Abstract
BACKGROUND
Bacillus cereus (B. cereus) is known to cause 2 types of foodborne diseases; the diarrheal and emetic syndromes. They are largely underreported due to their usually self-limiting course. Rare and sometimes fatal cases of liver failure, pulmonary hemorrhage and cerebral oedema have been reported mainly in children and young adults. We present here a case of liver failure associated with B. cereus food poisoning in a middle-aged patient.
CASE SUMMARY
A 48-year-old female patient presented to the emergency department for emesis, diarrhea, chills without fever, asthenia and diffuse abdominal cramps that started less than 30 minutes after eating a rice salad. Her past medical history was relevant for cholecystectomy and a cured Hashimoto’s disease. She did not take any medication, drugs and declared a consumption of one glass of wine per week. In the emergency department, she was treated with acetaminophen, metoclopramide, ondansetron, and an intravenous normal saline infusion. Blood gas analysis revealed a metabolic acidosis with hyperlactatemia, coagulation revealed a low prothrombin activity [32 %; normal values (N): 70-140] and a low Factor V activity (15%; N: > 70). Transaminases were elevated with hyperbilirubinemia, elevated lipase and rhabdomyolysis. N-acetylcysteine treatment was introduced. Abdominal echography revealed no signs of chronic hepatopathy or hepatomegaly. Day after the admission, psychomotor activity improved, transaminases and lipase started decreasing. Rhabdomyolysis gradually worsened to peak on day 3. Screening tests for liver disease were negative for viral and autoimmune cause of liver failure. Stools cultures were positive for colonies of the B. cereus group which were also identified in the rice salad samples processed whereas blood cultures were negative. The patient’s condition improved gradually including her liver function parameters and psychomotor activity which allowed her discharged home on day 9.
CONCLUSION
We describe a rare case of hepatocellular dysfunction due to a foodborne B. cereus intoxication in an adult patient. Even if it is uncommon, the severity of liver dysfunction reported and mechanism of the cereulide toxin toxicity on liver suggest that acetaminophen should be avoided in case of a foodborne intoxication and n-acetylcysteine could be a potential therapy helping to prevent hepatocytes necrosis due to the oxidative stress induced by mitochondrial dysfunction.
Keywords: Liver failure, Foodborne intoxication, Bacillus cereus, Toxin cereulide, Emetic syndrome, Acetaminophen, Case report
Core Tip: Bacillus cereus is known to cause two types of foodborne diseases: The diarrheal and emetic syndromes, which are usually self-limiting. Previous case studies have reported on children presenting with acute liver failure and rhabdomyolysis linked to mitochondrial dysfunction associated with the cereulide toxin. Herein, we report the first case of severe liver failure in an adult after a foodborne intoxication linked to a Bacillus cereus sequence type 26 strain harboring CES, SPH, and NHE genes. We review the literature and discuss the potential role of n-acetylcysteine therapy in these patients.
INTRODUCTION
Bacterial toxins [from Bacillus cereus (B. cereus), Clostridium botulinum, Clostridium perfringens, Staphylococcus aureus] and unspecified toxins (positive molecular assays without bacterial identification) are the third most common causative agents of foodborne illness after viruses and enteropathogenic bacteria, such as Campylobacter, Salmonella, Listeria monocytogenes. The European union food safety authority reported 10555 such cases in 2019, representing 19.3% of all registered foodborne outbreaks[1]. Among them, 1636 cases, 44 hospitalizations, and 7 deaths have been attributed to B. cereus although the incidence of the intoxication is likely underreported due to its usually self-limiting course. Because of its highly resistant endospore, this facultative anaerobic gram-positive bacterium is ubiquitously found in the soil and on vegetables, and has been linked to livestock feces contaminating the food chain[2]. Alongside extraintestinal infections, such as endophthalmitis, respiratory tract and gas-gangrene-like infections, B. cereus may cause two types of foodborne diseases: The diarrheal and emetic syndromes[3].
The diarrheal type is characterized by abdominal cramps and diarrhea occurring 8 to 16 hours after the ingestion of a high volume of vegetative cells (> 106 cfu/g). Symptoms are usually self-limiting and last 12 to 24 hours. They are likely caused by three pore-forming enterotoxins (hemolysin BL, non-hemolytic enterotoxin, cytotoxin K), which act during the exponential growth phase of vegetative cells, following the germination of spores in the intestines[4]. This syndrome is mostly associated with ingestion of vegetables, meat, and milk products[3].
Meanwhile, in the emetic type, a foodborne intoxication, nausea, and vomiting appear 30 min to 4 hours after the ingestion of the heat-stable and acidic-resistant cereulide toxin, which stimulates the vagus nerve through the serotonin 5-hydroxytryptamine type 3 receptors in the stomach and small intestine[5]. The depsipeptide toxin cereulide is encoded by the plasmid genes (CES locus encoding the genetic determinants essential for non-ribosomal assembly of the cereulide toxin), present in up to 10% of the B. cereus strain and is produced during its growth phase in foodstuffs (103-105 B. cereus cfu/g)[6]. High content of starch, carbohydrates, vitamins, trace elements, neutral potential of hydrogen (pH), and medium or high-water activity (aw) seem a favorable environment for cereulide production[6]. Emetic-type symptoms are usually mild and last less than 24 hours. Rare and sometimes fatal cases of liver failure, pulmonary hemorrhage and cerebral oedema have been reported mainly in children, with only two cases reported in young adults[7-17].
We present a case of liver failure associated with B. cereus food poisoning in a middle-aged patient, which has been rarely reported in an adult and we review the previous cases of this condition. The patient provided written consent and all data were anonymized.
CASE PRESENTATION
Chief complaints
A 48-year-old woman presented to the emergency department with emesis, diarrhea, chills without fever, asthenia, and diffuse abdominal cramps.
History of present illness
Symptoms started less than 30 min after eating a rice salad with ham and salmon. The meal had been cooked 3 days before and conserved in a malfunctioning fridge, which maintained its content at 8 °C. The patient’s husband and her 18-month-old child, who ate the same food, presented similar although less severe symptoms. At the emergency department, she was treated with acetaminophen (1000 mg), metoclopramide (10 mg), ondansetron (4 mg), and an intravenous normal saline infusion for food poisoning with mild dehydration. She rapidly became lethargic and had a persistent Glasgow coma score of 14/15 with confusion.
History of past illness
The patient’s medical history revealed cholecystectomy 16 years before and remitted Hashimoto’s disease.
Personal and family history
She had an intrauterine device implanted for contraception and did not take any medication or drugs and declared a consumption of one glass of wine per week. She reported no remarkable history nor liver or similar disease in her family.
Physical examination
Her vital signs showed tachycardia (105 pulses/min) with normal blood pressure (130/80 mmHg), tachypnea (28 breaths/min), and normal oxygen saturation (98%). Physical examination revealed skin mottling and an abdomen diffusely sensitive to palpation without guarding or flapping tremor.
Laboratory examinations
Blood gas analysis revealed a metabolic acidosis with hyperlactatemia [pH: 7.14, normal values (N): 7.35-7.45; lactate: 13.9 mmol/L, N: 0.5-1.6; carbonic acid hydrogen radical: 4.6 mmol/L, N: 22-28; partial pressure of carbon dioxide: 1.8 kPa, N: 4.27-6]. Blood analysis showed hemoconcentration (hemoglobin: 175 G/L, N: 120-160), leukocytosis (leucocytes: 30.9 G/L, N: 4-11) with neutrophilic predominance (segmented neutrophil: 25.3 G/L, N: 1.5-7.5; unsegmented neutrophil: 1.5 G/L, N: 0-0.05), and normal thrombocyte count (390 G/L, N: 150-350). Coagulation revealed a low prothrombin activity (32%, N: 70-140) with a spontaneous high international normalized ratio (2.0). Liver tests revealed elevated transaminases [aspartate transaminase (AST): 3536 U/L, N: 11-42; alanine transaminase (ALT): 3248 U/L, N: 9-42), slight elevation of gamma-glutamyl transferase: 79 U/L, N: 9-35], normal phosphatase alkaline (125 U/L, N: 25-102) and hyperbilirubinemia (total: 55.2 μmol/L, N: 7-25; conjugate: 46.1 μmol/L, N: 0.5-9.5). Acute renal failure kidney disease improving global outcome stage 2 (creatinine: 124 μmol/L, N: 44-80; uremia: 10.0 mmol/L, N: 3.2-7.5) and elevated lipase levels at 272 U/L (N: 13-60 U/L) were concomitantly present. A complement of blood analysis revealed low activity of factor V (15%, N: > 70) with rhabdomyolysis [creatinine kinase (CK): 1525 U/L, N: 33-187), high lactate dehydrogenase (2224 U/L, N: 87-210), and nontoxic acetaminophen blood level (0.066 mmol/L, nontoxic: < 0.2).
Screening tests for liver disease failed to identify a cause of liver failure: The patient was immune against hepatitis virus A, parvovirus B19 and Epstein–Barr virus with negative viremia. Other serologies and viremia were negative for hepatitis B, C, and E viruses, and human immunodeficiency virus. Total immunoglobulin G, anti-smooth muscle antibody, anti-actin antibody, anti-liver-kidney-microsomal antibody, anti-nuclear antibody, antimitochondrial antibody, and anticentromere antibody levels were negative.
Stool cultures were positive for colonies of the B. cereus group which were also identified in the rice salad samples processed, whereas blood cultures were negative. Polymerase chain reaction analyses of stool samples for the detection of Clostridium difficile toxin, Campylobacter jejuni/coli, Salmonella, Shigella, Yersinia enterocolitica, and diarrhoeagenic Escherichia coli (Enteropathogenic, enteroaggregative, enterotoxigenic, enteroinvasive) were all negative.
Whole-genome sequencing of the clinical and food derived isolates: The B. cereus group isolates from the patient’s stool cultures and rice salad samples were characterized by whole genome sequencing. Genomic DNA of the isolates was extracted from subcultures obtained from single colonies grown for 24 hours at 37 °C on sheep blood agar using the DNeasy Blood and Tissue Kit (Qiagen, Hilden, Germany). Libraries were prepared using the Nextera DNA Flex Library Preparation Kit (Illumina, CA, United States) and sequenced on the Illumina MiniSeq platform (2 × 150 bp). Illumina read adapters and low-quality bases were trimmed with fastp (https://github.com/OpenGene/fastp) and draft genomes were assembled using SPAdes 3.14.1 implemented in shovill 1.1.0 (https://github.com/tseemann/shovill). Assembly quality was assessed using QUAST 5.0.2 (https://github.com/ablab/quast/releases) and CheckM v1.1.3 (https://ecogenomics.github.io/CheckM/). Multi-locus sequence types and virulence genes were identified using BTyper3 v3.2.0 (https://pypi.org/project/btyper3/). Pairwise cg single nucleotide polymorphism (SNP) distances were determined using the Center for Food Safety and Applied Nutrition SNP pipeline 2.2.1 (https://snp-pipeline.readthedocs.io/en/Latest/readme.html) with the chromosome of strain AH187 (B. cereus ST26; GCF_000021225.1) as the reference. Sequencing data were deposited in GenBank and the Sequence Read Archive under BioProject accession number PRJNA992990. The accession numbers of the genomes are N21-2405 (clinical strain) JAUKFW000000000 and N21-2406 (food isolate) JAUKFX000000000.
Imaging examinations
Abdominal echography revealed air in the portal vessels with no sign of chronic hepatopathy or hepatomegaly. An abdominal computed tomography (CT) showed diffuse paralytic ileus with mild peritoneal effusion, congestive oedema and pneumatosis of a short portion of the jejunum without signs of ischemia or pancreatic lesions.
FINAL DIAGNOSIS
After exclusion of all other causes, the diagnosis was a B. cereus food poisoning with severe hepatic dysfunction.
TREATMENT
Because of the severe hepatocellular dysfunction, n-acetylcysteine treatment was administered (150 mg/kg IV over 1 hour, followed by 50 mg/kg over 4 hours, then 100 mg/kg over 16 hours). Ceftriaxone with metronidazole was initiated regarding the jejunal pneumatosis on CT scan despite absence of overt sepsis sign.
OUTCOME AND FOLLOW-UP
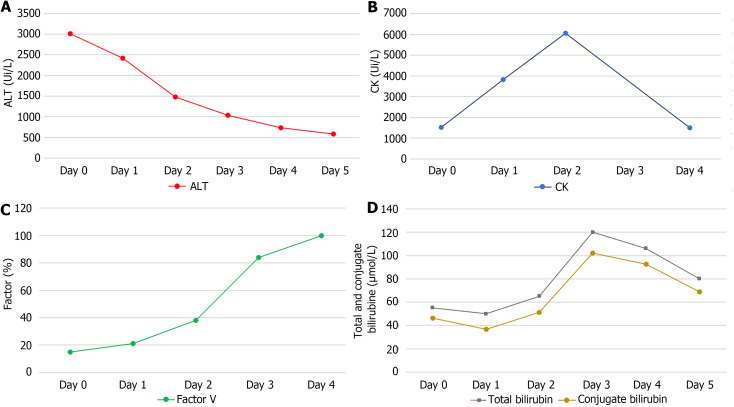
The day after the admission, psychomotor activity improved, transaminase levels started decreasing (AST: 2228 U/L, ALT: 2423 U/L), and hepatic and kidney function improved (factor V: 21%, total bilirubin: 50 μmol/L, conjugate bilirubin: 40 μmol/L, creatinine: 59 μmol/L) (Figure 1). Rhabdomyolysis gradually worsened to peak on day 3 (CK total: 6047 U/L) and lipase blood levels decreased the next day, only to rise again to peak at 496 U/L on day 5 without pancreatic symptomatology. The patient’s condition improved gradually, including her liver function parameters and psychomotor activity, which allowed her to be discharged from the intensive care unit and transferred to our hepatology unit. Given this improvement, liver tissue biopsy was not performed. Considering the disease severity and persistence of diarrhea, we administered ciprofloxacin for 5 days. A follow-up abdominal CT scan at day 8 after hospitalization showed a colonic and intestinal hypervascularization with a persistence of a mild thickening wall without either pancreatic or hepatic abnormalities. Following improved clinical presentation and psychomotor recovery, the patient was discharged home on day 9.
Figure 1.
blood levels over the duration of hospitalization in days. A: Alanine transaminase; B: Creatinine kinase; C: Factor V; D: Bilirubin. Day 0 is the first day of hospitalization in our hospital; Alanine transaminase and creatinine kinase are expressed in unit per L, factor V in percent and bilirubin in micromole per L. ALT: Alanine transaminase; CK: Creatine kinase.
DISCUSSION
This otherwise healthy patient presented with liver failure due to a foodborne intoxication linked to the B. cereus ST26 strain, harboring CES, SPH, NHE genes. This entity has, to our knowledge, only been described previously in the pediatric population but not in adults. Herein, the liver screening tests and investigations for hepatocellular dysfunction were negative and we initially suspected ischemic hepatitis secondary to circulatory failure due to hypovolemia. However, the severe metabolic acidosis, associated with hyperlactatemia suggested a significant dysfunction of the relative hemodynamic stability and absence of other severe organ failure. Thus, hypovolemia could have explained some but not all liver test results. In addition, the elevated CK blood levels are not usually a feature of ischemic hepatitis in hepatocellular dysfunction. Consequently, we suspected B. cereus food poisoning as the alternative diagnosis. Given the extremely short incubation time and clinical presentation without evidence of another pathogen, emetic type of B. cereus food poisoning was the most likely diagnosis. This was supported by identification of B. cereus group strains both in the stool and rice salad sample cultures. Whole-genome sequencing was conducted on both isolates showing that they harbored SPH, CESAB, AND NHEABC genes, and both belonged to ST26. Comparison of their genome sequences (0 single nucleotide polymorphisms) conclusively showed that both isolates were from the same strain.
The main limitation of our case report is the lack of liver tissue biopsy results. As the patient improved, a biopsy was not considered clinically essential. However, as a result, we cannot conclusively state that no other disease entity was involved in patient presentation.
Thirteen pediatric cases of liver failure due to B. cereus foodborne intoxications have been published[8-16]. Three (23.1%) patients died, one (7.7%) had an uncomplicated liver transplantation, and nine (69.2%) had an excellent outcome (Table 1). All cases occurred due to improperly stored pasta or rice. Out of these 13 patients, 8 patients had rhabdomyolysis, whereas the relevant data were not reported for 5 patients[8,10-16]. The median age of these patients was 9 (interquartile range: 7-11) years. Adults who ate the same food in three studies had less severe symptoms than the pediatric patients did, and hepatic dysfunction was not reported[8,10,11]. To date, only two cases of adult patients aged 20 and 25 years, respectively, were reported[7,17]. One of this two young adults recovered well, and liver tissue biopsy was not undertaken, whereas the other patient died with the reported leading cause of death being liver necrosis on histology, combined with pulmonary oedema and necrotic enteritis.
Table 1.
Previous case reports of hepatocellular dysfunctions and liver failures induced by Bacillus cereus food poisoning
|
Sex and age
|
1Year
|
Country
|
Ref.
|
Food
|
n
|
Symptoms
|
pH at admission
|
Transaminase levels (peak value) (U/L)
|
CK at admission or peak (U/L)
|
Lactate (mmol/L)
|
Factor V or PT (%)
|
Imaging
|
Prehospital treatment
|
In-hospital treatment
|
| M, 17 years old | 1997 | Switzerland | Mahler et al[8] | Spaghetti with pesto | 4 | Emesis, lethargy, jaundice | 7.27 | AST: 2.140; ALT: 5.270 | 2.56 | 14.4 | PT: 12 | NA | Aspirin (1 g), acetaminophen (1 g), thiethylperazine, meclizine, domperidone | Unspecified antibiotic |
| F, 7 years old | 2005 | Belgium | Dierick et al[9] | Pasta | 3 | Vomiting, respiratory distress | NA | AST: 3.123; ALT: 2.636 | NA | NA | PT: < 10 | NA | NA | Cardiopulmonary resuscitation |
| M, 9 years old | 2005 | Belgium | Dierick et al[9] | Pasta | 3 | Nausea, vomiting | 7.16 | AST: 1.858; ALT: 5.181 | NA | 9.5 | PT: 23.1 | NA | NA | Endotracheal intubation with mechanical ventilation. vitamin K, lactuloses (oral and rectal), n-acetylcysteine, neomycin |
| F, 9 years old | 2005 | Belgium | Dierick et al[9] | Pasta | 3 | Nausea, emesis | 7.37 | AST: 239; ALT: 447 | NA | 13.8 | PT: 83.7 | NA | NA | Bicarbonate |
| F, 10 years old | 2005 | Belgium | Dierick et al[9] | Pasta | 3 | Nausea, emesis | 7.37 | AST: 5.327; ALT: 4.101 | NA | 8.8 | PT: 30.6 | NA | NA | Bicarbonate |
| F, 10 years old | 2008 | Switzerland | Pósfay-Barbe et al[10] | Pasta | 2 | Abdominal pain, emesis, diarrhea, convulsion | 6.74 | AST: 14.622; ALT: 9.018 | 22 | 17 | FV: 25 | CT scan: Intestinal wall thickening | NA | OTI, meropenem, clindamycin, vasopressors |
| M, 1 years old | 2008 | Japan | Shiota et al[11] | Fried rice | 1 | Emesis lethargy, unconsciousness, cardiac arrest | 6.25 | AST: 164; ALT: 131 | 175 | 23 | NA | NA | NA | Cardiopulmonary resuscitation |
| F, 2 years old | 2008 | Japan | Shiota et al[11] | Fried rice | 1 | Emesis, lethargy, unconsciousness | 7.18 | AST: 104; ALT: 34 | 114 | 11.6 | NA | NA | NA | Plasma exchange followed by hemodialysis |
| M, 11 years old | 2010 | Japan | Ichikawa et al[12] | Fried rice | 1 | Vomiting, convulsions | 7.29 | AST: 10.399; ALT: 6.118 | 458 | 11.8 | PT: 38 | NA | NA | Methylprednisolone, cyclosporin A, therapeutic hypothermia, mannitol, edaravone |
| M, 20 years old | 2011 | Belgium | Naranjo et al[7] | Spaghetti with tomato sauce | 5 | Emesis, diarrhea | NA | NA | NA | NA | NA | NA | NA | NA |
| M, 15 years old | 2012 | France | Saleh et al[13] | Pasta | NA | Abdominal pain, vomiting, diarrhea, fever | 7.41 | AST: 3.829; ALT: 4.823 | 12.592 | 5.4 | FV: 35 | Liver ultrasound: Normal; CT scan: Hepatomegaly | NA | Piperacillin-tazobactam |
| M, 13 years old | 2015 | Germany | Tschied et al[14] | Rice | 2 | Abdominal pain, nausea, vomiting, lethargy | NA | AST: 38.811; ALT: 19.976 | 141 (Peak: 389.355) | 12 | FV: < 5 | Liver ultrasound: Hepatomegaly | NA | Vasopressors, hemodialysis, transplantation (living donor) |
| M, 25 years old | 2021 | Austria | Schreiber et al[17] | Rice | 3 | Nausea, vomiting, diarrhea | NA | AST: 11.578; ALT: 8.959 | 161 (Peak: 2186) | 9.9 | FV: 8; PT: 4.3 | NA | NA | Lactulose, piperacillin/tazobactam, rifaximin, n-acetylcysteine |
| F, 11 years old | 2022 | France | Thery et al[15] | Lasagna | 3 | Abdominal pain, nausea. vomiting | 7.26 | AST: 5.912; ALT: 8.571 | 2.134 (Peak: 36.666) | 12.1 | FV: 26; PT: 19 | NA | NA | OTI, cefotaxime, micafungin, benzylpenicillin, vancomycin, n-acetylcysteine, hemodialysis, norepinephrine |
| M, 4 years old | 2022 | Japan | Kubota et al[16] | Rice | NA | Vomiting, diarrhea, convulsion, loss of consciousness | NA | NA | NA | NA | NA | NA | NA | NA |
| F, 48 years old (This case) | 2023 | Switzerland | Chatelanat et al | Rice | 2 | Abdominal pain, nausea, vomiting, diarrhea, asthenia | 7.14 | AST: 2.228; ALT: 2.423 | 1.525 (Peak: 6.047) | 13.9 | FV: 15 | Liver ultrasound: Air in portal vessels | Acetaminophen (1 g) | N-acetylcysteine |
Year: Year of publication.
F: Female; M: Male; n: Number of day(s) elapsed between cooking and consumption; pH: Potential of hydrogen; AST: Aspartate transaminase; ALT: Alanine transaminase; FV: Factor V; OTI: Orotracheal intubation; CK: Creatin kinase; PT: Prothrombin time; NA: Not reported; CT: Computed tomography.
Recent in vitro studies have improved our understanding of the pathophysiology associated with liver dysfunction. The molecular structure of the cereulide toxin is similar to that of valinomycin, allowing it to act as a potent potassium-selective ionophore[18]. By affecting the electrochemical gradient, this property causes mitochondrial depolarization, inducing respiratory chain dysfunction with hyperlactatemia, steatosis, accumulation of reactive oxygen species, swelling of mitochondria and cell apoptosis[8,19,20]. This molecular picture is supported by liver tissue biopsy findings from previous case reports showing microvesicular steatosis, hepatocyte necrosis, and cell vacuolization, corresponding to swollen mitochondria (Table 1)[6,10-14]. The cause of rhabdomyolysis is unclear but could be the result of the same mechanism. In addition to the effects of the cereulide toxin, rhabdomyolysis could be exacerbated by acidosis[21]. A study using a porcine model reported that traces of ingested cereulide toxin can be found in many organs, such as kidneys, liver, muscles, and fat tissues[22] and that it could cross the blood-brain-barrier, suggesting its effects on potassium concentration in the cerebrospinal fluid through its ionophore characteristics. This mechanism could explain why patients develop lethargy, convulsion, or coma, without the typical signs of hepatic encephalopathy despite the severe hepatocellular dysfunction, as shown in our case. Our patient had a slight elevation of lipase blood levels without pancreatitis which was not reported previously. A previous study described cereulide toxin effects on the pancreas as affecting its beta cells due to a mitochondrial defect, resulting in apoptosis, and thus preventing insulin secretion[23].
It remains unclear why the toxin mostly affects the pediatric population; the disproportionate case reporting could be due to publication bias or poorly understood increased toxicity of the cereulide toxin in children. If cases in adults are indeed uncommon rather than underreported, it is possible that congenital subclinical mitochondrial dysfunction could have predisposed our patient to cereulide toxin. Nevertheless, no event in her past medical or family history suggested such an illness. Given these considerations, n-acetylcysteine could be a potential treatment, as it has antioxidant effects and may provide cysteine for glutathione synthesis, which acts as a potent antioxidant[24]. In contrast, even low-dose acetaminophen, as in our case, could exacerbate this dysfunction and precipitate necrosis by triggering glutathione depletion and oxidative stress.
CONCLUSION
We describe a rare case of hepatocellular dysfunction due to foodborne B. cereus intoxication in an adult patient. The observed severity of liver dysfunction and suspected mechanism of the cereulide toxin toxicity on the liver suggest that acetaminophen should be avoided in patients with this presentation. Instead, n-acetylcysteine may help prevent hepatocyte necrosis due to oxidative stress induced by mitochondrial dysfunction.
ACKNOWLEDGEMENTS
The authors thank Cernela N, Stevens M and Biggel M for their help with sequencing and bioinformatic analysis.
Footnotes
Informed consent statement: The patient provided written consent and all data were anonymized.
Conflict-of-interest statement: The authors declare that they have no conflict of interest.
CARE Checklist (2016) statement: The authors have read the CARE Checklist (2016), and the manuscript was prepared and revised according to the CARE Checklist (2016).
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country of origin: Switzerland
Peer-review report’s classification
Scientific Quality: Grade B, Grade C
Novelty: Grade B, Grade B
Creativity or Innovation: Grade B, Grade B
Scientific Significance: Grade B, Grade B
P-Reviewer: Talmon GA; Zhao YC S-Editor: Fan M L-Editor: A P-Editor: Cai YX
Contributor Information
Olivier Chatelanat, Department of Gastroenterology and Hepatology, Geneva University Hospitals, Geneva 1205, Switzerland. olivier.chatelanat@gmail.com.
Mikaël de Lorenzi-Tognon, Department of Infectious Diseases, Geneva University Hospitals, Geneva 1205, Switzerland.
Laurent Spahr, Department of Gastroenterology and Hepatology, Geneva University Hospitals, Geneva 1205, Switzerland.
Abdessalam Cherkaoui, Department of Genetics and Laboratory Medicine, Geneva University Hospitals, Geneva 1205, Switzerland.
Roger Stephan, Institute for Food Safety and Hygiene, University of Zürich, Zurich 8057, Switzerland.
Marie Ongaro, Department of Gastroenterology and Hepatology, Geneva University Hospitals, Geneva 1205, Switzerland.
Laurent Kaiser, Department of Infectious Diseases, Geneva University Hospitals, Geneva 1205, Switzerland.
Nicolas Goossens, Department of Gastroenterology and Hepatology, Geneva University Hospitals, Geneva 1205, Switzerland.
References
- 1.European Food Safety Authority European Centre for Disease Prevention and Control. The European Union One Health 2019 Zoonoses Report. EFSA J. 2021;19:e06406. doi: 10.2903/j.efsa.2021.6406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Choi W, Kim SS. Outbreaks, Germination, and Inactivation of Bacillus cereus in Food Products: A Review. J Food Prot. 2020;83:1480–1487. doi: 10.4315/0362-028X.JFP-19-429. [DOI] [PubMed] [Google Scholar]
- 3.Enosi Tuipulotu D, Mathur A, Ngo C, Man SM. Bacillus cereus: Epidemiology, Virulence Factors, and Host-Pathogen Interactions. Trends Microbiol. 2021;29:458–471. doi: 10.1016/j.tim.2020.09.003. [DOI] [PubMed] [Google Scholar]
- 4.Dietrich R, Jessberger N, Ehling-Schulz M, Märtlbauer E, Granum PE. The Food Poisoning Toxins of Bacillus cereus. Toxins (Basel) 2021;13 doi: 10.3390/toxins13020098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Agata N, Ohta M, Mori M, Isobe M. A novel dodecadepsipeptide, cereulide, is an emetic toxin of Bacillus cereus. FEMS Microbiol Lett. 1995;129:17–20. doi: 10.1016/0378-1097(95)00119-P. [DOI] [PubMed] [Google Scholar]
- 6.Messelhäusser U, Frenzel E, Blöchinger C, Zucker R, Kämpf P, Ehling-Schulz M. Emetic Bacillus cereus are more volatile than thought: recent foodborne outbreaks and prevalence studies in Bavaria (2007-2013) Biomed Res Int. 2014;2014:465603. doi: 10.1155/2014/465603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Naranjo M, Denayer S, Botteldoorn N, Delbrassinne L, Veys J, Waegenaere J, Sirtaine N, Driesen RB, Sipido KR, Mahillon J, Dierick K. Sudden death of a young adult associated with Bacillus cereus food poisoning. J Clin Microbiol. 2011;49:4379–4381. doi: 10.1128/JCM.05129-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mahler H, Pasi A, Kramer JM, Schulte P, Scoging AC, Bär W, Krähenbühl S. Fulminant liver failure in association with the emetic toxin of Bacillus cereus. N Engl J Med. 1997;336:1142–1148. doi: 10.1056/NEJM199704173361604. [DOI] [PubMed] [Google Scholar]
- 9.Dierick K, Van Coillie E, Swiecicka I, Meyfroidt G, Devlieger H, Meulemans A, Hoedemaekers G, Fourie L, Heyndrickx M, Mahillon J. Fatal family outbreak of Bacillus cereus-associated food poisoning. J Clin Microbiol. 2005;43:4277–4279. doi: 10.1128/JCM.43.8.4277-4279.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pósfay-Barbe KM, Schrenzel J, Frey J, Studer R, Korff C, Belli DC, Parvex P, Rimensberger PC, Schäppi MG. Food poisoning as a cause of acute liver failure. Pediatr Infect Dis J. 2008;27:846–847. doi: 10.1097/INF.0b013e318170f2ae. [DOI] [PubMed] [Google Scholar]
- 11.Shiota M, Saitou K, Mizumoto H, Matsusaka M, Agata N, Nakayama M, Kage M, Tatsumi S, Okamoto A, Yamaguchi S, Ohta M, Hata D. Rapid detoxification of cereulide in Bacillus cereus food poisoning. Pediatrics. 2010;125:e951–e955. doi: 10.1542/peds.2009-2319. [DOI] [PubMed] [Google Scholar]
- 12.Ichikawa K, Gakumazawa M, Inaba A, Shiga K, Takeshita S, Mori M, Kikuchi N. Acute encephalopathy of Bacillus cereus mimicking Reye syndrome. Brain Dev. 2010;32:688–690. doi: 10.1016/j.braindev.2009.09.004. [DOI] [PubMed] [Google Scholar]
- 13.Saleh M, Al Nakib M, Doloy A, Jacqmin S, Ghiglione S, Verroust N, Poyart C, Ozier Y. Bacillus cereus, an unusual cause of fulminant liver failure: diagnosis may prevent liver transplantation. J Med Microbiol. 2012;61:743–745. doi: 10.1099/jmm.0.038547-0. [DOI] [PubMed] [Google Scholar]
- 14.Tschiedel E, Rath PM, Steinmann J, Becker H, Dietrich R, Paul A, Felderhoff-Müser U, Dohna-Schwake C. Lifesaving liver transplantation for multi-organ failure caused by Bacillus cereus food poisoning. Pediatr Transplant. 2015;19:E11–E14. doi: 10.1111/petr.12378. [DOI] [PubMed] [Google Scholar]
- 15.Thery M, Cousin VL, Tissieres P, Enault M, Morin L. Multi-organ failure caused by lasagnas: A case report of Bacillus cereus food poisoning. Front Pediatr. 2022;10:978250. doi: 10.3389/fped.2022.978250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kubota N, Kobayashi J, Kasai A, Nasuno M, Murai T, Minami K, Ohta M. Detection of Bacillus cereus as a causative agent of emetic food poisoning by an unconventional culture procedure. J Infect Chemother. 2022;28:1575–1577. doi: 10.1016/j.jiac.2022.07.011. [DOI] [PubMed] [Google Scholar]
- 17.Schreiber N, Hackl G, Reisinger AC, Zollner-Schwetz I, Eller K, Schlagenhaufen C, Pietzka A, Czerwenka C, Stark TD, Kranzler M, Fickert P, Eller P, Ehling-Schulz M. Acute Liver Failure after Ingestion of Fried Rice Balls: A Case Series of Bacillus cereus Food Poisonings. Toxins (Basel) 2021;14 doi: 10.3390/toxins14010012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mikkola R, Saris NE, Grigoriev PA, Andersson MA, Salkinoja-Salonen MS. Ionophoretic properties and mitochondrial effects of cereulide: the emetic toxin of B. cereus. Eur J Biochem. 1999;263:112–117. doi: 10.1046/j.1432-1327.1999.00476.x. [DOI] [PubMed] [Google Scholar]
- 19.Decleer M, Jovanovic J, Vakula A, Udovicki B, Agoua REK, Madder A, De Saeger S, Rajkovic A. Oxygen Consumption Rate Analysis of Mitochondrial Dysfunction Caused by Bacillus cereus Cereulide in Caco-2 and HepG2 Cells. Toxins (Basel) 2018;10 doi: 10.3390/toxins10070266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li D, Lin R, Xu Y, Chen Q, Deng F, Deng Y, Wen J. Cereulide Exposure Caused Cytopathogenic Damages of Liver and Kidney in Mice. Int J Mol Sci. 2021;22 doi: 10.3390/ijms22179148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lim AK. Abnormal liver function tests associated with severe rhabdomyolysis. World J Gastroenterol. 2020;26:1020–1028. doi: 10.3748/wjg.v26.i10.1020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bauer T, Sipos W, Stark TD, Käser T, Knecht C, Brunthaler R, Saalmüller A, Hofmann T, Ehling-Schulz M. First Insights Into Within Host Translocation of the Bacillus cereus Toxin Cereulide Using a Porcine Model. Front Microbiol. 2018;9:2652. doi: 10.3389/fmicb.2018.02652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vangoitsenhoven R, Maris M, Overbergh L, Van Loco J, Mathieu C, Van der Schueren B. Cereulide food toxin, beta cell function and diabetes: Facts and hypotheses. Diabetes Res Clin Pract. 2015;109:1–5. doi: 10.1016/j.diabres.2015.04.029. [DOI] [PubMed] [Google Scholar]
- 24.de Andrade KQ, Moura FA, dos Santos JM, de Araújo OR, de Farias Santos JC, Goulart MO. Oxidative Stress and Inflammation in Hepatic Diseases: Therapeutic Possibilities of N-Acetylcysteine. Int J Mol Sci. 2015;16:30269–30308. doi: 10.3390/ijms161226225. [DOI] [PMC free article] [PubMed] [Google Scholar]