Abstract
Background
Sepsis is a global health problem with high morbidity and mortality. Low- and middle-income countries have a higher incidence and poorer outcome with sepsis. Large epidemiological studies in sepsis using Sepsis-3 criteria, addressing the process of care and deriving predictors of mortality are scarce in India.
Method
A multicentre, prospective sepsis registry was conducted using Sepsis 3 criteria of suspected or confirmed infection and SOFA score of 2 or more in 19 ICUs in India over a period of one year (August 2022–July 2023). All adult patients admitted to the Intensive Care Unit who fulfilled the Sepsis 3 criteria for sepsis and septic shock were included. Patient infected with Covid 19 were excluded. Patients demographics, severity, admission details, initial resuscitation, laboratory and microbiological data and clinical outcome were recorded. Performance improvement programs as recommended by the Surviving Sepsis guideline were noted from the participating centers. Patients were followed till discharge or death while in hospital.
Results
Registry Data of 1172 patients with sepsis (including 500 patients with septic shock) were analysed. The average age of the study cohort was 65 years, and 61% were male. The average APACHE II and SOFA score was 21 and 6.7 respectively. The majority of patients had community-acquired infections, and lung infections were the most common source. Of all culture positive results, 65% were gram negative organism. Carbapenem-resistance was identified in 50% of the gram negative blood culture isolates. The predominant gram negative organisms were Klebsiella spp (25%), Escherechia coli (24%) and Acinetobacter Spp (11%). Tropical infections (Dengue, Malaria, Typhus) constituted minority (n = 32, 2.2%) of sepsis patients. The observed hospital mortality for the entire cohort (n = 1172) was 36.3%, for those without shock (n = 672) it was 25.6% and for those with shock (n = 500) it was 50.8%. The average length of ICU and hospital stay for the study cohort was 8.64 and 11.9 respectively. In multivariate analysis adequate source control, correct choice of empiric antibiotic and the use of intravenous thiamine were protective.
Conclusion
The general demographics of the sepsis population in the Indian Sepsis Registry is comparable to Western population. The mortality of sepsis cohort was higher (36.3%) but septic shock mortality (50.8%) was comparable to Western reports. Gram negative infection was the predominant cause of sepsis with a high incidence of carbapenem resistance. Eschericia coli, Klebsiella Spp and Acinetobacter Spp were the predominant causative organism. Tropical infection constituted a minority of sepsis population with low hospital mortality. The SOFA score on admission was a comparatively better predictor of poor outcome. Sepsis secondary to nosocomial infections had the worst outcomes, while source control, correct empirical antibiotic selection, and intravenous thiamine were protective.
CTRI Registration CTRI:2022/07/044516.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13054-024-05176-8.
Keywords: Sepsis, Registry, Epidemiology, Intensive Care Unit, India
Background
Sepsis, a dysregulated host response to an infection, is a global health problem and an important cause of hospitalization and mortality in the ICU [1]. Reports from Global burden of disease study had estimated 48·9 million cases of sepsis and 110 million sepsis-related deaths worldwide, representing 19·7% of all global deaths [2]. The highest burden of sepsis incidence and mortality is from low- and middle-income countries (LMIC). Most of the epidemiological data on sepsis has been published from high-income countries, reporting lower mortality [3] Sepsis epidemiological data from India has been mostly retrospective, single-centre studies with small sample sizes using older definitions of sepsis, or a subset of large datasets from point prevalence studies [4, 5, 10, 19]. There have been a few multi-centre point prevalence studies using the current criteria of sepsis but many studies have focused mainly on the incidence of antibiotic resistance in Indian polulation [6]. We conducted a multicentre, prospective study on sepsis using a sepsis registry with a large sample size using Sepsis 3 criteria to define sepsis and septic shock admitted to the ICU. Predictors of adverse outcomes during hospital stays were derived from the observed data. We aimed to contribute to existing literature and identify clinical practice variability and areas of further research on the subject.
Method
The study is a research initiative of Sepsis Forum India (SFI—https://sepsisforum.in), which is a scientific organization created to improve awareness, promote educational activities, and research in sepsis. The scientific committee of SFI was responsible for conceptualisation, study design, site selection, and constructing the electronic case record (e-CRF.) form. Site selection was based on previous experience with sepsis research of the principal investigator (PI), who expressed interest in the project and the availability of adequate infrastructure. Nineteen sites were selected that satisfied the criteria for Level III ICU (for tertiary care hospitals with more than 150 beds including medical colleges and corporate hospitals) with all advanced ICU facilities including extracorporeal support as defined by the Indian Society of Critical Care Medicine (ISCCM) [7]. The Sepsis registry steering committee held multiple meetings with the PI and the data collection team prior to the commencement of the registry. Patient enrolment in the Sepsis registry was conducted over one-year period starting on 1st August 2022 until 31st July, 2023. All adult patients (aged > 18 years) admitted to ICU during the study period with suspected (subsequently confirmed) or confirmed infection as per the treating team, based on clinical and laboratory variables, were identified. SOFA scores were calculated within 24 h of screening and if a score of 2 or more (or 2 more than the baseline if known) was found, those patients were enrolled in the registry. Both newly admitted patients and those already in the ICU who fulfilled the above criteria were enrolled. Sepsis and septic shock were defined according to the Sepsis-3 definition [8]. Patients less than 18 years of age, those with no suspicion of infection, SOFA scores of less than two, and, those infected with SARS-COV-2 were excluded from the study. All data were collected prospectively and patients were followed until hospital discharge or death.
Data collection and entry into the registry were performed in a standardized manner, using an electronic case record form (Supplement 1). Systematic data collection was conducted under ten different sections for all patients entered into the registry. This dataset captured over one hundred different parameters related to patient demographics, initial resuscitation laboratory values, microbiological data, processes of sepsis care, and outcomes (Supplement 2).
The objective was to study the demographics of this population, determine the prognostic ability of severity scoring markers and mortality outcomes, analyse sepsis care processes such as initial resuscitation, clinical and laboratory parameters, antibiotic use, microbiology details, course in the hospital, and adjunctive sepsis treatment. This study was conducted in compliance with the principles of the Declaration of Helsinki and local regulatory requirements. Ethical approval was obtained from the individual Institutional Review Board or Ethics Committee of the participating centres. Informed consent was obtained if required by the ethical committee of centres, from study participants or their legal representatives before enrolment in the registry. The registry was also registered with the Trial Registry Database in India (CTRI) (CTRI:2022/07/044516). Registry data was collected, analysed and reported as per Registry reporting guideline [45].
Statistical analysis
Descriptive statistics were used to summarize the demographic and clinical characteristics of the study cohort. Continuous variables were reported as means with standard deviations or medians with interquartile ranges (IQR) based on their distribution. Categorical variables were presented as frequencies and percentages. For comparing parameters between those with or without the outcome of death, Chi-square test or Fisher’s exact test was used for categorical data comparisons, while t-tests or Mann–Whitney U tests were used for continuous data comparisons, as appropriate. We conducted a comprehensive univariate analysis of the dataset comprising 117 variables for predicting hospital mortality. Variables found to be significant were further explored through multivariate binary logistic regression analysis, with the aim of constructing predictive models that determine the binary outcome of interest. Goodness of fit model showed an accuracy rate of 89.4%. Moreover, the Hosmer and Lemeshow Test, with a p-value of 0.428, suggested that the model adequately fit the data.
Mortality predictions of severity scoring methods (SIRS, qSOFA and SOFA) were constructed using Receiver Operating Curve (ROC) curve testing. Discriminatory power was determined by comparing the area under the receiver operating curve (AUC ROC) for each of SIRS, qSOFA and SOFA scores individually (unadjusted analysis) with a 95% confidence interval (CI). Z statistic as described by Hanley and McNeil was used to compare the differences in the AUC of the scores in the sepsis patients. All statistical tests were 2-sided, and p < 0.05 was considered statistically significant.
The results from the logistic regression models and the ROC curve analysis were used to identify significant predictors of mortality and evaluate predictive accuracy in the context of specific patient subgroups. All statistical analyses were conducted using statistical software (e.g., SPSS version 26 and R.03, MedCalc ® Statistical Software version 22.021).
Results
Over a period of one year, data were prospectively collected from the 19 participating centres, identifying 1457 patients as having sepsis or septic shock as per the sepsis 3 criteria. The final cohort after data cleaning consisted of 1172 ICU patients, of which 672 (58%) patients had sepsis and 500 (42%) patients had septic shock.
The average age of the entire study cohort was 65 years, with 61% being male. The average APACHE II and SOFA scores were 21 and 6.7, respectively. The respective figures for patients with septic shock were 60 years, 60% male, APACHE II of 26, and SOFA of 8.3. The majority of patients were transferred from the emergency department and had community-acquired infections, which were the primary reasons for ICU admission. The average BMI was 20 and Charlson Comorbidity index was 3 for the entire study cohort. The lung was the most common source of infection (38%) followed by urosepsis (32%) (Table 1).
Table 1.
Demographic of the study cohort
| Variables | Group | Entire cohort N = 1172 | Septic shock cohort (n) = 500 | Sepsis (n) = 672 |
|---|---|---|---|---|
| Age (mean (SD)) | 62.12 (16.83) | 60.25 (17.84) | 63.50 (15.90) | |
| Gender (%) | Male | 719(61.3) | 306 ( 61.2) | 413 ( 61.5) |
| Female | 453(38.7) | 194 ( 38.8) | 259 ( 38.5) | |
| BMI (mean (SD)) | 22.16 (6.29) | 22.84 (6.43) | 21.66 (6.13) | |
| Charlson Comorbidity Index (mean (SD)) | 3.52 (2.46) | 3.50 (2.67) | 3.53 (2.29) | |
| SOFA score (mean (SD)) | 6.70 (3.64) | 8.30 (3.78) | 5.51 (3.02) | |
| APACHE score (mean (SD)) | 21.87 (12.45) | 26.22 (14.36) | 18.63 (9.62) | |
| ICU transferred from (%) | ICU transferred From another hospital (%) | 58(4.9) | 35 ( 7.0) | 23 ( 3.4) |
| ICU Transferred from Emergency (%) | 844(72) | 314 ( 62.8) | 530 ( 78.9) | |
| ICU Transferred From OT (%) | 31(2.6) | 17 ( 3.4) | 14 ( 2.1) | |
| ICU Transferred From WARD (%) | 166(14.2) | 92 ( 18.4) | 74 ( 11.0) | |
| Community acquired infection(%) | 960(81.9) | 384 (76.8) | 576 (85.7) | |
| Site of infection (%) | Abdomen | 12 ( 2.1) | 10 ( 3.7) | 2 ( 0.6) |
| Blood | 241 ( 41.6) | 127 (46.9) | 114 (36.9) | |
| Respiratory Tract | 139 ( 24.0) | 52 (19.2) | 87 (28.2) | |
| Urine | 145 ( 25.0) | 60 (22.1) | 85 (27.5) | |
| Wound | 30 ( 5.2) | 16 ( 5.9) | 14 ( 4.5) | |
| Others | 13 ( 2.2) | 6 ( 2.2) | 7 ( 2.3) |
Other site of infections: Central Nervous System, Heart, Bones/Joints, Female Reproductive System, Head/Neck
The observed hospital mortality rate of the study cohort (n = 1172), for sepsis without shock (n = 672), and for septic shock (n = 500), was 36.3%, 25.6% and 50.8% (p < 0.001) respectively. The predicted hospital mortality rate (PMR) as per APACHE II was 40%, 25% and 55%, and the standardised mortality ratio (SMR = Observed Mortality Rate /PMR) was 0.9,1.02 and 0.92 respectively (Table 2).
Table 2.
Hospital mortality and length of stay (LOS) of total study cohort, septic shock and sepsis without shock
| Total cohort N = 1172 | Septic shock = 500 | Sepsis = 672 | p value | |
|---|---|---|---|---|
| APACHEII score (mean (SD)) | 21.87 (12.45) | 26.22 (14.36) | 18.63 (9.62) | < 0.001 |
| Observed hospital mortality | 36.34%(n = 426) | 50.8%(n = 254) | 25.6 (172) | < 0.001 |
| Predicted hospital mortality (PMR) (as per mean APACHE II score) | 40% | 55% | 25% | |
| Standardised mortality ratio (SMR) | 0.9 | 0.92 | 1.024 | |
| Average ICU LOS (mean (SD)) | 8.64 (10.12) | 8.47 (8.56) | 8.76 (11.15) | 0.617 |
| Average hospital LOS (mean (SD)) | 11.99 (12.34) | 11.71 (11.42) | 12.19 (12.99) | 0.504 |
The mean length of stay in the ICU for the entire study cohort, sepsis and septic shock was 8.64, 8.76 and 8.47 days (p = 0.617), respectively. The corresponding figures for Hospital length of stay were 11.99, 12.19 and 11 0.71 days (p = 0.50), respectively (Table 2). Discharge against Medical Advice (DAMA) was noted in 14% of patients.
SOFA, qSOFA and SIRS were assessed on the day of sepsis diagnosis. SOFA score exhibited a significantly higher Area under the curve (AUC) of 0.768 as opposed to qSOFA and SIRS of 0.70 and 0.54 respectively.
An initial univariate analysis was conducted on the dataset comprising 99 variables. The demographic, clinical, and laboratory characteristics in the study cohorts are presented in Table 3, which lists the variables and performs a comparative analysis among these parameters between deceased and surviving subjects (Supplement 4).
Table 3.
Univariate analysis of study variables and its effect on hospital mortality
| Study variables | All (N = 1172) | Alive_at discharge (n = 746) | In Hospital death (n = 426) | p value | |
|---|---|---|---|---|---|
| SOFA score (mean (SD)) | 6.7(3.639) | 5.51 (3.13) | 8.78 (3.53) | < 0.001 | |
| Q SOFA score (mean (SD)) | 1.87 (0.904) | 1.63 (0.89) | 2.30 (0.75) | < 0.001 | |
| SIRS (mean (SD)) | 2.86 (0.931) | 2.80 (0.99) | 2.98 (0.81) | 0.001 | |
| APACHE II score (median [IQR]) | 21[14.00, 26.00] | 17.00 [12.00, 24.00] | 23.00 [19.00, 31.00] | < 0.001 | |
| Charlson comorbidity index (median [IQR]) | 3 [2.00, 5.00] | 3.00 [2.00, 5.00] | 4.00 [2.00, 5.00] | 0.004 | |
| Age (median [IQR]) | 65[52.75, 74.00] | 65.00 [53.00, 74.00] | 64.50 [52.25, 74.00] | 0.866 | |
| Gender (%) | 719(61.3) | Male | 434 (58.2) | 285 (66.9) | 0.003 |
| 453(38.7) | Female | 312 (41.8) | 141 (33.1) | ||
| BMI (median [IQR]) | 20.59 [17.81, 25.00] | 19.95 [17.30, 23.30] | 23.44 [19.12, 30.36] | < 0.001 | |
| Suspected origin of infection (%) | |||||
| 960(81.9) | Community acquired infection(%) | 637 (85.4) | 323 (75.8) | < 0.001 | |
| 165(14.1) | Hospital acquired infection (%) | 89 (11.9) | 76 (17.8) | 0.007 | |
| 47(4) | ICU acquired infection(%) | 20 (2.7) | 27 (6.3) | 0.003 | |
| Clinical details (On the day of sepsis diagnosis) | |||||
| Septic shock as per sepsis 3 criteria(%) | 500(42.6) | 246 (33.0) | 254 (59.6) | < 0.001 | |
| Clinical Feature of hypoperfusion (%) | 338(28.8) | 166 (22.3) | 172 (40.4) | < 0.001 | |
| Risk factor for antibiotic resistant infection infection (%) | 371(31.7) | 248 (33.2) | 123 (28.9) | 0.133 | |
| Organ support (%) | 64(5.5) | Dialysis (%) | 42 (5.6) | 22 (5.2) | 0.79 |
| 394(33.6) | Ventilation (%) | 165 (22.1) | 229 (53.8) | < 0.001 | |
| 71(6.1) | Ventilation and dialysis (%) | 29 (3.9) | 42 (9.9) | < 0.001 | |
| Laboratory variables (On the day of sepsis diagnosis) | |||||
| PaO2 (median [IQR]) (mmHg) | 212 [110.00, 320.25] | 263.95 [167.51, 350.00] | 130.85 [70.25, 231.48] | < 0.001 | |
| Arterial pH (median [IQR]) | 7.36 [7.26, 7.43] | 7.39 [7.30, 7.45] | 7.30 [7.20, 7.40] | < 0.001 | |
| Serum HCO3 (mmol/L) (median [IQR]) | 20.50 [16.00, 24.20] | 21.00 [17.10, 24.40] | 19.78 [15.03, 23.37] | 0.003 | |
| Platelets count (median [IQR])(µL) | 1,,60000 [100000.00, 240000.00] | 1,71374.86 [133000.00, 254000.00] | 1,35766.13 [70000.00, 203750.00] | < 0.001 | |
| Serum creatinine (median [IQR]) (mg/dl) | 1.60 [0.98, 2.80] | 1.50 [0.95, 2.66] | 1.80 [1.00, 2.93] | 0.052 | |
| Serum Albumin (median [IQR]) (g/dl) | 3.0 [2.50, 3.40] | 3.20 [2.80, 3.52] | 2.58 [2.20, 3.00] | < 0.001 | |
| Blood glucose (mg/dl) (median [IQR]) | 169 [130.00, 213.57] | 180.00 [134.00, 226.18] | 152.39 [126.00, 189.77] | < 0.001 | |
| Initial resucitation | |||||
| Fluid infused type (%) | 753 (64.2) | Normal saline (%) | 502 (67.3) | 251 (58.9) | 0.004 |
| 34 (2.9) | Ringers lactate (%) | 17 (2.3) | 17 (4.0) | 0.104 | |
| 264 (22.5) | Other balanced (%) | 145 (19.4) | 119 (27.9) | 0.001 | |
| Amount . of fluid infused in 1 h (ml) (median [IQR]) | 200 [83.00, 500.00] | 210.02 [100.00, 500.00] | 181.93 [60.00, 500.00] | 0.001 | |
| Amount . of fluid infused in 3 h (ml) (median [IQR]) | 387.59 ([200.00, 786.65] | 430.76 [240.00, 800.00] | 300.00 [150.00, 758.75] | 0.001 | |
| Albumin infusion within 3 h (%) | 233(19.9) | 56 (7.5) | 177 (41.5) | < 0.001 | |
| Lactate level 1 h value (mmol/l) (median [IQR]) | 2.30 [1.40, 3.90] | 2.10 [1.30, 3.35] | 2.76 [1.60, 4.72] | < 0.001 | |
| Antibiotic therapy | |||||
| Correct empiric choice (%) | 889 (75.9) | 583 (78.2) | 306 (71.8) | 0.016 | |
| Beta lactam given as extented infusion (%) | 773 (66) | 458 (61.4) | 315 (73.9) | < 0.001 | |
| Loading dose of antibiotic (%) | 1009 (86.1) | 656 (87.9) | 353 (82.9) | 0.018 | |
| Descalation of antibiotic (%) | 416 (35.5) | 266 (35.7) | 150 (35.2) | 0.899 | |
| Microbiology | |||||
| Blood culture positive (%) | 577 (49.2) | 349 (46.8) | 228 (53.5) | 0.029 | |
| Culture sensitivity (%) | 327 (27) | Carba Resistant Organism (%) | 194 (26.0) | 133 (31.2) | 0.058 |
| 240 (20) | Carba Sensitive Organism (%) | 149 (20.0) | 91 (21.4) | 0.599 | |
| Adjunctive therapy for sepsis | |||||
| Corticosteroids (%) | 474 (40.4) | 210 (28.2) | 264 (62.0) | < 0.001 | |
| Vitamin C (%) | 46 (3.9) | 30 (4.0) | 16 (3.8) | 0.877 | |
| Thiamine (%) | 382 (32.6) | 317 (42.5) | 65 (15.3) | < 0.001 | |
| Extracorporeal therapy (%) | 105 (9) | 26 (3.5) | 79 (18.5) | < 0.001 | |
| Bicarbonate therapy (%) | 260 (22.2) | 110 (14.7) | 150 (35.2) | < 0.001 | |
| Course in hospital | |||||
| NIV (%) | 286 (24.4) | 182 (24.4) | 104 (24.4) | 1 | |
| HFNO (%) | 275 (23.5) | 183 (24.5) | 92 (21.6) | 0.282 | |
| Invasive ventilation (%) | 563 (48) | 206 (27.6) | 357 (83.8) | < 0.001 | |
| Reintubation (%) | 41 (3.5) | 18 (9.6) | 23 (6.7) | 0.24 | |
| Prone (%) | 39 (3.3) | 16 (8.6) | 23 (6.7) | 0.486 | |
| Recruitment maneuver (%) | 67 (5.7) | 18 (9.9) | 49 (14.6) | 0.169 | |
| Renal support (%) | 357 (30.5) | 156 (20.9) | 201 (47.2) | < 0.001 | |
| Encephalopathy (%) | 369 (31.5) | 135 (18.1) | 234 (54.9) | < 0.001 | |
| Coagulopathy (%) | 169 (14.4) | 87 (11.7) | 82 (19.2) | 0.001 | |
| Liver dysfunction (%) | 173 (14.8) | 93 (12.5) | 80 (18.8) | 0.005 | |
| Nosocomial infection (%) | 385 (32.8) | 79 (10.6) | 306 (71.8) | < 0.001 | |
| Nutrition started early (%) | 980 (83.6) | 645 (86.5) | 335 (78.6) | 0.001 | |
| Blood transfusion (%) | 321 (27.4) | 162 (21.7) | 159 (37.3) | < 0.001 | |
| Platelet transfusion (%) | 102 (8.7) | 39 (5.2) | 63 (14.8) | < 0.001 | |
| FFP transfusion (%) | 114 (9.7) | 34 (4.6) | 80 (18.8) | < 0.001 | |
| DVT prophylaxis (%) | 711 (60.7) | 455 (61.0) | 256 (60.1) | 0.804 | |
| Stress ulcer prophylaxis (%) | 1108 (94.5) | 718 (96.2) | 390 (91.5) | 0.001 | |
| Continuous infusion sedation (%) | 308 (26.3) | 114 (15.3) | 194 (45.5) | < 0.001 | |
| Neuromuscular blocker infusion (%) | 97 (8.3) | 36 (4.8) | 61 (14.3) | < 0.001 | |
| Clinical outcome | |||||
| Clinical improvement (%) | 628 (53.6) | 610 (81.8) | 18 (4.2) | < 0.001 | |
| lCU Discharge status (%) | 758 (64.7) | Alive | 746 (100.0) | 12 (2.8) | < 0.001 |
| Dead | 0 (0.0) | 414 (97.2) | |||
| Hospital length of stay (median [IQR]) | 9 (12.34) | 10.00 [6.00, 14.00] | 8.00 [4.00, 15.00] | < 0.001 | |
| ICU length of stay (median [IQR]) | 6 (10.123) | 5.50 [4.00, 9.00] | 6.00 [3.00, 13.00] | 0.286 | |
| Unanticipated cardiac arrest (%) | 172 (14.7) | 10 (1.3) | 162 (38.0) | < 0.001 |
SOFA score: Sequential (or sepsis-related) organ failure assessment score. qSOFA score: quick sequential (or sepsis-related) organ failure assessment score. SIRS: Systemic inflammatory response syndrome. APACHE score: Acute physiology and chronic health evaluation score. BMI: Body mass index. NIV: Non-invasive ventilation. HFNO: High-flow nasal oxygen. DVT: deep vein thrombosis. FFP: fresh frozen plasma. TOF: Train-of-four (used in neuromuscular monitoring). TDM: Therapeutic drug monitoring
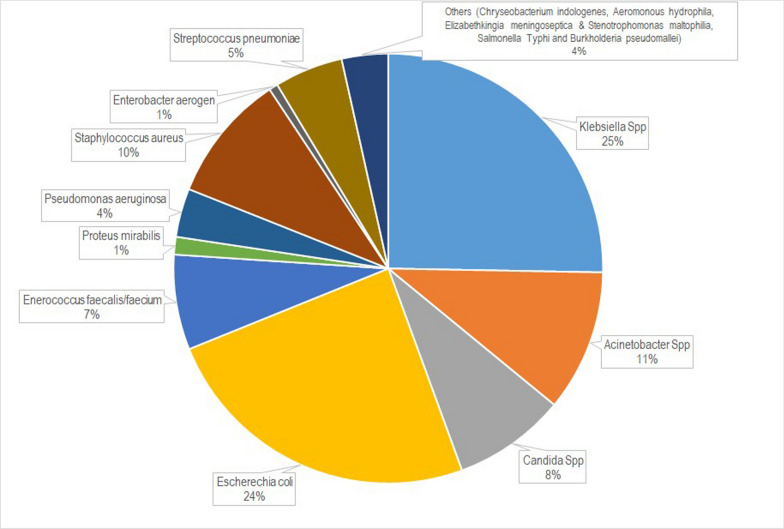
In univariate analyses, multiple demographic variables were associated with unfavorable outcomes. Significant among these were male gender, high BMI, high Charlson comorbidity index, transfer to ICU from the ward, and nosocomial infection. Certain clinical variables like septic shock, and clinical features of hypoperfusion were also associated with worse outcomes. Laboratory parameters such as low pH, low bicarbonate, low platelet count, high sodium, high creatinine, lower albumin and low glucose values were significantly associated with poor outcomes. During initial resuscitation, less fluid infused within 1 and 3 h, increased lactate, vasopressor use and albumin infusion were associated with poor outcomes. Normal saline was the most common resuscitation fluid used. Risk factors for drug-resistant bacterial infections were not associated with increased mortality. The majority of patients received empirical antibiotics. Factors significantly associated with better outcomes were source control, correct choice of empirical antibiotic and administration of a loading dose of antibiotic. Early enteral nutrition support, successful extubation, and stress ulcer prophylaxis were also associated with better outcomes whereas the use of extended infusion of beta-lactam antibiotics and de-escalation of antibiotics were not. Blood cultures were sent in the majority of patients and were positive in 50% of patients. Gram -negative organism constituted 80% of all positive blood cultures. The site of infection with microorganism spectra is described in Supplement 3. Major Gram-negatives were Klebsiella pneumoniae (25%), E. coli (25%), Acinetobacter spp (19%), and Pseudomonas aeruginosa (13.4%) (Fig. 1). Carbapenem-resistant organisms were detected in 57% of all positive blood cultures. Unadjusted hospital mortality in patients infected with carbapenem resistant organism was 31% as opposed to 21% in patients infected with carbapenem sensitive organism (Table 3). MRSA was detected in a minority of patients only. Empirical antifungal therapy was associated with worse outcomes. All patients received empiric antibiotic. Single antibiotic (16%) and dual antibiotics (83%) were used in this patient population. The empiric antibiotic choice was correct when compared to subsequent culture results in 62% of patients. Empiric antibiotic choices consisted of Meropenem (55%), Piperacillin Tazobactam (22%), Polymyxin B (11%), Colistin (4%), Ceftazidime Avibactam (5%), Minocycline (4%), Doxycycline (12%), Teicoplanin (24%) and Vancomycin (2%). Tropical infections (Dengue, Malaria, Typhus) with sepsis constituted 2.2% (n = 32) of the study population. Average age 45 years (SD 20), 65% Male (n = 21), SOFA 4.41 and APACHE II 8.75. Hospital mortality 3.13%, much lower than the general septic cohort and the average length of stay was 6 days. Renal support was needed in 30% of patients, with Slow extended Dialysis (SLEDD) in 38%, Continuous renal replacement therapy (CRRT) in 14% and the rest requiring Intermittent Hemodialysis (IHD). The reasons for starting renal support were low urine output (35%), rising creatinine (25%), worsening acidosis, (22%) volume overload (5%) and hyperkalaemi (2%). 60% of patients were successfully weaned off the renal support.
Fig. 1.
Microorganisms identified in positive blood cultures
The use of adjunctive therapies like corticosteroid, extracorporeal therapy, and bicarbonate infusion was associated with poor outcomes while the use of thiamine was protective (Table 3).
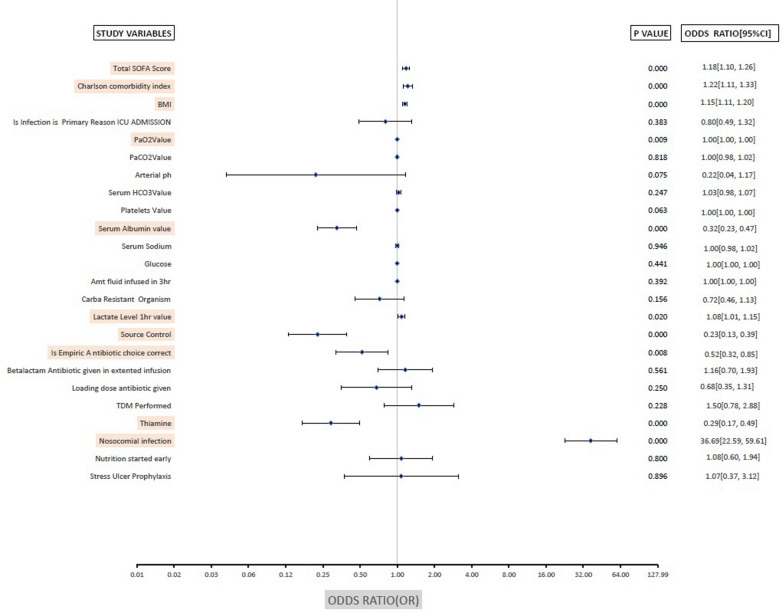
Significant variables identified in the univariate analysis were further subjected to multivariate analysis. Independent predictors of increased hospital mortality were high SOFA score, high BMI, High Charlson comorbidity index and nosocomial infection. Conversely, independent factors for improved survival included the correct empirical choice of antibiotic, source control, the use of intravenous thiamine, high albumin and high PaO2 (Table 4, Fig. 2).
Table 4.
Independent predictors of outcome
| Predictors | Odds ratio (OR) | Confidence interval (CI) |
|---|---|---|
| SOFA score | 1.216 | (1.112–1.331) |
| Charlson comorbidity index | 1.216 | (1.112–1.1331) |
| BMI | 1.153 | (1.108–1.199) |
| Nosocomial infection | 36.695 | (22.587–59.615) |
| PaO2 | 0.997 | (0.996–0.999) |
| Serum albumin | 0.324 | (0.225–0.467) |
| Source control | 0.227 | (0.132–0.389) |
| Correct empiric antibiotic | 0.519 | (0.171–0.495) |
| Intravenous Thiamine | 0.291 | (0.196–0.587) |
Fig. 2.
Identifying predictors of mortality in multivariable binary logistic regression
Performance improvement programs as recommended by the Surviving Sepsis guideline were noted in the participating centres [32]. A screening policy for high-risk sepsis patients was performed by 15 centres, the majority of whom conducted screening through manual chart reviews. Written SOP for sepsis management were maintained by 12 centres, a sepsis team was present in 10 centres, and periodic performance audits were done by 13 participating centres.
Discussion
This study is one of the largest multicentre prospective registry in India of sepsis and septic shock identified by Sepsis-3 criteria. The demographics of the study cohort regarding age, gender, severity scores, site of infection, and comorbidity index, are similar to other sepsis epidemiological studies conducted in India and globally including North America and Australia/Newzeland [9–14]. The predominant infections in our study were caused by Gram-negative organism, of which 62% were carbapenem resistant, similar to another study from India [10], but this was lower than the 80% resistance reported in other single centre Indian studies [15, 16]. Data from Western literature describes the incidence of Gram-negative sepsis at around 40%, with a much lower incidence of carbapenem-resistant Gram-negative organisms [17, 18].
Mortality from sepsis and septic shock varies widely in the literature, from 10 to 52%, depending on the source, timing of data collection, definitions used, and patients treatment related factors. In the original data set from which the Sepsis 3 definition was derived, unadjusted hospital mortality rates due to sepsis from two different hospital consortiums in USA (University of Pittsburgh Medical Center and Kaiser Permanente Northern California), were 20% and 8% respectively. Hospital mortality from septic shock (Hypotension requiring vasopressors and lactate more than 2 mmol/l) from the same two databases was 54% and 35%, respectively [8]. Unadjusted hospital mortality for sepsis patients was significantly higher in our study (36.34%), while septic shock mortality (55%) was similar to the University of Pittsburgh dataset. In a single-centre prospective observational study over a 5-year period from India that used, Sepsis-1 criteria hospital mortality of sepsis including septic shock was 63% among 282 patients [9]. In a recent multicentre observational study of ICU population in India, the INDICAP-II study, ICU mortality in the subset of 1368 sepsis patient was 32.9%, and it was 53.4% with septic shock [19]. In another cross-sectional study of sepsis from India the hospital mortality was 30% [10]. Sepsis mortality was similar in reports from China (33%) [20], but higher than that reported from East and South East Asia (13–16%) [11, 21] and Japan (23%) [22] and less than in Brazil (55.7%) [23] and Turkey (55.7%) [24]. These differences in the outcome across various studies may reflect a true difference or heterogeneity of reporting. The average SOFA and APACHE II score of sepsis and septic shock patients from Western studies are comparable to our population [25, 26]. The reasons for higher mortality in sepsis patients in Indian studies compared to Western studies, despite similar severity score, are difficult to ascertain; however, we speculate that they are likely multifactorial. This may reflect infection-related, host-related or health care delivery-related aspects. However, similar mortality rates are noted in septic shock patients in recent sepsis studies from India, comparable to Western studies. The SMR of sepsis and septic shock patients is not routinely reported in studies. The SMR of sepsis in a study conducted by the ANZICs group was 0.68, while a similar study from India reported a SMR of 1.4 [9, 13, 27]. Our study provides the most recent SMR report on sepsis and septic shock showing an improvement of 0.9 and 0.92 respectively, from the previous Indian study.
ICU and hospital LOS are a marker of cost-effectiveness and quality control for patients discharged alive from the hospital. In Western studies, the average ICU LOS ranges from 7 to 10 days for sepsis and 10–14 days for septic shock, while hospital LOS is somewhat longer for the same cohort [28]. Our study also showed an average ICU and hospital LOS of 8 and 11 days for sepsis and septic shock respectively. An earlier study on sepsis conducted in India has reported a longer hospital stay [9].
Dataset from the Sepsis-3 consensus definition has reported AUCs of SIRS, qSOFA and SOFA as 0.64, 0.66 and 0.75 respectively, for hospital mortality [26] which has been further validated in another large dataset [29]. Our study cohort had AUCs of 0.54, 0.70 and 0.76 for SIRS, qSOFA and SOFA respectively for hospital mortality. It is interesting to note that the AUC of SOFA for hospital mortality was similar in all the studies and is consistently found to be the best discriminator for hospital mortality [30]. Screening or prognostic ability of SIRS, qSOFA and SOFA are reported variably in the literature.
Many variables have been described in the literature as predictors of mortality in sepsis [4]. In our univariate analysis, multiple variables were found to be associated with worse outcomes, consistent with previous literature, reflecting the internal validity of the dataset [32, 33]. Moreover, limitations of interpreting univariate analysis consist of confounding variables and overfitting risks. Most of these variables correlate with the underlying severity of sepsis. It was interesting to note that normal saline, rather than balanced crystalloid, was the most common resuscitation fluid used and the amount of fluid infused in the first 3 h was much less than recommended in the current sepsis guideline. The risk factor for drug-resistant infection was not associated with worse outcomes, which was surprising and may reflect the practice of empirical broad-spectrum antibiotic use. The use of extended infusion of beta-lactam antibiotics was not associated with better outcomes, a finding similar to a recent randomised study [43]. In a large dataset like ours, statistical significance may not always correlate with clinical significance, which is again a subjective interpretation. Multivariable analysis of this large dataset is important to identify independent predictors of mortality. Nosocomial infections associated with sepsis or organ failure were the highest independent predictor of mortality. This has also been observed in other studies [31]. The empirical choice of appropriate antibiotic and adequate source control, similar to surviving sepsis guideline data, was also noticed in our dataset [32, 33]. Low serum albumin as an independent predictor of mortality is a meaningful observation, as many of our patients are malnourished, emphasising the importance of nutritional screening and appropriate management upon admission [34]. Interestingly, the independent use of intravenous thiamine has been significantly associated with improved survival in our study. Variable results have been reported in the literature on the use of intravenous thiamine along with a combination of vitamin C and hydrocortisone [35]. literatures on the independent use of thiamine in sepsis patients is sparse; however, its usefulness is described in patients with acute myocardial infarction and its association with a decreased incidence of delirium has been described [36, 37]. There is also a physiological and biological rationale for using thiamine in sepsis [38].
The site of infection and spread of microorganisms was comparable to INDICAP II study [19]. The demographics and outcome of a minority subset of patients with tropical infections were comparable to INDICAP II point prevalence study with similar incidence constituting 2.9% of the study population, and similar age and SOFA score as in our study. The mortality rate was 13.9% which was lower than the general septic population and was higher than in our study.
This study uniquely addresses the performance improvement programs at the participating centres which is a strong recommendation in the current surviving sepsis guidelines [32, 33]. Automated monitoring tools for sepsis screening is being utilized in many Western centres with equivocal results compared to standard manual monitoring [25]. In our study, most of the participating centres utilized manual chart review for sepsis screening. Sepsis improvement programs in low and middle income countries have not yielded encouraging results [39]. The impact of sepsis performance improvement programs on guideline compliance or patient outcome was not assessed due to the small sample size of 19 participating centres in our study.
Strengths of this study includes its prospective and multicentric design. The large sample size of more than 1000 patients and analysis of over 100 variables contributed to the big dataset from this study. The use of the current Sepsis 3 definition as the inclusion criteria for sepsis and septic shock, along with the one-year study period, as opposed to the point prevalence design, enhances the robustness of the study findings. The processes of sepsis care such as initial resuscitation, hospital course, adjunctive therapies, and organisation of sepsis care have not been addressed in previous epidemiological studies of sepsis from India.
Limitations of the study are specific to the registry design where data interpretation can imply association only. Nevertheless, the large sample size strengthens these associations. Participation in centres was voluntary, and selection was not randomised; it was restricted to experienced tertiary care centres, which may introduce selection bias. Although the centres were chosen from across the country, they were mainly from metro cities, which may not represent the septic population in non-metro ICUs. Systematic Screening of all patients admitted to the ICU to select those who fulfill inclusion criteria may not have been as rigorous as in a closely monitored observational study, leading to a potential selection bias at the patient level. Due to the collection of more than 100 variables and a large data set, some missing data were encountered during the data cleaning process. Moreover, details of drug dosing were not noted during the data collection.
The future of epidemiological studies like these lies in applying machine learning models to large data set to develop better predictive models [40]. Currently we are limited to short-term outcomes such as ICU and hospital mortality; however studies conducted in the West indicate the need to study long-term outcome of sepsis patients at six months or a year on various domains, including mortality, physical and mental functioning [41]. Presently, epidemiological studies are concentrating on demographics, outcome, and treatment related variables, but phenotyping patients with distinctive characterstics may be beneficial for future research. Biobanking of blood samples and metabolic profiling may promote the genomic and proteomic analysis of sepsis patients, potentially leading to meaningful differences from the Western studies [42]. Finally, Registry-based clinical trials may serve as a cost-effective means of further research in resource-constrained environment [44].
Conclusion
This large registry was a multicentre, prospective study on a cohort of more than 1000 patients with sepsis admitted to ICUs in India according to sepsis 3 criteria of sepsis and septic shock. Hospital mortality was 25% and 50% in sepsis and septic shock respectively, which was higher than that reported in Western studies. The SOFA score was the best predictive score for hospital mortality. One third of infections were due to Carbapenem-resistant Gram-negative organisms. Nosocomial infection had the worst odds for mortality, while the correct empirical choice of antibiotics, source control, and intravenous thiamine were protective.
Supplementary Information
Acknowledgements
List of Other Contributors to SEPSIS registry: Dr Fehmeena Begum, Care Hospital, Hydrabad, India Dr Mubeena Qadir, Care Hospital, Hydrabad, India Dr Sourabh S Ambapkar, Sanjeevan Hospital, Pune, India Dr Saanvi S Ambapkar, Sanjeevan Hospital, Pune, India Dr D Soma Sekhar, Apollo Hospital, Chennai, India Dr Ramesh Shanmugam, Apollo Hospital, Chennai, India Dr Ashok P, JSS Medical College, Mysuru, Karnataka, India. Dr Chetak KB, JSS Medical College, Mysuru, Karnataka, India. Dr Abhijit Deshmukh, Ruby Hall Clinic, Pune, India Dr Anand Tiwari, Ruby Hall Clinic, Pune, India Dr Anita Salgar, Sahyadri Hospital, Pune, India Dr Puja Kaushik, Sahyadri Hospital, Pune, India Dr Sushmita Basu, Manipal Hospital, Mukundapur, India Dr Mayur Shah, Nanavati Max Super Speciality Hospital, Mumbai, India Dr Chitra Mehta, Medanta-the Medicity, Haryana, India. Dr Sweta J Patel, Medanta-the Medicity, Haryana, India. Dr Rahul Hurne, Medanta -the Medicity, Haryana, India. Dr Anjana Srivastav, Tata Hospital, Mumbai, India Dr Gaurav Kochhar, Medanta -the Medicity, Haryana, India. Acknowledgement: 1. Shibani Kayal, Critical Care and Medical Education Trust, Kolkata. 2. Chandrani Das, Prajna Pattanayak, Dr Sounak Ghosh and Tapas Kayal, Manipal Hospital Dhakuria, Kolkata, India. 3. Dr Saurabh Nanda and Mr. Joby V George, Medanta–The Medicity, Delhi.
Abbreviations
- APACHE
Acute physiology and chronic health evaluation score
- AUC
Area under the curve
- BMI
Body mass index
- CTRI
Clinical trials registry-India
- DVT
Deep vein thrombosis
- FFP
Fresh frozen plasma
- HFNO
High-flow nasal oxygen
- ICU
Intensive care unit
- IQR
Interquartile ranges
- LMIC
Low and middle-income countries
- LOS
Length of stay
- NIV
Non-invasive ventilation
- qSOFA
Quick sequential (or Sepsis-related) organ failure assessment score
- ROC
Receiver operating curve
- SFI
Sepsis forum India
- SIRS
Systemic inflammatory response syndrome
- SOFA
Sequential (or Sepsis-related) organ failure assessment score
- SOP
Standard operating procedure
- TDM
Therapeutic drug monitoring
- TOF
Train-of-four (used in neuromuscular monitoring)
Author contributions
Todi S was responsible for conceptualization, participation in the study, manuscript preparation and submission. Zirpe K, Mehta Y, Dixit S, Kulkarni AP responsible for conceptualization, reviewing manuscript and participation in the study. Saha A responsible for data cleaning, analyzing data, statistical analysis and manuscript preparation. Rest authors participated in the study.
Funding
Educational Grant from Pfizer Ltd.
Availability of data and materials
No datasets were generated or analysed during the current study.
Declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
Yes.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Subhash Todi, Email: drsubhashtodi@gmail.com.
Other contributors to SEPSIS Registry:
Fehmeena Begum, Mubeena Qadir, Sourabh S. Ambapkar, Saanvi S. Ambapkar, D. Soma Sekhar, Ramesh Shanmugam, P. Ashok, K. B. Chetak, Abhijit Deshmukh, Anand Tiwari, Anita Salgar, Puja Kaushik, Sushmita Basu, Mayur Shah, Chitra Mehta, Sweta J. Patel, Rahul Hurne, Anjana Srivastav, and Gaurav Kochhar
References
- 1.Marshall JC, Vincent JL, Guyatt G, Angus DC, Abraham E, Bernard G, et al. Outcome measures for clinical research in sepsis: a report of the 2nd Cambridge colloquium of the international sepsis forum. Crit Care Med. 2005;33:1708–16. [DOI] [PubMed] [Google Scholar]
- 2.Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the global burden of disease study. Lancet. 2020;395(10219):200–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rhee C, Dantes R, Epstein L, Murphy DJ, Seymour CW, Iwashyna TJ, CDC Prevention Epicenter Program, et al. Incidence and trends of sepsis in US hospitals using clinical vs. claims data, 2009–2014. JAMA. 2017;318(13):1241–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mohamed AKS, Mehta AA, James P. Predictors of mortality of severe sepsis among adult patients in the medical intensive care unit. Lung India. 2017;34(4):330–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stephen AH, Montoya RL, Aluisio AR. Sepsis and septic shock in low- and middle-income countries. Surg Infect (Larchmt). 2020;21(7):571–8. [DOI] [PubMed] [Google Scholar]
- 6.Sulis G, Sayood S, Gandra S. Antimicrobial resistance in low- and middle-income countries: current status and future directions. Expert Rev Anti Infect Ther. 2022;20(2):147–60. [DOI] [PubMed] [Google Scholar]
- 7.Rungta N, Zirpe KG, Dixit SB, Mehta Y, Chaudhry D, Govil D, et al. Indian society of critical care medicine experts committee consensus statement on ICU planning and designing, 2020. Indian J Crit Care Med. 2020;24(Suppl 1):S43–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):801–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chatterjee S, Bhattacharya M, Todi SK. Epidemiology of adult-population sepsis in India: a single center 5 year experience. Indian J Crit Care Med. 2017;21(9):573–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hammond NE, Kumar A, Kaur P, Tirupakuzhi Vijayaraghavan BK, Sepsis in India Prevalence Study (SIPS) Investigator Network. Estimates of sepsis prevalence and outcomes in adult patients in the ICU in India: a cross-sectional study. Chest. 2022;161(6):1543–54. [DOI] [PubMed] [Google Scholar]
- 11.Sakr Y, Jaschinski U, Wittebole X, Szakmany T, ICON Investigators. Sepsis in intensive care unit patients: worldwide data from the intensive care over nations audit. Open Forum Infect Dis. 2018;5(12):313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kaukonen KM, Bailey M, Suzuki S, Pilcher D, Bellomo R. Mortality related to severe sepsis and septic shock among critically ill patients in Australia and New Zealand, 2000–2012. JAMA. 2014;311(13):1308–16. [DOI] [PubMed] [Google Scholar]
- 13.Finfer S, Bellomo R, Lipman J, French C, Dobb G, Myburgh J. Adult-population incidence of severe sepsis in Australian and New Zealand intensive care units. Intensive Care Med. 2004;30(4):589–96. [DOI] [PubMed] [Google Scholar]
- 14.Kadri SS, Rhee C, Strich JR, Morales MK. Estimating ten-year trends in septic shock incidence and mortality in United States academic medical centers using clinical data. Chest. 2017;151(2):278–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gill JS, Arora S, Khanna SP, Kumar KH. Prevalence of multidrug-resistant, extensively drug-resistant, and pandrug-resistant Pseudomonas aeruginosa from a tertiary level intensive care unit. J Glob Infect Dis. 2016;8(4):155–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Gupta R, Malik A, Rizvi M. Epidemiology of multidrug-resistant Gram-negative pathogens isolated from ventilator-associated pneumonia in ICU patients. J Glob Antimicrob Resist. 2017;9:47–50. [DOI] [PubMed] [Google Scholar]
- 17.Beale R, Reinhart K, Brunkhorst FM, PROGRESS Advisory Board. Promoting global research excellence in severe sepsis (PROGRESS): lessons from an international sepsis registry. Infection. 2009;37(3):222–32. [DOI] [PubMed] [Google Scholar]
- 18.Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet. 2022;399(10325):629–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Divatia JV, Mehta Y, Govil D, Zirpe K, Amin PR, Ramakrishnan N, The INDICAPS-II Investigators, et al. Intensive care in India in 2018–2019: the second Indian intensive care case mix and practice patterns study. Indian J Crit Care Med. 2021;25(10):1093–107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhou J, Qian C, Zhao M, Yu X, China Critical Care Clinical Trials Group. Epidemiology and outcome of severe sepsis and septic shock in intensive care units in mainland China. PLoS ONE. 2014;9(9):e107181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Southeast Asia Infectious Disease Clinical Research Network. Causes and outcomes of sepsis in southeast Asia: a multinational multicentre cross-sectional study. Lancet Glob Health. 2017;5(2):e157–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Abe T, Ogura H, Shiraishi A, Kushimoto S, JAAM FORECAST Group. Characteristics, management, and in-hospital mortality among patients with severe sepsis in intensive care units in Japan: the FORECAST study. Crit Care. 2018;22(1):322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Machado FR, Cavalcanti AB, Bozza FA, SPREAD Investigators, Latin American Sepsis Institute Network. The epidemiology of sepsis in Brazilian intensive care units (the sepsis PREvalence assessment database, SPREAD): an observational study. Lancet Infect Dis. 2017;17(11):1180–9. [DOI] [PubMed] [Google Scholar]
- 24.Baykara N, Akalın H, Arslantaş MK, Sepsis Study Group. Epidemiology of sepsis in intensive care units in Turkey: a multicenter, point-prevalence study. Crit Care. 2018;22(1):93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Warttig S, Alderson P, Evans DJ. Automated monitoring compared to standard care for the early detection of sepsis in critically ill patients. Cochrane Database Syst Rev. 2018;6(6):CD012404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Seymour CW, Liu VX, Iwashyna TJ. Assessment of clinical criteria for sepsis: for the third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):762–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Dabhi AS, Khedekar SS, Mehalingam V. A prospective study of comparison of APACHE-IV & SAPS-II scoring systems and calculation of standardised mortality rate in severe sepsis and septic shock patients. J Clin Diagn Res. 2014;8(10):09–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Angus DC, Linde-Zwirble WT, Lidicker J, Clermont G, Carcillo J, Pinsky MR. Epidemiology of severe sepsis in the United States: analysis of incidence, outcome, and associated costs of care. Crit Care Med. 2001;29(7):1303–10. [DOI] [PubMed] [Google Scholar]
- 29.Raith EP, Udy AA, Bailey M, McGloughlin S, MacIsaac C, Bellomo R, Australian and New Zealand Intensive Care Society (ANZICS) Centre for Outcomes and Resource Evaluation (CORE), et al. Prognostic accuracy of the SOFA score, SIRS criteria, and qSOFA score for in-hospital mortality among adults with suspected infection admitted to the intensive care unit. JAMA. 2017;317(3):290–300. [DOI] [PubMed] [Google Scholar]
- 30.Bhattacharya PK, Subrahmanya Murti V, Jamil M, Barman B, Marak PS. Comparison of systemic inflammatory response syndrome, sequential organ failure assessment, and quick sequential organ failure assessment scores to predict mortality in sepsis. J Assoc Phys India. 2022;70(8):11–2. [DOI] [PubMed] [Google Scholar]
- 31.Markwart R, Saito H, Harder T, Tomczyk S, Cassini A, Fleischmann-Struzek C, et al. Epidemiology and burden of sepsis acquired in hospitals and intensive care units: a systematic review and meta-analysis. Intensive Care Med. 2020;46(8):1536–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. 2021;47(11):1181–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Damiani E, Donati A, Serafini G, Rinaldi L, Adrario E, Pelaia P, et al. Effect of performance improvement programs on compliance with sepsis bundles and mortality: a systematic review and meta-analysis of observational studies. PLoS ONE. 2015;10(5):e0125827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mulherin DW, Cogle SV. Updates in nutrition support for critically ill adult patients. Hosp Pharm. 2017;52(1):17–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yao R, Zhu Y, Yu Y, Li Z, Wang L, Zheng L, et al. Combination therapy of thiamine, vitamin C and hydrocortisone in treating patients with sepsis and septic shock: a meta-analysis and trial sequential analysis. Burns Trauma. 2021;9:tkab040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yue S, Wang J, Zhao Y, Ye E, Niu D, Huang J, et al. Thiamine administration may increase survival benefit in critically ill patients with myocardial infarction. Front Nutr. 2023;29(10):1227974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sedhai YR, Shrestha DB, Budhathoki P. Effect of thiamine supplementation in critically ill patients: a systematic review and meta-analysis. J Crit Care. 2021;65:104–15. [DOI] [PubMed] [Google Scholar]
- 38.Costa NA, Pereira AG, Sugizaki CSA, Vieira NM, Garcia LR, de Paiva SAR, et al. Insights into thiamine supplementation in patients with septic shock. Front Med (Lausanne). 2022;28(8): 805199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Morton B, Stolbrink M, Kagima W, Rylance J, Mortimer K. The early recognition and management of sepsis in Sub-Saharan African adults: a systematic review and meta-analysis. Int J Environ Res Public Health. 2018;15(9):2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yang J, Hao S, Huang J, Chen T, Liu R, Zhang P, Feng M, He Y, Xiao W, Hong Y, Zhang Z. The application of artificial intelligence in the management of sepsis. Med Rev. 2023;3(5):369–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shankar-Hari M, Rubenfeld GD. Understanding long-term outcomes following sepsis: implications and challenges. Curr Infect Dis Rep. 2016;18(11):37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Engoren M, Jewell ES, Douville N, Moser S, Maile MD, Bauer ME. Genetic variants associated with sepsis. PLoS ONE. 2022;17(3):e0265052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Monti G, Bradic N, Marzaroli M, Konkayev A, Fominskiy E, Kotani Y, MERCY Investigators. Continuous vs intermittent meropenem administration in critically ill patients with sepsis: the MERCY randomized clinical trial. JAMA. 2023;330(2):141–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li G, Sajobi TT, Menon BK, Korngut L, Lowerison M, James M, 2016 Symposium on Registry-Based Randomized Controlled Trials in Calgary. Registry-based randomized controlled trials- what are the advantages, challenges, and areas for future research? J Clin Epidemiol. 2016;80:16–24. [DOI] [PubMed] [Google Scholar]
- 45.Gliklich RE, Leavy MB, Dreyer NA, (eds). Registries for evaluating patient outcomes: a user’s guide [Internet]. 4th edition. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Sep. Chapter 13, Analysis, Interpretation, and Reporting of Registry Data To Evaluate Outcomes.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.