Abstract
Background
Dissecting intracranial pseudoaneurysms represent a challenge for treatment both endovascularly and surgically.
Methods
We review the treatment course of a ruptured anterior inferior cerebellar artery (AICA) dissecting pseudoaneurysm in a 50-year-old patient and review the history of dissecting aneurysms and AICA aneurysms treatment.
Results
An aneurysm cure was achieved using flow diversion in the AICA. The recent introduction of flow diversion devices that can be deployed through 0.017” microcatheters represents a new avenue for treatment of aneurysms.
Conclusion
To the best of our knowledge this is the first published case of flow diversion in the(?) AICA to treat a dissecting aneurysm. The introduction of Silk Vista Baby and similar future devices is likely to widen the scope of aneurysm treatment utilizing flow diversion in distal distribution with small parent arteries.
Keywords: Anterior inferior cerebellar artery, aneurysm, dissection, flow diversion, Silk Vista Baby
Introduction
Dissecting intracranial aneurysms are characterized by irregular appearance, rapid growth, re-rupture, and tapered parent artery. They represent a challenge to treat through endovascular or neurosurgical avenues.1,2 Treatment may require parent vessel sacrifice, which may lead to significant neurologic deficit. 3
Flow Diverters have been used for the treatment of dissecting aneurysms in several case. 4 Flow diverters function as reconstructive devices which make them ideal for treatment of dissecting aneurysms while maintaining parent vessel patency and preserving side branches. 5
Anterior inferior cerebellar artery (AICA) aneurysms are rare accounting for less than 1.5% of all intracranial aneurysms. 6 Endovascular therapies have been reported for the treatment AICA aneurysms with the most common procedure being parent artery occlusion.7–9 Primary coil embolization and stent assisted coil embolization have been reported and appear to provide feasible and safe option with technical success rate of 78%, however, these aneurysms were not dissecting in nature.10,11
Case report
Here we describe the complex treatment course of a ruptured dissecting aneurysm of the lateral pontine segment of the right AICA. This 50-year-old patient presented with thunderclap headache, tinnitus, and deafness, followed by syncope.
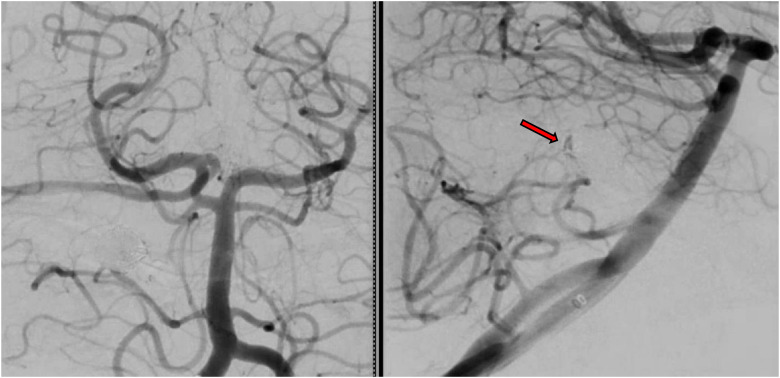
Patient was found to have subarachnoid hemorrhage in the posterior. Digital subtraction angiography (DSA) documented a 7.4 mm aneurysm of the right AICA (Figure 1). Selective microcatheter injection documented the aneurysm to involve the lateral pontine segment of an irregular and tapered parent vessel, consistent with underlying dissection (Figure 2).
Figure 1.
Digital subtraction angiography of the right vertebral artery documenting the irregular right AICA aneurysm (arrow). (Left in anterior posterior view, right is later view).
Figure 2.
Selective microcatheter injection of the right AICA documenting the dissecting aneurysm involving the proximal lateral pontine segment of the AICA (Arrow left, aneurysm and segment are demarcated). Notice that this is immediately distal to the bifurcation of the AICA into the medial anterior pontine segments.
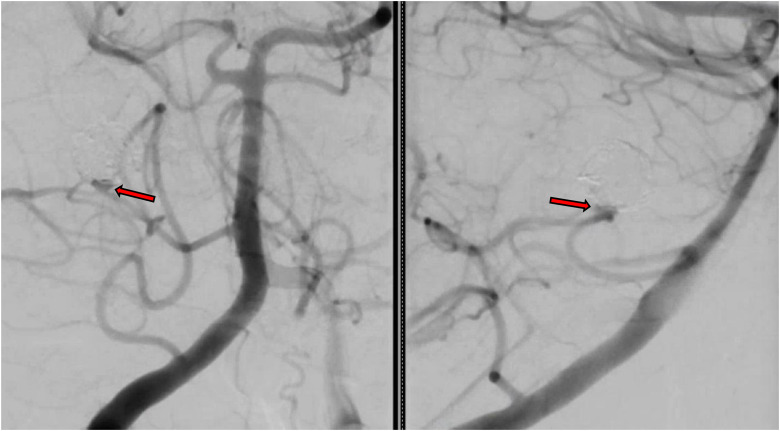
Initial primary endovascular coiling of the aneurysm was successful and preserved the patency of the AICA with very slow flow into the posterior pole of the aneurysm (Figure 3). More coiling was not possible at this point. Due to the dissecting etiology, DSA was repeated within one week. This documented resolution of the filling in the posterior pole, however, there appeared to be a small new filling at the neck (Figure 4).
Figure 3.
Post primary coiling documenting minor filling of the posterior lobe of the aneurysm. This is seen on lateral views on right (Arrow). (Left: anterior posterior view, right: later view).
Figure 4.
One week follow-up DSA documenting minor aneurysm growth at the neck (Arrow). (Left: anterior posterior view, right: later view).
The patient was discharged with a modified Rankin Scale of 1, at 17 days with right hearing loss and dizziness.
At six weeks, the patient's headaches increased in severity and frequency. Brain MRI suggested increasing aneurysm size and filling. DSA confirmed significant interval growth of the aneurysm with migration of the coil mass (Figure 5).
Figure 5.
Six-week follow up DSA with large growth of the aneurysm (Left: anterior posterior view, right: later view).
Treatment options
Multidisciplinary discussions were carried out in the neuroscience department to consider treatment options. Vessel sacrifice was considered, however, there was a concern for lateral pontine stroke.
Primary clipping of dissecting aneurysms is generally considered unfavorable. Surgical vessel sacrifice and bypass surgery was believed to be of high risk and likely to fail, given the small size of the parent vessel.
While stent coiling has been described in this location, it was not believed to have added benefit given the dissecting nature. Also, the aneurysm had a well-demarcated neck.12
Flow diversion has been used with good success for dissecting aneurysms.7 However, to the best of our knowledge this has not been attempted in the AICA. The size of the AICA presents a significant challenge as the flow divertors available in the USA at the time of the writing of this paper included devices that can only go through an 021″ or 027″ inner diameter (ID) microcatheters, both of which are too large to safely fit in this AICA. Silk vista baby (Balt Montmorency, France), a CE market approved device, was previously described in Posterior Inferior Cerebellar Artery (PICA) aneurysms with success. 12
All options, including our recommendation for flow diversion, were discussed with the patient who consented to the procedure.
Procedure
After obtaining emergency compassionate use authorization from the Food and Drug Administration (FDA), we proceeded with aneurysm embolization. The patient was on daily oral Aspirin 81 mg. Intravenous Cangrelor was initiated before deploying the device using a protocol described by Aguilar-Salinas et al. 13
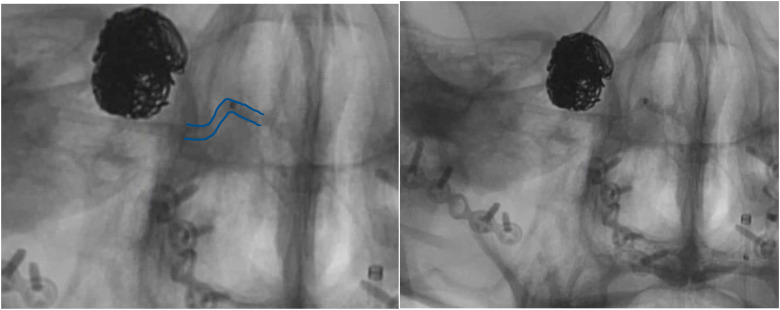
The patient was heparinized to an adequate Activated Clotting Time (ACT) level. We started by fully coiling the aneurysm before jailing by the flow diverter, as we had no guarantee that it would be successful. Coil embolization was performed with a Headway 17 microcatheter (Microvention, CA, USA). After coiling, the catheter was redirected over an 014″ microwire to the flocculopeduncular segment of the AICA, distal to the dissected vessel segment. A Silk Vista Baby 2.25 × 10 mm device (Balt, Montmorency, France) was deployed across the aneurysm neck (Figure 6).
Figure 6.
Left: Positioning of Silk Vista Baby before deployment, notice new coil mass in the inferior pole of the aneurysm. Right: Native image documenting deployment of the device in the AICA covering the bifurcation of the AICA.
After deployment of the flow divertor device, complete occlusion of the vessel was noted (Figure 7). Our initial interpretation of this occlusion was either thrombosis or vasospasm of the vessel. Injections of intra-arterial Eptifibatide followed by intra-arterial Verapamil were not successful in recanalizing the vessel. After careful analysis of the images, we realized that the occlusion was caused by a severe kink immediately proximal to the device, which changed the trajectory of the AICA – a loose artery in the cerebrospinal fluid (CSF) (Figure 8). We re-accessed the device and deployed a second, partially overlapping Silk Vista Baby device to straighten the vessel and restore flow (Figure 9). This successfully achieved regaining flow in the AICA (Figure 10 and Video1). Small distal emboli in the AICA territory were noted and successfully treated with intra-arterial Eptifibatide administration (2 mg). The patient tolerated the procedure well, without new neurologic deficits except for transient subjective horizontal diplopia that resolved after 24 h. She was transitioned to oral Ticagrelor 90 mg BID along with daily ASA 81 mg.
Figure 7.
Post deployment of the Silk Vista Baby documenting occlusion of the AICA proximal to device.
Figure 8.
Native image documenting the kink in the right AICA post deployment of the device.
Figure 9.
Placement of second device to correct the occlusive kink.
Figure 10.
Post treatment in AP (Left) and lateral (Right) views. Notice the course of AICA being straight at this point due to stents adjusting the trajectory of the vessel. No residual aneurysm at this time.
A 3-week follow up DSA documented continued growth of the aneurysm (Figures 11 and 12). The appearance of the lateral pontine segment at the neck of the aneurysm was lost at this point. The branch had transformed into a dissecting aneurysm. We opted to observe this and repeated angiogram at a 5-week interval, demonstrating further enlargement (Figures 13 and 14). The patient's symptoms continued to worsen with increasing headaches, paresthesia of the tongue. Treatment options were re-visited, followed by discussions with the patient.
Figure 11.
Three weeks post Silk Vista Baby device deployment documenting regrowth of the aneurysm in 2D views.
Figure 12.
Three weeks follow up angiogram 3D reconstruction images showing the lateral pontine segment to have become a dissecting aneurysm without a discrete branch.
Figure 13.
Five weeks post Silk Vista baby device deployment showing the aneurysm residual continuing to grow. Also, the total diameter of the aneurysm has increased from its initial 7.4 mm to 12 mm.
Figure 14.
The growth of the aneurysm between week 3 (right) and 5 (left) post Silk Vista Baby device deployment.
Given the previously discussed risks of an open surgical approach, the patient agreed to switch to monotherapy with aspirin only. Simultaneously, an FDA application for compassionate use of a second device was submitted. The plan was to deploy it inside the previous construct to enhance the flow diversion.
During the 6 weeks waiting for device approval and delivery, the patient's symptoms improved. DSA with intention of intervention, failed to show aneurysm filling. However, when paying close attention to the coil mass, we noticed that it was not in the vicinity of the flow diverters; a sign sometimes appreciated in dissecting aneurysms with partial thrombosis that can be deceiving without transaxial imaging (Figure 15). Nevertheless, an MRI and MRA of the head showed no evidence of aneurysm filling.
Figure 15.
Twelve weeks post deployment of the Silk Vista Baby. While there is no filling of the aneurysm visualized, the coil mass is clearly not adjacent to the stent.
At 6 months interval from the last DSA, while the patient was maintained on Aspirin 81 mg daily, the patient presented with worsening paresthesia of the tongue. An MRI and MRA documented aneurysmal recurrence (Figure 16). DSA, including selective injection of the AICA, demonstrated slow filling of the aneurysm through the superior lateral pontine segment (Figure 17).
Figure 16.
Axial MP Rage T1 post gadolinium MRI brain in the posterior fossa. (A) shows patency of the stent (White arrow). (B) Partially shows patency of the stent (White arrow) and sign of aneurysm recurrence (Grey arrow). (C and D) showing aneurysm regrowth and compression of the pons with the mass effect from the aneurysm (Grey arrow).
Figure 17.
Selective injection of the right AICA showing slow flow into the aneurysm through a possible side branch; the lateral pontine segment. (Left: Anteroposterior, right: lateral).
Balloon test occlusion of the AICA was performed using a 6 mm × 7 mm Eclipse balloon (Balt, Montmorency, France). This failed to document sufficient pial collaterals to support parent artery sacrifice (Figure 18).
Figure 18.
Balloon test occlusion of the right AICA utilizing a 6 × 7 mm Eclipse balloon demonstrating poor leptomeningeal collaterals from SCA and PICA into the distal distribution of the AICA from vertebral injection.
Given the slow flow of the aneurysm we decided after discussion with patient to discontinue the Aspirin to enhance thrombosis and monitor the progression very closely. After two weeks of stopping the Aspirin the patient's symptoms resolved.
Repeated DSA two months later documented the aneurysm to have regressed without any residual filling appreciated, and with maintained patency of the stent construct (Figures 19 and 20). A repeat MRI brain with gadolinium 18 months from initial ictus confirms persistent occlusion of the aneurysm with persistent device patency (Figure 21). The patient continues to be symptom-free with improving hearing impairment.
Figure 19.
Final angiogram documenting no evidence of aneurysm filling. Notice that the coil mass is adjacent to the stent in this stage.
Figure 20.
Migration of coil mass to abut the stent over the course of treatment signifying resolution of the aneurysm. Left when associated with aneurysm filling. Right when associated with no filling of the aneurysm. (Green: Silk vista baby. Red: Coil mass).
Figure 21.
18 months from ictus MRI documenting patency of the stent (White) and resolution of the aneurysm (Black).
Discussion
Dissecting brain aneurysms are challenging to treat both endovascularly and via open neurosurgery due to the circumferentially diseased wall of the parent artery.
Dissecting brain aneurysms result from interruption of the cohesiveness of the arterial wall layers leading to wall friability and the formation of pseudoaneurysms. Due to the underlying pathophysiology, they are typically associated with significantly higher rate of morbidity and mortality. 1,2 They are more frequent in young and middle-aged men, with a predilection for the posterior circulation, and especially the vertebral arteries. Appearance is usually fusiform, with sidewall outpouching being less frequently observed. 14
In general, there is increasing evidence supporting the treatment of intracranial dissecting aneurysms, in different distributions, utilizing reconstructive flow diversion with good success rate and reasonable risks. 4
Anterior inferior cerebellar aneurysms in general are exceptionally rare, amounting for less than 1–1.5% of all brain aneurysms. 15
Neurosurgical treatment of anterior inferior cerebellar aneurysms has been reported, including via resection and bypass as shown by Huang et al. 15
Endovascular treatment with stent assisted coiling has also been reported. 16 However, per our literature review, there is no significant data pertaining to dissecting AICA aneurysms.
In general, there is increasing evidence supporting the reconstructive flow diversion treatment of intracranial dissecting aneurysms in different distributions. 4
The size of the AICA is a challenge as the flow divertors available in the USA at the time of the writing of this paper included devices that can only go through an 0.021″ or 0.027″ inner diameter (ID) microcatheters, both of which are too large to safely fit in the AICA. Silk vista baby (Balt Montmorency, France), a CE market approved device was described in compassionate use to be used previously in Posterior Inferior Cerebellar Arteries (PICAs) with success in a case series of 15 cases.17,18 Our case report suggests that this is possibly reproducible in the AICA, even though it typically has a lower caliber.
Here, we presented a rare case of ruptured dissecting anterior inferior cerebellar artery pseudoaneurysm. The failure of the initial, angiographically satisfactory coiling, with aneurysm recanalization and growth was the expected course based on our understanding of this disease process. We treated this aneurysm via compassionate use of Silk Vista Baby, as the only available flow divertor deployable through an 0.017″ microcatheter at the time of treatment. From our review of literature to the best of our knowledge this is the first treatment of its kind in this location.
The ability to deploy this flow divertor through an 0.017″ catheter makes this technology feasible to apply in smaller caliber and more distal arteries. This application is associated with additional, and at times unpredictable potential complications. As our case demonstrates, distal and smaller calibers arteries, such as the AICA, are free floating in the CSF and their vector can remarkably change with the deployment of intraluminal devices. This may lead to “pinching” the vessel just proximal or distal to the device. Using longer devices that can extend close to the origin of the artery, and avoiding deployment ending at a curvature, maybe helpful in avoiding this complication.
Our case also demonstrates the limitations in the effectiveness of flow diversion. The mismatch between the device and the arterial diameter gives rise to the known phenomenon of stretching of the braided structure of the device, leading to larger opening of its cells. This leads to decreased flow diversion effect. 18 We speculate that this may have at least partially contributed to the complicated course.
As established in animal models, endothelization of the flow diverter device, and ultimately the arterial defect, is necessary for the isolation of the aneurysm from the circulation. It seemed that the early interruption of the blood thinners helped enhance the thrombosis of the aneurysm and facilitated the process of endothelialization, suggesting that in certain dissecting aneurysms faster interruption of the antiplatelets maybe beneficial to avoid recanalization and expansion of the aneurysm. This strategy is a double-edged sword, as it may lead to a clinically significant thrombotic event. Currently, our understanding is very limited in this field. Therefore, a case-by-case thoughtful approach is necessary when treating complex dissecting intracranial aneurysms, especially in smaller vessels. Larger studies may help understand the role of this device in the distal smaller caliber circulation.
Supplemental Material
Video 1.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors received no financial support for the research, authorship, and/or publication of this article.
ORCID iD: Omar Kass-Hout https://orcid.org/0000-0001-9738-6824
Supplemental material: Supplemental material for this article is available online.
References
- 1.Jin SC, Kwon DH, Choi CG, et al. Endovascular strategies for vertebrobasilar dissecting aneurysms. AJNR Am J Neuroradiol 2009; 30: 1518–1523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dmytriw AA, Alrashed A, Enriquez-Marulanda A, et al. Unruptured intradural posterior circulation dissecting/fusiform aneurysms natural history and treatment outcome. Interv Neuroradiol 2023; 29: 56–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ohkuma H, Suzuki S, Ogane K. Dissecting aneurysms of intracranial carotid circulation. Stroke 2002; 33: 941–947. [DOI] [PubMed] [Google Scholar]
- 4.de Barros Faria M, Castro RN, Lundquist J, et al. The role of the pipeline embolization device for the treatment of dissecting intracranial aneurysms. AJNR Am J Neuroradiol 2011; 32: 2192–2195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kan P, Sweid A, Srivatsan Aet al. et al. Expanding indications for flow diverters: ruptured aneurysms, blister aneurysms, and dissecting aneurysms. Neurosurgery 2020; 86: S96–s103. [DOI] [PubMed] [Google Scholar]
- 6.Drake CG. The treatment of aneurysms of the posterior circulation. Clin Neurosurg 1979; 26: 96–144. [DOI] [PubMed] [Google Scholar]
- 7.Suzuki K, Meguro K, Wada M, et al. Embolization of a ruptured aneurysm of the distal anterior inferior cerebellar artery: case report and review of the literature. Surg Neurol 1999; 51: 509–512. [DOI] [PubMed] [Google Scholar]
- 8.Mitsos AP, Corkill RA, Lalloo S, et al. Idiopathic aneurysms of distal cerebellar arteries: endovascular treatment after rupture. Neuroradiology 2008; 50: 161–170. [DOI] [PubMed] [Google Scholar]
- 9.Peluso JP, van Rooij WJ, Sluzewski Met al. et al. Distal aneurysms of cerebellar arteries: incidence, clinical presentation, and outcome of endovascular parent vessel occlusion. AJNR Am J Neuroradiol 2007; 28: 1573–1578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Suh SH, Kim DJ, Kim DI, et al. Management of anterior inferior cerebellar artery aneurysms: endovascular treatment and clinical outcome. AJNR Am J Neuroradiol 2011; 32: 159–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hou K, Xu K, Yu J. Endovascular treatment of anterior inferior cerebellar artery trunk aneurysms. Interv Neuroradiol 2022; 28: 604–612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Benalia VHC, Cortez GM, Brasiliense LBC, et al. Silk vista baby for the treatment of Complex posterior Inferior cerebellar artery aneurysms. Neurosurgery 2022; 91: 547–554. [DOI] [PubMed] [Google Scholar]
- 13.Aguilar-Salinas P, Agnoletto GJ, Brasiliense LBC, et al. Safety and efficacy of cangrelor in acute stenting for the treatment of cerebrovascular pathology: preliminary experience in a single-center pilot study. J Neurointerv Surg 2019; 11: 347–351. [DOI] [PubMed] [Google Scholar]
- 14.Yamaura A, Watanabe Y, Saeki N. Dissecting aneurysms of the intracranial vertebral artery. J Neurosurg 1990; 72: 183–188. [DOI] [PubMed] [Google Scholar]
- 15.Huang C, Qin S, Huang Wet al. et al. Anterior Inferior cerebellar aneurysm treated by aneurysm resection and intracranial artery anastomosis in situ: a. Case Report and Literature Review. Front Surg 2021; 8: 669433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sabharwal PS, Saini J, Hanumanthapura Ramalingaiah A. Stent assisted coil embolization of wide-necked bilobed anterior inferior cerebellar artery aneurysm with incorporated artery arising from the dome: a technical note. J Neurointerv Surg 2013; 5: e35. [DOI] [PubMed] [Google Scholar]
- 17.Hanel RA, Cortez GM, Benalia VHC, et al. Patient outcomes after treatment of brain aneurysm in small diameter vessels with the silk vista baby flow diverter: a systematic review. Interv Neuroradiol 2022: 15910199221091645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bhogal P, Chudyk J, Bleise C, et al. The use of flow diverters to treat aneurysms of the posterior inferior cerebellar artery: report of three cases. Interv Neuroradiol 2018; 24: 489–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Video 1.