Abstract
Background
Sexually transmitted infections (STIs) are a significant public health concern worldwide. These infections can have severe consequences for women’s health, including pelvic inflammatory disease, infertility, and increased risk of HIV acquisition. Understanding the prevalence and factors associated with STIs among women in Sierra Leone is crucial to developing effective prevention and treatment strategies. This study investigated the provincial distribution and factors associated with self-reported STIs (SR-STIs) and STI symptoms among women in Sierra Leone.
Methods
The study used data from the 2019 Sierra Leone Demographic and Health Survey, comprising 13,965 women aged 15 to 49 who have ever had sex. Provincial variations in the prevalence of SR-STIs and STI symptoms were presented using a spatial map. A binary logistic regression analysis was performed to assess the factors associated with SR-STIs and STI symptoms among women in Sierra Leone. The results were presented as adjusted odds ratios (aOR) with a 95% confidence interval (CI).
Results
The overall prevalence of SR-STIs and their symptoms was 21.6% [20.4, 22.9] among women in Sierra Leone. Higher odds of SR-STIs and their symptoms were associated with being aged 25–29 (aOR = 1.35, 95% CI = 1.10,1.66), 30–34 (aOR = 1.38, 95% CI = 1.09,1.74), 35–39 (aOR = 1.39, 95% CI = 1.10, 1.77), having tested for HIV (aOR = 1.16, 95% CI = 1.04, 1.29), and having multiple sexual partners (aOR = 2.25, 95% CI = 1.80, 2.83) compared to those in the reference category. Being in the rich wealth index (aOR = 1.32, 95% CI = 1.11, 1.57), living in the Northern (aOR = 2.43, 95% CI = 2.08, 2.83) and Northwestern province (aOR = 3.28, 95% CI = 2.81, 3.83), and being covered by health insurance (aOR = 3.19, 95% CI = 2.62, 3.88) were associated with higher odds of SR-STIs and their symptoms. Lower odds of SR-STIs and their symptoms were found among women with one (aOR = 0.81, 95% CI = 0.68, 0.96), two (aOR = 0.75, 95% CI = 0.61, 0.93), three (aOR = 0.66, 95% CI = 0.53, 0.83), and four or more children (aOR = 0.62, 95% CI = 0.49, 0.77], those residing in rural areas (aOR = 0.81, 95% CI = 0.69, 0.94), and women living in the Southern province (aOR = 0.67, 95% CI = 0.57, 0.80) compared to those in the reference category.
Conclusion
The findings indicate that SR-STIs and their symptoms are prevalent among women in Sierra Leone. Our findings underscore the urgent need for targeted public health interventions to address the SR-STIs and their symptoms among women in Sierra Leone. The increased risk among women aged 25–39, those with multiple sexual partners and those from wealthier backgrounds, suggests that educational programmes focusing on safe sexual practices and STI prevention should be prioritized in urban and affluent areas. Additionally, the lower odds of SR-STIs among women with more children and those in rural regions indicate that family dynamics and geographic factors may offer protective benefits, which could be leveraged in public health messaging. Our findings highlight the importance of tailoring health services and outreach efforts to effectively reach and support vulnerable populations, ultimately aiming to reduce the incidence of SR-STIs.
Keywords: Sexually transmitted infections, Prevalence, Women, Sierra Leone, Demographic and Health Survey
Introduction
Sexually transmitted infections (STIs) present a substantial global public health issue, disproportionately affecting women’s reproductive health and resulting in severe health consequences if not treated [1]. The World Health Organization (WHO) reports 374 million new cases of chlamydia, gonorrhoea, syphilis, and trichomoniasis among individuals 15–49 in 2020 [2], resulting in 2.5 million deaths each year [3]. In low- and middle-income countries, STIs account for a substantial burden of diseases, with the majority of highly burdened countries in sub-Saharan Africa [4]; this constitutes 40% of the worldwide burden of STIs [5]. STIs can lead to lifelong complications, including infertility and chronic pain, with women being the most affected [6]. It also increases the risk of peripartum morbidity and deaths in industrialised areas as well as among disadvantaged populations in developed countries [7]. The WHO estimates that around 300 million women are infected with human papillomavirus [2], and in the WHO African Region, women have the highest prevalence of herpes simplex virus type 2 (HSV-2), which heightens their risk of HIV infection [8].
Factors associated with the prevalence of STIs among women are multifaceted. Biological factors, such as the anatomical differences between men and women make women more susceptible to certain infections such as STIs [8]. Social determinants, including education level [9, 10], socio-economic status [11, 12], and sexual violence [13], also play a critical role in STI prevalence. Behavioural factors, such as the number of sexual partners, condom use, and substance abuse, further influence the risk of acquiring STIs [14–16]. Previous studies in sub-Saharan Africa among women have addressed the following significant factors associated with STIs: age, educational attainment, economic advantage, having multiple sexual partners, rural-urban residence, and distance to health facilities [17].
In Sierra Leone, the prevalence of STIs among women is particularly concerning due to limited access to healthcare, socio-economic challenges, and cultural factors that influence sexual health behaviours [18]. These issues are compounded by post-conflict health infrastructure and prevailing gender inequalities. Several historically risky factors have engulfed Sierra Leone, such as the civil war between 1992 and 2002 [19], the Ebola outbreak (2014–2016) [20] and the recent COVID-19 pandemic [21]. Adverse situations and conflicts can elevate the probability of engaging in risky behaviours, such as those related to sexual activity [22]. The 2019 Sierra Leone Demographic and Health Survey (SLDHS) report revealed concerning trends, with high-risk behaviours prevalent among young people, including engaging in unprotected sex, having multiple sexual partners, and participating in transactional sex [23]. In many regions, including Sierra Leone, limited access to sexual health services exacerbates the problem, as women may not receive timely diagnosis and treatment [24].
Despite the contribution of self-reported behaviour in the control and prevention of STIs, little attention has been given to self-reporting of these infections. No study in Sierra Leone has specifically investigated this issue using a nationally representative sample. Understanding (SR-STIs) provides valuable insights into the burden of these infections and how women in Sierra Leone seek healthcare. Therefore, this study aims to examine the provincial distribution and factors associated with SR-STIs and STI symptoms among women in Sierra Leone.
Methods
Study design and sampling methods
The 2019 SLDHS was used for this study, a periodically cross-sectional survey that gathers data on demographic, health, and nutritional indicators among women and men of reproductive age as well as children. The survey was conducted over four months (from May 2019 to August 2019) [23], using a stratified, two-stage cluster sampling design. In this study, we included 13,965 women who have ever had sex and were aged 15 to 49. This study adhered to the Strengthening Reporting of Observational Studies in Epidemiology (STROBE) guidelines [25].
Variables
Outcome variable
The outcome variable in the study was SR-STIs and their symptoms. During the survey, women who had engaged in sexual activity were asked if they had SR-STIs and their symptoms in the last 12 months. The symptoms encompass vaginal discharge, along with the existence of a sore or ulcer in the vaginal region. There were three response options: no, yes, and don’t know. Women who responded “no” and “don’t know” were coded as 0 = no (no SR-STIs and their symptoms), whilst those who responded “yes” were coded as 1, as done in the literature [26, 27] that utilised the DHS dataset.
Explanatory variables
The study included 15 explanatory variables. The selection of the variables was determined by thoroughly examining the existing literature [11, 12, 16, 26–28] and their presence in the 2019 SLDHS dataset. The variables were the age of the respondents, level of education, marital status, current employment status, age at first sexual intercourse, history of HIV testing, and number of sexual partners in the past year. Others were visits to healthcare facilities in the past year, health insurance coverage, exposure to mass media, parity, wealth index, place of residence, and province. Table 1 displays the categories of the explanatory variables.
Table 1.
Background characteristics of the women in Sierra Leone (n = 13965)
| Variables | Weighted sample(n) | Weighted percentage(%) |
|---|---|---|
| Woman’s age (years) | ||
| 15–19 | 1,929 | 13.8 |
| 20–24 | 2,534 | 18.1 |
| 25–29 | 2,718 | 19.5 |
| 30–34 | 1,941 | 13.9 |
| 35–39 | 2,222 | 15.9 |
| 40–44 | 1,335 | 9.6 |
| 45–49 | 1,287 | 9.2 |
| Marital status | ||
| Never in union | 3,459 | 24.8 |
| Married | 9,097 | 65.2 |
| Cohabiting | 607 | 4.3 |
| Previously married | 800 | 5.7 |
| Religion | ||
| Christianity | 3,235 | 23.2 |
| Islamic | 10,730 | 76.8 |
| Level of education | ||
| No education | 6,919 | 49.5 |
| Primary | 1,768 | 12.7 |
| Secondary | 4,635 | 33.2 |
| Higher | 643 | 4.6 |
| Current working status | ||
| Not working | 3,623 | 26.0 |
| Working | 10,341 | 74.0 |
| Parity | ||
| None | 2,516 | 18.0 |
| One | 2,478 | 17.7 |
| Two | 2,214 | 15.9 |
| Three | 1,910 | 13.7 |
| Four or more | 4,847 | 34.7 |
| Exposed to mass media | ||
| No | 6,656 | 47.7 |
| Yes | 7,309 | 52.3 |
| Visited health facility in the last 12 months | ||
| No | 5,974 | 42.8 |
| Yes | 7,991 | 57.2 |
| Covered by health insurance | ||
| No | 13,382 | 95.8 |
| Yes | 583 | 4.2 |
| Ever tested for HIV | ||
| No | 5,334 | 38.2 |
| Yes | 8,631 | 61.8 |
| Multiple sexual partnerships | ||
| No | 13,385 | 95.8 |
| Yes | 580 | 4.2 |
| Age at first sex | ||
| 16 years and below | 9,321 | 66.7 |
| 17 years and above | 4,644 | 33.3 |
| Wealth index | ||
| Poor | 5,233 | 37.5 |
| Middle | 2,691 | 19.3 |
| Rich | 6,041 | 43.2 |
| Place of residence | ||
| Urban | 6,154 | 44.1 |
| Rural | 7,811 | 55.9 |
| Province | ||
| Eastern | 2,786 | 20.0 |
| Northern | 2,985 | 21.4 |
| Northwestern | 2,323 | 16.6 |
| Southern | 2,627 | 18.8 |
| Western | 3,244 | 23.2 |
Data analyses
The statistical analyses were performed utilising Stata version 17.0 (Stata Corporation, College Station, TX, USA). First, we utilised a spatial map to visually present the geographical variations in the prevalence of SR-STIs and their symptoms among women in Sierra Leone. A Pearson chi-square analysis was performed to test the independent associations between the variables. Finally, we performed a binary logistic regression analysis to identify the factors associated with SR-STIs and their symptoms. The results were reported as adjusted odds ratios (aORs) and their corresponding 95% confidence intervals (CI). All analyses were weighted and the svyset command in Stata was used to account for the complex sampling design of the DHS data. Weighting was necessary to ensure that the results were representative of the target population, as the DHS employed a two-stage sampling approach.
Ethical consideration
Due to the dataset being publicly available, ethical clearance was not required for this work. Before using the dataset, we requested permission from the Monitoring and Evaluation to Assess and Use Results Demographic and Health Surveys (MEASURE DHS) to utilise the SLHDS.
Results
Background characteristics of women in Sierra Leone
Table 1 shows the background characteristics of the women in Sierra Leone. The modal age group was 20-29. The age distribution showed that 13.8% were aged 15–19, while 19.5% fell within the 25–29 age group. In terms of marital status, 65.2% were married, and 24.8% had never been in a union. The majority identified as Islamic (76.8%), and nearly half (49.5%) had no formal education, with only 4.6% achieving higher education. The results showed that 74.0% were working, and 34.7% had four or more children. Media exposure was noted in 52.3% of respondents, and 57.2% had visited a health facility in the past year. For health insurance coverage, 95.8% were lacking it, and 61.8% had undergone HIV testing. Most women (95.8%) reported no multiple sexual partnerships, and 66.7% experienced their first sexual intercourse at 16 years or younger. With wealth index, 37.5% were classified as poor, with 43.2% identified as rich, and residentially, 55.9% lived in rural areas. In terms of province, the majority of the women were in the Western area.
Prevalence of self-reported STIs and STI symptoms among women in Sierra Leone
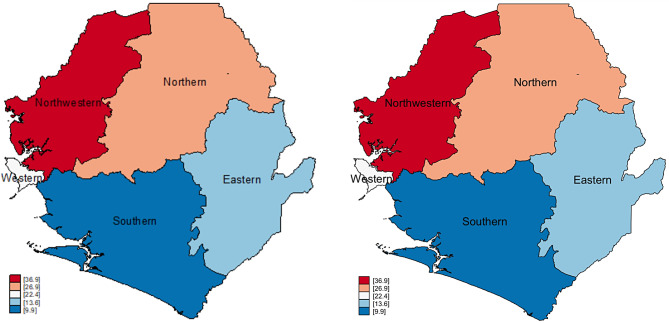
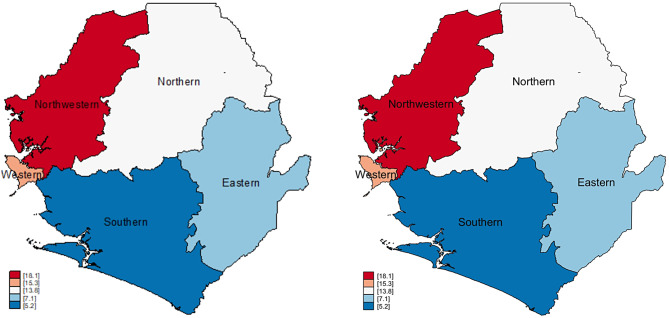
Table 2 shows the prevalence of SR-STIs and STI symptoms among women in Sierra Leone. The prevalence of SR-STIs and STI symptoms was 21.6% [20.4,22.9], any STIs (any STI that a respondent reports having had in their lifetime) was 11.9% [10.9,13.0], genital sore or ulcer was 9.6% [8.6,10.6], and genital discharge was 17.0% [16.0,18.2]. In Fig. 1, we present results on the provincial distribution of SR-STIs and STI symptoms among women in Sierra Leone. The North Western province, had the highest prevalence of SR-STIs and their symptoms (36.7%), while the Southern province had the lowest (9.9%). Figure 2 shows the provincial distribution of genital sores/ulcers among women in Sierra Leone. The Northern province had the highest prevalence of genital sore/ulcer (16.0%), whilst the Southern province had the lowest prevalence (1.7%). Figure 3 has results on the provincial distribution of genital discharge among women in Sierra Leone. The North Western province had the highest prevalence of genital discharge (29.6%), whilst the Southern province had the lowest prevalence of 5.8%. As shown in Fig. 4, the North Western province had the highest prevalence of any STIs (18.1%), whilst the Southern province had the lowest prevalence of 5.2%.
Table 2.
Bivariate analysis of self-reported STIs and symptoms among women in Sierra Leone using a chi-square test
| Variable | Genital sore/ulcer | Genital discharge | Any STIs | SR-STIs and their symptoms |
|---|---|---|---|---|
| Prevalence | 9.6% [8.6,10.6] | 17.0% [16.0,18.2] | 11.9% [10.9,13.0] | 21.6% [20.4,22.9] |
| Age of women (years) | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| 15–19 | 9.5 [8.1,11.2] | 17.6 [15.6,19.8] | 12.2 [10.3,14.4] | 22.7 [20.4,25.2] |
| 20–24 | 10.8 [9.2,12.5] | 19.8 [17.9,21.9] | 12.4 [10.8,14.3] | 24.3 [22.2,26.6] |
| 25–29 | 10.8 [9.0,12.9] | 18.4 [16.5,20.5] | 14.8 [13.0,16.8] | 23.7 [21.6,25.9] |
| 30–34 | 9.3 [7.9,10.8] | 17.4 [15.2,19.8] | 12.5 [10.5,14.7] | 22.1 [19.7,24.7] |
| 35–39 | 10.3 [8.7,12.3] | 17.0 [15.3,19.0] | 11.5 [10.0,13.3] | 21.5 [19.5,23.7] |
| 40–44 | 7.8 [6.1,9.8] | 13.9 [12.0,16.0] | 9.0 [7.4,10.9] | 18.2 [16.0,20.7] |
| 45–49 | 5.7 [4.3,7.6] | 10.6 [8.7,12.7] | 7.0 [5.5,8.9] | 13.4 [11.4,15.6] |
| Marital status | p = 0.019 | p = 0.008 | p = 0.021 | p = 0.022 |
| Never in union | 11.3 [9.9,12.9] | 19.0 [17.1,21.0] | 13.9 [12.2,15.7] | 24.0 [22.0,26.2] |
| Married | 9.0 [7.9,10.2] | 16.7 [15.4,18.0] | 11.3 [10.2,12.5] | 20.9 [19.5,22.4] |
| Cohabiting | 8.3 [6.0,11.4] | 12.4 [9.7,15.7] | 11.2 [8.3,15.0] | 19.2 [15.5,23.5] |
| Previously married | 10.0 [7.6,13.0] | 16.0 [12.9,19.7] | 10.5 [7.8,14.0] | 21.0 [17.3,25.2] |
| Religion | p = 0.074 | p = 0.011 | p = 0.678 | p = 0.025 |
| Christianity | 8.3 [7.0,9.9] | 15.1 [13.4,16.9] | 11.6 [9.9,13.5] | 19.6 [17.6,21.8] |
| Islam | 10.0 [8.9,11.1] | 17.6 [16.4,18.9] | 12.0 [10.8,13.2] | 22.2 [20.9,23.6] |
| Level of education | p = 0.021 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| No education | 8.4 [7.4,9.6] | 15.4 [14.2,16.6] | 10.6 [9.4,11.9] | 19.7 [18.3,21.3] |
| Primary | 9.4 [6.9,12.8] | 17.0 [14.3,20.2] | 10.9 [8.6,13.6] | 20.3 [17.5,23.4] |
| Secondary | 11.1 [9.8,12.6] | 18.9 [17.2,20.6] | 13.6 [12.1,15.2] | 24.2 [22.2,26.2] |
| Tertiary | 11.0 [8.3,14.5] | 21.8 [17.9,26.2] | 16.9 [13.2,21.3] | 27.1 [22.7,32.0] |
| Current working status | p = 0.563 | p = 0.067 | p = 0.542 | p = 0.123 |
| Not working | 9.2 [7.9,10.7] | 15.6 [13.9,17.6] | 12.3 [10.7,14.1] | 20.4 [18.5,22.5] |
| Working | 9.7 [8.6,10.9] | 17.5 [16.4,18.8] | 11.8 [10.7,12.9] | 22.1 [20.7,23.4] |
| Parity | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| None | 11.0 [9.5,12.8] | 20.7 [18.7,22.9] | 14.7 [12.8,16.7] | 26.1 [23.8,28.5] |
| One | 10.6 [9.0,12.4] | 19.0 [17.0,21.3] | 13.7 [11.9,15.8] | 23.8 [21.6,26.2] |
| Two | 11.3 [9.4,13.5] | 18.7 [16.7,20.8] | 13.0 [11.4,14.8] | 23.5 [21.3,25.8] |
| Three | 9.3 [7.8,10.9] | 15.2 [13.5,17.2] | 10.8 [9.2,12.6] | 20.3 [18.3,22.4] |
| Four or more | 7.7 [6.6,8.9] | 14.1 [12.8,15.5] | 9.5 [8.3,10.8] | 17.9 [16.4,19.5] |
| Exposed to mass media | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| No | 8.2 [7.1,9.5] | 15.2 [13.8,16.6] | 10.2 [9.0,11.6] | 19.6 [18.1,21.3] |
| Yes | 10.8 [9.7,12.1] | 18.7 [17.4,20.2] | 13.4 [12.1,14.8] | 23.4 [21.8,25.2] |
| Visited health facility in last 12 months | p = 0.078 | p = 0.078 | p = 0.775 | p = 0.076 |
| No | 8.8 [7.7,10.1] | 16.1 [14.6,17.6] | 11.8 [10.4,13.3] | 20.6 [19.0,22.3] |
| Yes | 10.2 [9.0,11.4] | 17.8 [16.4,19.2] | 12.0 [10.8,13.3] | 22.4 [20.9,24.0] |
| Health insurance | p = 0.444 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| No | 9.6 [8.7,10.7] | 16.3 [15.2,17.4] | 10.6 [9.7,11.5] | 20.5 [19.3,21.7] |
| Yes | 8.3 [5.5,12.2] | 34.5 [27.7,42.1] | 41.7 [32.3,51.8] | 48.2 [37.7,58.8] |
| Ever tested for HIV | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| No | 8.0 [7.1,9.1] | 15.3 [14.0,16.7] | 9.7 [8.6,11.0] | 19.8 [18.2,21.4] |
| Yes | 10.5 [9.3,11.9] | 18.1 [16.7,19.5] | 13.2 [12.0,14.6] | 22.8 [21.3,24.4] |
| Multiple sexual partners | p = < 0.001 | p = < 0.001 | p = 0.116 | p = < 0.001 |
| No | 9.1 [8.2,10.0] | 16.5 [15.4,17.7] | 11.8 [10.7,12.9] | 21.0 [19.8,22.4] |
| Yes | 21.7 [17.9,26.0] | 28.7 [24.4,33.4] | 14.6 [11.3,18.7] | 35.2 [30.9,39.9] |
| Age at first sex | p = 0.660 | p = 0.688 | p = 0.314 | p = 0.313 |
| 16 years and below | 9.7 [8.7,10.8] | 16.9 [15.7,18.2] | 12.2 [11.0,13.5] | 22.0 [20.5,23.5] |
| 17 years and above | 9.4 [8.0,10.9] | 17.3 [15.7,19.0] | 11.3 [10.0,12.8] | 20.9 [19.2,22.8] |
| Wealth index | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| Poor | 7.5 [6.1,9.3] | 14.4 [12.9,16.0] | 8.8 [7.5,10.2] | 18.1 [16.4,19.9] |
| Middle | 9.0 [7.5,10.7] | 15.9 [14.1,18.0] | 9.8 [7.9,12.1] | 20.7 [18.4,23.2] |
| Rich | 11.6 [10.2,13.1] | 19.8 [18.0,21.8] | 15.5 [13.9,17.3] | 25.1 [23.0,27.3] |
| Place of residence | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| Urban | 11.4 [10.0,12.9] | 19.1 [17.4,21.0] | 15.3 [13.7,17.2] | 24.7 [22.7,26.9] |
| Rural | 8.2 [6.9,9.6] | 15.4 [14.0,16.9] | 9.2 [7.9,10.6] | 19.2 [17.6,20.8] |
| Province | p = < 0.001 | p = < 0.001 | p = < 0.001 | p = < 0.001 |
| Eastern | 6.3 [5.1,7.9] | 9.7 [8.0,11.7] | 7.1 [5.6,9.0] | 13.6 [11.4,16.1] |
| Northern | 16.0 [13.6,18.8] | 24.1 [21.7,26.7] | 13.8 [11.9,16.0] | 26.9 [24.3,29.5] |
| Northwestern | 12.2 [9.9,14.9] | 29.6 [26.7,32.7] | 18.1 [14.3,22.6] | 36.7 [32.9,40.7] |
| Southern | 1.7 [1.2,2.3] | 5.8 [4.6,7.2] | 5.2 [4.3,6.4] | 9.9 [8.4,11.7] |
| Western | 10.9 [8.9,13.4] | 16.9 [14.5,19.7] | 15.3 [13.0,17.9] | 22.4 [19.5,25.6] |
| p = p-value from the chi-square test | ||||
Fig. 1.
Prevalence of SR-STIs and STI symptoms among women in Sierra Leone
Fig. 2.
Prevalence of genital sore/ulcer among women in Sierra Leone
Fig. 3.
Prevalence of genital discharge among women in Sierra Leone
Fig. 4.
Prevalence of any STIs among women in Sierra Leone
Bivariate results of self-reported STIs and STI symptoms among women in Sierra Leone using a chi-square test
Table 2 shows the bivariate results of SR-STIs and STI symptoms among women in Sierra Leone. SR-STIs and their symptoms were higher among women aged 20–24 (24.3%), never married women (24.0%), those who were Muslims (22.2%), those with tertiary education (27.1%), women with no children (26.1%), and women exposed to media (23.4%). The results further show that women covered by health insurance (48.2%), those who tested for HIV (22.8%), women with multiple sexual partners (35.2%), those with riche wealth index (25.3%), women who reside in urban areas (24.7%) and those living in the North Western province (36.7%). Apart from age at first sex, health facility visits, and working status, the remaining explanatory variables were significantly associated with SR-STIs and their symptoms among women in Sierra Leone at p < 0.05.
Factors associated with SR-STIs and their symptoms among women in Sierra Leone
Table 3 presents the results of the factors associated with SR-STIs and their symptoms among women in Sierra Leone. Women aged 25–39 had higher odds of SR-STIs and their symptoms than those aged 15–19, with the highest odds among those aged 35–39 (aOR = 1.39, 95% CI = 1.10, 1.77). The odds of SR-STIs and STI symptoms were higher among women who had tested for HIV (aOR = 1.16, 95% CI = 1.04, 1.29) compared to those who had not. Women with multiple sexual partners (aOR = 2.25, 95% CI = 1.80, 2.83) had higher odds of SR-STIs and their symptoms compared to those who did not have. Women of the rich wealth index (aOR = 1.32, 95% CI = 1.11, 1.57), those living in the Northern (aOR = 2.43, 95% CI = 2.08, 2.83) and North West provinces (aOR = 3.28, 95% CI = 2.81, 3.83) and those covered by health insurance (aOR = 3.19, 95% CI = 2.62, 3.88) had higher odds of SR-STIs and their symptoms than those of the poor wealth index, those living in the Eastern province, and those without health insurance respectively. Women with at least one child had lower odds of SR-STIs and their symptoms compared to those with no child, with the lowest odds among those with four or more children (aOR = 0.62, 95% CI = 0.49, 0.77). Women residing in rural areas (aOR = 0.81, 95% CI = 0.69, 0.94) had lower odds of SR-STIs and their symptoms than those living in urban areas. Also, women in the Southern province (aOR = 0.67, 95% CI = 0.57, 0.80) had lower odds of SR-STIs and their symptoms than those in the Eastern province.
Table 3.
Factors associated with genital ulcer, discharge, and any STIs among women in Sierra Leone
| Variables | Genital sore or ulcer | Genital discharge | Any STIs | SR-STIs and their symptoms | ||||
|---|---|---|---|---|---|---|---|---|
| Woman’s age (years) | aOR | [95% CI] | aOR | [95% CI] | aOR | [95% CI] | aOR | [95% CI] |
| 15–19 | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| 20–24 | 1.07 | [0.84,1.38] | 1.25* | [1.03,1.51] | 1.00 | [0.80,1.24] | 1.15 | [0.97,1.38] |
| 25–29 | 1.29 | [0.95,1.76] | 1.36** | [1.09,1.71] | 1.45** | [1.12,1.88] | 1.35** | [1.10,1.66] |
| 30–34 | 1.26 | [0.91,1.76] | 1.43** | [1.11,1.84] | 1.41* | [1.07,1.88] | 1.38** | [1.09,1.74] |
| 35–39 | 1.55* | [1.10,2.18] | 1.46** | [1.13,1.90] | 1.37* | [1.01,1.84] | 1.39** | [1.10,1.77] |
| 40–44 | 1.12 | [0.76,1.67] | 1.19 | [0.89,1.59] | 1.08 | [0.77,1.50] | 1.16 | [0.89,1.51] |
| 45–49 | 0.89 | [0.58,1.37] | 0.91 | [0.66,1.25] | 0.90 | [0.64,1.28] | 0.84 | [0.63,1.12] |
| Level of education | ||||||||
| No education | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Primary | 1.17 | [0.89,1.53] | 1.21 | [1.00,1.47] | 1.01 | [0.81,1.27] | 1.07 | [0.89,1.27] |
| Secondary | 1.12 | [0.92,1.37] | 1.07 | [0.92,1.25] | 0.94 | [0.79,1.12] | 1.06 | [0.92,1.22] |
| Higher | 1.00 | [0.71,1.41] | 1.08 | [0.82,1.42] | 0.90 | [0.67,1.21] | 0.96 | [0.74,1.25] |
| Parity | ||||||||
| None | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| One | 0.98 | [0.76,1.25] | 0.84 | [0.69,1.01] | 0.78* | [0.63,0.96] | 0.81* | [0.68,0.96] |
| Two | 1.07 | [0.79,1.45] | 0.77* | [0.61,0.96] | 0.69** | [0.53,0.89] | 0.75** | [0.61,0.93] |
| Three | 0.91 | [0.65,1.27] | 0.63*** | [0.50,0.81] | 0.59*** | [0.44,0.78] | 0.66*** | [0.53,0.83] |
| Four or more | 0.82 | [0.59,1.13] | 0.64*** | [0.50,0.81] | 0.57*** | [0.44,0.76] | 0.62*** | [0.49,0.77] |
| Exposed to mass media | ||||||||
| No | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Yes | 0.98 | [0.84,1.16] | 1.06 | [0.94,1.19] | 1.01 | [0.88,1.16] | 1.03 | [0.92,1.14] |
| Ever tested for HIV | ||||||||
| No | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Yes | 1.24** | [1.06,1.45] | 1.17* | [1.03,1.31] | 1.39*** | [1.21,1.60] | 1.16** | [1.04,1.29] |
| Multiple sexual partnerships | ||||||||
| No | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | ||
| Yes | 2.71*** | [2.06,3.57] | 2.24*** | [1.77,2.84] | 2.25*** | [1.80,2.83] | ||
| Wealth index | ||||||||
| Poor | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Middle | 1.08 | [0.88,1.32] | 0.98 | [0.84,1.15] | 0.96 | [0.81,1.15] | 1.00 | [0.87,1.15] |
| Rich | 1.37* | [1.07,1.75] | 1.48*** | [1.23,1.79] | 1.40** | [1.12,1.76] | 1.32** | [1.11,1.57] |
| Residence | ||||||||
| Urban | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Rural | 0.84 | [0.68,1.03] | 0.90 | [0.76,1.06] | 0.72** | [0.59,0.88] | 0.81** | [0.69,0.94] |
| Province | ||||||||
| Eastern | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Northern | 3.06*** | [2.50,3.76] | 3.05*** | [2.58,3.62] | 2.29*** | [1.88,2.79] | 2.43*** | [2.08,2.83] |
| Northwestern | 2.04*** | [1.64,2.52] | 3.53*** | [2.97,4.19] | 3.47*** | [2.85,4.22] | 3.28*** | [2.81,3.83] |
| Southern | 0.26*** | [0.19,0.36] | 0.53*** | [0.43,0.66] | 0.80* | [0.64,1.00] | 0.67*** | [0.57,0.80] |
| Western | 1.31* | [1.04,1.67] | 1.15 | [0.95,1.41] | 1.59*** | [1.28,1.97] | 1.17 | [0.98,1.40] |
| Marital status | ||||||||
| Never in union | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] |
| Married | 0.83 | [0.66,1.03] | 1.04 | [0.88,1.23] | 0.97 | [0.80,1.18] | 1.03 | [0.88,1.21] |
| Cohabiting | 0.77 | [0.52,1.14] | 0.82 | [0.61,1.12] | 0.87 | [0.63,1.20] | 0.97 | [0.74,1.27] |
| Previously married | 0.87 | [0.60,1.26] | 0.95 | [0.72,1.27] | 0.80 | [0.58,1.10] | 1.00 | [0.77,1.29] |
| Religion | ||||||||
| Christian | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | ||||
| Muslim | 1.06 | [0.93,1.21] | 1.08 | [0.96,1.22] | ||||
| Covered by health insurance | ||||||||
| No | 1.00 | [1.00,1.00] | 1.00 | [1.00,1.00] | ||||
| Yes | 2.15*** | [1.74,2.64] | 3.19*** | [2.62,3.88] | ||||
| N | 13965 | 13965 | 13965 | 13965 | ||||
| pseudo R2 | 0.082 | 0.090 | 0.058 | 0.083 | ||||
Exponentiated coefficients; 95% confidence intervals in brackets
*p < 0.05, **p < 0.01, ***p < 0.001
Discussion
In this study, we report the prevalence and correlates of SR-STIs and their symptoms among women in Sierra Leone. Factors associated with SR-STIs and their symptoms in women include women’s age, having tested for HIV, having multiple sexual partners, household wealth index, residence, health insurance cover, parity, and province of residence.
STIs remain a significant public health problem because of their ability to cause tremendous health and economic burdens when they are not adequately managed [29]. The findings of this study showed that one in five women reported SR-STIs and their symptoms. The relatively high proportion of SR-STIs and STI symptoms among these women reflects a generally high STI prevalence in Sierra Leone, as well as the sub-Saharan African region, where 40% of global STI cases are recorded [30]. The finding indicate that Sierra Leone has one of the highest STI prevalence in SSA [31]. Consistent with the present finding, one-third of college students in Sierra Leone reported that they had experienced STIs [32]. STIs such as Syphilis and HIV have been reported to be high among different populations in the country [18, 32–34]. Based on this study’s findings regarding the high prevalence of SR-STIs, Sierra Leone still has a considerable effort to make in achieving the global health strategy for STIs, which aims to ensure improved sexual and reproductive health among women. To achieve this, it is essential to understand the sociodemographic and socio-economic factors contributing to the burden of STIs among women.
The factors associated with SR-STIs and STI symptoms in this study were age, parity, HIV testing, multiple sexual partnerships, wealth, place of residence, and possession of health insurance. We observed that SR-STIs and STI symptoms was high among reproductive-age women, especially those between 25 and 39 years, compared to younger teenagers between 15 and 19 years. As observed in the present study, younger women in South Africa were more likely than older women to have an STI [35]. Several factors may explain why SR-STIs and STI symptoms may be higher among older women than adolescents in Sierra Leone. The first is the difference in the utilisation of condoms and other contraceptives for protection. In Sierra Leone, differences exist in the utilisation of condoms and contraceptives for sexual protection among different age groups [36, 37]. The use of condoms remains, for a very long time, one of the effective strategies offering protection against STIs by Chlamydia trachomatis, Neisseria gonorrhoeae, Trichomonas vaginalis, Treponema pallidum, Cytomegalovirus and HIV [38, 39]. Given the changing dynamics of sexual activity in Sierra Leone, with decreasing age at sexual debut and increasing risk [32, 40, 41], the rate of condom use could be a determining factor accounting for the difference in SR-STIs and STI symptoms prevalence among different age groups. Aside from condom use patterns, women below the age of 20 years may not have experienced an increased multiple sexual debuts or had increased multiple sexual partnerships as their older counterparts. Older women are generally more sexually active than teenagers, which could contribute to increased rates of SR-STIs and STI symptoms. However, it is important to acknowledge that reporting biases may also play a role. Teenage girls may be less likely to disclose their sexual history due to concerns about stigma and judgment from interviewers or peers. Of course, a large set of studies have reported that the odds of having an STI increases when a woman has a high number of lifetime or previous year sexual partners [42–45]. This is also in line with the observed increased likelihood of women with multiple sexual partners reporting having STIs in the present study. Engaging in numerous sexual partnerships tends to increase exposure to STIs, given that a lot of people in Sierra Leone do not even know their status and may carry infections without knowing [46].
Pregnancy occurs when there is unprotected sexual intercourse. Therefore, an association between parity and SR-STI and STI symptoms is not an unexpected finding. Women with more children were less likely to have SR-STI and STI symptoms compared with their counterparts with no or few children, consistent with a similar study in South Africa [35]. Decreased SR-STI and STI symptoms prevalence among women with increased parity could be a result of an increased number of visits to health facilities. By visiting the hospital regularly, these women are educated on issues to improve their well-being and that of their children.
Economic status was associated with SR-STI and STI symptoms, consistent with a previous study [47]. In a study conducted in the United States and Bangladesh, poorer participants were found to have increased STI odds compared with the rich [48, 49]. Poor women are expected to be at increased risk of STI because of the likelihood of having low education, decreased healthcare access, and engaging in sexual activity for economic gain. This finding is the direct opposite of what we observed in women in Sierra Leone, where more affluent women were more likely to report STIs and their symptoms compared to those in the poorest wealth quantiles. A possible reason for increased SR-STI and STI symptoms in the rich is increased mobility for business or holidays and access to social events and networks, which is likely to lead to higher exposure to multiple sexual partners [50]. It is important to note that our analysis of economic status may be limited, as it does not account for individual income levels, which could influence STI risk. Future research should consider a more nuanced approach to measuring economic status, incorporating both individual income and education, to better elucidate their relationships with STI risk.
Place of residence was associated with STI, which is consistent with other studies [11]. We observed that women living in rural areas were less likely to report STIs and their symptoms than those in urban areas. In Sierra Leone, urban residents are more likely to engage in risky sexual behaviours such as having multiple sexual partners, engaging in transactional sex, and having condomless sex [18]. Migrants from rural regions occasionally populate urban areas, and the anonymity associated with foreign nationality may encourage riskier sexual behaviours, including engaging in multiple sexual partnerships, commercial sex workers, or alcohol and drug abuse [51]. We found notable variations in SR-STI and their symptoms prevalence based on the provinces where participants resided. Specifically, women from the Northern, Northwestern, and Western provinces were more likely to report STIs and their symptoms compared to those from the Eastern province, while those in the Southern province exhibited lower rates of STIs and their symptoms reporting. These variations may be attributed to differences in the rates of risky sexual behaviors, as well as social, cultural, and economic factors unique to each province. Additionally, it is important to consider disparities in access to sexual and reproductive health services across these provinces. For instance, women in the Northwestern province, which is characterized by high economic and mining activities, have been found to engage in riskier sexual behaviors, while access to health services may be limited [18]. Conversely, women in the Southern province tend to report lower levels of risky behaviors, which may be linked to better access to sexual and reproductive health resources [18]. By incorporating these factors, we can better understand the provincial disparities in STI prevalence and the underlying influences that contribute to these differences. Finally, we made a critical observation, which showed that STI was more likely among women who seemed to pay attention to their healthcare. We found that women who had tested for HIV and those with health insurance were more likely to report that they have STIs and their symptoms than their counterparts who had never tested for HIV or without health insurance. This finding raises important questions about the characteristics of women with health insurance. It is essential to consider whether these women are predominantly residing in urban areas, where access to health services is generally better. Urban living can be associated with increased exposure to risky sexual behaviors, including having multiple sexual partners, which are known risk factors for STIs [52].Our finding contradicts other studies where health insurance coverage is relatively lower in patients with STIs [52]. Several factors may explain this observation. First, women who have tested their HIV status and those with health insurance may be aware of their STI status and have taken these steps to manage it. This is particularly important because coinfection of HIV with other STIs is expected because of their shared mode of transmission and decreased immunity caused by each other [53]. This observation is encouraging, given that HIV testing is low and health insurance is relatively expensive for most Sierra Leoneans [54]. Our findings underscore the importance of integrated sexual health services that address both HIV testing and STIs, as seen in efforts like those described by Joseph et al. in Haiti [55].
Strengths and limitation
The findings of this study should be considered in light of the following limitations. One major limitation is the inability to establish causality since a cross-sectional design was employed in the survey. As with examining self-reported behaviour and STI, social desirability may have influenced the reports’ validity. Social desirability bias is particularly challenging because respondents may intentionally answer questions incorrectly by underreporting stigmatised behaviours or by overreporting normative activities if their conduct is considered socially undesirable. Despite these limitations, our study is the first to assess the provincial distribution of SR-STIs and STI symptoms and their determinants in Sierra Leone using a nationally representative dataset.
Conclusion
Our study has shown that STIs and their symptoms are prevalent among women in Sierra Leone. Our findings suggest that the country needs a comprehensive STI screening, surveillance, and treatment system. In designing STI elimination and management strategies, it is essential to consider the demographics of women, such as their age, residence, parity and economic status. It is also necessary to discourage risky lifestyles such as multiple sexual partnerships, condomless sex, and engaging in sexual activity for money among women through education and policy formulation. Through these interventions, increased STI prevalence and their adverse effects would be curbed among women.
Acknowledgements
We thank the MEASURE DHS Program for their support and for making the dataset freely accessible.
Abbreviations
- aOR
Adjusted Odds Ratio
- CI
Confidence Interval
- DHS
Demographic and Health Survey
- STIs
Sexually Transmitted Infections
- MEASURE DHS
Monitoring and Evaluation to Assess and Use Results Demographic and Health Surveys
- STROBE
Strengthening the Reporting of Observational Studies in Epidemiology
Author contributions
AO, RGA and BOA contributed to the study design and conceptualisation. AO, RGA and BOA performed the analysis. AO, MAE, FGW, RGA, CB, and BOA developed the initial draft. All the authors critically reviewed the manuscript for its intellectual content. All authors read and amended drafts of the paper and approved the final version. AO had the final responsibility of submitting it for publication.
Funding
The study received no funding.
Data availability
The data used for this study is freely available at https://dhsprogram.com/data/dataset/Sierra-Leone_Standard-DHS_2019.cfm?flag=0.
Declarations
Ethics approval and consent to participate
Ethical clearance was not sought for the study since the secondary dataset is freely available in the public domain. A detailed description of the ethical issues regarding the DHS and its dataset usage can be assessed at http://goo.gl/ny8T6X.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Wynn A, Bristow CC, Cristillo AD, Murphy SMC, Van Den Broek N, Muzny C et al. Sexually Transmitted Infections in Pregnancy and Reproductive Health: Proceedings of the STAR Sexually Transmitted Infection Clinical Trial Group Programmatic Meeting. Sex Transm Dis. 2020;47(1):5–11. [DOI] [PMC free article] [PubMed]
- 2.World Health Organization. Sexually transmitted infections (STIs). 2024. Available from https://www.who.int/news-room/fact-sheets/detail/sexually-transmitted-infections-(stis).
- 3.United Nations. WHO reports a major increase in sexually transmitted infections. 2024. Available from https://news.un.org/en/story/2024/05/1150061
- 4.World Health Organization. Global progress report on HIV, viral hepatitis and sexually transmitted infections, 2021. Accountability for the global health sector strategies 2016–2021: actions for impact. Geneva: World Health Organization. 2021.
- 5.Voth ML, Akbari RP. Sexually transmitted proctitides. Clin Colon Rectal Surg. 2007;20(1):58–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rietmeijer CA, Kissinger PJ, Guilamo-Ramos V, Gaydos CA, Hook EW, Mead A, et al. Report from the National Academies of Sciences, Engineering and Medicine—STI: adopting a sexual health Paradigm—A synopsis for sexually transmitted infection practitioners, clinicians, and researchers. Sex Transm Dis. 2022;49(2):169–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Van Gerwen OT, Muzny CA, Marrazzo JM. Sexually transmitted infections and female reproductive health. Nat Microbiol. 2022;7(8):1116–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.World Health Organization. Massive proportion of world’s population are living with herpes infection. 2020.
- 9.Semwogerere M, Dear N, Tunnage J, Reed D, Kibuuka H, Kiweewa F, et al. Factors associated with sexually transmitted infections among care-seeking adults in the African cohort study. BMC Public Health. 2021;21(1):1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brima N, Burns F, Fakoya I, Kargbo B, Conteh S, Copas A. Factors associated with HIV prevalence and HIV testing in Sierra Leone: findings from the 2008 Demographic Health Survey. PLoS ONE. 2015;10(10):1–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Seidu AA, Ahinkorah BO, Dadzie LK, Tetteh JK, Agbaglo E, Okyere J, et al. A multi-country cross-sectional study of self-reported sexually transmitted infections among sexually active men in sub-saharan Africa. BMC Public Health. 2020;20(1):1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huda MN, Ahmed MU, Uddin MB, Hasan MK, Uddin J, Dune TM. Prevalence and demographic, Socio-economic, and behavioral risk factors of self-reported symptoms of sexually transmitted infections (STIs) among ever-married women: evidence from nationally representative surveys in Bangladesh. Int J Environ Res Public Health. 2022;19(3):1906. [DOI] [PMC free article] [PubMed]
- 13.Leblanc NM, Alexander K, Carter S, Crean H, Ingram L, Kobie J, et al. The effects of Trauma, Violence, and Stress on Sexual Health Outcomes among Female Clinic Clients in a small northeastern U.S. Urban Center. Women’s Heal Rep. 2020;1(1):132–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sentís A, Martin-Sanchez M, Arando M, Vall M, Barbera MJ, Ocaña I, et al. Sexually transmitted infections in young people and factors associated with HIV coinfection: an observational study in a large city. BMJ Open. 2019;9(5):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gottlieb SL, Low N, Newman LM, Bolan G, Kamb M, Broutet N. Toward global prevention of sexually transmitted infections (STIs): The need for STI vaccines. Vaccine. 2014;32(14):1527–35. 10.1016/j.vaccine.2013.07.087 [DOI] [PMC free article] [PubMed]
- 16.Seidu AA, Agbaglo E, Dadzie LK, Tetteh JK, Ahinkorah BO. Self-reported sexually transmitted infections among sexually active men in Ghana. BMC Public Health. 2021;21(1):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shewarega ES, Fentie EA, Asmamaw DB, Negash WD, Fetene SM, Teklu RE et al. Sexually transmitted infections related care-seeking behavior and associated factors among reproductive age women in East Africa: a multilevel analysis of demographic and health surveys. BMC Public Health. 2022;22(1):1–11. 10.1186/s12889-022-14120-w [DOI] [PMC free article] [PubMed]
- 18.Kawuki J, Kamara K, Sserwanja Q. Prevalence of risk factors for human immunodeficiency virus among women of reproductive age in Sierra Leone: a 2019 nationwide survey. BMC Infect Dis. 2022;22(1):1–9. 10.1186/s12879-022-07037-7 [DOI] [PMC free article] [PubMed]
- 19.Alemu MM. Youth unemployment challenges and opportunities: the case of Sierra Leone. Int J Soc Sci Stud. 2016;4(10).
- 20.Lamin SA, Teboh C. Situational analysis of the Emergence and Spread of Ebola in Sierra Leone. Glob Soc Welf. 2017;4(1):41–9. [Google Scholar]
- 21.Akbarialiabad H, Shidhaye R, Shidhaye P, Cuijpers P, Weaver MR, Bahrololoom M, et al. Impact of major disease outbreaks in the third millennium on adolescent and youth sexual and reproductive health and rights in low and/or middle-income countries: a systematic scoping review protocol. BMJ Open. 2022;12(3):1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pratto DJ, Jessor R, Jessor SL. Problem Behavior and Psychosocial Development: a longitudinal study of growth. Soc Forces. 1979;57(3):1025. [Google Scholar]
- 23.Statistics Sierra Leone (Stats SL), ICF. Sierra Leone Demographic and Health Survey 2019. Freetown: Stats SL and ICF. 2020. https://dhsprogram.com/pubs/pdf/FR365/FR365.pdf
- 24.Ninsiima LR, Chiumia IK, Ndejjo R. Factors influencing access to and utilisation of youth-friendly sexual and reproductive health services in sub-saharan Africa: a systematic review. Reprod Health. 2021;18(1):135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Von Elm E, Altman DG, Egger M, et al. Strobe Initiative. Strengthening the reporting of Observational studies in Epidemiology (STROBE) Statement: guidelines for reporting observational studies. Int J Surg. 2014;12(12):1495–9. [DOI] [PubMed] [Google Scholar]
- 26.Appiah CK, Dowou RK, Balame SK, Adzigbli LA, Yeboah PA, Aboagye RG, Cadri A. Self-reported sexually transmitted infections among adolescent girls and young women in Mali: analysis of prevalence and predictors. BMJ Open. 2023;13(4):e069226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dadzie LK, Agbaglo E, Okyere J, Aboagye RG, Arthur-Holmes F, Seidu AA, Ahinkorah BO. Self-reported sexually transmitted infections among adolescent girls and young women in sub-saharan Africa. Int Health. 2022;14(6):545–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Adu C, Mohammed A, Budu E, Frimpong JB, Tetteh JK, Ahinkorah BO, Seidu AA. Sexual autonomy and self-reported sexually transmitted infections among women in sexual unions. Archives Public Health. 2022;80(1):40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zheng Y, Yu Q, Lin Y, Zhou Y, Lan L, Yang S, Wu J. Global burden and trends of sexually transmitted infections from 1990 to 2019: an observational trend study. Lancet Infect Dis. 2022;22(4):541–51. [DOI] [PubMed] [Google Scholar]
- 30.Korenromp EL, Wi T, Resch S, Stover J, Broutet N. Costing of national STI program implementation for the global STI control strategy for the health sector, 2016–2021. PLoS ONE. 2017;12(1):e0170773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ekholuenetale M, Nzoputam CI, Okonji OC. Sub-regional variations in sexually transmitted infections manifesting as Vaginitis among Reproductive-aged women in Sub-saharan Countries. Venereology. 2022;1(3):245–61. [Google Scholar]
- 32.Small E, Kim YK, Yu M. Sexually transmitted diseases among college students in Sierra Leone: a life course ecological analysis. Sex Cult. 2021;25(3):884–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jiba DF, Lakoh S, Wang S, Sun W, Barrie U, Kamara MN, Jalloh AT, Tamba FK, Yendewa GA, Song JW, Yang G. Sero-prevalence of syphilis infection among people living with HIV in Sierra Leone: a cross-sectional nationwide hospital-based study. BMC Infect Dis. 2023;23(1):762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Osborne A, James PB, Bangura C, Williams SM, Wadsworth R, Lebbie A. Knowledge, prevalence, and risk factors for self-reported sexually transmitted diseases among University students in Sierra Leone. Sierra Leone J Biomedical Res. 2023;14(2):81–91. [Google Scholar]
- 35.Wand H, Reddy T, Dassaye R, Moodley J, Naidoo S, Ramjee G. Estimating prevalence and incidence of sexually transmitted infections among South African women: implications of combined impacts of risk factors. Int J STD AIDS. 2020;31(11):1093–101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Labat A, Medina M, Elhassein M, Karim A, Jalloh MB, Dramaix M, Zhang W-H, Alexander S, Dickson KE. Contraception determinants in youths of Sierra Leone are largely behavioral. Reproductive Health. 2018;15(1):66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sserwanja Q, Turimumahoro P, Nuwabaine L, Kamara K, Musaba MW. Association between exposure to family planning messages on different mass media channels and the utilisation of modern contraceptives among young women in Sierra Leone: insights from the 2019 Sierra Leone Demographic Health Survey. BMC Women’s Health. 2022;22(1):376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Beksinska M, Wong R, Smit J. Male and female condoms: their key role in pregnancy and STI/HIV prevention. Mod Methods Contracept. 2020;66:55–67. [DOI] [PubMed] [Google Scholar]
- 39.Marfatia YS, Pandya I, Mehta K. Condoms: past, present, and future. Indian J Sexually Transmitted Dis AIDS. 2015;36(2):133–9. [DOI] [PMC free article] [PubMed]
- 40.James PB, Osborne A, Bah AJ, Margao EK, Conteh-Barrat M. Sexual risk behaviour among school-going adolescents in Sierra Leone and Liberia: a secondary analysis of the 2017 global school-based student health surveys. Contracept Reproductive Med. 2022;7(1):27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ueda P, Mercer CH, Ghaznavi C, Herbenick D. Trends in frequency of sexual activity and number of sexual partners among adults aged 18 to 44 years in the US, 2000–2018. JAMA Netw Open. 2020;3(6):e203833–203833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dagnew GW, Asresie MB, Fekadu GA. Factors associated with sexually transmitted infections among sexually active men in Ethiopia. Further analysis of 2016 Ethiopian demographic and health survey data. PLoS ONE. 2020;15(5):e0232793. [DOI] [PMC free article] [PubMed]
- 43.Marcus U, Mirandola M, Schink SB, Gios L, Schmidt AJ. Changes in the prevalence of self-reported sexually transmitted bacterial infections from 2010 and 2017 in two large European samples of men having sex with men–is it time to re-evaluate STI-screening as a control strategy?. PLoS ONE. 2021;16(3):e0248582. [DOI] [PMC free article] [PubMed]
- 44.Regushevskaya E, Dubikaytis T, Laanpere M, Nikula M, Kuznetsova O, Karro H, Haavio-Mannila E, Hemminki E. The determinants of sexually transmitted infections among reproductive age women in St. Petersburg, Estonia and Finland. Int J Public Health. 2010;55(6):581–9. [DOI] [PubMed] [Google Scholar]
- 45.Winston SE, Chirchir AK, Muthoni LN, Ayuku D, Koech J, Nyandiko W, Carter EJ, Braitstein P. Prevalence of sexually transmitted infections including HIV in street-connected adolescents in western Kenya. Sex Transm Infect. 2015;91(5):353–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Brima N, Burns F, Fakoya I, Kargbo B, Conteh S, Copas A. Factors associated with HIV prevalence and HIV testing in Sierra Leone: findings from the 2008 demographic health survey. PLoS ONE. 2015;10(10):e0137055. [DOI] [PMC free article] [PubMed]
- 47.Springer YP, Samuel MC, Bolan G. Socioeconomic gradients in sexually transmitted diseases: A geographic information system–based analysis of poverty, race/ethnicity, and gonorrhea rates in California, 2004–2006. American J Public Health. 2010;100(6):1060–7. [DOI] [PMC free article] [PubMed]
- 48.Harling G, Subramanian SV, Bärnighausen T, Kawachi I. Socioeconomic disparities in sexually transmitted infections among young adults in the United States: examining the interaction between income and race/ethnicity. Sexually Transmitted Dis. 2013;40(7):575–81. [DOI] [PMC free article] [PubMed]
- 49.Rahman M, Nakamura K, Seino K, Kizuki M. Intimate partner violence and symptoms of sexually transmitted infections: are the women from low socio-economic strata in Bangladesh at increased risk. Int J Behav Medicine. 2014;21:348–57. [DOI] [PubMed]
- 50.Coffee M, Lurie MN, Garnett GP. Modelling the impact of migration on the HIV epidemic in South Africa. Aids. 2007;21(3):343–50. [DOI] [PubMed]
- 51.Tiruneh K, Wasie B, Gonzalez H. Sexual behavior and vulnerability to HIV infection among seasonal migrant laborers in Metema district, northwest Ethiopia: a cross-sectional study. BMC Public Health. 2015;15:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sathiyasusuman A. Associated risk factors of STIs and multiple sexual relationships among youths in Malawi. PLoS ONE. 2015;10(8):e0134286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Montgomery MC, Raifman J, Nunn AS, Bertrand T, Uvin AZ, Marak T, et al. Insurance coverage and utilization at a sexually transmitted disease clinic in a Medicaid expansion state. Sexually Transmitted Dis. 2017;44(5):313–7. [DOI] [PMC free article] [PubMed]
- 54.Poudel KC, Poudel-Tandukar K, Palmer PH, Mizoue T, Jimba M, Kobayashi J, Acharya B, Pandey BD, Oka S. Coinfection of sexually transmitted infections among HIV-Positive Individuals: cross-sectional results of a community-based positive living with HIV (POLH) study in Nepal. J Int Association Providers AIDS Care (JIAPAC). 2017;16(4):338–46. [DOI] [PubMed] [Google Scholar]
- 55.Joseph F, Jean Simon D, Kondo Tokpovi VC, Kiragu A, Toudeka MR, Nazaire R. Trends and factors associated with recent HIV testing among women in Haiti: a cross-sectional study using data from nationally representative surveys. BMC Infect Dis. 2024;24(1):74. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data used for this study is freely available at https://dhsprogram.com/data/dataset/Sierra-Leone_Standard-DHS_2019.cfm?flag=0.