Abstract
There is an increased prevalence of atherosclerotic cardiovascular disease (ASCVD) in patients with inflammatory rheumatic diseases (IRD) including rheumatoid arthritis, systemic lupus erythematosus, psoriatic arthritis, and systemic sclerosis. The mechanism for the development of ASCVD in these conditions has been linked not only to a higher prevalence and undertreatment of traditional cardiovascular (CV) risk factors but importantly to chronic inflammation and a dysregulated immune system which contribute to impaired endothelial and microvascular function, factors that may contribute to accelerated atherosclerosis. Accurate ASCVD risk stratification and optimal risk management remain challenging in this population with many barriers that include lack of validated risk calculators, the remitting and relapsing nature of underlying disease, deleterious effect of medications used to manage rheumatic diseases, multimorbidity, decreased mobility due to joint pain, and lack of clarity about who bears the responsibility of performing CV risk assessment and management (rheumatologist vs. primary care provider vs. cardiologist). Despite recent advances in this field, there remain significant gaps in knowledge regarding the best diagnostic and management approach. The evolving field of Cardio-Rheumatology focuses on optimization of cardiovascular care and research in this patient population through collaboration and coordination of care between rheumatologists, cardiologists, radiologists, and primary care providers. This review aims to provide an overview of current state of knowledge about ASCVD risk stratification in patients with IRD, contributing factors including effect of medications, and review of the current recommendations for cardiovascular risk management in patients with inflammatory disease with a focus on hypertension as a key risk factor.
Keywords: atherosclerotic cardiovascular risk, cardio-rheumatology, hypertension, inflammation, inflammatory rheumatic diseases
Introduction
Cardiovascular diseases (CVD) are the leading cause of death globally.1 There is a higher prevalence of a broad range of CVD including atherosclerotic cardiovascular disease (ASCVD) in patients with inflammatory rheumatic diseases (IRD) compared to the general population.2–4
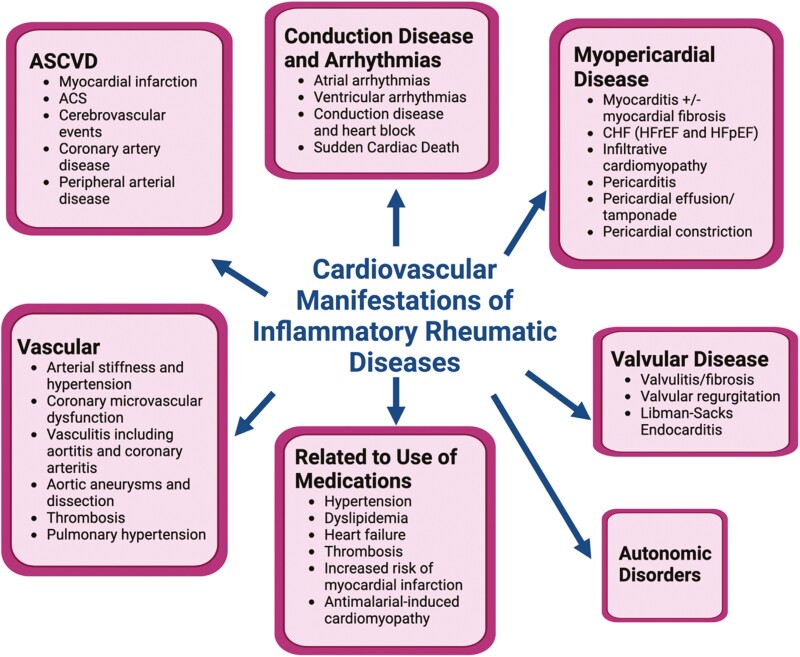
The mechanism for development of ASCVD in the presence of systemic inflammatory disease has been linked not only to a higher prevalence and undertreatment of traditional cardiovascular (CV) risk factors but importantly to chronic inflammation and a dysregulated immune system which contribute to impaired endothelial and microvascular function, factors that may contribute to accelerated atherogenesis.5–7 Other than a predisposition to premature ASCVD which is the focus of this review, patients with IRD are at high risk for a wide range of other cardiac and vascular problems, including myocarditis, ischemic, and non-ischemic cardiomyopathy, pericardial disease, valvulitis, and valvular dysfunction, conduction disease and arrhythmias, pulmonary hypertension, and vasculitis. A hypercoagulable state may be present in some IRDs, predisposing patients to thrombotic events including vascular thrombosis and nonbacterial thrombotic endocarditis (Figure 1).
Figure 1.
Range of cardiovascular problems in inflammatory rheumatic diseases. ASCVD, atherosclerotic cardiovascular disease; ACS, acute coronary syndromes; CHF, congestive heart failure; HfrEF, heart failure with reduced ejection fraction; HfpEF, heart failure with preserved ejection fraction.
The increased prevalence of CVD in patients with IRDs and the worse associated CV outcomes, highlight CVD prevention and management as a vital component of care in this patient population. The evolving field of Cardio-Rheumatology focuses on the optimization of cardiovascular care and research in patients with IRDs through collaboration and coordination between rheumatologists, cardiologists, radiologists, and primary care providers. In this review, we aim to provide an overview of current state of knowledge about ASCVD risk stratification in patients with IRD, contributing factors including effect of medications, and review of the current recommendations for risk management in this population with a focus on hypertension (HTN) as a potent and modifiable risk factor.
CARDIOVASCULAR RISK IN RHEUMATIC DISEASES
Increased risk of CVD morbidity and mortality has been reported in a wide range of IRDs. Rheumatoid arthritis (RA), the most common type of chronic inflammatory arthritis, affects approximately 1% of the adult population and has been consistently shown to be associated with an increase in CV risk. CVD mortality in RA has been reported to be increased by 50% compared to the general population.8 Patients with RA have also been reported to have a 1.7-fold increased risk of myocardial infarction (MI) compared to those without RA2 and poorer prognosis after an MI.9 A 2-fold increased risk for developing congestive heart failure (CHF), both due to HF with reduced ejection fraction (HFrEF) and with preserved ejection fraction (HFpEF),10,11 have also been reported in patients with RA compared to those without RA, a risk that is not fully explained by presence of ischemic heart disease and is associated with underlying disease activity and rheumatoid factor seropositivity.11 Furthermore, there is a higher prevalence of atrial fibrillation and strokes in individuals with RA, especially in those with elevated markers of disease activity including severe extra-articular RA-related manifestations, such as rheumatoid nodules (HR 3.29, 95% CI 1.98–5.48) and elevated inflammatory markers (HR 2.04, 95% CI 1.19–3.5).12,13 Systemic lupus erythematosus (SLE) is an autoimmune systemic condition, predominantly affecting women, with multiple organ involvement including the heart. In a recent nationwide Danish cohort study, patients with SLE had a higher associated risk of CHF, ASCVD, arrhythmias, and sudden cardiac death compared to matched control subjects.4 Psoriatic arthritis (PsA), a seronegative type of inflammatory arthritis, affects a third of patients with psoriasis. Both psoriasis and PsA are strongly associated with obesity and metabolic syndrome. A meta-analysis of observational studies in patients with PsA reported 43% and 22% increase in CV and cerebrovascular morbidity, respectively.14 Increased risk of CHF in PsA was associated with a combination of known traditional CV risk factors and measures of disease activity, particularly in non-ischemic heart failure, suggesting that the effect of inflammation on CHF may be partially independent of atherosclerotic disease.15 Increased risk is also observed in a wide range of other rheumatic inflammatory conditions, such as ankylosing spondylitis, systemic sclerosis, giant cell arteritis, and Takayasu arteritis, though less well studied.
CHALLENGES OF MANAGING ATHEROSCLEROTIC CARDIOVASCULAR RISK IN RHEUMATIC DISEASES
Despite growing knowledge of the increased ASCVD risk and its mechanisms over the last decade, there remain significant gaps in diagnosis and treatment of traditional CV risk factors including HTN in this population (Figure 2).16,17 CV risk assessment and management in patients with underlying IRDs can be challenging due to a number of different barriers including lack of validated risk calculators, the remitting and relapsing nature of underlying inflammatory disease, deleterious effect of medications used to manage rheumatic diseases, multimorbidity, decreased mobility due to joint pain, and lack of clarity about who bears the responsibility of performing CV risk assessment and management (rheumatologist vs. primary care provider vs. cardiologist).
Figure 2.
Risk factors for atherosclerotic cardiovascular disease in patients with inflammatory rheumatic diseases.
CURRENT STATE OF ASCVD RISK STRATIFICATION IN PATIENTS WITH RHEUMATIC DISEASE
The commonly used ASCVD risk calculators, such as the Framingham Risk Score and the Pooled Cohort Equations typically underestimate risk in this patient population.16 Efforts to include nontraditional risk factors, disease-specific parameters, multipliers, and biomarkers have not yet been successful at improving risk estimates for primary prevention.18,19 Exposure to traditional risk factors, such as HTN, can vary markedly over time in patients with a chronic relapsing-and-remitting disease partly due to variable disease activity and treatment-related factors, such as use of non-steroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and selected disease-modifying anti-rheumatic drugs (DMARDs) that can affect blood pressure such as leflunomide.17 Cumulative exposure overtime to risk factors, such as dyslipidemia and HTN, are better able to quantify ASCVD risk,18 though these dynamic risk factors are difficult to capture in a single point-in-time model, likely contributing to the poor performance of current snapshot risk calculators. Additionally, some risk factors behave paradoxically in the inflammatory milieu; for example, lower Body Mass Index (BMI) in RA has been associated with worse CV outcomes.20 Similarly, lipid levels may appear falsely low in the presence of active inflammation and start to rise once inflammation is better controlled, the so-called lipid paradox.21
The European League Against Rheumatism (EULAR) recommended a multiplier of 1.5 to the calculated score using traditional ASCVD risk models for RA.22 Despite this recommendation, validation studies have shown that using this multiplication factor does not significantly improve risk prediction.23,24 Literature to support recommendations for other rheumatic conditions is even more scarce. As such, EULAR did not endorse the use of any particular risk calculators or multipliers for SLE or other IRDs in their latest recommendations in 2021 due to lack of validated disease-specific tools. Instead, they recommended a thorough assessment of traditional and disease-specific factors to guide prevention efforts.25 The American College of Cardiology/American Heart Association (ACC/AHA) 2018 guidelines for ASCVD risk management do recognize chronic inflammatory conditions, such as RA, as a risk-enhancing factor to guide clinician–patient risk discussion in favor of use of statins in patients that would otherwise be placed in an intermediate risk category.26 Similarly, the Canadian Cardiovascular Society 2021 guidelines for the management of dyslipidemia, recognize individuals with inflammatory diseases to be at higher ASCVD risk and suggest screening for dyslipidemia regardless of age for all individuals with systemic inflammatory disease.27 Use of risk enhancers, such as elevation in lipoprotein (a) (Lp(a)), an LDL-like atherogenic lipid particle, and high-sensitivity C-Reactive Protein (hs-CRP), an inflammatory biomarker, are recommended to help further risk stratify patients in the general population. Data from both primary and secondary ASCVD prevention studies suggest that Lp(a)-associated ASCVD risk is enhanced in the presence of inflammation, highlighting the utility of these risk enhancers in patients with systemic inflammatory disease.28,29 In addition, vascular imaging biomarkers can also be used as risk modifiers to further fine-tune ASCVD risk assessment. There is ample evidence in the general population for the use of noninvasive vascular imaging for detection of subclinical atheroma, such as non-contrast coronary artery calcium measurement and/or carotid ultrasound, to improve appropriate selection of patients that are at high risk of ASCVD events.30–35 In the absence of a reliable clinical risk calculator in patients with IRD, consideration of additional risk enhancers, such as Lp(a), along with more liberal use of vascular imaging to screen for subclinical atherosclerosis appears to be the most effective way of risk stratification in patients that would otherwise be considered at low or intermediate risk by traditional risk calculators.
INTERSECTION WITH SEX AND GENDER
It is important to recognize that many IRDs, including RA and SLE, disproportionately affect women. Of the approximately 8% of the general population affected by IRDs, 80% are women,36 with many of them affected by these conditions starting at a young age, exposed to chronic inflammation, effect of medications, as well as debilitating physical symptoms and psychological burden of a disrupted sense of identity, loss of independence, societal stigma, and complicated reproductive health issues that may coexist. Traditional ASCVD risk calculators, blind to these important factors, would commonly place them in a low-risk category, leading to underestimation and undertreatment of their true ASCVD risk. This underscores the need for a more thorough ASCVD risk assessment in this population than offered by traditional risk calculators.
EFFECT OF MEDICATIONS USED IN IRD ON CV RISK
Medications used in the treatment of IRDs have the potential to further alter ASCVD risk. DMARDs are the cornerstone of treatment of inflammatory arthritis, aiming to control systemic inflammation leading to improvement in symptoms and prevention of joint and extra-articular damage. Corticosteroids and NSAIDs are typically used either as bridging therapy for short-term effects or together with DMARDs for control of symptoms and disease activity. Over the past two decades, a variety of targeted biologic and synthetic DMARD therapies have been approved for the management of rheumatic conditions. Despite effective control of inflammation, research has shown that different therapies have differential effects on CV risk. While medications such as systemic corticosteroids, NSAIDs, and some DMARDs, like leflunomide, may contribute to an increased CV risk, others, like methotrexate and anti-Tumour Necrosis Factor (TNF) therapies, have been shown to have cardioprotective effects.37–39 Corticosteroid use is associated with a variety of adverse CV effects including HTN, premature atherosclerosis, risk of MI, arrhythmias, and HF.8 However, observational studies in RA suggested that low-dose prednisone (<5 mg daily) used as maintenance therapy does not increase CV risk.40,41 Use of both nonselective and cyclo-oxygenase-2 (COX-2)-selective NSAIDs have been found to increase the risk of CV events. A number of studies, including a large nationwide Danish cohort study, have found that the CV risk associated with NSAID use in RA patients was significantly lower than in non-RA patients, possibly related to potential inflammatory mechanisms shared between RA and CV disease or better pain control and subsequent increase in mobility.42 Tofacitinib, a targeted synthetic DMARD that inhibits Janus Kinase (JAK), has been linked to higher risk of major adverse cardiovascular events (MACE) compared to anti-TNF therapy among patients with RA with a higher CV risk. In the ORAL surveillance study, a safety, non-inferiority trial mandated by the Food and Drug Administration (FDA), patients with RA aged ≥50 years who had at least 1 CV risk factor were randomized to tofacitinib or to a TNF inhibitor. The study showed that the risk of developing MACE and venous thromboembolism was higher in those on tofacitinib than TNF inhibitors, thus failing its non-inferiority criteria.43 This study led to the addition of a label warning by regulatory agencies, including Health Canada and the FDA, assigned to all JAK inhibitors for all clinical indications. A number of anti-inflammatory medications including canakinumab (anti-IL 1b) have been shown in randomized controlled trials to be effective in secondary prevention of ASCVD events in the general population44,45 but efficacy and safety with regards to CVD outcomes in IRD have not been studied. Finally, medications such as the antimalarial drugs, chloroquine and hydroxychloroquine, can have both important cardioprotective as well as uncommon, but potentially life-threatening cardiotoxic side effects in a small number of patients who develop cardiomyopathy, typically with long-term use.46
A FOCUS ON HYPERTENSION IN PATIENTS WITH IRD
HTN is a leading modifiable risk factor for CV death. Nearly half of adults in the US (48.1%, 119.9 million) have HTN, defined by the ACC/AHA guidelines as a systolic BP > 130 mmHg or a diastolic BP > 80 mmHg, or are taking medication for HTN.47,48 HTN control is a key component of ASCVD risk reduction and significantly lowers risk both in primary as well as in secondary prevention and reduces mortality from CV events.49,50
Numerous studies have reported a higher prevalence of HTN in patients with IRD than in the general population.51–55 The exact prevalence is difficult to determine with wide ranges reported in various studies, partially related to underdiagnosis of HTN in this population, impact of disease-specific therapies, varied sample size and significant differences in definition of HTN used over time and across jurisdictions due to variations in expert society guidelines and criteria. In patients with RA, studies have shown conflicting results with some case-control studies showing a significant increase in prevalence of HTN compared to healthy individuals,51,56 whereas other studies have not.57,58 The best evidence for higher prevalence of HTN comes from large community-based studies demonstrating that HTN was significantly more prevalent in patients with RA than the general population.51,59 One study found an increased prevalence of HTN in patients with RA (31%) compared to the general population (17%).51 In another study, prevalence of HTN in patients with RA was found to be 70% in both men and women of all age groups with a significant proportion (35% of males and 41% of females) undiagnosed and untreated, higher than that of the general population. Age, elevated BMI, and steroid use were independently associated with presence of HTN.55 Importantly, undiagnosed HTN was much more common in younger patients with RA.55 Similarly, in PsA and ankylosing spondylitis, large population-based case-control studies have demonstrated a higher prevalence of HTN compared to the general population.60–62 In SLE, HTN has been found to be more prevalent than in healthy individuals, particularly in younger women.54,63 HTN has been shown to be prevalent in patients with scleroderma, especially with renal involvement.64 Additionally, studies have shown that BP fluctuates considerably in patients with IRD due to variations in disease activity and subsequent change in medications including increased use of NSAIDs and corticosteroids, as well as potential kidney involvement in some IRDs at various stages of disease.65
Despite its high prevalence, HTN is underrecognized and undertreated among patients with IRD.16,53 Many patients are not prescribed appropriate antihypertensive therapy and many of those taking antihypertensive medications are not at the recommended treatment targets. In their study of frequencies of indication for CV preventative drugs in patients with inflammatory joint disease, Ikdahl et al. found an indication for antihypertensive treatment in 53% of patients of whom 48% were untreated. They also found that of those treated with antihypertensive medications, approximately 50% were not at guideline-recommended treatment targets recommended for the general population.66
There are multiple potential reasons for the underdiagnosis and undertreatment of HTN and other traditional CV risk factors in this patient population. Studies that have evaluated barriers to CV risk management among rheumatologists and PCPs have identified lack of clarity about who is responsible for CV risk management (rheumatologist or PCP), lack of time, lack of knowledge of current guidelines for CV risk management, and lack of care coordination as some of the main perceived reasons.67,68 Additionally, some clinicians may not be aware of the increased risk in patients with IRD. Due to frequent visits to their rheumatologist centered on joint and extra-articular symptoms, patients may not visit their PCP regularly and may be less regularly screened for various common risk factors. Finally, polypharmacy and “medication-fatigue” in patients with underlying chronic inflammatory disease can also contribute to this problem.
MECHANISMS OF HYPERTENSION IN IRD
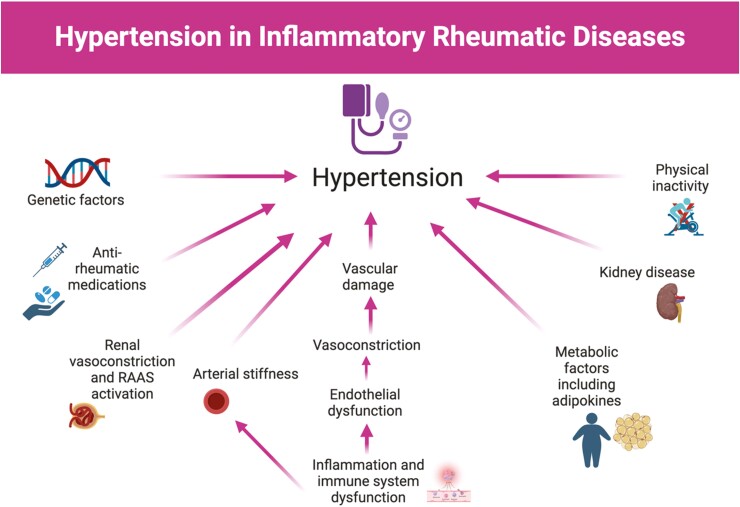
The mechanisms that contribute to HTN in patients with IRDs are multifactorial and are not fully understood but are at least in part dependent on disease-related factors including degree of inflammation, renal involvement, and disease-specific therapeutics (Figure 3). A number of overlapping genetic and environmental risk factors exist for development of both IRD and HTN.69 For example, adipokines such as leptin, secreted by white adipose tissue, have been implicated to have proinflammatory effects in autoimmune conditions such as SLE and RA.70 Leptin is also known to increase BP through impaired natriuresis, vascular endothelial dysfunction, oxidative stress, and increased renal tubular sodium absorption.71 Inflammation and immune-mediated damage of the vessel wall can lead to endothelial dysfunction and arterial stiffness leading to HTN.72 In conditions such as SLE, renal disease and progressive kidney dysfunction may also contribute to increased risk of HTN. Lupus nephritis occurs in nearly 50% of patients with SLE and has a strong correlation with HTN, though HTN may develop independently of lupus nephritis.73 Additionally, renal vasoconstriction and ischemia-induced activation of the renin–angiotensin–aldosterone system (RAAS) are important factors in development of HTN in vasculitis affecting renal arteries and in scleroderma renal crisis.
Figure 3.
Factors contributing to the development of hypertension in inflammatory rheumatic diseases; RAAS, renin–angiotensin–aldosterone system.
Physical inactivity exacerbated by joint pain, stiffness, and permanent joint damage may lead to a sedentary lifestyle and obesity, synergistically contributing to HTN in these patients. Recommendations for even mild physical exercise including walking or swimming that may be better tolerated in patients with joint disease should be encouraged. Design and implementation of specific structured exercise programs for these patients may provide more pronounced benefits.
IMPACT OF MEDICATIONS USED IN IRD ON HYPERTENSION
The effect of medications used for the treatment of underlying IRD must also be considered. All NSAIDs, both nonselective and selective, at doses required to control inflammation, can increase blood pressure though the effect on BP is variable.74–79 NSAIDs can also diminish the efficacy of many antihypertensive medications, with the exception of calcium channel blockers (CCB).74,75 There has been an ongoing interest in identifying which NSAIDs have the best safety profile with regard to HTN and CV outcomes, but this remains uncertain. A meta-analysis of randomized trials studying the effect of NSAIDs on blood pressure found that when pooled, NSAIDs elevated mean blood pressure by 5.0 mmHg (95% CI, 1.2–8.7 mmHg).76 Additionally, long-term NSAID use may also have a negative impact on kidney function, further contributing to HTN. Using the smallest dose required for the shortest duration of time possible with careful monitoring of BP is therefore recommended with all NSAIDs. There are no RCTs evaluating the effect of corticosteroids on BP in patients with IRD. Long-term (>6 months) and moderate-to-high doses (prednisone > 7.5 mg daily) have been associated with increased risk of developing HTN in RA, independently of other risk factors.80 The DMARD leflunomide has been reported to induce HTN in 2–5% of patients with RA, thought to be related to an increase in autonomic sympathetic tone, displacement of free fraction of any concomitant NSAID from protein binding, and increasing salt and water retention.81 Cyclosporine can also induce HTN through vasoconstrictive effect on the renal circulation resulting in reduced kidney function and should be avoided if possible in patients with HTN.82 The effect of biologic therapies on BP remains uncertain due to paucity of data, though TNF inhibitors have been shown to have beneficial effects on arterial stiffness in RA.83 In a small study of patients with RA, treatment with the TNF inhibitor infliximab was shown to result in a significant reduction in BP which was also associated with reduction in disease activity,84 although the study was limited by short duration of follow up.
BP TARGETS IN IRD
Despite known high prevalence of HTN and associated increased ASCVD risk, there is little data on optimal threshold for initiation and BP targets of therapy for patients with IRD and typically recommendations for the general population are applied to this group of patients. The 2023 European Society of Hypertension (ESH) guidelines recognize immune-mediated inflammatory diseases as being associated with increased prevalence of HTN that is underdiagnosed and poorly controlled, but no specific recommendations with regard to specific systolic and diastolic BP criteria for diagnosis, thresholds for initiation of therapy or for targets of therapy were suggested for this population. The 2017 ACC/AHA guidelines defined HTN at ≥ 130/80 mmHg, however, no special consideration or recommendation was provided for management of HTN in patients with IRDs. Tselios et al. studied the impact of this new definition of HTN on patients with SLE and showed that patients with a sustained mean BP of 130–139/80–89 mmHg over 2 years had a 2.5-fold increased risk of ASCVD events compared with those with BP < 130/80 mmHg. After adjustment for traditional and disease-related atherosclerotic risk factors, this level of BP conferred a 73% increased risk for atherosclerotic vascular events and therefore, they proposed a treatment BP target < 130/80 mmHg in patients with SLE to effectively reduce CV risk.85 The EULAR is also now recommending a target BP < 130/80 mmHg in patients with SLE. Due to paucity of data and in keeping with the ESH guidelines, the EULAR recommendation for management of HTN in other IRDs including RA, PsA, and ankylosing spondylitis is to follow recommendations used for the general population.22,25 BP thresholds for initiation of antihypertensive therapy and treatment targets, even in the general population, remain to be harmonized with variations across different practice guidelines.48,86 More research, particularly in the IRD population, is needed to define the best initiation threshold and targets of therapy.
CHOICE OF ANTIHYPERTENSIVE DRUGS IN IRD
The optimal choice of antihypertensive medication in patients with IRD is not well studied. EULAR recommends antihypertensive management in accordance with national guidelines for the general population. The 2023 ESH guidelines recommend lowering BP to targets recommended in the general population, preferentially with CCB and RAAS inhibitors because of the evidence of an overactive RAAS in these conditions,87 while also focusing on reducing systemic inflammation, and avoiding high dose NSAIDs. Multiple studies have demonstrated that concomitant use of nonselective NSAIDs attenuates the antihypertensive effect of angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, diuretics, and beta blockers.76,88,89 However, NSAIDs do not appear to alter the antihypertensive effect of CCBs90,91 and, therefore, in patients with HTN who require daily NSAID use, a dihydropyridine CCB such as amlodipine should be considered. Beta blockers may trigger or worsen psoriasis92 and should be used with caution if there is a compelling indication such as CHF. Similarly, use of beta blockers is avoided in patients with Raynaud’s as this can worsen symptoms and CCBs are favored due to their beneficial effects on Raynaud’s. Future research is needed to fill the knowledge gaps in this area and help delineate the most effective regimens in various IRDs with regard to BP control and long-term CV outcomes.
PRACTICAL IMPLICATIONS FOR SCREENING, DIAGNOSIS, AND TREATMENT OF HTN IN IRD
Given the high prevalence of HTN and its underdiagnosis and undertreatment in patients with IRD, it is prudent that patients undergo regular annual screening with their PCP and at visits with their rheumatologist, particularly after change in medications. Careful reevaluation of blood pressure and its control should also be considered at the time of development of comorbidities such as ASCVD, diabetes, or proteinuria. It is also advisable that for patients who are physically and financially able to do home BP measurements, to have ongoing self-monitoring to help exclude both white coat and masked hypertension. When unclear, use of Ambulatory Blood Pressure Monitoring can be helpful in more precise BP assessment, though access particularly related to cost coverage may be limiting in some jurisdictions. With regard to the choice of antihypertensive therapy, following societal guidelines with consideration of comorbidities, potential contraindications related to other conditions, and interactions with concurrent medications should be considered. As for targets of therapy, national societal guidelines recommendations are to be followed for the general population with the exception of a BP target of <130/80 mmHg for SLE given specific EULAR recommendations in this condition.25
CONCLUSIONS AND FUTURE DIRECTIONS
Individuals with IRD are at higher risk of developing ASCVD and experiencing worse CV outcomes than the general population. Despite significant advances over the last decade in understanding pathophysiological mechanisms underlying this increased risk, accurate ASCVD risk stratification and management remains suboptimal. Key overarching strategies for prevention of CV complications include aggressive management of traditional risk factors as well as achievement of effective control of systemic inflammation. Despite HTN being one of the most important and prevalent modifiable traditional risk factors in patients with IRD, it remains underdiagnosed, undertreated, and under-researched in this population. There is increasing recognition that an interdisciplinary model of care is crucial for optimal management of CV risk in these patients. Fostering collaborations between rheumatologists and cardiologists, such as that done in dedicated Cardio-Rheumatology programs, provides a valuable space for designing tailored screening protocols and management strategies for individuals with IRD, while allowing for combined research efforts to fill current knowledge gaps and improve quality of cardiovascular care offered to this patient group.
Contributor Information
Shadi Akhtari, Division of Cardiology, Department of Medicine, Women’s College Hospital, Toronto, ON, Canada; Women’s College Research Institute, Women’s College Hospital, Toronto, ON, Canada; Department of Medicine, University of Toronto, Toronto, ON, Canada.
Paula J Harvey, Division of Cardiology, Department of Medicine, Women’s College Hospital, Toronto, ON, Canada; Women’s College Research Institute, Women’s College Hospital, Toronto, ON, Canada; Department of Medicine, University of Toronto, Toronto, ON, Canada.
Lihi Eder, Division of Rheumatology, Department of Medicine, Women’s College Hospital, Toronto, ON, Canada; Women’s College Research Institute, Women’s College Hospital, Toronto, ON, Canada; Department of Medicine, University of Toronto, Toronto, ON, Canada.
FUNDING
The authors delared no source of funding.
CONFLICT OF INTEREST
S.A.: Honoraria: AbbVie and Novartis; P.J.H.: Honoraria for Continuing Medical Education from AbbVie and unrestricted grant funding support from Bios; L.E.: AbbVie, Novartis, UCB, Eli Lilly, Pfizer, Fresenius Kabi, BMS, Janssen—Advisory board/consult and grants.
DATA AVAILABILITY STATEMENT
No new data were generated or analysed in support of this research.
REFERENCES
- 1. Vaduganathan M, Mensah GA, Turco JV, Fuster V, Roth GA.. The global burden of cardiovascular diseases and risk: a compass for future health. J Am Coll Cardiol 2022; 80:2361–2371. [DOI] [PubMed] [Google Scholar]
- 2. Avina-Zubieta JA, Thomas J, Sadatsafavi M, Lehman AJ, Lacaille D.. Risk of incident cardiovascular events in patients with rheumatoid arthritis: a meta-analysis of observational studies. Ann Rheum Dis 2012; 71:1524–1529. [DOI] [PubMed] [Google Scholar]
- 3. Manzi S, Meilahn EN, Rairie JE, Conte CG, Medsger TA Jr, Jansen-McWilliams L, D'Agostino RB, Kuller LH.. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am J Epidemiol 1997; 145:408–415. [DOI] [PubMed] [Google Scholar]
- 4. Yafasova A, Fosbol EL, Schou M, Baslund B, Faurschou M, Docherty KF, Jhund PS, McMurray JJV, Sun G, Kristensen SL, Torp-Pedersen C, Køber L, Butt JH.. Long-term cardiovascular outcomes in systemic lupus erythematosus. J Am Coll Cardiol 2021; 77:1717–1727. [DOI] [PubMed] [Google Scholar]
- 5. Ciftci O, Yilmaz S, Topcu S, Caliskan M, Gullu H, Erdogan D, Pamuk BO, Yildirir A, Muderrisoglu H.. Impaired coronary microvascular function and increased intima-media thickness in rheumatoid arthritis. Atherosclerosis 2008; 198:332–337. [DOI] [PubMed] [Google Scholar]
- 6. Liao KP, Huang J, He Z, Cremone G, Lam E, Hainer JM, Morgan V, Bibbo C, Di Carli M.. Coronary microvascular dysfunction in rheumatoid arthritis compared to diabetes mellitus and association with all-cause mortality. Arthritis Care Res (Hoboken) 2021; 73:159–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Ikonomidis I, Lekakis JP, Nikolaou M, Paraskevaidis I, Andreadou I, Kaplanoglou T, Katsimbri P, Skarantavos G, Soucacos PN, Kremastinos DT.. Inhibition of interleukin-1 by anakinra improves vascular and left ventricular function in patients with rheumatoid arthritis. Circulation 2008; 117:2662–2669. [DOI] [PubMed] [Google Scholar]
- 8. Avina-Zubieta JA, Choi HK, Sadatsafavi M, Etminan M, Esdaile JM, Lacaille D.. Risk of cardiovascular mortality in patients with rheumatoid arthritis: a meta-analysis of observational studies. Arthritis Rheum 2008; 59:1690–1697. [DOI] [PubMed] [Google Scholar]
- 9. Palomaki A, Kerola AM, Malmberg M, Rautava P, Kyto V.. Patients with rheumatoid arthritis have impaired long-term outcomes after myocardial infarction: a nationwide case-control registry study. Rheumatology (Oxford) 2021; 60:5205–5215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Nicola PJ, Maradit-Kremers H, Roger VL, Jacobsen SJ, Crowson CS, Ballman KV, Gabriel SE.. The risk of congestive heart failure in rheumatoid arthritis: a population-based study over 46 years. Arthritis Rheum 2005; 52:412–420. [DOI] [PubMed] [Google Scholar]
- 11. Mantel A, Holmqvist M, Andersson DC, Lund LH, Askling J.. Association between rheumatoid arthritis and risk of ischemic and nonischemic heart failure. J Am Coll Cardiol 2017; 69:1275–1285. [DOI] [PubMed] [Google Scholar]
- 12. Ungprasert P, Srivali N, Kittanamongkolchai W.. Risk of incident atrial fibrillation in patients with rheumatoid arthritis: a systematic review and meta-analysis. Int J Rheum Dis 2017; 20:434–441. [DOI] [PubMed] [Google Scholar]
- 13. Maradit-Kremers H, Nicola PJ, Crowson CS, Ballman KV, Gabriel SE.. Cardiovascular death in rheumatoid arthritis: a population-based study. Arthritis Rheum 2005; 52:722–732. [DOI] [PubMed] [Google Scholar]
- 14. Polachek A, Touma Z, Anderson M, Eder L.. Risk of cardiovascular morbidity in patients with psoriatic arthritis: a meta-analysis of observational studies. Arthritis Care Res (Hoboken) 2017; 69:67–74. [DOI] [PubMed] [Google Scholar]
- 15. Koppikar S, Colaco K, Harvey P, Akhtari S, Chandran V, Gladman DD, et al. Incidence of and risk factors for heart failure in patients with psoriatic disease—a cohort study. Arthritis Care Res (Hoboken) 2022; 74(8):1244–1253. [DOI] [PubMed] [Google Scholar]
- 16. Eder L, Harvey P, Chandran V, Rosen CF, Dutz J, Elder JT, Rahman P, Ritchlin CT, Rohekar S, Hayday R, Barac S, Feld J, Zisman D, Gladman DD.. Gaps in diagnosis and treatment of cardiovascular risk factors in patients with psoriatic disease: an international multicenter study. J Rheumatol 2018; 45:378–384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kuriya B, Akhtari S, Movahedi M, Udell JA, Lawler PR, Farkouh ME, Keystone EC, Hanneman K, Nguyen E, Harvey PJ, Eder L.. Statin use for primary cardiovascular disease prevention is low in inflammatory arthritis. Can J Cardiol 2022; 38:1244–1252. [DOI] [PubMed] [Google Scholar]
- 18. Colaco K, Ocampo V, Ayala AP, Harvey P, Gladman DD, Piguet V, Eder L.. Predictive utility of cardiovascular risk prediction algorithms in inflammatory rheumatic diseases: a systematic review. J Rheumatol 2020; 47:928–938. [DOI] [PubMed] [Google Scholar]
- 19. Sivakumaran J, Harvey P, Omar A, Tayer-Shifman O, Urowitz MB, Gladman DD, Anderson N, Su J, Touma Z.. Assessment of cardiovascular risk tools as predictors of cardiovascular disease events in systemic lupus erythematosus. Lupus Sci Med 2021; 8:e000448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Kremers HM, Nicola PJ, Crowson CS, Ballman KV, Gabriel SE.. Prognostic importance of low body mass index in relation to cardiovascular mortality in rheumatoid arthritis. Arthritis Rheum 2004; 50:3450–3457. [DOI] [PubMed] [Google Scholar]
- 21. Myasoedova E, Crowson CS, Kremers HM, Roger VL, Fitz-Gibbon PD, Therneau TM, Gabriel SE.. Lipid paradox in rheumatoid arthritis: the impact of serum lipid measures and systemic inflammation on the risk of cardiovascular disease. Ann Rheum Dis 2011; 70:482–487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Agca R, Heslinga SC, Rollefstad S, Heslinga M, McInnes IB, Peters MJ, Kvien TK, Dougados M, Radner H, Atzeni F, Primdahl J, Södergren A, Wallberg Jonsson S, van Rompay J, Zabalan C, Pedersen TR, Jacobsson L, de Vlam K, Gonzalez-Gay MA, Semb AG, Kitas GD, Smulders YM, Szekanecz Z, Sattar N, Symmons DPM, Nurmohamed MT.. EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann Rheum Dis 2017; 76:17–28. [DOI] [PubMed] [Google Scholar]
- 23. Navarini L, Margiotta DPE, Caso F, Currado D, Tasso M, Angeletti S, Ciccozzi M, Scarpa R, Afeltra A, Costa L.. Performances of five risk algorithms in predicting cardiovascular events in patients with Psoriatic Arthritis: an Italian bicentric study. PLoS One 2018; 13:e0205506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Arts EE, Popa CD, Den Broeder AA, Donders R, Sandoo A, Toms T, Rollefstad S, Ikdahl E, Semb AG, Kitas GD, Van Riel PLCM, Fransen J.. Prediction of cardiovascular risk in rheumatoid arthritis: performance of original and adapted SCORE algorithms. Ann Rheum Dis 2016; 75:674–680. [DOI] [PubMed] [Google Scholar]
- 25. Drosos GC, Vedder D, Houben E, Boekel L, Atzeni F, Badreh S, Boumpas DT, Brodin N, Bruce IN, González-Gay MA, Jacobsen S, Kerekes G, Marchiori F, Mukhtyar C, Ramos-Casals M, Sattar N, Schreiber K, Sciascia S, Svenungsson E, Szekanecz Z, Tausche A-K, Tyndall A, van Halm V, Voskuyl A, Macfarlane GJ, Ward MM, Nurmohamed MT, Tektonidou MG.. EULAR recommendations for cardiovascular risk management in rheumatic and musculoskeletal diseases, including systemic lupus erythematosus and antiphospholipid syndrome. Ann Rheum Dis 2022; 81:768–779. [DOI] [PubMed] [Google Scholar]
- 26. Grundy SM, Stone NJ, Bailey AL, Beam C, Birtcher KK, Blumenthal RS, Braun LT, de Ferranti S, Faiella-Tommasino J, Forman DE, Goldberg R, Heidenreich PA, Hlatky MA, Jones DW, Lloyd-Jones D, Lopez-Pajares N, Ndumele CE, Orringer CE, Peralta CA, Saseen JJ, Smith SC, Sperling L, Virani SS, Yeboah J.. 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: a report of the American College of Cardiology/American Heart Association task force on clinical practice guidelines. Circulation 2019; 139:e1082–e1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Pearson GJ, Thanassoulis G, Anderson TJ, Barry AR, Couture P, Dayan N, Francis GA, Genest J, Grégoire J, Grover SA, Gupta M, Hegele RA, Lau D, Leiter LA, Leung AA, Lonn E, Mancini GBJ, Manjoo P, McPherson R, Ngui D, Piché M-E, Poirier P, Sievenpiper J, Stone J, Ward R, Wray W.. 2021 Canadian Cardiovascular Society Guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in adults. Can J Cardiol 2021; 37:1129–1150. [DOI] [PubMed] [Google Scholar]
- 28. Zhang W, Speiser JL, Ye F, Tsai MY, Cainzos-Achirica M, Nasir K, Herrington DM, Shapiro MD.. High-sensitivity C-reactive protein modifies the cardiovascular risk of lipoprotein(a): multi-ethnic study of atherosclerosis. J Am Coll Cardiol 2021; 78:1083–1094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Puri R, Nissen SE, Arsenault BJ, St John J, Riesmeyer JS, Ruotolo G, McErlean E, Menon V, Cho L, Wolski K, Lincoff AM, Nicholls SJ.. Effect of C-reactive protein on lipoprotein(a)-associated cardiovascular risk in optimally treated patients with high-Risk vascular disease: a prespecified secondary analysis of the ACCELERATE trial. JAMA Cardiol 2020; 5:1136–1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Peters SA, den Ruijter HM, Bots ML, Moons KG.. Improvements in risk stratification for the occurrence of cardiovascular disease by imaging subclinical atherosclerosis: a systematic review. Heart 2012; 98:177–184. [DOI] [PubMed] [Google Scholar]
- 31. McClelland RL, Jorgensen NW, Budoff M, Blaha MJ, Post WS, Kronmal RA, Bild DE, Shea S, Liu K, Watson KE, Folsom AR, Khera A, Ayers C, Mahabadi A-A, Lehmann N, Jöckel K-H, Moebus S, Carr JJ, Erbel R, Burke GL.. 10-Year coronary heart disease risk prediction using coronary artery calcium and traditional risk factors: derivation in the MESA (Multi-Ethnic Study of Atherosclerosis) with validation in the HNR (Heinz Nixdorf Recall) study and the DHS (Dallas Heart Study). J Am Coll Cardiol 2015; 66:1643–1653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Galarza-Delgado DA, Azpiri-Lopez JR, Colunga-Pedraza IJ, Guajardo-Jauregui N, Rodriguez-Romero AB, Lugo-Perez S, Cardenas-de la Garza JA, Arvizu-Rivera RI, Flores-Alvarado DE, Ilizaliturri-Guerra O, Garcia-Arellano G, Garza-Acosta AC.. Cardiovascular risk reclassification according to six cardiovascular risk algorithms and carotid ultrasound in psoriatic arthritis patients. Clin Rheumatol 2022; 41:1413–1420. [DOI] [PubMed] [Google Scholar]
- 33. Sobchak C, Akhtari S, Harvey P, Gladman D, Chandran V, Cook R, Eder L.. Value of carotid ultrasound in cardiovascular risk stratification in patients with psoriatic disease. Arthritis Rheumatol 2019; 71:1651–1659. [DOI] [PubMed] [Google Scholar]
- 34. Corrales A, Gonzalez-Juanatey C, Peiro ME, Blanco R, Llorca J, Gonzalez-Gay MA.. Carotid ultrasound is useful for the cardiovascular risk stratification of patients with rheumatoid arthritis: results of a population-based study. Ann Rheum Dis 2014; 73:722–727. [DOI] [PubMed] [Google Scholar]
- 35. Evans MR, Escalante A, Battafarano DF, Freeman GL, O’Leary DH, del Rincon I.. Carotid atherosclerosis predicts incident acute coronary syndromes in rheumatoid arthritis. Arthritis Rheum 2011; 63:1211–1220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Mavrogeni SI, Markousis-Mavrogenis G, Koutsogeorgopoulou L, Dimitroulas T, Vartela V, Rigopoulos A, Noutsias M, Kolovou G.. Pathophysiology and imaging of heart failure in women with autoimmune rheumatic diseases. Heart Fail Rev 2019; 24:489–498. [DOI] [PubMed] [Google Scholar]
- 37. Westlake SL, Colebatch AN, Baird J, Kiely P, Quinn M, Choy E, Ostor AJK, Edwards CJ.. The effect of methotrexate on cardiovascular disease in patients with rheumatoid arthritis: a systematic literature review. Rheumatology (Oxford) 2010; 49:295–307. [DOI] [PubMed] [Google Scholar]
- 38. Choi HK, Hernan MA, Seeger JD, Robins JM, Wolfe F.. Methotrexate and mortality in patients with rheumatoid arthritis: a prospective study. Lancet 2002; 359:1173–1177. [DOI] [PubMed] [Google Scholar]
- 39. Guin A, Chatterjee Adhikari M, Chakraborty S, Sinhamahapatra P, Ghosh A.. Effects of disease modifying anti-rheumatic drugs on subclinical atherosclerosis and endothelial dysfunction which has been detected in early rheumatoid arthritis: 1-year follow-up study. Semin Arthritis Rheum 2013; 43:48–54. [DOI] [PubMed] [Google Scholar]
- 40. So H, Lam TO, Meng H, Lam SHM, Tam LS.. Time and dose-dependent effect of systemic glucocorticoids on major adverse cardiovascular event in patients with rheumatoid arthritis: a population-based study. Ann Rheum Dis 2023; 82:1387–1393. [DOI] [PubMed] [Google Scholar]
- 41. Ocon AJ, Reed G, Pappas DA, Curtis JR, Kremer JM.. Short-term dose and duration-dependent glucocorticoid risk for cardiovascular events in glucocorticoid-naive patients with rheumatoid arthritis. Ann Rheum Dis 2021; 80:1522–1529. [DOI] [PubMed] [Google Scholar]
- 42. Lindhardsen J, Gislason GH, Jacobsen S, Ahlehoff O, Olsen AM, Madsen OR, Torp-Pedersen C, Hansen PR.. Non-steroidal anti-inflammatory drugs and risk of cardiovascular disease in patients with rheumatoid arthritis: a nationwide cohort study. Ann Rheum Dis 2014; 73:1515–1521. [DOI] [PubMed] [Google Scholar]
- 43. Ytterberg SR, Bhatt DL, Mikuls TR, Koch GG, Fleischmann R, Rivas JL, Germino R, Menon S, Sun Y, Wang C, Shapiro AB, Kanik KS, Connell CA; ORAL Surveillance Investigators. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med 2022; 386:316–326. [DOI] [PubMed] [Google Scholar]
- 44. Tardif JC, Kouz S, Waters DD, Bertrand OF, Diaz R, Maggioni AP, Pinto FJ, Ibrahim R, Gamra H, Kiwan GS, Berry C, López-Sendón J, Ostadal P, Koenig W, Angoulvant D, Grégoire JC, Lavoie M-A, Dubé M-P, Rhainds D, Provencher M, Blondeau L, Orfanos A, L'Allier PL, Guertin M-C, Roubille F.. Efficacy and safety of low-dose colchicine after myocardial infarction. N Engl J Med 2019; 381:2497–2505. [DOI] [PubMed] [Google Scholar]
- 45. Ridker PM, Everett BM, Thuren T, MacFadyen JG, Chang WH, Ballantyne C, Fonseca F, Nicolau J, Koenig W, Anker SD, Kastelein JJP, Cornel JH, Pais P, Pella D, Genest J, Cifkova R, Lorenzatti A, Forster T, Kobalava Z, Vida-Simiti L, Flather M, Shimokawa H, Ogawa H, Dellborg M, Rossi PRF, Troquay RPT, Libby P, Glynn RJ; CANTOS Trial Group. Antiinflammatory therapy with Canakinumab for atherosclerotic disease. N Engl J Med 2017; 377:1119–1131. [DOI] [PubMed] [Google Scholar]
- 46. Tselios K, Deeb M, Gladman DD, Harvey P, Urowitz MB.. Antimalarial-induced cardiomyopathy: a systematic review of the literature. Lupus 2018; 27:591–599. [DOI] [PubMed] [Google Scholar]
- 47. Ritchey MD, Gillespie C, Wozniak G, Shay CM, Thompson-Paul AM, Loustalot F, Hong Y.. Potential need for expanded pharmacologic treatment and lifestyle modification services under the 2017 ACC/AHA Hypertension Guideline. J Clin Hypertens (Greenwich) 2018; 20:1377–1391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Whelton PK, Carey RM, Aronow WS, Casey DE Jr, Collins KJ, Dennison Himmelfarb C, DePalma SM, Gidding S, Jamerson KA, Jones DW, MacLaughlin EJ, Muntner P, Ovbiagele B, Smith SC, Spencer CC, Stafford RS, Taler SJ, Thomas RJ, Williams KA, Williamson JD, Wright JT.. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2018; 71:e127–e248. [DOI] [PubMed] [Google Scholar]
- 49. Karmali KN, Lloyd-Jones DM, Berendsen MA, Goff DC Jr, Sanghavi DM, Brown NC, Korenovska L, Huffman MD.. Drugs for primary prevention of atherosclerotic cardiovascular disease: an overview of systematic reviews. JAMA Cardiol 2016; 1:341–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Bansilal S, Castellano JM, Fuster V.. Global burden of CVD: focus on secondary prevention of cardiovascular disease. Int J Cardiol 2015; 201:S1–S7. [DOI] [PubMed] [Google Scholar]
- 51. Han C, Robinson DW Jr, Hackett MV, Paramore LC, Fraeman KH, Bala MV.. Cardiovascular disease and risk factors in patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis. J Rheumatol 2006; 33:2167–2172. [PubMed] [Google Scholar]
- 52. Nikpour M, Urowitz MB, Ibanez D, Harvey PJ, Gladman DD.. Importance of cumulative exposure to elevated cholesterol and blood pressure in development of atherosclerotic coronary artery disease in systemic lupus erythematosus: a prospective proof-of-concept cohort study. Arthritis Res Ther 2011; 13:R156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Al-Herz A, Ensworth S, Shojania K, Esdaile JM.. Cardiovascular risk factor screening in systemic lupus erythematosus. J Rheumatol 2003; 30:493–496. [PubMed] [Google Scholar]
- 54. Sabio JM, Vargas-Hitos JA, Navarrete-Navarrete N, Mediavilla JD, Jimenez-Jaimez J, Diaz-Chamorro A, Jiménez-Alonso J; Grupo Lupus Virgen de las Nieves. Prevalence of and factors associated with hypertension in young and old women with systemic lupus erythematosus. J Rheumatol 2011; 38:1026–1032. [DOI] [PubMed] [Google Scholar]
- 55. Panoulas VF, Douglas KM, Milionis HJ, Stavropoulos-Kalinglou A, Nightingale P, Kita MD, Tselios AL, Metsios GS, Elisaf MS, Kitas GD.. Prevalence and associations of hypertension and its control in patients with rheumatoid arthritis. Rheumatology (Oxford) 2007; 46:1477–1482. [DOI] [PubMed] [Google Scholar]
- 56. Chung CP, Oeser A, Solus JF, Avalos I, Gebretsadik T, Shintani A, Raggi P, Sokka T, Pincus T, Stein CM.. Prevalence of the metabolic syndrome is increased in rheumatoid arthritis and is associated with coronary atherosclerosis. Atherosclerosis 2008; 196:756–763. [DOI] [PubMed] [Google Scholar]
- 57. Muller R, Kull M, Polluste K, Aart A, Eglit T, Lember M, Kallikorm R.. The metabolic profile in early rheumatoid arthritis: a high prevalence of metabolic obesity. Rheumatol Int 2017; 37:21–27. [DOI] [PubMed] [Google Scholar]
- 58. Solomon DH, Curhan GC, Rimm EB, Cannuscio CC, Karlson EW.. Cardiovascular risk factors in women with and without rheumatoid arthritis. Arthritis Rheum 2004; 50:3444–3449. [DOI] [PubMed] [Google Scholar]
- 59. Siebert S, Lyall DM, Mackay DF, Porter D, McInnes IB, Sattar N, Pell JP.. Characteristics of rheumatoid arthritis and its association with major comorbid conditions: cross-sectional study of 502 649 UK Biobank participants. RMD Open 2016; 2:e000267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Jafri K, Bartels CM, Shin D, Gelfand JM, Ogdie A.. Incidence and management of cardiovascular risk factors in psoriatic arthritis and rheumatoid arthritis: a population-based study. Arthritis Care Res (Hoboken) 2017; 69:51–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Ahmed N, Prior JA, Chen Y, Hayward R, Mallen CD, Hider SL.. Prevalence of cardiovascular-related comorbidity in ankylosing spondylitis, psoriatic arthritis and psoriasis in primary care: a matched retrospective cohort study. Clin Rheumatol 2016; 35:3069–3073. [DOI] [PubMed] [Google Scholar]
- 62. Haque N, Lories RJ, de Vlam K.. Comorbidities associated with psoriatic arthritis compared with non-psoriatic spondyloarthritis: a cross-sectional study. J Rheumatol 2016; 43:376–382. [DOI] [PubMed] [Google Scholar]
- 63. Bruce IN, Urowitz MB, Gladman DD, Ibanez D, Steiner G.. Risk factors for coronary heart disease in women with systemic lupus erythematosus: the Toronto Risk Factor Study. Arthritis Rheum 2003; 48:3159–3167. [DOI] [PubMed] [Google Scholar]
- 64. Shanmugam VK, Steen VD.. Renal manifestations in scleroderma: evidence for subclinical renal disease as a marker of vasculopathy. Int J Rheumatol 2010; 2010:1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Tselios K, Koumaras C, Urowitz MB, Gladman DD.. Do current arterial hypertension treatment guidelines apply to systemic lupus erythematosus patients? A critical appraisal. Semin Arthritis Rheum 2014; 43:521–525. [DOI] [PubMed] [Google Scholar]
- 66. Ikdahl E, Wibetoe G, Rollefstad S, Salberg A, Bergsmark K, Kvien TK, Olsen IC, Soldal DM, Bakland G, Lexberg A, Fevang BTS, Gulseth HC, Haugeberg G, Semb AG.. Guideline recommended treatment to targets of cardiovascular risk is inadequate in patients with inflammatory joint diseases. Int J Cardiol 2019; 274:311–318. [DOI] [PubMed] [Google Scholar]
- 67. Navarro-Millan I, Cornelius-Schecter A, O’Beirne RJ, Morris MS, Lui GE, Goodman SM, Cherrington AL, Fraenkel L, Curtis JR, Safford MM.. Views of primary care physicians and rheumatologists regarding screening and treatment of hyperlipidemia among patients with rheumatoid arthritis. BMC Rheumatol 2020; 4:14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Esmaeilbeigi F, Pope JE.. Appropriate cardiovascular disease risk assessment in systemic lupus erythematosus may be lacking in rheumatology practice. Clin Exp Rheumatol 2018; 36:526–532. [PubMed] [Google Scholar]
- 69. Barbhaiya M, Costenbader KH.. Environmental exposures and the development of systemic lupus erythematosus. Curr Opin Rheumatol 2016; 28:497–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Tian G, Liang JN, Wang ZY, Zhou D.. Emerging role of leptin in rheumatoid arthritis. Clin Exp Immunol 2014; 177:557–570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Koh KK, Park SM, Quon MJ.. Leptin and cardiovascular disease: response to therapeutic interventions. Circulation 2008; 117:3238–3249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Bartoloni E, Alunno A, Gerli R.. Hypertension as a cardiovascular risk factor in autoimmune rheumatic diseases. Nat Rev Cardiol 2018; 15:33–44. [DOI] [PubMed] [Google Scholar]
- 73. Bertsias GK, Tektonidou M, Amoura Z, Aringer M, Bajema I, Berden JH, Boletis J, Cervera R, Dörner T, Doria A, Ferrario F, Floege J, Houssiau FA, Ioannidis JPA, Isenberg DA, Kallenberg CGM, Lightstone L, Marks SD, Martini A, Moroni G, Neumann I, Praga M, Schneider M, Starra A, Tesar V, Vasconcelos C, van Vollenhoven RF, Zakharova H, Haubitz M, Gordon C, Jayne D, Boumpas DT; European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association. Joint European League Against Rheumatism and European Renal Association-European Dialysis and Transplant Association (EULAR/ERA-EDTA) recommendations for the management of adult and paediatric lupus nephritis. Ann Rheum Dis 2012; 71:1771–1782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. White WB. Cardiovascular effects of the cyclooxygenase inhibitors. Hypertension 2007; 49:408–418. [DOI] [PubMed] [Google Scholar]
- 75. Warner TD, Mitchell JA.. COX-2 selectivity alone does not define the cardiovascular risks associated with non-steroidal anti-inflammatory drugs. Lancet 2008; 371:270–273. [DOI] [PubMed] [Google Scholar]
- 76. Johnson AG, Nguyen TV, Day RO.. Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann Intern Med 1994; 121:289–300. [DOI] [PubMed] [Google Scholar]
- 77. Morrison A, Ramey DR, van Adelsberg J, Watson DJ.. Systematic review of trials of the effect of continued use of oral non-selective NSAIDs on blood pressure and hypertension. Curr Med Res Opin 2007; 23:2395–2404. [DOI] [PubMed] [Google Scholar]
- 78. Pope JE, Anderson JJ, Felson DT.. A meta-analysis of the effects of nonsteroidal anti-inflammatory drugs on blood pressure. Arch Intern Med 1993; 153:477–484. [PubMed] [Google Scholar]
- 79. Ruschitzka F, Borer JS, Krum H, Flammer AJ, Yeomans ND, Libby P, Lüscher TF, Solomon DH, Husni ME, Graham DY, Davey DA, Wisniewski LM, Menon V, Fayyad R, Beckerman B, Iorga D, Lincoff AM, Nissen SE.. Differential blood pressure effects of ibuprofen, naproxen, and celecoxib in patients with arthritis: the PRECISION-ABPM (Prospective Randomized Evaluation of Celecoxib Integrated Safety Versus Ibuprofen or Naproxen Ambulatory Blood Pressure Measurement) Trial. Eur Heart J 2017; 38:3282–3292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Panoulas VF, Douglas KM, Stavropoulos-Kalinoglou A, Metsios GS, Nightingale P, Kita MD, Elisaf MS, Kitas GD.. Long-term exposure to medium-dose glucocorticoid therapy associates with hypertension in patients with rheumatoid arthritis. Rheumatology (Oxford) 2008; 47:72–75. [DOI] [PubMed] [Google Scholar]
- 81. Fox RI, Herrmann ML, Frangou CG, Wahl GM, Morris RE, Strand V, Kirschbaum BJ.. Mechanism of action for leflunomide in rheumatoid arthritis. Clin Immunol 1999; 93:198–208. [DOI] [PubMed] [Google Scholar]
- 82. Panoulas VF, Metsios GS, Pace AV, John H, Treharne GJ, Banks MJ, Kitas GD.. Hypertension in rheumatoid arthritis. Rheumatology (Oxford) 2008; 47:1286–1298. [DOI] [PubMed] [Google Scholar]
- 83. Agca R, Heslinga SC, van Halm VP, Nurmohamed MT.. Atherosclerotic cardiovascular disease in patients with chronic inflammatory joint disorders. Heart 2016; 102:790–795. [DOI] [PubMed] [Google Scholar]
- 84. Yoshida S, Takeuchi T, Kotani T, Yamamoto N, Hata K, Nagai K, Shoda T, Takai S, Makino S, Hanafusa T.. Infliximab, a TNF-alpha inhibitor, reduces 24-h ambulatory blood pressure in rheumatoid arthritis patients. J Hum Hypertens 2014; 28:165–169. [DOI] [PubMed] [Google Scholar]
- 85. Tselios K, Gladman DD, Su J, Urowitz M.. Impact of the new American College of Cardiology/American Heart Association definition of hypertension on atherosclerotic vascular events in systemic lupus erythematosus. Ann Rheum Dis 2020; 79:612–617. [DOI] [PubMed] [Google Scholar]
- 86. Mancia G, Kreutz R, Brunstrom M, Burnier M, Grassi G, Januszewicz A, Muiesan ML, Tsioufis K, Agabiti-Rosei E, Algharably EAE, Azizi M, Benetos A, Borghi C, Hitij JB, Cifkova R, Coca A, Cornelissen V, Cruickshank JK, Cunha PG, Danser AHJ, de Pinho Rosa Maria, Delles C, Dominiczak AF, Dorobantu M, Doumas M, Fernández-Alfonso MS, Halimi J-M, Járai Z, Jelaković B, Jordan J, Kuznetsova T, Laurent S, Lovic D, Lurbe E, Mahfoud F, Manolis A, Miglinas M, Narkiewicz K, Niiranen T, Palatini P, Parati G, Pathak A, Persu A, Polonia J, Redon J, Sarafidis P, Schmieder R, Spronck B, Stabouli S, Stergiou G, Taddei S, Thomopoulos C, Tomaszewski M, Van de Borne P, Wanner C, Weber T, Williams B, Zhang Z-Y, Kjeldsen SE.. 2023 ESH Guidelines for the management of arterial hypertension The Task Force for the management of arterial hypertension of the European Society of Hypertension: endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA). J Hypertens 2023; 41:1874–2071. [DOI] [PubMed] [Google Scholar]
- 87. Flammer AJ, Sudano I, Hermann F, Gay S, Forster A, Neidhart M, Künzler P, Enseleit F, Périat D, Hermann M, Nussberger J, Luscher TF, Corti R, Noll G, Ruschitzka F.. Angiotensin-converting enzyme inhibition improves vascular function in rheumatoid arthritis. Circulation 2008; 117:2262–2269. [DOI] [PubMed] [Google Scholar]
- 88. Wong DG, Spence JD, Lamki L, Freeman D, McDonald JW.. Effect of non-steroidal anti-inflammatory drugs on control of hypertension by beta-blockers and diuretics. Lancet 1986; 1:997–1001. [DOI] [PubMed] [Google Scholar]
- 89. Fogari R, Zoppi A, Carretta R, Veglio F, Salvetti A, Italian Collaborative Study G.. Effect of indomethacin on the antihypertensive efficacy of valsartan and lisinopril: a multicentre study. J Hypertens 2002; 20:1007–1014. [DOI] [PubMed] [Google Scholar]
- 90. Klassen DK, Jane LH, Young DY, Peterson CA.. Assessment of blood pressure during naproxen therapy in hypertensive patients treated with nicardipine. Am J Hypertens 1995; 8:146–153. [DOI] [PubMed] [Google Scholar]
- 91. Morgan T, Anderson A.. Interaction of indomethacin with felodipine and enalapril. J Hypertens Suppl 1993; 11:S338–S339. [PubMed] [Google Scholar]
- 92. Azzouz B, De Guizelin A, Lambert A, Fresse A, Morel A, Trenque T.. Psoriasis risk after beta-blocker exposure: description of a pharmacovigilance signal. Br J Clin Pharmacol 2022; 88:3813–3818. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were generated or analysed in support of this research.