Abstract
Most addiction research has focused on reward- and impulsivity-related neurocircuitry. However, the impact of the withdrawal/negative affect stage in the addiction cycle has been somewhat overlooked, despite it being commonly evident in the clinic. This stage crucially drives negative reinforcement of repeated drug use and relapse, yet less is known about its neural underpinnings. How negative emotional processing is dysregulated in substance dependence is incompletely understood and may manifest differentially across the types of substances. In turn, the regions involved in negative emotional processing may show different patterns of dysregulation. Understanding how neurocircuitry involved in negative states differs across various substances may help inform new targets for treatments. Following a comprehensive literature search of studies examining negative emotional processing in substance dependence, a quantitative approach was deemed inappropriate. Instead, we employed a narrative approach to exploring neural responses to tasks involving emotional processing in alcohol, cocaine, opioid and cannabis dependence. Regions that were found to be dysregulated included the amygdala, insula, anterior cingulate, and medial prefrontal cortex. However, patterns of reactivity differed across alcohol, cocaine, opioid and cannabis dependence. Brain activation in alcohol dependence broadly appeared blunted in response to negative affective stimuli and emotional faces, whilst conversely appeared heightened in cocaine dependence. In opioid dependence, the amygdala was consistently implicated, whilst the insula, anterior cingulate, and medial prefrontal cortex were implicated in cannabis dependence. However, there was wide variability amongst the studies, with very few studies investigating opioid and cannabis dependence. These findings suggest emotional dysregulation varies according to the type of substance dependence. However, the variability in findings and lack of studies highlights the need for more research in this area. Further characterisation of emotional dysregulation in substance dependence will enable identification of treatment targets. More targeted treatments that modulate negative emotional processing could substantially improve outcomes by aiding relapse prevention.
Subject terms: Neuroscience, Addiction, Prognostic markers
Introduction
Overview
Whilst there has been substantial research about the neurobiology of addiction over the last 20 years, translation into widespread clinical use has been more limited [1, 2]. Addiction has been conceptualised as consisting of three stages that drive repeated drug use, including binge/intoxication, withdrawal/negative affect and preoccupation/anticipation [3]. The focus of addiction research has largely been on dopaminergic reward- and impulsivity-related neurocircuitry implicated in the stages of binge/intoxication and preoccupation/anticipation. There is less evidence from clinical populations about the neurobiological underpinnings of the withdrawal/negative affect stage. Clinically, persistent addiction is often driven by negative states such as emotional dysregulation and stress. Whilst there is preclinical evidence about approaches to attenuate stress-related relapse in alcohol dependence, clinical trials in humans lacked efficacy [4, 5]. A greater understanding of the neurocircuitry involved in negative states in clinical populations would therefore inform development of new approaches and identification of new targets for treatment [6, 7]. Initially, we sought to conduct a quantitative review of the literature concerning negative emotional processing in substance dependence. However, following a comprehensive search of the literature, the limited number of studies precluded a meaningful meta-analysis. Therefore, the aim of this review is to provide a narrative overview of fMRI research that examines brain reactivity patterns during negative emotional processing in addiction during treatment and/or abstinence; identify key regions that are dysregulated during these responses; and identify the gaps in our knowledge to highlight useful future research.
Emotional dysregulation is central to the chronic relapsing nature of addiction
Koob and Le Moal [8] refer to the term hyperkatifeia, ‘derived from katifeia for dejection or negative emotional state’, which can be used to describe the greater intensity of negative emotional and motivational states during withdrawal [9]. This experience of heightened negative emotional states can be further enhanced through genetic vulnerability, environmental factors, and comorbid psychiatric diagnoses. Individuals suffering from pre-existing heightened stress, anxiety or anhedonia are at greater risk of developing substance dependences when drugs are used to self-medicate negative emotions [10]. Together, these factors contribute to compulsive drug use through negative reinforcement to alleviate negative affect, which in turn may further result in compensatory neuroadaptations and allostasis.
Preclinical evidence has shown the important role of the extended amygdala in repeated drug use to alleviate negative affect during withdrawal [11]. The extended amygdala is a key hub that receives inputs from other brain regions involved in emotional processing, including the anterior insula, anterior cingulate cortex (ACC), and medial prefrontal cortex (mPFC); Fig. 1; [3]. The ACC is involved in cognitive inhibition and motivational drive [12], and the anterior insula is involved in the conscious urge to take drugs and integrating interoceptive signals into emotional and motivational responses [13]. The mPFC has similar roles, including motivation, reward-related decision-making, and appraisal of emotional stimuli [14, 15]. Prefrontal regions exhibit top-down control of subcortical regions, allowing for modulation of processes such as emotional regulation and impulse control [16].
Fig. 1. Emotional processing is governed by brain regions including the extended amygdala, insula, ACC, and mPFC.
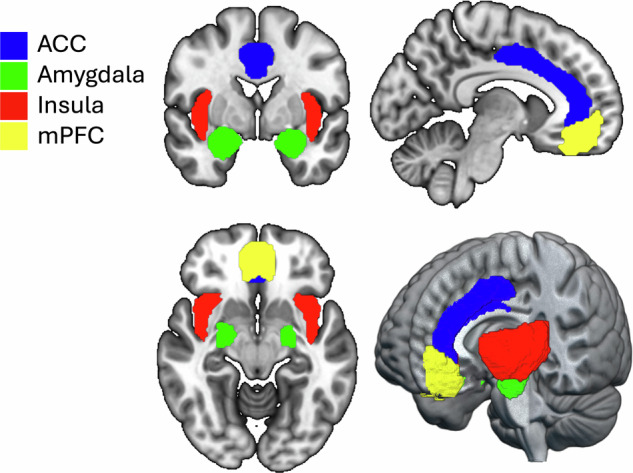
Abbreviations: ACC anterior cingulate cortex, mPFC medial prefrontal cortex. Image created with MRIcroGL [https://www.nitrc.org/projects/mricrogl].
Neuroimaging for addiction research
Neuroimaging is fundamental for characterising the structure and function of the brain. fMRI allows images of the brain to be captured whilst participants perform specific tasks inside a scanner. Changes in blood oxygenation indirectly indicate increases or decreases in brain activity, known as the blood-oxygen-level-dependent (BOLD) response.
A variety of neuroimaging tasks have been developed to probe different aspects of emotional processing. Tasks can be passive, such as passively viewing emotional faces or aversive images, or active, such as requiring conscious emotion regulation strategies, like reappraisal. These tasks can be modified further depending on the aspect of emotional processing being investigated. For example, tasks can generalised or individualised, or include distractor tasks like letter discrimination that require additional cognitive strategies. Together, examining the outcomes of studies utilising a variety of fMRI techniques in emotional processing may help characterise dysregulation in brain regions in substance dependence.
Methods
This review is a non-systematic narrative of task-based fMRI studies investigating negative emotional processing and emotional dysregulation in humans with alcohol, cocaine, opioid and cannabis dependence (terminology used throughout). The search strategy for publications involved PubMed searches for terms including combinations and variations of “addiction”, “dependence”, “fMRI”, “aversive”, “emotional processing”, “negative emotional processing”, “emotional dysregulation”, “relapse”, “stress”, “anxiety”, “alcohol”, “cocaine”, “opioid”, “opiate”, “heroin”, “cannabis”, “marijuana”. Additional papers were identified from citations in other reviews and studies. For inclusion in the current review, studies must have utilised task-based fMRI (including task-based functional connectivity) examining negative emotional processing in alcohol, cocaine, opioid or cannabis dependence.
A total of 24 studies were found exploring brain responses to a range of emotionally salient stimuli (see Tables 1, 2, 3 and 4). The majority of studies were in alcohol (n = 11) and cocaine (n = 6) with few in cannabis (n = 3) and opioids (n = 4). Tables 2 and 3 contain the same information but organised differently, either according to substance (Table 2) or by task category. This is to enable easier reference to the effect of drug class or the breadth of tasks utilised.
Table 1.
Overview of studies included.
| First author, date | Study location | Participant dependence status | N (dependent) |
|---|---|---|---|
| Alcohol, n = 11 | |||
| Alba-Ferrara 2016 | California, USA | Early (<12 months) or sustained (>12 months) remission | 10 |
| Charlet 2014 | Mannheim, Germany | 4–24 days abstinent | 33 |
| Gilman 2008 | Maryland, USA | 3 weeks abstinent | 12 |
| Jansen 2019 | Amsterdam, Netherlands | Minimum 3 weeks abstinent | 39 |
| Kim 2014 | Seoul, South Korea | 3–21 days abstinent | 38 |
| Marinkovic 2009 | Massachusetts, USA | 4 weeks abstinent | 15 |
| O’Daly 2012 | London, UK | Minimum 2 weeks abstinent | 29 (12 MSDs) |
| Salloum 2007 | Maryland, USA | Mean 28 days since last drink ± 15 days | 11 |
| Seo 2013 | Connecticut, USA | 4–8 weeks abstinent | 45 |
| Zakiniaiez 2017 | Connecticut, USA | 4–6 weeks abstinent (mean 34 days) | 45 |
| Yang 2013 | Texas, USA | 3–5 weeks abstinent | 15 |
| Cocaine, n = 6 | |||
| Albein-Urios 2014 | Grenada, Spain | Minimum 15 days abstinent | 17 |
| Asensio 2010 | Valencia, Spain | 72 hours abstinent | 32 |
| Canterberry 2016 | USA (unspecified) | Not stated | 20 |
| Potenza 2012 | Connecticut, USA | Minimum 2 weeks abstinent | 30 |
| Sinha 2005 | Connecticut, USA | Minimum 2 weeks abstinent | 20 |
| Xu 2013 | Connecticut, USA | 2–4 weeks in treatment | 67 |
| Opioids, n = 4 | |||
| Schmidt 2014 | Basel, Switzerland | Maintained on heroin for at least 6 months | 22 |
| Schmidt 2015 | Basel, Switzerland | Maintained on heroin for at least 6 months | 22 (as above) |
| Wang 2010 | Shantou, China | Abstinent 2–5 months | 17 |
| Smoski 2011 | Washington, USA | Maintained on buprenorphine/naloxone for average 14.8 weeks | 12 |
| Cannabis, n = 3 | |||
| Wesley 2016 | Kentucky, USA | Abstinent the day of scanning | 16 |
| Wetherill 2014 | Pennsylvania, | Self-reported abstinence 1.5 ± 1.5 days | 20 |
| Zimmermann 2018 | Bonn, Germany | At least 28 days abstinent | 19 |
Abbreviations: MSD medically supervised detoxification, USA United States of America.
Table 2.
Overview of tasks and contrasts by substance.
| Author, date | Task/Stimulus | Contrasts |
|---|---|---|
| Alcohol | ||
| Alba-Ferrara 2016 | Alcohol-Emotion-Picture fMRI paradigm | alcoholic > non-alcoholic beverages, angry > happy faces, sad > happy faces, angry > sad faces |
| Charlet 2014 | Implicit facial information processing task | aversive faces > neutral shapes |
| Gilman 2008 | High-arousal negative and positive images paired with alcoholic or non-alcoholic cues | negative non-alcohol > positive non-alcohol |
| Jansen 2019 | Emotion reappraisal task | attend > reappraise, reappraise > attend, attend alcohol > attend neutral, attend negative > attend neutral, attend positive > attend neutral |
| Kim 2014 | Craving-inducing cues (CIC) and aversion-inducing cues (AIC) | AIC > control, CIC > control |
| Marinkovic 2009 | Emotional (negative, positive, neutral) words and faces encoding and recognition | faces > words, deep > shallow, negative > positive > neutral |
| O’Daly 2012 | Facial expression recognition task (fear) | fear > neutral |
| Salloum 2007 | Facial emotion-decoding task | high emotional facial expression > baseline, high emotional facial expression > low emotional facial expression |
| Seo 2013 | Individualised script-driven imagery of stressful personal experiences | stress > neutral-relaxing, alcohol > neutral-relaxing |
| Zakiniaiez 2017 | Individualised script-driven imagery of stressful personal experiences | alcohol > neutral, stress > neutral |
| Yang 2013 | CS paired with uncertain, physically painful unconditioned stressor | high threat > low threat |
| Cocaine | ||
| Albein-Urios 2014 | Negative emotion re-appraisal task | maintain (negative) > observe (neutral), suppress (negative) > maintain (negative) |
| Asensio 2010 | Letter discrimination task with affective pictures as background | unpleasant > neutral, neutral > unpleasant, pleasant > unpleasant, unpleasant > pleasant, pleasant > neutral, neutral > pleasant |
| Canterberry 2016 | Passive viewing of high-arousal negative and positive and emotionally neutral standardised images | positive > neutral, negative > eutral |
| Potenza 2012 | Individualised script-driven imagery of drug, stress, and neutral situations | stress > neutral, drug > neutral |
| Sinha 2005 | Guided imagery and stress recall | personal stress > personal neutral |
| Xu 2013 | Personalised script-driven imagery | stress > neutral, drug > neutral |
| Opioids | ||
| Schmidt 2014 | Fearful faces processing | 100% fearful faces > neutral faces, 50% fearful faces > neutral faces |
| Schmidt 2015 | Fearful faces processing | 100% fearful faces > neutral faces, 50% fearful faces > neutral faces |
| Wang 2010 | Passive viewing of standardised affective pictures | neutral > rest, negative > neutral, positive > neutral |
| Smoski 2011 | Emotional oddball task | negative > neutral distractor |
| Cannabis | ||
| Wesley 2016 | Evaluation of emotional (i.e., emotional evaluation) or neutral (i.e., neutral evaluation) stimuli | positive + negative > neutral, positive > neutral, negative > neutral |
| Wetherill 2014 | Exposure to cannabis, sexual, and aversive cues presented in a backward-masking paradigm | sexual > neutral, cannabis > sexual, aversive > neutral, cannabis > aversive |
| Zimmermann 2018 | Passive viewing of negative, positive, and neutral standardised images | negative > neutral, positive > neutral |
AIC aversion-inducing cues, CIC craving-inducing cues, CS conditioned stimulus, fMRI functional magnetic resonance imaging.
Table 3.
Overview of tasks and contrasts by category.
| Author, date | Drug | Task/Stimulus | Contrasts |
|---|---|---|---|
| Facial emotions | |||
| Alba-Ferrara 2016 | Alcohol | Alcohol-Emotion-Picture fMRI paradigm | alcoholic > non-alcoholic beverages, angry > happy faces, sad > happy faces, angry > sad faces |
| Charlet 2014 | Alcohol | Implicit facial information processing task | aversive faces > neutral shapes |
| O’Daly 2012 | Alcohol | Facial expression recognition task (fear) | fear > neutral |
| Salloum 2007 | Alcohol | Facial emotion-decoding task | high emotional facial expression > baseline, high emotional facial expression > low emotional facial expression |
| Marinkovic 2009 | Alcohol | Emotional (negative, positive, neutral) words and faces encoding and recognition | faces > words, deep > shallow, negative > positive > neutral |
| Schmidt 2014 | Opioids | Fearful faces processing | 100% fearful faces > neutral faces, 50% fearful faces > neutral faces |
| Schmidt 2015 | Opioids | Fearful faces processing | 100% fearful faces > neutral faces, 50% fearful faces > neutral faces |
| Affective images | |||
| Gilman 2008 | Alcohol | High-arousal negative and positive images paired with alcoholic or non-alcoholic cues | negative non-alcohol > positive non-alcohol |
| Kim 2014 | Alcohol | Craving-inducing cues (CIC) and aversion-inducing cues (AIC) | AIC > control, CIC > control |
| Wetherill 2014 | Cannabis | Exposure to cannabis, sexual, and aversive cues presented in a backward-masking paradigm | sexual > neutral, cannabis > sexual, aversive > neutral, cannabis > aversive |
| Zimmermann 2018 | Cannabis | Passive viewing of negative, positive and neutral standardised images | negative > neutral, positive > neutral |
| Asensio 2010 | Cocaine | Letter discrimination task with affective pictures as background | unpleasant > neutral, neutral > unpleasant, pleasant > unpleasant, unpleasant > pleasant, pleasant > neutral, neutral > pleasant |
| Canterberry 2016 | Cocaine | Passive viewing of high-arousal negative and positive and emotionally neutral standardised images | positive > neutral, negative > neutral |
| Wang 2010 | Opioids | Passive viewing of standardised affective pictures | neutral > rest, negative > neutral, positive > neutral |
| Emotion regulation (reappraisal or evaluation) | |||
| Jansen 2019 | Alcohol | Emotion reappraisal task | attend > reappraise, reappraise > attend, attend alcohol > attend neutral, attend negative > attend neutral, attend positive > attend neutral |
| Wesley 2016 | Cannabis | Evaluation of emotional (i.e., emotional evaluation) or neutral (i.e., neutral evaluation) stimuli | positive + negative > neutral, positive > neutral, negative > neutral |
| Albein-Urios 2014 | Cocaine | Negative emotion re-appraisal task | maintain (negative) > observe (neutral), suppress (negative) > maintain (negative) |
| Script-driven imagery | |||
| Seo 2013 | Alcohol | Individualised script-driven imagery of stressful personal experiences | stress > neutral-relaxing, alcohol > neutral-relaxing |
| Zakiniaiez 2017 | Alcohol | Individualised script-driven imagery of stressful personal experiences | alcohol > neutral, stress > neutral |
| Potenza 2012 | Cocaine | Individualised script-driven imagery of drug, stress and neutral situations | stress > neutral, drug > neutral |
| Sinha 2005 | Cocaine | Guided imagery and stress recall | personal stress > personal neutral |
| Xu 2013 | Cocaine | Personalised script-driven imagery | stress > neutral, drug > neutral |
| Other | |||
| Yang 2013 | Alcohol | CS paired with uncertain, physically painful unconditioned stressor | high threat > low threat |
| Smoski 2011 | Opioids | Emotional oddball task | negative > neutral distractor |
AIC aversion-inducing cues, CIC craving-inducing cues, CS conditioned stimulus, fMRI functional magnetic resonance imaging.
Table 4.
Overview of main effects and regions implicated.
| Author/date | Overall effect* | Regions* (WB vs ROI) |
|---|---|---|
| Alcohol | ||
| Alba-Ferrara 2016 | Alcohol dependent < controls |
dlPFC, mPFC, aPFC, left putamen, left anterior insula (activation) hippocampus (connectivity) |
| Charlet 2014 | Alcohol dependent < controls |
left rACC, bilateral fusiform gyrus (ROI) right middle frontal gyrus, right inferior parietal gyrus, left cerebellum (WB) No difference amygdala (ROI) |
| Gilman 2008 | Negative images > positive images |
amygdala (VOI) bilateral insula, bilateral inferior frontal gyrus, left lingual gyrus, right parahippocampal gyrus, right medial temporal gyrus (WB) |
| Jansen 2019 | Alcohol dependent < controls |
posterior insula, precuneus, operculum, superior temporal gyrus (WB) No effect amygdala (ROI) |
| Kim 2014 | Alcohol dependent < controls |
right inferior temporal gyrus, bilateral inferior occipital gyrus (WB) right amygdala, right middle temporal gyrus, pons, left superior frontal gyrus (WB) |
| Marinkovic 2009 | Alcohol dependent < controls during emotional faces vs neutral | amygdala and hippocampus (ROI) |
| O’Daly 2012 |
Alcohol dependent < controls (activation) Repeated MSDs < fewer MSDs (connectivity) |
OFC and insula (activation) insula-amygdala and prefrontal-globus pallidus (connectivity) |
| Salloum 2007 |
Alcohol dependent < controls Alcohol dependent > controls |
rACC (fear, disgust, and sad), insula (disgust) insula (angry, happy, sad and fear) |
| Seo 2013 | Alcohol dependent < controls during stress trials | vmPFC, ventral ACC, insula, precuneus (WB) |
| Zakiniaiez 2017 | Alcohol dependent < controls during stress vs neutral | ventral ACC, MCC and PCC |
| Yang 2013 | Alcohol dependent < controls during high threat | pgACC, mPFC, PCC, bilateral parietal/occipital cortex, right hippocampus (WB) |
| Cocaine | ||
| Albein-Urios 2014 |
Cocaine dependent > controls in maintain vs observe Cocaine dependent < controls in suppress vs maintain |
right dlPFC and bilateral temporoparietal junction right inferior frontal gyrus, PCC, insula and fusiform gyrus |
| Asensio 2010 | Cocaine dependent < controls when pleasant pictures | dorsal and ventral striatum (including NAcc), thalamus, parietal cortex and dmPFC |
| Canterberry 2016 | Cocaine dependent < controls to positive and negative images | mPFC and ventral ACC |
| Potenza 2012 |
Cocaine dependent women > controls during stress and neutral-relaxing Cocaine dependent women > men during stress and neutral-relaxing |
amygdala, hippocampus, insula, ACC, vlPFC, vmPFC, dmPFC, dlPFC in women (WB) |
| Sinha 2005 |
Cocaine dependent > controls during stress Cocaine dependent < controls during stress |
caudate and dorsal striatum dACC, left hippocampal/parahippocampal region, right fusiform gyrus, right postcentral gyrus |
| Xu 2013 | Cocaine dependent with mutation > cocaine dependent without mutation during stress and cue vs neutral | right amygdala, hippocampus |
| Opioids | ||
| Schmidt 2014 | Opioid dependent > controls | left amygdala |
| Schmidt 2015 | Opioid dependent > controls | connectivity left fusiform gyrus-left amygdala, right amygdala-right OFC |
| Wang 2010 | Opioid dependent < controls to negative affective images | right amygdala |
| Smoski 2011 | Opioid dependent < controls to negative affective images | dACC and amygdala |
| Cannabis | ||
| Wesley 2016 | Cannabis group < controls | mPFC |
| Wetherill 2014 | Aversive cues > neutral cues (no group comparison) | left anterior insula and pgACC |
| Zimmermann 2018 | Cannabis dependent > controls to negative emotional stimuli |
mOFC (activation) mOFC-dorsal striatum, mPFC-amygdala (connectivity) |
ACC anterior cingulate cortex, aPFC anterior prefrontal cortex, BOLD blood-oxygen-level-dependent, dACC dorsal anterior cingulate cortex, dlPFC dorsolateral prefrontal cortex, dmPFC dorsomedial prefrontal cortex, MCC midcingulate gyrus, mOFC medial orbitofrontal cortex, MSD medically supervised detoxification, mPFC medial prefrontal cortex, NAcc nucleus accumbens, PCC posterior cingulate gyrus, pgACC pregenual anterior cingulate cortex, rACC rostral anterior cingulate cortex, OFC orbitofrontal cortex, ROI region of interest, vmPFC ventromedial prefrontal cortex, vlPFC ventrolateral prefrontal cortex, VOI volume of interest, WB whole brain.
Results and discussion
fMRI studies of emotional dysregulation in alcohol dependence
Facial emotion processing
In alcohol dependence, differences in brain responses were seen in tasks of facial emotion processing. Blunted ACC activity was observed in recently detoxified alcohol-dependent individuals in response to both fear and disgust compared with controls [17]. In line with this, higher rACC activation in recently detoxified alcohol-dependent patients in response to aversive faces vs neutral cues was associated with better treatment outcomes [18]. In the same study, blunted activation was also observed in the fusiform gyrus [18]. Lower prefrontal and left anterior insula activation, and less hippocampal connectivity, was observed in response to happy, sad, and angry faces in alcohol-dependent individuals during both early and sustained remission compared with controls [19]. Therefore, it is possible that blunting of brain activity in response to facial emotions may persist into longer-term abstinence and recovery.
In line with this, recently abstinent alcohol-dependent individuals compared with controls demonstrated deficits in recognising fearful faces in conjunction with lower insula and orbitofrontal cortex (OFC) activity [20]. In the same study, alcohol-dependent participants with a history of repeated detoxifications demonstrated lower insula and OFC activation and insula-amygdala connectivity compared with those who had fewer detoxification attempts [20]. Lower amygdala and hippocampal activation was also observed in response to emotional faces relative to neutral faces in recently detoxified long-term alcohol-dependent men, without the same repeated detoxification history, compared with controls [21]. However, another study found no differences in amygdala activity in response to negative emotional faces between recently detoxified inpatient alcohol-dependent men and controls [17].
Aversive stimuli
Blunted ACC and mPFC activity were observed in recently detoxified alcohol-dependent individuals in response to anticipatory anxiety and threat [22]. Similar blunting in ACC, midcingulate cortex (MCC) and posterior cingulate cortex (PCC) connectivity was seen in response to individualised stressful experience cues in recently detoxified alcohol-dependent individuals compared with healthy controls [23]. Blunted activation was also observed following exposure to individualised stressful experience cues relative to neutral in the vmPFC and ACC in recently-detoxified alcohol-dependent individuals [24]. This higher activation in the vmPFC and ACC during the neutral condition was correlated with high levels of stress-induced craving in the recently detoxified alcohol-dependent group [24]. Lower activation in the right amygdala was observed in a recently detoxified alcohol-dependent population compared with healthy controls in response to aversive images [25]. Right amygdala activity was also negatively correlated with severity of alcohol abuse [25]. Recently detoxified alcohol-dependent individuals demonstrated blunted activation in the posterior insula compared with controls whilst focusing on, i.e. ‘sustaining’, emotional images compared with neutral images in an emotion reappraisal task [26]. However, in response to high-arousal negative images in the absence of alcohol cues, recently detoxified alcohol-dependent individuals demonstrated heightened activation in the bilateral insula and amygdala compared with controls [27], in contrast to the aforementioned studies which mostly demonstrated blunted activity.
Summary
Together, these findings suggest that alcohol-dependent individuals demonstrate blunted activation to negative affective stimuli and negative emotional faces compared with controls. In response to negative emotional faces, alcohol-dependent individuals showed blunted activation in the ACC, insula, fusiform gyrus, hippocampus, OFC, and amygdala compared with controls, and reduced amygdala-insula and hippocampal connectivity. In line with this, the ACC, insula, and fusiform gyrus have previously been shown to be involved in facial emotion processing [28, 29]. Therefore, blunted responses in these regions may be associated with deficits in facial emotion recognition, and subsequent poorer treatment outcomes [18]. In response to aversive images or cues, alcohol-dependent individuals also demonstrated overall blunted cingulate connectivity, and blunted activation in the ACC, insula, mPFC, vmPFC, and amygdala. However, some results were conflicting, as heightened activation was also observed in the insula and amygdala. Overall, these results suggest that alcohol dependence may be associated with blunted corticolimbic activity, as the hypoactivation in these regions may cause deficits in facial emotion recognition and processing of negative emotional stimuli. Further studies should clarify whether deficits in facial emotion recognition only result in diminished recognition capabilities or whether this may lead to misinterpretation of emotions, which could have social implications. Furthermore, it is challenging to account for the impact of variation in abstinence length on brain activity during the tasks (Table 1), which could be more clearly elucidated through longitudinal studies.
fMRI studies of emotional dysregulation in cocaine dependence
Compared with alcohol, fewer fMRI studies have investigated emotional dysregulation in cocaine dependence, each using tasks of varying design. In a reappraisal task, recently abstinent cocaine-dependent individuals demonstrated higher activation in the bilateral dlPFC, left IFG and bilateral temporoparietal junction compared with healthy controls whilst ‘sustaining’ emotions elicited by negative affective images [30]. Whilst ‘suppressing’ the emotions via reappraisal, lower activation was observed in the insula, PCC, bilateral IFG and thalamus in the cocaine-dependent participants compared with healthy controls [30]. Higher right dlPFC-amygdala and insula-OFC connectivity was observed when ‘sustaining’ negative emotions, although lower right IFG-amygdala connectivity was observed during reappraisal [30].
In response to individualised imagery of stressful situations, recently detoxified cocaine-dependent women exhibited higher activation in the insula, ACC, PCC, and striatum compared with men [31], although the association with stress-induced cocaine craving is inconsistent. Higher activations were not found to be correlated with stress-induced cravings [31], although heightened dorsal striatum activity has previously been shown to be associated with stress-induced cocaine craving [32]. Furthermore, cocaine-dependent women compared with healthy women demonstrated higher activation in response to stress cue imagery in the amygdala, hippocampus, striatum, insula, ACC, and lPFC, vmPFC, dmPFC and dlPFC [31]. However, following a similar task of personalised stress situations, lower activation was observed in the ACC, left hippocampus and right fusiform gyrus of recently abstinent cocaine-dependent participants compared with controls [32, 33]. Blunted reactivity was also observed in the ACC and mPFC in non-treatment seeking cocaine-dependent individuals compared with controls in response to both positive and negative emotional stimuli [34]. Interestingly, blunting of activity was more pronounced in women compared with men [34]. However, in another study, no differences in activation were observed between cocaine-dependent individuals and healthy controls in response to negative versus neutral affective images [35].
Due to the heterogenous nature of the studies with a range of ‘addiction status’ and different tasks, it is challenging to ascertain the specific roles that each region plays during negative emotional processing in cocaine dependence. As the ACC is associated with control and regulation of emotion, lower activation in this area may be associated with diminished ability to exercise control during stressful scenarios, subsequently leading to craving and heightened risk of relapse. The disparity in these findings may be due to gender differences and treatment-seeking status. Participants in Potenza, Hong [31] were treatment seeking, thus possibly exerting more effortful control in response to the negative images compared with non-treatment seeking participants in Canterberry, Peltier [34], an effect which may be mediated by the ACC. Further, the abstinence length of the participants in Canterberry, Peltier [34] is unclear. It is possible this differs to the other reviewed studies, where most participants were abstinent for at least 2 weeks.
fMRI studies of emotional dysregulation in opioid dependence
All four neuroimaging studies investigating negative emotional processing in opioid dependence show evidence of amygdala dysregulation. In heroin-dependent individuals, left amygdala activity was higher in response to fearful faces compared with healthy controls [36]. This response was positively correlated with state anxiety levels, suggesting an association between amygdala hyperactivity and subjective anxiety. Furthermore, both state anxiety and amygdala hyperactivation were attenuated towards levels in healthy controls following acute heroin administration [36]. Increased fear-induced brain connectivity was observed in heroin-dependent individuals compared with healthy controls [37]. Likewise, this response was both positively correlated with state anxiety and attenuated following acute heroin administration [37]. Together, these findings suggest that increased amygdala-cortical connectivity and amygdala hyperactivity may underlie the heightened state anxiety and stress-related responses observed in the studies. In turn, these responses can be ‘normalised’ following acute heroin administration similar to healthy control levels [36, 37].
However, blunted amygdala and ACC reactivity was observed in opioid-dependent individuals maintained on buprenorphine/naloxone (suboxone) with comorbid borderline personality disorder (BPD) in response to negative stimuli compared with healthy controls [38]. Abstinent heroin-dependent individuals also demonstrated lower amygdala reactivity in response to aversive images compared with controls [39]. The divergent amygdala responses during these studies may be attributed to abstinence status of the participants, as participants were either abstinent from all opioids for 1 year [39]; currently on suboxone [38]; or dependent on heroin during the study [36, 37]. Further, it is challenging to disentangle whether the observed effect in Smoski, Salsman [38] is due to the presence of suboxone, as the study did not include a BPD group without suboxone. Together, the studies demonstrate distinct patterns of activity during different stages of opioid dependence treatment, which suggest that longer-term abstinence may help attenuate or normalise dysregulated brain reactivity to negative affective stimuli indicative of clinical benefit.
fMRI studies of emotional dysregulation in cannabis dependence
There have been only two neuroimaging studies examining negative emotional processing in cannabis dependence, and one in heavy use and/or dependence. A backward-masking paradigm was utilised by Wetherill et al. [40], whereby participants were briefly shown target cue images immediately followed by masking images, such that the target images are prevented from being consciously processed. In cannabis-dependent individuals, activation was observed during the task in the left anterior insula and pgACC in response to aversive vs neutral images [40]. In response to passive viewing of negative images, higher activation was observed in the mOFC in cannabis-dependent individuals compared with controls [41]. Higher mOFC-amygdala and mOFC-dorsal striatum connectivity was also observed [41]. However, in heavy cannabis users, where dependence status was unspecified, lower activation was seen in the mPFC, including portions of the ACC and vmPFC, during evaluation of emotional images relative to neutral [42]. Additionally, no activation was seen in the amygdala and IFG in cannabis users, which was observed in healthy controls. Hypoactivation of the mPFC was also observed compared with controls in response to both positive and negative emotional evaluation [42]. These results suggest that dependence status, i.e., the transition from heavy use to dependence, may cause functional adaptations in the brain. This may result in the differential brain responses observed, as greater activation in cortical areas was observed in the Zimmermann, Yao [41] dependent group, but not in the Wesley, Lile [42] heavy users group.
However, with such few studies it is not possible to determine the reliability of the dysregulated brain responses observed. For example, the study conducted by Wetherill, Childress [40] did not use a control group for comparison. Therefore, the activations observed in this population may not be different to non-users of cannabis. Furthermore, the participants in the studies were abstinent for different lengths of time, as the participants were either allowed to use cannabis up until the day of study [40, 42], or were abstinent for at least 28 days prior to the study [41]. These differences in abstinence length could result in differential responses due to brain adaptions occurring over the 28 day abstinence period.
Understanding the brain regions implicated in emotional dysregulation in addiction
Amygdala in addiction
Although amygdala hyperactivation is commonly demonstrated in neuroimaging studies investigating emotional processing in negative affective disorders [43, 44], this review found inconsistent amygdala activation in substance dependence. Amygdala dysregulation was reliably observed in the reviewed studies in alcohol dependence, although inconsistent in direction. Differential amygdala activation was observed between groups in response to negative images [27], but not to emotional faces [17, 18, 20] or high threat stimuli [22]. Together, these findings suggest that the type of emotional stimulus may affect the direction of the amygdala reactivity in those with alcohol dependence. In contrast, in cocaine dependence, amygdala hyperactivity was only observed in Potenza, Hong [31], suggesting other regions may be more centrally involved in the contribution to the emotional dysregulation and negative reinforcement observed in cocaine dependence. In Potenza, Hong [31], amygdala hyperactivity was observed in women compared with men, suggesting women with cocaine dependence may be more susceptible to amygdala dysregulation following the experience of stress. However, more studies are required to elucidate the impact of sex differences in cocaine dependence. In opioid dependence, dysregulation of amygdala activity was reliably observed across the reviewed studies. However, the nature of the dysregulation was inconsistent, as higher activity and connectivity were observed in response to emotional faces during heroin treatment [36, 37], but not in response to negative images during abstinence [39], or the emotional oddball task during suboxone treatment [38]. Therefore, the divergent results may be attributable to the varying abstinence statuses of the participants. As previously discussed, longer-term abstinence may help attenuate or normalise dysregulated brain reactivity to negative affective stimuli. Treatment with opioid agonists like heroin, such as in Schmidt, Borgwardt [36], Schmidt, Walter [37], or other OST, are also likely to have inhibitory effects in the amygdala due to the high expression of inhibitory mu opioid receptors. Further, the variety of tasks used are likely to engage different cognitive processes and thus recruit amygdala activity in differing ways.
Hypoactivity in the amygdala could be attributable to down-regulation by modulatory ACC/mPFC networks during tasks requiring more cognitive effort [45]. For example, there was no amygdala activation during emotion reappraisal in alcohol dependence [26]. This is in line with neuroimaging research, which demonstrates heightened activity in prefrontal areas alongside reduced activity in the amygdala [46]. As prefrontal regions are involved in emotion regulation and the amygdala is considered emotion generating, lack of amygdala activity during an emotion regulation task is expected. Additionally, blunted amygdala responses may be due to habituation following repeated exposure of participants to negative emotional stimuli [16, 47], such as in Wang, Zhang [39]. However, in people with alcohol use disorder and adverse childhood experiences, a lack of habituation in the amygdala was observed in response to negative emotional faces [48]. The results suggest repeated exposure could cause sensitisation to negative affective stimuli, which may be mediated by neuroplastic alterations in the amygdala [49]. These changes may perpetuate the dysphoria and anhedonia that drive negative reinforcement [49].
Anterior cingulate cortex in addiction
The ACC is a key region implicated in addiction, as the neural networks mediating emotion regulation and cognitive control are dysregulated. In the studies currently reviewed, individuals with substance dependence generally demonstrated dampened ACC activity during tasks of emotional processing and regulation [17, 22–24, 32, 34, 38]. Together, these findings suggest that regulatory control governed by the ACC may be compromised in dependence, resulting in impaired emotional regulation. Impaired top-down control of emotion generating regions such as the amygdala may manifest as heightened impulsivity and craving. This effect likely occurs following exposure to conditioned cues including emotional states like stress, which subsequently triggers craving characteristic of the preoccupation/anticipation stage [3]. In line with this, reduced ACC activity is observed during tasks of attention and inhibitory control in users of cocaine, heroin, alcohol and cannabis [14]. This impairment in inhibitory control subsequently heightens impulsivity and contributes to relapse [3]. Modulating the rACC, considered the emotional hub of the ACC, may prevent craving caused by emotional states, by ameliorating hypohedonia or negative affect, thus preventing the transition to the preoccupation/anticipation stage [12].
Prefrontal cortex in addiction
Multiple roles governed by the PFC are dysregulated in dependence during negative emotional processing and contribute to the negative reinforcement observed in addiction. As previously described, in alcohol dependence, lower activation was observed compared with controls in the dlPFC, mPFC and anterior portions of the PFC in response to emotional faces [19], and in the OFC in response to fearful faces [20]. These subregions are collectively involved in emotion regulation and cognitive control [50], characterised by increased activity in response to negative affective stimuli to downregulate emotion generating regions like the amygdala [51]. Given its role in cognitive appraisal, diminished activity in the dlPFC suggests impairments in evaluating and interpreting emotional facial stimuli. Indeed, reduced activity in the dlPFC is associated with impairments in negative facial emotion processing in people with mood disorders [52], suggesting similar patterns of emotion dysregulation in people with alcohol dependence. Connections of the OFC and mPFC to the amygdala are known for modulating social cognition and emotional responses [53, 54]. Thus, lower activity in the mPFC and OFC in alcohol dependence may indicate impairments in the ability to process emotional faces due to their aberrant connectivity with the amygdala [55, 56]. Lower vmPFC activity was observed in response to stress imagery, which was predictive of heavier drinking after relapse [24]. In line with this, blunted vmPFC response to stress in young healthy adults was also associated with increased binge drinking and emotional dysregulation [57]. Given its role in integrating affective valuations made by regions like the amygdala [58], diminished activity in the vmPFC is indicative of more dysregulated emotional processing, which may confer greater risk of relapse, heavy drinking, and treatment failure [24, 59, 60].
In opioid dependence, increased amygdala-OFC connectivity was observed under placebo in heroin-maintained dependent individuals compared with controls in response to fearful faces, which was reduced by heroin treatment [61]. Amygdala-OFC connectivity is a known connection mediating anxiety responses [53, 54], suggesting that heroin dependence may disrupt the normal connectivity of this response and increase anxiety responses to fearful faces in the absence of heroin, which is restored following heroin treatment [61]. Similarly, amygdala-OFC connectivity and OFC activity was heightened in cannabis dependence in response to negative affective images [41], and reduced mPFC activity was also observed in long-term heavy cannabis users during evaluation of emotional stimuli [42]. The patterns of emotional dysregulation in cannabis dependence are similar to that of alcohol dependence, suggesting cannabis and alcohol may recruit similar neural pathways that subsequently become dysregulated with repeated use.
In cocaine dependence, higher activation in the dlPFC and higher dlPFC-amygdala connectivity was observed during reappraisal [30], whilst hyperactivation was observed in the dmPFC, dlPFC, vmPFC, and vlPFC in in response to stress imagery [31]. These results suggest that people with cocaine dependence demonstrate increased sensitisation to negative emotions compared with controls and subsequent dysregulation of the associated emotional state [30]. This is in contrast to alcohol dependence, where prefrontal activity was blunted in response to negative affective stimuli and facial emotions. Interestingly, in polydrug users with primary cocaine dependence, activity was lower in the mPFC in response high arousal negative and positive images compared with controls [34]. As prefrontal activity diminished in polydrug dependence, it is possible that heightened prefrontal activity is facilitated by purer cocaine use, whilst the dampening of its activity may be due to additional drugs like alcohol and cannabis. Together, the results suggest that different drugs of dependence may lead to variance in PFC functioning, disrupting a variety of emotional regulation processes.
Insula cortex in addiction
In anxiety disorders, the role of the insula in perceiving the internal state of feeling anxious can become maladaptive and manifest as heightened subjective anxiety [10, 62]. However, this effect is less consistently observed within substance dependence. The results of the studies reviewed showed inconsistent patterns of insula reactivity and connectivity during negative emotional processing. Insula activity was heightened during tasks involving passive viewing of aversive stimuli in alcohol [27, 31, 40], and dampened during tasks of reappraisal in cocaine [26, 30]. During emotion reappraisal in the healthy population, the insula consistently demonstrates reduced activity [16, 46, 63], which suggests downregulation by regions like the ACC and mPFC [16, 45, 63]. Across all substances, reactivity of the insula was consistent depending on the type of task, i.e. heightening of insula reactivity during passive viewing of aversive stimuli and dampening of insula reactivity during reappraisal. This suggests that the type of task used, and the subsequent cognitive process employed, may be the driving factor in modulating insula activity, rather than the prolonged use of the drug of dependence.
Tasks of facial emotion processing showed different patterns of insula activity in alcohol dependence [17, 19, 20]. In response to happy, sad and angry faces, anterior insula activity was lower compared with controls in Alba-Ferrara, Muller-Oehring [19], but higher in Salloum, Ramchandani [17]. In response to fearful faces, anterior insula activity was lower in O’Daly, Trick [20] but higher in Salloum, Ramchandani [17]. Additionally, anterior insula activity was lower in response to disgust [17]. Given the differences in reactivity to the same emotions, it is possible these differences can be attributed to abstinence length of the participants. Insula activity occurs following homeostatic changes, such as during withdrawal from drugs. Therefore, the length of time the participants were abstinent likely affects insula activity during emotion processing. It is likely that the longer the participants were abstinent, the less hyperactive the insula would be during tasks of negative emotional processing. This is in line with the reviewed studies, as participants in Alba-Ferrara, Muller-Oehring [19] were either in early remission (<12 months) or late remission (>12 months), compared with Salloum, Ramchandani [17] and O’Daly, Trick [20], where all the participants were in early abstinence for <4 weeks. As early remission was considered up to 12 months in Alba-Ferrara, Muller-Oehring [19], but early abstinence was considered up to 4 weeks in Salloum, Ramchandani [17] and O’Daly, Trick [20], participants were likely abstinent for considerably longer in Alba-Ferrara, Muller-Oehring [19]. Therefore, reduced insula hyperactivity following exposure to facial emotions, particularly negative emotions, during longer periods of remission may indicate therapeutic benefit by reducing the negative subjective experience of feelings, which may help prevent craving and relapse.
Summary of brain reactivity in negative emotional processing in substance dependence
In the studies reviewed, the patterns of reactivity following negative emotional processing were variable amongst the discussed brain regions and substances. Blunting in activation was broadly seen in alcohol dependence in response to emotional faces and affective images. This may suggest that emotional networks that are recruited whilst processing affective stimuli may subsequently become dysregulated during alcohol dependence. In cocaine dependence, the results were also variable but broadly demonstrated heightened activation in response to stressful and aversive stimuli and blunted activation in response to positive affective stimuli. This is in line with previous evidence demonstrating blunted responding to natural rewards in cocaine addiction [14, 64, 65]. Patterns in brain reactivity were also variable in the studies investigating opioid dependence, although the amygdala was consistently implicated. Fewer conclusions can be drawn regarding cannabis dependence due to the lower number of studies. Although, regions that were implicated in other studies like the mPFC, anterior insula and ACC were also dysregulated in cannabis dependence. Together, the findings demonstrate alterations in activity and connectivity of these regions, which may result in impairments to negative emotional processing in drug dependence. This effect could be due to pre-existing maladaptations or functional adaptions following repeated drug exposure, or a combination of both, although is difficult to ascertain without longitudinal studies. Results following the completion of the ABCD and IMAGEN studies will hopefully shed light on the causality of developing substance use and addictive behaviour [66, 67]. Together, the results indicate that there may not be a broad level mechanism of dysfunction across substance dependences, but rather each class causes dysregulation in diverging ways.
Given the lack of neuroimaging research on emotional dysregulation in the substance dependence field, conclusively characterising brain reactivity patterns during negative emotional processing remains a challenge. Particularly, differences in study design, statistics and participant demographics contribute variability to the results, making interpretation increasingly challenging. Brain regions with significant anatomical overlap and indistinct boundaries may also present challenges when attributing dysfunction to a specific region. The ambiguity between boundaries increases potential for misinterpreting findings, making it difficult to attribute the results within the context of one specific region. This may lend itself to interpreter bias, whereby the effect may be interpreted in line with the hypothesis or current trends in research. The best way to account for this is with Activation Likelihood Estimation (ALE) meta-analyses, which can estimate the probability of overlap between activations across various studies [68]. However, given there are insufficient studies in this domain to make ALE possible, whilst the field grows, it is important to ensure all data being published is transparent moving forward. Following new results being published in the future, previously published data can be more easily re-evaluated and re-interpreted considering the new findings and be utilised in quantitative meta-analyses. In particular, whilst data in the field remains relatively sparse, a Bayesian approach may be appropriate as it can combine prior knowledge with small or heterogenous study results, reducing uncertainty and stabilising effect sizes.
Considerations from current evidence
Statistical considerations
Since the first studies, the methodology and statistical approaches used in fMRI have developed hugely, resulting in more caution about conclusions from earlier studies. Statistical methods commonly used in early fMRI statistical packages were found to allow substantially higher false positive rates than initially thought [69]. Some of the earlier studies in this review may not have accounted for multiple comparisons (Table 5), increasing the likelihood of erroneous interpretations. Some statistics in early fMRI studies were uncorrected, and instead the significance threshold was increased to help prevent false positives. However, this is less effective than FWE or FDR, which are more powerful at controlling for false positives when conducting multiple hypothesis tests [70]. Furthermore, studies using whole brain analyses only (see Table 1) are subject to increased variance in the results. Using a priori ROI hypothesis-driven approaches may help mitigate the problems associated with greater variance at the whole brain level. Studies using fMRI are typically underpowered due to small sample sizes. Increasing sample sizes can help improve confidence in results by reducing the likelihood of both type I and type II errors [71]. Recently, sample sizes have been recommended to be in the thousands to improve replicability and decrease effect size inflation [72]. However, increasing sample size is often extremely challenging in addiction research.
Table 5.
Overview of statistical thresholding and anatomical normalisations.
| Author/Date | Statistical threshold | Normalisation |
|---|---|---|
| Alcohol | ||
| Alba-Ferrara 2016 | p <0.001 uncorrected | MNI |
| Charlet 2014 | p <0.05 FWE corrected (ROI), p <0.001 uncorrected (WB) | Talairach |
| Gilman 2008 | p <0.05 cluster corrected | Talairach |
| Jansen 2019 | p <0.05 FWE corrected | MNI |
| Kim 2014 | p <0.05 FDR corrected | Talairach |
| Marinkovic 2009 | p <0.0001 uncorrected | Talairach |
| O’Daly 2012 | p <0.05 cluster and SVC | MNI |
| Salloum 2007 | p <0.05 corrected | Talairach |
| Seo 2013 | p <0.01 whole brain corrected | MNI |
| Zakiniaiez 2017 | p <0.05 FWE corrected | ? |
| Yang 2013 | p <0.05 GRF corrected | MNI |
| Cocaine | ||
| Albein-Urios 2014 | p <0.005 uncorrected | MNI |
| Asensio 2010 | p <0.01 FWE corrected | MNI |
| Canterberry 2016 | p <0.05 Monte Carlo simulation corrected | MNI |
| Potenza 2012 | p <0.05 FWE corrected | Talairach |
| Sinha 2005 | p <0.01 cluster corrected (between-groups), p <0.005 uncorrected (correlations) | Talairach |
| Xu 2013 | p <0.05 FWE corrected | MNI |
| Opioids | ||
| Schmidt 2014 | p <0.05 whole brain or SVC corrected | MNI |
| Schmidt 2015 | p <0.05 FWE cluster-level, p <0.001 FWE voxel-level | ? |
| Wang 2010 | voxel-wise p <0.005, p <0.05 corrected | Talairach |
| Smoski 2011 | p <0.01 FDR corrected | MNI |
| Cannabis | ||
| Wesley 2016 | p <0.05 FWE SVC cluster-level, p <0.01 voxel-level | MNI |
| Wetherill 2014 | p <0.05 cluster corrected | MNI |
| Zimmermann 2018 | P <0.05 FWE corrected | MNI |
FDR, false discovery rate; FWE, family wise error; GRF, Gaussian random field; MNI, Montreal Neurological Institute; ROI, region of interest; SVC, small volume correction; WB, whole brain.
Sex differences
A critical factor contributing variability in results is sex differences, as there are broad neurobiological differences between males and females [73]. Across the substance dependence literature, the majority of study participants are typically men. This prevents the results from being accurately attributed to how addiction manifests in females. In the reviewed studies, only Potenza, Hong [31] and Canterberry, Peltier [34] addressed sex differences in cocaine dependence. In Potenza, Hong [31], hyperactivity was observed in corticostriatal-limbic circuitry in women compared with men following exposure to stress cues. Meanwhile, no differences between sexes were observed in Canterberry, Peltier [34] in response to negative images. It is possible that the divergence in findings may be explained by co-occurring drug dependencies, as dependence on alcohol and cannabis were allowed in Potenza, Hong [31] and Canterberry, Peltier [34] respectively. Together, these discrepancies highlight the need to better address the impact of sex differences on emotional dysregulation in substance dependence, as well as comorbid polydrug dependencies.
Comorbid diagnoses
It is rare that an individual is dependent on one substance, particularly if nicotine is included, but such ‘polydrug’ dependence is rarely acknowledged. Nicotine dependence is particularly common in substance dependences and so is often allowed in studies. This makes it particularly challenging to disentangle the effect of nicotine relative to the substance being studied. Further, the majority will meet criteria for other psychiatric diagnoses, such as anxiety, depression, post-traumatic stress disorder and personality disorders [74]. As these conditions share similar but also distinct neural networks, attributing the results to a particular condition is incongruous [44]. Individuals with substance use disorders and comorbid psychiatric disorders demonstrate alterations in brain regions associated with reward processing, emotional regulation, and cognitive control [3]. However, these brain changes can vary depending on the specific combination of disorders, as well as the severity of symptoms and the stage of addiction. Additionally, the frequency of comorbid substance dependences and psychiatric diagnoses creates a recruitment challenge, as recruiting a ‘clean’ sample of individuals without current or past psychiatric disorders or substance dependencies is extremely difficult. Together, these challenges highlight the importance of carefully characterising the study population and accounting for the complexity of polydrug use and psychiatric comorbidity when designing and interpreting research studies.
Abstinence
As previously discussed, differences in abstinence length of the participants likely contributes to the disparity in findings across studies. During early abstinence, within days to a few weeks, the brain initially adapts to the lack of the substance and is more hyperreactive to negative affective stimuli, manifesting as increases in anxiety, emotional distress, craving and irritability, as it rebalances without the influence of substance [75]. Heightened reactivity to negative affective stimuli may intensify the feelings of negative emotions, culminating as heightened stress or anxiety responses [76]. As abstinence continues into weeks and months, neural circuitry associated with emotion processing may begin to stabilise, which may be accompanied by reduced craving and reactivity to stressful triggers. Longer-term abstinence, from months to years, may facilitate further recovery of brain reactivity to negative stimuli [77]. However, long-term neuroadaptations can persist into protracted withdrawal [78]. However, individual variation must be considered, due to factors such as dependence duration and severity, co-morbid psychiatric diagnoses, and concomitant behavioural intervention [79].
Considerations for the future
Standardising tasks
To further improve the reliability of findings in task-based fMRI, we should standardise the tasks being used, as this will help improve replicability [16]. Many studies used different tasks, falling into different categories regarding the paradigms used (Table 2), making comparisons in results between task categories difficult. Further, even studies using similar tasks but with small differences (Table 3), can produce profound differences in brain activation (Table 4). Small differences in task design may recruit different neural processes and engage different emotion networks [41], making it challenging to compare studies with each other directly. From a psychological perspective, the tasks utilised only measure singular aspects of emotion processing, which is a more complex construct. Given the variation in task categories and individual design within categories (Table 3), the differences between tasks likely mean that various processes are being measured that have neural underpinnings that are distinct from one another. Further, with other variables contributing to heterogenous results, such as sex, abstinence length and co-morbid diagnoses, using consistent task designs should improve replicability. Whilst the IAPS is good for standardising images used within studies examining affect, further standardisation in emotional processing task design is necessary. A shift towards consistency through the utilisation of reporting checklists [80], akin to what is observed in the cue reactivity literature with the formation of the ENIGMA Addiction Cue Reactivity Initiative [81, 82], would help improve reliability and replicability of results. A similar initiative should be developed within the field of emotional processing. A collaborative network like ENIGMA would help define and validate standardised tasks, which can be used consistently across different studies and research groups and ensure their sensitivity to measuring the relevant cognitive and behavioural processes. Alongside providing a centralised database where researchers can access the standardised tasks, together, this approach would help facilitate replication and comparison of findings in the field of emotional processing in substance dependence going forward.
Standardising terminology
In addition to standardising tasks, standardising terminology across the wider emotional processing field would greatly impact the interpretation of results. In the emotional processing field, terms such as ‘emotion’, ‘affect’ and ‘mood’ are often used interchangeably, although they refer to distinct psychological constructs [83]. The DSM-5 characterises ‘affect’ as observable expressions and objective measures of emotion, and ‘mood’ as a more prolonged and sustained emotional state [84], whilst ‘emotion’ generally refers to an immediate and transient response to a stimulus [85]. However, these terms are used inconsistently across the field, which may influence how findings are conceptualised, compared across studies, and applied in clinical context. For example, inconsistent use of terminology may hinder the synthesis of results in meta-analyses or systematic reviews. Without clear definitions for these terms, studies may not be investigating the same emotional constructs that they appear to be, which may subsequently compromise the ability to accurately interpret the findings and draw conclusions. Translating this into the clinic presents more challenges, as the subjective experience of the patient may be the same across different psychological constructs. For example, stress and anxiety are often conflated even though their neurobiological and psychological processes differ. Moving forward, precise terminology should be adopted across the emotional processing field, similar to Wellcome’s initiative for identifying and adopting common questionnaires in mental health research [86], in order to ensure that research outcomes are valid and that findings can be compared across studies.
Therapeutic interventions
Future studies should incorporate more techniques to modulate dysregulated brain processes in therapeutically beneficial ways. At the neural level, fMRI studies should include more pharmacological or psychological intervention. Incorporating these interventions into fMRI may help further elucidate biomarkers of emotional dysregulation by characterising whether they modulate brain reactivity in a manner consistent with therapeutic benefit. Protocols should carefully consider the timing of drug administration relative to the imaging session and have this kept consistent across studies.
Moreover, non-invasive brain stimulation (NIBS) techniques, including repetitive transcranial magnetic stimulation (rTMS), transcranial direct current stimulation (tDCS), and temporal interference (TI) stimulation have recently emerged as having potential clinical utility in treating substance dependence [87, 88]. In comparison to fMRI, which is primarily used as a tool to identify brain reactivity patterns and subsequent neural markers of psychiatric disorders, NIBS is advantageous as it can potentially modulate brain function in a therapeutic manner. Further, NIBS has been shown to modulate brain networks rather than individual regions, suggesting it may be useful for treating complex psychiatric disorders where multiple regions within a network are dysregulated [89]. Recent research has highlighted the dlPFC as an efficacious target region for substance dependence and may be a beneficial stimulation site across various substances [90]. rTMS of the dlPFC was shown to modulate emotion regulation during an emotion reappraisal task in alcohol use disorder [91]. Relative to sham procedure, rTMS reduced the emotional experience to positive and negative images in alcohol use disorder participants, whereas rTMS increased the emotional experience to neutral and positive images in healthy controls. The results suggest a possible role for rTMS modulating emotional states related to relapse, by reducing the negative emotional experience to negative images. However, since there were no associated reductions in subjective craving, further work is needed to characterise the effect of NIBS on emotion processing networks on clinically relevant measures of substance dependence.
However, whilst few studies exist examining the effect of NIBS on emotion regulation in substance dependence, more studies have been conducted investigating NIBS in cue reactivity paradigms [92]. Studies have demonstrated a personalised approach to NIBS, where individualised stimulation of deeper-brain targets in combination with individualised cue-reactivity paradigms have substantially decreased craving and substance use in heroin dependence [93, 94]. Recent advances in NIBS have advanced the ability to modulate deeper-brain structures more commonly associated with emotional circuitry, further enhancing the translational relevance of NIBS for substance dependence [93–95]. Further incorporation of NIBS with fMRI studies will help continue establishing the causal relationships between brain reactivity patterns and cognitive and behavioural processes. As a relatively new field, there are important limitations to consider, such as cost and accessibility [96], studies with small sample sizes and lack of controls, as well as methodological differences between studies resulting in inconsistent findings [87]. Nonetheless, the therapeutic potential of NIBS in modulating neural circuits that are implicated in addiction in an individualised manner is substantial and warrants further investigation to elucidate its treatment potential.
Conclusion
fMRI research has been instrumental for enhancing our understanding of the roles of various brain regions over the last two decades. Emotion processing has been relatively well characterised, implicating a complex interplay between regions such as the amygdala, insula, ACC, and prefrontal cortex, amongst others. However, the dysregulated neural mechanisms associated with negative emotion processing during substance dependence is less clear. Therefore, given the few studies that have examined this domain, elucidating clear biomarkers for developing targeted treatments remains a challenge. From the studies conducted, the key brains regions involved in emotion processing, namely the prefrontal cortex, ACC, insula, and amygdala, appear to be dysregulated in substance dependence, suggesting potentially useful target sites for intervention. However, whether this dysregulation manifests as blunted activity or hyperreactivity was variable across the different substances, with more clarity within alcohol and cocaine research given the larger number of studies compared with opioid and cannabis research. The uniqueness by which each drug class seems to dysregulate brain function suggests there is no ‘shared’ mechanism of addiction, making treatment development challenging still. With more studies, we can begin to better characterise emotion dysregulation in dependence. By standardising tasks and incorporating methods to modulate dysregulated neural processes with pharmacology and non-invasive brain stimulation, we can elucidate specific treatment targets that may enable the development of more effective relapse prevention methods following exposure to negative affective experiences.
Author contributions
LH conducted the literature review, synthesised the findings, and wrote the manuscript. LP and ALH contributed to the writing process by providing feedback and helping revise the manuscript. All authors reviewed and approved the final version of the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Greenfield SF, Weiss RD. Addiction-25 years later. Harv Rev Psychiatry. 2017;25:97–100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Heilig M. Columbia university press blog: Columbia university press wesbite. https://cupblog.org/2015/05/19/addiction-research-and-addiction-treatment/. Accessed10 October 2015.
- 3.Koob GF, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology. 2010;35:217–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kwako LE, Spagnolo PA, Schwandt ML, Thorsell A, George DT, Momenan R, et al. The corticotropin releasing hormone-1 (CRH1) receptor antagonist pexacerfont in alcohol dependence: a randomized controlled experimental medicine study. Neuropsychopharmacology. 2015;40:1053–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schwandt ML, Cortes CR, Kwako LE, George DT, Momenan R, Sinha R, et al. The CRF1 antagonist verucerfont in anxious alcohol-dependent women: translation of neuroendocrine, but not of anti-craving effects. Neuropsychopharmacology. 2016;41:2818–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Carmichael O, Schwarz AJ, Chatham CH, Scott D, Turner JA, Upadhyay J, et al. The role of fMRI in drug development. Drug Discov Today. 2018;23:333–48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gibson BC, Claus ED, Sanguinetti J, Witkiewitz K, Clark VP. A review of functional brain differences predicting relapse in substance use disorder: actionable targets for new methods of noninvasive brain stimulation. Neurosci Biobehav Rev. 2022:104821. [DOI] [PubMed]
- 8.Koob GF, Le Moal M. Addiction and the brain antireward system. Annu Rev Psychol. 2008;59:29–53. [DOI] [PubMed] [Google Scholar]
- 9.Koob GF. Drug addiction: hyperkatifeia/negative reinforcement as a framework for medications development. Pharmacol Rev. 2021;73:163–201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Beauchaine TP, Crowell SE, Garland EL, Bell S, Atchley RM, Froeliger B. Emotion dysregulation in addiction. The oxford handbook of emotion dysregulation. Oxford University Press; 2020. pp. 516.
- 11.Kwako LE, Koob GF. Neuroclinical framework for the role of stress in addiction. Chronic. Stress. 2017;1:1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhao Y, Sallie SN, Cui H, Zeng N, Du J, Yuan T, et al. Anterior cingulate cortex in addiction: new insights for neuromodulation. Neuromodulation. 2020;24:187–96. [DOI] [PubMed] [Google Scholar]
- 13.Naqvi NH, Bechara A. The hidden island of addiction: the insula. Trends Neurosci. 2009;32:56–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Goldstein RZ, Volkow ND. Dysfunction of the prefrontal cortex in addiction: neuroimaging findings and clinical implications. Nat Rev Neurosci. 2011;12:652–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kalisch R, Wiech K, Critchley HD, Dolan RJ. Levels of appraisal: a medial prefrontal role in high-level appraisal of emotional material. Neuroimage. 2006;30:1458–66. [DOI] [PubMed] [Google Scholar]
- 16.Wilcox CE, Pommy JM, Adinoff B. Neural circuitry of impaired emotion regulation in substance use disorders. Am J Psychiatry. 2016;173:344–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Salloum JB, Ramchandani VA, Bodurka J, Rawlings R, Momenan R, George D, et al. Blunted rostral anterior cingulate response during a simplified decoding task of negative emotional facial expressions in alcoholic patients. Alcohol Clin Exp Res. 2007;31:1490–504. [DOI] [PubMed] [Google Scholar]
- 18.Charlet K, Schlagenhauf F, Richter A, Naundorf K, Dornhof L, Weinfurtner CE, et al. Neural activation during processing of aversive faces predicts treatment outcome in alcoholism. Addict Biol. 2014;19:439–51. [DOI] [PubMed] [Google Scholar]
- 19.Alba-Ferrara L, Muller-Oehring EM, Sullivan EV, Pfefferbaum A, Schulte T. Brain responses to emotional salience and reward in alcohol use disorder. Brain Imaging Behav. 2016;10:136–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.O’Daly OG, Trick L, Scaife J, Marshall J, Ball D, Phillips ML, et al. Withdrawal-associated increases and decreases in functional neural connectivity associated with altered emotional regulation in alcoholism. Neuropsychopharmacology. 2012;37:2267–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Marinkovic K, Oscar-Berman M, Urban T, O’Reilly CE, Howard JA, Sawyer K, et al. Alcoholism and dampened temporal limbic activation to emotional faces. Alcohol Clin Exp Res. 2009;33:1880–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Yang H, Devous MD, Briggs RW, Spence JS, Xiao H, Kreyling N, et al. Altered neural processing of threat in alcohol-dependent men. Alcohol Clin Exp Res. 2013;37:2029–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zakiniaeiz Y, Scheinost D, Seo D, Sinha R, Constable RT. Cingulate cortex functional connectivity predicts future relapse in alcohol dependent individuals. Neuroimage Clin. 2017;13:181–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Seo D, Lacadie CM, Tuit K, Hong KI, Constable RT, Sinha R. Disrupted ventromedial prefrontal function, alcohol craving, and subsequent relapse risk. JAMA Psychiatry. 2013;70:727–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kim SM, Han DH, Min KJ, Kim BN, Cheong JH. Brain activation in response to craving- and aversion-inducing cues related to alcohol in patients with alcohol dependence. Drug Alcohol Depend. 2014;141:124–31. [DOI] [PubMed] [Google Scholar]
- 26.Jansen JM, van den Heuvel OA, van der Werf YD, de Wit SJ, Veltman DJ, van den Brink W, et al. Emotion processing, reappraisal, and craving in alcohol dependence: a functional magnetic resonance imaging study. Front Psychiatry. 2019;10:227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gilman JM, Hommer DW. Modulation of brain response to emotional images by alcohol cues in alcohol-dependent patients. Addict Biol. 2008;13:423–34. [DOI] [PubMed] [Google Scholar]
- 28.Zhao Y, Rutgen M, Zhang L, Lamm C. Pharmacological fMRI provides evidence for opioidergic modulation of discrimination of facial pain expressions. Psychophysiology. 2021;58:e13717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kanwisher N, Yovel G. The fusiform face area: a cortical region specialized for the perception of faces. Philos Trans R Soc Lond B Biol Sci. 2006;361:2109–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Albein-Urios N, Verdejo-Roman J, Asensio S, Soriano-Mas C, Martinez-Gonzalez JM, Verdejo-Garcia A. Re-appraisal of negative emotions in cocaine dependence: dysfunctional corticolimbic activation and connectivity. Addict Biol. 2014;19:415–26. [DOI] [PubMed] [Google Scholar]
- 31.Potenza MN, Hong KI, Lacadie CM, Fulbright RK, Tuit KL, Sinha R. Neural correlates of stress-induced and cue-induced drug craving: influences of sex and cocaine dependence. Am J Psychiatry. 2012;169:406–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sinha R, Lacadie C, Skudlarski P, Fulbright RK, Rounsaville BJ, Kosten TR, et al. Neural activity associated with stress-induced cocaine craving: a functional magnetic resonance imaging study. Psychopharmacology (Berl). 2005;183:171–80. [DOI] [PubMed] [Google Scholar]
- 33.Sinha R, Lacadie, C, Li, C-S, Skudlarski, P, Kosten, TR. Stress-related fMRI brain activity predicts cocaine relapse outcomes. Proceedings of the 67th Annual Scientific Meeting of the College on Problems of Drug Dependence; 2005; Orlando, FL: NIDA Research Monograph Series.
- 34.Canterberry M, Peltier MR, Brady KT, Hanlon CA. Attenuated neural response to emotional cues in cocaine-dependence: a preliminary analysis of gender differences. Am J Drug Alcohol Abuse. 2016;42:577–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Asensio S, Romero MJ, Palau C, Sanchez A, Senabre I, Morales JL, et al. Altered neural response of the appetitive emotional system in cocaine addiction: an fMRI Study. Addict Biol. 2010;15:504–16. [DOI] [PubMed] [Google Scholar]
- 36.Schmidt A, Borgwardt S, Gerber H, Wiesbeck GA, Schmid O, Riecher-Rossler A, et al. Acute effects of heroin on negative emotional processing: relation of amygdala activity and stress-related responses. Biol Psychiatry. 2014;76:289–96. [DOI] [PubMed] [Google Scholar]
- 37.Schmidt A, Walter M, Gerber H, Seifritz E, Brenneisen R, Wiesbeck GA, et al. Normalizing effect of heroin maintenance treatment on stress-induced brain connectivity. Brain. 2015;138:217–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Smoski MJ, Salsman N, Wang L, Smith V, Lynch TR, Dager SR, et al. Functional imaging of emotion reactivity in opiate-dependent borderline personality disorder. Personal Disord. 2011;2:230–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wang ZX, Zhang JX, Wu QL, Liu N, Hu XP, Chan RC, et al. Alterations in the processing of non-drug-related affective stimuli in abstinent heroin addicts. Neuroimage. 2010;49:971–6. [DOI] [PubMed] [Google Scholar]
- 40.Wetherill RR, Childress AR, Jagannathan K, Bender J, Young KA, Suh JJ, et al. Neural responses to subliminally presented cannabis and other emotionally evocative cues in cannabis-dependent individuals. Psychopharmacology (Berl). 2014;231:1397–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zimmermann K, Yao S, Heinz M, Zhou F, Dau W, Banger M, et al. Altered orbitofrontal activity and dorsal striatal connectivity during emotion processing in dependent marijuana users after 28 days of abstinence. Psychopharmacology (Berl). 2018;235:849–59. [DOI] [PubMed] [Google Scholar]
- 42.Wesley MJ, Lile JA, Hanlon CA, Porrino LJ. Abnormal medial prefrontal cortex activity in heavy cannabis users during conscious emotional evaluation. Psychopharmacology (Berl). 2016;233:1035–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sibille E, Wang Y, Joeyen-Waldorf J, Gaiteri C, Surget A, Oh S, et al. A molecular signature of depression in the amygdala. Am J Psychiatry. 2009;166:1011–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.McTeague LM, Rosenberg BM, Lopez JW, Carreon DM, Huemer J, Jiang Y, et al. Identification of common neural circuit disruptions in emotional processing across psychiatric disorders. Am J Psychiatry. 2020;177:411–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Etkin A, Egner T, Kalisch R. Emotional processing in anterior cingulate and medial prefrontal cortex. Trends Cogn Sci. 2011;15:85–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Goldin PR, McRae K, Ramel W, Gross JJ. The neural bases of emotion regulation: reappraisal and suppression of negative emotion. Biol Psychiatry. 2008;63:577–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Denny BT, Fan J, Liu X, Guerreri S, Mayson SJ, Rimsky L, et al. Insula-amygdala functional connectivity is correlated with habituation to repeated negative images. Soc Cogn Affect Neurosci. 2014;9:1660–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gerhardt S, Berhe O, Moessnang C, Horning M, Kiefer F, Tost H, et al. Lack of amygdala habituation to negative emotional faces in alcohol use disorder and the relation to adverse childhood experiences. Addict Biol. 2022;28:e13251. [DOI] [PubMed] [Google Scholar]
- 49.Shurman J, Koob GF, Gutstein HB. Opioids, pain, the brain, and hyperkatifeia: a framework for the rational use of opioids for pain. Pain Med. 2010;11:1092–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ochsner KN, Gross JJ. The cognitive control of emotion. Trends Cogn Sci. 2005;9:242–9. [DOI] [PubMed] [Google Scholar]
- 51.Colibazzi T, Posner J, Wang Z, Gorman D, Gerber A, Yu S, et al. Neural systems subserving valence and arousal during the experience of induced emotions. Emotion. 2010;10:377–89. [DOI] [PubMed] [Google Scholar]
- 52.Zhong M, Wang X, Xiao J, Yi J, Zhu X, Liao J, et al. Amygdala hyperactivation and prefrontal hypoactivation in subjects with cognitive vulnerability to depression. Biol Psychol. 2011;88:233–42. [DOI] [PubMed] [Google Scholar]
- 53.Mao Y, Zuo XN, Ding C, Qiu J. OFC and its connectivity with amygdala as predictors for future social anxiety in adolescents. Dev Cogn Neurosci. 2020;44:100804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sladky R, Hoflich A, Kublbock M, Kraus C, Baldinger P, Moser E, et al. Disrupted effective connectivity between the amygdala and orbitofrontal cortex in social anxiety disorder during emotion discrimination revealed by dynamic causal modeling for FMRI. Cereb Cortex. 2015;25:895–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Klenowski PM. Emerging role for the medial prefrontal cortex in alcohol-seeking behaviors. Addict Behav. 2018;77:102–6. [DOI] [PubMed] [Google Scholar]
- 56.Gorka SM, Fitzgerald DA, King AC, Phan KL. Alcohol attenuates amygdala-frontal connectivity during processing social signals in heavy social drinkers: a preliminary pharmaco-fMRI study. Psychopharmacology (Berl). 2013;229:141–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sinha R, Lacadie CM, Constable RT, Seo D. Dynamic neural activity during stress signals resilient coping. Proc Natl Acad Sci USA. 2016;113:8837–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Ochsner KN, Silvers JA, Buhle JT. Functional imaging studies of emotion regulation: a synthetic review and evolving model of the cognitive control of emotion. Ann N Y Acad Sci. 2012;1251:E1–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Blaine SK, Wemm S, Fogelman N, Lacadie C, Seo D, Scheinost D, et al. Association of prefrontal-striatal functional pathology with alcohol abstinence days at treatment initiation and heavy drinking after treatment Initiation. Am J Psychiatry. 2020;177:1048–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sinha R. Alcohol’s negative emotional side: the role of stress neurobiology in alcohol use disorder. Alcohol Res. 2022;42:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Schmidt A, Denier N, Magon S, Radue EW, Huber CG, Riecher-Rossler A, et al. Increased functional connectivity in the resting-state basal ganglia network after acute heroin substitution. Transl Psychiatry. 2015;5:e533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ibrahim C, Rubin-Kahana DS, Pushparaj A, Musiol M, Blumberger DM, Daskalakis ZJ, et al. The insula: a rain stibmulation target for the treatment of addiction. Front Pharmacol. 2019;10:720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Frank DW, Dewitt M, Hudgens-Haney M, Schaeffer DJ, Ball BH, Schwarz NF, et al. Emotion regulation: quantitative meta-analysis of functional activation and deactivation. Neurosci Biobehav Rev. 2014;45:202–11. [DOI] [PubMed] [Google Scholar]
- 64.Volkow ND, Wang GJ, Telang F, Fowler JS, Logan J, Childress AR, et al. Cocaine cues and dopamine in dorsal striatum: mechanism of craving in cocaine addiction. J Neurosci. 2006;26:6583–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Balodis IM, Potenza MN. Anticipatory reward processing in addicted populations: a focus on the monetary incentive delay task. Biol Psychiatry. 2015;77:434–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lisdahl KM, Sher KJ, Conway KP, Gonzalez R, Feldstein Ewing SW, Nixon SJ, et al. Adolescent brain cognitive development (ABCD) study: overview of substance use assessment methods. Dev Cogn Neurosci. 2018;32:80–96. [DOI] [PMC free article] [PubMed]
- 67.Mascarell Maricic L, Walter H, Rosenthal A, Ripke S, Quinlan EB, Banaschewski T, et al. The IMAGEN study: a decade of imaging genetics in adolescents. Mol Psychiatry. 2020;25:2648–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Turkeltaub PE, Eden GF, Jones KM, Zeffiro TA. Meta-analysis of the functional neuroanatomy of single-word reading: method and validation. Neuroimage. 2002;16:765–80. [DOI] [PubMed] [Google Scholar]
- 69.Eklund A, Nichols TE, Knutsson H. Cluster failure: Why fMRI inferences for spatial extent have inflated false-positive rates. Proc Natl Acad Sci USA. 2016;113:7900–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Bennett CM, Miller MB, Wolford GL. Neural correlates of interspecies perspective taking in the post-mortem Atlantic salmon: an argument for proper multiple comparisons correction. 2009;1:1–5.
- 71.Button KS, Ioannidis JP, Mokrysz C, Nosek BA, Flint J, Robinson ES, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. 2013;14:365–76. [DOI] [PubMed] [Google Scholar]
- 72.Marek S, Tervo-Clemmens B, Calabro FJ, Montez DF, Kay BP, Hatoum AS, et al. Reproducible brain-wide association studies require thousands of individuals. Nature. 2022;603:654–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fonseca F, Robles-Martinez M, Tirado-Munoz J, Alias-Ferri M, Mestre-Pinto JI, Coratu AM, et al. A gender perspective of addictive disorders. Curr Addict Rep. 2021;8:89–99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.NIDA. Common comorbidities with substance use disorders research report. https://www.ncbi.nlm.nih.gov/books/NBK571451/. Accessed April 2022. [PubMed]
- 75.Sinha R. The role of stress in addiction relapse. Curr Psychiatry Rep. 2007;9:388–95. [DOI] [PubMed] [Google Scholar]
- 76.Fox HC, Hong KI, Siedlarz K, Sinha R. Enhanced sensitivity to stress and drug/alcohol craving in abstinent cocaine-dependent individuals compared to social drinkers. Neuropsychopharmacology. 2008;33:796–805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Parvaz MA, Rabin RA, Adams F, Goldstein RZ. Structural and functional brain recovery in individuals with substance use disorders during abstinence: a review of longitudinal neuroimaging studies. Drug Alcohol Depend. 2022;232:109319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Volkow ND. Fowler JS. Addiction, a disease of compulsion and drive: involvement of the orbitofrontal cortex. Cereb Cortex. 2000;10:318–25. [DOI] [PubMed] [Google Scholar]
- 79.Sinha R. New findings on biological factors predicting addiction relapse vulnerability. Curr Psychiatry Rep. 2011;13:398–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Ekhtiari H, Zare-Bidoky M, Sangchooli A, Valyan A, Abi-Dargham A, Cannon DM, et al. Reporting checklists in neuroimaging: promoting transparency, replicability, and reproducibility. Neuropsychopharmacology. 2024. 10.1038/s41386-024-01973-5. [DOI] [PMC free article] [PubMed]
- 81.Ekhtiari H, Zare-Bidoky M, Sangchooli A, Janes AC, Kaufman MJ, Oliver JA, et al. A methodological checklist for fMRI drug cue reactivity studies: development and expert consensus. Nat Protoc. 2022;17:567–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ekhtiari H, Secretariat A. A systematic review on fMRI drug cue reactivity studies. https://osf.io/eb972/. Accessed 26 September 2020.
- 83.Ketal R. Affect, mood, emotion, and feeling: semantic considerations. Am J Psychiatry. 1975;132:1215–7. [DOI] [PubMed] [Google Scholar]
- 84.Association AP. Diagnostic and statistical manual of mental disorders. 5th edn. DSM Library; 2013.
- 85.Palomero-Gallagher N, Amunts K. A short review on emotion processing: a lateralized network of neuronal networks. Brain Struct Funct. 2022;227:673–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Farber GK, Gage S, Kemmer D, White R. Common measures in mental health: a joint initiative by funders and journals. Lancet Psychiatry. 2023;10:465–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Stein ER, Gibson BC, Votaw VR, Wilson AD, Clark VP, Witkiewitz K. Non-invasive brain stimulation in substance use disorders: implications for dissemination to clinical settings. Curr Opin Psychol. 2019;30:6–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dunlop K, Hanlon CA, Downar J. Noninvasive brain stimulation treatments for addiction and major depression. Ann N Y Acad Sci. 2017;1394:31–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.To WT, De Ridder D, Hart J Jr., Vanneste S. Changing brain networks through non-invasive neuromodulation. Front Hum Neurosci. 2018;12:128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Liu Q, Yuan T. Noninvasive brain stimulation of addiction: one target for all? Psychoradiology. 2021;1:172–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jansen JM, van den Heuvel OA, van der Werf YD, de Wit SJ, Veltman DJ, van den Brink W, et al. The effect of high-frequency repetitive tanscranial magnetic stimulation on emotion processing, reappraisal, and craving in alcohol use disorder patients and healthy controls: a functional magnetic resonance imaging study. Front Psychiatry. 2019;10:272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kurtin DL, Prabhu AM, Groen A, Amer MJ, Lingford-Hughes A, Paterson LM. Differences in fMRI-based functional connectivity during abstinence or interventions between heroin-dependent individuals and healthy controls. Rev Transl Psychiatry. 2024. 10.1101/2024.06.11.598479.
- 93.Mahoney JJ, Thompson-Lake DGY, Ranjan M, Marton JL, Carpenter JS, Zheng W, et al. Low-intensity focused ultrasound targeting the bilateral nucleus accumbens as a potential treatment for substance use disorder: a first-in-human report. Biol Psychiatry. 2023;94:e41–e3. [DOI] [PubMed] [Google Scholar]
- 94.Mahoney JJ, Haut MW, Carpenter J, Ranjan M, Thompson-Lake DGY, Marton JL, et al. Low-intensity focused ultrasound targeting the nucleus accumbens as a potential treatment for substance use disorder: safety and feasibility clinical trial. Front Psychiatry. 2023;14:1211566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Violante IR, Alania K, Cassara AM, Neufeld E, Acerbo E, Carron R, et al. Non-invasive temporal interference electrical stimulation of the human hippocampus. Nat Neurosci. 2023;26:1994–2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Goldbloom DS, Gratzer D. Barriers to brain stimulation therapies for treatment-resistant depression: beyond cost effectiveness. Can J Psychiatry. 2020;65:193–5. [DOI] [PMC free article] [PubMed] [Google Scholar]


