Abstract
Diabetic peripheral neuropathy (DPN), a common complication of diabetes mellitus, is primarily characterized by damage to Schwann cells caused by oxidative stress under hyperglycemic conditions. Recently, we demonstrated the ability of coumarin-rich Ficus formosana Maxim. to alleviate DPN in ovariectomized diabetic mice. However, the underlying mechanisms remain unclear. In this study, we established an in vitro DPN model using RSC96 Schwann cells exposed to high glucose levels. Daphnetin, a natural coumarin found abundantly in Ficus formosana Maxim., was co-incubated with Schwann cells in a high-glucose medium to investigate its protective effects against DPN. The free radical scavenging capacity of daphnetin was evaluated, along with assessments of cell viability, apoptosis, H2O2 levels, and the expression of proteins by the nuclear factor erythroid 2-related factor 2 (Nrf2)/glutamate–cysteine ligase catalytic subunit (GCLC) pathway in RSC96 Schwann cells. The results showed that daphnetin was non-toxic within the tested concentration range of 6.25 μM to 50 μM in RSC96 Schwann cells. Moreover, daphnetin significantly improved cell viability, exhibited strong antioxidant activity, reduced H2O2 levels, and regulated the Nrf2/GCLC pathway protein expressions in RSC96 cells cultured in high-glucose medium. Additionally, daphnetin influenced apoptosis-related proteins by decreasing the expression levels of Bax and Caspase 3, while increasing the Bcl-2 expression level in high-glucose-treated RSC96 cells. These findings suggest that daphnetin may alleviate oxidative stress induced by high glucose levels through activation of the Nrf2/GCLC pathway and inhibition of Schwann cell apoptosis, underscoring its potential as a therapeutic agent for DPN.
Keywords: diabetic peripheral neuropathy, Schwann cells, Ficus formosana Maxim., daphnetin, Nrf2, RSC96 cells, Bcl-2, Bax, apoptosis
1. Introduction
Diabetes mellitus (DM) is a chronic metabolic disorder characterized by hyperglycemia and is associated with numerous complications. Diabetic peripheral neuropathy (DPN) is one of the most common complications, affecting approximately half of all diabetic patients worldwide [1]. DPN is a debilitating condition that results in sensory loss, pain, and a higher risk of foot ulcers and amputations [1]. Schwann cells, the main glial cells of the peripheral nervous system, play a crucial role in nerve regeneration and repair by maintaining axonal integrity and promoting myelination [2,3]. Under hyperglycemic conditions, Schwann cells are highly vulnerable to oxidative stress, leading to apoptosis and impaired nerve function [2,4].
Oxidative stress, driven by the overproduction of reactive oxygen species (ROS), is a key contributor to the development of DPN [4]. The cellular imbalance between ROS and the antioxidant defenses results in mitochondrial dysfunction, lipid peroxidation, and ultimately Schwann cell death [1,5,6]. The nuclear factor erythroid 2-related factor 2 (Nrf2) pathway plays a crucial role in regulating the cellular antioxidant defense system [7]. When activated, Nrf2 enhances the expression of several antioxidant enzymes, including the glutamate–cysteine ligase catalytic subunit (GCLC), which protect Schwann cells from oxidative damage [8,9,10].
In our previous research, we demonstrated that coumarin-rich Ficus formosana Maxim., a traditional medicine used in the treatment of DM in the Orient, alleviated peripheral neuropathy by reducing oxidative damage in ovariectomized diabetic mice [6]. Coumarin is a group of natural compounds abundant in plants, known for their various biological activities, including antioxidant [11], anti-inflammatory [12], and anticancer properties [13]. Daphnetin (Figure 1), a coumarin derivative, has been shown to modulate oxidative stress responses in cell models [10]. Additionally, daphnetin has been reported to activate Nrf2 expression and its downstream antioxidant targets, providing neuroprotective effects against oxidative stress in vitro [8,10]. Given the severe side effects of conventional drugs, there is an urgent need to explore natural therapeutic strategies for DM and its complications, such as DPN. However, the potential of daphnetin to prevent DPN remains inconclusive. This study aims to investigate the therapeutic potential of daphnetin in protecting Schwann cells from high-glucose-induced oxidative injury by modulating the Nrf2/GCLC signaling pathway.
Figure 1.
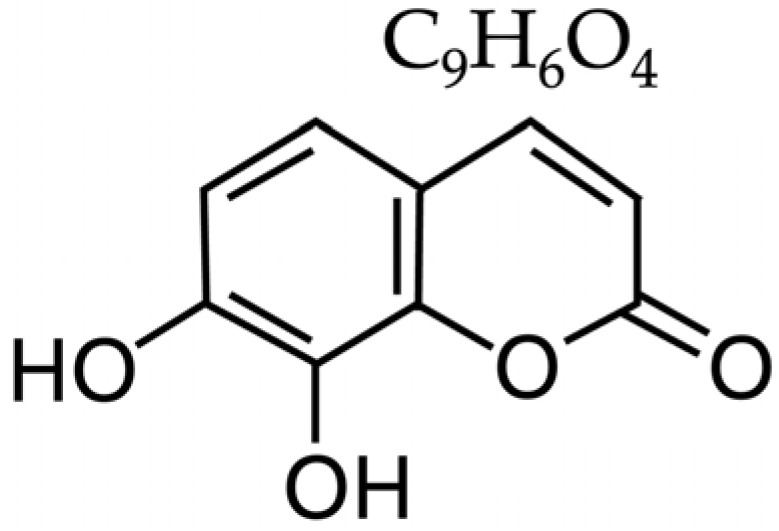
The chemical structure of daphnetin.
2. Results
2.1. Effect of Daphnetin on RSC96 Schwann Cells’ Viability Under High-Glucose Conditions
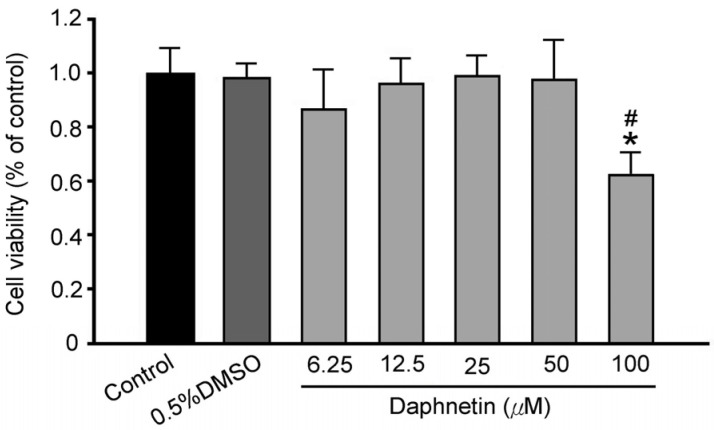
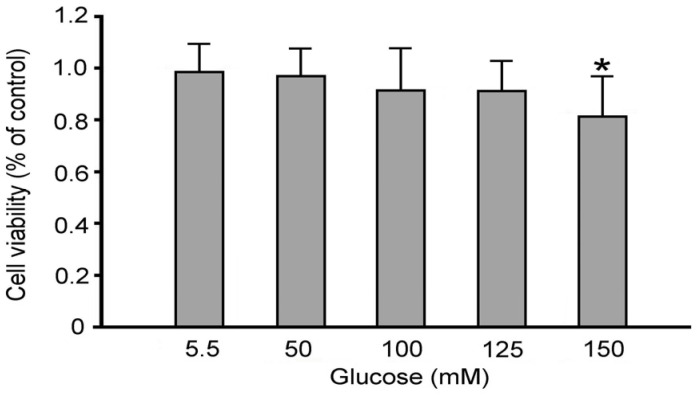
The results demonstrated that the maximum non-toxic concentration of daphnetin for RSC96 Schwann cells was 50 μM (Figure 2), which was subsequently used in further experiments. Increasing glucose concentrations resulted in a progressive reduction in cell viability. This concentration was then used in further experiments in this study. Additionally, increasing glucose concentrations resulted in a progressive decrease in cell viability. Various glucose concentrations (5.5 mM, 50 mM, 100 mM, 125 mM, and 150 mM) were used to simulate both normal and hyperglycemic conditions in RSC96 Schwann cells. Cell viability decreased by 17% at 150 mM (high glucose, HG group) compared to 5.5 mM (normal glucose, NG group) (Figure 3), and thus 150 mM glucose was used in subsequent experiments.
Figure 2.
Effect of daphnetin concentration on cell viability of RSC96 Schwann cells. * p < 0.05 compared to the NG (5.5 mM glucose) group; # p < 0.05 compared to the NG + DMSO group.
Figure 3.
Effect of glucose concentration on cell viability of RSC96 Schwann cells. * p < 0.05 as compared to the NG (5.5 mM glucose) group.
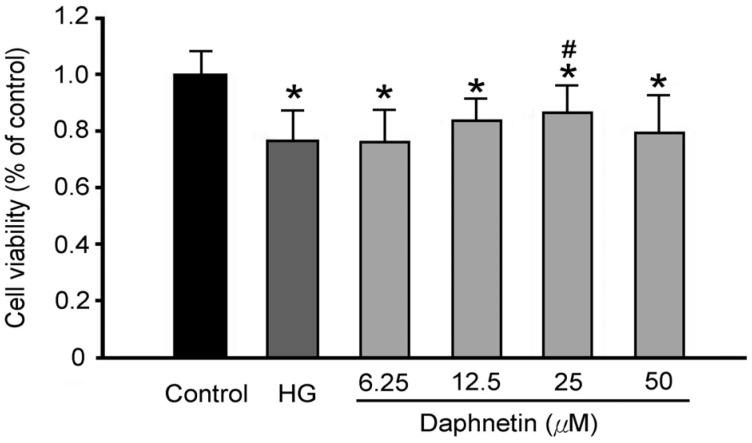
Figure 4 shows that, compared to the NG group, the viability of RSC96 cells in the HG group was significantly reduced, suggesting that a high-glucose environment could damage Schwann cells and even cause their death. However, daphnetin significantly increased cell viability by 7% compared to the HG group, indicating that daphnetin may reduce Schwann cell damage in a high-glucose environment.
Figure 4.
Effect of daphnetin concentration on cell viability of RSC96 Schwann cells incubated in a high-glucose medium. * p < 0.05 compared to the NG (5.5 mM glucose) group; # p < 0.05 compared to the HG (150 mM glucose) group.
2.2. Free Radical Scavenging Ability of Daphnetin
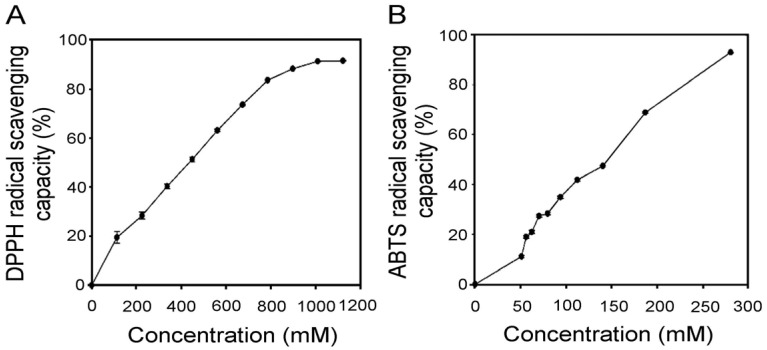
The 2,2-Diphenyl-1-picrylhydrazyl (DPPH) assay was used to evaluate the hydrogen-donating ability of the antioxidants, and daphnetin had an IC50 value of 435.4 mM for scavenging DPPH free radicals (Figure 5A). This indicates that daphnetin has a strong hydrogen proton-donating ability. In addition, the ABTS assay showed an IC50 of 146.1 mM for daphnetin (Figure 5B), indicating its excellent antioxidant capacity.
Figure 5.
Effects of daphnetin on radical scavenging capacity of (A) DPPH and (B) ABTS in RSC96 Schwann cells incubated in a high-glucose medium. Data are expressed as mean ± SD from triplicate experiments.
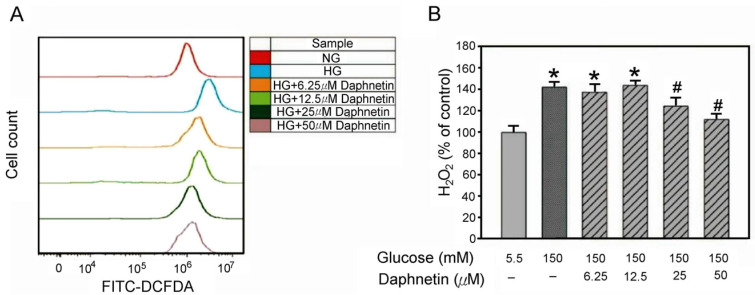
2.3. Impact of Daphnetin on Hydrogen Peroxide (H2O2) Content of RSC96 Schwann Cells Under High-Glucose Conditions
Generally, 2′, 7′-dichlorodihydrofluorescein diacetate (DCFH-DA) is used to measure the H2O2 content in “live” cells at different time points in order to avoid errors caused by apoptotic cells. In this study, the highest concentration of H2O2 in Schwann cells in a high-glucose environment occurred during the sixth hour. Figure 6 shows that high glucose levels increase oxidative stress in Schwann cells, as evidenced by the higher H2O2 content in the HG group when compared to the NG group. In contrast, daphnetin at a concentration of 25 μM reduced H2O2 levels by around 20% compared to the HG group (Figure 6A,B), demonstrating its inhibiting ability on H2O2 production in RSC96 Schwann cells in a high-glucose medium.
Figure 6.
Effect of daphnetin on H2O2 levels in RSC96 cells incubated in a high-glucose medium. Fluorescence intensity measured by flow cytometry (A) and Quantitative analysis of fluorescence intensity (B). * p < 0.05 compared to the NG (5.5 mM glucose) group; # p < 0.05 compared to the HG (150 mM glucose) group.
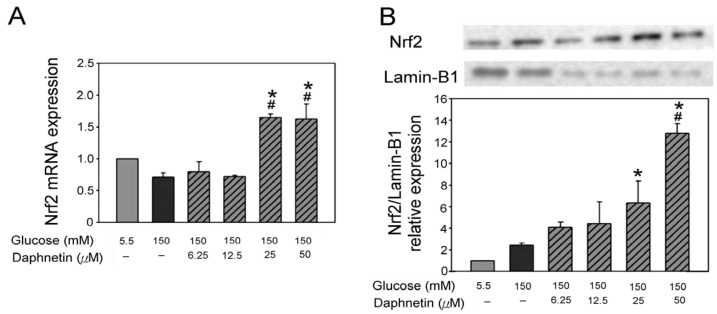
2.4. Regulation of Nrf2 Expression in RSC96 Schwann Cells by Daphnetin Under High-Glucose Conditions
The Nrf2 mRNA levels of RSC96 cells cultured with daphnetin in high-glucose conditions are shown in Figure 6A. The results revealed that 25 μM and 50 μM daphnetin significantly increased Nrf2 mRNA levels by 96% and 94%, respectively, compared to the HG group (Figure 7A). Moreover, it was observed that 50 μM daphnetin significantly increased nuclear Nrf2 expression by 6-fold compared to the HG group (Figure 7B).
Figure 7.
Nrf2 mRNA expression (A) and nuclear Nrf2 protein expression (B) in RSC96 cells incubated in a high-glucose medium with daphnetin. * p < 0.05 compared to the NG (5.5 mM glucose) group; # p < 0.05 compared to the HG (150 mM glucose) group.
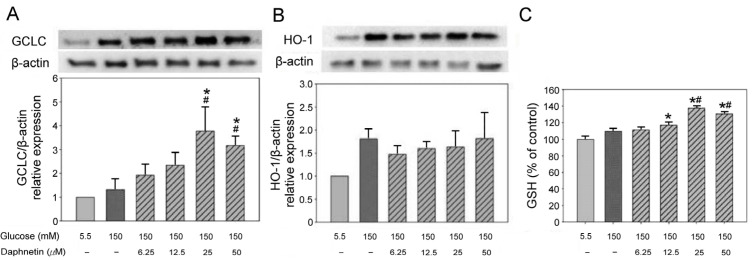
2.5. Regulation of Antioxidant Proteins and Glutathione (GSH) Levels in RSC96 Schwann Cells by Daphnetin Under High-Glucose Conditions
The protein expression of GCLC and heme oxygenase-1 (HO-1) in RSC96 Schwann cells was determined by co-culture with daphnetin in a high-glucose medium. The results showed that 25 μM and 50 μM daphnetin increased GCLC expression by 84% and 38% (Figure 8A), respectively; however, no significant difference was observed for HO-1compared to the HG group (Figure 8B). In addition, 25 μM and 50 μM daphnetin significantly increased GSH levels compared to the HG group (Figure 8C).
Figure 8.
Protein expression of GCLC (A), HO-1 (B) and GSH content (C) in RSC96 Schwann cells incubated in a high-glucose medium with daphnetin. * p < 0.05 compared to the NG (5.5 mM glucose) group; # p < 0.05 compared to the HG (150 mM glucose) group.
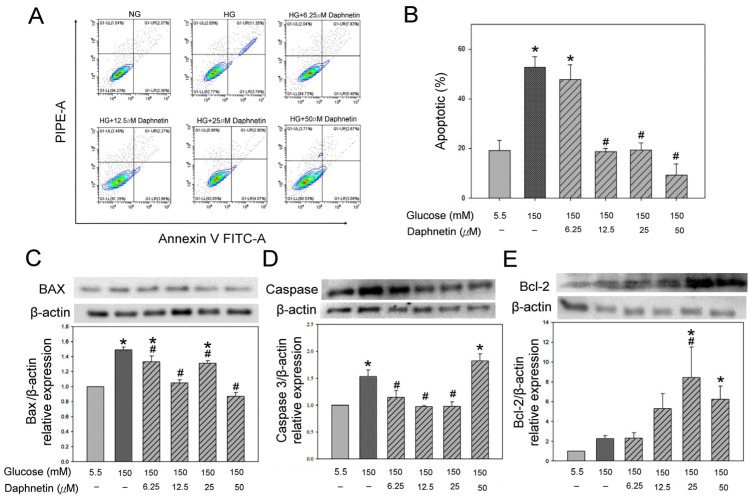
2.6. Regulation of Apoptosis of RSC96 Schwann Cells by Daphnetin Under High-Glucose Conditions
Figure 9A,B shows that the HG group significantly increased apoptosis 2.7-fold; however, 12.5 μM, 25 μM, and 50 μM daphnetin significantly reduced cell apoptosis by 60–80% compared to the NG group. In addition, the protein expression of Bax and Caspase 3 in the HG group was found to be approximately 50% higher than that in the NG group (Figure 9C,D). However, 6.25–50 μM and 6.25–25 μM daphnetin significantly inhibited Bax expression by 10–40% and Caspase 3 expression by 25–35%, respectively, in RSC96 Schwann cells compare to the HG group (Figure 9C,D). Moreover, daphnetin also significantly increased the level of anti-apoptotic protein Bcl-2 protein expression in Schwann cells compared to the HG group (Figure 9E). These results suggested that daphnetin inhibits the pro-apoptotic proteins Bax and Caspase 3, and increases the anti-apoptotic protein Bcl-2 in high-glucose-induced apoptosis in Schwann cells.
Figure 9.
Fluorescence intensity measurement by flow cytometry (A), quantitative analysis (B), protein expression of Bax (C), Caspase 3 (D) and Bcl-2 (E) in RSC96 Schwann cells incubated in a high-glucose medium with daphnetin. * p < 0.05 compared to the NG (5.5 mM glucose) group; # p < 0.05 compared to the HG (150 mM glucose) group.
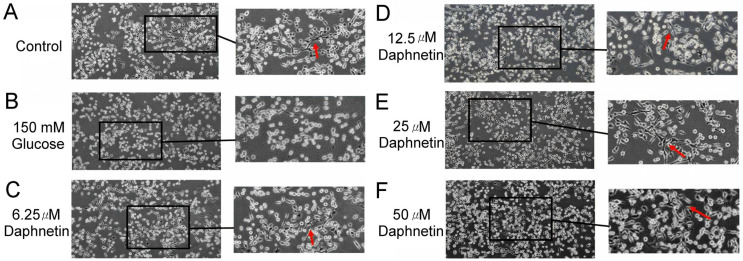
2.7. Daphnetin-Induced Morphological Alterations of RSC96 Schwann Cells in High-Glucose Environments
In this study, the RSC96 Schwann cells remained oval-shaped in the HG group, indicating that a high-glucose medium impairs the differentiation of cells (Figure 10B). However, a spindle-shaped or bipolar-shaped cell morphology was observed after co-culturing with daphnetin in a high-glucose medium (Figure 10C–F), suggesting that daphnetin promoted Schwann cells differentiation in a high-glucose environment.
Figure 10.
Effect of daphnetin on the morphology of RSC96 Schwann cells incubated in a high-glucose medium (A) control, (B) 150 mM glucose, (C–F) 6.25-50 μM daphnetin. (100× magnification, arrows denote spindle-shaped cells).
3. Discussion
Our study demonstrates that daphnetin significantly protects Schwann cells from high glucose-induced oxidative stress and apoptosis by modulating the Nrf2/GCLC signaling pathway. These findings are consistent with previous research, which underscores the role of oxidative stress in DPN and the critical function of the Nrf2 pathway in reducing oxidative damage.
Previous studies have shown that the progression of DPN is associated with poor blood glucose control and a long duration of diabetes [14]. In patients with type 2 DM, the prevalence of DPN exceeds 19% after 5–10 years of disease [14]. Excess oxidative stress caused by high blood glucose levels plays a key role in the development of DPN [4]. Phytochemicals from plants, including vegetables and herbs, have been identified for their strong antioxidant properties [11]. Coumarin, recognized for its distinctive aroma, is widely used as a food flavoring agent following its synthesis [15]. In humans, coumarin is metabolized by the enzyme 7-hydroxylase in liver microsomes into 7-hydroxycoumarin, which is then conjugated and excreted in urine [16].
This study investigated the effects of daphnetin, a natural coumarin derivative, on the viability of RSC96 Schwann cells under high-glucose conditions. While standard culture medium contains approximately 5.5 mM glucose [17], we observed that cell viability decreased as glucose concentrations increased, consistent with previous findings [2]. However, daphnetin improved Schwann cell viability in these high-glucose environments. In DPN models, Schwann cells have been cultured in glucose concentrations ranging from 44.4 to 150 mM to induce diabetic neuropathy [8,17,18,19,20,21]. In the present study, we used a high glucose concentration of 150 mM to simulate DPN in Schwann cells, similar to previous studies.
Schwann cells are essential for maintaining peripheral nerve function, and oxidative stress plays a significant role in their dysfunction under hyperglycemic conditions [4]. This study also revealed that daphnetin possesses strong antioxidant properties. Both the DPPH and ABTS assays confirmed daphnetin’s potent antioxidant capacity. Notably, daphnetin effectively reduced oxidative stress markers, such as H2O2, indicating its protective effect on RSC96 Schwann cells incubated in high-glucose conditions. This antioxidant capacity may be linked to the hydroxyl groups present in the chemical structure of daphnetin [22]. Moreover, this study demonstrated that daphnetin exerts its protective effects by activating the Nrf2 pathway, which plays a crucial role in mitigating oxidative stress and promoting Schwann cell survival [9]. Nrf2 is a transcription factor that, upon translocation from the cytoplasm to the nucleus, regulates the expression of antioxidant enzymes. In this study, the increased expression of Nrf2 mRNA and elevated nuclear Nrf2 protein levels resulted in the upregulation of GCLC, promoting intracellular GSH synthesis and effectively reducing oxidative damage under high-glucose conditions [8,23]. The observed increase in GSH levels and reduction in H2O2 are consistent with findings that enhancing the antioxidant response via the Nrf2 pathway improves cell viability and reduces oxidative stress markers [24]. Additionally, the significant decrease in H2O2 levels suggests that the antioxidant effects of daphnetin are mediated through the Nrf2/GCLC pathway, with no significant changes in HO-1 expression. These findings highlight the potential of daphnetin in counteracting oxidative stress related to DPN.
Oxidative stress is known to overwhelm cellular antioxidant defenses, leading to apoptosis, a hallmark of DPN [1,5,6]. Under hyperglycemic conditions, Schwann cells are particularly vulnerable to apoptosis driven by excessive ROS production [25], with excess H2O2 contributing to the apoptosis of these cells [26]. To assess this, we used flow cytometry and Western blotting to measure the extent of apoptosis in RSC96 cells and the expression of apoptosis-related proteins, including Bax, Caspase 3, and Bcl-2. It is well established that Bcl-2 is negatively associated with apoptosis, while Bax and Caspase 3 are positively associated with it [27]. In this study, daphnetin was found to modulate the expression of these apoptosis-related proteins, reducing pro-apoptotic Bax and Caspase 3 levels while increasing the anti-apoptotic Bcl-2 levels in Schwann cells exposed to high-glucose conditions. These findings are consistent with previous research demonstrating similar protective effects against hyperglycemia-induced oxidative damage in Schwann cells [17].
Normally, RSC96 Schwann cells exhibit an oval shape before differentiation, transitioning to a spindle-shaped or bipolar morphology after differentiation [28]. High-glucose-induced damage to RSC96 Schwann cells often results in morphological changes, such as swelling and vacuolation, which lead to the destruction of organelles [29]. The results of the current study suggest that daphnetin not only inhibits apoptosis but also improves cell morphology and enhances the proliferation of Schwann cells cultured in a high-glucose environment.
This study has several limitations. In vitro experiments may not fully replicate the complexity of Schwann cells in the in vivo environment in diabetic patients. Future research should build on these findings by exploring additional cell lines to better understand cellular responses under diabetic conditions, as well as conducting in vivo studies to evaluate the therapeutic potential of daphnetin in diabetic models. Furthermore, it is essential to assess the long-term effects and safety profiles of these compounds in clinical settings if they are to be developed as therapeutic agents.
4. Materials and Methods
4.1. Cell Culture
The RSC96 rat Schwann cell line was purchased from the Food Industry Research and Development Institute (Hsinchu, Taiwan). The cells were cultured in Dulbecco’s Modified Eagle Medium (Thermo Fisher, Waltham, MA, USA) containing 10% fetal bovine serum (FBS; Wellington, CO, USA) and 1% non-essential amino acids (Beit-Haemek, Israel), and maintained at 37 °C in a 5% CO2 incubator.
4.2. Treatment and Viability Assay
RSC96 Schwann cells were incubated in media containing 5.5, 50, 100, 125, and 150 mM glucose, 0.5% DMSO medium contain 150 mM glucose with fresh prepared 6.25, 12.5, 50, 100, 125, and 150 μM daphnetin (C9H6O4; Purity ≥ 97%; Sigma-Aldrich, St. Louis, MO, USA), respectively, for 24 h. For the viability assay, cells were treated with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT; Sigma-Aldrich, St. Louis, MO, USA) for 4 h, and then analyzed spectrophotometrically at 517 nm. Viability was determined as a percentage of control cells treated with the vehicle alone.
4.3. DPPH Radical Scavenging Activity
The DPPH radical scavenging assay was used to evaluate the antioxidant activity of the samples [30]. A 0.1 mM solution of DPPH (Sigma-Aldrich, St. Louis, MO, USA) in methanol was prepared. Samples were mixed with the DPPH solution and incubated in the dark for 30 min. The absorbance was measured at 517 nm.
4.4. ABTS Radical Scavenging Activity
The ABTS radical cation decolorization assay was used to measure the ability of antioxidants to scavenge ABTS [30]. ABTS was generated by mixing 7 mM ABTS (Sigma-Aldrich, St. Louis, MO, USA) with 2.45 mM potassium persulfate and allowing the mixture to react in the dark for 12–16 h. The ABTS solution was then diluted to an absorbance of 0.7 at 734 nm. Samples were mixed with the ABTS solution, incubated for 6 min in the dark, and the absorbance was measured at 734 nm.
4.5. GSH Content Determination
GSH levels were measured using a colorimetric assay. Cells were treated with various concentrations of samples. After incubation, cells were lysed, and GSH levels were determined by reacting cell lysates with 5,5′-dithiobis-(2-nitrobenzoic acid) (Sigma-Aldrich, St. Louis, MO, USA) and measuring the absorbance at 412 nm.
4.6. H2O2 Content Determination
Intracellular H2O2 levels were measured using DCFH-DA (Sigma-Aldrich, St. Louis, MO, USA). Cells were incubated with DCFH-DA, which is converted to the fluorescent compound dichlorodihydrofluorescein in the presence of H2O2. The fluorescence intensity was measured using a flow cytometer with excitation at 485 nm and emission at 535 nm [31].
4.7. Cell Apoptosis Analysis
Apoptosis was assessed using the Annexin V-FITC apoptosis detection kit (Dojindo, Munich, Germany), followed by flow cytometry. Cells were stained with annexin V-FITC and propidium iodide (PI; Thermo Fisher Scientific, Waltham, MA, USA) to distinguish between live, apoptotic, and necrotic cells. The percentage of apoptotic cells was analyzed using flow cytometry.
4.8. Real-Time Quantitative PCR Analysis
Total RNA was extracted from cells using the TRIzol reagent (Zymo Research, Irvine, CA, USA). cDNA was synthesized using a reverse transcription kit (Protech, Taipei, Taiwan). Real-time quantitative PCR was performed using specific primers for target genes and the SYBR Green master mix. Primer sequences of Nrf2 and β-actin are (5′-GTACAACCCTTGTCACCATCTC-3′, 5′-TCCGATGACCAGGACTTACA-3′ and 5′-CCGTCTTCCCCTCCATCG-3′, 5′-GTCCCAGTTGGTGACGATGC-3′, respectively. The relative expression levels of genes were calculated using the ΔΔCt method with β-actin as the internal control.
4.9. Western Blot Analysis
The protein extraction methods used in this study were based on our previous protocol [32]. The protein concentration in the cell extract was determined using a Bio-Rad protein assay dye reagent (Richmond, VA, USA). Aliquots of the supernatant protein were separated by standard SDS-PAGE and then electrophoretically transferred to polyvinylidene difluoride membranes. The membranes were incubated overnight at 4 °C with the following primary antibodies: anti-Nrf2 (1:1000, Cat. No. 33649, Cell Signaling Technology), anti-caspase-3 (1:1000, Cat. No. 9662, Cell Signaling Technology, Danvers, MA, USA), anti-HO-1 (1:1000, Cat. No. 82206, Cell Signaling Technology, Danvers, MA, USA), anti-Lamin-b1 (1:1000, Cat. No. 13435, Cell Signaling Technology), anti-GCLC (1:2000, Cat. No. ab190685, Abcam, Cambridge, UK), anti-Bcl-2 (1:1000, Cat. No. ab182858, Abcam), anti-Bax (1:200, Cat. No. ab32503, Abcam), or anti-β-actin (1:1000, Cat. No. ab8226, Abcam, Cambridge, UK). Subsequently, the membranes were incubated with anti-mouse IgG or anti-rabbit IgG secondary antibodies and washed three times for 5 min each. Protein bands were detected and captured using the UVP Biospectrum imaging system (Level, Cambridge, UK). Finally, all relative protein expressions were normalized to β-actin.
4.10. Statistical Analysis
The experimental data were analyzed using SPSS version 22 (SPSS Inc., Chicago, IL, USA). The means and standard errors of each group were calculated, and one-way ANOVA was used to determine significant differences between groups. Post hoc tests were performed using Duncan’s multiple range test to identify significant differences. Statistical significance was defined as p < 0.05.
5. Conclusions
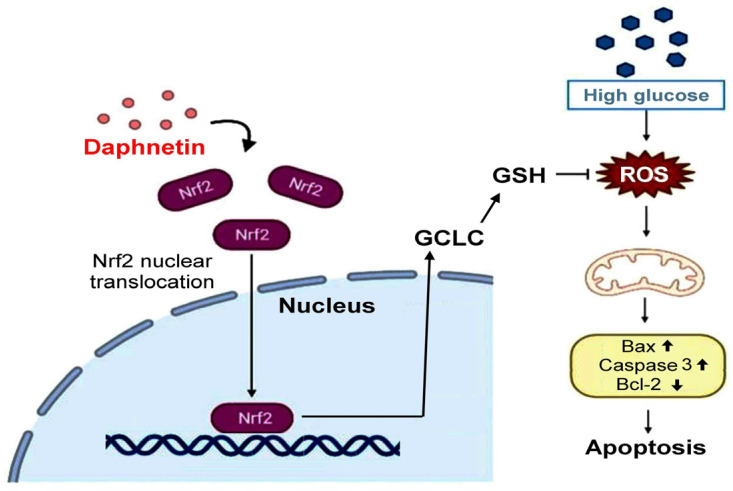
This study highlights the protective effects of daphnetin, a natural coumarin derivative, on RSC96 Schwann cells exposed to high-glucose conditions. Daphnetin facilitated the nuclear translocation of Nrf2, increased the expression of antioxidant proteins such as GCLC and GSH but not HO-1, and consequently reduced oxidative stress damage in the Schwann cells. Additionally, daphnetin inhibited apoptosis by regulating the expression of Bax, Caspase 3, and Bcl-2 proteins through the suppression of oxidative stress in Schwann cells (Figure 11). In summary, daphnetin may mitigate oxidative damage caused by hyperglycemia in DM and shows potential as a therapeutic option for managing DPN.
Figure 11.
The possible mechanism of daphnetin in reducing high-glucose-induced Schwann cell apoptosis.
Acknowledgments
The authors thank anonymous reviewers for very helpful comments on a draft of the paper.
Author Contributions
Conceptualization, C.-Y.K. and S.-C.S.; Methodology, R.-T.M., C.-H.W. and Y.-E.C.; Software, Y.-E.C. and C.-H.W.; Validation, R.-T.M. and Y.-E.C.; Formal Analysis, Y.-E.C., T.K.N.N. and C.-Y.K.; Investigation, Y.-E.C.; Resources, T.K.N.N., J.S.-B.W. and S.-C.S.; Data Curation, Y.-E.C. and S.-C.S.; Writing—Original Draft Preparation, C.-Y.K., W.-C.H. and S.-C.S.; Writing—Review and Editing, C.-Y.K., T.K.N.N., W.-C.H. and S.-C.S.; Visualization, Y.-E.C., C.-H.W. and C.-Y.K.; Supervision, S.-C.S.; Project Administration, S.-C.S.; Funding Acquisition, C.-Y.K. and S.-C.S. All authors have read and agreed to the published version of the manuscript.
Data Availability Statement
Data are contained within the article.
Conflicts of Interest
The authors declare that they do not have any conflicts of interest.
Funding Statement
Ministry of Science and Technology of the Republic of China (ROC) (No. NSTC 111-2320-B-003-005-MY3) and the Fujian Provincial Health and Family Planning Commission (Serial No. 2022GGA038).
Footnotes
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content.
References
- 1.Yang H., Sloan G., Ye Y., Wang S., Duan B., Tesfaye S., Gao L. New perspective in diabetic neuropathy: From the periphery to the brain, a call for early detection, and precision medicine. Front. Endocrinol. 2020;10:929. doi: 10.3389/fendo.2019.00929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tatsumi Y., Kato A., Sango K., Himeno T., Kondo M., Kato Y., Kamiya H., Nakamura J., Kato K. Omega-3 polyunsaturated fatty acids exert anti-oxidant effects through the nuclear factor (erythroid-derived 2)-related factor 2 pathway in immortalized mouse Schwann cells. J. Diabetes Investig. 2019;10:602–612. doi: 10.1111/jdi.12931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fallon M., Tadi P. StatPearls. StatPearls Publishing; Treasure Island, FL, USA: 2024. Histology, Schwann cells. [PubMed] [Google Scholar]
- 4.Wu L., Wang X.J., Luo X., Zhang J., Zhao X., Chen Q. Diabetic peripheral neuropathy based on Schwann cell injury: Mechanisms of cell death regulation and therapeutic perspectives. Front. Endocrinol. 2024;15:1427679. doi: 10.3389/fendo.2024.1427679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ko C.Y., Lin R.H., Lo Y.M., Chang W.C., Huang D.W., Wu J.S., Chang Y.F., Huang W.C., Shen S.C. Effect of Ruellia tuberosa L. on aorta endothelial damage-associated factors in high-fat diet and streptozotocin-induced type 2 diabetic rats. Food Sci. Nutr. 2019;7:3742–3750. doi: 10.1002/fsn3.1233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ko C.Y., Wu C.H., Nguyen T.K.N., Chen L.W., Wu J.S., Huang W.C., Shen S.C. Alleviative effect of Ficus formosana extract on peripheral neuropathy in ovariectomized diabetic mice. Plants. 2023;12:3774. doi: 10.3390/plants12213774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kaspar J.W., Niture S.K., Jaiswal A.K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic. Biol. Med. 2009;47:1304–1309. doi: 10.1016/j.freeradbiomed.2009.07.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yang X., Yao W., Shi H., Liu H., Li Y., Gao Y., Liu R., Xu L. Paeoniflorin protects Schwann cells against high glucose induced oxidative injury by activating Nrf2/ARE pathway and inhibiting apoptosis. J. Ethnopharmacol. 2016;185:361–369. doi: 10.1016/j.jep.2016.03.031. [DOI] [PubMed] [Google Scholar]
- 9.Sen Z., Weida W., Jie M., Li S., Dongming Z., Xiaoguang C. Coumarin glycosides from Hydrangea paniculata slow down the progression of diabetic nephropathy by targeting Nrf2 anti-oxidation and smad2/3-mediated profibrosis. Phytomedicine. 2019;57:385–395. doi: 10.1016/j.phymed.2018.12.045. [DOI] [PubMed] [Google Scholar]
- 10.Hassanein E.H.M., Sayed A.M., Hussein O.E., Mahmoud A.M. Coumarins as modulators of the Keap1/Nrf2/ARE signaling pathway. Oxidative Med. Cell. Longev. 2020;2020:1675957. doi: 10.1155/2020/1675957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pérez-Cruz K., Moncada-Basualto M., Morales-Valenzuela J., Barriga-González G., Navarrete-Encina P., Núñez-Vergara L., Squella J., Olea-Azar C. Synthesis and antioxidant study of new polyphenolic hybrid-coumarins. Arab. J. Chem. 2018;11:525–537. doi: 10.1016/j.arabjc.2017.05.007. [DOI] [Google Scholar]
- 12.Liu Y.P., Yan G., Xie Y.T., Lin T.C., Zhang W., Li J., Wu Y.J., Zhou J.Y., Fu Y.H. Bioactive prenylated coumarins as potential anti-inflammatory and anti-HIV agents from Clausena lenis. Bioorg. Chem. 2020;97:103699. doi: 10.1016/j.bioorg.2020.103699. [DOI] [PubMed] [Google Scholar]
- 13.Emami S., Dadashpour S. Current developments of coumarin-based anti-cancer agents in medicinal chemistry. Eur. J. Med. Chem. 2015;102:611–630. doi: 10.1016/j.ejmech.2015.08.033. [DOI] [PubMed] [Google Scholar]
- 14.Amthor K.F., Dahl-Jørgensen K., Berg T.J., Heier M.S., Sandvik L., Aagenaes O., Hanssen K.F. The effect of 8 years of strict glycaemic control on peripheral nerve function in IDDM patients: The Oslo study. Diabetologia. 1994;37:579–584. doi: 10.1007/BF00403376. [DOI] [PubMed] [Google Scholar]
- 15.Krüger S., Winheim L., Morlock G.E. Planar chromatographic screening and quantification of coumarin in food, confirmed by mass spectrometry. Food Chem. 2018;239:1182–1191. doi: 10.1016/j.foodchem.2017.07.058. [DOI] [PubMed] [Google Scholar]
- 16.Barot K.P., Jain S.V., Kremer L., Singh S., Ghate M.D. Recent advances and therapeutic journey of coumarins: Current status and perspectives. Med. Chem. Res. 2015;24:2771–2798. doi: 10.1007/s00044-015-1350-8. [DOI] [Google Scholar]
- 17.Dong J., Li H., Bai Y., Wu C. Muscone ameliorates diabetic peripheral neuropathy through activating AKT/mTOR signalling pathway. J. Pharm. Pharmacol. 2019;71:1706–1713. doi: 10.1111/jphp.13157. [DOI] [PubMed] [Google Scholar]
- 18.Yu Y., Ma X., Yang T., Li B., Xie K., Liu D., Wang G., Yu Y. Protective effect of hydrogen-rich medium against high glucose-induced apoptosis of Schwann cells in vitro. Mol. Med. Rep. 2015;12:3986–3992. doi: 10.3892/mmr.2015.3874. [DOI] [PubMed] [Google Scholar]
- 19.Kusano K., House S.B., Gainer H. Effects of osmotic pressure and brain-derived neurotrophic factor on the survival of postnatal hypothalamic oxytocinergic and vasopressinergic neurons in dissociated cell culture. J. Neuroendocrinol. 1999;11:145–152. doi: 10.1046/j.1365-2826.1999.00296.x. [DOI] [PubMed] [Google Scholar]
- 20.Wang P.Y., Liang X.C., Zhang H., Zhao L., Sun Q., Huang W.Z., Li B.W. Effect of serum containing Jinmaitong Capsule on rats’ Schwann cell apoptosis induced by high glucose concentration. Chin. J. Integr. Med. 2013;19:517–523. doi: 10.1007/s11655-013-1506-7. [DOI] [PubMed] [Google Scholar]
- 21.Tiong Y.L., Ng K.Y., Koh R.Y., Ponnudurai G., Chye S.M. Melatonin Prevents Oxidative Stress-Induced Mitochondrial Dysfunction and Apoptosis in High Glucose-Treated Schwann Cells via Upregulation of Bcl2, NF-κB, mTOR, Wnt Signalling Pathways. Antioxidants. 2019;8:198. doi: 10.3390/antiox8070198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Annunziata F., Pinna C., Dallavalle S., Tamborini L., Pinto A. An overview of coumarin as a versatile and readily accessible scaffold with broad-ranging biological activities. Int. J. Mol. Sci. 2020;21:4618. doi: 10.3390/ijms21134618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Simpson T., Pase M., Stough C. Bacopa monnieri as an antioxidant therapy to reduce oxidative stress in the aging brain. Evid. Based Complement. Alternat. Med. 2015;2015:615384. doi: 10.1155/2015/615384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Albert-Garay J.S., Riesgo-Escovar J.R., Salceda R. High glucose concentrations induce oxidative stress by inhibiting Nrf2 expression in rat Müller retinal cells in vitro. Sci. Rep. 2022;12:1261. doi: 10.1038/s41598-022-05284-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zhu L., Hao J., Cheng M., Zhang C., Huo C., Liu Y., Du W., Zhang X. Hyperglycemia-induced Bcl-2/Bax-mediated apoptosis of Schwann cells via mTORC1/S6K1 inhibition in diabetic peripheral neuropathy. Exp. Cell Res. 2018;367:186. doi: 10.1016/j.yexcr.2018.03.034. [DOI] [PubMed] [Google Scholar]
- 26.He B., Tao H.Y., Liu S.Q. Neuroprotective effects of carboxymethylated chitosan on hydrogen peroxide induced apoptosis in Schwann cells. Eur. J. Pharmacol. 2014;740:127–134. doi: 10.1016/j.ejphar.2014.07.008. [DOI] [PubMed] [Google Scholar]
- 27.Fröhlich M., Jaeger A., Weiss D.G., Kriehuber R. Inhibition of BCL-2 leads to increased apoptosis and delayed neuronal differentiation in human ReNcell VM cells in vitro. Int. J. Dev. Neurosci. 2016;48:9. doi: 10.1016/j.ijdevneu.2015.10.004. [DOI] [PubMed] [Google Scholar]
- 28.Liu D., Liang X., Zhang H. Effects of high glucose on cell viability and differentiation in primary cultured Schwann cells: Potential role of ERK signaling pathway. Neurochem. Res. 2016;41:1281–1290. doi: 10.1007/s11064-015-1824-6. [DOI] [PubMed] [Google Scholar]
- 29.Chung Y.C., Lim J.H., Oh H.M., Kim H.W., Kim M.Y., Kim E.N., Kim Y., Chang Y.S., Kim H.W., Park C.W. Calcimimetic restores diabetic peripheral neuropathy by ameliorating apoptosis and improving autophagy. Cell Death Dis. 2018;9:1163. doi: 10.1038/s41419-018-1192-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ko C.Y., Chen X.Y., Chang W.C., Zeng Y.M., Lin R.H., Zhang X.B., Wu J.S., Shen S.C. Effects of Maillard reaction products in a glucose-glycine alcoholic solution on antioxidative and antimutagenic activities. J. Sci. Food Agric. 2018;98:5242–5247. doi: 10.1002/jsfa.9062. [DOI] [PubMed] [Google Scholar]
- 31.Xin X., Gong T., Hong Y. Hydrogen peroxide initiates oxidative stress and proteomic alterations in meningothelial cells. Sci. Rep. 2022;12:14519. doi: 10.1038/s41598-022-18548-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ko C.Y., Xu J.H., Chang Y.W., Lo Y.M., Wu J.S., Huang W.C., Shen S.C. Effects of α-lipoic acid on phagocytosis of oligomeric beta-amyloid1-42 in BV-2 mouse microglial cells. Front. Aging Neurosci. 2022;13:788723. doi: 10.3389/fnagi.2021.788723. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data are contained within the article.