Abstract
Background
Congenitally missing permanent canines (CMPC) have a significant impact on function and aesthetics. The prevalence is low, and the availability of rigorous studies on this issue in China is limited. This study was designed to investigate the prevalence and distribution of CMPC in patients and concomitant anomalies. Additionally, we aimed to compare the prevalence of CMPC between two age groups.
Methods
A total of 10,447 patient’s panoramic radiograph (PR) images from August 2021 to December 2023 were reviewed and data regarding the location and number of CMPCs were recorded. Age and gender were obtained from the clinical records. Additionally, concomitant anomalies such as persistent primary canines, congenitally missing other permanent teeth, supernumerary teeth and microdontia were also recorded in patients with CMPC. 10,447 patients were divided into two groups: group A (< 121 months of age) and group B (≥ 121 months of age) to record the number of patients with CMPC. The data were presented as counts (n) and percentages (%), and statistically analysed using the Chi-square test.
Results
The average age of patients with CMPC was 9.2 years, with a prevalence of 0.69% (n = 72). The difference between genders was not statistically significant (p = 0.950). The prevalence of CMPC in group B (1.08%, 26/2400) was significantly greater than that in group A (0.57%, 46/8047) (p = 0.008). 89.42% (93/104) of the missing permanent canines had visible primary canines in patients. CMPC were more likely to occur in the maxilla (91.67%) than in the mandible (6.94%) (p<0.001). No significant difference was found between the left and right sides (p = 0.844). With respect to concomitant anomalies among those with CMPC, 22 patients had 58 congenitally other permanent teeth missing, 3 patients had 3 supernumerary teeth, and 12 maxillary lateral incisors from 7 patients presented with microdontia.
Conclusions
The prevalence of CMPC was 0.69%. CMPC were more likely to occur in the maxilla in the presence of persistent primary canines and were not influenced by gender or side. Early panoramic radiographs can improve the detection rate of CMPC, thereby facilitating prompt intervention and timely management.
Keywords: Congenitally missing teeth, Permanent teeth, Canines, Panoramic radiograph
Background
Congenitally missing teeth, which refer to teeth that fail to develop or form during the process of odontogenesis [1], have a prevalence ranging from 4.4 to 13.4% [2]. These dental anomalies can be divided into isolated and syndromic types on the basis of their association with systemic diseases [1]. Research on this issue is crucial as it can provide more direct guidance to improve patients’ oral health and promote standardized treatment approaches.
Canines, commonly known as “fang teeth” or “eye teeth”, are located at the front corners of the mouth and have relatively thick roots, the longest length, and the longest retention time in the oral cavity, serving functions such as tearing food, guiding occlusion, and providing support to facial soft tissue. Permanent canines can be used as abutments and play an important role in prosthodontic treatment. Congenitally missing permanent canines (CMPC) have a significant impact on function and aesthetics [3]. Patients without complaints at the time of presentation usually have persistent primary canines that mask the problem [3]. CMPC can lead to several developmental abnormalities in the maxillary bone, including hypoplasia and asymmetry. This condition often results in insufficient support for facial soft tissues, which may cause facial collapse and contribute to an aged appearance. Additionally, CMPC are associated with various malocclusion conditions, such as dental crowding, altered occlusion, and functional impairments. Furthermore, it may induce resorption and morphological alterations in the alveolar bone of the affected area.
The etiology of CMPC is multifactorial, and involves genetic regulation and environmental factors, with the former playing a more important role [3, 4]. Genetic influence often appears as a familial trait involving multiple genes such as PAX9, MSX1, AXIN2, and EDA, which play roles in signalling pathways and signal transduction cascades [3]. Environmental factors, including early childhood radiation therapy, maternal rubella during pregnancy, and thalidomide exposure, can lead to tooth agenesis [3].
The reported prevalence of CMPC varies widely, such as 0.04 ~ 0.14% in America [5], 0.18% in Japan [6], 0.23% in Sweden [7], 0.29% in Hungary [8], 0.45% in Hong Kong, China [9], 0.51% in China [10], and 0.76% in Israel [11]. CMPC are rare and existing studies on this topic are predominantly case reports, with limited large sample studies [6, 10, 12], particularly in China. Previous studies often combined data from both children and adults in their analysis [6, 11], which may obscure age-specific trends. Furthermore, the complaints differed between patients who visited before and after the average age of canine eruption, suggesting that the prevalence of CMPC may vary between different age groups.
Dental anomalies are associated with CMPC [17]. Microdontia often appear conical and deviate from normal tooth morphology. Research indicates that mutations in genes responsible for tooth germ development and size, such as PAX9 and MSX1, can lead to congenital tooth absence or microdontia.
This retrospective study aimed to investigate the prevalence and distribution of CMPC using PR among Chinese outpatient population under 18 years of age, and to compare the differences in the prevalence of CMPC between the two different age groups, and genders. Additionally, the presence of concomitant dental anomalies such as persistent primary canines, congenitally missing other permanent teeth, microdontia were analysed.
Methods
Sample size calculation
To investigate the prevalence of CMPC, the sample size estimation in this study was calculated using the formula  , where n represents the required sample size, α is the significance level set at 0.05, δ is the allowable error set at 0.0015, and π is the prevalence of CMPC.A literature [10] revealed that the domestic prevalence of CMPC was 0.51%. When the formula, a sample size of 8664 individuals was needed. Considering a 10% dropout rate, a minimum of 9627 individuals needed to be examined.
, where n represents the required sample size, α is the significance level set at 0.05, δ is the allowable error set at 0.0015, and π is the prevalence of CMPC.A literature [10] revealed that the domestic prevalence of CMPC was 0.51%. When the formula, a sample size of 8664 individuals was needed. Considering a 10% dropout rate, a minimum of 9627 individuals needed to be examined.
Study subjects
PR images of a total of 10,447 patients admitted to outpatient clinics at Beijing Children’s Hospital in China during the period from August 2021 to December 2023 were selected for the study, without consideration of gender. Instances of repeated visits by the same patient were counted as a single case. Panoramic radiographs were obtained using panoramic Cranex D (Soredex, Tuusula, Finland) operating at 70 kV, 14 mA and an exposure time of 12 s.
The inclusion criteria were patients of Chinese origin aged ≤ 18 years, and good quality PR images devoid of distortion, facilitating the identification of patients with missing teeth. In cases where documentation was lacking, guardians were contacted by phone to ascertain the cause of the missing permanent canine. The exclusion criteria were as follows: incomplete information; history of permanent canine extraction for orthodontic reasons, trauma, jaw cysts or other pathologic lesions; syndromes or fusion between the permanent lateral incisor and canine; and distorted or blurred PR images.
Procedures
Two experienced clinicians reviewed the 10,447 panoramic radiograph images and made the diagnoses of CMPCs. If the two clinicians were consistent with the diagnoses of congenitally missing permanent canine, then the diagnosis was accepted; if not, a third well-experienced clinician was consulted to make a diagnosis which was then recorded. If the diagnoses of the three clinicians were not consistent, they consulted together to reach a consensus or the case was excluded.
Microdontia was typically diagnosed by two clinicians, with any disagreements resolved by a third experienced clinician. The diagnostic criteria include a tooth that is smaller than normal, deviates from the normal tooth morphology, and exhibits a tapering coronal form, where the incisal mesio-distal width of the crown is narrower than the cervical width [13].
Data collection
For patients diagnosed with CMPC, the age, gender, location and number of missing permanent canines were recorded. Additionally, the number of missing primary canines, the location and numbers of congenitally missing other permanent teeth and the presence of concomitant anomalies such as persistent primary canines, supernumerary teeth and microdontia were recorded.
Based on the average age of permanent canine eruption, all the study PR images of patients were divided into two groups based on age: group A (< 121 months of age) and group B (≥ 121 months of age). The number of patients with CMPC in the two groups was recorded.
Statistical analysis
The qualitative data were presented as counts (percentages) and were analysed using the χ² test in SPSS (SPSS Inc. Chicago, IL) (P < 0.05).
Results
Prevalence and distribution of congenitally missing permanent canines
Of 10,447 patients (5842 males and 4605 females), resulting in a male-to-female ratio of 1.27:1, the overall prevalence of CMPC was 0.69% (72/10447), with an average age of 9.2 years. Among the 72 patients, 40 males and 32 females were documented (male-to-female ratio 1.25:1). The prevalence of CMPC was lower in males (0.68%; 40/5842) than in females (0.69%; 32/4605), but the difference was not statistically significant (χ²=0.004, p = 0.950) (Table 1). 66 patients (91.67%) had maxillary permanent canines affected, 5 patients (6.94%) had mandibular canines affected, and 1 patient (1.39%) had both maxillary and mandibular canines affected (Table 2). The prevalence of CMPC in group B (1.08%, 26/2400) was significantly greater than that in group A (0.57%, 46/8047) (χ²=7.072, p = 0.008) (Table 3).
Table 1.
Gender distribution of patients with congenitally missing permanent canines
| Male | Female | Total | |
|---|---|---|---|
| canines congenitally missing | 40(0.68%) | 32(0.69%) | 72(0.69%) |
| canines normal | 5802(99.32%) | 4573(99.31%) | 10,375(99.31%) |
| 5842(55.92%) | 4605(44.08%) | 10,447 |
Table 2.
Distribution of congenitally missing permanent canines based on the jaws
| Male | Female | Total | |
|---|---|---|---|
| Maxilla | 36(50%) | 30(41.67%) | 66(91.67%) |
| Mandible | 3(4.17%) | 2(2.78%) | 5(6.94%) |
| Maxilla and Mandible | 1(1.39%) | 0 | 1(1.39%) |
| Total | 40(55.56%) | 32(44.44%) | 72 |
Table 3.
Age distribution of patients with congenitally missing permanent canines
| Age group (m) | congenitally missing canines | Total | χ² |
|---|---|---|---|
| <121 m | 46(0.57%) | 8047(77.03%) | 7.072 |
| ≥ 121 m | 26(1.08%) | 2400(22.97%) | |
| Total | 72(0.69%) | 10,447 |
A total of 104 congenitally missing permanent canines were found (96 in the maxilla, 8 in the mandible). CMPC were significantly more likely to occur in the maxilla (χ²=74.647, p < 0.001). 53 permanent canines were congenitally missing on the left side and 51 were missing on the right side, showing no statistically significant difference (χ²=0.039, p = 0.844) (Table 4).
Table 4.
Distribution of congenitally missing permanent canines based on the number of teeth
| Location | Male | Female | total | |
|---|---|---|---|---|
|
1 canine congenitally missing |
Right Maxilla Left Maxilla Left Mandible Right Mandible |
9(8.65%) 9(8.65%) 2(1.92%) 1(0.96%) |
10(9.62%) 9(8.65%) 0 0 |
19(18.27%) 18(17.31%) 2(1.92%) 1(0.96%) |
| Total | 21(20.19%) | 19(18.27%) | 40(38.46%) | |
|
2 canines congenitally missing |
Right Maxilla Left Maxilla Left Mandible Right Mandible |
19(18.27%) 18(17.31%) 1(0.96%) 2(1.92%) |
11(10.58%) 11(10.58%) 1(0.96%) 1(0.96%) |
30(28.85%) 29(27.89%) 2(1.92%) |
| 3(2.88%) | ||||
| Total | 40(38.46%) | 24(23.08%) | 64(61.54%) | |
| Total | 61(58.65%) | 43(41.35%) | 104 |
The number of congenitally missing permanent canines
One permanent canine was congenitally missing for 40 patients, accounting for 55.56% (40/72) (Fig. 1). 32 patients (44.44%, 32/72) had two congenitally missing permanent canines, with all but one exhibiting bilateral absence (Figs. 2, 3 and 4). Cases with three or four congenitally missing permanent canines were not found.
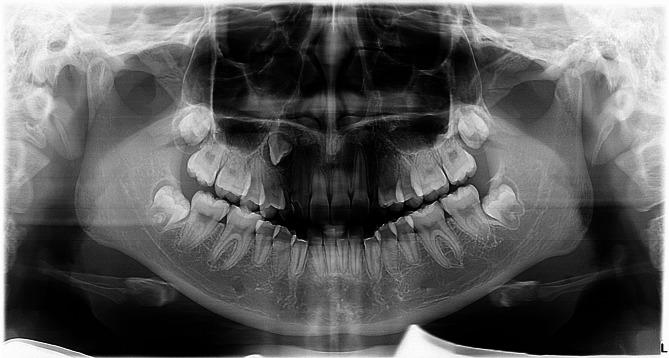
Fig. 1.
Panoramic radiograph of a 13-year-old female showing congenitally missing left maxillary permanent canine, impacted right maxillary permanent canine and bilateral persistent primary canines with root resorption
Fig. 2.
Panoramic radiograph of a 12-year-old female showing bilateral congenitally missing maxillary permanent canines with persistent primary canines showing root resorption, and bilateral microdontia of maxillary lateral incisors
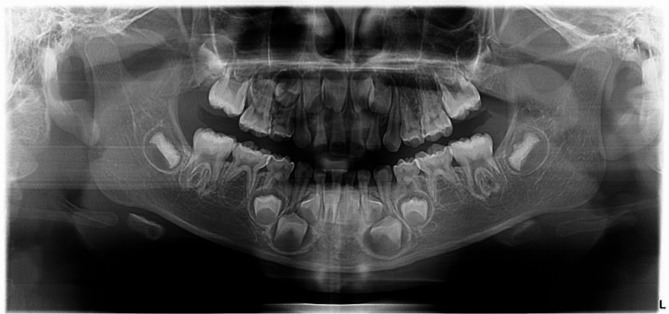
Fig. 3.
Panoramic radiograph of an 8-year-old male showing evidence of bilateral congenitally missing mandibular permanent canines
Fig. 4.
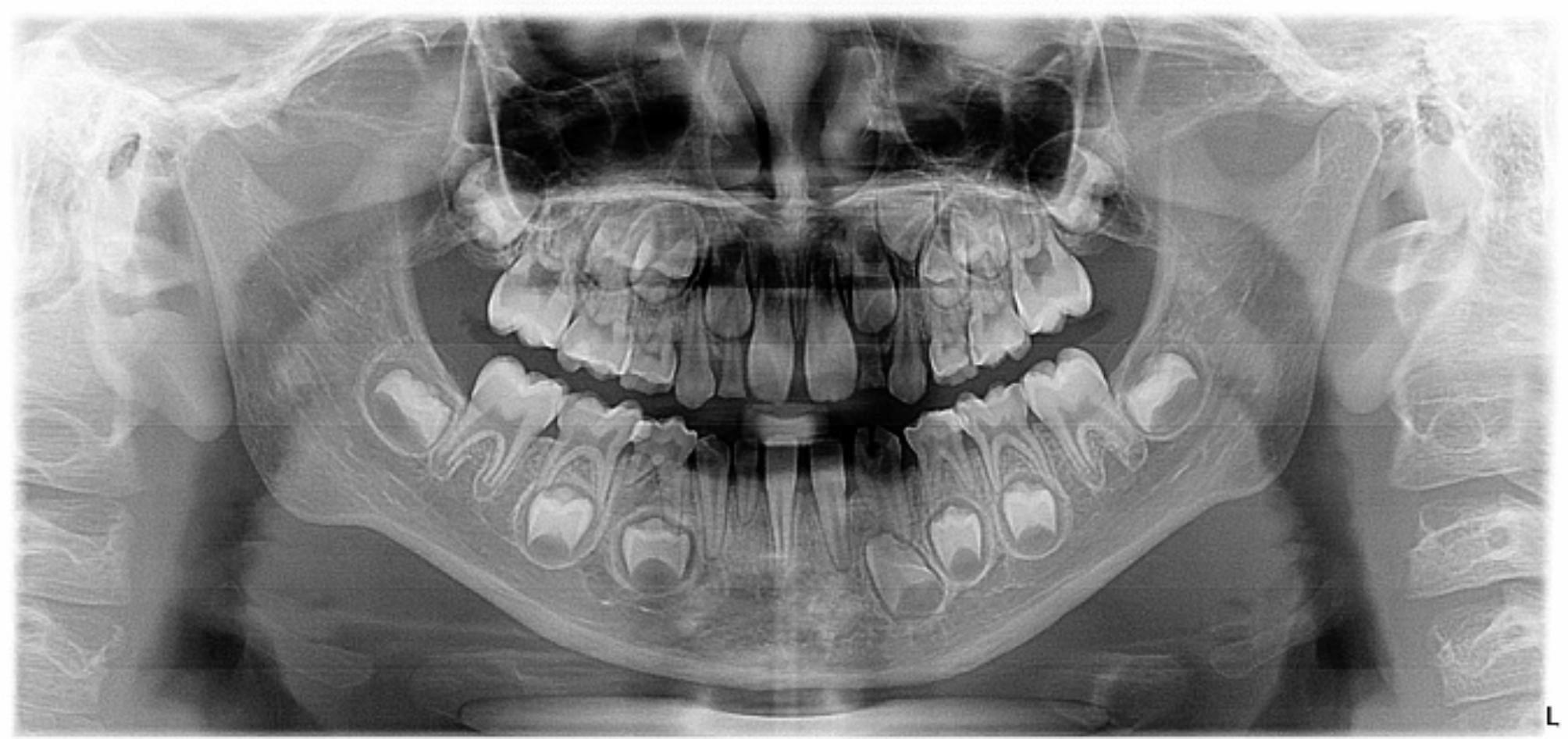
Panoramic radiograph of a 9-year-old male showing evidence of congenitally missing right maxillary and mandibular permanent canines, and mandibular permanent lateral incisors
In this study, 104 congenitally missing canines were observed. Of these, 93 (89.42% ,93/104) primary canines were present, while 11 were missing.
Congenitally missing permanent canines and other concomitant dental anomalies
Among patients with CMPC, 58.33% (42/72) exhibited only permanent canine loss, with no other dental anomalies, excluding persistent primary canines, whereas 30.56% (22/72) had congenitally missing other permanent teeth. Among patients with CMPC, a total of 58 other non-canine permanent teeth were missing. Among the 58 missing teeth, 39.66% (23/58) were associated with the absence of second premolars, followed by 25.86% (15/58) with first premolars (Fig. 5), 22.41% (13/58) with lateral incisors, and 12.07% (7/58) with mandibular central incisors. 3 patients presented with congenital absence of maxillary permanent canines and supernumerary teeth (Fig. 6) 0.12 maxillary permanent lateral incisors exhibited microdontia in 7 patients, and impacted permanent canines could also be observed (Fig. 1) (Table 5).
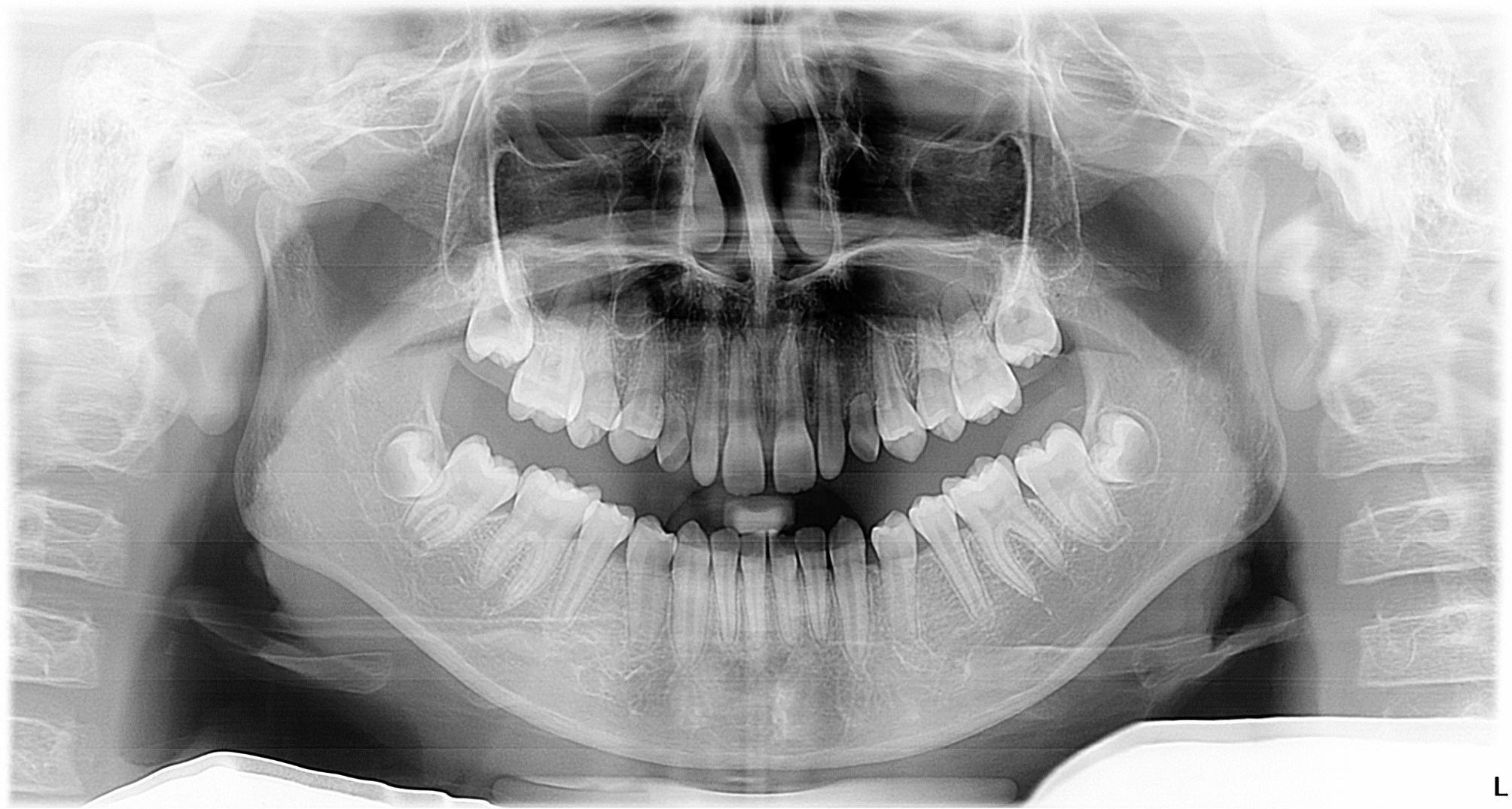
Fig. 5.
Panoramic radiograph of a 5-year-old male showing congenital absence of bilateral maxillary permanent canines, bilateral mandibular and left maxillary second premolars, and left maxillary first premolar
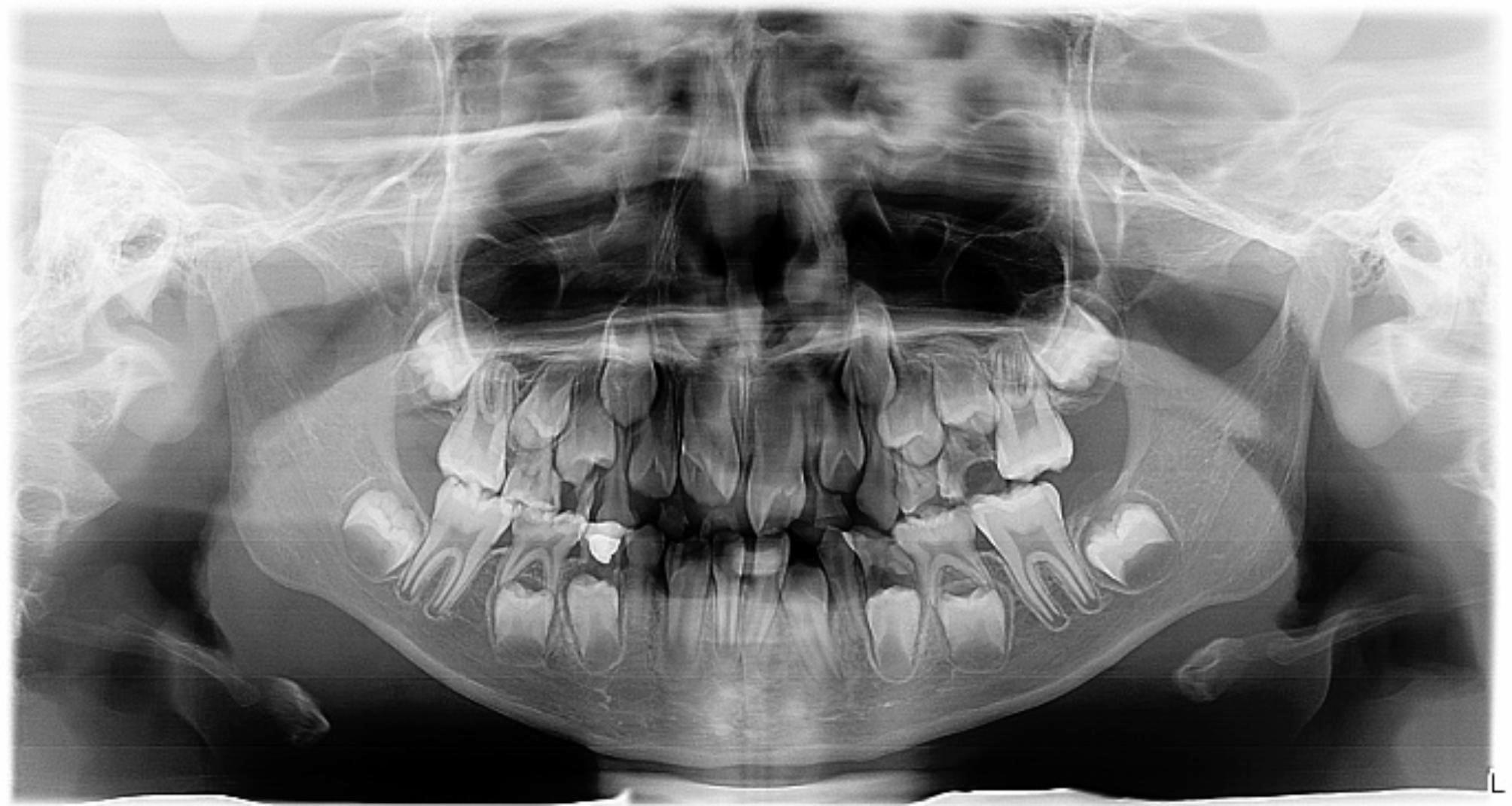
Fig. 6.
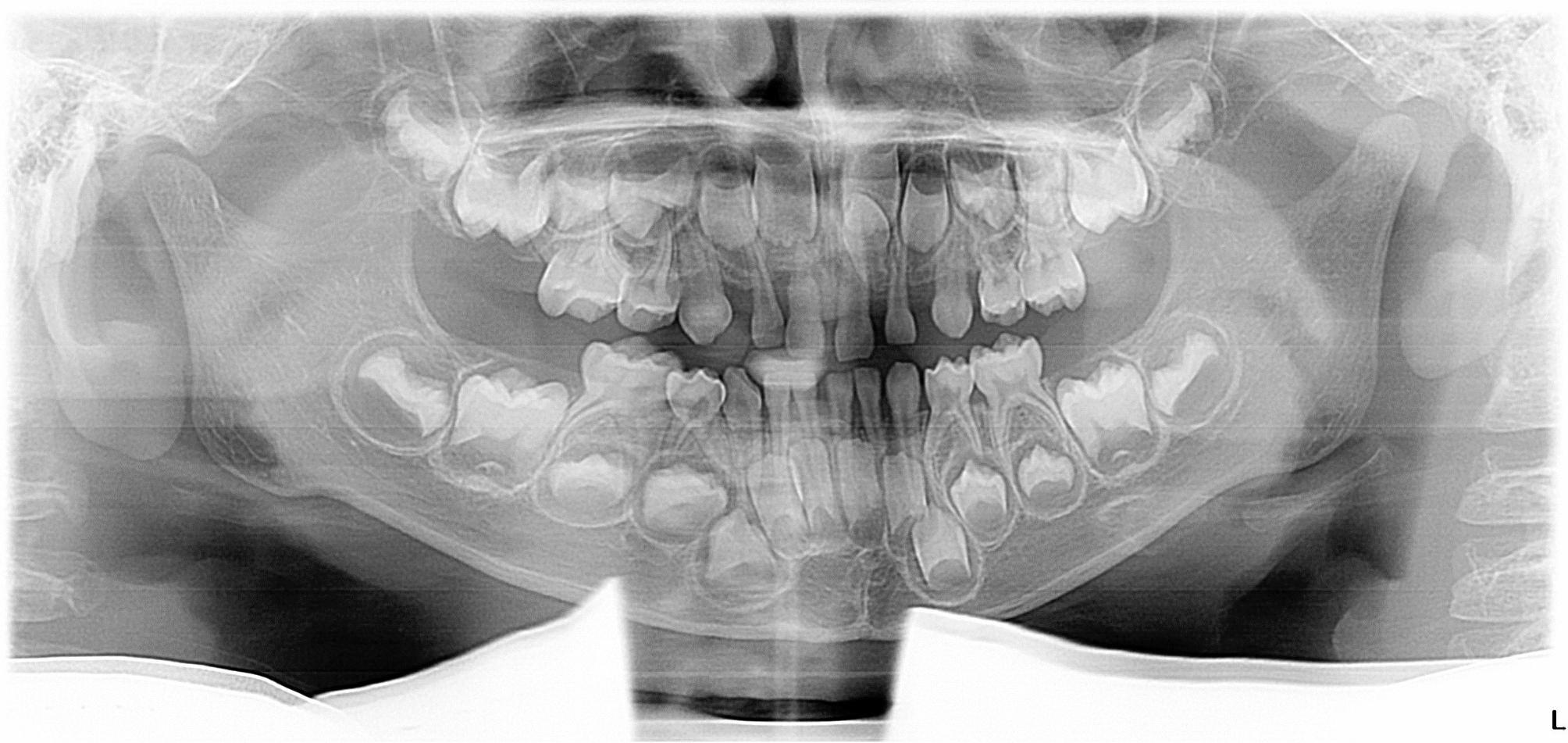
Panoramic radiograph of a 4-year-old male showing bilateral congenital absence of maxillary permanent canines, and evidence of a supernumerary tooth in the maxillary incisor region
Table 5.
Congenital anomalies associated with congenitally missing permanent canines
| Congenital anomalies | Tooth | no. |
|---|---|---|
| Congenitally missing permanent teeth (excluding third molars) | 35.45 | 17 |
|
14.24 15.25 31.41 32.42 12.22 34.44 |
11 6 7 5 8 4 |
|
| Primary canines visible | 93 | |
| Supernumerary teeth | 11.21 | 3 |
| Microdontia of upper lateral incisors | 12.22 | 12 |
Teeth identified using the FDI two-digit system
Discussion
This study focused on isolated congenitally missing permanent canines, a complex condition influenced by polygenic inheritance, as well as environmental and epigenetic factors. The genetic patterns of congenital tooth agenesis may include autosomal dominant, autosomal recessive, or X-linked inheritance [14]. Research [15] indicated that EDARADD (c.308 C > T, p.Ser103Phe) and COL5A1 (c.1588G > A, p.Gly530Ser) were specifically associated with canine agenesis. A study [16] on a family with congenital maxillary canine agenesis identified ITGAV as a potential pathogenic gene. Whilst, some studies suggested that the absence of maxillary canines were associated with WNT10A gene [17–20].
In this study, the overall prevalence of CMPC was 0.69%. In the younger age group, the prevalence of CMPC was 0.57%, aligning closely with findings of Qiu (0.51%) [10] and Davis (0.45%) [9] in China. A Study [11] has also reported CMPC in the orthodontic population in Israel, with a prevalence of 0.76%. It is speculated that racial factors significantly influence this process. Research findings [21] indicated that racial differences exist in the occurrence of congenitally missing teeth. Genetic processes such as genetic drift, gene flow, and natural selection lead to variations in genomic compositions among different races and populations [22].The variations from other studies may also be attributed to differences in regional populations, sample sizes, and sample selection. Samples from specialist hospitals were more likely to detect missing teeth. Different complaints for dental visits before and after the expected eruption age of permanent canines should be considered. Younger children were less likely to have chief complaint of non-eruption of canines, which could better indicate the true prevalence of CMPC. Research [23] has shown that mandibular canines erupt at approximately 9 years of age, whereas maxillary canines erupt at approximately 10 years of age, with an average of approximately 10.1 years or 121 months. This study categorized participants into two groups based on the age of 121 months. The significantly greater prevalence of CMPC in older children(1.08%), which is twice that of younger children(0.57%), may be attributed to parents observing no eruption of their children’s canines and prompting them to seek medical attention. The result in older age group (1.08%) was similar to those(0.76%) in orthodontic patients reported by Finkelstein [11].
Previous research has indicated a predominance of females in cases of Hypodontia [3]. Whilst, this study demonstrated no gender predominance for CMPC, aligning with earlier findings [10]. Nonetheless, a study suggested that women were more affected by CMPC and that CMPC differed from hypodontia, exhibiting different gender tendencies [6].
No significant difference was found in the prevalence of CMPC between the left and right sides, which is consistent with previous research [10]. CMPC are more frequently found in the maxilla, which is consistent with the findings of previous studies [5–7, 9, 11, 12]. Some studies have reported [6, 7] a greater prevalence of congenitally missing left maxillary canines. Cleft lip and alveolar anomalies are more likely to affect the maxilla and may be associated with dental abnormalities [21]. It’s suspected that CMPC were more prevalent in the maxilla, likely for similar reasons. However, right mandibular dominance has been reported in CMPC, which remains unexplained [6]. In this study, of the 4 quadrants, maxillary canines were more commonly affected than mandibular canines, with no other similar site dominance observed.
CMPC typically involved the absence of one or two permanent canines with persistent primary canines, with no cases exceeding the absence of three found in this study. However, previous studies have reported cases in which three to four permanent canines were congenitally missing [6, 10]. This discrepancy may be attributed to the extremely low prevalence of congenitally missing mandibular permanent canines. This study found that 89.42% (93/104) of the missing permanent canines had visible primary canines in patients with CMPC, aligning with Qiu’s research [10]. The absence of permanent successors delayed normal resorption of primary tooth roots, resulting in primary teeth being retained for 40 or 50 years [3].
The presence of associations among various tooth anomalies is significant in clinical practice [24], as the early detection of one anomaly may suggest an increased risk for additional anomalies [25]. A total of 58.33% of patients with CMPC had no other dental abnormalities. There was a suspicion that CMPC demonstrated a tendency toward isolated absence. Among 58 accompanying permanent tooth absence, second premolars (39.66%) were most observed, followed by first premolars (25.86%) and lateral incisors (22.41%). These characteristics were consistent with those observed when these anomalies occur independently. 7 patients with CMPC exhibited microdontia of maxillary lateral incisors. Microdontia and hypodontia exhibit significant and intimate genetic associations [26]. Additionally, a rare combination of abnormal tooth numbers was observed in 3 patients with CMPC, who also presented with supernumerary teeth in the maxilla. Disturbances in the differentiation, migration and proliferation of neural crest cells are associated with interactions between epithelial and mesenchymal cells during odontogenesis initiation and may be responsible for “concomitant hypo-hyperdontia” [27, 28].
In multistage restorative therapy for CMPC, many factors should be considered, such as the early diagnosis of agenesis, malocclusion and the facial skeleton. Ephraim [29] et al. emphasized the impact of the congenital absence of teeth on developing dentition, underscoring the significance of early diagnosis of mixed dentition to prevent malocclusion from evolving. Primary canines, when not replaced by their permanent successors, usually exhibit minimal or no root resorption, which helps preserve the alveolar bone for future prosthetic rehabilitation [30]. Therefore, preservation of primary canines is recommended whenever feasible. However, despite their prolonged retention time, primary canines still face the issues of root resorption and tooth loss. Consulting an orthodontist and restorative dentist is necessary to prevent difficulties caused by space loss and jaw atrophy resulting from premature primary canine loss in the developing dentition. To optimize the restoration process, clinicians should focus on minimizing or consolidating the edentulous space and reducing the required number of implants. For instance, the consideration of guided eruption and orthodontic treatment for premolar substitution in cases where permanent maxillary canines are missing can be beneficial [11, 31, 32]. One study reported the successful replacement of primary canines with implants [33]. Mini-implants can be utilized for the temporary restoration of missing permanent teeth in adolescent patients [32, 34]. This alternative approach facilitates vertical development of the alveolar process and maintains bone density and alveolar morphology, obviating the need for additional surgical procedures for dental implantation and leading to favourable long-term outcomes.
One limitation of this study was that it might not accurately represent the prevalence and distribution of CMPC among the Chinese population; hence, the generalizability of the results is limited. To validate the prevalence observed in this study, multicenter research utilizing electronic health records (EHR) systems or other comprehensive medical databases is recommended. Additionally, long-term follow-up to assess the absence of second permanent molars and other dental anomalies, such as taurodontism is recommended. Further genetic investigations into the pathogenic genes associated with CMPC in affected patients may provide additional insights.
Conclusions
This study provides valuable data on CMPC and their co-occurrence with other dental anomalies in the Chinese population. The prevalence of CMPC was 0.69%. CMPC were more likely to occur in the maxilla with persistent primary canines, showing no gender or side trends. Early panoramic radiograph is recommended to facilitate early diagnosis, intervention and referral for treatment, thereby minimizing or preventing the functional and aesthetic complications associated with CMPC.
Acknowledgements
Not applicable.
Abbreviations
- PR
Panoramic radiograph
- CMPC
Congenitally missing permanent canines
Author contributions
SL: study conception, study design, data collection and analysis, manuscript writing and editing. YL: data collection and analyzation. GY: project administration, supervision, manuscript review & editing. All Authors reviewed and approved the final manuscript.
Funding
There was no funding received for this study.
Data availability
All data generated or analysed during this study are included in this published article.
Declarations
Ethics approval and consent to participate
This study, conducted in adherence to the Declaration of Helsinki, was approved by the Ethics Committee of Beijing Children’s Hospital, Capital Medical University ([2024]-E-025-R). The Beijing Children’s Hospital Ethics Committee provided an exemption from obtaining informed consent from the subjects.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Aslam K, Jabeen S, Jafri SS, Saeed A, Anjum I. The molecular genetics of selective tooth agenesis. J Pak Med Assoc. 2020;70(11):2023–7. [DOI] [PubMed] [Google Scholar]
- 2.Khalaf K, Miskelly J, Voge E, Macfarlane TV. Prevalence of hypodontia and associated factors: a systematic review and meta-analysis. J Orthod. 2014;41(4):299–316. [DOI] [PubMed] [Google Scholar]
- 3.Al-Ani AH, Antoun JS, Thomson WM, Merriman TR, Farella M. Hypodontia: An Update on Its Etiology, Classification, and Clinical Management. BioMed research international 2017, 2017:9378325. [DOI] [PMC free article] [PubMed]
- 4.Choi SJ, Lee JW, Song JH. Dental anomaly patterns associated with tooth agenesis. Acta Odontol Scand. 2017;75(3):161–5. [DOI] [PubMed] [Google Scholar]
- 5.Muller TP, Hill IN, Peterson AC, Blayney JR. A survey of congenitally missing permanent teeth. Journal of the American Dental Association (1939) 1970, 81(1):101–107. [DOI] [PubMed]
- 6.Fukuta Y, Totsuka M, Takeda Y, Yamamoto H. Congenital absence of the permanent canines: a clinico-statistical study. J Oral Sci. 2004;46(4):247–52. [DOI] [PubMed] [Google Scholar]
- 7.Bergström K. An orthopantomographic study of hypodontia, supernumeraries and other anomalies in school children between the ages of 8–9 years. An epidemiological study. Swed Dent J. 1977;1(4):145–57. [PubMed] [Google Scholar]
- 8.Rozsa N, Nagy K, Vajo Z, Gabris K, Soos A, Alberth M, Tarjan I. Prevalence and distribution of permanent canine agenesis in dental paediatric and orthodontic patients in Hungary. Eur J Orthod. 2009;31(4):374–9. [DOI] [PubMed] [Google Scholar]
- 9.Davis PJ. Hypodontia and hyperdontia of permanent teeth in Hong Kong schoolchildren. Commun Dent Oral Epidemiol. 1987;15(4):218–20. [DOI] [PubMed] [Google Scholar]
- 10.Fenfang Q, Shan M, Wenwen H. Clinical analysis of non-syndromic congenital permanent canines agenesis in children. J Oral Sci Res 2022(07):632–5.
- 11.Finkelstein T, Shapira Y, Pavlidi AM, Schonberger S, Shpack N. Agenesis of Permanent canines in Orthodontic patients: Prevalence, Location, Treatment options and outcomes. J Dentistry Child (Chicago Ill). 2018;85(3):133–8. [PubMed] [Google Scholar]
- 12.Rose JS. A survey of congenitally missing teeth, excluding third molars, in 6000 orthodontic patients. Dent Practitioner Dent Record. 1966;17(3):107–14. [PubMed] [Google Scholar]
- 13.Backman B, Wahlin YB. Variations in number and morphology of permanent teeth in 7-year-old Swedish children. Int J Pediatr Dent. 2001;11(1):11–7. [DOI] [PubMed] [Google Scholar]
- 14.Abid MF, Simpson MA, Petridis C, Cobourne MT, Sharpe PT. Non-syndromic severe hypodontia caused by a novel frameshift insertion mutation in the homeobox of the MSX1 gene. Arch Oral Biol. 2017;75:8–13. [DOI] [PubMed] [Google Scholar]
- 15.Barbato E, Traversa A, Guarnieri R, Giovannetti A, Genovesi ML, Magliozzi MR, Paolacci S, Ciolfi A, Pizzi S, Giorgio RD, et al. Whole exome sequencing in an Italian family with isolated maxillary canine agenesis and canine eruption anomalies. Arch Oral Biol. 2018;91:96–102. [DOI] [PubMed] [Google Scholar]
- 16.Fan Y, Siyue Y, Xin Y, Liwen F, Yongchu P, Lin W. Identification of pathogenic genes in a canine agenesis family. Stomatology 2020(06):513–6.
- 17.Kantaputra P, Kaewgahya M, Kantaputra W. WNT10A mutations also Associated with Agenesis of the Maxillary Permanent canines, a separate entity. Am J Med Genet Part A. 2014;164(2):360–3. [DOI] [PubMed] [Google Scholar]
- 18.Park H, Song JS, Shin TJ, Hyun HK, Kim YJ, Kim JW. WNT10A mutations causing oligodontia. Arch Oral Biol. 2019;103:8–11. [DOI] [PubMed] [Google Scholar]
- 19.Zeng Y, Baugh E, Akyalcin S, Letra A. Functional effects of WNT10A Rare variants Associated with Tooth Agenesis. J Dent Res. 2021;100(3):302–9. [DOI] [PubMed] [Google Scholar]
- 20.Živković M, Stefanović N, Glišić B, Brajović G, Miličić B, Kostić M, Popović B. WNT10A and RUNX2 mutations associated with non-syndromic tooth agenesis. Eur J Oral Sci. 2022;130(6):e12896. [DOI] [PubMed] [Google Scholar]
- 21.Larmour CJ, Mossey PA, Thind BS, Forgie AH, Stirrups DR. Hypodontia–a retrospective review of prevalence and etiology. Part I. Quintessence Int. 2005;36(4):263–70. [PubMed] [Google Scholar]
- 22.Li L, Comi TJ, Bierman RF, Akey JM. Recurrent gene flow between neanderthals and modern humans over the past 200,000 years. Volume 385. New York, NY): Science; 2024. p. eadi1768. 6705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fekonja A. Evaluation of the eruption of permanent teeth and their association with malocclusion. Clin Experimental Dent Res. 2022;8(4):836–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Townsend G, Bockmann M, Hughes T, Brook A. Genetic, environmental and epigenetic influences on variation in human tooth number, size and shape. Odontology. 2012;100(1):1–9. [DOI] [PubMed] [Google Scholar]
- 25.Schonberger S, Kadry R, Shapira Y, Finkelstein T. Permanent tooth agenesis and Associated Dental Anomalies among orthodontically treated children. Child (Basel Switzerland) 2023, 10(3). [DOI] [PMC free article] [PubMed]
- 26.Matošić Ž, Šimunović L, Jukić T, Granić R, Meštrović S. Examining the link between tooth agenesis and papillary thyroid cancer: is there a risk factor? Observational study. Prog Orthodont. 2024;25(1):12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Anthonappa RP, Lee CK, Yiu CKY, King NM. Hypohyperdontia: literature review and report of seven cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontology. 2008;106(5):E24–30. [DOI] [PubMed] [Google Scholar]
- 28.Zadurska M, Sieminska-Piekarczyk B, Maciejak D, Wyszomirska-Zdybel B, Kurol J. Concomitant hypodontia and hyperodontia-An analysis of nine patients. Acta Odontol Scand. 2012;70(2):154–9. [DOI] [PubMed] [Google Scholar]
- 29.Ephraim R, Rajamani T, Feroz TM, Abraham S. Agenesis of multiple primary and permanent teeth unilaterally and its possible management. J Int oral Health : JIOH. 2015;7(5):68–70. [PMC free article] [PubMed] [Google Scholar]
- 30.Laverty DP, Fairbrother K, Addison O. The current evidence on retaining or prosthodontically replacing retained deciduous teeth in the adult Hypodontia patient: a systematic review. Eur J Prosthodont Restor Dent. 2018;26(1):2–15. [DOI] [PubMed] [Google Scholar]
- 31.Borzabadi-Farahani A. Orthodontic considerations in Restorative Management of Hypodontia patients with endosseous implants. J Oral Implantology. 2012;38(6):779–91. [DOI] [PubMed] [Google Scholar]
- 32.Borzabadi-Farahani A. Bilateral agenesis of maxillary permanent canines: review of the literature. J Orthodontic Sci. 2015;4(1):26–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Zuiderveld EG, Meijer HJA, Vissink A, Raghoebar GM. Immediate placement and provisionalization of an implant after removal of an impacted maxillary canine: two case reports. Int J Implant Dentistry. 2015;1(1):13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ciarlantini R, Melsen B. Semipermanent replacement of missing maxillary lateral incisors by mini-implant retained pontics: a follow-up study. Am J Orthod Dentofac Orthop. 2017;151(5):989–94. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data generated or analysed during this study are included in this published article.