Abstract
Ocular drug delivery presents significant challenges, attributed to the various anatomical and physiological barriers, as well as the limitations associated with conventional ocular formulations including low bioavailability, necessitating frequent dosing. The objective of the essay was to design sustained release nanofibrous inserts loaded with ceftazidime (CAZ), an antibiotic effective against gram-negative and gram-positive microorganisms, for the treatment of ocular infections. These nanofibers were fabricated using the electrospinning technique, employing biodegradable polymers such as polyvinyl alcohol (PVA), polycaprolactone (PCL) and Eudragit® (EUD). The nanofibrous inserts exhibited adequate mechanical strength for ocular use with an average diameter < 250 nm. In the initial 12-h period, a burst drug release was observed, followed by a controlled release for 120 h. Cell viability test confirmed the non-toxicity and safety of the nanofibers. The in vivo study demonstrated that the inserts sustain a drug concentration exceeding the minimum inhibitory concentration (MIC) of Pseudomonas aeruginosa and Staphylococcus aureus for 4 and 5 days, respectively. The AUC0–120 for CAZ-PVA-PCL was reported 11,882.81 ± 80.5 μg·h/ml and for CAZ-PVA-EUD was 9649.39 ± 86.84 μg·h/ml. The nanofibrous inserts' extended drug release maintains effective antimicrobial concentrations, avoids the fluctuations of eye drops, and, by being preservative-free, eliminates cytotoxicity.
Keywords: Ocular drug delivery, Nanofibrous inserts, Ocular infection, Sustained delivery, Ceftazidime
Graphical abstract
Highlights
-
•
Ceftazidime-loaded nanofibers were fabricated by electrospinning using polyvinyl alcohol, polycaprolactone, and Eudragit.
-
•
The sustained-release nanofibrous inserts exhibited sufficient mechanical strength for ocular use.
-
•
In vivo study showed that the inserts maintained drug levels above the minimum inhibitory concentration for about 5 days.
-
•
Extended drug release from the preservative-free inserts avoids eye drop fluctuations and eliminates cytotoxicity.
1. Introduction
Ocular infections pose a significant threat to vision and conventional drug delivery methods have not effectively addressed this issue. Pseudomonas aeruginosa is a major pathogen responsible for ocular infection, particularly bacterial keratitis. This type of keratitis, caused by P. aeruginosa, is often more severe at initial diagnosis compared to keratitis caused by other bacterial pathogens. Such cases require more intensive therapeutic intervention and are frequently associated with poorer visual outcomes than keratitis of other etiologies (Sy et al., 2012). P. aeruginosa is also primarily responsible for vision-threatening corneal diseases in otherwise healthy individuals who use contact lenses (Yildiz et al., 2012; Ng et al., 2015).
Ceftazidime (CAZ) is currently the most potent cephalosporin against P. aeruginosa. It functions as an antimicrobial agent by binding to penicillin-binding proteins in bacterial cell walls, thereby preventing the synthesis of new cell walls and inhibiting bacterial growth. CAZ is resistant to various β-lactamases (Chang et al., 2008; Daikos et al., 2021). It is effective against a broad spectrum of bacteria, encompassing both gram-positive and gram-negative species, even those resistant to other antibiotics (Bouattour et al., 2021). However, its primary use is for treating infections caused by gram-negative bacteria (Chen et al., 2021). Despite its efficacy, ceftazidime degrades rapidly in water, leading to a loss of effectiveness. Due to this instability, there are no commercially available eye drops containing ceftazidime in water-based solutions (Wijesooriya et al., 2013).
Conventional drug delivery methods like eye drops face several challenges, including poor drug bioavailability, low drug loading, uncontrolled release of drug molecules, and unsustainable delivery methods. Only a small percentage (1–5 %) of the drug delivered via eye drops reaches the intended eye tissues. This is because the drops are quickly drained from the eye surface and face barriers such as the tear film and corneal epithelium. Eye drops are rapidly washed away by tears and blinking, resulting in minimal contact time with the ocular surface. Additionally, a significant amount of the drug can be absorbed systemically through the nasolacrimal duct, potentially causing side effects and diminishing efficacy at the target site. The need for frequent administration and the discomfort associated with eye drops can lead to poor patient compliance, further reducing the treatment's effectiveness. To address these issues, significant advancements are being made in designing efficient alternative systems for the ophthalmic administration of antimicrobial agents. These innovative approaches include the development of hydrogels (Khalil et al., 2020), nanoparticles (Silva et al., 2017), liposomes (Wijesooriya et al., 2013), contact lenses (Ubani-Ukoma et al., 2019), and nanofibrous inserts (Taghe et al., 2023). While information on novel ocular drug delivery systems for ceftazidime is limited (Wijesooriya et al., 2013; Silva et al., 2017), ongoing research and development efforts are focused on enhancing the delivery of this antibiotic. Various advanced ocular delivery systems, including inserts, are being explored to improve the efficacy and precision of ceftazidime administration.
Sustained release inserts are important because they increase the residence time of the drug in the eye, ensuring that the drug remains effective for a longer period. By maintaining a consistent drug concentration, these inserts enhance the bioavailability of the drug compared to eye drops. They also reduce the frequency of administration, which can substantially influence the drug's concentration in ocular tissue. This reduction in frequency helps to minimize fluctuations, thereby impacting both the drug's efficacy and the incidence of side effects. Additionally, sustained release systems can minimize systemic absorption and local side effects by delivering the drug directly to the target site in a controlled manner. These benefits make sustained release ocular inserts a valuable system in the treatment of ocular diseases (Kumari et al., 2010). Furthermore, these systems offer stability and eliminate the need for preservatives, thereby reducing potential side effects (Rozi and Mohmad Sabere, 2021). Nanofiber-based ocular inserts provide a safe and efficient method for the noninvasive delivery of nanoscale drugs (Omer and Zelkó, 2021).
Electrospinning is an adaptable and economical method used to produce nanoscale fibers from polymer solutions through the application of a strong electric field (Sofi et al., 2020). Nanofibers can absorb moisture and swell when placed in the cul-de-sac, potentially providing prolonged drug retention and sustained release. This can help achieve effective drug levels within the eye. (Chandel and Kandav, 2024; Taghe et al., 2024a). Nanofibers have the advantage of accommodating a significant quantity of drugs and demonstrating high encapsulation efficiency (Luraghi et al., 2021). Moreover, fibers can facilitate drug permeation across physiological barriers, reduce premature drug release, and enable targeted delivery to specific tissues, thereby minimizing drug dispersion to other areas of the body (Zupancic et al., 2016). Various polymers can be utilized for preparing ocular nanofibers including polyvinyl alcohol (PVA), polycaprolactone (PCL) and Eudragit® (EUD). PVA is a water-soluble synthetic polymer known for its excellent mechanical properties, biocompatibility, non-toxicity, and biodegradability. Previous studies have developed PVA-based electrospun fiber containing ketoprofen (Kenawy et al., 2007) and ciprofloxacin (Zhou et al., 2016) with sustained release capabilities. Incorporating hydrophobic polymers like PCL and EUD into blend fibers can enhance the properties of the nanofibrous structure (Mirzaeei et al., 2021). PCL, a biodegradable polyester, was selected for the preparation of nanofibers due to its demonstrated safety, biocompatibility, and non-toxicity. Previous research has shown that PCL-based nanofibers are effective when combined with other natural polymers to enhance scaffold structural integrity and regulate the release of antimicrobial substances (Mohamady Hussein et al., 2021). Studies have also indicated that PCL ocular nanofibers provide sustained drug release in ocular tissues(Da Silva et al., 2019; Taghe et al., 2024a; Taghe et al., 2024c). EUD, FDA-approved polymers derived from methylmethacrylate, offer advantages as biocompatible materials. Researchers have explored EUD RL100 polymeric nanofibers as vehicles for delivering antimicrobial agents such as vancomycin (Abdel-Rahman et al., 2020). These specific polymers (PVA, PCL and EUD) are chosen for their high compatibility with biological tissues, meaning they do not cause adverse reactions when in contact with the body. This is crucial for ocular drug delivery, where the materials must be safe for sensitive eye tissues. These polymers can maintain their structure and function in the biological environment, ensuring that the drug delivery system remains effective over time. The polymers can be engineered to control the rate at which the drug is released, allowing for a steady release of medication over an extended period and reducing the need for frequent dosing. By controlling the release profile, these polymers ensure that the drug reaches the desired site of action in the eye at therapeutic levels, enhancing treatment efficacy and minimizing side effects.
In the present study, ocular CAZ-PVA-EUD and CAZ-PVA-PCL nanofibers were designed and prepared through electrospinning using PVA, PCL and EUD RL100. This innovative approach marks the first instance of designing ceftazidime nanofibers with these specific polymers for ocular drug delivery systems. The formulations were assessed for physiochemical and mechanical properties. Quantification of CAZ in rabbit eyes was performed using a specific animal-friendly method, and cell viability tests were conducted after in vitro characterization. The nanofibrous formulations are expected to offer significant benefits, such as maintaining a consistent drug release rate in the eye and improving pharmacokinetic efficacy, thereby reducing the frequency of dosing.
2. Materials and methods
2.1. Materials
Ceftazidime and EUD RL100 were acquired from Sina Darou (Tehran, Iran) and Evonik Industries (Darmstadt, Germany), respectively. Trypsin, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazoliumbromide (MTT), and modified Dulbecco's Modified Eagle Medium (DMEM) were acquired from Sigma Aldrich (Darmstadt, Germany). Fetal bovine serum (FBS) was obtained from Gibco (Carlsbad, CA, USA). Polycaprolactone (PCL) (Mw = 80 kDa), polyvinyl alcohol (PVA) (99 % hydrolyzed, average Mw = 89–98 kDa), the solutions containing methanol, DCM (dichloromethane), DMF (dimethylformamide), and ACT (acetone), as well as the culture media including Sabouraud Dextrose Broth, Thioglycolate Sodium Liquid, Fluid Casein Digest Medium, and Tryptic Soy Agar, were obtained from Merck (Darmstadt, Germany).
2.2. Synthesis of ceftazidime nanofiber
In order to synthesize ceftazidime nanofibers, a high-voltage direct current (DC) power supply electrospinning device (Fanavaran Nano-Meghyas, Iran) was utilized. Separate solutions were formulated, each with a concentration of 10 % (w/v) consisting of PVA, EUDRL100, and PCL in distilled water, methanol, and a solvent blend of DCM: DMF: ACT in a 4:3:1 (v/v) ratio, at room temperature for 24, 3, and 2 h, respectively with stirring at 1000 rpm to obtain transparent solutions. The PVA solution containing 30 % w/v CAZ was then prepared.
A dual-source, dual-power electrospinning device was used to prepare CAZ-PVA-EUD and CAZ-PVA-PCL. This device comprises of a rotating drum collector covered with aluminum foil. To prepare CAZ-PVA-PCL, two syringes were used, one containing 10 ml of PCL solution without the drug, prepared as previously explained, and another syringe filled with 10 ml of drug-containing PVA solution. The procedure for preparing CAZ-PVA-EUD, was exactly as described above, except instead of PCL solution, one syringe contained 10 ml of EUD solution without the drug. Then, the solutions were simultaneously directed towards the electrospinning collector (8 cm diameter) at a flow rate of 2 ml/h. The entire process took place at a temperature of 25 °C. Aseptic conditions were maintained throughout the entire process of preparing the nanofibrous inserts. Furthermore, the prepared inserts were exposed to UV radiation for 30 min to ensure sterility. Fig. 1 depicts the methodology for fabricating the CAZ nanofibrous inserts.
Fig. 1.

Process of fabrication of ocular nanofibrous inserts.
2.3. Physicochemical properties
2.3.1. Thickness and weight uniformity test
The thickness of the samples was measured using a Tork Craft Digital Micrometer with a range of 0-25 mm (Model ME30025, Quanzhou, China). This was done by preparing nanofibers and measuring them at various points to calculate the average and standard deviation for thickness uniformity assessment. To evaluate the uniformity of weight, pieces from various sections of the prepared nanofiber were weighed using a digital scale.
2.3.2. Folding endurance and tensile strength
The flexibility of the nanofibers was evaluated through a folding endurance test, which measures the number of folds a fiber can withstand at a single point before tearing becomes apparent. For this procedure, the nanofiber, with a specific size of 2 × 2 cm2 and a thickness of around 0.2 mm, was repeatedly folded at the same spot until a tear or crack appeared. The folding endurance was quantified by averaging the number of folds achieved before damage, across three repetitions.
To measure the tensile strength of the nanofiber, a mechanical testing device (STM-5, Santam, Iran) was utilized. Pieces of various nanofibrous inserts were cut, measuring 3 × 1.5 cm2 with a thickness of around 0.2 mm. The samples were pulled at a constant speed of 5 mm/min until they tore. Elongation as flexibility and ultimate stress resistance as tensile strength were examined.
2.3.3. Entrapment efficiency percentage
For the analysis of entrapment efficiency percentage, a specified amount of inserts was stirred in 2 ml of methanol for CAZ-PVA-EUD and in a solvent blend of DCM: DMF: ACT (4:3:1 v/v) for CAZ-PVA-PCL in glass flasks at 100 rpm to remove the EUD and PCL nanofibers from the inserts. The drug-loaded PVA electrospun nanofiber was isolated by centrifuging at a speed of 2000 rpm for a duration of 30 min, separating it from the PCL-containing DCM: DMF: ACT and EUD-containing methanol. Subsequently, drug-loaded PVA electrospun nanofiber underwent immersion in distilled water to quantify the CAZ loaded within the PVA nanofiber. The drug content was measured in three samples, and eventually, the amount of ceftazidime was measured in the solution by HPLC method. The Entrapment efficiency was calculated using Eq. 1.
| (1) |
2.3.4. Swelling percentage, moisture uptake, and moisture loss study
Initially, the weight of the designed nanofibrous samples was recorded. The samples were then soaked in a phosphate buffer solution (PBS) with a pH of 7.4 (physiological pH) and a temperature of 37 °C. After 3, 6, and 12 h, the inserts were extracted, any surplus PBS was removed using a paper, and a subsequent weight measurement was evaluated. The swelling ratio was calculated based on the weight difference before and after immersion, as outlined in Eq. 2.
| (2) |
Pre-weighted inserts were kept in a desiccator filled with anhydrous calcium chloride and aluminum chloride, maintaining a moisture level of 79.5 %, to evaluate the physical stability of the inserts under dry and humid conditions, respectively. After a period of three days, the nanofibers were removed and their weight was measured again. Using Eqs. (3), (4), the percentages of moisture uptake and loss were calculated.
| (3) |
| (4) |
2.3.5. Surface pH Test
To determine the surface pH of the prepared inserts, the fabricated nanofibers were placed in a dish filled with a designated volume of PBS at a pH level of 7.4. Afterward, the surface pH levels of the nanofibers were determined utilizing a pH meter.
2.3.6. Surface hydrophilicity
The measurement of contact angle was performed to characterize surface moisture properties. Surface hydrophilicity properties were characterized using the sessile drop technique. A test set-up for contact angle analysis was arranged using a digital microscope (Dino-Lite, AM7815, AnMo Electronics Corp, Taiwan) set to capture at an interval of 1 frame every 500 milliseconds. Subsequently, distilled deionized water at room temperature was dropped onto the sample surface using a micropipette. The images captured at the moment of contact were examined using ImageJ software to estimate the contact angle and evaluate the hydrophilicity. The average contact angle was calculated from three separate tests (Turanlı and Acartürk, 2021).
2.3.7. Ex-vivo mucoadhesion time
A freshly removed sheep cornea (3 cm2) was cleaned with PBS before being taped to a glass slide (Tofighia et al., 2017). A disintegration apparatus was then filled with a specific amount of PBS (pH = 7.4), and the assembled slide was fixed vertically on it. Additionally, pieces of the inserts (1 cm2) were moistened on one side with PBS, applied to the cornea, and then reciprocated upward and downward at 37 °C. Finally, the mucoadhesion time—the amount of time needed for these inserts to completely separate from the cornea—was reported (n = 3).
2.4. Assessment of ceftazidime using high-performance liquid chromatography (HPLC)
The separation process in high-performance liquid chromatography (HPLC) was conducted utilizing a Knauer-V7603-Germany model linked to a Knauer Smartline 2500 UV detector, which was set to a 255 nm wavelength. The chromatographic analysis was carried out using a column measuring 250 mm in length and 4.6 mm in internal diameter, filled with C18 material (Mainz, Germany) at 25 °C. The mobile phase consisted of a 10:90 v/v% mix of methanol and a triethylamine buffer (0.1 % concentration, pH 3.0), used for separating ceftazidime at a flow rate of 0.7 ml/min over a total run time of 6 min. Samples of the drug were prepared at varying concentrations, from the lowest (3.12 μg/ml) to the highest (100 μg/ml), and were introduced into the system. The retention time was found to be about 1.7 min. The calibration curve and regression equation derived from this data were then applied to determine the concentrations of the drug in unknown samples.
2.5. Fourier transform infrared (FTIR) spectroscopy studies
FTIR spectra were obtained using an IR Prestige-21 spectrometer (Shimadzu Corp, Japan). The samples were blended with KBr powder and subjected to a pressure of 9 tons, causing the KBr to melt and create a background matrix. The target sample was then dispersed on the surface of this matrix. After the pressure was released, a transparent disc was formed. FTIR spectra within the range of 4000 to 400 cm−1 were recorded for ceftazidime, the nanofibers, and the polymers used in the nanofiber fabrication process.
2.6. Differential scanning calorimetry (DSC)
The DSC Studies were conducted to investigate the thermal characteristics and phase transitions of CAZ and the polymers within the nanofibrous structures. Approximately 5 mg of each sample was placed in an aluminum pan. The samples were analyzed under a nitrogen gas flow, with the temperature range set from 40 to 250 °C and a scanning rate of 5 °C per minute, using a simultaneous thermal analyzer (STA 6000, Perkin Elmer).
2.7. Morphology of ceftazidime–loaded nanofibers
The morphological characteristics of the prepared nanofibrous inserts were determined using electron microscopy. The surface characteristics and morphology of the formulations, after being coated with a specified amount of gold-covered samples, were examined using a Hitachi SU3500 Scanning Electron Microscope (SEM). The voltage factor in high vacuum mode was selected to be above 20 kV.
2.8. In vitro drug release test
To determine the quantity of CAZ released from the prepared nanofibers, a specific amount of the formulated inserts was placed in a donor compartment filled with 0.5 ml of PBS at a pH of 7.4. The donor chamber, containing the nanofiber inserts, was separated from the receptor chamber, which was filled with 15.5 ml of medium, by a dialysis membrane. This membrane, with a molecular weight cutoff of 12,000 to 14,000 Da (Delchimica Scientific Glassware, Milan, Italy), was maintained at 37 °C and 100 rpm. Predetermined quantities of the sample were methodically extracted from the receptor chamber at set intervals (1, 2, 4, 6, 12, 24, 36, 48, 60, 72, 96, and 120 h) and immediately replaced with 15.5 ml of fresh PBS. Considering the experimental conditions and drug solubility, sink conditions were maintained during the experiment. The concentration of CAZ in the receptor phase was determined using the specified HPLC technique.
Release data were evaluated through various kinetic models, such as Korsmeyer-Peppas, first-order, zero-order, and Higuchi. The model that demonstrated the lowest Akaike Information Criterion (AIC) and the highest correlation coefficient(R2) was chosen to determine the in vitro drug release mechanisms.
2.9. Cell toxicity
Cultured (L929 (mouse fibroblast)) cells were plated in a 96-well and underwent an incubation period of 48 h at a temperature of 37 °C in an atmosphere containing 5 % CO2. One row of the 96-well plate served as a control without any formulation, while the other rows received different concentrations (4.68, 9.375, 18.75, 37.5, 75, 50, 150 and 300 μg/ml) of the prepared nanofibers. Nanofibrous pieces with different drug concentrations were sectioned and incubated in 10 ml of sterile DMEM for a specified period. Subsequently, the supernatant was removed to allow for direct interaction with the cells. The cell viability was evaluated by conducting the MTT assay on the fibroblast cells after a 24-h period. The culture medium was removed, the plate was cleansed with PBS, and then trypsin was applied. In the next step, 10 μl of MTT solution (5 mg/ml) were added to all wells. The plate underwent a 4-h incubation period in accordance with standard laboratory conditions. Subsequently, the medium that included the MTT solution was discarded, and Dimethyl Sulfoxide (DMSO) was introduced to solubilize the formazan crystals. After a 30-min incubation period, the absorbance was recorded at 540 nm with the use of a microplate reader. The gathered data was then analyzed using the specified equation:
2.10. Sterility and antimicrobial efficacy test
Samples of the ocular inserts were placed into various culture mediums: Sabouraud Dextrose broth for fungi incubated at 25 ± 0.5 °C, Thioglycolate Sodium Liquid for anaerobic bacteria, and Fluid Casein Digest Medium for aerobic bacteria, both at 35 ± 0.5 °C. This process was to confirm that no living microorganisms were present, suitable for ophthalmic use. Following this, the cultures were placed in an incubator.
For the purpose of determining the antimicrobial properties of CAZ-infused nanofibers, the study utilized two commonly encountered ocular pathogens. A McFarland standard suspension of P. aeruginosa (ATCC 9637) and the S. aureus (ATCC 6538) was uniformly grown on Tryptic Soy Agar (TSA) plates. Subsequently, designated segments of the nanofibers were positioned on the TSA plates. After a 24-h incubation at 35 °C, the antimicrobial effectiveness of nanofibrous samples was determined by measuring the inhibition zones.
2.11. Rabbit eye irritation test and in vivo studies
12 male New Zealand white rabbits with specific weight (3.7 to 4.2 kg) were used for the ocular irritation test. Before the experiments began, the New Zealand White rabbits were housed individually in standard laboratory conditions. They were kept on a 12-h light/dark cycle, at a temperature of 20–22 °C, and with a relative humidity of 50–60 %. The rabbits had access to a standard diet and water at all times. These conditions were maintained throughout the experiment. A 100 μl of sterile PBS along with ceftazidime-loaded nanofiber were inserted into one eye of the rabbits. A similar volume of sterile PBS was instilled in the other eye, which served as a control. The ocular irritation test was performed over a period of 3 days using clinical assessment methods to evaluate symptoms such as eye discomfort, tearing, or redness. These evaluations were based on the scoring criteria specified by the ISO-10993-10 standard.
12 male New Zealand white rabbits were divided into 2 groups: a) 6 rabbits were selected for CAZ-PVA-PCL, and b) 6 rabbits were treated with CAZ-PVA-EUD. Firstly, the rabbit's eye was washed with sterile PBS, and then, a single dose of the ocular inserts, with different formulations containing approximately 30 mg of fibers and a specified amount of sterile PBS, was placed into the conjunctival sac of the right eye of rabbits. The left eyes received only PBS. Approximately 100 μl of PBS was dropped into the eyes and then sampling of tear fluid was carried out after 30 s using a micropipette at specific time intervals (1, 2, 4, 6, 12, 24, 36, 48, 60, 72, 96, 120 h). The HPLC method described in section 2.5 was employed to determine the CAZ concentration in the tear fluid samples. The samples were centrifuged at 1000 g for 10 min, and the supernatant was stored in Eppendorf tubes at −20 °C until analysis. Dilution considerations were also factored into the calculations. To analyze the concentration of CAZ in the tear fluid, we utilized an HPLC method as detailed in the manuscript. Importantly, this method allowed us to obtain the required measurements without sacrificing any animals. Our team measured the concentration of Ceftazidime in rabbit eyes using a unique, self-designed, animal-friendly technique. Post-experiment, the animals were not sacrificed, due to the methodology used. The procedure was conducted in compliance with the guidelines for animal research, as sanctioned by the Laboratory Animal Ethics Committee at Kermanshah University of Medical Sciences (Approval No. IR.KUMS.AEC.1403.002.).
2.12. Statistical analysis
The experiments were performed in triplicate, and the outcomes were presented as the mean value ± the standard deviation (SD). For the statistical evaluation, the Kruskal-Wallis and t-tests were applied. A p-value less than 0.05 was deemed to indicate a statistically significant difference.
3. Results and discussion
3.1. Preparation of ceftazidime-loaded nanofibers
The synthesis of CAZ-loaded nanofibers involves a delicate balance of parameters to ensure the structural integrity and bioactivity of the drug. The process includes optimizing the conditions for electrospinning, such as voltage, distance between the needle and collector, and flow rate, as well as selecting appropriate solvents used, and the levels of polymer concentration. The developed formulations were subsequently utilized to manufacture nanofiber inserts containing CAZ. PCL, PVA, and EUD have been extensively studied for their potential in delivering drugs to the eye, owing to their notable mechanical strength, slow degradation, compatibility with biological tissues, and lack of toxic effects on the eyes. The controlled release of drugs using nanofibers composed of after mentioned polymers has garnered significant interest owing to these advantageous attributes (Da Silva et al., 2015; Sahu et al., 2022; Nikam et al., 2023). This study's objective was to create innovative nanofibrous inserts for the prolonged release of ceftazidime into the eye, and to assess how the incorporation of PCL and EUD nanofibers alongside PVA nanofiber containing drug influences the physicochemical properties and release profile of the inserts.
3.2. Physicochemical properties
3.2.1. Thickness and weight uniformity test
The ophthalmic ceftazidime inserts exhibited weights within the range of 31.6 ± 0.59 to 31.88 ± 0.16 mg, displaying low standard deviations, indicating consistency in the weight of the prepared formulations. As displayed in Table 1, the measured nanofiber thickness was reported as 0.211 ± 0.025 and 0.234 ± 0.008 mm for CAZ-PVA-EUD and CAZ-PVA-PCL respectively, with low standard deviations, underscoring uniformity in the thickness of the prepared formulations. In general, the ophthalmic inserts were found to possess appropriate weight and thickness for placement within the eye sac ensuring that the nanofibers won't cause any animal discomfort or irritations to the ocular tissue after administration (Sun et al., 2016). The nanofibers were considered comfortable for patients, falling within the thickness range of 250 μm (Omer and Zelkó, 2021).
Table 1.
The physicochemical and mechanical properties of the inserts.
|
Physiochemical properties |
Formulations |
|
|---|---|---|
| CAZ-PVA-PCL | CAZ-PVA-EUD | |
| Thickness (mm) | 0.234 ± 0.008 | 0.211 ± 0.025 |
| Weight Uniformity (mg) | 31.88 ± 0.16 | 31.6 ± 0.59 |
| Entrapment Efficacy% (EE%) | 98.01 ± 0.56 | 98.8 ± 0.61 |
|
Folding Endurance (times) |
275.66 ± 9.86 | 217.66 ± 16.25 |
| Elongation at Break (%) | 91.6 ± 1.77 | 82.6 ± 1.57 |
| Time at Break (min) | 6.96 ± 0.06 | 4.86 ± 0.32 |
| Tensile strength (MPa) | 1.93 ± 0.05 | 1.4 ± 0.09 |
| Surface pH (mean ± SD) | 6.69 ± 0.12 | 6.52 ± 0.15 |
| Moisture loss (%) | 0.96 ± 0.06 | 1.193 ± 0.12 |
| Moisture uptake(%) | 1.27 ± 0.24 | 1.33 ± 0.15 |
| %Swelling 3 h | 127.31 ± 7.76 | 153.05 ± 3.64 |
| %Swelling 6 h | 162.16 ± 6.18 | 182.06 ± 4.17 |
| %Swelling 12 h | 179.92 ± 7.5 | 207.35 ± 10.4 |
| Mucoadhesion Time (s) | 407.66 ± 12.05 | 523.33 ± 21.82 |
3.2.2. Folding endurance and tensile strength
Table 1 shows the result for the folding endurance and tensile strength of the prepared ocular inserts. A high folding endurance in the nanofibrous inserts indicates their elevated rigidity, potentially leading to ocular irritation. On the other hand, low folding endurance suggests a degree of fragility during placement within the ocular sac, which could result in breakage and expulsion from the eye due to blinking and eye movements. Therefore, achieving an appropriate level of folding endurance is crucial. The range of folding endurance for the prepared nanofibers is 217.66 ± 16.25 to 275.66 ± 9.86. All the prepared nanofibrous inserts demonstrate suitable tensile strength for ocular application which is consistent with previous studies with a folding endurance of 272–320 folds for PVA-HA nanofibrous inserts (Lokhande et al., 2023), and folding endurance range of 186–379 folds for PCL-EUD and PCL nanofibers (Mirzaeei et al., 2022).
The tensile strength test results showed that the CAZ-PVA-EUD formulation, with a break time of 4.86 ± 0.32 min and an elongation of approximately 82.6 ± 1.57 %, experienced a faster break under tension compared to the other formulation. CAZ-PVA-PCL nanofiber, with a break time of around 6.96 ± 0.06 min and an elongation of about 91.6 ± 1.77 %, demonstrated higher elongation and break time. All the prepared nanofiber formulations exhibited appropriate tensile strength for use in ocular applications. According to previous research, PCL nanofibrous inserts possess lower break time and higher elongation and strength compared to EUD based nanofibers (Hashemi et al., 2022; Taghe et al., 2024a).
3.2.3. Entrapment efficiency percentage
The elevated drug concentration in the fabricated nanofibers enables the utilization of smaller ophthalmic inserts for placement in the eye. The drug content was calculated 98.01 ± 0.56 % and 98.8 ± 0.61 % for CAZ-PVA-PCL and CAZ-PVA-EUD, respectively as shown in Table 1. The adoption of small sized nanofibers in ophthalmic applications diminishes the risk of redness, eye irritation, and patient dissatisfaction. In the passive drug loading approach, the therapeutic compound is integrated into the polymeric matrix prior to the spinning process, thereby facilitating enhanced efficacy in drug entrapment. However, there is a noticeable decrease in entrapment efficiency, potentially could be attributable to the reduced capacity of the collector to drain. This occurs because the fibers that have built up on the surface of the collector might cause some fibers to be diverted from the target collection area as reported in a previous study(Garg et al., 2014). In another research, the slight reduction in EE percentage may have been caused by a small quantity of the drug that was not extracted into the distilled water to quantify the drug loaded within the nanofiber(Patrojanasophon et al., 2020). A significant benefit of this method is the capacity to load substantial drug quantities into more compact dosage forms. Electrospun nanofibers are expected to possess a substantial drug content capacity, owing to their exceptionally large surface area. This is also attributed to the passive method of drug loading, that involves incorporating drugs into the polymeric solution prior to electrospinning. As the result, the risk of losing the drug during this procedure was minimal.
3.2.4. Swelling percentage, moisture uptake, and moisture loss study
The degree of swelling in the fabricated inserts holds significant importance in determining the release behavior of the entrapped drug. The CAZ-PVA-EUD nanofibers indeed exhibited higher swelling percentages compared to CAZ-PVA-PCL at the specified time intervals (Table.1). After 12 h, the swelling percentages were 207.35 ± 10.4 % for CAZ-PVA-EUD and 179.92 ± 7.5 % for CAZ-PVA-PCL. The reduced swelling percentage in CAZ-PVA-PCL may be ascribed to the incorporation of PCL nanofiber, a polymer with relatively lower hydrophilicity compared to EUD, into the PVA structure. This alteration leads to a nanofiber that exhibits a decreased capacity for water absorption (Putri et al., 2020).
To assess the formulation's stability under both humid and dry environments, the moisture uptake and loss of the nanofibers were quantified (Taghe et al., 2023). The results for the prepared ocular nanofibers were reported >1.5 % (Table 1). These findings from the moisture loss and uptake tests offer valuable insights into the physical stability and consistency of the ocular inserts under varying environmental conditions, particularly in relation to dryness and humidity.
3.2.5. Surface pH
The pH values of the prepared nanofibers range from 6.52 ± 0.1 to 6.69 ± 0.12, demonstrating a compatibility akin to that of the pH level found in tear fluid (Table 1). Consequently, their application is not associated with adverse effects such as redness, burning sensation, or sensitivity on the ocular surface (Abelson et al., 1981).
3.2.6. Ex-vivo mucoadhesion time
Table 1 shows the mucoadhesion time values measured for the nanofibrous inserts, which range from approximately 407.66 ± 12.05 to 523.33 ± 21.82 s. This represents a significant improvement over conventional eye formulations, which are usually eliminated within the first few seconds of administration (Mirzaeei et al., 2022). The extended mucoadhesion time can enhance drug delivery effectiveness by ensuring that the medication remains in contact with the eye for a longer duration, potentially improving therapeutic outcomes. The CAZ-PVA-EUD formulation demonstrated higher mucoadhesion to the cornea due to its greater wettability and hydrophilicity compared to the CAZ-PVA-PCL insert. The nanofibers are anticipated to exhibit longer residence time due to the inflated test conditions compared to the typical ocular environment and cul-de-sac. The in vivo study involved positioning the inserts in the conjunctival sac of rabbits, which can retain the inserts securely (Taghe et al., 2024a).
3.2.7. Surface contact angle
The ceftazidime-loaded inserts exhibits contact angles below 90 degrees, CAZ-PVA-EUD with a mean value of 68.56 ± 3.27° and CAZ-PVA-PCL with a mean contact angle of 71.88 ± 3.16°(Fig. 2), indicative of an appropriate degree of surface hydrophilicity (Tiyek et al., 2019). This attribute holds importance in enhancing mucosal adhesion and establishing a favorable site of contact with the conjunctiva. In a previous study, the contact angle for methotrexate-loaded PCL and EUD nanofibers were evaluated as 117 ± 5° and 128 ± 4° which indicates that PCL-based nanofibers are more hydrophobic than EUD nanofibers (Hashemi et al., 2022). This result has illustrated that the integration of hydrophobic polymers such as PCL and EUD to hydrophilic polymers like PVA, enables the fabrication of fibers with a desired contact angle (Du et al., 2016).
Fig. 2.

Measured surface contact angle for different formulations.
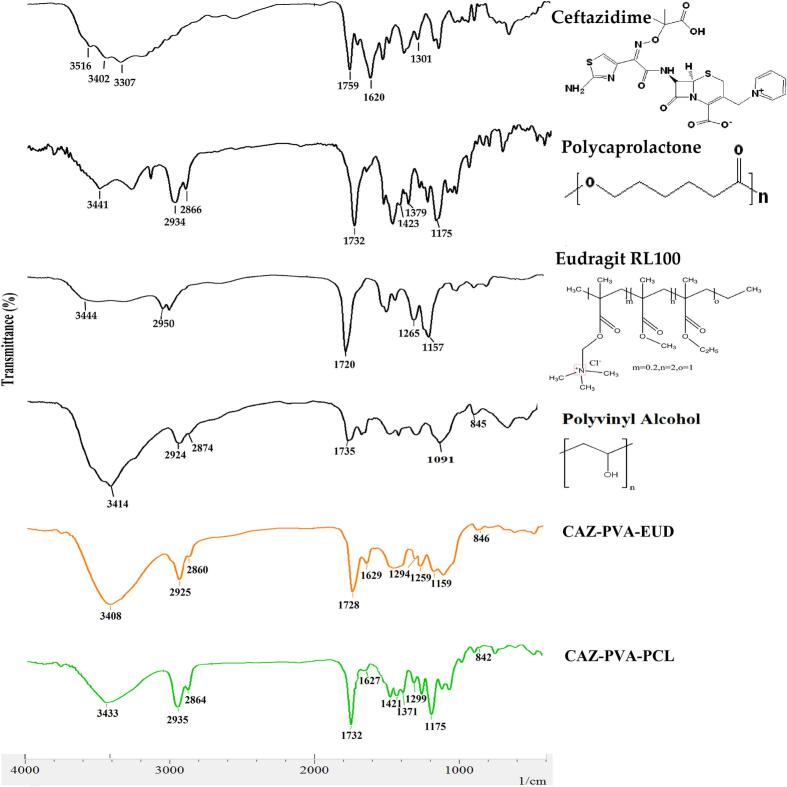
3.3. Fourier transform infrared (FTIR) spectroscopy study
Fig. 3 presents the absorption spectra of ceftazidime, CAZ-PVA-PCL, CAZ-PVA-EUD nanofibers, along with the polymers employed in the fabrication of ophthalmic inserts.
Fig. 3.
The FTIR spectrum of CAZ, polymers, and prepared nanofibers.
EUD RL 100 exhibited distinctive infrared absorption bands, such as OH stretching at approximately 3400 cm−1, CH stretching at 2950 cm−1, C—O stretching at 1720 cm−1, and quaternary ammonium group stretching at 1265 and 1157 cm−1 are linked to stretching steric carbonyl groups. These absorption bands are indicative of their chemical composition and structure. In the present study, the prominent peaks observed for PVA comprise of hydroxyl group at 3414 cm−1. Peaks at 2924 and 2874 cm−1 are attributed to the asymmetrical -CH2 groups. Peak at 1735 cm−1 corresponds to the stretching vibrations of C O. Additionally, the peak at 1091 cm−1 pertains to the stretching vibration of C—O. Furthermore, the peak at 845 cm−1 is associated with the bending vibration of C—H. Similar FTIR spectra were obtained for PCL. Noteworthy peaks include the hydroxyl group at 3441 cm−1, C—H stretching at 2934 and 2866 cm−1, stretching vibrations of -C=O at 1732 cm−1, and C—H bending vibrations of –CH2 and –CH3 groups at 1423 and 1379 cm−1, respectively. The peak at 1175 cm−1 is attributed to C—O stretching. The CAZ spectrum exhibits distinctive bands characteristic of cephalosporin compounds. These include bands in the range of 3600–3250 cm−1, corresponding to the NH and OH groups. Additionally, stretching band at 1759 cm−1 arises from the carboxylic groups stretching. Peak at 1301 cm−1 corresponding to C—N stretching and 1620 cm−1 resulting from stretching of amide group C O of β-lactam ring (Moreno and Salgado, 2012; Osório et al., 2018). The FTIR spectra of CAZ-PVA-EUD nanofibers include OH stretching peak at 3408 cm−1, peaks at 2900–2800 cm−1 attributing to CH stretching, 1728 cm−1 corresponding to C—O stretching are all peaks also seen in each polymer. The peaks at 1259 and 1159 cm−1 are the shifted peaks of EUD attributing to stretching steric carbonyl groups. The characteristic peaks of the drug were also seen in the nanofibers at 1629 and 1294 cm−1 respectively contributing to C O of β-lactam ring and C—N stretching. The peak at 846 cm−1 also corresponds to C—H bending vibration of PVA. The CAZ-PVA-PCL nanofiber exhibits distinctive peaks in its FTIR spectrum. Notably, the peak at 3433 cm−1 is attributed to the hydroxyl group present in both PCL and PVA. Furthermore, the peak at 2935 cm−1 is related to C—H stretching and the peak at 1732 cm−1 corresponds to the C O group in PCL and PVA. Additionally, the peaks observed at 1421, 1371 and 1175 cm−1 are attributed to bending vibrations of –CH2 and –CH3 groups the C—O stretching vibration of PCL, respectively. C—H bending vibration of PVA can also be observed at 842 cm−1. Peaks at 1627 and 1299 cm−1 are the drug's characteristic peaks relating to amide group C O of β-lactam ring and C—N stretching, respectively. These spectral characteristics provide valuable insights into the chemical composition of the nanofiber. The FTIR spectrum of the nanofibers exhibits characteristic peaks corresponding to both the drug and the polymer. This observation suggests that there is no discernible interaction between the drug and the polymer during the manufacturing process.
3.4. Differential scanning calorimetry (DSC) study
The DSC curves for CAZ and the nanofibers, along with the polymers employed in the fabrication of ophthalmic inserts are presented in Fig. 4. The DSC thermogram of PVA displayed two peaks at about 68 °C and 199 °C, indicating the glass transition and melting points of PVA, respectively. The DSC curves of PCL exhibit endothermic melting peaks at around 57 °C. The DSC thermograms of Eudragit RL100 indicated a glass transition temperature of approximately 63 °C. The thermogram of CAZ displays an endothermic peak at around 114 °C, corresponding to the drug's characteristic melting point. Fig. 4 illustrates that the sharp melting peak of CAZ in the nanofibrous structure has disappeared, potentially because of the molecular dispersion of CAZ within the nanofibrous structure or its amorphous state. Moreover, the endothermic peak seen for PVA at approximately 190 °C is absent in the DSC thermogram of the nanofibrous inserts. This omission is attributed to the formation of a non-crystalline structure caused by the rapid solidification process during electrospinning.
Fig. 4.
The DSC thermogram of CAZ, polymers, and the prepared nanofibers.
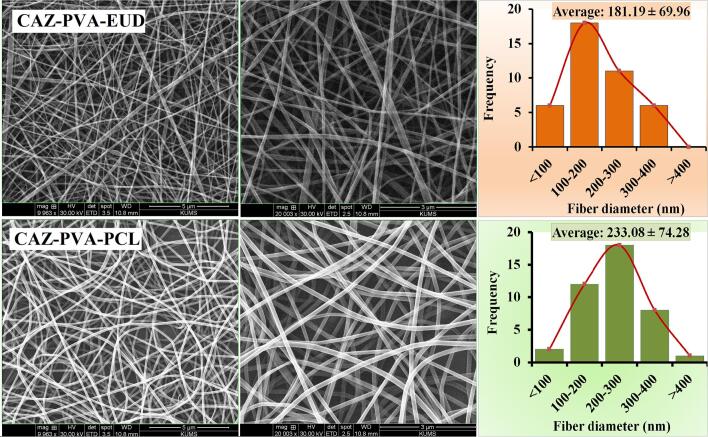
3.5. Morphology
The morphology of the ophthalmic inserts was analyzed using SEM. The nanofiber structures in the prepared formulation exhibited a continuous and randomly oriented pattern, showcasing an average diameter of 181.19 ± 69.96 nm for CAZ-PVA-EUD and 233.08 ± 74.28 nm for CAZ-PVA-PCL (Fig. 5). The average diameter of the synthesized nanofibers was determined to be below 700 nm. The tensile properties of nanofibers are influenced by various parameters, notably the chemical composition of the polymer and the dimensions of the nanofibers. It has been observed that a reduction in fiber diameter to approximately 700 nm significantly enhances the tensile modulus and strength. Therefore, both formulations exhibit considerable tensile strength. This adjustment is anticipated to yield a more consistent structure, thereby enhancing the durability of the inserts (Wong et al., 2008).
Fig. 5.
The SEM image and mean diameter of designed ocular nanofibrous inserts.
3.6. In vitro release study
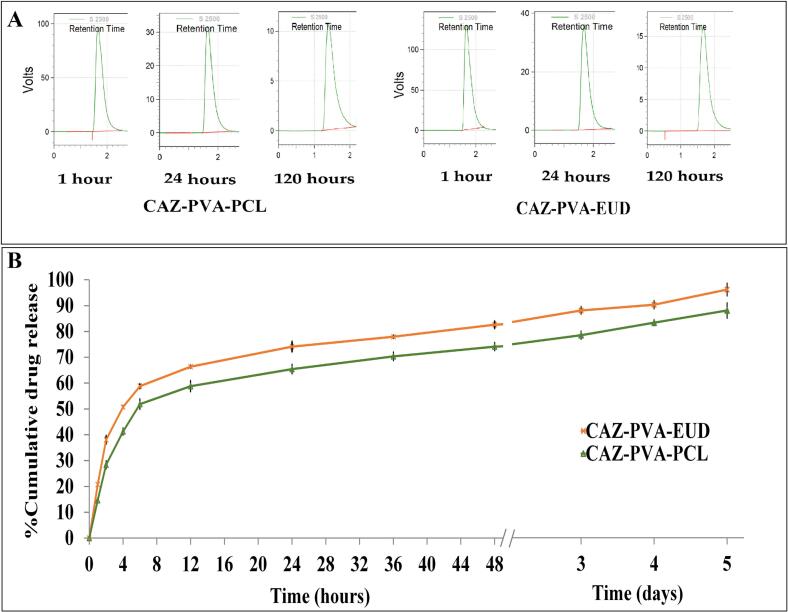
The peaks from various concentrations of ceftazidime were found to be suitable and distinct, as shown in Fig. 6. The calibration curve for the HPLC analysis was obtained by plotting the peak height against the drug concentration. The calibration curve demonstrated linearity within the concentration range from 100 to 3.125 μg/ml. The correlation equation was reported as y = 928.46× + 3407.9 with a coefficient (R2) of 0.9981. The percentage of variation coefficients for prepared standards ranged from 0.89 to 1.04. The values for the Limit of Detection (LOD) and Limit of Quantification (LOQ) for ceftazidime were determined to be 0.87 μg/ml and 2.65 μg/ml, respectively. The interday precision, expressed as coefficient of variation (CV%), and the accuracy (%), were observed to be within the ranges of 0.2–0.85 % and 89.03–106.36 %, respectively. The conclusion can be drawn that HPLC is an appropriate method for obtaining dependable outcomes.
Fig. 6.
A: The HPLC peaks obtained at 1 h, 24 h and 120 h for different formulation during in vitro study and B: Comparison of the drug release percentage of ceftazidime in the time interval of 0 to 120 h from CAZ-PVA-EUD and CAZ-PVA-PCL nanofibers in in vitro conditions with pH = 7.4 and temperature of 37 °C.
The in vitro release test for ceftazidime was conducted to study how the drug is released from the prepared nanofibers by HPLC method. Several studies on testing drug delivery systems for ceftazidime have been conducted. In a previous study Silva et al., prepared chitosan nanoparticles with different formulations for delivering ceftazidime to the eyes, which resulted in a prolonged release of ceftazidime over a period of 24 h (Silva et al., 2017). In yet another study, ceftazidime was loaded into the first generation of poly(propyleneimine) dendrimer in order to achieve an effective drug delivery system against P. aeruginosa. The dendrimer was able to sustain release the drug for up to72 hours, with an initial burst release during the first 15 h, in which about 50 % of the drug was released (Aghayari et al., 2015). In other studies, antibiotics, ofloxacin and ciprofloxacin, were selected as model drugs and loaded in hydrophilic layers from multi-layered nanofibrous ocular inserts that were appropriate systems to control drug release rate due to embedment between two hydrophobic layers of electrospun Eudragit® and PCL (Mirzaeei et al., 2021; Taghe et al., 2023).
Hydrophilic drugs, such as CAZ, are effectively encapsulated in hydrophilic polymers like PVA, though they exhibit rapid release rates. Conversely, these drugs are not as efficiently encapsulated in hydrophobic polymers like PCL and EUD, but these materials offer the benefit of controlled and slower drug release. The benefit of utilizing mixed-structured nanofibrous membranes lies in their ability to encapsulate a high quantity of drugs like CAZ, facilitating a prolonged drug delivery to the eye. Fig. 6 illustrates the drug release percentage of ceftazidime at specific time intervals from ophthalmic nanofibers. This study found that the prepared nanofibers release the drug in two phases: they begin with a rapid initial release and then transition to a sustained release. In the first 12 h, approximately 58.78 ± 1.18 and 49.93 ± 3.84 % of the drug was released from CAZ-PVA-EUD and CAZ-PVA-PCL, respectively. The process involved a rapid initial release of CAZ from the surface, followed by the penetration of PBS into the nanofibrous structure. This approach could enhance the infection healing process by initially providing a loading dose through the initial release, followed by a maintenance dose through the extended release. The total drug release after 120 h was around 90.58 ± 3.6 and 80.86 ± 2.3 % for CAZ-PVA-EUD and CAZ-PVA-PCL, respectively. As anticipated, CAZ-PVA-EUD exhibited greater drug release and swelling percentage in comparison to CAZ-PVA-PCL, attributed to EUD's higher level of swelling compared to PCL.
Analysis of the release data using kinetic models revealed that CAZ-PVA-PCL and CAZ-PVA-EUD formulations adhered to the Korsmeyer-Peppas model with the highest correlation coefficients (R2) and the minimum AIC values reported as 0.9917 and − 20.34 for CAZ-PVA-PCL, respectively (Table 2). R2 and AIC were also calculated as 0.9779 and − 27.35 for CAZ-PVA-EUD. The Korsmeyer-Peppas model demonstrated that diffusion was the primary factor in the release process. This process entailed an initial fast rate of release of CAZ from the surface, which was then followed by the infiltration of PBS into the nanofibrous structure, facilitating a gradual diffusion of CAZ into the medium. The ‘n’ value, which is the release exponent in the Korsmeyer-Peppas kinetic model, serves to predict the release mechanism of CAZ. An ‘n’ value that is >0.5 indicates that the release of CAZ from the formulations primarily adhered to the Fickian diffusion mechanism. Prior research has shown that the release of the drug from PVA and PCL/PVA nanofibrous inserts aligns with the Korsmeyer-Peppas model (Taghe et al., 2024b).
Table 2.
Values of parameters in different kinetic models for CAZ-PVA-PCL and CAZ-PVA-EUD.
|
Kinetic models |
Formulations |
|
|---|---|---|
| CAZ-PVA-PCL | CAZ-PVA-EUD | |
| Zero order | ||
| K | 0.006 | 0.006 |
| R2 | 0.72 | 0.67 |
| AIC | −15.6 | −22.14 |
| first order | ||
| K | 0.012 | 0.016 |
| R2 | 0.74 | 0.78 |
| AIC | −15.76 | −22.5 |
| Higuchi | ||
| K | 0.063 | 0.06 |
| R2 | 0.91 | 0.93 |
| AIC | −18.07 | −24.94 |
| Peppas | ||
| K | 0.31 | 0.26 |
| R2 | 0.99 | 0.97 |
| AIC | −20.34 | −27.35 |
| n | 0.31 | 0.26 |
3.7. Cell toxicity
As observed in Fig. 7, by examining the percentage of cell survival treated with nanofibers at different concentrations, it can be concluded that the prepared ophthalmic inserts exhibit low cellular toxicity. Previous research has demonstrated that nanostructured scaffolds crafted from PCL did not provoke inflammatory or immune reactions, nor did they exhibit toxicity upon implantation in the vitreous cavity of rat eyes. This substantiates their biocompatibility within the intraocular environment (Da Silva et al., 2015). PVA nanostructures also have been proved to be non-toxic, non-carcinogenic and biocompatible(Kenawy et al., 2007). The outcomes of earlier studies conclusively demonstrate that EUD RL 100 is an advantageous adjunct for ocular drug delivery, attributed to its inherent biocompatibility, absence of immunogenicity, and any toxic or irritative properties (Khan et al., 2013). The findings indicate a negative correlation between CAZ concentration and cell viability. Optimal cell viability, exceeding 96 %, was observed at the minimal drug concentration of 4.68 μg/ml. Notably, cell viability remained above 80 % even at a concentration of 300 μg/ml for both formulations. In accordance with ISO-10993-5 guidelines, cell viability above 80 % is classified as non-toxic. Our results demonstrate that the prepared inserts possess appropriate biocompatibility for ophthalmic drug delivery. These formulations can serve as an effective and non-toxic carrier for ophthalmic application.
Fig. 7.
A: Cell viability percentage after treatment with different concentrations of ophthalmic formulation after a 24-h period using MTT method (n = 3). B: Antimicrobial activity of CAZ-PVA-PCL and CAZ-PVA-EUD ocular inserts against P. aeruginosa and S. aureus.
3.8. Sterility and Antimicrobial Efficacy Test
It is crucial to confirm the sterility of the prepared nanofibrous inserts prior to initiating animal studies. The negative controls displayed no signs of growth, affirming the sterility of the test and the maintenance of aseptic conditions. Additionally, the absence of turbidity in the tubes containing the nanofibrous inserts serves as evidence of their sterility. This assures that the prepared ocular formulations can be utilized without risk of causing infection.
After a 24-h incubation period, distinct zones of inhibition were evident for the nanofibers loaded with the drug, indicating the preservation of CAZ antibacterial properties following incorporation. The average diameters of the inhibition zones were measured to be 32.42 ± 0.85 and 36.45 ± 1.29 mm against P. aeruginosa, and 24.86 ± 0.5 mm and 26.17 ± 0.62 mm against S. aureus for CAZ-PVA-PCL and CAZ-PVA-EUD, respectively. Evidently, the nanofibers demonstrated greater antimicrobial effectiveness against P. aeruginosa in contrast to S. aureus, owing to the higher efficacy of CAZ against gram-negative microorganisms like P. aeruginosa.(Kodati et al., 2017; Afzal et al., 2021). The negligible difference between EUD and PCL-based nanofibers could be due to the EUD's slightly higher swelling capability compared to PCL, causing a higher drug release percentage (Fig. 7B).
3.9. Rabbit eye irritation test and ocular pharmacokinetic studies
In the conducted animal studies, the nanofibers were inserted in the cul-de-sac of the rabbit's eye without causing any damage or surgery. The ocular irritability level for the prepared formulations was assessed for up to 3 days. No signs of ocular irritability such as redness, swelling, or tearing were observed in the animal's eye (Fig. 8). This indicates that no reaction occurred in the rabbit's eyes due to the placement of the nanoscale insets making them biocompatible systems.
Fig. 8.
a: Inserting the nanofibers in the cul-de-sac of the rabbit's eye. b: Observations of the rabbit's eye condition and the irritation ratings acquired through eye irritation assessments over a three-day period. c: The position of the nanofiber inserts at the end of the experiment after 72 h.
Measurement of Ceftazidime Concentration carried out by our team in rabbit eyes using a unique, animal-friendly technique. The ceftazidime ophthalmic inserts can provide sustained drug delivery within the eye over an extended period. The left eye of the rabbits served as the control, receiving only sterile PBS. Since no drug-loaded ocular insert was administered to the left eye, the recorded drug concentration was either zero or fell below the detection threshold. Drug release testing in the animal eye was performed after simply placing the ophthalmic inserts in the eye sac non-invasively (Fig. 9A). The measured drug concentration in the eye tears for pharmacokinetic parameters after placing the ophthalmic nanofibers is specified in Table 3. The maximum measured concentration (Cmax) for the prepared formulations was obtained after one hour. The maximum concentrations reported for CAZ-PVA-PCL and CAZ-PVA-EUD were 2263.98 ± 97.3 and 2638.85 ± 71.57 μg/ml, respectively, and the prepared formulations subsequently provided a stable drug release in the tear fluid for up to 120 h. The AUC0–120 factor was calculated for different formulations. The AUC0–120 factor for CAZ-PVA-PCL and CAZ-PVA-EUD were approximately 11,882.81 ± 80.5 and 9649.39 ± 86.84, respectively. As evident, the MRT factor for CAZ-PVA-PCL and CAZ-PVA-EUD were approximately 20.79 ± 0.8 and 18.41 ± 0.58, respectively. Overall, it can be concluded that the nanofiber inserts have high AUC0–120 and MRT values, this is due to the drug being encapsulated within the nanofibrous inserts, ensuring a steady and extended release of the drug from the nanosystems that were formulated. Earlier studies on PVA, PCL and EUD-based nanofibrous inserts for ocular drug delivery have been carried out. Sahu et al. developed a gatifloxacin-PVA loaded nanofiber which demonstrated anti-inflammatory properties for 24 h (Sahu et al., 2022). In another study, PCL nanofibers loaded with timolol maleate were prepared by electrospinning technique, maintaining the reduction in IOP for up to 72 h (Garg et al., 2014). Taghe et al. designed and prepared Eudragit® and PCL ocular nanofibrous inserts for ketorolac tromethamine delivery. These inserts are characterized by a rapid initial release within the first two hours in rabbit eyes, which is then followed by a steady and controlled release over a period of 144 h. (Taghe et al., 2024a). An innovative nanofiber insert has been fabricated for the extended administration of ofloxacin. This involves encapsulating hydrophilic chitosan/polyvinyl alcohol (CS/PVA) nanofibers within multiple layers of the hydrophobic polymer EUD RL100. In vivo experiments conducted on rabbit eyes using ofloxacin nanofibrous inserts demonstrated a consistent and sustained release of the drug for a duration of up to 96 h (Mirzaeei et al., 2021).
Fig. 9.
A: The HPLC peaks obtained at different hours for the formulations during in vivo study, B: Ceftazidime release from in vivo studies in rabbit eyes over a time interval of 0 to 120 h, and C: over a time interval of 50 to 120 h.
Table 3.
In Vivo Pharmacokinetic characteristics of ceftazidime.
| Formulation | Cmax | AUC0–120 | MRT |
|---|---|---|---|
| CAZ-PVA-EUD | 2638.85 ± 71.57 | 9649.39 ± 86.84 | 18.41 ± 0.58 |
| CAZ-PVA-PCL | 2263.98 ± 97.3 | 11,882.81 ± 80.5 | 20.794 ± 0.8 |
MIC90 refers to the minimum concentration of an antibiotic required to inhibit 90 % of the bacterial isolates. MIC90 of ceftazidime against P. aeruginosa and S. aureus has been reported as 2 and 22.4 μg/ml, respectively (Sueke et al., 2010). The in vivo findings validate that the formulated ocular inserts sustain a drug concentration (24.67–25.65 μg/ml) exceeding the S. aureus MIC90 for a duration of 96 h. The concentration of drug was 11.01–14.97 μg/ml for formulated ocular inserts after 120 h, which was 5.5- to 7.48-fold over the P. aeruginosa MIC90 (Fig. 9B).
4. Conclusion
In order to overcome the drawbacks of conventional ocular formulations such as eye drops, CAZ-loaded nanofibrous inserts were developed utilizing biodegradable polymers including PVA, PCL, and EUD ensuring safety and a sustained drug release profile. The development of nanofibers composed of CAZ-PVA-PCL and CAZ-PVA-EUD for the delivery of CAZ represents a novel approach for the therapy of bacterial keratitis, a severe ocular infectious disease, caused by P. aeruginosa. To our knowledge, this is the first documented instance of employing these specific nanofibers for such a purpose. The findings from this research confirmed that the nanofibers exhibit sustained release of drugs at a steady rate, enhanced bioavailability, decrease in the frequency of drug administration and minimal cytotoxicity and adverse effects due to the lack of preservatives and drug fluctuations. Moreover, the patterns of drug release documented in in vitro experiment within this study indicated a phase of an initial rapid release phase, followed by an extended release lasting up to 5 days. The in vivo results demonstrated that a single-dose administration of the inserts maintains drug concentrations above the MIC90 of ceftazidime against P. aeruginosa and S. aureus for 96 and 120 h, respectively.
Ethical approval
The protocol was approved by the Local Ethical Committee of Kermanshah University of Medical Sciences; approval number: IR.KUMS.AEC.1403.002.
CRediT authorship contribution statement
Shiva Taghe: Writing – review & editing, Writing – original draft, Visualization, Validation, Software, Methodology, Investigation, Formal analysis, Data curation, Conceptualization. Shahla Mirzaeei: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
The authors would like to acknowledge the Research Council of Kermanshah University of Medical Sciences (Grant number: 4030142) for financial support of this work. Thank Rahesh Daru Novin pharmaceutical start up for providing materials and equipment.
Contributor Information
Shiva Taghe, Email: shivataghe@gmail.com.
Shahla Mirzaeei, Email: shahlamirzaeei@gmail.com, smirzaeei@kums.ac.ir.
Data availability
The authors are unable or have chosen not to specify which data has been used.
References
- Abdel-Rahman L.M., Eltaher H.M., Abdelraouf K., Bahey-El-Din M., Ismail C., Kenawy E.-R.S., El-Khordagui L.K. Vancomycin-functionalized Eudragit-based nanofibers: Tunable drug release and wound healing efficacy. Int. J. Drug Deliv. Technol. 2020;58 doi: 10.1016/j.jddst.2020.101812. [DOI] [Google Scholar]
- Abelson M.B., Udell I.J., Weston J.H. Normal human tear pH by direct measurement. Arch. Ophthalmol. 1981;99:301. doi: 10.1001/archopht.1981.03930010303017. [DOI] [PubMed] [Google Scholar]
- Afzal M., Vijay A.K., Stapleton F., Willcox M.D. Susceptibility of ocular Staphylococcus aureus to antibiotics and multipurpose disinfecting solutions. Antibiotics. 2021;10:1203. doi: 10.3390/antibiotics10101203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aghayari M., Salouti M., Kazemizadeh A.R., Zabihian A., Hamidi M., Shajari N., Moghtader F. Enhanced antibacterial activity of ceftazidime against pseudomonas aeruginosa using poly(propyleneimine) dendrimer as a nanocarrier. Sci. Iran. 2015;22:1330–1336. https://scientiairanica.sharif.edu/article_3724.html [Google Scholar]
- Bouattour Y., Neflot-Bissuel F., Traïkia M., Biesse-Martin A.-S., Frederic R., Yessaad M., Jouannet M., Wasiak M., Chennell P., Sautou V. Cyclodextrins allow the combination of incompatible vancomycin and ceftazidime into an ophthalmic formulation for the treatment of bacterial keratitis. Int. J. Mol. Sci. 2021;22(19):10538. doi: 10.3390/ijms221910538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandel A., Kandav G. Insights into ocular therapeutics: a comprehensive review of anatomy, barriers, diseases and nanoscale formulations for targeted drug delivery. Int. J. Drug Deliv. Technol. 2024;105785 doi: 10.1016/j.jddst.2024.105785. [DOI] [Google Scholar]
- Chang S.-C., Lee M.-J., Lin H.-M. Preparation of nano-and micrometric ceftazidime particles with supercritical anti-solvent technique. J. Phys. Chem. C. 2008;112(38):14835–14842. doi: 10.1021/jp8046664. [DOI] [Google Scholar]
- Chen K.-J., Sun M.-H., Hou C.-H., Chen H.-C., Chen Y.-P., Wang N.-K., Liu L., Wu W.-C., Chou H.-D., Kang E.Y.-C., Lai C.-C. Susceptibility of bacterial endophthalmitis isolates to vancomycin, ceftazidime, and amikacin. Sci. Rep. 2021;11(1):15878. doi: 10.1038/s41598-021-95458-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Da Silva G.R., Lima T.H., Oréfice R.L., Fernandes-Cunha G.M., Silva-Cunha A., Zhao M., Behar-Cohen F. In vitro and in vivo ocular biocompatibility of electrospun poly (ɛ-caprolactone) nanofibers. Eur. J. Pharm. Sci. 2015;73:9–19. doi: 10.1016/j.ejps.2015.03.003. [DOI] [PubMed] [Google Scholar]
- Da Silva G.R., Lima T.H., Fernandes-Cunha G.M., Oréfice R.L., Da Silva-Cunha A., Zhao M., Behar-Cohen F. Ocular biocompatibility of dexamethasone acetate loaded poly (ɛ-caprolactone) nanofibers. Eur. J. Pharm. Biopharm. 2019;142:20–30. doi: 10.1016/j.ejpb.2019.05.010. [DOI] [PubMed] [Google Scholar]
- Daikos G.L., da Cunha C.A., Rossolini G.M., Stone G.G., Baillon-Plot N., Tawadrous M., Irani P. Review of ceftazidime-avibactam for the treatment of infections caused by Pseudomonas aeruginosa. Antibiotics. 2021;10(9):1126. doi: 10.3390/antibiotics10091126. https://www.mdpi.com/2079-6382/10/9/1126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du L., Xu H., Li T., Zhang Y., Zou F. Fabrication of silver nanoparticle/polyvinyl alcohol/polycaprolactone hybrid nanofibers nonwovens by two-nozzle electrospinning for wound dressing. Fibers Polym. 2016;17:1995–2005. doi: 10.1007/s12221-016-6813-0. [DOI] [Google Scholar]
- Garg T., Malik B., Rath G., Goyal A.K. Development and characterization of nano-fiber patch for the treatment of glaucoma. Eur. J. Pharm. Sci. 2014;53:10–16. doi: 10.1016/j.ejps.2013.11.016. [DOI] [PubMed] [Google Scholar]
- Hashemi S., Mortazavi S.A., Moghimi H.R., Darbasizadeh B. Development and evaluation of a novel methotrexate-loaded electrospun patch to alleviate psoriasis plaques. Drug Dev. Ind. Pharm. 2022;48(8):355–366. doi: 10.1080/03639045.2022.2117373. [DOI] [PubMed] [Google Scholar]
- Kenawy E.-R., Abdel-Hay F.I., El-Newehy M.H., Wnek G.E. Controlled release of ketoprofen from electrospun poly (vinyl alcohol) nanofibers. Mater. Sci. Eng. A. 2007;459(1–2):390–396. doi: 10.1016/j.msea.2007.01.039. [DOI] [Google Scholar]
- Khalil I.A., Saleh B., Ibrahim D.M., Jumelle C., Yung A., Dana R., Annabi N. Ciprofloxacin-loaded bioadhesive hydrogels for ocular applications. Biomater. Sci. 2020;8:5196–5209. doi: 10.1039/d0bm00935k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan M.S., Vishakante G.D., Bathool A. Development and characterization of pilocarpine loaded Eudragit nanosuspensions for ocular drug delivery. J. Biomed. Nanotechnol. 2013;9(1):124–131. doi: 10.1166/jbn.2013.1475. [DOI] [PubMed] [Google Scholar]
- Kodati S., Eller A.W., Kowalski R.P. The susceptibility of bacterial endophthalmitis isolates to vancomycin, ceftazidime, and amikacin: a 23-year review. Ophthalmol. Retina. 2017;1(3):206–209. doi: 10.1016/j.oret.2016.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumari A., Sharma P.K., Garg V.K., Garg G. Ocular inserts—Advancement in therapy of eye diseases. J. Adv. Pharm. Technol. Res. 2010;1(3):291–296. doi: 10.4103/0110-5558.72419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lokhande S., Bhandari Y., Ansari M., Khairnar S., Singh K. Development and evaluation of biodegradable nanofiber insert containing hyaluronic acid for treatment of dry eye syndrome. J. Drug Deliv. Technol. 2023;87 doi: 10.1016/j.jddst.2023.104847. [DOI] [Google Scholar]
- Luraghi A., Peri F., Moroni L. Electrospinning for drug delivery applications: a review. J. Control. Release. 2021;334:463–484. doi: 10.1016/j.jconrel.2021.03.033. [DOI] [PubMed] [Google Scholar]
- Mirzaeei S., Taghe S., Asare-Addo K., Nokhodchi A. Polyvinyl alcohol/chitosan single-layered and polyvinyl alcohol/chitosan/eudragit rl100 multi-layered electrospun nanofibers as an ocular matrix for the controlled release of ofloxacin: an in vitro and in vivo evaluation. AAPS Pharm. Sci. Tech. 2021;22(5):170. doi: 10.1208/s12249-021-02051-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirzaeei S., Faryadras F.B., Mehrandish S., Rezaei L., Daneshgar F., Karami A. Development and evaluation of polycaprolactone-based electrospun nanofibers containing timolol maleate as a sustained-release device for treatment of glaucoma: in vivo evaluation in equine eye. Res. Pharm. Sci. 2022;17(5):468–481. doi: 10.4103/1735-5362.355196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohamady Hussein M.A., Guler E., Rayaman E., Cam M.E., Sahin A., Grinholc M., Sezgin Mansuroglu D., Sahin Y.M., Gunduz O., Muhammed M., El-Sherbiny I.M., Megahed M. Dual-drug delivery of Ag-chitosan nanoparticles and phenytoin via core-shell PVA/PCL electrospun nanofibers. Carbohydr. Polym. 2021;270 doi: 10.1016/j.carbpol.2021.118373. [DOI] [PubMed] [Google Scholar]
- Moreno A., Salgado H. Development and validation of the quantitative analysis of ceftazidime in powder for injection by infrared spectroscopy. Phys. Chem. 2012;2:6–11. doi: 10.5923/j.pc.20120201.02. [DOI] [Google Scholar]
- Ng A.L., Te K.K., Choi C.C., Yuen L.H., Yim S.M., Chan K.S., Lai J.S., Wong I.Y. Predisposing factors, microbial characteristics, and clinical outcome of microbial keratitis in a tertiary centre in Hong Kong: a 10-year experience. J. Ophthalmol. 2015;2015 doi: 10.1155/2015/769436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikam A., Sahoo P.R., Musale S., Pagar R.R., Paiva-Santos A.C., Giram P.S. A systematic overview of Eudragit® based copolymer for smart healthcare. Pharmaceutics. 2023;15(2):587. doi: 10.3390/pharmaceutics15020587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omer S., Zelkó R. A systematic review of drug-loaded electrospun nanofiber-based ophthalmic inserts. Pharmaceutics. 2021;13(10):1637. doi: 10.3390/pharmaceutics13101637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osório L.R., Meneguin A.B., da Silva H.B., Barreto H.M., Osajima J.A., da Silva Filho E.C. Evaluation of physico-chemical properties and antimicrobial synergic effect of ceftazidime-modified chitosan. J. Therm. Anal. Calorim. 2018;134(3):1629–1636. doi: 10.1007/s10973-018-7332-6. [DOI] [Google Scholar]
- Patrojanasophon P., Tidjarat S., Opanasopit P., Ngawhirunpat T., Rojanarata T. Influence of nanofiber alignment on the release of a water-soluble drug from cellulose acetate nanofibers. Saudi Pharm. J. 2020;28(10):1210–1216. doi: 10.1016/j.jsps.2020.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Putri N.R.E., Kurniawan D.A., Pradana B.A., Imani N.A.C., Kusumastuti Y. Preparation of chitosan-polycaprolactone (PCL) composite nanofiber as potential for annulus fibrosus regeneration. Key Eng. Mater. 2020;840:368–376. doi: 10.4028/www.scientific.net/KEM.840.368. [DOI] [Google Scholar]
- Rozi M.F., Mohmad Sabere A.S. A review on conventional and novel topical ocular drug delivery system. Aust. J. Pharm. 2021;1(1):19–26. doi: 10.31436/jop.v1i1.32. [DOI] [Google Scholar]
- Sahu D.K., Pradhan D., Halder J., Biswasroy P., Kar B., Ghosh G., Rath G. Design and optimization of gatifloxacin loaded polyvinyl alcohol nanofiber for the treatment of dry eye infection: in vitro and in vivo evaluation. Int. J. Drug Deliv. Technol. 2022;76 doi: 10.1016/j.jddst.2022.103651. [DOI] [Google Scholar]
- Silva M.M., Calado R., Marto J., Bettencourt A., Almeida A.J., Gonçalves L.M.D. Chitosan nanoparticles as a mucoadhesive drug delivery system for ocular administration. Mar. Drugs. 2017;15(12):370. doi: 10.3390/md15120370. https://www.mdpi.com/1660-3397/15/12/370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sofi H.S., Abdal-Hay A., Ivanovski S., Zhang Y.S., Sheikh F.A. Electrospun nanofibers for the delivery of active drugs through nasal, oral and vaginal mucosa: current status and future perspectives. Mater. Sci. Eng. C. 2020;111 doi: 10.1016/j.msec.2020.110756. [DOI] [PubMed] [Google Scholar]
- Sueke H., Kaye S., Neal T., Murphy C., Hall A., Whittaker D., Tuft S., Parry C. Minimum inhibitory concentrations of standard and novel antimicrobials for isolates from bacterial keratitis. Invest. Ophthalmol. Vis. Sci. 2010;51(5):2519–2524. doi: 10.1167/iovs.09-4638. [DOI] [PubMed] [Google Scholar]
- Sun X., Yu Z., Cai Z., Yu L., Lv Y. Voriconazole composited polyvinyl alcohol/hydroxypropyl-β-cyclodextrin nanofibers for ophthalmic delivery. PLoS One. 2016;11(12) doi: 10.1371/journal.pone.0167961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sy A., Srinivasan M., Mascarenhas J., Lalitha P., Rajaraman R., Ravindran M., Oldenburg C.E., Ray K.J., Glidden D., Zegans M.E., McLeod S.D., Lietman T.M., Acharya N.R. Pseudomonas aeruginosa keratitis: outcomes and response to corticosteroid treatment. Invest. Ophthalmol. Vis. Sci. 2012;53(1):267–272. doi: 10.1167/iovs.11-7840. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taghe S., Mirzaeei S., Ahmadi A. Preparation and evaluation of nanofibrous and film-structured ciprofloxacin hydrochloride inserts for sustained ocular delivery: pharmacokinetic study in rabbit’s eye. Life. 2023;13(4):913. doi: 10.3390/life13040913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taghe S., Mirzaeei S., Bagheri M. Preparation of polycaprolactone and polymethacrylate nanofibers for controlled ocular delivery of ketorolac tromethamine: pharmacokinetic study in rabbit's eye. Eur. J. Pharm. Sci. 2024;192 doi: 10.1016/j.ejps.2023.106631. [DOI] [PubMed] [Google Scholar]
- Taghe S., Mirzaeei S., Hosseinkhani T. Design and development of dual drug-loaded nanofibrous inserts for ophthalmic sustained delivery of AMK and VAN: pharmacokinetic study in rabbit’s eye. Int. J. Pharm. 2024;124056 doi: 10.1016/j.ijpharm.2024.124056. [DOI] [PubMed] [Google Scholar]
- Taghe S., Mirzaeei S., Pakdaman N., Kazemi A., Nokhodchi A. Macrolide-loaded nanofibrous inserts with polycaprolactone and cellulose acetate base for sustained ocular delivery: pharmacokinetic study in rabbit’s eye. Int. J. Pharm. 2024;124699 doi: 10.1016/j.ijpharm.2024.124699. [DOI] [PubMed] [Google Scholar]
- Tiyek I., Gunduz A., Yalcinkaya F., Chaloupek J. Influence of electrospinning parameters on the hydrophilicity of electrospun polycaprolactone nanofibres. J. Nanosci. Nanotechnol. 2019;19(11):7251–7260. doi: 10.1166/jnn.2019.16605. [DOI] [PubMed] [Google Scholar]
- Tofighia P., Soltani S., Montazam S.H., Montazam S.A., Jelvehgari M. Formulation of tolmetin ocuserts as carriers for ocular drug delivery system. Iran. J. Pharm. Res. 2017;16(2):432. [PMC free article] [PubMed] [Google Scholar]
- Turanlı Y., Acartürk F. Fabrication and characterization of budesonide loaded colon-specific nanofiber drug delivery systems using anionic and cationic polymethacrylate polymers. Int. J. Drug Deliv. Technol. 2021;63 doi: 10.1016/j.jddst.2021.102511. [DOI] [Google Scholar]
- Ubani-Ukoma U., Gibson D., Schultz G., Silva B.O., Chauhan A. Evaluating the potential of drug eluting contact lenses for treatment of bacterial keratitis using an ex vivo corneal model. Int. J. Pharm. 2019;565:499–508. doi: 10.1016/j.ijpharm.2019.05.031. [DOI] [PubMed] [Google Scholar]
- Wijesooriya C., Budai M., Budai L., Szilasi M.E., Petrikovics I. Optimization of liposomal encapsulation for ceftazidime for developing a potential eye drop formulation. J. Basic. Clin. Pharm. 2013;4(3):73–75. doi: 10.4103/0976-0105.118810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong S.-C., Baji A., Leng S. Effect of fiber diameter on tensile properties of electrospun poly (ɛ-caprolactone) Polymer. 2008;49(21):4713–4722. doi: 10.1016/j.polymer.2008.08.022. [DOI] [Google Scholar]
- Yildiz E.H., Airiani S., Hammersmith K.M., Rapuano C.J., Laibson P.R., Virdi A.S., Hongyok T., Cohen E.J. Trends in contact lens-related corneal ulcers at a tertiary referral center. Cornea. 2012;31(10):1097–1102. doi: 10.1097/ICO.0b013e318221cee0. [DOI] [PubMed] [Google Scholar]
- Zhou X.-H., Wei D.-X., Ye H.-M., Zhang X., Meng X., Zhou Q. Development of poly (vinyl alcohol) porous scaffold with high strength and well ciprofloxacin release efficiency. Mater. Sci. Eng. C. 2016;67:326–335. doi: 10.1016/j.msec.2016.05.030. [DOI] [PubMed] [Google Scholar]
- Zupancic S., Sinha-Ray S., Sinha-Ray S., Kristl J., Yarin A.L. Long-term sustained ciprofloxacin release from PMMA and hydrophilic polymer blended nanofibers. Mol. Pharm. 2016;13(1):295–305. doi: 10.1021/acs.molpharmaceut.5b00804. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The authors are unable or have chosen not to specify which data has been used.