Abstract
Background
The Non-Healthy Diet Index (NHDI) and the Pro-Healthy Diet Index (PHDI) are two novel indices that evaluate the healthiness of a diet based on the consumption of several food groups. This study aimed to evaluate the association between adherence to the PHDI and NHDI and colorectal cancer (CRC) risk in the Iranian population.
Methods
The current study was conducted as a hospital-based research using a case (n = 71)- matched-controls (n = 142) design in Tehran, Iran. A semi-quantitative food frequency questionnaire was utilized to determine participants’ dietary intake after confirming the diagnosis of CRC and at the time of the interview. The PHDI-10 was employed to assess the consumption of foods with positive health effects, which is linked to the frequency of consuming 10 food groups, and the NHDI-14 was used to assess the consumption of foods that have detrimental effects on health, based on the frequency of 14 food groups. Logistic regression was used to evaluate the association between continuous PHDI and NHDI scores and their tertiles with CRC.
Results
The results indicated that individuals in the highest tertile of the PHDI showed a lower CRC risk compared to those in the lowest tertile (adjusted model- odds ratio (OR) = 0.25; 95% confidence interval (CI): 0.10–0.61; P = 0.002). Also, lower odds of CRC risk were seen with each unit change in the total score of PHDI in the adjusted model (OR = 0.86; 95% CI: 0.76–0.96; P = 0.009). In contrast, individuals in the highest tertile of the NHDI showed a higher risk of CRC compared to those in the lowest tertile (OR = 2.62; 95% CI: 1.09–6.27; P = 0.030) in the adjusted model. Also, higher odds of CRC risk were observed with each unit increase in the total score of NHDI in the adjusted model (OR = 1.13; 95% CI: 1.03–1.25; P = 0.008).
Conclusions
The present study showed that higher adherence to PHDI and NHDI is associated with lower and higher CRC risk, respectively. These results provide valuable insights into the roles of healthy and unhealthy diets in CRC prevention.
Keywords: Diet index, Diet quality, Colorectal neoplasms, Iran
Introduction
In recent years, colorectal cancer (CRC) has become the third most common cancer worldwide [1]. The incidence rate of CRC from 1996 to 2000 in Iran has been reported as 7–8 per 100,000 for both females and males [2], while this rate reached 16.5 and 11.8 per 100,000 in men and women in 2014, respectively [3]. There is also a trend of rising occurrence in younger patients, termed “early-onset” disease [4]. According to the statistics provided by the International Agency for Research on Cancer in 2022, the incidence of CRC in Iran is reported to be 9.4% [5].
This type of cancer, like many others, is influenced by two main categories of genetic and environmental factors [6]. One of the central environmental factors is nutrition and diet [7, 8]. A diet with adequate vegetables, fruits, and fish, as well as rich in fiber, antioxidants, and healthy micronutrients, has been associated with a decreased risk [6]. On the other hand, high red/processed meat, excess dietary fat, and alcohol consumption may be carcinogenic [8–10]. Also, obesity is a significant risk factor for CRC within the range of nutritional impact [9].
Dietary patterns or indices that consider the complex interactions between foods and nutrients may offer a more comprehensive approach to investigating the relationship between diet and disease than focusing on individual foods [11, 12]. The number of studies examining the potential impact of dietary patterns on CRC in developing countries is limited [13], and as a result, this evidence is limited and inconclusive [14]. Diet quality is broadly defined. As shown by a systematic review, the majority of results in large samples show an existing association between low diet quality and CRC [15]. However, the evidence is insufficient to draw a conclusion and establish dietary recommendations [15–19].
The Non-Healthy Diet Index (NHDI) and the Pro-Healthy Diet Index (PHDI) are two novel indices that evaluate the healthiness of a diet based on the consumption of several food groups [20]. These two indices focus on almost all the important food groups, which may be better indicators of diet quality than singular nutrients or food items. The PHDI consists of possibly healthy food groups such as whole grains, fish, legumes, fruits, and vegetables. While there are limited studies assessing diet quality using the PHDI, it is evident that consuming foods included in the index has a beneficial effect on health, as these products are recommended in the Mediterranean diet [21]. Meanwhile, the NHDI comprises relatively unhealthy food groups such as refined cereals, white bread, fast foods, processed meat, sweets, and sweetened beverages [20]. This indicator also has a negative impact on health [22].
To our knowledge, the association between PHDI/NHDI and cancers such as CRC has not been previously examined. However, a case-control study showed that consuming a pro-healthy diet is inversely related to the incidence of breast cancer [23]. Another case-control study further showed a positive correlation between a greater degree of nutritional knowledge and adopting a pro-healthy diet. Moreover, it showed a significant reduction in the likelihood of developing lung cancer in men and breast cancer in women [24]. The dietary patterns of the Iranian population are deteriorating, with an increasing preference for fast foods. This shift can significantly affect the health of different social groups. The nutrition transition is a global health challenge, and understanding these food patterns is crucial for informing the development of nutrition policies [25]. Hence, this study aimed to evaluate the association between adherence to PHDI and NHDI and CRC risk in the Iranian population.
Methods
Study population
The current study was conducted as a hospital-based research using a case-matched-control design. It was performed in three general hospitals (Ayatollah Taleghani, Imam Hossein, and Shariati) and 19 CRC surgical units of the Cancer Institute of Imam Khomeini Hospital Complex in Tehran City, Iran. The inclusion criteria for the case group included age 40 to 75 years and recent definite diagnosis of CRC. This group had no history of inflammatory bowel disease and did not have any previous diagnosis of cancer in other organs. The matched-control group consisted of individuals (40–75 years old) randomly selected from the same hospitals. These individuals were hospitalized for non-neoplastic and acute conditions at the time of selection, excluding those with chronic diseases related to diet. The most prevalent causes for hospitalization included disc disorders, bone and joint disorders, fractures, and sprains. The exclusion criteria included the following: unwillingness to participate in the study, pregnancy, total energy intake outside the mean ± 3 standard deviations (SDs), and incomplete completion of the food frequency questionnaire (FFQ). Every CRC patient was matched with two controls by sex and age. The study population was calculated based on a previous study, with odds ratio (OR) = 0.45, β = 0.2, and α = 0.05 [26]. It should be noted that certain aspects of the current study have been previously published [27–29].
Dietary evaluation and diet quality scores
A semi-quantitative FFQ was utilized to determine participants’ dietary intake in the last year. This questionnaire consisted of 168 foods and drinks commonly consumed by Iranian people. The questionnaire’s validity has already been assessed [30]. The frequency of each food was recorded based on the day, week, month, and year. Then, to simplify the assessment of food type and portion size, a set of household measuring items was used (e.g., teaspoons, tablespoons, glasses, cups, bowls, plates, and spatulas) [31]. Then, each food item’s portion size was changed to grams, and finally, the total intake of each food item was calculated by multipling by the frequency of daily intake. Finally, the Nutritionist IV software (version 7.0) was employed to calculate nutrients and energy.
The two dietary indices, the PHDI and NHDI, have recently been developed and validated in the Polish population [19]. Initially, they were derived from the assessment of dietary habits, lifestyle, and nutrition knowledge (KomPAN) questionnaire in a study conducted in Poland [25]. Other versions of PHDI and NHDI with different items can be found in the literature [31, 32]. Therefore, we have labeled these indicators with the respective number of items to differentiate them from other versions. The PHDI-10 was employed to assess the consumption of foods with positive health effects, which is linked to the frequency of consuming 10 food groups: whole grain products, whole bread, fresh cheeses, white meats, fermented dairy products, milk, fish, fruits, vegetables, and legume seeds. At first, food group intake (total grams) was divided into tertiles and then scored 0–2 for the first, second and last tertiles. Finally, the PHDI-10 ranged from 0 to 20 [32].
Most diet quality indices combine healthy and unhealthy food components to judge a diet, but the NHDI only scores the unhealthiness of the diets; therefore, the higher the score, the unhealthier the diet. The NHDI-14 was used to assess the consumption of foods that have detrimental effects on health, based on the frequency of 14 food groups: fried foods, refined cereal products, white bread, butter, lard, fast foods, processed cheeses, sweets, canned meat, red meat, processed meat products, sweetened beverages, alcoholic beverages, and energy drinks. However, three items were not included in the present study because these items were not assessed in the Iranian version of the FFQ (energy drinks, alcoholic beverages, and lard). Afterward, each food group was divided into tertiles and assigned a score ranging from 0 to 2 based on their tertile placement (score 2 for the first, 1 for the second, and 0 for the last tertiles). Thus, the maximum NHDI-14 score was 22. A higher score for each index indicates greater adherence to the PHDI-10 and NHDI-14 indices [32–34].
Personal characteristics and anthropometric assessments
Characteristics such as a history of CRC in the family (yes, no), smoking habits (never, former, current), medication information (ibuprofen (yes, no), aspirin (yes, no), and acetaminophen (yes, no)), education (no formal education, elementary, junior/senior/ high school, and diploma/college/university), and income (dollars/month) were obtained by a checklist, and dietary intake and physical activity (metabolic equivalent of task (MET)-hour/day) were extracted by a questionnaire. Participants’ height was determined by a SECA body meter to the nearest 0.1 cm and weight was measured with a precision of 0.1 kg. In addition, the waist-to-hip ratio (WHR) was assessed by measuring waist circumference (WC) and hip circumference (HC) with a tape measure (minimum waist/maximum hip). Additionally, the body mass index (BMI) was determined using the following formula: weight (kg) divided by height (m) squared.
Statistical analysis
For data analysis, SPSS version 26.0 was used. Additionally, the Kolmogorov-Smirnov test was employed to assess the normality of the data. The independent samples T-test or Mann-Whitney test was used for continuous variables, and the chi-square test was used for categorical variables. The values for continuous variables are reported as mean ± SD or median (interquartile range [IQR]). Categorical variables are reported as percentages. Furthermore, in order to assess the intakes of food groups, we first converted the PHDI and NHDI into tertiles based on the total population. We then conducted an analysis using one-way analysis of variance (ANOVA). Logistic regression was used to evaluate the association between continuous PHDI and NHDI scores and their tertiles with CRC. In the second model, the role of family history of CRC (yes, no), smoking (never, former, current), physical activity (MET-hour/day), energy intake (kcal/day), BMI (kg/m2), vegetable (raw/fresh, boiled, fried, fried/frozen) and meat (fried, fried/boiling, smoking/grilling) cooking methods, and taking acetaminophen (yes, no), aspirin (yes, no), and ibuprofen (yes, no), were adjusted (potential confounders were selected using previous studies [35–37]). In both models, the first tertile was considered the reference category. A p-value < 0.05 was considered statistically significant.
Results
A total of 267 patients were initially screened for the current study. However, 36 controls and 18 cases were excluded for two reasons: their total energy intake fell outside the range of ± 3 SDs from the mean energy intake, and they provided incomplete dietary intake data by FFQ. Finally, a total of 142 controls and 71 cases were included for statistical analysis.
Table 1 represents the baseline features of the present study. The mean of PHDI (P = 0.005) and fiber intake (P ˂ 0.001) was significantly higher in the control group, but WHR was significantly higher in the case groups (P = 0.002). Also, a family history of CRC was significantly more prevalent in the case group (P = 0.017), but the use of acetaminophen (P = 0.004) and aspirin (P = 0.016) was significantly higher in the control group.
Table 1.
The baseline features of the study population
| Variables | Cases (71) | Controls (142) | P-value |
|---|---|---|---|
| Quantitative Variables | |||
| Age (years) 1 | 58.2 ± 10.4 | 57.7 ± 10.4 | 0.746 |
| Pro-healthy diet index score 1 | 9.5 ± 3.0 | 10.9 ± 3.2 | 0.005 |
| Non-healthy diet index score 1 | 11.8 ± 3.4 | 10.7 ± 4.1 | 0.067 |
| Energy (kcal/day) 1 | 2262.3 ± 450.1 | 2255.2 ± 341.2 | 0.908 |
| Fiber (g/day) 1 | 18.9 ± 2.3 | 20.4 ± 3.1 | <0.001 |
| Income (dollars/month) 2 | 393.0 (253.0) | 402.0 (302.0) | 0.206 |
| Physical activity (MET-h/day) 1 | 36.8 ± 3.6 | 36.7 ± 4.8 | 0.932 |
| BMI (kg/m2) 1 | 27.6 ± 4.2 | 26.6 ± 4.2 | 0.362 |
| WHR1 | 0.97 ± 0.08 | 0.94 ± 0.06 | 0.002 |
| Qualitative Variables | |||
| Gender, male 3 | 35 (49.3) | 70 (49.3) | 0.558 |
| Smoking 3 | 0.164 | ||
|
Never Former Current |
57 (80.2) 8 (11.3) 6 (8.5) |
101 (70.1) 15 (10.6) 26 (18.3) |
|
| Education 3 | 0.147 | ||
|
No formal education Elementary Junior/Senior/ High school Diploma/College/University |
28 (39.3) 22 (31.0) 7 (9.9) 14 (19.7) |
36 (25.4) 45 (31.6) 19 (13.4) 42 (29.6) |
|
| Common ways of cooking meat 3 | 0.282 | ||
|
Fried Fried / Boiling Smoking / Grilling |
20 (28.2) 35 (49.3) 16 (22.5) |
28 (19.7) 71 (50.0) 43 (30.3) |
|
| Common ways of preparing vegetables 3 | 0.083 | ||
|
Raw / Fresh Boiled Fried, Fried / Frozen |
29 (40.8) 8 (11.3) 34 (47.9) |
78 (54.9) 18 (12.7) 46 (32.4) |
|
| Family history of CRC, yes 3 | 7 (9.9) | 3 (2.1) | 0.017 |
| Ibuprofen, yes 3 | 5 (7.0) | 22 (15.5) | 0.059 |
| Aspirin, yes 3 | 1 (1.4) | 14 (9.9) | 0.016 |
| Acetaminophen, yes 3 | 4 (5.6) | 28 (19.7) | 0.004 |
MET: metabolic equivalent of task, BMI: body mass index, WHR: waist-to-hip ratio, CRC: colorectal cancer
-Using the chi-square test for categorical variables and the Mann-Whitney or independent samples T-test for continuous variables
1Values are mean ± SD
2Values are median (IQR)
3Values are number (percent)
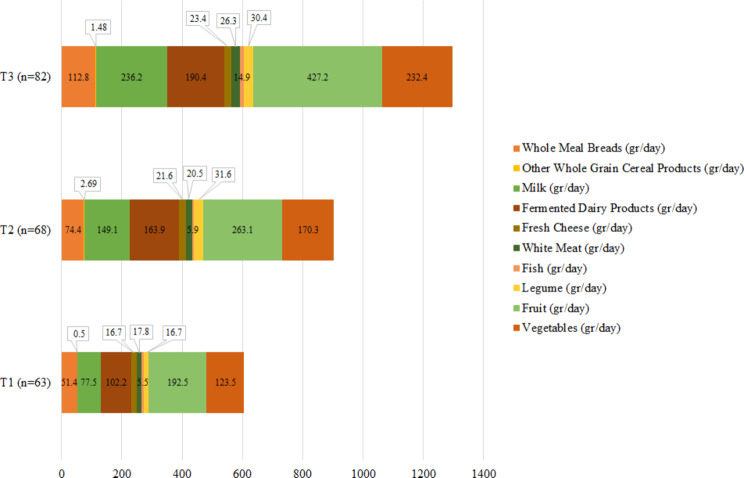
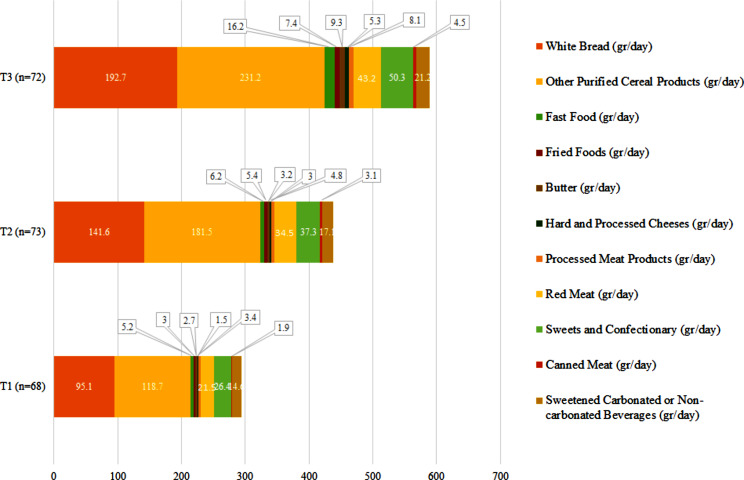
The PHDI and NHDI components are shown in Figs. 1 and 2, respectively. In the highest tertile, consumption of all PHDI components (legumes, vegetables, fruits, white meat, fish, fermented dairy products, milk, fresh cheese, whole breads, and whole grains) was significantly higher compared to the lowest tertile. Additionally, all NHDI components, except for sweetened carbonated or non-carbonated beverages, were significantly higher in the highest tertile.
Fig. 1.
The consumption of components of the PHDI across the tertiles of the PHDI
Fig. 2.
The consumption of components of the NHDI across the tertiles of the NHDI
Based on the data presented in Table 2, the results indicate lower odds of CRC risk for each unit change in the total score of PHDI in crude (OR = 0.87; 95% confidence interval (CI): 0.79–0.96; P = 0.006) and adjusted models (OR = 0.86; 95% CI: 0.76–0.96; P = 0.009). Also, individuals in the highest tertile (T3) of the PHDI showed a lower CRC risk compared to those in the lowest tertile (T1). This association holds for both the crude (OR = 0.30; 95% CI: 0.14–0.63; P = 0.001) and adjusted models (OR = 0.25; 95% CI: 0.10–0.61; P = 0.002).
Table 2.
Association between pro- and non-healthy diet indices and colorectal cancer
| Tertiles of Indices | Case/Control | Model 1 | Model 2 | ||||
|---|---|---|---|---|---|---|---|
| OR | 95% CI | P-value | OR | 95% CI | P-value | ||
| Pro-Healthy Diet Index | |||||||
| Total score | 71/141 | 0.87 | 0.79–0.96 | 0.006 | 0.86 | 0.76–0.96 | 0.009 |
| T1 (≤ 8) | 29/34 | 1.00 | Ref. | - | 1.00 | Ref. | - |
| T2 (9–11) | 25/43 | 0.68 | 0.33–1.37 | 0.282 | 0.45 | 0.20–1.04 | 0.062 |
| T3 (≥ 12) | 17/65 | 0.30 | 0.14–0.63 | 0.001 | 0.25 | 0.10–0.61 | 0.002 |
| Non-Healthy Diet Index | |||||||
| Total score | 71/141 | 1.07 | 0.99–1.15 | 0.069 | 1.13 | 1.03–1.25 | 0.008 |
| T1 (≤ 9) | 18/50 | 1.00 | Ref. | - | 1.00 | Ref. | - |
| T2 (10–12) | 24/49 | 1.36 | 0.65–2.81 | 0.407 | 1.88 | 0.82–4.28 | 0.132 |
| T3 (≥ 13) | 29/43 | 1.87 | 0.91–3.82 | 0.086 | 2.62 | 1.09–6.27 | 0.030 |
OR: odds ratio, CI: confidence interval, Ref: reference, T: tertile
Model 1: crude model
Model 2: adjusted for energy intake, BMI, physical activity, smoking, family history of CRC, vegetable and meat cooking methods, and use of ibuprofen, aspirin, and acetaminophen
-Obtained from logistic regression
-These values are odds ratios (95% CIs)
-Significant values are shown in bold
On the contrary, the results showed higher odds of CRC risk for each unit increase in the total score of NHDI in the adjusted model (OR = 1.13; 95% CI: 1.03–1.25; P = 0.008). Also, in the adjusted model, individuals in the highest tertile of the NHDI showed a greater risk of CRC compared to those in the lowest tertile (OR = 2.62; 95% CI: 1.09–6.27; P = 0.030). However, in the crude NHDI model, the risk of CRC in the highest tertile compared to the lowest tertile was not statistically significant (OR = 1.87; 95% CI: 0.91–3.82; P = 0.086).
Discussion
The present study examined the association between the PHDI and NHDI, which are indicators of a healthy and unhealthy diet, respectively, with CRC in an Iranian sample. The results showed that the PHDI had an inverse association with the risk of CRC, while the NHDI had a direct association with this condition.
To our knowledge, the association between the PHDI/NHDI and cancers such as CRC has not been previously assessed. However, the association of CRC with various dietary patterns and diet quality indices (with almost similar components) has been explored by several previous studies [15, 38, 39]. In line with our results, Thanikachalam et al. showed that a “healthy” dietary pattern, commonly characterized by substantial consumption of fish, whole grains, fruits, vegetables, nuts, and dairy products, was associated with a reduced risk of CRC [6]. Furthermore, Castelló et al. showed that the “Mediterranean” dietary pattern had a protective effect against CRC among a sample of Spanish adults [40]. The findings of the present study also showed that the consumption of legumes, vegetables, and fruits was higher in the highest tertile of the PHDI compared to the lowest tertile. These plant-based foods are rich in fiber, micronutrients, antioxidants, and phytochemicals, all of which are protective against cancer [41–44]. Also, our results showed that people with CRC had a lower intake of fiber, which supports this explanation. In addition to plant-based foods, individuals in the highest tertile of the PHDI also indicated greater consumption of fresh cheese. As mentioned above, dairy products are also a part of healthy patterns. In fact, calcium, the main micronutrient provided by dairy products in the diet, is inversely associated with CRC [45].
Concerning the inverse index, the NHDI, individuals in the highest tertile showed elevated consumption of butter, fried foods, fast foods, refined cereal products, white bread, hard and processed cheeses, red meat, processed meat, canned meat, sweets, and sweetened beverages. Most diet quality indices combine healthy and unhealthy food components to judge a diet, but the NHDI only scores the unhealthiness of the diets; therefore, the higher the score, the unhealthier the diet. As the NHDI does not include healthy food groups, it is more appropriate to complement it with the PHDI to assess a given diet. In the present study, the results of these complementing indices indicated an overall poorer diet in the cases than in the controls. Comparable results have been found for other “unhealthy” diet indicators.
Thanikachalam et al. showed that a dietary pattern characterized as “unhealthy,” which includes high consumption of refined grains, processed and red meat, sugar-sweetened beverages, and other sugary foods, is linked to an elevated risk of CRC [39]. A similar indicator of an unhealthy diet is the “Western” dietary pattern, characterized by excessive consumption of junk food, high-calorie beverages, refined grains, sweets, as well as red and processed meat [40]. It has been shown that lower adherence to this dietary pattern is correlated with a decreased risk of CRC [40, 46]. The NHDI and unhealthy dietary patterns, like the Western pattern, assess food groups that can induce systemic inflammation, a process that contributes to the progression of cancer [47]. Furthermore, refined carbohydrates and simple sugars in the food lists of these dietary patterns may cause a high glycemic response and, ultimately, insulin resistance. These consequences may cause this type of cancer [48–50]. Also, the high saturated fat content in animal food groups and, in some instances, trans fats and carcinogenic substances in fast foods and processed meats may be involved in the development of CRC [51–53].
Studies on the association between the PHDI/NHDI and cancers need to be included in the literature, and more research is needed to establish a definitive association. However, a few studies on the PHDI and NHDI have found an association with CRC-related conditions. One study showed that the PHDI had a strong inverse association with homocysteine [54]. An elevated homocysteine level is associated with CRC and adenomatous polyps [55]. Since this association may be independent of oxidative stress indices [56], it requires further investigation as a mediating factor. Furthermore, a comprehensive two-year cohort study showed a strong correlation between a reduction in the PHDI and an elevated risk of obesity in individuals adhering to two distinct lifestyle patterns. In one of the lifestyle patterns, a lower NHDI score was found to be correlated with lower levels of fasting blood glucose [57]. Because of the abundance of calorie-dense or high sugar/fat food choices in the NHDI, this association seems reasonable. In this regard, one study showed a significant association between metabolic syndrome and CRC and suggested that high BMI or WC may be responsible [58]. In the present study, BMI and WHR were found to be higher in the case group compared to the control group. However, it is important to note that only the latter showed statistically significant differences. These mechanisms can further clarify the association between these dietary indices and CRC.
Inflammation is a significant contributor to tumorigenesis in CRC. Evidence shows that the use of anti-inflammatory drugs may help prevent or delay the onset of CRC, highlighting the critical role of inflammation in the development and progression of all forms of CRC [59, 60]. Gut microbiota can impact tumor growth in various ways, one of which is by producing metabolites that induce genetic instability in host cells [61]. Dysbiosis has been found in patients with CRC, who tend to have reduced microbial diversity compared to healthy individuals [62]. Additionally, the composition of gut microbiota can affect the presence of immune cells that produce pro-inflammatory cytokines. This, in turn, can lead to a pro-inflammatory environment that promotes tumor growth [63]. Adhering to healthy diets appears to be associated with a reduction in inflammation [64]. Additionally, dietary habits can significantly influence the composition of gut microbiota [65]. A healthier eating pattern is associated with lower inflammatory markers as well as greater richness of gut microbiota [66].
One limitation of the current study is that the sample size is relatively small, which reduces the ability to draw meaningful conclusions from the results. Also, response biases are inevitable in a retrospective and case-control design, especially when people have to recall food intake from previous times. The disease diagnosis itself may divert participants’ minds from their actual diets. Also, more details about CRC diagnosis were not available. Furthermore, a causal association cannot be inferred in a case-control design, so other studies, such as prospective cohorts, should investigate causality. To highlight the strengths of this study, we should first mention the novelty of the PHDI and NHDI in assessing the association between diet quality and CRC. In addition, to our knowledge, this is the first time these indices have been applied in Iran or the Middle East region. Also, participants in the control group were matched based on sex and age.
Conclusions
In summary, this study has identified a significant association between the PHDI/NHDI and the risk of CRC. Therefore, increasing the consumption of legumes, fruits, and vegetables, while reducing the intake of butter, fried foods, fast foods, processed meats, refined grains, and sweets, may lower the risk of CRC. These results provide valuable insights into the role of healthy and unhealthy diets in CRC prevention. If other studies confirm the results, healthcare professionals can apply these findings in health promotion programs to reduce complications and costs associated with this type of cancer.
Acknowledgements
We sincerely thank all field investigators, staff, and participants of the present study.
Author contributions
M.R.Z., I.K., A.K., Z.S., M.M.R. and M.N; Contributed to writing the first draft. Z.S. and M.N.; Contributed to statistical analysis, and interpretation of data. M.N and B.R.; Contributed to the research concept, supervised the work and revised the manuscript. All authors read and approved the final manuscript.
Funding
Not applicable.
Data availability
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was conducted in accordance with the ethical standards of the Declaration of Helsinki and was approved by the Medical Research and Ethics Committee of Shiraz University of Medical Sciences (IR.SUMS.SCHEANUT.REC.1401.011). Informed consent was obtained from all subjects and their legal guardian(s).
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Mehran Nouri, Email: mehran_nouri71@yahoo.com.
Bahram Rashidkhani, Email: rashidkhani@yahoo.com.
References
- 1.Xi Y, Xu P. Global colorectal cancer burden in 2020 and projections to 2040. Translational Oncol. 2021;14(10):101174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ansari R, Mahdavinia M, Sadjadi A, Nouraie M, Kamangar F, Bishehsari F, Fakheri H, Semnani S, Arshi S, Zahedi M-J. Incidence and age distribution of colorectal cancer in Iran: results of a population-based cancer registry. Cancer Lett. 2006;240(1):143–7. [DOI] [PubMed] [Google Scholar]
- 3.Roshandel G, Ghanbari-Motlagh A, Partovipour E, Salavati F, Hasanpour-Heidari S, Mohammadi G, Khoshaabi M, Sadjadi A, Davanlou M, Tavangar S-M. Cancer incidence in Iran in 2014: results of the Iranian National Population-based Cancer Registry. Cancer Epidemiol. 2019;61:50–8. [DOI] [PubMed] [Google Scholar]
- 4.Siegel RL, Jakubowski CD, Fedewa SA, Davis A, Azad NS. Colorectal cancer in the young: epidemiology, prevention, management. Am Soc Clin Oncol Educational Book. 2020;40:e75–88. [DOI] [PubMed]
- 5.Costamagna D, Costelli P, Sampaolesi M, Penna F. Role of inflammation in muscle homeostasis and myogenesis. Mediat Inflamm. 2015;2015(1):805172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Thanikachalam K, Khan G. Colorectal cancer and nutrition. Nutrients. 2019;11(1):164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Song M, Chan AT. Environmental factors, gut microbiota, and Colorectal Cancer Prevention. Clin Gastroenterol Hepatol. 2019;17(2):275–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Murphy N, Moreno V, Hughes DJ, Vodicka L, Vodicka P, Aglago EK, Gunter MJ, Jenab M. Lifestyle and dietary environmental factors in colorectal cancer susceptibility. Mol Aspects Med. 2019;69:2–9. [DOI] [PubMed] [Google Scholar]
- 9.Ye P, Xi Y, Huang Z, Xu P. Linking obesity with colorectal cancer: epidemiology and mechanistic insights. Cancers. 2020;12(6):1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ocvirk S, Wilson AS, Appolonia CN, Thomas TK, O’Keefe SJD. Fiber, Fat, and Colorectal Cancer: New Insight into Modifiable Dietary Risk factors. Curr Gastroenterol Rep. 2019;21(11):62. [DOI] [PubMed] [Google Scholar]
- 11.Jacques PF, Tucker KL. Are dietary patterns useful for understanding the role of diet in chronic disease? 12. Am J Clin Nutr. 2001;73(1):1–2. [DOI] [PubMed] [Google Scholar]
- 12.Hu FB. Dietary pattern analysis: a new direction in nutritional epidemiology. Curr Opin Lipidol. 2002;13(1):3–9. [DOI] [PubMed] [Google Scholar]
- 13.Tayyem RF, Bawadi HA, Shehadah I, Agraib LM, AbuMweis SS, Al-Jaberi T, Al-Nusairr M, Bani-Hani KE, Heath DD. Dietary patterns and colorectal cancer. Clin Nutr. 2017;36(3):848–52. [DOI] [PubMed] [Google Scholar]
- 14.Tabung FK, Brown LS, Fung TT. Dietary patterns and colorectal cancer risk: a review of 17 years of evidence (2000–2016). Curr Colorectal cancer Rep. 2017;13:440–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moazzen S, van der Sloot KWJ, Bock GHd, Alizadeh BZ. Systematic review and meta-analysis of diet quality and colorectal cancer risk: is the evidence of sufficient quality to develop recommendations? Crit Rev Food Sci Nutr. 2021;61(16):2773–82. [DOI] [PubMed] [Google Scholar]
- 16.Schwingshackl L, Schwedhelm C, Galbete C, Hoffmann G. Adherence to Mediterranean diet and risk of cancer: an updated systematic review and meta-analysis. Nutrients. 2017;9(10):1063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schwingshackl L, Hoffmann G. Diet quality as assessed by the healthy eating Index, the alternate healthy eating Index, the Dietary approaches to stop hypertension score, and health outcomes: a systematic review and meta-analysis of cohort studies. J Acad Nutr Dietetics. 2015;115(5):780–800. e785. [DOI] [PubMed] [Google Scholar]
- 18.Jones P, Cade JE, Evans CE, Hancock N, Greenwood DC. The Mediterranean diet and risk of colorectal cancer in the UK women’s Cohort Study. Int J Epidemiol. 2017;46(6):1786–96. [DOI] [PubMed] [Google Scholar]
- 19.Haslam A, Robb SW, Hébert JR, Huang H, Wirth MD, Shivappa N, Ebell MH. The association between Dietary Inflammatory Index scores and the prevalence of colorectal adenoma. Public Health Nutr. 2017;20(9):1609–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kowalkowska J, Wadolowska L, Czarnocinska J, Galiński G, Długosz A, Łoboda D, Czlapka-Matyasik M. Data-Driven Dietary Patterns and Diet Quality Scores: Reproducibility and Consistency in Sex and Age Subgroups of Poles Aged 15–65 Years. 2020. [DOI] [PMC free article] [PubMed]
- 21.Ratajczak J, Czerniak U, Wieliński D, Ciekot-Sołtysiak M, Zieliński J, Gronek P, Demuth A. Pro-healthy diet properties and its determinants among aging masters athletes. Int J Environ Res Public Health. 2021;18(14):7614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gacek M, Kosiba G, Wojtowicz A. Sense of generalised self-efficacy and body mass index, diet health quality and pro-health behaviours of nursing students and active professional nurses. Med Pracy. 2023;74(1):251–61. [DOI] [PubMed] [Google Scholar]
- 23.Stasiewicz B, Wadolowska L, Biernacki M, Slowinska MA, Drozdowski M. Hybrid dietary-blood inflammatory profiles and postmenopausal breast cancer: a case-control study. Nutrients. 2020;12(11):3503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hawrysz I, Krusinska B, Slowinska M, Wadolowska L, Czerwinska A, Biernacki M. Nutritional knowledge, diet quality and breast or lung cancer risk: a case-control study of adults from Warmia and Mazury region in Poland. Roczniki Państwowego Zakładu Higieny 2016, 67(1). [PubMed]
- 25.Abdi F, Atarodi Z, Mirmiran P, Esteki T. Review of nutritional status in Iranian population. Focus Sci. 2016;2(3):1–4. [Google Scholar]
- 26.Terry P, Giovannucci E, Michels KB, Bergkvist L, Hansen H, Holmberg L, Wolk A. Fruit, vegetables, dietary fiber, and risk of colorectal cancer. J Natl Cancer Inst. 2001;93(7):525–33. [DOI] [PubMed] [Google Scholar]
- 27.Kahrizsangi MA, Ebrahimi Z, Shateri Z, Mansouri F, Zangene A, Rajabzadeh-Dehkordi M, Nouri M, Rashidkhani B. Carbohydrate quality indices and colorectal cancer risk: a case-control study. BMC Cancer. 2023;23(1):347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bentyaghoob S, Dehghani F, Alimohammadi A, Shateri Z, Kahrizsangi MA, Nejad ET, Nouri M, Rashidkhani B. Oxidative balance score and dietary phytochemical index can reduce the risk of colorectal cancer in Iranian population. BMC Gastroenterol. 2023;23(1):183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yarmand S, Rashidkhani B, Alimohammadi A, Shateri Z, Shakeri M, Sohrabi Z, Nouri M. A healthful plant-based diet can reduce the risk of developing colorectal cancer: case-control study. J Health Popul Nutr. 2024;43(1):111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mirmiran P, Esfahani FH, Mehrabi Y, Hedayati M, Azizi F. Reliability and relative validity of an FFQ for nutrients in the Tehran lipid and glucose study. Public Health Nutr. 2010;13(5):654–62. [DOI] [PubMed] [Google Scholar]
- 31.Ghaffarpour M, Houshiar-Rad A, Kianfar H. The manual for household measures, cooking yields factors and edible portion of foods. Tehran: Nashre Olume Keshavarzy. 1999;7(213):42–58. [Google Scholar]
- 32.Mahmoodi M, Gabal BC, Mohammadi F, Ibrahim FM, Jalilpiran Y, Nouri M, Faghih S. The association between healthy and unhealthy dietary indices with prostate cancer risk: a case-control study. J Health Popul Nutr. 2024;43(1):90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gacek M, Kosiba G, Wojtowicz A. Personality determinants of diet quality among Polish and Spanish physical education students. Int J Environ Res Public Health. 2021;18(2):466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kowalkowska J, Wadolowska L, Czarnocinska J, Galinski G, Dlugosz A, Loboda D, Czlapka-Matyasik M. Data-driven dietary patterns and diet quality scores: reproducibility and consistency in sex and age subgroups of poles aged 15–65 years. Nutrients. 2020;12(12):3598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Demb J, Earles A, Martínez ME, Bustamante R, Bryant AK, Murphy JD, Liu L, Gupta S. Risk factors for colorectal cancer significantly vary by anatomic site. BMJ open Gastroenterol. 2019;6(1):e000313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Eldridge RC, Doubeni CA, Fletcher RH, Zauber AG, Corley DA, Doria-Rose VP, Goodman M. Uncontrolled confounding in studies of screening effectiveness: an example of colonoscopy. J Med Screen. 2013;20(4):198–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Aleksandrova K, Pischon T, Jenab M, Bueno-de-Mesquita HB, Fedirko V, Norat T, Romaguera D, Knüppel S, Boutron-Ruault M-C, Dossus L, et al. Combined impact of healthy lifestyle factors on colorectal cancer: a large European cohort study. BMC Med. 2014;12(1):168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Safari A, Shariff ZM, Kandiah M, Rashidkhani B, Fereidooni F. Dietary patterns and risk of colorectal cancer in Tehran Province: a case-control study. BMC Public Health. 2013;13:222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tabung FK, Brown LS, Fung TT. Dietary patterns and colorectal Cancer risk: a review of 17 years of evidence (2000–2016). Curr Colorectal Cancer Rep. 2017;13(6):440–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Castelló A, Amiano P, Fernández de Larrea N, Martín V, Alonso MH, Castaño-Vinyals G, Pérez-Gómez B, Olmedo-Requena R, Guevara M, Fernandez-Tardon G, et al. Low adherence to the western and high adherence to the mediterranean dietary patterns could prevent colorectal cancer. Eur J Nutr. 2019;58(4):1495–505. [DOI] [PubMed] [Google Scholar]
- 41.Xu M, Chen Y-M, Huang J, Fang Y-J, Huang W-Q, Yan B, Lu M-S, Pan Z-Z, Zhang C-X. Flavonoid intake from vegetables and fruits is inversely associated with colorectal cancer risk: a case–control study in China. Br J Nutr. 2016;116(7):1275–87. [DOI] [PubMed] [Google Scholar]
- 42.Song Y, Liu M, Yang FG, Cui LH, Lu XY, Chen C. Dietary fibre and the risk of colorectal cancer: a case- control study. Asian Pac J Cancer Prev. 2015;16(9):3747–52. [DOI] [PubMed] [Google Scholar]
- 43.Ding S, Xu S, Fang J, Jiang H. The protective effect of polyphenols for colorectal cancer. Front Immunol. 2020;11. [DOI] [PMC free article] [PubMed]
- 44.Afrin S, Giampieri F, Gasparrini M, Forbes-Hernández TY, Cianciosi D, Reboredo-Rodriguez P, Zhang J, Manna PP, Daglia M, Atanasov AG, et al. Dietary phytochemicals in colorectal cancer prevention and treatment: a focus on the molecular mechanisms involved. Biotechnol Adv. 2020;38:107322. [DOI] [PubMed] [Google Scholar]
- 45.Meng Y, Sun J, Yu J, Wang C, Su J. Dietary intakes of Calcium, Iron, Magnesium, and potassium elements and the risk of Colorectal Cancer: a Meta-analysis. Biol Trace Elem Res. 2019;189(2):325–35. [DOI] [PubMed] [Google Scholar]
- 46.Garcia-Larsen V, Morton V, Norat T, Moreira A, Potts JF, Reeves T, Bakolis I. Dietary patterns derived from principal component analysis (PCA) and risk of colorectal cancer: a systematic review and meta-analysis. Eur J Clin Nutr. 2019;73(3):366–86. [DOI] [PubMed] [Google Scholar]
- 47.Zitvogel L, Pietrocola F, Kroemer G. Nutrition, inflammation and cancer. Nat Immunol. 2017;18(8):843–50. [DOI] [PubMed] [Google Scholar]
- 48.Sieri S, Krogh V, Agnoli C, Ricceri F, Palli D, Masala G, Panico S, Mattiello A, Tumino R, Giurdanella MC, et al. Dietary glycemic index and glycemic load and risk of colorectal cancer: results from the EPIC-Italy study. Int J Cancer. 2015;136(12):2923–31. [DOI] [PubMed] [Google Scholar]
- 49.Yoon YS, Keum N, Zhang X, Cho E, Giovannucci EL. Hyperinsulinemia, insulin resistance and colorectal adenomas: a meta-analysis. Metabolism. 2015;64(10):1324–33. [DOI] [PubMed] [Google Scholar]
- 50.Ma X, Nan F, Liang H, Shu P, Fan X, Song X, Hou Y, Zhang D. Excessive intake of sugar: an accomplice of inflammation. Front Immunol. 2022;13. [DOI] [PMC free article] [PubMed]
- 51.Jaworowska A, Blackham T, Davies IG, Stevenson L. Nutritional challenges and health implications of takeaway and fast food. Nutr Rev. 2013;71(5):310–8. [DOI] [PubMed] [Google Scholar]
- 52.Hur SJ, Yoon Y, Jo C, Jeong JY, Lee KT. Effect of Dietary Red Meat on Colorectal Cancer Risk—A Review. Compr Rev Food Sci Food Saf. 2019;18(6):1812–24. [DOI] [PubMed] [Google Scholar]
- 53.Hodge AM, Williamson EJ, Bassett JK, MacInnis RJ, Giles GG, English DR. Dietary and biomarker estimates of fatty acids and risk of colorectal cancer. Int J Cancer. 2015;137(5):1224–34. [DOI] [PubMed] [Google Scholar]
- 54.Osadnik T, Pawlas N, Lejawa M, Lisik M, Osadnik K, Fronczek M, Bujak K, Gąsior M. Genetic and environmental factors associated with homocysteine concentrations in a population of healthy young adults. Analysis of the MAGNETIC study. Nutr Metabolism Cardiovasc Dis. 2020;30(6):939–47. [DOI] [PubMed] [Google Scholar]
- 55.Shiao SPK, Lie A, Yu CH. Meta-analysis of homocysteine-related factors on the risk of colorectal cancer. Oncotarget. 2018;9(39):25681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chiang F-F, Wang H-M, Lan Y-C, Yang M-H, Huang S-C, Huang Y-C. High homocysteine is associated with increased risk of colorectal cancer independently of oxidative stress and antioxidant capacities. Clin Nutr. 2014;33(6):1054–60. [DOI] [PubMed] [Google Scholar]
- 57.Lonnie M, Wadolowska L, Morze J, Bandurska-Stankiewicz E. Associations of Dietary-Lifestyle Patterns with Obesity and Metabolic Health: Two-Year Changes in MeDiSH(®) Study Cohort. Int J Environ Res Public Health 2022, 19(20). [DOI] [PMC free article] [PubMed]
- 58.Shen X, Wang Y, Zhao R, Wan Q, Wu Y, Zhao L, Wu X. Metabolic syndrome and the risk of colorectal cancer: a systematic review and meta-analysis. Int J Colorectal Dis. 2021;36(10):2215–25. [DOI] [PubMed] [Google Scholar]
- 59.Long AG, Lundsmith ET, Hamilton KE. Inflammation and colorectal cancer. Curr Colorectal cancer Rep. 2017;13:341–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Friis S, Riis AH, Erichsen R, Baron JA, Sørensen HT. Low-dose aspirin or nonsteroidal anti-inflammatory drug use and colorectal cancer risk: a population-based, case–control study. Ann Intern Med. 2015;163(5):347–55. [DOI] [PubMed] [Google Scholar]
- 61.Wang X, Huycke MM. Extracellular superoxide production by Enterococcus faecalis promotes chromosomal instability in mammalian cells. Gastroenterology. 2007;132(2):551–61. [DOI] [PubMed] [Google Scholar]
- 62.Mira-Pascual L, Cabrera-Rubio R, Ocon S, Costales P, Parra A, Suarez A, Moris F, Rodrigo L, Mira A, Collado M. Microbial mucosal colonic shifts associated with the development of colorectal cancer reveal the presence of different bacterial and archaeal biomarkers. J Gastroenterol. 2015;50:167–79. [DOI] [PubMed] [Google Scholar]
- 63.Ivanov II, de Llanos Frutos R, Manel N, Yoshinaga K, Rifkin DB, Sartor RB, Finlay BB, Littman DR. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe. 2008;4(4):337–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Hart MJ, Torres SJ, McNaughton SA, Milte CM. Dietary patterns and associations with biomarkers of inflammation in adults: a systematic review of observational studies. Nutr J. 2021;20(1):1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rinninella E, Cintoni M, Raoul P, Lopetuso LR, Scaldaferri F, Pulcini G, Miggiano GAD, Gasbarrini A, Mele MC. Food components and dietary habits: keys for a healthy gut microbiota composition. Nutrients. 2019;11(10):2393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kong LC, Holmes BA, Cotillard A, Habi-Rachedi F, Brazeilles R, Gougis S, Gausserès N, Cani PD, Fellahi S, Bastard J-P. Dietary patterns differently associate with inflammation and gut microbiota in overweight and obese subjects. PLoS ONE. 2014;9(10):e109434. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.