Abstract
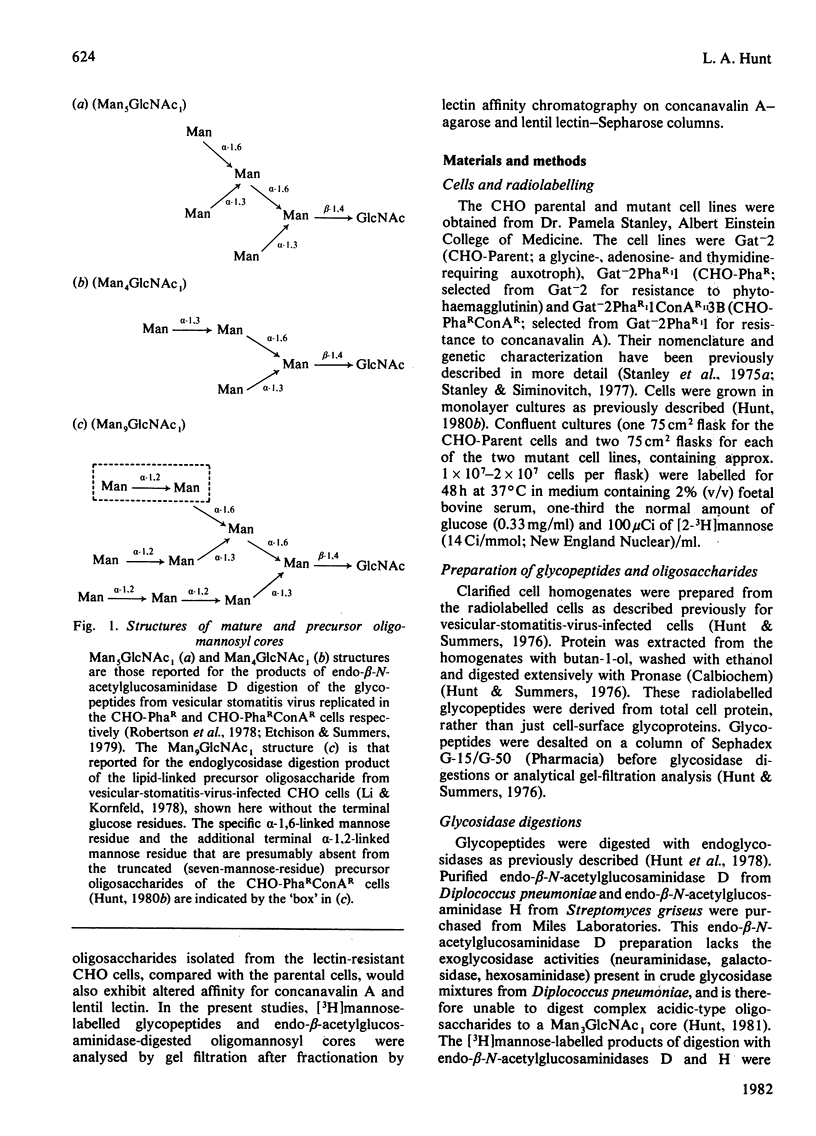
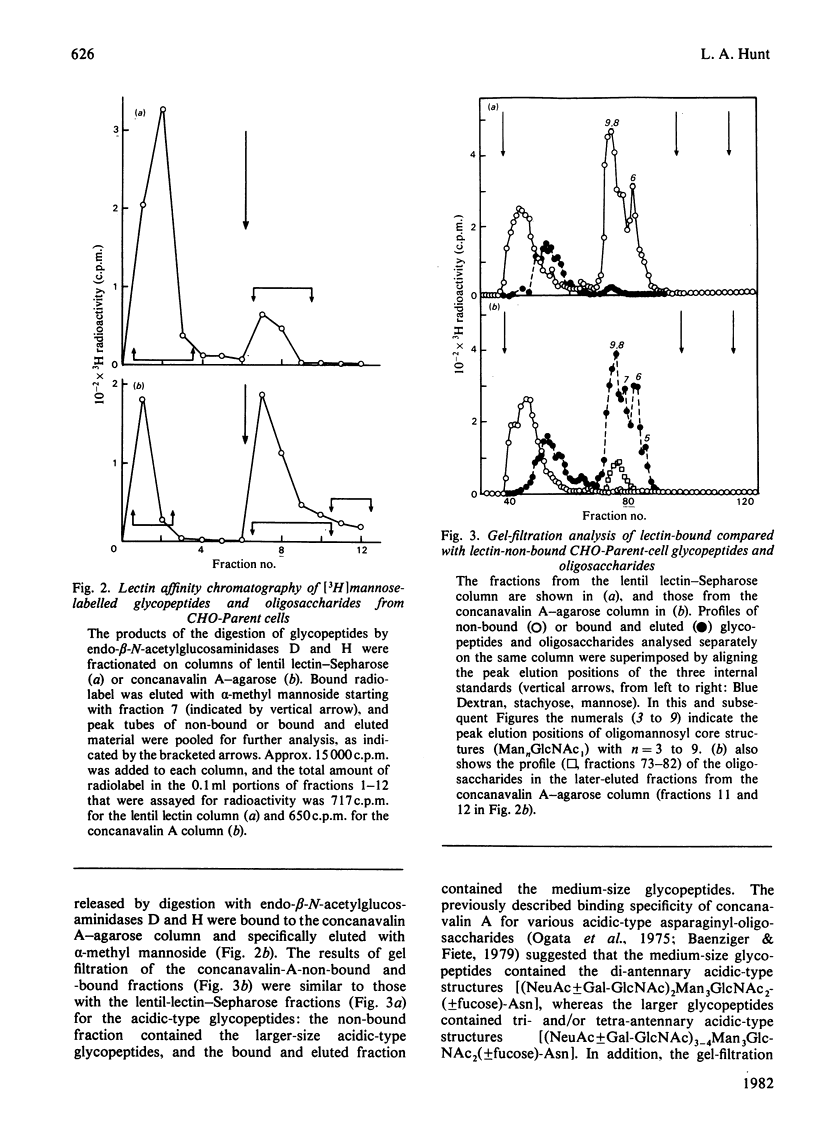
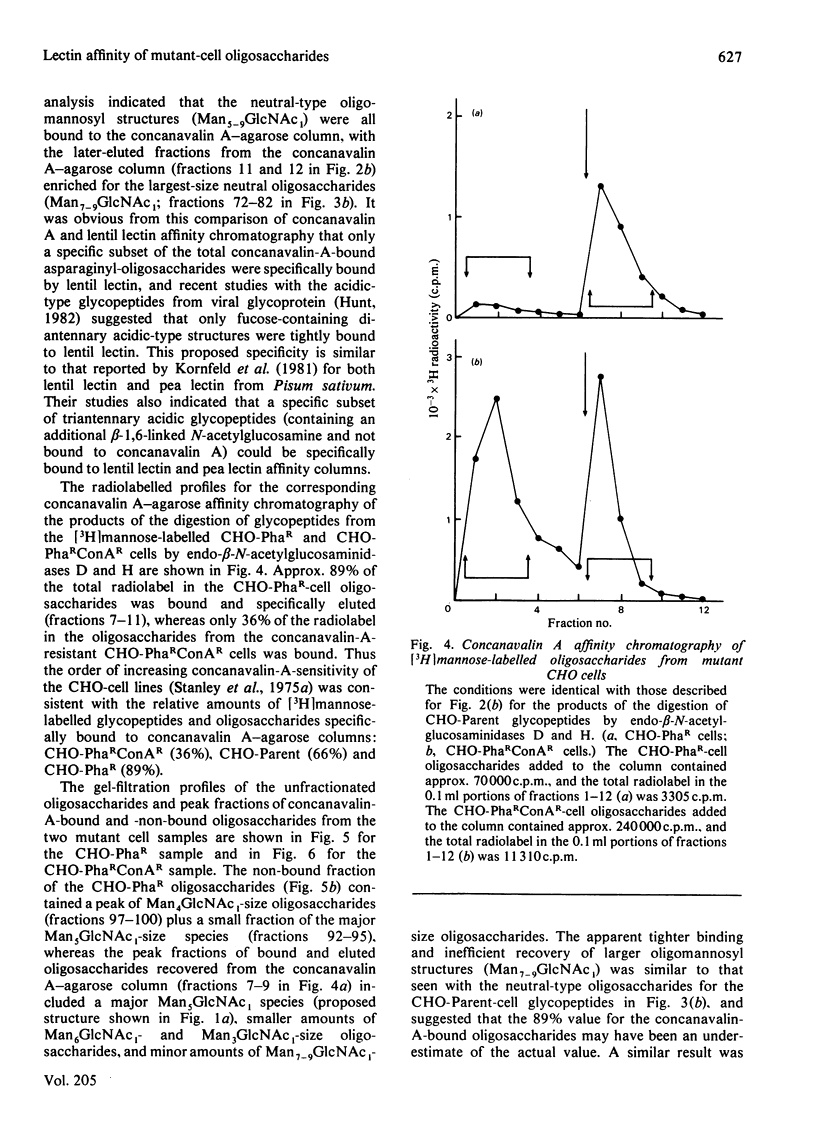
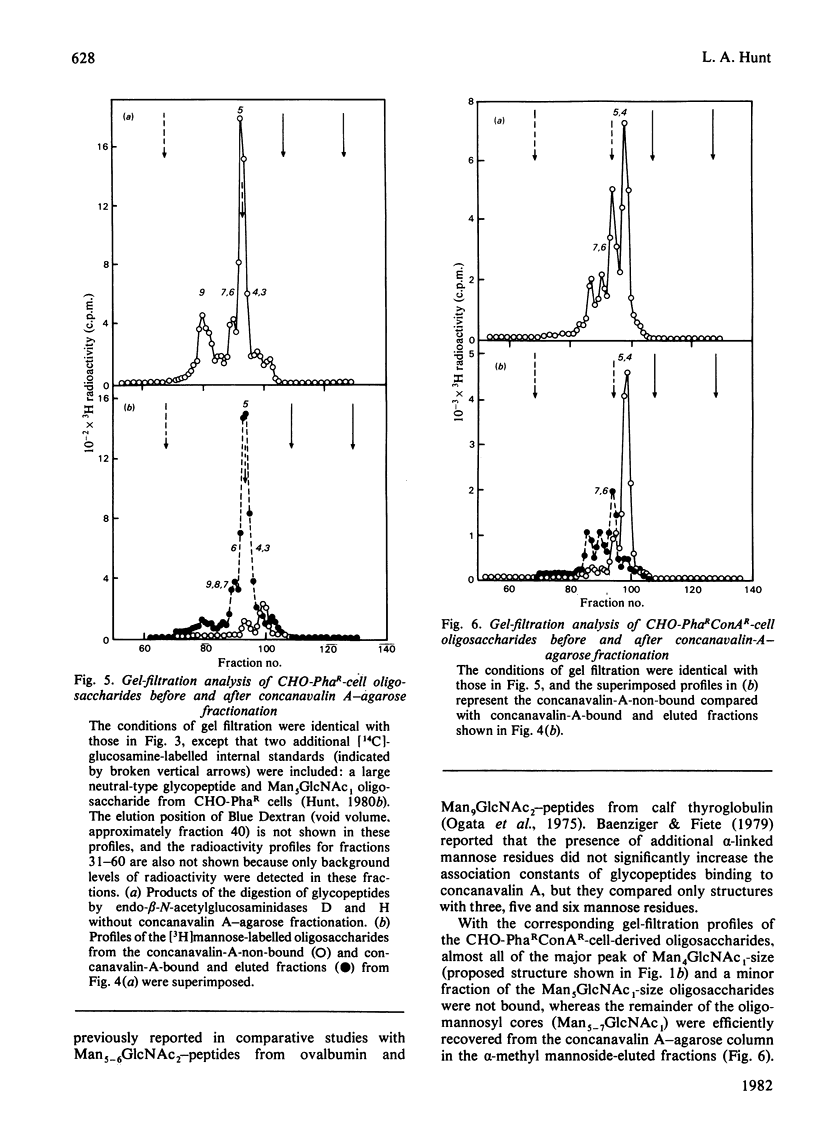
The [3H]mannose-labelled glycopeptides from two lectin-resistant lines of Chinese-hamster ovary cells were fractionated by chromatography on lentil lectin-Sepharose and concanavalin A-agarose columns and subsequently analysed by gel filtration in comparison with the glycopeptides of the parental cell line. Essentially all of the [3H]mannose-labelled asparaginyl-oligosaccharides from the 'single-mutant' cells selected for resistance to phytohaemagglutinin and the 'double-mutant' cells selected for additional resistance to concanavalin A were not bound to lentil lectin, whereas approximately one-sixth of the parental-cell glycopeptides were bound and specifically eluted with alpha-methyl mannoside. These bound and eluted glycopeptides represented a specific subset of the complex acidic-type asparaginyl-oligosaccharides. The percentage of radiolabelled glycopeptides and oligosaccharides from each cell line that were specifically bound to concanavalin A was consistent with the relative sensitivities of the three cell lines to this lectin. The major radiolabelled species in the endoglycosidase digest of the 'double-mutant'-cell glycopeptides (Man4GlcNAc1-size neutral oligosaccharides) were not bound to concanavalin A, whereas essentially all of the other neutral-type oligosaccharides were bound. In addition, the larger neutral-type oligosaccharides (Man8--9GlcNAc1) were more strongly bound to concanavalin A than were either the smaller neutral-type or the di-antennary acidic-type structures.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baenziger J. U., Fiete D. Structural determinants of concanavalin A specificity for oligosaccharides. J Biol Chem. 1979 Apr 10;254(7):2400–2407. [PubMed] [Google Scholar]
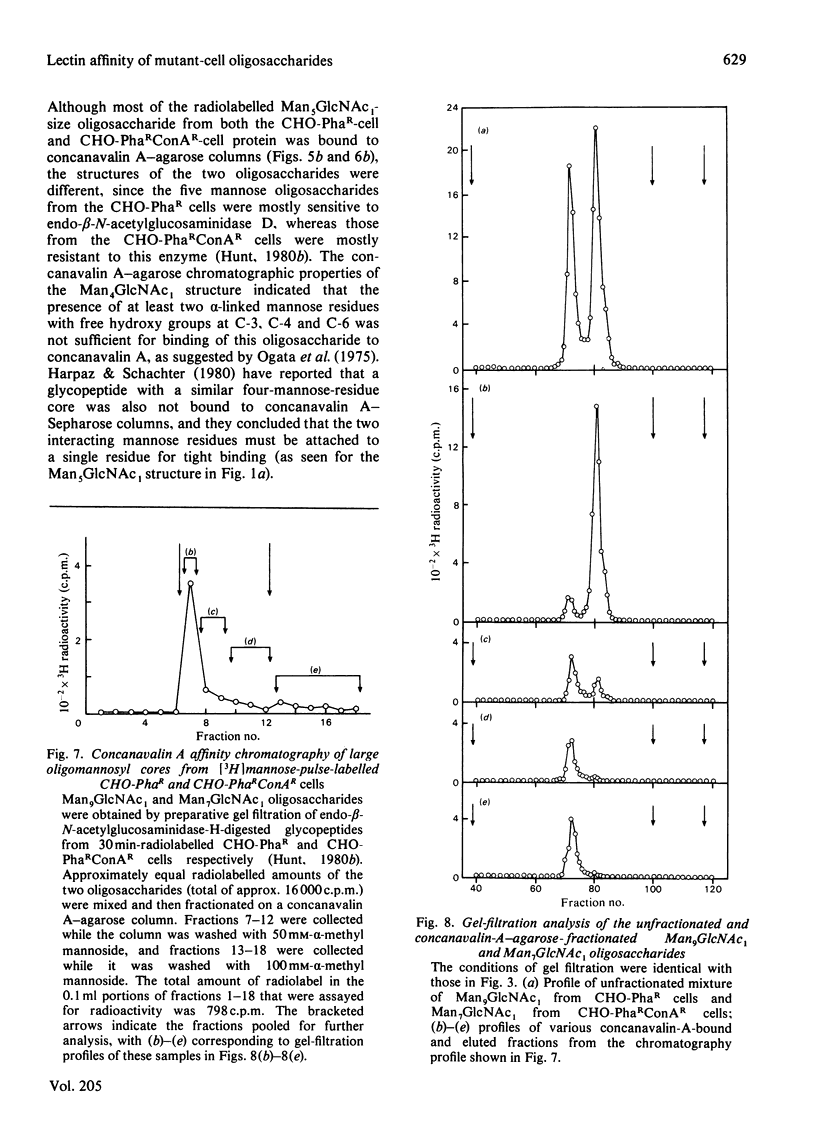
- Harpaz N., Schachter H. Control of glycoprotein synthesis. Processing of asparagine-linked oligosaccharides by one or more rat liver Golgi alpha-D-mannosidases dependent on the prior action of UDP-N-acetylglucosamine: alpha-D-mannoside beta 2-N-acetylglucosaminyltransferase I. J Biol Chem. 1980 May 25;255(10):4894–4902. [PubMed] [Google Scholar]
- Hunt L. A. Altered synthesis and processing of oligosaccharides of vesicular stomatitis virus glycoprotein in different lectin-resistant Chinese hamster ovary cell lines. J Virol. 1980 Aug;35(2):362–370. doi: 10.1128/jvi.35.2.362-370.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A. CHO cells selected for phytohemagglutinin and con A resistance are defective in both early and late stages of protein glycosylation. Cell. 1980 Sep;21(2):407–415. doi: 10.1016/0092-8674(80)90477-8. [DOI] [PubMed] [Google Scholar]
- Hunt L. A., Etchison J. R., Summers D. F. Oligosaccharide chains are trimmed during synthesis of the envelope glycoprotein of vesicular stomatitis virus. Proc Natl Acad Sci U S A. 1978 Feb;75(2):754–758. doi: 10.1073/pnas.75.2.754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A., Lamph W., Wright S. E. Transformation-dependent alterations in the oligosaccharides of Prague C Rous sarcoma virus glycoproteins. J Virol. 1981 Jan;37(1):207–215. doi: 10.1128/jvi.37.1.207-215.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt L. A. Lectin affinity chromatography of Sindbis and Rous sarcoma virus glycopeptides and oligosaccharides. J Virol Methods. 1982 May;4(4-5):283–295. doi: 10.1016/0166-0934(82)90075-1. [DOI] [PubMed] [Google Scholar]
- Hunt L. A. Sindbis virus glycoproteins acquire unusual neutral oligosaccharides in both normal and lectin-resistant Chinese Hamster ovary cell lines. Virology. 1981 Sep;113(2):534–543. doi: 10.1016/0042-6822(81)90181-1. [DOI] [PubMed] [Google Scholar]
- Hunt L. A., Summers D. F. Glycosylation of vesicular stomatitis virus glycoprotein in virus-infected HeLa cells. J Virol. 1976 Dec;20(3):646–657. doi: 10.1128/jvi.20.3.646-657.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld K., Reitman M. L., Kornfeld R. The carbohydrate-binding specificity of pea and lentil lectins. Fucose is an important determinant. J Biol Chem. 1981 Jul 10;256(13):6633–6640. [PubMed] [Google Scholar]
- Kornfeld S., Rogers J., Gregory W. The nature of the cell surface receptor site for Lens culinaris phytohemagglutinin. J Biol Chem. 1971 Nov;246(21):6581–6586. [PubMed] [Google Scholar]
- Li E., Tabas I., Kornfeld S. The synthesis of complex-type oligosaccharides. I. Structure of the lipid-linked oligosaccharide precursor of the complex-type oligosaccharides of the vesicular stomatitis virus G protein. J Biol Chem. 1978 Nov 10;253(21):7762–7770. [PubMed] [Google Scholar]
- Ogata S., Muramatsu T., Kobata A. Fractionation of glycopeptides by affinity column chromatography on concanavalin A-sepharose. J Biochem. 1975 Oct;78(4):687–696. doi: 10.1093/oxfordjournals.jbchem.a130956. [DOI] [PubMed] [Google Scholar]
- Robertson M. A., Etchison J. R., Robertson J. S., Summers D. F., Stanley P. Specific changes in the oligosaccharide moieties of VSV grown in different lectin-resistnat CHO cells. Cell. 1978 Mar;13(3):515–526. doi: 10.1016/0092-8674(78)90325-2. [DOI] [PubMed] [Google Scholar]
- Stanley P., Caillibot V., Siminovitch L. Selection and characterization of eight phenotypically distinct lines of lectin-resistant Chinese hamster ovary cell. Cell. 1975 Oct;6(2):121–128. doi: 10.1016/0092-8674(75)90002-1. [DOI] [PubMed] [Google Scholar]
- Stanley P., Carver J. P. Lectin receptors and lectin resistance in chinese hamster ovary cells. Adv Exp Med Biol. 1977;84:265–284. doi: 10.1007/978-1-4684-3279-4_13. [DOI] [PubMed] [Google Scholar]
- Stanley P., Narasimhan S., Siminovitch L., Schachter H. Chinese hamster ovary cells selected for resistance to the cytotoxicity of phytohemagglutinin are deficient in a UDP-N-acetylglucosamine--glycoprotein N-acetylglucosaminyltransferase activity. Proc Natl Acad Sci U S A. 1975 Sep;72(9):3323–3327. doi: 10.1073/pnas.72.9.3323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanley P., Siminovitch L. Complementation between mutants of CHO cells resistant to a variety of plant lectins. Somatic Cell Genet. 1977 Jul;3(4):391–405. doi: 10.1007/BF01542968. [DOI] [PubMed] [Google Scholar]


