Abstract
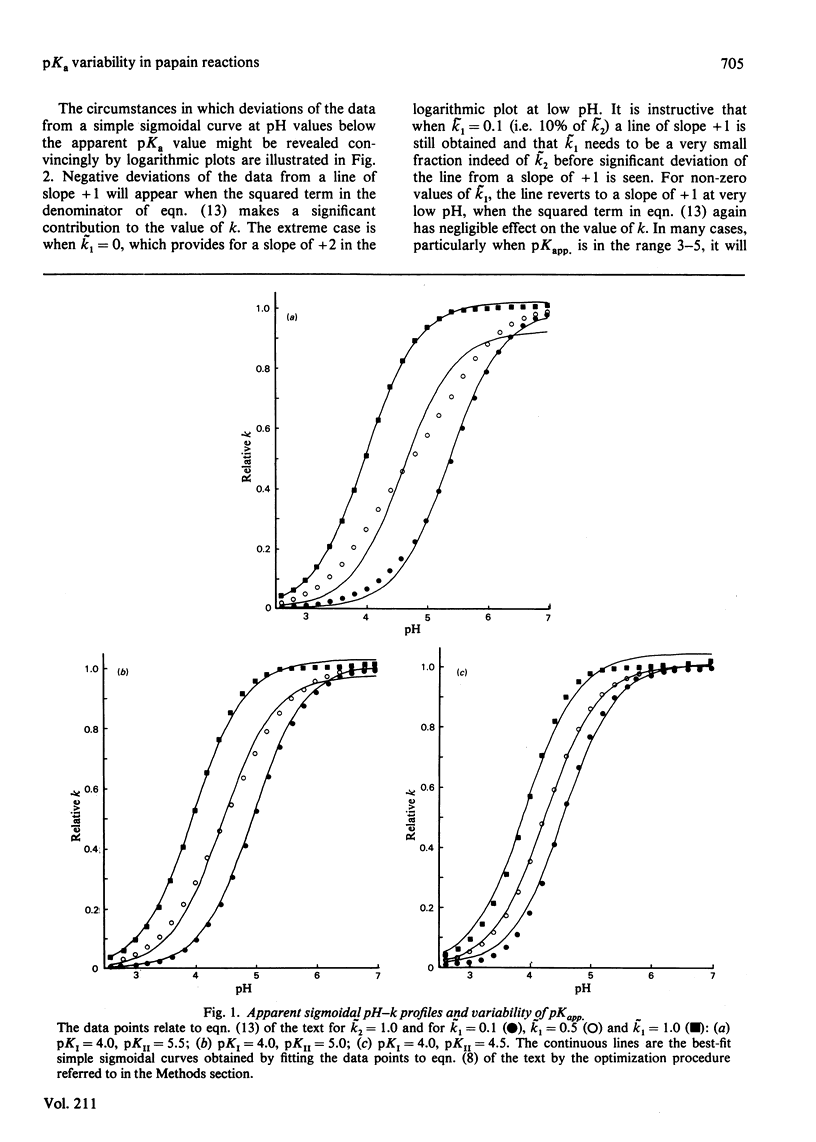
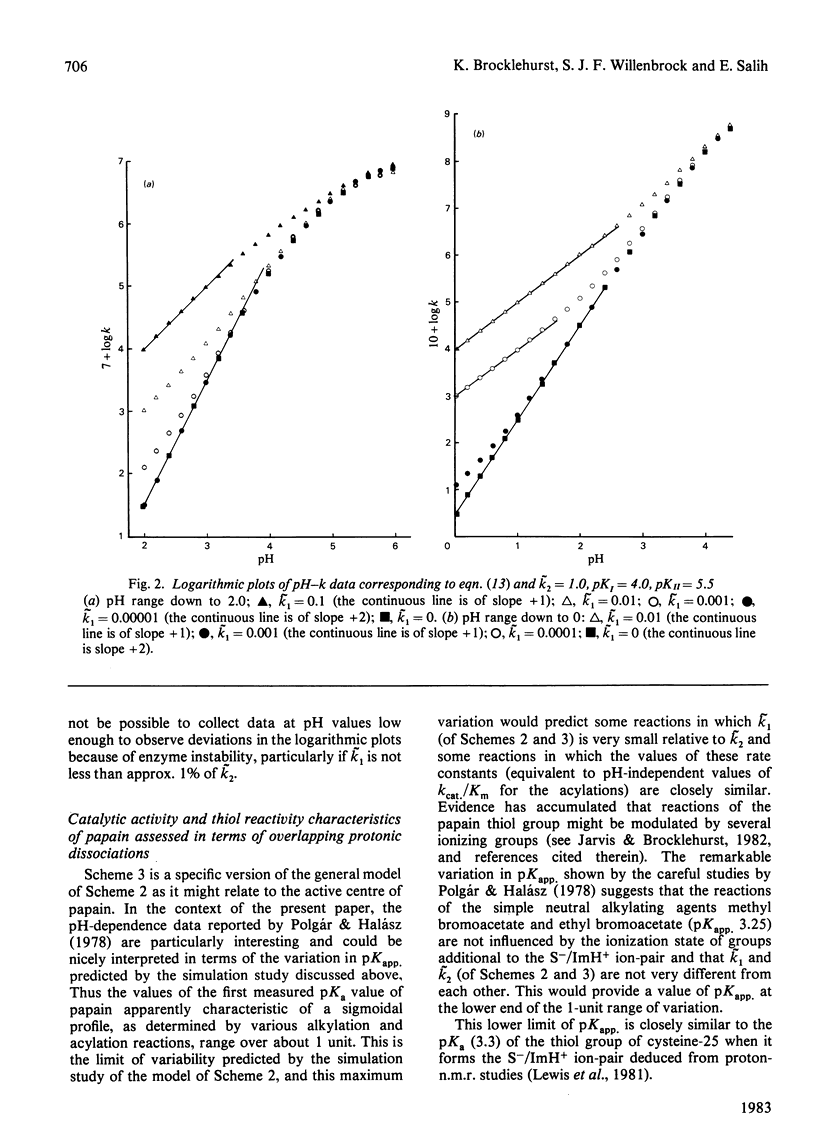
The effects of selection by a small molecule, when binding to a protein, of a particular conformation from an equilibrium stereopopulation on the characteristics of the pH-dependence of reaction with a reactivity probe or substrate were determined by analysis of an appropriate kinetic model. For reaction in one protonic state containing an equilibrium mixture of two conformational isomers, the pH-second-order rate constant (k) profile is of conventional sigmoidal form. The apparent pKa value is a composite of the pKa values of the two conformational states. The value of pKapp. for a given enzyme under given experimental conditions will always be the same (provided that the site-specificity assumed in the model is maintained) irrespective of whether only one conformation reacts or both react, with the same or with different rate constants. The experimentally determined pH-independent rate constant (kapp.) is an average of the reactivities of the two conformational states weighted in favour of the predominant form. The presence of an additional but unreactive conformational state also affects the value of kapp. The possibility that overlapping acid dissociations that affect the reactivity of the enzyme might provide pH-k profiles often indistinguishable in practice from simple sigmoidal dissociation curves and subject to variability in apparent pKa values was evaluated by a simulation study. If two reactive protonic states of the enzyme respond differently to changes in the structure of the substrate or site-specific reactivity probe, differences in apparent pKa values of up to approx. 1 unit can be exhibited without deviation from sigmoidal behaviour being reliably observed. Differences in apparent pKa values observed in some site-specific reactions of papain and their possible consequences for its catalytic mechanism are discussed.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allen K. G., Stewart J. A., Johnson P. E., Wettlaufer D. G. Identification of the functional ionic groups of papain by pH/rate profile analysis. Eur J Biochem. 1978 Jul 3;87(3):575–582. doi: 10.1111/j.1432-1033.1978.tb12409.x. [DOI] [PubMed] [Google Scholar]
- Angelides K. J., Fink A. L. Cryoenzymology of papain: reaction mechanism with an ester substrate. Biochemistry. 1978 Jun 27;17(13):2659–2668. doi: 10.1021/bi00606a032. [DOI] [PubMed] [Google Scholar]
- Angelides K. J., Fink A. L. Mechanism of Action of papain with a specific anilide substrate. Biochemistry. 1979 May 29;18(11):2355–2363. doi: 10.1021/bi00578a034. [DOI] [PubMed] [Google Scholar]
- Angelides K. J., Fink A. L. Mechanism of thiol protease catalysis: detection and stabilization of a tetrahedral intermediate in papain catalysis. Biochemistry. 1979 May 29;18(11):2363–2369. doi: 10.1021/bi00578a035. [DOI] [PubMed] [Google Scholar]
- Baines B. S., Brocklehurst K. Characterization of papaya peptidase A as a cysteine proteinase of Carica papaya L. with active-centre properties that differ from those of papain by using 2,2'-dipyridyl disulphide and 4-chloro-7-nitrobenzofurazan as reactivity probes. Use of two-protonic-state electrophiles in the identification of catalytic-site thiol groups. Biochem J. 1982 Jul 1;205(1):205–211. doi: 10.1042/bj2050205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bendall M. R., Lowe G. Co-operative ionisation of aspartic-acid-158 and histidine-159 in papain. Evidence from 19F nuclear-magnetic-resonance and fluorescence spectroscopy. Eur J Biochem. 1976 Jun 1;65(2):481–491. doi: 10.1111/j.1432-1033.1976.tb10364.x. [DOI] [PubMed] [Google Scholar]
- Brocklehurst K., Dixon H. B. PH-dependence of the steady-state rate of a two-step enzymic reaction. Biochem J. 1976 Apr 1;155(1):61–70. doi: 10.1042/bj1550061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Dixon H. B. The pH-dependence of second-order rate constants of enzyme modification may provide free-reactant pKa values. Biochem J. 1977 Dec 1;167(3):859–862. doi: 10.1042/bj1670859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Herbert J. A., Norris R., Suschitzky H. Evidence for association-activation effects in reactions of papain from studies on its reactivity towards isomeric two-protonic-state reactivity probes. Biochem J. 1979 Nov 1;183(2):369–373. doi: 10.1042/bj1830369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Malthouse J. P. Evidence for a two-state transition in papain that may have no close analogue in ficin. Differences in the disposition of cationic sites and hydrophobic binding areas in the active centres of papain and ficin. Biochem J. 1980 Dec 1;191(3):707–718. doi: 10.1042/bj1910707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Malthouse J. P. Mechanism of the reaction of papain with substrate-derived diazomethyl ketones. Implications for the difference in site specificity of halomethyl ketones for serine proteinases and cysteine proteinases and for stereoelectronic requirements in the papain catalytic mechanism. Biochem J. 1978 Nov 1;175(2):761–764. doi: 10.1042/bj1750761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Malthouse J. P., Shipton M. Evidence that binding to the s2-subsite of papain may be coupled with catalytically relevant structural change involving the cysteine-25-histidine-159 diad. Kinetics of the reaction of papain with a two-protonic-state reactivity probe containing a hydrophobic side chain. Biochem J. 1979 Nov 1;183(2):223–231. doi: 10.1042/bj1830223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K., Mushiri S. M., Patel G., Willenbrock F. Evidence for a close similarity in the catalytic sites of papain and ficin in near-neutral media despite differences in acidic and alkaline media. Kinetics of the reactions of papain and ficin with chloroacetate. Biochem J. 1982 Jan 1;201(1):101–104. doi: 10.1042/bj2010101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brocklehurst K. Two-protonic-state electrophiles as probes of enzyme mechanisms. Methods Enzymol. 1982;87:427–469. doi: 10.1016/s0076-6879(82)87026-2. [DOI] [PubMed] [Google Scholar]
- Chaiken I. M., Smith E. L. Reaction of chloroacetamide with the sulfhydryl group of papain. J Biol Chem. 1969 Oct 10;244(19):5087–5094. [PubMed] [Google Scholar]
- Chaiken I. M., Smith E. L. Reaction of the sulfhydryl group of papain with chloroacetic acid. J Biol Chem. 1969 Oct 10;244(19):5095–5099. [PubMed] [Google Scholar]
- Drenth J., Kalk K. H., Swen H. M. Binding of chloromethyl ketone substrate analogues to crystalline papain. Biochemistry. 1976 Aug 24;15(17):3731–3738. doi: 10.1021/bi00662a014. [DOI] [PubMed] [Google Scholar]
- Jolley C. J., Yankeelov J. A., Jr Reaction of papain with -bromo- -(5-imidazolyl)propionic acid. Biochemistry. 1972 Jan 18;11(2):164–169. doi: 10.1021/bi00752a005. [DOI] [PubMed] [Google Scholar]
- Lewis S. D., Johnson F. A., Shafer J. A. Effect of cysteine-25 on the ionization of histidine-159 in papain as determined by proton nuclear magnetic resonance spectroscopy. Evidence for a his-159--Cys-25 ion pair and its possible role in catalysis. Biochemistry. 1981 Jan 6;20(1):48–51. doi: 10.1021/bi00504a009. [DOI] [PubMed] [Google Scholar]
- Polgár L., Halász P. Current problems in mechanistic studies of serine and cysteine proteinases. Biochem J. 1982 Oct 1;207(1):1–10. doi: 10.1042/bj2070001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polgár L., Halász P. Evidence for multiple reactive forms of papain. Eur J Biochem. 1978 Aug 1;88(2):513–521. doi: 10.1111/j.1432-1033.1978.tb12477.x. [DOI] [PubMed] [Google Scholar]
- Shipton M., Brochlehurst K. Characterization of the papain active centre by using two-protonic-state electrophiles as reactivity probes. Evidence for nucleophilic reactivity in the un-interrupted cysteine-25-histidine-159 interactive system. Biochem J. 1978 May 1;171(2):385–401. doi: 10.1042/bj1710385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipton M., Brocklehurst K. Benzofuroxan as a thiol-specific reactivity probe. Kinetics of its reactions with papain, ficin, bromelain and low-molecular-weight thiols. Biochem J. 1977 Dec 1;167(3):799–810. doi: 10.1042/bj1670799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shipton M., Stuchbury T., Brocklehurst K. 4-Chloro-7-nitrobenzo-2-oxa-1,3-diazole as a reactivity probe for the investigation of the thiol proteinases. evidence that ficin and bromelain may lack carboxyl groups conformationally equivalent to that of aspartic acid-158 of papain. Biochem J. 1976 Nov;159(2):235–244. doi: 10.1042/bj1590235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallenfels K., Eisele B. Stereospecific alkylation with asymmetric reagents. Eur J Biochem. 1968 Jan;3(3):267–275. doi: 10.1111/j.1432-1033.1968.tb19526.x. [DOI] [PubMed] [Google Scholar]



