Abstract
Fungal pathogens cause a wide range of infections in humans, from superficial to disfiguring, allergic syndromes, and life-threatening invasive infections, affecting over a billion individuals globally. With an estimated 1.5 million deaths annually attributable to them, fungal pathogens are a major cause of mortality in humans, especially people with underlying immunosuppression. The continuous increase in the population of individuals at risk of fungal infections in sub-Saharan Africa, such as HIV patients, tuberculosis patients, intensive care patients, patients with haematological malignancies, transplant (haematopoietic stem cell and organ) recipients and the growing global threat of multidrug-resistant fungal strains, raise the need for an appreciation of the region’s perspective on antifungal usage and resistance. In addition, the unavailability of recently introduced novel antifungal drugs in sub-Saharan Africa further calls for regular evaluation of resistance to antifungal agents in these settings. This is critical for ensuring appropriate and optimal use of the limited available arsenal to minimise antifungal resistance. This review, therefore, elaborates on the multifaceted nature of fungal resistance to the available antifungal drugs on the market and further provides insights into the prevalence of fungal infections and the use of antifungal agents in sub-Saharan Africa.
Keywords: Antifungal agents, antifungal resistance, fungal pathogens, prevalence, sub-Saharan Africa
Introduction
Fungi are a major threat to the lives of immunocompromised individuals. 1 They can colonise different sites of their human hosts, including the gastrointestinal tract, respiratory tract, bloodstream, and female genitals, 2 which are of public health concern. Fungal infections could either be endogenous in origin or be transmitted via inhalation, direct skin contact with humans or animals, or close proximity to contaminated surfaces. 3
The prevalence of invasive fungal infections (IFIs) is increasing worldwide due to the expansion of existing at-risk populations and the emergence of new at-risk groups, such as persons with recent COVID-19 infections. 4 The global attributable death toll from these infections exceeds 2.5 million, with more than seven million individuals affected annually. 5 In sub-Saharan Africa, fungal infections significantly contribute to the overall disease burden, primarily driven by factors such as HIV, tuberculosis, admission to intensive care, haematological malignancies, transplantation (haematopoietic stem cells and organs) and poverty. 6 Available data from epidemiological and other studies suggest a high burden of fungal infections in sub-Saharan Africa.5,7
The impact of fungal infections has been exacerbated by the increasing occurrence of antifungal-resistant strains, which can be attributed to the widespread suboptimal and inappropriate use of antifungal drugs, as well as the agricultural application of analogues of these drugs. 8 Although fungi are also implicated in a variety of infections in sub-Saharan Africa, a greater attention has been given to bacteria, with very little interest shown in fungal species. 9 This is largely attributed to inadequate awareness and poor access to essential diagnostics and antifungal drugs. 10 Consequently, these historical challenges have led to a decreased focus on efforts that ensure early detection and optimal management of fungal infections in the region. The continuous increase in populations in sub-Saharan Africa that are at risk for fungal infections 11 and the growing global threat of multidrug-resistant fungal strains 12 raise the need for an appreciation of the region’s perspective on antifungal usage and resistance. This review, therefore, aimed to summarise the prevalence of fungal infections, the use of antifungal agents, and antifungal resistance, in the context of sub-Saharan Africa, focusing on the major fungal species associated with IFIs.
Methods
A detailed search was conducted in multiple databases, including PubMed, Scopus, ScienceDirect, Google Scholar and African Journal Online, for each of the 48 countries in the sub-Saharan Africa, using the following keywords: (‘fungal infections’ OR ‘tinea capitis’ OR ‘cryptococcosis’ OR ‘pneumocystosis’ OR ‘disseminated histoplasmosis’ OR ‘chronic pulmonary aspergillosis’ OR ‘candidiasis’ OR ‘candidaemia’ OR ‘mucormycosis’ OR ‘fungal keratitis’) AND (‘antifungal agents’) AND (‘antifungal resistance’) AND (‘Angola’ OR ‘Benin’ OR ‘Botswana’ OR ‘Burkina Faso’ OR ‘Burundi’ OR ‘Cabo Verde’ OR ‘Cameroon’ OR ‘Central African Republic’ OR ‘Chad’ OR ‘Comoros’ OR ‘Democratic Republic of the Congo’ OR ‘Republic of Congo’ OR ‘Cote d’Ivoire’ OR ‘Djibouti’ OR ‘Equatorial Guinea’ ‘Eritrea’ OR ‘Eswatini’ OR ‘Ethiopia’ OR ‘Gabon’ OR ‘Gambia’ OR ‘Ghana’ OR ‘Guinea’ OR ‘Guinea-Bissau’ OR ‘Kenya’ OR ‘Lesotho’ OR ‘Liberia’ OR ‘Madagascar’ OR ‘Malawi’ OR ‘Mali’ OR ‘Mauritania’ OR ‘Mauritius’ OR ‘Mozambique’ OR ‘Namibia’ OR ‘Niger’ OR ‘Nigeria’ ‘Rwanda’ OR ‘Sao Tome and Principe’ OR ‘Senegal’ OR ‘Seychelles’ OR ‘Sierra Leone’ OR ‘Somalia’ OR ‘South Africa’ OR ‘South Sudan’ OR ‘Sudan’ OR ‘Tanzania’ OR ‘Togo’ OR ‘Uganda’ OR ‘Zambia’ OR ‘Zimbabwe’). The search was limited to articles published in English up to 2023, focusing on the most recent data on IFIs in each country. Apart from epidemiological data on IFIs, articles presenting the treatment of fungal infections and the mechanism of resistance exhibited by common pathogenic fungal species were included in this review. The references of the articles were also screened for relevant studies.
The burden of fungal infections in sub-Saharan Africa
In many sub-Saharan African countries, surveillance and available data on fungal disease burden are limited, contributing to challenges in determining the precise prevalence of IFIs. Despite these challenges, individual countries have reported high burdens of severe or serious fungal infections. In Ghana, for example, approximately 4% of the population, equivalent to 1,147,228 individuals, is affected by severe fungal infections, with 35,000 experiencing severe morbidity and mortality related to these infections. 13 This prevalence is higher than the reported 3% in Tanzania 14 but lower than the projected 11.8% in Nigeria. 15 In the Democratic Republic of the Congo (DRC), it is estimated that approximately 5,177,000 people (5.4% of the general population) suffer from serious fungal infections each year. 16 Elsewhere in Senegal, approximately 12.5% of the population (approximately 1,743,507 individuals) is affected by fungal infections. 17 In Sierra Leone, however, 4.92% of the population is affected by serious fungal infections, and a prevalence of 2.9% has been observed among people living with HIV. 18 Moreover, research conducted in Zimbabwe indicates that approximately 14.9% of the inhabitants experiences fungal infections annually, with tinea capitis accounting for 80% of these cases. 19 A summary of the prevalence of fungal infections in sub-Saharan Africa is provided in Table 1, and the burden of the major fungal infections in the subregion is detailed in Table 2.
Table 1.
Summary of the prevalence of fungal infections in sub-Saharan Africa.
| Country | Estimated prevalence (%) | Population affected | Year of study | Reference |
|---|---|---|---|---|
| Burkina Faso | 7.51 | 1,400,000 | 2018 | Bamba et al. 20 |
| Cameroon | – | 1,236,332 | 2018 | Mandengue et al. 21 |
| Côte d’Ivoire | 7.25 | 1,800,000 | 2021 | Koffi et al. 22 |
| Republic of the Congo | 5.6 | 293,918 | 2020 | Amona et al. 23 |
| Democratic Republic of the Congo | 5.4 | 5,177,000 | 2021 | Kamwiziku et al. 16 |
| Ethiopia | 8 | – | 2019 | Tufa et al. 24 |
| Ghana | 4 | 1,147,228 | 2019 | Ocansey et al. 13 |
| Kenya | 11.57 | – | 2023 | Ratemo et al. 25 |
| Malawi | 7.54 | 1,338,523 | 2018 | Kalua et al. 26 |
| Mali | 12.8 | 2,729,670 | 2023 | Doumbo et al. 27 |
| Mozambique | 6.9 | 1,834,947 | 2018 | Sacarlal et al. 28 |
| Namibia | 5 | 112,870 | 2019 | Dunaiski et al. 29 |
| Nigeria | 11.8 | – | 2014 | Oladele et al. 15 |
| Senegal | 12.5 | 1,743,507 | 2015 | Badiane et al. 17 |
| Sierra Leone | 4.92 | – | 2021 | Lakoh et al. 18 |
| Sudan | 10 | 5,000,000 | 2023 | Ahmed et al. 30 |
| Tanzania | 3 | – | 2015 | Faini et al. 14 |
| Togo | 5.29 | 7,265,286 | 2021 | Dorkenoo et al. 31 |
| Zimbabwe | 14.9 | – | 2021 | Pfavayi et al. 19 |
| South Africa | 5.7 | 3,220,014 | 2019 | Schwartz et al. 32 |
Table 2.
Burden of major fungal infections in sub-Saharan Africa.
| Country | Tinea capitis (in children) (cases per year) | Cryptococcosis (cases per year) | Pneumocystosis (cases per year) | Disseminated histoplasmosis (cases per year) | Chronic pulmonary aspergillosis (cases per year) | Vulvovaginal candidiasis (cases per year) | Candidaemia (cases per year) | Mucormycosis (cases per year) | Fungal keratitis (cases per year) | References |
|---|---|---|---|---|---|---|---|---|---|---|
| Burkina Faso | 1,132,781 | 459 | 1013 | – | 1120 | – | 906 | – | – | Bamba et al. 20 |
| Cameroon | 721,000 | 6720 | 9000 | 1800 | 1265 | 350,000 | – | 5 | – | Mandengue et al. 21 |
| Democratic Republic of the Congo | 3,551,900 | 9265 | 2800 | – | 54,700 | 1,202,640 | – | – | – | Kamwiziku et al.
16
Zono et al. 33 |
| Côte d’Ivoire | – | 4590 | 2640 | 513 | 6568 | 421,936 | – | – | 3350 | Koffi et al. 22 |
| Ethiopia | 7,051,700 | 9900 | 11,500 | 1 | 15,200 | 1,469,100 | 5300 | – | – | Tufa et al. 24 |
| Ghana | 598,840 | 6275 | 12,610 | 724 | 12,620 | 442,621 | 1,446 | 58 | 810 | Ocansey et al. 13 |
| Kenya | 4,905,249 | 8975 | 25,429 | 2,244 | 100,570 | 915,795 | 2,804 | 109 | 7929 | Ratemo et al. 25 |
| Malawi | 670,900 | 8200 | 10,800 | – | 2911 | 326,960 | – | 30 | 1825 | Kalua et al. 26 |
| Mali | 2,300,000 | 611 | 1393 | 180 | 7290 | 272,460 | >1060 | 43 | 2820 | Doumbo et al. 27 |
| Mozambique | 1,181,686 | 18,640 | 33,380 | 153 | 18,475 | 348,179 | 1321 | 53 | – | Sacarlal et al. 28 |
| Namibia | 53,784 | 543 | 836 | – | 453 | 37,390 | 125 | 5 | – | Dunaiski et al. 29 |
| Nigeria | 15,581,400 | 57,894 | 74,594 | 120,753 | 1,521,520 | 9284 | 300 | Oladele et al. 15 | ||
| Senegal | 1,523,700 | 366 | 1149 | – | 2700 | 191,228 | – | 26 | – | Badiane et al. 17 |
| Sierra Leone | 266,450 | 302 | 643 | 0 | 1275 | 85,440 | 382 | 15 | 1017 | Lakoh et al. 18 |
| Sudan | 4,127,760 | 462 | 571 | – | 14,950 | 631,300 | 2340 | 94 | 6550 | Ahmed et al. 30 |
| Tanzania | 420,000 | 6394 | 9600 | 135 | 10,437 | 759,500 | 2181 | – | – | Faini et al. 14 |
| Togo | 232,271 | 1,342 | 1650 | 330 | 191 | 108,979 | – | – | 981 | Dorkenoo et al. 31 |
| Zimbabwe | 1,806,700 | 6086 | 9429 | 57 | 2448 | 203,585 | 743 | – | 2080 | Pfavayi et al. 19 |
| Republic of the Congo | 178,400 | 560 | 830 | 120 | 3420 | 85,440 | – | – | – | Amona et al. 23 |
| South Africa | 1,003,490 | 8357 | 4452 | 60 | 89,416 | 1,002,499 | 5421 | 113 | – | Schwartz et al. 32 |
Tinea capitis
Tinea capitis, a fungal infection that affects the scalp, skin, and hair, is the most prevalent type of dermatophyte infection and constitutes 52% of all fungal infections. 34 African children are particularly susceptible, with an estimated 138 million cases recorded among them. 35 In Ghana, tinea capitis is the predominant fungal disease, accounting for 44% of fungal infections. 13 The prevalence among Ghanaian school children ranges from 8.4% to 8.7%, which is lower than the prevalence reported in other sub-Saharan African countries. 36 Another study revealed that 598,840 Ghanaian children, or 2070 cases per 100,000 individuals, have tinea capitis. 13 In Nigeria, at least 20% of school-aged children were reported to have tinea capitis. 37 Zimbabwe has approximately 1,806,700 affected children, 19 while Senegal has a 25% prevalence among children, corresponding to approximately 1.5 million individuals. 17 In Malawi, 670,900 cases of tinea capitis were reported among school children in 2018. 26 Similarly, in Rwanda, approximately 21% or 34,995 school children were identified to have tinea capitis. 36 The DRC detected approximately 3,551,900 cases of tinea capitis out of the 5,177,000 cases of fungal infections. 16 Approximately 178,400 school children in the Republic of the Congo (RC) suffer from tinea capitis of the 293,918 cases of fungal infections. 23
Cryptococcosis
Cryptococcosis, caused by Cryptococcus neoformans and Cryptococcus gattii complexes, is a leading cause of meningitis in HIV-infected patients in sub-Saharan Africa. 38 On average, 152,100 HIV patients worldwide were estimated to contract cryptococcal meningitis in 2020, leading to 112,000 deaths, which constituted 19% of all AIDS-related deaths. 7 In sub-Saharan Africa, 61,000–101,000 people living with HIV were diagnosed with cryptococcal meningitis in 2020, resulting in 71,000 deaths. 7 The prevalence of cryptococcosis among HIV patients is notably high in sub-Saharan Africa. 7 For instance, one study estimated that 21.7 per 100,000 cases of cryptococcal meningitis occurred among AIDS patients in Ghana. 13 Another study, conducted in Zimbabwe, projected an annual prevalence of 6086 cases of cryptococcal meningitis among HIV/AIDS patients, with an estimated rate of 41/100,000 people per year. 19 In Senegal, 366 of 59,000 HIV-positive individuals developed cryptococcal meningitis. 17 In addition, the DRC has an annual prevalence of 9265 cases of cryptococcal meningitis in adults. 16
Candidiasis
Candidiasis, whose spectrum of manifestations includes oral, vaginal and vulvovaginal candidiasis (VVC), as well as systemic candidiasis (such as candidaemia), is a severe and potentially fatal disease that poses a significant threat to critically ill individuals, with mortality rates ranging from 29% to 76%. 39 In sub-Saharan Africa, a study by Mushi et al. 40 found that 33.5% of people living with HIV had oral candidiasis, which was caused by various Candida species, including both albicans and non-albicans, such as Candida glabrata, Candida krusei, Candida tropicalis, Candida parapsilosis, and others.
Candidaemia is a common bloodstream infection characterised by the presence of Candida species in the blood. 41 The most prevalent Candida species associated with candidaemia include Candida albicans, C. krusei, C. parapsilosis, C. tropicalis, C. glabrata, and the usually multidrug-resistant, biofilm-forming species Candida auris. 42 In a single-site surveillance of Candida bloodstream infections conducted in Kenya over a six-year period, C. auris comprised 38% of all isolates, closely followed by C. albicans, which accounted for 25% of the isolates. 43 By contrast, a retrospective study conducted in South Africa between 2009 and 2010 indicated that C. albicans was the predominant cause of candidaemia, accounting for 23.8% of cases. 44 Similarly, retrospective studies conducted in hospitals in both South Africa and Nigeria found C. albicans to be the primary cause of candidaemia, accounting for 73% and 77% of cases, respectively. 45
Regarding vulvovaginal candidiasis, one study reported its diagnosis among 21% of patients receiving care at a gynaecological clinic in the Middle Belt of Ghana. 46 VVC has a pooled prevalence of 33%, affecting both pregnant and non-pregnant women in sub-Saharan Africa. 47 The projected prevalence of recurrent vulvovaginal candidiasis (RVVC) among adult women in Ghana’s overall healthy population is estimated to be approximately 442,621 cases, occurring annually at a rate of 1530 females per 100,000 persons. 13 Elsewhere in Namibia, RVVC affects 37,390 adult women (33.1% of 112,870 cases of fungal infections), 29 while in Zimbabwe, its burden is 2739 cases per 100,000 people. 19 Moreover, it is predicted that each year, 1,000,000 South African women will be affected by RVVC out of every 56,521,947 persons diagnosed with fungal infections. 32 Furthermore, the reported prevalence of RVVC in the DRC is 1,202,640 of the total 5,177,000 fungal infections, 16 while in the RC, RVVC is estimated to account for approximately 85,440 of the 293,918 cases of fungal infections, occuring among adult women. 23
Oral candidiasis, the most common opportunistic fungal infection among immunocompromised individuals, affects 7.6%–75.0% of HIV patients in sub-Saharan Africa. 40 Although C. albicans is the predominant cause, non-albicans Candida species account for more than 30% of the cases. 40 Similarly, oesophageal candidiasis is considered an AIDS-defining condition and affects approximately 12% of HIV/AIDS patients in the region. 48 In Senegal, 1946 of 59,000 HIV-positive individuals were determined to develop oesophageal candidiasis. 17 Additionally, a Zimbabwean study among persons living with HIV reported that oral candidiasis affects 77,143 patients, while oesophageal candidiasis affects 63,571 patients. 19 In the DRC, oral and oesophageal candidiasis were observed in 50,470 and 28,800 HIV-infected patients, respectively, 16 whereas in Ghana, they were reported to account for 27,100 of the 1,147,228 cases of fungal infections. 13
Invasive aspergillosis
Invasive aspergillosis (IA) is a potentially fatal infection that primarily affects individuals with a weakened immune system. 49 It is caused by Aspergillus spp., which are ubiquitous and release airborne conidia into the environment. Inhaling these conidia can lead to various clinical manifestations, including allergies, chronic pulmonary aspergillosis (CPA) and IA. 50
The prevalence of IA in Africa varies across different regions, with rates ranging from 0.05 to 10.9 cases per 100,000 people. 51 In Ghana, there were 12,620 estimated cases of CPA and 1254 cases of IA out of the 1,147,228 cases of fungal infections. 13 Southern Africa has a prevalence of 4.8–16 per 100,000 people, Western Africa has 0.3–6.25 per 100,000 people, Northern Africa has 7.1–10.7 per 100,000 people and Central Africa has 3.2–6.9 per 100,000 people. 51 Among AIDS patients, the prevalence of invasive aspergillosis was found to be 380 cases per year in the DRC. 16 Histological evaluations indicated a prevalence of 2.6% in South Africa and 1.15% in Uganda. 52 An estimated 2448 cases of IA occur annually in Zimbabwe, with a rate of 16 cases per 100,000 people. 19 The primary Aspergillus species identified are Aspergillus flavus and Aspergillus niger, which are more predominant in sub-Saharan Africa, given that the tropical climate of the region is ideal for their survival. 53 To a lesser extent, other species, such as Aspergillus fumigatus, Aspergillus ochraceus, Aspergillus tubingensis, and Aspergillus westerdijkiae have also been identified in the region. 54
Pneumocystis jirovecii pneumonia
Pneumocystis jirovecii, a member of the Ascomycota fungal group, is known for causing severe pneumonia in individuals with a weakened immune system. 55 A comprehensive analysis of studies conducted in hospitals across 18 sub-Saharan African countries revealed that the overall occurrence of Pneumocystis jirovecii pneumonia (PJP) among persons living with HIV was 15.4%, with high prevalence among individuals exhibiting respiratory symptoms (18.8%), hospitalised patients (22.4%) and hospitalised patients with respiratory symptoms (24%). 56 In Ghana, it was estimated that there were 12,610 cases of Pneumocystis jirovecii among individuals living with HIV/AIDS. 13 Screening of randomly selected patients in a facility in Cameroon revealed that 82% of these patients had significant antibody titres and were, therefore, exposed to Pneumocystis jirovecii. 57 Zimbabwe reported an annual prevalence of 63 cases per 100,000 people for PJP among HIV/AIDS patients. 19 In Senegal, it was predicted that out of 59,000 HIV-positive individuals, 1149 would develop PJP. 17 Moreover, South Africa reported 4452 cases of PJP out of 56,521,947 cases of fungal infections, 32 while the DRC had an estimated annual prevalence of 2800 cases of PJP among adults out of the 5,177,000 cases of fungal infection. 16
Histoplasmosis
Histoplasmosis is a fungal infection that primarily affects individuals living in endemic regions. 58 It is particularly problematic for people with advanced HIV disease, as it can cause opportunistic infections. 58 Infection occurs when individuals inhale spores of Histoplasma capsulatum, which has two variants: Histoplasma capsulatum var. capsulatum (HCC) and Histoplasma capsulatum var. duboisii (HCD). 59
Histoplasmosis has significantly expanded in Africa alongside the HIV epidemic but remains largely underestimated. Limited information and epidemiological data are available for histoplasmosis in Ghana; however, a recent study reported a prevalence of 4.7% (5 out of 107) among HIV patients. 60 A comprehensive analysis of histoplasmosis in Africa revealed a total of 470 documented cases spanning a period of six decades (1952–2017). Among these cases, 38% (178) was reported in HIV-infected individuals. 61 A study also reported that West Africa had the highest number of histoplasmosis cases in Africa, followed by Southern Africa. 61 Research conducted in Cameroon revealed that 13% of HIV-positive individuals who presented with persistent fever, cough, and skin lesions were diagnosed with histoplasmosis. 62 Specific studies reported 17 of the 7,265,286 cases of fungal infections in Lomé, Togo, between 2002 and 2016 63 and 36 patients infected with HCD out of the 5,177,000 fungal infection cases in Kimpese, DRC. 64
Sporotrichosis
Sporotrichosis, caused by the Sporothrix schenckii complex, is a chronic fungal infection primarily transmitted through traumatic inoculation, such as thorn pricks or scratches, among individuals involved in occupations or recreational activities, such as gardening, farming, fishing, hunting and, horticulture. 65 Although sporotrichosis in Africa has received limited attention, it has been reported in some countries, particularly Madagascar. 66 In South Africa, sporotrichosis cases have primarily been documented among mine workers, with an estimated number of undocumented cases surpassing 3300. 67 The infection has been observed in various regions in the southern part of Africa, such as Pretoria, Botswana, Mpumalanga, and the Northwest Province, particularly among employees in gold mines. 9 However, due to the limited availability of medical mycology laboratories in sub-Saharan Africa, the true extent of sporotrichosis spread across the continent remains poorly documented. 9
Chromoblastomycosis
Chromoblastomycosis (CBM) is a fungal infection caused by melanised fungi, specifically Fonsecaea pedrosoi and Cladophialophora carrionii. 68 The extent of the impact of CBM in Africa is not accurately understood due to a lack of thorough epidemiological studies. However, it is noteworthy that Madagascar has the highest number of reported CBM cases among African countries. 68 In one study in the country, chromoblastomycosis was diagnosed in 50 out of 148 patients who had chronic subcutaneous lesions suggestive of dermatomycosis. 66 A recent study examining the global burden of CBM spanning from 1914 to 2020 revealed that Africa ranks as the second highest region in terms of documented cases, following South America. Across 22 countries in Africa, a total of 1875 out of 7740 CBM cases were reported. 69
Other fungal infections
Emergomycosis, previously known as Emmonsia, is a fungal infection caused by a fungus of the genus Emergomyces that can spread throughout the body. There are five recognised species of Emergomyces worldwide, namely, Emergomyces pasteurianus, Emergomyces africanus, Emergomyces canadensis, Emergomyces orientalis and Emergomyces europaeus. Of these, Es. pasteurianus and Es. africanus are notably prevalent in Africa, with numerous cases reported in Lesotho, Uganda, and South Africa. 70 Mucormycosis and fungal keratitis are two other significant diseases affecting the African population. Approximately 58 out of 100,000 individuals in Ghana are affected by mucormycosis, while fungal keratitis affects approximately 810 people out of 100,000. 13 However, in Namibia, only five cases of mucormycosis have been reported. 29
Mode of action of the different antifungal agents
In ancient times, plant extracts were utilised to treat skin and nail fungal infections, contributing to the development of antifungal agents as drugs for combating fungal infections. 71 The earliest available pharmacologic agents for the treatment of fungal infections included undecylenic acid, phenolic dye and weak acid. 71 The modern era of antifungal therapy began in the mid-20th century, 71 following the discovery of griseofulvin in 1939, the first synthetic antifungal drug, which was approved for clinical use in early 1960 and became available for oral use that year. 71 Griseofulvin belongs to the antifungal class of polyenes, was isolated from the fungus Penicillium griseofulvum, 72 and remains effective against dermatophyte infections. 73 Besides being available for oral use, antifungal medications, are available as topical and intravenous agents as well, and are used to treat different types of fungal infections; however, all these routes of administration have their respective limitations and side effects. 74
Numerous antifungals have since been developed after the discovery of the first antifungal agent in the 20th century. 75 These are grouped into classes including azoles, echinocandins, polyenes, and flucytosine, with each class exhibiting different mechanisms of action. 76 Therefore, appropriate combination therapy may ultimately yield a positive outcome, even for patients who have drug-resistant fungal infections. However, not all these antifungals are readily available in sub-Saharan African countries. In Ghana, for instance, the antifungals that are easily accessible are 5-flucytosine 77 and fluconazole, 78 while amphotericin B is available only in Benin, Zambia, South Africa, and Ethiopia. 77
Azoles
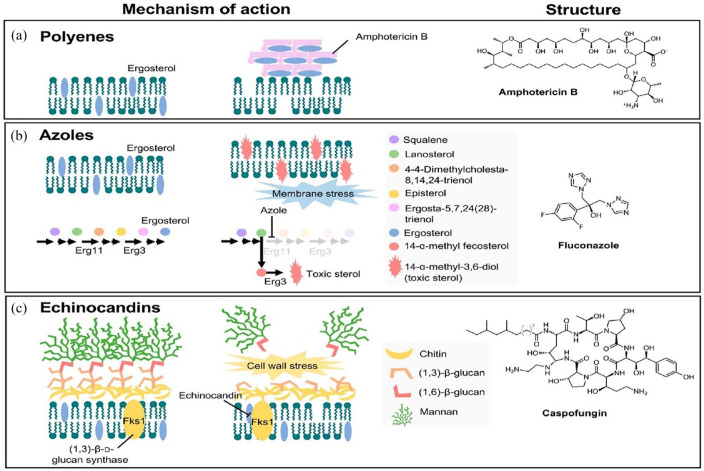
Azoles are heterocyclic compounds within whose chemical structures are imidazole or triazole rings, which are bound to the rest of the structure via nitrogen-carbon bonds. 79 This structure confers a weak basic character to azole antifungals. 79 It is believed that the nitrogen atoms N-3 and N-4 in the imidazoles and triazoles, respectively, bind to the heme portion of cytochrome P-450, inhibiting the demethylation of lanosterol. 80 The balance of the lipophilicity/hydrophilicity of azoles is determined by the number of aromatic rings and halogen substituents, with the latter imparting a hydrophilic character and the former imparting a lipophilic character. 80 These azoles exhibit antifungal activity by blocking the synthesis of ergosterol, a key constituent of the fungal cell membrane, which leads to disruptions in permeability, membrane stability, and the function of membrane-bound enzymes. 81 The blockage of ergosterol synthesis occurs by inhibiting lanosterol 14-α-demethylase encoded by ERG11 in C. albicans and Cryptococcus neoformans, Cyp51A/Cyp51B in moulds, 82 and cyp51B and cyp51A in A. fumigatus, 81 leading to the build-up of dangerous sterol intermediates, such as the product of Erg3, 14-methyl-3,6-diol. 83 When incorporated into the fungal membrane, this creates membrane stress 82 and ultimately inhibits proliferation. 84
Azoles are the most frequently and widely used antifungals in clinical practice for the treatment of fungal infections. 81 They are fungistatic and are often administered long-term. 85 The currently available azoles that are clinically approved are isavuconazole, voriconazole, itraconazole, fluconazole and posaconazole, and they primarily possess fungistatic activity against yeasts, such as Cryptococcus and Candida spp. 84 Compared to other antifungals, many azole drugs have excellent oral bioavailability and are offered in both intravenous and oral formulations. 79 However, this does not make azoles harmless consumable drugs, as they have a high potential to interact with other drugs. In addition, they inhibit mammalian cytochrome P450 enzymes, which are responsible for drug metabolism. 86 New azoles that have a higher specificity for fungal enzymes (VT-1161, VT-1129 and VT-1598) are currently being developed to address this restriction. 81
Considering that fluconazole (an azole) has been recommended by previous guidelines as the initial treatment for Candida infections, 87 it is worth noting that fungal resistance to azoles (including fluconazole) has risen over time following the continuous exposure of the drugs to fungi. For instance, C. parapsilosis resistance to fluconazole has increased over time. 88
A recent study demonstrated increased efficacy of combination therapy involving azoles and a target of rapamycin (TOR) inhibitor (AZD8055) against azole-resistant A. fumigatus and multidrug-resistant C. auris. 89 The authors further reported that both TORC1 and TORC2 are inhibited by the binding of AZD8055 to the adenosine triphosphate (ATP)-binding cleft of TOR kinase. Investigations have shown that in the pathogenesis of fungal infections, the TOR signalling pathway plays important roles. 90 The expression of genes associated with cellular adhesion, aggregation and morphogenesis, which are attributed to the virulence of C. albicans, is regulated by TOR 91 and the DNA damage response “machinery”. 92
Despite the promise of this class of antifungals in contemporary fungal treatment regimens, its overuse, particularly in sub-Saharan Africa, has driven the emergence of resistance. Additionally, the widespread use of azole fungicides (14a-demethylase inhibitors) in agricultural fields in Africa could be a contributing factor to the selection of azole-resistant isolates. 93 For example, multiazole-resistant A. fumigatus strains have been isolated from the environment in Tanzania, 94 and azole-resistant C. parapsilosis strains causing bloodstream infection have been isolated in South Africa. 95 Hamdy et al. considered numerous novel azoles, such as S-alkylated azole derivatives (UoST5, 7, 8, and 11), for further development due to their low MIC50 and safety in mammalian cells. 96
Echinocandins
Echinocandins have a cyclic hexapeptide as their core and are acylated by having a different fatty acid attached to the dihydroxyornithine amino group 75 ; this lipid residue is necessary for bioactivity because it anchors the drug to the cell membrane. 97 Filamentous fungi naturally produce echinocandins, such as echinocandin B. 75 Semisynthetic cyclic lipopeptides with antifungal activity include rezafungin, anidulafungin, micafungin, and caspofungin. 75
This class of antifungals has lipopeptide molecules that execute their antifungal activity by noncompetitively inhibiting β-1,3-D-glucan synthase in the fungal cell wall, 98 which oversees the production of β-1,3-D-glucan, a key structural element of fungal cell walls. 99 Caspofungin, micafungin, and anidulafungin are some of the accepted antifungal agents, and they are fungicidal against most Candida species, including those resistant to polyenes and azoles. 88 Rezafungin (CD101), a novel echinocandin with a prolonged half-life, is currently being evaluated in Phase 3 trials. 100 A study conducted by Song et al. 101 revealed that isolates of C. krusei, C. albicans, C. tropicalis, and C. parapsilosis resistant to one or more echinocandins are notably rare. However, resistance to anidulafungin, caspofungin, and micafungin was most noticeable among C. glabrata isolates. While the echinocandin class is fungicidal, 102 it is fungistatic to some pathogenic filamentous fungi, including Aspergillus spp. 101
Unlike polyenes, which are toxic owing to their slight affinity for human cholesterol, the therapeutic potential of echinocandins is outstanding, and they are unlikely to cause serious drug interactions or renal or hepatic toxicity. 103 In most cases, when caspofungin and posaconazole were combined, they worked well in a synergistic manner. 104 Interestingly, synergistic effects were also detected in azole-resistant isolates of A. fumigatus. 104
The wide availability of the accepted antifungal agents in this class in sub-Saharan Africa has not been determined, but studies in South Africa, 105 Ethiopia, 106 and Cameroon 107 have indicated the availability of caspofungin and anidulafungin.
Polyenes
The oldest class of antifungal medications used to treat systemic fungal infections are polyenes, which are organic, amphipathic molecules. The most widely used polyene, amphotericin B, was first used in medicine in the 1950s and has potent activity against a variety of clinically important fungal species, including several species of Aspergillus, Cryptococcus, and Candida. 108 The distinguishable feature of polyenes that characterises and differentiates them from other antifungal classes is the multiple conjugated double bonds of the hydroxylated chromophore. 80 Depending on the quantity of conjugated double bonds they contain, amphotericin B and natamycin are categorised as heptaene and tetraene polyene antifungal drugs, respectively. 80 The all-trans conformation of the conjugated double-bond lactone chromophore is necessary for the stability and antifungal activity of natamycin and amphotericin B. 109 The aforementioned polyene antifungals are lipophilic because of the conjugated double bonds and hydrophilic because of the hydroxylation of the chromophore. 80 Hydrophilicity allows the drug to dissolve in water, while lipophilicity impacts drug uptake, metabolism, and drug interactions with the target protein. 80
Traditionally, polyenes are believed to directly bind to ergosterol to form drug-lipid complexes that intercalate into the membranes of fungal cells, resulting in leakage of intracellular components and eventually cell death. 110 Recent structural and biophysical studies, however, cast doubt on this model, showing that amphotericin B forms extra membranous aggregates that act as a fungicidal ‘sterol sponge’ to draw ergosterol from fungal cell membranes, as shown in Figure 1. 111
Figure 1.
Illustration showing the mechanism of action of the three classes of antifungal agents.
Source: Figure adopted from Lee et al. 83
Polyenes have a wide range of fungicidal effects. The target of amphotericin B is ergosterol, which is a sterol lipid; unlike proteins, it is not encoded by genes; hence, resistance to polyenes is relatively infrequent. 112 Its resistance may arise when there is ergosterol replacement or depletion that results in alteration of the cell membrane composition. 84 According to a recent study, clinicians found that a single high dose of amphotericin B is as effective as a one-week course of daily amphotericin B. 60 The administration of polyenes, such as amphotericin B, is disfavoured due to nephrotoxicity, 113 dose-dependent toxic effects towards the host and poor oral bioavailability. 110
Flucytosine
Flucytosines are pyrimidine analogues that are antimycotic, lack intrinsic antifungal capacity and have limited clinical uses. 81 After intake, flucytosine is transformed into the toxic substance 5-fluorouracil (via the activity of cytosine deaminase, which is absent in humans), which inhibits RNA and DNA synthesis, as well as various metabolic pathways. 81 Flucytosines are rarely used alone owing to the rapid emergence of drug resistance during monotherapy, and they are combined with amphotericin B when used to treat cryptococcal meningitis and other fungal infections. 81 Flucytosine monotherapy is used only for treating chromoblastomycosis and vaginal and lower urinary tract candidiasis. 114 Unlike some other antifungals, flucytosines are currently not widely available in sub-Saharan Africa. 115
Tolerance and resistance mechanism of different antifungal classes
Numerous environmental and clinical fungal isolates can resist or tolerate antifungal drugs. The ability of fungi to grow at antifungal drug concentrations that stop growth or kill most fungal isolates is known as antifungal resistance. 116 On the other hand, tolerance refers to the capacity of drug-sensitive cells to proliferate at drug concentrations greater than the minimum inhibitory concentration (MIC), and it involves a variety of general stress responses, spore formation and/or epigenetic pathways. 116 Some fungal species exhibit intrinsic resistance, resulting from the inability of drugs to effectively bind to their target or the removal of the drug by efflux pumps, as observed in C. krusei, Aspergillus spp., and intrinsic (100.0%) fluconazole resistance in C. krusei. 117 In contrast to intrinsic resistance, some fungal species exhibit acquired resistance – acquisition of resistance mechanisms that give them an upper hand in thriving and proliferating in higher antifungal drug concentrations than their corresponding wild types. 85 This resistance can be acquired via evolution or vertical or horizontal gene transfer from the resistant mutant to the wild type. 118
As fungi are eukaryotes, they are usually multicellular and have multinucleate genomic organisation, which increases the potential for genetic changes promoting adaptations and the development of resistance. Some genetic alterations that confer antifungal resistance include hypermutator fungal lineages in C. glabrata and Cryptococcus spp., gene duplications, transposon insertions (~10−3 to 10−4 per cell per generation) and point mutations (~10−6 to 10−8 per cell per generation). 85
The environmental resistance of fungi to antiungal agents can emerge following the prior exposure of human pathogenic fungi to fungicides in nature. 119 Resistance to all major classes of fungicides, including anilinopyrimidines, benzimidazoles, strobilurins, sterol demethylation inhibitors, including azoles and succinate dehydrogenase inhibitors, is evolving due to environmental pressure from fungicides. 120 Furthermore, some species exhibit a nongenetic route of conferring resistance or tolerance via a physiologically sessile state of multimorphic cells known as biofilm formation. 85 The extracellular matrix produced by the collective fungal cells in biofilms serves as a drug sink, lowering the effective drug concentration for the cells in the biofilm. 121 Alterations at the molecular level and in the environment result in many adaptive mechanisms of antifungal resistance and tolerance. This includes overexpression or drug target alteration, activation of stress responses and upregulation of multidrug transporters. 122 Due to these multiple ways of obtaining resistance, resistance to azoles and echinocandins is much more common, 82 but resistance to polyenes is still incredibly rare. 123
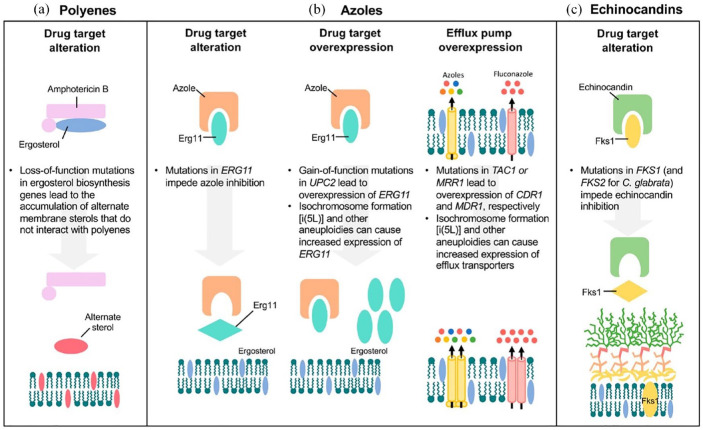
Resistance to polyenes
Fungal resistance to polyenes is mediated by alterations in enzymes, such as ERG2 and ERG11, 124 which deplete ergosterol from the membrane or reduce drug-binding affinity, 83 as illustrated in Figure 2.
Figure 2.
A picture showing the mechanism of antifungal resistance.
Source: Image adapted from Lee et al. 83
Furthermore, a study by Vandeputte et al. 125 on clinical isolates of C. glabrata that were less susceptible to polyenes revealed a nonsense mutation in the ERG6 gene, which encodes an enzyme involved in the final stages of the pathway for ergosterol biosynthesis and has been attributed to be the cause of this reduction in susceptibility. Additionally, the ERG6 mutation caused the overexpression of almost all the genes that catalyse the final stages of the ergosterol biosynthesis pathway, supporting the idea that this metabolic pathway is blocked in the wild type. 125
Resistance to echinocandins
Fungal resistance to the echinocandin class is principally mediated by mutations in the FKS genes, as shown in Figure 2. These mutations increase the MIC, resulting in cross-resistance to various echinocandins and significantly reducing the IC50 of the glucan synthase enzyme. 126 Pfaller et al. 88 confirmed variable cross-resistance between voriconazole and fluconazole when examining the common species of Candida that cause invasive candidiasis.
Although prolonged exposure to two different classes of agents occurs when the multidrug resistance (MDR) phenotype is most frequently found, 88 recent studies have shown that under stressful circumstances, mutations in the MSH2 DNA mismatch repair gene may result in a hypermutable state and accelerate the development of resistance to echinocandin, amphotericin B, and fluconazole. 127 These findings may provide a potential explanation for why some studies reported echinocandin resistance in Candida strains exposed to fluconazole, even though the drugs have different targets and resistance mechanisms. 128
A study conducted by Pfaller et al. 88 revealed that drug-resistant strains of C. albicans, which are specifically resistant to caspofungin and micafungin, exhibited mutations in the FKS1 HS1 gene. These mutations included F641I, F641S, S654P, S629P and S645P (each present in four isolates). Additionally, all C. glabrata isolates analysed in the study showed modifications in the FKS HS genes. The most prevalent alterations observed were F659S/V/Y in FKS2 HS1 and S663P in FKS2 HS1. 88 Another study on C. albicans revealed that mutations occurring at positions S641 and S645 are not only the most frequently observed but also result in the most pronounced resistance phenotypes. 98 According to earlier reports by Pfaller et al., 88 isolated C. glabrata harbouring the S663F mutation did not respond to high doses of anidulafungin in vivo but responded to high doses of micafungin or caspofungin under similar conditions. By contrast, isolates carrying the S629P mutation were insensitive to any of the three echinocandins, even at the highest dose. 129 Echinocandin therapy typically has positive results for patients whose C. glabrata infections are caused by strains with the I1379V and I634V mutations (susceptible to both caspofungin and anidulafungin). 130 Echinocandin-resistant strains displayed either FKS1 or FKS2 hotspot mutations. 127 Perlin et al. further reported that the main cause of clinical resistance to echinocandins is FKS1 or FKS2 hotspot mutations. 98
Resistance to azoles
Fungistatic azoles exert strong directional selection pressure on fungal pathogens, such as Cryptococcus and Candida pathogens, without causing their death, thereby facilitating the development of antifungal drug resistance. As a result, different Candida species exhibit varying levels of susceptibility to azoles. C. krusei is inherently resistant to the commonly used azole fluconazole, whereas resistance in C. glabrata is increasing, and C. albicans generally remains susceptible. 83
Numerous mechanisms of azole resistance have been identified; one of the most common involves the drug target gene ERG11/CYP51A/CYP51B being altered or overexpressed, with resistant isolates of Candida and Aspergillus frequently having amino acid substitutions in regions close to the enzyme’s heme-binding site. 131 It has also been reported that the transcription factors SRE1 and SRBA in C. neoformans and A. fumigatus, respectively, are involved in resistance to this antifungal agent. 132 Furthermore, azole resistance can be enabled following alterations to the other steps in the biosynthesis of ergosterol, such as loss of function of the Δ-5,6-desaturase enzyme ERG3. Cross-resistance to azoles and polyenes frequently results from the depletion of ergosterol and the accumulation of alternative sterols, all of which result from ERG3 mutations. 133
The overexpression of CDR1 and CDR2, which are transporters, is often associated with azole resistance, 82 as shown in Figure 2. Among these two transporters, CDR1 plays a more significant role in determining azole resistance. Removing CDR1 from a clinical isolate reduces azole resistance by fourfold to eightfold. Conversely, deleting CDR2 typically has a comparatively weaker impact. 134 SNQ2 is also involved in azole resistance. 135 C. albicans acquires azole resistance by altering the ergosterol biosynthesis pathway through loss of function mutations in ERG3. These mutations affect the 5,6-desaturase enzyme encoded by ERG3, preventing the cellular accumulation of 14-α-methyl-3,6-diol, a toxic sterol intermediate generated as a result of the inhibition of ERG11 by azoles. 136 Alternatively, fungal growth and replication in the presence of azoles are made possible and enhanced by the incorporation of 14-α-methyl-fecosterol into the fungal cell membrane. Another study by Spettel et al. 137 revealed that C. albicans resistance to azoles can be attributed to five missense mutations in ERG3 (A353T, G261E, S191P, T329S and A168V) and two additional nonsense mutations (Y190* and Y325*), leading to loss of function.
Resistance to flucytosine
Flucytosine enters fungal cells through cytosine permease, where it is converted into fluorouracil, which then interferes with pyrimidine salvage pathways, subsequently inhibiting DNA, RNA and protein synthesis. 138 Primarily, resistance to flucytosine arises from mutations in the FCY2 gene, which encodes a cytosine permease enzyme, leading to decreased uptake of the drug by the enzyme. 139 Fungal pathogens can also acquire resistance to flucytosine by suppressing the conversion of 5-fluorocytosine (5FC) to 5-fluorouracil (5FU) or by inhibiting the binding of 5-fluorouracil to uracil phosphoribosyl transferase. 138 The genes involved in this resistance mechanism are FCY1 and FUR1, which encode the enzymes cytosine deaminase and uracil phosphoribosyl transferase, respectively. 138 These resistance mechanisms have been identified in Candida spp., S. cerevisiae, Cryptococcus spp. and other fungal pathogens. 138 Kern et al. 140 reported that the R134S mutation in the FUR1 gene of S. cerevisiae confers resistance to 5FU. Furthermore, the G28D/S29L and A176G mutations in C. albicans as well as the A15D/G11D/W148R/G210D/L136R/T84L/I384F and G246S/I384F mutations in C. glabrata result in defective cytosine deaminase and cytosine permease enzymes. 141 Billmyre et al. 142 reported that the D306G/Y217C/1520delT/1182insC mutation in the UXS1 gene of C. deuterogattii leads to the accumulation of UDP glucuronic acid and the alteration of nucleotide metabolism, resulting in the inhibition of both 5FC and 5FU toxicity. Additionally, defective DNA mismatch repair pathways frequently contribute to acquired 5FC resistance in C. deuterogattii isolates. 142
Studies have proposed that each fungal pathogen may possess different mechanisms of resistance to 5-fluorocytosine other than the Fcy2-Fcy1-Fur1 pathway. 139 In a chemogenomic analysis by Costa et al., 143 it was discovered that 183 genes contribute to resistance to flucytosine by affecting various stages of fungal metabolic processes. Notably, within the domain of DNA metabolism, 13 genes were found to be associated with flucytosine resistance, comprising eight genes responsible for chromatin remodelling and five genes involved in DNA repair. Although flucytosine does not primarily target the cell membrane, eight lipid metabolism genes are required for 5-flucytosine tolerance. 143 These genes included the ergosterol biosynthetic genes ERG4 and ERG3, as well as six others that affect plasma membrane phospholipid composition, such as the transcription factor encoding gene MGA2, which has a role in regulating the desaturase-encoding genes OLE1, and OPI1, which is a negative regulator of phospholipid biosynthesis genes. 143 The authors also discovered that cell wall-encoding genes, specifically CWP2 and SED1, play a role in conferring resistance to flucytosine. Moreover, the deletion of CGFPS1 and CGFPS2 was found to increase the susceptibility of fungi to flucytosine, 143 suggesting that these genes play a major role in resistance because they are related to osmotic stress resistance. 144 The discovery of five genes encoding arginine metabolic enzymes that confer 5-flucytosine resistance within these processes suggested that arginine itself may be required for 5-flucytosine resistance. 143
Antifungal resistance cases reported in sub-Saharan Africa
Microbiological resistance occurs when antifungal medications are unable to effectively eliminate or hinder the growth of fungi under controlled laboratory conditions. 145 This resistance can either be inherent to fungi or acquired over time. 146 The emergence of resistance is a major factor contributing to treatment failures and the high mortality rates observed in systemic fungal infections. 147 The efficacy of available antifungal therapies is compromised due to the widespread presence of resistance, which is likely a result of the extensive and repeated use of these drugs. 148 Additionally, factors such as limited systemic usage due to toxicity concerns and the emergence of new strains of fungal infections further undermine the effectiveness of antifungal medications. 149 Notably, fungal pathogens belonging to Candida, Aspergillus, Cryptococcus, and Pneumocystis species demonstrate significant rates of resistance to antifungal agents. 146
Candida
The global prevalence of antifungal resistance in yeasts is on the rise, particularly due to the emergence of non-albicans Candida species. 128 Similar to bacteria, there is growing concern over multidrug-resistant yeasts, such as C. auris and C. glabrata. 150 A study conducted in Ghana revealed that C. albicans was the most frequently isolated species from clinical samples, with resistance levels ranging from 4.5% to 22.2%. 151 However, another study focusing on 267 HIV-infected individuals with oropharyngeal candidiasis in Ghana revealed a shift in the distribution of Candida species, with an increase in non-albicans species that often exhibit resistance to fluconazole. 152 Recent research conducted in Ghana demonstrated that Candida species showed the highest rates of resistance to fluconazole (48.1%), followed by voriconazole (37.0%) and nystatin (9.3%). 153 The significant resistance to fluconazole among Candida isolates may be attributed to its predominant use for the empirical treatment of vulvovaginal candidiasis. 153 As a result, current treatment guidelines recommend the use of alternative effective antifungals, such as amphotericin B and flucytosine. 154
Resistance to antifungal drugs has been observed in non-albicans Candida species, such as C. glabrata and C. krusei, in several African countries, including South Africa, 155 Cameroon, 107 Nigeria, 156 Ghana, 151 Tanzania, 157 and Ethiopia. 158 Azole resistance has increased in candidiasis patients, leading to increased mortality rates, 159 and echinocandins have been recommended as alternative treatments. However, coresistance to both echinocandins and azoles has been reported in clinical isolates of C. glabrata, 130 and cases of echinocandin-resistant C. glabrata infections have been documented in South Africa. 45 Fluconazole resistance in C. glabrata and C. tropicalis has been observed in various sub-Saharan African countries, including Tanzania. 160 Amphotericin B resistance has also been identified in Kenya 161 and Ghana. 151
Studies from Southwest Cameroon have indicated intermediate resistance to clotrimazole and amphotericin B, suggesting the need for higher dosages for effective treatment. 107 Topical antifungals, such as econazole and nystatin, are recommended for localised Candida infections, but resistance to nystatin has been reported in Ivory Coast, Ethiopia, Kenya, South Africa, and Cameroon, 162 while econazole resistance has been found in Southwest Cameroon. 163 A new multidrug-resistant species, C. auris, has been isolated in South Africa. 164 When comparing drug resistance patterns in South Africa and Cameroon, C. albicans showed significantly greater resistance to azoles, while C. glabrata generally exhibited low azole resistance. 107 A recent systematic review by Mushi et al. 47 on VVC in sub-Saharan Africa revealed that resistance to fluconazole in C. albicans ranged from 6.8% in Cameroon to 53.7% in Ethiopia.
Cryptococcus
Treatment of cryptococcosis involves a combination of amphotericin B deoxycholate and flucytosine for at least two weeks, followed by fluconazole for a minimum of eight weeks. 165 However, due to the widespread unavailability and inaccessibility of flucytosine in sub-Saharan Africa, treatment primarily relies on amphotericin B deoxycholate and fluconazole, despite their limited effectiveness. 166 Fluconazole is the most commonly prescribed antifungal drug for treating cryptococcal meningitis in the region, although it is less effective than amphotericin B. 167 Moreover, reports indicate the emergence of fluconazole resistance in C. neoformans, with associated chromosomal changes in the fungus. 168 Widespread resistance to fluconazole and, to a lesser extent, amphotericin B deoxycholate has significantly impacted treatment outcomes. 169 The C. gattii species complex generally exhibits greater resistance to azoles than isolates from the C. neoformans species complex. 170
Studies conducted in different sub-Saharan African countries have reported an increase in antifungal drug resistance among clinical isolates of C. neoformans. In Uganda, resistance to amphotericin B is rare, while resistance to fluconazole appears to be emerging. 171 According to the research conducted by Atim et al., 172 43.9% of the 310 clinical isolates of C. neoformans had IC50 values ⩾8 µg/ml. In vitro susceptibility testing of C. neoformans isolates from Egypt revealed that out of 29 isolates, only three exhibited resistance to the tested antifungal drugs. 173 Another study reported a decrease in fluconazole susceptibility among clinical isolates of C. neoformans in South Africa over a decade. 174 Similarly, a study in Cameroon found that 92.7% of C. neoformans with reduced susceptibility to fluconazole. 175 Recently, Bive et al. 176 found that two C. neoformans isolates out of 23 (8.7%) were resistant to fluconazole and one isolate out of 23 (4.3%) isolates was resistant to flucytosine. Research conducted in Nairobi, Kenya, highlighted the presence of azole resistance in C. neoformans isolates from clinical sources. 177 These findings emphasise the challenges faced in effectively treating cryptococcosis in sub-Saharan Africa due to the development of antifungal drug resistance.
Aspergillus
Azole-resistant Aspergillus species have emerged as a growing problem in sub-Saharan Africa. 178 In a Tanzanian study, all five clinical isolates of A. fumigatus were found to be resistant to triazole antifungal drugs, indicating a prevalence rate of 100%. 179 Surveillance studies conducted in various regions have also reported an increase in triazole-resistant A. fumigatus, raising concerns about the management of fungal diseases caused by this particular species. 180 The presence of azole-resistant A. fumigatus isolates has been documented in Tanzania, 94 and the first isolate of A. fumigatus resistant to azole antifungal agents was identified in Burkina Faso. 181 The resistance of filamentous fungi, notably A. fumigatus, to antifungal drugs, particularly azoles, has been attributed to their increased use in both clinical and environmental settings. 182 The increase in azole-resistant Aspergillus species poses significant challenges for the effective treatment of fungal infections in sub-Saharan Africa.
Dermatophytes
A study conducted in Nigeria revealed that every single isolate (100.0%) of T. rubrum, which is one of the main fungi responsible for tinea capitis, demonstrated resistance to ketoconazole, fluconazole, itraconazole, griseofulvin, and terbinafine. 183 There are limited data on T. rubrum resistance in sub-Saharan Africa.
Current and future interventions
Diagnosis and surveillance
Fungal diseases often receive insufficient attention in terms of funding, research, and health policies, with medical mycology accounting for only approximately 3% of infectious disease research expenditures. 184 Although effective species prevalence and antifungal monitoring programmes have been successfully implemented in Europe, the Asia-Pacific region, Latin America, and North America, comprehensive epidemiological data on fungal infections worldwide, particularly in sub-Saharan Africa, are lacking. For instance, a recent assessment of clinical mycology in Africa involving 40 institutions from 21 countries revealed that the majority of institutions (n = 39, 97.5%) reported having microbiology laboratories. However, regarding mycological diagnostic tools, three institutions (7.5%) reported no access to such services at all. 185 In cases of suspected fungal infection, 21 institutions (52.5%) conducted direct microscopy on clinical specimens, 34 (85.0%) used microscopy for diagnosing cryptococcosis, and only eight (20.0%) employed silver staining when pneumocystosis was suspected. 185 Similarly, data collected in 48 African countries revealed that a significant portion of the population in 14 sub-Saharan African countries, approximately 74% or 1.041 billion people, lacked access to histoplasmosis diagnostics, and 78.5% or 1.105 billion people lacked access to Pneumocystis pneumonia diagnostics. 186 Moreover, fungal culture, another diagnostic method, was available in 41 countries, covering a population of 94% or 1.289 billion people, whereas magnetic resonance imaging was routinely accessible to only 32.2% or 453.59 million people in sub-Saharan Africa. 186 A recent study investigating the diagnostic capacity for cutaneous fungal diseases in sub-Saharan Africa revealed that skin biopsies were regularly collected in 46% (22) of the 47 countries studied, while direct microscopy and histopathological examination of tissue were frequently performed in 42% (20) and 40% (19) of the countries, respectively. 187 The cost of diagnostics for patients was identified as a significant hindrance to the utilisation of these diagnostic methods. 187 Additionally, a study focusing on diagnostic options for pulmonary fungal diseases in sub-Saharan African countries with populations exceeding one million revealed that chest X-rays and computed tomography were frequently utilised in the public sector, while bronchoscopy and spirometry were commonly conducted in tertiary health facilities. 188
In sub-Saharan Africa, there is a deficiency in surveillance of antifungal medication resistance. 162 Surveillance programs play a vital role in the transition from empirical antifungal therapy, which often faces challenges due to varying resistance levels and the presence of inherently resistant organisms. 162 The optimal selection of effective medication and accurate identification of the causative fungal species are crucial for the management of many fungal infections. C. auris, a multidrug-resistant pathogen first discovered in South Africa 164 in 2009 and Kenya 43 in 2011, serves as a notable example. However, only a few laboratories in sub-Saharan Africa can accurately identify C. auris using current technologies, such as matrix-assisted laser desorption or ionisation time of flight (MALDI-ToF), or molecular techniques. 189 In a study focused on identifying fungi at the species level, biochemical tests were the predominant method utilised for species-level identification of fungi in 28 out of the 40 institutions (70%), while seven institutions (17.5%) employed MALDI-ToF, eight institutions (20%) used DNA sequencing and 19 institutions (47.5%) had access to automated blood culture monitoring. 185 These studies have revealed notable variations in both the accessibility and quality of diagnostic tests throughout sub-Saharan Africa. While a few instances showcased exceptional diagnostic capabilities and superior care, such occurrences were infrequent. Due to the limited availability of routine diagnostic facilities in most sub-Saharan African countries, many patients receive treatment without knowledge of the specific fungal species they are infected with, and there is a lack of updated guideline data for the appropriate prescription of antifungal medications. 162 Establishing diagnostic molecular microbiology laboratories, particularly in underdeveloped nations, such as those in Africa, presents significant challenges. 190 However, strengthening healthcare laboratories and overall system capacity is urgently needed in sub-Saharan Africa to address the burden of fungal infections.
Treatment
Unlike bacterial infections, which have more promising alternative treatments, 191 fungal infections still face limitations because there are currently limited therapeutic and combination options, thereby emphasising the importance of exploring new targets for the development of antifungal therapies. Africa encounters various challenges concerning fungal infections, including inadequate awareness among healthcare professionals and policymakers, as well as concerns regarding the cost, toxicity, availability, and accessibility of antifungal treatments. 192 The WHO Essential Medicines List presently includes amphotericin B, clotrimazole, fluconazole, flucytosine, griseofulvin, itraconazole, nystatin, and voriconazole. These medications have been identified and approved by the World Health Organisation as essential treatments for various fungal infections. 193 The scarcity of these essential medications in sub-Saharan Africa is a worrisome issue. One potential approach to address the emergence of antifungal resistance is the utilisation of combination therapies, which involve combining antifungal drugs with different mechanisms of action. 194 These combinations can synergistically enhance the elimination of pathogens, potentially enabling lower dosages of the drugs to mitigate toxicity concerns. 8 For instance, in the treatment of cryptococcal meningitis, the addition of flucytosine to amphotericin B has proven effective in preventing the selection of resistant colonies and improving treatment outcomes. 195 Combinations of flucytosine, fluconazole, and amphotericin B have also shown promise in accelerating infection clearance and reducing mortality rates. 196 The extensive use of antifungal medications is believed to contribute to the development of drug resistance, emphasising the need for a deeper understanding of resistance mechanisms and underlying biological processes.
A significant challenge faced in the sub-Saharan Africa region is the lack of comprehensive and current data regarding the burden of fungal infections, as well as management and treatment practices and antifungal resistance. This scarcity is due to limited research conducted on fungal infections within the region. As a result, this review falls short of capturing the full impact of these infections in the region and recommending treatment strategies to mitigate their burden.
Conclusion
This extensive review has revealed significant evidence on fungal infections in sub-Saharan Africa, a considerable proportion of which remain untreated or unreported. Most antifungal susceptibility tests have focused on species of the Candida genus. Given the increasing populations at risk of fungal infections, however, it is crucial to evaluate antifungal resistance in various invasive fungal diseases across the region. Giving additional attention to fungal infections is crucial to overcoming the multifaceted resistance of fungi to the limited spectrum of antifungal arsenals. Regulatory bodies need to monitor the use of antifungal agents across sub-Saharan Africa, allowing the use of potent antifungals and further discouraging the overuse of some antifungal agents. Additionally, active surveillance needs to be implemented to keep track of the diagnosis and antifungal treatment of fungal infections.
Acknowledgments
None.
Footnotes
ORCID iDs: Alex Odoom  https://orcid.org/0000-0001-5761-1564
https://orcid.org/0000-0001-5761-1564
Abdul-Halim Osman  https://orcid.org/0000-0002-6791-0442
https://orcid.org/0000-0002-6791-0442
Onyansaniba K. Ntim  https://orcid.org/0009-0006-9791-1848
https://orcid.org/0009-0006-9791-1848
Fleischer C. N. Kotey  https://orcid.org/0000-0003-0286-3638
https://orcid.org/0000-0003-0286-3638
Contributor Information
Emily Boakye-Yiadom, Department of Medical Microbiology, University of Ghana Medical School, Accra, Ghana; Department of Microbiology and Immunology, University of Health and Allied Sciences, Ho, Ghana.
Alex Odoom, Department of Medical Microbiology, University of Ghana Medical School, Accra, Ghana.
Abdul-Halim Osman, Department of Medical Microbiology, University of Ghana Medical School, Accra, Ghana.
Onyansaniba K. Ntim, Department of Medical Microbiology, University of Ghana Medical School, Accra, Ghana
Fleischer C. N. Kotey, Department of Medical Microbiology, University of Ghana Medical School, Accra, Ghana
Bright K. Ocansey, Division of Evolution, Infection and Genomics, Faculty of Biology, Medicine and Health, University of Manchester, Manchester Academic Health Science Centre, Manchester, UK
Eric S. Donkor, Department of Medical Microbiology, University of Ghana Medical School, Accra, P.O. Box KB 4236, Ghana.
Declarations
Ethics approval and consent to participate: Not applicable.
Consent for publication: Not applicable.
Author contributions: Emily Boakye-Yiadom: Data curation, Formal analysis, Investigation, Resources, Validation, Writing – original draft, Writing – review & editing.
Alex Odoom: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing.
Abdul-Halim Osman: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing.
Onyansaniba K. Ntim: Data curation, Formal analysis, Investigation, Methodology, Project administration, Visualization, Writing – original draft, Writing – review & editing.
Fleischer C. N. Kotey: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing.
Bright K. Ocansey: Data curation, Formal analysis, Investigation, Methodology, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing.
Eric S. Donkor: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
Funding: The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This review paper was supported by the Fogarty International Center of the National Institutes of Health, USA, through the Research and Capacity Building in Antimicrobial Resistance in West Africa (RECABAW) Training Programme hosted at the Department of Medical Microbiology, University of Ghana Medical School (Award Number: D43TW012487). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Competing interests: The authors declare that there is no conflict of interest.
Availability of data and materials: The data supporting the findings of this study are available within the article.
References
- 1. White SK, Schmidt RL, Walker BS, et al. (1→3)-β-D-glucan testing for the detection of invasive fungal infections in immunocompromised or critically ill people. Cochrane Database Syst Rev 2020; 7(7): CD009833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Hall RA, Noverr MC. Fungal interactions with the human host: exploring the spectrum of symbiosis. Curr Opin Microbiol 2017; 40: 58–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Carpouron JE, de Hoog S, Gentekaki E, et al. Emerging animal-associated fungal diseases. J Fungi 2022; 8: 611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ibe C. The impact of COVID-19 pandemic on invasive fungal infections in Africa: what have we learned? PLoS Negl Trop Dis 2022; 16: e0010720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Denning D, Beyene T. Estimated burden of serious fungal infections in Ethiopia. J Infect Public Health 2019; 12: 125. [DOI] [PubMed] [Google Scholar]
- 6. Bongomin F, Kibone W, Okot J, et al. Fungal diseases in Africa: epidemiologic, diagnostic and therapeutic advances. Ther Adv Infect Dis 2022; 9: 20499361221081440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Rajasingham R, Govender NP, Jordan A, et al. The global burden of HIV-associated cryptococcal infection in adults in 2020: a modelling analysis. Lancet Infect Dis 2022; 22: 1748–1755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Gow NAR, Johnson C, Berman J, et al. The importance of antimicrobial resistance in medical mycology. Nat Commun 2022; 13: 5352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Rodrigues ML, Nosanchuk JD. Fungal diseases as neglected pathogens: a wake-up call to public health officials. PLoS Negl Trop Dis 2020; 14: e0007964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Dangarembizi R, Wasserman S, Hoving JC. Emerging and re-emerging fungal threats in Africa. Parasite Immunol 2023; 45: e12953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bongomin F, Ekeng BE, Kibone W, et al. Invasive fungal diseases in Africa: a critical literature review. J Fungi (Basel) 2022; 8: 1236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Osset-Trénor P, Pascual-Ahuir A, Proft M. Fungal drug response and antimicrobial resistance. J Fungi (Basel) 2023; 9: 565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Ocansey BK, Pesewu GA, Codjoe FS, et al. Estimated burden of serious fungal infections in Ghana. J Fungi (Basel) 2019; 5: 38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Faini D, Maokola W, Furrer H, et al. Burden of serious fungal infections in Tanzania. Mycoses 2015; 58: 70–79. [DOI] [PubMed] [Google Scholar]
- 15. Oladele R, Otu AA, Olubamwo O, et al. Evaluation of knowledge and awareness of invasive fungal infections amongst resident doctors in Nigeria. Pan Afr Med J 2020; 36: 297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Kamwiziku GK, Makangara JC, Orefuwa E, et al. Serious fungal diseases in Democratic Republic of Congo – Incidence and prevalence estimates. Mycoses 2021; 64: 1159–1169. [DOI] [PubMed] [Google Scholar]
- 17. Badiane AS, Ndiaye D, Denning DW. Burden of fungal infections in Senegal. Mycoses 2015; 58: 63–69. [DOI] [PubMed] [Google Scholar]
- 18. Lakoh S, Orefuwa E, Kamara MN, et al. The burden of serious fungal infections in Sierra Leone: a national estimate. Ther Adv Infect Dis 2021; 8: 20499361211027996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Pfavayi LT, Denning DW, Baker S, et al. Determining the burden of fungal infections in Zimbabwe. Sci Rep 2021; 11: 13240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Bamba S, Zida A, Sangaré I, et al. Burden of severe fungal infections in Burkina Faso. J Fungi (Basel) 2018; 4: 35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Mandengue CE, Denning DW. The burden of serious fungal infections in Cameroon. J Fungi (Basel) 2018; 4: 44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Koffi D, Bonouman IV, Toure AO, et al. Estimates of serious fungal infection burden in Côte d’Ivoire and country health profile. J Mycol Med 2021; 31: 101086. [DOI] [PubMed] [Google Scholar]
- 23. Amona FM, Denning DW, Moukassa D, et al. Current burden of serious fungal infections in Republic of Congo. Mycoses 2020; 63: 543–552. [DOI] [PubMed] [Google Scholar]
- 24. Tufa TB, Denning DW. The burden of fungal infections in Ethiopia. J Fungi (Basel) 2019; 5: 109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Ratemo SN, Denning DW. Burden of fungal infections in Kenya. Mycology 2023; 14: 142–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Kalua K, Zimba B, Denning DW. Estimated burden of serious fungal infections in Malawi. J Fungi (Basel) 2018; 4: 61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Doumbo SN, Cissoko Y, Dama S, et al. The estimated burden of fungal diseases in Mali. J Mycol Med 2023; 33: 101333. [DOI] [PubMed] [Google Scholar]
- 28. Sacarlal J, Denning DW. Estimated burden of serious fungal infections in Mozambique. J Fungi (Basel) 2018; 4: 75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Dunaiski CM, Denning DW. Estimated burden of fungal infections in Namibia. J Fungi (Basel) 2019; 5: 75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Ahmed SA, Ismail M, Albirair M, et al. Fungal infections in Sudan: an underestimated health problem. PLoS Negl Trop Dis 2023; 17: e0011464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Dorkenoo AM, Adjetey-Toglozombio AK, Ocansey BK, et al. Estimated burden of serious fungal infections in Togo. Mycoses 2021; 64: 1535–1541. [DOI] [PubMed] [Google Scholar]
- 32. Schwartz I, Denning D. The estimated burden of fungal diseases in South Africa. J Infect Public Health 2019; 12: 124. [Google Scholar]
- 33. Zono BB, Kasumba DM, Situakibanza Nani-Tuma H, et al. Cryptococcosis in the Democratic Republic of Congo from 1953 to 2021: a systematic review and meta-analysis. Mycoses 2022; 65: 580–589. [DOI] [PubMed] [Google Scholar]
- 34. Hay R, Denning DW, Bonifaz A, et al. The diagnosis of fungal neglected tropical diseases (Fungal NTDs) and the Role of investigation and laboratory tests: an expert consensus report. Trop Med Infect Dis 2019; 4: 122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Bongomin F, Olum R, Nsenga L, et al. Burden of tinea capitis among children in Africa: protocol for a systematic review and meta-analysis of observational studies, 1990-2020. BMJ Open 2020; 10: e041230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Hogewoning A, Amoah A, Bavinck JNB, et al. Skin diseases among schoolchildren in Ghana, Gabon, and Rwanda. Int J Dermatol 2013; 52: 589–600. [DOI] [PubMed] [Google Scholar]
- 37. Coulibaly O, L’Ollivier C, Piarroux R, et al. Epidemiology of human dermatophytoses in Africa. Med Mycol 2018; 56: 145–161. [DOI] [PubMed] [Google Scholar]
- 38. Ellis J, Bangdiwala AS, Cresswell FV, et al. The changing epidemiology of HIV-associated adult meningitis, Uganda 2015-2017. Open forum Infect Dis 2019; 6: ofz419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Khairat SM, Sayed AM, Nabih M, et al. Prevalence of Candida blood stream infections among children in tertiary care hospital: detection of species and antifungal susceptibility. Infect Drug Resist 2019; 12: 2409–2416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Mushi MF, Bader O, Taverne-Ghadwal L, et al. Oral candidiasis among African human immunodeficiency virus-infected individuals: 10 years of systematic review and meta-analysis from sub-Saharan Africa. J Oral Microbiol 2017; 9: 1317579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Hussain M, Whitelaw A, Parker A. A five-year retrospective descriptive study on the clinical characteristics and outcomes of candidaemia at a tertiary hospital in South Africa. IJID Reg 2022; 3: 79–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Shoham S, Levitz SM. The immune response to fungal infections. Br J Haematol 2005; 129: 569–582. [DOI] [PubMed] [Google Scholar]
- 43. Adam RD, Revathi G, Okinda N, et al. Analysis of Candida auris fungemia at a single facility in Kenya. Int J Infect Dis 2019; 85: 182–187. [DOI] [PubMed] [Google Scholar]
- 44. Naicker SD, Govender N, Patel J, et al. Comparison of species-level identification and antifungal susceptibility results from diagnostic and reference laboratories for bloodstream Candida surveillance isolates, South Africa, 2009-2010. Med Mycol 2016; 54: 816–824. [DOI] [PubMed] [Google Scholar]
- 45. Naicker SD, Magobo RE, Zulu TG, et al. Two echinocandin-resistant Candida glabrata FKS mutants from South Africa. Med Mycol Case Rep 2016; 11: 24–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Abruquah HH. Prevalence and antifungal susceptibility of Candida species isolated from women attending a gynaecological clinic in Kumasi, Ghana. J Sci Technol 2012; 32: 39–45. [Google Scholar]
- 47. Mushi MF, Olum R, Bongomin F. Prevalence, antifungal susceptibility and etiology of vulvovaginal candidiasis in sub–Saharan Africa: a systematic review with meta-analysis and meta-regression. Med Mycol 2022; 60: myac037. [DOI] [PubMed] [Google Scholar]
- 48. Olum R, Baluku JB, Okidi R, et al. Prevalence of HIV-associated esophageal candidiasis in sub-Saharan Africa: a systematic review and meta-analysis. Trop Med Health 2020; 48: 82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Dagenais TRT, Keller NP. Pathogenesis of Aspergillus fumigatus in invasive aspergillosis. Clin Microbiol Rev 2009; 22: 447–465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Kosmidis C, Denning DW. The clinical spectrum of pulmonary aspergillosis. Thorax 2014; 70: 270–277. [DOI] [PubMed] [Google Scholar]
- 51. Yerbanga IW, Nakanabo Diallo S, Rouamba T, et al. A systematic review of epidemiology, risk factors, diagnosis, antifungal resistance, and management of invasive aspergillosis in Africa. J Mycol Med 2023; 33: 101328. [DOI] [PubMed] [Google Scholar]
- 52. Kwizera R, Bongomin F, Lukande R. Deep fungal infections diagnosed by histology in Uganda: a 70-year retrospective study. Med Mycol 2020; 58: 1044–1052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Rudramurthy SM, Paul RA, Chakrabarti A, et al. Invasive aspergillosis by Aspergillus flavus: epidemiology, diagnosis, antifungal resistance, and management. J Fungi (Basel) 2019; 5: 55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Gheith S, Saghrouni F, Bannour W, et al. Characteristics of invasive aspergillosis in neutropenic haematology patients (Sousse, Tunisia). Mycopathologia 2014; 177: 281–289. [DOI] [PubMed] [Google Scholar]
- 55. Skalski JH, Kottom TJ, Limper AH. Pathobiology of Pneumocystis pneumonia: life cycle, cell wall and cell signal transduction. FEMS Yeast Res 2015; 15: fov046. [DOI] [PubMed] [Google Scholar]
- 56. Wasserman S, Engel ME, Griesel R, et al. Burden of Pneumocystis pneumonia in HIV-infected adults in sub-Saharan Africa: a systematic review and meta-analysis. BMC Infect Dis 2016; 16: 482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Nkinin SW, Daly KR, Walzer PD, et al. Evidence for high prevalence of Pneumocystis jirovecii exposure among Cameroonians. Acta Trop 2009; 112: 219–224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Azar MM, Hage CA. Laboratory diagnostics for histoplasmosis. J Clin Microbiol 2017; 55: 1612–1620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Loulergue P, Bastides F, Baudouin V, et al. Literature review and case histories of Histoplasma capsulatum var. duboisii infections in HIV-infected patients. Emerg Infect Dis 2007; 13: 1647–1652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Ocansey BK, Otoo B, Asamoah I, et al. Cryptococcal and histoplasma antigen screening among people with human immunodeficiency virus in Ghana and comparative analysis of OIDx histoplasma lateral flow assay and IMMY histoplasma enzyme immunoassay. Open Forum Infect Dis 2022; 9: ofac277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Oladele RO, Ayanlowo OO, Richardson MD, et al. Histoplasmosis in Africa: an emerging or a neglected disease? PLoS Negl Trop Dis 2018; 12: e0006046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Mandengue CE, Ngandjio A, Atangana PJA. Histoplasmosis in HIV-infected persons, Yaoundé, Cameroon. Emerg Infect Dis 2015; 21: 2094–2096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Darré T, Saka B, Mouhari-Touré A, et al. Histoplasmosis by Histoplasma capsulatum var. duboisii observed at the laboratory of pathological anatomy of Lomé in Togo. J Pathog 2017; 2017: 2323412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Pakasa N, Biber A, Nsiangana S, et al. African histoplasmosis in HIV-negative patients, Kimpese, Democratic Republic of the Congo. Emerg Infect Dis 2018; 24: 2068–2070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Arenas R, Sánchez-Cardenas CD, Ramirez-Hobak L, et al. Sporotrichosis: from KOH to molecular biology. J Fungi (Basel) 2018; 4: 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Rasamoelina T, Rakotozandrindrainy N, Raberahona M, et al. Chromoblastomycosis and sporotrichosis in Madagascar: epidemiology, molecular diagnostic and perspectives. J Mycol Med 2016; 26: e15. [Google Scholar]
- 67. Lurie HI. Five unusual cases of sporotrichosis from South Africa showing lesions in muscles, bones, and viscera. Br J Surg 1963; 50: 585–591. [DOI] [PubMed] [Google Scholar]
- 68. Queiroz-Telles F, de Hoog S, Santos DWCL, et al. Chromoblastomycosis. Clin Microbiol Rev 2017; 30: 233–276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Santos DWCL, de Azevedo C de MPES, Vicente VA, et al. The global burden of chromoblastomycosis. PLoS Negl Trop Dis 2021; 15: e0009611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Dukik K, Muñoz JF, Jiang Y, et al. Novel taxa of thermally dimorphic systemic pathogens in the Ajellomycetaceae (Onygenales). Mycoses 2017; 60: 296–309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Smith EB. History of antifungals. J Am Acad Dermatol 1990; 23: 776–778. [DOI] [PubMed] [Google Scholar]
- 72. Campoy S, Adrio JL. Antifungals. Biochem Pharmacol 2017; 133: 86–96. [DOI] [PubMed] [Google Scholar]
- 73. Gupta AK, Mays RR, Versteeg SG, et al. Tinea capitis in children: a systematic review of management. J Eur Acad Dermatol Venereol 2018; 32: 2264–2274. [DOI] [PubMed] [Google Scholar]
- 74. Kaur N, Bains A, Kaushik R, et al. A review on antifungal efficiency of plant extracts entrenched polysaccharide-based nanohydrogels. Nutrients 2021; 13: 2055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Szymański M, Chmielewska S, Czyz·ewska U, et al. Echinocandins – structure, mechanism of action and use in antifungal therapy. J Enzyme Inhib Med Chem 2022; 37: 876–894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Ghannoum MA, Rice LB. Antifungal agents: mode of action, mechanisms of resistance, and correlation of these mechanisms with bacterial resistance. Clin Microbiol Rev 1999; 12: 501–517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Ibe C, Otu AA, Torres HA, et al. Recent advances and challenges in the early diagnosis and management of invasive fungal infections in Africa. Diagn Microbiol Infect Dis 2003; 22: 25–28. [DOI] [PubMed] [Google Scholar]
- 78. Ocansey BK, Dadzie EA, Eduful SK, et al. Improving awareness, diagnosis and management of invasive fungal infections in Ghana: establishment of the Ghana Medical Mycology Society. Med Mycol 2022; 60: myac069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Allen D, Wilson D, Drew R, et al. Azole antifungals: 35 years of invasive fungal infection management. Expert Rev Anti Infect Ther 2015; 13: 787–798. [DOI] [PubMed] [Google Scholar]
- 80. Lakhani P, Patil A, Majumdar S. Challenges in the polyene- and azole-based pharmacotherapy of ocular fungal infections. J Ocul Pharmacol Ther 2019; 35: 6–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Perfect JR. The antifungal pipeline: a reality check. Nat Rev Drug Discov 2017; 16: 603–616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Revie NM, Iyer KR, Robbins N, et al. Antifungal drug resistance: evolution, mechanisms and impact. Curr Opin Microbiol 2018; 45: 70–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Lee Y, Puumala E, Robbins N, et al. Antifungal drug resistance: molecular mechanisms in Candida albicans and beyond. Chem Rev 2021; 121: 3390–3411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Robbins N, Caplan T, Cowen LE. Molecular evolution of antifungal drug resistance. Annu Rev Microbiol 2017; 71: 753–775. [DOI] [PubMed] [Google Scholar]
- 85. Fisher MC, Alastruey-Izquierdo A, Berman J, et al. Tackling the emerging threat of antifungal resistance to human health. Nat Rev Microbiol 2022; 20: 557–571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Roemer T, Krysan DJ. Antifungal drug development: challenges, unmet clinical needs, and new approaches. Cold Spring Harb Perspect Med 2014; 4: a019703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Pappas PG, Kauffman CA, Andes D, et al. Clinical practice guidelines for the management of candidiasis: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis 2009; 48: 503–535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Pfaller MA, Diekema DJ, Turnidge JD, et al. Twenty years of the SENTRY antifungal surveillance program: results for Candida species from 1997-2016. Open Forum Infect Dis 2019; 6: S79–S94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Sun Y, Tan L, Yao Z, et al. In vitro and in vivo interactions of TOR inhibitor AZD8055 and azoles against pathogenic fungi. Microbiol Spectr 2022; 10: e0200721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Eltschinger S, Loewith R. TOR Complexes and the maintenance of cellular homeostasis. Trends Cell Biol 2016; 26: 148–159. [DOI] [PubMed] [Google Scholar]
- 91. Madeira JB, Masuda CA, Maya-Monteiro CM, et al. TORC1 inhibition induces lipid droplet replenishment in yeast. Mol Cell Biol 2015; 35: 737–746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. So Y-S, Lee D-G, Idnurm A, et al. The TOR pathway plays pleiotropic roles in growth and stress responses of the fungal pathogen Cryptococcus neoformans. Genetics 2019; 212: 1241–1258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Drakulovski P, Krasteva D, Bellet V, et al. Exposure of Cryptococcus neoformans to seven commonly used agricultural azole fungicides induces resistance to fluconazole as well as cross-resistance to voriconazole, posaconazole, itraconazole and isavuconazole. Pathog (Basel) 2023; 12: 662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Chowdhary A, Sharma C, van den Boom M, et al. Multi-azole-resistant Aspergillus fumigatus in the environment in Tanzania. J Antimicrob Chemother 2014; 69: 2979–2983. [DOI] [PubMed] [Google Scholar]
- 95. Govender NP, Patel J, Magobo RE, et al. Emergence of azole-resistant Candida parapsilosis causing bloodstream infection: results from laboratory-based sentinel surveillance in South Africa. J Antimicrob Chemother 2016; 71: 1994–2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Hamdy R, Fayed B, Hamoda AM, et al. Essential oil-based design and development of novel anti-candida azoles formulation. Molecules 2020; 25: 1463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Hüttel W. Echinocandins: structural diversity, biosynthesis, and development of antimycotics. Appl Microbiol Biotechnol 2021; 105: 55–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Perlin DS. Mechanisms of echinocandin antifungal drug resistance. Ann N Y Acad Sci 2015; 1354: 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Onishi J, Meinz M, Thompson J, et al. Discovery of novel antifungal (1,3)-beta-D-glucan synthase inhibitors. Antimicrob Agents Chemother 2000; 44: 368–377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Aruanno M, Glampedakis E, Lamoth F. Echinocandins for the treatment of invasive aspergillosis: from laboratory to bedside. Antimicrob Agents Chemother 2019; 63: e00399-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Song Y, Chen X, Yan Y, et al. Prevalence and antifungal susceptibility of pathogenic yeasts in China: A 10-year retrospective study in a teaching hospital. Front Microbiol 2020; 11: 1401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Ingham CJ, Schneeberger PM. Microcolony imaging of Aspergillus fumigatus treated with echinocandins reveals both fungistatic and fungicidal activities. PLoS One 2012; 7: e35478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Kofla G, Ruhnke M. Pharmacology and metabolism of anidulafungin, caspofungin and micafungin in the treatment of invasive candidosis: review of the literature. Eur J Med Res 2011; 16: 159–166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Mavridou E, Meletiadis J, Rijs A, et al. The strength of synergistic interaction between posaconazole and caspofungin depends on the underlying azole resistance mechanism of Aspergillus fumigatus. Antimicrob Agents Chemother 2015; 59: 1738–1744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Grey A, Joubert R, Steyn S, et al. Prescribing patterns of echinocandins in adult patients in a private hospital in Gauteng, South Africa. S Afr J Infect Dis 2023; 38: 470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Gebretekle GB, Fentie AM, Gebremariam GT, et al. Cost-utility analysis of caspofungin and fluconazole for primary treatment of invasive candidiasis and candidemia in Ethiopia. BMC Health Serv Res 2022; 22: 1302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. dos Santos Abrantes PM, McArthur CP, Africa CWJ. Multi-drug resistant oral Candida species isolated from HIV-positive patients in South Africa and Cameroon. Diagn Microbiol Infect Dis 2014; 79: 222–227. [DOI] [PubMed] [Google Scholar]
- 108. Robbins N, Wright GD, Cowen LE. Antifungal drugs: the current armamentarium and development of new agents. Microbiol Spectr 2016; 4: 10. [DOI] [PubMed] [Google Scholar]
- 109. Tevyashova AN, Olsufyeva EN, Solovieva SE, et al. Structure-antifungal activity relationships of polyene antibiotics of the amphotericin B group. Antimicrob Agents Chemother 2013; 57: 3815–3822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. Ostrosky-Zeichner L, Casadevall A, Galgiani JN, et al. An insight into the antifungal pipeline: selected new molecules and beyond. Nat Rev Drug Discov 2010; 9: 719–727. [DOI] [PubMed] [Google Scholar]
- 111. Anderson TM, Clay MC, Cioffi AG, et al. Amphotericin forms an extramembranous and fungicidal sterol sponge. Nat Chem Biol 2014; 10: 400–406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Moosa M-YS. Resistance to amphotericin B does not emerge during treatment for invasive aspergillosis. J Antimicrob Chemother 2002; 49: 209–213. [DOI] [PubMed] [Google Scholar]
- 113. Sabra R, Branch RA. Amphotericin B nephrotoxicity. Drug Saf 1990; 5: 94–108. [DOI] [PubMed] [Google Scholar]
- 114. Vermes A. Flucytosine: a review of its pharmacology, clinical indications, pharmacokinetics, toxicity and drug interactions. J Antimicrob Chemother 2000; 46: 171–179. [DOI] [PubMed] [Google Scholar]
- 115. Miot J, Leong T, Takuva S, et al. Cost-effectiveness analysis of flucytosine as induction therapy in the treatment of cryptococcal meningitis in HIV-infected adults in South Africa. BMC Health Serv Res 2021; 21: 305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Berman J, Krysan DJ. Drug resistance and tolerance in fungi. Nat Rev Microbiol 2020; 18: 319–331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Pappas PG, Kauffman CA, Andes DR, et al. Executive summary: clinical practice guideline for the management of Candidiasis: 2016 update by the Infectious Diseases Society of America. Clin Infect Dis 2016; 62: 409–417. [DOI] [PubMed] [Google Scholar]
- 118. Anderson JB. Evolution of antifungal-drug resistance: mechanisms and pathogen fitness. Nat Rev Microbiol 2005; 3: 547–556. [DOI] [PubMed] [Google Scholar]
- 119. Verweij PE, Lucas JA, Arendrup MC, et al. The one health problem of azole resistance in Aspergillus fumigatus: current insights and future research agenda. Fungal Biol Rev 2020; 34: 202–214. [Google Scholar]
- 120. Fisher MC, Hawkins NJ, Sanglard D, et al. Worldwide emergence of resistance to antifungal drugs challenges human health and food security. Science 2018; 360: 739–742. [DOI] [PubMed] [Google Scholar]
- 121. Zarnowski R, Sanchez H, Covelli AS, et al. Candida albicans biofilm-induced vesicles confer drug resistance through matrix biogenesis. PLoS Biol 2018; 16: e2006872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Shapiro RS, Robbins N, Cowen LE. Regulatory circuitry governing fungal development, drug resistance, and disease. Microbiol Mol Biol Rev 2011; 75: 213–267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Vincent BM, Lancaster AK, Scherz-Shouval R, et al. Fitness trade-offs restrict the evolution of resistance to amphotericin B. PLoS Biol 2013; 11: e1001692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Jensen RH, Astvad KMT, Silva LV, et al. Stepwise emergence of azole, echinocandin and amphotericin B multidrug resistance in vivo in Candida albicans orchestrated by multiple genetic alterations. J Antimicrob Chemother 2015; 70: 2551–2555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Vandeputte P, Tronchin G, Larcher G, et al. A nonsense mutation in the ERG6 gene leads to reduced susceptibility to polyenes in a clinical isolate of Candida glabrata. Antimicrob Agents Chemother 2008; 52: 3701–3709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Garcia-Effron G, Katiyar SK, Park S, et al. A naturally occurring proline-to-alanine amino acid change in Fks1p in Candida parapsilosis, Candida orthopsilosis, and Candida metapsilosis accounts for reduced echinocandin susceptibility. Antimicrob Agents Chemother 2008; 52: 2305–2312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Healey KR, Zhao Y, Perez WB, et al. Prevalent mutator genotype identified in fungal pathogen Candida glabrata promotes multi-drug resistance. Nat Commun 2016; 7: 11128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Lamoth F, Lockhart SR, Berkow EL, et al. Changes in the epidemiological landscape of invasive candidiasis. J Antimicrob Chemother 2018; 73: i4–i13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Arendrup MC, Perlin DS, Jensen RH, et al. Differential in vivo activities of anidulafungin, caspofungin, and micafungin against Candida glabrata isolates with and without FKS resistance mutations. Antimicrob Agents Chemother 2012; 56: 2435–2442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Alexander BD, Johnson MD, Pfeiffer CD, et al. Increasing echinocandin resistance in Candida glabrata: clinical failure correlates with presence of FKS mutations and elevated minimum inhibitory concentrations. Clin Infect Dis 2013; 56: 1724–1732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131. Balashov SV, Gardiner R, Park S, et al. Rapid, high-throughput, multiplex, real-time PCR for identification of mutations in the cyp51A gene of Aspergillus fumigatus that confer resistance to itraconazole. J Clin Microbiol 2005; 43: 214–222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Willger SD, Puttikamonkul S, Kim K-H, et al. A sterol-regulatory element binding protein is required for cell polarity, hypoxia adaptation, azole drug resistance, and virulence in Aspergillus fumigatus. PLoS Pathog 2008; 4: e1000200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Cowen LE, Sanglard D, Howard SJ, et al. Mechanisms of antifungal drug resistance. Cold Spring Harb Perspect Med 2014; 5: a019752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Tsao S, Rahkhoodaee F, Raymond M. Relative contributions of the Candida albicans ABC transporters Cdr1p and Cdr2p to clinical azole resistance. Antimicrob Agents Chemother 2009; 53: 1344–1352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Torelli R, Posteraro B, Ferrari S, et al. The ATP-binding cassette transporter–encoding gene CgSNQ2 is contributing to the CgPDR1-dependent azole resistance of Candida glabrata. Mol Microbiol 2008; 68: 186–201. [DOI] [PubMed] [Google Scholar]
- 136. Kelly SL, Lamb DC, Kelly DE, et al. Resistance to fluconazole and cross-resistance to amphotericin B in Candida albicans from AIDS patients caused by defective sterol Δ5,6-desaturation. FEBS Lett 1997; 400: 80–82. [DOI] [PubMed] [Google Scholar]
- 137. Spettel K, Barousch W, Makristathis A, et al. Analysis of antifungal resistance genes in Candida albicans and Candida glabrata using next generation sequencing. PLoS One 2019; 14: e0210397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Papon N, Noël T, Florent M, et al. Molecular mechanism of flucytosine resistance in Candida lusitaniae: contribution of the FCY2, FCY1, and FUR1 genes to 5-fluorouracil and fluconazole cross-resistance. Antimicrob Agents Chemother 2007; 51: 369–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Delma FZ, Al-Hatmi AMS, Brüggemann RJM, et al. Molecular mechanisms of 5-fluorocytosine resistance in yeasts and filamentous fungi. J Fungi (Basel) 2021; 7: 909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Kern L, de Montigny J, Lacroute F, et al. Regulation of the pyrimidine salvage pathway by the FUR1 gene product of Saccharomyces cerevisiae. Curr Genet 1991; 19: 333–337. [DOI] [PubMed] [Google Scholar]
- 141. Vandeputte P, Pineau L, Larcher G, et al. Molecular mechanisms of resistance to 5-fluorocytosine in laboratory mutants of Candida glabrata. Mycopathologia 2010; 171: 11–21. [DOI] [PubMed] [Google Scholar]
- 142. Billmyre RB, Applen Clancey S, Li LX, et al. 5-fluorocytosine resistance is associated with hypermutation and alterations in capsule biosynthesis in Cryptococcus. Nat Commun 2020; 11: 127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Costa C, Ponte A, Pais P, et al. New mechanisms of flucytosine resistance in C. glabrata unveiled by a chemogenomics analysis in S. cerevisiae. PLoS One 2015; 10: e0135110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Beese-Sims SE, Pan S-J, Lee J, et al. Mutants in the Candida glabrata glycerol channels are sensitized to cell wall stress. Eukaryot Cell 2012; 11: 1512–1519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Ben-Ami R, Kontoyiannis DP. Resistance to antifungal drugs. Infect Dis Clin North Am 2021; 35: 279–311. [DOI] [PubMed] [Google Scholar]
- 146. Arastehfar A, Gabaldón T, Garcia-Rubio R, et al. Drug-resistant fungi: an emerging challenge threatening our limited antifungal armamentarium. Antibiotics (Basel) 2020; 9: 877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Lestrade PPA, Meis JF, Melchers WJG, et al. Triazole resistance in Aspergillus fumigatus: recent insights and challenges for patient management. Clin Microbiol Infect 2019; 25: 799–806. [DOI] [PubMed] [Google Scholar]
- 148. Jia X-M, Ma Z-P, Jia Y, et al. RTA2, a novel gene involved in azole resistance in Candida albicans. Biochem Biophys Res Commun 2008; 373: 631–636. [DOI] [PubMed] [Google Scholar]
- 149. Sanglard D, Kuchler K, Ischer F, et al. Mechanisms of resistance to azole antifungal agents in Candida albicans isolates from AIDS patients involve specific multidrug transporters. Antimicrob Agents Chemother 1995; 39: 2378–2386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Arendrup MC, Patterson TF. Multidrug-resistant Candida: epidemiology, molecular mechanisms, and treatment. J Infect Dis 2017; 216: S445–S451. [DOI] [PubMed] [Google Scholar]
- 151. Feglo P. Prevalence and antifungal susceptibility patterns of yeast isolates at the Komfo Anokye Teaching Hospital (KATH), Kumasi, Ghana. Br Microbiol Res J 2012; 2: 10–22. [Google Scholar]
- 152. Kwamin F, Nartey NO, Codjoe FS, et al. Distribution of Candida species among HIV-positive patients with oropharyngeal candidiasis in Accra, Ghana. J Infect Dev Ctries 2013; 7: 41–45. [DOI] [PubMed] [Google Scholar]
- 153. Waikhom SD, Afeke I, Kwawu GS, et al. Prevalence of vulvovaginal candidiasis among pregnant women in the Ho municipality, Ghana: species identification and antifungal susceptibility of Candida isolates. BMC Pregnancy Childbirth 2020; 20: 266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Johnson MD, Perfect JR. Use of antifungal combination therapy: agents, order, and timing. Curr Fungal Infect Rep 2010; 4: 87–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Blignaut E, Messer S, Hollis RJ, et al. Antifungal susceptibility of South African oral yeast isolates from HIV/AIDS patients and healthy individuals. Diagn Microbiol Infect Dis 2002; 44: 169–174. [DOI] [PubMed] [Google Scholar]
- 156. Taura DW, Maje MH, Koki AM, et al. Antifungal resistance among Candida species from patients with genitourinary tract infection at Muhammad Abdullahi Wase Specialist Hospital, Kano – Nigeria. Nig J Basic Appl Sci 2013; 21: 33–38. [Google Scholar]
- 157. Hamza OJM, Matee MIN, Moshi MJ, et al. Species distribution and in vitro antifungal susceptibility of oral yeast isolates from Tanzanian HIV-infected patients with primary and recurrent oropharyngeal candidiasis. BMC Microbiol 2008; 8: 135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Mulu A, Kassu A, Anagaw B, et al. Frequent detection of ‘azole’ resistant Candida species among late presenting AIDS patients in northwest Ethiopia. BMC Infect Dis 2013; 13: 82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Yoo J, Il, Choi CW, Lee KM, et al. Gene expression and identification related to fluconazole resistance of Candida glabrata strains. Osong Public Health Res Perspect 2010; 1: 36–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Pfaller MA, Moet GJ, Messer SA, et al. Geographic variations in species distribution and echinocandin and azole antifungal resistance rates among Candida bloodstream infection isolates: report from the SENTRY Antimicrobial Surveillance Program (2008 to 2009). J Clin Microbiol 2011; 49: 396–399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Bii CC, Ouko TT, Amukoye E, et al. Antifungal drug susceptibility of Candida albicans. East Afr Med J 2002; 79: 143–145. [DOI] [PubMed] [Google Scholar]
- 162. Africa CWJ, Abrantes PMDS. Candida antifungal drug resistance in sub-Saharan African populations: a systematic review. F1000Res 2016; 5: 2832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163. Njunda AL, Nsagha DS, Assob JCN, et al. In vitro antifungal susceptibility patterns of Candida albicans from HIV and AIDS patients attending the Nylon Health District Hospital in Douala, Cameroon. J Public Health Afr 2012; 3: e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Magobo RE, Corcoran C, Seetharam S, et al. Candida auris-associated candidemia, South Africa. Emerg Infect Dis 2014; 20: 1250–1251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165. Perfect JR, Dismukes WE, Dromer F, et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the Infectious Diseases Society of America. Clin Infect Dis 2010; 50: 291–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. Molloy SF, Kanyama C, Heyderman RS, et al. Antifungal combinations for treatment of cryptococcal meningitis in Africa. N Engl J Med 2018; 378: 1004–1017. [DOI] [PubMed] [Google Scholar]
- 167. Jarvis JN, Leeme TB, Molefi M, et al. Short-course high-dose liposomal amphotericin B for human immunodeficiency virus-associated cryptococcal meningitis: a phase 2 randomized controlled trial. Clin Infect Dis 2019; 68: 393–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168. Hope W, Stone NRH, Johnson A, et al. Fluconazole monotherapy is a suboptimal option for initial treatment of cryptococcal meningitis because of emergence of resistance. MBio 2019; 10: e02575-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Ibe C, Okoye CA. Integrated healthcare approach can curb the increasing cases of cryptococcosis in Africa. PLoS Negl Trop Dis 2022; 16: e0010625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Herkert PF, Hagen F, Pinheiro RL, et al. Ecoepidemiology of Cryptococcus gattii in developing countries. J Fungi (Basel) 2017; 3: 62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171. Smith KD, Achan B, Hullsiek KH, et al. Increased antifungal drug resistance in clinical isolates of Cryptococcus neoformans in Uganda. Antimicrob Agents Chemother 2015; 59: 7197–7204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172. Atim PB, Meya DB, Gerlach ES, et al. Lack of Association between fluconazole susceptibility and ERG11 nucleotide polymorphisms in Cryptococcus neoformans clinical isolates from Uganda. J Fungi (Basel) 2022; 8: 508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173. Abdel-Salam HA. In vitro susceptibility of Cryptococcus neoformans clinical isolates from Egypt to seven antifungal drugs. Mycoses 2005; 48: 327–332. [DOI] [PubMed] [Google Scholar]
- 174. Naicker SD, Mpembe RS, Maphanga TG, et al. Decreasing fluconazole susceptibility of clinical South African Cryptococcus neoformans isolates over a decade. PLoS Negl Trop Dis 2020; 14: e0008137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Kammalac Ngouana T, Dongtsa J, Kouanfack C, et al. Cryptoccocal meningitis in Yaoundé (Cameroon) HIV infected patients: diagnosis, frequency and Cryptococcus neoformans isolates susceptibility study to fluconazole. J Mycol Med 2015; 25: 11–16. [DOI] [PubMed] [Google Scholar]
- 176. Bive BZ, Sacheli R, Mudogo CN, et al. Correlation of antifungal susceptibility and sequence types within Cryptococcus neoformans VNI from HIV patients, and ERG11 gene polymorphism. J Med Mycol 2023; 33: 101428. [DOI] [PubMed] [Google Scholar]
- 177. Bii CC, Makimura K, Abe S, et al. Antifungal drug susceptibility of Cryptococcus neoformans from clinical sources in Nairobi, Kenya. Mycoses 2006; 50: 25–30. [DOI] [PubMed] [Google Scholar]
- 178. Otu A, Osaigbovo I, Orefuwa E, et al. Collaborative One Health approaches can mitigate increasing azole-resistant Aspergillus fumigatus in Africa. Lancet Microbe 2021; 2: e490–e491. [DOI] [PubMed] [Google Scholar]
- 179. Mushi MF, Buname G, Bader O, et al. Aspergillus fumigatus carrying TR34/L98H resistance allele causing complicated suppurative otitis media in Tanzania: call for improved diagnosis of fungi in sub-Saharan Africa. BMC Infect Dis 2016; 16: 464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180. Lestrade PP, Bentvelsen RG, Schauwvlieghe AFAD, et al. Voriconazole resistance and mortality in invasive aspergillosis: a multicenter retrospective cohort study. Clin Infect Dis 2018; 68: 1463–1471. [DOI] [PubMed] [Google Scholar]
- 181. Yerbanga IW, Resendiz-Sharpe A, Bamba S, et al. First investigative study of azole-resistant Aspergillus fumigatus in the environment in Burkina Faso. Int J Environ Res Public Health 2021; 18: 2250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182. Berger S, El Chazli Y, Babu AF, et al. Azole resistance in Aspergillus fumigatus: a consequence of antifungal use in agriculture? Front Microbiol 2017; 8: 1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183. Ayodele EH, Charles N, Abayomi F. Prevalence, identification and antifungal susceptibility of dermatophytes causing tinea capitis in a locality of north central Nigeria. African J Infect Dis 2020; 15: 1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184. Brown GD, Denning DW, Levitz SM. Tackling human fungal infections. Science 2012; 336: 647. [DOI] [PubMed] [Google Scholar]
- 185. Driemeyer C, Falci DR, Oladele RO, et al. The current state of clinical mycology in Africa: a European Confederation of Medical Mycology and International Society for Human and Animal Mycology survey. Lancet Microbe 2022; 3: e464–e470. [DOI] [PubMed] [Google Scholar]
- 186. Lakoh S, Kamudumuli PS, Penney ROS, et al. Diagnostic capacity for invasive fungal infections in advanced HIV disease in Africa: a continent-wide survey. Lancet Infect Dis 2023; 23: 598–608. [DOI] [PubMed] [Google Scholar]
- 187. Badiane AS, Ramarozatovo LS, Doumbo SN, et al. Diagnostic capacity for cutaneous fungal diseases in the African continent. Int J Dermatol 2023; 62: 1131–1141. [DOI] [PubMed] [Google Scholar]
- 188. Mushi MF, Zaki SM, Penney ROS, et al. Diagnostic options for pulmonary fungal diseases in Africa. ERJ Open Res 2023; 9: 00397-02022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189. Jamalian A, Freeke J, Chowdhary A, et al. Fast and accurate identification of Candida auris by high resolution mass spectrometry. J Fungi 2023; 9: 267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190. Singh-Moodley A, Ismail H, Perovic O. Molecular diagnostics in South Africa and challenges in the establishment of a molecular laboratory in developing countries. J Infect Dev Ctries 2020; 14: 236–243. [DOI] [PubMed] [Google Scholar]
- 191. Osman A-H, Kotey FCN, Odoom A, et al. The potential of bacteriophage-antibiotic combination therapy in treating infections with multidrug-resistant bacteria. Antibiotics (Basel) 2023; 12: 1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192. Friedman DZP, Schwartz IS. Emerging fungal infections: new patients, new patterns, and new pathogens. J Fungi (Basel) 2019; 5: 67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193. WHO Model Lists of Essential Medicines, https://www.who.int/groups/expert-committee-on-selection-and-use-of-essential-medicines/essential-medicines-lists (accessed 16 December 2023).
- 194. Chassot F, Venturini TP, Piasentin FB, et al. Activity of antifungal agents alone and in combination against echinocandin-susceptible and -resistant Candida parapsilosis strains. Rev Iberoam Micol 2019; 36: 44–47. [DOI] [PubMed] [Google Scholar]
- 195. Stone NR, Rhodes J, Fisher MC, et al. Dynamic ploidy changes drive fluconazole resistance in human cryptococcal meningitis. J Clin Invest 2019; 129: 999–1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196. Nussbaum JC, Jackson A, Namarika D, et al. Combination flucytosine and high-dose fluconazole compared with fluconazole monotherapy for the treatment of cryptococcal meningitis: a randomized trial in Malawi. Clin Infect Dis 2010; 50: 338–344. [DOI] [PMC free article] [PubMed] [Google Scholar]