Summary
Background:
In April 2022, French Lentil and Leek Crumble (FLLC), a new frozen food preparation manufactured by Daily Harvest™ (containing Tara flour) was offered as a natural high protein meal product. Soon thereafter, widespread anecdotal reports of acute gastrointestinal symptoms with liver injury were reported, leading to its voluntary withdrawal in June 2022, after shipment of 28,000 preparations.
Aims:
To summarize the clinical and laboratory features of 17 patients with FLLC associated liver injury from the Drug Induced Liver Injury Network (DILIN).
Methods:
Patients with FLLC associated liver injury were enrolled into a prospective protocol and followed for six months. Cases were adjudicated by expert opinion causality assessment with summary statistics for data analysis.
Results:
Enrolled subjects had a mean age of 41 years, 82% were female with mean BMI of 24kg/m2. All were Caucasian without underlying liver disease. In most cases, abdominal pain and nausea arose within hours of FLLC ingestion. Mean days from ingestion to identification of liver injury was 3.1 days (±2.8). On enrollment, 53% had jaundice, 47% nausea, 24% fever, 59% abdominal pain, 41% itching, and 12% rash. The mean initial serum ALT was 475 U/L (± 302), AST 315 U/L (± 315), alkaline phosphatase 190 U/L (± 76), with a total bilirubin of 2.6 mg/dL (± 2). 63% presented with a hepatocellular pattern of liver injury, 6% cholestatic, and 31% mixed as determined by the R value. 24% of patients were hospitalized, and there were no fatalities or liver transplants. Liver biopsy in one subject revealed acute hepatitis with mild ductular reaction, mild lymphocytic and eosinophilic portal inflammation, mild lobular inflammation, preserved bile ducts and absence of interface hepatitis, steatosis, granulomatous reaction, or cholestasis. Phylogenetic analysis confirmed the presence of Tara spinosa, the source of Tara flour.
Conclusions:
Natural food products are increasingly ubiquitous and may unexpectedly cause significant illness. All clinicians should inquire whether patients are consuming natural food products or herbal supplements and consider them as a potential cause of liver injury.
Introduction
In the United States, there has been increased public interest in natural products for improved health with significant increase in the use of dietary supplements and herbal products1,2. As part of this movement, plant based diets have also increased in popularity3. Daily Harvest™ in April 2022 introduced a new 12-ounce high protein frozen food product named French Lentil and Leek Crumble (FLLC), which included Tara flour, a new source of plant-based protein. FLCC was sold online and available nationwide4. Soon thereafter, numerous complaints of gastrointestinal, liver, bile duct, and/or gallbladder illnesses after ingestion of FLLC were reported to the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention. In response to these complaints, the company voluntarily recalled the product on June 17, 2022 after shipment of 28,000 preparations4. As of October 21, 2022, the FDA reported 393 adverse events, 133 hospitalizations, but no deaths related to this product in reports from 39 states4 with the last injury reported on September 4, 2022.
Methods
The National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) established the Drug-Induced Liver Injury Network (DILIN) in 2003 to collect and characterize liver injury due to prescription and over-the-counter drugs, herbal products and supplements using a standardized protocol. The DILIN’s Prospective Study enrolls patients with suspected drug induced liver injury within six months of onset. Patients eligible for enrollment meet pre-defined laboratory criteria of serum aspartate aminotransferase (AST) or alanine aminotransferase (ALT) >5 times or serum alkaline phosphatase (ALP) levels >2 times the upper limit of normal (or baseline before exposure) on two consecutive occasions. Those with a total serum bilirubin > 2.5 mg/dL or an INR >1.5 after an exposure are also eligible. A more in-depth review of eligibility, evaluation, and enrollment in the DILIN Prospective Study has been described in prior publications.5 The diagnosis of drug-induced liver injury relies on the exclusion of other causes of liver injury and temporal evidence linking the injury to a specific drug or agent. DILIN uses expert opinion to determine the degree of association between the implicated agent(s) and liver injury6. A panel of DILIN investigators score the case and implicated agents from one (definite) to five (unlikely), reflecting the likelihood of attribution for liver injury. Cases are graded as definite (>95% likelihood), highly likely (75-95%), probable (50-74%), possible (25-49%), or unlikely (<25%) DILI. Expert adjudication occurs six months after the injury and data have been collected. Chemical analysis for hepatoxins and heavy metals of six FLLC preparations collected from the subjects were performed and the presence of baikiain was confirmed as described7.
Results
A total of 18 cases of liver injury from FLLC were identified and adjudicated as definite (n=8), highly likely (n=7), and probable (n=2). One case was adjudicated as possible, leaving 17 cases for the final analysis. The mean age was 41 years, and 82% were women. The mean BMI was 24 kg/m2, indicating a non-obese group of individuals, and all self-reported as being white or Caucasian without history of underlying liver disease. All patients presented in May or June of 2022, with none either before or after.
Clinical presentation and laboratory findings
As illustrated in Table 1, the mean days from ingestion to finding of abnormal liver tests qualifying for enrollment was 3.1 days (± 2.8). On enrollment, 53% had jaundice, 47% nausea, 24% fever, 59% abdominal pain, 41% itching, and 12% rash. The mean initial serum ALT was 475 U/L (± 302), AST 315 U/L (± 315), alkaline phosphatase 190 U/L (± 76), with a total bilirubin of 2.6 mg/dL (± 2). 63% presented with a hepatocellular pattern of liver injury, 6% cholestatic and 31% mixed as determined by the R value [R = (ALT/ULN) ÷ (Alkaline phosphatase/ULN), a formula used to clinically characterize drug induced liver injury (https://www.ncbi.nlm.nih.gov/books/NBK548733/). The serum ALT level at onset was the peak value in all except one patient, indicating the rapid resolution of the injury after cessation. 24% of patients were hospitalized, but there were no fatalities or liver transplants. No patients were treated with corticosteroids, and liver enzymes normalized in all patients.
Table 1:
Clinical Features and Labs on Presentation
| N=17* | |
|---|---|
|
| |
| Age, mean (±SD) | 41 (8.77) |
|
| |
| Gender (Female) | 82% |
|
| |
| BMI (kg/m2) (n=16) | |
| Mean (±SD) | 24 (3.99) |
| Median (Q1, Q3) | 22.4 (20.4,26.8) |
|
| |
| Race/Ethnicity | |
| White | 100% |
|
| |
| Days from DILI onset till qualifying for enrollment, (n=14) | |
| Mean (±SD) | 3.1(2.76) |
| Median (Q1,Q3) | 2.5 (1.0,5.0) |
| ALT at DILI onset (U/L), (n=16) | |
| Mean (±SD) | 475(302) |
| Median (Q1,Q3) | 379 (314,597) |
| AST at DILI onset (U/L), (n=16) | |
| Mean (±SD) | 315(315) |
| Median (Q1,Q3) | 184 (81,524) |
| ALK at DILI onset (U/L), (n=16) | |
| Mean (±SD) | 189(76) |
| Median (Q1,Q3) | 177 (131,227) |
| Total bilirubin (mg/dL), (n=16) | |
| Mean (±SD) | 2.6(2) |
| Median (Q1,Q3) | 2 (1,3.6) |
|
| |
| Symptoms on presentation | |
| Jaundice | 53% |
| Nausea | 47% |
| Fever | 24% |
| Abdominal Pain | 59% |
| Itching | 41% |
| Rash | 12% |
|
| |
| Outcomes | |
| Hospitalized | 24% |
| Liver death | 0 |
| Transplantation | 0 |
| Chronic injury | 0 |
N = 17 unless noted
One patient underwent liver biopsy which revealed an acute hepatitis with mild ductular reaction, mild lymphocytic and eosinophilic portal inflammation with preserved bile ducts, without interface hepatitis with mild lobular necroinflammation without steatosis, granulomatous reaction, or cholestasis (Figure 1a,b,c). Chemical analysis in six FLLC samples provided by affected patients had no detectable amatoxins, phallotoxins, aflatoxins, microcystins, or pyrrolizidine alkaloids within limits of detection (10-25 ppb) and concentrations of heavy metals (As, Pb, Hg, Cr, Cd) were all within acceptable limits7. All six did contain multiple botanical components including those of Tara flour.
Figure 1:
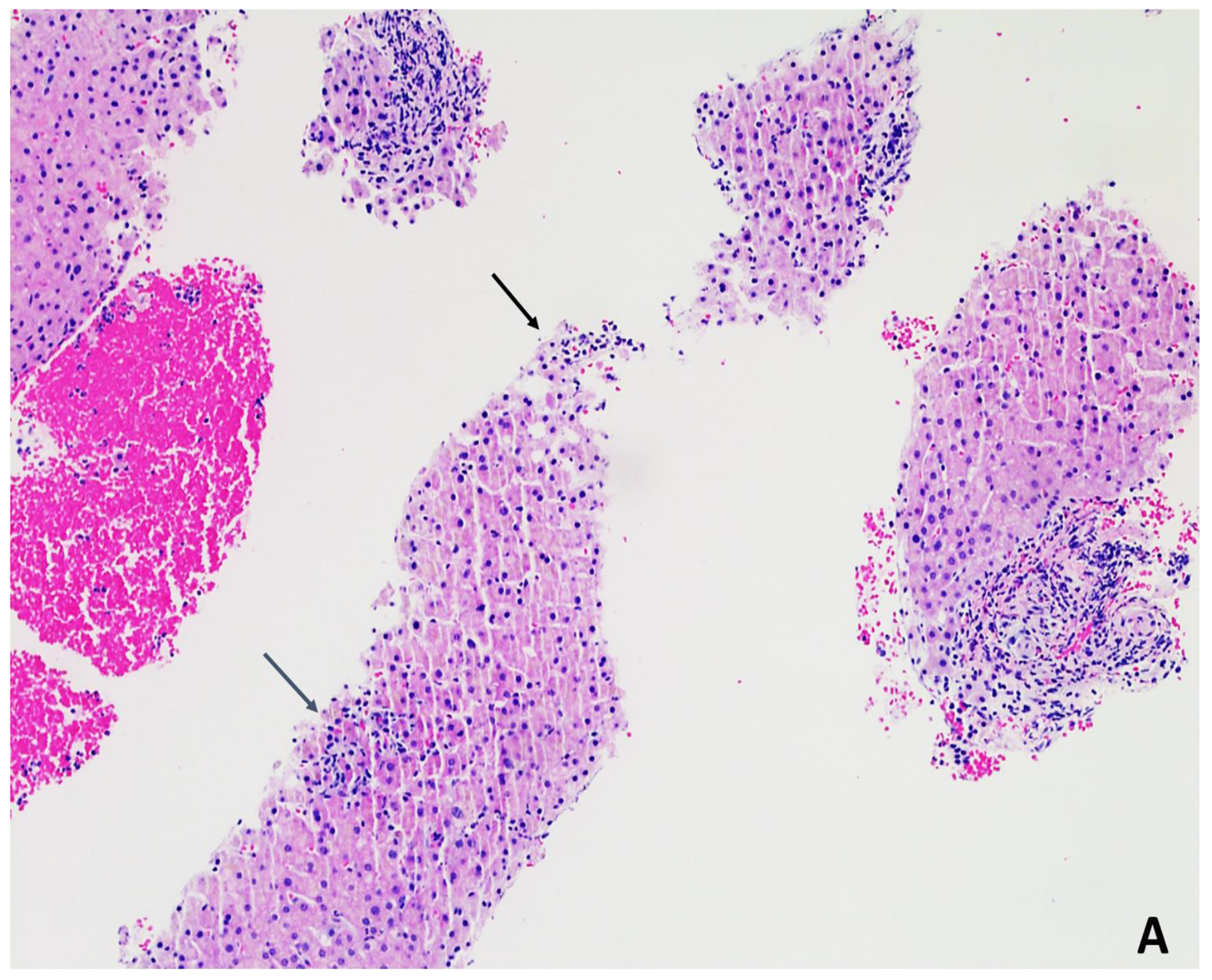
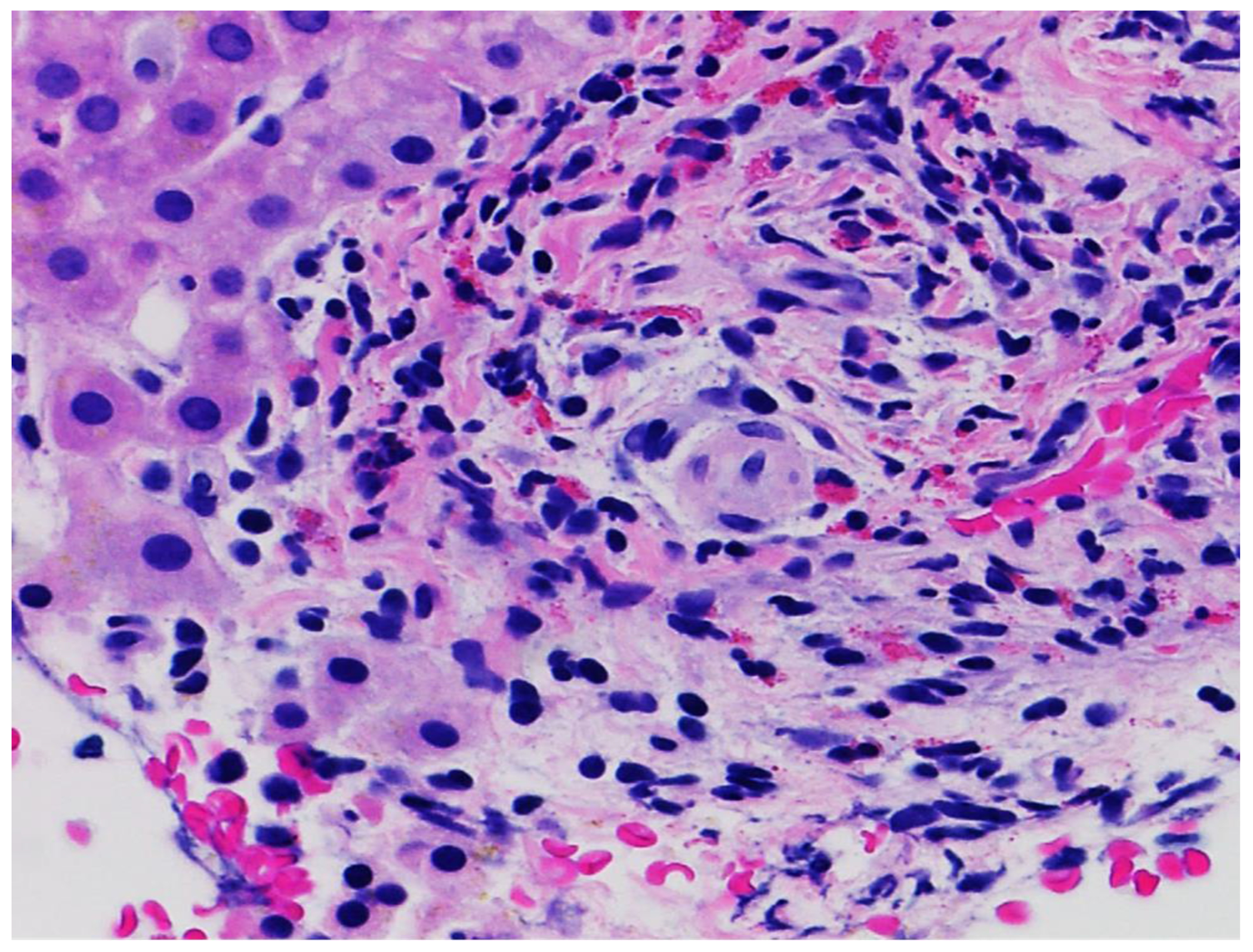
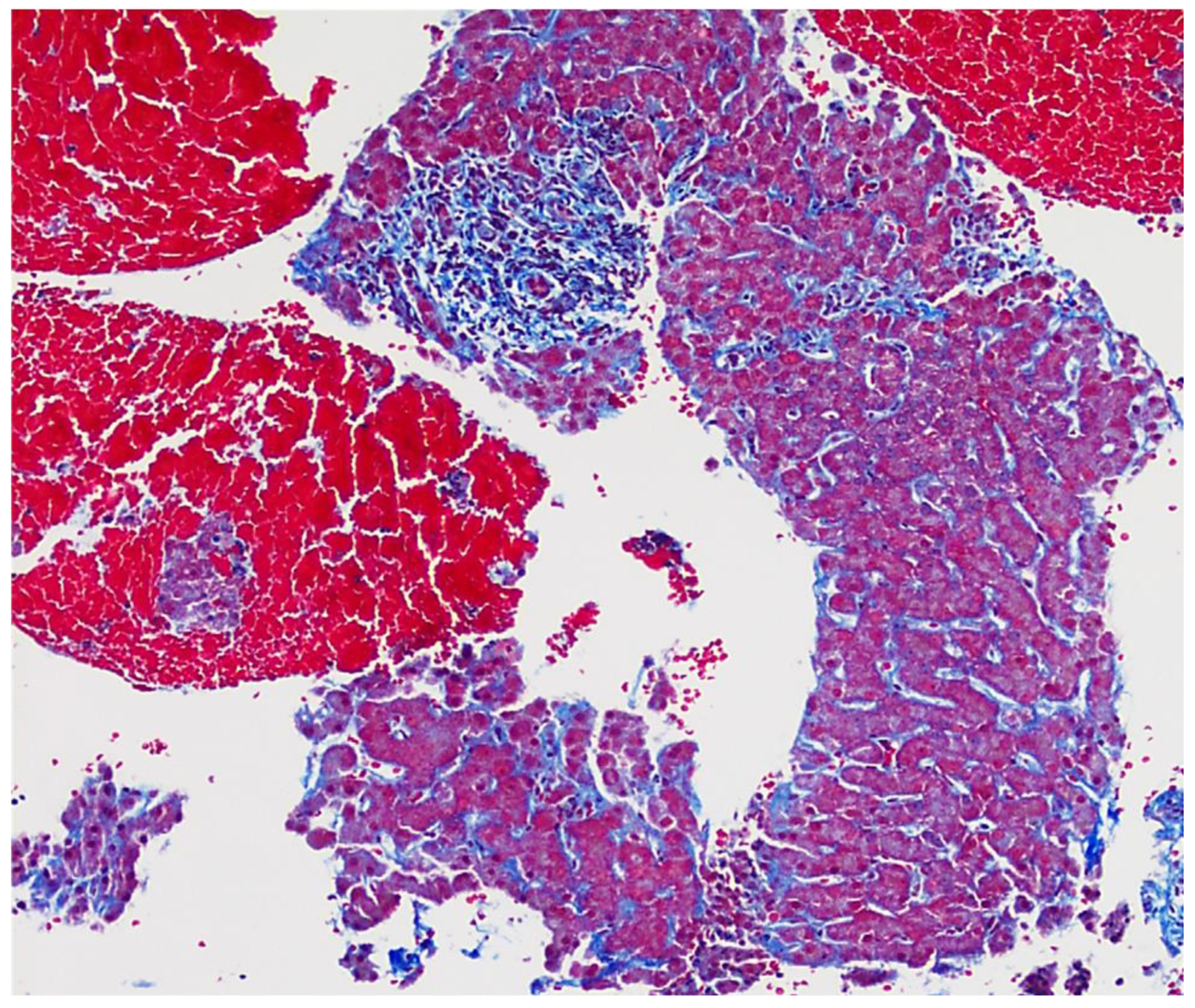
Liver histopathology
a. Liver biopsy specimen in one subject, obtained through endoscopic ultrasound, revealed fragmented liver tissue with foci of parenchymal necrosis as indicated by arrows.
b. A few complete portal tracts were present, showing focal fibrosis, mild lymphocytic and eosinophilic infiltration, mild ductular reaction and no interface hepatitis without steatosis, granulomatous reaction, or cholestasis.
c. Trichrome stain confirms focal portal fibrosis without fibrous septum formation.
Discussion
This paper describes and summarizes the clinical and laboratory features of 17 patients with FLLC associated liver injury which contained a new plant based, high protein food source from Tara flour. The account benefits from a chemical analysis of the product which identified the likely culprit agent and mechanism of injury. Out of the 27 different components in FLLC, Tara flour was the suspected cause of the injury as this ingredient had never been used in prior Daily Harvest™ products7. Tara flour is a plant-based protein extracted from the seeds of the South American tree Tara spinosa (Feuillé ex Molina) Britton & Rose (synonym: Caesalpinia spinosa (Molina) Kuntze). Tara spinosa is primarily cultivated in Peru which is responsible for more than 80% of the world supply8. The rapid onset of gastrointestinal symptoms and liver injury soon after exposure to the Tara flour suggested toxic as opposed to immune-mediated liver injury, which is typical of herbal and dietary supplements, including green tea extract, turmeric and garcinia cambogia9,10,11.
Implication that Tara flour was the etiology for injury is supported by the chemical analysis performed on FLLC and another report of similar injury pattern after ingestion of a smoothie product containing Tara four. Chemical analysis of FLLC confirmed the presence of Tara flour and absence of toxic compounds, microbial pathogens, mycotoxins, major allergens, pesticides, hepatitis A, norovirus and therefore, the Tara flour itself was implicated as the likely source of hepatotoxin7. The component in Tara flour responsible for the injury was assessed in studies by Chittiboyina et al. who demonstrated high concentrations of baikiain, an uncommon, non-protein amino acid. Baikiain was considered the likely cause as under physiological conditions, it could be metabolized into a reactive intermediate – 4,5-epoxypipecolic acid presumably via cytochrome P450 oxidation or 4- or 5-hydroxypipecolic acid via hydration that could lead to glutathione depletion by inactivation of detoxification enzymes, such as flavoenzyme l-pipecolate oxidase (PIPOX), whose reduction in tissue culture models can potentiate hydrogen peroxide mediated injury12. Thus, inhibition of these pathways may lead to oxidant induced liver injury, a key feature of acetaminophen hepatotoxicity13.
Using a multi-pronged approach, the authors demonstrated that crude extracts of Tara flour itself did not cause oxidant stress. However, oral administration of baikiain in ND4 mice resulted in marked increases in serum ALT levels and a reduction in liver glutathione, a well-recognized consequence of oxidant injury known to exacerbate and perpetuate liver injury7,14,15. This suggested that intestinal metabolism either by the microbiome or enterocytes may be required to produce liver injury.
There are two other references in the literature describing Tara flour induced liver injury. One described a similar case of hepatobiliary complications in a patient after consuming lentil crumbles from a mail-order vegan food company, presumably related to FLLC. This 35 year old woman also presented with abdominal pain, jaundice, and pruritus. Her liver biopsy showed non-specific portal lymphocytic inflammation without features of autoimmune hepatitis or cholangitis, including absence of hepatitis, fibrosis, plasma cells, or bile duct changes.16 A second publication described two cases of epigastric pain and acute liver injury who presented independently after consumption of a smoothie product containing Tara flour unrelated to FLLC. Rechallenge with the Tara flour containing smoothie led to recurrence of symptoms. It was thought that Tara flour was also the cause of the hepatotoxicity.17 As of October 21, 2022, the FDA received 393 CFSAN Adverse Event Reporting System (CAERS) reports and Consumer Complaints related to this product. There were 133 hospitalizations and no deaths in reports from 39 states with the last injury reported on September 4, 2022. On June 17, 2022, in response to consumer complaints submitted to the company, Daily Harvest™ voluntarily recalled FLLC.4
Baikiain is not the only example of a toxic, non-protein amino acid produced by plants. Another example is L-canavanine, a toxic amino acid similar to L-arginine which is found in many legumes and thought to protect them from predators18,19. Mimosine, found in the legume Leucaena leucocephala, demonstrates similar properties20. A recent report from France which involved plant toxins in undercooked kidney beans is yet another example of a plant derived toxin21. Baikiain, L-canavanine, and mimosine are all examples of plant-based food sources, in contrast to the experience with hepatotoxicity due to OxyELITE Pro. OxyELITE Pro was the commercial name for a variety of multi-ingredient dietary supplements marketed for weight loss. In 2013, the “Super Thermogenic” product was implicated in more than 50 cases of acute hepatitis and fatal, acute liver failure. In this instance, a synthetic (of unknown purity) product of aegeline (active ingredients include tannins, fatty oils, furocoumarins and furoquinolin alkaloids) was added to the commercial product and thought to be the culprit ingredient.22
Given the purported toxicity of baikiain, we speculate that oxidant injury due to its metabolism may account for the adverse events in both the intestine and liver, and that the degree of injury may be proportional to the amount consumed. As we hypothesize that baikiain metabolism is required to generate its toxicity, potential genetic variants in metabolic pathways may predispose to injury in certain populations or possible metabolism by the individual’s microbiome and thus may explain why only a small proportion of persons who consumed the product became ill. In addition, we speculate that given the cohort mean BMI 24, metabolic associated liver disease or steatohepatitis were unlikely to contribute to liver injury. In contrast to herbal supplements in which older age was associated with higher use2, the mean age of 41 in our study suggests a younger population group is more likely to consume natural food preparations and therefore be at risk for injury due to food supplements.
Conclusion
This work describes the clinical features of a plant based, high protein food source, Tara flour, that led to acute liver injury. The account benefits from a detailed chemical analysis of the product, which identified the potential culprit agent baikiain. Despite being composed of natural components, the frozen food supplement FLLC was the apparent cause of an outbreak of acute hepatotoxicity, probably due to inclusion of Tara flour that contained significant amounts of the non-protein amino acid baikiain, which in a murine model caused acute liver injury after oral ingestion. This report emphasizes the potential for unintended consequences of natural, plant based protein sources in foods including substantial illness. Thus, in assessing patients with liver injury, particularly in younger age groups, clinicians should query patients about natural foods in addition to herbal supplements.
Key Points.
An outbreak of acute liver injury took place in 2022 associated with introduction of a Daily Harvest™ product entitled French Lentil and Leek Crumble (FLLC), which contained a new plant based, high protein food source from Tara flour.
Acute liver injury associated with gastrointestinal symptoms occurred soon after ingestion, suggesting a toxin exposure.
Consumption of plant based food supplements composed of natural components may cause acute liver injury due to non-protein amino acids.
Financial Support:
Research reported in this publication was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK) of the National Institutes of Health under award numbers U01DK065211 (Indiana University), U01DK065184 (University of Michigan), U01DK065201 (University of North Carolina-Chapel Hill), U01DK083020 (University of Southern California), U01DK083027 (Thomas Jefferson University/Albert Einstein Medical Center), U01DK100928 (Icahn School of Medicine at Mount Sinai), and U24DK065176 (Duke University). Additional support is provided by the intramural programs of the NIDDK and National Cancer Institute. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
Disclosures: No related conflicts of interest.
References
- 1.Kelly JP, Kaufman DW, Kelley K, Rosenberg L, Anderson TE, Mitchell AA. Recent trends in use of herbal and other natural products. Arch Intern Med. 2005;165(3):281–286. doi: 10.1001/archinte.165.3.281 [DOI] [PubMed] [Google Scholar]
- 2.Rashrash M, Schommer JC, Brown LM. Prevalence and Predictors of Herbal Medicine Use Among Adults in the United States. J Patient Exp. 2017;4(3):108–113. doi: 10.1177/2374373517706612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Key TJ, Papier K, Tong TY. Plant-based diets and long-term health: findings from the EPIC-Oxford study. Proc Nutr Soc. 2022;81(2):190–198. doi: 10.1017/S0029665121003748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nutrition C for FS and A. Investigation of Adverse Event Reports: French Lentil & Leek Crumbles (June 2022). FDA. Published online October 16, 2023. Accessed December 21, 2023. https://www.fda.gov/food/outbreaks-foodborne-illness/investigation-adverse-event-reports-french-lentil-leek-crumbles-june-2022 [Google Scholar]
- 5.Fontana RJ, Watkins PB, Bonkovsky HL, et al. Drug-Induced Liver Injury Network (DILIN) prospective study: rationale, design and conduct. Drug Saf. 2009;32(1):55–68. doi: 10.2165/00002018-200932010-00005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rockey DC, Seeff LB, Rochon J, et al. Causality assessment in drug-induced liver injury using a structured expert opinion process: comparison to the Roussel-Uclaf causality assessment method. Hepatol Baltim Md. 2010;51(6):2117–2126. doi: 10.1002/hep.23577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chittiboyina AG, Ali Z, Avula B, et al. Is Baikiain in Tara Flour a Causative Agent for the Adverse Events Associated with the Recalled Frozen French Lentil & Leek Crumbles Food Product? - A Working Hypothesis. Chem Res Toxicol. 2023;36(6):818–821. doi: 10.1021/acs.chemrestox.3c00100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Acevedo-Rodríguez P, Strong MT. Catalogue of Seed Plants of the West Indies. Published online 2012. Accessed December 21, 2023. http://repository.si.edu/xmlui/handle/10088/17551
- 9.Hoofnagle JH, Bonkovsky HL, Phillips EJ, et al. HLA-B*35:01 and Green Tea Induced Liver Injury. Hepatol Baltim Md. 2021;73(6):2484–2493. doi: 10.1002/hep.31538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vuppalanchi R, Bonkovsky HL, Ahmad J, et al. Garcinia cambogia, Either Alone or in Combination With Green Tea, Causes Moderate to Severe Liver Injury. Clin Gastroenterol Hepatol Off Clin Pract J Am Gastroenterol Assoc. 2022;20(6):e1416–e1425. doi: 10.1016/j.cgh.2021.08.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Halegoua-DeMarzio D, Navarro V, Ahmad J, et al. Liver Injury Associated with Turmeric-A Growing Problem: Ten Cases from the Drug-Induced Liver Injury Network [DILIN]. Am J Med. 2023;136(2):200–206. doi: 10.1016/j.amjmed.2022.09.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Natarajan SK, Muthukrishnan E, Khalimonchuk O, Mott JL, Becker DF. Evidence for Pipecolate Oxidase in Mediating Protection Against Hydrogen Peroxide Stress. J Cell Biochem. 2017;118(7):1678–1688. doi: 10.1002/jcb.25825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Jaeschke H, McGill MR, Ramachandran A. Oxidant stress, mitochondria, and cell death mechanisms in drug-induced liver injury: lessons learned from acetaminophen hepatotoxicity. Drug Metab Rev. 2012;44(1):88–106. doi: 10.3109/03602532.2011.602688 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lu SC. Dysregulation of glutathione synthesis in liver disease. Liver Res. 2020;4(2):64–73. doi: 10.1016/j.livres.2020.05.003 [DOI] [Google Scholar]
- 15.Updates on our voluntary recall of French Lentil + Leek Crumbles. Accessed December 21, 2023. https://www.daily-harvest.com/content/french-lentil-leek-crumbles-advisory [Google Scholar]
- 16.Gaumnitz J, DuBroff J, Aguilar M. S3752 Novel Case of Tara Flour-Induced Liver Injury. Off J Am Coll Gastroenterol ACG. 2023;118(10S):S2420. [Google Scholar]
- 17.Chan SE, Smith CA. A food product as a potential serious cause of liver injury. Clin Toxicol Phila Pa. 2023;61(8):616–619. doi: 10.1080/15563650.2023.2256469 [DOI] [PubMed] [Google Scholar]
- 18.Bence AK, Crooks PA. The mechanism of L-canavanine cytotoxicity: arginyl tRNA synthetase as a novel target for anticancer drug discovery. J Enzyme Inhib Med Chem. 2003;18(5):383–394. doi: 10.1080/1475636031000152277 [DOI] [PubMed] [Google Scholar]
- 19.Rosenthal GA. L-Canavanine: a higher plant insecticidal allelochemical. Amino Acids. 2001;21(3):319–330. doi: 10.1007/s007260170017 [DOI] [PubMed] [Google Scholar]
- 20.Yeung PKK, Wong FTW, Wong JTY. Mimosine, the Allelochemical from the leguminous tree Leucaena leucocephala, selectively enhances cell proliferation in dinoflagellates. Appl Environ Microbiol. 2002;68(10):5160–5163. doi: 10.1128/AEM.68.10.5160-5163.2002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Desk N. Plant toxin in undercooked kidney beans behind French outbreak. Food Safety News. Published August 20, 2023. Accessed December 21, 2023. https://www.foodsafetynews.com/2023/08/plant-toxin-in-undercooked-kidney-beans-behind-french-outbreak/ [Google Scholar]
- 22.OxyELITE Pro In: LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases; 2012. Accessed April 28, 2024. http://www.ncbi.nlm.nih.gov/books/NBK548935/ [PubMed] [Google Scholar]


