Abstract
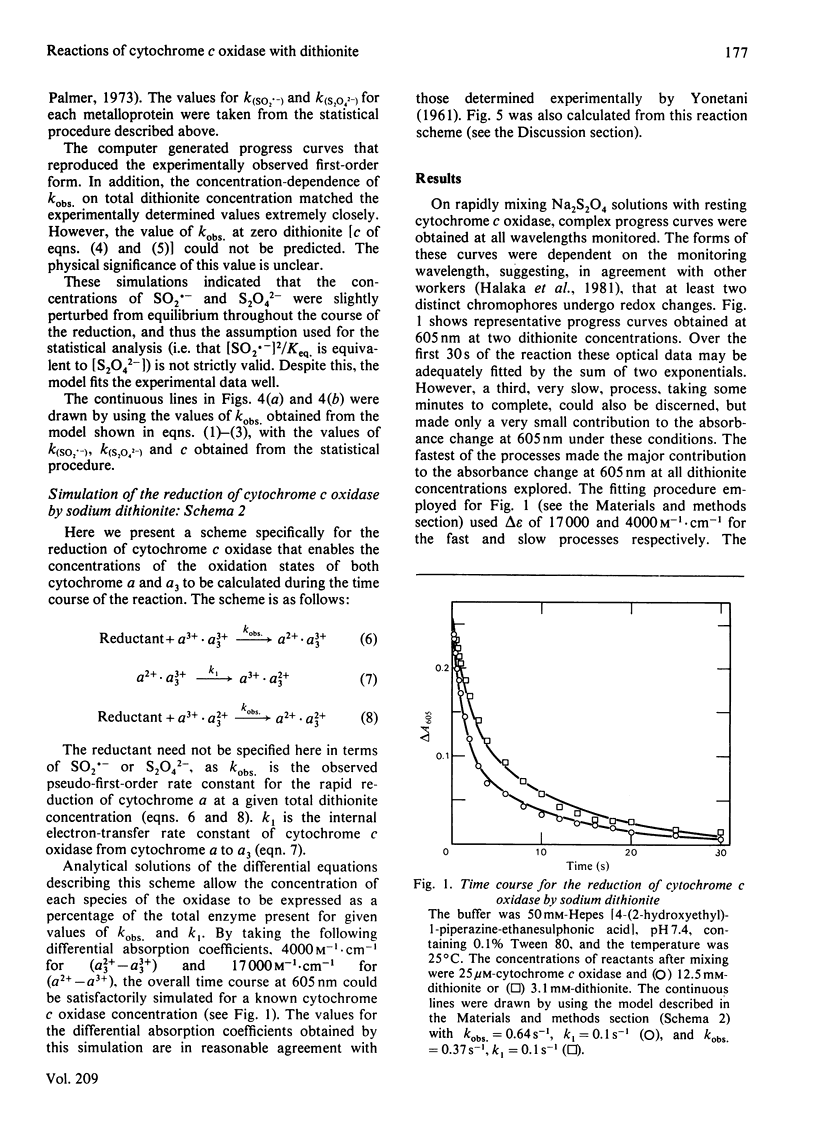
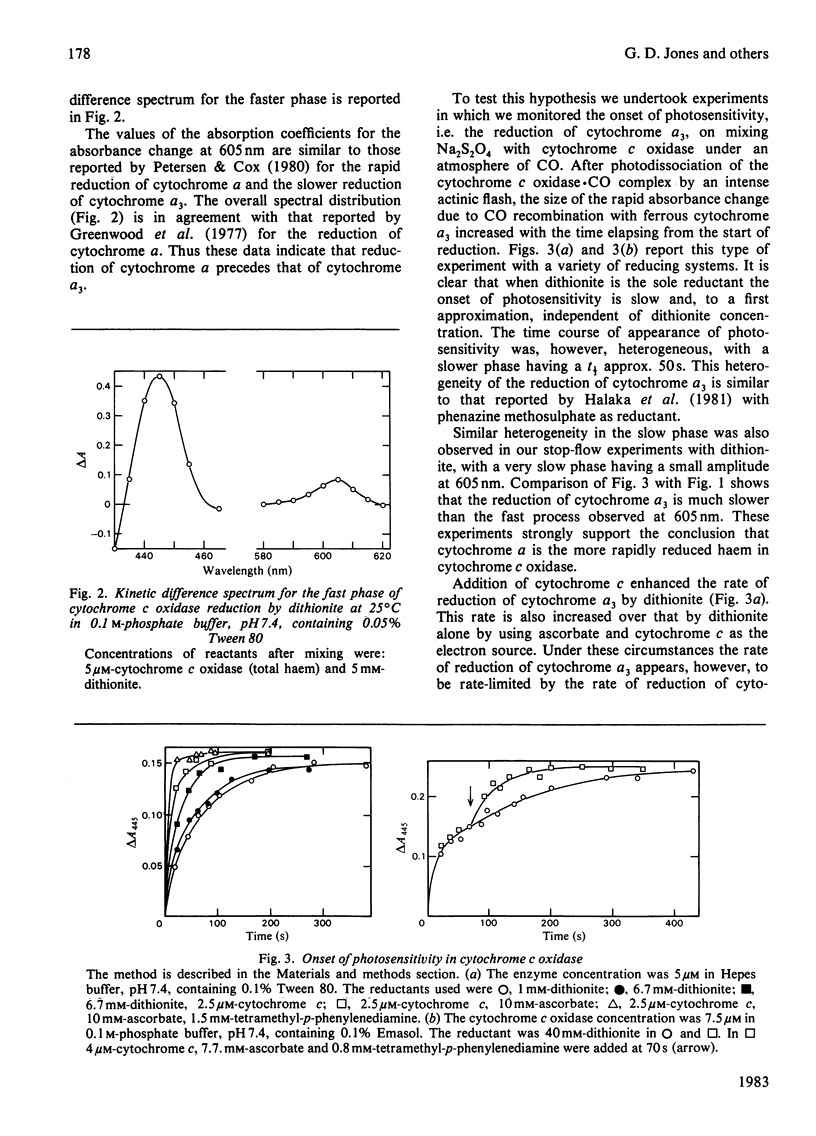
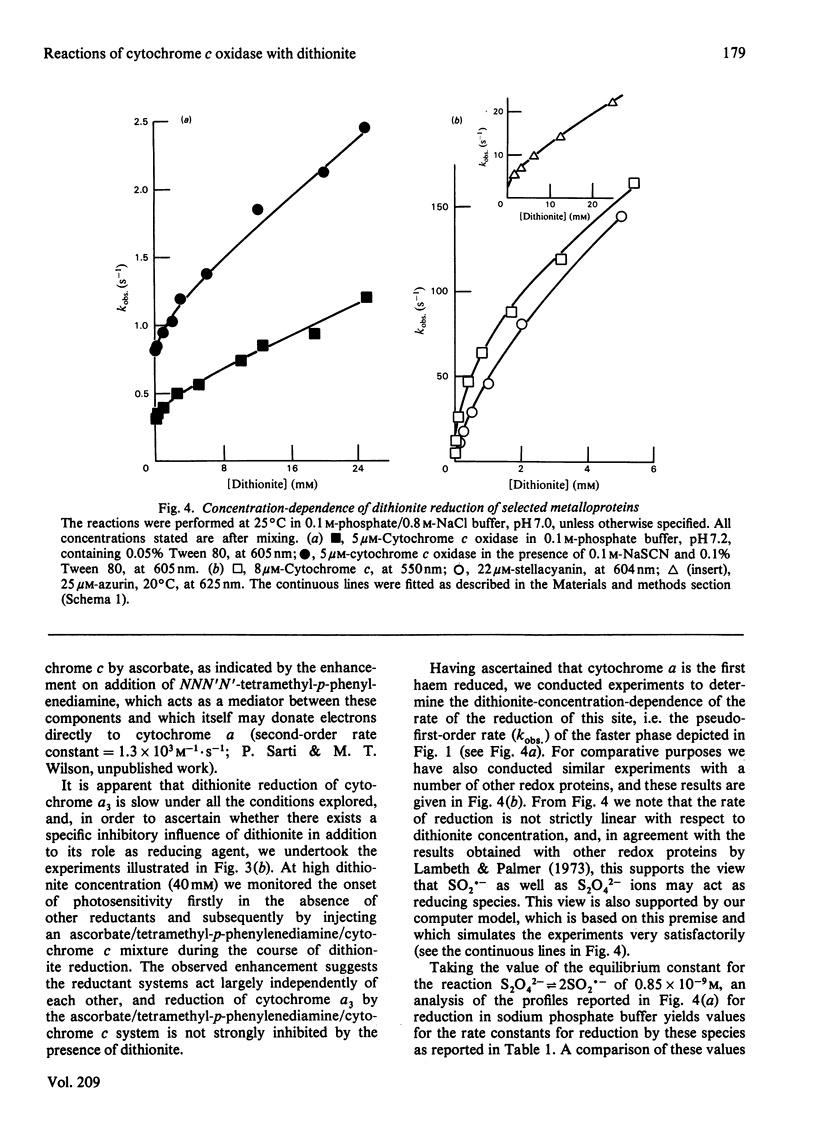
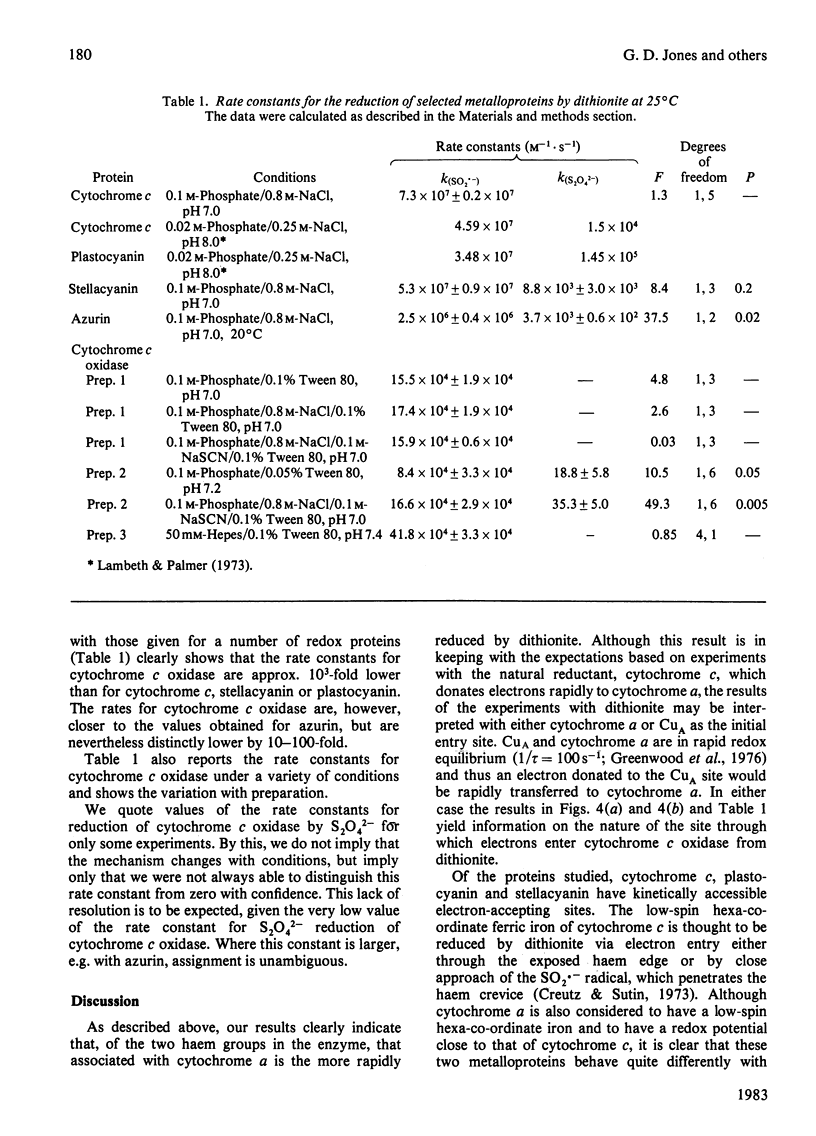
The reduction of cytochrome c oxidase (EC 1.9.3.1) by dithionite was investigated by stopped-flow spectrophotometry and flow-flash techniques in the presence of CO. Of the two haem groups present in the enzyme, that associated with cytochrome alpha is the first reduced. The second-order rate constants for reduction of a number of redox proteins (cytochrome c, stellacyanin and azurin) by the S2O4(2-) and SO2.- anions are reported, and the values are compared with those determined for cytochrome c oxidase. These results are discussed in terms of the accessibility and charge distribution of the electron-entry site of cytochrome c oxidase.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Al-Ayash A. I., Wilson M. T. The mechanism of reduction of single-site redox proteins by ascorbic acid. Biochem J. 1979 Feb 1;177(2):641–648. doi: 10.1042/bj1770641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brudvig G. W., Stevens T. H., Morse R. H., Chan S. I. Conformations of oxidized cytochrome c oxidase. Biochemistry. 1981 Jun 23;20(13):3912–3921. doi: 10.1021/bi00516a039. [DOI] [PubMed] [Google Scholar]
- Creutz C., Sutin N. Reduction of ferricytochrome c by dithionite ion: electron transfer by parallel adjacent and remote pathways. Proc Natl Acad Sci U S A. 1973 Jun;70(6):1701–1703. doi: 10.1073/pnas.70.6.1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cummins D., Gray H. B. Electron-transfer protein reactivities. Kinetic studies of the oxidation of horse heart cytochrome c, Chromatium vinosum high potential iron-sulfur protein, Pseudomonas aeruginosa azurin, bean plastocyanin, and Rhus vernicifera stellacyanin by pentaamminepyridineruthenium(III). J Am Chem Soc. 1977 Jul 20;99(15):5158–5167. doi: 10.1021/ja00457a042. [DOI] [PubMed] [Google Scholar]
- Dawson J. W., Gray H. B., Holwerda R. A., Westhead E. W. Kinetics of the reduction of metalloproteins by chromous ion (laccase-cytochrome c-plastocyanins-temperature-rate constants). Proc Natl Acad Sci U S A. 1972 Jan;69(1):30–33. doi: 10.1073/pnas.69.1.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GIBSON Q. H., GREENWOOD C. THE REACTION OF CYTOCHROME OXIDASE WITH CYTOCHROME C. J Biol Chem. 1965 Feb;240:888–894. [PubMed] [Google Scholar]
- Greenwood C., Brittain T., Brunori M., Wilson M. T. Chromous ion reduction of mammalian cytochrome oxidase and some of its derivatives. Biochem J. 1977 Aug 1;165(2):413–416. doi: 10.1042/bj1650413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenwood C., Brittain T. Studies on partially reduced mammalian cytochrome oxidase reactions with ferrocytochrome c. Biochem J. 1976 Sep 1;157(3):591–598. doi: 10.1042/bj1570591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Halaka F. G., Babcock G. T., Dye J. L. Kinetic distinction between cytochromes a and a3 in cytochrome c oxidase. Rapid scanning stopped flow study of anaerobic reduction by a neutral and a negatively charged donor. J Biol Chem. 1981 Feb 10;256(3):1084–1087. [PubMed] [Google Scholar]
- Koppenol W. H., Vroonland C. A., Braams R. The electric potential field around cytochrome c and the effect of ionic strength on reaction rates of horse cytochrome c. Biochim Biophys Acta. 1978 Sep 7;503(3):499–508. doi: 10.1016/0005-2728(78)90149-4. [DOI] [PubMed] [Google Scholar]
- Lambeth D. O., Palmer G. The kinetics and mechanism of reduction of electron transfer proteins and other compounds of biological interest by dithionite. J Biol Chem. 1973 Sep 10;248(17):6095–6103. [PubMed] [Google Scholar]
- Parr S. R., Barber D., Greenwood C. A purification procedure for the soluble cytochrome oxidase and some other respiratory proteins from Pseudomonas aeruginosa. Biochem J. 1976 Aug 1;157(2):423–430. doi: 10.1042/bj1570423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen L. C., Cox R. P. On the mechanism of the cytochrome c oxidase reaction. Bound Fe(III) cytochrome c as an intermediate in the reductive half reaction. Eur J Biochem. 1980 Apr;105(2):321–327. doi: 10.1111/j.1432-1033.1980.tb04504.x. [DOI] [PubMed] [Google Scholar]
- Reinhammar B. Purification and properties of laccase and stellacyanin from Rhus vernicifera. Biochim Biophys Acta. 1970 Apr 7;205(1):35–47. doi: 10.1016/0005-2728(70)90059-9. [DOI] [PubMed] [Google Scholar]
- Saleem M. M., Wilson M. T. Kinetic studies on the reduction of cytochrome c. Reaction with dihydroxy conjugated compounds (catechols and quinols). Biochem J. 1982 Mar 1;201(3):433–444. doi: 10.1042/bj2010433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sutin N., Yandell J. K. Mechanisms of the reactions of cytochrome c. Rate and equilibrium constants for ligand binding to horse heart ferricytochrome c. J Biol Chem. 1972 Nov 10;247(21):6932–6936. [PubMed] [Google Scholar]
- Van Buuren J. H., Van Gelder B. F., Wilting J., Braams R. Biochemical and biophysical studies on cytochrome C oxidase. XIV. The reaction with cytochrome as studied by pulse radiolysis. Biochim Biophys Acta. 1974 Mar 26;333(3):421–429. doi: 10.1016/0005-2728(74)90126-1. [DOI] [PubMed] [Google Scholar]
- Wilson M. T., Peterson J., Antonini E., Brunori M., Colosimo A., Wyman J. A plausible two-state model for cytochrome c oxidase. Proc Natl Acad Sci U S A. 1981 Nov;78(11):7115–7118. doi: 10.1073/pnas.78.11.7115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- YONETANI T. Studies on cytochrome oxidase. I. Absolute and difference absorption spectra. J Biol Chem. 1960 Mar;235:845–852. [PubMed] [Google Scholar]
- YONETANI T. Studies on cytochrome oxidase. III. Improved preparation and some properties. J Biol Chem. 1961 Jun;236:1680–1688. [PubMed] [Google Scholar]



