Abstract
Background
Large tidal volumes during de novo acute hypoxemic respiratory failure (AHRF) may promote patient self-inflicted lung injury. Tidal volume assessment under high flow nasal cannula (HFNC) is not routinely feasible at the bedside. Our objective was to determine whether tidal volume during low-level continuous positive airway pressure (CPAP) could predict tidal volume during HFNC and to compare the physiological effects of HFNC and low-level CPAP.
Methods
Prospective, single-center study including 29 de novo AHRF patients treated with HFNC (50 to 60 L.min− 1). Patients were monitored using electrical impedance tomography during HFNC then CPAP at 4 cmH2O. Tidal volume during HFNC was calculated based on tidal impedance variation. The ability of tidal volume under low-level CPAP to predict tidal volume under HFNC was explored using Bland-Altman analysis. CPAP and HFNC were compared in terms of tidal volume, minute ventilation, respiratory comfort, dyspnea, oxygenation, ventilation distribution, end-expiratory lung volume, thoraco-abdominal asynchrony and recruitment.
Results
Under HFNC, patients had a tidal volume of 6.6 (5.9–8.7) mL.kg− 1 PBW. 20 (69%) patients exhibited a tidal volume between 4 and 8 mL.kg− 1 PBW, while in 5 (17%) patients it exceeded 9 mL.kg− 1 PBW. Tidal volume under CPAP was higher (9.4 (8.3–11) mL.kg− 1 PBW, p < 0.001). Tidal volumes under CPAP and under HFNC were modestly correlated (Spearman r = 0.50, p = 0.005). Bland-Altman analysis showed a bias of 2.4 mL.kg− 1, with limits of agreement ranging from − 1.1 mL.kg− 1to 5.9 mL.kg− 1. Nevertheless, a larger (> 11.5 mL.kg− 1 PBW ) tidal volume under low-level CPAP predicted a larger (> 9 mL.kg− 1 PBW ) tidal volume under HFNC with 80% sensitivity and 96% specificity. Low-level CPAP was associated with increased minute ventilation, end-expiratory lung volume, and oxygenation as compared to HFNC. It decreased signs of respiratory distress in the most severe patients but was associated with lower comfort compared to HFNC.
Conclusion
Among ICU patients with de novo AHRF, tidal volume under HFNC was mostly protective. Tidal volume during CPAP at 4 cmH2O did not predict tidal volume during HFNC. Such low-level CPAP was associated with increased tidal volume, minute ventilation, end-expiratory volume, and oxygenation.
Trial registration
ClinicalTrials.gov ID NCT03919331. Registration date: 2019-03-26.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13613-024-01408-w.
Keywords: Acute hypoxemic respiratory failure, High flow nasal cannula, Continuous positive airway pressure, Noninvasive ventilation, Respiratory support
Introduction
During de novo acute hypoxemic respiratory failure (AHRF), large tidal volumes are associated with poor outcome in patients under noninvasive ventilation (NIV) [1, 2], raising concern about the potential involvement of patient self-inflicted lung injury (P-SILI) [3, 4]. Several devices are available to provide oxygen to such patients, with high flow oxygen through nasal cannula (HFNC) being widely adopted in intensive care units due to its clinical benefits compared to NIV and conventional oxygen therapy [5–8]. However, assessing tidal volume under HFNC is not routinely feasible at the bedside and data are scarce in the literature, leaving the prognostic impact of tidal volume under HFNC during de novo AHRF unknown.
HFNC has been shown to produce several physiological effects compared to conventional oxygen therapy [9], including an increase in end-expiratory lung volume due to the generation of positive airway pressure [6, 9]: with a flow rate of 50–60 L.min− 1, HFNC can generate a positive airway pressure approximating 4 cmH2O [10–12].
We hypothesized that Continuous Positive Airway Pressure (CPAP) set at a similarly low level of positive airway pressure to that generated by HFNC would result in comparable tidal volumes and could therefore be used to predict HFNC’s effect on tidal volume in clinical practice. Our primary objective was to compare the tidal volume during a low level of CPAP with that measured during HFNC in patients with de novo AHRF.
Additionally, CPAP has regained interest due to its frugality [13], making it suitable for use outside the ICU or constrained conditions [14–17]. Notably, most CPAP devices available for use outside the ICU provide a low level of CPAP [15, 17]. Physiological effects of such low-level CPAP have not been compared to those of HFNC. Therefore, our secondary objective was to compare the effects of low-level CPAP and HFNC on end-expiratory lung volume, oxygenation, distribution of ventilation, and thoraco-abdominal asynchrony.
Methods
This was a prospective, single-center, physiological study conducted in the medical intensive care unit of Henri Mondor Hospital, Créteil, France. The study was approved by the ethics committee “Comité de Protection des Personnes Ile de France 4” (2018-A02972-53). This research had been registered on clinicaltrial.gov (NCT03919331). A detailed description of the methods is provided in the online supplementary material.
Patients
Adult patients, hospitalized for de novo acute hypoxemic respiratory failure (defined as PaO2/FiO2 < 300 mmHg and PaCO2 ≤ 45 mmHg), with a respiratory rate greater than 25.min-1, and placed under HFNC as part of standard care, were eligible. Non-inclusion criteria were systolic arterial pressure < 90 mmHg, mean arterial pressure < 65 mm Hg, use of vasopressors, impaired consciousness (Glasgow coma score ≤ 12), contraindication to CPAP (e.g., facial trauma), contraindication to electrical impedance tomography (EIT), AHRF explained solely by cardiogenic pulmonary edema, asthma or chronic obstructive pulmonary disease exacerbation.
Protocol
The study consisted of three steps:
Initial HFNC step (HFNC 1): A 10-minute period during which FiO2 and gas flow were left as set by the clinician.
A CPAP step: A 10-minute period during which patients were placed under CPAP using an ICU ventilator via an oronasal mask. A heat and moisture exchanger was inserted in the ventilator circuit. The CPAP level was set at 4 cmH2O, and FiO2 was kept unchanged. Absence of significant leakage was verified by comparing expired and insufflated volumes.
Second HFNC step (HFNC 2): A 10-minute period with the same FiO2 and flow rate as HFNC 1.
All measurements were done during the last minute of each step.
Collected data
General data
During each step, blood pressure, heart rate, respiratory rate (RR), and transcutaneous oxygen saturation (SpO2) were collected. Arterial blood gases were taken at the end of the HFNC 1 and CPAP steps if an arterial catheter was present. Otherwise, a single arterial blood gas was collected at the end of HFNC 1. Changes in discomfort and dyspnea between HFNC 1 and CPAP were assessed using a 5-point Likert scale ranging from − 2 (much less discomfort or dyspnea) to + 2 (much more discomfort or dyspnea).
Electrical impedance tomography (EIT)
Patients were monitored using the Enlight 1800 EIT device (TIMPEL SA, São Paulo, Brazil). Two hemi-belts applied to the patient’s thorax collected thoracic impedance variations, that are proportional to intrathoracic aeration variations. During the HFNC phases, only electrical impedance was collected. During the CPAP phase, a pneumotachograph was inserted on the ventilator circuit to record tidal volume.
Thoraco-abdominal asynchrony
Thoracic and abdominal respiratory movements were recorded using two stretch-sensitive respiratory transducers connected to a wireless transmitter (BioNomadix® Respiration Transducer, Biopac systems, Goleta, CA, USA). The signals were then processed using AcqKnowledge version 4.3 (Biopac systems, Goleta, CA, USA).
Computed data
Tidal volume estimation under HFNC
The tidal volume under HFNC was calculated from the tidal impedance changes. The proportionality ratio between volume changes and electrical impedance changes was calculated during the CPAP step and used to convert tidal impedance changes during HFNC into tidal volumes [9]. Corrected minute ventilation was computed as minute ventilation multiplied by the ratio of the patient’s PaCO2 to 40 mm Hg [9].
Pendelluft
Pendelluft was calculated using a previously published method [18, 19]. A detailed description of this method is available in the supplement.
End-expiratory lung volume
Changes in end-expiratory lung volume (EELV) between steps were calculated from the changes in end-expiratory lung impedance (EELI). EELI was first averaged over the last minute of each step. Then the proportionality ratio between changes in volume and changes in electrical impedance was applied. EELI during HFNC 1 was taken as the reference.
Ventilation maps
Ventilation maps representing the distribution of ventilation at the pixel level were obtained for each condition using dedicated software (TIMPEL SA, São Paulo, Brazil). Each map was composed of 1024 pixels. To account for potential tidal volume variations between conditions, ventilated pixels were defined as follows [20, 21]: for each map, the volume received by each pixel was calculated by multiplying the tidal volume by the fraction of ventilation received by that pixel. The pixel receiving the most volume (Pixelmax) was identified for each map. Among the three Pixelmax obtained in a given patient (HFNC1, CPAP, HFNC2), the one receiving the lowest volume was taken as reference (PixelREF). A pixel was considered ventilated if it received a volume greater than 10% of the volume received by the PixelREF. A pixel was considered to represent potentially aerated lung if it was ventilated in at least one condition.
From the ventilation maps were calculated.
The number of ventilated pixels (PixelVENT), estimating the functional size of the lung. The changes in functional lung size between the three conditions were then calculated in absolute change (Δ PixelVENT, ABS) and relative value (Δ PixelVENT, REL = Δ PixelVENT, ABS / PixelVENT).
The global inhomogeneity index (GI) [22].
The distribution of ventilation (anterior versus posterior), expressed as a percentage of total ventilation.
Thoraco-abdominal asynchrony
The thoraco-abdominal phase angle, an objective measure of thoraco-abdominal asynchrony, was calculated [23–25]. Detailed description of the calculation is provided in the supplementary material.
Statistics
Continuous variables are expressed as median (interquartile range); categorical variables are expressed as number (percentage). Differences between the three conditions were examined using a Friedman test, followed by Wilcoxon paired tests with Benjamini-Hochberg correction. The relationship between tidal volume under HFNC and CPAP was assessed using Spearman correlation and Bland-Altman analysis. The performance of a larger tidal volume under CPAP in diagnosing a larger tidal volume under HFNC (i.e., > 9 mL.kg− 1 PBW a threshold associated with intubation or death in patients under NIV [26]) was assessed by ROC curve analysis (to determine the best cut-off for CPAP tidal volume), sensitivity and specificity. Patients were separated into two groups according to the median ROX index [27] at inclusion to determine the differential effects of CPAP in the most severe patients. Statistics were performed using GraphPad Prism software version 9.4.1 (GraphPad Software, LLC).
Ethics
Patients were included after receiving oral and written information and signing a consent form.
Results
Patients
Thirty patients were assessed. Data from the first patient were excluded due to unstable EIT recordings. Characteristics of the 29 analyzed patients, included 15 (11–19) hours after starting HFNC, are provided in Table 1.
Table 1.
Patient’s characteristics
| Variables | All patients (n = 29) |
|---|---|
| Age (years) | 63 (54–69) |
| Male sex – n (%) | 23 (79) |
| BMI (kg.m− 2) | 24 (20–27) |
| SAPS 2 at admission | 39 (27–48) |
| Current or past smoking – n (%) | 10 (34) |
| Reason for de novo acute respiratory failure – n (%) | |
| Community-acquired pneumonia | 18 (62) |
| Including COVID-19 | 7 (24) |
| Including pneumonia related to immunosuppression | 5 (17) |
| Hospital-acquired pneumonia | 5 (17) |
| Including pneumonia related to immunosuppression | 3 (10) |
| Aspiration – n (%) | 1 (3) |
| Other – n (%) | 5 (17) |
| Bilateral pulmonary infiltrates – n (%) | 20 (69) |
| Respiratory rate (breaths.min− 1) | 24 (19–28) |
| Heart rate (beat.min− 1) | 87 (72–97) |
| Systolic arterial pressure (mm Hg) | 123 (117–139) |
| Diastolic arterial pressure (mm Hg) | 69 (57–75) |
| HFNC data | |
| Flow (L.min− 1) | 50 (50–60) |
| FiO2 (%) | 70 (50–100) |
| Arterial Blood Gas | |
| pH | 7,46 (7,43 − 7,49) |
| [HCO3−] (mmol.L− 1) | 24,4 (22,1–26,9) |
| PaCO2 (mm Hg) | 33 (30,5–37) |
| PaO2 (mm Hg) | 78 (68–109) |
| PaO2/FiO2 (mm Hg) | 123 (101–169) |
| SaO2 (%) | 95 (93–98) |
| Lactate (mmol.L− 1) | 1.2 (0.8–1.9) |
| Ventilatory ratio | 1.4 (1.12–1.8) |
BMI: Body Mass Index; SAPS 2: Simplified Acute Physiology Score 2 (Le Gall JR, Lemeshow S, Saulnier F. A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA. 1993 Dec 22–29;270 [24]:2957-63.); HFNC: High Flow Nasal Cannula; SpO2: oxygen saturation by pulse oximeter; FiO2: inspired fraction of oxygen; PaCO2: arterial carbon dioxide pressure; PaO2: arterial oxygen pressure; SaO2: arterial oxygen saturation
Tidal volume and minute ventilation
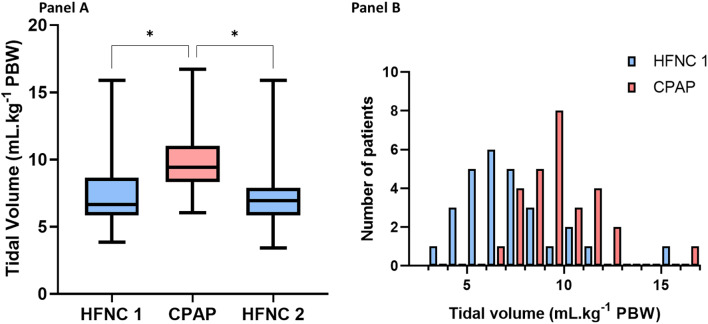
Ventilatory data during the HFNC 1 and CPAP are summarized in Table 2. Data comparing the three conditions are available in Table S1. Under HFNC 1 (at baseline), patients had a tidal volume of 6.6 (5.9–8.7) mL.kg− 1 PBW. 20 (69%) patients exhibited a tidal volume between 4 and 8 mL.kg− 1 PBW while the tidal volume exceeded 9 mL.kg− 1 PBW in 5 (17%) patients (Fig. 1).
Table 2.
Comparison between HFNC 1 and CPAP
| Variable | HFNC 1 | CPAP | Adjusted p-value |
|---|---|---|---|
| FiO2 (%) | 70 (50–100) | 70 (50–100) | NA |
| Flow (L.min− 1) | 50 (50–60) | NA | NA |
| Respiratory rate (.min− 1) | 24 (19–28) | 24 (20–30) | 0.09 |
| Tidal volume (mL) | 453 (353–578) | 629 (551–686) | < 0.001 |
| Tidal volume/PBW (mL.kg− 1 PBW) | 6.6 (5.9–8.7) | 9.4 (8.3–11) | < 0.001 |
| Minute ventilation (L.min− 1) | 10.6 (7.9–13.8) | 15.8 (12.5–17.5) | < 0.001 |
| Tidal impedance variation pixel−level (arbitrary units) | 37.9 (29.3–50.6) | 46.9 (38.9–65.7) | < 0.001 |
| Pendelluft (%) | 12.1 (8.3–18.5) | 9.8 (6.3–14.1) | 0.002 |
| EELV variation (mL) | 0 | 170 (43–399) | < 0.001 |
| EELV variation/PBW (mL.kg− 1 PBW) | 0 | 2.5 (0.6–5.4) | < 0.001 |
| Anterior EELV variation/PBW (mL.kg− 1 PBW) | 0 | 1.1 (0.2–3.1) | < 0.001 |
| Posterior EELV variation/PBW (mL.kg− 1 PBW) | 0 | 1.1 (0.1–2.7) | < 0.001 |
| Thoraco-abdominal phase angle (°) (N = 20) | 15 (9–34) | 10 (7–21) | 0.51 |
| SpO2 (%) | 96 (95–99) | 99 (97–100) | < 0.001 |
| Dyspnea (n = 23) | 0 | 0 (-1-1) | 0.15 |
| Discomfort (n = 23) | 0 | 1 (0–1) | 0.02 |
| ROX | 5.7 (4.2–8.2) | 5.5 (4.1–8.5) | 0.55 |
| Anterior distribution (%) | 46 (41–54) | 41 (37–50) | 0.01 |
| Global inhomogeneity index | 0.56 (0.53–0.60) | 0.58 (0.53–0.60) | 0.78 |
| Ventilated pixels (n) | 327 (292–356) | 358 (341–394) | < 0.001 |
| Hemodynamic parameters | |||
| Heart rate (beat.min− 1) | 87 (72–97) | 87 (71–103) | 0.09 |
| SAP (mm Hg) | 123 (117–139) | 134 (115–146) | < 0.01 |
| DAP (mm Hg) | 69 (57–75) | 74 (61–78) | < 0.01 |
| PP (mm Hg) | 55 (49–68) | 58 (48–77) | 0.28 |
| Arterial blood gases ( n = 12) | |||
| PaO2/FiO2 (mm Hg) | 109 (77–160) | 153 (109–194) | < 0.01 |
| PaCO2 (mm Hg) | 33 (31–35) | 35 (34–37) | < 0.001 |
| pH | 7.48 (7.44–7.5) | 7.45 (7.41–7.48) | < 0.001 |
| [HCO3−] (mmol.L− 1) | 24.6 (24-26.5) | 25.6 (23.6–26.8) | 0.11 |
| Corrected minute ventilation (L.min− 1) | 8.5 (7.4–11.3) | 14.6 (11.4–15.8) | < 0.001 |
| Ventilatory ratio | 1.39 (1.11–1.67) | 2.15 (1.91–2.6) | 0.001 |
Adjusted p-values were computed by the mean of Wilcoxon paired tests with Benjamini-Hochberg correction
Tidal impedance variation pixel−level refers to the sum of the tidal impedance variations of every pixel, with the breath cycle defined for each pixel, thus considering the Pendelluft. End-expiratory lung volume during CPAP is expressed as difference from HFNC1. The change in discomfort and dyspnea between HFNC 1 and CPAP was assessed using a 5-point Likert scale ranging from − 2 (much less discomfort or dyspnea to + 2: much more discomfort or dyspnea)
HFNC: High Flow Nasal Cannula; CPAP: Continuous Positive Airway Pressure; FiO2: inspired fraction of oxygen; PBW: predicted body weight; EELV: end-expiratory lung volume; SpO2: oxygen saturation by pulse oximeter; SAP: systolic arterial pressure; DAP: diastolic arterial pressure; PP: pulse pressure; PaO2: arterial oxygen pressure; FiO2: inspired fraction of oxygen; PaCO2: arterial carbon dioxide pressure
Fig. 1.
Comparison of tidal volumes between HFNC and CPAP. Legend: Panel A. Statistical comparison of tidal volume between HFNC 1, CPAP and HFNC 2. * denotes p < 0.01. Panel B. Distribution of tidal volumes among 29 patients under HFNC 1 (blue) and CPAP (red). HFNC: High Flow Nasal Cannula; CPAP: Continuous Positive Airway Pressure; PBW: predicted body weight
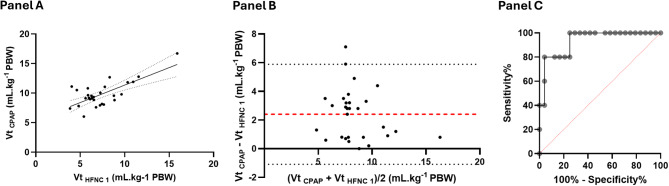
Tidal volume under CPAP was significantly higher compared to HFNC (Table 2; Fig. 1). The median tidal volume increase between HFNC 1 and CPAP was 2.8 (0.8–3.4) mL.kg− 1 PBW (Figure S2). Tidal volume under HFNC 1 and CPAP were modestly correlated (Spearman r = 0.50, p = 0.005) (Fig. 2A). Bland-Altman analysis showed a bias of 2.4 mL.kg− 1, with limits of agreement ranging from − 1.1 to 5.9 mL.kg− 1 PBW (Fig. 2B). The change in tidal volume between HFNC1 and CPAP was inversely correlated to the tidal volume during HFNC, with patients with higher tidal volume having the smallest change or no change at all (Figure S3). Regarding intra-tidal gas redistribution, CPAP was associated with significantly less Pendelluft than HFNC 1: 9.8% (6.3–14.1) vs. 12.1% (8.3–18.5), p = 0.002.
Fig. 2.
Accuracy of tidal volume during low-level CPAP to predict tidal volume during HFNC. Legend: Panel A. Correlation between tidal volume under HFNC and tidal volume under CPAP. Panel B. Bland-Altman graph comparing tidal volume under HFNC and tidal volume under CPAP. Panel C. ROC curve of tidal volume during CPAP to diagnose tidal volume under HFNC > 9mL.kg-1 PBW. HFNC: High Flow Nasal Cannula; CPAP: Continuous Positive Airway Pressure; PBW: predicted body weight. Vt: tidal volume
The area under the ROC curve for a tidal volume under CPAP to predict a larger (> 9 mL.kg− 1 PBW) tidal volume under HFNC was 0.93 (95% CI: 0.83 to 1) (Fig. 2). A tidal volume > 11.5 mL.kg− 1 PBW under CPAP predicted a tidal volume > 9mL.kg− 1 PBW under HFNC with 80% (95% CI: 38 to 99) sensitivity and 96% (95% CI: 80 to 100) specificity. 11 (38%) patients were eventually intubated. Their tidal volumes were not significantly higher than those of non-intubated patients (HFNC 1: 7.2 mL.kg− 1 PBW [5.2–8.1] vs. 6.1 mL.kg− 1 PBW [6.3–8.8], p = 0.17; CPAP: 9.3 mL.kg− 1 PBW [7.9–9.8] vs. 9.5 mL.kg− 1 PBW [8.7–11.1], p = 0.72). A tidal volume > 9 mL.kg-1 PBW during HFNC or > 11.5 mL.kg-1 PBW during CPAP was not associated with intubation, as determined by Fisher exact test (P > 0.99 for both comparisons) (Table S2).
Minute ventilation was significantly higher with CPAP compared to HFNC.
Gas exchange
At the same FiO2, CPAP resulted in higher SpO2 (Table 2). Among the 12 patients with blood gases measured under CPAP, the PaO2/FiO2 ratio significantly increased (Table 2). Changes in PaO2/FiO2 ratio between HFNC 1 and CPAP did not correlate with changes in EELV, tidal volume, or tidal volume distribution.
Despite increased tidal volume and minute ventilation, CPAP was associated with higher PaCO2 [35 mmHg (34-37) for CPAP vs. 33 mmHg (31-35) for HFNC 1, p < 0.01], suggesting increased dead space. As a result, corrected minute ventilation and ventilatory ratio were higher during CPAP. Detailed data from the 12 patients with CPAP blood gases are presented in Table S3.
End-expiratory lung volume, recruitment and ventilation distribution
EELV under CPAP was significantly higher than under HFNC (median 2.5 mL.kg− 1 PBW vs. 0 for CPAP and HFNC respectively, p < 0.001) (Table 2). The increase in EELV was similarly distributed between anterior and posterior regions (p = 0.77 for anterior variation vs. posterior variation). CPAP increased functional lung size, estimated by the number of ventilated pixels (median 358 vs. 327, p < 0.001) (Table 2). The relative increase in ventilated pixels between HFNC 1 and CPAP was 13% (7-20). Ventilation distribution slightly favored posterior areas under both supports, with CPAP resulting in more posterior ventilation (median anterior distribution of ventilation 41% vs. 46% for CPAP and HFNC respectively, p = 0.01) (Table 2). Ventilatory inhomogeneity, measured by the global inhomogeneity index, did not significantly differ between HFNC and CPAP (p = 0.78).
Thoraco-abdominal phase angle, discomfort and dyspnea
Thoraco-abdominal phase angle was evaluated in 20 patients (not performed in 7 COVID-19 patients and 2 patients with highly resistant bacteria due to potential transmission concerns). The phase angle was similar between CPAP and HFNC (p = 0.51) (Table 2). In patients with greater respiratory distress (phase angle greater than the median value), CPAP was associated with a significant decrease in thoraco-abdominal phase angle [10.6° (5.6; 20) for CPAP versus 33.5° (24.4–42.7) for HFNC 1, p = 0.03].
Although CPAP was associated with more discomfort (p = 0.02), it did not increase dyspnea (p = 0.15) (Table 2).
The results of the analysis according to ROX score at inclusion is available in the supplement (Table S4).
Discussion
The main results of our study are as follows: (i) tidal volume under HFNC was mostly in a protective range in our series; (ii) tidal volume under low-level CPAP does not accurately predict tidal volume under HFNC; (iii) low-level CPAP has different physiological effects than HFNC when used in patients during de novo AHRF. The original features of our study include the conversion of electrical impedance data into absolute volumes specifically during HFNC. While others have made similar conversions in different settings, this is the first application with HFNC. We also provided a comprehensive physiological comparison of HFNC and low-level CPAP.
Tidal volume under HFNC
We report the largest cohort of patients under HFNC for AHRF with absolute values of tidal volume. Large tidal volumes could represent a significant contributor of P-SILI [3, 26]. In our series, most patients exhibited protective tidal volumes, and only 5 (17%) had tidal volumes greater than 9 mL.kg− 1 PBW. This finding is consistent with previously published smaller cohorts [28].
We found that tidal volume under low-level CPAP failed to accurately predict tidal volume under HFNC. Despite significant correlation between both tidal volumes, Bland-Altman analysis showed substantial bias and broad limits of agreement. Nevertheless, a larger tidal volume during CPAP (> 11.5 mL.kg− 1 PBW) exhibited good sensitivity and specificity for diagnosing a larger high tidal volume under HFNC (> 9 mL.kg− 1 PBW). Whether tidal volume has the same prognostic value under HFNC as under NIV in de novo AHRF remains unknown and deserves future studies. In our series, neither tidal volume under HFNC nor CPAP was associated with the need for intubation, consistent with previous observations [29].
Comparison of the physiological effects of CPAP and HFNC
To our knowledge, this is the first study comparing the detailed physiological effects of HFNC and low-level CPAP in patients with de novo AHRF. Compared to HFNC, low-level CPAP increased tidal volume, minute ventilation, end-expiratory volume, posterior distribution of ventilation, and oxygenation. The rise in EELV and the rise in tidal volume were in the same range. It also decreased signs of respiratory distress in the most severe patients. These physiological effects are important because some CPAP devices, such as certain open valves, which are particularly used in constrained conditions and outside intensive care units [15, 17], typically result in low levels of CPAP.
The increase in tidal volume and minute ventilation under CPAP, despite increased PaCO2, suggests an increase in dead space. This can be attributed to: (1) increased instrumental dead space related to the oronasal mask; (2) disappearance of anatomical dead space washout present under HFNC [28, 30, 31]; and (3) possible increase in alveolar dead space under CPAP. The increase in tidal volume varied significantly among patients (0 to 7.1 mL.kg− 1 PBW). This suggests that while instrumental dead space and loss of anatomical dead space washout are certain, alveolar dead space may increase in some patients and decrease in others. The negative correlation between the difference in tidal volume from HFNC 1 to CPAP and tidal volume under HFNC 1 suggests that dead space washout’s contribution to carbon dioxide clearance under HFNC is prominent when tidal volume is low.
Higher tidal volumes can result from increased respiratory effort and/or decreased respiratory system impedance. Our data suggest alveolar recruitment with CPAP, evidenced by increased EELV and ventilated pixels, which could increase respiratory system compliance. In our study, the thoraco-abdominal phase angle was not significantly different between CPAP and HFNC. Previous studies [18, 32] indicate similar respiratory effort under CPAP and HFNC, suggesting that increased tidal volume under CPAP is accompanied by increased compliance, allowing higher tidal volumes with similar muscle pressure and driving pressure.
Our study demonstrates that low-level CPAP is associated with higher EELV and recruitment compared to HFNC, suggesting higher end-expiratory pressure. HFNC induces a positive expiratory pressure [9, 28, 33], which can exceed 4 cmH2O [34] in healthy volunteers [33, 34] or patients without de novo AHRF [35]. Studies on AHRF patients showed expiratory pressures < 4 cmH2O for gas flows used in clinical practice [10], confirming the impact of breathing pattern on airway pressure [36]. Mouth closure’s impact on airway pressure during HFNC is well established [12, 34]. Our study did not instruct patients to keep their mouths closed, reflecting real-life physiological effects of HFNC. The heterogeneity in end-expiratory lung impedance variation may partly relate to whether the mouth was open or closed. The choice of 4 cmH2O for low-level CPAP is therefore questionable. Whether lower levels of CPAP may better predict tidal volume under HFNC in the clinical scenario warrants further research.
Low-level CPAP improved oxygenation compared to HFNC, consistent with previous studies [29, 32]. This improvement may result from: (i) alveolar recruitment improving ventilation/perfusion ratios; (ii) redistribution of tidal volume to posterior lung zones reducing low ventilation/perfusion areas; and (iii) potentially lower delivered FiO2 during HFNC compared to set FiO2, especially in the case of significant respiratory drive and effort [29, 36, 37].
Limits
Our study has several limitations. First, it is a single-center study with a relatively small number of patients, limiting its power, especially in assessing the prognostic impact of tidal volume. However, our diverse population of de novo AHRF patients, similar tidal volumes under HFNC to other studies [5], and comparable intubation rates to large cohorts [5] tend to make our results generalizable. Second, we did not measure inspiratory effort. The observed increase in EELV, ventilated pixels, dorsal ventilation and improved oxygenation during CPAP could alternatively be explained by increased respiratory effort, leading to intra-tidal recruitment, which may be harmful. Although the findings of decreased Pendelluft and similar levels of dyspnea do not support this explanation, the lack of inspiratory effort measurement precludes definitive conclusions. Third, the physiological effects of prolonged CPAP were not studied. Fourth, we did not standardize mouth closure or opening in order to reflect clinical practice. Fifth, Pendelluft values in our patients were lower than those described elsewhere [18, 19, 38]. This may be due to differences in patient selection and/or discrepancies in pixel-by-pixel impedance data accuracy between the two different EIT devices used. Nevertheless, the interpretation of this parameter’s variation between the two different ventilatory supports remains relevant. Sixth, the volume data from EIT do not cover the entire lung field, possibly missing some localized aeration variations. Seventh, using a pragmatic approach, we included patients who still exhibited hypoxemia and high respiratory rate despite receiving high-flow nasal oxygen. This may constitute an inclusion bias. Finally, the impact of the physiological differences between HFNC and low-level CPAP on patient outcomes cannot be predicted from our study.
Conclusion
Among ICU patients with de novo AHRF, tidal volume under HFNC was mostly protective. Tidal volume during CPAP 4 cmH2O did not predict tidal volume during HFNC 50 to 60 L.min− 1. Low-level CPAP was associated with increased tidal volume, minute ventilation, end-expiratory volume, and oxygenation.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
Not applicable.
Abbreviations
- ARDS
Acute respiratory distress syndrome
- BMI
Body Mass Index
- CPAP
Continuous Positive Airway Pressure
- EELV
End-expiratory lung volume
- FiO2
Inspired fraction of oxygen
- GI
Global inhomogeneity index
- HFNC
High Flow Nasal Cannula
- PaCO2
Arterial carbon dioxide pressure
- PaO2
Arterial oxygen pressure
- PBW
Predicted body weight
- RR
Respiratory rate
- SaO2
Arterial oxygen saturation
- SAPS 2
Simplified Acute Physiology Score 2
- SOFA
Sequential Organ Failure Assessment
- SpO2
Oxygen saturation by pulse oximeter
- Vt
Tidal volume
Author contributions
ST was involved in study design, data collection, analysis and interpretation, and script writing. MAB, AFH, PL and EM were involved in data collection, analysis and interpretation.TM and FP were involved in study design, data collection and analysis. SG and ML were involved in data analysis. EV and AMD were involved in data interpretation and script writing. GC was involved in study design, data analysis and interpretation and script writing. All authors read and approved the final manuscript.
Funding
None.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by an ethic committee “Comité de Protection des Personnes Ile de France 4” (2018-A02972-53). All participants received oral and written information and signed a consent form.
Consent for publication
NA.
Competing interests
AF. Haudebourg reports personal fees from Air Liquide Medical System, Drager, Medtronic and Fisher & Paykel, outside the submitted work. A. Mekontso Dessap reports grants from Fisher and Paykel and fees fom Air Liquide Medical System and Fisher and Paykel, outside the submitted work. G. Carteaux reports grant from Hamilton, personal fees from Air Liquide Medical System, GE Healthcare, Dräger, Fisher and Paykel, Medtronic and Löwenstein, outside the submitted work. Samuel Tuffet, Mohamed Ahmed Boujelben, Tommaso Maraffi, François Perier, Pascale Labedade, Elsa Moncomble, Ségolène Gendreau, Matthieu Lacheny and Emmanuel Vivier report no competing interest.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Carteaux G, Millán-Guilarte T, De Prost N, Razazi K, Abid S, Thille AW, et al. Failure of noninvasive ventilation for De Novo Acute Hypoxemic Respiratory failure: role of tidal Volume*. Crit Care Med. 2016;44(2):282–90. [DOI] [PubMed] [Google Scholar]
- 2.Frat JP, Ragot S, Coudroy R, Constantin JM, Girault C, Prat G, et al. Predictors of intubation in patients with Acute Hypoxemic respiratory failure treated with a Noninvasive Oxygenation Strategy*. Crit Care Med. 2018;46(2):208–15. [DOI] [PubMed] [Google Scholar]
- 3.Brochard L, Slutsky A, Pesenti A. Mechanical ventilation to minimize progression of Lung Injury in Acute Respiratory failure. Am J Respir Crit Care Med. 2017;195(4):438–42. [DOI] [PubMed] [Google Scholar]
- 4.Carteaux G, Perier F, Maraffi T, Razazi K, De Prost N, Mekontso Dessap A. Patient self-inflicted lung injury: Ce que le réanimateur doit connaître. Méd Intensive Réa. 2019;28(1):11–20. [Google Scholar]
- 5.Frat JP, Thille AW, Mercat A, Girault C, Ragot S, Perbet S, et al. High-Flow Oxygen through Nasal Cannula in Acute Hypoxemic Respiratory failure. N Engl J Med. 2015;372(23):2185–96. [DOI] [PubMed] [Google Scholar]
- 6.Ricard JD, Roca O, Lemiale V, Corley A, Braunlich J, Jones P, et al. Use of nasal high flow oxygen during acute respiratory failure. Intensive Care Med. 2020;46(12):2238–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ospina-Tascón GA, Calderón-Tapia LE, García AF, Zarama V, Gómez-Álvarez F, Álvarez-Saa T, et al. Effect of High-Flow Oxygen Therapy vs Conventional Oxygen Therapy on Invasive Mechanical Ventilation and clinical recovery in patients with severe COVID-19: a Randomized Clinical Trial. JAMA. 2021;326(21):2161–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Frat JP, Quenot JP, Badie J, Coudroy R, Guitton C, Ehrmann S, et al. Effect of High-Flow Nasal Cannula Oxygen vs Standard Oxygen Therapy on Mortality in patients with respiratory failure due to COVID-19: the SOHO-COVID Randomized Clinical Trial. JAMA. 2022;328(12):1212–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mauri T, Turrini C, Eronia N, Grasselli G, Volta CA, Bellani G, et al. Physiologic effects of High-Flow Nasal Cannula in Acute Hypoxemic Respiratory failure. Am J Respir Crit Care Med. 2017;01(9):1207–15. [DOI] [PubMed] [Google Scholar]
- 10.Parke RL, Eccleston ML, McGuinness SP. The effects of Flow on Airway pressure during Nasal High-Flow Oxygen Therapy. Respir Care. 2011;56(8):1151–5. [DOI] [PubMed] [Google Scholar]
- 11.Pinkham M, Tatkov S. Effect of flow and cannula size on generated pressure during nasal high flow. Crit Care. 2020;24(1):248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Parke R, McGuinness S, Eccleston M. Nasal high-flow therapy delivers low level positive airway pressure. Br J Anaesth. 2009;103(6):886–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Beaufort E, Carteaux G, Morin F, Lesimple A, Haudebourg A, Fresnel E, et al. A new reservoir-based CPAP with low oxygen consumption: the Bag-CPAP. Crit Care. 2023;27:262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bellani G, Grasselli G, Cecconi M, Antolini L, Borelli M, De Giacomi F, et al. Noninvasive ventilatory support of patients with COVID-19 outside the Intensive Care units (WARd-COVID). Ann Am Thorac Soc. 2021;18(6):1020–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alviset S, Riller Q, Aboab J, Dilworth K, Billy PA, Lombardi Y, et al. Continuous positive Airway pressure (CPAP) face-mask ventilation is an easy and cheap option to manage a massive influx of patients presenting acute respiratory failure during the SARS-CoV-2 outbreak: a retrospective cohort study. PLoS ONE. 2020;15(10):e0240645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mekontso Dessap A, Richard JCM, Baker T, Godard A, Carteaux G. Technical Innovation in critical care in a World of constraints: lessons from the COVID-19 pandemic. Am J Respir Crit Care Med. 2023;207(9):1126–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Carteaux G, Pons M, Morin F, Tuffet S, Lesimple A, Badat B, et al. Continuous positive airway pressure for respiratory support during COVID-19 pandemic: a frugal approach from bench to bedside. Ann Intensive Care. 2021;11(1):38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Menga LS, Delle Cese L, Rosà T, Cesarano M, Scarascia R, Michi T, et al. Respective effects of Helmet pressure support, continuous positive Airway pressure, and Nasal High-Flow in Hypoxemic Respiratory failure: a randomized crossover clinical trial. Am J Respir Crit Care Med. 2023;207(10):1310–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Grieco DL, Delle Cese L, Menga LS, Rosà T, Michi T, Lombardi G, et al. Physiological effects of awake prone position in acute hypoxemic respiratory failure. Crit Care. 2023;27(1):315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Spinelli E, Kircher M, Stender B, Ottaviani I, Basile MC, Marongiu I, et al. Unmatched ventilation and perfusion measured by electrical impedance tomography predicts the outcome of ARDS. Crit Care. 2021;25(1):192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pavlovsky B, Pesenti A, Spinelli E, Scaramuzzo G, Marongiu I, Tagliabue P, et al. Effects of PEEP on regional ventilation-perfusion mismatch in the acute respiratory distress syndrome. Crit Care. 2022;26(1):211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhao Z, Möller K, Steinmann D, Frerichs I, Guttmann J. Evaluation of an electrical impedance tomography-based global inhomogeneity index for pulmonary ventilation distribution. Intensive Care Med. 2009;35(11):1900. [DOI] [PubMed] [Google Scholar]
- 23.Sivan Y, Deakers TW, Newth CJL. Thoracoabdominal Asynchrony in Acute Upper Airway obstruction in small children. Am Rev Respir Dis. 1990;142(3):540–4. [DOI] [PubMed] [Google Scholar]
- 24.Agostoni E, Mognoni P. Deformation of the chest wall during breathing efforts. J Appl Physiol. 1966;21(6):1827–32. [DOI] [PubMed] [Google Scholar]
- 25.Hammer J, Newth CJL. Assessment of thoraco-abdominal asynchrony. Paediatr Respir Rev. 2009;10(2):75–80. [DOI] [PubMed] [Google Scholar]
- 26.Carteaux G, Millán-Guilarte T, De Prost N, Razazi K, Abid S, Thille AW, et al. Failure of noninvasive ventilation for De Novo Acute Hypoxemic Respiratory failure: role of tidal volume. Crit Care Med. 2016;44(2):282–90. [DOI] [PubMed] [Google Scholar]
- 27.Roca O, Caralt B, Messika J, Samper M, Sztrymf B, Hernández G, et al. An Index combining respiratory rate and oxygenation to Predict Outcome of Nasal High-Flow Therapy. Am J Respir Crit Care Med. 2019;199(11):1368–76. [DOI] [PubMed] [Google Scholar]
- 28.Mauri T, Alban L, Turrini C, Cambiaghi B, Carlesso E, Taccone P, et al. Optimum support by high-flow nasal cannula in acute hypoxemic respiratory failure: effects of increasing flow rates. Intensive Care Med. 2017;43(10):1453–63. [DOI] [PubMed] [Google Scholar]
- 29.Dargent A, Hombreux A, Roccia H, Argaud L, Cour M, Guérin C. Feasibility of non-invasive respiratory drive and breathing pattern evaluation using CPAP in COVID-19 patients. J Crit Care. 2022;69:154020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Onodera Y, Akimoto R, Suzuki H, Okada M, Nakane M, Kawamae K. A high-flow nasal cannula system with relatively low flow effectively washes out CO2 from the anatomical dead space in a sophisticated respiratory model made by a 3D printer. Intensive Care Med Exp. 2018;6(1):7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Möller W, Feng S, Domanski U, Franke KJ, Celik G, Bartenstein P, et al. Nasal high flow reduces dead space. J Appl Physiol. 2017;122(1):191–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vargas F, Saint-Leger M, Boyer A, Bui NH, Hilbert G. Physiologic effects of High-Flow nasal cannula oxygen in critical care subjects. Respir Care. 2015;60(10):1369–76. [DOI] [PubMed] [Google Scholar]
- 33.Parke RL, Bloch A, McGuinness SP. Effect of very-high-Flow nasal therapy on Airway pressure and end-expiratory lung impedance in healthy volunteers. Respir Care. 2015;60(10):1397–403. [DOI] [PubMed] [Google Scholar]
- 34.Vieira F, Bezerra FS, Coudroy R, Schreiber A, Telias I, Dubo S, et al. High-flow nasal cannula compared with continuous positive airway pressure: a bench and physiological study. J Appl Physiol. 2022;132(6):1580–90. [DOI] [PubMed] [Google Scholar]
- 35.Parke RL, McGuinness SP. Pressures delivered by nasal high Flow Oxygen during all phases of the respiratory cycle. Respir Care. 2013;58(10):1621–4. [DOI] [PubMed] [Google Scholar]
- 36.Sun YH, Dai B, Peng Y, Tan W, Zhao HW. Factors affecting FiO2 and PEEP during high-flow nasal cannula oxygen therapy: a bench study. Clin Respir J. 2019;13(12):758–64. [DOI] [PubMed] [Google Scholar]
- 37.Chikata Y, Onodera M, Oto J, Nishimura M. FIO2 in an adult Model Simulating High-Flow nasal cannula therapy. Respir Care. 2017;62(2):193–8. [DOI] [PubMed] [Google Scholar]
- 38.Bello G, Giammatteo V, Bisanti A, Delle Cese L, Rosà T, Menga LS, et al. High vs low PEEP in patients with ARDS exhibiting intense Inspiratory Effort during assisted ventilation: a randomized crossover trial. Chest. 2024;165(6):1392–405. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.