Abstract
The brain-derived neurotrophic factor (BDNF) gene plays an important role in modulating the stress-response axis and inflammation, which can be regulated by epigenetic mechanisms. BNDF methylation has been associated with stress-related psychiatric disorders such as depression, anxiety and post-traumatic stress. Previous studies have reported that stressful events are involved with long-lasting alterations in DNA methylation (DNAm) of the BNDF exon IV promoter, suggesting that glucocorticoids and inflammatory cytokines can regulate this process. We previously found that perceived psychological stress is modulated by inflammatory cytokines, such as interleukin (IL)-6, IL-8 and IL-10, and IL-12p70, suggesting their role in mediating the stress response. However, the epigenetic mechanism mediating this response has yet to be fully understood. In this study, we propose that high perceived stress and high serum levels of inflammatory cytokines may correlate with specific methylation sites within the BNDF exon IV promoter. To address these questions, we conducted a cross-sectional study of 82 adult women teachers working in basic education in Brazil. The perceived stress scale was used to assess stress and blood samples were collected for the measurement of inflammatory markers and BNDF methylation through flow cytometry assay and DNA pyrosequencing, respectively. We detected differentially methylated CpG sites in the BNDF gene, where 5 CpG sites were directly correlated with high stress levels. However, 4 CpG sites showed inverse effects, indicating that changes in methylation levels in those sites could lead to a protective effect on perceived stress. About inflammatory markers, IL-6 and IL-8 were associated with high perceived stress. However, only IL-8 and IL-10 showed simultaneous modulation of perceived stress, while IL-10 and IL12p70 correlated with DNAm. We found that higher levels in IL-10 and IL-12p70 serum decrease methylation in CpG11. A direct relationship was also found to IL-12p70, where higher levels in serum increase methylation in CpG5 and 13, respectively. Taken as a whole, our findings reinforce the hypothesis regarding stress-sensitive regions within the BDNF gene, mainly for CpG5, 11, and 13. In addition to these results, CpG7 and 9 may be regarded as stress-protective regions. Our data suggest that BDNF DNAm in the blood may represent a novel biomarker for early detection of adverse effects of chronic exposure to stress in healthy individuals.
Keywords: BDNF DNA methylation, Stress, Inflammatory cytokines, Healthy
Highlights
-
•
Our study identifies specific methylation sites within the BDNF gene correlated with stress levels and inflammatory cytokines, highlighting CpG5, 11, and 13 as stress-sensitive regions and CpG7 and 9 as stress-protective regions, suggesting BDNF DNAm as a potential biomarker for chronic stress.
-
•
IL-6 and IL-8 were associated with high perceived stress, and IL-8 and IL-10 showed simultaneous modulation of perceived stress, with IL-10 and IL-12p70 influencing BDNF methylation levels.
-
•
BDNF DNA methylation in blood samples could serve as a novel biomarker for early detection of the adverse effects of chronic stress exposure in healthy individuals.
1. Introduction
Stress can arise from a wide range of adverse life experiences and has long been recognized as playing a crucial role in the health/disease interface. The impact of stress is influenced by individual responses to stressful stimuli, such as genetic factors and situational perceptions, as well as the nature of the stressor, including its duration and timing of its occurrence (Epel et al., 2018; McEwen, 2012; Slavich et al., 2023). This complex interplay has been frequently associated with the onset and progression of psychiatric disorders like depression, anxiety, and post-traumatic stress (PTSD) disorders (Cohen et al., 2016; Cohen et al., 2007; Mazure et al., 2023; McEwen and Akil, 2020). Despite this, the cellular and molecular mechanisms underlying these stress-related psychopathologies remain unknown. Epigenetic mechanisms, however, emerge as promising and plausible candidates for explaining how exposure to chronic stress leads to long-lasting DNA methylation (DNAm) changes, which can subsequently affect gene expression by modifying the accessibility of the DNA to transcriptional machinery. This regulation is particularly relevant in the context of stress.
Brain-derived neurotrophic factor (BDNF) is a neurotrophic protein essential in neuronal development, maintenance, and synaptic plasticity (Gray et al., 2013; Notaras and van den Buuse, 2020; Wang et al., 2022). Numerous studies have shown that BDNF gene and protein expression changes significantly in response to stress (de Assis and Gasanov, 2019; Herhaus et al., 2023; Hötting et al., 2016; Lambert et al., 2013; Linz et al., 2019). Additionally, BDNF plays a crucial role in modulating the body's stress response by interacting directly with the hypothalamic-pituitary-adrenal (HPA) axis (Chen et al., 2017; Gray et al., 2013; Lambert et al., 2013; Tapia-Arancibia et al., 2004). Research has also identified an association between stress-related events and BNDF DNAm in humans (Bakusic et al., 2020; Song et al., 2014) and rodents (Blaze et al., 2017; Dong et al., 2015; Roth et al., 2011). Given its association with psychiatric disorders, BDNF DNA methylation is considered a valuable marker for tracking the onset of stress-related mental health issues in clinical settings (Ikegame et al., 2013a; Lin and Huang, 2020; Miao et al., 20200; Song et al., 2014; Zheleznyakova et al., 2016b).
The human BNDF gene is located on chromosome 11p14.1 (Ikegame et al., 2013a; Özçelik et al., 1991). It has a complex structure with 11 exons (I-V, Vh, VI-VIII, VIIIh, IX), 9 of which exons (I–VII, IX) contain functional promoters (Ikegame et al., 2013a; Zheleznyakova et al., 2016b). The promoter region of exon IV has gained interest in recent years, as it has shown that this region is especially sensitive to environmental stressors, and its methylation status can significantly alter BDNF gene expression. Studies have shown increased methylation levels in patients with depression when compared to healthy controls in this region (Ferrer et al., 2019b; Januar et al., 2015). Interestingly, an elevated level of BDNF methylation (BNDFm) at promoter IV has been associated with burnout and depressive symptoms compared to a healthy control group (Bakusic et al., 2020). Studying these epigenetic changes in healthy individuals is essential. It offers a baseline to understand how DNAm patterns shift over time, revealing early biomarkers of stress susceptibility and offering valuable insights into molecular changes that lead to psychiatric disorders.
Regarding the association between immune function, BNDF, and psychological stress is well-established (Calabrese et al., 2014; Carpenter et al., 2010; Cattaneo et al., 2016; Knight et al., 2021). Evidence suggests that inflammation can directly modify DNA methylation patterns, contributing to the development of stress-related disorders like depression in humans (Crawford et al., 2018) and depression-like behavior in rodents (Zhao et al., 2023). Other factors related to inter-individual variability as body mass index (BMI), age, smoking, and depression or anxiety symptoms influencing DNA methylation (Ferrer et al., 2019b; He et al., 2019; Ikegame et al., 2013a; Quaioto et al., 2023; Song et al., 2014).
To our knowledge, our study is the first one to focus specifically on the involvement of BDNF DNAm in perceived stress and immune function. We hypothesized that the specific methylation site of BDNF gene promoter IV could be associated with high levels of perceived stress and inflammatory markers. Our goal was to explore the influence of each specific CpG site within the BDNF gene promoter IV on perceived psychological stress and immune function, considering inter-individual variability as a potential external modulator. We also hypothesize that BDNF methylation could act as a modulator in stress inflammation, potentially influencing individual responses to stress. We addressed these questions in a group of women teachers who were presently active (i.e., were not at health treatment license and not impaired in their functions) at several elementary basic public schools, having made use of peripheral blood cells to study such biomarkers.
2. Material and methods
Study design and participants – The sample partially overlaps the one used in the previous study (de Oliveira et al., 2021), which explored different hypotheses. Eighty-two teachers actively working in public schools, who had participated in previous studies and were previously excluded for practicing current meditation, yoga, or Tai-Chi practices, were included in this study, following the updated inclusion and exclusion criteria. Inclusion criteria were: (a) being Brazilian; (b) woman; (c) age between 23 and 65 years, and (d) actively teaching at the time of the study. Exclusion criteria: (a) presence of a clinical condition that prevents participation in activities; (b) psychiatric condition that is life-threatening; (c) suicidal ideation present at the time of the study; (d) use of psychotropic drugs; (e) presence of an inflammatory process in the last 2 weeks prior to the study; and (f) use of anti-inflammatories or antibiotics.
This study utilized the Hospital Anxiety and Depression Scale (HADS), Perceived Stress Scale (PSS), and questionnaires on socioeconomic, demographic, and lifestyle factors, along with blood samples, all of which were part of our established database. The current participants agree to provide blood samples for DNA extraction, inflammatory markers, and DNA methylation studies. The research protocol for a cross-sectional study was approved by the Research Ethics Committee of the Universidade Federal de São Paulo – UNIFESP, Brazil (Number: 2.825.371 - CAAE: 92876418.2.0000.5505). All participants provided written informed consent after receiving a full explanation of the study. The study was registered (https://www.clinicaltrials.gov/ct2/show/NCT04955457) under the number NCT04955457 in August 2018.
2.1. Stress assessment
Perceived Stress Scale (PSS-10) is an instrument used to measure how much the events of an individual's life, according to their perception, seem stressful (subjective stress assessment). The test consists of 10 items, 4 of which are positive aspects and 6 are negative aspects and must be answered within a Likert-type frequency scale (ranging from 0 – Never, to 4 – Always). For its evaluation, all responses must be added to obtain a total perceived stress score, where zero is the lowest score and forty is the highest. The higher the score, the higher the level of perceived stress. As for its validation, the internal consistency was verified using Cronbach's alpha coefficient and the construct validity, by exploratory factor analysis with orthogonal rotation by the varimax method, and the complete version showed similar internal consistency (r = 0.83) (Reis et al., 2010). In this study, scores ranging from 0 to 26 were categorized as indicating low to moderate stress, while scores from 27 to 40 were classified as high stress.
2.2. Anxiety and depression assessment
The Hospital Anxiety and Depression Scale (HADS) was developed to identify probable and possible levels of Anxiety and Depression in clinical and non-clinical populations. It consists of 14 items divided into two subscales of 7 items each, where one comprises questions to identify signs of anxiety disorders and the other of depression. The answers range from 0 to 3 points, ranging from absent to very frequent, with a maximum score of 21 points. The cut-off point for both subscales is ≥ 9 points, with the higher the score, the higher the level of anxiety and/or depression symptoms. The anxiety (HADS-A) and depression (HADS-D) subscales showed internal consistency of 0.68 and 0.77, respectively, and the correlation of the items of both scales suggests that they have convergent, non-discriminating validity. Sensitivity and specificity were 93.7% and 72.6% for anxiety, and 84.6% and 90.3% for depression (Pais-Ribeiro et al., 2007).
2.3. DNA methylation analysis
Genomic DNA was obtained by collecting peripheral blood from the subjects in EDTA tubes (BD vacutainer®), followed by centrifugation to collect the buffy coat. The buffy coat samples were stored at −80 °C. DNA was extracted and bisulfite-converted directly from these samples using the EZ® DNA methylation-Direct Kit (Zymo Research, Irvine, CA, USA), following the manufacturer's recommendations. Pyrosequencing was performed using a PSQ 96 ID Pyrosequencer (Qiagen, Valencia, CA, USA) with PyroMark Gold Q96 Reagent Kit (Qiagen, Valencia, CA), according to the manufacturer's protocol. Pyrosequencing conditions were used as previously described (Alece Arantes Moreno et al., 2023). The percentage of methylation was recorded for each CpG from 5 to 13 and evaluated in PyroMark Q96 ID Software 2.5 version 2.5.10.7.
A representative scheme of the amplified exon IV promoter region of BDNF and the nine CpG sites analyzed (chr11: 27,701,519 to 27,701,826; GRCh38/hg38, GenBank NCBI—Access number: NC_000011.10) using bisulfite-pyrosequencing assays are shown in Fig. 1. This region was selected due to its consistent associations with stress-related disorders and cognition (Alece Arantes Moreno et al., 2023; Ferrer et al., 2019a; Zheleznyakova et al., 2016a).
Fig. 1.
Schematic representation of human BDNF gene indicating promoter regions. The BDNF promoter IV sequence is detailed in the enlarged box. The CpGs studied (5–13) are represented in orange and numbered in red. GeneBanK NCBI-Access number: NC_000011.10. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
2.4. Assessment of inflammatory cytokine measures
For the measurement of serum cytokines, interleukin-8 (IL-8), interleukin-6 (IL-6), interleukin-10 (IL-10), and interleukin −12p70 (IL-12p70) levels were assessed using the Cytometric Bead Array (CBA) Human Inflammatory Cytokines Kit (BD Biosciences, San Jose, CA), as previously described (Oliveira et al., 2021; Wilson et al., 2022). For values that were below the kit's limit of detection, a value equal to the cytokine's limit of detection was imputed to avoid bias in the analysis.
2.5. Statistical analyses
Sociodemographic, clinical, physical, and lifestyle variables were described using means and standard deviations, frequencies, or percentages (Thackray, 1981), according to the nature of the variables. The Generalized Linear Models (GzLM), introduced by Nelder and Wedderburn (1972), are models that comprise linear, logistic, and Poisson regressions in a unified tool, which allows the analysis of variables with the non-uniform distribution. GLzM models were used in this work to explore the influence of each specific CpG site within the BDNF gene promoter IV on perceived psychological stress and immune function. Age, body mass index (BMI), HADS.A and HADS.D were considered covariates. These variables were selected based on literature demonstrating their association with changes in DNA methylation patterns (Ikegame et al., 2013a; Song et al., 2014; Wiechmann et al., 2019). Additionally, including these covariates helps account for individual variability, ensuring that our analysis is not confounded by these known factors. For the selection of models, the following criteria were considered: (1) adjustment of the model by the Akaike's Information Criteria (AIC, Akaike's Information Criteria), in which lower values of AIC represent greater quality and simplicity (Akaike, 1972); (2) the principle of parsimony to obtain a more succinct model, disregarding covariates and interactions with high P-value; logical interpretation of the model within the hypothesis studied (Marioni et al., 2018). The parameters used to obtain the models considered the distribution of the response variable for the selection of the model type and linkage function. The best models were obtained with the Gamma distribution parameters, logarithmic linkage method, and a maximum of 100 iterations (see Supplementary Table 1). P values were presented based on Wald's chi-square test (Wald ꭓ2) and were adjusted by False Discovery Rate (FDR). The FDR considers the type I error rate in null hypothesis tests when performing multiple comparisons (Benjamini, Hochberg, 1995). Moderation regressions were calculated through PROGRESS macro in SPSS software (Hayes, 2017) with a confidence interval of 95%. The moderator variable, the sum of CpG5 to CpG13, was divided into three portions, according to the 16th, 50th, and 84th percentiles established by Hayes (2017) (Hayes, 2017). Continuous variables with different metrics were standardized by z-score. Statistical analyses were performed in IBM SPSS version 25.0 (IBM Corp., Armonk, NY, USA) and adjusted P values were calculated in Microsoft Excel. Interleukin data that were below the kit's limit of detection (BD Biosciences, San Jose, CA), a value equal to half the cytokine's limit of detection was imputed to avoid bias in the analysis (Beal, 2001). Non-parametric Kruskal-Wallis’ tests were used to compare BDNF methylation sites, inflammation, and oxidative stress measures divided by age ranges and Mann-Whitney's test for perceived stress ranges. Heatmap with a hierarchical cluster of studied variables was obtained in R Studio version 2022.12.0 + 353 (RStudio, Boston, MA, 2020) (R version 3.3.0+) (Team, 2020), applying the ggplot2 (H, W., 2016) and complex heatmap packages (Gu, 2022).
3. Results
3.1. Socioeconomic, demographic, and lifestyle characteristics
Socioeconomic, demographic, and lifestyle variables are shown in Table 1. Ages ranged from 31 to 60 years old, with a median age of 45 years (IQR = 37–42), 51.2% were married, 52.4% had an education level of more than 15 years. Laborally, 75.6% worked more than 40 hours per week. As expected, 64.6% of women in this study showed high perceived stress (median = 28, IQR = 3–40). The prevalence of symptoms of anxiety and depression were, respectively, 51.2% (median = 9, IQR = 1–20) and 30.5% (median = 6, IQR = 1–15).
Table 1.
Socioeconomic, demographic, and lifestyle characteristics.
| Characteristics | All (N = 82) |
|---|---|
| Age, years | |
| Age, mean (±SD) | 44.87 (8.57) |
| Education level, n (%) | |
| <15 years of study | 39 (47.6) |
| ≥15 years of study | 43 (52.4) |
| Relationship status, n (%) | |
| Single | 27 (32.9) |
| In a relationship | 6 (7,3) |
| Married | 42 (51.2) |
| Divorced | 7 (8.5) |
| Individual Income (R$ 954.00), n (%) | |
| <4770.00 | 49 (59.7) |
| ≥4770.00 | 33 (40.3) |
| Working hours, n (%) | |
| <40 | 20 (24.4) |
| ≥40 | 62 (75.6) |
| Clinical and lifestyle characteristics | |
| Psychological health, mean (±SD) | |
| HADS-A score | 8.78 (3.48) |
| HADS-A <9 | 5 (1.6) |
| HADS-A ≥9 | 10.44 (2.70) |
| HADS-D score | 7.04 (3.47) |
| HADS-D <9 | 4.57 (1.70) |
| HADS-D ≥9 | 10.70 (1.72) |
| PSS score | 29.89 (6.51) |
| PSS <27 | 18.83 (5.80) |
| PSS ≥27 | 33 (4.74) |
| BMI, mean (±SD) | 27.43 (5.40) |
| Alcohol comsumption (gr/day), n (%) | |
| none | 50 (61.0) |
| <49 gr/day | 25 (30.5) |
| >50 gr/day | 7 (8.5) |
| Smoking habits, n (%) | |
| Non-smoker | 64 (78.0) |
| Former smoker | 5 (6.1) |
| Smoker | 13 (15.9) |
| Physical activity (times per week), n (%) | |
| None | 9 (11.0) |
| Less than 3 | 43 (52.4) |
| More than 3 | 30 (36.6) |
Notes: Means with standard deviations (SD) and number of cases with percentages (%). Hospital Anxiety and Depression Scale (HADS), Hospital Anxiety and Depression Scale-anxiety symptoms (HADS-A), Hospital Anxiety and Depression Scale-depression symptoms (HADS-D), Perceived Stress Scale (PSS) and Body mass index (BMI).
3.2. Inflammatory biomarkers and DNA methylation profile
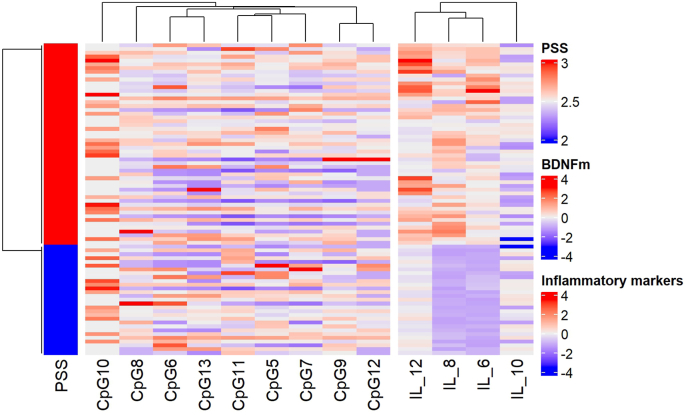
The high-stress group showed high serum IL-6, IL-8, and IL-12p70 levels compared to low-moderate perceived stress (P < 0.001, Table 2). The heatmap further illustrates distinct clustering patterns, where participants with low to moderate perceived stress formed a clear group with consistently lower serum levels of IL-6, IL-8, and IL-12p70 (Fig. 2). Notably, in Cluster A (Fig. 2), participants from the low-moderate stress group showed markedly reduced levels of these inflammatory markers, highlighting the clear differentiation in inflammatory response between the stress groups. No significant differences were observed in IL-10 levels (P = 0.329, Table 2).
Table 2.
BDNF methylation sites and inflammatory markers according to perceived stress classification.
| Variables (n = 82) |
Perceived stress |
||||
|---|---|---|---|---|---|
| Low stress |
High to moderate stress |
Group comparisons (P value)∗ | |||
| median (IQR) | mean (±SD) | (n = 29) mean (±SD) | (n = 53) mean (±SD) | ||
| IL-6 (pg/mL) | 5.04 (2.50–6.48) | 5.09 (2.90) | 2.51 (0.13) | 6.49 (2.72) | <0.001 |
| IL-8 (pg/mL) | 5.64 (3.60–8.22) | 6.33(2.86) | 3.57 (3.60) | 7.84 (2.48) | <0.001 |
| IL-10 (pg/mL) | 8.53 (6.67–8.77) | 7.94 (1.210 | 8.18 (0.91) | 7.93 (1.01) | 0.329 |
| IL-12p70 (pg/mL) | 2.00 (1.90–4.31) | 3.32 (2.07) | 2.10 (0.38) | 3.98 (2.31) | <0.001 |
| BDNF - CpG5 (%) | 4.64 (3.15–3.67) | 4.57 (3.07) | 4.81 (3.66) | 4.46 (2.66) | 0.802 |
| BDNF - CpG6 (%) | 3.60 (2.21–5.13) | 3.87 (2.63) | 4.13 (3.11) | 3.73 (2.32) | 0.623 |
| BDNF - CpG7 (%) | 5.08 (3.98–6.68) | 5.11 (2.83) | 5.07 (3.57) | 5.12 (2.36) | 0.888 |
| BDNF - CpG8 (%) | 4.20 (2.90–6.44) | 4.84 (4.87) | 5.19 (6.96) | 4.73 (3.19) | 0.595 |
| BDNF - CpG9 (%) | 11.26 (9.39–13.87) | 11.59 (5.32) | 10.91 (4.13) | 11.85 (6.08) | 0.956 |
| BDNF - CpG10 (%) | 1.51 (0–2.88) | 1.45 (1.76) | 1.55 (1.78) | 1.48 (1.79) | 0.824 |
| BDNF - CpG11 (%) | 6.28 (4.93–8.14) | 6.47 (2.69) | 6.65 (2.95) | 6.30 (2.70) | 0.512 |
| BDNF - CpG12 (%) | 3.48 (0–4.96) | 3.36 (3.44) | 3.31 (2.67) | 3.40 (3.83 | 0.697 |
| BDNF - CpG13 (%) | 4.1 (2.86–6.03) | 4.07 (2.84) | 3.72 (2.83) | 4.35 (2.81) | 0.474 |
Notes: Median with interquartile range (IQR), means with standard deviations (SD), ∗non-parametric Kruskal-Wallis’ test. BDNF-CpG: methylation of CpG sites of brain-derived neurotrophic factor gene.
Fig. 2.
Heatmap of perceived stress scale (PSS), inflammatory markers and BDNF DNA methylation with differentially methylated CpGs.
Data showed in z-score. Perceived stress scale (PSS), BDNF DNA methylation and inflammatory markers were scaled from −4 to 4, with red indicating high levels of inflammatory cytokines or high methylation or perceived stress, while blue low levels of inflammatory cytokines or low methylation or low perceived stress. (For interpretation of the references to colour in this figure legend, the reader is referred to the Web version of this article.)
BDNF methylation levels did not differ between the groups across 9 CpG sites analyzed (P > 0.05, Table 2). The heatmap analysis did not reveal substantial differences between the clusters (Fig. 2). Participants across the identified clusters showed similar patterns across 9 CpG sites, with no clear separation based on perceived stress levels.
3.3. Differentially methylated CpG sites in the BDNF gene are associated with perceived stress and inflammatory markers
As hypothesized, methylation levels were significantly associated with perceived stress levels (Table 3, see Supplementary Figures 1: Q-Q plots and residuals). CpG sites 5 (β = 0.143, χ 2 = 5.092, P adj. = 0.033), 6 (β = 0.095, χ 2 = 5.524, P adj. = 0.028), 11 (β = 0.398, χ 2 = 11.965, P adj. = 0.006), 12 (β = 0.767, χ 2 = 20.692, P adj. = 0.005), and 13 (β = 0.410, χ 2 = 19.117, P adj. = 0.004), presented a direct association with perceived stress level, indicating that DNA methylation in those sites is related to perceived stress. An inverse relationship was observed in GpG sites 7 (β = −0.260, χ 2 = 11.642, P adj. = 0.017), 8 (β = −0.196, χ 2 = 21.435, P adj. = 0.011), 9 (β = −0.364, χ 2 = 20.626, P adj. = 0.009), and 10 (β = −0.962, χ 2 = 23.555, P adj. = 0.007), where higher methylation predicted better results in perceived stress.
Table 3.
Differential methylation patterns in specific CpG sites within BDNF gene in relation to perceived stress and inflammatory cytokines.
| GzLM models - Perceived Stress | |||||||
|---|---|---|---|---|---|---|---|
| β | SE | 95% CI |
Wald ꭓ2 | P-value | P-value adja | ||
| Min | Max | ||||||
| DNA methylation (BDNF - CpGs %) | |||||||
| CpG5 | 0.143 | 0.063 | 0.019 | 0.267 | 5.092 | 0.024 | 0.033 |
| CpG6 | 0.095 | 0.040 | 0.016 | 0.174 | 5.524 | 0.019 | 0.028 |
| CpG7 | −0.260 | 0.076 | −0.409 | −0.110 | 11.642 | <0.001 | 0.017 |
| CpG8 | −0.196 | 0.042 | −0.279 | −0.113 | 21.435 | <0.001 | 0.011 |
| CpG9 | −0.364 | 0.080 | −0.521 | −0.207 | 20.626 | <0.001 | 0.009 |
| CpG10 | −0.962 | 0.198 | −1.350 | −0.573 | 23.555 | <0.001 | 0.007 |
| CpG11 | 0.398 | 0.115 | 0.173 | 0.624 | 11.965 | <0.001 | 0.006 |
| CpG12 | 0.767 | 0.169 | 0.437 | 1.098 | 20.692 | <0.001 | 0.005 |
| CpG13 | 0.410 | 0.094 | 0.226 | 0.594 | 19.117 | <0.001 | 0.004 |
| Inflamatory markers | |||||||
| IL-6 (pg/mL) | 0.036 | 0.014 | 0.008 | 0.063 | 6.487 | 0.011 | 0.033 |
| IL-8 (pg/mL) | 0.049 | 0.013 | 0.024 | 0.074 | 15.189 | <0.001 | <0.0001 |
| IL-10 (pg/mL) | −0.020 | 0.029 | −0.077 | 0.038 | 0.442 | 0.506 | 0.759 |
| IL-12p70 (pg/mL)) | 0.015 | 0.018 | −0.021 | 0.050 | 0.651 | 0.420 | 0.944 |
Note: Generalized linear models obtained with Gamma distribution. P-values were presented based on Wald's chi-square test (Wald ꭓ2) and were adjusted by FDR (False Discovery Rate). P < 0.05 (IC = 95%); BMI: body mass index; HADS-A and HADS-D: Hospital Anxiety and Depression Scale; CpG: DNA methylation sites of BDNF; IL: interleukin.
FDR: False Dicovery Rate.
Considering inflammatory markers, we found that high levels of IL-6 and IL-8 serum levels suggest an increase in perceived stress levels, suggesting that those cytokines are associated with perceived stress (P = 0.033 and < 0.0001, respectively). See Table 3 for additional details.
Given that IL-6 and IL-8 were associated with high perceived stress (see Table 2), we further investigated their correlations with BDNF methylation levels. No significant differences were found in IL-6 and IL-8 (P > 0.05). Surprisingly, an inverse relationship was observed in IL-10, where a higher serum level of IL-10 decreases methylation on CpG11 (β = −0.093, χ2 = 9.341, Padj. = 0.010). Additionally, IL-12p70 was directly associated with CpG sites 5 (β = 0.287, χ2 = 6.492, Padj. = 0.022), and 13 (β = 0.514, χ2 = 13.918, Padj. = 0.005), however, an inverse relationship was observed for CpG site 7 (β = −0.263, χ2 = 11.576, Padj. = 0.005), 9 (β = −0.274, χ2 = 5.031, Padj. = 0.040), and 11 (β = −0.455, χ2 = 9.867, Padj. = 0.006). For more details, see Table 4 and see Supplementary Material 1: Q-Q plots and residuals.
Table 4.
The effects of BDNF-CpGs by Inflammatory markers.
| GzLM models - Inflamatory markers | |||||||
|---|---|---|---|---|---|---|---|
| β | SE | 95% CI |
Wald ꭓ2 | P-value | P-value adja | ||
| Min | Max | ||||||
| IL-6 (pg/mL)versus %DNA methylation (BDNF - CpGs) | |||||||
| CpG5 | 0.235 | 0.119 | 0.001 | 0.468 | 3.887 | 0.049 | 0.243 |
| CpG6 | −0.031 | 0.026 | −0.082 | 0.019 | 1.498 | 0.221 | 0.473 |
| CpG7 | −0.028 | 0.024 | −0.075 | 0.018 | 1.406 | 0.236 | 0.393 |
| CpG8 | −0.009 | 0.014 | −0.036 | 0.018 | 0.450 | 0.502 | 0.685 |
| CpG9 | −0.005 | 0.019 | −0.042 | 0.032 | 0.071 | 0.790 | 0.911 |
| CpG10 | 0.006 | 0.036 | −0.064 | 0.077 | 0.032 | 0.858 | 0.919 |
| CpG11 | −0.004 | 0.024 | −0.051 | 0.044 | 0.023 | 0.879 | 0.879 |
| CpG12 | −0.027 | 0.030 | −0.085 | 0.031 | 0.828 | 0.363 | 0.544 |
| CpG13 | 0.051 | 0.027 | −0.001 | 0.103 | 3.640 | 0.056 | 0.211 |
| IL-8 (pg/mL)versus%DNA methylation (BDNF - CpGs) | |||||||
| CpG5 | −0.022 | 0.018 | −0.058 | 0.014 | 1.424 | 0.233 | 0.436 |
| CpG6 | −0.034 | 0.022 | −0.077 | 0.009 | 2.348 | 0.125 | 0.471 |
| CpG7 | −0.020 | 0.020 | −0.059 | 0.019 | 0.982 | 0.322 | 0.439 |
| CpG8 | 0.006 | 0.011 | −0.015 | 0.027 | 0.330 | 0.566 | 0.707 |
| CpG9 | 0.019 | 0.015 | −0.009 | 0.048 | 1.741 | 0.187 | 0.467 |
| CpG10 | 0.031 | 0.029 | −0.026 | 0.089 | 1.142 | 0.285 | 0.428 |
| CpG11 | −0.030 | 0.019 | −0.068 | 0.008 | 2.341 | 0.126 | 0.378 |
| CpG12 | −0.028 | 0.023 | −0.074 | 0.018 | 1.436 | 0.231 | 0.495 |
| CpG13 | 0.047 | 0.021 | 0.005 | 0.089 | 4.869 | 0.027 | 0.205 |
| IL-10 (pg/mL)versus%DNA methylation (BDNF - CpGs) | |||||||
| CpG5 | −0.009 | 0.005 | −0.019 | 0.002 | 2.732 | 0.098 | 0.239 |
| CpG6 | 0.004 | 0.006 | −0.007 | 0.016 | 0.585 | 0.444 | 0.581 |
| CpG7 | 0.008 | 0.006 | −0.003 | 0.018 | 1.822 | 0.177 | 0.301 |
| CpG8 | −0.002 | 0.003 | −0.007 | 0.004 | 0.413 | 0.520 | 0.590 |
| CpG9 | 0.003 | 0.004 | −0.005 | 0.010 | 0.433 | 0.510 | 0.620 |
| CpG10 | −0.001 | 0.026 | −0.052 | 0.050 | 0.002 | 0.962 | 1.000 |
| CpG11 | −0.093 | 0.030 | −0.152 | −0.033 | 9.341 | 0.002 | 0.010 |
| CpG12 | 0.000 | 0.006 | −0.012 | 0.012 | 0.000 | 0.995 | 0.995 |
| CpG13 | −0.007 | 0.006 | −0.018 | 0.005 | 1.224 | 0.269 | 0.380 |
| IL-12p70 (pg/mL)versus%DNA methylation (BDNF - CpGs) | |||||||
| CpG5 | 0.287 | 0.113 | 0.066 | 0.508 | 6.492 | 0.011 | 0.022 |
| CpG6 | 0.027 | 0.071 | −0.113 | 0.167 | 0.142 | 0.707 | 0.707 |
| CpG7 | −0.263 | 0.077 | −0.415 | −0.112 | 11.576 | 0.001 | 0.005 |
| CpG8 | 0.089 | 0.048 | −0.004 | 0.183 | 3.512 | 0.061 | 0.077 |
| CpG9 | −0.274 | 0.122 | −0.513 | −0.035 | 5.031 | 0.025 | 0.040 |
| CpG10 | 0.057 | 0.036 | −0.014 | 0.128 | 2.494 | 0.114 | 0.137 |
| CpG11 | −0.455 | 0.145 | −0.739 | −0.171 | 9.867 | 0.002 | 0.006 |
| CpG12 | 0.051 | 0.058 | −0.063 | 0.165 | 0.766 | 0.382 | 0.398 |
| CpG13 | 0.514 | 0.138 | 0.244 | 0.784 | 13.918 | 0.000 | 0.005 |
Note: Generalized linear models obtained with Gamma distribution. P-values were presented based on Wald's chi-square test (Wald ꭓ2) and were adjusted by FDR (False Discovery Rate). P < 0.05 (IC = 95%); CpG: DNA methylation sites of BDNF.
FDR: False Dicovery Rate.
3.4. Interactions of inter-individual variability influencing BDNF DNA methylation and inflammatory markers and perceived stress
We also assessed the interaction of confounding factors including age, BMI, anxiety, and depression symptoms, on BDNF methylation. We observed significant direct interactions of age (CpG sites 9 and 10), BMI (CpG sites 7, 8, 9, and 10), anxiety (CpG site 9), and depression symptoms (CpG sites 6 and 10), in which every increase in methylation values was associated with a high level of perceived stress. An inverse interaction was also observed for age (CpG sites 12 and 13), BMI (CpG 5, CpG 11, CpG 12 and CpG13), anxiety (CpG 6), and depression (CpG 12) symptoms (see Supplementary Table 2).
3.5. Moderation effect of methylation levels on the association between inflammatory markers and perceived stress
Moderation effects of methylation levels were investigated with the association of significant inflammatory makers (IL-6 and IL-8) obtained from GzLM models and perceived stress (see Supplementary Table 3). For each interleukin was obtained one model, and both models showed statistically significant moderation effects considering the levels of methylation on BDNF (IL-6 ∗ sum of CpGs: β = −0.451, P = 0.004; IL-8 sum of CpGs: β = −0.381, P = 0.041). Considering the conditional effects of methylation levels, the association between perceived stress and IL-6 and IL-8 on low levels of methylation (percentile 16th) showed an increased effect (IL-6 (16th): β = 0.653, p < 0.0001; IL-8 (16th): β = 0.710, P < 0.0001). This effect decreases when increase methylation levels (IL-6 (84th): β = 0.473, P < 0.0001; IL-8 (84th): β = 0.579, P < 0.0001). For more details, see Supplementary Material 2.
4. Discussion
To the best of our knowledge, this is the first study to evaluate the associations involving perceived stress, and inflammatory markers in relation to BDNF DNAm in women free of stress-related disorders and diseases. Three main findings emerge from this study. First, we demonstrated that 5 CpG sites were directly associated with high levels of perceived stress, whereas 4 CpGs sites exhibited inverse effects. Second, IL-8 and IL-6 showed simultaneous modulation of perceived stress, while IL-10 and IL-12p70 showed a correlation with DNAm. Moreover, we found that higher levels in IL-10 and IL-12p70 serum were inverse-associated with methylation in CpG11. A direct relationship was also observed with IL-12p70, where higher serum levels were associated with increased methylation at CpGs 5 and 13 respectively. Taken together, our findings further support our hypothesis regarding stress-sensitive regions within the BDNF gene, particularly highlighting CpGs 5,6, 11–13. In addition to these results, CpGs 7 and 9 may be regarded as stress-adaptive regions. Significant interactions were detected between age and depressive or anxiety symptoms in relation to BDNF DNAm.
Exploring the relationship between perceived stress and BDNF gene regulation through DNAm, we found that DNAm at CpG sites 5, 6, 11, 12, and 13 correlate to an increase in the perception of being under overwhelming current stressful life events. These results suggest that hypermethylation in those sites correlates with increased anxiety and depression symptoms. Another interesting association was found in our study to CpGs 7, 8, 9, and 10, in which increased DNAm levels were strongly associated with decreased perceived stress levels. These results suggest that hypermethylation in those sites might have a protective effect on stress response, suggesting that this region could help maintain the allostasis. To the best of our knowledge, this finding is the first scientific evidence to be published pointing out a possible existence of stress-responsive regions within the BDNF gene. The mechanism through which differential methylation occurs in the BDNF gene promoter IV is unclear and should be focused on future studies.
Further extending these findings, BDNF promoter IV contains a specific binding site for the cyclic-AMP-responsive element-binding protein (CREB), located at the CpG5 region, which is known to be particularly sensitive to stress (Braithwaite et al., 2015; Kundakovic et al., 2015). Methylation changes in this region can significantly affect gene expression, leading to alterations in neuroplasticity and stress responses. Our study found that methylation at this region and in other regions, such as CpG6, 11–13, is associated with increased perceived stress, indicating its potential role in stress response mechanisms. Conversely, we also identified 4 CpGs sites that exhibited inverse effects in stress response. According to Gatta and collaborators (2021), adaptive stress responses involve mechanisms that promote resilience and recovery, while maladaptive responses can exacerbate stress and lead to stress-related disorders (Gatta et al., 2021). The findings from our study suggest that GpG sites 5–6 and 11–13 methylation changes might be linked to maladaptive stress responses, potentially contributing to vulnerability to stress. This observation supports our hypothesis, supporting the idea that stress-sensitive regions within the BDNF, such as GpG5, 6, and 11–13, play a crucial role in these processes since these regions are located close to each other within the BDNF IV promoter. This hypothesis is supported by the bidirectional interaction between BDNF and glucocorticoid signaling, where stress influences the expression of specific BDNF (Blaze et al., 2017; Chen et al., 2017; Kundakovic et al., 2015; Roth et al., 2011; Zheleznyakova et al., 2016a).
Our results are in accordance with previous data that reported an association between DNA methylation at BDNF promoters in blood samples of stressed women with depressive symptoms (Peng et al., 2018; Song et al., 2014) and burnout (Bakusic et al., 2020). BDNF serum levels implicated in the regulation of stress (Linz et al., 2019) further supporting its role in stress-related psychiatric disorders, including major depressive disorder (MDD) (Ferrer et al., 2019b) as well as anxiety (Martinowich et al., 2007) and schizophrenia (Fernandes et al., 2015). These findings highlight the broader implications of BDNF methylation in stress response and its potential as a biomarker for psychiatric conditions.
In the present sample, IL-8 was directly associated with perceived stress levels. Previous evidence in the literature showed that reduced levels of serum IL-8 were inversely associated with high levels of perceived stress (Glaser et al., 1999), or that it was not associated with stress (Glaser et al., 1999; Liu et al., 2022). The contradictory findings in the present sample may be due to the exclusion of chronic and psychiatric conditions and/or anti-inflammatory and psychotropic drugs, as we focused only on healthy women, while Glaser and co-authors were evaluating inflammation at wound sites. However, considering the limited number of studies investigating the association between IL-8 and stress in healthy women and the complexity of inflammatory signaling, our result, albeit thrilling, cautions us to strive for further and more specific studies, in order to clarify the role of IL-8 in acute and chronic stress response in healthy individuals. We also found that IL-12p70 levels were directly associated with the methylation level of CpG13, suggesting that an increase in IL-12p70 level could be associated with DNAm changes in this site. Our results corroborate literature data that showed that hypermethylation of BDNF exon IV was associated with higher levels of serum IL-6 in patients with acute coronary syndrome (Kim et al., 2019). Another relevant finding in our study is the direct association of perceived stress level and IL-6, as would be intuitively expected. This finding corroborates previous observations of the intimate relationship between inflammation and stress as seen in stress-related disorders such as depression (Hiles et al., 2012; Liu et al., 2022; Roohi et al., 2021; Ting et al., 2020) and PTSD (Lima et al., 2019; Renner et al., 2022). The mechanism by which inflammation changes the BDNF methylation is unclear and should be the object of future studies. Those results corroborate our hypothesis that some CpG sites on BDNF exon IV promoter are sensitive to inflammatory responses led by stress.
Our results substantiate the role of BDNF methylation in moderating the relationship between perceived stress and inflammatory markers IL-6 and IL-8. The moderation analysis reveals that lower levels of perceived stress and these inflammatory markers are linked to BDNF methylation. This suggests that individuals with reduced BDNF methylation may exhibit greater sensitivity to stress-related inflammation. Specifically, at lower methylation levels, the impact of perceived stress on IL-6 and IL-8 is more pronounced, indicating an increased vulnerability to stress-induced inflammatory responses. These findings highlight the potential of BDNF methylation as a critical factor in understanding individual differences in stress reactivity and inflammation, underscoring the need for further investigation into how BDNF methylation influences stress and its associated health outcomes. This pattern supports the hypothesis that BDNF methylation could act as a modulator in the stress-inflammation pathway, potentially influencing individual responses to stress. These findings highlight the complexity of the stress response system and the potential for BDNF methylation to serve as a marker for susceptibility or resilience to stress-related inflammatory conditions. Further research is needed to explore these relationships in more detail and assess the potential clinical implications of BDNF methylation as a target for interventions aimed at mitigating stress-related inflammatory responses.
However, some limitations restrict the scope of the conclusions hereafter derived and must be addressed. First, the sample was predominantly women, meaning that additional research is needed in order to allow a larger generalization than those of the present study. A further examination of sex differences in inflammation mechanisms might be useful here: more advanced studies in DNAm and stress might show relevance for a mechanistic understanding of inflammation, both in healthy and clinical populations (Mengelkoch and Slavich, 2024). Secondly, related to the previous consideration, a future measure of estrogen levels at the time of evaluation might be helpful in shedding further light on our results of inflammation and DNA methylation, since estrogen effects have been implicated in cognition, especially in aging populations (Alivizatou and Psarropoulou, 2024; Au et al., 2016; Korol and Pisani, 2015), and also in inflammation (Bowling et al., 2014; Chakrabarti et al., 2008; Novella et al., 2012). Third, the small sample size might have reduced the statistical power of our analysis. This was due to the constraints of using a pre-existing database with specific inclusion criteria. Future studies with larger sample sizes are needed to validate our findings and address additional research questions. Fourth, we did not evaluate the influence of Val66Met polymorphism and its impact on BDNF methylation in this sample. Since its crucial participation in reactivity at the amygdala, hence on emotional regulation, such an evaluation might discriminate the participation of each factor (Redlich et al., 2020). Fifth, we accessed the neuroinflammation and neuroepigenetic changes in peripheral blood cells, and although probably these peripheral cells may not adequately detect the changes in brain tissue, research in this field might also shed light on the peripheral effects of BDNF epigenetic alterations, which have also been found to be related to other major clinical pictures, besides anxiety and depression (Chang et al., 2014; Ikegame et al., 2013b).
The current literature has abundant evidence supporting the neurotrophic hypothesis of both pathophysiology and treatment response in depression (Cavaleri et al., 2023; Polyakova et al., 2015), anxiety (Suliman et al., 2013) neurocognitive and neurodegenerative conditions such as Alzheimer (Cattaneo et al., 2016; Nagahara and Tuszynski, 2011), or schizophrenia (Cattaneo et al., 2016; Favalli et al., 2012; Nieto et al., 2013), and our findings align with this hypothesis.
Although not the primary focus of this study, it is important to note that classical stress-related disorders, such as PTSD and depression, are closely linked to acute and chronic stress, as outlined in diagnostic guidelines. In such disorders, which are credited as stress-inducible illnesses, a basic disturbance of cognition and emotion, we also find a fundamental participation of BDNF (Notaras and van den Buuse, 2020). The findings of this study, including the observed effects on BDNF methylation, may have relevance to understanding resilience and vulnerability to such conditions (Miao et al., 2020; Zheleznyakova et al., 2016b). BDNF methylation, given its association with psychiatric disorders, is emerging as a valuable marker for tracking stress-related mental health issues. Studying these epigenetic changes in healthy individuals helps establish a baseline for identifying early biomarkers of stress susceptibility and offers insights into the molecular pathways leading to psychiatric disorders.
Given the growing body of evidence supporting the role of neurotrophic mechanisms in stress-related and other mental health conditions, there is a strong case for further research to explore new strategies and clinical interventions targeting these pathways.
CRediT authorship contribution statement
Luciana Fungaro Rissatti: Writing – review & editing, Writing – original draft, Methodology, Data curation, Conceptualization. David Wilson: Writing – review & editing, Writing – original draft, Methodology, Investigation, Data curation, Conceptualization. Fanny Palace-Berl: Writing – review & editing, Writing – original draft, Methodology, Formal analysis, Data curation. Bárbara de Mello Ponteciano: Writing – review & editing, Methodology, Data curation. Flávia Sardela de Miranda: Writing – review & editing, Methodology, Data curation. Ivana Alece Arantes Moreno: Writing – review & editing, Methodology, Data curation. Tamires dos Santos Vieira: Writing – review & editing, Methodology, Data curation. Bruna Pereira Sorroche: Writing – review & editing, Methodology, Data curation. Lidia Maria Rebolho Batista Arantes: Writing – review & editing, Resources, Methodology. Adriana Madeira Alvares da Silva: Writing – review & editing, Resources, Methodology. Vânia D'Almeida: Review & Editing, Resources, Methodology, Conceptualization. Marcelo Demarzo: Review & Editing, Resources, Conceptualization. Daniela Rodrigues de Oliveira: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Resources, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this article.
Acknowledgments
DRO and other co-authors appreciate the support provided by grant 424329/2018-2 from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq). DRO appreciates the support provided by the Post-doctoral fellowship grant 2017/16155-4, Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP). We also appreciate the support provided by the Diretoria Regional de Educação Santo Amaro (DRE), Núcleo de Apoio e Acompanhamento para Aprendizagem (NAAPA), São Paulo/SP/Brazil. We would also like to express our gratitude to Ana Claudia de Paula Correa, Kátia Maria Vergne, and Debora Lieber de Paula for the support and partnership of this study and all the teachers and staff in the Public Schools from São Paulo city. We especially thank all volunteers who took part in this study. We express our gratitude to Fanny Palace-Berl, our statistical bioinformatics, for her support in the statistical analysis of this article. VD’A and MD are recipients of CNPq fellowships.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.bbih.2024.100900.
Contributor Information
Luciana Fungaro Rissatti, Email: lufungaro@hotmail.com.
David Wilson, Email: davidwilsonbrasileiro@gmail.com.
Fanny Palace-Berl, Email: fanny.palace@gmail.com.
Bárbara de Mello Ponteciano, Email: bmponteciano@gmail.com.
Flávia Sardela de Miranda, Email: flavia.sardela@gmail.com.
Ivana Alece Arantes Moreno, Email: ivanaarantesm@gmail.com.
Tamires dos Santos Vieira, Email: tamiresvieiraalim@gmail.com.
Bruna Pereira Sorroche, Email: brupsorroche@gmail.com.
Lidia Maria Rebolho Batista Arantes, Email: lirebolho@hotmail.com.
Adriana Madeira Alvares da Silva, Email: adriana.biomol@gmail.com.
Vânia D'Almeida, Email: vaniadalmeida@icloud.com.
Marcelo Demarzo, Email: marcelodemarzo@gmail.com.
Daniela Rodrigues de Oliveira, Email: doliveira@mcw.edu, danielaoliveira.phd@gmail.com.
Appendix A. Supplementary data
The following are the Supplementary data to this article.
Data availability
Data will be made available on request.
References
- Alece Arantes Moreno I., Rodrigues de Oliveira D., Ribeiro Borçoi A., Fungaro Rissatti L., Vitorino Freitas F., Arantes L.M.R.B., Oliveira Mendes S., dos Santos Vieira T., Risse Quaioto B., Cerbino Doblas P. Methylation of BDNF gene in association with episodic memory in women. Front. Neurosci. 2023;17 doi: 10.3389/fnins.2023.1092406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alivizatou A.-M., Psarropoulou C. vol. 1. STUDENT INTERNATIONAL BIOLOGICAL & BIOMEDICAL RESEARCH; 2024. (Effects of Estrogen on Brain and Cognitive Senescence). [Google Scholar]
- Au A., Feher A., McPhee L., Jessa A., Oh S., Einstein G. Estrogens, inflammation and cognition. Front. Neuroendocrinol. 2016;40:87–100. doi: 10.1016/j.yfrne.2016.01.002. [DOI] [PubMed] [Google Scholar]
- Bakusic J., Ghosh M., Polli A., Bekaert B., Schaufeli W., Claes S., Godderis L. Epigenetic perspective on the role of brain-derived neurotrophic factor in burnout. Transl. Psychiatry. 2020;10:354. doi: 10.1038/s41398-020-01037-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beal S.L. Ways to fit a PK model with some data below the quantification limit. J. Pharmacokinet. Pharmacodyn. 2001;28:481–504. doi: 10.1023/a:1012299115260. [DOI] [PubMed] [Google Scholar]
- Blaze J., Asok A., Borrelli K., Tulbert C., Bollinger J., Ronca A.E., Roth T.L. Intrauterine exposure to maternal stress alters Bdnf IV DNA methylation and telomere length in the brain of adult rat offspring. Int. J. Dev. Neurosci. 2017;62:56–62. doi: 10.1016/j.ijdevneu.2017.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bowling M.R., Xing D., Kapadia A., Chen Y.-F., Szalai A.J., Oparil S., Hage F.G. Estrogen effects on vascular inflammation are age dependent: role of estrogen receptors. Arterioscler. Thromb. Vasc. Biol. 2014;34:1477–1485. doi: 10.1161/ATVBAHA.114.303629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braithwaite E., Kundakovic M., Ramchandani P., Murphy S., Champagne F. Maternal prenatal depressive symptoms predict infant NR3C1 1F and BDNF IV DNA methylation. Epigenetics. 2015;10:408–417. doi: 10.1080/15592294.2015.1039221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calabrese F., Rossetti A.C., Racagni G., Gass P., Riva M.A., Molteni R. Brain-derived neurotrophic factor: a bridge between inflammation and neuroplasticity. Front. Cell. Neurosci. 2014;8:430. doi: 10.3389/fncel.2014.00430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carpenter L.L., Gawuga C.E., Tyrka A.R., Lee J.K., Anderson G.M., Price L.H. Association between plasma IL-6 response to acute stress and early-life adversity in healthy adults. Neuropsychopharmacology. 2010;35:2617–2623. doi: 10.1038/npp.2010.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cattaneo A., Cattane N., Begni V., Pariante C., Riva M. The human BDNF gene: peripheral gene expression and protein levels as biomarkers for psychiatric disorders. Transl. Psychiatry. 2016;6 doi: 10.1038/tp.2016.214. e958-e958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavaleri D., Moretti F., Bartoccetti A., Mauro S., Crocamo C., Carrà G., Bartoli F. The role of BDNF in major depressive disorder, related clinical features, and antidepressant treatment: insight from meta-analyses. Neurosci. Biobehav. Rev. 2023 doi: 10.1016/j.neubiorev.2023.105159. [DOI] [PubMed] [Google Scholar]
- Chakrabarti S., Lekontseva O., Davidge S.T. Estrogen is a modulator of vascular inflammation. IUBMB Life. 2008;60:376–382. doi: 10.1002/iub.48. [DOI] [PubMed] [Google Scholar]
- Chang L., Wang Y., Ji H., Dai D., Xu X., Jiang D., Hong Q., Ye H., Zhang X., Zhou X. Elevation of peripheral BDNF promoter methylation links to the risk of Alzheimer's disease. PLoS One. 2014;9 doi: 10.1371/journal.pone.0110773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H., Lombès M., Le Menuet D. Glucocorticoid receptor represses brain-derived neurotrophic factor expression in neuron-like cells. Mol. Brain. 2017;10:12. doi: 10.1186/s13041-017-0295-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen S., Gianaros P.J., Manuck S.B. A stage model of stress and disease. Perspect. Psychol. Sci. 2016;11:456–463. doi: 10.1177/1745691616646305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen S., Janicki-Deverts D., Miller G.E. Psychological stress and disease. JAMA. 2007;298:1685–1687. doi: 10.1001/jama.298.14.1685. [DOI] [PubMed] [Google Scholar]
- Crawford B., Craig Z., Mansell G., White I., Smith A., Spaull S., Imm J., Hannon E., Wood A., Yaghootkar H. DNA methylation and inflammation marker profiles associated with a history of depression. Hum. Mol. Genet. 2018;27:2840–2850. doi: 10.1093/hmg/ddy199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Assis G.G., Gasanov E.V. BDNF and cortisol integrative system–plasticity vs. degeneration: implications of the Val66Met polymorphism. Front. Neuroendocrinol. 2019;55 doi: 10.1016/j.yfrne.2019.100784. [DOI] [PubMed] [Google Scholar]
- de Oliveira D.R., Wilson D., Palace-Berl F., de Mello Ponteciano B., Rissatti L.F., de Miranda F.S., Pollizi V.P., Fuscella J.C., Terzi A.M., Lepique A.P. Mindfulness meditation training effects on quality of life, immune function and glutathione metabolism in service healthy female teachers: a randomized pilot clinical trial. Brain, behavior, & immunity-health. 2021;18 doi: 10.1016/j.bbih.2021.100372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong E., Dzitoyeva S.G., Matrisciano F., Tueting P., Grayson D.R., Guidotti A. Brain-derived neurotrophic factor epigenetic modifications associated with schizophrenia-like phenotype induced by prenatal stress in mice. Biol. Psychiatr. 2015;77:589–596. doi: 10.1016/j.biopsych.2014.08.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epel E.S., Crosswell A.D., Mayer S.E., Prather A.A., Slavich G.M., Puterman E., Mendes W.B. More than a feeling: a unified view of stress measurement for population science. Front. Neuroendocrinol. 2018;49:146–169. doi: 10.1016/j.yfrne.2018.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Favalli G., Li J., Belmonte-de-Abreu P., Wong A.H., Daskalakis Z.J. The role of BDNF in the pathophysiology and treatment of schizophrenia. J. Psychiatr. Res. 2012;46:1–11. doi: 10.1016/j.jpsychires.2011.09.022. [DOI] [PubMed] [Google Scholar]
- Fernandes B., Steiner J., Berk M., Molendijk M., Gonzalez-Pinto A., Turck C., Nardin P., Gonçalves C. Peripheral brain-derived neurotrophic factor in schizophrenia and the role of antipsychotics: meta-analysis and implications. Mol. Psychiatr. 2015;20:1108–1119. doi: 10.1038/mp.2014.117. [DOI] [PubMed] [Google Scholar]
- Ferrer A., Labad J., Salvat-Pujol N., Barrachina M., Costas J., Urretavizcaya M., de Arriba-Arnau A., Crespo J.M., Soriano-Mas C., Carracedo Á. BDNF genetic variants and methylation: effects on cognition in major depressive disorder. Transl. Psychiatry. 2019;9:1–10. doi: 10.1038/s41398-019-0601-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferrer A., Labad J., Salvat-Pujol N., Barrachina M., Costas J., Urretavizcaya M., de Arriba-Arnau A., Crespo J.M., Soriano-Mas C., Carracedo Á. BDNF genetic variants and methylation: effects on cognition in major depressive disorder. Transl. Psychiatry. 2019;9:265. doi: 10.1038/s41398-019-0601-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gatta E., Saudagar V., Auta J., Grayson D.R., Guidotti A. International Review of Neurobiology. Elsevier; 2021. Epigenetic landscape of stress surfeit disorders: key role for DNA methylation dynamics; pp. 127–183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glaser R., Kiecolt-Glaser J.K., Marucha P.T., MacCallum R.C., Laskowski B.F., Malarkey W.B. Stress-related changes in proinflammatory cytokine production in wounds. Arch. Gen. Psychiatr. 1999;56:450–456. doi: 10.1001/archpsyc.56.5.450. [DOI] [PubMed] [Google Scholar]
- Gray J., Milner T., McEwen B. Dynamic plasticity: the role of glucocorticoids, brain-derived neurotrophic factor and other trophic factors. Neuroscience. 2013;239:214–227. doi: 10.1016/j.neuroscience.2012.08.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu Z. Complex heatmap visualization. Imeta. 2022;1:e43. doi: 10.1002/imt2.43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- H W. Springer-Verlag; 2016. ggplot2: Elegant Graphics for Data Analysis. [Google Scholar]
- Hayes A.F. Guilford publications; 2017. Introduction to Mediation, Moderation, and Conditional Process Analysis: A Regression-Based Approach. [Google Scholar]
- He F., Berg A., Imamura Kawasawa Y., Bixler E.O., Fernandez-Mendoza J., Whitsel E.A., Liao D. Association between DNA methylation in obesity-related genes and body mass index percentile in adolescents. Sci. Rep. 2019;9:2079. doi: 10.1038/s41598-019-38587-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herhaus B., Heni M., Bloch W., Petrowski K. Acute and chronic psychosocial stress by the brain-derived neurotrophic factor in male humans: a highly standardized and controlled study. medRxiv. 2023 2023.2009. 2029.23296327. [Google Scholar]
- Hiles S.A., Baker A.L., de Malmanche T., Attia J. A meta-analysis of differences in IL-6 and IL-10 between people with and without depression: exploring the causes of heterogeneity. Brain Behav. Immun. 2012;26:1180–1188. doi: 10.1016/j.bbi.2012.06.001. [DOI] [PubMed] [Google Scholar]
- Hötting K., Schickert N., Kaiser J., Röder B., Schmidt-Kassow M. The effects of acute physical exercise on memory, peripheral BDNF, and cortisol in young adults. Neural Plast. 2016;2016 doi: 10.1155/2016/6860573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ikegame T., Bundo M., Murata Y., Kasai K., Kato T., Iwamoto K. DNA methylation of the BDNF gene and its relevance to psychiatric disorders. J. Hum. Genet. 2013;58:434–438. doi: 10.1038/jhg.2013.65. [DOI] [PubMed] [Google Scholar]
- Ikegame T., Bundo M., Sunaga F., Asai T., Nishimura F., Yoshikawa A., Kawamura Y., Hibino H., Tochigi M., Kakiuchi C. DNA methylation analysis of BDNF gene promoters in peripheral blood cells of schizophrenia patients. Neurosci. Res. 2013;77:208–214. doi: 10.1016/j.neures.2013.08.004. [DOI] [PubMed] [Google Scholar]
- Januar V., Ancelin M.-L., Ritchie K., Saffery R., Ryan J. BDNF promoter methylation and genetic variation in late-life depression. Transl. Psychiatry. 2015;5 doi: 10.1038/tp.2015.114. e619-e619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J.-M., Stewart R., Kim J.-W., Kang H.-J., Lee J.-Y., Kim S.-Y., Kim S.-W., Shin I.-S., Hong Y.J., Ahn Y. Modifying effects of depression on the association between BDNF methylation and prognosis of acute coronary syndrome. Brain Behav. Immun. 2019;81:422–429. doi: 10.1016/j.bbi.2019.06.038. [DOI] [PubMed] [Google Scholar]
- Knight E.L., Jiang Y., Rodriguez-Stanley J., Almeida D.M., Engeland C.G., Zilioli S. Perceived stress is linked to heightened biomarkers of inflammation via diurnal cortisol in a national sample of adults. Brain Behav. Immun. 2021;93:206–213. doi: 10.1016/j.bbi.2021.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korol D.L., Pisani S.L. Estrogens and cognition: friends or foes?: an evaluation of the opposing effects of estrogens on learning and memory. Horm. Behav. 2015;74:105–115. doi: 10.1016/j.yhbeh.2015.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kundakovic M., Gudsnuk K., Herbstman J.B., Tang D., Perera F.P., Champagne F.A. DNA methylation of BDNF as a biomarker of early-life adversity. Proc. Natl. Acad. Sci. USA. 2015;112:6807–6813. doi: 10.1073/pnas.1408355111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lambert W.M., Xu C.-F., Neubert T.A., Chao M.V., Garabedian M.J., Jeanneteau F.D. Brain-derived neurotrophic factor signaling rewrites the glucocorticoid transcriptome via glucocorticoid receptor phosphorylation. Molecular and cellular biology. 2013 doi: 10.1128/MCB.00150-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lima B.B., Hammadah M., Wilmot K., Pearce B.D., Shah A., Levantsevych O., Kaseer B., Obideen M., Gafeer M.M., Kim J.H. Posttraumatic stress disorder is associated with enhanced interleukin-6 response to mental stress in subjects with a recent myocardial infarction. Brain Behav. Immun. 2019;75:26–33. doi: 10.1016/j.bbi.2018.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin C.-C., Huang T.-L. Brain-derived neurotrophic factor and mental disorders. Biomed. J. 2020;43:134–142. doi: 10.1016/j.bj.2020.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linz R., Puhlmann L., Apostolakou F., Mantzou E., Papassotiriou I., Chrousos G., Engert V., Singer T. Acute psychosocial stress increases serum BDNF levels: an antagonistic relation to cortisol but no group differences after mental training. Neuropsychopharmacology. 2019;44:1797–1804. doi: 10.1038/s41386-019-0391-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X., Huang J., Jiang Y., Cao Z., Wu M., Sun R., Chen Z., Yu P., Ma J., Chen Y. IL-6 and IL-8 are likely associated with psychological status in treatment naïve general population. J. Affect. Disord. 2022;298:337–344. doi: 10.1016/j.jad.2021.10.042. [DOI] [PubMed] [Google Scholar]
- Marioni R.E., McRae A.F., Bressler J., Colicino E., Hannon E., Li S., Prada D., Smith J.A., Trevisi L., Tsai P.-C. Meta-analysis of epigenome-wide association studies of cognitive abilities. Mol. Psychiatr. 2018;23:2133–2144. doi: 10.1038/s41380-017-0008-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinowich K., Manji H., Lu B. New insights into BDNF function in depression and anxiety. Nat. Neurosci. 2007;10:1089–1093. doi: 10.1038/nn1971. [DOI] [PubMed] [Google Scholar]
- Mazure C.M., Husky M.M., Pietrzak R.H. Stress as a risk factor for mental disorders in a gendered environment. JAMA Psychiatr. 2023;80:1087–1088. doi: 10.1001/jamapsychiatry.2023.3138. [DOI] [PubMed] [Google Scholar]
- McEwen B.S. vol. 109. Proceedings of the National Academy of Sciences; 2012. pp. 17180–17185. (Brain on Stress: How the Social Environment Gets under the Skin). [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwen B.S., Akil H. Revisiting the stress concept: implications for affective disorders. J. Neurosci. 2020;40:12–21. doi: 10.1523/JNEUROSCI.0733-19.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mengelkoch S., Slavich G.M. Sex differences in stress susceptibility as a key mechanism underlying depression risk. Curr. Psychiatr. Rep. 2024:1–9. doi: 10.1007/s11920-024-01490-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miao Z., Wang Y., Sun Z. The relationships between stress, mental disorders, and epigenetic regulation of BDNF. Int. J. Mol. Sci. 2020;21:1375. doi: 10.3390/ijms21041375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagahara A.H., Tuszynski M.H. Potential therapeutic uses of BDNF in neurological and psychiatric disorders. Nat. Rev. Drug Discov. 2011;10:209–219. doi: 10.1038/nrd3366. [DOI] [PubMed] [Google Scholar]
- Nieto R., Kukuljan M., Silva H. BDNF and schizophrenia: from neurodevelopment to neuronal plasticity, learning, and memory. Front. Psychiatr. 2013;4 doi: 10.3389/fpsyt.2013.00045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Notaras M., van den Buuse M. Neurobiology of BDNF in fear memory, sensitivity to stress, and stress-related disorders. Mol. Psychiatr. 2020;25:2251–2274. doi: 10.1038/s41380-019-0639-2. [DOI] [PubMed] [Google Scholar]
- Novella S., Heras M., Hermenegildo C., Dantas A.P. Effects of estrogen on vascular inflammation: a matter of timing. Arterioscler. Thromb. Vasc. Biol. 2012;32:2035–2042. doi: 10.1161/ATVBAHA.112.250308. [DOI] [PubMed] [Google Scholar]
- Oliveira D.R.d., Wilson D., Berl F.P., Ponteciano B.d.M., Rissatti L.F., Miranda F.S.d., Pollizi V.P., Fuscella J.C., Terzi A.M., Lepique A.P. Mindfulness meditation training effects on quality of life, immune function and glutathione metabolism in service healthy female teachers: arandomized pilot clinical trial. Brain, Behavior, & Immunity-Health. 2021;18:1–12. doi: 10.1016/j.bbih.2021.100372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Özçelik T., Rosenthal A., Francke U. Chromosomal mapping of brain-derived neurotrophic factor and neurotrophin-3 genes in man and mouse. Genomics. 1991;10:569–575. doi: 10.1016/0888-7543(91)90437-j. [DOI] [PubMed] [Google Scholar]
- Pais-Ribeiro J., Silva I., Ferreira T., Martins A., Meneses R., Baltar M. Validation study of a Portuguese version of the hospital anxiety and depression scale. Psychol. Health Med. 2007;12:225–237. doi: 10.1080/13548500500524088. [DOI] [PubMed] [Google Scholar]
- Peng H., Zhu Y., Strachan E., Fowler E., Bacus T., Roy-Byrne P., Goldberg J., Vaccarino V., Zhao J. Childhood trauma, DNA methylation of stress-related genes, and depression: findings from two monozygotic twin studies. Psychosom. Med. 2018;80:599. doi: 10.1097/PSY.0000000000000604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polyakova M., Stuke K., Schuemberg K., Mueller K., Schoenknecht P., Schroeter M.L. BDNF as a biomarker for successful treatment of mood disorders: a systematic & quantitative meta-analysis. J. Affect. Disord. 2015;174:432–440. doi: 10.1016/j.jad.2014.11.044. [DOI] [PubMed] [Google Scholar]
- Quaioto B.R., Borçoi A.R., Mendes S.O., Doblas P.C., dos Santos Vieira T., Moreno I.A.A., Dos Santos J.G., Hollais A.W., Olinda A.S., de Souza M.L.M. Tobacco use modify exon IV BDNF gene methylation levels in depression. J. Psychiatr. Res. 2023;159:240–248. doi: 10.1016/j.jpsychires.2023.01.038. [DOI] [PubMed] [Google Scholar]
- Redlich R., Schneider I., Kerkenberg N., Opel N., Bauhaus J., Enneking V., Repple J., Leehr E.J., Grotegerd D., Kähler C. The role of BDNF methylation and Val66Met in amygdala reactivity during emotion processing. Hum. Brain Mapp. 2020;41:594–604. doi: 10.1002/hbm.24825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reis R.S., Hino A., Añez C. Perceived stress scale. J. Health Psychol. 2010;15:107–114. doi: 10.1177/1359105309346343. [DOI] [PubMed] [Google Scholar]
- Renner V., Joraschky P., Kirschbaum C., Schellong J., Petrowski K. Pro-and anti-inflammatory cytokines Interleukin-6 and Interleukin-10 predict therapy outcome of female patients with posttraumatic stress disorder. Transl. Psychiatry. 2022;12:472. doi: 10.1038/s41398-022-02230-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roohi E., Jaafari N., Hashemian F. On inflammatory hypothesis of depression: what is the role of IL-6 in the middle of the chaos? J. Neuroinflammation. 2021;18:1–15. doi: 10.1186/s12974-021-02100-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roth T.L., Zoladz P.R., Sweatt J.D., Diamond D.M. Epigenetic modification of hippocampal Bdnf DNA in adult rats in an animal model of post-traumatic stress disorder. J. Psychiatr. Res. 2011;45:919–926. doi: 10.1016/j.jpsychires.2011.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slavich G.M., Roos L.G., Mengelkoch S., Webb C.A., Shattuck E.C., Moriarity D.P., Alley J.C. Social Safety Theory: conceptual foundation, underlying mechanisms, and future directions. Health Psychol. Rev. 2023;17:5–59. doi: 10.1080/17437199.2023.2171900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song Y., Miyaki K., Suzuki T., Sasaki Y., Tsutsumi A., Kawakami N., Shimazu A., Takahashi M., Inoue A., Kan C. Altered DNA methylation status of human brain derived neurotrophis factor gene could be useful as biomarker of depression. Am. J. Med. Genet. Part B: Neuropsychiatric Genetics. 2014;165:357–364. doi: 10.1002/ajmg.b.32238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suliman S., Hemmings S.M., Seedat S. Brain-Derived Neurotrophic Factor (BDNF) protein levels in anxiety disorders: systematic review and meta-regression analysis. Front. Integr. Neurosci. 2013;7:55. doi: 10.3389/fnint.2013.00055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tapia-Arancibia L., Rage F., Givalois L., Arancibia S. Physiology of BDNF: focus on hypothalamic function. Front. Neuroendocrinol. 2004;25:77–107. doi: 10.1016/j.yfrne.2004.04.001. [DOI] [PubMed] [Google Scholar]
- Team R. Integrated Development for R. RStudio, PBC; Boston, MA: 2020. RStudio. [Google Scholar]
- Thackray R. vol. 43. 1981. pp. 165–176. (The Stress of Boredom and Monotony: A Consideration of the Evidence). [DOI] [PubMed] [Google Scholar]
- Ting E.Y.-C., Yang A.C., Tsai S.-J. Role of interleukin-6 in depressive disorder. Int. J. Mol. Sci. 2020;21:2194. doi: 10.3390/ijms21062194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang C.S., Kavalali E.T., Monteggia L.M. BDNF signaling in context: from synaptic regulation to psychiatric disorders. Cell. 2022;185:62–76. doi: 10.1016/j.cell.2021.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiechmann T., Röh S., Sauer S., Czamara D., Arloth J., Ködel M., Beintner M., Knop L., Menke A., Binder E.B. Identification of dynamic glucocorticoid-induced methylation changes at the FKBP5 locus. Clin. Epigenet. 2019;11:1–14. doi: 10.1186/s13148-019-0682-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson D., de Oliveira D.R., Palace-Berl F., de Mello Ponteciano B., Rissatti L.F., Pollizi V.P., de Miranda F.S., D'Almeida V., Demarzo M. Fostering emotional self-regulation in female teachers at the public teaching network: a mindfulness-based intervention improving psychological measures and inflammatory biomarkers. Brain, behavior, & immunity-health. 2022;21 doi: 10.1016/j.bbih.2022.100427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao T., Piao L.-H., Li D.-P., Xu S.-H., Wang S.-Y., Yuan H.-B., Zhang C.-X. BDNF gene hydroxymethylation in hippocampus related to neuroinflammation-induced depression-like behaviors in mice. J. Affect. Disord. 2023;323:723–730. doi: 10.1016/j.jad.2022.12.035. [DOI] [PubMed] [Google Scholar]
- Zheleznyakova G.Y., Cao H., Schiöth H.B. BDNF DNA methylation changes as a biomarker of psychiatric disorders: literature review and open access database analysis. Behav. Brain Funct. 2016;12:1–14. doi: 10.1186/s12993-016-0101-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheleznyakova G.Y., Cao H., Schiöth H.B. BDNF DNA methylation changes as a biomarker of psychiatric disorders: literature review and open access database analysis. Behav. Brain Funct. 2016;12:1–14. doi: 10.1186/s12993-016-0101-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.