(75)90156-7. [DOI] [PubMed] [Google Scholar]
- Graham R. C., Jr, Karnovsky M. J. The early stages of absorption of injected horseradish peroxidase in the proximal tubules of mouse kidney: ultrastructural cytochemistry by a new technique. J Histochem Cytochem. 1966 Apr;14(4):291–302. doi: 10.1177/14.4.291. [DOI] [PubMed] [Google Scholar]
- Guesdon J. L., Ternynck T., Avrameas S. The use of avidin-biotin interaction in immunoenzymatic techniques. J Histochem Cytochem. 1979 Aug;27(8):1131–1139. doi: 10.1177/27.8.90074. [DOI] [PubMed] [Google Scholar]
- HANKS J. H. The longevity of chick tissue cultures without renewal of medium. J Cell Physiol. 1948 Apr;31(2):235–260. doi: 10.1002/jcp.1030310209. [DOI] [PubMed] [Google Scholar]
- Hedman K., Kurkinen M., Alitalo K., Vaheri A., Johansson S., Hök M. Isolation of the pericellular matrix of human fibroblast cultures. J Cell Biol. 1979 Apr;81(1):83–91. doi: 10.1083/jcb.81.1.83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedman K., Vaheri A., Wartiovaara J. External fibronectin of cultured human fibroblasts is predominantly a matrix protein. J Cell Biol. 1978 Mar;76(3):748–760. doi: 10.1083/jcb.76.3.748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heggeness M. H., Ash J. F. Use of the avidin-biotin complex for the localization of actin and myosin with fluorescence microscopy. J Cell Biol. 1977 Jun;73(3):783–788. doi: 10.1083/jcb.73.3.783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hsu S. M., Raine L., Fanger H. Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem. 1981 Apr;29(4):577–580. doi: 10.1177/29.4.6166661. [DOI] [PubMed] [Google Scholar]
- Huang C. C., Aminoff D. Enzymes that destroy blood group specificity. V. The oligosaccharase of Clostridium perfringens. J Biol Chem. 1972 Nov 10;247(21):6737–6742. [PubMed] [Google Scholar]
- Hubbard A. L., Cohn Z. A. The enzymatic iodination of the red cell membrane. J Cell Biol. 1972 Nov;55(2):390–405. doi: 10.1083/jcb.55.2.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubbard B. D., Lazarides E. Copurification of actin and desmin from chicken smooth muscle and their copolymerization in vitro to intermediate filaments. J Cell Biol. 1979 Jan;80(1):166–182. doi: 10.1083/jcb.80.1.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hynes R. O. Alteration of cell-surface proteins by viral transformation and by proteolysis. Proc Natl Acad Sci U S A. 1973 Nov;70(11):3170–3174. doi: 10.1073/pnas.70.11.3170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irimura T., Kawaguchi T., Terao T., Osawa T. Carbohydrate-binding specificity of the so-called galactose-specific phytohemagglutinins. Carbohydr Res. 1975 Feb;39(2):317–327. doi: 10.1016/s0008-6215(00)86141-8. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Lotan R., Nicolson G. L. Purification of cell membrane glycoproteins by lectin affinity chromatography. Biochim Biophys Acta. 1979 Dec 20;559(4):329–376. doi: 10.1016/0304-4157(79)90010-8. [DOI] [PubMed] [Google Scholar]
- Lotan R., Skutelsky E., Danon D., Sharon N. The purification, composition, and specificity of the anti-T lectin from peanut (Arachis hypogaea). J Biol Chem. 1975 Nov 10;250(21):8518–8523. [PubMed] [Google Scholar]
- Muramatsu T., Koide N., Ceccarini C., Atkinson P. H. Characterization of mannose-labeled glycopeptides from human diploid cells and their growth-dependent alterations. J Biol Chem. 1976 Aug 10;251(15):4673–4679. [PubMed] [Google Scholar]
- Nicolson G. L., Blaustein J., Etzler M. E. Characterization of two plant lectins from Ricinus communis and their quantitative interaction with a murine lymphoma. Biochemistry. 1974 Jan 1;13(1):196–204. doi: 10.1021/bi00698a029. [DOI] [PubMed] [Google Scholar]
- Nicolson G. L., Singer S. J. Ferritin-conjugated plant agglutinins as specific saccharide stains for electron microscopy: application to saccharides bound to cell membranes. Proc Natl Acad Sci U S A. 1971 May;68(5):942–945. doi: 10.1073/pnas.68.5.942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pena S. D. Giant axonal neuropathy: an inborn error of organization of intermediate filaments. Muscle Nerve. 1982 Feb;5(2):166–172. doi: 10.1002/mus.880050215. [DOI] [PubMed] [Google Scholar]
- Pena S. D., Gordon B. B., Karpati G., Carpenter S. Lectin histochemistry of human skeletal muscle. J Histochem Cytochem. 1981 Apr;29(4):542–546. doi: 10.1177/29.4.6166659. [DOI] [PubMed] [Google Scholar]
- Pena S. D., Mills G., Hughes R. C. Two-dimensional electrophoresis of surface glycoproteins of normal BHK cells and ricin resistant mutants. Biochim Biophys Acta. 1979 Jan 5;550(1):100–109. doi: 10.1016/0005-2736(79)90118-4. [DOI] [PubMed] [Google Scholar]
- Phillips D. R., Morrison M. Exposed protein on the intact human erythrocyte. Biochemistry. 1971 May 11;10(10):1766–1771. doi: 10.1021/bi00786a006. [DOI] [PubMed] [Google Scholar]
- Spiro R. G. Glycoproteins. Adv Protein Chem. 1973;27:349–467. doi: 10.1016/s0065-3233(08)60451-9. [DOI] [PubMed] [Google Scholar]
- Steck T. L., Dawson G. Topographical distribution of complex carbohydrates in the erythrocyte membrane. J Biol Chem. 1974 Apr 10;249(7):2135–2142. [PubMed] [Google Scholar]
- Tanner M. J., Anstee D. J. A method for the direct demonstration of the lectin-binding components of the human erythrocyte membrane. Biochem J. 1976 Feb 1;153(2):265–270. doi: 10.1042/bj1530265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Verlinden J., Van Leuven F., Cassiman J. J., Van den Berghe H. Immunochemical characterization of a 150 000 dalton human fibroblast surface glycoprotein. Biochim Biophys Acta. 1981 Jan 30;667(1):1–14. doi: 10.1016/0005-2795(81)90061-1. [DOI] [PubMed] [Google Scholar]
- Wallach D. F., Lin P. S. A critical evaluation of plasma membrane fractionation. Biochim Biophys Acta. 1973 Nov;300(3):211–254. doi: 10.1016/0304-4157(73)90005-1. [DOI] [PubMed] [Google Scholar]