Abstract
In recent years, the incidence of prostate cancer has been increasing globally. Early stage of the disease can obtain a better clinical prognosis from surgery and endocrine therapy. The progression of advanced stage varies significantly between individuals, with some patients developing metastatic castration-resistant prostate cancer after standardized treatment. Therefore, staging of prostate cancer by accurate imaging is particularly important for the clinical management of patients. Simultaneously, the development of targeted therapy is also urgent for the treatment of advanced prostate cancer. Prostate specific membrane antigen as a prostate specific target has been widely used in the diagnosis and treatment of prostate cancer. This review summarizes the latest research progress of targeted prostate specific membrane antigen in the diagnosis and treatment of prostate cancer in detail, analyzes their value and challenges.
Keywords: prostate cancer, PSMA, PET, radioligand therapy, CAR-T, DCs vaccine
Introduction
Prostate cancer (PCa) is the second most common malignancy in men worldwide and the most diagnosed cancer in European countries.1 The United States reported 268,490 new cases and 34,500 deaths in 2022.2 Because of the long course of PCa and the variation in clinical progress between individual patients,3,4 clinicians tend to view the diagnosis and treatment of PCa as a lengthy process.5 If PCa is diagnosed at an early stage, the life expectancy of the patients can be more than 10 years, up to 99%.6 However, for some other patients, recurrent PCa after standard treatment may be aggressive and may not respond to current standard treatment under certain circumstances. About 5% of patients with PCa are clinically diagnosed with distant metastases, of which 15% are diagnosed with local regional metastasis.7 If the disease progresses to advanced PCa, the 5-year overall survival rate is only 30%.7 Therefore, the selection of the most appropriate diagnostic strategy for accurate staging of the disease has a crucial role in guiding the management of PCa patients. Furthermore, the implementation of targeted therapeutic approaches for the management of recurrent PCa holds significant significance.
Currently, prostate-specific antigen (PSA) is used as a preliminary screening indicator for PCa, and further testing will be performed for patients with abnormal PSA values. In recent years, studies have confirmed that PSA screening is clear in reducing the specific mortality of PCa.8 Gleason score is used to determine the degree of aggressiveness of PCa by pathological methods, and can predict the prognosis of PCa patients.9 The multi-parameter magnetic resonance imaging (mpMRI) is recommended for local staging in moderate-high risk patients.10 Computed tomography (CT) and bone scan (BS) for cross-sectional abdominal and pelvic imaging is recommended for the assessment of distant metastasis.11 As an alternative to conventional PCa imaging, molecular imaging is increasingly used to guide treatment decisions in PCa patients in recent years.12 Furthermore, the development of specific targeted therapies for PCa can benefit many patients with advanced PCa. As a diagnostic and therapeutic target for PCa, prostate specific membrane antigen (PSMA) has gained considerable attention. It can be used for positron emitter labeling for positron emission tomography (PET) imaging, as well as β and α emitter labeling for targeted radionuclide therapy.13 In addition, due to its specific expression and internalization in PCa, PSMA can be used in tumor immunotherapy and carry drugs to kill tumors.
PSMA, also known as glutamate carboxypeptidase II, is a transmembrane protein composed of 750 amino acids, which is a membrane protein on the cell surface.14,15 PSMA protein is divided into three parts, namely intracellular part, transmembrane part and extracellular part.16,17 The extracellular portion makes up 95% of the PSMA protein and is the target portion accessible to small molecule and antibody drugs for imaging and therapy. Cell surface PSMA receptor binding proteins can migrate PSMA labeled radioisotopes into cells through internalization, which determines that PSMA can be used as a target for radiodiagnosis and therapy.18 Moreover, studies have shown that the expression of PSMA is increased in 85–100% PCa, especially in metastatic PCa. In cancerous tissues, PSMA expression is significantly upregulated, facilitating tumor progression and metastasis.19–21 PSMA is also low expressed in healthy prostate, small intestine, central nervous system, proximal renal tubules, salivary glands, and lacrimal glands, it may be involved in folate metabolism and cellular processes.22,23 The relatively specific expression of PSMA in PCa determines that it will play a crucial role in the targeted diagnosis and treatment of PCa.
In recent years, numerous studies have shown that PSMA as a diagnostic and therapeutic target is changing the management of PCa.24–27 It was found that PSMA expression was strongly correlated with Gleason score and serum PSA value.28 Furthermore, PSMA expression level was negatively correlated with androgen level. Therefore, the diagnosis and treatment of targeted PSMA can be used in castration-resistant PCa (CRPC) with low androgen activity.29–31 The use of highly specific radio-labeled PSMA ligands for molecular imaging and targeted therapy is an important trend in PCa diagnosis and treatment in recent years.14,15 Therapies based on targeted PSMA have evolved from antibodies and nanoparticles to small molecules.32,33 Small molecule ligand targeting PSMA have been developed and made remarkable progress, mainly 68 gallium (68Ga) and 18 fluorine (18F) labeled PSMA tracers for imaging,34,35, 177 lutetium (177Lu) and 225 actinium (225Ac) labeled PSMA small molecules for treatment.36,37 Researchers use PSMA to bind to therapeutic drugs and rapidly internalize them to preferentially deliver radiopharmaceuticals, antibody-drug couplings, and vaccines to tumors, thus achieving accurate diagnosis and treatment.38–40 More recently, researchers have evaluated the expression of PSMA in circulating tumor cells (CTCs) in order to apply PSMA targeting therapies to the diagnosis of CTCs and to evaluate therapeutic efficacy.41,42 This review summarizes in detail the research progress of targeted PSMA in the diagnosis and treatment of PCa, discusses its clinical application value, and provides the diagnosis and treatment of PCa ideas for clinicians and researchers.
Materials and Methods
To obtain relevant studies on the use of targeted PSMA in the diagnosis and treatment of PCa, we conducted searches in PubMed and Web of Science databases. Using the Boolean operator, we combine the terms PCa and PSMA. We considered articles published in English between August 2016 and August 2023. The published articles included were prospective and retrospective clinical studies, systematic reviews, and meta analyses, all of which reported the application of targeted PSA in the diagnosis and treatment of PCa. A total of 328 articles were obtained through a comprehensive search, PubMed and Web of Science. Then, the retrieved studies were screened, and reviews (218) and some basic studies43 were excluded. Finally, 64 relevant studies were included in this review for analysis (Figure 1).
Figure 1.
The review flow diagram of selected studies.
Application of PET Targeting PSMA in PCa Diagnosis
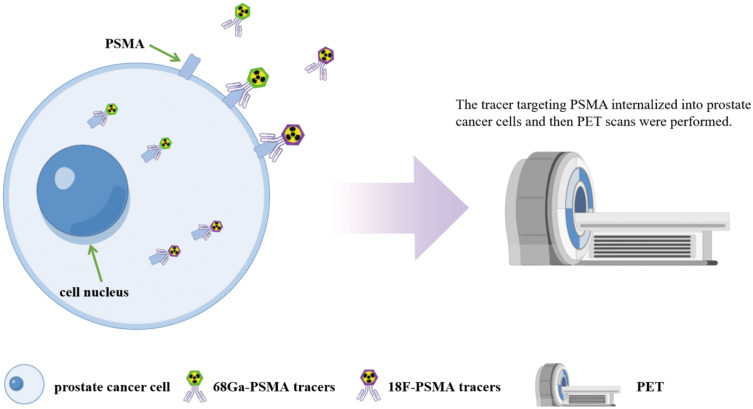
Studies have shown that routine imaging examinations such as CT and magnetic resonance imaging (MRI) to detect lymph node metastasis are insufficient for newly diagnosed PCa. Radiolabeled PSMA PET is increasingly used in PCa worldwide. The technique first uses the radioactive element labeled PSMA tracer injected into the patient, and then the radioactive element labeled PSMA tracer specifically binds to the PSMA protein internalizes into the PCa cell. Finally, PET imaging is used to achieve the purpose of molecular imaging (Figure 2). Currently, there are mainly 68Ga and 18F labeled PSMA tracers. PET technology targeting PSMA has been successfully used in the diagnosis of primary PCa and the staging of biochemical recurrent PCa. Moreover, researchers combined PSMA PET with MRI and CT respectively, which can achieve accurate location of the lesions and display the pathophysiological characteristics of the lesions in the diagnosis of PCa, so as to conduct a good staging of PCa, and thus improve the accuracy of diagnosis. Researchers have also conducted a large number of clinical studies on the diagnostic efficacy of these two tracers (Table 1). In addition, the diagnostic efficacy of PSMA PET was compared with mpMRI and BS. The results of clinical studies indicate that PSMA-PET has great potential in the diagnosis of PCa, and can effectively stage PCa patients and optimize treatment strategies.
Figure 2.
The principle of PSMA PET imaging in PCa diagnosis is illustrated by Figdraw. The technique first uses the radioactive element labeled PSMA tracer injected into the patient, and then the radioactive element labeled PSMA tracer specifically binds to the PSMA protein on the surface of the PCa cells and internalizes into the cell. Finally, PET imaging is used to achieve the purpose of molecular imaging.
Table 1.
Studies on the Application of PET Targeting PSMA in the Diagnosis of PCa
| Interventions | Type of Study | Objectives | Number of Studies and/or Patients | Conclusion | References |
|---|---|---|---|---|---|
| 18F-PSMA PET/CT | Prospective non-randomized controlled study | Researchers evaluated the utility of 18F-PSMA PET/CT in patients with metastatic PCa. | 18 patients | 18F-PSMA PET/CT can detect a large number of suspected PCa sites, many of which are hidden in conventional imaging mode. | Rowe SP et al, 201644 |
| BS and 68Ga-PSMA PET | Retrospective cohort study | This study compared the diagnostic efficacy of 68Ga-PSMA PET and (99m)Tc BS for bone metastases in PCa. | 126 patients | 68Ga-PSMA PET is superior to BS in the detection of the affected bone area and the determination of the overall bone involvement in PC patients. | Pyka T et al, 201645 |
| mpMRI and 68Ga-PSMA PET | Prospective non-randomized controlled study | This study compared the diagnostic efficacy of Multiparametric MRI and 68Ga-PSMA PET in detecting and locating tumors in the prostate. | 20 patients | 68Ga-PSMA PET combined with multiparametric MRI can improve the local staging of retropubic PCa patients undergoing radical resection. | Rhee H et al, 201646 |
| 18F-PSMA PET and 68Ga-PSMA PET | Retrospective cohort study | The purpose of this study was to evaluate diagnostic efficiency of 18F-PSMA PET and 68Ga-PSMA PET | 191 patients | The diagnostic efficiency of 18F-PSMA PET is not inferior to that of 68Ga-PSMA PET, and has the advantage of 18F labeling. | Dietlein F et al, 201747 |
| 68Ga-PSMA PET/CT and mpMRI | Prospective non-randomized controlled study | This study compared the value of 68Ga-PSMA PET/CT and mpMRI in staging of high-risk PCa. | 36 patients | Ga-PSMA PET/CT can be used for lymph node and metastatic staging of high-risk PCa. | Tulsyan S et al, 201748 |
| 68Ga-PSMA PET/CT | A systematic review | This study investigated the diagnostic value of 68Ga-PSMA PET/CT in bone metastasis of PCa. | 37 studies | 68Ga-PSMA PET/CT showed more lesions than bone scan, and its diagnostic performance improved in the primary stage of PCa, but not in mCRPC. | Zacho HD et al, 201749 |
| 68Ga-PSMA-PET | A systematic review | This study systematically evaluated the application value of 68Ga-PSMA-PET in the primary staging of high-risk PCa | 12 studies /322 patients |
The diagnosis of 68Ga-PSMA PET appears to be superior to conventional imaging in the initial staging of PCa. | Corfield J et al, 201850 |
| 68Ga-PSMA PET/CT | A systematic review | This study evaluated the role of 68Ga-PSMA PET/CT scanning in patients with recurrent PCa. | 37 studies | 68Ga-PSMA PET/CT is also effective for recurrence localization with extremely low PSA levels. | Eissa A et al, 201851 |
| 68Ga-PSMA PET/CT and PET/ MRI | A systematic review and meta-analysis | This study evaluated 68Ga-PSMA PET/CT or PET/ MRI for PCa staging, diagnostic test accuracy, and adverse reactions to restaging of PSA elevation after initial treatment. | 15 studies/ 1256 patients | 68Ga-PSMA PET/CT has clinical significance in detecting the recurrence site of PSA in patients with PSA < 1.0 ng/mL after radical prostatectomy. | Von Eyben FE et al, 201852 |
| 68Ga-PSMA PET/CT | Prospective non-randomized controlled study | This study evaluated the 68Ga-PSMA PET/CT detection rate for PCa recurrence sites. | 332 patients | 68Ga-PSMA-11 PET/CT has a good diagnostic value for PCa patients with BCR after radical surgery. | Ceci F et al, 201953 |
| 68Ga-PSMA PET | Meta-analysis | This study investigated the diagnostic value of 68Ga-PSMA PET for initial staging and BCR of PCa. | 29 studies | 68Ga-PSMA has good localization value in the initial stage and BCR of metastatic PCa. | Hope TA et al, 201954 |
| 18F-PSMA PET/CT | A Systematic review and meta-analysis | This study evaluated the detection rate of 18F-PSMA PET/CT in biochemical recurrent PCa. | 6 studies /645 patients | 18F-PSMA PET/CT has a good detection rate for biochemical recurrent PCa, and the detection rate is correlated with PSA value. | Treglia G et al, 201955 |
| 68Ga-PSMA PET | Prospective multicenter non-randomized controlled study | A study was conducted to determine whether 68Ga-PSMA PET could be used to treat recurrent pancreatic cancer. | 635 patients | 68Ga-PSMA PET can identify the recurrence sites of more than half of the patients with BCR of PCa, which can provide a basis for the change of treatment plan. | Fendler WP et al, 202056 |
| 68Ga-PSMA PET | A systematic review and meta-analysis | This study investigated the diagnostic value of 68Ga-PSMA PET for lymph node metastases in PCa. | 14 studies /462 patients | In patients with BCR after radical treatment for PCa, PMAS-PET before salvage lymph node dissection has high accuracy. | Kimura S et al, 202057 |
| 68Ga-PSMA PET/CT | A diagnostic meta-analysis | This study evaluated the diagnostic value of 68Ga-PSMA PET/CT in lymph node staging in patients. | 10 studies/ 701 patients | In patients with moderate to high-risk PCa, 8Ga-PSMA PET/CT has a high diagnostic value for lymph node staging. | Peng L et al, 202058 |
| 68Ga-PSMA PET | An expedited systematic review | This study investigated the preoperative value of 68Ga-PSMA PET for primary lymph node staging. | 18 studies /969 patients | According to the pathological results, 68Ga-PSMA PET has high diagnostic accuracy. | Petersen LJ et al, 202059 |
| 18F-PSMA PET/CT | Prospective non-randomized controlled study | This study evaluated the positive rate of 18F-PSMA PET/CT in patients with BCR of PCa. | 72 patients | 18F-PSMA PET/CT has a high positive rate and affects the clinical treatment of 60% patients. | Song H et al, 202060 |
| 68Ga-PSMA PET/CT | A diagnostic meta-analysis | This study evaluated the accuracy of 68Ga-PSMA PET/CT in preoperative lymph node staging in patients with moderate to high risk PCa. | 11 studies /904 patients | 68Ga-PSMA PET/CT can be used for preoperative lymph node staging, and patients without lymph node metastasis have a low misdiagnosis rate. | Tu X et al, 202061 |
| 68Ga-PSMA PET | A systematic review and meta-analysis | This study compared 68Ga-PSMA PET with MRI in the diagnosis of lymph node metastatic staging of PCa. | 13 studies/ 1597 patients | Ga-PSMA PET is more sensitive than MRI in detecting metastases in moderate and high-risk PCa before treatment, and may be a more effective weapon for predicting lymph node metastasis before radical surgery. | Wu H et al, 202062 |
| 18F-PSMA PET/CT | Prospective, multicenter cohort study | This study investigated the diagnostic value of 18F-PSMA PET/CT in lymph node staging of primary PCa. | 117 patients | 18F-PSMA PET/CT showed high specificity but limited sensitivity in the diagnosis of pelvic lymph node metastasis of primary PCa. | Jansen BHE et al, 202163 |
| 68Ga-PSMA PET | Prospective and retrospective cross-sectional studies | This study identified the accuracy of 68Ga-PSMA PET. | 35 studies / 3910 patients | 68Ga-PSMA PET has higher sensitivity and specificity than conventional imaging in the diagnosis of PCa. | Matushita CS et al, 202164 |
| 68Ga-PSMA PET/CT | A meta-analysis | This study compared 68Ga-PSMA PET/CT with mpMRI for preoperative pelvic lymph node staging in patients with PCa. | 9 studies/ 640 patients | Compared with mpMRI, 68Ga-PSMA PET/CT showed a trend of higher sensitivity and diagnostic accuracy. | Wang X et al, 202165 |
| 68Ga-PSMA PET/CT | A meta-analysis | This study evaluated the value of 68Ga-PSMA-PET/CT and BSin the diagnosis of PCa. | 4 studies/ 318 patients | 68Ga-PSMA-PET/CT has higher diagnostic efficacy for bone metastasis of PCa, which is worthy of clinical application. | Zhao R et al, 202166 |
| 68Ga-PSMA PET/CT | A systematic review and meta-analysis | This study investigated the feasibility of 68Ga-PSMA PET/CT imaging in the diagnosis of primary PCa. | 9 studies/ 547 patients | Compared with conventional imaging, 68Ga-PSMA PET/CT has higher sensitivity and specificity in the diagnosis of primary PCa. | Hu X et al, 202243 |
| 18F-PSMA PET/CT | A systematic review and meta-analysis | This study evaluated the performance of 18 F-PSMA PET/CT in patients with primary PCa. | 12 studies/ 540 patients | 18F-PSMA PET/CT has excellent performance in detecting localized prostate tumor lesions and regional lymph node metastases. | Huang YT et al, 202267 |
| PSMA-PET-TB | A systematic review and meta-analysis | This study evaluated the accuracy of SMA-PET-TB in diagnosing csPCa. | 5 studies/ 497 patients | PSMA-PET-TB has good diagnostic accuracy for csPCa, and combined with MRI can improve the diagnostic accuracy. | Kawada T et al, 202268 |
| 18F-PSMA PET/CT | A systematic review and meta-analysis | This study systematically evaluated the detection rate of 18F-PSMA-1007 PET/CT in PCa patients. | 15 studies/ 1022 patients | 18F-PSMA-1007 PET/CT has high application value in PCa. | Liu X et al, 202269 |
| 18F-PSMA PET/CT | A systematic review and meta-analysis | This study evaluated the diagnostic value of 18f- PSMA PET/CT in primary or BCR PCa. | 11 studies/ 799 patients | 18F-PSMA PET/CT has high diagnostic value for primary and BCR PCa. As serum PSA levels increase, diagnostic accuracy improves. | Liu X et al, 202270 |
| 68Ga-PSMA PET/CT | Prospective no randomized controlled study | This study investigated the application value of 68Ga-PSMA PET/CT in N and M staging in patients with PCa. | 81 patients | 68Ga-PSMA PET/CT has high diagnostic efficacy in N and M stages of middle and high risk PCa patients, and is superior to pelvic mpMRI in detecting local regional lymph node metastasis. | Szigeti F et al, 202271 |
| 68Ga-PSMA PET/CT | A meta-analysis | This study compared the diagnostic efficacy of 68Ga-PSMA PET/CT with (99m)Tc-MDP BS for bone metastases associated with PCa. | 6 studies/ 546 patients | 68Ga-PSMA PET/CT was superior to (99m)Tc-MDP BS in the diagnosis of PCa bone metastases. | Joshi A et al, 202272 |
68Ga-PSMA PET/CT
PET with 68Ga-labeled PSMA (68Ga-PSMA) is an emerging imaging method, which has been widely used in the diagnostic study of PCa due to its ability to locate the focus of PCa. Corfield et al50 systematically evaluated the application value of 68Ga-PSMA PET in the primary diagnosis of high-risk PCa. 68Ga-PSMA PET was used in the diagnosis of primary PCa. The ability of 68Ga-PSMA PET to detect malignant lesions was found to be evident in various studies, most of which showed an improved detection rate of 68Ga-PSMA PET compared to conventional imaging modalities (CIM).73,74 Similarly, Hu X et al43 explored the feasibility of 68Ga-PSMA PET/CT in the diagnosis of primary PCa through a systematic review and meta-analysis. A total of nine studies were included for analysis, 47 patients with primary PCa and 443 lesions underwent 68Ga-PSMA PET/CT scans. The results showed that the sensitivity of 68Ga-PSMA PET/CT in the diagnosis of primary PCa was 67% to 97%. Compared with conventional imaging, 68Ga-PSMA PET/CT has higher sensitivity and specificity in the diagnosis of primary PCa. In addition, Matushita CS et al64 confirmed the accuracy of 68Ga-PSMA PET technology in PCa diagnosis through systematic review. The summary sensitivity and specificity of diagnosis were 0.90 (0.86~0.93) and 0.90 (0.82~0.96), respectively. These results indicate that 68Ga-PSMA PET has higher sensitivity and specificity in the diagnosis of PCa than conventional imaging.73 These results indicate that 68Ga-PSMA PET has greater diagnostic efficacy than conventional imaging in the diagnosis of PCa, and therefore has great potential. Based on its advantages of high detection rate, 68Ga-PSMA PET/CT should be popularized in the detection of primary PCa.
Recurrence of PCa after initial treatment is one of the major challenges facing urologists. Therefore, it is urgent to develop an imaging technique for early detection of recurrent lesions. In recent years, the diagnostic efficacy of 68Ga-PSMA PET for recurrent PCa has been extensively studied. Ceci et al53 evaluated the detection rate of 68Ga-PSMA PET/CT for PCa recurrence sites through a prospective study. In this study, 24.7% of cases were found to be limited to pelvic disease, and 28.9% of the cases had at least one distant lesion. This study showed that 68Ga-PSMA PET/CT has good diagnostic value for PCa patients with treatment failure. Eissa et al51 evaluated the role of 68Ga-PSMA PET/CT scanning in patients with recurrent PCa after radical treatment through a systematic review of the literature. After biochemical recurrence (BCR) of PCa, the primary objective is to locate the site of the recurrent lesion. The results of this study confirm that 68Ga-PSMA PET/CT also appears to be effective in the detection of recurrence with PSA < 0.5 ng/mL, thus enabling early selection of the optimal treatment strategy for some patients. Similarly, von Eyben et al52 evaluated the diagnostic value of 68Ga-PSMA PET/CT for recurrent PCa through meta-analysis. The results showed that 68Ga-PSMA PET/CT has clinical significance in detecting the recurrence site of PSA patients with PSA < 1.0 ng/mL after radical prostatectomy. At present, the guidelines still do not recommend salvage lymph node dissection (sLND) for patients with BCR of PCa due to the low accuracy of existing conventional imaging diagnosis. Kimura et al57 studied the diagnostic value of 68Ga-PSMA PET for lymph node metastasis of PCa confirmed by sLND pathology. The results showed that in patients with BCR of PCa after effective treatment, the diagnosis of PSMA-PET before sLND surgery had high accuracy, with high positive and negative predictive values. 68Ga-PSMA PET may benefit patients with sLND, making it possible to precisely remove lesions or regional lymph nodes. The value of 68Ga-PSMA PET in the diagnosis of recurrent PCa is that it can change the treatment of PCa patients. Fendler et al56 described the influence of 68Ga-PSMA PET on the management of recurrent PCa. The study was a prospective multicentre trial that reported treatment changes in patients after 68Ga-PSMA PET diagnosis. The results showed that 68Ga-PSMA PET could identify the recurrence site of more than half of the patients with BCR of PCa, and translate into changes in treatment plan.
Due to the diagnostic value of 68Ga-PSMA PET for different stages of PCa, it is also used for pathological staging of PCa patients. Petersen et al59 confirmed that PSMA PET is a promising method for staging primary lymph nodes of PCa through a systematic review. The included studies took the pathological results as reference, and finally confirmed that PSMA PET has high diagnostic accuracy for primary lymph node staging. Peng et al58 evaluated the diagnostic value of 68Ga-PSMA PET/CT for lymph node staging in PCa patients through a meta-analysis of diagnostic tests. The diagnostic sensitivity was 0.84 (95% CI 0.55~0.95) and specificity was 0.95 (95% CI 0.87~0.98). This study also reaffirmed the overall diagnostic value of 68Ga-PSMA PET/CT for lymph node staging in patients with moderate and high-risk PCa. Besides, Hope et al54 investigated the value of 68Ga-PSMA PET in the initial staging of PCa through a meta-analysis. The results showed that the sensitivity and specificity of 68Ga-PSMA PET in the initial stage diagnosis of PCa were 0.74 (95% CI: 0.51~0.89) and 0.96 (95% CI: 0.85~0.99), respectively. The results of this study indicate that 68Ga-PSMA has good localization value in the initial stage of metastatic PCa. Similarly, Tu et al61 evaluated the accuracy of 68Ga-PSMA PET/CT for preoperative lymph node staging in patients with moderate and high risk PCa, using the pathological examination results of pelvic lymph node dissection as the reference standard. The results indicate that 68Ga-PSMA PET/CT can be used for preoperative lymph node staging, which can effectively reduce the misdiagnosis rate of patients without lymph node metastasis.
In addition, recent studies have shown that PSMA-PET targeted biopsy (PSMA-PET-TB) is associated with the detection of clinically significant PCa (csPCa). Kawada et al68 evaluated the accuracy of PSMA-PET-TB in the diagnosis of csPCa. The combined sensitivity and specificity of PSMA-PET-TB for csPCa detection were 0.89 (95% CI: 0.85~0.93) and 0.56 (95% CI: 0.29~0.80), respectively. The results of this study indicated that PSMA-PET-TB had good diagnostic accuracy for csPCa, and it was found that PSMA-PET-TB combined with MRI could improve the diagnostic accuracy. PSMA-PET has a good clinical application prospect in the detection of csPCa. More prospective studies are needed to further explore the true clinical efficacy of SMA-PET-TB in order to promote clinical application.
18f-PSMA Pet/Ct
Currently, PET/CT using radioactive element labeled PSMA has been recommended to detect primary PCa. Compared with other PSMA-targeting tracers, 18F-PSMA has been found to have lower urinary tract clearance and better image resolution, which is more beneficial for the treatment of primary tumors.35,75 In order to more accurately evaluate the detection performance of 18F-PSMA PET/CT in patients with primary PCa, Huang et al67 conducted a related systematic review and meta-analysis. A total of 12 studies were included in the study, and results showed that the pooled detection rate of 18F-PSMA was 94%, the positive predictive value of lymph node metastases in the test area was 0.94, and the positive predictive value of localized prostate tumors was 0.84. This study demonstrated the superior performance of 18F-PSMA in detecting localized PCa lesions and regional lymph node metastases.
There have also been numerous studies on the application of 18F-PSMA PET/CT in the diagnosis of metastatic PCa. The results show that it has clinical significance in the location of metastatic PCa. Rowe et al44 evaluated the utility of 18F-PSMA PET/CT in patients with metastatic PCa. Eighteen patients with suspected recurrence or recurrence of PCa underwent 18F-PSMA PET/CT imaging. This study compared the detection of suspected metastatic PCa foci with PET and CIM. A total of 139 positive sites for metastatic 18F-PSMA uptake were detected in 8 patients, while only 45 lesions were found in CIM. These results indicate that 18F-PSMA imaging agent can detect a large number of suspected PCa sites, many of which are hidden or uncertain in CIM. Similarly, Jansen et al63 discussed the diagnostic value of 18F-PSMA PET/CT in lymph node staging of primary PCa through a prospective, multicenter cohort study. The results showed that 18F-PSMA PET/CT had high specificity (94.4%) but limited sensitivity (41.2%) in the diagnosis of pelvic lymph node metastasis of primary PCa. These studies provide strong evidence for the use of 18F-PSMA PET imaging agents in the detection of metastatic PCa, and further support the importance of PET imaging targeting PSMA in PCa.
Furthermore, researchers evaluated the value of 18F-PSMA PET/CT in the diagnosis of biochemical recurrent PCa in a number of clinical studies. Song et al60 evaluated the positive rate of 18F-PSMA PET/CT in patients with BCR of PCa using a single-center prospective study. The overall positive rate of 18F-PSMA PET/CT was 85%, which increased with the increase of PSA level. The study also found that 18F-PSMA PET detected more lesions than conventional imaging. This study shows that 18F-PSMA PET/CT is a promising diagnostic tool. Liu et al69 systematically evaluated the detection rate of 18F-PSMA PET/CT in PCa patients with different serum PSA levels. The results showed that 18F-PSMA PET/CT had a 90% to 100% detection rate in newly diagnosed PCa patients and a 47% to 100% PCa detection rate in BCR patients. This study confirmed that 18F-PSMA PET/CT has high application value in PCa including primary tumor and BCR. In addition, systematic review studies have demonstrated that the diagnostic accuracy of 18F-PSMA PET/CT improves with the increase of serum PSA level.55,70 All these studies confirmed the diagnostic value of 18F-PSMA PET/CT in biochemical recurrent PCa, and found that serum PSA level was a factor affecting the diagnostic efficiency.
There has also been a study comparing the diagnostic efficacy of 68Ga-PSMA PET to 18F-PSMA PET. Dietlein et al47 compared the sensitivity of PSA stratification to the diagnosis of PCa patients. In this study, 191 patients with BCR received 18F-PSMA PET (62 cases) or 68Ga-PSMA PET (129 cases) scan. This study confirmed that the 18F-PSMA PET is not inferior to the 68Ga-PSMA PET and found that 18F-PSMA imaging can even improve sensitivity to locate recurrent tumors in patients with moderately elevated PSA levels after prostatectomy. This study suggests that 18F-PSMA PET may have a greater advantage in the diagnosis of PCa, and more prospective randomized controlled studies are needed to confirm this.
Comparison of Diagnostic Performance of PSMA- PET and Multiparametric MRI Imaging
The mpMRI has high sensitivity and specificity in the detection of PCa, and is currently the most widely used imaging technology for tumor localization and cancer staging. mpMRI exerts a crucial role in risk stratification of patients, active surveillance of low-risk patients, and eventual post-treatment recurrence monitoring.76 The diagnostic efficacy of mpMRI was compared with that of 68Ga-PSMA PET. The results also showed that 68Ga-PSMA PET had a higher value for medium and high risk PCa metastases and lymph node metastases. Tulsyan et al48 conducted a prospective non-randomized controlled study comparing the value of 68Ga-PSMA PET versus mpMRI for high-risk PCa staging. The study involved 36 cases with high-risk PCa. Results demonstrate that 68Ga-PSMA PET/CT can be used to stage high-risk PCa’s lymph nodes and metastatic sites. Wu et al62 compared the diagnostic efficacy of 68Ga-PSMA PET and mpMRI for lymph node metastasis staging of PCa through systematic review and meta-analysis. The results showed that the sensitivity of 68Ga-PSMA PET to detect metastasis was higher than that of MRI, and the specificity was slightly different in moderate and high-risk PCa before treatment. This study also confirmed that 68Ga-PSMA PET is a more effective predictor of lymph node metastasis before radical surgery. Subsequently, Wang et al65 compared the diagnostic efficacy of 68Ga-PSMA PET/CT and mpMRI for preoperative pelvic lymph node staging in patients with PCa. This study found that the 68Ga-PSMA PET/CT showed greater sensitivity and diagnostic accuracy in detecting pelvic lymph node metastasis in PCa patients compared to mpMRI. Furthermore, Szigeti et al71 discussed the application value of 68Ga-PSMA PET/CT in N and M staging before radical prostatectomy and enlarged pelvic lymph node dissection for PCa patients at high risk. The results showed that 68Ga-PSMA PET/CT had high diagnostic efficacy in N and M stages of medium-high risk PCa patients, and was superior to pelvic mpMRI in detecting local regional lymph node metastasis. The above studies once again confirmed the diagnostic value of 68Ga-PSMA PET in the staging of medium-high risk PCa, which is conducive to further accurate radical treatment of PCa.
In addition, the investigators also found that PSMA PET combined with mpMRI had better diagnostic value in the staging of PCa. Rhee et al46 compared the diagnostic efficiency of prostate mpMRI imaging and PSMA PET for localized PCa, and confirmed the diagnosis by using in vitro prostate histopathology to determine the true sensitivity and specificity of these imaging modes. In the prospective clinical trial, 20 patients with localized PCa were enrolled and scheduled for radical prostatectomy. All patients underwent mpMRI and PSMA PET examination before surgery, and were directly compared with pathological sections. The results found that a significant proportion of cancers with both imaging methods were likely to be missed or underestimated. PSMA PET combined with mpMRI (PSMA PET/MRI) is helpful to optimize the staging of retropubic PCa patients undergoing radical resection. This study suggests that PSMA PET/MRI may also be an excellent option in the diagnosis of PCa.
Comparison of Diagnostic Performance of PSMA-PET and Bone Scintigraphy
Currently, BS using (99m)TC-labeled methylene bisphosphate (99m)Tc-MDP remains the recommended imaging mode for detecting bone metastasis in patients with PCa. These studies have confirmed that PSMA PET/CT is increasingly recognized as an effective means of assessing disease severity in patients with PCa. Therefore, a large number of studies have been conducted to compare the diagnostic capabilities of the two in PCa bone metastasis. The results show that PSMA PET/CT is better than BS in the diagnosis of PCa bone metastasis, and has great value in the diagnosis of middle and advanced PCa.
Pyka et al45 compared the diagnostic efficacy of 68Ga-PSMA PET and (99m)Tc BS in PCa bone metastasis through a retrospective study. This study retrospectively analyzed the diagnostic value of BS and PSMA PET in 126 patients with PCa. The cohort was divided into clinical subgroups: primary PCa, BCR PCa, and metastatic castration-resistant PCa(mCRPC). The results of this study showed that 68Ga-PSMA PET was superior to BS in detecting the affected bone area and determining the overall bone involvement. Zacho et al49 discussed the diagnostic value of 68Ga-PSMA PET/CT for bone metastasis of PCa through a systematic review. This study found that 68Ga-PSMA PET/CT has a broad application prospect in the diagnosis of PCa. 68Ga-PSMA PET/CT showed more lesions than bone imaging, and the diagnostic efficiency of mCRPC was improved compared with bone imaging. Researchers have also demonstrated the ability of 68Ga-PSMA-PET/CT to detect malignant bone lesions through systematic analysis and whether this method is superior to existing BS methods. Zhao et al66 evaluated the value of 68Ga-PSMA PET/CT and BS in the clinical diagnosis of PCa from the perspective of evidence-based medicine. By comparing the diagnostic results of 68Ga-PSMA-PET/CT with bone imaging, it has higher sensitivity and specificity than bone imaging, and has higher diagnostic efficacy for bone metastasis of PCa, which is worthy of clinical application. In addition, Joshi et al72 conducted a meta-analysis to compare the diagnostic efficacy of 68Ga-PSMA PET/CT and (99m)Tc BS for bone metastasis of PCa. The results showed that the combined sensitivity and specificity of 68Ga-PSMA PET/CT were 98% and 97%, and that of (99m)Tc BS were 83% and 68%, respectively. The results showed that the 68Ga-PSMA PET/CT was superior to (99m)Tc BS in the diagnosis of PCa bone metastasis. These studies confirm the superior efficacy of PSMA PET/CT in the diagnosis of bone metastasis in PCa. More prospective multi-center studies are needed to clarify its diagnostic performance and its superiority over existing methods before 68Ga-PSMA PET/CT can be widely used in the clinic.
Application of Targeting PSMA Therapy in PCa Treatment
The progression of PCa varies significantly between individuals, with some patients developing mCRPC after standardized treatment. Currently, the main treatments for mCRPC include endocrine therapy with abiraterone and Enzalutamide and chemotherapy represented by docetaxel. However, the median survival improvement for mCRPC was not ideal. In recent years, targeted therapy has brought hope to patients with middle and advanced tumors, and has achieved initial efficacy. Due to the specific and stable expression of PSMA in advanced PCa tissues, PSMA has become the most promising target for PCa. Therefore, researchers have attempted to apply PSMA targeted therapy (radioligand therapy, immunotherapy, antibody-drug conjugate, etc) in the treatment of mCRPC and conducted a large number of studies (Tables 2–4), and obtained preliminary efficacy. The results suggest that PSMA targeted therapy has great potential in the treatment of advanced PCa.
Table 2.
Studies on the Application of RLT Targeting PSMA in the Treatment of PCa
| Interventions | Type of Study | Objectives | Number of Studies and/or Patients | Results | References |
|---|---|---|---|---|---|
| 177Lu-PSMA-617 RLT | Retrospective study | This study examined the value of 177Lu-PSMA-617 in the treatment of mCRPC. | 30 patients | 177Lu-PSMA-617 is a promising new treatment option for mCRPC. | Kratochwil C et al, 201677 |
| 177Lu-PSMA-617 RLT | Systematic review and meta-analysis |
This study evaluated 177 the efficacy of Lu-PSMA in mCRPC. | 10 studies/ 369 patients | About two-thirds of the patients responded to treatment with 177Lu-PSMA-617. | Calopedos RJS et al, 201778 |
| 177Lu- PSMA-617 RLT | Prospective single-group clinical trial | Lu-PSMA-617 was evaluated in this study for its efficacy and safety in the treatment of mCRPC. | 31 patients | 177Lu-PSMA-617 was a safe and effective method for the treatment of mCRPC. | Yadav MP et al, 201779 |
| 177Lu-PSMA-617 RLT | Prospective single-group clinical trial | Patients with mCRPC were enrolled in this study in order to evaluate the efficacy of 177Lu-PSMA-617 and its effect on quality of life. | 30 patients | Radionuclide therapy with 177Lu-PSMA-617 has a high response rate, low toxicity, and reduced pain in patients with mCRPC that has progressed after conventional treatment. | Hofman MS et al, 201880 |
| 177Lu-PSMA-617 RLT | A meta-analysis | This study examined the effect of 177Lu-PSMA-617 nuclide therapy on patients with mCRPC. | 10 studies/ 455 patients | After one cycle of Lu-PSMA-617 RLT in mCRPC patients, PSA decreased in two-thirds of patients and PSA decreased more than 50% in one-third of patients. | Kim YJ et al, 201881 |
| 177Lu-PSMA-617 RLT | A systematic review | This study investigated whether 177Lu-PSMA RLT had similar efficacy and adverse effects to third-line treatment. | 12 studies/ 669 patients | For mCRPC patients, 177Lu-PSMA-617 RLT showed better efficacy and fewer adverse reactions than third-line treatment. | Von Eyben FE et al, 201882 |
| 177Lu-PSMA-617 RLT | Multicenter prospective randomized controlled study | This study compared the therapeutic activity and safety of cabazitaxel chemotherapy with 177Lu-PSMA-617 RLT in patients with mCRPC. | 200 patients | 177Lu-PSMA-617 RLT offers a potential life-prolonging treatment option for patients with mCRPC. | Hofman MS et al, 201983 |
| 177Lu-PSMA-617 RLT | Prospective non-randomized controlled study | This study determined the outcomes of patients who were not treated with 177Lu-PSMA-617 RLT due to low PSMA expression. | 16 patients | The 177Lu-PSMA-617 RLT may be an effective therapy for patients with advanced PCa that has progressed after standard treatment. | Thang SP et al, 201984 |
| 177Lu-PSMA-617 RLT | A systematic review and meta-analysis | The purpose of this study was to determine whether 177Lu-PSMA RLT was effective and safe for the treatment of metastatic CRPC. | 17 studies/ 744 patients | 177Lu-PSMA RLT is an effective treatment for mCRPC. | Yadav MP et al, 201936 |
| 177Lu-PSMA-617 RLT | Retrospective single-group clinical trial | This study evaluated the clinical efficacy of 177Lu-PSMA-617 RLT in the treatment of mCRPC. | 30 patients | 177Lu-PSMA-617 RLT appears to be an effective treatment method for patients with mCRPC because it improves their quality of life by increasing health and relieving disease-related pain. | Marinova M et al, 202085 |
| 177Lu-PSMA-617 RLT | A systematic review and meta-analysis | This study evaluated the efficacy of mCRPC patients receiving 177Lu-PSMA-617 RLT. | 36 studies/ 2346 patients | Patients treated with intensive regimen 177Lu-PSMA-617 RLT survived longer than those treated with conventional regimen. PSA decreased by more than 50% in half of patients. | Von Eyben FE et al, 202086 |
| 177Lu-PSMA-617 RLT | Prospective non-randomized controlled study | This study evaluated the safety and efficacy of 177Lu PSA-617 combined with idronoxil (NOX66) in the treatment of end-stage mCRPC patients. | 32 patients | Lu PSMA-617 combined NOX66 is a safe and feasible therapeutic strategy. | Crumbaker M et al, 202187 |
| 177Lu-PSMA RLT | A systematic review and meta-analysis | This study evaluated the efficacy and toxicity of 177Lu-PSMA RLT in the treatment of PCa | 24 studies / 1192 patients | 177Lu-PSMA RLT had a relatively high PSA response rate and a relatively low rate of severe toxic reaction in the treatment of PCa. | Sadaghiani MS et al, 202188 |
| 177Lu-PSMA-617 RLT | Prospective randomized controlled study | This study evaluated the efficacy of 177Lu-PSMA-617 RLT in the treatment of mCRPC. | 831 patients | The addition of 177Lu-PSMA-617 RLT to standard treatment in mCRPC patients prolonged radiologically progression-free survival and overall survival. | Sartor O et al, 202189 |
| 225Ac-PSMA RLT | A systematic review and meta-analysis | This study systematically evaluated the role of 225Ac-PSMA RLT in mCRPC. | 10 studies/ 256 patients | 225Ac-PSMA RLT is an effective option for patients with mCRPC. | Satapathy S et al, 202190 |
| 225Ac-PSMA RLT | Prospective randomized controlled study | The objective was to evaluate the role of the 225Ac-PSMA RLT as a salvage treatment option for mCRPC. | 40 patients | 225Ac-PSMA-617 RLT showed good biochemical reaction in the treatment of mCRPC patients and improved the survival rate of patients. | Ballal S et al, 202191 |
| 177Lu-PSMA-617 RLT | A meta-analysis | It was evaluated in this study whether 177Lu-PSMA-617 RLT was effective and safe for the treatment of mCRPC. | 12 studies/ 508 patients | It is safe and effective to treat mCRPC with 177Lu-PSMA-617 RLT. | Zhang H et al, 202192 |
| 225Ac-PSMA RLT | A meta-analysis | This study evaluated the efficacy of 225Ac-PSMA RLT in patients with mCRPC. | 9 studies/ 263 patients | After treatment with 225Ac-PSMA RLT in mCRPC patients, PSA decreased by more than 50% in 61% of patients and decreased in 84% of patients. | Lee DY et al, 202293 |
| 225Ac-PSMA-617 RLT | A systematic review and meta-analysis | This study evaluated the efficacy and safety of 225 AC-PSMA-617 RLT in patients with mCRPC. | 6 studies/ 201 patients | 225Ac-PSMA-617 RLT is an effective and safe treatment option for mCRPC patients with relatively low toxicity. | Ma J et al, 202237 |
| 177Lu-PSMA-617 RLT | Retrospective single-group clinical trial | This study investigated the safety and efficacy of 177Lu-PSMA-617 in combination with NOX66. | 56 patients | NOX66 combined with 177Lu-PSMA-617 RLT is a safe and feasible treatment strategy in patients with mCRPC. | Pathmanandavel S et al, 202294 |
| 177Lu-PSMA RLT | A systematic review and meta-analysis | This study evaluated the effectiveness of targeting 177Lu-PSMA RLT in the treatment of CRPC. | 69 studies/4157 patients | A higher proportion of patients with PSA decreased by ≥50% after treatment with 177Lu-PSMA RLT compared to the control group. | Sadaghiani MS et al, 202295 |
| 177Lu-PSMA-617 RLT | Prospective randomized controlled study | This study compared the efficacy and safety of 177Lu-PSMA-617 RLT with docetaxel in patients with mCRPC. | 40 patients | 177Lu-PSMA-617 RLT has been shown to be safe in the treatment of mCRPC and is not inferior to docetaxel. | Satapathy S et al, 202296 |
Table 3.
Studies on the Application of Immunotherapy Targeting PSMA in the Treatment of PCa
| Interventions | Type of Study | Objectives | Number of Studies and/or Patients | Results | References |
|---|---|---|---|---|---|
| BPX101 (PSMA Targeted Autoantigen Presenting Cell Vaccine) | Prospective single-group clinical trial | The study investigated the safety and activity of BPX101 in patients with mCRPC | 18 patients | BPX101 has no dose-limiting toxicity. Immunoupregulation and antitumor activity were observed through PSA reduction and objective tumor regression. | Sonpavde G et al, 201797 |
| DCs targeted therapy loaded PSMA | A systematic review and meta-analysis | This study evaluated the efficacy of DCs loaded PSMA targeted therapy in mCRPC patients. | 6 studies/153 patients | This study did not prove that DCs loaded PSMA targeted therapy had a significant therapeutic effect on mCRPC. However, no significant adverse effects were observed. | Mohammadzadeh M et al, 201898 |
| CAR engineered NK-92 cells targeting PSMA (PSMA-CAR-NK-92) | Basic Research | This study explored the potential efficacy of PSMA-CAR-NK-92 for PCa treatment. | - | PSMA-CAR-NK-92 cells significantly inhibited the growth of cell tumors and improved the survival rate of PCa mice. | Montagner IM et al, 202099 |
| CAR-T cells co-expressing TGF-β/IL-7 inverted cytokine receptor (ICR) targeting PSMA (TGF-β/IL-7-PSMA-CAR-T) | Basic Research | This study investigated the lethal activity of TGF-β/IL-7-PSMA-CAR-T cells against PCa. | - | TGF-β/IL-7-PSMA-CAR-T cells may serve as a novel PCa treatment strategy and support the translation of this enhanced approach in the clinical setting. | Weimin S et al, 2020100 |
| CAR-T cells co-expressing IL23 targeting PSMA CAR-T cells (IL23-PSMA-CAR-T) | Basic Research | This study investigated the antitumor activity of IL23-PSMA-CAR T cells in PCa. | - | The efficacy of IL23-SMA-CAR-T cells in the treatment of PCa was superior to that of SMA-CAR- T cells. | Wang D et al, 2020101 |
| PSMA-CAR-T cells | Basic Research | This study investigated the antitumor activity of PSMA-CAR-T in PCa. | - | Local injection of PSMA-CAR-T cells can eradicate the transplanted tumor in the xenograft model of PCa. | Alzubi J et al, 2020102 |
| PSMA-targeting TGFβ-insensitive armored CAR-T cells (PSMA- TGFβ-CAR-T) | Prospective single-group clinical trial | This study investigated the feasibility and safety of PSMA-TGFβ-CAR-T cells in clinical application. | 18 patients | The clinical application of PSMA- TGFβ-CAR T cells is feasible and generally safe. | Narayan V et al, 2022103 |
| PSMA-CAR-T cells | Basic Research | This study investigated whether PSMA-CAR-T cells combined with docetaxel were more effective in treating PCa. | - | PSMA-CAR-T cells combined with docetaxel significantly improved the antitumor effect of xenograft PCa models. | Zhang X et al, 2022104 |
Table 4.
Studies on the Application of Other Treatment Methods Targeting PSMA in the Treatment of PCa
| Interventions | Type of Study | Objectives | Number of Studies and/or Patients | Results | References |
|---|---|---|---|---|---|
| Targeting PSMA docetaxel nanoparticles (BIND-014) | Prospective single-group clinical trial | This study investigated the safety and efficacy of BIND-014 in the treatment of mCRPC. | 42 patients | BIND-014 treatment is effective and well tolerated in mCRPC patients who have not received chemotherapy. | Autio KA et al, 2018105 |
| PSMA ADC | Prospective single-group clinical trial | This study evaluated the safety and initial antitumor effects of the PSMA ADC in patients with refractory PCa. | 52 patients | PSMA ADC showed acceptable toxicity in an extensively pretreated mCRPC population. Antitumor activity was observed at doses over and including 2.5 mg/kg. | Petrylak DP et al, 2019106 |
| PSMA ADC | Prospective single-group clinical trial | This study investigated the tumor killing activity of PSMA ADC against PCa. | 119 patients | PSMA ADC showed certain tumor killing activity of PCa through PSA reduction and TC reduction. | Petrylak DP et al, 2020107 |
| A humanized monoclonal antibody targeting PSMA (MLN2704) | Prospective non-randomized controlled study | This study evaluated the dose-limiting toxicity and maximum tolerated dose of MLN2704. | 62 patients | MLN2704 has limited activity in metastatic castration-resistant PCa. | Milowsky MI et al, 2016108 |
Radioligand Therapy (RLT) Targeting PSMA
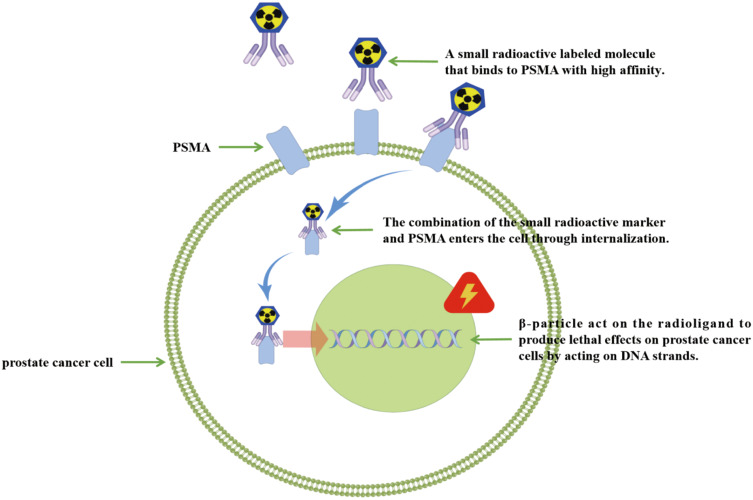
Progressive mCRPC is a highly fatal disease, and new effective therapeutic agents are urgently needed to improve patient outcomes. PSMA is an excellent target for radionuclide therapy in mCRPC. In addition to having a high affinity with PCa cells and a long retention period, vipivotide tetraxetan (PSMA-617) has low renal uptake, making it a good choice for therapeutic applications. RLT is a tumor killing technique that targets cell membrane proteins. PSMA-617, labeled by radioactive elements, specifically binds to PSMA, and the binding molecules enter the cell through internalization. The β particle treatment then kills PCa cells by acting on binding molecules that disrupt their DNA strands (Figure 3). Preliminary clinical studies on the treatment of PCa based on this principle have achieved gratifying results (Table 2).
Figure 3.
The principle of RLT treatment is shown in the figure by Figdraw. PSMA-617, labeled by radioactive elements, specifically binds to PSMA, and the binding molecules enter the cell through internalization. The β particle treatment then kills PCa cells by acting on binding molecules that disrupt their DNA strands.
Labeling of PSMA ligands with lutetium-177Lu has shown promising therapeutic results. 177Lu-PSMA-617 is a small radioactive labeled molecule that binds to PSMA with high affinity, thereby achieving β-particle therapy targeting metastatic castration-resistant PCa. It has been determined that this approach is safe and effective by most published studies to date.36,77–80,85,86,92 These studies found that the treatment of 177Lu-PSMA-617 was achieved by lowering the level of PSA in response to mCRPC. Sadaghiani et al95 systematically evaluated the effectiveness of RLT targeting PSMA in CRPC. The results of the study found that a higher proportion of patients with PSA decreased by more than 50% after RLT treatment compared to the control group. A decrease in PSA after treatment with RLT or a greater than 50% decrease in PSA prolongs survival. Kim et al81 studied mCRPC patients through a meta-analysis and found that about two-thirds of PSA decreased and one-third of PSA decreased by more than 50% after the first cycle of Lu-PSMA-617 RLT. Decreased PSA indicated prolonged survival after Lu-PSMA-617 RLT. Moreover, Thang et al84 conducted a prospective trial to determine outcomes in patients screened but not treated with 177Lu-PSMA-617 radioligand due to low PSMA expression. The results of this study found that low PSMA expression was associated with poor prognosis and low survival in mCRPC patients who progressed after conventional treatment. These studies suggest that 177Lu-PSMA-617 RLT may be an effective therapy for patients with high PSMA expression in patients with advanced PCa that has progressed after standard therapy.
Currently, the efficacy of 177Lu-PSMA-617 RLT in mCRPC patients compared with third-line treatment is controversial. von Eyben et al82 discussed whether 177Lu-PSMA RLT has similar efficacy and adverse reactions to third-line treatment. The results of the study showed that 177Lu-PSMA-617 RLT had better efficacy and fewer adverse reactions in mCRPC patients than the third-line treatment (enzalutamide and carpacasel). Similarly, the researchers compared the activity and safety of carbimazole chemotherapy with that of 177Lu-PSMA RLT. The results also found that 177Lu-PSMA-617 RLT offers a potential life-prolonging treatment option for patients with mCRPC.83 In addition, Satapathy et al96 compared the efficacy and safety of 177Lu-PSMA-617 RLT and docetaxel in the treatment of mCRPC patients through a prospective non-inferior trial. The results of the study found that 177Lu-PSMA-617 RLT was shown to be safe and not inferior to docetaxel in the treatment of mCRPC, and it could be used earlier in the course of the disease, rather than only for advanced end-stage disease.
Trials of 177Lu-PSMA-617 RLT in patients with mCRPC have shown favorable safety and efficacy, and the investigators hypothesized that the combined strategy may be better for improved outcomes. Idronoxil (NOX66) is a synthetic flavonoid derivative with radiosensitizing properties. Crumbaker et al87 evaluated the safety and efficacy of 177Lu-PSMA-617 RLT combined with NOX66 in the treatment of end-stage mCRPC patients. The results of the study found NOX66 in combination with 177Lu-PSMA-617 RLT to be a safe and feasible treatment strategy in mCRPC patients receiving third-line treatment. Similarly, Pathmanandavel et al94 reported the same results in a Phase I/II clinical study combining 177Lu-PSMA-617 RLT and NOX66. Besides, the efficacy of 177Lu-PSMA-617 RLT in addition to standard treatment for mCRPC was also discussed.89 The results of this study showed that the addition of 177Lu-PSMA-617 RLT in addition to standard treatment extended radiologic progression-free survival and overall survival in patients with advanced PSMA-positive mCRPC. These studies suggest that the 177Lu-PSMA-617 RLT combined with standard PCa therapy and radiosensitizers is a promising direction for the treatment of mCRPC.
225Ac has high linear energy transfer and short path length, which can induce DNA double strands break. In recent years, it has been found that 225AC-labeled PSMA ligand (225Ac-PSMA-617) targeting radionuclide therapy is another promising treatment mode for mCRPC, with good efficacy and safety.90,93 Ma et al37 conducted a meta-analysis on the efficacy and safety of 225Ac-PSMA-617 RLT in the treatment of mCRPC based on existing clinical evidence. The results showed that 225Ac-PSMA-617 RLT is an effective and safe treatment option for mCRPC patients with relatively low toxic effects. Similarly, Ballal et al91 investigated the role of 225AC-SMA −617 RLT as a salvage treatment for mCRPC. The results show that 225Ac-PSMA-617 RLT has a favorable biochemical response in the treatment of mCRPC patients, improves patient survival and reduces treatment-related toxicity, and is a promising treatment option. However, most of the above studies are retrospective studies, and prospective randomized controlled studies are still needed in the future to judge the efficacy and survival benefits compared with existing clinical therapies.
Immunotherapy Targeting PSMA
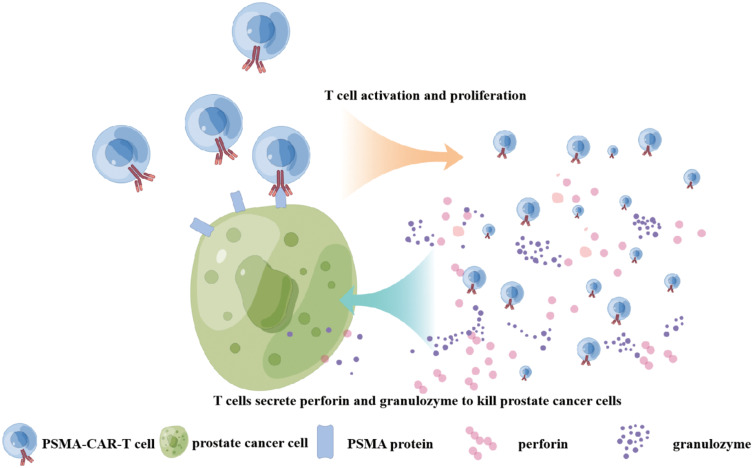
Adoptive immunotherapy is a promising strategy, especially for controlling advanced tumor stages by specifically targeting the tumor through chimeric antigen receptor T cells (CAR-T) therapy. In recent years, CAR-T cells technology has made great progress in the treatment of hematologic malignancies, and researchers are also trying to apply CAR-T cells technology to the treatment of solid tumors. PSMA, as a membrane protein specifically expressed in PCa cells, has been widely used in the study of CAR-T cells therapy. The single chain antibody variable region gene fragment (scFv) of PSMA is combined with T cells by gene editing technique to construct PSMA-targeting CAR-T cells (PSMA-CAR-T). PSMA-CAR-T cells can bind to PSMA on the surface of PCa cells to activate CAR-T cells. Activated CAR-T cells can kill PCa by secreting cytokines such as perforin and granulozyme (Figure 4). Immunotherapy targeting PSMA for PCa has been preliminarily studied and confirmed to have certain potential in the treatment of PCa (Table 3).
Figure 4.
The killing mechanism of PSMA-CAR T cells on PCa cells is shown in the figure by Figdraw. PSMA-CAR-T cells bind to PSMA on the surface of PCa cells to activate CAR-T cells. Activated CAR-T cells can kill PCa by secreting cytokines such as perforin and granulozyme.
Alzubi et al102 designed a PSMA-CAR-T capable of recognizing one epitope in PSMA, and conducted related preclinical studies on anti-PCa. The results of this study demonstrated that local injection of PSMA-CAR T cells eradicated established human PCa xenografts in a preclinical mouse model. In addition, intravenous application of PSMA-CAR-T cells in combination with low-dose docetaxel chemotherapy significantly inhibited tumor growth, while docetaxel or CAR-T cells alone did not. This study suggests that PSMA is a viable target for the treatment of PCa by CAR-T cell technology, but its killing efficacy needs to be optimized. With the development of four-generation CAR-T cells technology, researchers have also attempted to enhance the tumor killing function of PSMA-CAR-T cells by co-expressing IL-7, IL23100,101. The tumor killing activity of optimized PSMA-CAR-T cells was significantly enhanced in vitro and in vivo. Narayan et al103 reported the results of a Phase 1 clinical trial that PSMA-CAR-T cells treat TGF-β-resistant CRPC (NCT03089203). The results confirmed that the CAR-T cells were feasible and safe in the treatment of TGF-β-resistant CRPC. Furthermore, Zhang et al104 explored whether the combined application of docetaxel and PSMA-CAR-T cells could be more effective in the treatment of PCa. The results demonstrated the PSMA-CAR-T cells combined with docetaxel significantly improved the antitumor effect of PCa in mice.
Recently, dendritic cells (DCs) have been used as vaccines in many malignancies to induce immunity against specific tumor antigens. Sonpavde et al97 investigated the safety and activity of DCs targeted therapy loaded with PSMA (BPX101) in mCRPC patients through a Phase I clinical trial. Eighteen advanced mCRPC patients were enrolled in the study who received three intradermal doses of BPX101. The results showed that BPX101 had no dose-limiting toxicity, and showed robust tumor killing efficacy through PSA reduction and objective tumor regression. In addition, Mohammadzadeh et al98 evaluated the proportion of mCRPC patients receiving DCs targeted therapy loaded with PSMA in a meta-analysis, and investigated possible adverse effects caused by this treatment mode. This study did not prove that DCs had a significant therapeutic effect on mCRPC. However, no significant adverse effects were observed. Therefore, DCs targeted therapy loaded with PSMA should be optimized to achieve better tumor killing efficacy.
Antibody-Drug Conjugates (ADCs) Targeting PSMA
PSMA has emerged as a pivotal target in the diagnosis and treatment of prostate cancer. ADCs targeting PSMA represent a promising therapeutic strategy due to their ability to deliver cytotoxic agents directly to cancer cells, thereby minimizing off-target effects.109 The mechanism of PSMA-targeted ADCs involves the utilization of monoclonal antibodies that specifically bind to PSMA on the surface of prostate cancer cells. Once bound, the ADCs is internalized, and the cytotoxic payload is released within the cancer cell, leading to cell death.110 This targeted approach enhances the therapeutic index compared to conventional chemotherapy.
Researchers have also tried ADCs targeting PSMA in the treatment of PCa, and have shown some promise (Table 4). In preclinical models of advanced PCa, PSMA ADCs show effective and selective antitumor activity. Petrylak et al106 evaluated the safety, pharmacokinetics and initial antitumor effects of ADCs targeting PSMA in patients with refractory PCa. The results of this study found that the ADCs targeting PSMA showed acceptable toxicity in an extensively pretreated mCRPC population. Antitumor activity was observed at doses over and including 2.5 mg/kg. The observed antitumor activity supports further evaluation of this novel agent for the treatment of advanced metastatic PCa. In addition, Petrylak et al107 evaluated the efficacy of the ADCs targeting PSMA in mCRPC patients who progressed after abiraterone/enzalutamide treatment. The results of the study found that the ADCs targeting PSMA showed some activity in PSA reduction, CTCs reduction, and radiological evaluation in treated mCRPC patients. These studies confirm the efficacy of the ADCs targeting PSMA in the treatment of PCa, and more prospective randomized controlled studies are needed to confirm the efficacy.
BIND-014 is docetaxel-encapsulating nanoparticle decorated with small-molecule PSMA targeting ligands. The modified nanoparticle is enriched in the tumor microenvironment and can be internalized on binding to a PSMA-expressing cell, enabling the preferential delivery of docetaxel to the tumor.111 Delivery of PSMA-targeted BIND-014 to tumors showed significant therapeutic efficacy, and selective reduction of PSMA-positive CTCs after treatment, which is of significance for disease monitoring in patients. Autio et al105 investigated the safety and efficacy of PSMA targeted BIND-014 in the treatment of mCRPC (NCT01812746). The results of this study suggest that BIND-014 therapy is effective and well tolerated in mCRPC patients who have not received chemotherapy. Antitumor activity may correlate with the level of PSMA expression on CTCs, suggesting that patients who might benefit from this treatment can be identified before treatment begins. Besides, a study have attempted to apply MLN2704, a humanized monoclonal antibody targeting PSMA, to the treatment of PCa, but its activity in the treatment of mCRPC is limited.108 More prospective studies are needed to examine its antitumor activity in PCa.
However, challenges remain in the clinical application of PSMA-targeted ADCs. Resistance mechanisms, including the downregulation of PSMA expression and alterations in internalization pathways, pose significant hurdles. Furthermore, identifying the most appropriate patient populations and optimal treatment regimens continues to be an area of active research. In summary, PSMA-targeted ADCs hold great promise in the treatment of prostate cancer, yet ongoing studies are needed to fully understand their efficacy and address the challenges associated with their use.
Clinical Trials Related to PMSA Targeted Therapy
PSMA-targeted therapies have generated substantial interest and promising clinical results in recent years. As clinical trials continue to explore innovative treatment strategies, it is expected that these approaches will lead to improved outcomes for patients with prostate cancer, further solidifying the role of PSMA as a pivotal target in oncology. Among these advances, both radioligand therapy (RLT) and CAR T-cell therapy have featured prominently (Table 5). Numerous studies have produced promising preliminary results, while many others remain ongoing and are expected to expand our understanding of PSMA-targeted therapies.
Table 5.
Clinical Trials of Targeted PMSA Therapy Strategies for PCa Treatment
| Identifying code | Patients (n) | Clinical stage | Study Title | Interventions | First Posted | Status |
|---|---|---|---|---|---|---|
| NCT05204927 | 439 | Phase 3 | 177Lu-PSMA-I&T for Metastatic Castration-Resistant Prostate Cancer | Drug: 177Lu-PSMA-I&T Drug: Abiraterone with Prednisone or Enzalutamide |
2022–01-24 | Active, not recruiting |
| NCT05521412 | 30 | Phase 1Phase 2 | EValuation of radIOLigand Treatment in mEn With Metastatic Castration-resistant Prostate Cancer With [161Tb] Tb-PSMA-I&T | Drug: [161 Tb] Tb PSMA I&T | 2022–08-30 | Active, not recruiting |
| NCT05150236 | 93 | Phase 2 | EVOLUTION: 177Lu-PSMA Therapy Versus 177Lu-PSMA in Combination with Ipilimumab and Nivolumab for Men With mCRPC | Drug: 177Lu-PSMA-617 Drug: Ipilimumab Drug: Nivolumab |
2021–12-09 | Active, not recruiting |
| NCT03454750 | 43 | Phase 2 | Radiometabolic Therapy (RMT) With 177Lu PSMA 617 in Advanced Castration Resistant Prostate Cancer (CRPC) | Drug: 177Lu-PSMA | 2018–03-06 | Active, not recruiting |
| NCT05555017 | 60 | Phase 2 | Tracer-Guided Surgery for Recurrent Prostate Cancer | Procedure: PSMA-radioguided surgery Drug: ADT |
2022–09-26 | Recruiting |
| NCT04429451 | 100 | Phase 1Phase 2 | PSMA-specific CAR-T Cell Therapy | Biological: 4SCAR-PSMA T cells | 2020–06-12 | Recruiting |
| NCT05893381 | 70 | Phase 2 | Lu-PSMA and Stereotactic Radiotherapy Versus Radiotherapy Alone for Prostate Cancer (LUST) | Drug: [177Lu] Lu-PSMA I&T Radiation: Stereotactic Radiotherapy |
2023–06-07 | Recruiting |
| NCT03658447 | 37 | Phase 1 Phase 2 |
PRINCE (PSMA-lutetium Radionuclide Therapy and ImmuNotherapy in Prostate CancEr) | Drug: Pembrolizumab Drug: 177Lu-PSMA |
2018–09-05 | Completed |
| NCT05383079 | 36 | Phase 1Phase 2 | Combination of Radium-223 and Lutetium-177 PSMA-I&T in Men with Metastatic Castration-Resistant Prostate Cancer | Drug: Lutetium-177 PSMA-I&T Drug: Radium-223 |
2022–05-19 | Recruiting |
| NCT04443062 | 58 | Phase 2 | Lutetium-177-PSMA-617 in Oligo-metastatic Hormone Sensitive Prostate Cancer | Drug: 177Lu-PSMA-617 | 2020–06-23 | Recruiting |
| NCT06402331 | 60 | Phase 2Phase 3 | FPI-2265 (225Ac-PSMA-I&T) for Patients With PSMA-Positive Metastatic Castration-Resistant Prostate Cancer (mCRPC) | Drug: FPI-2265 | 2024–05-07 | Recruiting |
| NCT03828838 | 10 | Phase 1Phase 2 | Lutetium-177-PSMA-617 in Low Volume Metastatic Prostate Cancer | Drug: Lu-177 PSMA-617 | 2019–02-04 | Completed |
| NCT06220188 | 20 | Phase 2 | PSMA-RLT in Biochemically Recurrent PCa | Drug: [177Lu] Lu-PSMA I&T | 2024–01-23 | Recruiting |
Notes: All clinical trials were download at http://www.clinical trials.gov (access date: September 28, 2024).
One of the landmark studies in this domain is the VISION trial, a Phase 3 randomized study evaluating the efficacy of radioligand therapy with [177Lu]Lu-PSMA-617 in patients with mCRPC. The trial reported substantial improvements in overall survival and progression-free survival for patients receiving this treatment compared to standard care alone, making it a cornerstone in our approach to managing advanced prostate cancer.112 Moreover, the TheraP trial has directly compared [177Lu] Lu-PSMA-617 to cabazitaxel, establishing the radioligand’s superiority in reducing PSA levels and improving quality of life in mCRPC patients.113 These outcomes underscore the potential of PSMA-targeted therapies to alter the treatment landscape for advanced prostate cancer.
In addition to RLT, newer PSMA-targeted immunotherapies have been under evaluation. A notable example is the Phase 1 trial involving PSMA-targeting CAR T-cells, which demonstrated effective targeting of PSMA-expressing cancer cells, providing an innovative route of immunotherapy in advanced prostate cancer.103 The results suggested these engineered T-cells can overcome some of the challenges related to TGFβ signaling in the tumor microenvironment, which generally hampers the immune response. Clinical trials investigating combination therapies are also emerging. The trial evaluating the efficacy of a single dose of [177Lu] Lu-PSMA-617 followed by maintenance pembrolizumab demonstrated encouraging results in mCRPC patients, indicating potential synergistic effects between radioligand therapy and checkpoint inhibitors.114
Furthermore, ongoing investigations are exploring the administration of [177Lu] Lu-PSMA-617 prior to radical prostatectomy in high-risk localized prostate cancer cases, aiming to enhance surgical outcomes and reduce recurrence rates. 115 Each of these studies reflects a nuanced understanding of how PSMA-targeted therapies can fit into various stages of prostate cancer management. Challenges remain in the integration of these therapies into routine practice, particularly regarding patient selection, the heterogeneity of PSMA expression, and the development of resistance mechanisms. Additionally, there are ongoing discussions surrounding optimizing dosage protocols in PSMA therapy to minimize adverse effects while maximizing therapeutic outcomes.
Discussion
This review summarized the research progress of the diagnosis and treatment of targeted PSMA in patients with PCa. It was found that PCa patients could benefit from the application of targeted PSMA due to the specific expression of PSMA on the surface of PCa cells. These results indicate that PET with radionuclide PSMA targeting radionuclide tracer has a higher detection rate in primary and recurrent tumors, and a higher diagnostic efficacy in staging of medium-high risk PCa patients compared with conventional imaging. In addition, PSMA-targeting RLT, immunotherapy, and ADC showed good safety and efficacy in the treatment of mCRPC compared with third-line therapy. Therefore, with the gradual deepening of relevant research on the diagnosis and treatment technology of targeted PSMA, it is believed that it may play a greater role in the diagnosis and treatment of PCa.
As we all know, mpMRI has high sensitivity and specificity in the detection of PCa, and is currently the most widely used imaging technology for tumor localization and cancer staging.76 However, for the past few years, with the development of molecular imaging technology and the strengthening of clinical demands for accurate diagnosis of PCa, molecular imaging technology targeting PSMA has been widely used in the diagnosis of PCa, and has achieved better imaging results. Numerous studies have shown that 68Ga-PSMA PET scan can be used in the staging of recurrent or metastatic PCa because of its excellent diagnostic efficacy in primary, metastatic and metastatic lymph nodes.43,50–53,64 Second, the 18F-PSMA PET was found to be no worse than the 68Ga-PSMA PET in detecting localized prostate tumor lesions and regional lymph node metastases.47 These findings suggest that PSMA-targeting molecular imaging has great potential in the diagnostic staging of PCa.
In recent years, targeted therapy of PSMA has also made some achievements in mCRPC. Clinical studies have shown that 177Lu-PSMA-617 and 225Ac-PSMA-617 RLT have good biochemical reactions in the treatment of mCRPC patients, improve the survival rate of patients, reduce treatment-related toxicity, and are a promising salvage treatment option.37,81,84,91,95 The study also compared the efficacy of 177Lu-PSMA-617 RLT with docetaxel in mCRPC. It was found that 177Lu-PSMA-617 RLT was proven to be safe and not inferior to docetaxel. The study also suggests that 177Lu-PSMA-617 RLT may be used earlier in the course of the disease, rather than only for late end-stage disease. Furthermore, the investigators found that 177Lu-PSMA-617 combined with standard PCa treatment and radiosensitizers may lead to better outcomes in the treatment of mCRPC. Therefore, targeted PSMA RLT has great potential in the treatment of PCa, especially mCRPC.
Immunotherapy of PCa is also the direction of researchers’ efforts. Several preclinical studies of PSMA-targeted CAR-T cells technology in PCa have also confirmed the efficacy and safety in the treatment of PCa.102,103 Current clinical trials have initially confirmed the safety of PSMA-CAR-T in humans, and ongoing studies further confirm its tumor killing efficacy. Meanwhile, in order to overcome the suppressive immune microenvironment of PCa, researchers are trying to modify the PSMA-CAR-T structure by gene editing technology to enhance its tumor killing function.100,101 DCs targeted therapy with PSMA loading has also achieved initial efficacy, and its clinical safety has been confirmed. However, studies on its efficacy have produced mixed results.97,98 Therefore, there is still a long way to go before immunotherapy targeting PSMA can be widely used in PCa. In addition, it has been found that PSMA ADC can deliver anti-tumor drugs to tumor tissues to achieve precise tumor killing, and can kill CTCs.106,107,111 This technique has some potential for PCa treatment, but has been poorly studied. More clinical studies are needed to confirm its efficacy.
Conclusions
In summary, the progression of PCa can not be accurately diagnosed by staging, resulting in the failure of accurate management of PCa patients. Therefore, some patients progressed to mCRPC. Even if the treatment measures for mCRPC are very timely and adequate, patients still cannot get a very good clinical prognosis. Targeted PSMA diagnostics can accurately diagnose the clinical progression of PCa at the molecular level, thereby optimizing the clinical management of the patients. In addition, targeted PSMA therapy also holds promise for the treatment of mCRPC. It is believed that with the rapid development of targeted diagnosis and treatment technology, PCa will certainly benefit from the diagnosis and treatment of targeted PSMA.
Acknowledgments
We would like to thank all the authors who participated in the writing of this review, and also thank the Third Affiliated Hospital of Gansu University of Traditional Chinese Medicine for facilitating the publication of this review, and thank the FIGDRAW online graphics website for the convenience of making the images in this article.
Funding Statement
This work was supported by Baiyin Science and Technology Plan Project (Clinical Application research on early screening of Prostate cancer in Baiyin Area: 2022-3-6Y) and Baiyin First People’s Hospital Science and Technology Plan Project (2019YK-08).
Abbreviations
PCa, prostate cancer; mpMRI, multi-parameter magnetic resonance imaging; CT, computed tomography; PSMA, prostate specific membrane antigen; PSA, prostate-specific antigen; CRPC, castration-resistant prostate cancer; 68Ga, 68 gallium; 18F, 18 fluorine; 177Lu, 177 lutetium; 225Ac, 225 actinium; CTCs, circulating tumor cells; PET, positron emission tomography; MRI, magnetic resonance imaging; 68Ga-PSMA, 68Ga-labeled PSMA; CIM, conventional imaging modalities; BCR, biochemical recurrence; sLND, salvage lymph node dissection; PSMA-PET-TB, PSMA-PET targeted biopsy; csPCa, clinically significant prostate cancer; PSMA-PET-TB, PSMA-PET targeted biopsy; BS, bone scan; mCRPC, metastatic castration-resistant prostate cancer; RLT, radioligand therapy; 225Ac-PSMA-617, 225AC-labeled PSMA ligand; CAR-T, chimeric antigen receptor T cells; scFv, single chain antibody variable region gene fragment; PSMA-CAR-T, PSMA-targeting CAR-T cells; DCs, dendritic cells; ADC, Antibody-Drug Conjugate; ALP, Alkaline Phosphatase.
Data Sharing Statement
The datasets used and/or analyzed during the current study are available from the corresponding authors on reasonable request.
Ethics Approval and Consent to Participate
The study was approved by the Ethics Committee of Third Affiliated Hospital of Gansu University of Traditional Chinese Medicine and is consistent with the Declaration of Helsinki.
Author Contributions
All authors made a significant contribution to the work reported, whether that is in the conception, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no conflict of interest.
References
- 1.Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clinicians. 2021;71:209–249. doi: 10.3322/caac.21660 [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. Ca a Cancer J Clinicians. 2022;72:7–33. doi: 10.3322/caac.21708 [DOI] [PubMed] [Google Scholar]
- 3.Rebbeck TR. Prostate cancer genetics: variation by race, ethnicity, and geography. Semin Radiat Oncol. 2017;27:3–10. doi: 10.1016/j.semradonc.2016.08.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Siegel DA, O’Neil ME, Richards TB, Dowling NF, Weir HK. Prostate Cancer Incidence and Survival, by Stage and Race/Ethnicity - United States, 2001-2017. Morbid Mortal Week Rep. 2020;69:1473–1480. doi: 10.15585/mmwr.mm6941a1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Elemam NM, Hotait HY, Saleh MA, et al. Prostate cancer: a tale of two sides. Nat Rev Urol. 2019;16:141. doi: 10.1038/s41585-019-0152-z [DOI] [PubMed] [Google Scholar]
- 6.Rebello RJ, Oing C, Knudsen KE, et al. Prostate cancer. Nat Rev Dis Prim. 2021;7:8. doi: 10.1038/s41572-021-00249-2 [DOI] [PubMed] [Google Scholar]
- 7.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. Ca a Cancer J Clinicians. 2018;68:7–30. doi: 10.3322/caac.21442 [DOI] [PubMed] [Google Scholar]
- 8.Fleshner K, Carlsson SV, Roobol MJ. The effect of the USPSTF PSA screening recommendation on prostate cancer incidence patterns in the USA. Nat Rev Urol. 2017;14:26–37. doi: 10.1038/nrurol.2016.251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang X, Zhang Y, Ji Z, Yang P, Tian Y. Old men with prostate cancer have higher risk of Gleason score upgrading and pathological upstaging after initial diagnosis: a systematic review and meta-analysis. World J Surg Oncol. 2021;19:18. doi: 10.1186/s12957-021-02127-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Annamalai A, Fustok JN, Beltran-Perez J, Rashad AT, Krane LS, Triche BL. interobserver agreement and accuracy in interpreting mpMRI of the prostate: a systematic review. Curr Urol Rep. 2022;23:1–10. doi: 10.1007/s11934-022-01084-y [DOI] [PubMed] [Google Scholar]
- 11.Mottet N, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent. Europ urol. 2017;71:618–629. doi: 10.1016/j.eururo.2016.08.003 [DOI] [PubMed] [Google Scholar]
- 12.de Kouchkovsky I, Aggarwal R, Hope TA. Prostate-specific membrane antigen (PSMA)-based imaging in localized and advanced prostate cancer: a narrative review. Translat Androl Urol. 2021;10:3130–3143. doi: 10.21037/tau-20-1047 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kaewput C, Vinjamuri S. Update of PSMA theranostics in prostate cancer: current applications and future trends. J Clin Med. 2022;11:2738. doi: 10.3390/jcm11102738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jones W, Griffiths K, Barata PC, Paller CJ. PSMA theranostics: review of the current status of PSMA-targeted imaging and radioligand therapy. Cancers. 2020;12:1367. doi: 10.3390/cancers12061367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lütje S, Slavik R, Fendler W, Herrmann K, Eiber M. PSMA ligands in prostate cancer - Probe optimization and theranostic applications. Methods. 2017;130:42–50. [DOI] [PubMed] [Google Scholar]
- 16.Chang SS, Reuter VE, Heston WD, Bander NH, Grauer LS, Gaudin PB. Five different anti-prostate-specific membrane antigen (PSMA) antibodies confirm PSMA expression in tumor-associated neovasculature. Cancer Res. 1999;59:3192–3198. [PubMed] [Google Scholar]
- 17.Chang SS, Heston WD. The clinical role of prostate-specific membrane antigen (PSMA). Urolog Oncol. 2002;7:7–12. doi: 10.1016/S1078-1439(01)00124-7 [DOI] [PubMed] [Google Scholar]
- 18.Liu H, Moy P, Kim S, et al. Monoclonal antibodies to the extracellular domain of prostate-specific membrane antigen also react with tumor vascular endothelium. Cancer Res. 1997;57:3629–3634. [PubMed] [Google Scholar]
- 19.Wright GL, Haley C, Beckett ML, Schellhammer PF. Expression of prostate-specific membrane antigen in normal, benign, and malignant prostate tissues. Urolog Oncol. 1995;1:18–28. doi: 10.1016/1078-1439(95)00002-Y [DOI] [PubMed] [Google Scholar]
- 20.Mannweiler S, Amersdorfer P, Trajanoski S, Terrett JA, King D, Mehes G. Heterogeneity of prostate-specific membrane antigen (PSMA) expression in prostate carcinoma with distant metastasis. Pathol Oncol Res. 2009;15:167–172. doi: 10.1007/s12253-008-9104-2 [DOI] [PubMed] [Google Scholar]
- 21.Kusumi T, Koie T, Tanaka M, et al. Immunohistochemical detection of carcinoma in radical prostatectomy specimens following hormone therapy. Pathol Inter. 2008;58:687–694. doi: 10.1111/j.1440-1827.2008.02294.x [DOI] [PubMed] [Google Scholar]
- 22.Bouchelouche K, Choyke PL, Capala J. Prostate specific membrane antigen- a target for imaging and therapy with radionuclides. Discov Med. 2010;9:55–61. [PMC free article] [PubMed] [Google Scholar]
- 23.Backhaus P, Noto B, Avramovic N, et al. Targeting PSMA by radioligands in non-prostate disease-current status and future perspectives. Eur J Nucl Med Mol Imaging. 2018;45:860–877. doi: 10.1007/s00259-017-3922-y [DOI] [PubMed] [Google Scholar]
- 24.Shahrokhi P, Masteri Farahani A, Tamaddondar M, Rezazadeh F. The utility of radiolabeled PSMA ligands for tumor imaging. Chem Biol Drug Design. 2022;99:136–161. doi: 10.1111/cbdd.13946 [DOI] [PubMed] [Google Scholar]
- 25.Sheehan B, Guo C, Neeb A, et al. Prostate-specific membrane antigen biology in lethal prostate cancer and its therapeutic implications. European Urology Focus. 2022;8:1157–1168. doi: 10.1016/j.euf.2021.06.006 [DOI] [PubMed] [Google Scholar]
- 26.Wang F, Li Z, Feng X, Yang D, Lin M. Advances in PSMA-targeted therapy for prostate cancer. Prost Can Prost Dis. 2022;25:11–26. doi: 10.1038/s41391-021-00394-5 [DOI] [PubMed] [Google Scholar]
- 27.Savir-Baruch B, Werner RA, Rowe SP, Schuster DM. PET imaging for prostate cancer. Radiol Clin North Am. 2021;59:801–811. doi: 10.1016/j.rcl.2021.05.008 [DOI] [PubMed] [Google Scholar]
- 28.Cimadamore A, Gasparrini S, Scarpelli M, et al. Epigenetic modifications and modulators in prostate cancer. Crit Rev Oncogen. 2017;22:439–450. doi: 10.1615/CritRevOncog.2017020964 [DOI] [PubMed] [Google Scholar]
- 29.Pinto JT, Suffoletto BP, Berzin TM, et al. Prostate-specific membrane antigen: a novel folate hydrolase in human prostatic carcinoma cells. Clin Can Res. 1996;2:1445–1451. [PubMed] [Google Scholar]
- 30.Carter RE, Feldman AR, Coyle JT. Prostate-specific membrane antigen is a hydrolase with substrate and pharmacologic characteristics of a neuropeptidase. Proceed Nat Acad Sci United States Am. 1996;93:749–753. doi: 10.1073/pnas.93.2.749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Halsted CH, Ling EH, Luthi-Carter R, Villanueva JA, Gardner JM, Coyle JT. Folylpoly-gamma-glutamate carboxypeptidase from pig jejunum. Molecular characterization and relation to glutamate carboxypeptidase II. J Biol Chem. 1998;273:20417–20424. doi: 10.1074/jbc.273.32.20417 [DOI] [PubMed] [Google Scholar]
- 32.Uspenskaya AA, Nimenko EA, Machulkin AE, Beloglazkina EK, Majouga AG. The importance of linkers in the structure of PSMA ligands. Curr Med Chem. 2022;29:268–298. doi: 10.2174/0929867328666210804092200 [DOI] [PubMed] [Google Scholar]
- 33.Rizzo A, Dall’Armellina S, Pizzuto DA, et al. PSMA radioligand uptake as a biomarker of neoangiogenesis in solid tumours: diagnostic or theragnostic factor? Cancers;2022. 14. doi: 10.3390/cancers15010014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Albisinni S, Sarkis J, Diamand R, De Nunzio C. Prebiopsy (68)Ga-PSMA PET imaging: can we improve the current diagnostic pathway for prostate cancer? Prost Can Prost Dis. 2022;2022:1. [DOI] [PubMed] [Google Scholar]
- 35.Ferrari M, Treglia G. (18)F-PSMA-1007 PET in biochemical recurrent prostate cancer: an updated meta-analysis. Cont Med Molec Imag. 2021;2021:3502389. doi: 10.1155/2021/3502389 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yadav MP, Ballal S, Sahoo RK, Dwivedi SN, Bal C. Radioligand therapy with (177)Lu-PSMA for metastatic castration-resistant prostate cancer: a systematic review and meta-analysis. Am J Roentgenol. 2019;213:275–285. doi: 10.2214/AJR.18.20845 [DOI] [PubMed] [Google Scholar]
- 37.Ma J, Li L, Liao T, Gong W, Zhang C. Efficacy and Safety of (225)Ac-PSMA-617-targeted alpha therapy in metastatic castration-resistant prostate cancer: a systematic review and meta-analysis. Front Oncol. 2022;12:796657. doi: 10.3389/fonc.2022.796657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Vallabhajosula S, Nikolopoulou A, Babich JW, et al. 99mTc-labeled small-molecule inhibitors of prostate-specific membrane antigen: pharmacokinetics and biodistribution studies in healthy subjects and patients with metastatic prostate cancer. J Nucl Med. 2014;55:1791–1798. doi: 10.2967/jnumed.114.140426 [DOI] [PubMed] [Google Scholar]
- 39.Tagawa ST, Milowsky MI, Morris M, et al. Phase II study of Lutetium-177-labeled anti-prostate-specific membrane antigen monoclonal antibody J591 for metastatic castration-resistant prostate cancer. Clin Can Res. 2013;19:5182–5191. doi: 10.1158/1078-0432.CCR-13-0231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Weber JS, Vogelzang NJ, Ernstoff MS, et al.A phase 1 study of a vaccine targeting preferentially expressed antigen in melanoma and prostate-specific membrane antigen in patients with advanced solid tumors. J Immunoth. 2011;(34):556–567. doi: 10.1097/CJI.0b013e3182280db1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Josefsson A, Linder A, Flondell Site D, et al. Circulating tumor cells as a marker for progression-free survival in metastatic castration-naïve prostate cancer. Prostate. 2017;77:849–858. doi: 10.1002/pros.23325 [DOI] [PubMed] [Google Scholar]
- 42.Theil G, Fischer K, Weber E, et al. The use of a new cellcollector to isolate circulating tumor cells from the blood of patients with different stages of prostate cancer and clinical outcomes - a proof-of-concept study. PLoS One. 2016;11:e0158354. doi: 10.1371/journal.pone.0158354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hu X, Wu Y, Yang P, Wang J, Wang P, Cai J. Performance of 68Ga-labeled prostate-specific membrane antigen ligand positron emission tomography/computed tomography in the diagnosis of primary prostate cancer: a systematic review and meta-analysis. International Braz J Urol. 2022;48:891–902. doi: 10.1590/s1677-5538.ibju.2020.0986 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Rowe SP, Macura KJ, Mena E, et al. PSMA-Based [(18)F]DCFPyL PET/CT is superior to conventional imaging for lesion detection in patients with metastatic prostate cancer. Molec Imag Biol. 2016;18:411–419. doi: 10.1007/s11307-016-0957-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Pyka T, Okamoto S, Dahlbender M, et al. Comparison of bone scintigraphy and (68)Ga-PSMA PET for skeletal staging in prostate cancer. Eur J Nucl Med Mol Imaging. 2016;43:2114–2121. doi: 10.1007/s00259-016-3435-0 [DOI] [PubMed] [Google Scholar]
- 46.Rhee H, Thomas P, Shepherd B, et al. Prostate specific membrane antigen positron emission tomography may improve the diagnostic accuracy of multiparametric magnetic resonance imaging in localized prostate cancer. J Urol. 2016;196:1261–1267. doi: 10.1016/j.juro.2016.02.3000 [DOI] [PubMed] [Google Scholar]
- 47.Dietlein F, Kobe C, Neubauer S, et al. PSA-Stratified Performance of (18)F- and (68)Ga-PSMA PET in patients with biochemical recurrence of prostate cancer. J Nucl Med. 2017;58:947–952. doi: 10.2967/jnumed.116.185538 [DOI] [PubMed] [Google Scholar]
- 48.Tulsyan S, Das CJ, Tripathi M, Seth A, Kumar R, Bal C. Comparison of 68Ga-PSMA PET/CT and multiparametric MRI for staging of high-risk prostate cancer68Ga-PSMA PET and MRI in prostate cancer. Nucl Med Commun. 2017;38:1094–1102. doi: 10.1097/MNM.0000000000000749 [DOI] [PubMed] [Google Scholar]
- 49.Zacho HD, Nielsen JB, Haberkorn U, Stenholt L, Petersen LJ. (68) Ga-PSMA PET/CT for the detection of bone metastases in prostate cancer: a systematic review of the published literature. Clin Physiol Funct Imag. 2017. doi: 10.1111/cpf.12480 [DOI] [PubMed] [Google Scholar]
- 50.Corfield J, Perera M, Bolton D, Lawrentschuk N. (68)Ga-prostate specific membrane antigen (PSMA) positron emission tomography (PET) for primary staging of high-risk prostate cancer: a systematic review. World J Urol. 2018;36:519–527. doi: 10.1007/s00345-018-2182-1 [DOI] [PubMed] [Google Scholar]
- 51.Eissa A, Elsherbiny A, Coelho RF, et al. The role of 68Ga-PSMA PET/CT scan in biochemical recurrence after primary treatment for prostate cancer: a systematic review of the literature. Miner Urolog e Nefrolog. 2018;70:462–478. doi: 10.23736/S0393-2249.18.03081-3 [DOI] [PubMed] [Google Scholar]
- 52.von Eyben FE, Picchio M, von Eyben R, Rhee H, Bauman G. (68)Ga-labeled prostate-specific membrane antigen ligand positron emission tomography/computed tomography for prostate cancer: a systematic review and meta-analysis. European Urology Focus. 2018;4:686–693. doi: 10.1016/j.euf.2016.11.002 [DOI] [PubMed] [Google Scholar]
- 53.Ceci F, Castellucci P, Graziani T, et al. (68)Ga-PSMA-11 PET/CT in recurrent prostate cancer: efficacy in different clinical stages of PSA failure after radical therapy. Eur J Nucl Med Mol Imaging. 2019;46:31–39. doi: 10.1007/s00259-018-4189-7 [DOI] [PubMed] [Google Scholar]
- 54.Hope TA, Goodman JZ, Allen IE, Calais J, Fendler WP, Carroll PR. Metaanalysis of (68)Ga-PSMA-11 PET accuracy for the detection of prostate cancer validated by histopathology. J Nucl Med. 2019;60:786–793. doi: 10.2967/jnumed.118.219501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Treglia G, Annunziata S, Pizzuto DA, Giovanella L, Prior JO, Ceriani L. Detection Rate of (18)F-Labeled PSMA PET/CT in biochemical recurrent prostate cancer: a systematic review and a meta-analysis. Cancers. 2019;12:11. doi: 10.3390/cancers12010011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Fendler WP, Ferdinandus J, Czernin J, et al. Impact of (68)Ga-PSMA-11 PET on the management of recurrent prostate cancer in a prospective single-arm clinical trial. J Nucl Med. 2020;61:1793–1799. doi: 10.2967/jnumed.120.242180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kimura S, Abufaraj M, Janisch F, et al. Performance of [(68)Ga] Ga-PSMA 11 PET for detecting prostate cancer in the lymph nodes before salvage lymph node dissection: a systematic review and meta-analysis. Prost Can Prost Dis. 2020;23:1–10. doi: 10.1038/s41391-019-0156-z [DOI] [PubMed] [Google Scholar]
- 58.Peng L, Li J, Meng C, et al. Can (68)Ga-prostate specific membrane antigen positron emission tomography/computerized tomography provide an accurate lymph node staging for patients with medium/high risk prostate cancer? A diagnostic meta-analysis. Radiat Oncol. 2020;15:227. doi: 10.1186/s13014-020-01675-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Petersen LJ, Zacho HD. PSMA PET for primary lymph node staging of intermediate and high-risk prostate cancer: an expedited systematic review. Can Imag. 2020;20:10. doi: 10.1186/s40644-020-0290-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Song H, Harrison C, Duan H, et al. Prospective Evaluation of (18)F-DCFPyL PET/CT in biochemically recurrent prostate cancer in an academic center: a focus on disease localization and changes in management. J Nucl Med. 2020;61:546–551. doi: 10.2967/jnumed.119.231654 [DOI] [PubMed] [Google Scholar]
- 61.Tu X, Zhang C, Liu Z, et al. The Role of 68Ga-PSMA positron emission tomography/computerized tomography for preoperative lymph node staging in intermediate/high risk patients with prostate cancer: a diagnostic meta-analysis. Front Oncol. 2020;10:1365. doi: 10.3389/fonc.2020.01365 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wu H, Xu T, Wang X, et al. Diagnostic Performance of (68)gallium labelled prostate-specific membrane antigen positron emission tomography/computed tomography and magnetic resonance imaging for staging the prostate cancer with intermediate or high risk prior to radical prostatectomy: a systematic review and meta-analysis. World J Men’s Health. 2020;38:208–219. doi: 10.5534/wjmh.180124 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Jansen BHE, Bodar YJL, Zwezerijnen GJC, et al. Pelvic lymph-node staging with (18)F-DCFPyL PET/CT prior to extended pelvic lymph-node dissection in primary prostate cancer - The SALT trial. Eur J Nucl Med Mol Imaging. 2021;48:509–520. doi: 10.1007/s00259-020-04974-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Matushita CS, da Silva AMM, Schuck PN, et al. 68Ga-Prostate-specific membrane antigen (PSMA) positron emission tomography (pet) in prostate cancer: a systematic review and meta-analysis. International Braz J Urol. 2021;47:705–729. doi: 10.1590/s1677-5538.ibju.2019.0817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wang X, Wen Q, Zhang H, Ji B. Head-to-Head Comparison of (68)Ga-PSMA-11 PET/CT and Multiparametric MRI for pelvic lymph node staging prior to radical prostatectomy in patients with intermediate to high-risk prostate cancer: a meta-analysis. Front Oncol. 2021;11:737989. doi: 10.3389/fonc.2021.737989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhao R, Li Y, Nie L, Qin K, Zhang H, Shi H. The meta-analysis of the effect of 68Ga-PSMA-PET/CT diagnosis of prostatic cancer compared with bone scan. Medicine. 2021;100:e25417. doi: 10.1097/MD.0000000000025417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Huang YT, Tseng NC, Chen YK, et al. The detection performance of 18 F-prostate-specific membrane antigen-1007 PET/CT in primary prostate cancer: a systemic review and meta-analysis. Clin Nucl Med. 2022;47:755–762. doi: 10.1097/RLU.0000000000004228 [DOI] [PubMed] [Google Scholar]
- 68.Kawada T, Yanagisawa T, Rajwa P, et al. Diagnostic performance of prostate-specific membrane antigen positron emission tomography-targeted biopsy for detection of clinically significant prostate cancer: a systematic review and meta-analysis. Europ Urol Oncol. 2022;5:390–400. doi: 10.1016/j.euo.2022.04.006 [DOI] [PubMed] [Google Scholar]
- 69.Liu X, Jiang T, Gao C, et al. Detection rate of fluorine-18 prostate-specific membrane antigen-1007 PET/CT for prostate cancer in primary staging and biochemical recurrence with different serum PSA levels: a systematic review and meta-analysis. Front Oncol. 2022;12:911146. doi: 10.3389/fonc.2022.911146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Liu X, Wang Q, Zhang B, Jiang T, Zeng W. Diagnostic accuracy of (18)F-PSMA-1007 PET/CT for prostate cancer in primary staging and biochemical recurrence with different serum PSA levels: a systematic review and meta-analysis. Hell J Nucl Med. 2022;25:88–102. doi: 10.1967/s002449912438 [DOI] [PubMed] [Google Scholar]
- 71.Szigeti F, Schweighofer-Zwink G, Meissnitzer M, et al. Incremental Impact of [(68) Ga]Ga-PSMA-11 PET/CT in Primary N and M staging of prostate cancer prior to curative-intent surgery: a prospective clinical trial in comparison with mpMRI. Molecul Imag Biol. 2022;24:50–59. doi: 10.1007/s11307-021-01650-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zhao G, Ji B. Head-to-head comparison of (68)Ga-PSMA-11 PET/CT and (99m)Tc-MDP bone scintigraphy for the detection of bone metastases in patients with prostate cancer: a meta-analysis. Am J Roentgenol. 2022;219:386–395. doi: 10.2214/AJR.21.27323 [DOI] [PubMed] [Google Scholar]
- 73.Maurer T, Gschwend JE, Rauscher I, et al. Diagnostic Efficacy of (68)Gallium-PSMA positron emission tomography compared to conventional imaging for lymph node staging of 130 consecutive patients with intermediate to high risk prostate cancer. The Journal of Urology. 2016;195:1436–1443. doi: 10.1016/j.juro.2015.12.025 [DOI] [PubMed] [Google Scholar]
- 74.Herlemann A, Wenter V, Kretschmer A, et al. (68)Ga-PSMA positron emission tomography/computed tomography provides accurate staging of lymph node regions prior to lymph node dissection in patients with prostate cancer. Europ Urol. 2016;70:553–557. doi: 10.1016/j.eururo.2015.12.051 [DOI] [PubMed] [Google Scholar]
- 75.Hennrich U, Eder M. [(68)Ga]Ga-PSMA-11: the First FDA-Approved (68)Ga-Radiopharmaceutical for PET imaging of prostate cancer. Pharmaceuticals. 2021;14:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Midiri F, Vernuccio F, Purpura P, Alongi P, Bartolotta TV. Multiparametric MRI and radiomics in prostate cancer: a review of the current literature. Diagnostics. 2021;12:11. doi: 10.3390/diagnostics12010011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kratochwil C, Giesel FL, Stefanova M, et al. PSMA-targeted radionuclide therapy of metastatic castration-resistant prostate cancer with 177Lu-Labeled PSMA-617. J Nucl Med. 2016;57:1170–1176. doi: 10.2967/jnumed.115.171397 [DOI] [PubMed] [Google Scholar]
- 78.Calopedos RJS, Chalasani V, Asher R, Emmett L, Woo HH. Lutetium-177-labelled anti-prostate-specific membrane antigen antibody and ligands for the treatment of metastatic castrate-resistant prostate cancer: a systematic review and meta-analysis. Prost Can Prost Dis. 2017;20:352–360. doi: 10.1038/pcan.2017.23 [DOI] [PubMed] [Google Scholar]
- 79.Yadav MP, Ballal S, Tripathi M, et al. (177)Lu-DKFZ-PSMA-617 therapy in metastatic castration resistant prostate cancer: safety, efficacy, and quality of life assessment. Eur J Nucl Med Mol Imaging. 2017;44:81–91. doi: 10.1007/s00259-016-3481-7 [DOI] [PubMed] [Google Scholar]
- 80.Hofman MS, Violet J, Hicks RJ, et al. [(177)Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): a single-centre, single-arm, phase 2 study. Lancet Oncol. 2018;19:825–833. doi: 10.1016/S1470-2045(18)30198-0 [DOI] [PubMed] [Google Scholar]
- 81.Kim YJ, Kim YI. Therapeutic responses and survival effects of 177Lu-PSMA-617 radioligand therapy in metastatic castrate-resistant prostate cancer: a meta-analysis. Clin Nucl Med. 2018;43:728–734. doi: 10.1097/RLU.0000000000002210 [DOI] [PubMed] [Google Scholar]
- 82.von Eyben FE, Roviello G, Kiljunen T, et al. Third-line treatment and (177)Lu-PSMA radioligand therapy of metastatic castration-resistant prostate cancer: a systematic review. Eur J Nucl Med Mol Imaging. 2018;45:496–508. doi: 10.1007/s00259-017-3895-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Hofman MS, Emmett L, Violet J, et al. TheraP: a randomized phase 2 trial of (177) Lu-PSMA-617 theranostic treatment vs cabazitaxel in progressive metastatic castration-resistant prostate cancer (Clinical Trial Protocol ANZUP 1603). BJU International. 2019;124 Suppl 1:5–13. doi: 10.1111/bju.14876 [DOI] [PubMed] [Google Scholar]
- 84.Thang SP, Violet J, Sandhu S, et al. Poor outcomes for patients with metastatic castration-resistant prostate cancer with low prostate-specific membrane antigen (PSMA) expression deemed ineligible for (177)Lu-labelled PSMA radioligand therapy. Europ Urol Oncol. 2019;2:670–676. doi: 10.1016/j.euo.2018.11.007 [DOI] [PubMed] [Google Scholar]
- 85.Marinova M, Alamdar R, Ahmadzadehfar H, et al. Improving quality of life in patients with metastatic prostate cancer following one cycle of 177Lu-PSMA-617 radioligand therapy: a pilot study. Nuklearmedizin Nucl Med. 2020;59:409–414. doi: 10.1055/a-1234-5891 [DOI] [PubMed] [Google Scholar]
- 86.von Eyben FE, Bauman G, von Eyben R, et al. Optimizing PSMA radioligand therapy for patients with metastatic castration-resistant prostate cancer. a systematic review and meta-analysis. Int J Mol Sci. 2020;21:9054. doi: 10.3390/ijms21239054 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Crumbaker M, Pathmanandavel S, Yam AO, et al. Phase I/II Trial of the Combination of (177)Lutetium Prostate specific Membrane Antigen 617 and Idronoxil (NOX66) in men with end-stage metastatic castration-resistant prostate cancer (LuPIN). Europ Urol Oncol. 2021;4:963–970. doi: 10.1016/j.euo.2020.07.002 [DOI] [PubMed] [Google Scholar]
- 88.Sadaghiani MS, Sheikhbahaei S, Werner RA, et al. A systematic review and meta-analysis of the effectiveness and toxicities of lutetium-177-labeled prostate-specific membrane antigen-targeted radioligand therapy in metastatic castration-resistant prostate cancer. Europ Urol. 2021;80:82–94. doi: 10.1016/j.eururo.2021.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sartor O, de Bono J, Chi KN, et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. New Engl J Med. 2021;385:1091–1103. doi: 10.1056/NEJMoa2107322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Satapathy S, Sood A, Das CK, Mittal BR. Evolving role of (225)Ac-PSMA radioligand therapy in metastatic castration-resistant prostate cancer-a systematic review and meta-analysis. Prost Can Prost Dis. 2021;24:880–890. doi: 10.1038/s41391-021-00349-w [DOI] [PubMed] [Google Scholar]
- 91.Ballal S, Yadav MP, Sahoo RK, Tripathi M, Dwivedi SN, Bal C. (225) Ac-PSMA-617-targeted alpha therapy for the treatment of metastatic castration-resistant prostate cancer: a systematic review and meta-analysis. Prostate. 2021;81:580–591. doi: 10.1002/pros.24137 [DOI] [PubMed] [Google Scholar]
- 92.Zhang H, Xin P, Zhang YX. Efficacy and safety of the 177Lu-PSMA-617 therapy in the treatment of metastatic castration-resistant prostate cancer: a meta-analysis. Zhonghua Nan Ke Xue. 2021;27:63–69. [PubMed] [Google Scholar]
- 93.Lee DY, Kim YI. Effects of (225)Ac-labeled prostate-specific membrane antigen radioligand therapy in metastatic castration-resistant prostate cancer: a meta-analysis. J Nucl Med. 2022;63:840–846. doi: 10.2967/jnumed.121.262017 [DOI] [PubMed] [Google Scholar]
- 94.Pathmanandavel S, Crumbaker M, Yam AO, et al. (177)Lu-PSMA-617 and idronoxil in men with end-stage metastatic castration-resistant prostate cancer (LuPIN): patient outcomes and predictors of treatment response in a phase I/II trial. J Nucl Med. 2022;63:560–566. doi: 10.2967/jnumed.121.262552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Sadaghiani MS, Sheikhbahaei S, Werner RA, et al. (177) Lu-PSMA radioligand therapy effectiveness in metastatic castration-resistant prostate cancer: an updated systematic review and meta-analysis. The Prostate. 2022;82:826–835. doi: 10.1002/pros.24325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Satapathy S, Mittal BR, Sood A, et al. (177)Lu-PSMA-617 versus docetaxel in chemotherapy-naïve metastatic castration-resistant prostate cancer: a randomized, controlled, phase 2 non-inferiority trial. Eur J Nucl Med Mol Imaging. 2022;49:1754–1764. doi: 10.1007/s00259-021-05618-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Sonpavde G, McMannis JD, Bai Y, et al. Phase I trial of antigen-targeted autologous dendritic cell-based vaccine with in vivo activation of inducible CD40 for advanced prostate cancer. Can Immunol. 2017;66:1345–1357. doi: 10.1007/s00262-017-2027-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Mohammadzadeh M, Shirmohammadi M, Ghojazadeh M, Nikniaz L, Raeisi M, Aghdas SAM. Dendritic cells pulsed with prostate-specific membrane antigen in metastatic castration-resistant prostate cancer patients: a systematic review and meta-analysis. Prost Internat. 2018;6:119–125. doi: 10.1016/j.prnil.2018.04.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Montagner IM, Penna A, Fracasso G, et al. Anti-PSMA CAR-engineered NK-92 Cells: an Off-The-shelf Cell Therapy for Prostate Cancer. Cells. 2020;9:1382. doi: 10.3390/cells9061382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Weimin S, Abula A, Qianghong D, Wenguang W. Chimeric cytokine receptor enhancing PSMA-CAR-T cell-mediated prostate cancer regression. Cancer Biology & Therapy. 2020;21:570–580. doi: 10.1080/15384047.2020.1739952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wang D, Shao Y, Zhang X, Lu G, Liu B. IL-23 and PSMA-targeted duo-CAR T cells in Prostate Cancer Eradication in a preclinical model. J Translat Med. 2020;18:23. doi: 10.1186/s12967-019-02206-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Alzubi J, Dettmer-Monaco V, Kuehle J, et al. PSMA-Directed CAR T cells combined with low-dose docetaxel treatment induce tumor regression in a prostate cancer xenograft model. Molec Therap Oncolyt. 2020;18:226–235. doi: 10.1016/j.omto.2020.06.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Narayan V, Barber-Rotenberg JS, Jung IY, et al. PSMA-targeting TGFβ-insensitive armored CAR T cells in metastatic castration-resistant prostate cancer: a phase 1 trial. Natu Med. 2022;28:724–734. doi: 10.1038/s41591-022-01726-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zhang X, Sun S, Miao Y, et al. Docetaxel enhances the therapeutic efficacy of PSMA-specific CAR-T cells against prostate cancer models by suppressing MDSCs. J Cancer Res Clin Oncol. 2022;148:3511–3520. doi: 10.1007/s00432-022-04248-y [DOI] [PubMed] [Google Scholar]
- 105.Autio KA, Dreicer R, Anderson J, et al. Safety and Efficacy of BIND-014, a docetaxel nanoparticle targeting prostate-specific membrane antigen for patients with metastatic castration-resistant prostate cancer: a phase 2 clinical trial. JAMA Oncology. 2018;4:1344–1351. doi: 10.1001/jamaoncol.2018.2168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Petrylak DP, Kantoff P, Vogelzang NJ, et al. Phase 1 study of PSMA ADC, an antibody-drug conjugate targeting prostate-specific membrane antigen, in chemotherapy-refractory prostate cancer. Prostate. 2019;79:604–613. doi: 10.1002/pros.23765 [DOI] [PubMed] [Google Scholar]
- 107.Petrylak DP, Vogelzang NJ, Chatta K, et al. PSMA ADC monotherapy in patients with progressive metastatic castration-resistant prostate cancer following Abiraterone and/or enzalutamide: efficacy and safety in open-label single-arm phase 2 study. Prostate. 2020;80:99–108. doi: 10.1002/pros.23922 [DOI] [PubMed] [Google Scholar]
- 108.Milowsky MI, Galsky MD, Morris MJ, et al. Phase 1/2 multiple ascending dose trial of the prostate-specific membrane antigen-targeted antibody drug conjugate MLN2704 in metastatic castration-resistant prostate cancer. Urolog Oncol. 2016;34:530.e515–530.e521. doi: 10.1016/j.urolonc.2016.07.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Ghose A, Lapitan P, Apte V, et al. Antibody drug conjugates in urological cancers: a review of the current landscape. Curr Oncol Rep. 2024;26:633–646. doi: 10.1007/s11912-024-01524-7 [DOI] [PubMed] [Google Scholar]
- 110.Mjaess G, Aoun F, Rassy E, Diamand R, Albisinni S, Roumeguère T. Antibody-drug conjugates in prostate cancer: where are we? Clin Genitour Can. 2023;21:171–174. doi: 10.1016/j.clgc.2022.07.009 [DOI] [PubMed] [Google Scholar]
- 111.Von Hoff DD, Mita MM, Ramanathan RK, et al. Phase I Study of PSMA-targeted docetaxel-containing nanoparticle BIND-014 in patients with advanced solid tumors. Clin Can Res. 2016;22:3157–3163. doi: 10.1158/1078-0432.CCR-15-2548 [DOI] [PubMed] [Google Scholar]
- 112.Chi KN, Armstrong AJ, Krause BJ, et al. Safety Analyses of the Phase 3 VISION Trial of [(177)Lu]Lu-PSMA-617 in patients with metastatic castration-resistant prostate cancer. Europ urol. 2024;85:382–391. doi: 10.1016/j.eururo.2023.12.004 [DOI] [PubMed] [Google Scholar]
- 113.Hofman MS, Emmett L, Sandhu S, et al. [(177)Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet. 2021;397:797–804. doi: 10.1016/S0140-6736(21)00237-3 [DOI] [PubMed] [Google Scholar]
- 114.Aggarwal R, Starzinski S, de Kouchkovsky I, et al. Single-dose (177)Lu-PSMA-617 followed by maintenance pembrolizumab in patients with metastatic castration-resistant prostate cancer: an open-label, dose-expansion, phase 1 trial. Lancet Oncol. 2023;24:1266–1276. doi: 10.1016/S1470-2045(23)00451-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Eapen RS, Buteau JP, Jackson P, et al. Administering [(177)Lu]Lu-PSMA-617 Prior to Radical Prostatectomy in Men with High-risk Localised Prostate Cancer (LuTectomy): a Single-centre, Single-arm, Phase 1/2 Study. Europ urol. 2024;85:217–226. doi: 10.1016/j.eururo.2023.08.026 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding authors on reasonable request.