Abstract
Background:
Valve-in-valve (ViV) transcatheter aortic valve implantation (TAVI) is quickly becoming a routine and effective means by which to treat degenerated bioprosthetic valves. A known complication of ViV-TAVI is patient-prosthesis mismatch, which substantially affects survival. Bioprosthetic valve fracture is a method by which to reduce the risk of patient-prosthesis mismatch and post–ViV-TAVI transvalvular gradients. This study sought to determine the safety and efficacy of post–ViV-TAVI bioprosthetic valve fracture.
Methods:
Patients with a history of surgical aortic valve replacement undergoing ViV-TAVI bioprosthetic valve fracture (N = 25) at the corresponding institution from 2015 to 2022 were cataloged for a retrospective analysis. The implanted transcatheter valves were Medtronic Evolut R, Evolut PRO, and Evolut PRO+. Gradients were assessed before and after implantation and after fracturing using transthoracic echocardiogram.
Results:
The mean left ventricular ejection fraction of patients who underwent fracturing was 55.04%. The average (SD) peak and mean (SD) transvalvular gradients before the intervention were 68.17 (19.09) mm Hg and 38.98 (14.37) mm Hg, respectively. After ViV-TAVI, the same gradients were reduced to 27.25 (12.27) mm Hg and 15.63 (6.47) mm Hg, respectively. After bioprosthetic valve fracture, the gradients further decreased to 17.59 (7.93) mm Hg and 8.860 (3.334) mm Hg, respectively. The average reduction in peak gradient associated with fracturing was 12.07 mm Hg (95% CI, 5.73-18.41 mm Hg; P = .001). The average reduction in mean gradient associated with valve fracturing was 6.97 mm Hg (95% CI, 3.99-9.74 mm Hg; P < .001).
Conclusion:
Bioprosthetic valve fracture is a viable option for reducing residual transvalvular gradients after ViV-TAVI and should be considered in patients with elevated gradients (>20 mm Hg) or with concern for patient-prosthesis mismatch in patients who have an unacceptable risk for a redo sternotomy and surgical aortic valve replacement.
Keywords: Transcatheter aortic valve replacement, heart valve prosthesis, balloon valvuloplasty
Key Points
A complication of ViV-TAVI is patient-prosthesis mismatch; severe patient-prosthesis mismatch can negatively affect patient survival.
Bioprosthetic valve fracture can reduce patient-prosthesis mismatch and transvalvular gradients.
Bioprosthetic valve fracture is safe to perform and should be considered for use in patients with elevated gradients or concern for patient-prosthesis mismatch.
Introduction
The long-standing standard of care for patients with severe aortic valve stenosis has been surgical aortic valve replacement. In the past 2 decades, there has been a considerable increase in both bioprosthetic and transcatheter valve use as a result of patient and physician preference.1–3 In parallel, it is expected that there will be an upward trend in patients presenting with degeneration of implanted cardiac valves that will require intervention, but redo sternotomy is associated with increased risk and is not possible in all patients.4–6 As a result, valve-in-valve (ViV) transcatheter aortic valve implantation (TAVI) is becoming a routinely performed, less invasive alternative therapy for the treatment of degenerated bioprosthetic heart valves.7–9
Despite feasibility and successful reported outcomes with ViV-TAVI, patient-prosthesis mismatch remains a challenge. Patient-prosthesis mismatch refers to the clinical situation in which an intact and functional prosthetic heart valve does not allow adequate cardiac output given a specific patient’s body size.10 Severe patient-prosthesis mismatch is defined by an indexed effective orifice area smaller than 0.65 cm2/m2 or by a residual transvalvular gradient greater than 20 mm Hg.11–13 Patient-prosthesis mismatch has been associated with decreased New York Heart Association functional class, decreased exercise tolerance, higher rates of hospitalization related to heart failure, accelerated heart valve degeneration, and increased mortality.14,15 The risk of patient-prosthesis mismatch following ViV-TAVI is particularly high because the transcatheter heart valve is implanted within the frame of the failed bioprosthetic valve, preventing complete expansion and restricting the effective orifice area.12,13 The 1-year mortality rate in patients has been shown to be higher in small (<21 mm) and intermediate-sized (<25 mm) valves compared with larger (>25 mm) valves.16 In the Global ViV Registry, the incidence rate of severe patient-prosthesis mismatch after ViV-TAVI was 28.4%.16 Patient-prosthesis mismatch has also been documented as problematic in surgical heart valve replacement because the suturing ring limits the valve size in patients not considered low risk for developing patient-prosthesis mismatch.
The fracturing of implanted bioprosthetic surgical heart valves has emerged as a viable intervention for managing patient-prosthesis mismatch. Fracturing uses high-pressure balloon inflation to dilate, fracture, and expand the bioprosthetic surgical valve, allowing for a more physiologic and patient-appropriate effective orifice area.17–19 Several studies have discussed the safety and efficacy of this technique following ViV-TAVI.17,20,21 The objective of the current study was to determine whether bioprosthetic valve fracturing is a safe and effective method to reduce postprocedural transvalvular gradients.
Patients and Methods
This study was a single-center, institutional review board–approved, retrospective observational analysis of patients with a history of surgical aortic valve replacement who underwent ViV-TAVI with bioprosthetic valve fracture between 2015 and 2022 at Kettering Health Main Campus in Kettering, Ohio. All patients who underwent TAVI in the designated time period were screened. Patients who had undergone ViV-TAVI were selected, and outcomes were assessed based on whether the intervention (fracturing) was performed. The indication for TAVI was symptomatic severe aortic valve stenosis (American College of Cardiology/American Heart Association stage D). The indication for ViV-TAVI was severe symptomatic structural heart valve degeneration (stage 3). Anesthesia was used for all procedures, with the majority of procedures performed under moderate sedation and a select few under general endotracheal anesthesia because of patient-related factors. All patients were of at least a moderate surgical risk for redo sternotomy and surgical aortic valve replacement.
The decision to proceed with bioprosthetic valve fracturing was at the discretion of the operator at the time of valve implantation. The indication for bioprosthetic valve fracture in all patients was suboptimal valve hemodynamics, defined as a residual mean transvalvular gradient above 20 mm Hg, determined by intraoperative transthoracic echocardiography or the inability of the TAVI valve to expand fully. Transvalvular hemodynamics were obtained before and after implantation and after fracturing. The method of valve fracturing was determined by the operator at the time of valve implantation. Bioprosthetic valve fracturing was performed after ViV-TAVI in all cases. In anticipation of possible bioprosthetic valve fracturing in patients undergoing TAVI, all patients were assessed for the virtual valve to coronary distance to determine whether fracturing could safely be pursued, if necessary.
Statistical Analysis
Statistical analysis was performed using SPSS Statistics 28.0 (IBM Corp). Continuous variables were expressed as mean (SD). Categorical variables were expressed as frequencies and percentages. A Shapiro-Wilk test of continuous variables showed no significant departure from normality. A paired t test was performed to compare the post–ViV-TAVI mean and peak pressure gradients with the post–bioprosthetic valve fracturing mean and peak pressure gradients. P < .05 was deemed statistically significant for all analyses.
Results
A total of 78 patients underwent ViV-TAVI, of whom 25 underwent ViV-TAVI with bioprosthetic valve fracturing; these 25 patients were included in this study. Transcatheter heart valves implanted included the Medtronic Evolut R, Evolut PRO, and Evolut PRO+ valves. Bioprosthetic valve fracturing was performed in patients who had undergone initial surgical valve implantation with the Mitroflow (Sorin Group), Mosaic Ultra (Medtronic), Magna (Edwards Lifesciences), Magna Ease (Edwards Lifesciences), and PERIMOUNT 2800 (Edwards Lifesciences) valves. The mean (SD) age of the patients who underwent bioprosthetic value fracture was 74.4 (9.37) years (range, 56-90 years); the mean (SD) left ventricular ejection fraction in these patients was 55.04% (11.74%). The average (SD) age of the initial surgical valve at the time of ViV-TAVI with bioprosthetic valve fracturing was 9.88 (3.98) years. See Table I for a complete list of patient characteristics and valve details.
Table I.
Patient Characteristics and Heart Valve Information
| Patient | Age, y | Sex | Left ventricular ejection fraction, % | Surgical aortic valve replacement valve, model(manufacturer) | Surgical aortic valve replacement size, mm | Age of surgical aortic valve replacement, y | TAVI valve, model (manufacturer) | TAVI size, mm | Bioprosthetic valve fracture balloon, model (manufacturer) | Bioprosthetic valve fracture balloon size, mm | Bioprosthetic valve fracture balloon pressure, atm |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 75 | Female | 57.5 | Mitroflow (Sorin Group) | 19 | 8 | Evolut PRO (Medtronic) | 23 | TRUE (BARD Medical) | 20 | 16 |
| 2 | 80 | Female | 62.5 | Mitroflow (Sorin Group) | 21 | 5 | Evolut R (Medtronic) | 23 | TRUE (BARD Medical) | 20 | 12 |
| 3 | 78 | Female | 57.5 | Mitroflow (Sorin Group) | 21 | 11 | Evolut PRO+ (Medtronic) | 23 | TRUE (BARD Medical) | 20 | 15 |
| 4 | 87 | Female | 57.5 | Mitroflow (Sorin Group) | 23 | 6 | Evolut PRO (Medtronic) | 23 | TRUE (BARD Medical) | 24 | 15 |
| 5 | 63 | Female | 57.5 | Mitroflow (Sorin Group) | 23 | 8 | Evolut PRO (Medtronic) | 23 | Z-MED II (B. Braun Interventional Systems Inc) | 20 | 13 |
| 6 | 77 | Male | 59 | Mitroflow (Sorin Group) | 23 | 8 | Evolut PRO+ (Medtronic) | 26 | TRUE (BARD Medical) | 20 | 14 |
| 7 | 80 | Male | 46 | Mitroflow (Sorin Group) | 25 | 12 | Evolut PRO+ (Medtronic) | 26 | TRUE (BARD Medical) | 22 | 16 |
| 8 | 79 | Female | 67 | Magna Ease (Edwards Lifesciences) | 21 | 12 | Evolut R (Medtronic) | 23 | TRUE (BARD Medical) | 23 | 16 |
| 9 | 69 | Female | 56 | Magna Ease (Edwards Lifesciences) | 23 | 6 | Evolut R (Medtronic) | 23 | TRUE (BARD Medical) | 23 | 14 |
| 10 | 62 | Female | 62 | Magna Ease (Edwards Lifesciences) | 23 | 8 | Evolut PRO+ (Medtronic) | 26 | TRUE (BARD Medical) | 22 | 10 |
| 11 | 77 | Female | 55 | Magna Ease (Edwards Lifesciences) | 23 | 15 | Evolut PRO+ (Medtronic) | 26 | Z-MED II (B. Braun Interventional Systems Inc) | 20 | 10 |
| 12 | 71 | Male | 65 | Magna Ease (Edwards Lifesciences) | 25 | 3 | Evolut R (Medtronic) | 29 | TRUE (BARD Medical) | 26 | 14 |
| 13 | 56 | Male | 35 | Magna Ease (Edwards Lifesciences) | 25 | 7 | Evolut PRO (Medtronic) | 26 | TRUE (BARD Medical) | 26 | 14 |
| 14 | 90 | Male | 57.5 | Magna Ease (Edwards Lifesciences) | 25 | 18 | Evolut R (Medtronic) | 29 | TRUE (BARD Medical) | 24 | 16 |
| 15 | 67 | Male | 20 | Magna Ease (Edwards Lifesciences) | 27 | 6 | Evolut PRO+ (Medtronic) | 29 | TRUE (BARD Medical) | 22 | 18 |
| 16 | 73 | Female | 67 | Magna (Edwards Lifesciences) | 21 | 11 | Evolut R (Medtronic) | 23 | TRUE (BARD Medical) | 21 | 18 |
| 17 | 74 | Male | 57.5 | Magna (Edwards Lifesciences) | 23 | 14 | Evolut PRO+ (Medtronic) | 26 | TRUE (BARD Medical) | 21 | 10 |
| 18 | 60 | Male | 27.5 | Magna (Edwards Lifesciences) | 25 | 10 | Evolut PRO+ (Medtronic) | 26 | TRUE (BARD Medical) | 22 | 16 |
| 19 | 85 | Male | 57.5 | Magna (Edwards Lifesciences) | 25 | 16 | Evolut PRO+ (Medtronic) | 29 | TRUE (BARD Medical) | 24 | 16 |
| 20 | 64 | Male | 68 | PERIMOUNT 2800 (Edwards Lifesciences) | 23 | 9 | Evolut PRO (Medtronic) | 23 | TRUE (BARD Medical) | 22 | 12 |
| 21 | 90 | Male | 60 | PERIMOUNT 2800 (Edwards Lifesciences) | 23 | 10 | Evolut R (Medtronic) | 26 | TRUE (BARD Medical) | 21 | 13 |
| 22 | 79 | Male | 48 | PERIMOUNT 2800 (Edwards Lifesciences) | 25 | 11 | Evolut R (Medtronic) | 29 | TRUE (BARD Medical) | 24 | 13 |
| 23 | 70 | Male | 57 | PERIMOUNT 2800 (Edwards Lifesciences) | 25 | 16 | Evolut R (Medtronic) | 26 | TRUE (BARD Medical) | 24 | 16 |
| 24 | 86 | Female | 57.5 | Mosaic Ultra (Medtronic) | 21 | 13 | Evolut R (Medtronic) | 23 | TRUE (BARD Medical) | 20 | 13 |
| 25 | 69 | Male | 61 | Mosaic Ultra (Medtronic) | 23 | 4 | Evolut R (Medtronic) | 23 | TRUE (BARD Medical) | 22 | 16 |
TAVI, transcatheter aortic valve implantation.
The average (SD) peak and mean (SD) transvalvular gradients before intervention were 68.17 (19.09) mm Hg and 38.98 (14.37) mm Hg, respectively. After ViV-TAVI, the same gradients were reduced to 27.25 (12.27) mm Hg and 15.63 (6.47), respectively. After ViV-TAVI with bioprosthetic valve fracturing, the gradients decreased to 17.59 (7.93) mm Hg and 8.860 (3.334) mm Hg, respectively (Fig. 1). The average reduction in peak pressure gradient associated with fracturing was 12.07 mm Hg (95% CI, 5.73-18.41 mm Hg; P = .001). The average reduction in mean pressure gradient associated with fracturing was 6.97 mm Hg (95% CI, 3.99-9.74 mm Hg; P < .001) (Fig. 1). The gradients remained within acceptable limits (<20 mm Hg) at 1 month and 12 months postoperatively; the mean (SD) pressure gradient in the ViV-TAVI bioprosthetic valve fracturing group was 13.2 (4.31) mm Hg at 1 month and 12.46 (5.29) mm Hg at 12 months. See Table II for a complete list of hemodynamic data. The post–ViV-TAVI gradient was not obtained for 1 patient. Following ViV-TAVI implantation, the newly implanted heart valve was constrained by the bioprosthetic valve, so bioprosthetic valve fracturing was pursued before gradients could be obtained. In addition, fracturing that was performed because of a visible waist on the TAVI valve on fluoroscopy did not result in a reduction in transvalvular gradients following fracture but rather in a visual improvement of valve expansion, as confirmed by the operator.
Fig. 1.
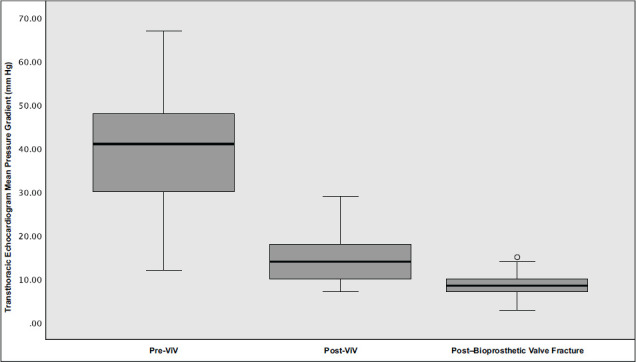
Effects of post–ViV-TAVI bioprosthetic valve fracturing on mean transvalvular gradient are shown. The mean (SD) transvalvular gradient before ViV-TAVI was 38.98 (14.37) mm Hg. After ViV-TAVI, the mean (SD) gradient was reduced to 14.159 (5.95) mm Hg. After ViV-TAVI with fracturing, the mean (SD) gradient was further reduced to 8.860 (3.334) mm Hg. The average reduction in mean pressure gradient associated with fracturing was 5.136 mm Hg (95% CI, 2.768-7.505 mm Hg; P < .001). P < .05 was considered statistically significant.
ViV, valve-in-valve.
Table II.
Preoperative and Postoperative Mean Transaortic Pressure Gradients in ViV-TAVI Bioprosthetic Valve Fracture
| Patient | Baseline mean gradient, mm Hg | After TAVI, mm Hg | After bioprosthetic valve fracture, mm Hg | 1-mo follow-up, mm Hg | 12-mo follow-up, mm Hg |
|---|---|---|---|---|---|
| 1 | 43 | 12 | 9 | 25 | 23 |
| 2 | 42 | 7 | 5 | 10 | 6 |
| 3 | 40 | 8 | 8 | 19 | 20 |
| 4 | 12 | 21 | 15 | 9 | 11 |
| 5 | 45 | 10 | 7 | 22 | 25 |
| 6 | 46 | 7 | 7 | 10 | 11 |
| 7 | 30 | 15 | 14 | 13 | 14 |
| 8 | 49 | 15 | 9 | 14 | 15 |
| 9 | 44 | 12 | 13 | 17 | 19 |
| 10 | 67 | 15 | 10 | 14 | 13 |
| 11 | 33 | 12 | 7 | 11 | 13 |
| 12 | 47 | 28 | 10 | 9 | N/A |
| 13 | 54 | 17 | 3 | 16 | 17 |
| 14 | 51 | 15 | 7 | 8 | 7 |
| 15 | 19 | 12 | 9 | 11 | 12 |
| 16 | 37 | 18 | 11 | 9 | 9 |
| 17 | 27 | 10 | 7 | 10 | 8 |
| 18a | 40 | 18 | 8 | 14 | 8 |
| 19 | 37 | 10 | 6 | 9 | 10 |
| 20 | 41 | 28 | 12 | 14 | 10 |
| 21a | 50 | 23 | 11 | 17 | 14 |
| 22 | 60 | 20 | 11 | 13 | 7 |
| 23 | 48 | 29 | 8 | 9 | 11 |
| 24 | 41 | 13 | 8 | 14 | 12 |
| 25a | 32 | N/A | 4 | 13 | 4 |
N/A, not applicable; TAVR, transcatheter aortic valve implantation.
SI conversion factor: To convert mm Hg to kPa, multiply by 0.133.
Post-TAVI gradients were not obtained for patients 18, 21, and 25 because the TAVI valve could not fully expand secondary to restriction by the bioprosthetic valve. Bioprosthetic valve fracturing was therefore promptly pursued.
There were no major immediate postoperative complications (aortic root rupture, left ventricular perforation, coronary occlusion, myocardial infarction, or death). Only 1 patient required permanent pacemaker implantation for complete heart block. There was no substantial perivalvular or valvular regurgitation in any case. There was a 96% one-year survival rate in the ViV-TAVI bioprosthetic valve fracturing cohort; the single mortality in this group was attributed to a noncardiac cause. There was a 91% three-year survival rate in the ViV-TAVI bioprosthetic valve fracturing cohort; the additional instance of mortality was also attributed to a noncardiac cause.
Discussion
This study found that post–ViV-TAVI bioprosthetic valve fracture appears to be a safe and effective option for the reduction of residual transvalvular gradients. Fracturing of the original surgical valves resulted in a statistically significant reduction in peak and mean transvalvular gradients. There were some individual patients for whom there was no change in gradient following fracturing. In these specific patients, the indication to perform bioprosthetic valve fracturing was TAVI “waisting” visible on intraoperative fluoroscopy, suggesting that the valve was constrained without compromising gradients. Following fracturing, the waisting resolved without a decrease in gradients. Because of the frequency of TAVI valve restriction by the bioprosthetic valve following deployment, it is important to have fracturing readily available in centers performing ViV-TAVI to rapidly allow for the full expansion of the TAVI valve. Bioprosthetic valve fracturing allows for a more ideal physiologic size match between the patient and the bioprosthesis, reducing the risk for patient-prosthesis mismatch and improving valve longevity and function.11
Patient selection and preprocedural planning are critical because not all valves are amenable to fracturing or remodeling.22,23 Valve fracturing, also known as valve “fracking,” refers to the act of dilating the existing ring of a surgical heart valve until fracture occurs (Fig. 2), allowing for unrestricted expansion of a new transcatheter valve. Valve remodeling refers to surgical heart valves that cannot be fractured but can be stretched or bent to a larger diameter, which also allows implantation of larger transcatheter valves. Prosthetic heart valves that can be fractured include the Magna, Magna Ease, PERIMOUNT 2800, Mitroflow (CarboMedics), Mosaic, Biocor (St Jude Medical), and Epic (Abbott) valves.22,23 Valves that can be remodeled or modified include the Carpentier-Edwards Standard (Edwards Lifesciences), Carpentier-Edwards Supra-Annular Valve (Edwards Lifesciences), PERIMOUNT 2700 (Edwards Lifesciences), INSPIRIS (Edwards Lifesciences), and Trifecta (Abbott) valves.22,23 The Carpentier-Edwards Standard, Supra-Annular Valve, and PERIMOUNT 2700 valves have expandable valve rings. The INSPIRIS valve has an expandable valve ring joint. The Trifecta valve has a nonexpandable titanium valve ring but has valve posts that can be bent or modified to allow for ViV-TAVI. Valves that cannot be fractured or remodeled include the Hancock II (Medtronic) and the Avalus (Medtronic) valves.22,23
Fig. 2.
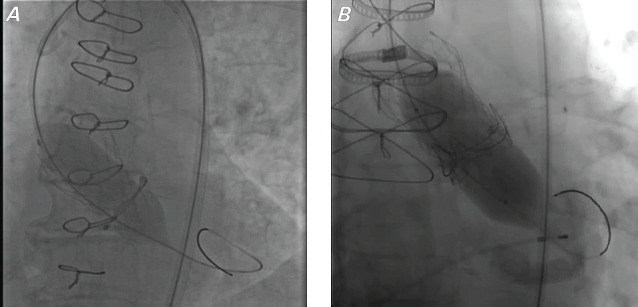
Cinefluoroscopy images of surgical valve valvuloplasty show (A) the initial inflation of a valvuloplasty balloon after ViV-TAVI, with notable balloon “waisting” that suggests that the original surgical valve was not fractured or modified sufficiently, and (B) a fully expanded valvuloplasty balloon with a fractured surgical valve ring after ViV-TAVI with bioprosthetic valve fracture.
TAVI, transcatheter aortic valve implantation; ViV, valve-in-valve.
Fracturing is performed by high-pressure inflation of a noncompliant valvuloplasty balloon. Patient selection and preprocedural planning are important to avoid the increased risk of coronary obstruction associated with ViV-TAVI. Observance of the virtual transcatheter heart valve to coronary distance is critical because one must anticipate a 3-mm to 4-mm increase in the diameter of the surgical valve after fracturing.22 Patients who have a virtual valve to coronary distance less than 3 mm are at the highest risk of coronary obstruction. It is preferred to have a valve to coronary distance greater than 6 mm.22 Another factor to consider is the height of the sinotubular junction. Preoperative analysis by computed tomography angiography with virtual valve assessments should be performed to rule out leaflet sinotubular junction sequestration and coronary obstruction. Valve-in-valve TAVI valve size selection is also important and should consider the true internal diameter of the surgical heart valve, with an increase of 3 mm to 4 mm to account for fracturing.22 Valvuloplasty balloon sizing may vary and depends on the ViV-TAVI valve. When using a SAPIEN 3 (Edwards Lifesciences) transcatheter heart valve, the sizing ratio of the valvuloplasty balloon to the transcatheter heart valve is 1:1.22 When using the CoreValve (Medtronic) transcatheter heart valve platform, the valvuloplasty balloon must be no larger than 2 mm greater than the width of the constrained area because of the risk of damage to the valve leaflet stent adhesion point.22 It is also critical to position the shoulder of the balloon at or below the constrained area to minimize the risk of leaflet damage. Maximum inflator pressure will vary based on the type of surgical valve that is being fractured (Table III).
Table III.
Protocol and Comments for Bioprosthetic Valve Fracturing
| Equipment | Basic steps | Comments |
|---|---|---|
|
|
|
Discussion continues regarding when to perform bioprosthetic valve fracturing because performing the procedure before and after ViV-TAVI is each associated with different risks and benefits. Performing fracturing before ViV-TAVI allows implantation of a larger transcatheter valve and reduces the risk of trauma to the new valve, but it is associated with an increased risk of debris embolization and acute severe aortic valve regurgitation. Performing fracturing after ViV-TAVI ensures full valve expansion but exposes the new valve to high-pressure dilation, which may result in damage to the valve and affect its longevity. The current recommendation is to fracture the bioprosthetic valve after ViV-TAVI to optimize safety while still allowing for full expansion of the valve.21,22 Recent studies have highlighted the use of the Boston Scientific SENTINEL Cerebral Embolic Protection system during TAVI, although the PROTECTED TAVR trial showed that routine use of a cerebral protection device does not result in a lower risk of stroke within 72 hours among patients undergoing transfemoral TAVI for aortic valve stenosis.24 In the current study, all patients undergoing ViV-TAVI, regardless of fracturing status, were protected using the SENTINEL Cerebral Embolic Protective system. No procedural cerebrovascular events were noted in the ViV-TAVI or post–ViV-TAVI bioprosthetic valve fracturing cohorts. Although the current study had no embolic cerebrovascular events, its cohort size was small, and additional studies are warranted. Recently analyzed registry data suggest that bioprosthetic valve fracture may also be associated with increased in-hospital mortality.25
Although this dataset was small, the study’s findings are consistent with findings reported in larger studies.25,26 Consolidating the currently available data and the findings detailed in this report, the analysis suggests that post–ViV-TAVI fracturing can be performed safely in select patients, resulting in lower residual transvalvular gradients and larger effective valve areas.25,26 This finding is important because contemporary data suggest patient-prosthesis mismatch is associated with smaller valve size.26
Study Limitations
Because of the retrospective nature of this analysis and the use of data from a single center, this study may not be representative of the general population. There may have also been unmeasured confounding factors and selection bias. Clinical factors such as hemodynamic status, variability in echocardiographic techniques, and measurement variation are also subjective.
Conclusion
Bioprosthetic valve fracture is a viable option for the reduction of residual transvalvular gradients after ViV-TAVI and should be considered in patients with postimplantation residual gradients or anticipated patient-prosthesis mismatch. Improvement in transvalvular gradients has been linked to both clinically and statistically significant symptom reduction and long-term mortality outcomes. Future research is needed to develop guidelines on desired postimplantation transvalvular gradients.
Abbreviations and Acronyms
- TAVI
transcatheter aortic valve implantation
- ViV
valve-in-valve
Article Information
Open Access: © 2024 The Authors. Published by The Texas Heart Institute®. This is an Open Access article under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC, https://creativecommons.org/licenses/by-nc/4.0/), which permits use and distribution in any medium, provided the original work is properly cited, and the use is noncommercial.
Author Contributions: Osama Hallak, MD, general cardiology fellow, performed the literature review and drafted the manuscript, formatted the images and figures, and formatted the manuscript and prepared it for submission. Karley Fischer, MD, internal medicine resident, performed the literature review, drafted the manuscript, and formatted the images and figures. Shaina Ailawadi, MD, internal medicine resident, performed the literature review, drafted the manuscript, and formatted the images and figures. Damian Valencia, MD, interventional fellow, performed the procedures, collected and analyzed hemodynamic data, and performed a preliminary review and editing of the manuscript. Yan Yatsynovich, MD, interventional fellow, performed the procedures, collected and analyzed hemodynamic data, and performed a preliminary review and editing of the manuscript. Raja Nazir, MD, attending interventional-structural cardiologist and co-principal investigator, performed the procedures, supervised the study, and performed the final review of the manuscript. Brian Schwartz, MD, senior author, attending interventional-structural cardiologist, and co-principal investigator, performed the procedures, supervised the study, and performed the final review of manuscript.
Conflict of Interest Disclosure: The authors report no financial relationships or conflicts of interest regarding the content herein.
Funding/Support: None.
References
- 1.Thourani VH, Suri RM, Gunter RL, et al. Contemporary real-world outcomes of surgical aortic valve replacement in 141,905 low-risk, intermediate-risk, and high-risk patients. Ann Thorac Surg. 2015;99(1):55–61. doi: 10.1016/j.athoracsur.2014.06.050. doi: [DOI] [PubMed] [Google Scholar]
- 2.Isaacs AJ, Shuhaiber J, Salemi A, Isom OW, Sedrakyan A. National trends in utilization and in-hospital outcomes of mechanical versus bioprosthetic aortic valve replacements. J Thorac Cardiovasc Surg. 2015;149(5):1262–1269.e3. doi: 10.1016/j.jtcvs.2015.01.052. doi: [DOI] [PubMed] [Google Scholar]
- 3.Brennan JM, Edwards FH, Zhao Y, et al. DEcIDE AVR (Developing Evidence to Inform Decisions about Effectiveness–Aortic Valve Replacement) Research Team. Long-term safety and effectiveness of mechanical versus biologic aortic valve prostheses in older patients: results from the Society of Thoracic Surgeons Adult Cardiac Surgery National Database. Circulation. 2013;127(16):1647–1655. doi: 10.1161/CIRCULATIONAHA.113.002003. doi: [DOI] [PubMed] [Google Scholar]
- 4.Onorati F, Biancari F, De Feo M, et al. Mid-term results of aortic valve surgery in redo scenarios in the current practice: results from the multicentre European RECORD (REdo Cardiac Operation Research Database) initiative. Eur J Cardiothorac Surg. 2015;47(2):269–280. doi: 10.1093/ejcts/ezu116. doi: [DOI] [PubMed] [Google Scholar]
- 5.Maganti M, Rao V, Armstrong S, Feindel CM, Scully HE, David TE. Redo valvular surgery in elderly patients. Ann Thorac Surg. 2009;87(2):521–525. doi: 10.1016/j.athoracsur.2008.09.030. doi: [DOI] [PubMed] [Google Scholar]
- 6.Balsam LB, Grossi EA, Greenhouse DG, et al. Reoperative valve surgery in the elderly: predictors of risk and long-term survival. Ann Thorac Surg. 2010;90(4):1195–1200. doi: 10.1016/j.athoracsur.2010.04.057. doi: [DOI] [PubMed] [Google Scholar]
- 7.Adams DH, Popma JJ, Reardon MJ, et al. US CoreValve Clinical Investigators. Transcatheter aortic-valve replacement with a self-expanding prosthesis. N Engl J Med. 2014;370(19):1790–1798. doi: 10.1056/NEJMoa1400590. doi: [DOI] [PubMed] [Google Scholar]
- 8.Leon MB, Smith CR, Mack M, et al. PARTNER Trial Investigators. Transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. N Engl J Med. 2010;363(17):1597–1607. doi: 10.1056/NEJMoa1008232. doi: [DOI] [PubMed] [Google Scholar]
- 9.Smith CR, Leon MB, Mack MJ, et al. PARTNER Trial Investigators. Transcatheter versus surgical aortic-valve replacement in high-risk patients. N Engl J Med. 2011;364(23):2187–2198. doi: 10.1056/NEJMoa1103510. doi: [DOI] [PubMed] [Google Scholar]
- 10.Rahimtoola SH. The problem of valve prosthesis-patient mismatch. Circulation. 1978;58(1):20–24. doi: 10.1161/01.cir.58.1.20. doi: [DOI] [PubMed] [Google Scholar]
- 11.Dayan V, Vignolo G, Soca G, Paganini JJ, Brusich D, Pibarot P. Predictors and outcomes of prosthesis-patient mismatch after aortic valve replacement. JACC Cardiovasc Imaging. 2016;9(8):924–933. doi: 10.1016/j.jcmg.2015.10.026. doi: [DOI] [PubMed] [Google Scholar]
- 12.Sathananthan J, Sellers S, Barlow AM, et al. Valve-in-valve transcatheter aortic valve replacement and bioprosthetic valve fracture comparing different transcatheter heart valve designs: an ex vivo bench study. JACC Cardiovasc Interv. 2019;12(1):65–75. doi: 10.1016/j.jcin.2018.10.043. doi: [DOI] [PubMed] [Google Scholar]
- 13.Sathananthan J, Fraser R, Hatoum H, et al. A bench test study of bioprosthetic valve fracture performed before versus after transcatheter valve-in-valve intervention. EuroIntervention. 2020;15(16):1409–1416. doi: 10.4244/EIJ-D-19-00939. doi: [DOI] [PubMed] [Google Scholar]
- 14.Pibarot P, Simonato M, Barbanti M, et al. Impact of pre-existing prosthesis-patient mismatch on survival following aortic valve-in-valve procedures. JACC Cardiovasc Interv. 2018;11(2):133–141. doi: 10.1016/j.jcin.2017.08.039. doi: [DOI] [PubMed] [Google Scholar]
- 15.Pibarot P, Magne J, Leipsic J, et al. Imaging for predicting and assessing prosthesis-patient mismatch after aortic valve replacement. JACC Cardiovasc Imaging. 2019;12(1):149–162. doi: 10.1016/j.jcmg.2018.10.020. doi: [DOI] [PubMed] [Google Scholar]
- 16.Dvir D, Webb J, Brecker S, et al. Transcatheter aortic valve replacement for degenerative bioprosthetic surgical valves: results from the global valve-in-valve registry. Circulation. 2012;126(19):2335–2344. doi: 10.1161/CIRCULATIONAHA.112.104505. doi: [DOI] [PubMed] [Google Scholar]
- 17.Nielsen-Kudsk JE, Christiansen EH, Terkelsen CJ, et al. Fracturing the ring of small Mitroflow bioprostheses by high-pressure balloon predilatation in transcatheter aortic valve-in-valve implantation. Circ Cardiovasc Interv. 2015;8(8):e002667. doi: 10.1161/CIRCINTERVENTIONS.115.002667. doi: [DOI] [PubMed] [Google Scholar]
- 18.Brown SC, Cools B, Gewillig M. Cracking a tricuspid PERIMOUNT bioprosthesis to optimize a second transcatheter SAPIEN valve-in-valve placement. Catheter Cardiovasc Interv. 2016;88(3):456–459. doi: 10.1002/ccd.26507. doi: [DOI] [PubMed] [Google Scholar]
- 19.Tanase D, Grohmann J, Schubert S, Uhlemann F, Eicken A, Ewert P. Cracking the ring of Edwards PERIMOUNT bioprosthesis with ultrahigh pressure balloons prior to transcatheter valve in valve implantation. Int J Cardiol. 2014;176(3):1048–1049. doi: 10.1016/j.ijcard.2014.07.175. doi: [DOI] [PubMed] [Google Scholar]
- 20.Allen KB, Chhatriwalla AK, Saxon JT, Sathananthan J, Dvir D, Webb JG. Bioprosthetic valve fracture to facilitate valve-in-valve transcatheter aortic valve repair. Ann Cardiothorac Surg. 2020;9(6):528–530. doi: 10.21037/acs-2020-av-20. doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Allen KB, Chhatriwalla AK, Saxon JT, et al. Bioprosthetic Valve Fracture Investigators. Bioprosthetic valve fracture: technical insights from a multicenter study. J Thorac Cardiovasc Surg. 2019;158(5):1317–1328.e1. doi: 10.1016/j.jtcvs.2019.01.073. doi: [DOI] [PubMed] [Google Scholar]
- 22.Allen KB, Chhatriwalla AK, Saxon JT, et al. Bioprosthetic valve fracture: a practical guide. Ann Cardiothorac Surg. 2021;10(5):564–570. doi: 10.21037/acs-2021-tviv-25. doi: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chhatriwalla AK, Sorajja P. Expanding indications for bioprosthetic valve fracture and bioprosthetic valve remodeling. Circ Cardiovasc Interv. 2018;11(8):e007017. doi: 10.1161/CIRCINTERVENTIONS.118.007017. doi: [DOI] [PubMed] [Google Scholar]
- 24.Kapadia SR, Makkar R, Leon M, et al. PROTECTED TAVR Investigators. Cerebral embolic protection during transcatheter valve replacement. N Engl J Med. 2022;387(14):1253–1263. doi: 10.1056/NEJMoa2204961. doi: [DOI] [PubMed] [Google Scholar]
- 25.Garcia S, Bapat V, Depta J, et al. Paper presented at: Transcatheter Cardiovascular Therapeutics Congress; Boston, MA: Sep 16-19, 2022. Frequency and safety of bioprosthetic valve fracture in patients undergoing valve in valve TAVR for failed surgical valves using the SAPIEN 3/Ultra Valves: Insights from real-world data. [Google Scholar]
- 26.Chhatriwall AK, Allen KB, Saxon JT, et al. Bioprosthetic valve fracture improves the hemodynamic results of valve-in-valve transcatheter aortic valve replacement. 2017;10(7):e005216. doi: 10.1161/CIRCINTERVENTIONS.117.005216. [DOI] [PubMed] [Google Scholar]


