Abstract
Brain cancer pose significant life-threats by destructively invading normal brain tissues, causing dysneuria, disability and death, and its therapeutics is limited by underdosage and toxicity lying in conventional drug delivery that relied on passive delivery. The application of nanorobots-based drug delivery systems is an emerging field that holds great potential for brain cancer active targeting and controllable treatment. The ability of nanorobots to encapsulate, transport, and supply therapies directly to the lesion site through blood–brain barriers makes it possible to deliver drugs to hard-to-reach areas. In order to improve the efficiency of drug delivery and problems such as precision and sustained release, nanorobots are effectively realized by converting other forms of energy into propulsion and motion, which are considered as high-efficiency methods for drug delivery. In this article, we described recent advances in the treatment of brain cancer with nanorobots mainly from three aspects: firstly, the development history and characteristics of nanorobots are reviewed; secondly, recent research progress of nanorobots in brain cancer is comprehensively investigated, like the driving mode and mechanism of nanorobots are described; thirdly, the potential translation of nanorobotics for brain diseases is discussed and the challenges and opportunities for future research are outlined.
Supplementary Information
The online version contains supplementary material available at 10.1186/s11671-024-04131-4.
Keywords: Targeted drug delivery nanorobots, Active targeting, Controllable therapeutics, Precise oncology, Brain cancer
Introduction
Malignant brain tumor poses serious risks to health, causing cognitive impairment, seizures, sensory disturbances, paralysis, speech issues, personality changes, and vision impairments [1]. Primary brain tumors, particularly glioblastoma (GBM), are the most prevalent and devastating brain-related diseases, originating in the brain or spine [2], and it can infiltrate surrounding brain tissue, rendering the tumor's boundaries imperceptible during surgical removal. Timely medical attention is crucial for accurate diagnosis and treatment, while brain cancer presents significant therapeutic challenges due to the blood–brain barrier (BBB), limited drug penetration, and the need for precise targeting to avoid damage to healthy tissues. Traditional drug delivery methods often struggle to achieve effective concentrations at tumor sites while minimizing systemic toxicity. Regrettably, less than one percent of nanoparticles manage to accumulate in tumors [3]. Nanorobots, with their unique ability to autonomously navigate complex biological environments and actively target diseased tissues, offer a promising solution for brain cancer treatment. And nanorobots have been diversely applied to targeted drug delivery, surgery, sensing, and detoxification [4, 5], such as mediating drugs pass through the BBB, driven by the special microenvironment of brain cancer like high oxidative stress, hypoxia, and angiogenesis [6]. Additionally, the side effects and toxicity associated with chemotherapy drugs can be substantially reduced through the use of nanorobots [7]. Nanorobots offer a versatile approach to targeted therapy, as they can be precisely delivered to the desired site via local injection, intranasal application, or systemic administration [8].
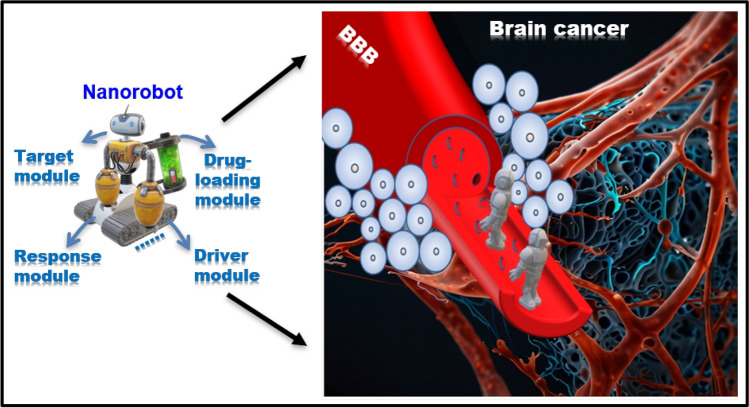
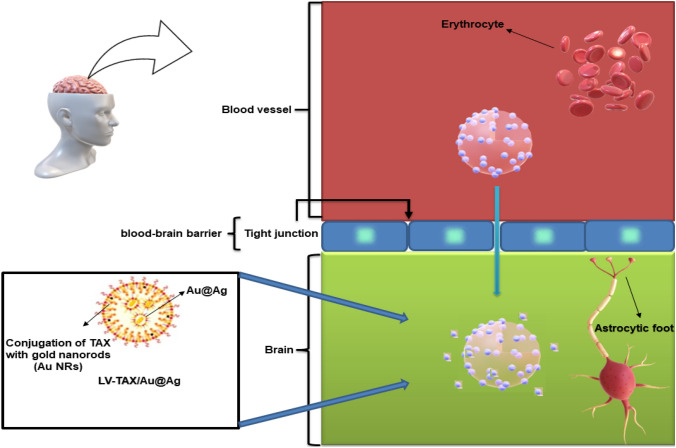
This review focuses on the emerging role of nanorobots in mediating drug delivery for brain cancer, highlighting their potential in active targeting, enhanced BBB penetration, and controllable therapeutics. By leveraging advanced navigation, real-time responsiveness, and localized drug release mechanisms, nanorobots are poised to revolutionize brain cancer therapy, offering new pathways to more effective and safer treatments. The BBB plays a significant role in maintaining the homeostasis and integrity of the brain. It acts as a selectively permeable barrier, allowing only certain molecules to pass through while blocking others. This protective mechanism is crucial for maintaining the brain's delicate environment and preventing harmful substances from entering. However, this same barrier presents challenges when it comes to treating various brain diseases [9]. BBB is a complex structure that prevents most macromolecules, including peptides, recombinant proteins, monoclonal antibodies, drugs based on RNA interference technology, and more than 98% of small molecule drugs from crossing it to reach the brain cancer site for cancer cell ablation. This makes it difficult to treat brain tumors effectively [10]. Nanotechnology's approach has been shown to enhance the delivery of therapeutic agents to brain by improving targeting, crossing the BBB, controlling release, and reducing toxic side effects by reducing the amount of anticancer drugs [11]. To address the limitations of clinical surgery in treating brain diseases, nanoparticles smaller than 200 nm can be engineered as nanorobots through various strategies to enhance their ability to cross the BBB. The structures of different nanocarriers vary, and so does their mode of transformation across the BBB. Drugs can be delivered to brain cancer via passive transport, carrier-mediated transport, receptor-mediated transport, and adsorption-mediated transport through the BBB. The nanorobots were illustrated in the Fig. 1 including mediating drug delivery to brain cancer by active targeting and controllable therapeutics.
Fig. 1.
Schematic illustration of nanorobots mediated drug delivery for brain cancer active targeting and controllable therapeutics
Intelligent nanorobots are employed to address the challenges of drug delivery to specific lesion sites in the complex brain biological environment. These nanorobots have the capability to reach the lesion area either autonomously or under guidance. Once there, they can accurately identify disease-specific markers on cells or pathogens, facilitating targeted treatment. This approach is referred to as "nanorobotic" therapy [12]. The basic components of nanorobots include sensors, propulsion, data storage, power supply, magnetic plates, and wireless communicators [7]. Nanorobots can provide minimally invasive, precisely targeted local treatments for targeted drug delivery, biopsies, hyperthermia, radiation therapy, stents, in vivo ablation, stents, sensing and labeling procedures [13]. Minimally invasive surgery performed by nanorobots has multiple benefits for patients like reducing recovery time, medical complications, risk of infection, and postoperative pain [14]. Nanorobots are generally designed to have sizes ranging from 1 nm (nm) to 200 nm. These extremely tiny robots are at the forefront of technological advancements, especially in the field of medicine, where they can be used for precise interventions at the cellular or even molecular level. Their small size allows them to navigate and operate within microscopic environments, such as inside living organisms, making them ideal for targeted drug delivery and other applications requiring high precision and control [15]. Nanorobots are characterized by their super-fast response times, a capability that is directly linked to the size of the sensor. This relationship arises because smaller sensors can detect species more rapidly, leading to enhanced performance in tasks such as targeted drug delivery and environmental monitoring. The diminutive dimensions of these devices not only contribute to their swiftness but also confer additional advantages. Their small size, lightweight nature, and high surface-to-volume ratio make them exceptionally suitable for achieving high signal amplification and trace detection, capabilities that were previously considered unachievable. To fulfill their intended functions effectively, nanorobots should be equipped with at least four (or more) basic modules. The first is a targeting module, which allows the device to identify and navigate toward specific cells or molecules within an organism or environment. The second is a drug loading module, responsible for carrying and releasing therapeutic agents precisely where they are needed. The third is a response module, which enables the device to react to changes in its environment, such as pH levels or the presence of certain biomarkers, triggering the release of drugs or other actions. Finally, the power module is crucial for energizing the device, ensuring it has the necessary energy to perform its tasks without relying on external power sources. These modular components work together seamlessly, allowing nanorobots to operate efficiently and effectively in a wide range of applications, revolutionizing fields such as medicine, environmental monitoring, and materials science [16, 17]. Materials used in the construction of nanorobots can be broadly categorized into three main classes, each with its unique set of properties and applications. The first category comprises molecular robots, which are built using biomolecules such as DNA and RNA. These materials are particularly attractive for their programmability and self-assembly capabilities, allowing for the creation of complex structures and functions inspired by biological systems. The second category is constituted by countless nanoparticle robots. These devices are typically fabricated from a wide array of nanoscale particles, including metallic, ceramic, and polymeric materials. Nanoparticles offer exceptional mechanical, electrical, and magnetic properties that can be harnessed for various tasks, including precision manipulation, sensing, and drug delivery. Their small size also provides a high surface area to volume ratio, making them highly efficient for surface-based reactions and interactions. The third category encompasses biological inorganic hybrid robots. These devices integrate both organic and inorganic components, leveraging the synergistic advantages of both worlds. For instance, they might combine the recognition capabilities of biomolecules with the robustness and functional diversity of synthetic materials. This hybrid approach opens up new possibilities for designing nanorobots that can operate in complex environments, respond to multiple stimuli, and perform sophisticated tasks with high specificity and efficiency. Hence, the materials used in nanorobot technology span a diverse range, offering designers and engineers a palette of options to tailor devices for specific applications. From the molecular precision of DNA and RNA constructs to the functional versatility of nanoparticles and the integrative potential of hybrid systems, these materials are paving the way for revolutionary advances in fields ranging from healthcare to environmental remediation.
Though the application of these nanorobots in biomedicine highlights their developmental potential, there's a notable scarcity of intelligent nanorobots designed for brain drug delivery, with even fewer instances of clinical use [18]. Given this scenario, there's an urgent desire to see advancements in this area. In this review, we present the cutting-edge advancements in medical nanorobotics for targeted drug delivery to treat brain disorders and offer a historical overview of nanorobot-mediated drug transport to cancerous regions. We also cover real-time imaging techniques of nanorobots within living organisms and address associated safety concerns. Initially, we delineate the hypothetical mechanisms and functionalities of nanorobots. Subsequently, we explore the prospective locations within a living body where these nanorobots could operate and examine their propulsion and control systems. Lastly, we contemplate on the prospective applications that are on the horizon in the near future.
The development of targeted drug delivery nanorobots
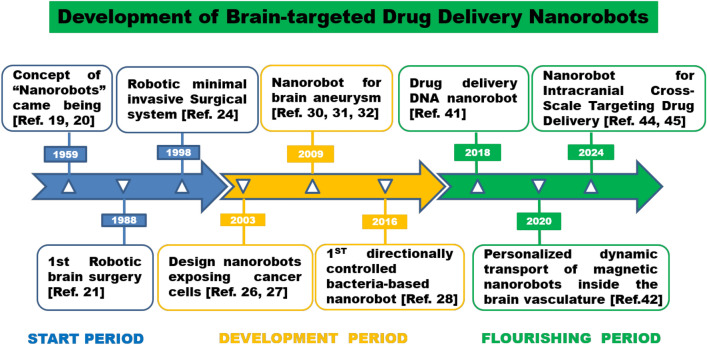
In recent years, the advancement of nanotechnology has opened up new frontiers in the field of medicine, particularly in drug delivery systems. Targeted drug delivery nanorobots represent a significant leap forward in addressing the limitations of traditional drug therapies. These nanorobots are designed to deliver therapeutic agents directly to specific cells or tissues, reducing systemic side effects and improving treatment efficacy. The early origins of the concept of nanomedicine stem from a visionary idea by Nobel physicist Richard that tiny nanorobots and related machines could be designed, manufactured, and introduced into the human body to carry out cellular repair at the molecular level [19]. The prototype concept of a nanorobot appeared in 1959, when Richard Feynman first proposed the concept of a miniaturized machine [20]. In 1988, the Kwoh team first applied a robot to brain surgery with a robotic CT scanner properly connected, and once a target was identified on a CT image, a simple command allowed the robot to move to a position so that the end-effector probe was directed toward the target [21]. In 1990, British physiologists and neurosurgeons Horsley and Clarke first proposed a completely fixed unit for the head of an animal [22], it provides a new idea for the treatment of brain diseases by intelligent robots in the future. In 1996, the first magnetic stereotactic system to perform magnetic micromotion was born [23]. A robotic minimally invasive Surgical system––Intuitive Surgical's Da Vinci Surgical System success, it represents the opening of the era of intelligent robot application in clinical surgery [24].
In 2003, as illustrated in Fig. 3A, Shlomi Dolev developed a carbon nanotube-polymer hybrid nanomaterials-based nanorobots as drug delivery system [25]. In the same year, Cavalcanti's team used computer simulations to design a dynamic collision detection nanosystem based on the motion control of autonomous nanorobots using neural networks [26]. And this is the first directionally controlled bacteria-based nanorobot (consisting of polymer microbeads attached to magnetotactic bacteria) [27]. Traditional craniotomy in 2005 involves drilling holes into the skull. However, in order to keep the skull intact, nanorobots are inserted into the lumbar spine to intervene in the brain, and this principle of percutaneous intraperitoneal navigation is used in brain surgery [28]. On March 24, 2007, a nanoscale 25-dimensional shape created using DNA origami was demonstrated [29], it provides a new idea for the future DNA nanorobots. In 2008, it was first proposed to combine magnetism with a usable airborne sensory device for nanorobots [30]. In 2009, Cavalcanti team [31] proposed that nanorobots can help early detection of cerebral aneurysms, and computer simulation is used to simulate the conduction in vivo. The sensors of nanorobots are activated due to the overexpression of proteins, so as to perform intracranial prognostic treatment of cerebrovascular diseases. In 2011, Deng Zhidong's team proposed the SSVEP asynchronous humanoid robot control system brain-computer interface to control humanoid robots through human brain electrical signals [32]. In 2012, Gao et al. [33] used magnetically driven nanorobots to achieve the first example of targeted delivery of drug-carrying magnetic polymer particles. In 2013, a platform and related approach called a magnetic nanorobot system were first described and validated, which is capable of controlling displacement and enabling the accumulation of magnetic nanorobots in deep physiological targets such as tumors [34]. In the same year, Hamdi's team [35] proposed that superparamagnetic nanorobot capsules could pass through the BBB using magnetic forces compatible with medical constraints. In 2014, Loscri et al. [36] proposes a special nanorobot structure called "nanobee", which uses a grid of cells in the spinal cortex to simulate the space in which nanorobots move and act. The structure communicates with the vibrating sound signals of the nanorobots, allowing decentralized control to treat endogenous diseases of the brain. And the development of brain-targeted drug delivery nanorobots were illustrated in Fig. 2.
Fig. 3.
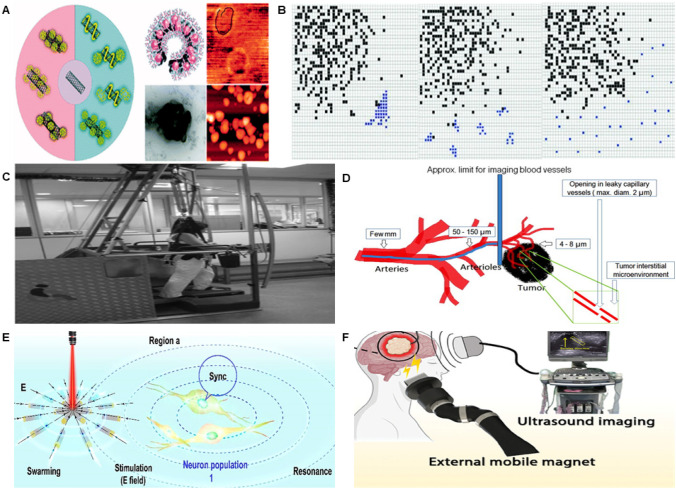
All kinds of nanorobots developed based on different materials and driving mode. A Carbon nanotube-polymer hybrid nanomaterials as biomacromolecules for anticancer drug delivery systems. Reproduced under the terms of the Creative Commons CC BY license [45]. Copyright © 2024, Informa UK Limited. B Aggregation, dispersion and isolation of nanorobots in a grid of cells in the spinal cortex (black for nanorobots, blue for cancer cells).Reproduced with permission from ref [36]. Copyright © 2014, Institute for Computer Sciences, Social Informatics and Telecommunications Engineering. C Eight-week intensive rehabilitation, including robotic and manual gait training, was well tolerated by patients with early stroke and was associated with significant increases in function. Patients with moderate gait dysfunction showed the strongest improvement after robot training. Reproduced under the terms of the Creative Commons CC BY license [46].Copyright © 2012, Conesa et al.; licensee BioMed Central Ltd. D A simplified representation of the vascular network that must pass through to reach the tumor under closed loop navigation control. Reproduced with permission from ref [27]. Copyright ©Rights managed by AIP Publishing. E The local neuronal population 1 is activated rhythmically, and the resonant response between the synchronous periodic ripple cell populations is observed within the neurons without the need for direct synaptic contact. Reproduced with permission from ref [43]. Copyright © 2023, American Chemical Society. F Schematic diagram of a robotic system for intracranial cross-scale targeted drug delivery. Reproduced with permission from ref [47]. Copyright © 2023, Wiley‐VCH GmbH
Fig. 2.
The development of brain-targeted drug delivery nanorobots
As shown in Fig. 3B, each nanorobot is able to recognize cancer cells, announce the presence of brain cancer to other nanorobots, and destroy it by emitting an audible signal. In 2015, as illustrated in Fig. 3C, nanorobot-assisted training reduced upper limb impairment in stroke and traumatic brain injury patients [37]. In the same year, Amir's team [38] built a DNA-origami nanorobot that can be covalently linked with functionalized iron oxide nanoparticles that detect brain damage with electrical signals to release its fragmented antibody payload. In 2016, as shown in Fig. 3D, Martel et al. [27] proposed that swimming microorganisms acting as nanorobots, summarize a simplified representation of the vascular network that must pass through to reach the tumor. In 2018, Li et al. [39] developed magnetic nanorobots for carrying and delivering target cells that can use external magnetic fields to direct nanoparticles to selected brain regions. In the same year, Bradley et al. [40] discovered the ability of DNA-Th nanorobots to target tumor blood vessels in vivo. After intravenous injection, TH-nanorobots gradually accumulated in in-situ MDA-MB-231 breast cancer tumors in mice, transporting and delivering protease payloads to tumor-associated blood vessels, triggering a cascade of clotting, vascular occlusion, and necrosis. In 2020, Chen et al. [41] proposed that magnetic nanorobots could achieve personalized dynamic transport and deliver drugs to the brain of mice in the cerebral vascularization system to treat brain tumors. Nanorobots can precisely manipulate and deliver drugs, and even swim in cerebrospinal fluid to treat brain diseases or repair cells, a key step toward controlled drug delivery for non-invasive brain therapy that has great potential for treating diseases such as brain tumors, stroke and Parkinson's disease. With the continuous development of technology, a brain-driven robotic arm system using a non-invasive hybrid brain-computer interface and shared control strategy is proposed in 2021 [42], the nanorobot's assistance to the human body is becoming more and more perfect, and researchers also pay attention to the research of the treatment direction of brain diseases by nanorobots in vivo. In 2023, as illustrated in Fig. 3E, Chen et al. [43] described how black (B)-TiO2@N/Au nanorobots induced periodic bee colony oscillation light in electrophoretically programmed near-infrared by photocatalytic pure water splitting, resulting in periodic chemical electric field oscillation, field oscillation of synchronous neurons and neural transmission to further trigger resonant oscillation contact of non-synaptic neuron groups. As the technology continues to develop, more new types of nanorobots will be developed. As shown in Fig. 3F, Wu et al. [44] proposed a dual-response nanorobot system for intracranial cross-scale targeted drug delivery system, which provides a novel intracranial local treatment strategy and is a key milestone in the development of glioblastoma treatment platform.
Categorization, design and construction of nanorobots
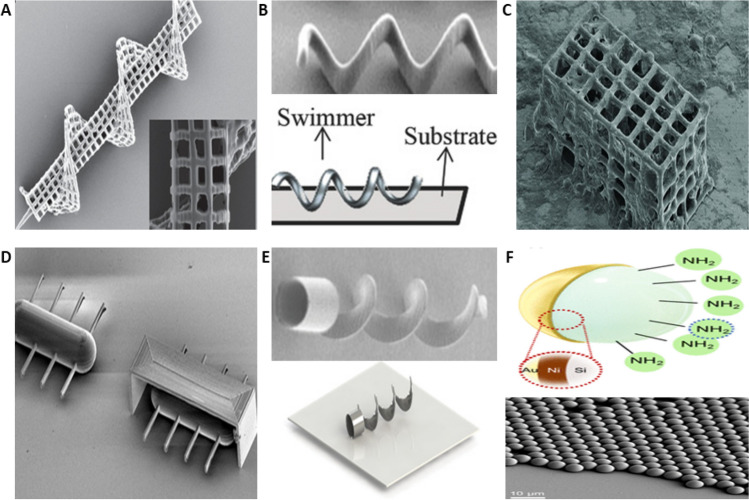
The categorization of general nanorobots based on their shape is a fundamental aspect of their design and functionality. As shown in Fig. 4, these nanorobots can adopt an array of forms, each suited for specific tasks and environments. One such form is the needle shape [48], which is particularly advantageous for penetrating tissues or materials with minimal damage, making it ideal for medical applications like drug delivery or biopsy. Screw-type nanorobots [49], on the other hand, are designed to mimic the motion of screws, allowing them to navigate through viscous environments or tight spaces by rotating their bodies. This shape is beneficial in scenarios where linear movement is impeded, such as in the human body or inside machinery. Bracket-type nanorobots [50] are characterized by their structural framework, which provides stability and support for attachments or payloads. This design is useful for carrying sensors or other devices into hard-to-reach areas, maintaining positional accuracy despite external forces. Ciliary-type nanorobots [51] take inspiration from the cilia found in living organisms, using hair-like extensions to generate movement. This form is well-suited for fluidic environments, where the cilia can propel the robot or manipulate particles within the fluid. Capsule-type nanorobots are enclosed structures that can protect their contents from external conditions, making them ideal for delivering sensitive substances or for use in harsh environments [52]. Their rounded shape also minimizes resistance as they move through fluids or soft tissues. Nickel-based sphere-Janus nanorobots [53] combine the symmetrical properties of a sphere with the asymmetric functionalization of a Janus particle. This results in directional movement and control, enabling precise manipulation and targeting. Nanorods and microcoils are elongated structures that offer unique magnetic and mechanical properties. Nanorods can be used for targeted heating or sensing, while microcoils can generate localized magnetic fields for manipulation or power transmission. Tubular structures are hollow and cylindrical, providing a cavity for storage or transport of substances. They can also be used as conduits for fluid flow or as components in more complex robotic systems [54]. Therefore, the variety of shapes available for general nanorobots allows for specialized designs that cater to different operational requirements and environmental challenges. From the precision of needle tips to the robustness of tubular frames, each form brings its own set of advantages to the field of nanorobotics.
Fig. 4.
Various shapes of different types of nanorobots. A Needle type. Adapted with permission. [48] Copyright 2020 WILEY‐VCH Verlag GmbH. B Screw-type. Adapted with permission. [49] Copyright 2015 WILEY‐VCH Verlag GmbH. C Bracket type. Adapted with permission. [50] Copyright 2013 WILEY‐VCH Verlag GmbH. D Ciliary type. Reproduced under the terms of the Creative Commons CC BY license. [51] Copyright 2020, the Authors, published by Scientific Reports. E Capsule type. Adapted with permission [52].Copyright 2020 WILEY‐VCH Verlag GmbH. F Nickel-based spherical Janus. Reproduced under the terms of the CC BY-NC 4.0 license. [53] Copyright 2022, the Authors, published by SCIENCE ADVANCES
The design and construction of nanorobots involve a variety of synthetic strategies, which can generally be categorized into physical vapor deposition (PVD), template electrodeposition, and self-assembly methods [55]. For instance, magnetic nanorobots are equipped with flexible nanowire motors that are fabricated using materials such as Au/Ni/Au/Pt-CNT. These motors enable directional motion along predetermined paths within polymer microchannel networks [56]. The fabrication of these motors typically involves the template electrodeposition method, where metal layers are deposited to generate magnetic properties and enhance biocompatibility [57]. Other methods for nanorobot fabrication include thermal decomposition, hydrothermal synthesis, and the polyol method [58]. In the context of biomimetic medical nanorobots, manufacturing techniques primarily encompass 3D printing, microforming, and template-assisted layer-by-layer assembly processes [59]. The power module and response module of nanorobots enable them to perform autonomous migration, thereby facilitating the controlled distribution of nanoparticles to challenging areas. These nanorobots are equipped with a set of application-specific integrated circuit (ASIC) modules that address functional requirements and offer asynchronous interfaces for antennas, sensors, and logic nanoprocessors [60]. Nanorobots possess the ability to execute intricate sequences of operations, which are often supervised by clinicians or can be directly and remotely operated. Owing to the low Reynolds number characteristic of fluids such as blood and mucus, coupled with the effects of Brownian motion, specialized driving methods are necessary to induce movement. These methods ensure effective motion at the nanoscale level. The propulsion techniques for nanorobots can be categorized into external physical field drives, including optical, magnetic, electric, ultrasonic, and bubble drives; chemical fuel drives like hydrogen peroxide and enzyme drives [61, 62], as well as self-decomposition mechanisms, microbial drives and hybrid drives that combine these modes to achieve optimal functionality. Nanorobots made with magnetic materials respond well to external magnetic fields, offering high controllability in fluidic environments like blood or tissue. Magnetic fields can penetrate deeply into biological tissues, making this method effective for in vivo applications, particularly in areas like the brain. However, precise navigation requires sophisticated equipment, and controlling multiple robots simultaneously can be challenging. Optical techniques, often using lasers, provide high precision and localized control. Light-responsive nanomaterials or structures enable nanorobots to move when exposed to specific wavelengths. This method is ideal for highly targeted therapies, but it is limited by light's shallow penetration depth in biological tissues, which restricts its use to surface-level treatments or transparent tissues. Chemical propulsion typically involves catalyzed reactions, such as the decomposition of hydrogen peroxide (H₂O₂) by catalytic materials on the nanorobot’s surface. This generates a concentration gradient that drives movement. Chemical propulsion can function autonomously and efficiently, especially in fluid environments. However, its reliance on chemical fuel raises concerns about biocompatibility, toxicity, and the need for continuous fuel supply. Utilizing biological entities such as sperm, bacteria, or algae offers excellent biocompatibility and energy efficiency. These organisms can naturally navigate and target specific tissues, making them promising for drug delivery. However, their integration with synthetic nanomaterials presents challenges in control, scalability, and immune response management. In comparison, magnetic and biological propulsion tend to perform better in complex, viscous environments, while optical propulsion offers precision but is more suitable for superficial applications. Magnetic methods provide deep tissue control, optical methods allow precise spatial manipulation, chemical propulsion is autonomous, and biological methods offer high energy efficiency and natural targeting. Magnetic propulsion requires complex equipment, optical methods face tissue penetration limits, chemical methods risk toxicity, and biological propulsion faces challenges in control and immune response. Understanding these distinctions is critical for selecting the appropriate propulsion method based on the nanorobot's intended application, environment, and therapeutic goals.
Transformation and application of nanorobots for targeted drug delivery
Nanorobots have been transformed and applied to targeted drug delivery based on the above features [63]. A diverse range of medical nanorobots has been proposed and designed, including an artificial mechanical phagocytic nanorobot specifically designed to eradicate microbial pathogens present in human blood through digestion and electrical discharge [64]. In the field of nanodentistry, overall oral health is maintained using nanomaterials, encompassing tissue engineering and the eventual deployment of dental nanorobots [65]. One of the pioneering examples of biomimetic nanorobots is the red blood cell (RBC) sponge integrated with a state-of-the-art fuel-free man-made nanomotor [66]. This construct is composed of magnetic spiral nanomotors cloaked with invisible human platelet plasma membranes, enabling it to blend with adult platelets and exhibit effective propulsion within the bloodstream over extended periods [67]. Another notable development is the cell membrane functionalized magnesium-based Janus micromotor nanorobot, which is powered by water and emulates the natural movement of cells [68]. It demonstrates efficient and guided propulsion in water without any external fuel, as well as in biological media rich in albumin, without causing evident biological contamination, thereby mimicking the motion of naturally moving cells. Hydrodynamic propulsion further enhances detoxification efficiency by augmenting the interaction between Janus motors and toxins that mimic cellular behavior [69]. Acid-powered zinc nanorockets have led to the creation of a new category of nanorocket pellets that can be administered in the stomach [70]. Nanorobots are capable of eliminating fatty buildups from the interior surfaces of blood vessels, and one method is through ultrasonic ablation, where these tiny machines use resonant mechanical structures to produce ultrasonic pressure waves to obliterate substances like kidney stones. Fuel-free nanomotors allow for the transport of drugs by flexible nanorobots directly to HeLa cancer cells via magnetic propulsion [33]. The existence of these nanorobots underscores the viability of targeted drug delivery.
The energy source for nanorobots can be derived from either chemical fuels (natural or synthetic) or physical sources [71]. For instance, a magnetic robot's motion is regulated by an external magnetic field, and its movement is influenced by factors such as magnetization, friction, and magnetic strength. However, the use of chemical drives in nanorobots, which rely on fuels like high concentrations of hydrogen peroxide that are harmful to the human body, poses limitations for their application within biological sy artificial intelligence stems [72]. This has led to a preference for mimicking natural bionic fuel-free nanomotors [73], or employing biocompatible chemical fuels such as glucose, urea, water, collagen, and endogenous hydrogen peroxide. In order to emulate the functions of natural motors more effectively and expand their potential in diverse and robust biomedical and sensing applications [74], biomolecules including enzymes, antibodies, nucleic acids, lectins, and aptamers have been utilized to functionalize nanomotors. These enhancements enable them to perform complex tasks ranging from biological isolation to targeted drug delivery.
The drug loading module and targeting module of nanorobots are crucial components for their potential application in active drug delivery, which is considered one of the most widespread and promising scenarios for these nanorobots, as highlighted in reference [75]. The process of drug delivery by nanorobots can be likened to a GPS-equipped car, where the drug is targeted to reach the tumor cell with precision. Active targeting nanoparticles play a significant role in this process. These nanoparticles are linked to a multitude of targeting ligands that are specifically designed based on the differences in gene expression between target cells and normal cells. These ligands encompass a wide range of molecules, including antibodies (such as EGFR, among others), antibody fragments, peptides (like transferrin, among others), small molecules (such as folate, among others), and various other different receptor ligands. The efficacy of this targeted drug delivery system is enhanced by the ability of nanoscale structures to penetrate tissue systems. This penetration facilitates the easy uptake of drugs by cells, allows for efficient drug delivery, and ensures that the drugs act at the intended target location, as mentioned in reference [76]. This precise targeting mechanism not only maximizes the therapeutic effect but also minimizes the adverse effects on healthy cells, making nanorobot-based drug delivery a promising avenue for future medical treatments.
Nanorobots employ two primary methods for drug delivery: passive delivery, where the drug is incorporated into the inner cavity of the structure through hydrophobic action, and self-delivery, in which the drug intended for release is directly coupled to the carrier nanostructured material. Medical nanorobots, with dimensions ranging from 1 to 2 microns, have the capacity to carry drug payloads of up to 1 cubic meter. These payloads are stored in onboard tanks that utilize molecular sorting pumps for mechanical unloading. Drug delivery platforms often feature helical and tubular lipid structures. There are three types of neuro-nanorobots designed to cross the BBB, enter the brain parenchyma, and even enter a single human brain cell: the endoneural robot, glial robot, and synaptic robot, as discussed in reference [77]. The components of nanorobots used in tumorigenic drug delivery systems are based on two main criteria: nanorobot navigation and attachment to targeted cancer cells. These nanorobots must navigate through the bloodstream without causing damage to other cells and avoid collisions. Nanocarriers serve as drug delivery systems capable of transporting macromolecules or cells to targeted sites. They possess the capability to overcome traditional obstacles presented by the glioma microenvironment, such as the BBB. Some nanoparticles in the loaded part can be designed to encapsulate antigens and adjuvants, offering protection not only to the adjuvant from degradation but also shielding the body from the systemic toxicity of the adjuvant. The benefits of these loaded parts of nanomaterials are significant, holding potential applications in drug delivery and nanoscale patterning. Catalytic nanorobots require precise control over their direction and speed [78]. The most common method to direct magnetic nanoscale motors involves the use of an external magnetic field [79]. Nanorobots controlled by external magnetic fields are considered safe, exhibiting minimal deformation or decay inside the body, as stated in reference. After completing their task, nanorobots are ultimately removed from the body through endocytosis [80, 81].
Nanorobots-mediated drug delivery across the blood–brain barrier
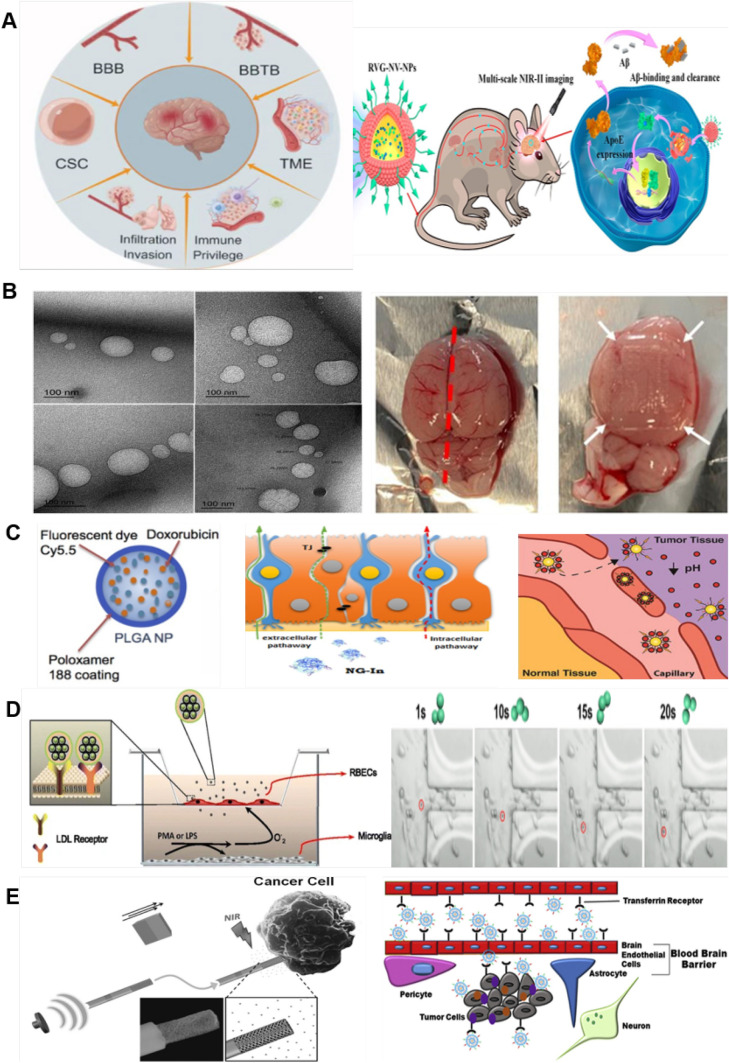
As one of the plates of nanorobots, nanocarriers that can deliver drugs in vivo have formed a complete system platform and are expected to be developed into nanorobots in newer iterations. In the treatment of brain diseases, there are a variety of ways to increase BBB transport, such as osmotic shock, ultrasound and the use of CPP, magnetic gradients can also significantly increase the transport of magnetic nanoparticles through the sieve plate into the olfactory bulb of the brain, there's also focused ultrasound [82]. The majority of nanomaterials that can cross BBB are polymers (PLBA, PLGA, and PLA) and gold nanoparticles, liposomes, micelles, dendrimers, exosomes, and nanoantibodies, and nanorobots have proven to be the most promising targeted drug carriers [83]. As illustrated in Fig. 5, in addition to carrying the chemotherapy drug doxorubicin as an effective drug delivery system for glioblastoma, it can also be functionalized and modified by β-amyloid specific peptides as a photothermal absorbent to treat Alzheimer's disease [84], while its ability to cross the BBB is affected by insulin encapsulation [85]. Targeted drug delivery for brain diseases often requires crossing the BBB. Nanorobot drug delivery mechanisms, such as those using gold nanoparticles, show promise in this area. For instance, Huang et al. [86] developed a traceable CNS delivery system that used neural stem cell (NSC) membranes to enhance BBB crossover and target nerve cells, offering therapeutic potential for Alzheimer’s disease (AD) as illustrated in Fig. 6A(ii). Law et al. [87] demonstrated the use of CEST MRI to noninvasively detect liposome delivery from the nose to the brain, a technique valuable for drug delivery studies. As shown in Fig. 6, Sabry et al. [88] used solid lipid nanoparticles (SLN) to deliver valsartan to the brain, mitigating stroke effects. A microneedle patch for targeted delivery of cannabidiol (CBD) to brain tumor sites was also developed, showing safety and efficacy in preclinical glioblastoma models [89]. Nanorobots have successfully delivered doxorubicin (DOX) to glioblastoma cells [90]. For example, Li et al. [91] developed a pH-sensitive dual-target carrier that transported DOX to glioma cells, enhancing BBB transportation and drug accumulation. These nanorobots are engineered to release drugs in response to the acidic environment typically found in tumor tissues. Tumors often have lower pH than healthy tissues due to their abnormal metabolism. pH-sensitive nanorobots take advantage of this by undergoing structural changes in the acidic microenvironment. For example, the pH-sensitive dual-target drug carrier G4-DOX-PEG-Tf-TAM utilizes a pH-sensitive acylhydrazone bond. Under acidic conditions, the bond breaks, allowing controlled drug release at the tumor site. This targeted release reduces non-specific toxicity during circulation, enhancing drug accumulation in glioma cells. Meng et al. [92] created ultrasound-responsive nanorobots that accumulated in tumors and released drugs triggered by ultrasound. Ultrasound-triggered nanorobots are activated by external ultrasound waves. The mechanical vibrations generated by ultrasound cause the nanorobot’s shell or matrix to destabilize and release its drug payload. For instance, ultrasound-responsive nanorobots, like the alkaline nanorobots developed by Meng et al., contain nanoparticles that vaporize into bubbles upon exposure to ultrasound. This mechanism improves drug release efficiency by targeting tumors with enhanced permeability and retention (EPR), allowing the drug to accumulate specifically in tumor tissues. Ultrasound also enhances deep tissue penetration, ensuring that drugs reach the tumor site more effectively, overcoming barriers posed by dense tumor tissues.
Fig. 5.
Schematic illustration of gold nanoplatform drug delivery across the blood–brain barrier
Fig. 6.
A (i) the challenge of treating brain cancer. Reproduced with permission from ref [117]. Copyright © 2024Informa UK Limited. (ii) Genetically engineered NSCs membrane coated nanoformulations and their application in drug delivery in the central nervous system. Reproduced with permission from ref [118]. Copyright © 2023, American Chemical Society. (iii)Load TEM images of Rhodamine's optimized SLN formulation. Reproduced with permission from ref [119]. Copyright © 2024Informa UK Limited. (iv) Release CBD NPs in vitro in the rat brain. Reproduced with permission from ref [120]. © 2022 Elsevier B.V. All rights reserved. Mechanism of drug delivery by different nanocarriers B (i) Effective delivery of adriamycin to PLGA structures in human glioma cells. Reproduced with permission from ref. [90] Copyright 2017 Elsevier B.V. All rights reserved. (ii) Different route schemes for nanobots to administer drugs from the nose to the brain in mice. Reproduced with permission from ref [121]. © 2019 Elsevier B.V. All rights reserved. (iii) Schematic diagram of the therapeutic and diagnostic efficacy of Au NP-mediated brain tumors. Reproduced with permission from ref. [96] Copyright 2014 WILEY‐VCH Verlag GmbH & Co. KGaA, Weinheim. (iv) Silicon dioxide nanoparticles carry RSV across BBB release schematic. (MSNPS are recognized and internalized by the LDL receptors of RBEC, and PMA or LPS stimulate and activate the inflammatory response of microglia, releasing reactive substances). Reproduced with permission from ref [122]. Copyright 2018, the Authors, published by Journal of Nanobiotechnology. (v)An experiment of targeted cancer cell therapy using magnetoactuated three-bead/azo /DOX nanorobots. Reproduced under the terms of the CC BY-NC 4.0 license [123]. Copyright 2021, the Authors, published by Scientific Reports (Sci Rep). (vi) Schematic illustration of ultrasound-driven motion of a nanoporous Au nanomotor loaded with drugs and triggering drug release around cancer cells. Reproduced with permission from ref [124]. © 2014 WILEY–VCH Verlag GmbH. (vii) A schematic showing the translocation of Dox and ERLO-loaded Tf-Pen liposomes across BBB and then endocytosis into glioblastoma tumor cells. Reproduced with permission from ref [125]. © 2019 Elsevier B.V. All rights reserved
Other studies have explored different drug delivery systems for brain diseases. For instance, FK506-modified liposomes targeted ischemic areas and protected neurons, while polymer-based nanogels delivered insulin via the nasal route for potential neurodegenerative disease treatment [93]. Cell-penetrating peptide-modified nanoparticles [95] and transcriptional TAT peptide-modified gold nanoparticles have been developed for effective BBB penetration and drug delivery for glioma treatment [96]. Further innovations include alginate-loaded nanoparticles for depression treatment [97], mesoporous silica nanorobots for cancer therapy [98], and nanoparticles targeting the LAT-1 receptor for brain cancer treatment [99]. Polymer-based nanocarriers and other advanced drug delivery platforms are being explored for efficient BBB crossing and localized brain targeting [100]. Su et al. [101] developed hollow carbon nitride nanospheres with a well-oriented copper (II) phthalocyanine layer and hyaluronic acid assembly. These nanospheres significantly enhance the SERS signal and photosynthetic simulation properties of heterogeneous structures. They have been used in circulating tumor cell (CTC) diagnosis in tumor xenotransplantation models and SERS image-guided cancer catalytic therapy. Shen's team [102] created a polylactic acid (PLA)-coated mesoporous silica nanoparticle for RSV delivery. This vector was coupled with a low-density LDLR ligand peptide to improve transendocytosis across the BBB. Another example of bifunctional liposomes was achieved by Singh's team. A three-bead near-infrared responsive nanorobot composed of biotin /NH coupled to streptavidin-coated beads via an azo group, selectively releasing DOX due to photoinduced bond breakage of the azobenzene unit, ultimately leading to lung cell apoptosis [103]. Barbara's team encapsulated curcumin in PLGA nanoparticles and modified them with g7 ligands to cross the BBB for Alzheimer's treatment. The same system was also used for targeted delivery of cisplatin and the antioxidant boldine for brain cancer therapy [104], it can also be used as a target specific delivery of cisplatin and the antioxidant boldine in vivo for brain cancer treatment [105]. Fonseca et al. [106] proposed amphiphilic methacrylate copolymer functionalized poly (ε-caprolactone) nanocapsules as a mucosal adhesion system to deliver olanzapine through intranasal administration. Jafarieh's team [107] prepared ropiniero hydrochloride-supported chitosan nanoparticles by ionic gel method. The polymer nanoparticles bypass the BBB by directly transporting the anti-Parkinson's disease drug ropiniero from nose to brain. Nanoporous gold-based nanorobots for light-triggered drug release. Gold nanowires have a strong absorption capacity in the near-infrared region, which allows near-infrared light to be efficiently converted into heat. This heat causes structural changes in the polymer that release drugs, such as DOX [108]. Bifunctional liposomes for effective co-delivery of anti-cancer chemotherapy drugs for the treatment of glioblastoma [109]. Song et al. [110] developed liposome RGD-TMZ/NLCs, which can effectively and selectively deliver temozolomide to U87MG cells for targeted delivery and treatment of glioma cells, thus effectively inhibiting tumor with high inhibitory efficacy. Lohan's team [111] developed a multi-walled carbon nanotube to adsorb berberine, an isoquinoline alkaloid, for the treatment of dementia and other neurological diseases, with the potential to reduce amyloid-induced Alzheimer's disease. Alvarez-Erviti's team delivered siRNA to mouse brains by targeting exosomes with systemic injections [112], the therapeutic potential of RNA drug delivery is realized. PLGA-loaded microspheres connected by micron particle magnetic Ni-Ag nanorobots (5–6 μm in length and 200 nm in diameter) could be used to carry Dox and deliver it to HeLa cancer cells, with a nanoparticle transport rate of more than 10 μm/s in vitro [113]. EC-K1 phagocytic maltodextrin (MD)-modified therapeutics via the bacteria-specific MD transporter pathway is inactivated by ultraviolet irradiation and dead bacteria can carry a therapeutic drug (e.g., indocyanine green (ICG)) and bypass BBB together after being injected intravenously into mice, demonstrating that the developed strategy is capable of treating bacterial meningitis and glioblastoma in mice. Based on these nanoparticles, it is possible to develop a part of targeted drug delivery nanorobots for brain cancer, such as exosomes [114], carbohydrate nanoparticles [115], mucosal adhesion nanoparticles (chitosan), mucopermeable nanoparticles (polymerized or coupled), ligand-linked nanoparticles, cell-penetrating peptides and transferrin nanoparticles [116] and other nanoformation for drug delivery.
Based on these advancements, several targeted drug delivery nanorobots can be developed for brain cancer treatment. Examples include exosomes, carbohydrate nanoparticles, chitosan-based mucosal adhesion nanoparticles, mucopermeable nanoparticles, ligand-linked nanoparticles, cell-penetrating peptides, and transferrin nanoparticles. Overall, the application of nanorobots in brain cancer treatment is progressing rapidly. And the use of nanorobots for mediating drug delivery in the treatment of brain cancer has been summarized and detailed in Table 1. This table provides an overview of the current state of research, highlighting the various types of nanorobots being developed and their respective mechanisms of action. The information presented includes the specific drugs that can be delivered, the targeted areas within the brain, and any notable successes or challenges encountered in the experimental and clinical trial phases. This summary serves as a comprehensive resource for researchers and medical professionals interested in advancing the application of nanorobots in combating brain cancer.
Table 1.
Nanorobots mediated drug delivery in brain cancer
| NPs-based Nanorobots | Drug delivery | Mode of active targeting | Site of action | References |
|---|---|---|---|---|
| RVG-MSP-FLAG | GAPDH siRNA | Intravenous targeted delivery | Neuro-2A | [112] (2011) |
| G4-DOX-PEG-Tf-TAM | DOX | Drugs are coupled by acylhydrazone bonds and transported by the BBB | C6 glioma cells | [91] (2012) |
| FK506-Liposome | FK506 | The drug is passively targeted and effectively delivered to the I/R region | PC12 cells | [93] (2013) |
| TAT-Au NP | DOX | Cross the BBB and deliver the BBB impermeable drug to the brain tumor tissue | U87 | [96] (2014) |
| VLF AG-NPs | VLF | NP is carried to the central nervous system via the olfactory neuroepithelial cells via the trigeminal nervous system and olfactory neural pathways | Brain | [97] (2014) |
| MMA-b-DMAEMA | Olanzapine | By nasal drip | Brain | [106] (2015) |
| RGD-TMZ/NLC | TMZ | Intravenous targeted delivery | U-87 MG | [110] (2016) |
| Cy5.5-PLGA | DOX | Passes through the BBB and accumulates into glial cells through endocytosis | U87 cells | [90] (2017) |
| BRB-MWCNT | BRB | Targeted drug delivery in BBB after intravenous injection | SH-SY5Y cells | [111] (2017) |
| PLGA-g7 | Curcumin | Delivery via BBB-targeted drugs | Hippocampal cells | [104] (2017) |
| NG-In | Insulin | After penetrating the nasal cavity, it can reach the brain via the trigeminal nerve and the olfactory pathway | Tritencephalon | [94] (2018) |
| CSV | SAX | Carrying drugs across the blood–brain barrier | Brain | [99] (2018) |
| PLGA NPs | IRI or MET | Injected through a stereotactic device | U-87 MG | [100] (2018) |
| PLA-MSNP-LDLR | RSV | Cross the blood–brain barrier | HAPI cells | [102] (2018) |
| DSPE-PEG(2000)-Tf | Dox and erlotinib | Translocation across BBB to glioblastoma tumor | U-87 MG | [109] (2019) |
| Tri-bead microrobots | DOX | Magnetically guided targeted chemo-photothermal therapy in a microchannel | H1299 cancer cells | [102] (2021) |
| Bionic HCNCH Nanoplatform | HA | Intravenous injection | HeLa cells | [100] (2022) |
| RVG-NV-NPs | BEX | Targeted drug delivery across the blood–brain barrier | NSC | [85](2023) |
| SLN | VAL | It's transported through the nasal cavity to the brain | Brain | [87] (2023) |
| MN | CBD | The patch is inserted into the brain parenchyma, and the drug is targeted and diffused | The hemisphers of the brain | [88](2023) |
| EC-K1 | ICG | Bypass the BBB and target delivery | Brain | [112] (2023) |
Driving mode of nanorobots and its application in the brain cancer
Bio-powered nanorobots
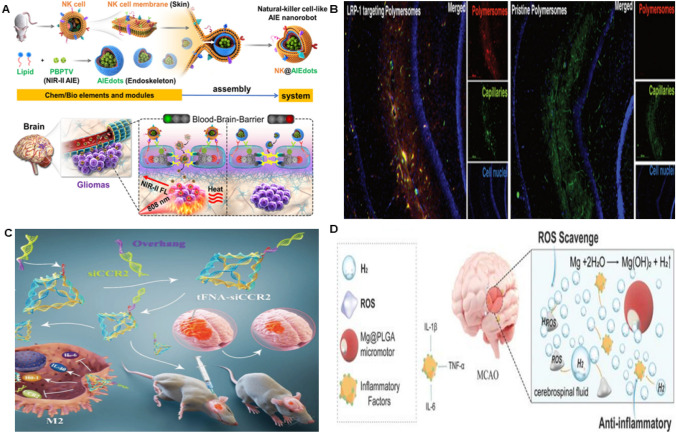
Bio-powered nanorobots have emerged as a promising tool for targeted drug delivery and imaging in various medical applications, including the treatment of brain cancer. One such example is the NK@AIEdots nanorobot, inspired by natural killer cells, which utilizes the unique phenomenon of aggregation-induced emission (AIE) to enhance imaging capabilities. This nanorobot is designed to cross the BBB and actively accumulate in glioblastoma cells within the complex brain stroma. Its ability to perform immune surveillance in the central nervous system and deliver drugs across the BBB makes it a potential tool for high-contrast cranial tumor imaging and photothermal therapy (PTT) [118], as shown in Fig. 7A. Another innovative approach is the DNA-based medical nanorobot developed by Joseph et al. These nanorobots can navigate autonomously within tissues and organs using active diffusion, guided by endogenous chemical gradients. They are synthesized as biocompatible nanoscale polymer vesicles, or polymeric objects, which serve as effective carriers. The nanorobots' propulsion relies on two driving forces: the asymmetry of the nanoscale polymer and the encapsulation of glucose oxidase or catalase for chemotactic movement. This technology demonstrates the potential for nanorobots to revolutionize targeted drug delivery and diagnostic imaging in brain cancer treatment [126], as illustrated in Fig. 7B. As shown in Fig. 7C, self-assembled DNA nanorobots based on Watson–Crick base pairing rules injected tFNA-siCCR2 into intraventricular tubes to treat intracranial hemorrhage. TFNA-siCCR2 can be used to regulate the polarization of microglia, inhibit the release of inflammatory mediators, and increase the expression of anti-inflammatory factors. Treatment with tFNA-siCCR2 can inhibit neuroinflammation [120], accelerate hematoma absorption and partially preserve motor nerve function, which has the potential of clinical transformation.
Fig. 7.
Biological and chemically-driven nanorobots for brain diseases. A NK cells simulate the preparation and assembly process of AIE nanoparticles and NK@AIEdots "smart" modulated TJ penetration of the BBB for brain tumor targeted lighting and inhibition. Reproduced with permission from ref. [9] Copyright 2020 American Chemical Society. B Immunofluorescence histology of polymerized rat hippocampal sections. Reproduced under the terms of the CC BY-NC 4.0 license [126]. Copyright 2017, the Authors, published by SCIENCE ADVANCES. C Diagram of DNA nanorobot treating mice. Adapted with permission. [120] Copyright 2021 WILEY‐VCH GmbH. D Mechanism of action and structure of HPMs for in vivo treatment of acute ischemic stroke. Adapted with permission. [121] Copyright 2021 WILEY‐VCH GmbH
Chemically driven nanorobots
Traditional drug delivery systems laid the foundation for tumor-targeted therapy but suffer from limited recognition range and low targeting efficiency. In contrast, nanorobots, with their ability to autonomously move and navigate, offer a promising new platform for more efficient and safer brain cancer treatment. By modifying the oxidative stress of gliomas to treat brain diseases, as illustrated in Fig. 7D, a hydrogen-powered magnesium nanorobot HPMs that can be used in the treatment of acute ischemic stroke in the brain. The robot can be accurately injected into the lateral ventricle of the middle cerebral artery occlusion by using the stereopositioning device. After the injection, the motion of HPMs is activated, and the magnesium particles in the magnesium (Mg) nanorobot react with water to locally produce hydrogen and actively transport H2 in the body. The speed of HPMs in artificial cerebrospinal fluid is up to 51.1 μm/s [121], which not only has the effect of motion propellant, but also can relieve oxidative stress and remove excessive ROS in addition to treating ischemic brain stroke. HPMs has excellent biocompatibility, and the remaining polylactic-glycolic acid copolymer (PLGA) can be gradually biodegraded after H2 release. For example, Li et al. introduced swarming responsive photonic nanorobots (RPNRs) that can navigate hard-to-reach biological environments and map local physicochemical conditions in real-time. The RPNRs, composed of magnetic Fe₃O₄ nanoparticles within a hydrogel shell, perform multiple functions such as magnetically-driven swarming, responsive structural color changes, and photothermal conversion. These capabilities allow them to map abnormal conditions (e.g., pH, temperature) and guide localized photothermal treatment [127], offering potential advancements in cancer and inflammatory disease therapies. This innovation paves the way for intelligent nanosensors and multifunctional nanotheranostics [128].
Nanorobots driven by physical fields
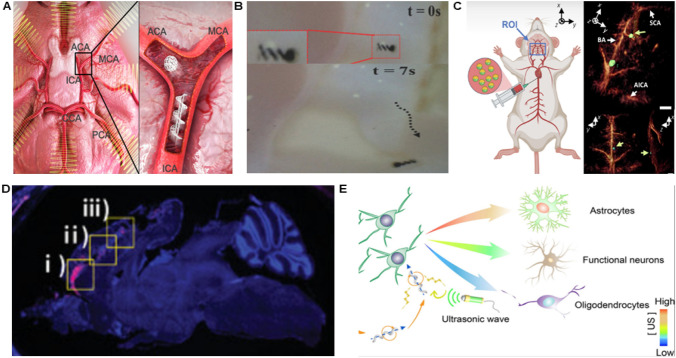
Transplanted neurons can be generated from cultured stem cells, and the adult brain generates new neurons from its own stem cells in response to damage [122]. According to this feature, Jeon et al. [123] designed with 3D microelectromechanical system technology to create a magnetized nanorobot that was attached, proliferated and differentiated by hippocampal neural stem cells, and was biocompatible by deposition of nickel and titanium layers. As illustrated in Fig. 8A. The nanorobot is injected into the internal carotid artery. Controlled by an external rotating magnetic field, the nanorobot can maneuver its direction to roll to the ventricle and can deliver therapeutic agents to the ventricles and blood vessels. As shown in Fig. 8B a magnetically controlled capsule nanorobot that exhibits a "pick and drop" (P&D) motion. The capsule nanorobot can transport suspended or attached cells, drugs or cells with minimal or incomplete exposure to the surrounding fluid, and safely transport particles in the closed volume of the capsule through P&D motion. The release of the lid from the capsule-type nanorobot transported olfactory receptor neuron cells to a target site on a rat brain slice, which offers potential for brain-targeted therapy [52]. Wrede et al. [53] designed a nickel-based spherical magnetic Janus nanorobot with a microcapillary size, which was composed of indocyanine green nano-liposome [liposome ICG (Lipo-ICG)] and nickel layer combined with gold (Au) coated silica particles with a diameter of about 5 to 10 μm. As illustrated in Fig. 8C.The average speed in the cerebral blood vessel can reach 7 mm/s, much higher than the blood flow speed. The real-time 3D tracking of the nanorobot circulating in the cerebral vasculature system is realized by using molecular imaging function and magnetic manipulation, this paves the way for the efficient and safe operation of cell-sized nanorobots in clinically relevant intravascular environments. Jeon et al. [124] proposed a magnetic-based stem cell nanorobot "Cellbot". Cellbot was developed by internalizing superparamagnetic iron oxide nanoparticles into human turbinate stem cells and using external magnetic fields to manipulate their orientation and position to target stem cells to the brain through intranasal channels [129]. Cellbot has good biocompatibility without any effect (including morphology, cell viability and neuronal differentiation) in human turbinate stem cells. As shown in Fig. 8D, cellular robots are capable of multiplying and differentiating into neurons, neuroprecursor cells and glial cells, and have the potential to effectively treat central nervous system diseases in a minimally invasive manner. Liu et al. [125] developed an intelligent robot using a minimally invasive micromotor with superparamagnetic Fe3O4 nanoparticles to functionalize biodegradable spirulina (S. platensis) as a framework, and piezoelectric BaTiO3 nanoparticles as a built-in functional unit. With a low-intensity rotating magnetic field, the magnetic nanoparticles facilitate the magnetic drive of the micromotor, and this integrated micromotor system can precisely navigate through biological fluids and achieve single neural stem cell targeting with an average maximum speed of 68.5 ± 11.5 μm/s. Stem cells are directed to differentiate into astrocytes, functional neurons (dopamine neurons, cholinergic neurons), and oligodendrocytes, so neural stem cells have the potential to combat various neurodegenerative diseases. As illustrated in Fig. 8E, the nanoplatform of the intelligent robot could be a controllable tool for treating neuronal regeneration. Felfoul et al. [30] synthesized nanorobot Mc1-lp, which proved that the magnetotactic bacterium Magnetococcus marinus strain MC-1 could be used to transport the nanoliposomes containing drug SN-38 to the anoxic region of tumors. In addition, nanorobots also offer promise for intravascular therapy. B Wang et al. [130] developed a retrievable magnetic colloidal microswarm composed of tPA-nbots for enhanced thrombolysis with low side effects and the ability to be retrieved via CMAFIS.
Fig. 8.
Magnetically powered nanorobots for brain cancer. A In vitro magnetic drive of a nanorobot in the brain. Reproduced with permission from ref [123].Copyright 2019, the Authors, published by SCIENCE ROBOTICS. B The capsule-shaped nanorobot releases the lid and tranships the ORN cells to a target point on the rat brain slice. Adapted with permission [52]. Copyright 2020 WILEY‐VCH Verlag GmbH. C Noninvasive photoacoustic tomography (OAT) and OAT imaging of a nanorobot in the cerebral vasculature of a mouse. Reproduced under the terms of the CC BY-NC 4.0 license. [53] Copyright 2022, the Authors, published by SCIENCE ADVANCES. D Intracellular robot delivery to the target brain region through intranasal administration and magnetic guidance. Adapted with permission. [124] Copyright 2021 WILEY‐VCH GmbH. E Magnetic power supply voltage SP@Fe3O4@BaTiO3 micromotion targeting neural stem cells schematic diagram. Reproduced with permission from ref. [125] Copyright 2020 American Chemical Society
One of the major challenges is ensuring that nanorobots are biocompatible. Materials used in nanorobots, such as metals, polymers, or nanoparticles, must not induce adverse immune responses or toxicity when introduced into the body. Ensuring that these materials are non-toxic and do not provoke inflammatory responses is crucial for their safe use in medical applications. The degradation products of nanorobots must also be non-toxic and safely eliminated from the body. Effective real-time tracking of nanorobots in clinical settings is challenging. While imaging technologies such as MRI, CT, or fluorescence microscopy can be used, they often require complex setups and can be costly. Moreover, the resolution of these imaging techniques might not always be sufficient to monitor nanorobots accurately in real-time. Developing reliable and minimally invasive tracking methods that can provide continuous feedback on the nanorobots' location and behavior is essential for their practical application. Scaling up the production of nanorobots from laboratory settings to clinical applications presents several challenges. Issues such as reproducibility of nanorobot production, cost of materials, and manufacturing techniques need to be addressed. Additionally, ensuring that nanorobots maintain their functionality and performance when produced in larger quantities is crucial for widespread use. Nanorobots driven by physical fields, such as magnetic or optical fields, face limitations related to the strength and precision of these fields. For instance, magnetic fields need to be strong enough to control nanorobots effectively but not so strong as to cause harm to surrounding tissues. Similarly, optical fields used for controlling light-responsive nanorobots must be precisely focused, which can be difficult in deep tissue areas. The biological environment within the human body is highly complex and variable. Nanorobots must navigate through diverse tissue types and physiological conditions, which can affect their performance. Ensuring that nanorobots can adapt to or overcome these variations without losing efficacy is a significant challenge. The development and deployment of nanorobots in medicine also involve regulatory and ethical considerations. Regulatory agencies require thorough testing to ensure safety and efficacy before approval for clinical use. Additionally, ethical concerns regarding privacy, consent, and long-term effects need to be addressed.
Hybrid driven nanorobots
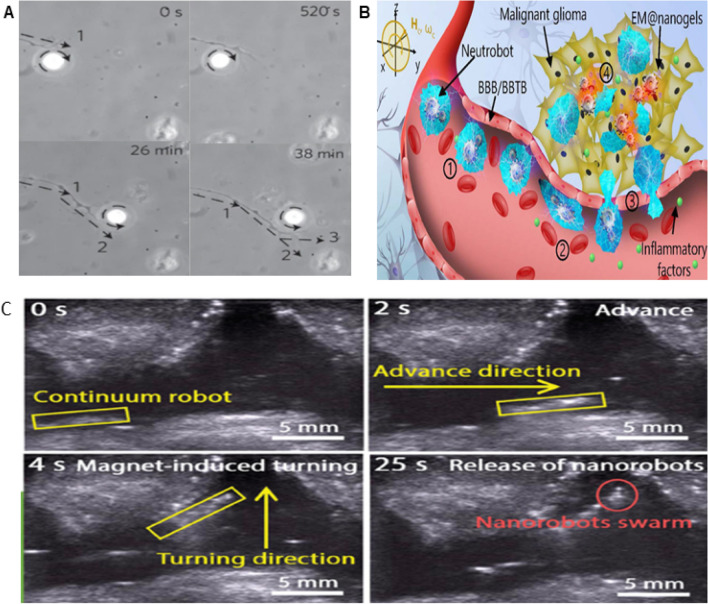
As illustrated in Fig. 9A, Wu et al. [131] developed an option-based system that uses a laser-driven rotating birefringent ball to precisely control the growth direction of a single axon (nerve fiber), which has great potential for the treatment of initial development, nerve repair and regeneration of the brain nervous system. Zhang et al. [132] developed a nanorobot ("neutrobot") with dual response mixing neutrophils. The robot is composed of a magnetic nanogel wrapped in the outer membrane of E. coli swallowed by NEs. As shown in Fig. 9B, when rotating the magnetic field, it can control its movement in the blood vessel, cross the blood–brain barrier along the positive chemotactic movement of the inflammatory factor gradient, and autonomously accumulate in the brain to deliver drugs to the malignant glioma in the body. Wu et al. [133] developed a dual-responsive nanorobot based marsupial robotic system, which integrated chemical and magnetic hybrid nanorobots with a miniature magnetic continuum robot, and the robot entered the cranial cavity through a minimally invasive channel in the skull. And deliver the nanorobots to the disease-causing area, and after bypassing the blood–brain barrier, the released nanorobots perform secondary targeting on a nanoscale. As illustrated in Fig. 9C, which reported a marsupial robot system that integrates chemical/magnetic hybrid nanorobots (child robots) with miniature magnetic continuum robots (mother robots) for intracranial cross-scale targeted drug delivery.
Fig. 9.
Light-driven and hybrid driven nanorobots for brain diseases. A The growth direction of axons can be changed by changing the position and rotation direction of aragonite particles. Reproduced with permission from ref. [131] Copyright 2011, Springer Nature Limited. B Schematic diagram of in vivo active therapy for dual response neutral robot. Reproduced with permission from ref [132]. Copyright 2021,the Authors,published by SCIENCE ADVANCE. C Intracranial drug delivery in GBM therapy provides a real-time in vivo imaging approach. Reproduced with permission from ref [133]. Copyright © 2023 Wiley‐VCH GmbH
The concept of nanorobots mediated controllable brain therapeutics has been thoroughly summarized in Table 2. This table provides an exhaustive overview of the latest advancements and research in the field of nanorobot-mediated controlled brain therapy, elucidating the various types of nanorobots currently being developed and evaluated for this purpose. It delineates the specific mechanisms through which these nanorobots can be controlled and directed to specific regions of the brain, encompassing the different drugs or therapeutic agents they can deliver. Furthermore, the table also highlights the potential efficacy of these treatments, any side effects or risks associated with them, and the stages of research or clinical trials they are currently in. This comprehensive summary aims to serve as a valuable resource for scientists, researchers, and medical practitioners interested in exploring and furthering the application of nanorobots in controllable brain therapeutics. However, the field of nanorobots currently also faces several challenges. Initially, surgeons had to rely on two-dimensional endoscopic images to navigate and operate robots within the surgical site, which were low in resolution [138]. And advancements have been made with the integration of MRI, CT, PET, and ultrasound technologies, leading to not only reduced health impact but also higher frame rates. Despite these improvements, achieving real-time image transmission over distances still poses problems. As robots are miniaturized to microscopic dimensions, fundamental physical challenges arise, particularly in electricity generation and storage, severely hampering the evolution of medical nanorobots. Additionally, the side effects associated with drug delivery systems are of significant concern. These are influenced by various factors including the size, shape, structural complexity, biocompatibility, degradation rate, chemical makeup, solubility, and propensity for aggregation of the nanomedical platforms [83]. The safety aspects of intelligent robots are not yet flawless. In the power module of nanorobots, the drive fuel propulsion micromotors typically incorporate additional components such as precious metals. The optimal approach would be to engineer these motors to be entirely biodegradable and capable of autonomous disassembly, similar to the Mg/Si Janus micromotor which propels using 5.0 M NaHCO3 and subsequently degrades completely [139]. However, the majority of micro and nanorobot actuators are still constructed from inorganic materials like SiO2, mesoporous silicon, and Fe3O4. These materials often exhibit poor system toxicity or lack biodegradability, rendering them unsuitable for biological systems [140]. Furthermore, the methodologies for characterizing intelligent nanomachines both in vivo and in vitro remain imperfect [141]. Addressing these issues represents significant future challenges, alongside ensuring the long-term durability of therapeutic effects, precise localization and navigation within the brain, and resolving ethical concerns.
Table 2.
Nanorobots Mediated Controllable Brain Therapeutics
| Intelligent robot | Driving mode | Mode of action | Treat the lesion | References |
|---|---|---|---|---|
| Spin the birefringent ball | Optical drive | Control the direction of growth of individual axons | Cranial nervous system | [131] (2012) |
| DNA polymer vesicles | Biological drive | Cross the BBB into the central nervous system for targeted therapy | Central nervous system | [134] (2017) |
| Capsule robot | Magnetic drive | P&D movement | Transporting cells to the brain | [135] (2018) |
| Hippocampal stem cells attached to microrobots | Magnetic drive | Targeted injection into the internal carotid artery | The drug is delivered to the ventricle | [123] (2019) |
| NK@AIEdots | Biological drive | Targeted accumulation across the blood–brain barrier into glioblastoma cells | Brain glioma | [136] (2020) |
| neutrobot | Hybrid drive | Target accumulation across the blood–brain barrier to glioblastoma cells | Brain glioma | [132] (2021) |
| SP@Fe3O4@BaTiO3 | Magnetic drive | Single neural stem cell targeting | Neurodegenerative disease | [125] (2021) |
| Cellbot | Magnetic drive | Target stem cell transport to the brain | Central nervous system disease | [124] (2021) |
| DNA nanorobot | Biological drive | Entering the intracranial with tFNA-siCCR2 targeted therapy | Intracranial hemorrhage | [120] (2021) |
| Janus magnetic microrobot | Magnetic drive | Real-time tracking in cerebral vasculature | Cerebral vasculature | [53] (2022) |
| HPMS | Chemical drive | Targeted injection of middle cerebral artery occlusion | Acute cerebral ischemia | [137] (2024) |
Conclusion and outlook
In conclusion, this review provides a comprehensive overview of recent advances in the treatment of brain cancer using nanorobots. It covers three main aspects: the development history and characteristics of nanorobots, recent research progress in brain cancer treatment with nanorobots, and the potential translation of nanorobotics for brain diseases. And the review highlights the promising potential of nanorobots in improving the treatment of brain cancer. The development of nanorobots has led to significant advancements in drug delivery and targeting, which can enhance the efficacy of cancer therapy while reducing side effects. The ability of nanorobots to navigate and target cancerous regions within the brain offers new therapeutic possibilities, supported by real-time imaging technologies that enhance their precision and control. This review also emphasizes the importance of further research and development to overcome challenges such as nanorobot design, biocompatibility, and safety concerns. One key challenge is enhancing the precision with which nanorobots deliver drugs to specific target sites, such as tumor cells, without affecting surrounding healthy tissues. Improvements in targeting mechanisms, such as incorporating multi-targeting capabilities or enhancing responsiveness to biochemical cues (e.g., pH, temperature), could address this. The long-term effects of nanorobot presence in the body, including the potential for nanomaterials to accumulate in tissues or organs, require thorough investigation. Studies focusing on the degradation and elimination of nanorobots after completing their function are needed. And one pressing safety concern is the potential for nanorobots to interact unintentionally with non-target cells or tissues, leading to off-target effects or toxicity. More sophisticated targeting and localization mechanisms are needed to mitigate these risks. Even if nanorobots are biocompatible, their movement through the body may still activate immune responses. The design should account for stealth characteristics, such as using biocompatible coatings that allow the nanorobots to evade detection by the immune system. Another critical area is the development of better real-time monitoring systems to track nanorobots’ behavior in the body. If malfunctions occur, such as unintended aggregation or migration to non-target areas, immediate corrective actions must be possible to avoid potential harm. Although safety concerns remain, ongoing research into the propulsion, control, and operational mechanisms of nanorobots shows promise for overcoming these challenges by emphasizing the following points:
(1) Driving modes and mechanisms
Magnetic and Ultrasound Propulsion: While these modes show promise for controlled and non-invasive navigation of nanorobots, further research is needed to optimize their use in complex biological environments. Studies should focus on improving the precision of magnetic field control for deeper tissue navigation and enhancing the sensitivity of ultrasound-responsive nanorobots to minimize potential tissue damage.
Chemical and Enzyme-Based Propulsion: These propulsion methods offer high biocompatibility and autonomous movement but often face limitations in energy efficiency and control. Future research should aim to enhance the stability and reactivity of chemical and enzyme-driven nanorobots to ensure sustained propulsion over longer durations, particularly in low-energy environments such as the bloodstream.
Hybrid Propulsion Systems: Combining different propulsion techniques (e.g., chemical with magnetic) could increase control and efficiency. Investigating hybrid systems could lead to more adaptable nanorobots capable of switching propulsion modes based on their environment.
(2) Energy Efficiency:
Energy Harvesting Mechanisms: Research should explore novel ways for nanorobots to harvest energy from their surroundings, such as utilizing body heat, biochemical reactions, or light for propulsion. This would extend the operational lifespan of nanorobots and reduce the need for external energy inputs.
Miniaturized Power Sources: Development of ultra-small, yet efficient power sources (such as nanoreactors or nanogenerators) that can sustain nanorobot functions over extended periods is an essential area for future research.
(3) Precision Targeting:
pH and Temperature Sensitivity: More work is needed to refine pH-sensitive and thermally responsive nanorobots, particularly for targeting tumor microenvironments. Fine-tuning their sensitivity to subtle changes in the physiological conditions of diseased tissues could enhance targeting specificity and reduce off-target effects.
Multi-Targeting Capabilities: Future nanorobots could be equipped with multi-targeting mechanisms that respond to multiple biomarkers or environmental cues. This would improve their ability to navigate complex biological environments and enhance treatment effectiveness.
(4) Scalability and Manufacturing:
Scalable Production Techniques: One significant bottleneck in translating nanorobots from the lab to clinical settings is the difficulty in scaling up production. Research into more cost-effective and reproducible manufacturing processes, such as advanced nanofabrication or 3D printing techniques, is necessary to make large-scale production feasible.
Standardization and Quality Control: There is also a need to develop standardized protocols for nanorobot manufacturing to ensure consistent quality and functionality across different batches, which will be essential for regulatory approval and widespread clinical use.
(5) Real-Time Tracking and Monitoring:
Advanced Imaging Techniques: Future research should also focus on improving real-time imaging and monitoring techniques, such as integrating nanorobots with contrast agents for MRI, PET, or optical imaging. This would enable better tracking of nanorobot location and activity in clinical settings, ensuring both efficacy and safety.
Non-Invasive Monitoring Systems: Research on non-invasive or minimally invasive methods to track nanorobot movement and function in real-time will be crucial, as current methods may not always be practical or safe for patients.
Looking forward, nanorobots have the potential to revolutionize brain tumor treatment by enabling more precise, targeted, and controlled therapeutic interventions, potentially improving patient outcomes and minimizing harmful side effects, and we suggest that future research should focus on developing more advanced nanorobots with improved driving modes and mechanisms for better control and precision in drug delivery. Additionally, collaboration between interdisciplinary fields such as robotics, materials science, and neuroscience is crucial for the successful translation of nanorobotics into clinical practice for treating brain diseases. Overall, the review provides valuable insights into the potential of nanorobots in revolutionizing brain cancer treatment and paves the way for future research and innovation in this field.
Appendix A. Supplementary Information
The citations of the pictures with copyright obtained are summarized in the supporting information (Additional file 1). Supplementary materials associated with this article can be found in the online version at 10.1186/s11671-024-04131-4.
Acknowledgements
This work was supported by the Hospital-level scientific research fund of Yunfu People's Hospital(A20231006), the Start-Up Fund for Introduced Talents and Scientific Research at Beijing Normal University (28709-312200502501), Overseas Expert Project of Guangdong Provincial Department of Science and Technology (YKZ No. 205 (2024): 309257975056). National major project of brain science and brain-like research (2021ZD0204300), National major scientific research instrument development project (61827811), the Young Scientists Fund of the National Natural Science Foundation of China, Macao Science and Technology Development Fund (FDCT 0020/2019/AMJ and FDCT 0048/2021/AGJ), University of Macau (MYRGGRG2023-00038-FHS and MYRG2022-00054-FHS) and Higher Education Fund of Macao SAR Government Natural Science Foundation of Guangdong Province (EF017/FHS-YZ/2021/GDST).
Abbreviations
- GBM
Glioblastoma
- EPR
Enhanced permeability and retention
- BBB
Blood brain barrier
- PVD
Physical vapour deposition
- 3D
Three-dimensional
- ASIC
Application specific integrated circuits
- GPS
Global positioning system
- EGFR
Epidermal growth factor receptor
- RBC
Red blood cell
- MRI
Magnetic resonance imaging
- CT
Computed tomographic
- PET
Positron emission tomography
- MN
Manufactured polymeric microneedle
- CPP
Cell-penetrating peptides
- Tf
Transferrin
- NP
Nanoparticles
- TAM
Tamoxifen
- DOX
Doxorubicin
- FK506
Tacrolimus
- PC12 cells
Pheochromocytoma cells
- AD
Alzheimer’s disease
- TAT
Trans activator transcription
- IRI
Irinotecan hydrochloride
- MET
Metformin hydrochloride
- SAX
Saxagliptin
- HA
Hyaluronic acid
- CTC
Circulating tumor cell
- VLF
Venlafaxine
- RSV
Resveratrol
- LDLR
Lipoprotein receptor
- TMZ
Temozolomide
- BRB
Berberine
- RVG
Rabies viral glycoprotein
- MSP
Muscle-specific peptide
- NSC
Neural stem cell
- Bex
Bexarotene
- AIE
Aggregation-induced emission
- ACQ
Aggregation-induced quenching
- TJs
Tightly connected
- PTT
Photothermal therapy
- CNS
Central nervous system
- LRP-1
Low density lipoprotein receptor associated Protein 1
- ROS
Reactive oxygen species
Author contributions
The concept and framework of review was designed by M.X, Z.Y, X.L and L.P. The manuscript was equally prepared by M.X, Z.Q and Z.C. And S.W provided help in revising this manuscript. All authors have given approval to the final version of the manuscript.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Competing interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mengze Xu, Zhaoquan Qin and Zhichao Chen have contributed equally.
Contributor Information
Mengze Xu, Email: mengzexu@bnu.edu.cn.
Shichao Wang, Email: scwang@bnu.edu.cn.
Liang Peng, Email: pengliang8036@163.com.
Xiaoli Li, Email: xiaoli@bnu.edu.cn.
Zhen Yuan, Email: zhenyuan@um.edu.mo.
References
- 1.Miller KD, Ostrom QT, Kruchko C, Patil N, Tihan T, Cioffi G, Fuchs HE, Waite KA, Jemal A, Siegel RL, Barnholtz-Sloan JS. Brain and other central nervous system tumor statistics, 2021. CA Cancer J Clin. 2021;71(5):381–406. [DOI] [PubMed] [Google Scholar]
- 2.Arvanitis CD, Ferraro GB, Jain RK. The blood-brain barrier and blood-tumour barrier in brain tumours and metastases. Nat Rev Cancer. 2020;20(1):26–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Blanco E, Shen H, Ferrari M. Principles of nanoparticle design for overcoming biological barriers to drug delivery. Nat Biotechnol. 2015;33(9):941–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang J, Dong Y, Ma P, Wang Y, Zhang F, Cai B, Chen P, Liu BF. Intelligent micro-/nanorobots for cancer theragnostic. Adv Mater. 2022;34(52):2201051. [DOI] [PubMed] [Google Scholar]
- 5.Wu D, Chen Q, Chen X, Han F, Chen Z, Wang Y. The blood-brain barrier: structure, regulation, and drug delivery. Signal Transduct Target Ther. 2023;8(1):217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li X, Geng X, Chen Z, Yuan Z. Recent advances in glioma microenvironment-response nanoplatforms for phototherapy and sonotherapy. Pharmacol Res. 2022;179:106218. [DOI] [PubMed] [Google Scholar]
- 7.Kishore C, Bhadra P. Targeting brain cancer cells by nanorobot, a promising nanovehicle: new challenges and future perspectives. CNS Neurol Disord Drug Targets. 2021;20(6):531–9. [DOI] [PubMed] [Google Scholar]
- 8.Yoo J, Tang S, Gao W. Micro-and nanorobots for biomedical applications in the brain. Nat Rev Bioeng. 2023. 10.1038/s44222-023-00038-4. [Google Scholar]
- 9.Deng G, Peng X, Sun Z, Zheng W, Yu J, Du L, Chen H, Gong P, Zhang P, Cai L, Tang BZ. Natural-killer-cell-inspired nanorobots with aggregation-induced emission characteristics for near-infrared-ii fluorescence-guided glioma theranostics. ACS Nano. 2020;14(9):11452–62. [DOI] [PubMed] [Google Scholar]
- 10.Zhang T-T, Li W, Meng G, Wang P, Liao W. Strategies for transporting nanoparticles across the blood–brain barrier. Biomater Sci. 2016;4(2):219–29. [DOI] [PubMed] [Google Scholar]
- 11.Jain K. Use of nanoparticles for drug delivery in glioblastoma multiforme. Expert Rev Neurother. 2007;7(4):363–72. [DOI] [PubMed] [Google Scholar]
- 12.Li J, Esteban-Fernández B, de Ávila W, Gao LZ, Wang J. Micro/nanorobots for biomedicine: delivery, surgery, sensing, and detoxification. Sci Robot. 2017. 10.1126/scirobotics.aam6431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim K, Guo J, Xu X, Fan DL. Recent progress on man-made inorganic nanomachines. Small. 2015;11(33):4037–57. [DOI] [PubMed] [Google Scholar]
- 14.Nelson BJ, Kaliakatsos IK, Abbott JJ. Microrobots for minimally invasive medicine. Annu Rev Biomed Eng. 2010;12(1):55–85. [DOI] [PubMed] [Google Scholar]
- 15.Chen YY, Chen DX, Liang SZ, Dai YG, Bai X, Song B, Zhang DY, Chen HW, Feng L. Recent advances in field-controlled micro-nano manipulations and micro-nano robots. Adv Intell Syst. 2022. 10.1002/aisy.202100116. [Google Scholar]
- 16.Requicha AAG. Nanorobots, NEMS, and nanoassembly. Proc IEEE. 2003;91(11):1922–33. [Google Scholar]
- 17.Li JH, Mayorga-Martinez CC, Ohl CD, Pumera M. Ultrasonically Propelled Micro- and Nanorobots. Adv Funct Mater. 2022. 10.1002/adfm.202102265.37521161 [Google Scholar]
- 18.Kong X, Gao P, Wang J, Fang Y, Hwang KC. Advances of medical nanorobots for future cancer treatments. J Hematol Oncol. 2023;16(1):74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Freitas RA Jr. What is nanomedicine? Nanomed Nanotechnol Biol Med. 2005;1(1):2–9. [Google Scholar]
- 20.Feynman R. There’s plenty of room at the bottom. In Feynman and computation: CRC Press; 2018. p. 63–76. [Google Scholar]
- 21.Kwoh YS, Hou J, Jonckheere EA, Hayati S. A robot with improved absolute positioning accuracy for CT guided stereotactic brain surgery. IEEE Trans Biomed Eng. 1988;35(2):153–60. [DOI] [PubMed] [Google Scholar]
- 22.Marshall LH, Magoun HW, The Horsley–Clarke stereotaxic instrument: the beginning. Kopf Carrier October, 1990: 1–5.
- 23.Meeker DC, Maslen EH, Ritter RC, Creighton FM. Optimal realization of arbitrary forces in a magnetic stereotaxis system. IEEE Trans Magn. 1996;32(2):320–8. [Google Scholar]
- 24.Tendick F, Sastry SS, Fearing RS, Cohn M. Applications of micromechatronics in minimally invasive surgery. IEEE/ASME Trans Mechatron. 1998;3(1):34–42. [Google Scholar]
- 25.Dolev S, Narayanan RP, Rosenblit M. Design of nanorobots for exposing cancer cells. Nanotechnology. 2019;30(31):315501. [DOI] [PubMed] [Google Scholar]
- 26.Cavalcanti A, Freitas RA Jr. Nanosystem design with dynamic collision detection for autonomous nanorobot motion control using neural networks. Comput Graph Geom J. 2003;5:33–49. [Google Scholar]
- 27.Martel S. Swimming microorganisms acting as nanorobots versus artificial nanorobotic agents: a perspective view from an historical retrospective on the future of medical nanorobotics in the largest known three-dimensional biomicrofluidic networks. Biomicrofluidics. 2016. 10.1063/1.4945734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Purdy PD, Fujimoto T, Replogle RE, Giles BP, Fujimoto H, Miller SL. Percutaneous intraspinal navigation for access to the subarachnoid space: use of another natural conduit for neurosurgical procedures. Neurosurg Focus. 2005;19(1):1–5. [DOI] [PubMed] [Google Scholar]
- 29.Rothemund PW. Folding DNA to create nanoscale shapes and patterns. Nature. 2006;440(7082):297–302. [DOI] [PubMed] [Google Scholar]
- 30.Felfoul O, Mohammadi M, Taherkhani S, de Lanauze D, Yong Zhong X, Loghin D, Essa S, Jancik S, Houle D, Lafleur M, Gaboury L, Tabrizian M, Kaou N, Atkin M, Vuong T, Batist G, Beauchemin N, Radzioch D, Martel S. Magneto-aerotactic bacteria deliver drug-containing nanoliposomes to tumour hypoxic regions. Nat Nanotechnol. 2016;11(11):941–7. 10.1038/nnano.2016.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Cavalcanti A, Shirinzadeh B, Fukuda T, Ikeda S. Nanorobot for brain aneurysm. Int J Robot Res. 2009;28(4):558–70. [Google Scholar]
- 32.Deng Z, Li X, Zheng K, Yao W. A humanoid robot control system with SSVEP-based asynchronous brain-computer interface. Jiqiren. 2011;33(2):129–35. [Google Scholar]
- 33.Gao W, Kagan D, Pak OS, Clawson C, Campuzano S, Chuluun-Erdene E, Shipton E, Fullerton EE, Zhang L, Lauga E, Wang J. Cargo-towing fuel-free magnetic nanoswimmers for targeted drug delivery. Small. 2012;8(3):460–7. [DOI] [PubMed] [Google Scholar]
- 34.De Lanauze D, Felfoul O, Turcot J-P, Mohammadi M, Martel S. Three-dimensional remote aggregation and steering of magnetotactic bacteria microrobots for drug delivery applications. Int J Robot Res. 2014;33(3):359–74. [Google Scholar]
- 35.Hamdi M, Ferreira A. Guidelines for the design of magnetic nanorobots to cross the blood–brain barrier. IEEE Trans Rob. 2013;30(1):81–92. [Google Scholar]
- 36.Loscrì V, Natalizio E, Mannara V, Aloi G. In a novel communication technique for nanobots based on acoustic signals, bio-inspired models of network, information, and computing systems: 7th international ICST Conference, BIONETICS 2012, Lugano, Switzerland, December 10–11, 2012, Revised Selected Papers 7. Springer. 2014;2014:91–104. [Google Scholar]
- 37.Schwartz I, Meiner Z. Robotic-assisted gait training in neurological patients: who may benefit? Ann Biomed Eng. 2015;43(5):1260–9. [DOI] [PubMed] [Google Scholar]
- 38.Amir Y, Abu-Horowitz A, Bachelet I. Folding and characterization of a bio-responsive robot from DNA origami. JoVE (Journal of Visualized Experiments). 2015;106:e51272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Li J, Li X, Luo T, Wang R, Liu C, Chen S, Li D, Yue J, Cheng S-H, Sun D. Development of a magnetic microrobot for carrying and delivering targeted cells. Sci Robot. 2018;3(19):8829. [DOI] [PubMed] [Google Scholar]
- 40.Bradley CA. Drug delivery: DNA nanorobots - seek and destroy. Nat Rev Drug Discov. 2018;17(4):242. [DOI] [PubMed] [Google Scholar]
- 41.Chen J, Wang Y. Personalized dynamic transport of magnetic nanorobots inside the brain vasculature. Nanotechnology. 2020;31(49):495706. [DOI] [PubMed] [Google Scholar]
- 42.Cao L, Li G, Xu Y, Zhang H, Shu X, Zhang D. A brain-actuated robotic arm system using non-invasive hybrid brain–computer interface and shared control strategy. J Neural Eng. 2021;18(4):046045. [DOI] [PubMed] [Google Scholar]
- 43.Chen B, Tan H, Ding M, Liu L, Wang S, Peng X, Tian H, Jiang J, Gao J, Huang W. Nanorobot-mediated synchronized neuron activation. ACS Nano. 2023;17(14):13826–39. [DOI] [PubMed] [Google Scholar]
- 44.Wu J, Jiao N, Lin D, Li N, Ma T, Tung S, Cheng W, Wu A, Liu L. Dual-responsive nanorobot-based marsupial robotic system for intracranial cross-scale targeting drug delivery. Adv Mater. 2024;36(9):e2306876. [DOI] [PubMed] [Google Scholar]
- 45.Sireesha M, Jagadeesh Babu V, Kranthi Kiran AS, Ramakrishna S. A review on carbon nanotubes in biosensor devices and their applications in medicine. Nanocomposites. 2018;4(2):36–57. [Google Scholar]
- 46.Conesa L, Costa U, Morales E, Edwards DJ, Cortes M, León D, Bernabeu M, Medina J. An observational report of intensive robotic and manual gait training in sub-acute stroke. J Neuroeng Rehabil. 2012;9:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wu JF, Jiao ND, Lin DJ, Li N, Ma TY, Tung SV, Cheng W, Wu AH, Liu LQ. Dual-responsive nanorobot-based marsupial robotic system for intracranial cross-scale targeting drug delivery. Adv Mater. 2024. 10.1002/adma.202306876. [DOI] [PubMed] [Google Scholar]
- 48.Lee S, Kim JY, Kim J, Hoshiar AK, Park J, Lee S, Kim J, Pané S, Nelson BJ, Choi H. A needle-type microrobot for targeted drug delivery by affixing to a microtissue. Adv Healthcare Mater. 2020;9(7):1901697. [DOI] [PubMed] [Google Scholar]
- 49.Servant A, Qiu F, Mazza M, Kostarelos K, Nelson BJ. Controlled in vivo swimming of a swarm of bacteria-like microrobotic flagella. Adv Mater. 2015;27(19):2981–8. [DOI] [PubMed] [Google Scholar]
- 50.Kim S, Qiu F, Kim S, Ghanbari A, Moon C, Zhang L, Nelson BJ, Choi H. Fabrication and characterization of magnetic microrobots for three-dimensional cell culture and targeted transportation. Adv Mater. 2013;25(41):5863–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kim S, Lee S, Lee J, Nelson BJ, Zhang L, Choi H. Fabrication and manipulation of ciliary microrobots with non-reciprocal magnetic actuation. Sci Rep. 2016;6(1):30713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Lee S, Kim S, Kim S, Kim J-Y, Moon C, Nelson BJ, Choi H. A Capsule-type microrobot with pick-and-drop motion for targeted drug and cell delivery. Adv Healthcare Mater. 2018. 10.1002/adhm.201700985. [DOI] [PubMed] [Google Scholar]
- 53.Wrede P, Degtyaruk O, Kalva SK, Deán-Ben XL, Bozuyuk U, Aghakhani A, Akolpoglu B, Sitti M, Razansky D. Real-time 3D optoacoustic tracking of cell-sized magnetic microrobots circulating in the mouse brain vasculature. Sci Adv. 2022;8(19):eabm9132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Soto F, Chrostowski R. Frontiers of medical micro/nanorobotics: in vivo applications and commercialization perspectives toward clinical uses. Front Bioeng Biotechnol. 2018;6:170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Feng Y, An M, Liu Y, Sarwar MT, Yang H. Advances in chemically powered micro/nanorobots for biological applications: a review. Adv Func Mater. 2023;33(1):2209883. [Google Scholar]
- 56.Wang J, Manesh KM. Motion control at the nanoscale. Small. 2010;6(3):338–45. [DOI] [PubMed] [Google Scholar]
- 57.Sundararajan S, Lammert PE, Zudans AW, Crespi VH, Sen A. Catalytic motors for transport of colloidal cargo. Nano Lett. 2008;8(5):1271–6. [DOI] [PubMed] [Google Scholar]
- 58.Qiao R, Fu C, Forgham H, Javed I, Huang X, Zhu J, Whittaker AK, Davis TP. Magnetic iron oxide nanoparticles for brain imaging and drug delivery. Adv Drug Deliv Rev. 2023;197:114822. [DOI] [PubMed] [Google Scholar]
- 59.Hu NN, Ding LJ, Liu YY, Wang KM, Zhang B, Yin RX, Zhou WJ, Bi ZM, Zhang WJ. Development of 3D-Printed magnetic micro-nanorobots for targeted therapeutics: the state of art. Adv Nanobiomed Res. 2023. 10.1002/anbr.202300018. [Google Scholar]
- 60.Guo Y, Jing D, Liu S, Yuan Q. Construction of intelligent moving micro/nanomotors and their applications in biosensing and disease treatment. Theranostics. 2023;13(9):2993–3020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wu Z, Chen Y, Mukasa D, Pak OS, Gao W. Medical micro/nanorobots in complex media. Chem Soc Rev. 2020;49(22):8088–112. [DOI] [PubMed] [Google Scholar]
- 62.Pulgar VM. Transcytosis to cross the blood brain barrier, new advancements and challenges. Front Neurosci. 2019;12:1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gupta A, Soni S, Chauhan N, Khanuja M, Jain U. Nanobots-based advancement in targeted drug delivery and imaging: an update. J Control Release. 2022;349:97–108. [DOI] [PubMed] [Google Scholar]
- 64.Freitas RA Jr. Microbivores: artificial mechanical phagocytes using digest and discharge protocol. J Evol Technol. 2005;14(1):54–106. [Google Scholar]
- 65.Kanaparthy R, Kanaparthy R. The changing face of dentistry: nanotechnology. Int J Nanomed. 2011. 10.2147/IJN.S24353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wu Z, Li T, Gao W, Xu T, Jurado-Sánchez B, Li J, Gao W, He Q, Zhang L, Wang J. Cell-membrane-coated synthetic nanomotors for effective biodetoxification. Adv Func Mater. 2015;25(25):3881–7. [Google Scholar]
- 67.Li J, Angsantikul P, Liu W, Esteban-Fernandez-de-Avila B, Chang X, Sandraz E, Liang Y, Zhu S, Zhang Y, Chen C, Gao W, Zhang L, Wang J. Biomimetic platelet-camouflaged nanorobots for binding and isolation of biological threats. Adv Mater. 2018. 10.1002/adma.201704800. [DOI] [PubMed] [Google Scholar]
- 68.Mengze X, Yang Y, Yuan Z. Breast cancer cell membrane camouflaged lipid nanoparticles for tumor-targeted NIR-II phototheranostics. Pharmaceutics. 2022;14(7):1367. 10.3390/pharmaceutics14071367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wu Z, Lin X, Si T, He Q. Recent progress on bioinspired self-propelled micro/nanomotors via controlled molecular self-assembly. Small. 2016;12(23):3080–93. [DOI] [PubMed] [Google Scholar]
- 70.Mundaca-Uribe R, Esteban-Fernandez-de-Avila B, Holay M, Lekshmy-Venugopalan P, Nguyen B, Zhou J, Abbas A, Fang RH, Zhang L, Wang J. Zinc Microrocket pills: fabrication and characterization toward active oral delivery. Adv Healthc Mater. 2020;9(18):2000900. [DOI] [PubMed] [Google Scholar]
- 71.Guix M, Mayorga-Martinez CC, Merkoci A. Nano/micromotors in (bio)chemical science applications. Chem Rev. 2014;114(12):6285–322. [DOI] [PubMed] [Google Scholar]
- 72.Xu M, Chen Z, Zheng J, Zhao Q, Yuan Z. Artificial intelligence-aided optical imaging for cancer theranostics. Semin Cancer Biol. 2023;94:62–80. [DOI] [PubMed] [Google Scholar]
- 73.Liu D, Wang T, Lu Y. Untethered microrobots for active drug delivery: from rational design to clinical settings. Adv Healthc Mater. 2022;11(3):e2102253. [DOI] [PubMed] [Google Scholar]
- 74.Liu J, Xu M, Yuan Z. Immunoscore guided cold tumors to acquire “temperature” through integrating physicochemical and biological methods. BIO Integration. 2020;7(1):6. [Google Scholar]
- 75.Patra JK, Das G, Fraceto LF, Campos EVR, Rodriguez-Torres MDP, Acosta-Torres LS, Diaz-Torres LA, Grillo R, Swamy MK, Sharma S. Nano based drug delivery systems: recent developments and future prospects. J Nanobiotechnol. 2018;16(1):1–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Freitas RA. Pharmacytes: an ideal vehicle for targeted drug delivery. J Nanosci Nanotechnol. 2006;6(9–10):2769–75. [DOI] [PubMed] [Google Scholar]
- 77.Martins NR, Angelica A, Chakravarthy K, Svidinenko Y, Boehm FJ, Opris I, Lebedev MA, Swan M, Garan SA, Rosenfeld JV, Human brain/cloud interface. In Advances in Clinical Immunology, Medical Microbiology, COVID-19, and Big Data, Jenny Stanford Publishing: 2021; pp 485–538.
- 78.Mei Y, Solovev AA, Sanchez S, Schmidt OG. Rolled-up nanotech on polymers: from basic perception to self-propelled catalytic microengines. Chem Soc Rev. 2011;40(5):2109–19. [DOI] [PubMed] [Google Scholar]
- 79.Sanchez S, Soler L, Katuri J. Chemically powered micro- and nanomotors. Angew Chem Int Ed Engl. 2015;54(5):1414–44. [DOI] [PubMed] [Google Scholar]
- 80.Kumari S, Mg S, Mayor S. Endocytosis unplugged: multiple ways to enter the cell. Cell Res. 2010;20(3):256–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Frcitas RA. Exploratory design in medical nanotechnology: a mechanical artificial red cell. Artif Cells Blood Substit Biotechnol. 1998;26(4):411–30. [DOI] [PubMed] [Google Scholar]
- 82.Aryal M, Vykhodtseva N, Zhang Y-Z, Park J, McDannold N. Multiple treatments with liposomal doxorubicin and ultrasound-induced disruption of blood–tumor and blood–brain barriers improve outcomes in a rat glioma model. J Control Release. 2013;169(1–2):103–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zottel A, Videtič Paska A, Jovčevska I. Nanotechnology meets oncology: nanomaterials in brain cancer research, diagnosis and therapy. Materials. 2019;12(10):1588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ruff J, Hüwel S, Kogan MJ, Simon U, Galla H-J. The effects of gold nanoparticles functionalized with ß-amyloid specific peptides on an in vitro model of blood–brain barrier. Nanomed Nanotechnol Biol Med. 2017;13(5):1645–52. [DOI] [PubMed] [Google Scholar]
- 85.Betzer O, Shilo M, Opochinsky R, Barnoy E, Motiei M, Okun E, Yadid G, Popovtzer R. The effect of nanoparticle size on the ability to cross the blood–brain barrier: an in vivo study. Nanomedicine. 2017;12(13):1533–46. [DOI] [PubMed] [Google Scholar]
- 86.Huang D, Wang Q, Cao Y, Yang H, Li M, Wu F, Zhang Y, Chen G, Wang Q. Multiscale NIR-II imaging-guided brain-targeted drug delivery using engineered cell membrane nanoformulation for alzheimer’s disease therapy. ACS Nano. 2023;17(5):5033–46. [DOI] [PubMed] [Google Scholar]
- 87.Law LH, Huang J, Xiao P, Liu Y, Chen Z, Lai JHC, Han X, Cheng GWY, Tse KH, Chan KWY. Multiple CEST contrast imaging of nose-to-brain drug delivery using iohexol liposomes at 3T MRI. J Control Release. 2023;354:208–20. [DOI] [PubMed] [Google Scholar]
- 88.Sabry SA, Abd El Razek AM, Nabil M, Khedr SM, El-Nahas HM, Eissa NG. Brain-targeted delivery of Valsartan using solid lipid nanoparticles labeled with Rhodamine B; a promising technique for mitigating the negative effects of stroke. Drug Deliv. 2023;30(1):2179127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Muresan P, McCrorie P, Smith F, Vasey C, Taresco V, Scurr DJ, Kern S, Smith S, Gershkovich P, Rahman R, Marlow M. Development of nanoparticle loaded microneedles for drug delivery to a brain tumour resection site. Eur J Pharm Biopharm. 2023;182:53–61. [DOI] [PubMed] [Google Scholar]
- 90.Malinovskaya Y, Melnikov P, Baklaushev V, Gabashvili A, Osipova N, Mantrov S, Ermolenko Y, Maksimenko O, Gorshkova M, Balabanyan V. Delivery of doxorubicin-loaded PLGA nanoparticles into U87 human glioblastoma cells. Int J Pharm. 2017;524(1–2):77–90. [DOI] [PubMed] [Google Scholar]
- 91.Li Y, He H, Jia X, Lu W-L, Lou J, Wei Y. A dual-targeting nanocarrier based on poly (amidoamine) dendrimers conjugated with transferrin and tamoxifen for treating brain gliomas. Biomaterials. 2012;33(15):3899–908. [DOI] [PubMed] [Google Scholar]
- 92.Meng X, Xu Y, Lu Q, Sun L, An X, Zhang J, Chen J, Gao Y, Zhang Y, Ning X. Ultrasound-responsive alkaline nanorobots for the treatment of lactic acidosis-mediated doxorubicin resistance. Nanoscale. 2020;12(25):13801–10. [DOI] [PubMed] [Google Scholar]
- 93.Ishii T, Asai T, Oyama D, Agato Y, Yasuda N, Fukuta T, Shimizu K, Minamino T, Oku N. Treatment of cerebral ischemia-reperfusion injury with PEGylated liposomes encapsulating FK506. FASEB J. 2013;27(4):1362–70. [DOI] [PubMed] [Google Scholar]
- 94.Picone P, Sabatino MA, Ditta LA, Amato A, San Biagio PL, Mule F, Giacomazza D, Dispenza C, Di Carlo M. Nose-to-brain delivery of insulin enhanced by a nanogel carrier. J Control Release. 2018;270:23–36. [DOI] [PubMed] [Google Scholar]
- 95.Yan L, Wang H, Jiang Y, Liu J, Wang Z, Yang Y, Huang S, Huang Y. Cell-penetrating peptide-modified PLGA nanoparticles for enhanced nose-to-brain macromolecular delivery. Macromol Res. 2013;21:435–41. [Google Scholar]
- 96.Cheng Y, Dai Q, Morshed RA, Fan X, Wegscheid ML, Wainwright DA, Han Y, Zhang L, Auffinger B, Tobias AL. Blood-brain barrier permeable gold nanoparticles: an efficient delivery platform for enhanced malignant glioma therapy and imaging. Small. 2014;10(24):5137–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Haque S, Md S, Sahni JK, Ali J, Baboota S. Development and evaluation of brain targeted intranasal alginate nanoparticles for treatment of depression. J Psychiatr Res. 2014;48(1):1–12. [DOI] [PubMed] [Google Scholar]
- 98.Shao J, Xuan M, Zhang H, Lin X, Wu Z, He Q. Chemotaxis-guided hybrid neutrophil micromotors for targeted drug transport. Angew Chem Int Ed Engl. 2017;56(42):12935–9. [DOI] [PubMed] [Google Scholar]
- 99.Fernandes J, Ghate MV, Mallik SB, Lewis SA. Amino acid conjugated chitosan nanoparticles for the brain targeting of a model dipeptidyl peptidase-4 inhibitor. Int J Pharm. 2018;547(1–2):563–71. [DOI] [PubMed] [Google Scholar]
- 100.Taghizadehghalehjoughi A, Hacimuftuoglu A, Cetin M, Ugur AB, Galateanu B, Mezhuev Y, Okkay U, Taspinar N, Taspinar M, Uyanik A. Effect of metformin/irinotecan-loaded poly-lactic-co-glycolic acid nanoparticles on glioblastoma: in vitro and in vivo studies. Nanomedicine. 2018;13(13):1595–606. [DOI] [PubMed] [Google Scholar]
- 101.Su Y, Yuan B, Jiang Y, Wu P, Huang X, Zhu JJ, Jiang LP. A bioinspired hollow g-C(3)N(4)-CuPc heterostructure with remarkable SERS enhancement and photosynthesis-mimicking properties for theranostic applications. Chem Sci. 2022;13(22):6573–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Shen Y, Cao B, Snyder NR, Woeppel KM, Eles JR, Cui XT. ROS responsive resveratrol delivery from LDLR peptide conjugated PLA-coated mesoporous silica nanoparticles across the blood–brain barrier. J Nanobiotechnol. 2018;16(1):1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Song X, Chen Z, Zhang X, Xiong J, Jiang T, Wang Z, Geng X, Cheang UK. Magnetic tri-bead microrobot assisted near-infrared triggered combined photothermal and chemotherapy of cancer cells. Sci Rep. 2021;11(1):7907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Barbara R, Belletti D, Pederzoli F, Masoni M, Keller J, Ballestrazzi A, Vandelli MA, Tosi G, Grabrucker AM. Novel curcumin loaded nanoparticles engineered for blood-brain barrier crossing and able to disrupt abeta aggregates. Int J Pharm. 2017;526(1–2):413–24. [DOI] [PubMed] [Google Scholar]
- 105.Mondal J, Patra M, Panigrahi AK, Khuda-Bukhsh AR. Improved drug carriage and protective potential against Cisplatin-induced toxicity using Boldine-loaded PLGA nanoparticles. J Ayur Integr Med. 2020;11(1):24–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Fonseca FN, Betti AH, Carvalho FC, Gremião MP, Dimer FA, Guterres SS, Tebaldi ML, Rates SM, Pohlmann AR. Mucoadhesive amphiphilic methacrylic copolymer-functionalized poly (ε-caprolactone) nanocapsules for nose-to-brain delivery of olanzapine. J Biomed Nanotechnol. 2015;11(8):1472–81. [DOI] [PubMed] [Google Scholar]
- 107.Jafarieh O, Md S, Ali M, Baboota S, Sahni J, Kumari B, Bhatnagar A, Ali J. Design, characterization, and evaluation of intranasal delivery of ropinirole-loaded mucoadhesive nanoparticles for brain targeting. Drug Dev Ind Pharm. 2015;41(10):1674–81. [DOI] [PubMed] [Google Scholar]
- 108.Garcia-Gradilla V, Sattayasamitsathit S, Soto F, Kuralay F, Yardimci C, Wiitala D, Galarnyk M, Wang J. Ultrasound-propelled nanoporous gold wire for efficient drug loading and release. Small. 2014;10(20):4154–9. [DOI] [PubMed] [Google Scholar]
- 109.Lakkadwala S, Dos Santos Rodrigues B, Sun C, Singh J. Dual functionalized liposomes for efficient co-delivery of anti-cancer chemotherapeutics for the treatment of glioblastoma. J Control Release. 2019;307:247–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Song S, Mao G, Du J, Zhu X. Novel RGD containing, temozolomide-loading nanostructured lipid carriers for glioblastoma multiforme chemotherapy. Drug Delivery. 2016;23(4):1404–8. [DOI] [PubMed] [Google Scholar]
- 111.Lohan S, Raza K, Mehta S, Bhatti GK, Saini S, Singh B. Anti-Alzheimer’s potential of berberine using surface decorated multi-walled carbon nanotubes: a preclinical evidence. Int J Pharm. 2017;530(1–2):263–78. [DOI] [PubMed] [Google Scholar]
- 112.Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJ. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29(4):341–5. [DOI] [PubMed] [Google Scholar]
- 113.Lu JP, Ding JL, Chu BB, Ji C, Zhang Q, Xu YA, Song B, Wang HY, He Y. Inactive Trojan bacteria as safe drug delivery vehicles crossing the blood-brain barrier. Nano Lett. 2023;23(10):4326–33. [DOI] [PubMed] [Google Scholar]
- 114.Rehman FU, Liu Y, Zheng M, Shi B. Exosomes based strategies for brain drug delivery. Biomaterials. 2023;293:121949. [DOI] [PubMed] [Google Scholar]
- 115.Silant’ev VE, Shmelev ME, Belousov AS, Patlay AA, Shatilov RA, Farniev VM, Kumeiko VV. How to develop drug delivery system based on carbohydrate nanoparticles targeted to brain tumors. Polymers. 2023;15(11):2516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Sharma S, Dang S. Nanocarrier-based drug delivery to brain: interventions of surface modification. Curr Neuropharmacol. 2023;21(3):517–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Li X, Sun T, Jiang C. Intelligent delivery systems in tumor metabolism regulation: exploring the path ahead. Adv Mater. 2024;36(11):e2309582. [DOI] [PubMed] [Google Scholar]
- 118.Tan Y, Liu P, Li D, Wang D, Tang BZ. NIR-II aggregation-induced emission luminogens for tumor phototheranostics. Biosensors. 2022;12(1):46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Joseph A, Contini C, Cecchin D, Nyberg S, Ruiz-Perez L, Gaitzsch J, Fullstone G, Tian X, Azizi J, Preston J, Volpe G, Battaglia G. Chemotactic synthetic vesicles: design and applications in blood-brain barrier crossing. Sci Adv. 2017;3(8):e1700362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Fu W, Ma L, Ju Y, Xu J, Li H, Shi S, Zhang T, Zhou R, Zhu J, Xu R. Therapeutic siCCR2 loaded by tetrahedral framework DNA nanorobotics in therapy for intracranial hemorrhage. Adv Func Mater. 2021;31(33):2101435. [Google Scholar]
- 121.Wang S, Liu K, Zhou Q, Xu C, Gao J, Wang Z, Wang F, Chen B, Ye Y, Ou J, Jiang J, Wilson DA, Liu S, Peng F, Tu Y. Hydrogen-powered microswimmers for precise and active hydrogen therapy towards acute ischemic stroke. Adv Funct Mater. 2021;31(19):2009475. [Google Scholar]
- 122.Lindvall O, Kokaia Z, Martinez-Serrano A. Stem cell therapy for human neurodegenerative disorders–how to make it work. Nat Med. 2004;10(Suppl 7):S42–50. [DOI] [PubMed] [Google Scholar]
- 123.Jeon S, Kim S, Ha S, Lee S, Kim E, Kim SY, Park SH, Jeon JH, Kim SW, Moon C. Magnetically actuated microrobots as a platform for stem cell transplantation. Sci Robot. 2019;4(30):eaav4317. [DOI] [PubMed] [Google Scholar]
- 124.Jeon S, Park SH, Kim E, Kim JY, Kim SW, Choi H. A magnetically powered stem cell-based microrobot for minimally invasive stem cell delivery via the intranasal pathway in a mouse brain. Adv Healthcare Mater. 2021;10(19):2100801. [DOI] [PubMed] [Google Scholar]
- 125.Liu L, Wu J, Wang S, Kun L, Gao J, Chen B, Ye Y, Wang F, Tong F, Jiang J. Control the neural stem cell fate with biohybrid piezoelectrical magnetite micromotors. Nano Lett. 2021;21(8):3518–26. [DOI] [PubMed] [Google Scholar]
- 126.Joseph A, Contini C, Cecchin D, Nyberg S, Ruiz-Pérez L, Gaitzsch J, Fullstone G, Tian X, Azizi JB, Preston JE, Volpe G, Battaglia G. Chemotactic synthetic vesicles: design and applications in blood-brain barrier crossing. Sci Adv. 2017. 10.1126/sciadv.1700362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Xu M, Xue B, Wang Y, Wang D, Gao D, Yang S, Zhao Q, Zhou C, Ruan S, Yuan Z. Temperature-feedback nanoplatform for NIR-II penta-modal imaging-guided synergistic photothermal therapy and CAR-NK immunotherapy of lung cancer. Small. 2021;17(43):e2101397. [DOI] [PubMed] [Google Scholar]
- 128.Li L, Yu Z, Liu J, Yang M, Shi G, Feng Z, Luo W, Ma H, Guan J, Mou F. Swarming responsive photonic nanorobots for motile-targeting microenvironmental mapping and mapping-guided photothermal treatment. Nanomicro Lett. 2023;15(1):141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Xie X, Lu W, Ma D, Liu S, Duan Y, Li K, Cheng Z, Qiu J. Metabolic and textural changes in the brain of non-small cell lung cancer patients: a total-body positron emission tomography/computed tomography study. iRADIOLOGY. 2023;1(2):109–19. [Google Scholar]
- 130.Wang B, Wang QL, Chan KF, Ning ZP, Wang QQ, Ji FT, Yang HJ, Jiang S, Zhang ZF, Ip BYM, Ko H, Chung JPW, Qiu M, Han JG, Chiu PWY, Sung JJY, Du SW, Leung TWH, Yu SCH, Zhang L. tPA-anchored nanorobots for in vivo arterial recanalization at submillimeter-scale segments. Sci Adv. 2024. 10.1126/sciadv.adk8970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Wu T, Nieminen TA, Mohanty S, Miotke J, Meyer RL, Rubinsztein-Dunlop H, Berns MW. A photon-driven micromotor can direct nerve fibre growth. Nat Photon. 2012;6(1):62–7. [Google Scholar]
- 132.Zhang H, Li Z, Gao C, Fan X, Pang Y, Li T, Wu Z, Xie H, He Q. Dual-responsive biohybrid neutrobots for active target delivery. Sci Robot. 2021;6(52):eaaz9519. [DOI] [PubMed] [Google Scholar]
- 133.Wu J, Jiao N, Lin D, Li N, Ma T, Tung S, Cheng W, Wu A, Liu L. Dual-responsive nanorobot based marsupial robotic system for intracranial cross-scale targeting drug delivery. Adv Mater. 2023;36:2306876. [DOI] [PubMed] [Google Scholar]
- 134.Joseph A, Contini C, Cecchin D, Nyberg S, Ruiz-Perez L, Gaitzsch J, Fullstone G, Tian X, Azizi J, Preston J. Chemotactic synthetic vesicles: design and applications in blood-brain barrier crossing. Sci Adv. 2017;3(8):e1700362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lee S, Kim S, Kim S, Kim JY, Moon C, Nelson BJ, Choi H. A capsule-type microrobot with pick-and-drop motion for targeted drug and cell delivery. Adv Healthcare Mater. 2018;7(9):1700985. [DOI] [PubMed] [Google Scholar]
- 136.Deng G, Peng X, Sun Z, Zheng W, Yu J, Du L, Chen H, Gong P, Zhang P, Cai L. Natural-killer-cell-inspired nanorobots with aggregation-induced emission characteristics for near-infrared-II fluorescence-guided glioma theranostics. ACS Nano. 2020;14(9):11452–62. [DOI] [PubMed] [Google Scholar]
- 137.Wang S, Liu K, Zhou Q, Xu C, Gao J, Wang Z, Wang F, Chen B, Ye Y, Ou J. Hydrogen-powered microswimmers for precise and active hydrogen therapy towards acute Ischemic stroke. Adv Func Mater. 2021;31(19):2009475. [Google Scholar]
- 138.Matsuoka Y, Howe R. Robotics for surgery. Annu Rev Biomed Eng. 1999;1:211–40. [DOI] [PubMed] [Google Scholar]
- 139.Esteban-Fernández-de-Ávila B, Angsantikul P, Li J, Gao W, Zhang L, Wang J. Micromotors go in vivo: from test tubes to live animals. Adv Funct Mater. 2017. 10.1002/adfm.201705640. [Google Scholar]
- 140.Wang D, Gao C, Zhou C, Lin Z, He Q. Leukocyte membrane-coated liquid metal nanoswimmers for actively targeted delivery and synergistic chemophotothermal therapy. Research. 2020. 10.34133/2020/3676954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Zhou Y, Ye M, Hu C, Qian H, Nelson BJ, Wang X. Stimuli-responsive functional micro-/nanorobots: a review. ACS Nano. 2023;17(16):15254–76. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.