Abstract
An intense, stereotyped inflammatory response occurs in response to ischaemic and non-ischaemic injury to the myocardium. The NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome is a finely regulated macromolecular protein complex that senses the injury and triggers and amplifies the inflammatory response by activation of caspase 1, cleavage of pro-inflammatory cytokines, such as pro-IL-1β and pro-IL-18 in their mature forms, and inducing inflammatory cell death (pyroptosis). Inhibitors of the NLRP3 inflammasome and blockers of IL-1β and IL-18 activity have been shown to reduce injury to the myocardium and pericardium, favour resolution of the inflammation and preserve cardiac function. In this Review, we discuss the components of the NLRP3 inflammasome and how it is formed and activated in variety of ischaemic and non-ischaemic cardiac pathologies (acute myocardial infarction, cardiac dysfunction and remodelling, atherothrombosis, myocarditis and pericarditis, cardiotoxicity and cardiac sarcoidosis). We also summarize current preclinical and clinical evidence from studies of agents that target the NLRP3 inflammasome and related cytokines.
Introduction
An estimated 19 million deaths were attributed to cardiovascular diseases (CVD) globally in 2020, an increase of 18.7% from 20101. Ischaemic and non-ischaemic injury to the heart both induce acute cardiac dysfunction and can be life-threatening. A stereotyped inflammatory response follows cardiac injury, irrespective of its nature, promoting further injury and dysfunction and enhancing the risk of complications and death2,3. The NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome has emerged as a central mediator of the inflammatory response to tissue injury and sterile inflammation3,4, making this multiprotein complex an important therapeutic target.
In this Review, we discuss the components of the NLRP3 inflammasome and how it is formed and activated in variety of ischaemic and non-ischaemic cardiac pathologies (acute myocardial infarction (AMI), cardiac dysfunction and remodelling, atherothrombosis, myocarditis and pericarditis, cardiotoxicity and cardiac sarcoidosis). We also summarize the evidence that identified the NLRP3 inflammasome and derived cytokines, IL-1β and IL-18, and examine preclinical and clinical data from studies of drugs that target these molecules. We place emphasis on how our understanding of the mechanism and development of therapies has changed during the past 5 years3,4. The role of non-NLRP3 inflammasomes, such as the NLRP1, NLR family CARD domain-containing protein 4 (NLRC4) and interferon-inducible protein (AIM2) inflammasomes, in CVD is not well established and is beyond the scope of this Review.
Inflammatory response to cardiac injury
AMI is the prototypical model of non-infectious, ischaemic injury ensuing sterile inflammation, which results in resorption of the wound and the formation of a scar. Early reperfusion in AMI reduces the extent of the injury. Inflammation was once considered necessary for the formation of infarct scar and to prevent wall rupture. However, it is now clear that overzealous inflammation promotes further injury, dysfunctional healing and loss of cardiac function5,6. An intense inflammatory response also promotes wall rupture, however, such an event is now unlikely due to the reduced incidence of transmural infarction with reperfusion1,7–9.
The inflammatory response to both AMI and non-ischaemic injury induces a paracrine effect on the myocardium in the infarct border zone, a portion of the tissue that was salvaged by reperfusion but remained in jeopardy. A systemic response also occurs, regulated by IL-1β and downstream mediators10. Pro-inflammatory cytokines promote cardiac dysfunction and unfavourable remodelling, in a similar fashion to neurohormonal activation6. Sensing is essential to initiate inflammation. In the case of infection, sensing the pathogen allows for a targeted immune response. In sterile inflammation, cell debris provides the trigger and initiates the response3,10.
The NLRP3 inflammasome as a sensor
Inflammasomes are macromolecular protein complexes that have evolved to activate inflammatory caspases (1, 4, 5 and 11)11,12. Caspase 1 is the effector enzyme that, in humans as well as in mice, promotes the maturation and secretion of powerful pro-inflammatory cytokines, such as IL-1β and IL-18, and a form of inflammatory, lytic cell death termed ‘pyroptosis’11,12. Caspases 4 and 5 in humans, and their murine ortholog caspase 11, mediate non-canonical inflammasome activation13,14. The classic structure of the NLRP3 inflammasome sensing component is tripartite, with a central nucleotide oligomerization domain (defined as NACHT), a C-terminal leucine-rich repeats (LRR) domain and an N-terminal effector domain (pyrin domain, or PYD), which interacts with downstream signalling molecules11,12. NLRP3 activation, due to microbial pathogen-derived molecules and extracellular or intracellular damage-associated molecular patterns (DAMPs), induces the formation of NLRP3 disk-like oligomers15. The ATPase activity of the NACHT domain is essential for NLRP3 activation, and NACHT and LRR domains participate in oligomer formation and conformational changes15. Active NLRP3 engages a scaffold protein, the apoptosis speck-like protein containing a caspase recruiting domain (ASC), facilitating the polymerization of ASC into a stable, long filament structure favouring the formation of multiple star-like caspase 1 filaments, thereby increasing caspase 1 density to promote contact with substrates15,16. Caspase 1, also known as IL-1β-converting enzyme (ICE), cleaves and converts pro-IL-1β and pro-IL-18 into active forms17,18. In addition, caspase 1 cleaves gasdermin D (GSDMD), leading to multimerization of its N-terminal fragments (GSDMD-NT). GSDMD-NT form 21 nm pores on the plasma membrane, enabling extracellular release of IL-1β and IL-18 and loss of membrane integrity, eventually leading to cell death19,20. Mature IL-1β and IL-18 are 17 and 18 KDa, respectively, and measure ~4 nm (for comparison, serum albumin is a protein of 66 KDa and 7.5 nm21). Pro-IL-1α, another member of the IL-1 family, lacks a caspase 1 cleavage site, is already active in the pro-form and functions as an alarmin10. Cleavage of pro-IL-1α by calpains in the cell (or neutrophil elastase, granzyme B and chimase outside the cell) increases the affinity of IL-1α for the IL-1 receptor type I (IL-1RI)22. Although not responsible for its cleavage, caspase 1 activation facilitates pro-IL-1α release23. In the mouse, forced overexpression of caspase 1 in the heart leads to apoptosis without cytokine release24. Signalling downstream of the IL-1 receptor is mediated by myeloid differentiation primary response protein MyD88 (MyD88) and interleukin-1 receptor-associated kinases (IRAKs) leading to nuclear factor κB (NF-κB) activation10,17.
Pyroptosis
Inflammasome-dependent activation of caspases promotes pyroptosis, a GSDMD-dependent form of lytic cell death19,25, which is associated with the active release of IL-1β and IL-18 and exacerbates the inflammatory response. Caspase 1 is the main pyroptosis effector. Unlike apoptosis, a typical ‘silent’ form of programmed cell death, pyroptosis is associated with increased membrane permeability, mediated by GSDMD-NT, leading to cell swelling and ultimately, through the activity of ninjurin 1, to plasma membrane rupture25,26. Other gasdermins (i.e. GSDMB in humans and GSDME in humans and mice) induce pyroptosis through inflammasome-independent mechanisms27. GSDMD-NT have a high affinity for phosphatidylinositol phosphates and phosphatidylserine, which are mostly present on the intracellular-facing side of the plasma membrane, thus reducing damage to membranes of neighbouring cells19. Studies of GSDMD-NT pore structure have revealed that they favour the passage of neutral and positively charged proteins, and reduce the transit of negatively charged proteins19. This selectivity promotes the passage of mature IL-1β and IL-18 and increases the cytoplasmic permanence of pro-IL-1β and pro-IL-18, which have an acidic/negatively charged pro-domain. GSDMD-NT can also regulate the release of pro-IL-1α and active IL-1α28. At low concentrations, GSDMD-NT form non-lytic pores, thus favouring the release of pro-inflammatory cytokines rather than cell death19. Caspase 1 cleaves enzymes of the Krebs cycle, dramatically reducing energy production29,30, as well as other substrates (e.g. actin, cytoplasmic 1 (also known as β-actin), calpastatin and E3 ubiquitin-protein ligase parkin). Caspases 4 and 5 in humans, or caspase 11 in mice, bind to cytosolic lipopolysaccharide leading to GSDMD cleavage and non-canonical NLRP3 inflammasome activation and pyroptosis31.
Activation of the NLRP3 inflammasome
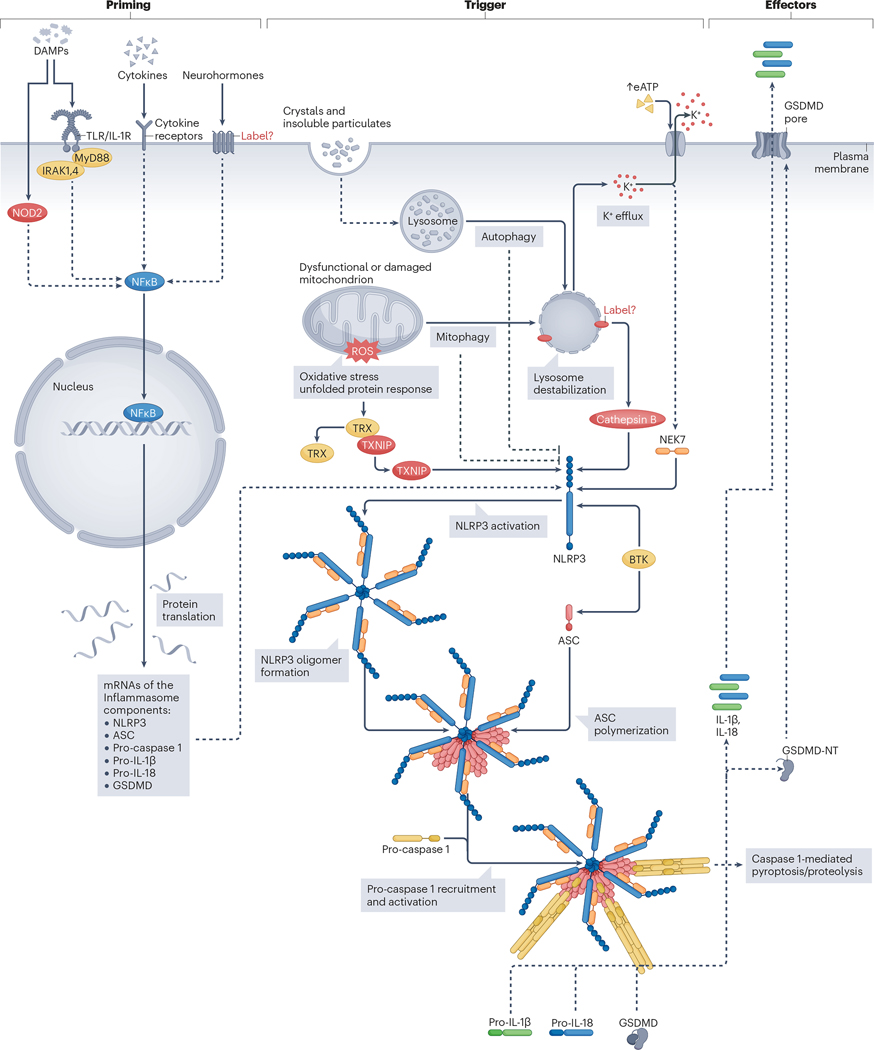
Activation of the NLRP3 inflammasome in cardiomyocytes and other cardiac resident cells is a two-step process32. In the healthy, uninjured heart, the expression of components of the NLRP3 inflammasome is very low, insufficient to promote oligomerization and polymerization32. Priming — transcriptional regulation through activation of NF-κB — is the first necessary step in activation and is regulated by pro-inflammatory cytokines, neurohormones, cellular debris or microbial products, which function as DAMPs or pathogen-associated molecular patterns (PAMPs) and bind to the Toll-like receptors (TLRs)15 (Fig. 1). Nucleotide-binding oligomerization domain-containing protein 2 (NOD2), a member of the innate immune receptor family, contributes to inflammasome priming by inducing NF-κB activation, and exacerbates myocardial injury in mice with AMI33. NOD2 deletion reduces myocardial damage and the expression of pro-inflammatory markers, including IL-1β4,33. Once a critical mass of NLRP3 inflammasome components is reached, activation signals (or triggers) converging on NLRP3, and leading to K+ efflux from the cell, catalyse the formation of the inflammasome. Extracellular ATP binding to the purinergic P2X receptor 7 (P2X7) is a classical triggering signal that leads to K+ efflux. In addition, lysosome destabilization, by cholesterol/uric acid crystals or pathogens, leads to intracellular cathepsin B leakage and K+ efflux34,35 (Fig. 1).
Fig. 1 |. Activation of the NLRP3 inflammasome pathways in cardiomyocytes.
The NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome forms in myocardial cells following a two-step process. Firstly, ‘priming’ that leads to transcription and translation of the NLRP3 inflammasome components and substrates (IL-1β and IL-18). Nuclear factor-kB (NF-κB) activation drives the priming process, initiated by the membrane Toll-like receptors (TLRs) or IL-1 receptor (IL-1R) family. Downstream signalling is mediated by myeloid differentiation primary response protein MyD88 (MyD88) and interleukin-1 receptor-associated kinases (IRAKs). Nucleotide-binding oligomerization domain-containing protein 2 (NOD2) promotes a similar priming signal. Secondly, activation or ‘triggering’ of NLRP3. NLRP3 activation is mediated by extracellular and intracellular pathways. Increasing the concentration of extracellular ATP (eATP) activates the P2X purinoceptor 7 and leads to K+ efflux, a step that triggers the activation of NLRP3. The serine/threonine-protein kinase NEK7 senses K+ efflux and binds to NLRP3, allowing its activation. Lysosomal destabilization by crystals and indigestible material represent another mechanism that leads to NLRP3 activation, through leakage of the lysosomal enzyme cathepsin B and by induction of K+. Mitochondrial damage and dysfunction produce reactive oxygen species (ROS), leading to dissociation of thioredoxin-interacting protein (TXNIP) from thioredoxin (TRX), and TXNIP binding to NLRP3. Ineffective clearance of dysfunctional mitochondria through mitophagy contributes to lysosomal destabilization. By contrast, effective mitophagy and autophagy limit the activation of NLRP3. Tyrosine-protein kinase BTK (also known as Bruton kinase) also binds to NLRP3,as well as to apoptosis speck-like protein containing a caspase recruiting domain (ASC), contributing to inflammasome activation. The active NLRP3 bound to NEK7 oligomerizes and forms a disc-like structure that is platform for the polymerization of ASC into a central filament, facilitating assembly of caspase 1 into filaments that form a star-like structure. Active caspase 1 cleaves the inactive pro-IL-1β and pro-IL-18 into the active forms, IL-1 and IL-18. Caspase 1 also cleaves gasdermin D (GSDMD) producing N-terminal fragments (GSDMD-NT) that oligomerize and form pores in the cell membrane, leading to the extracellular release of active IL-1β and IL-18 and pyroptosis. DAMPs, damage-associated molecular patterns.
The process of NLRP3 inflammasome formation and activation in the heart is cell-type specific36–38. Circulating monocytes constitutively express inflammasome components and require no, or minimal priming to produce large amounts of IL-1β when triggered39. Experimental activation of the NLRP3 inflammasome in resident macrophages, fibroblasts and endothelial cells, leads to the release of large amounts of IL-1β, whereas it is minimal in cardiomyocytes36–38. By contrast, cardiomyocytes with NLRP3 inflammasome formation have active caspase 1, secrete IL-18 and die of pyroptosis40–43.
Important to note is that some inflammasome-independent functions of NLRP3 have been reported in colon and kidney cells, although their role in the heart has not been established44,45. Deletion of NLRP3, but not ASC, inhibits ex vivo ischaemic preconditioning through an IL-1-independent effect46.
Oxidative stress and kinase-mediated activation
Several cytoplasmic adaptor proteins and kinases participate in the process of NLRP3 inflammasome activation. A member of the never in mitosis A-related kinase family, serine/threonine-protein kinase NEK7, is highly expressed in the heart, where it senses K+ efflux, binds NLRP3 and induces its oligomerization47. Tyrosine-protein kinase BTK (also known as Bruton kinase) is required for the activation of NLRP3 in vivo, and also interacts with ASC48. BTK inhibition suppresses inflammasome activation in vivo, reduces infarct size after cerebral ischaemia and improves heart function in a mouse model of sepsis 48,49.
Thioredoxin-interacting protein (TXNIP) links oxidative stress to NLRP3 inflammasome activation50–52 (Fig. 1). In healthy conditions, TXNIP is bound to thioredoxin, but increased levels of reactive oxygen species (ROS) and the unfolded protein response release TXNIP from thioredoxin, promoting NLRP3 activation and consequent inflammasome formation. TXNIP silencing, using small interfering RNAs, reduces NLRP3 activation and infarct size after AMI50. However, in some models, such as murine bone marrow-derived cells treated with classical NLRP3 inducers, activation of NLRP3 is independent of TXNIP53,54.
The role of autophagy
Autophagy is a homeostatic, proteolytic process that removes misfolded or damaged proteins and organelles and regulates inflammasome function55. Inhibition of the autophagic pathway in human monocytes and macrophages enhances NLRP3 inflammasome formation, whereas increasing the autophagic flux reduces IL-1β secretion56–58. In a mouse model of heart failure secondary to pressure overload due to transaortic constriction (TAC), cardiomyocyte-restricted gene silencing of the renin receptor (Atp6ap2), a component of the vacuolar-type ATPase required for autophagy, reduced autophagic flux, increased NLRP3 activation and worsened myocardial damage and remodelling59. NLRP3 inhibition blunted myocardial damage, implying inflammasome activation driven by an autophagy impairment59. In mice with diabetes mellitus, MI induced by coronary artery ligation impaired autophagic flux, increased expression of NLRP3 inflammasome components and worsened cardiac remodelling60. Induction of autophagy with rapamycin reversed these effects, to a degree similar to NLRP3 inhibition60,61.
Mitochondrial injury and mitophagy
Mitochondrial dysfunction and damage are major contributors to NLRP3 activation62. Mitochondria generate ROS and determine cell fate under stress conditions, regulating multiple cell death pathways (i.e. necrosis, apoptosis and pyroptosis)62–65. Dysfunctional mitochondria increase ROS production and K+ efflux, both of which are NLRP3 triggers62,64,65. Three distinct pathways regulate the fate of dysfunctional and damaged mitochondria — mitophagy (which clears mitochondria through autophagy), mitochondrial fission (division) and mitochondrial fusion55,66,67. Ineffective mitophagy increases activation of caspase 1 and IL-1β due to sustained ROS production and cytoplasmic accumulation of mitochondrial DNA62. In activated macrophages, mitophagy is reduced by pro-IL-1α binding to cardiolipin (a mitochondrial lipid that binds and activates NLRP368) exacerbating NLRP3 inflammasome activity69. In another model, deletion of dynamin-1-like protein, 1 (DRP1) impaired mitochondrial fission and promoted NLRP3-dependent caspase 1 activation and IL-1β secretion66. Mitochondrial fusion on the other hand prevents NLRP3 activation70.
The NLRP3 inflammasome in AMI
Ischaemia and reperfusion
The expression of NLRP3 inflammasome components in the healthy heart is negligible. However, human pathology studies show evidence of NLRP3 inflammasome activation during AMI71. Patients with AMI also have elevated plasma levels of NLRP3 and caspase 172. The time-dependent activation of the NLRP3 inflammasome in the heart has been examined in animal studies40,73 (Fig. 2). In a mouse model of AMI, the expression of NLRP3 inflammasome components increased over time — activation occurred 3–24 hours after reperfusion, peaking after 1 or 3 days in mice with reperfused and non-reperfused AMI, respectively40,73–75. Histology studies showed inflammasome specks in endothelial cells and cardiomyocytes from the ischaemic area and border zones early in the course of AMI, followed by activation in fibroblasts and then infiltrating leukocytes. As inflammation resolves, inflammasome specks are seen in isolated cardiomyocytes or fibroblasts40,76
Fig. 2 |. Temporal expression of the NLRP3 inflammasome components and window of opportunity for effective inhibition in ischaemia–reperfusion injury.
Progressive expression of the NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome components (black line) and of infarct area growth (orange line) over time are shown. The infarct area (orange) in the ventricular wall is shown below the graph. For the initial hour after ischaemia–reperfusion injury, the expression of NLRP3 components and the inflammasome activity in the myocardium remains low. The expression of the inflammasome components increases between 1 and 3 hours after injury, leading to effective priming and facilitation of inflammasome formation during this time and in subsequent hours. The size of the infarct continues to grow after reperfusion. The time that precedes the activation of the inflammasome represents a therapeutic window for intervention with NLRP3 inhibitors before the inflammasome forms.
Response to injury
Inflammasome formation is a finely-regulated and energy-requiring process. From an evolutionary standpoint, the formation of the inflammasome is a ‘sacrifice’ by which injured (often infected) cells activate the inflammatory response, sacrificing those ‘sentinel’ cells77. Therefore, when the inflammasome is formed in injured cardiomyocytes it becomes a mechanism of cell death40,78. Compared with wild-type mice, those that lack Nlrp3, ASC or Casp1 genes, or have undergone Nlrp3 siRNA silencing, have a smaller infarct size and preserved cardiac function50,71,79 (Fig. 3) . Of note, differences between Nlrp3 knock-out and wild-type mice were evident early during ischaemia, a finding consistent with the limited expression of inflammasome components during the first 3 hours after injury73,74,79 (Fig. 2).
Fig. 3 |. Prevention of NLRP3 inflammasome formation reduces damage in animal models of ischaemia–reperfusion injury.
Coronary plaque growth and instability are promoted by NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome formation. An atherothrombotic event causes ischaemic ventricular damage, leading to the death of cardiac muscle. During reperfusion, inflammasome-mediated damage promotes further damage and infarct growth. Preventing the formation of the inflammasome — by deleting genes of the inflammasome components (NLRP3, apoptosis speck-like protein containing a caspase recruiting domain (ASC) or caspase 1), by reducing expression of NLRP3 using small interfering RNA (siRNA) or by administering selective NLRP3 inhibitors — reduces infarct size.
Healing and remodelling
In mouse models, delaying treatment with an NLRP3 inhibitor by 1 hour after the beginning of reperfusion did not limit the ability to reduce infarct size. However, a delay of 3 hours led to failure to reduce infarct size73,80 (Fig. 2). These data suggest that inhibition of the NLRP3 inflammasome within 3 hours of reperfusion salvages the portion of myocardium that is rescued by reperfusion but remains in jeopardy and is eventually lost by pyroptosis. Accordingly, inhibition of other inflammasome components limits pyroptosis and infarct size71.
NLRP3 inhibition did not reduce infarct size in a non-reperfused AMI model43. Nevertheless, inhibition of the NLRP3 inflammasome in non-reperfused AMI prevents adverse remodelling, independent of infarct size40,43. Inflammasome formation is present in cardiomyocytes and fibroblasts for several days after AMI40. IL-1β production is increased during AMI and modulates cardiac function and remodelling (Fig. 4). IL-1 blockade with the recombinant human IL-1 receptor antagonist, anakinra (Fig. 5), IL-1trap (analogous to rilonacept) or a mouse anti-IL-1β antibody (analogous to canakinumab) reduced cardiomyocyte cell death, preserved cardiac function and restored β-adrenergic receptor responsiveness in models of ischaemic cardiomyopathy81–85.
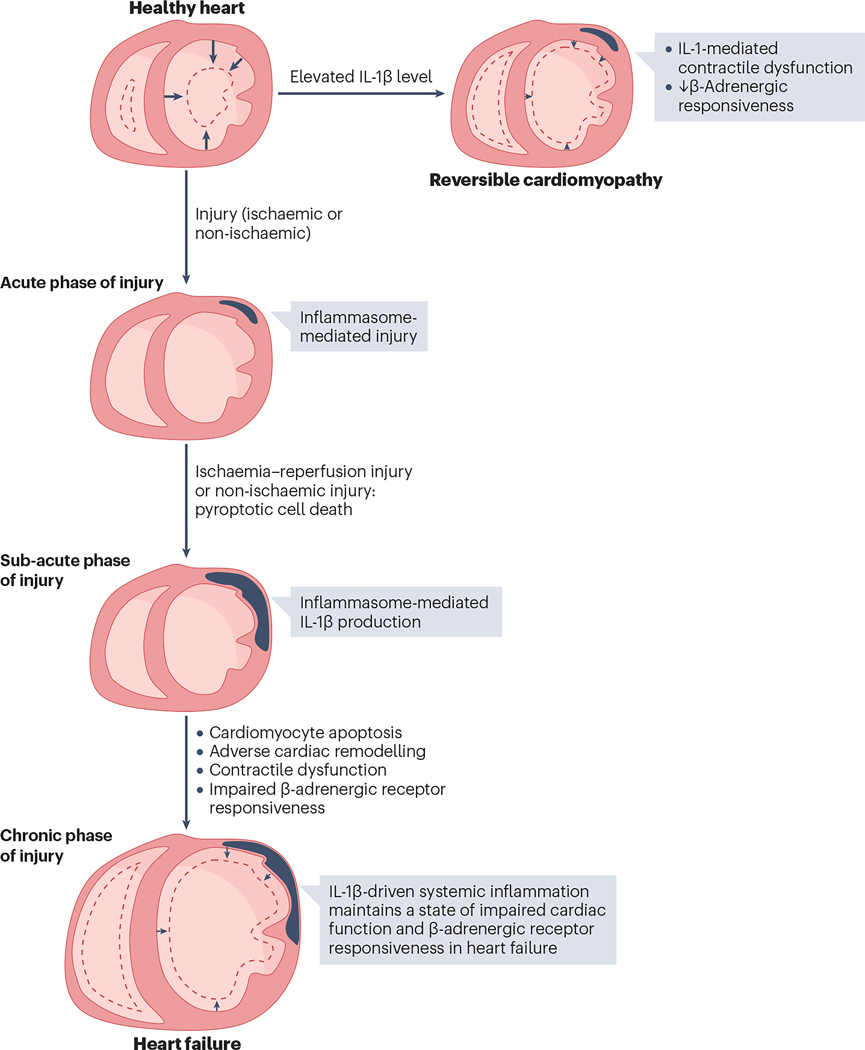
Fig. 4 |. Role of IL-1β in acute injury and progression to heart failure.
The left and right ventricular cavities in diastole and in systole (dotted lines) are shown. IL-1β decreases myocardial contractility and the response to β-adrenergic receptor agonists in cardiomyocytes, even in the absence of acute myocardial injury. In the acute phase that follows ischaemic or non-ischaemic myocardial injury, NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome formation promotes myocardial cell death due to pyroptosis. In the subacute phase, IL-1β induces cardiomyocyte apoptosis, favouring adverse cardiac remodelling and heart failure. Enhanced IL-1β activity in the subacute and chronic phases contribute to impaired myocardial contractility and β-adrenergic responsiveness in heart failure.
Fig. 5 |. Mechanism of action of NLRP3 inflammasome inhibitors tested in experimental models of ischaemic and non-ischaemic injury.
Several NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome inhibitors have been tested in animal models of acute myocardial infarction. Colchicine can act downstream of NLRP3 by inhibiting the polymerization of apoptosis speck-like protein containing a caspase recruiting domain (ASC). OLT1177, BAY 11–7082, INF4E, MCC950 and CY-09 inhibit the ATPase activity of NLRP3. ZYIL1, selnoflast, DFV890 and JC-121 (also known as 16673–34-0) prevent oligomerization of NLRP3 and the interaction with ASC. Ibrutinib inhibits activation of NLRP3 mediated by tyrosine-protein kinase BTK (also known as Bruton kinase). RRx-001 binds NLRP3 and inhibits its interaction with serine/threonine-protein kinase NEK7. IL-1β activity is blocked by the IL-1 inhibitors anakinra (a recombinant IL-1 receptor antagonist), canakinumab (an anti-IL-1β antibody), rilonacept (an IL-1 trap) and goflikicept (a different IL-1 trap). IL-18 activity is blocked by tadekinig α (a recombinant form of IL-18 binding protein; IL-18BP) and by GSK1070806 (an anti-IL-18 antibody). eATP, extracellular ATP.
Treatment of mice with AMI using an anti-IL-1α antibody significantly reduced the acute inflammatory response and infarct size, whereas an IL-1β antibody did not86,87. This finding reflects the fact that IL-1α is an IL-1 isoform that is constitutively expressed in the heart and is active in its precursor form, pro-IL-1α84,86,88. By contrast, IL-18 activity is regulated by the inflammasome. IL-18 contributes to contractile dysfunction in isolated human myocardial strips exposed to simulated ischaemia–reperfusion injury89. In mouse models of AMI, treatment with an anti-IL-18 antibody or recombinant IL-18 binding protein (IL-18BP) reduce infarct size, ischaemia–reperfusion injury, and myocardial inflammation90–92.
Cardiac dysfunction and heart failure
Activation of NLPR3 (triggering), without induction of the scaffold and effector proteins (priming) is insufficient to induce cardiac dysfunction, due to low constitutive expression of inflammasome components32. As such, patients with mutations in the NLRP3 gene (previously known as CIAS1), causing constitutively active, cryopyrin-associated periodic syndromes, show no generalized signs of cardiac dysfunction, but might be vulnerable to loss of cardiac function during the clinically active phases of the disease93. Pro-inflammatory stimuli cause cardiac dysfunction through activation of the NLRP3 inflammasome and release of IL-1β and IL-1832,93–95. IL-1β induces simultaneous changes in the expression and function of L-type calcium channels, phospholamban and sarcoplasmic/endoplasmic reticulum calcium ATPase 2, leading to a functional desensitization of the β-adrenergic receptors, consistent with changes in patients with heart failure96 (Fig. 4). IL-18 seems to mediate many of the effects of IL-1β97. Of note, cardiac dysfunction induced by IL-1β is, at least in part, reversible98.
The NLRP3 inflammasome in non-ischaemic injury
As discussed above, the inflammatory response to injury is a stereotyped response in which the NLRP3 inflammasome is central. Priming and triggering can occur through the membrane and cytoplasmic sensors (i.e. Toll-like receptors and P2X purinoceptor 2 on the membrane and NOD-like receptors in the cell (Fig. 1). Several non-ischaemic processes can induce injury to the heart and cardiovascular system and stimulate an NLRP3 inflammasome-mediated response38.
Atherothrombosis
The NLRP3 inflammasome has a role in the atherothrombotic process that leads to myocardial ischaemia and infarction99. Atherosclerosis refers to changes in the vessel wall characterized by inflammation and accumulation of intracellular and extracellular cholesterol crystals. When the atherosclerotic plaque is complicated by thrombosis (atherothrombosis), blood flow in the vessel is abruptly threatened causing ischaemia and infarction (Fig. 3). Cholesterol and calcium phosphate crystals found in atherosclerotic plaque lead to lysosomal instability, cathepsin B release and NLRP3 inflammasome activation100. Deletion of the Nlrp3, Asc and Il-1α/β genes in the bone marrow of mice with hypercholesterolaemia prevented the development and modified the characteristics of atherosclerotic plaques100–103. In addition, caspase 4 expression in human peripheral blood mononuclear cells increases with the severity of coronary artery disease, and deletion of its ortholog in mice, caspase 11, reduced the progression of atherosclerosis104.
Myocarditis
Myocarditis is an inflammatory process that occurs in response to direct injury of the myocardium by a virus, or a pathological immune response (antigen mimicry or autoimmunity). Myocardial injury stimulates the stereotyped NLRP3 inflammasome-mediated response that promotes further damage and progression to heart failure105,106 (Fig. 4). Caspase 11 activation, part of the non-canonical inflammasome activation pathway, has been reported in the hearts of mice with coxsackievirus B3-induced myocarditis107.
Sarcoidosis
The formation of the NLRP3 inflammasome has also been described in cardiac sarcoidosis. This distinct form of myocarditis is generally chronic, often part of a systemic illness and characterized by myocardial injury mediated by non-caseating granulomatous inflammation with the formation of multinucleated giant cells108 (Fig. 4). Kron et al. found that granulomas in samples of myocardial tissue from three patients with cardiac sarcoidosis and end-stage heart failure contained the inflammasome components ASC, NLRP3 and caspase 1108. Similarly, the presence of NLRP3 inflammasome components in granulomas was also seen in lung samples, and the induction of sarcoid granulomas in mouse lungs using trehalose 6,6’-dimycolate was attenuated by Nlrp3 deletion, pharmacological NLRP3 inhibition or IL-1β blockade109.
Sepsis and septic cardiomyopathy
Sepsis refers to a systemic inflammatory response to infection, in which inflammation is excessive and often a mechanism for multiple organ failure. Cardiac dysfunction during sepsis is referred to as septic cardiomyopathy. Activation of the NLRP3 inflammasome in the heart during sepsis provides a source of cytokines, inducing a paracrine and systemic effect on contractility95,110 (Fig. 4).
Cardiotoxicity
The myocardium is sensitive to toxic injury, stimulating NLRP3 inflammasome-mediated sterile inflammation. A classic example of non-ischaemic, non-infectious injury is caused by the cancer chemotherapeutic agent, doxorubicin, which induces the NLRP3 inflammasome in leukocytes and cardiac cells111 (Fig. 4). Nlrp3–/– or Casp1–/– mice are protected from doxorubicin-induced cardiomyopathy112, and treatment with an NLRP3 inhibitor has been shown to prevent cardiac dysfunction and fibrosis in wild-type mice43.
Chronic non-ischaemic cardiomyopathy
Non-ischaemic cardiomyopathy refers to a broad category of conditions, in which injury to the myocardium is not the result of coronary artery disease or AMI. Cardiac remodelling and heart failure are often the result of pressure overload secondary to hypertension or valvular diseases (Fig. 4). Chronic pressure overload stimulates a pro-inflammatory response in the heart with NLRP3 inflammasome activation113. Calcium/calmodulin-dependent protein kinase II δ seems to be necessary for NLRP3 inflammasome activation in response to pressure overload due to TAC or infusion of angiotensin II in cardiomyocytes41,42. NLRP3 signalling in cardiomyocytes promoted maladaptive left ventricular remodelling114. TAC-induced β-adrenergic signalling contributed to inflammasome priming and NLRP3 activation, leading to ventricular dysfunction, fibrosis and hypertrophy in rats114.
Diabetic cardiomyopathy refers to structural changes in the hearts of patients with diabetes that are not explained by other factors, and that lead heart failure. The consequences of the pro-inflammatory stimuli in diabetes are, at least partly, mediated by the NLRP3 inflammasome115. In mice, a high fat diet (HFD) induces obesity and type 2 diabetes, cardiac remodelling and heart failure with preserved ejection fraction (HFpEF). The adverse metabolic effects, as well as cardiac remodelling and dysfunction, induced by a HFD are mitigated in Nlrp3–/– or Asc–/– mice116. The use of an NLRP3 inhibitor also improved diastolic function in mice fed a HFD117. In a Dahl/salt-sensitive rat model of HFpEF, colchicine treatment increased survival and reduced cardiac dysfunction and fibrosis118. In a HFD mouse model of the nitric oxide synthetases inhibitor, NΩ-nitro-L-arginine methyl ester (L-NAME), the development of HFpEF was associated with increased inflammasome priming and active IL-1β and IL-18 in heart tissue119. Atrial tissue from patients with arrhythmias express elevated levels of NLRP3 inflammasome components and active caspase 1120. Mutant mice expressing cardiomyocyte-specific constitutively active NLRP3 developed arrhythmias that were prevented by NLRP3 inhibition120.
Cardiomyopathies can also be genetically determined and mediated by low-grade chronic injury, as in Duchenne muscular dystrophy. Activation of the NLRP3 inflammasome in such conditions can amplify injury and promote progression to cardiomyopathy121.
Pericarditis
Injury to the mesothelial cells constituting the pericardial sac often causes an inflammatory response. In some cases, such inflammation is perpetuated by activation of the NLRP3 inflammasome and IL-1α and IL-1β released by dying cells and inflammatory cells, respectively122,123 (Fig. 6).
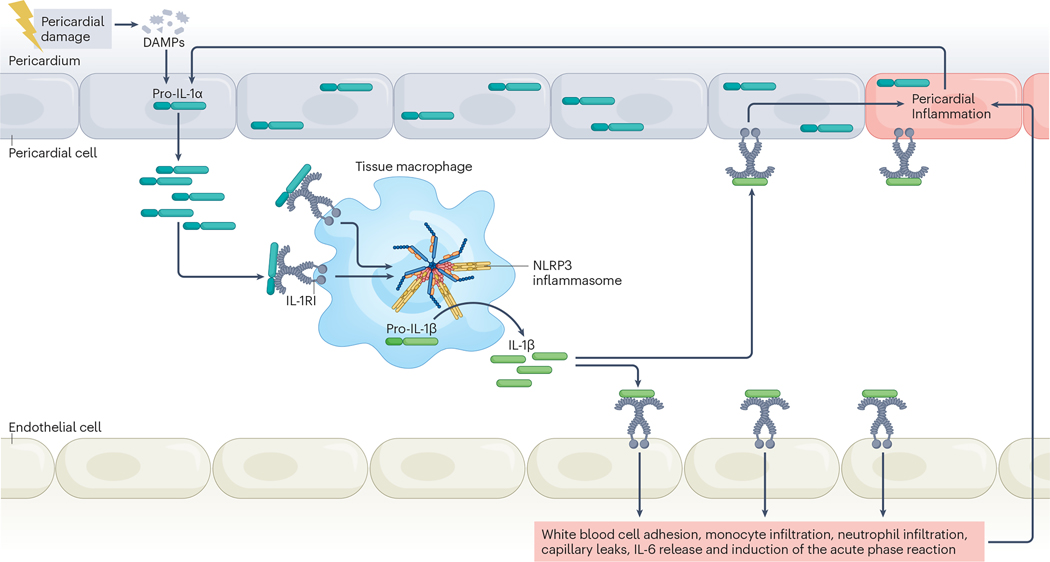
Fig. 6 |. Role of IL-1α and IL-1β in the pathophysiology of recurrent pericarditis.
Injury to the pericardial cells induces the release of intracellular contents. (Pro)-IL-1α released outside the cell is already active, binds the IL-1 receptor type I (IL-1RI) on macrophages and activates the signal, functioning as an alarmin. Macrophages respond to the alarmins by forming and triggering the NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome that processes and releases active IL-1β in its active form. This process amplifies the inflammatory response and induces further injury, which in turn leads to the release of more pro-IL-1α. IL-1β also induces the release of IL-6, which mediates the acute phase reaction associated with inflammation. DAMPs, damage-associated molecular patterns.
NLRP3 inflammasome inhibitors
Preclinical data
Several pharmacological agents are being developed to inhibit NLRP3 inflammasome activation through various mechanisms, including NLRP3–NEK7 interaction and NLRP3 oligomerization. Table 1 and Fig. 5 show the characteristics and proposed sites of action of the NLRP3 inflammasome inhibitors tested in animal models of AMI. Although these inhibitors vary in structure, site of action, efficacy and specificity, the ATPase activity of NLRP3 is a preferred site of action for several agents. The data generated in studies of NLRP3 inflammasome inhibitors, paired with matching genetic mouse models, are important for translational and clinical research. Some of the inhibitors have not been tested in CVD and, therefore, their utility is unknown.
Table 1 |.
NLRP3 inflammasome inhibitors in experimental models of AMI
| Name, chemical name | Chemical structure | Mechanism of action | Doses, route | Animal model | Findings | Refs |
|---|---|---|---|---|---|---|
| JC-121 (or 16673–34-0) 5-chloro-2-methoxy-N-[2-(4- sulfamoylphenyl) ethyl]benzamide and JC-124 5-chloro-N-[2-(4-methyl-sulfamoyl-phenyl)-ethyl]-2-methoxy-benzamide |

|
Prevention of oligomerization in the interaction between NLRP3 and ASC | 5–100 mg/kg, intraperitoneal. Given prior to or up to 1 h in reperfused/non-reperfused AMI | Mouse, coronary artery ligation AMI | Reduction in infarct size at 24 h (measured by pathology, echocardiography and troponin I plasma levels) | 43, 125, 126 |
| Improvement in cardiac remodelling and left ventricular dysfunction in reperfused and non-reperfused AMI | ||||||
| Reduction in caspase 1 activation and cardiomyocyte pyroptosis in the early phases of AMI, and reduction in apoptosis and myocardial fibrosis in the late phases of AMI | ||||||
| MCC950 N-((1,2,3,5,6,7-hexahydro-s-indacen-4-yl)carbamoyl)-4-(2-hydroxypropan-2-yl)furan-2-sulfonamide; N-[[(1,2,3,5,6,7-hexahydro-s-indacen-4-yl)amino]carbonyl]-4-(1-hydroxy-1-methylethyl)-2-furansulfonamide |

|
Inhibition of NLRP3 activation and ATPase activity | Pig model: 3 or 6 mg/kg, intravenous. Given 15 min before reperfusion, then repeated every 24 h for 6 days; (mouse model): 10 mg/kg intraperitoneal, given after ischaemia and for 14 days (mouse model) | Pig model: balloon angioplasty inflation;mouse model: surgical coronary artery ligation | Reduction in infarct size (measured by pathology, echocardiography and troponin I plasma levels) | 128, 129 |
| Improvement in cardiac remodelling and prevention of left ventricular dysfunction | ||||||
| Reduction in local and systemic inflammatory response | ||||||
| OLT1177 (dapansutrile) 3-(methanesulfonyl)propanenitrile |

|
Inhibition of ATPase activity | Ligation model: 6, 60 or 600 mg/kg, intraperitoneal. Given at the same time as, or up to 30 min after, reperfusion; ischaemic cardiomyopathy model: 3.75 or 7.5 mg/kg in chow diet started 7 days after MI and given daily up to 10 weeks | Mouse, surgical coronary artery ligation or chronic ischaemic cardiomyopathy | Reduction in infarct size (measured by pathology and echocardiography) and improvement in contractile reserve | 80, 136 |
| Reduction in end diastolic pressure | ||||||
| BAY 11–7082 3-[(4-methylphenyl)sulfonyl]-(2E)-propenenitrile |

|
Inhibition of ATPase activity | Mouse model: pretreatment (dose not specified) prior to ischaemia or reperfusion; rat model: pretreatment (130 μg/kg) 30 min before ischaemia | Mouse model: ischaemia–reperfusion; rat model: ischaemia–reperfusion | Reduction in infarct size (measured by pathology) | 50, 139, 140 |
| Reduction in inflammasome activity (caspase 1 activity, IL-1β levels) | ||||||
| Preservation of cardiac function | ||||||
| INF4E Ethyl 2-((2-Chlorophenyl)(hydroxy)methyl)acrylate |

|
Inhibition of ATPase activity | 50 μM given 20 minutes prior to ischaemia | Ex vivo myocardial model | Reduction of infarct size at 1 h (measured by pathology, cardiac markers, and contractility) | 75 |
| Reduction in caspase 1 activity, and levels of cleaved IL-1β and gasdermin D levels at 1 h. | ||||||
| CY-09 4-[[4-Oxo-2-thioxo-3-[[3-(trifluoromethyl)phenyl]methyl]-5-thiazolidinylidene]methyl]benzoic acid |

|
Prevention of NLRP3 oligomerization | 5 mg/kg, intraperitoneal. Given three times per week for 4 weeks | Mouse, permanent surgical coronary artery ligation | Reduction in fibrosis and improvement in ventricular function (measured by pathology and echocardiography) | 143 |
| Colchicine N-[(7S)-1,2,3,10-tetramethoxy-9-oxo-6,7-dihydro-5H-benzo[a]heptalen-7-yl]acetamide |

|
Inhibition of NLPR3 and ASC polymerization | 0.1 mg/kg, oral. Given 1 h after surgery then daily for the following 6 days | Mouse, permanent surgical coronary artery ligation | Reduced infarct size (measured by pathology) | 144, 145 |
| Reduction in cytokines and in infiltrating granulocytes and macrophages | ||||||
| Reduction in inflammasome activity (expression of inflammasome components, caspase 1 activity) | ||||||
| Prevention of adverse remodelling and heart failure | ||||||
| Improved survival |
AMI, acute myocardial infarction; ASC, apoptosis speck-like protein containing a caspase recruiting domain; NLRP3, NACHT, LRR and PYD domains-containing protein 3.
Sulfonamides and related molecules.
Glyburide is a sulfonylurea that increases insulin release by pancreatic β cells and is used to treat diabetes. Glyburide inhibits the NLRP3 inflammasome in vitro without suppressing its ATPase activity124,125. However, at the concentration required to inhibit the NLRP3 in vivo, glyburide causes lethal hypoglycaemia in mice125. JC121 (also known as 16673–34-0) and JC124 are sulfonamides that are derived from glyburide, but lack the cycloexylurea moiety group. JC121 and JC124 inhibit the NLRP3 inflammasome, without affecting insulin or inducing hypoglycaemia43,125,126. These agents prevented NLRP3 oligomerization, significantly reduced infarct size and preserved cardiac function in mice with ischaemia–reperfusion injury and those with non-ischaemic injury43,125,126.
MCC950 is a sulfonylurea that selectively inhibits NLRP3 activation and its ATPase activity15,127. MCC950 significantly reduced infarct size in a pig model of ischaemia induced by balloon occlusion of the left anterior descending artery128. The effects of MCC950 in AMI have been confirmed in a mouse model of permanent left main coronary occlusion, in which it reduced fibrosis, ameliorated remodelling and improved cardiac function129. MCC950 was also shown to reduce left ventricular dysfunction, hypertrophy, fibrosis and inflammation in a model of TAC in obese mice130. Furthermore, MCC950 improved survival and reduced post-resuscitation myocardial dysfunction in rats undergoing cardiac arrest and resuscitation131. The development of atherosclerosis in susceptible mice fed a HFD is promoted by the NLRP3 inflammasome and its cytokines, IL-1β and IL-183. MCC950 was reported to reduce atherosclerosis in apolipoprotein E-deficient mice fed a HFD132. In another study, MCC950 prevented aortic aneurism and dissection in mice fed a HFD and given angiotensin II, by reducing caspase 1 and IL-1-dependent activation of matrix metalloproteinase 9133.
OLT1177 (dapansutrile).
A β-sulfonyl nitrile compound that selectively inhibits NLRP3 ATPase activity134,135. Administration of OLT1177 reduced infarct size in mice that underwent experimental ischaemia–reperfusion injury80. OLT1177 has also been tested in mice with severe ischaemic cardiomyopathy. Treatment started 1 week after permanent coronary artery ligation improved diastolic function and increased cardiac reserve compared with control mice136.
RRx-001.
A dinitroazetidine anticancer compound that covalently binds to haemoglobin, increases oxygen delivery and catalyses the production of nitric oxide and ROS in tumour cells. RRx-001 promotes epigenetic modulation and upregulates the expression of the antioxidant protein nuclear factor erythroid 2-related factor 2137. In addition, RRx-001 covalently binds to NLRP3 and prevents its interaction with NEK7138. In vivo, RRx-001 reduced systemic inflammation in separate mouse models of sepsis, colitis and autoimmune encephalomyelitis, all diseases driven by NLRP3 activity138.
BAY 11–7082.
An NF-κB inhibitor that also inhibits NLRP3 ATPase activity139. Mice with experimental ischaemia–reperfusion AMI that were given BAY 11–7082 intraperitoneally, 10 minutes prior to reperfusion, displayed reduced myocardial inflammasome formation and decreased infarct size50. Similarly, BAY 11–7082 administered 30 minutes prior to reperfusion in rats, significantly reduced infarct size and preserved cardiac function140.
INF4E.
An acrylamide derivative that covalently binds to NLRP3 and blocks its ATPase activity141. INF4E administered ex vivo (50 μM) 20 minutes prior to ischaemia on a Langendorff apparatus, reduced inflammasome activity and infarct size75.
CY-09.
Inhibits NLRP3 and its oligomerization through covalent binding to the ATPase domain142. In mice that underwent permanent occlusion of the coronary artery, CY-09 given three times per week for 4 weeks reduced fibrosis and improved heart function143.
Colchicine.
Used for millennia to treat inflammatory disease, colchicine is known to interfere with microtubule assembly and is recognized as an inhibitor of inflammasome aggregation (ASC and NLRP3 polymerization through the pyrin domain)144. Colchicine also inhibits the activation of P2X7 by extracellular ATP144. In mice with non-reperfused AMI, colchicine (0.1 mg/kg/day) significantly reduced inflammasome activation measured after 24 hours, decreased infarct size and ventricular remodelling and increased 7-day survival145.
Ibrutinib.
A small molecule drug that irreversibly binds to BTK, thereby inhibiting Toll-like receptor signalling and NLRP3 inflammasome activation. In a mouse model of sepsis, ibrutinib (30 mg/kg) given intravenously 1 hour after surgery ameliorated ventricular dysfunction associated with sepsis49.
Clinical data
Clinical experience with targeted NLRP3 inflammasome inhibitors is limited. Indirect evidence of the central role of the NLRP3 inflammasome is derived from non-selective NLRP3 inhibitors, such as colchicine, or blockers of IL-1 and IL-18.
Colchicine.
Colchicine is frequently used in patients with acute and chronic inflammatory diseases144,146. In patients with ST-segment elevation AMI, colchicine significantly reduced infarct size in some trials, but not in others (Table 2)147–149. Deftereos et al. reported that colchicine given after percutaneous coronary intervention with a bare-metal stent in patients with diabetes was associated with reduced in-stent restenosis and neointimal hyperplasia150. In COLCOT151, a study of patients with recent (<30 days) AMI, colchicine 0.5 mg daily significantly reduced the risk of ischaemic cardiovascular events when compared with placebo (Table 2). A similar benefit was seen with colchicine 0.5 mg daily administered to patients with stable coronary artery disease in the LoDoCo152 and LoDoCo2153 trials, with a significant reduction in the incidence of atherothrombotic complications compared with placebo in both studies (Table 2). However, colchicine 0.5 mg twice daily did not induce a benefit compared with placebo in patients with stable, chronic heart failure154 (Table 2). Data from several trials of patients with pericarditis indicate that colchicine effectively treats the initial episode and prevents recurrences155–159 (Table 3). However, an unexpected small increase in the rates of non-cardiac death and pneumonia was seen in three separate colchicine trials151,153,160, as well as in a meta-analysis including all available studies161. As of June 2023, colchicine is approved by the FDA for the prevention of atherothrombotic complications in patients with established CVD or multiple cardiovascular risk factors and, although not approved for the treatment of pericarditis, colchicine is considered first-line therapy for initial and recurrent events.
Table 2 |.
Clinical trials of colchicine and IL-1 blockers in AMI, CAD and HF
| Study (year) | Indication (n) | Study design and drug regimen | Main findings for study drug | Refs |
|---|---|---|---|---|
| Colchicine | ||||
| LoDoCo (2013) | Stable CAD (532) | Randomization 1:1 to colchicine 0.5 mg or placebo daily (median duration 2.36 years) | Reduction in the combined cardiovascular end point, due to a large reduction in the incidence of ACS | 152 |
| Colchicine for the prevention of restenosis of bare-metal stents in diabetic patients (2013) | Stable CAD and diabetes mellitus undergoing bare-metal stent implantation (196) | Randomization 1:1 to colchicine 0.5 mg twice daily or placebo for 6 months | Reduction in angiographic in-stent restenosis; significantly less lumen area loss | 150 |
| Colchicine in stable chronic HF (2014) | Stable systolic HF (LVEF ≤40%) (267) | Randomized, double-blinded, trial of colchicine 0.5 mg daily or placebo for 6 months | No significant effect on the >1 class NYHA improvement; no significant effect on treadmill exercise time; significant reduction in CRP levels | 154 |
| Anti-inflammatory treatment with colchicine in AMI (2015) | ST-segment elevation AMI (151) | Randomization 1:1 to colchicine or placebo; loading dose 2.0 mg (1.5 mg initially followed by 0.5 mg after 1 h) and continuing with 0.5 mg twice daily or placebo for 5 days | Reduction in infarct size (measured by cardiac magnetic resonance and by area-under-the-curve for cardiac markers); reduction in peak C reactive protein levels | 151 |
| COLCOT (2019) | Stable CAD (4,745) | Randomization 1:1 to colchicine 0.5 mg or placebo daily (median duration 22.6 months) | Reduction in the composite of death from cardiovascular causes, resuscitated cardiac arrest, AMI, stroke or urgent hospitalization for angina leading to coronary revascularization, due to a large reduction in urgent revascularization | 151 |
| LoDoCo2 (2020) | Stable CAD (5,522) | Randomization 1:1 to colchicine 0.5 mg or placebo daily (median duration 28.6 months) | Reduction in the composite of cardiovascular death, AMI, ischaemic stroke and ischaemia-driven revascularization, due to a large reduction in the rate of AMI and revascularization | 153 |
| The incidence of death from non-cardiovascular causes was higher in the colchicine group than in the placebo group (0.7 vs. 0.5 events per 100 person-years; HR 1.51, 95% CI 0.99–2.31). | ||||
| COPS (2020) | ACS (795) | Randomization 1:1 to colchicine 0.5 mg twice daily for 1 month followed by 0.5 mg daily or placebo for 11 months | No significant effect on the composite end point of death from any cause, ACS, ACS-driven urgent revascularization, and non-cardioembolic ischaemic stroke | 160 |
| The incidence of death from any cause was higher in the colchicine group than in the placebo group (HR 8.20, 95% CI 1.03–65.6). | ||||
| Colchicine and major adverse cardiac events after ACS (2021) | ACS (361) | Randomization 1:1 to colchicine 0.5 mg or placebo daily for 6 months | Reduction in major adverse cardiac events due to a large reduction in recurrent ACS | 212 |
| COPE-PCI (2022) | Stable angina or non-ST-segment elevation AMI (50) | Randomization 1:1 to colchicine 1.0 mg followed by 0.5 mg after 1 h or matching placebo 6–24 hours before intervention | No significant difference in the number of patients with coronary microvascular dysfunction; no significant effect on CRP levels; reduction in troponin levels and blood neutrophil count | 148, 213 |
| PodCAST-PCI (2022) | ST-segment elevation AMI (321) | Randomization 1:1 to colchicine 1.0 mg followed by 0.5 mg daily or placebo until discharge | No significant effect on no-reflow, infarct size or levels of inflammatory biomarkers | 149 |
| IL-1 blockers | ||||
| VCU-ART (2010) | ST-segment elevation AMI (10) | Randomization 1:1 to anakinra 100 mg once daily or placebo for 14 days | Anakinra was safe and favourably affected left ventricular remodelling. Anakinra also blunted the inflammatory response during AMI | 162 |
| VCU-ART2 (2013) | ST-segment elevation AMI (30) | Randomization 1:1 to anakinra 100 mg once daily or placebo for 14 days | Blunting of the inflammatory response during AMI and a trend toward reduced incidence of adverse remodelling or HF at 3 months and at long-term follow up | 163 |
| VCU-ART3 (2020) | ST-segment elevation AMI (99) | Randomization 1:1:1 to anakinra 100 mg once daily, 100 mg twice daily, or placebo for 14 days | Blunting of the inflammatory response during AMI; reduced incidence of new-onset HF and hospitalization for HF; no difference between anakinra once daily versus twice daily | 164, 165 |
| MRC-ILA Heart Study (2015) | Non-ST-segment elevation AMI (182) | Randomization 1:1 to anakinra 100 mg or placebo once daily for 14 days | Blunting of the inflammatory response during AMI; no differences in major adverse cardiac events at 30 days and 3 months, but more events occurring after 6 months in the anakinra group | 166 |
| AIR-HF (2012) | Stable NYHA II–III systolic HF with CRP>2 mg/l (10) | Single arm, open-label; treatment with anakinra 100 mg once daily for 2 weeks | Reduction in CRP levels; improvement in peak aerobic exercise capacity and ventilator efficiency at 2 weeks | 167 |
| ADHF (2016) | Acute decompensated systolic HF with CRP >5 mg/l (30) | Randomized, double-blinded, trial of anakinra 100 mg or placebo twice daily for 3 days and then once daily for 2 weeks | Reduction in CRP levels at 72 h and 14 days and a trend toward more favourable effects on signs of congestion and LVEF | 170 |
| REDHART (2017) | Recently (within 2 weeks of hospital discharge) decompensated systolic HF with CRP >2 mg/l (60) | Randomized, double-blinded trial of anakinra 100 mg once daily continued for 2 weeks or for 12 weeks or placebo for 2 weeks (1:1:1) | Reduction in CRP levels; improvement in peak aerobic exercise capacity and quality of life questionnaires, and a trend toward reduced HF readmissions at 6 months, with 12-week treatment | 168 |
| D-HART (2014) | Stable NYHA II–III diastolic HF with LVEF >50% and CRP >2 mg/l (12) | Randomized, double-blinded, cross-over trial of anakinra 100 mg or placebo once daily for 2 weeks | Reduction in CRP levels; improvement in peak aerobic exercise capacity at 2 weeks | 167 |
| D-HART2 (2018) | Stable NYHA II–III diastolic HF with LVEF >50% and CRP >2 mg/l (31) | Randomized, double-blinded trial of anakinra 100 mg once daily or placebo for 12 weeks (2:1) | No improvement in peak aerobic exercise capacity or ventilatory efficiency. Reduction in CRP and NT-proBNP levels and increase in exercise time. | 171 |
| CANTOS (2017) | Prior AMI with CRP >2 mg/l (at least 30 days after AMI) (10,060) | Randomized, double-blinded trial of canakinumab 50 mg, 150 mg or 300 mg or placebo (1:1:1:1.5) (median duration 3.5 years) | For canakinumab 150 mg: significant reduction in the incidence of the composite end point of cardiac death, non-fatal AMI or non-fatal stroke (canakinumab 150 mg); significant reduction in the incidence of recurrent AMI, unstable angina and need for revascularization | 179, 181 |
| Significant reduction in the rate of HF-related hospitalizations and death (p for trend = 0.025) | ||||
| For canakinumab 150 mg and 300 mg: small reduction in cancer-related mortality | ||||
| Small increase in the risk of fatal infections with canakinumab (all doses combined) | ||||
ACS, acute coronary syndromes; AMI, acute myocardial infarction; CAD, coronary artery disease; CRP, C reactive protein; HF, heart failure; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association.
Table 3 |.
Clinical trials of colchicine and IL-1 blockers in pericarditis
| Study (year) | Indication (n) | Study design and drug regimen | Main findings for study drug | Refs |
|---|---|---|---|---|
| Colchicine | ||||
| COPE trial (2005) | Acute pericarditis (120) | Randomization 1:1 to colchicine loading dose of 1.0–2.0 mg followed by 0.5–1.0 mg daily plus aspirin versus aspirin alone for 3 months | Resolution of symptoms at 72 h and reduction in recurrence rate at 18 months | 155 |
| CORP trial (2011) | Recurrent pericarditis – first episode (120) | Randomization 1:1 to colchicine loading dose of 1.0–2.0 mg followed by 0.5–1.0 mg daily or placebo in addition to standard of care for 3 months | Resolution of symptoms at 72 h and reduction in recurrence rate at 18 months | 157 |
| ICAP trial (2013) | Acute pericarditis – first episode (240) | Randomization 1:1 to colchicine 0.5–1.0 mg or placebo for 3 months | Resolution of symptoms at 72 h; reduction in the rate of incessant or recurrent pericarditis and in hospitalizations; increase in number of patients in remission at 1 week | 158 |
| CORP2 trial (2014) | Recurrent pericarditis – multiple episodes (240) | Randomization 1:1 to colchicine 0.5–1.0 daily or placebo in addition to standard of care for 6 months | Resolution of symptoms at 72 h; reduction in the rate of incessant or recurrent pericarditis and in pericarditis-related hospitalizations | 159 |
| IL-1 blockers | ||||
| AIRTRIP trial (2016) | Recurrent, colchicine resistant, corticosteroid dependent pericarditis (21) | Open-label run-in with anakinra 100 mg daily for 2 months, followed by randomized withdrawal to anakinra continuation or placebo for 6 months | Reduction of recurrences; increase in flare-free survival; frequent injection site reactions | 176 |
| Acute effects of anakinra in patients with acute pericarditis (2020) | Symptomatic acute pericarditis despite initial treatment with colchicine and NSAIDs (5) | Open-label treatment with anakinra 100 mg | Significant reduction in pain at 6 hand 24 h; reduction of IL-6 at 24 h | 175 |
| RHAPSODY trial (2021) | Recurrent pericarditis, unresponsive to standard treatment (86 run-in; 61 randomized) | Open-label run-in with rilonacept 320 mg loading dose, followed by 160 mg weekly, followed by randomized withdrawal to rilonacept continuation or placebo | Resolution of acute flares (open label run-in phase); reduction in the recurrence rate | 186 |
| Goflikicept in recurrent pericarditis (2022) | Recurrent pericarditis, unresponsive to standard treatment (22) | Open-label run-in with goflikicept 80 mg every 2 weeks (with or without loading dose), followed by randomized withdrawal to goflikicept continuation or placebo | Resolution of acute flares (open label run-in phase); reduction in the recurrence rate | 187 |
NSAIDs, non-steroidal anti-inflammatory drugs.
IL-1 blockers.
Several IL-1 blockers are available for clinical use and are being studied in patients with CVD3. Anakinra (Kineret, Sobi), a recombinant human IL-1 receptor antagonist, was first studied in VCU-ART162, in 10 patients with ST-segment elevated AMI, at the standard dose of 100 mg daily for up to 14 days. Two follow-up phase II studies (VCU-ART2163 and VCU-ART3164) followed, with 30 and 99 patients, respectively. Anakinra was well tolerated, blunted the acute systemic inflammatory response associated with myocardial injury (measured by C-reactive protein serum levels) and reduced the incidence of heart failure and related complications165 (Table 2; Fig. 7). In the MRC-ILA Heart study166 of 182 patients with non-ST-segment elevation AMI, anakinra administered for 14 days reduced the acute inflammatory response, albeit with a paradoxical increase in late atherothrombotic events (Table 2; Fig. 7). When studied in patients with acute and chronic systolic and diastolic heart failure, anakinra (once or twice daily) reduced C-reactive protein levels, improved cardiorespiratory fitness, Doppler echocardiographic parameters and quality of life measures and showed a trend toward a reduction in hospitalizations167–171 (Table 2; Fig. 6). A systematic review of studies reporting on IL-1-targeted therapies showed that anakinra is effective in treating myocarditis and pericarditis associated with Still disease (a rare type of inflammatory arthritis)172. A case report from 2016 suggests a role for anakinra in the treatment of viral and idiopathic myocarditis173. The first randomized clinical trial of IL-1 blockade in patients with acute myocarditis (the ARAMIS trial174) was completed in 2023. In patients with pericarditis, anakinra reduces systemic inflammation and improves pain within 24 hours175, and in patients with recurrent pericarditis refractory to first-line therapy (including colchicine and prednisone), anakinra treated the acute event and prevented recurrence176 (Table 3). Finally, a small pilot study of patients with rheumatoid arthritis showed that anakinra improved vascular and left ventricular function177,178.
Fig. 7 |. Overview of phase II–III trials of IL-1 blockers in acute myocardial infarction and heart failure.
a, The CANTOS trial. Canakinumab 150 mg every 3 months significantly reduced the incidence of cardiovascular death, non-fatal myocardial infarction and stroke in patients with prior myocardial infarction (MI) and residual inflammatory risk. b, A pooled analysis of the VCU-ART trials. Anakinra 100 mg daily (or twice daily) for 14 days significantly reduced the incidence of heart failure or death in patients with ST-segment elevation MI (STEMI). c, The MRC-ILA Heart trial. Anakinra 100 mg daily for 14 days was associated with an unexpected increase in late cardiac and cerebral ischaemic events in patients with non-ST segment elevation MI (NSTEMI). d, The REDHART trial. Anakinra 100 mg daily for 12 weeks, but not for 2 weeks only, showed a favourable rate of hospital readmission for heart failure in patients with recently decompensated systolic heart failure as compared with placebo and an observational registry.
Canakinumab (Ilaris, Novartis) is an antibody that selectively blocks IL-1β (Table 2; Fig. 5). In CANTOS179, the largest cytokine trial ever completed, 10,061 patients with prior AMI and elevated C-reactive protein (>2 mg/l) were randomly assigned to receive canakinumab (50 mg, 150 mg or 300 mg) or placebo every 3 months (median treatment duration 3.5 years). In the canakinumab 150 mg arm, the incidence of cardiovascular death, AMI, stroke, urgent revascularization and heart failure-related hospitalizations was significantly reduced, independent of the effects on classic cardiovascular risk factors179–182. Canakinumab improved cardiac function and cardiorespiratory fitness in patients with heart failure and reduced ejection fraction (HFrEF), and walking distance in patients with low-extremity peripheral arterial disease183,184. The benefits of canakinumab seemed to be most pronounced in the subgroup of patients with somatic mutations associated with clonal hematopoiesis185.
Rilonacept (Arcalyst, Regeneron Pharmaceuticals, Inc.) is a recombinant chimeric fusion protein functioning as a ‘trap’ for IL-1β and IL-1α3 (Table 3; Fig. 5). In the RHAPSODY trial186 of patients with recurrent pericarditis, rilonacept (320 mg loading dose, then 160 mg weekly) effectively treated the acute episode and significantly reduced the recurrence rate (Table 3). Consequently, rilonacept is now approved by the FDA for the treatment of recurrent pericarditis186.
Goflikicept (RPH104) is also a recombinant chimeric fusion trap protein, structurally different from rilonacept, binding both IL-1β and IL-1α187 (Table 3; Fig. 5). Goflikicept significantly reduced the risk of recurrence in patients with recurrent or refractory pericarditis in a phase II–III trial187. Goflikicept is also being studied in a phase II trial of patients with ST-segment elevation AMI188.
IL-18 and IL-6 blockers.
Data from CANTOS189 have shown that substantial residual inflammatory risk, related to both IL-18 and IL-6, remains after IL-1β inhibition with canakinumab. Potential future therapies to address this residual risk include GSK1070806, a monoclonal antibody directed against IL-18, and tadekinig α, a recombinant human IL-18 binding protein that sequesters IL-18. However, these drugs are not available for clinical use and data on cardiovascular outcomes are lacking190,191 (Fig. 5).
IL-6 is a secondary cytokine, acting downstream of IL-1β and other cytokines, and often considered a surrogate for IL-1 activity192. Several IL-6 blockers are available, or under development, for clinical use across a wide range of inflammatory conditions. Tocilizumab (Actemra/Ro-Actemra, Chugai Pharmaceutical Co. Ltd (Roche)), an IL-6 receptor blocking antibody, was shown to increase the myocardial salvage ratio in the ASSAIL-MI trial193 of patients with ST-segment elevation AMI, and to reduce myocardial injury in patients with out-of-hospital cardiac arrest (the IMICA trial194). Tocilizumab, however, interfered with the cardiovascular and metabolic benefits of exercise training in abdominally obese, but otherwise healthy, adults195. Another IL-6 blocking antibody, ziltivekimab, was shown to reduce biomarkers of inflammation and thrombosis relevant to atherosclerosis in the phase II, dose-ranging RESCUE trial196. Ziltivekimab 15 mg monthly is now under investigation in two large phase III cardiovascular outcome studies (the ZEUS197 and HERMES198 trials).
Targeted inflammasome inhibitors.
As discussed above, several targeted inflammasome inhibitors are under development (Table 1), but the clinical data for these agents are scarce at this time. OLT1177 (dapansutrile) has a favourable safety profile in healthy individuals as well as in patients with HFrEF199. Efficacy was dose-dependent, with favourable signals for 2000 mg per day in patients with heart failure199 or acute monoarticular gout flares200. DFV890 (IFM-2427) is a sulfonimidamide with a structure similar to MCC950, but with some key modifications, which is being studied across a wide spectrum of inflammatory disease. DFV890 had acceptable safety and biological activity in a phase IIa trial of patients with severe COVID 19-associated pneumonia201, and is in phase II testing to treat osteoarthritis202. ZYIL1, a NLRP3 inhibitor derived from MCC950, is in the early phase of clinical development. ZYIL1 showed an acceptable safety profile and biological activity in healthy volunteers and is being studied in patients with the autoinflammatory disease cryopyrin-associated periodic syndromes (CAPS)203,204. No data are yet available on the effects of ZYIL1 in CVD. Selnoflast (RG6418/IZD334) is a sulfonamide carboxamide compound that showed a good safety profile in a phase Ib study of patients with ulcerative colitis205 (a disease driven by NLRP3 inflammasome activity). This agents is under investigation in phase I clinical trials of healthy volunteers and patients with CAPS206. However, a study in individuals at high cardiovascular risk was discontinued prematurely207. HT-6184, which targets NEK7, is being tested in healthy individuals208. Several small molecule inhibitors of NLRP3 are being studied in clinical trials for various indications — VTX2735 for CAPS209, NT-0249 and NT-0796 for neuro-inflammation210 and RRx-001 for advanced small cell lung cancer211.
Conclusions
The NLRP3 inflammasome is a macromolecular intracellular structure that functions as a sensor for injury. This complex amplifies the inflammatory response and tissue injury by regulating the processing and release of IL-1β and IL-18, and causing inflammatory cell death (pyroptosis). The preclinical experience with genetically-modified mouse models and the use of targeted inhibitors has stimulated a clinical research programme in CVD. During the past 5 years, selective NLRP3 inhibitors have been studied in phase I–II clinical trials. Colchicine, a non-selective NLRP3 inflammasome inhibitor, has been shown to be efficacious not only in the treatment of pericarditis, for which it is considered part of the standard of care, but also in atherothrombotic risk reduction in patients with coronary artery disease, and is now approved for this indication by the FDA. IL-1 blockers, already clinically available and routinely used for various chronic inflammatory conditions, have been shown in phase Ib–III to reduce cardiovascular risk and morbidity across a wide range of cardiovascular pathologies, including AMI, heart failure, myocarditis and, most recently, recurrent pericarditis, for which rilonacept now has a FDA-approved indication. Inhibitors of IL-6, downstream of IL-1, are also clinically available and are being studied in phase Ib–III trials of patients with CVD. Although not yet available for clinical use, IL-18 blockers represent a potential future approach to treatment.
Key points.
The NACHT, LRR and PYD domains-containing protein 3 (NLRP3) inflammasome is a macromolecular, intracellular structure that functions as a sensor for injury.
NLRP3 inflammasome activation amplifies the inflammatory response and tissue injury by regulating the processing and release of IL-1β and IL-18, and causing cell death by pyroptosis.
Preclinical studies with genetically-modified mouse models and the use of targeted inhibitors have shown that inhibiting activation of the NLRP3 inflammasome reduces inflammatory injury and adverse remodelling.
Colchicine, a non-selective NLRP3 inflammasome inhibitor, has been shown to be efficacious in the treatment of pericarditis and in reducing atherothrombotic risk in patients with coronary artery disease.
IL-1 blockers have been shown in phase Ib–III to reduce cardiovascular risk and morbidity across a wide range of cardiovascular diseases, including myocardial infarction, heart failure, acute myocarditis and recurrent pericarditis.
Targeted NLRP3 inflammasome inhibitors and blockers of IL-18 and IL-6, downstream of IL-1, are in clinical testing for a variety of cardiovascular diseases.
Acknowledgements
A.A. is supported by National Institute of Health, Bethesda, MD, USA, grants AG076360, HL139943, and HL150115 and S.T. is supported by a National Institute of Health, Bethesda grant HL150115.
Footnotes
Competing interests
A.A. has served as a consultant to Cardiol, Implicit Bioscience, Janssen, Kiniksa, Novo Nordisk, Olatec, R-Pharm, Sanofi and Serpin Pharma. S.T. has received research grants from Cardiol, Kiniksa and Olatec.
References
- 1.Tsao CW et al. Heart Disease and Stroke Statistics-2022 Update: A Report From the American Heart Association. Circulation 145, e153–e639 (2022). [DOI] [PubMed] [Google Scholar]
- 2.Lenz A, Franklin GA & Cheadle WG Systemic inflammation after trauma. Injury 38, 1336–1345 (2007). [DOI] [PubMed] [Google Scholar]
- 3.Abbate A. et al. Interleukin-1 and the Inflammasome as Therapeutic Targets in Cardiovascular Disease. Circ. Res 126, 1260–1280 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Toldo S. & Abbate A. The NLRP3 inflammasome in acute myocardial infarction. Nat. Rev. Cardiol 15, 203–214 (2018). [DOI] [PubMed] [Google Scholar]
- 5.Westman PC et al. Inflammation as a Driver of Adverse Left Ventricular Remodeling After Acute Myocardial Infarction. J. Am. Coll. Cardiol 67, 2050–2060 (2016). [DOI] [PubMed] [Google Scholar]
- 6.Seropian IM, Toldo S, Van Tassell BW & Abbate A. Anti-inflammatory strategies for ventricular remodeling following ST-segment elevation acute myocardial infarction. J. Am. Coll. Cardiol 63, 1593–1603 (2014). [DOI] [PubMed] [Google Scholar]
- 7.Gao X-M, White DA, Dart AM & Du X-J Post-infarct cardiac rupture: recent insights on pathogenesis and therapeutic interventions. Pharmacol. Ther 134, 156–179 (2012). [DOI] [PubMed] [Google Scholar]
- 8.Abbate A. et al. Alterations in the interleukin-1/interleukin-1 receptor antagonist balance modulate cardiac remodeling following myocardial infarction in the mouse. PloS One 6, e27923 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Savvatis K. et al. Interleukin-23 deficiency leads to impaired wound healing and adverse prognosis after myocardial infarction. Circ. Heart Fail 7, 161–171 (2014). [DOI] [PubMed] [Google Scholar]
- 10.Dinarello CA Overview of the IL-1 family in innate inflammation and acquired immunity. Immunol. Rev 281, 8–27 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Swanson KV, Deng M. & Ting JP-Y The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat. Rev. Immunol 19, 477–489 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zheng D, Liwinski T. & Elinav E. Inflammasome activation and regulation: toward a better understanding of complex mechanisms. Cell Discov. 6, 1–22 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Viganò E. et al. Human caspase-4 and caspase-5 regulate the one-step non-canonical inflammasome activation in monocytes. Nat. Commun 6, 8761 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Matikainen S, Nyman TA & Cypryk W. Function and Regulation of Noncanonical Caspase-4/5/11 Inflammasome. J. Immunol. Baltim. Md 1950 204, 3063–3069 (2020). [DOI] [PubMed] [Google Scholar]
- 15.Ma Q. Pharmacological Inhibition of the NLRP3 Inflammasome: Structure, Molecular Activation, and Inhibitor-NLRP3 Interaction. Pharmacol. Rev 75, 487–520 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lu A. et al. Unified polymerization mechanism for the assembly of ASC-dependent inflammasomes. Cell 156, 1193–1206 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dinarello CA Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 117, 3720–3732 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Westermann D. et al. Cardioprotective and anti-inflammatory effects of interleukin converting enzyme inhibition in experimental diabetic cardiomyopathy. Diabetes 56, 1834–1841 (2007). [DOI] [PubMed] [Google Scholar]
- 19.Xia S. et al. Gasdermin D pore structure reveals preferential release of mature interleukin-1. Nature 593, 607–611 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Santa Cruz Garcia AB, Schnur KP, Malik AB & Mo GCH Gasdermin D pores are dynamically regulated by local phosphoinositide circuitry. Nat. Commun 13, 52 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Erickson HP Size and Shape of Protein Molecules at the Nanometer Level Determined by Sedimentation, Gel Filtration, and Electron Microscopy. Biol. Proced. Online 11, 32–51 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rider P, Carmi Y, Voronov E. & Apte RN Interleukin-1α. Semin. Immunol. 25, 430–438 (2013). [DOI] [PubMed] [Google Scholar]
- 23.Gross O. et al. Inflammasome activators induce interleukin-1α secretion via distinct pathways with differential requirement for the protease function of caspase-1. Immunity 36, 388–400 (2012). [DOI] [PubMed] [Google Scholar]
- 24.Merkle S. et al. A Role for Caspase-1 in Heart Failure. Circ. Res 100, 645–653 (2007). [DOI] [PubMed] [Google Scholar]
- 25.Liu X. et al. Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores. Nature 535, 153–158 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kayagaki N. et al. NINJ1 mediates plasma membrane rupture during lytic cell death. Nature 591, 131–136 (2021). [DOI] [PubMed] [Google Scholar]
- 27.Magnani L, Colantuoni M. & Mortellaro A. Gasdermins: New Therapeutic Targets in Host Defense, Inflammatory Diseases, and Cancer. Front. Immunol 13, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tsuchiya K. et al. Gasdermin D mediates the maturation and release of IL-1α downstream of inflammasomes. Cell Rep. 34, 108887 (2021). [DOI] [PubMed] [Google Scholar]
- 29.Shao W, Yeretssian G, Doiron K, Hussain SN & Saleh M. The caspase-1 digestome identifies the glycolysis pathway as a target during infection and septic shock. J. Biol. Chem 282, 36321–36329 (2007). [DOI] [PubMed] [Google Scholar]
- 30.Shen J. et al. Caspase-1 recognizes extended cleavage sites in its natural substrates. Atherosclerosis 210, 422–429 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Downs KP, Nguyen H, Dorfleutner A. & Stehlik C. An overview of the non-canonical inflammasome. Mol. Aspects Med 76, 100924 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Toldo S. et al. Independent roles of the priming and the triggering of the NLRP3 inflammasome in the heart. Cardiovasc. Res 105, 203–212 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li X. et al. NOD2 deficiency protects against cardiac remodeling after myocardial infarction in mice. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol 32, 1857–1866 (2013). [DOI] [PubMed] [Google Scholar]
- 34.Chevriaux A. et al. Cathepsin B Is Required for NLRP3 Inflammasome Activation in Macrophages, Through NLRP3 Interaction. Front. Cell Dev. Biol 8, 167 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rajamäki K. et al. Cholesterol Crystals Activate the NLRP3 Inflammasome in Human Macrophages: A Novel Link between Cholesterol Metabolism and Inflammation. PLoS ONE 5, e11765 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Toldo S, Mauro AG, Cutter Z. & Abbate A. Inflammasome, pyroptosis, and cytokines in myocardial ischemia-reperfusion injury. Am. J. Physiol. Heart Circ. Physiol 315, H1553–H1568 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Takahashi M. NLRP3 inflammasome as a novel player in myocardial infarction. Int. Heart. J 55, 101–105 (2014). [DOI] [PubMed] [Google Scholar]
- 38.Mezzaroma E, Abbate A. & Toldo S. The inflammasome in heart failure. Curr. Opin. Physiol 19, 105–112 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Netea MG et al. Differential requirement for the activation of the inflammasome for processing and release of IL-1beta in monocytes and macrophages. Blood 113, 2324–2335 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mezzaroma E. et al. The inflammasome promotes adverse cardiac remodeling following acute myocardial infarction in the mouse. Proc. Natl. Acad. Sci. U. S. A 108, 19725–19730 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Suetomi T. et al. Inflammation and NLRP3 Inflammasome Activation Initiated in Response to Pressure Overload by Ca2+/Calmodulin-Dependent Protein Kinase II δ Signaling in Cardiomyocytes Are Essential for Adverse Cardiac Remodeling. Circulation 138, 2530–2544 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Willeford A. et al. CaMKIIδ-mediated inflammatory gene expression and inflammasome activation in cardiomyocytes initiate inflammation and induce fibrosis. JCI Insight 3, e97054, 97054 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Marchetti C. et al. Pharmacologic Inhibition of the NLRP3 Inflammasome Preserves Cardiac Function After Ischemic and Nonischemic Injury in the Mouse. J. Cardiovasc. Pharmacol 66, 1 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang H. et al. Inflammasome-independent NLRP3 is required for epithelial-mesenchymal transition in colon cancer cells. Exp. Cell Res 342, 184–192 (2016). [DOI] [PubMed] [Google Scholar]
- 45.Wang W. et al. Inflammasome-independent NLRP3 augments TGF-β signaling in kidney epithelium. J. Immunol. Baltim. Md 1950 190, 1239–1249 (2013). [DOI] [PubMed] [Google Scholar]
- 46.Zuurbier CJ et al. Deletion of the Innate Immune NLRP3 Receptor Abolishes Cardiac Ischemic Preconditioning and Is Associated with Decreased Il-6/STAT3 Signaling. PLOS ONE 7, e40643 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.He Y, Zeng MY, Yang D, Motro B. & Núñez G. NEK7 is an essential mediator of NLRP3 activation downstream of potassium efflux. Nature 530, 354–357 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ito M. et al. Bruton’s tyrosine kinase is essential for NLRP3 inflammasome activation and contributes to ischaemic brain injury. Nat. Commun 6, 7360 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.O’Riordan CE et al. Bruton’s Tyrosine Kinase Inhibition Attenuates the Cardiac Dysfunction Caused by Cecal Ligation and Puncture in Mice. Front. Immunol 10, 2129 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 50.Liu Y. et al. TXNIP mediates NLRP3 inflammasome activation in cardiac microvascular endothelial cells as a novel mechanism in myocardial ischemia/reperfusion injury. Basic Res. Cardiol 109, 415 (2014). [DOI] [PubMed] [Google Scholar]
- 51.He K. et al. Inhibition of NLRP3 inflammasome by thioredoxin-interacting protein in mouse Kupffer cells as a regulatory mechanism for non-alcoholic fatty liver disease development. Oncotarget 8, 37657–37672 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Elshaer SL et al. Deletion of TXNIP Mitigates High-Fat Diet-Impaired Angiogenesis and Prevents Inflammation in a Mouse Model of Critical Limb Ischemia. Antioxid. Basel Switz 6, 47 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Masters SL et al. Activation of the NLRP3 inflammasome by islet amyloid polypeptide provides a mechanism for enhanced IL-1β in type 2 diabetes. Nat. Immunol 11, 897–904 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Muri J, Thut H, Feng Q. & Kopf M. Thioredoxin-1 distinctly promotes NF-κB target DNA binding and NLRP3 inflammasome activation independently of Txnip. eLife 9, e53627 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sun Q, Fan J, Billiar TR & Scott MJ Inflammasome and Autophagy Regulation: A Two-way Street. Mol. Med 23, 188–195 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Saitoh T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1beta production. Nature 456, 264–268 (2008). [DOI] [PubMed] [Google Scholar]
- 57.Nakahira K. et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat. Immunol 12, 222–230 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shi C-S et al. Activation of autophagy by inflammatory signals limits IL-1β production by targeting ubiquitinated inflammasomes for destruction. Nat. Immunol 13, 255–263 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Li L. et al. ATP6AP2 knockdown in cardiomyocyte deteriorates heart function via compromising autophagic flux and NLRP3 inflammasome activation. Cell Death Discov. 8, 161 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mao S, Chen P, Pan W, Gao L. & Zhang M. Exacerbated post-infarct pathological myocardial remodelling in diabetes is associated with impaired autophagy and aggravated NLRP3 inflammasome activation. ESC Heart Fail. 9, 303–317 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhang D. et al. Activation of autophagy inhibits nucleotide-binding oligomerization domain-like receptor protein 3 inflammasome activation and attenuates myocardial ischemia-reperfusion injury in diabetic rats. J. Diabetes Investig 11, 1126–1136 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Paik S, Kim JK, Silwal P, Sasakawa C. & Jo E-K An update on the regulatory mechanisms of NLRP3 inflammasome activation. Cell. Mol. Immunol 18, 1141–1160 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kim JS, He L, Qian T. & Lemasters JJ Role of the mitochondrial permeability transition in apoptotic and necrotic death after ischemia/reperfusion injury to hepatocytes. Curr. Mol. Med 3, 527–535 (2003). [DOI] [PubMed] [Google Scholar]
- 64.Zhou R, Yazdi AS, Menu P. & Tschopp J. A role for mitochondria in NLRP3 inflammasome activation. Nature 469, 221–225 (2011). [DOI] [PubMed] [Google Scholar]
- 65.Sadatomi D. et al. Mitochondrial function is required for extracellular ATP-induced NLRP3 inflammasome activation. J. Biochem. (Tokyo) 161, 503–512 (2017). [DOI] [PubMed] [Google Scholar]
- 66.Park S. et al. Defective mitochondrial fission augments NLRP3 inflammasome activation. Sci. Rep 5, 15489 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mills EL, Kelly B. & O’Neill LAJ Mitochondria are the powerhouses of immunity. Nat. Immunol 18, 488–498 (2017). [DOI] [PubMed] [Google Scholar]
- 68.Iyer SS et al. Mitochondrial Cardiolipin Is Required for Nlrp3 Inflammasome Activation. Immunity 39, 311–323 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Dagvadorj J. et al. Recruitment of pro-IL-1α to mitochondrial cardiolipin, via shared LC3 binding domain, inhibits mitophagy and drives maximal NLRP3 activation. Proc. Natl. Acad. Sci. U. S. A 118, e2015632118 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Pereira AC et al. Mitochondria Fusion upon SERCA Inhibition Prevents Activation of the NLRP3 Inflammasome in Human Monocytes. Cells 11(3):433 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kawaguchi M. et al. Inflammasome activation of cardiac fibroblasts is essential for myocardial ischemia/reperfusion injury. Circulation 123, 594–604 (2011). [DOI] [PubMed] [Google Scholar]
- 72.Wang L, Qu P, Zhao J. & Chang Y. NLRP3 and downstream cytokine expression elevated in the monocytes of patients with coronary artery disease. Arch. Med. Sci. AMS 10, 791–800 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Toldo S. et al. Inhibition of the NLRP3 inflammasome limits the inflammatory injury following myocardial ischemia–reperfusion in the mouse. Int. J. Cardiol 209, 215–220 (2016). [DOI] [PubMed] [Google Scholar]
- 74.Sandanger Ø et al. The NLRP3 inflammasome is up-regulated in cardiac fibroblasts and mediates myocardial ischaemia-reperfusion injury. Cardiovasc. Res 99, 164–174 (2013). [DOI] [PubMed] [Google Scholar]
- 75.Mastrocola R. et al. Pharmacological Inhibition of NLRP3 Inflammasome Attenuates Myocardial Ischemia/Reperfusion Injury by Activation of RISK and Mitochondrial Pathways. Oxid. Med. Cell. Longev 2016, 5271251 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Takahashi M. Cell-Specific Roles of NLRP3 Inflammasome in Myocardial Infarction. J. Cardiovasc. Pharmacol 74, 188 (2019). [DOI] [PubMed] [Google Scholar]
- 77.Toldo S. et al. The inflammasome in myocardial injury and cardiac remodeling. Antioxid. Redox Signal 22, 1146–1161 (2015). [DOI] [PubMed] [Google Scholar]
- 78.Toldo S. et al. Induction of microRNA-21 with exogenous hydrogen sulfide attenuates myocardial ischemic and inflammatory injury in mice. Circ. Cardiovasc. Genet 7, 311–320 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Jong WMC et al. Nlrp3 plays no role in acute cardiac infarction due to low cardiac expression. Int. J. Cardiol 177, 41–43 (2014). [DOI] [PubMed] [Google Scholar]
- 80.Toldo S. et al. The NLRP3 Inflammasome Inhibitor, OLT1177 (Dapansutrile), Reduces Infarct Size and Preserves Contractile Function After Ischemia Reperfusion Injury in the Mouse. J. Cardiovasc. Pharmacol 73, 215–222 (2019). [DOI] [PubMed] [Google Scholar]
- 81.Abbate A. et al. Anakinra, a recombinant human interleukin-1 receptor antagonist, inhibits apoptosis in experimental acute myocardial infarction. Circulation 117, 2670–2683 (2008). [DOI] [PubMed] [Google Scholar]
- 82.Toldo S. et al. Interleukin-1β blockade improves left ventricular systolic/diastolic function and restores contractility reserve in severe ischemic cardiomyopathy in the mouse. J. Cardiovasc. Pharmacol 64, 1–6 (2014). [DOI] [PubMed] [Google Scholar]
- 83.Toldo S. et al. Interleukin-1β blockade improves cardiac remodelling after myocardial infarction without interrupting the inflammasome in the mouse. Exp. Physiol 98, 734–745 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lugrin J. et al. Cutting edge: IL-1α is a crucial danger signal triggering acute myocardial inflammation during myocardial infarction. J. Immunol. Baltim. Md 1950 194, 499–503 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Van Tassell BW et al. Interleukin-1 trap attenuates cardiac remodeling after experimental acute myocardial infarction in mice. J. Cardiovasc. Pharmacol 55, 117–122 (2010). [DOI] [PubMed] [Google Scholar]
- 86.Mauro AG et al. Reduction of Myocardial Ischemia-Reperfusion Injury by Inhibiting Interleukin-1 Alpha. J. Cardiovasc. Pharmacol 69, 156–160 (2017). [DOI] [PubMed] [Google Scholar]
- 87.Sager HB et al. Targeting Interleukin-1β Reduces Leukocyte Production After Acute Myocardial Infarction. Circulation 132, 1880–1890 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.England H, Summersgill HR, Edye ME, Rothwell NJ & Brough D. Release of Interleukin-1α or Interleukin-1β Depends on Mechanism of Cell Death. J. Biol. Chem 289, 15942–15950 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Pomerantz BJ, Reznikov LL, Harken AH & Dinarello CA Inhibition of caspase 1 reduces human myocardial ischemic dysfunction via inhibition of IL-18 and IL-1beta. Proc. Natl. Acad. Sci. U. S. A 98, 2871–2876 (2001). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Venkatachalam K. et al. Neutralization of interleukin-18 ameliorates ischemia/reperfusion-induced myocardial injury. J. Biol. Chem 284, 7853–7865 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gu H. et al. The protective role of interleukin-18 binding protein in a murine model of cardiac ischemia/reperfusion injury. Transpl. Int. Off. J. Eur. Soc. Organ Transplant 28, 1436–1444 (2015). [DOI] [PubMed] [Google Scholar]
- 92.Quader M, Mezzaroma E, Kenning K. & Toldo S. Modulation of Interleukin-1 and −18 Mediated Injury in Donation after Circulatory Death Mouse Hearts. J. Surg. Res 257, 468–476 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Pörksen G. et al. Periodic fever, mild arthralgias, and reversible moderate and severe organ inflammation associated with the V198M mutation in the CIAS1 gene in three German patients--expanding phenotype of CIAS1 related autoinflammatory syndrome. Eur. J. Haematol 73, 123–127 (2004). [DOI] [PubMed] [Google Scholar]
- 94.Kumar A. et al. Tumor necrosis factor alpha and interleukin 1beta are responsible for in vitro myocardial cell depression induced by human septic shock serum. J. Exp. Med 183, 949–958 (1996). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Fujimura K. et al. NLRP3 inflammasome-driven IL-1β and IL-18 contribute to lipopolysaccharide-induced septic cardiomyopathy. J. Mol. Cell. Cardiol 180, 58–68 (2023). [DOI] [PubMed] [Google Scholar]
- 96.Van Tassell BW, Toldo S, Mezzaroma E. & Abbate A. Targeting interleukin-1 in heart disease. Circulation 128, 1910–1923 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Toldo S. et al. Interleukin-18 mediates interleukin-1-induced cardiac dysfunction. Am. J. Physiol. Heart Circ. Physiol 306, H1025–1031 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Van Tassell BW, Seropian IM, Toldo S, Mezzaroma E. & Abbate A. Interleukin-1β induces a reversible cardiomyopathy in the mouse. Inflamm. Res. Off. J. Eur. Histamine Res. Soc. Al 62, 637–640 (2013). [DOI] [PubMed] [Google Scholar]
- 99.Takahashi M. NLRP3 inflammasome as a key driver of vascular disease. Cardiovasc. Res 118, 372–385 (2022). [DOI] [PubMed] [Google Scholar]
- 100.Duewell P. et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 464, 1357–1361 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Alexander MR et al. Genetic inactivation of IL-1 signaling enhances atherosclerotic plaque instability and reduces outward vessel remodeling in advanced atherosclerosis in mice. J. Clin. Invest 122, 70–79 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Dragoljevic D. et al. Inhibition of interleukin-1β signalling promotes atherosclerotic lesion remodelling in mice with inflammatory arthritis. Clin. Transl. Immunol 9, e1206 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Hettwer J. et al. Interleukin-1β suppression dampens inflammatory leucocyte production and uptake in atherosclerosis. Cardiovasc. Res 118, 2778–2791 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Jiang M. et al. Caspase-11-Gasdermin D-Mediated Pyroptosis Is Involved in the Pathogenesis of Atherosclerosis. Front. Pharmacol 12, (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Toldo S. et al. Formation of the inflammasome in acute myocarditis. Int. J. Cardiol 171, e119–121 (2014). [DOI] [PubMed] [Google Scholar]
- 106.Pappritz K. et al. Colchicine prevents disease progression in viral myocarditis via modulating the NLRP3 inflammasome in the cardiosplenic axis. ESC Heart Fail. 9, 925–941 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Yu Y. et al. Inhibition of calpain alleviates coxsackievirus B3-induced myocarditis through suppressing the canonical NLRP3 inflammasome/caspase-1-mediated and noncanonical caspase-11-mediated pyroptosis pathways. Am. J. Transl. Res 12, 1954–1964 (2020). [PMC free article] [PubMed] [Google Scholar]
- 108.Kron J. et al. Inflammasome Formation in Granulomas in Cardiac Sarcoidosis. Circ. Arrhythm. Electrophysiol 12, e007582 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Huppertz C. et al. The NLRP3 inflammasome pathway is activated in sarcoidosis and involved in granuloma formation. Eur. Respir. J 55, 1900119 (2020). [DOI] [PubMed] [Google Scholar]
- 110.Zhang W. et al. Cardiac fibroblasts contribute to myocardial dysfunction in mice with sepsis: the role of NLRP3 inflammasome activation. PloS One 9, e107639 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Mauro AG et al. NLRP3-mediated inflammation in cardio-oncology: sterile yet harmful. Transl. Res. J. Lab. Clin. Med 252, 9–20 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Zeng C. et al. NLRP3 inflammasome-mediated pyroptosis contributes to the pathogenesis of non-ischemic dilated cardiomyopathy. Redox Biol. 34, 101523 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Li R. et al. Triptolide attenuates pressure overload-induced myocardial remodeling in mice via the inhibition of NLRP3 inflammasome expression. Biochem. Biophys. Res. Commun 485, 69–75 (2017). [DOI] [PubMed] [Google Scholar]
- 114.Dang S. et al. Blockade of β-adrenergic signaling suppresses inflammasome and alleviates cardiac fibrosis. Ann. Transl. Med 8, 127 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Zhang L, Ai C, Bai M, Niu J. & Zhang Z. NLRP3 Inflammasome/Pyroptosis: A Key Driving Force in Diabetic Cardiomyopathy. Int. J. Mol. Sci 23, 10632 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Sokolova M. et al. NLRP3 Inflammasome Promotes Myocardial Remodeling During Diet-Induced Obesity. Front. Immunol 10, 1621 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Carbone S. et al. An Orally Available NLRP3 Inflammasome Inhibitor Prevents Western Diet-Induced Cardiac Dysfunction in Mice. J. Cardiovasc. Pharmacol 72, 303–307 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Shen S. et al. Colchicine alleviates inflammation and improves diastolic dysfunction in heart failure rats with preserved ejection fraction. Eur. J. Pharmacol 929, 175126 (2022). [DOI] [PubMed] [Google Scholar]
- 119.Xia Y-Y et al. Involvement of pyroptosis pathway in epicardial adipose tissue - myocardium axis in experimental heart failure with preserved ejection fraction. Biochem. Biophys. Res. Commun 636, 62–70 (2022). [DOI] [PubMed] [Google Scholar]
- 120.Yao C. et al. Enhanced Cardiomyocyte NLRP3 Inflammasome Signaling Promotes Atrial Fibrillation. Circulation 138, 2227–2242 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Dubuisson N. et al. Inhibiting the inflammasome with MCC950 counteracts muscle pyroptosis and improves Duchenne muscular dystrophy. Front. Immunol 13, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mauro AG et al. The Role of NLRP3 Inflammasome in Pericarditis: Potential for Therapeutic Approaches. JACC Basic Transl. Sci 6, 137–150 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Vecchié A. et al. Interleukin-1 and the NLRP3 Inflammasome in Pericardial Disease. Curr. Cardiol. Rep 23, 157 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Lamkanfi M. et al. Glyburide inhibits the Cryopyrin/Nalp3 inflammasome. J. Cell Biol 187, 61–70 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Marchetti C. et al. A novel pharmacologic inhibitor of the NLRP3 inflammasome limits myocardial injury after ischemia-reperfusion in the mouse. J. Cardiovasc. Pharmacol 63, 316–322 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Fulp J. et al. Structural Insights of Benzenesulfonamide Analogues as NLRP3 Inflammasome Inhibitors: Design, Synthesis, and Biological Characterization. J. Med. Chem 61, 5412–5423 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Coll RC et al. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat. Med 21, 248–255 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.van Hout GPJ et al. The selective NLRP3-inflammasome inhibitor MCC950 reduces infarct size and preserves cardiac function in a pig model of myocardial infarction. Eur. Heart J 38, 828–836 (2017). [DOI] [PubMed] [Google Scholar]
- 129.Gao R. et al. The selective NLRP3-inflammasome inhibitor MCC950 reduces myocardial fibrosis and improves cardiac remodeling in a mouse model of myocardial infarction. Int. Immunopharmacol 74, 105575 (2019). [DOI] [PubMed] [Google Scholar]
- 130.Wang M. et al. MCC950, a Selective NLRP3 Inhibitor, Attenuates Adverse Cardiac Remodeling Following Heart Failure Through Improving the Cardiometabolic Dysfunction in Obese Mice. Front. Cardiovasc. Med 9, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zheng G. et al. The Selective NLRP3-inflammasome inhibitor MCC950 Mitigates Post-resuscitation Myocardial Dysfunction and Improves Survival in a Rat Model of Cardiac Arrest and Resuscitation. Cardiovasc. Drugs Ther 37, 423–433 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.van der Heijden T. et al. NLRP3 Inflammasome Inhibition by MCC950 Reduces Atherosclerotic Lesion Development in Apolipoprotein E-Deficient Mice-Brief Report. Arterioscler. Thromb. Vasc. Biol 37, 1457–1461 (2017). [DOI] [PubMed] [Google Scholar]
- 133.Ren P. et al. Targeting the NLRP3 Inflammasome With Inhibitor MCC950 Prevents Aortic Aneurysms and Dissections in Mice. J. Am. Heart Assoc 9, e014044 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Marchetti C. et al. OLT1177, a β-sulfonyl nitrile compound, safe in humans, inhibits the NLRP3 inflammasome and reverses the metabolic cost of inflammation. Proc. Natl. Acad. Sci. U. S. A 115, E1530–E1539 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Marchetti C. et al. NLRP3 inflammasome inhibitor OLT1177 suppresses joint inflammation in murine models of acute arthritis. Arthritis Res. Ther 20, 169 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Aliaga J. et al. Preservation of Contractile Reserve and Diastolic Function by Inhibiting the NLRP3 Inflammasome with OLT1177® (Dapansutrile) in a Mouse Model of Severe Ischemic Cardiomyopathy Due to Non-Reperfused Anterior Wall Myocardial Infarction. Mol. Basel Switz 26, 3534 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Oronsky B. et al. Discovery of RRx-001, a Myc and CD47 Downregulating Small Molecule with Tumor Targeted Cytotoxicity and Healthy Tissue Cytoprotective Properties in Clinical Development. J. Med. Chem 64, 7261–7271 (2021). [DOI] [PubMed] [Google Scholar]
- 138.Chen Y. et al. RRx-001 ameliorates inflammatory diseases by acting as a potent covalent NLRP3 inhibitor. Cell. Mol. Immunol 18, 1425–1436 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Juliana C. et al. Anti-inflammatory compounds parthenolide and Bay 11–7082 are direct inhibitors of the inflammasome. J. Biol. Chem 285, 9792–9802 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Kim YS et al. BAY 11–7082, a nuclear factor-κB inhibitor, reduces inflammation and apoptosis in a rat cardiac ischemia-reperfusion injury model. Int. Heart. J 51, 348–353 (2010). [DOI] [PubMed] [Google Scholar]
- 141.Cocco M. et al. Design, Synthesis, and Evaluation of Acrylamide Derivatives as Direct NLRP3 Inflammasome Inhibitors. ChemMedChem 11, 1790–1803 (2016). [DOI] [PubMed] [Google Scholar]
- 142.Jiang H. et al. Identification of a selective and direct NLRP3 inhibitor to treat inflammatory disorders. J. Exp. Med 214, 3219–3238 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Gao R-F et al. The covalent NLRP3-inflammasome inhibitor Oridonin relieves myocardial infarction induced myocardial fibrosis and cardiac remodeling in mice. Int. Immunopharmacol 90, 107133 (2021). [DOI] [PubMed] [Google Scholar]
- 144.Zhang F. et al. Therapeutic potential of colchicine in cardiovascular medicine: a pharmacological review. Acta Pharmacol. Sin 43, 2173–2190 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Fujisue K. et al. Colchicine Improves Survival, Left Ventricular Remodeling, and Chronic Cardiac Function After Acute Myocardial Infarction. Circ. J. Off. J. Jpn. Circ. Soc 81, 1174–1182 (2017). [DOI] [PubMed] [Google Scholar]
- 146.Imazio M. & Nidorf M. Colchicine and the heart. Eur. Heart J 42, 2745–2760 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Deftereos S. et al. Anti-Inflammatory Treatment With Colchicine in Acute Myocardial Infarction: A Pilot Study. Circulation 132, 1395–1403 (2015). [DOI] [PubMed] [Google Scholar]
- 148.Cole J. et al. Colchicine to Prevent Periprocedural Myocardial Injury in Percutaneous Coronary Intervention: The COPE-PCI Pilot Trial. Circ. Cardiovasc. Interv 14, e009992 (2021). [DOI] [PubMed] [Google Scholar]
- 149.Hosseini SH et al. Preprocedural Colchicine in Patients With Acute ST-elevation Myocardial Infarction Undergoing Percutaneous Coronary Intervention: A Randomized Controlled Trial (PodCAST-PCI). J. Cardiovasc. Pharmacol 80, 592–599 (2022). [DOI] [PubMed] [Google Scholar]
- 150.Deftereos S. et al. Colchicine treatment for the prevention of bare-metal stent restenosis in diabetic patients. J. Am. Coll. Cardiol 61, 1679–1685 (2013). [DOI] [PubMed] [Google Scholar]
- 151.Tardif J-C et al. Efficacy and Safety of Low-Dose Colchicine after Myocardial Infarction. N. Engl. J. Med 381, 2497–2505 (2019). [DOI] [PubMed] [Google Scholar]
- 152.Nidorf SM, Eikelboom JW, Budgeon CA & Thompson PL Low-dose colchicine for secondary prevention of cardiovascular disease. J. Am. Coll. Cardiol 61, 404–410 (2013). [DOI] [PubMed] [Google Scholar]
- 153.Nidorf SM et al. Colchicine in Patients with Chronic Coronary Disease. N. Engl. J. Med 383, 1838–1847 (2020). [DOI] [PubMed] [Google Scholar]
- 154.Deftereos S. et al. Anti-inflammatory treatment with colchicine in stable chronic heart failure: a prospective, randomized study. JACC Heart Fail. 2, 131–137 (2014). [DOI] [PubMed] [Google Scholar]
- 155.Imazio M. et al. Colchicine in addition to conventional therapy for acute pericarditis: results of the COlchicine for acute PEricarditis (COPE) trial. Circulation 112, (2005). [DOI] [PubMed] [Google Scholar]
- 156.Imazio M. et al. Colchicine as first-choice therapy for recurrent pericarditis: results of the CORE (COlchicine for REcurrent pericarditis) trial. Arch. Intern. Med 165, 1987–1991 (2005). [DOI] [PubMed] [Google Scholar]
- 157.Imazio M. et al. Colchicine for recurrent pericarditis (CORP): a randomized trial. Ann. Intern. Med 155, 409–414 (2011). [DOI] [PubMed] [Google Scholar]
- 158.Imazio M. et al. A randomized trial of colchicine for acute pericarditis. N. Engl. J. Med 369, 1522–1528 (2013). [DOI] [PubMed] [Google Scholar]
- 159.Imazio M. et al. Efficacy and safety of colchicine for treatment of multiple recurrences of pericarditis (CORP-2): a multicentre, double-blind, placebo-controlled, randomised trial. Lancet Lond. Engl 383, 2232–2237 (2014). [DOI] [PubMed] [Google Scholar]
- 160.Tong DC et al. Colchicine in Patients With Acute Coronary Syndrome. Circulation (2020) doi: 10.1161/CIRCULATIONAHA.120.050771. [DOI] [PubMed] [Google Scholar]
- 161.Fiolet ATL et al. Efficacy and safety of low-dose colchicine in patients with coronary disease: a systematic review and meta-analysis of randomized trials. Eur. Heart J 42, 2765–2775 (2021). [DOI] [PubMed] [Google Scholar]
- 162.Abbate A. et al. Interleukin-1 Blockade With Anakinra to Prevent Adverse Cardiac Remodeling After Acute Myocardial Infarction (Virginia Commonwealth University Anakinra Remodeling Trial [VCU-ART] Pilot Study). Am. J. Cardiol 105, 1371–1377.e1 (2010). [DOI] [PubMed] [Google Scholar]
- 163.Abbate A. et al. Effects of interleukin-1 blockade with anakinra on adverse cardiac remodeling and heart failure after acute myocardial infarction [from the Virginia Commonwealth University-Anakinra Remodeling Trial (2) (VCU-ART2) pilot study]. Am. J. Cardiol 111, 1394–1400 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Abbate A. et al. Interleukin-1 Blockade Inhibits the Acute Inflammatory Response in Patients With ST-Segment-Elevation Myocardial Infarction. J. Am. Heart Assoc 9, e014941 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Abbate A. et al. Interleukin-1 blockade with anakinra and heart failure following ST-segment elevation myocardial infarction: results from a pooled analysis of the VCUART clinical trials. Eur. Heart J. Cardiovasc. Pharmacother 8, 503–510 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Morton AC et al. The effect of interleukin-1 receptor antagonist therapy on markers of inflammation in non-ST elevation acute coronary syndromes: the MRC-ILA Heart Study. Eur. Heart J 36, 377–384 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Van Tassell BW et al. Effects of interleukin-1 blockade with anakinra on aerobic exercise capacity in patients with heart failure and preserved ejection fraction (from the D-HART pilot study). Am. J. Cardiol 113, 321–327 (2014). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Van Tassell BW et al. Interleukin-1 Blockade in Recently Decompensated Systolic Heart Failure: Results From REDHART (Recently Decompensated Heart Failure Anakinra Response Trial). Circ. Heart Fail 10, e004373 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Van Tassell BW et al. Enhanced interleukin-1 activity contributes to exercise intolerance in patients with systolic heart failure. PloS One 7, e33438 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Van Tassell BW et al. Interleukin-1 Blockade in Acute Decompensated Heart Failure: A Randomized, Double-Blinded, Placebo-Controlled Pilot Study. J. Cardiovasc. Pharmacol 67, 544–551 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Tassell Van B. W. et al. IL-1 Blockade in Patients With Heart Failure With Preserved Ejection Fraction. Circ Heart Fail 11(8):e005036 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Arnold DD, Yalamanoglu A. & Boyman O. Systematic Review of Safety and Efficacy of IL-1-Targeted Biologics in Treating Immune-Mediated Disorders. Front. Immunol 13, (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Cavalli G. et al. Treating Life-Threatening Myocarditis by Blocking Interleukin-1. Crit. Care Med 44, e751–754 (2016). [DOI] [PubMed] [Google Scholar]
- 174.Kerneis M. et al. Rationale and design of the ARAMIS trial: Anakinra versus placebo, a double blind randomized controlled trial for the treatment of acute myocarditis. Arch Cardiovasc Dis. doi: 10.1016/j.acvd.2023.07.004 (2023). [DOI] [PubMed] [Google Scholar]
- 175.Wohlford GF et al. Acute Effects of Interleukin-1 Blockade Using Anakinra in Patients With Acute Pericarditis. J. Cardiovasc. Pharmacol 76, 50–52 (2020). [DOI] [PubMed] [Google Scholar]
- 176.Brucato A. et al. Effect of Anakinra on Recurrent Pericarditis Among Patients With Colchicine Resistance and Corticosteroid Dependence: The AIRTRIP Randomized Clinical Trial. JAMA 316, 1906–1912 (2016). [DOI] [PubMed] [Google Scholar]
- 177.Ikonomidis I. et al. Inhibition of interleukin-1 by anakinra improves vascular and left ventricular function in patients with rheumatoid arthritis. Circulation 117, 2662–2669 (2008). [DOI] [PubMed] [Google Scholar]
- 178.Ikonomidis I. et al. Lowering interleukin-1 activity with anakinra improves myocardial deformation in rheumatoid arthritis. Heart Br. Card. Soc 95, 1502–1507 (2009). [DOI] [PubMed] [Google Scholar]
- 179.Ridker PM et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med 377, 1119–1131 (2017). [DOI] [PubMed] [Google Scholar]
- 180.Everett BM et al. Anti-Inflammatory Therapy With Canakinumab for the Prevention of Hospitalization for Heart Failure. Circulation 139, 1289–1299 (2019). [DOI] [PubMed] [Google Scholar]
- 181.Everett BM et al. Inhibition of Interleukin-1β and Reduction in Atherothrombotic Cardiovascular Events in the CANTOS Trial. J. Am. Coll. Cardiol 76, 1660–1670 (2020). [DOI] [PubMed] [Google Scholar]
- 182.Rothman AM et al. Effects of Interleukin-1β Inhibition on Blood Pressure, Incident Hypertension, and Residual Inflammatory Risk: A Secondary Analysis of CANTOS. Hypertens. Dallas Tex 1979 75, 477–482 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Trankle CR et al. Usefulness of Canakinumab to Improve Exercise Capacity in Patients With Long-Term Systolic Heart Failure and Elevated C-Reactive Protein. Am. J. Cardiol 122, 1366–1370 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Ks R. et al. A randomized, placebo-controlled trial of canakinumab in patients with peripheral artery disease. Vasc. Med. Lond. Engl 24, (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Svensson EC et al. TET2-Driven Clonal Hematopoiesis and Response to Canakinumab: An Exploratory Analysis of the CANTOS Randomized Clinical Trial. JAMA Cardiol. 7, 521–528 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Klein AL et al. Phase 3 Trial of Interleukin-1 Trap Rilonacept in Recurrent Pericarditis. N. Engl. J. Med 384, 31–41 (2021). [DOI] [PubMed] [Google Scholar]
- 187.Myachikova VY et al. Treatment of Idiopathic Recurrent Pericarditis With Goflikicept: Phase II/III Study Results. J Am Coll Cardiol 82(1):30–40 (2023). [DOI] [PubMed] [Google Scholar]
- 188.Samsonov M, Bogin V, Van Tassell BW & Abbate A. Interleukin-1 blockade with RPH-104 in patients with acute ST-elevation myocardial infarction: study design and rationale. J. Transl. Med 19, 169 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Ridker PM, MacFadyen JG, Thuren T. & Libby P. Residual inflammatory risk associated with interleukin-18 and interleukin-6 after successful interleukin-1β inhibition with canakinumab: further rationale for the development of targeted anti-cytokine therapies for the treatment of atherothrombosis. Eur. Heart J 41, 2153–2163 (2020). [DOI] [PubMed] [Google Scholar]
- 190.Mistry P. et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of single-dose antiinterleukin- 18 mAb GSK1070806 in healthy and obese subjects. Int. J. Clin. Pharmacol. Ther 52, 867–879 (2014). [DOI] [PubMed] [Google Scholar]
- 191.Gabay C. et al. Open-label, multicentre, dose-escalating phase II clinical trial on the safety and efficacy of tadekinig alfa (IL-18BP) in adult-onset Still’s disease. Ann. Rheum. Dis 77, 840–847 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Ridker PM & Rane M. Interleukin-6 Signaling and Anti-Interleukin-6 Therapeutics in Cardiovascular Disease. Circ. Res 128, 1728–1746 (2021). [DOI] [PubMed] [Google Scholar]
- 193.Broch K. et al. Randomized Trial of Interleukin-6 Receptor Inhibition in Patients With Acute ST-Segment Elevation Myocardial Infarction. J. Am. Coll. Cardiol 77, 1845–1855 (2021). [DOI] [PubMed] [Google Scholar]
- 194.Meyer MAS et al. Treatment Effects of Interleukin-6 Receptor Antibodies for Modulating the Systemic Inflammatory Response After Out-of-Hospital Cardiac Arrest (The IMICA Trial): A Double-Blinded, Placebo-Controlled, Single-Center, Randomized, Clinical Trial. Circulation 143, 1841–1851 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Christensen RH et al. Aerobic Exercise Induces Cardiac Fat Loss and Alters Cardiac Muscle Mass Through an Interleukin-6 Receptor-Dependent Mechanism: Cardiac Analysis of a Double-Blind Randomized Controlled Clinical Trial in Abdominally Obese Humans. Circulation 140, 1684–1686 (2019). [DOI] [PubMed] [Google Scholar]
- 196.Ridker PM et al. IL-6 inhibition with ziltivekimab in patients at high atherosclerotic risk (RESCUE): a double-blind, randomised, placebo-controlled, phase 2 trial. Lancet Lond. Engl 397, 2060–2069 (2021). [DOI] [PubMed] [Google Scholar]
- 197.NIH National Library of Medicine. ZEUS - A Research Study to Look at How Ziltivekimab Works Compared to Placebo in People With Cardiovascular Disease, Chronic Kidney Disease and Inflammation (ZEUS). https://clinicaltrials.gov/study/ClinicalTrials.gov NCT05021835 (2023). [Google Scholar]
- 198.NIH National Library of Medicine. A Research Study to Look at How Ziltivekimab Works Compared to Placebo in People With Heart Failure and Inflammation (HERMES). ClinicalTrials.gov. https://clinicaltrials.gov/study/NCT05636176 (2023). [Google Scholar]
- 199.Wohlford GF et al. Phase 1B, Randomized, Double-Blinded, Dose Escalation, Single-Center, Repeat Dose Safety and Pharmacodynamics Study of the Oral NLRP3 Inhibitor Dapansutrile in Subjects With NYHA II-III Systolic Heart Failure. J. Cardiovasc. Pharmacol 77, 49–60 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Klück V. et al. Dapansutrile, an oral selective NLRP3 inflammasome inhibitor, for treatment of gout flares: an open-label, dose-adaptive, proof-of-concept, phase 2a trial. Lancet Rheumatol. 2, e270–e280 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Madurka I. et al. DFV890: a new oral NLRP3 inhibitor-tested in an early phase 2a randomised clinical trial in patients with COVID-19 pneumonia and impaired respiratory function. Infection 51, 641–654 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.NIH National Library of Medicine. Study of Efficacy, Safety and Tolerability of DFV890 in Patients With Knee Osteoarthritis. NCT04886258. ClinicalTrials.gov https://clinicaltrials.gov/study/NCT04886258. [Google Scholar]
- 203.Parmar DV et al. Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of the Oral NLRP3 Inflammasome Inhibitor ZYIL1: First-in-Human Phase 1 Studies (Single Ascending Dose and Multiple Ascending Dose). Clin. Pharmacol. Drug Dev 12, 202–211 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Zydus Lifesciences Limited. A Phase 2a, Prospective, Open-Label Study to Evaluate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of ZYIL1 in Subjects With Cryopyrin Associated Periodic Syndromes (CAPS). https://clinicaltrials.gov/ct2/show/NCT05186051 (2022). [Google Scholar]
- 205.Klughammer B. et al. P805 Selnoflast, a potent NLRP3 inhibitor - results from a phase 1b experimental medicine study in patients with Ulcerative Colitis. J. Crohns Colitis 17, i938 (2023). [Google Scholar]
- 206.Inflazome UK Ltd. A Phase 1, Randomised, Double-Blind, Placebo-Controlled, Single and Multiple Ascending Dose Study to Determine the Safety, Tolerability, Pharmacokinetics, Pharmacodynamics and Food Effect of IZD334 in Healthy Adult Participants as Well as an Open-label Cohort to Confirm the Safety, Pharmacokinetics, and Pharmacodynamics in Adult Patients With Cryopyrin-Associated Periodic Syndromes. https://clinicaltrials.gov/ct2/show/NCT04086602 (2020). [Google Scholar]
- 207.Clinical Trials Register. https://www.clinicaltrialsregister.eu/ctr-search/trial/2020-000942-32/NL.
- 208.Therapeutics Halia, Inc. A Phase 1, Single-Center, Randomized, Placebo-controlled, Single Ascending Dose Study to Evaluate the Safety, Tolerability, Pharmacokinetics, and Pharmacodynamics of HT-6184 in Healthy Human Subjects https://clinicaltrials.gov/ct2/show/NCT05447546 (2022). [Google Scholar]
- 209.Zomagen Biosciences, Ltd. A Phase 2A, Single-Arm Study to Evaluate the Safety and Clinical Activity of VTX2735 in Participants With Cryopyrin-Associated Periodic Syndrome (CAPS). https://clinicaltrials.gov/ct2/show/NCT05812781 (2023).
- 210.Pipeline. Nodthera https://www.nodthera.com/pipeline/.
- 211.EpicentRx, Inc. A Phase 3, Controlled, Open-label, Randomized Study of RRx-001 Administered Sequentially With a Platinum Doublet or a Platinum Doublet in Third-Line or Beyond Small Cell Lung Cancer. https://clinicaltrials.gov/ct2/show/NCT03699956 (2022). [Google Scholar]
- 212.Akrami M. et al. Effects of colchicine on major adverse cardiac events in next 6-month period after acute coronary syndrome occurrence; a randomized placebo-control trial. BMC Cardiovasc Disord. 21, 583 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Cole J. et al. COlchicine to Prevent PeriprocEdural Myocardial Injury in Percutaneous Coronary Intervention (COPE-PCI): Coronary Microvascular Physiology Pilot Substudy. J Interv Cardiol. 2022, 1098429 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]