Abstract
Background
Paediatric patients undergoing surgery for congenital heart disease (CHD) are at risk for postoperative low cardiac output syndrome (LCOS) and mortality. LCOS affects up to 25% of children after heart surgery. It consists of reduced myocardial function and increases postoperative morbidity, prolongs mechanical ventilation, and lengthens the duration of intensive care unit (ICU) stay. Pharmacological prophylaxis involves inotropes, including catecholamines, phosphodiesterase III inhibitors, or calcium sensitisers, to enhance myocardial contractility. It is unclear whether they are effective in preventing LCOS or death in this vulnerable population.
Objectives
1. To evaluate the relative benefits and harms of inotropes for the prevention of LCOS and mortality in paediatric patients undergoing surgery for CHD.
2. To generate a clinically useful ranking of prophylactic inotropes for the prevention of LCOS and mortality in paediatric patients undergoing surgery for CHD according to benefits and harms.
Search methods
We searched CENTRAL, MEDLINE, Embase, Web of Science, and clinical trial registries, most recently in December 2023 and April 2024. We also checked reference lists from identified studies and review articles. We did not apply any language restrictions.
Selection criteria
We included randomised controlled trials comparing inotropes from one drug class (catecholamines, phosphodiesterase type III inhibitors, calcium sensitisers) to another (either alone or in combination) or placebo, in paediatric patients (birth to 18 years of age) undergoing surgery for CHD.
Data collection and analysis
Two review authors independently selected studies, extracted data, assessed risk of bias, and rated the certainty of evidence using the CINeMA framework. We performed random‐effects network and pairwise meta‐analyses comparing the relative effects of each possible pair of medications with each other or placebo. Where meta‐analysis was not possible, we provided a narrative description of the results. We ranked the prophylactic medications according to their effects relative to each other. The primary outcomes were all‐cause mortality within 30 days, time to death, and LCOS incidence; secondary outcomes were length of ICU stay, length of hospital stay, duration of mechanical ventilation, inotrope score, mechanical circulatory support, and adverse events.
Main results
We included 13 studies with 937 participants. All except two multicentre studies were conducted at single tertiary care hospitals. Participants comprised children from birth to 14 years of age undergoing surgery for different types of CHD on cardiopulmonary bypass. Five studies compared levosimendan versus milrinone; two compared levosimendan versus placebo; two compared milrinone versus placebo (one comparing two different doses); one compared levosimendan versus dobutamine, another milrinone versus dobutamine. Two studies used combinations of inotropes. Study duration was between less than one year and 5.3 years, with follow‐up mostly during ICU or hospital stay. Funding sources included governmental bodies and hospital departments, but also drug manufacturers. We downgraded the certainty of evidence for high risk of bias at study level, or imprecision at comparison level.
Primary outcomes
Compared to placebo, levosimendan likely results in a large reduction in mortality (risk ratio (RR) 0.57, 95% confidence interval (CI) 0.15 to 2.13) and milrinone likely results in no difference (RR 0.97, 95% CI 0.11 to 8.49), whereas for dobutamine, no effect was estimable; all moderate‐certainty evidence (9 studies, 557 participants, 14 events).
LCOS was largely reduced with levosimendan (RR 0.45, 95% CI 0.24 to 0.83; high‐certainty evidence), likely largely reduced with milrinone (RR 0.46, 95% CI 0.24 to 0.89; moderate‐certainty evidence), and may be reduced with low‐dose milrinone (RR 0.7, 95% CI 0.39 to 1.28; low‐certainty evidence), compared with placebo (5 studies, 513 participants, 85 events).
Secondary outcomes
The length of ICU stay may be no different with levosimendan (ratio of means (ROM) 1.12, 95% CI 0.77 to 1.63; low‐certainty evidence), and is likely no different with milrinone (ROM 1.13, 95% CI 0.75 to 1.69) or with dobutamine (ROM 1.11, 95% CI 0.66 to 1.86), compared with placebo (9 studies, 577 participants); both moderate‐certainty evidence.
The length of hospital stay, compared with placebo, is likely no different with levosimendan (ROM 1.03, 95% CI 0.84 to 1.27) or with milrinone (ROM 1, 95% CI 0.78 to 1.3), but is likely reduced with dobutamine (ROM 0.68, 95% CI 0.37 to 1.26); all moderate‐certainty evidence (7 studies, 297 participants).
The duration of mechanical ventilation, compared with placebo, is likely increased with levosimendan (ROM 1.17, 95% CI 0.65 to 2.12) or with milrinone (ROM 1.25, 5% CI 0.67 to 2.36) and is likely no different with dobutamine (ROM 1.04, 95% CI 0.45 to 2.38); all moderate‐certainty evidence (9 studies, 577 participants).
There is moderate‐certainty evidence that adverse events are likely increased with levosimendan (incidence rate ratio (IRR) 1.23, 95% CI 0.78 to 1.96) or dobutamine (IRR 1.24, 95% CI 0.75 to 2.03) and low‐certainty evidence that they may be increased with milrinone (IRR 1.31, 95% CI 0.96 to 1.79) and decreased with low‐dose milrinone (IRR 0.84, 95% CI 0.47 to 1.5), compared with placebo (8 studies, 706 participants, 380 events).
Authors' conclusions
Levosimendan likely results in a large reduction in mortality compared to placebo in paediatric patients undergoing surgery for congenital heart disease, whereas milrinone likely results in no difference, and the effect of dobutamine is unknown.
Low cardiac output syndrome (LCOS) is largely reduced with levosimendan, likely largely reduced with milrinone, and may be reduced with low‐dose milrinone, compared to placebo.
The length of ICU stay may be no different with levosimendan and is likely no different with milrinone or with dobutamine, compared to placebo.
The length of hospital stay is likely no different with levosimendan or with milrinone, but is likely reduced with dobutamine, compared to placebo.
The duration of mechanical ventilation is likely increased with levosimendan or with milrinone and is likely no different with dobutamine, compared to placebo.
Adverse events are likely increased with levosimendan or dobutamine, and may be increased with milrinone and decreased with low‐dose milrinone, compared to placebo.
The evidence is based on few, heterogeneous studies, with small numbers of patients and short follow‐up periods. Future research should include large numbers of patients, consistently report all co‐interventions, and ensure the longest possible follow‐up.
Keywords: Adolescent; Child; Child, Preschool; Humans; Infant; Infant, Newborn; Bias; Cardiac Output, Low; Cardiac Output, Low/mortality; Cardiac Output, Low/prevention & control; Cardiac Surgical Procedures; Cardiac Surgical Procedures/adverse effects; Cardiac Surgical Procedures/mortality; Cardiotonic Agents; Cardiotonic Agents/therapeutic use; Heart Defects, Congenital; Heart Defects, Congenital/mortality; Heart Defects, Congenital/surgery; Length of Stay; Milrinone; Milrinone/therapeutic use; Network Meta-Analysis; Postoperative Complications; Postoperative Complications/mortality; Postoperative Complications/prevention & control; Randomized Controlled Trials as Topic; Simendan; Simendan/therapeutic use
Plain language summary
Do drugs that enhance the beating force of the heart prevent low cardiac output syndrome and death in children undergoing surgery for congenital heart disease?
Key messages
‐ Using levosimendan as a prevention makes it less likely that children undergoing heart surgery will die. The risk for reduced heart function is largely decreased with levosimendan and likely also with milrinone, compared to placebo. Side effects are likely more common with levosimendan or dobutamine and may be more common with milrinone but less common with low‐dose milrinone, compared to placebo.
‐ Our confidence in the evidence is limited because only a small number of patients received each medication, the patients' ages and diseases differed, and there was short follow‐up or incomplete reporting of outcomes.
What is low cardiac output syndrome?
Children who undergo heart surgery are at risk of reduced heart function after surgery (low cardiac output syndrome, LCOS). This affects up to one‐quarter of these children, and there is a risk of death. Reduced heart function causes children to stay longer on breathing machines and longer in the intensive care unit (ICU).
What did we want to find out?
We wanted to find out if any medications that increase the pumping force of the heart prevent reduced heart function or death in children following heart surgery, if there are associated harms, and if we could rank the treatments. These medications are used when LCOS is present, but sometimes also to prevent it. There have been a few studies comparing some of these drugs against each other, each with few patients. By conducting a network meta‐analysis, where several different treatments can be compared to each other, we hoped to provide further information on how to prevent low heart function or death in children who undergo heart surgery.
What did we do?
We searched medical literature databases for planned, ongoing, and completed studies that compared any of these medications to each other, to standard care (measures commonly performed on the ICU after heart surgery), or to placebo (a solution that looks the same as the medication infusion, but has no active ingredient). Two review authors collected and reviewed the data independently. We then calculated how effective the drugs were when studies' results were combined, summarised the results, and rated our confidence in the evidence.
What did we find?
We found 13 studies comparing levosimendan, milrinone, dobutamine, dopamine, combinations of these with other drugs, or placebo, with 937 total participants. Two of the studies were conducted at multiple hospitals. Participants differed between the studies; for example, by age, from newborns less than one month of age to children aged 14 years old. All studies included children undergoing surgery for congenital heart defects by temporarily using a heart‐lung machine. Some studies examined only children with two heart chambers, one only children with one heart chamber, and some both.
Five studies compared levosimendan versus milrinone, two levosimendan versus placebo, and two milrinone versus placebo. One study compared levosimendan versus dobutamine, another milrinone versus dobutamine, and two studies used combination treatments. Patients were watched mostly for the duration of their stay in the ICU or in the hospital. The study authors received funding from governments, hospitals, and non‐profit organisations, but also from drug manufacturers; in some cases, we do not know how they were funded.
Key results
Levosimendan results in fewer patients dying after childhood heart surgery than placebo, milrinone likely does not change that risk, and for dobutamine, we do not know the risk (14 events, 557 participants, 9 studies).
The risk of reduced heart function was largely reduced with levosimendan and likely largely reduced with milrinone, and may be reduced with low‐dose milrinone, compared to placebo (85 events, 513 participants, 5 studies).
The length of ICU stay may be no different with levosimendan and is likely no different with milrinone or with dobutamine, compared to placebo (9 studies, 577 participants).
The length of hospital stay is likely no different with levosimendan or with milrinone, but is likely reduced with dobutamine, compared to placebo (7 studies, 297 participants).
The time children need to be on a respirator after heart surgery is likely longer with levosimendan or milrinone and is likely no different with dobutamine, compared to placebo (9 studies, 577 participants).
Side effects in general are likely more common with levosimendan or dobutamine and may be more common with milrinone and less common with low‐dose milrinone, compared to placebo (8 studies, 706 participants, 380 events).
What are the limitations of the evidence?
We found few studies with different trial designs where often not all of these effects were looked at. Our confidence in the evidence is limited due to the small number of included patients for each drug treatment, and because we were not always convinced that all patients in a study would have been treated in a completely comparable way, that they were followed up for long enough, or that all study results were completely reported. Therefore, all results of our network meta‐analysis must be viewed cautiously.
How up‐to‐date is this evidence?
This evidence is up‐to‐date to December 2023.
Summary of findings
Summary of findings 1. All‐cause mortality within 30 days.
| Estimate of effects, confidence intervals, and certainty of the evidence for prevention of mortality for paediatric patients undergoing surgery for congenital heart disease | ||||||||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: levosimendan, milrinone, dobutamine, placebo, levosimendan + milrinone + dopamine, milrinone + dopamine, levosimendan + dobutamine, milrinone + dobutamine Comparator (reference): placebo or combination of milrinone + dopamine or combination of milrinone + dobutamine Outcome: prevention of mortality Setting: inpatient |
Figure 1 | |||||||
|
Total studies: 9 Total participants: 557 Total events: 14 |
Relative effect (95% CI) | Anticipated absolute effect (95% CI) | No. of participants (studies) | Certainty of the evidence | Ranking (SUCRA*) | Comments | ||
| Without intervention | With intervention | Difference | ||||||
| Levosimendan |
RR 0.57 (0.15 to 2.13) Network estimate |
43 per 1000a | 25 per 1000 | 18 fewer per 1000 (37 fewer to 49 more) | 216 (7 RCTs) |
Moderate Due to imprecision |
0.76 | Levosimendan likely results in a large reduction in mortality compared to placebo |
| Milrinone |
RR 0.97 (0.11 to 8.49) Network estimate |
43 per 1000a | 42 per 1000 | 1 fewer per 1000 (38 fewer to 322 more) | 147 (6 RCTs) |
Moderate Due to imprecision |
0.39 | Milrinone likely results in no difference in mortality compared to placebo |
| Dobutamine | Excluded due to absence of events | ‐ | ‐ | ‐ | 51 (2 RCTs) |
‐ | Excluded due to absence of events | ‐ |
| Placebo | RR 1 | Not estimable | Not estimable | Not estimable | 143 (3 RCTs) |
Reference comparator | 0.35 | ‐ |
| Heterogeneity: tau2 = 0; I2 = 0% (95% CI 0% to 89.6%) Q total 0.86 (df = 2; P = 0.65) Q within designs 0.86 (df = 2; P = 0.65) Q between designs 0 (df = 0) | ||||||||
*SUCRA: surface under the cumulative ranking curve, calculated with R package netmeta
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Explanatory footnotes
aBaseline risk (assumed control risk) obtained from Wang 2019, placebo group.
Certainty of the evidence for each intervention was downgraded one step to 'Moderate' due to imprecision.
Abbreviation(s) CI: confidence interval; df: degrees of freedom; No.: number; RCT: randomised controlled trial; RR: risk ratio; SUCRA: surface under the cumulative ranking curve.
1.
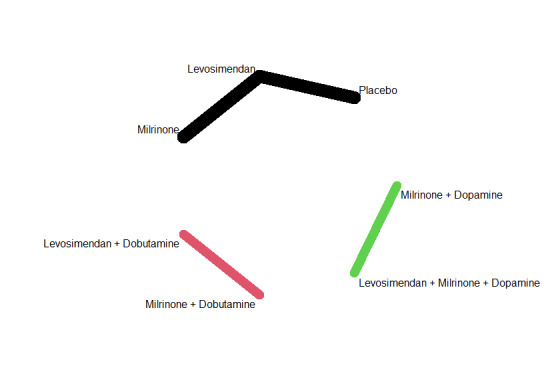
Networks for outcome 'All‐cause mortality within 30 days'.
Key There are three networks, one with levosimendan/placebo/milrinone, one with milrinone + dobutamine versus levosimendan + dobutamine, and one with levosimendan + milrinone + dopamine versus milrinone + dopamine.
Summary of findings 2. Time to death.
| Estimate of effects, confidence intervals, and certainty of the evidence for time to death in paediatric patients undergoing surgery for congenital heart disease | |||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: levosimendan Comparator (reference): placebo Outcome: time to death Setting: inpatient | Figure 2 | ||
| Total studies: 1 Total participants: 187 Total events: 7 | Relative effect (95% CI) | No. of participants (studies) | Comments |
| Levosimendan | HR 0.74 (0.16 to 3.28) | 94 (1 RCT) | Levosimendan likely results in a reduction of momentary probability of death compared to placebo |
| Placebo | HR 1 | 93 (1 RCT) | ‐ |
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Abbreviation(s) CI: confidence interval; HR: hazard ratio; No.: number; RCT: randomised controlled trial.
2.
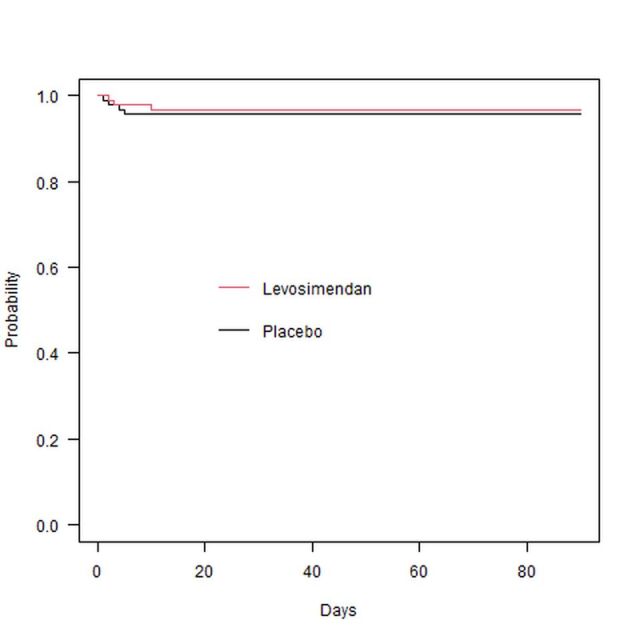
Time to death curves ‐ direct evidence only, from Wang 2019. The 'Probability' axis shows the probability of being alive, the 'Days' axis shows the number of days after surgery.
Summary of findings 3. Low cardiac output syndrome.
| Estimate of effects, confidence intervals, and certainty of the evidence for prevention of LCOS for paediatric patients undergoing surgery for congenital heart disease | ||||||||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: milrinone, milrinone low dose, levosimendan Comparator (reference): placebo or combination of milrinone + dopamine Outcome: prevention of LCOS Setting: inpatient |
Figure 3 | |||||||
|
Total studies: 5 Total participants: 513 Total events: 85 |
Relative effect (95% CI) | Anticipated absolute effect (95% CI) | No. of participants (studies) | Certainty of the evidence | Ranking (SUCRA*) | Comments | ||
| Without intervention | With intervention | Difference | ||||||
| Levosimendan |
RR 0.45 (0.24 to 0.83) Network estimate |
267 per 1000a | 120 per 1000 | 147 fewer per 1000 (203 fewer to 45 fewer) | 144 (4 RCTs) |
High Due to heterogeneity, but large magnitude of effect |
0.80 | Levosimendan results in a large reduction in LCOS, compared to placebo |
| Milrinone |
RR 0.46 (0.24 to 0.89) Network estimate |
267 per 1000a | 123 per 1000 | 144 fewer per 1000 (203 fewer to 29 fewer) |
122 (4 RCTs) |
Moderate Due to within‐study bias and heterogeneity, but large magnitude of effect |
0.77 | Milrinone likely results in a large reduction in the incidence of LCOS, compared to placebo |
| Milrinone low‐dose |
RR 0.70 (0.39 to 1.28) Network estimate |
267 per 1000a | 187 per 1000 | 80 fewer per 1000 (163 fewer to 75 more) |
79 (1 RCT) |
Low Due to within‐study bias and imprecision |
0.39 | Milrinone low dose may reduce the incidence of LCOS, compared to placebo |
| Placebo | RR 1 | Not estimable | Not estimable | Not estimable | 168 (2 RCTs) |
Reference comparator | 0.05 | ‐ |
| Heterogeneity: tau2 = 0; I2 = 0% (95% CI 0% to 89.6%). Q total 1.2 (df = 2, P = 0.55) Q within designs 0.05 (df = 1, P = 0.82) Q between designs 1.14 (df = 1, P = 0.29) | ||||||||
*SUCRA: surface under the cumulative ranking curve, calculated with R package netmeta
df: degrees of freedom
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Explanatory footnotes
aBaseline risk (assumed control risk) obtained from Hoffman 2003, placebo group.
Certainty of the evidence for levosimendan was downgraded one step due to heterogeneity, but upgraded one step due to large magnitude of effect, resulting in 'High'.
Certainty of the evidence for milrinone was downgraded two steps due to within‐study bias and heterogeneity, but upgraded one step due to large magnitude of effect, resulting in 'Moderate'.
Certainty of the evidence for milrinone low dose was downgraded two steps due to within‐study bias and imprecision, resulting in 'Low'.
Abbreviation(s) CI: confidence interval; df: degrees of freedom; LCOS: low cardiac output syndrome; No.: number; RCT: randomised controlled trial; RR: risk ratio; SUCRA: surface under the cumulative ranking curve.
3.
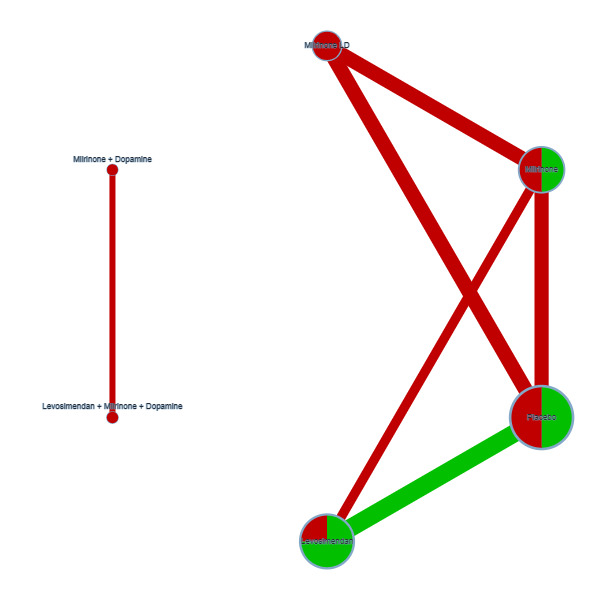
Networks for outcome 'Low cardiac output syndrome'.
Key Colours represent risk of bias (green = low risk, red = high risk). The size of the nodes represents the number of participants who received a certain treatment. Line thicknesses represent the sample size, while line colours represent the highest risk of bias among the relevant studies. There are two networks, one with levosimendan/placebo/milrinone/milrinone LD, and one with levosimendan + milrinone + dopamine versus milrinone + dopamine.
Abbreviation(s) LD: low‐dose.
Summary of findings 4. Length of intensive care stay.
| Estimate of effects, confidence intervals, and certainty of the evidence for length of intensive care stay for paediatric patients undergoing surgery for congenital heart disease | ||||||||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: levosimendan, milrinone, dobutamine, placebo, levosimendan + milrinone + dopamine, milrinone + dopamine, levosimendan + dobutamine, milrinone + dobutamine Comparator (reference): placebo or combination of milrinone + dopamine or combination of milrinone + dobutamine Outcome: length of intensive care stay Setting: inpatient |
Figure 4 | |||||||
|
Total studies: 9 Total participants: 577 |
Relative effect (95% CI) | Anticipated absolute effect (95% CI) | No. of participants (studies) | Certainty of the evidence | Ranking (SUCRA*) | Comments | ||
| Without intervention | With intervention | Difference | ||||||
| Levosimendan |
ROM 1.12 (0.77 to 1.63) Network estimate |
4.9 daysa | 5.5 days | 0.6 days more (1.1 less to 3.1 more) | 225 (7 RCTs) |
Low Due to imprecision and heterogeneity |
0.42 | Levosimendan may result in little to no difference in length of intensive care stay, compared to placebo |
| Milrinone |
ROM 1.13 (0.75 to 1.69) Network estimate |
4.9 daysa | 5.5 days | 0.6 days more (1.2 less to 3.4 more) | 158 (6 RCTs) |
Moderate Due to imprecision |
0.41 | Milrinone likely results in little to no difference in length of intensive care stay, compared to placebo |
| Dobutamine |
ROM 1.11 (0.66 to 1.86) Network estimate |
4.9 daysa | 5.4 days | 0.5 days more (1.7 less to 4.2 more) | 51 (2 RCTs) |
Moderate Due to imprecision |
0.47 | Dobutamine likely results in little to no difference in length of intensive care stay, compared to placebo |
| Placebo | ROM 1 | Not estimable | Not estimable | Not estimable | 143 (3 RCTs) |
Reference comparator | 0.70 | ‐ |
| Heterogeneity: tau2 = 0.0608; I2 = 68.3% (95% CI 29.9% to 85.7%) Q total 18.94 (df = 6; P = 0.004) Q within designs 17.13 (df = 4; P = 0.002) Q between designs 1.81 (df = 2; P = 0.40) | ||||||||
*SUCRA: surface under the cumulative ranking curve, calculated with R package netmeta
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Explanatory footnotes
aBaseline risk (assumed control risk) obtained from Wang 2019, placebo group, median length of intensive care stay.
Certainty of the evidence for levosimendan was downgraded two steps due to imprecision and heterogeneity, resulting in 'Low'.
Certainty of the evidence for milrinone and for dobutamine was downgraded one step due to imprecision, resulting in 'Moderate'.
Abbreviation(s) CI: confidence interval; df: degrees of freedom; No.: number; RCT: randomised controlled trial; ROM: ratio of means; SUCRA: surface under the cumulative ranking curve.
4.
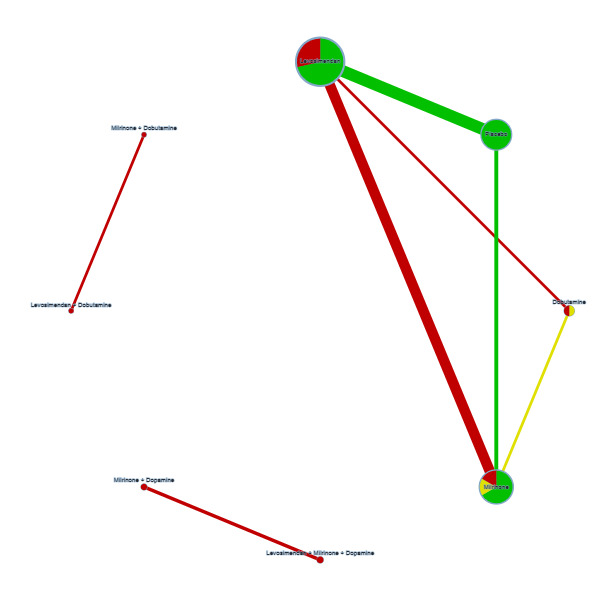
Network for outcome 'Length of intensive care stay'.
Key Colours represent risk of bias (green = low risk, yellow = unclear risk, red = high risk). The size of the nodes represents the number of participants who received a certain treatment. Line thicknesses represent the sample size, while line colours represent the highest risk of bias among the relevant studies. There are three networks, one with levosimendan/placebo/milrinone/dobutamine, one with milrinone + dobutamine versus levosimendan + dobutamine, and one with levosimendan + milrinone + dopamine versus milrinone + dopamine.
Summary of findings 5. Length of hospital stay.
| Estimate of effects, confidence intervals, and certainty of the evidence for length of hospital stay for paediatric patients undergoing surgery for congenital heart disease | ||||||||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: levosimendan, milrinone, dobutamine, placebo Comparator (reference): placebo Outcome: length of hospital stay Setting: inpatient |
Figure 5 | |||||||
|
Total studies: 7 Total participants: 497 |
Relative effect (95% CI) | Anticipated absolute effect (95% CI) | No. of participants (studies) | Certainty of the evidence | Ranking (SUCRA) | Comments | ||
| Without intervention | With intervention | Difference | ||||||
| Levosimendan |
ROM 1.03 (0.84 to 1.27) Network estimate |
20 daysa | 21 days | 1 day more (3 less to 5 more) | 185 (5 RCTs) |
Moderate Due to imprecision |
0.28 | Levosimendan likely results in no difference in length of hospital stay, compared to placebo |
| Milrinone |
ROM 1 (0.78 to 1.30) Network estimate |
20 daysa | 20 days | Same number of days (4 less to 6 more) | 222 (7 RCTs) |
Moderate Due to imprecision |
0.41 | Milrinone likely results in no difference in length of hospital stay, compared to placebo |
| Dobutamine |
ROM 0.68 (0.37 to 1.26) Network estimate |
20 daysa | 14 days | 6 days less (13 less to 5 more) | 26 (1 RCT) |
Moderate Due to imprecision |
0.91 | Dobutamine likely reduces the length of hospital stay, compared to placebo |
| Placebo | ROM 1 | Not estimable | Not estimable | Not estimable | 199 (3 RCTs) |
Reference comparator | 0.41 | ‐ |
| Heterogeneity: tau2 = 0.01; I2 = 31.2% (95% CI 0.0% to 73.6%) Q total 5.81 (df = 4; P = 0.21) Q within designs 5.46 (df = 3; P = 0.14) Q between designs 0.36 (df = 1; P = 0.55) | ||||||||
*SUCRA: surface under the cumulative ranking curve, calculated with R package netmeta
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Explanatory footnotes
aBaseline risk (assumed control risk) obtained from Wang 2019, placebo group, median length of hospital stay.
Certainty of the evidence for each intervention was downgraded one step due to imprecision, resulting in 'Moderate'.
Abbreviation(s) CI: confidence interval; df: degrees of freedom; No.: number; RCT: randomised controlled trial; ROM: ratio of means; SUCRA: surface under the cumulative ranking curve.
5.
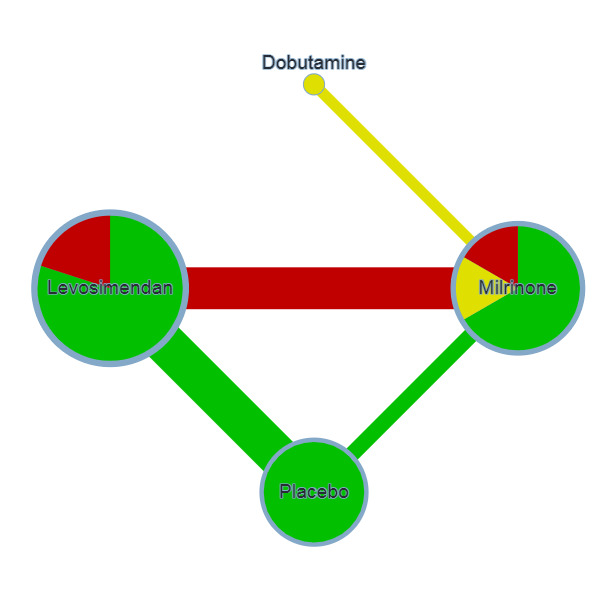
Network for outcome 'Length of hospital stay'.
Key Colours represent risk of bias (green = low risk, yellow = unclear risk, red = high risk). The size of the nodes represents the number of participants who received a certain treatment. Line thicknesses represent the sample size, while line colours represent the highest risk of bias among the relevant studies. There is one network with levosimendan/placebo/milrinone/dobutamine.
Summary of findings 6. Duration of mechanical ventilation.
| Estimate of effects, confidence intervals, and certainty of the evidence for duration of mechanical ventilation for paediatric patients undergoing surgery for congenital heart disease | ||||||||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: levosimendan, milrinone, dobutamine, placebo, levosimendan + milrinone + dopamine, milrinone + dopamine, levosimendan + dobutamine, milrinone + dobutamine Comparator (reference): placebo or combination of milrinone + dopamine or combination of milrinone + dobutamine Outcome: duration of mechanical ventilation Setting: inpatient |
Figure 6 | |||||||
|
Total studies: 9 Total participants: 577 |
Relative effect (95% CI) | Anticipated absolute effect (95% CI) | No. of participants (studies) | Certainty of the evidence | Ranking (SUCRA*) | Comments | ||
| Without intervention | With intervention | Difference | ||||||
| Levosimendan |
ROM 1.17 (0.65 to 2.12) Network estimate |
39.5 hoursa | 46.2 hours | 6.7 hours more (13.8 less to 44.2 more) | 225 (7 RCTs) |
Moderate Due to imprecision |
0.42 | Levosimendan likely increases the duration of mechanical ventilation slightly, compared to placebo |
| Milrinone |
ROM 1.25 (0.67 to 2.36) Network estimate |
39.5 hoursa | 49 hours | 9.9 hours more (13 less to 53.7 more) | 222 (7 RCTs) |
Moderate Due to imprecision |
0.30 | Milrinone likely increases the duration of mechanical ventilation, compared to placebo |
| Dobutamine |
ROM 1.04 (0.45 to 2.38) Network estimate |
39.5 hoursa | 41.1 hours | 1.6 hours more (21.7 less to 54.5 more) | 51 (2 RCTs) |
Moderate Due to imprecision |
0.61 | Dobutamine likely results in no difference in the duration of mechanical ventilation, compared to placebo |
| Placebo | ROM 1 | Not estimable | Not estimable | Not estimable | 214 (4 RCTs) |
Reference comparator | 0.66 | ‐ |
| Heterogeneity: tau2 = 0.1475; I2 = 67.9% (95% CI 28.7% to 85.5%) Q total 18.67 (df = 6; P = 0.0048) Q within designs 11.73 (df = 4; P = 0.0195) Q between designs 6.94 (df = 2; P = 0.0311) | ||||||||
*SUCRA: surface under the cumulative ranking curve, calculated with R package netmeta
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Explanatory footnotes
aBaseline risk (assumed control risk) obtained from Wang 2019, placebo group, median duration of mechanical ventilation.
Certainty of the evidence for each intervention was downgraded one step due to imprecision, resulting in 'Moderate'.
Abbreviation(s) CI: confidence interval; df: degrees of freedom; No.: number; RCT: randomised controlled trial; ROM: ratio of means; SUCRA: surface under the cumulative ranking curve.
6.
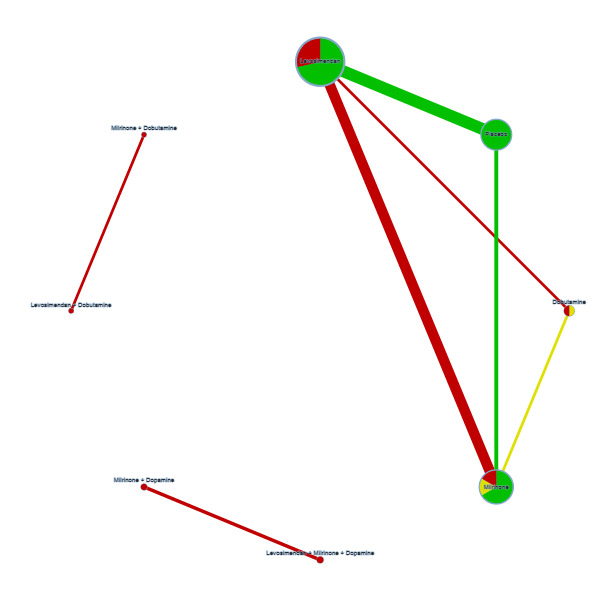
Networks for outcome 'Duration of mechanical ventilation'.
Key Colours represent risk of bias (green = low risk, yellow = unclear risk, red = high risk). The size of the nodes represents the number of participants who received a certain treatment. Line thicknesses represent the sample size, while line colours represent the highest risk of bias among the relevant studies. There are three networks, one with levosimendan/placebo/milrinone/dobutamine, one with milrinone + dobutamine versus levosimendan + dobutamine, and one with levosimendan + milrinone + dopamine versus milrinone + dopamine.
Summary of findings 7. Adverse events.
| Estimate of effects, confidence intervals, and certainty of the evidence for adverse events of inotropes for paediatric patients undergoing surgery for congenital heart disease | ||||||||
| Population: paediatric patients undergoing surgery for congenital heart disease Interventions: levosimendan, milrinone, milrinone low dose, dobutamine, placebo, levosimendan + dobutamine, milrinone + dobutamine Comparator (reference): placebo or combination of milrinone + dopamine or combination of milrinone + dobutamine Outcome: adverse events (ECMO or VAD, arrhythmia, hypotension, thrombocytopaenia, elevated liver enzymes, hypokalaemia, reduced LVEF or LVFS, requirement for renal replacement therapy) Setting: inpatient |
Figure 7 | |||||||
|
Total studies: 8 Total participants: 706 Total events: 380 |
Incidence rate ratio (95% CI)a | Anticipated absolute effect (95% CI) | No. of participants (studies) | Certainty of the evidence | Ranking (SUCRA*) | Comments | ||
| Without intervention | With intervention | Difference | ||||||
| Levosimendan | 1.23 (0.78 to 1.96) | 259 per 1000b | 319 per 1000 | 60 more per 1000 (57 fewer to 249 more) | 172 (5 RCTs) |
Moderate Due to imprecision |
0.36 | Levosimendan likely increases the incidence of adverse events, compared to placebo |
| Milrinone | 1.31 (0.96 to 1.79) | 259 per 1000b | 339 per 1000 | 80 more per 1000 (10 fewer to 205 more) | 204 (6 RCTs) |
Low Due to within‐study bias and imprecision |
0.22 | Milrinone may increase the incidence of adverse events, compared to placebo |
| Milrinone low dose | 0.84 (0.47 to 1.50) | 259 per 1000b | 218 per 1000 | 41 fewer per 1000 (137 fewer to 130 more) | 80 (1 RCT) |
Low Due to within‐study bias and imprecision |
0.85 | Milrinone low dose may decrease the incidence of adverse events, compared to placebo |
| Dobutamine | 1.24 (0.75 to 2.03) | 259 per 1000b | 321 per 1000 | 62 more per 1000 (65 fewer to 267 more) | 26 (1 RCT) |
Moderate Due to imprecision |
0.36 | Dobutamine likely increases the incidence of adverse events, compared to placebo |
| Placebo | 1 | Not estimable | Not estimable | Not estimable | 224 (4 RCTs) |
Reference comparator | 0.71 | ‐ |
| Heterogeneity: tau2 = 0; I2 = 0% (95% CI 0.0% to 74.6%) Q total 1.28 (df = 5; P = 0.9372) Q within designs 1.03 (df = 3; P = 0.7938) Q between designs 0.25 (df = 2; P = 0.8836) | ||||||||
*SUCRA: surface under the cumulative ranking curve, calculated with R package netmeta
GRADE Working Group grades of evidence (or certainty in the evidence)
High certainty: we are very confident that the true effect lies close to that of the estimate of the effect.
Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different.
Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect.
Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect.
Explanatory footnotes
aAn incidence rate ratio rather than a risk ratio was calculated, because there could be a higher number of cumulative adverse events than the number of patients in a treatment arm. bBaseline risk (assumed control risk) obtained from Hoffman 2003, placebo group.
Certainty of the evidence for levosimendan and for dobutamine was downgraded one step due to imprecision, resulting in 'Moderate'.
Certainty of the evidence for milrinone and for milrinone low‐dose was downgraded two steps due to within‐study bias and imprecision, resulting in 'Low'.
Abbreviation(s)
CI: confidence interval; df: degrees of freedom; ECMO: extracorporeal membrane oxygenation; LVEF: left ventricular ejection fraction; LVFS: left ventricular fractional shortening; No.: number; RCT: randomised controlled trial; SUCRA: surface under the cumulative ranking curve; VAD: ventricular assist device.
7.
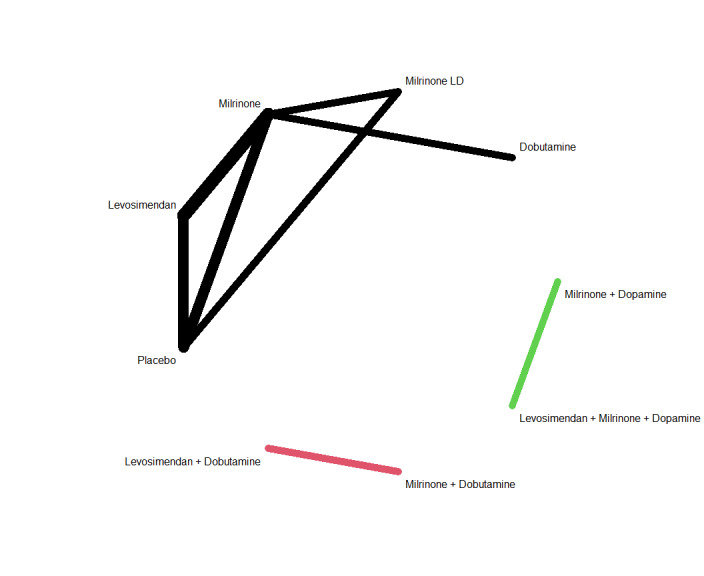
Networks for outcome 'All adverse events'. Colours distinguish the separate networks. There are three networks, one with levosimendan/placebo/milrinone/milrinone ld/dobutamine, one with milrinone + dobutamine versus levosimendan + dobutamine, and one with levosimendan + milrinone + dopamine versus milrinone + dopamine.
Abbreviation(s) LD: low‐dose
Background
Glossary in Appendix 1.
Description of the condition
Cardiac surgery for congenital heart disease (CHD) is often performed at a young patient age (Warnes 2001). Depending on the type of surgical intervention and patient age, postoperative low cardiac output syndrome (LCOS), also termed postoperative myocardial dysfunction, postoperative cardiocirculatory dysfunction, postsurgery heart failure, or postcardiotomy shock, is a frequent postoperative complication in children with CHD. Approximately 10% to 25% of children may be affected postoperatively (Du 2020; Wernovsky 1995). There are predisposing factors that increase the risk of postoperative LCOS, such as younger age, cardiac reoperation, longer duration of cardiopulmonary bypass and aortic cross clamping (causing initial ischaemia followed by reperfusion injury of the heart and systemic inflammation). In children with congenital heart defects and severe pulmonary hypertension, the risk of postoperative residual shunt seems to be higher, which, in turn, is a risk factor for LCOS (Wang 2023), among other risk factors. Different factors are thought to contribute to the pathophysiology of LCOS: a) blood exposure to foreign surfaces of the cardiopulmonary bypass (CPB), which leads to systemic inflammation and capillary leak, oedema, myocardial systolic and diastolic dysfunction; b) myocardial reperfusion injury after cardioplegia and cardiac arrest; c) pulmonary reperfusion injury with impaired oxygen supply; d) severe increase of systemic vascular resistance after CPB, which cannot be overcome by the weakened myocardium (Bailey 2004).
LCOS consists of inadequate oxygen delivery to the organs due to cardiocirculatory insufficiency (Stocker 2006). This, in turn, leads to multiorgan dysfunction; for example, renal insufficiency, intestinal ischaemia, or cerebral ischaemia. LCOS results in increased postoperative morbidity and mortality (Baysal 2010), a higher risk of cardiopulmonary resuscitation or extracorporeal life support (Delmo 2010), and prolonged mechanical ventilation and intensive care stay (Shi 2008). Prevention or early detection and treatment of LCOS, therefore, is important in order to improve patient outcomes. The diagnosis of LCOS is, however, not based on a uniform definition. Invasive measurement of cardiac output is preferred in adults but is often not possible in children (Teng 2011). Otherwise, a combination of signs of inadequate peripheral oxygen delivery may be used to detect LCOS; for example, tachycardia, low systolic blood pressure/high inotrope requirement (Shore 2001), renal dysfunction, or poor peripheral perfusion with an increased temperature difference between peripheral skin and body core, or need for certain treatments such as higher‐than‐intended inotropes, re‐sternotomy, or mechanical circulatory support (Jacobs 2008). An example of a composite LCOS score was developed by Ulate 2017 (combining symptoms of tachycardia, oliguria, decreased toe temperature, need for excess volume administration, decreased near‐infrared spectroscopy measurements, elevated arterial lactate, and need for inotropes in excess of milrinone at 0.5 μg/kg/min) and was associated with higher morbidity and durations of intensive care and hospital stay.
Description of the intervention
Pharmacological LCOS treatment consists mainly of vasoactive drugs and of drugs intended to increase cardiac output by acting on the myocardium. Examples include dobutamine, dopamine, epinephrine, norepinephrine (catecholamines), milrinone (phosphodiesterase III inhibitor), or levosimendan (calcium sensitiser).
Inotropes act by increasing myocardial contractility, vasodilators relax vascular smooth muscle cells leading to dilation of blood vessels and a reduction of vascular resistance, and vasopressors increase afterload and blood pressure by constricting blood vessels through contraction of vascular smooth muscle cells. Inodilators or inovasopressors provide a combination of inotrope effects and either dilation or constriction of blood vessels, respectively (Jentzer 2015).
Drugs from other classes, such as steroids (Cheema 2023), tri‐iodothyronine, nitric oxide, or sildenafil have also been studied as possible treatments for LCOS, but will not be part of this review; nor will pure vasodilators, such as nitroprusside or nitroglycerin, or pure vasopressors, such as arginine‐vasopressin (Loomba 2021).
Drugs used for treatment of LCOS may also be used prophylactically in order to prevent LCOS after cardiac surgery (Roeleveld 2018). Intravenous medications are primarily given by continuous infusion (or, with certain drugs, as a loading dose followed by continuous infusion). Drug administration is begun before, during, or immediately after separation from cardiopulmonary bypass, or within several hours, and can be continued for several days.
How the intervention might work
Therapeutic strategies to optimise cardiac output and minimise oxygen demand are not restricted to drugs acting on the heart itself or to drugs as such. It is important to optimise mechanical ventilation and oxygen supply (Liu 2023), volume load for the right and the left ventricles (Oh 2019), and body temperature, and, equally important, to prevent dysrhythmia (Zhang 1991). In addition to all these measures, pharmacological treatment may be needed to increase myocardial contractility (inotropic medications), to decrease afterload (vasodilators without inotropic effect), or a combination of these mechanisms (inodilators).
Catecholamines such as dobutamine, dopamine, epinephrine, and norepinephrine act on dopamine receptors or on alpha or beta (or both) adrenoceptors. These are located in the myocardium and in blood vessels. Some of the substances are synthesised by the body physiologically; for example, as stress hormones. The combination of its alpha and beta activity, which may vary based on drug dosing, determines the net effect of a given substance (Jentzer 2015). In the myocardium, catecholamines have positive inotropic, chronotropic, and dromotropic effects and increase myocardial oxygen consumption. In the vasculature, they can have vasopressor or vasodilatory effects (Prichard 1991).
Phosphodiesterase type III inhibitors, for example, amrinone, milrinone, olprinone, or enoximone (inodilators), reduce the degradation of cyclic adenosine monophosphate by the enzyme phosphodiesterase type III, thereby increasing phosphorylation of protein kinases that activate cardiac calcium channels, which has positive inotropic and lusitropic effects on the myocardium. In the vasculature, they act as dilators. In children undergoing congenital heart surgery, the prophylactic use of milrinone has so far not been sufficiently proven to prevent mortality or LCOS in clinical studies (Burkhardt 2015).
Calcium sensitisers (inodilatory effect), such as levosimendan, bind to troponin C in the myocardium and increase its responsiveness to calcium, which increases inotropy. They also open adenosine‐triphosphate‐sensitive potassium channels in the smooth muscle cells of systemic, pulmonary, and coronary vessels, which leads to vasodilation (Turanlahti 2004). Levosimendan is a long‐acting drug, as its active metabolite OR‐1896 has a half‐life of approximately 80 hours. Due to low‐certainty evidence in clinical studies, it is currently not clear whether levosimendan prevents mortality or LCOS in paediatric patients undergoing congenital heart surgery (Hummel 2017).
Why it is important to do this review
So far, there are no national or international guidelines on the safe and effective use of drugs for the prevention of LCOS in children undergoing congenital heart surgery. We have previously reviewed the use of milrinone and levosimendan for prevention of LCOS in this population, but did not find enough evidence for each drug separately among existing clinical trials (Burkhardt 2015; Hummel 2017). This is due to a small number of available studies and small numbers of paediatric patients, even in multicentre trials. New studies have been conducted or reported in the meantime, sometimes using both of these drugs in separate treatment arms. To synthesise all existing evidence on these two drugs and the potential of inotropic medications in general, a network meta‐analysis seemed appropriate. This review is an essential step to provide further information on how to prevent LCOS and mortality in paediatric patients undergoing surgery for congenital heart disease.
Objectives
To evaluate the relative benefits and harms of inotropes for the prevention of LCOS and mortality in paediatric patients undergoing surgery for CHD.
To generate a clinically useful ranking of prophylactic inotropes for the prevention of low cardiac output syndrome and mortality in paediatric patients undergoing surgery for congenital heart disease according to benefits and harms.
Methods
Criteria for considering studies for this review
Types of studies
We included parallel‐arm randomised controlled trials (RCTs). We did not include cluster‐randomised trials, because studies in this area focus on the individual patient level, nor trials with a cross‐over design, which would not be suitable for the prophylactic approach. We also did not consider quasi‐randomised trials, because randomisation is feasible in this scenario and reduces the influence of confounding variables.
Types of participants
We included paediatric patients from birth to 18 years of age who have undergone corrective or palliative heart surgery for congenital heart disease. We used pooled data for analysis. This benefited the transitivity needed for network meta‐analysis, where all interventions were considered legitimate alternatives and therefore jointly randomisable. There were no specific exclusion criteria for participants.
Types of interventions
We included studies comparing any pharmacological intervention(s) belonging to one of the following drug classes versus each other or versus standard of care treatment or placebo.
Catecholamines
Phosphodiesterase type III inhibitors
Calcium sensitisers
If combinations of drugs from different classes were used prophylactically, each combination represented a separate intervention of interest and was displayed as a separate node in the network. We examined the geometry of the network meta‐analysis (NMA) for all interventions (mono‐ and combination prophylaxis) versus mono‐prophylaxis alone. In a case when we could not analyse all combinations, we split the network and examined the single networks separately. Combination treatments can be split into their components, and the effects of the components can be analysed separately under certain assumptions, even for disconnected networks (Rücker 2019; Rücker 2021a). We did not plan to consider unspecified interventions for post hoc inclusion in the network within the context of jointly randomisable interventions. We expected co‐interventions, as explained above (How the intervention might work), but unless otherwise reported, we assumed that all other measures besides inotropic medications were exhausted for patients in all study arms of the included trials.
We defined prophylactic administration as starting during surgery for congenital heart disease or up to four hours after disconnecting from cardiopulmonary bypass, and it should have been continued for at least four hours. The lower dosing limits for a medication to count as an intervention of interest should be 0.2 μg/kg/min for milrinone, 0.05 μg/kg/min for levosimendan, 0.01 μg/kg/min for epinephrine and norepinephrine, and 5 μg/kg/min for dopamine and dobutamine. In the case of different dosages of the same drug, where one study arm used an unusually low or high dose of the drug, the intervention was used to constitute a separate node of the network, and the reasoning was explained.
Types of outcome measures
Reporting one or more of the outcomes listed here in the trial was not an inclusion criterion for the review. Where a published report did not appear to report one of these outcomes, we accessed the trial protocol and contacted the trial authors to ascertain whether the outcomes were measured but not reported. We included in the review, as part of the narrative, relevant trials that measured these outcomes but did not report the data at all, or not in a usable format.
Primary outcomes
We calculated the relative effects of the competing interventions using the following primary outcomes.
All‐cause mortality within 30 days.
Time to death (censored after three months).
-
Low cardiac output syndrome (at any time) defined as two or more of the following:
blood lactate > 3 mmol/L (27 mg/dL) or increase in blood lactate of at least 2 mmol/L (18 mg/dL) from baseline (Charpie 2000);
central venous oxygen saturation < 50% in biventricular physiology without shunts (Stocker 2006);
increase in arterial to central venous oxygen saturation difference by at least 20% from baseline prior to administration of the intervention of interest;
urine output < 0.5 mL/kg/h;
peripheral skin temperature to core body temperature difference of > 7 °C; and
cardiac index as determined by Doppler echocardiography of < 2.2 L/min/m² (Rao 1996).
Secondary outcomes
Length of intensive care stay (days).
Length of hospital stay (days).
Duration of mechanical ventilation (hours).
Inotrope score (as defined in Wernovsky 1995, if available).
Number of patients requiring mechanical circulatory support (e.g. extracorporeal membrane oxygenation (ECMO), pulsatile assist devices).
Number of patients requiring cardiac transplantation.
Number/proportion of patients experiencing adverse effects (adverse effects include: arrhythmia; hypotension defined as blood pressures below blood pressure appropriate for age or body surface area; headache; intraventricular haemorrhage; hypocalcaemia; hypokalaemia, bronchospasm; thrombocytopaenia defined as a platelet count < 50/nl or drop in platelet count of > 100% from baseline prior to administration of the intervention of interest; anaemia defined as a haemoglobin value below the age‐appropriate normal value; elevated serum levels of liver enzymes defined as serum enzymatic activities more than two‐fold the age‐appropriate normal values; left ventricular ejection fraction < 50% or left ventricular fraction of shortening < 28% as assessed by biplane or M‐mode echocardiography; and renal failure requiring replacement therapy).
Unless otherwise stated, we assessed the outcomes at the longest available follow‐up, which was mostly until hospital discharge or within one month after surgery.
Regarding the number/proportion of adverse effects, we analysed these separately as the individual components mentioned above. If there was insufficient information available, we reported the various adverse effects narratively.
The adverse effect outcome 'renal failure' was assessed in multiple ways in some studies (Abril‐Molina 2021; Costello 2014; Thorlacius 2020; Wang 2019), so we decided to compare 'renal failure requiring replacement therapy' across studies, which was also reported in other studies with fewer renal outcomes (Cavigelli‐Brunner 2018; Momeni 2011; Ricci 2012; Shah 2013).
Search methods for identification of studies
Electronic searches
We identified trials through systematic searches of the following bibliographic databases.
Cochrane Central Register of Controlled Trials (CENTRAL; 2023, Issue 12; 2 December 2023) in the Cochrane Library
MEDLINE (Ovid, from 1946 to 20 May 2021, last update to 19 April 2024)
Embase (Ovid, from 1980 to 20 May 2021 and Embase.com from 01 May 2021 to 25 April 2024)
Conference Proceedings Citation Index ‐ Science (CPCI‐S) on the Web of Science (1990 to 20 May 2021, last update to 19 April 2024)
We adapted the preliminary search strategy for MEDLINE (Ovid) for use in the other databases (Appendix 2). We applied the Cochrane sensitivity‐maximising RCT filter to MEDLINE (Ovid) and adaptations of it to the other databases, except CENTRAL, using terms for Embase as recommended in the Cochrane Handbook for Systematic Reviews of Interventions (Lefebvre 2019).
We also conducted searches of www.ClinicalTrials.gov and the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) search portal (apps.who.int/trialsearch) for ongoing or unpublished trials, last in April 2024.
We also searched regulatory data from the EMA (www.ema.europa.eu/ema), FDA (www.fda.gov/Drugs/InformationOnDrugs), and UK Medicines and Healthcare Products Regulatory Agency (https://products.mhra.gov.uk).
We searched all databases from their inception to April 2024 (CENTRAL from inception to 2 December 2023), and we imposed no restriction on language of publication or publication status.
We did not perform a separate search for adverse effects of interventions used for the prevention of LCOS and mortality in paediatric patients undergoing surgery for congenital heart disease. We considered adverse effects described in included studies as well as unpublished data provided by the study authors.
Searching other resources
We checked reference lists of all included studies and any relevant systematic reviews identified for additional references to trials. We also searched any relevant retraction statements and errata for included studies.
Data collection and analysis
Selection of studies
Two review authors (BB, JH) independently screened the titles and abstracts of all the potential studies we identified as a result of the search and coded them as 'retrieve' (eligible or potentially eligible/unclear) or 'do not retrieve'. We retrieved the full‐text study reports/publications, and two review authors (BB, JH) independently screened the full text and identified studies for inclusion, and identified and recorded reasons for exclusion of the ineligible studies. We resolved any disagreement through discussion. We identified and excluded duplicates and collated multiple reports of the same study so that each study rather than each report was the unit of interest in the review. We recorded the selection process in sufficient detail to complete a PRISMA flow diagram (Liberati 2009) and a 'Characteristics of excluded studies' table.
Data extraction and management
We used a data collection form, which we had piloted on one study in the review, for study characteristics and outcome data. Two review authors (BB, JH) independently extracted the information below from the included studies.
Methods: study design, total duration of study, number of study centres and location, study setting, and date of study.
Participants: number of participants randomised, number of participants lost to follow‐up/withdrawn, number of participants analysed, mean age, age range, age subgroups (if available), gender, type and severity of CHD (including univentricular versus biventricular CHD), diagnostic criteria, inclusion criteria, and exclusion criteria.
Interventions: intervention, comparison, concomitant medications, and excluded medications.
Outcome data: primary and secondary outcomes specified and collected, and time points reported. If additional data were needed to derive hazard ratios, we contacted the individual study authors and asked them to provide additional data.
Data on potential effect modifiers: 1) age: subgroups 0 to 1 month, 1 to 11 months, and children ≥ 12 months, 2) physiology: subgroups univentricular, biventricular.
Other data: funding for trial, and notable conflicts of interest of trial authors.
One review author (BB) transferred data into RevMan (RevMan 2024). We double‐checked that data were entered correctly by comparing the data presented in the systematic review with the data extraction form. A second review author (JH) spot‐checked study characteristics for accuracy against the trial report.
Assessment of risk of bias in included studies
Two review authors (BB, JH) independently assessed the risk of bias for each study using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions using the risk of bias tool (RoB 1) (Higgins 2017). We resolved any disagreements by discussion. We assessed the risk of bias according to the following domains (Higgins 2017).
Random sequence generation.
Allocation concealment.
Blinding of participants and personnel.
Blinding of outcome assessment.
Incomplete outcome data.
Selective outcome reporting.
Other bias.
We graded each potential source of bias as high, low, or unclear and provided a quote from the study report and/or another justification for our judgement in the risk of bias tables. We summarised the risk of bias judgements across different studies for each of the domains listed. Where information on risk of bias related to unpublished data or correspondence with a trialist, we noted this in the risk of bias table.
When considering treatment effects, we took into account the risk of bias for the studies that contributed to that outcome. For an overall risk of bias rating, we categorised studies with low risk of bias in all domains as raising 'no concerns', we categorised studies with possible risk of bias in any domain but no high risk of bias ratings as raising 'some concerns', and we categorised studies with high risk of bias in any domain as raising 'major concerns' regarding overall risk of bias. In the graphical network displays, the overall risk of bias of each contributing study is displayed with colour coding 'green' = no concerns, 'yellow' = some concerns, and 'red' = major concerns.
Relative treatment effects
We assessed dichotomous data with risk ratio (RR) as an effect measure (Higgins 2023). Based on the data available, we regarded the time variables (length of intensive care stay, length of hospital stay, duration of mechanical ventilation) as continuous outcomes. These can assume a wide range of values, so we used the ratio of means (ROM) with 95% confidence intervals (CI) to describe relative effects. We assessed adverse events as a group as count data (incidences) with the incidence rate ratio (IRR), because there was a possibility of one participant experiencing more than one type of adverse event, such that the number of events could be greater than the number of participants. When explored separately, we regarded adverse events as dichotomous outcomes, where patients were considered to experience a single event.
We calculated means and standard deviations (SD) from medians and interquartile ranges (IQR) where possible. We used the methods of Luo 2018 to approximate means and standard deviations from medians and interquartile ranges. More precisely, as implemented in R package meta (Balduzzi 2019), we used Luo’s equation no. 15 if sample size, median, interquartile range, and range were available, equation no. 11 if sample size, median, and interquartile range were available, and equation no. 7 if sample size, median, and range were available (Luo 2018).
Relative treatment ranking
For rankings, we calculated SUCRA (surface under the cumulative ranking curve; value closer to 1 = more desirable intervention, value closer to 0 = less desirable intervention, mean value = 0.5) using R package netmeta, for the outcomes 1) LCOS, 2) all‐cause mortality within 30 days, 3) length of intensive care stay, 4) length of hospital stay, 5) duration of mechanical ventilation, and 6) adverse events (cumulative incidence). We also estimated ranking probabilities using the R package nmarank (Nikolakopoulou 2023; Salanti 2011).
Unit of analysis issues
We included RCTs with two or more parallel treatment arms. For pairwise comparisons, in case of three or more treatment arms, we included these in the data set as a series of two‐arm comparisons, and adjusted the standard error of each of these comparisons for correlation between the arms. For this, we used back‐calculated standard errors in the weighted least‐square estimator to reflect the within‐study correlation (Rücker 2012; Rücker 2014).
Dealing with missing data
We contacted investigators or study sponsors in order to verify key study characteristics and obtain missing numerical outcome data where possible (e.g. when a study was identified as abstract only). Where possible, we calculated missing standard deviations using other data from the trial, such as confidence intervals, based on methods outlined in the Cochrane Handbook (Higgins 2019) and in Luo 2018. We performed analyses for efficacy outcomes, as far as possible, on an intention‐to‐treat (ITT) basis. In the case of missing data, we excluded all participants from the analysis for whom no outcome was available. Studies with no events for a certain outcome in any of the study arms were excluded from the analysis.
Pairwise comparisons
We described clinical and methodological heterogeneity regarding study and population baseline characteristics narratively. We assessed statistical heterogeneity using both Cochran’s Q test and the I² statistic. We regarded an I² value between 0% and 50% as a sign of low or moderate heterogeneity, and an I² value above 50% as representing substantial heterogeneity.
Network meta‐analysis
We described global inconsistency by splitting total Q into contributions from within‐design heterogeneity and between‐design inconsistency. We also investigated inconsistency locally based on the 'Separate Indirect from Direct Evidence' (SIDE) method (Dias 2010; König 2013; Higgins 2012; Jackson 2014).
Assessment of reporting biases
We had planned to create and examine a funnel plot to explore possible small‐study biases for the primary outcomes or missing/unpublished studies leading to reporting bias (Higgins 2011), and to examine funnel plot asymmetry according to Chaimani 2013 with a comparison‐adjusted funnel plot applied to the network meta‐analysis, but there were too few studies.
Data synthesis
We undertook meta‐analyses only where this was meaningful ‒ that is, if the treatments, participants, and the underlying clinical question were similar enough for pooling to make sense.
Pairwise comparisons
We expected methodological and clinical heterogeneity across the included studies and therefore performed standard pairwise meta‐analyses using a random‐effects model (inverse variance weighting for continuous outcomes and Mantel‐Haenszel method for dichotomous outcomes) in RevMan, versions 2.7.0 to 7.2.0 (RevMan 2024) as well as for cumulative adverse event count data in R (R core team 2023). Treatment comparisons with only one study are reported as well. As effect measures, we used the mean difference for continuous outcomes, the risk ratio (RR) for dichotomous outcomes, and the incidence rate ratio (IRR) for the number of adverse events.
Network meta‐analysis
We also performed random‐effects NMAs for the primary outcomes 'LCOS' and 'mortality', based on a frequentist framework in R, as described by Rücker 2014. For analysis of drug combinations, we used these as different nodes in the network, like single‐drug interventions. For the outcomes all‐cause mortality within 30 days, LCOS, length of ICU stay, duration of mechanical ventilation, and adverse events (cumulative), we separately analysed the data using component network meta‐analysis (CNMA) (Mills 2012; Rücker 2019; Welton 2009). The additive CNMA model assumes that the effects of combination interventions are the sum of their components and thus allows the disentanglement of the effects of single components. We assumed additive effects of inotrope medications used in combination. More general interaction models can also be applied, and the models can be compared using likelihood ratio tests. This was not performed here, given the small number of studies.
For each outcome, we first checked whether the network was connected. We then conducted network meta‐analysis (NMA) for each subnetwork and then, if the network was disconnected, we tried to connect it by using CNMA. This did not work in all analyses, as sometimes some components turned out to be not estimable due to the structure of the network. We report CNMA analyses where they seemed appropriate.
We analysed ranking of treatments (Rücker 2015) using SUCRA (surface under the cumulative ranking curve) (Salanti 2011), as calculated with the R package netmeta (Rücker 2021b), but without confidence intervals for the ranking scores (Veroniki 2018), for LCOS (all time points reported), all‐cause mortality within 30 days, length of ICU stay, length of hospital stay, duration of mechanical ventilation, and adverse events (cumulative) at their respective time points of assessment. We also estimated ranking probabilities using the R package nmarank (Nikolakopoulou 2023). Zero studies (studies with no events in either treatment arm for a given outcome) were excluded from the NMA.
Subgroup analysis and investigation of heterogeneity
Subgroup analysis was not possible due to insufficient information about the outcomes of subgroups in the included studies.
Sensitivity analysis
The available studies had different levels of risk of bias. We performed sensitivity analyses by re‐running the (component) meta‐analysis after excluding studies with a high risk of bias (in any of the four domains 'random sequence', 'allocation concealment', 'incomplete outcome data', and 'selective reporting'). Where we found it impossible to calculate missing numerical data, and we thought the missing data introduced serious bias, we explored the impact of excluding such studies in the overall assessment of results by a sensitivity analysis. We also assessed the effect of including any studies with an atypical population in sensitivity analysis. We restricted sensitivity analyses to the primary outcomes and to the NMA networks in cases of more than one subnetwork per outcome.
Summary of findings and assessment of the certainty of the evidence
We created summary of findings tables in RevMan Web using the following outcomes: all‐cause mortality within 30 days; low cardiac output syndrome defined as described above (during the intensive care stay); length of intensive care stay (at longest follow‐up); length of hospital stay (at longest follow‐up); duration of mechanical ventilation (at longest follow‐up); and adverse events (mostly timed until intensive care or hospital discharge).
We assessed the certainty of evidence using CINeMA (Confidence in Network Meta‐Analysis) (Nikolakopoulou 2020). Based on the GRADE framework, this assesses the evidence for the presence of the following six factors: (1) within‐study bias (referring to the impact of risk of bias in the included studies), (2) reporting bias (referring to publication and other reporting bias), (3) indirectness, (4) imprecision, (5) heterogeneity, and (6) incoherence. Depending on the presence of these factors, we downgraded the certainty of the evidence from high to moderate or low (Schünemann 2013). We created summary of findings tables for all primary outcomes and their time points mentioned above, modified from Yepes‐Nuñez 2019. We compared substance classes as above (catecholamines, phosphodiesterase type III inhibitors, calcium sensitisers) to each other and to placebo. We made comments to aid readers' understanding of the review where necessary.
Two review authors (BB, JH), working independently, made judgements about evidence certainty and resolved disagreements by discussion or by involving a third author (BS). We justified, documented, and incorporated judgements into the reporting of results for each outcome.
We extracted study data, formatted our comparisons in data tables, and prepared summary of findings tables before writing the results and conclusions of our review. Reporting followed the PRISMA checklist for network meta‐analysis (Hutton 2016).
Results
Description of studies
Results of the search
Electronic database searches yielded a total of 1256 records by May 2021. After removal of 259 duplicates, there were 997 records remaining. Non‐electronic searches yielded five additional records. A top‐up search in December 2023 across the CENTRAL database yielded another 62 records, 58 after de‐duplication, and a top‐up search across the other databases in April 2024 yielded another 969 records, 734 after de‐duplication. Overall, the searches identified 2292 records (2287 from searching electronic sources and 5 additional records from searching other resources). Following the removal of 498 duplicates, we screened 1794 records by title and abstract and excluded 1751 records as irrelevant. We retrieved 43 full‐text reports; 10 of these consisted of additional reports of the studies retrieved as full text (for example, one registry entry and one publication or one main trial report and one secondary analysis of the same trial). From these we identified two ongoing studies (Characteristics of ongoing studies), and three studies awaiting classification (Studies awaiting classification). Of the 28 remaining reports, we excluded 15 studies following full‐text review (Characteristics of excluded studies) and included 13 eligible studies in the qualitative and quantitative syntheses (Included studies). See Figure 8.
8.
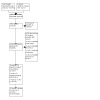
Study flow diagram
Included studies
We included 13 randomised, controlled, parallel‐arm studies (Characteristics of included studies), which fulfilled our inclusion criteria, with a total of 937 participants. Most studies were small, with population sizes between 9 or 11 patients per treatment arm (Pellicer 2013) or 94 or 93 patients per treatment arm (Wang 2019). There were two multi‐centre trials (Hoffman 2003; Thorlacius 2020), with the remainder of the studies conducted at one institution, located in Europe, China, or North America. Study durations varied between less than one year and 5.3 years. Funding sources included governmental bodies, hospital departments, and private donations/non‐profit organisations, but also drug manufacturers or were not stated.
The study population consisted of children of various ages: newborns less than one month of age (Pellicer 2013; Ricci 2012), newborns and infants less than one year of age (Jothinath 2021; Lechner 2012), infants between one month and one year of age (Thorlacius 2020), children more than one month of age (Abril‐Molina 2021; Costello 2014; Ebade 2013), and children aged from birth to more than one year and up to 14.2 years of age (Cavigelli‐Brunner 2018; Momeni 2011; Shah 2013; Wang 2019). Most studies included only children undergoing biventricular corrective heart surgery, except for one study including only patients with univentricular physiology (Costello 2014) and three studies including children with both uni‐ and biventricular hearts (Cavigelli‐Brunner 2018; Shah 2013; Wang 2019). None of the studies compared subgroups or contained sufficient information for further analysis of subgroups based on patient age or univentricular versus biventricular physiology.
There were five studies comparing levosimendan versus milrinone (Jothinath 2021; Lechner 2012; Momeni 2011; Pellicer 2013; Thorlacius 2020), two studies comparing levosimendan versus placebo (Abril‐Molina 2021; Wang 2019), and two studies comparing milrinone versus placebo (Costello 2014; Hoffman 2003), one of which included two milrinone treatment arms using different dosages (Hoffman 2003) and one including a third treatment arm with a non‐inotrope study drug (Costello 2014). One study compared levosimendan versus dobutamine (Ebade 2013), one study compared milrinone versus dobutamine (Cavigelli‐Brunner 2018), and two studies used combinations of inotropes: milrinone and dopamine with or without additional levosimendan (Ricci 2012), or levosimendan and dobutamine versus milrinone and dobutamine (Shah 2013).
The dosages of the study drugs were somewhat different across studies. Milrinone doses ranged from 0.4 μg/kg/min (Thorlacius 2020) to 0.75 μg/kg/min (Cavigelli‐Brunner 2018; Ricci 2012) or 0.5 μg/kg/min to 1 μg/kg/min (Costello 2014; Pellicer 2013), while Hoffman et al compared "high‐dose" milrinone at 0.75 μg/kg/min (which we regarded as standard dosing for the purpose of inter‐study comparability) versus "low‐dose" milrinone at 0.25 μg/kg/min (which was not used in a dose that low in any other study) versus placebo (Hoffman 2003). Levosimendan was given at doses between 0.05 μg/kg/min (Wang 2019) and 0.2 μg/kg/min (Abril‐Molina 2021). For dobutamine, a dose of 5 to 8 μg/kg/min (Shah 2013) or 6 μg/kg/min (Cavigelli‐Brunner 2018) was chosen, and for dopamine, 5 to 10 μg/kg/min (Ricci 2012).
Concomitant medications included other inotropes as needed on a clinical case‐to‐case basis in most studies. Some specified additional use of epinephrine or norepinephrine, or both (Cavigelli‐Brunner 2018; Jothinath 2021; Lechner 2012; Ricci 2012; Shah 2013), epinephrine, dopamine, and dobutamine (Pellicer 2013), or catecholamines in general (Hoffman 2003).
We included studies that reported at least one of our pre‐specified primary outcomes. We obtained additional information about our pre‐specified secondary outcomes from the study authors, where possible.
Excluded studies
We excluded 15 studies at full‐text stage as irrelevant. Four did not fulfil the inclusion criteria based on the intervention (e.g. Abdelbaser 2021), one assessed an ineligible comparator intervention (Abd Elraouf 2019), four assessed only outcomes that were not of interest to our review question (e.g. Innes 1994), and six were not randomised controlled trials (e.g. Wang 2018). See Characteristics of excluded studies.
Studies awaiting classification
After the search update in December 2023, we identified three more studies as potentially eligible and awaiting classification. One of these compared the interventions milrinone versus epinephrine (Abdallah 2003), the other two levosimendan versus milrinone (Gutiérrez‐Riveroll 2022; Nag 2023). See Characteristics of studies awaiting classification.
Ongoing studies
We found two ongoing studies, one comparing milrinone versus placebo in infants, with a co‐intervention of baseline catecholamines, with primary outcomes of 30‐day mortality and LCOS within 36 hours of surgery (NCT03823781), and the other (which has not started recruitment yet) comparing milrinone versus levosimendan in children up to 12 years of age after corrective surgery for Tetralogy of Fallot, with primary endpoints of LCOS and arrhythmias (CTRI/2023/07/055845). See Characteristics of ongoing studies.
Risk of bias in included studies
Out of the 13 included studies, we judged five to have an overall high risk of bias (defined as a high risk of bias in any domain: Ebade 2013, Hoffman 2003, Jothinath 2021, Ricci 2012, and Shah 2013). One study had an overall unclear risk of bias (defined as possible risk of bias in any domain but no high risk of bias ratings: Cavigelli‐Brunner 2018), and we considered seven studies to have an overall low risk of bias (defined as low risk of bias in all domains) (Figure 9; Figure 10).
9.

Risk of bias graph summarising bias risks across all included studies
10.

Risk of bias summary detailing the different risks in each included study
Allocation
Random sequence generation
We rated four studies at unclear risk of bias. One study did not describe allocation to the study arms (Shah 2013). In three other studies, the allocation and random sequence generation process or provider was not comprehensively described (Cavigelli‐Brunner 2018; Ebade 2013; Hoffman 2003). All other studies used computer‐generated random numbers based on general or stratified randomisation algorithms, and we rated them at low risk of bias.
Allocation concealment
We rated two studies at high risk of bias, because no allocation concealment measures are mentioned or because no allocation process was described at all and therefore concealment may have been a problem (Jothinath 2021; Shah 2013), and another study as at unclear risk of bias, because the allocation concealment process is not described, but was probably provided because of the multicentre structure administrated by an external clinical research organisation (Hoffman 2003). We rated all other studies at low risk of bias.
Blinding
Performance bias
In four studies, no blinding process of personnel was described (Ebade 2013; Jothinath 2021; Ricci 2012; Shah 2013). While one study was expressly "open‐label design" (Ricci 2012), in the other cases levosimendan was one of the study drugs, which is distinguishable by its yellow colour from other infusions. We rated these four studies at high risk of performance bias. All other studies used adequate mechanisms for blinding of participants and bedside personnel, so we rated these at low risk of performance bias.
Detection bias
We rated two studies at high risk of detection bias, because one study was expressly "open label" design (Ricci 2012) and another involved outcome assessments at the bedside by a cardiologist who would have easily seen which medications the patient was receiving at the time (Ebade 2013). Two other studies did not mention any blinding of the outcome assessors, and we therefore rated them at unknown risk of bias (Jothinath 2021; Shah 2013). We rated all other studies at low risk of bias.
Incomplete outcome data
We considered one study to have a high risk of attrition bias (Hoffman 2003), with several dropouts during the study, but unclear reporting about which study arm they had belonged to. Due to the long time elapsed since the conduct of the study, no unpublished data to clarify this could be retrieved from the company who was involved. There was also only partial information available about absolute numbers or percentages of outcomes in this study, and additional information could not be retrieved. The other 12 studies were deemed to have a low risk of attrition bias, because they reported to have either no dropouts or explained details about them.
Selective reporting
It is possible that there are studies that have not been published because they did not find significant improvements in the incidence of LCOS or mortality with prophylactic medication. Some trials may not have been reported because they were underpowered to find any differences in the setting of small study populations.
We assessed 12 studies as having low risk of reporting bias, because they reported all their specified outcomes. In one study, Hoffman 2003, information about mortality was very limited, so we rated it at high risk of bias.
Other potential sources of bias
We rated two studies at high risk of bias. One study, Hoffman 2003, was declared to be supported by the manufacturer of the study drug, whereas the other, Jothinath 2021, did not describe the method of participant selection. Another study, Shah 2013, did not report funding and possible conflicts of interest, so we rated it at unclear risk of bias. We rated the remaining studies at low risk of other bias.
Effects of interventions
See: Table 1; Table 2; Table 3; Table 4; Table 5; Table 6; Table 7
We performed numerical analyses for all pre‐specified outcomes for which enough data were available.
Baseline characteristics, such as patient age and severity of the disease, were often only available as means, with or without ranges, but without information about standard deviations (SD) or quantiles. Therefore, clinical heterogeneity could not be assessed statistically in a meaningful way. However, all participants were at least children and no adults participated in any of the included studies, therefore we assume transitivity for our review question dealing with paediatric patients.
There were three outcomes with some measures of mean effects, but with missing SD or quantiles: for 'duration of hospital stay', Hoffman 2003 (three study arms) did not provide SD, leaving seven other studies. For 'inotrope score', Ricci 2012 and Jothinath 2021 were missing SD, leaving six other studies. For 'duration of mechanical ventilation', Hoffman 2003 (three study arms) did not provide SD, leaving 11 studies in three subnetworks (nine comparisons, one comparison, and one comparison). However, since the SD of other studies varied greatly, we saw no grounds for imputation (especially as we already had to impute from quantiles for some of the other studies). Hence, we refrained from sensitivity analysis based on imputation of standard deviations.
Separation of indirect from direct evidence (SIDE) within networks using a back‐calculation method with a random‐effects model did not show any differences in direct versus indirect evidence for any outcome (Table 8; Table 9; Table 10; Table 11; Table 12). This applies to intervention comparisons where both types of evidence were available. Some treatment comparisons (e.g. dobutamine versus placebo) were only possible through indirect evidence.
1. Separating indirect from direct evidence: low cardiac output syndrome.
| Treatment comparison | Proportion of direct evidence | RoR | P value |
| Levosimendan versus milrinone | 0.61 | 0.4739 | 0.2852 |
| Levosimendan versus placebo | 0.73 | 2.1102 | 0.2852 |
| Milrinone versus milrinone LD | 0.82 | 0.3374 | 0.2852 |
| Milrinone versus placebo | 0.66 | 0.4739 | 0.2852 |
| Milrinone LD versus placebo | 0.97 | 0.1432 | 0.2852 |
Separation of indirect from direct evidence using a back‐calculation method, random‐effects model RoR: ratio of ratios (direct/indirect), with P value of test for disagreement
Abbreviations LD: low dose; NA: not applicable; RoR: ratio of ratios
2. Separating indirect from direct evidence: length of intensive care stay.
| Treatment comparison | Proportion of direct evidence | RoR | P value |
| Dobutamine versus levosimendan | 0.63 | 1.1501 | 0.7322 |
| Dobutamine versus milrinone | 0.49 | 0.8695 | 0.7322 |
| Dobutamine versus placebo, only indirect evidence | 0 | NA | NA |
| Levosimendan versus milrinone | 0.76 | 1.2063 | 0.5537 |
| Levosimendan versus placebo | 0.72 | 0.8302 | 0.6611 |
| Milrinone versus placebo | 0.39 | 1.2046 | 0.6611 |
Separation of indirect from direct evidence using a back‐calculation method, random‐effects model RoR: ratio of ratios (direct/indirect), with P value of test for disagreement
Abbreviations NA: not applicable; RoR: ratio of ratios
3. Separating indirect from direct evidence: length of hospital stay.
| Treatment comparison | Proportion of direct evidence | RoR | P value |
| Dobutamine versus levosimendan, only indirect evidence | 0 | NA | NA |
| Dobutamine versus milrinone, only direct evidence | 1 | NA | NA |
| Dobutamine versus placebo, only indirect evidence | 0 | NA | NA |
| Levosimendan versus milrinone | 0.96 | 1.2436 | 0.5981 |
| Levosimendan versus placebo | 0.93 | 0.8041 | 0.5981 |
| Milrinone versus placebo | 0.11 | 1.2436 | 0.5981 |
Separation of indirect from direct evidence using a back‐calculation method, random‐effects model RoR: ratio of ratios (direct/indirect), with P value of test for disagreement
Abbreviations NA: not applicable; RoR: ratio of ratios
4. Separating indirect from direct evidence: duration of mechanical ventilation.
| Treatment comparison | Proportion of direct evidence | RoR | P value |
| Dobutamine versus levosimendan | 0.65 | 2.2276 | 0.2274 |
| Dobutamine versus milrinone | 0.46 | 0.4489 | 0.2274 |
| Dobutamine versus placebo, only indirect evidence | 0 | NA | NA |
| Levosimendan versus milrinone | 0.78 | 1.7139 | 0.2276 |
| Levosimendan versus placebo | 0.68 | 0.8550 | 0.8099 |
| Milrinone versus placebo | 0.43 | 1.1696 | 0.8099 |
Separation of indirect from direct evidence using a back‐calculation method, random‐effects model RoR: ratio of ratios (direct/indirect), with P value of test for disagreement
Abbreviations NA: not applicable; RoR: ratio of ratios
5. Separating indirect from direct evidence: adverse events.
| Treatment comparison | Proportion of direct evidence | RoR | P value |
| Dobutamine versus levosimendan, only indirect evidence | 0 | NA | NA |
| Dobutamine versus milrinone, only direct evidence | 1 | NA | NA |
| Dobutamine versus milrinone LD, only indirect evidence | 0 | NA | NA |
| Dobutamine versus placebo, only indirect evidence | 0 | NA | NA |
| Levosimendan versus milrinone | 0.88 | 1.2788 | 0.6764 |
| Levosimendan versus milrinone LD, only indirect evidence | 0 | NA | NA |
| Levosimendan versus placebo | 0.2 | 0.782 | 0.6764 |
| Milrinone versus milrinone LD | 0.87 | 0.8494 | 0.8505 |
| Milrinone versus placebo | 0.92 | 1.2788 | 0.6764 |
| Milrinone LD versus placebo | 0.83 | 0.8627 | 0.8505 |
Separation of indirect from direct evidence using a back‐calculation method, random‐effects model RoR: ratio of ratios (direct/indirect), with P value of test for disagreement
Abbreviations LD: low dose; NA: not applicable; RoR: ratio of ratios
Comparison‐adjusted funnel plots could not be used because there were not enough studies included per network/outcome.
Results of the quantitative analysis are summarised in the summary of findings tables (Table 1; Table 2; Table 3; Table 4; Table 5; Table 6; Table 7).
Primary outcomes
1 All‐cause mortality within 30 days
The reported time frame for mortality was shorter than 30 days in some studies, but these were still included in the analysis: Momeni 2011 had a follow‐up period until the end of hospital stay, which may have missed a small number of deaths in patients who had received levosimendan or milrinone; Cavigelli‐Brunner 2018 and Thorlacius 2020 reported on 28‐day mortality (deemed comparable to 30‐day mortality, because most postoperative deaths occur early after surgery), and for Shah 2013, the follow‐up duration was unknown.
The studies that contributed to our analysis of mortality risk included different patient populations: neonates with biventricular physiology (Pellicer 2013; Ricci 2012: RACHS 3‐4), young children aged from birth to four years with uni‐ or biventricular physiology RACHS 2‐5 (Wang 2019), or young children with biventricular physiology aged 0.5 to eight years (Abril‐Molina 2021) or from birth to five years (Momeni 2011). One study provided age means and SD with mean age around four years (Shah 2013: levosimendan + dobutamine group = 4.1 ± 1.3 years, milrinone + dobutamine group = 4.0 ± 1.5 years).
There were three subnetworks (the largest one comprising nine studies with 14 events among 557 participants) (see Figure 4 in Appendix 3), one with levosimendan/placebo/milrinone/dobutamine, one with milrinone + dobutamine versus levosimendan + dobutamine, and one with levosimendan + milrinone + dopamine versus milrinone + dopamine. There were several studies with no events in both arms (Cavigelli‐Brunner 2018; Costello 2014; Ebade 2013; Lechner 2012; Thorlacius 2020), which were therefore not used in the primary analyses. This resulted in the exclusion of dobutamine from the NMA.
In the largest subnetwork, by NMA the pooled RR for mortality was lower with prophylactic levosimendan (7 studies, 216 participants) than with placebo (3 studies, 143 participants) (RR 0.57), with a wide 95% CI including 1 (0.15 to 2.13), indicating that levosimendan likely results in a large reduction of mortality, compared to placebo, with moderate certainty of the evidence (the true effect is likely close to the effect estimate, but could still be substantially different). Milrinone (6 studies, 147 participants) likely results in no difference in mortality, compared to placebo: RR 0.97, 95% CI 0.11 to 8.49 (moderate certainty of the evidence). Ranking of treatments revealed a SUCRA of 0.76 for levosimendan, 0.39 for milrinone, and 0.35 for placebo, thus showing that levosimendan is most preferable for prevention of mortality, milrinone is less preferable, and placebo is slightly less preferable than milrinone. Ranking probabilities are detailed in Table 13.
6. Ranking probabilities for intervention effectiveness to prevent all‐cause mortality within 30 days.
| Probabilities | Rank | Levosimendan | Milrinone | Placebo |
| 1 | 0.594 | 0.249 | 0.157 | |
| 2 | 0.353 | 0.286 | 0.361 | |
| 3 | 0.053 | 0.361 | 0.482 |
Ranking probabilities based on 1000 simulations, random‐effects model
Heterogeneity tests revealed tau2 = 0; I2 = 0% (95% CI 0.0% to 89.6%); Cochran's Q total 0.86 (degrees of freedom (df) = 2, P = 0.65), within designs 0.86 (df = 2, P = 0.65), and between designs 0 (df = 0).
For the outcome mortality, no separation of indirect from direct evidence was performed, because the comparisons of levosimendan versus milrinone and levosimendan versus placebo were only available as direct evidence, while milrinone versus placebo was only available as indirect evidence as a result of double‐zero studies.
One small, separate study (Ricci 2012), which added levosimendan to a regime of milrinone plus dopamine, found that the addition of levosimendan may result in a large reduction in mortality, compared to a milrinone plus dopamine combination (RR 0.32), but the 95% CI of the effect also included 1 (0.04 to 2.94), and the certainty of the evidence was deemed low due to within‐study bias and imprecision. Another small, separate study, Shah 2013, combined levosimendan with dobutamine versus milrinone with dobutamine and found that the levosimendan plus dobutamine combination may result in a large reduction in mortality, compared to a milrinone plus dobutamine combination: RR 0.33, 95% CI 0.04 to 2.99, with a low certainty of evidence due to within‐study bias and imprecision.
Sensitivity analysis excluding Shah 2013 due to high risk of bias and unknown duration of follow‐up was not applicable, because this study constituted its own subnetwork. The other two subnetworks were not affected by the exclusion of this study.
Sensitivity analysis excluding Costello 2014 due to a different study population (univentricular hearts only) was performed for the NMA subnetwork. Since there were no events of mortality in either study arm, the study could only be kept in the analysis by adding an increment of 0.5 to each study arm, which changed the result slightly. As adding such increments is controversial, the changes were minimal, and the ranking did not change either, the influence of Costello 2014 was deemed negligible.
We conducted two versions of CNMA, with and without assuming an active effect of placebo. We assumed an additive effect of combinations of inotropes. There was no evidence of an effect of levosimendan or of milrinone on mortality, each compared to placebo: levosimendan general RR 0.57 (95% CI 0.15 to 2.13; P = 0.41); levosimendan assuming placebo as inactive RR 0.49 (95% CI 0.16 to 1.53; P = 0.22); milrinone general RR 1.21 (95% CI 0.18 to 7.98; P = 0.84); milrinone assuming placebo as inactive RR 1.04 (95% CI 0.18 to 6.07; P = 0.97) (see Figure 5 in Appendix 3).
Pairwise analyses revealed no evidence of a difference in all‐cause mortality between levosimendan and milrinone (RR 0.59, 95% CI 0.11 to 3.29; P = 0.55; 4 studies, 169 participants; Analysis 1.1), and between levosimendan and placebo (RR 0.57, 95% CI 0.15 to 2.13; P = 0.41; 2 studies, 217 participants; Analysis 2.1).
1.1. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 1: All‐cause mortality within 30 days
2.1. Analysis.

Comparison 2: Pairwise analysis: levosimendan versus placebo, Outcome 1: All‐cause mortality within 30 days
2 Time to death
One study, Momeni 2011, reported one intraoperative death in each of its two study arms. Another study reporting the times of death of all affected participants was Wang 2019, where the survival curves show that the last death within 90 days occurred after 10 days in the levosimendan group (total 3/94 in levosimendan group – days 2, 3, 10; and 4/93 in placebo group – days 1, 2, 4, 5). For this direct evidence, the survival curves are shown in Figure 6, Appendix 3, and the hazard ratio was 0.73 (95% CI 0.16 to 3.28; P = 0.69), thus not proving a benefit for levosimendan versus placebo. In one study, Hoffman 2003, there was one death each on postoperative days 5 and 13, but the treatment group is unknown.
3 Low cardiac output syndrome
The studies that contributed to our analysis of LCOS risk included different patient populations: neonates with biventricular physiology (Pellicer 2013; Ricci 2012: Risk Adjusted Classification for Congenital Heart Surgery (RACHS) 3 to 4), infants with biventricular physiology from birth to one years of age (Jothinath 2021: RACHS 1 to 4, Lechner 2012: neonates from 36 weeks gestational age and infants up to one year of age: Aristotle risk score 2 to 4), young children aged from birth to four years with uni‐ or biventricular physiology RACHS 2 to 5 (Wang 2019), or young children aged from birth to six years with biventricular physiology (Hoffman 2003). We did not find enough information about the subgroups of neonates or infants in order to assess their outcomes separately.
For the outcome LCOS, there were two subnetworks with a total of six studies (see Figure 7 in Appendix 3), of which five studies comprising 513 participants and 85 total events formed the larger subnetwork and one study, Ricci 2012, formed the smaller subnetwork. NMA of the larger subnetwork resulted in benefits for levosimendan (4 studies, 144 participants), which results in a large reduction in the incidence of LCOS, compared to placebo (2 studies, 168 participants) (RR 0.45, 95% CI 0.24 to 0.83; high certainty of the evidence, due to heterogeneity but large magnitude of effect). Likewise, milrinone (4 studies, 122 participants) likely results in a large reduction in the incidence of LCOS, compared to placebo (RR 0.46, 95% CI 0.24 to 0.89; moderate certainty of the evidence due to within‐study bias and heterogeneity, but large magnitude of effect). Low‐dose milrinone (1 study, 79 participants) may reduce the incidence of LCOS, compared to placebo (RR 0.70, 95% CI 0.39 to 1.28; low certainty of the evidence due to within‐study bias and imprecision). The assessment of heterogeneity in the larger subnetwork yielded a tau2 of 0 and an I2 of 0% (95% CI 0% to 89.6%); Cochran's Q total 1.2 (degrees of freedom (df) = 2, P = 0.55), within designs 0.05 (df = 1, P = 0.82), and between designs 1.14 (df = 1, P = 0.29).
Sensitivity analysis after the exclusion of Hoffman 2003 and Jothinath 2021 due to high risk of bias showed a change in the NMA subnetwork. With the Mantel‐Haenszel method, including those two studies, levosimendan and milrinone seemed to have almost the same effect on LCOS incidence (RR 0.45 and 0.46, respectively); the difference between the two was greater and the 95% CI were wider (both including 1) after exclusion of the two studies: levosimendan: RR 0.55, 95% CI 0.27 to 1.1, milrinone: RR 0.67, 95% CI 0.15 to 3.1. Milrinone low‐dose (LD) is not evaluated in the case of exclusion of the two studies. The ranking results changed accordingly: when both studies were included, SUCRA (surface under the cumulative ranking curve, calculated with R package netmeta) was 0.80 for levosimendan, 0.77 for milrinone, and 0.05 for placebo. When both studies were excluded, SUCRA was similar for levosimendan at 0.78, but lower at 0.54 for milrinone, and mildly higher at 0.18 for placebo. Ranking, therefore, shows that levosimendan and milrinone are most preferable for prevention of LCOS, low‐dose milrinone is less preferable, and placebo is the least preferable of these four interventions. Ranking probabilities are detailed in Table 14.
7. Ranking probabilities for intervention effectiveness to prevent low cardiac output syndrome.
| Probabilities | Rank | Levosimendan | Milrinone | Milrinone LD | Placebo |
| 1 | 0.501 | 0.448 | 0.051 | 0.0000 | |
| 2 | 0.409 | 0.423 | 0.163 | 0.005 | |
| 3 | 0.088 | 0.124 | 0.628 | 0.16 | |
| 4 | 0.002 | 0.005 | 0.158 | 0.835 |
Ranking probabilities based on 1000 simulations, random‐effects model
Abbreviations LD: low dose
The study forming the smaller subnetwork, Ricci 2012, showed that the incidence of LCOS may be greatly reduced with the addition of levosimendan to a combination of milrinone and dopamine, with an RR of 0.61 and the 95% CI of the effect barely including 1 (0.36 to 1.04); however, this is low‐certainty evidence due to within‐study bias and imprecision.
We conducted two versions of CNMA, with and without assuming an active effect of placebo. We assumed an additive effect of combinations of inotropes. The risk ratio for LCOS was reduced with levosimendan (RR in general CNMA 0.45, 95% CI 0.24 to 0.83, RR assuming no effect of placebo 0.54, 95% CI 0.36 to 0.80) and for milrinone (RR in general CNMA 0.46, 95% CI 0.24 to 0.89, RR assuming no effect of placebo 0.50, 95% CI 0.27 to 0.93). The CNMA for low‐dose milrinone compared to placebo resulted in an effect with a 95% CI including 1 (see Figure 8 in Appendix 3).
Pairwise analysis revealed no evidence of a difference in the incidence of LCOS between levosimendan and milrinone (RR 0.72, 95% CI 0.31 to 1.70; P = 0.46; 3 studies, 99 participants; Analysis 1.2).
1.2. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 2: Low cardiac output syndrome
Secondary outcomes
1 Length of intensive care stay
There were 11 studies reporting the length of intensive care unit (ICU) stay of a total of 690 study participants in three subnetworks; one consisted of nine studies with 577 participants, and the two others consisted of only one study each (see Figure 9 in Appendix 3). The mean length of stay varied between means of 1.5 and 2.1 days, respectively, in the treatment arms of one study (Jothinath 2021), and 14 and 11 days, respectively, in the treatment arms of another study (Ricci 2012).
Network meta‐analysis showed that there may be little to no difference in the ratio of means (ROM) for the length of ICU stay in patients treated with levosimendan (7 studies, 225 participants) (ROM 1.12, 0.77 to 1.63; low certainty of the evidence due to imprecision and heterogeneity), and that there is likely little to no difference in the ratio of means (ROM) for the length of ICU stay in patients treated with milrinone (6 studies, 158 participants) (ROM 1.13, 0.75 to 1.69; moderate certainty of the evidence due to imprecision) or dobutamine (2 studies, 51 participants) (ROM 1.11, 0.66 to 1.86; moderate certainty of the evidence due to imprecision), compared to placebo (3 studies, 143 participants), and the 95% CI always included 1. Ranking probabilities for the different treatments are detailed in Table 15.
8. Ranking probabilities for intervention effectiveness regarding length of intensive care stay.
| Probabilities | Rank | Dobutamine | Levosimendan | Milrinone | Placebo |
| 1 | 0.254 | 0.094 | 0.112 | 0.54 | |
| 2 | 0.241 | 0.292 | 0.265 | 0.202 | |
| 3 | 0.208 | 0.353 | 0.317 | 0.112 | |
| 4 | 0.297 | 0.261 | 0.306 | 0.136 |
Ranking probabilities based on 1000 simulations, random‐effects model
There was heterogeneity within designs, but not between designs (inconsistency) for this outcome in the multi‐study subnetwork. Heterogeneity tests revealed tau2 = 0.06; I2 = 68.3% (95% CI 29.9% to 85.7%); Cochran's Q total 18.94 (degrees of freedom (df) = 6, P < 0.005), within designs 17.13 (df = 4, P = 0.002), and between designs 1.81 (df = 2, P = 0.40).
There may be no difference in the length of ICU stay by addition of levosimendan to a regimen of milrinone plus dopamine in one study disconnected from the main network, with low certainty of the evidence due to within‐study bias and imprecision (Ricci 2012; ROM 1.27, 95% CI 0.83 to 1.96), whereas the length of intensive care stay was shown to probably decrease in another study disconnected from the main network for the levosimendan plus dobutamine group (Shah 2013), compared to the milrinone plus dobutamine group: ROM 0.74, 95% CI 0.71 to 0.77, with moderate certainty of the evidence due to within‐study bias.
We conducted two versions of CNMA, with and without assuming an active effect of placebo. We assumed an additive effect of combinations of inotropes. All analyses for dobutamine, levosimendan, levosimendan + dobutamine, milrinone, and for milrinone + dobutamine, compared to placebo, resulted in effects with a 95% CI including 1 (see Figure 10 in Appendix 3).
Pairwise analyses revealed no evidence of a difference in the length of ICU stay between levosimendan and milrinone (mean difference 0.34 days, 95% CI ‐1.14 to 1.81; P = 0.65; 4 studies, 189 participants; Analysis 1.3) and between levosimendan and placebo (mean difference ‐0.06 days, 95% CI ‐1.16 to 1.04; P = 0.91; 2 studies, 217 participants; Analysis 2.2).
1.3. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 3: Length of intensive care stay
2.2. Analysis.

Comparison 2: Pairwise analysis: levosimendan versus placebo, Outcome 2: Length of intensive care stay
2 Length of hospital stay
Seven studies provided sufficient data about the length of their participants' hospital stay, reporting on a total of 497 patients (see Figure 11 in Appendix 3). This outcome ranged from 5 and 5.8 days in the treatment arms of one study (Jothinath 2021) to 20 days in each treatment arm of another study (Wang 2019).
There was only one single network for this outcome. Levosimendan and milrinone yielded likely no change in the length of hospital stay compared to placebo: levosimendan (5 studies, 185 participants) ROM 1.03, 95% CI 0.84 to 1.27 (moderate certainty of the evidence due to imprecision); milrinone (7 studies, 222 participants) ROM 1, 95% CI 0.78 to 1.30 (moderate certainty of the evidence due to imprecision). Dobutamine (1 study, 26 participants), on the other hand, likely reduces the length of hospital stay slightly, compared to placebo (3 studies, 199 participants): ROM 0.68, 95% CI 0.37 to 1.26 (moderate certainty of the evidence due to imprecision). Ranking probabilities are detailed in Table 16. We did not perform a component NMA for this outcome, because there were no combination treatments within the network.
9. Ranking probabilities for intervention effectiveness regarding length of hospital stay.
| Probabilities | Rank | Dobutamine | Levosimendan | Milrinone | Placebo |
| 1 | 0.843 | 0.023 | 0.055 | 0.079 | |
| 2 | 0.058 | 0.211 | 0.369 | 0.362 | |
| 3 | 0.031 | 0.362 | 0.396 | 0.211 | |
| 4 | 0.068 | 0.404 | 0.18 | 0.348 |
Ranking probabilities based on 1000 simulations, random‐effects model
Heterogeneity tests revealed tau2 = 0.01; I2 = 31.2% (95% CI 0.0% to 73.6%); Cochran's Q total 5.81 (degrees of freedom (df) = 4, P = 0.21), within designs 5.46 (df = 3, P = 0.14), and between designs 0.36 (df = 1, P = 0.55).
Pairwise analysis revealed no evidence of a difference in the length of hospital stay between levosimendan and milrinone (mean difference 0.40 days, 95% CI ‐1.42 to 2.22; P = 0.67); 4 studies, 189 participants; Analysis 1.4).
1.4. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 4: Length of hospital stay
3 Duration of mechanical ventilation
There were 11 studies in three subnetworks for this outcome (total 690 participants), of which the largest subnetwork comprised nine studies with 577 participants (see Figure 12 in Appendix 3). There were durations of mechanical ventilation between approximately 3 and 4 hours in the treatment arms of one study (Abril‐Molina 2021) and 166 and 142 hours in the treatment arms of another study (Ricci 2012). Levosimendan (7 studies, 225 participants) and milrinone (7 studies, 222 participants) likely increase the duration of mechanical ventilation, compared to placebo (4 studies, 214 participants) (levosimendan ROM 1.17, 95% CI 0.65 to 2.12; moderate certainty of the evidence due to imprecision; milrinone ROM 1.25, 95% CI 0.67 to 2.36; moderate certainty of the evidence due to imprecision), while dobutamine (2 studies, 51 participants) likely results in no difference (ROM 1.04, 95% CI 0.45 to 2.38; moderate certainty of the evidence due to imprecision), compared to placebo. There was heterogeneity within designs and between designs (inconsistency) for this outcome in the multi‐study subnetwork. Heterogeneity tests revealed tau2 = 0.15; I2 = 67.9% (95% CI 28.7% to 85.5%); Cochran's Q total 18.67 (degrees of freedom (df) = 6, P < 0.005), within designs 11.73 (df = 4, P = 0.02), and between designs 6.94 (df = 2, P = 0.03). Ranking probabilities are detailed in Table 17.
10. Ranking probabilities for intervention effectiveness regarding duration of mechanical ventilation.
| Probabilities | Rank | Dobutamine | Levosimendan | Milrinone | Placebo |
| 1 | 0.347 | 0.102 | 0.086 | 0.465 | |
| 2 | 0.28 | 0.305 | 0.193 | 0.222 | |
| 3 | 0.159 | 0.387 | 0.323 | 0.131 | |
| 4 | 0.214 | 0.206 | 0.398 | 0.182 |
Ranking probabilities based on 1000 simulations, random‐effects model
One study disconnected from the main network, Ricci 2012, added levosimendan to a regimen of milrinone plus dopamine and found that this may increase the duration of mechanical ventilation slightly (ROM 1.17, 95% CI 0.71 to 1.93), but with low certainty of the evidence due to within‐study bias and imprecision. The duration of mechanical ventilation is likely decreased, according to another study disconnected from the main network (Shah 2013), for the levosimendan plus dobutamine group, compared to the milrinone plus dobutamine group: ROM 0.71, 95% CI 0.65 to 0.78, with moderate certainty of the evidence due to within‐study bias.
We conducted two versions of CNMA, with and without assuming an active effect of placebo. We assumed an additive effect of combinations of inotropes. All analyses for dobutamine, levosimendan, levosimendan + dobutamine, milrinone, and for milrinone + dobutamine, compared to placebo, resulted in effects with a 95% CI including 1 (see Figure 13 in Appendix 3).
Pairwise analyses revealed no evidence of a difference in the duration of mechanical ventilation between levosimendan and milrinone (mean difference 3.95 hours, 95% CI ‐14.42 to 22.31; P = 0.67; 4 studies, 189 participants; Analysis 1.5) and between levosimendan and placebo (mean difference 1.21 hours, 95% CI ‐3.50 to 5.91; P = 0.62; 2 studies, 217 participants; Analysis 2.3).
1.5. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 5: Duration of mechanical ventilation
2.3. Analysis.

Comparison 2: Pairwise analysis: levosimendan versus placebo, Outcome 3: Duration of mechanical ventilation
4 Inotrope score
There were multiple definitions of inotrope scores in our included studies. Abril‐Molina 2021 reported on a vasoactive inotrope score (VIS), as did Jothinath 2021 while adding a distinction from "LVIS" including levosimendan: "dopamine dose (mcg/kg/min)+dobutamine dose (mcg/kg/min)+100×epinephrine dose (mcg/kg/min)+10×milrinone dose (mcg/kg/min)+10,000×vasopressin dose (U/kg/min)+100×norepinephrine dose (mcg/kg/min)+50×levosimendan dose (mcg/kg/min)" for the levosimendan treatment arm, after 12 hours. We used inotrope scores after 12 hours from Thorlacius 2020 as well. For Costello 2014, an inotrope score during the first 24 hours post surgery is reported ("dopamine dose (mcg/kg/min)+dobutamine dose (mcg/kg/min)+100×epinephrine dose (mcg/kg/min)"), and Wang 2019 also provided data for one day after surgery, calculated by "dopamine (μg/kg/min) + dobutamine (μg/kg/min) + [100 × epinephrine (μg/kg/min)] + [10 ×milrinone (μg/kg/min)] + [10,000 × vasopressin (U/kg/min)] + [100 × norepinephrine]". Ricci 2012 provided information for an inotrope score six hours after surgery to be read from their figure 1, defined as "dopamine mcg/kg/min x 1 + milrinone mcg/kg/min x 10 + epinephrine mcg/kg/min x 100".
While Jothinath 2021 and Ricci 2012 provided measures of means but not of distributions, there were six studies with this information available, of which three compared levosimendan to milrinone (Lechner 2012; Pellicer 2013; Thorlacius 2020), two compared levosimendan to placebo (Abril‐Molina 2021; Wang 2019), and one compared milrinone to placebo (Costello 2014). The inotrope score was decreased with levosimendan (ROM 0.84, 95% CI 0.73 to 0.97), compared to placebo, but not different with milrinone (ROM 1.04, 95% CI 0.82 to 1.31), compared to placebo. Ranking of treatments showed a SUCRA of 0.98 for levosimendan, 0.32 for placebo, and 0.20 for milrinone.
Heterogeneity tests revealed tau2 = 0; I2 = 0% (95% CI 0.0% to 79.2%); Cochran's Q total 3.31 (degrees of freedom (df) = 4; P = 0.51), within designs 3.27 (df = 3, P = 0.35), and between designs 0.04 (df = 1; P = 0.83).
Separating indirect from direct evidence resulted in a ratio of ratios close to 1 for all comparisons, with P = 0.83.
Pairwise analyses revealed no evidence of a difference in the inotrope score between levosimendan and milrinone (mean difference ‐0.88, 95% CI ‐2.42 to 0.66; P = 0.26; 3 studies, 129 participants; Analysis 1.6), but a lower inotrope score with levosimendan, compared to placebo (mean difference ‐1.06, 95% CI ‐2.04 to ‐0.08; P = 0.03; 2 studies, 217 participants; Analysis 2.4).
1.6. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 6: Inotrope score
2.4. Analysis.

Comparison 2: Pairwise analysis: levosimendan versus placebo, Outcome 4: Inotrope score
5 Number of patients requiring mechanical circulatory support
There were seven studies reporting on the incidence of mechanical circulatory support, consisting of ECMO or VAD. Of those, three had zero events in both of their treatment arms, and they were excluded from further analysis. There were two subnetworks from the four remaining studies, with one study (Shah 2013) disconnected from the remaining subnetwork (Cavigelli‐Brunner 2018; Costello 2014; Thorlacius 2020) and not showing a likely difference between levosimendan plus dobutamine versus milrinone versus dobutamine: RR 0.33, 95% CI 0.01 to 7.80. Ranking yielded a SUCRA of 0.72 for milrinone, 0.55 for levosimendan, and 0.23 for placebo. SIDE was not possible, because there were either only direct or only indirect comparisons between treatments for this outcome.
Heterogeneity tests revealed tau2 = 0; I2 = 0%; Cochran's Q total 0.19 (degrees of freedom (df) = 1, P = 0.66), within designs 0.19 (df = 1, P = 0.66), and between designs 0 (df = 0).
The main subnetwork did not prove an effect of either levosimendan (RR 0.33, 95% CI 0.01 to 7.57) or milrinone (RR 0.22, 95% CI 0.01 to 9.32), compared to placebo.
Pairwise analysis revealed no evidence of a difference in the number of patients requiring mechanical circulatory support between levosimendan and milrinone (RR 1.49, 95% CI 0.19 to 11.37; P = 0.70; 3 studies, 130 participants; Analysis 1.7).
1.7. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 7: Number of patients requiring mechanical circulatory support
6 Number of patients requiring cardiac transplantation
There were seven studies with a total of 344 participants reporting on the incidence of heart transplantation, but none of the studies had any of these events, so no further analysis of this outcome was possible.
7 Adverse events
We pooled the incidences of all our pre‐specified adverse events for a combined analysis. There was a possibility of one participant experiencing more than one adverse event, such that the number of events could be greater than the number of participants. Therefore, we used an incidence rate ratio (IRR) instead of a risk ratio. The results of this analysis are displayed in Table 7.
For the outcome of cumulative adverse events, we encountered a disconnected network. There were three subnetworks consisting of information from 10 studies with 819 participants experiencing a total of 415 events (see Figure 14 in Appendix 3). We conducted NMA for the largest subnetwork and two versions of CNMA, with and without assuming an active effect of placebo. All analyses for all interventions, compared to placebo, resulted in effects with a 95% CI including 1 (see Figure 15 in Appendix 3). NMA of the largest subnetwork (8 studies, 706 participants, 380 events) showed that low‐dose milrinone (1 study, 80 participants) may decrease the incidence of adverse events, compared to placebo (4 studies, 224 participants) (IRR 0.84, 95% CI 0.47 to 1.50; low certainty of the evidence due to within‐study bias and imprecision), while standard‐dose milrinone (6 studies, 204 participants) may increase the incidence of adverse events (IRR 1.31, 95% CI 0.96 to 1.79; low certainty of the evidence due to within‐study bias and imprecision). Dobutamine (1 study, 26 participants) (1.24, 955% CI 0.75 to 2.03; moderate certainty of the evidence due to imprecision) and levosimendan (5 studies, 172 participants) (1.23, 95% CI 0.78 to 1.96; moderate certainty of the evidence due to imprecision) likely increase the incidence of adverse events compared to placebo. All 95% CI included an IRR of 1. Ranking yielded a SUCRA (calculated with R package netmeta) of 0.8461 for low‐dose milrinone, 0.71 for placebo, 0.36 for dobutamine, 0.36 for levosimendan, and 0.22 for milrinone.
Heterogeneity tests revealed tau2 = 0; I2 = 0% (95% CI 0.0% to 74.6%); Cochran's Q total 1.28 (degrees of freedom (df) = 5; P = 0.94), within designs 1.03 (df = 3; P = 0.79), and between designs 0.25 (df = 2; P = 0.88).
Sensitivity analysis with exclusion of the study by Hoffman 2003 due to high risk of bias (Shah 2013 and Ricci 2012 also had high risk of bias, but were disconnected from the largest network) resulted in an IRR of 1.24 (95% CI 0.75 to 2.06) for levosimendan, an IRR of 1.32 (95% CI 0.91 to 1.92) for milrinone, and an IRR of 1.27 (95% CI 0.74 to 2.18) for dobutamine, compared to placebo (IRR = 1).
The smaller subnetworks included only one study each: Shah 2013 compared levosimendan + dobutamine versus milrinone + dobutamine, and the incidence of all adverse events was likely largely reduced with levosimendan + dobutamine compared to milrinone + dobutamine (IRR 0.29, 95% CI 0.12 to 0.71), with moderate certainty of the evidence due to within‐study bias. Ricci 2012 compared milrinone + dopamine to this regimen plus levosimendan, and there was likely no difference in the incidence of all adverse events (IRR 0.97, 95% CI 0.24 to 3.87), with moderate certainty of the evidence due to within‐study bias.
CNMA was possible including the largest subnetwork together with Shah 2013, with placebo assumed as inactive, resulting in an IRR of 1.04 (95% CI 0.66 to 1.64; P = 0.87) for levosimendan, IRR of 1.32 (95% CI 0.96 to 1.81; P = 0.08) for milrinone, IRR of 0.84 (95% CI 0.47 to 1.51; P = 0.57) for low‐dose milrinone, and IRR of 1.27 (95% CI 0.77 to 2.10; P = 0.36) for dobutamine. We assumed an additive effect of combinations of inotropes.
Pairwise analysis revealed no evidence of a difference in the number of adverse events between levosimendan and placebo (IRR 1.01, 95% CI 0.36 to 2.85; P = 0.98; 2 studies, 217 participants; Figure 16 in Appendix 3). There was also no evidence of more adverse events with prophylactic milrinone than with placebo in pairwise analysis (RR 1.34, 95% CI 0.97 to 1.85; P = 0.08; 2 studies, 219 participants; Figure 17 in Appendix 3).
7a Adverse events: incidence of arrhythmia
The definition of arrhythmia was not universal in our included studies. While Hoffman 2003 reported on the incidence of nodal arrhythmia, there was a case of atrio‐ventricular asynchrony in the comparison group in Pellicer 2013, and a reported incidence of junctional ectopic tachycardia in Thorlacius 2020. Pellicer 2013 and Thorlacius 2020 also reported on surgery‐related, high‐degree atrio‐ventricular block, but we did not add these numbers to the above due to the presumably surgery‐related rather than medication‐related aetiology of these atrio‐ventricular blocks.
Abril‐Molina 2021 reported two patients with arrhythmia in each group according to table 4 of their publication, but two in the levosimendan and three in the placebo group according to table 3 of their publication after 12 hours.
There were nine studies in two subnetworks, with one subnetwork made out of only one study (Shah 2013), comparing levosimendan plus dopamine versus milrinone versus dopamine. No evidence of a difference was found there (RR 0.33, 95% CI 0.07 to 1.50). For the larger subnetwork, we did not consider two studies without any events in their treatment arms (Lechner 2012; Momeni 2011), leaving six studies with mostly two treatment arms each (Cavigelli‐Brunner 2018; Costello 2014; Pellicer 2013; Thorlacius 2020; Wang 2019) or three treatment arms (Hoffman 2003).
The use of levosimendan (RR 1.20, 95% CI 0.46 to 3.12), dobutamine (RR 0.93, 95% CI 0.28 to 3.05), or low‐dose milrinone (RR 1.49, 95% CI 0.53 to 4.23) likely has no different risk compared to placebo regarding the incidence of arrhythmia, but there was an increased risk of arrhythmia with standard‐dose milrinone (RR 1.76, 95% CI 1.10 to 2.81), compared to placebo. Ranking of treatments resulted in a SUCRA of 0.71 for placebo, 0.70 for dobutamine, 0.53 for levosimendan, 0.37 for milrinone LD, and 0.18 for milrinone.
Heterogeneity tests revealed tau2 = 0; I2 = 0% (95% CI 0.0% to 84.7%); Cochran's Q total 2.35 (df = 3, P = 0.50), within designs 0.87 (df = 1, P = 0.35), and between designs 1.48 (df = 2, P = 0.48).
Separating indirect from direct evidence showed ratio of ratios (RoR) (direct/indirect) for levosimendan versus milrinone of 0.84 (P = 0.86), for levosimendan versus placebo of 1.19 (P = 0.86), for milrinone versus milrinone LD of 9.95 (P = 0.24), for milrinone versus placebo of 0.84 (P = 0.86), and for milrinone LD versus placebo of 3.57 (P = 0.24).
Pairwise analyses revealed no evidence of a difference in the incidence of arrhythmia between levosimendan and milrinone (RR 0.64, 95% CI 0.20 to 2.07; P = 0.46; 4 studies, 169 participants; Analysis 1.8) and between milrinone and placebo (RR 2.01, 95% CI 0.83 to 4.85; P = 0.12; 2 studies, 229 participants; Analysis 3.1).
1.8. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 8: Arrhythmia
3.1. Analysis.

Comparison 3: Pairwise analysis: milrinone versus placebo, Outcome 1: Arrhythmia
7b Adverse events: incidence of hypotension
The definition of hypotension was rarely the same as ours. We used numbers from Costello 2014 related to discontinuation of the study drug because of systolic blood pressures below 75 mmHg despite volume and dopamine infusion. The results from Shah 2013 implied that the patient required norepinephrine in addition to the study drugs. Wang 2019 used a definition of "systolic blood pressure (SBP) < the 5th percentile for normal individuals". Overall, five studies provided data for this outcome (Costello 2014; Hoffman 2003; Shah 2013; Thorlacius 2020; Wang 2019), of which Thorlacius 2020 did not find any incidence in either treatment arm.
Shah 2013 was separated from the remaining subnetwork and resulted in a lower incidence of hypotension with levosimendan plus dobutamine versus milrinone plus dobutamine (RR 0.1, 95% CI 0.01 to 0.72). The larger subnetwork did not show any differences compared to placebo with the use of levosimendan (RR 0.99, 95% CI 0.02 to 49.22), milrinone (RR 3.18, 95% CI 0.21 to 48.96), or low‐dose milrinone (RR 0.53, 95% CI 0.02 to 16.63). Ranking provided the following SUCRA scores: milrinone low‐dose = 0.69, placebo = 0.55, levosimendan = 0.53, and milrinone = 0.22.
Heterogeneity was high with tau2 = 2.99; I2 = 72.9% (95% CI 0.0% to 93.9%); Cochran's Q total 3.69 (df = 1, P = 0.05), of which within designs Q was 0 (df = 0).
Separating indirect from direct evidence was possible for milrinone versus milrinone LD, with RoR < 0.01 (P = 0.32) and milrinone LD versus placebo, with RoR < 0.01 (P = 0.32).
Pairwise analyses revealed no evidence of a difference in the incidence of hypotension between milrinone and placebo (RR 3.38, 95% CI 0.16 to 72.69; P = 0.44; 2 studies, 229 participants; Analysis 3.2).
3.2. Analysis.

Comparison 3: Pairwise analysis: milrinone versus placebo, Outcome 2: Hypotension
7c Adverse events: incidence of headache
Not reported in the included studies.
7d Adverse events: incidence of intraventricular haemorrhage
Not reported in the included studies.
7e Adverse events: incidence of hypocalcaemia
Not reported in the included studies.
7f Adverse events: incidence of hypokalaemia
As far as the definition of this outcome was described in the included studies, it was specified as serum potassium < 3.5 mEq/L in Cavigelli‐Brunner 2018 and Thorlacius 2020 (unpublished data) and as < 3 mEq/L in Costello 2014. Wang 2019 assessed hypokalaemia but did not report the results. Pellicer 2013 did not find any cases of hypokalaemia (unpublished data).
There were two subnetworks, with one of them comprising only one study (Shah 2013), which did not find evidence of a difference in this outcome for patients treated with levosimendan plus dobutamine versus those treated with milrinone plus dobutamine (RR 5.00, 95% CI 0.25 to 99.06), ranking SUCRA at 0.85 for the milrinone combination and 0.15 for the levosimendan combination.
The other subnetwork comprised three studies (Cavigelli‐Brunner 2018; Costello 2014; Thorlacius 2020). Treatment effect estimates compared to placebo were as follows: RR 1.07 (95% CI 0.60 to 1.89) for dobutamine, RR 3.67 (95% CI 1.01 to 13.29) for levosimendan, and RR 1.16 (95% CI 0.82 to 1.64) for milrinone. Ranking resulted in the following SUCRA scores: 0.79 for placebo, 0.67 for dobutamine, 0.51 for milrinone, and 0.03 for levosimendan.
Heterogeneity tests revealed tau2 = 0; I2 = 0%; Cochran's Q total 0.43 (degrees of freedom (df) = 1, P = 0.51), within designs 0.43 (df = 1, P = 0.51), and between designs 0 (df = 0).
Separating indirect from direct evidence was not possible, because all treatment combinations were only comparable either directly or indirectly, but not both.
Pairwise analysis revealed a higher incidence of hypokalaemia with levosimendan than with milrinone (RR 3.17, 95% CI 0.92 to 10.95; P = 0.07; 2 studies, 90 participants; Analysis 1.9).
1.9. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 9: Hypokalaemia
7g Adverse events: incidence of bronchospasm
Not reported in the included studies.
7h Adverse events: incidence of thrombocytopaenia
An explicit definition of thrombocytopaenia was provided by Cavigelli‐Brunner 2018, as a 20% or more drop from baseline platelet count upon arrival on the intensive care unit, which was experienced by a high proportion of participants in that study. A platelet count below 50 x 109/L was not experienced by any participant in the Thorlacius 2020 study (unpublished data). Pellicer 2013 did not find thrombocytopaenia in any of their treatment arms either and was excluded from the NMA.
There were four studies remaining for analysis (Cavigelli‐Brunner 2018; Costello 2014; Hoffman 2003; Shah 2013). The subnetwork of the single study by Shah 2013 did not show a difference between levosimendan plus dobutamine versus milrinone plus dobutamine regarding the incidence of thrombocytopaenia, even though the mean effect was well below an RR of 1 (RR 0.14, 95% CI 0.01 to 2.63). Ranking, therefore, favoured levosimendan plus dopamine (SUCRA = 0.90) above milrinone plus dobutamine (SUCRA = 0.10).
The larger subnetwork provided the following treatment estimates compared to placebo: dobutamine: RR 0.68 (95% CI 0.31 to 1.49), milrinone: RR 0.73 (95% CI 0.34 to 1.58), and milrinone LD: RR 0.86 (95% CI 0.36 to 2.05). Ranking showed SUCRA scores of 0.79 for dobutamine, 0.53 for milrinone, 0.44 for milrinone LD, and 0.25 for placebo.
Heterogeneity testing showed tau2 = 0; I2 = 0%; Cochran's Q total 0.16 (df = 1, P = 0.69), of which within designs Q was 0 (df = 0).
Separating indirect from direct evidence was possible for milrinone versus milrinone LD, with RoR 1.88 (P = 0.69) and milrinone LD versus placebo, with RoR 2.06 (P = 0.69).
Pairwise analyses revealed no evidence of a difference in the incidence of thrombocytopaenia between milrinone and placebo (RR 0.73, 95% CI 0.34 to 1.58; P = 0.43; 2 studies, 229 participants; Analysis 3.3).
3.3. Analysis.

Comparison 3: Pairwise analysis: milrinone versus placebo, Outcome 3: Thrombocytopaenia
7i Adverse events: incidence of anaemia
'Anaemia' results were only available from Thorlacius 2020, and no pooling with other studies was possible. In this one study, there were no cases of anaemia in either treatment group (with the study protocol specifying that postoperative haemoglobin should be maintained above 100 g/L).
7j Adverse events: incidence of elevated liver enzymes
The definition of elevated liver enzymes in Costello 2014 was "AST or ALT >400 IU/L after Fontan operation". Another study, Wang 2019, reported only absolute values, not a proportion of patients experiencing an elevation of liver enzymes above a certain cut‐off value. Pellicer 2013 did not find any incidence in either of their two treatment arms (unpublished data).
This left only the study Shah 2013, comparing levosimendan plus dobutamine versus milrinone plus dobutamine, with no evidence of a difference (RR of the former versus the latter 3.00, 95% CI 0.13 to 70.23) and the study by Costello 2014, comparing milrinone versus placebo, which also did not show a likely difference (milrinone: RR 0.49, 95% CI 0.10 to 2.49).
7k Adverse events: incidence of reduced LVEF < 50% or reduced LVFS < 28%
While Cavigelli‐Brunner 2018 performed echocardiography and reported LVFS < 28% two to six hours postoperatively, Pellicer 2013 reported the same outcome 24 hours postoperatively. Thorlacius 2020 performed postoperative echocardiography on the morning of the first day after surgery.
There was one network with three possible treatments. Using milrinone as a comparator, the RR (inverse variance method) for dobutamine was 1.85 (95% CI 0.74 to 4.63; P = 0.19) and for levosimendan 0.95 (95% CI 0.84 to 1.07; P = 0.40), and indirect comparison of dobutamine versus levosimendan resulted in a RR of 1.94 with dobutamine. Ranking showed SUCRA scores of 0.86 for levosimendan, 0.55 for milrinone, and 0.09 for dobutamine, with lower values indicating preferable treatments.
Separating indirect from direct evidence was not possible, because all treatment combinations were only comparable either directly or indirectly, but not both.
Pairwise analysis revealed no evidence of a difference in the incidence of reduced LVEF or reduced LVFS between levosimendan and milrinone (RR 0.95 for levosimendan, 95% CI 0.84 to 1.07; P = 0.40; 2 studies, 84 participants; Analysis 1.10) and between dobutamine and milrinone (RR 1.85 for dobutamine, 95% CI 0.74 to 4.63; 1 study, 50 participants).
1.10. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 10: Reduced left ventricular ejection fraction (< 50%) or shortening fraction (< 28%)
7l Adverse events: requirement for renal replacement therapy
There was different information regarding acute kidney injury or measures of kidney function in different studies. Therefore, an endpoint of requirement for renal replacement therapy was chosen to provide maximal comparability between studies. The incidence of renal replacement therapy was generally low (total reported: 17 cases among 527 patients = 3.2%).
We found three subnetworks among the studies, which reported cases of renal replacement therapy. The largest comprised five studies (Cavigelli‐Brunner 2018; Costello 2014; Momeni 2011; Thorlacius 2020; Wang 2019); the two smaller ones were made up of one single study each (Shah 2013 and Ricci 2012, respectively).
Treatment effect estimates using placebo as a reference were RR 0.50 (95% CI 0.01 to 24.74) for dobutamine, RR 1.54 (95% CI 0.17 to 14.03) for levosimendan, and 1.62 (95% CI 0.16; 16.15) for milrinone. Ranking of treatments resulted in SUCRA scores of 0.71 for dobutamine, 0.56 for placebo, 0.38 for levosimendan, and 0.35 for milrinone. Therefore, dobutamine seems to be a superior choice to avoid renal failure, even preferable to placebo.
Heterogeneity tests revealed tau2 = 0; I2 = 0% (95% CI 0.0% to 89.6%); Cochran's Q total 1.24 (df = 2, P = 0.54), within designs 0.96 (df = 1, P = 0.33), and between designs 0.28 (df = 1, P = 0.60).
Separating indirect from direct evidence showed an RoR (direct/indirect risk ratio) of 3.45 (P = 0.60) for levosimendan versus milrinone, 0.29 (P = 0.60) for levosimendan versus placebo, and 3.45 (P = 0.60) for milrinone versus placebo.
In the two separate studies, no difference between treatments regarding risk of requiring renal replacement therapy was proven: levosimendan + dobutamine versus milrinone + dobutamine: RR 0.33 (95% CI 0.01 to 7.80) (Shah 2013), and levosimendan + milrinone + dopamine versus milrinone + dopamine: RR 0.97 (95% CI 0.27 to 3.54) (Ricci 2012).
Pairwise analysis revealed no evidence of a difference in the incidence of requirement for renal replacement therapy between levosimendan and milrinone (RR 1.17, 95% CI 0.18 to 7.69; P = 0.87; 2 studies, 106 participants; Analysis 1.11).
1.11. Analysis.

Comparison 1: Pairwise analysis: levosimendan versus milrinone, Outcome 11: Renal failure requiring replacement therapy
Discussion
Summary of main results
Using the above search strategy, we identified 13 studies of prophylactic inotropes for the prevention of LCOS and mortality in paediatric patients undergoing surgery for congenital heart disease. There were a total of 937 participants in these studies. The studies were heterogeneous regarding study populations, interventions and co‐interventions, and reported outcomes. Grading of the evidence according to the CINeMA framework showed mostly moderate‐certainty evidence.
All‐cause mortality within 30 days
Regarding mortality, levosimendan was found to likely result in a large reduction in mortality compared to placebo, whereas milrinone may result in no difference. Ranking of treatments resulted in levosimendan with the highest, milrinone with the second lowest, and placebo with the lowest rank. In a single, small study, the addition of levosimendan to both milrinone and dopamine may result in a large reduction in mortality, compared to a milrinone and dopamine combination. Another single, small study showed that the addition of levosimendan to dobutamine may result in a large reduction in mortality, compared to the addition of milrinone to dobutamine. Overall, the evaluation of mortality is impeded by small patient numbers in the studies reporting this rare event.
Time to death
For the outcome time to death, there were insufficient data to perform a pooled analysis. Survival curves were provided in one study, which did not show a difference between levosimendan and placebo.
Low cardiac output syndrome
For our primary outcome of LCOS, the network evidence suggests that levosimendan results in a large reduction, and that milrinone (standard dosing) likely results in a large reduction in the incidence of LCOS compared to placebo. Milrinone low‐dose may reduce the incidence of LCOS compared to placebo as well, but the result was less precise, and the study using it had a high risk of bias. Ranking showed that levosimendan, followed by standard‐dose milrinone, is more preferable for prevention of LCOS than placebo. The incidence of LCOS may be greatly reduced when levosimendan is added to a combination of milrinone and dopamine in neonates, according to one separate study.
Secondary outcomes
For the secondary outcomes of length of intensive care stay and length of hospital stay, there was little to no evidence of a difference based on the intervention used.
The duration of mechanical ventilation was likely increased by milrinone and likely slightly increased by levosimendan, compared to placebo, while dobutamine may not result in any difference compared to placebo. In a single, small study, the addition of levosimendan to both milrinone and dopamine may slightly increase the duration of mechanical ventilation, compared to a milrinone and dopamine combination. Another single, small study showed that the addition of levosimendan to dobutamine likely reduces the duration of mechanical ventilation, compared to the addition of milrinone to dobutamine. There was a wide variation in the length of intensive care stay, hospital stay, and mechanical ventilation between the studies, which may be attributable to differences in age groups of patients and local settings.
Inotrope scores were defined quite differently and, thus, the value of the pooled analysis showing that levosimendan decreases the inotrope score when compared to placebo is questionable.
There was no evidence of a difference in the number of patients requiring mechanical circulatory support (ECMO or VAD) between levosimendan, milrinone, or placebo, or for a combination of levosimendan plus dobutamine versus a combination of milrinone versus dobutamine.
Adverse events were analysed separately and as a combined outcome. There seemed to be a higher risk of arrhythmia with standard‐dose milrinone compared to placebo. The incidence of hypotension (if comparable at all given the different definitions of this outcome) was not different between mono‐drug interventions and placebo. The same was true for thrombocytopaenia. No differences were found regarding the risk of liver enzyme elevation. Hypokalaemia seemed to be more common in patients treated with levosimendan compared to placebo. Left ventricular dysfunction, as seen on echocardiography, was not different between milrinone, levosimendan, or dobutamine. Regarding renal function, different studies reported different outcomes, such that we decided to use 'requirement for renal replacement therapy' as an adverse event, which was the most comparable outcome between studies. Dobutamine may decrease, while levosimendan and milrinone may increase the incidence of requiring renal replacement therapy. Taken together, the incidence of adverse events is likely to be increased with levosimendan or milrinone, and may be increased with dobutamine, compared to placebo, while adverse events may be decreased with low‐dose milrinone, compared to placebo. When used in addition to dobutamine, levosimendan likely results in a large reduction in adverse events, compared to milrinone in addition to dobutamine. The addition of levosimendan to a combination of milrinone and dopamine likely results in no difference in adverse events.
Overall completeness and applicability of evidence
The evidence compiled by this network meta‐analysis provides some estimates of the effects of different inotropes used for preventing LCOS and mortality in paediatric patients undergoing surgery for congenital heart disease. Through search updates, we have found additional studies, which are awaiting classification; therefore, this review is a high priority for updating. In many instances, there were only one or two studies comparing the same interventions. For most outcomes, we found disconnected subnetworks with few possibilities to compute indirect evidence in addition to direct evidence, which was always restricted to one greater subnetwork. This may be the reason why there was usually no inconsistency detected. Patient numbers within the largest subnetworks were greater than in any of the individual trials, but still low in comparison to similar meta‐analyses for adult populations (Weber 2019). The risk of bias in the included studies was variable, with seven studies assessed at low risk of bias in all categories, and five studies with high risk of bias in more than one category (Figure 10). Finally, our review only addresses prophylactic use of inotropes and is not intended to inform treatment decisions when LCOS is already present.
Participants
Populations differed between studies, sometimes including both neonates and older infants or children, and sometimes including patients with both uni‐ and biventricular hearts. We were unable to separate treatment effects based on population subgroups within studies, and therefore also between studies. All studies were rather small, with fewer than 100 participants in each treatment arm. We also found a limited number of studies, and many of them did not report on all our pre‐specified outcomes. There was heterogeneity in study populations (newborns, infants, and older children, albeit no adults), interventions (e.g. dosing, timing of administration), outcome definitions (e.g. for LCOS or hypotension), and outcome reporting. Unfortunately, age‐based subgroup analyses were not possible, because none of the studies that included more than one age group reported their outcomes separately by age. As neonates are a population at higher risk of LCOS and mortality after congenital heart surgery, inclusion of neonates in unknown numbers into treatment groups impairs the comparability of outcomes regarding LCOS and mortality. The other effect modifier that we had planned to evaluate in a subgroup analysis was severity of the underlying heart defect, but this too was not possible.
Interventions
Interventions were administered in different dosages or infusion rates. In addition, concomitant interventions often included other inotropes as needed, but without detailed information beyond inotrope scores in some of the studies. Therefore, it was not possible to use this information in the networks for other outcomes.
Co‐interventions and comparators
Concomitant use of other inotropes (catecholamines) in most studies introduces uncertainty into the evidence. From a trial design standpoint, it is necessary to allow catecholamine use as needed in patient care, as almost all patients require inotropes, but in various amounts. Restricting their use would lead to large percentages of participants not completing the study per protocol. However, since the exact amounts of additional catecholamines are not reported on a case‐by‐case basis, their relationship with all the outcomes of a certain study is unclear. However, only two of our included studies had a catecholamine (dobutamine) as a separate study drug (Cavigelli‐Brunner 2018; Ebade 2013). We used a node 'placebo', but none for 'no medication', because this was not applicable.
Outcomes
There was considerable discrepancy in the definitions or length of follow‐up for outcomes, which affected even our main outcomes of all‐cause mortality within 30 days, time to death within three months, and LCOS. Most studies only reported sparse data about adverse events and, sometimes, it may have been difficult to judge if a patient experienced a feature of the underlying disease and surgery, or if a medication side effect was observed (hypotension, reduced LVEF). We treated renal failure as an adverse event, but this event might, in turn, have implications for the effects of drugs by prolonging their clearance, thus increasing both desired and undesired drug effects.
Quality of the evidence
There were many downgrades in at least one domain of the CINeMA confidence ratings (within‐study bias, reporting bias, indirectness, imprecision, heterogeneity, and incoherence), mostly attributable to imprecision, heterogeneity, or within‐study bias (see summary of findings tables for details). For mortality, the certainty of the evidence was moderate, downgraded for imprecision. For the outcome LCOS, the certainty of the evidence ranged from low to high, with reasons for downgrading of within‐study bias, heterogeneity, and imprecision. The length of intensive care stay judgements of the certainty of the evidence were moderate or low, with downgrades due to imprecision and heterogeneity. For length of hospital stay, we judged the certainty of the evidence to be moderate, downgraded due to imprecision. Results for duration of mechanical ventilation were affected by imprecision, which caused the certainty of the evidence to be moderate. The certainty of the evidence for the combined adverse outcomes ranged from moderate to low, due to within‐study bias and imprecision.
Potential biases in the review process
We repeated the electronic database search eight months after the original search, which then also comprised the international clinical trial registries (May 2021). An updated search was conducted in December 2023 for CENTRAL and in April 2024 for the other databases. We attempted to obtain unpublished data by contacting study authors and received additional information from several of them. However, not all of our outcomes had been among the studies' outcomes, and not all of the data we would have needed were still available. Since only randomised trials have been included and the number and selection of adverse events reported in these studies was heterogeneous, this review may not have addressed all important side effects, especially as pertains to longer follow‐up periods.
Agreements and disagreements with other studies or reviews
This systematic review and network meta‐analysis is an update and extension of our previous reviews about milrinone (Burkhardt 2015) and levosimendan (Hummel 2017) for the prevention of LCOS and mortality in paediatric patients undergoing surgery for congenital heart disease. In these former reviews, we had found only low‐certainty evidence that milrinone reduces the incidence of LCOS, and there was insufficient evidence about whether levosimendan reduces the incidence of LCOS or whether either of the two medications reduces mortality. This network meta‐analysis now suggests that levosimendan or milrinone may reduce the risk for LCOS in paediatric patients undergoing surgery for congenital heart disease compared to placebo.
We are aware of two other systematic reviews and meta‐analyses of RCTs evaluating the effects of levosimendan in paediatric patients with cardiac dysfunction. In the article by Silvetti et al (Silvetti 2022), five RCTs are described, which are also included here. Their primary outcomes included haemodynamic and laboratory values, which we did not assess. There was no evidence of an effect of levosimendan compared to placebo on the secondary outcomes length of ICU stay, hospital stay, and mortality in their analysis. The systematic review and meta‐analysis by Lapere et al examined the perioperative use of levosimendan compared to placebo, milrinone, or dobutamine in children undergoing heart surgery (Lapere 2022). They found nine RCTs, which are also included here, with evidence of a reduction of LCOS in the treatment groups who received levosimendan, but not of mortality.
Authors' conclusions
Implications for practice.
There is currently some evidence for the prophylactic administration of inotropes for the prevention of low cardiac output syndrome (LCOS) or mortality in paediatric patients undergoing surgery for congenital heart disease. Levosimendan alone, or added to other regimens, likely results in a large reduction in mortality compared to placebo, and the incidence of LCOS is likely reduced with levosimendan or with milrinone, but the existing trials are heterogeneous regarding population, dosing, and outcome reporting. There may not be any difference in the length of intensive care unit (ICU) stay or hospital stay (although it may be shorter with dobutamine compared to placebo), while there are conflicting results regarding the influence of prophylactic inotropes on the duration of mechanical ventilation, which is very dependent on patient population and clinical setting. The incidence of any adverse events may be increased with levosimendan, standard‐dose milrinone, or dobutamine and may be decreased with low‐dose milrinone compared to placebo, but there are conflicting data, with small studies reporting either likely a much lower incidence with addition of levosimendan as opposed to milrinone or dobutamine, or likely no benefit of additional levosimendan to a regimen of milrinone and dopamine. There seems to be an elevated risk of arrhythmia with milrinone and of hypokalaemia with levosimendan; however, these results have to be regarded cautiously, since several studies did not report on these outcomes at all.
Implications for research.
The effect of inotropes in preventing LCOS and mortality should be further studied, ideally in multicentre trials that allow the inclusion of larger numbers of patients, and with detailed reporting of patient subgroups (for example, neonates versus (infants versus) older children, univentricular versus biventricular physiology). Concomitantly used catecholamines should also be assessed and reported consistently. Uniform definitions of LCOS and follow‐up periods at least until hospital discharge or ‐ better ‐ of 30 days and beyond would be relevant to obtain comparable and meaningful outcome data. Adverse events should be followed up comprehensively for the longest possible duration.
History
Protocol first published: Issue 8, 2020
Notes
This review will supersede the two previous systematic reviews 'Prophylactic milrinone for the prevention of low cardiac output syndrome and mortality in children undergoing surgery for congenital heart disease' (Burkhardt 2015) and 'Prophylactic levosimendan for the prevention of low cardiac output syndrome and mortality in paediatric patients undergoing surgery for congenital heart disease' (Hummel 2017).
Acknowledgements
We thank Cochrane Heart for their contributions to the review process, including:
Nicole Martin, Managing Editor, Cochrane Heart; and
Charlene Bridges, Information Specialist, Cochrane Heart.
We thank Martina Gosteli, Information Specialist, University of Zurich, Switzerland for her help with updating the literature search.
We thank all study authors who provided additional information about their studies upon request.
Editorial and peer reviewer contributions
Cochrane Heart supported the authors in the development of this intervention review.
The following people conducted the editorial process for this article.
Sign‐off Editor (final editorial decision): Cindy Farquhar, University of Auckland, Auckland;
Managing Editor (selected peer reviewers, provided editorial guidance to authors, edited the article): Ghazaleh Aali, Cochrane Heart; Joanne Duffield, Cochrane Central Editorial Service;
Editorial Assistant (conducted editorial policy checks, collated peer reviewer comments and supported the editorial team): Lisa Wydrzynski, Cochrane Central Editorial Service;
Copy Editor (copy editing and production): Jenny Bellorini, Cochrane Central Production Service;
Peer reviewers (provided comments and recommended an editorial decision): JL Guerrero Orriach, Institute of Biomedical Research in Malaga, Malaga, Spain; Department of Anesthesiology, Virgen de la Victoria University Hospital, Malaga, Spain; and Department of Pharmacology and Pediatrics, School of Medicine, University of Malaga, Malaga, Spain (clinical review); Jessica D'Urbano (consumer review), Sofia Tsokani, Cochrane MSU (statistical review); Andrew Bäck, Statistical Editor, Cochrane Methods Support Unit (statistical review); Charlene Bridges, Cochrane Heart (search review); and Jo Platt, Central Editorial Information Specialist (search review). Two additional peer reviewers provided clinical peer review but chose not to be publicly acknowledged.
Appendices
Appendix 1. Glossary
Blood lactate: lactic acid level in the blood, which is increased when the organs of the body receive insufficient amounts of oxygen
Central venous oxygen saturation: a measure in blood of a central vein of how much oxygen the organs have extracted from the blood
Chronotropic: increasing heart rate
CI: cardiac index, a measure of how much blood the heart can pump per minute, based on the patient's body surface area
CPR: cardiopulmonary resuscitation
Cyclic adenosine monophosphate: a messenger molecule inside cells
Doppler echocardiography: ultrasound of the heart with examination of blood flow
Dromotropic: speeding up electrical signal transmission between the heart muscle cells
ECMO: extra corporeal life support
Geometry of NMA (network meta‐analysis) network: relationships between interventions consisting of direct and indirect comparisons
Inodilatory: inotropic and vasodilatory, that is increasing the pumping force of the heart and making blood vessels wider
Inotropic: increasing the contractile force of the heart muscle cells
Inovasopressor: inotrope and vasopressor, that is increasing the pumping force of the heart and making blood vessels narrower
Lusitropic: improving relaxation of heart muscle cells
Metabolic acidosis: excessive acidity of the blood not attributable to causes of breathing
Mono: consisting of one component only
Myocardial: pertaining to the heart muscle
Parallel arm trial: a clinical study with at least two groups of patients, where each group receives another treatment
Peripheral perfusion: blood supply to the body periphery
Phosphodiesterase type 3: an enzyme
Phosphorylation of protein kinases: a molecular mechanism to regulate activity of protein kinase enzymes
Prophylactic: preventive
Troponin C: part of the contractile apparatus in heart muscle cells
Vasodilatory: making blood vessels wider
Vasopressor: making blood vessels narrower
Appendix 2. Search strategies
Databases
CENTRAL
#1 MeSH descriptor: [Catecholamines] explode all trees #2 Catecholamine #3 MeSH descriptor: [Sympathomimetics] explode all trees #4 Sympathomimetic* #5 Dopamine #6 Dobutamine #7 Dopexamine #8 Methylnoradrenalin* #9 Adrenalin* #10 Suprarenin #11 Epinephrine #12 Noradrenalin* #13 Norepinephrine #14 MeSH descriptor: [Phosphodiesterase Inhibitors] explode all trees #15 Phosphodiesterase inhibitor* #16 MeSH descriptor: [Milrinone] this term only #17 Milrinon* #18 Calcium sensitizer* #19 Calcium sensitiser* #20 Levosimendan #21 levosimedan #22 "OR‐1896" #23 {OR #1‐#22} #24 MeSH descriptor: [Heart Defects, Congenital] explode all trees #25 MeSH descriptor: [Heart Diseases] explode all trees and with qualifier(s): [congenital ‐ CN] #26 (heart NEAR/2 (defect* or abnormal* or malform*)) #27 (congenital NEAR/2 (heart or cardiac or cardio*)) #28 (aort* NEAR/3 coarctation*) #29 ((heart* or cor) NEAR/2 triatria*) #30 (subdivided NEAR/3 atrium*) #31 coronary vessel anomal* #32 criss‐cross heart* #33 crisscross heart* #34 criss cross heart* #35 (paten* NEAR/2 ductus NEAR/2 arteriosus) #36 (ebstein* NEAR/2 (anomal* or malform*)) #37 ((heart or cardiac) NEAR/2 septal defect*) #38 hypoplastic left heart syndrome #39 marfan* syndrome #40 (fallot* NEAR/2 tetralogy)~ #41 (transpos* NEAR/4 (arter* or vessel*)) #42 (tricuspid NEAR/2 atresia) #43 (fallot* NEAR/2 trilogy) #44 univentricular heart #45 arterial switch #46 norwood* #47 septal defect #48 sinus venosus defect #49 (anomalous pulmonary venous NEAR/3 (connection or return or drainage or total or partial)) #50 (PAPVC or PAPVR or APVC or TAPVC or APVD) #51 Scimitar #52 persistent ductus arteriosus #53 aortopulmonary window #54 interrupted aortic arch #55 (truncus arteriosus communis or common arterial trunk) #56 double aortic arch #57 congenital aortic stenosis #58 congenital pulmonary stenosis #59 ventricular outflow tract obstruction #60 absent pulmonary valve #61 cor triatriatum #62 double outlet #63 double inlet #64 double chambered right ventricle #65 single ventricle #66 {OR #24‐#65} #67 #23 AND #66
MEDLINE (Ovid)
1 exp Catecholamines/ 2 Catecholamine.tw. 3 exp Sympathomimetics/ 4 Sympathomimetic*.tw. 5 Dopamine.tw. 6 Dobutamine.tw. 7 Dopexamine.tw. 8 Methylnoradrenalin*.tw. 9 Adrenalin*.tw. 10 Suprarenin.tw. 11 Epinephrine.tw. 12 Noradrenalin*.tw. 13 Norepinephrine.tw. 14 exp Phosphodiesterase Inhibitors/ 15 Phosphodiesterase inhibitor*.tw. 16 Milrinone/ 17 Milrinon*.tw. 18 Calcium sensitizer*.tw. 19 Calcium sensitiser*.tw. 20 Levosimendan.tw. 21 levosimedan.tw. 22 OR‐1896.tw. 23 or/1‐22 24 exp Heart Defects, Congenital/ 25 exp Heart Diseases/cn [Congenital] 26 (heart adj2 (defect* or abnormal* or malform*)).tw. 27 (congenital adj2 (heart or cardiac or cardio*)).tw. 28 (aort* adj3 coarctation*).tw. 29 ((heart* or cor) adj2 triatria*).tw. 30 (subdivided adj3 atrium*).tw. 31 coronary vessel anomal*.tw. 32 criss‐cross heart*.tw. 33 crisscross heart*.tw. 34 criss cross heart*.tw. 35 (paten* adj2 ductus adj2 arteriosus).tw. 36 (ebstein* adj2 (anomal* or malform*)).tw. 37 ((heart or cardiac) adj2 septal defect*).tw. 38 hypoplastic left heart syndrome.tw. 39 marfan* syndrome.tw. 40 (fallot* adj2 tetralogy).tw. 41 (transpos* adj4 (arter* or vessel*)).tw. 42 (tricuspid adj2 atresia).tw. 43 (fallot* adj2 trilogy).tw. 44 univentricular heart.tw. 45 arterial switch.tw. 46 norwood*.tw. 47 septal defect.tw. 48 sinus venosus defect.tw. 49 (anomalous pulmonary venous adj3 (connection or return or drainage or total or partial)).tw. 50 (PAPVC or PAPVR or APVC or TAPVC or APVD).tw. 51 Scimitar.tw. 52 persistent ductus arteriosus.tw. 53 aortopulmonary window.tw. 54 interrupted aortic arch.tw. 55 (truncus arteriosus communis or common arterial trunk).tw. 56 double aortic arch.tw. 57 congenital aortic stenosis.tw. 58 congenital pulmonary stenosis.tw. 59 ventricular outflow tract obstruction.tw. 60 absent pulmonary valve.tw. 61 cor triatriatum.tw. 62 double outlet.tw. 63 double inlet.tw. 64 double chambered right ventricle.tw. 65 single ventricle.tw. 66 or/24‐65 67 23 and 66 68 randomized controlled trial.pt. 69 controlled clinical trial.pt. 70 randomized.ab. 71 placebo.ab. 72 clinical trials as topic.sh. 73 randomly.ab. 74 trial.ti. 75 68 or 69 or 70 or 71 or 72 or 73 or 74 76 exp animals/ not humans.sh. 77 75 not 76 78 67 and 77
EMBASE (Ovid)
1 exp catecholamine/ 2 Catecholamine.tw. 3 exp adrenergic receptor stimulating agent/ 4 Sympathomimetic*.tw. 5 Dopamine.tw. 6 Dobutamine.tw. 7 Dopexamine.tw. 8 Methylnoradrenalin*.tw. 9 Adrenalin*.tw. 10 Suprarenin.tw. 11 Epinephrine.tw. 12 Noradrenalin*.tw. 13 Norepinephrine.tw. 14 exp phosphodiesterase inhibitor/ 15 Phosphodiesterase inhibitor*.tw. 16 milrinone/ 17 Milrinon*.tw. 18 Calcium sensitizer*.tw. 19 Calcium sensitiser*.tw. 20 Levosimendan.tw. 21 levosimedan.tw. 22 OR‐1896.tw. 23 or/1‐22 24 exp congenital heart malformation/ 25 exp heart disease/cn [Congenital Disorder] 26 (heart adj2 (defect* or abnormal* or malform*)).tw. 27 (congenital adj2 (heart or cardiac or cardio*)).tw. 28 (aort* adj3 coarctation*).tw. 29 ((heart* or cor) adj2 triatria*).tw. 30 (subdivided adj3 atrium*).tw. 31 coronary vessel anomal*.tw. 32 criss‐cross heart*.tw. 33 crisscross heart*.tw. 34 criss cross heart*.tw. 35 (paten* adj2 ductus adj2 arteriosus).tw. 36 (ebstein* adj2 (anomal* or malform*)).tw. 37 ((heart or cardiac) adj2 septal defect*).tw. 38 hypoplastic left heart syndrome.tw. 39 marfan* syndrome.tw. 40 (fallot* adj2 tetralogy).tw. 41 (transpos* adj4 (arter* or vessel*)).tw. 42 (tricuspid adj2 atresia).tw. 43 (fallot* adj2 trilogy).tw. 44 univentricular heart.tw. 45 arterial switch.tw. 46 norwood*.tw. 47 septal defect.tw. 48 sinus venosus defect.tw. 49 (anomalous pulmonary venous adj3 (connection or return or drainage or total or partial)).tw. 50 (PAPVC or PAPVR or APVC or TAPVC or APVD).tw. 51 Scimitar.tw. 52 persistent ductus arteriosus.tw. 53 aortopulmonary window.tw. 54 interrupted aortic arch.tw. 55 (truncus arteriosus communis or common arterial trunk).tw. 56 double aortic arch.tw. 57 congenital aortic stenosis.tw. 58 congenital pulmonary stenosis.tw. 59 ventricular outflow tract obstruction.tw. 60 absent pulmonary valve.tw. 61 cor triatriatum.tw. 62 double outlet.tw. 63 double inlet.tw. 64 double chambered right ventricle.tw. 65 single ventricle.tw. 66 or/24‐65 67 23 and 66 68 random$.tw. 69 factorial$.tw. 70 crossover$.tw. 71 cross over$.tw. 72 cross‐over$.tw. 73 placebo$.tw. 74 (doubl$ adj blind$).tw. 75 (singl$ adj blind$).tw. 76 assign$.tw. 77 allocat$.tw. 78 volunteer$.tw. 79 crossover procedure/ 80 double blind procedure/ 81 randomized controlled trial/ 82 single blind procedure/ 83 68 or 69 or 70 or 71 or 72 or 73 or 74 or 75 or 76 or 77 or 78 or 79 or 80 or 81 or 82 84 (animal/ or nonhuman/) not human/ 85 83 not 84 86 67 and 85 87 limit 86 to embase
EMBASE.com (update search April 2024)
#89 #87 AND [embase]/lim NOT ([embase]/lim AND [medline]/lim)
#88 #87 AND [embase]/lim
#87 #67 AND #85 AND [01‐05‐2021]/sd
#86 #67 AND #85
#85 83 NOT ([animals]/lim NOT [humans]/lim)
#84 83 NOT ([animals]/lim NOT [humans]/lim)
#83 #68 OR #69 OR #70 OR #71 OR #72 OR #73 OR #74 OR #75 OR #76 OR #77 OR #78 OR #79 OR #80 OR #81 OR #82
#82 'single blind procedure'/de
#81 'randomized controlled trial'/de
#80 'double blind procedure'/de
#79 'crossover procedure'/de
#78 volunteer*:ti,ab
#77 allocat*:ti,ab
#76 assign*:ti,ab
#75 (singl* NEXT/1 blind*):ti,ab
#74 (doubl* NEXT/1 blind*):ti,ab
#73 placebo*:ti,ab
#72 'cross over*':ti,ab
#71 'cross over*':ti,ab
#70 crossover*:ti,ab
#69 factorial*:ti,ab
#68 random*:ti,ab
#67 #23 AND #66
#66 #24 OR #25 OR #26 OR #27 OR #28 OR #29 OR #30 OR #31 OR #32 OR #33 OR #34 OR #35 OR #36 OR #37 OR #38 OR #39 OR #40 OR #41 OR #42 OR #43 OR #44 OR #45 OR #46 OR #47 OR #48 OR #49 OR #50 OR #51 OR #52 OR #53 OR #54 OR #55 OR #56 OR #57 OR #58 OR #59 OR #60 OR #61 OR #62 OR #63 OR #64 OR #65
#65 'single ventricle':ti,ab
#64 'double chambered right ventricle':ti,ab
#63 'double inlet':ti,ab
#62 'double outlet':ti,ab
#61 'cor triatriatum':ti,ab
#60 'absent pulmonary valve':ti,ab
#59 'ventricular outflow tract obstruction':ti,ab
#58 'congenital pulmonary stenosis':ti,ab
#57 'congenital aortic stenosis':ti,ab
#56 'double aortic arch':ti,ab
#55 'truncus arteriosus communis':ti,ab OR 'common arterial trunk':ti,ab
#54 'interrupted aortic arch':ti,ab
#53 'aortopulmonary window':ti,ab
#52 'persistent ductus arteriosus':ti,ab
#51 scimitar:ti,ab
#50 papvc:ti,ab OR papvr:ti,ab OR apvc:ti,ab OR tapvc:ti,ab OR apvd:ti,ab
#49 ('anomalous pulmonary venous' NEAR/4 (connection OR return OR drainage OR total OR partial)):ti,ab
#48 'sinus venosus defect':ti,ab
#47 'septal defect':ti,ab
#46 norwood*:ti,ab
#45 'arterial switch':ti,ab
#44 'univentricular heart':ti,ab
#43 (fallot* NEAR/3 trilogy):ti,ab
#42 (tricuspid NEAR/3 atresia):ti,ab
#41 (transpos* NEAR/5 (arter* OR vessel*)):ti,ab
#40 (fallot* NEAR/3 tetralogy):ti,ab
#39 'marfan* syndrome':ti,ab
#38 'hypoplastic left heart syndrome':ti,ab
#37 ((heart OR cardiac) NEAR/3 'septal defect*'):ti,ab
#36 (ebstein* NEAR/3 (anomal* OR malform*)):ti,ab
#35 (paten* NEAR/3 ductus NEAR/3 arteriosus):ti,ab
#34 'criss cross heart*':ti,ab
#33 'crisscross heart*':ti,ab
#32 'criss‐cross heart*':ti,ab
#31 'coronary vessel anomal*':ti,ab
#30 (subdivided NEAR/4 atrium*):ti,ab
#29 ((heart* OR cor) NEAR/3 triatria*):ti,ab
#28 (aort* NEAR/4 coarctation*):ti,ab
#27 (congenital NEAR/3 (heart OR cardiac OR cardio*)):ti,ab
#26 (heart NEAR/3 (defect* OR abnormal* OR malform*)):ti,ab
#25 'heart disease'/exp/dm_cn
#24 'congenital heart malformation'/exp
#23 #1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7 OR #8 OR #9 OR #10 OR #11 OR #12 OR #13 OR #14 OR #15 OR #16 OR #17 OR #18 OR #19 OR #20 OR #21 OR #22
#22 'or 1896':ti,ab
#21 levosimedan:ti,ab
#20 levosimendan:ti,ab
#19 'calcium sensitiser*':ti,ab
#18 'calcium sensitizer*':ti,ab
#17 milrinon*:ti,ab
#16 'milrinone'/de
#15 'phosphodiesterase inhibitor*':ti,ab
#14 'phosphodiesterase inhibitor'/exp
#13 norepinephrine:ti,ab
#12 noradrenalin*:ti,ab
#11 epinephrine:ti,ab
#10 suprarenin:ti,ab
#9 adrenalin*:ti,ab
#8 methylnoradrenalin*:ti,ab
#7 dopexamine:ti,ab
#6 dobutamine:ti,ab
#5 dopamine:ti,ab
#4 sympathomimetic*:ti,ab
#3 'adrenergic receptor stimulating agent'/exp
#2 catecholamine:ti,ab
#1 'catecholamine'/exp
CPCI‐S
# 39 #38 AND #37
# 38 TS=(random* or blind* or allocat* or assign* or trial* or placebo* or crossover* or cross‐over*)
# 37 #36 AND #9
# 36 #35 OR #34 OR #33 OR #32 OR #31 OR #30 OR #29 OR #28 OR #27 OR #26 OR #25 OR #24 OR #23 OR #22 OR #21 OR #20 OR #19 OR #18 OR #17 OR #16 OR #15 OR #14 OR #13 OR #12 OR #11 OR #10
# 35 TS=(cor triatriatum or double outlet or double inlet or double chambered right ventricle or single ventricle)
# 34 TS=(double aortic arch or congenital aortic stenosis or congenital pulmonary stenosis or ventricular outflow tract obstruction or absent pulmonary valve)
# 33 TS=(truncus arteriosus communis or common arterial trunk)
# 32 TS=interrupted aortic arch
# 31 TS=aortopulmonary window
# 30 TS=persistent ductus arteriosus
# 29 TS=Scimitar
# 28 TS=(PAPVC or PAPVR or APVC or TAPVC or APVD)
# 27 TS=(anomalous pulmonary venous NEAR/3 (connection or return or drainage or total or partial))
# 26 TS=(univentricular heart or arterial switch or norwood* or septal defect or sinus venosus defect)
# 25 TS=(fallot* NEAR/2 trilogy)
# 24 TS=(tricuspid NEAR/2 atresia)
# 23 TS=(transpos* NEAR/4 (arter* or vessel*))
# 22 TS=(fallot* NEAR/2 tetralogy)
# 21 TS=marfan* syndrome
# 20 TS=hypoplastic left heart syndrome
# 19 TS=((heart or cardiac) NEAR/2 septal defect*)
# 18 TS=(ebstein* NEAR/2 (anomal* or malform*))
# 17 TS=(paten* NEAR/2 ductus NEAR/2 arteriosus)
# 16 TS=(criss‐cross heart* or crisscross heart* or criss cross heart*)
# 15 TS=coronary vessel anomal*
# 14 TS=(subdivided NEAR/3 atrium*)
# 13 TS=((heart* or cor) NEAR/2 triatria*)
# 12 TS=(aort* NEAR/3 coarctation*)
# 11 TS=(congenital NEAR/2 (heart or cardiac or cardio*))
# 10 TS=(heart NEAR/2 (defect* or abnormal* or malform*))
# 9 #8 OR #7 OR #6 OR #5 OR #4 OR #3 OR #2 OR #1
# 8 TS=OR‐1896
# 7 TS=(Levosimendan or levosimedan)
# 6 TS=Calcium sensitiser*
# 5 TS=Calcium sensitizer*
# 4 TS=Milrinon*
# 3 TS=Phosphodiesterase inhibitor*
# 2 TS=(Sympathomimetic* or Dopamine or Dobutamine or Dopexamine or Methylnoradrenalin* or Adrenalin* or Suprarenin or Epinephrine or Noradrenalin* or Norepinephrine)
# 1 TS=Catecholamine
Trial register search strategies
ClinicalTrials.gov
Condition or disease: Congenital Heart Disease
Intervention: Drug
Study Type: Interventional studies (Clinical trials)
WHO ICTRP
Condition: Congenital Heart Disease
Intervention: Drug
Appendix 3. Additional figures
11.
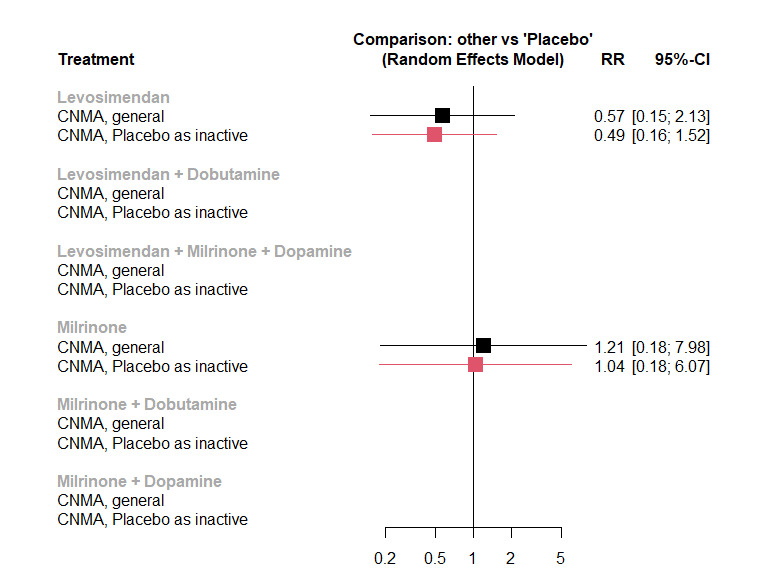
Component network meta‐analysis for outcome 'All‐cause mortality within 30 days'. General CNMA assumes additive effects of interventions, and CNMA with placebo as inactive assumes that there is no effect of placebo. There are no CNMA results for some combinations of interventions, because these occur only outside of the largest subnetwork. The combinations containing dobutamine or dopamine do not yield separate results, because those are part of both arms in their respective small subnetwork.
Abbreviation(s) CI: confidence interval; CNMA: component network meta‐analysis; RR: risk ratio; vs: versus.
12.
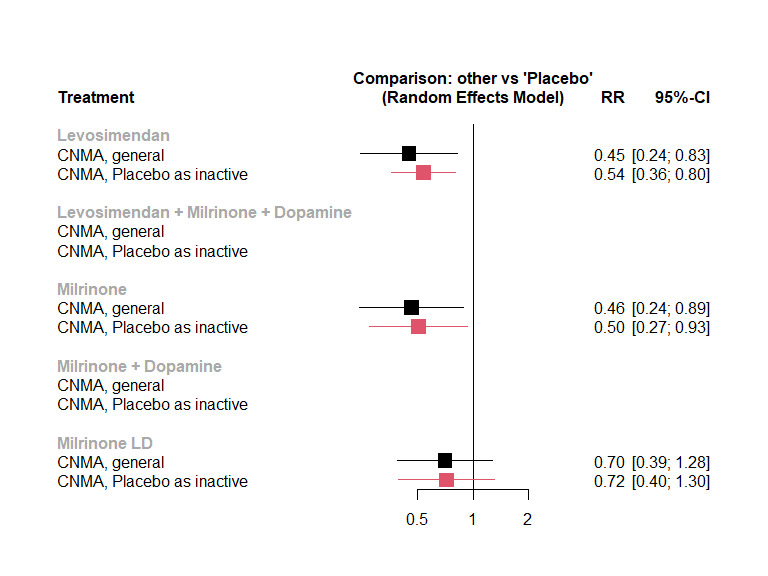
Component network meta‐analysis for outcome 'LCOS'. General CNMA assumes additive effects of interventions, and CNMA with placebo as inactive assumes that there is no effect of placebo. There are no CNMA results for some combinations of interventions, because these occur only outside of the largest subnetwork. The combinations containing dopamine do not yield separate results, because dopamine is part of both arms in the small subnetwork.
Abbreviation(s) CI: confidence interval; CNMA: component network meta‐analysis; LD: low dose; RR: risk ratio; vs: versus.
13.
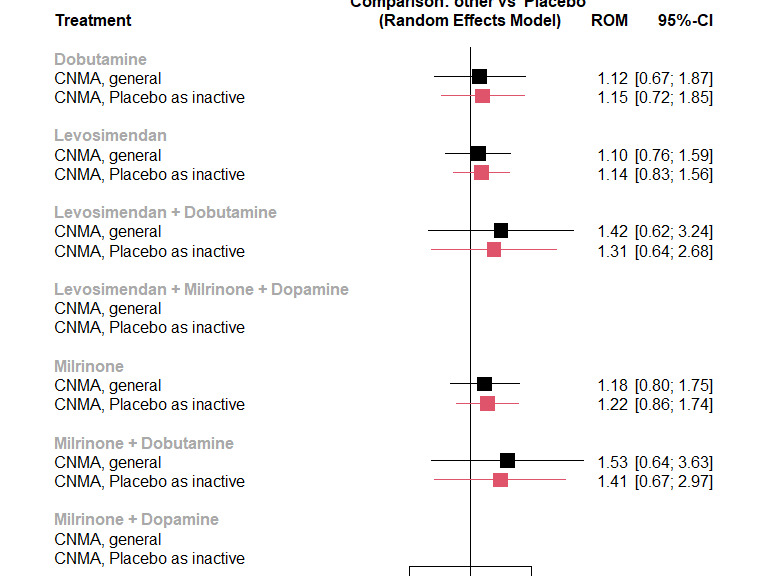
Component network meta‐analysis for outcome 'Length of intensive care stay'. General CNMA assumes additive effects of interventions, and CNMA with placebo as inactive assumes that there is no effect of placebo. There are no CNMA results for some combinations of interventions, because these occur only outside of the largest subnetwork. The combinations containing dopamine do not yield separate results, because dopamine is part of both arms in one of the small subnetworks.
Abbreviations CI: confidence interval; CNMA: component network meta‐analysis; ROM: ratio of means; vs: versus.
14.
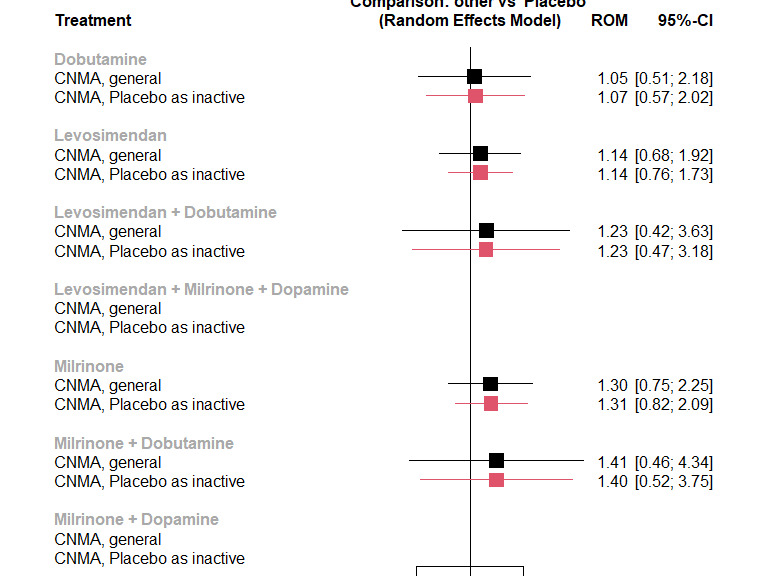
Component network meta‐analysis for outcome 'Duration of mechanical ventilation'. General CNMA assumes additive effects of interventions, and CNMA with placebo as inactive assumes that there is no effect of placebo. There are no CNMA results for some combinations of interventions, because these occur only outside of the largest subnetwork. The combinations containing dopamine do not yield separate results, because dopamine is part of both arms in one of the small subnetworks.
Abbreviation(s) CI: confidence interval; CNMA: component network meta‐analysis; ROM: ratio of means; vs: versus.
15.
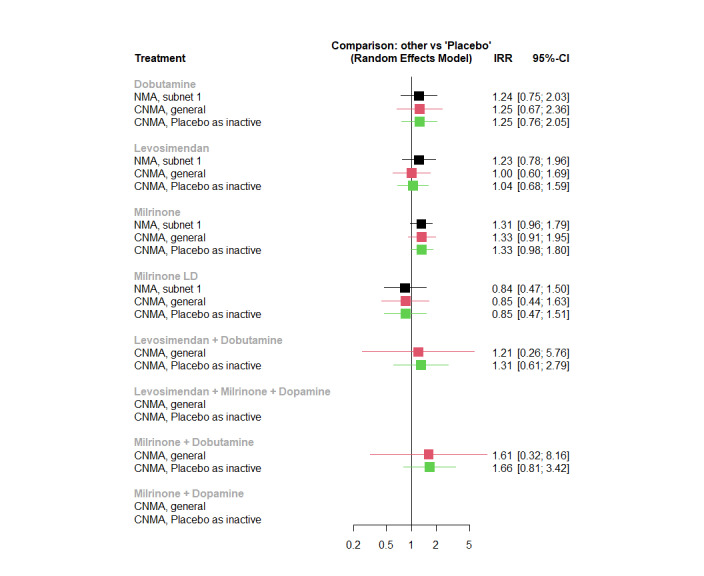
Component network meta‐analysis for outcome 'All adverse events'. Subnet 1 is the largest subnetwork, general CNMA assumes additive effects of interventions, and CNMA with placebo as inactive assumes that there is no effect of placebo. There are no NMA results for the last four combinations of interventions, because these occur only outside of the largest subnetwork. The combinations containing dopamine do not yield separate results, because dopamine is part of both arms in one of the small subnetworks.
Abbreviation(s) CI: confidence interval; CNMA: component network meta‐analysis; IRR: incidence rate ratio; LD: low dose; NMA: network meta‐analysis; vs: versus.
16.
Pairwise meta‐analysis for outcome 'All adverse events' comparing levosimendan and placebo. Left side of the forest plot: favours levosimendan; right side of the forest plot: favours placebo.
Abbreviations CI: confidence interval; IRR: incidence rate ratio; MH: Mantel‐Haenszel.
17.
Pairwise meta‐analysis for outcome 'All adverse events' comparing milrinone and placebo. Left side of the forest plot: favours milrinone; right side of the forest plot: favours placebo.
Abbreviations CI: confidence interval; IRR: incidence rate ratio; MH: Mantel‐Haenszel.
Appendix 4. Supplementary material: analyses for 'all‐cause mortality within 30 days'
Subnet 1
Results (random‐effects model): Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of observations: o = 277; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 |
| Momeni 2011 | Levosimendan | Milrinone | 0.5880 | 0.1052 | 3.2875 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.5880 | 0.1052 | 3.2875 |
| Wang 2019 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5721 | 0.1535 | 2.1319 | ‐0.83 | 0.4054 |
| Milrinone | 0.9730 | 0.1115 | 8.4901 | ‐0.02 | 0.9802 |
| Placebo | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.3333 | 0.0371 | 2.9910 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.3333 | 0.0371 | 2.9910 | ‐0.98 | 0.3264 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 3
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 63; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Ricci 2012 | Levosimendan + Milrinone + Dopamine | Milrinone + Dopamine | 0.3229 | 0.0355 | 2.9395 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Milrinone + Dopamine | 0.3229 | 0.0355 | 2.9395 | ‐1.00 | 0.3158 |
| Milrinone + Dopamine | reference | reference | reference | reference | reference |
Subnet 1: Inverse variance method with increments for zero studies
Results (random‐effects model): Number of studies: k = 9; Number of pairwise comparisons: m = 9; Number of observations: o = 557; Number of treatments: n = 4; Number of designs: d = 5
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.5826 | 0.1663 | 2.0410 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.8059 | 0.0478 | 13.5774 |
| Costello 2014 | Milrinone | Placebo | 0.8294 | 0.1474 | 4.6671 |
| Ebade 2013 | Dobutamine | Levosimendan | 1.1473 | 0.0681 | 19.3295 |
| Lechner 2012 | Levosimendan | Milrinone | 0.7024 | 0.1852 | 2.6640 |
| Momeni 2011 | Levosimendan | Milrinone | 0.7024 | 0.1852 | 2.6640 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.7024 | 0.1852 | 2.6640 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.7024 | 0.1852 | 2.6640 |
| Wang 2019 | Levosimendan | Placebo | 0.5826 | 0.1663 | 2.0410 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6684 | 0.0313 | 14.2609 | ‐0.26 | 0.7964 |
| Levosimendan | 0.5826 | 0.1663 | 2.0410 | ‐0.84 | 0.3983 |
| Milrinone | 0.8294 | 0.1474 | 4.6671 | ‐0.21 | 0.8320 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Additive model (random‐effects model): Number of studies: k = 6; Number of pairwise comparisons: m = 6; Number of treatments: n = 7; Number of active components: c = 3; Number of designs: d = 1; Number of subnetworks: s = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 | ‐0.83 | 0.4054 |
| Momeni 2011 | Levosimendan | Milrinone | 0.4737 | 0.1223 | 1.8349 | ‐1.08 | 0.2795 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.4737 | 0.1223 | 1.8349 | ‐1.08 | 0.2795 |
| Ricci 2012 | Levosimendan + Milrinone + Dopamine | Milrinone + Dopamine | 0.3229 | 0.0355 | 2.9395 | ‐1.00 | 0.3158 |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.4737 | 0.1223 | 1.8349 | ‐1.08 | 0.2795 |
| Wang 2019 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 | ‐0.83 | 0.4054 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5721 | 0.1535 | 2.1319 | ‐0.83 | 0.4054 |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 1.2078 | 0.1828 | 7.9786 | 0.20 | 0.8446 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Placebo | reference | reference | reference | reference | reference |
Incremental effect for components:
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.3229 | 0.0355 | 2.9395 | ‐1.00 | 0.3158 |
| Milrinone | 0.6817 | 0.0511 | 9.0940 | ‐0.29 | 0.7719 |
| Placebo | 0.5644 | 0.0432 | 7.3800 | ‐0.44 | 0.6628 |
CNMA for disconnected network, placebo assumed as inactive
Additive model (random‐effects model): Number of studies: k = 6; Number of pairwise comparisons: m = 6; Number of treatments: n = 7; Number of active components: c = 2; Number of designs: d = 4; Number of subnetworks: s = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.4925 | 0.1591 | 1.5250 | ‐1.23 | 0.2194 |
| Momeni 2011 | Levosimendan | Milrinone | 0.4737 | 0.1223 | 1.8349 | ‐1.08 | 0.2795 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.4737 | 0.1223 | 1.8349 | ‐1.08 | 0.2795 |
| Ricci 2012 | Levosimendan + Milrinone + Dopamine | Milrinone + Dopamine | 0.4925 | 0.1591 | 1.5250 | ‐1.23 | 0.2194 |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.4737 | 0.1223 | 1.8349 | ‐1.08 | 0.2795 |
| Wang 2019 | Levosimendan | Placebo | 0.4925 | 0.1591 | 1.5250 | ‐1.23 | 0.2194 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.4925 | 0.1591 | 1.5250 | ‐1.23 | 0.2194 |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 1.0398 | 0.1782 | 6.0674 | 0.04 | 0.9654 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Placebo | reference | reference | reference | reference | reference |
Incremental effect for components:
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.4925 [; ] 0.219 | 0.1591 | 1.5250 | ‐1.23 | 0.2194 |
| Milrinone | 1.0398 | 0.1782 | 6.0674 | 0.04 | 0.9654 |
Subnet 1: Exclude Costello 2014
Note that the first analysis is not affected, as Costello 2014 is a double‐zero study and therefore excluded anyway
Results (random‐effects model): Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of observations: o = 277; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 |
| Momeni 2011 | Levosimendan | Milrinone | 0.5880 | 0.1052 | 3.2875 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.5880 | 0.1052 | 3.2875 |
| Wang 2019 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5721 | 0.1535 | 2.1319 | ‐0.83 | 0.4054 |
| Milrinone | 0.9730 | 0.1115 | 8.4901 | ‐0.02 | 0.9802 |
| Placebo | reference | reference | reference | reference | reference |
Subnet 1: Inverse variance method with increments for zero studies
Results (random‐effects model): Number of studies: k = 8; Number of pairwise comparisons: m = 8; Number of observations: o = 486; Number of treatments: n = 4; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.8143 | 0.0479 | 13.8472 |
| Ebade 2013 | Dobutamine | Levosimendan | 1.1353 | 0.0668 | 19.3063 |
| Lechner 2012 | Levosimendan | Milrinone | 0.7172 | 0.1752 | 2.9356 |
| Momeni 2011 | Levosimendan | Milrinone | 0.7172 | 0.1752 | 2.9356 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.7172 | 0.1752 | 2.9356 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.7172 | 0.1752 | 2.9356 |
| Wang 2019 | Levosimendan | Placebo | 0.5721 | 0.1535 | 2.1319 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6495 | 0.0286 | 14.7680 | ‐0.27 | 0.7866 |
| Levosimendan | 0.5721 | 0.1535 | 2.1319 | ‐0.83 | 0.4054 |
| Milrinone | 0.7976 | 0.1160 | 5.4834 | ‐0.23 | 0.8182 |
| Placebo | reference | reference | reference | reference | reference |
Exclude: Shah
As Shah builds its own network component, no alternative analysis is necessary.
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 5. Supplementary material: analyses for 'time to death'
'Time to death' was not reported in most studies, only in Wang 2019, where the survival curves show that the last death within 90 days occurred after 10 days in the levosimendan group (total 3/94 in Levo group – days 2, 3, 10; and 4/93 in Placebo group – days 1, 2, 4, 5).
Survival analysis
Placebo:
| TIme | Number at risk | Number of events | Survival | Standard error | 95% CI lower limit | 95% CI upper limit |
| 1 | 93 | 1 | 0.989 | 0.0107 | 0.969 | 1 |
| 2 | 92 | 1 | 0.978 | 0.0150 | 0.949 | 1 |
| 4 | 91 | 1 | 0.968 | 0.0183 | 0.932 | 1 |
| 5 | 90 | 1 | 0.957 | 0.0210 | 0.917 | 0.999 |
Levosimendan:
| TIme | Number at risk | Number of events | Survival | Standard error | 95% CI lower limit | 95% CI upper limit |
| 2 | 94 | 1 | 0.989 | 0.0106 | 0.969 | 1 |
| 3 | 93 | 1 | 0.979 | 0.0149 | 0.950 | 1 |
| 10 | 92 | 1 | 0.968 | 0.0181 | 0.933 | 0.999 |
Concordance: 0.539 (SE 0.094) Likelihood ratio test: 0.16 on 1 df, P = 0.7 Wald test: 0.16 on 1 df, P = 0.7 Score (log rank) test = 0.16 on 1 df, P = 0.7
Footnotes
Abbreviations CI: confidence interval; df: degrees of freedom.
Appendix 6. Supplementary material: analyses for 'low cardiac output syndrome'
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 63; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Ricci 2012 | Levosimendan + Milrinone + Dopamine | Milrinone + Dopamine | 0.6118 | 0.3610 | 1.0370 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Milrinone + Dopamine | 0.6118 | 0.3610 | 1.0370 | ‐1.83 | 0.0680 |
| Milrinone + Dopamine | reference | reference | reference | reference | reference |
Mantel‐Haenszel method
Results (random‐effects model): Number of studies: k = 4; Number of pairwise comparisons: m = 6; Number of observations: o = 474; Number of treatments: n = 4; Number of designs: d = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Hoffman 2003 | Milrinone | Milrinone LD | 0.6593 | 0.3057 | 1.4218 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.7029 | 0.3870 | 1.2765 |
| Hoffman 2003 | Milrinone | Placebo | 0.4634 | 0.2422 | 0.8864 |
| Jothinath 2021 | Levosimendan | Milrinone | 0.9661 | 0.4960 | 1.8817 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9661 | 0.4960 | 1.8817 |
| Wang 2019 | Levosimendan | Placebo | 0.4477 | 0.2429 | 0.8251 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.4477 | 0.2429 | 0.8251 | ‐2.58 | 0.0100 |
| Milrinone | 0.4634 | 0.2422 | 0.8864 | ‐2.32 | 0.0201 |
| Milrinone LD | 0.7029 | 0.3870 | 1.2765 | ‐1.16 | 0.2468 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies
Results (random‐effects model): Number of studies: k = 5; Number of pairwise comparisons: m = 7; Number of observations: o = 513; Number of treatments: n = 4; Number of designs: d = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Hoffman 2003 | Milrinone | Milrinone LD | 0.6586 | 0.3058 | 1.4181 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.4628 | 0.2427 | 0.8825 |
| Hoffman 2003 | Milrinone | Placebo | 0.7027 | 0.3870 | 1.2759 |
| Jothinath 2021 | Levosimendan | Milrinone | 0.9685 | 0.5021 | 1.8682 |
| Lechner 2012 | Levosimendan | Milrinone | 0.9685 | 0.5021 | 1.8682 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9685 | 0.5021 | 1.8682 |
| Wang 2019 | Levosimendan | Placebo | 0.4482 | 0.2437 | 0.8242 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.4482 | 0.2437 | 0.8242 | ‐2.58 | 0.0098 |
| Milrinone | 0.4628 | 0.2427 | 0.8825 | ‐2.34 | 0.0193 |
| Milrinone LD | 0.7027 | 0.3870 | 1.2759 | ‐1.16 | 0.2463 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 5; Number of pairwise comparisons: m = 7; Number of treatments: n = 6; Number of active components: c = 4; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.6118 | 0.3610 | 1.0370 | ‐1.83 | 0.0680 |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 0.6333 | 0.2706 | 1.4819 | ‐1.05 | 0.2923 |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone LD | 0.9606 | 0.3661 | 2.5208 | ‐0.08 | 0.9349 |
| Placebo | 1.3667 | 0.6095 | 3.0646 | 0.76 | 0.4484 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.6118 | 0.3610 | 1.0370 | ‐1.83 | 0.0680 |
| Milrinone | 0.6333 | 0.2706 | 1.4819 | ‐1.05 | 0.2923 |
| Milrinone LD | 0.9606 | 0.3661 | 2.5208 | ‐0.08 | 0.9349 |
| Placebo | 1.3667 | 0.6095 | 3.0646 | 0.76 | 0.4484 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 5; Number of pairwise comparisons: m = 7; Number of treatments: n = 6; Number of active components: c = 4; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5355 | 0.3591 | 0.7984 | ‐3.06 | 0.0022 |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 0.5039 | 0.2734 | 0.9286 | ‐2.20 | 0.0280 |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone LD | 0.7160 | 0.3950 | 1.2979 | ‐1.10 | 0.2710 |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.5355 | 0.3591 | 0.7984 | ‐3.06 | 0.0022 |
| Milrinone | 0.5039 | 0.2734 | 0.9286 | ‐2.20 | 0.0280 |
| Milrinone LD | 0.7160 | 0.3950 | 1.2979 | ‐1.10 | 0.2710 |
Exclude: Hoffman, Jothinath (both in set p12)
Number of studies: k = 6; Number of pairwise comparisons: m = 8; Number of treatments: n = 6; Number of designs: d = 4; Number of subnetworks: 2
Mantel‐Haenszel method
Results (random‐effects model): Number of studies: k = 2; Number of pairwise comparisons: m = 2; Number of observations: o = 207; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Pellicer 2013 | Levosimendan | Milrinone | 0.8182 | 0.2151 | 3.1122 |
| Wang 2019 | Levosimendan | Placebo | 0.5496 | 0.2681 | 1.1268 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5496 | 0.2681 | 1.1268 | ‐1.63 | 0.1023 |
| Milrinone | 0.6718 | 0.1474 | 3.0614 | ‐0.51 | 0.6072 |
| Placebo | NA | NA | NA | NA | NA |
Inverse variance method with increments for zero studies
Results (random‐effects model): Number of studies: k = 3; Number of pairwise comparisons: m = 3; Number of observations: o = 246; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Lechner 2012 | Levosimendan | Milrinone | 0.8403 | 0.2377 | 2.9710 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.8403 | 0.2377 | 2.9710 |
| Wang 2019 | Levosimendan | Placebo | 0.5496 | 0.2681 | 1.1268 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5496 | 0.2681 | 1.1268 | ‐1.63 | 0.1023 |
| Milrinone | 0.6541 | 0.1530 | 2.7960 | ‐0.57 | 0.5668 |
| Placebo | NA | NA | NA | NA | NA |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 7. Supplementary material: analyses for 'length of intensive care stay'
Number of studies: k = 11; Number of pairwise comparisons: m = 11; Number of observations: o = 577; Number of treatments: n = 4; Number of designs: d = 5
Analyse all data
Subnet 1
Results (random‐effects model): Number of studies: k = 9; Number of pairwise comparisons: m = 9; Number of treatments: n = 8; Number of designs: d = 7; Number of subnetworks: 3
| Study | Treatment 1 | Treatment 2 | ROM | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 1.1206 | 0.7719 | 1.6266 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9818 | 0.6578 | 1.4653 |
| Costello 2014 | Milrinone | Placebo | 1.1284 | 0.7517 | 1.6941 |
| Ebade 2013 | Dobutamine | Levosimendan | 0.9887 | 0.6721 | 1.4543 |
| Jothinath 2021 | Levosimendan | Milrinone | 0.9930 | 0.7620 | 1.2941 |
| Lechner 2012 | Levosimendan | Milrinone | 0.9930 | 0.7620 | 1.2941 |
| Momeni 2011 | Levosimendan | Milrinone | 0.9930 | 0.7620 | 1.2941 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9930 | 0.7620 | 1.2941 |
| Wang 2019 | Levosimendan | Placebo | 1.1206 | 0.7719 | 1.6266 |
Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.1079 | 0.6593 | 1.8617 | 0.39 | 0.6989 |
| Levosimendan | 1.1206 | 0.7719 | 1.6266 | 0.60 | 0.5494 |
| Milrinone | 1.1284 | 0.7517 | 1.6941 | 0.58 | 0.5599 |
| Placebo | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | ROM | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.7413 | 0.7104 | 0.7734 |
Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.7413 | 0.7104 | 0.7734 | ‐13.81 | < 0.0001 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 3
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 63; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | ROM | 95% CI lower limit | 95% CI upper limit |
| Ricci 2012 | Levosimendan + Milrinone + Dopamine | Milrinone + Dopamine | 1.2727 | 0.8273 | 1.9581 |
Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Milrinone + Dopamine | 1.2727 | 0.8273 | 1.9581 | 1.10 | 0.2726 |
| Milrinone + Dopamine | reference | reference | reference | reference | reference |
CNMA for disconnected network
Results for combinations (additive model, random‐effects model): Number of studies: k = 11; Number of pairwise comparisons: m = 11; Number of treatments: n = 8; Number of active components: c = 4; Number of designs: d = 7; Number of subnetworks: s = 3
Results for combinations (additive model, random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2943 | 0.6123 | 2.7359 | 0.68 | 0.4993 |
| Levosimendan | 1.2727 | 0.6678 | 2.4256 | 0.73 | 0.4636 |
| Levosimendan + Dobutamine | 1.6473 | 0.4294 | 6.3203 | 0.73 | 0.4669 |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 1.3710 | 0.6910 | 2.7201 | 0.90 | 0.3667 |
| Milrinone + Dobutamine | 1.7745 | 0.4462 | 7.0579 | 0.81 | 0.4155 |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Placebo | 1.1604 | 0.5520 | 2.4393 | 0.39 | 0.6947 |
Results for components (random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2943 | 0.6123 | 2.7359 | 0.68 | 0.4993 |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 1.2727 | 0.6678 | 2.4256 | 0.73 | 0.4636 |
| Milrinone | 1.3710 | 0.6910 | 2.7201 | 0.90 | 0.3667 |
| Placebo | 1.1604 | 0.5520 | 2.4393 | 0.39 | 0.6947 |
CNMA for disconnected network, placebo assumed as inactive
Results for combinations (additive model, random‐effects model): Number of studies: k = 11; Number of pairwise comparisons: m = 11; Number of treatments: n = 8; Number of active components: c = 3; Number of designs: d = 7; Number of subnetworks: s = 3
Results for combinations (additive model, random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.1540 | 0.7180 | 1.8547 | 0.59 | 0.5541 |
| Levosimendan | 1.1360 | 0.8299 | 1.5550 | 0.80 | 0.4260 |
| Levosimendan + Dobutamine | 1.3110 | 0.6414 | 2.6794 | 0.74 | 0.4579 |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 1.2197 | 0.8562 | 1.7375 | 1.10 | 0.2714 |
| Milrinone + Dobutamine | 1.4075 | 0.6680 | 2.9657 | 0.90 | 0.3687 |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.1540 | 0.7180 | 1.8547 | 0.59 | 0.5541 |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 1.1360 | 0.8299 | 1.5550 | 0.80 | 0.4260 |
| Milrinone | 1.2197 | 0.8562 | 1.7375 | 1.10 | 0.2714 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable: ROM: ratio of means.
Appendix 8. Supplementary material: analyses for 'length of hospital stay'
Number of studies: k = 8; Number of pairwise comparisons: m = 10; Number of treatments: n = 5; Number of designs: d = 5; Number of subnetworks: 1
Analyse all data
Results (random‐effects model): Number of studies: k = 7; Number of pairwise comparisons: m = 7; Number of observations: o = 497; Number of treatments: n = 4; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | ROM | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.6821 | 0.3907 | 1.1908 |
| Costello 2014 | Milrinone | Placebo | 1.0034 | 0.7759 | 1.2976 |
| Jothinath 2021 | Levosimendan | Milrinone | 1.0294 | 0.8765 | 1.2089 |
| Lechner 2012 | Levosimendan | Milrinone | 1.0294 | 0.8765 | 1.2089 |
| Momeni 2011 | Levosimendan | Milrinone | 1.0294 | 0.8765 | 1.2089 |
| Thorlacius 2020 | Levosimendan | Milrinone | 1.0294 | 0.8765 | 1.2089 |
| Wang 2019 | Levosimendan | Placebo | 1.0329 | 0.8372 | 1.2744 |
Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6844 | 0.3705 | 1.2642 | ‐1.21 | 0.2258 |
| Levosimendan | 1.0329 | 0.8372 | 1.2744 | 0.30 | 0.7627 |
| Milrinone | 1.0034 | 0.7759 | 1.2976 | 0.03 | 0.9792 |
| Placebo | reference | reference | reference | reference | reference |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; ROM: ratio of means.
Appendix 9. Supplementary material: analyses for 'duration of mechanical ventilation'
Number of studies: k = 12; Number of pairwise comparisons: m = 14; Number of treatments: n = 9; Number of designs: d = 8; Number of subnetworks: 3
Subnet 1
Results (random‐effects model): Number of studies: k = 9; Number of pairwise comparisons: m = 9; Number of observations: o = 577; Number of treatments: n = 4; Number of designs: d = 5
| Study | Treatment 1 | Treatment 2 | ROM | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 1.1736 | 0.6482 | 2.1248 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.8257 | 0.4320 | 1.5780 |
| Costello 2014 | Milrinone | Placebo | 1.2541 | 0.6669 | 2.3583 |
| Ebade 2013 | Dobutamine | Levosimendan | 0.8823 | 0.4747 | 1.6399 |
| Jothinath 2021 | Levosimendan | Milrinone | 0.9358 | 0.6262 | 1.3985 |
| Lechner 2012 | Levosimendan | Milrinone | 0.9358 | 0.6262 | 1.3985 |
| Momeni 2011 | Levosimendan | Milrinone | 0.9358 | 0.6262 | 1.3985 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9358 | 0.6262 | 1.3985 |
| Wang 2019 | Levosimendan | Placebo | 1.1736 | 0.6482 | 2.1248 |
Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0355 | 0.4508 | 2.3787 | 0.08 | 0.9345 |
| Levosimendan | 1.1736 | 0.6482 | 2.1248 | 0.53 | 0.5971 |
| Milrinone | 1.2541 | 0.6669 | 2.3583 | 0.70 | 0.4823 |
| Placebo | reference | reference | reference | reference | reference |
Subnet 2
Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1 Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.7105 | 0.6494 | 0.7774 | ‐7.44 | < 0.0001 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 3
Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 63; Number of treatments: n = 2; Number of designs: d = 1
Treatment estimate:
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Milrinone + Dopamine | 1.1695 | 0.7091 | 1.9289 | 0.61 | 0.5397 |
| Milrinone + Dopamine | reference | reference | reference | reference | reference |
CNMA for disconnected network
Number of studies: k = 11; Number of pairwise comparisons: m = 11; Number of treatments: n = 8; Number of active components: c = 4; Number of designs: d = 7; Number of subnetworks: s = 3
Results for combinations (additive model, random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0820 | 0.4146 | 2.8238 | 0.16 | 0.8720 |
| Levosimendan | 1.1695 | 0.5250 | 2.6051 | 0.38 | 0.7016 |
| Levosimendan + Dobutamine | 1.2654 | 0.2343 | 6.8346 | 0.27 | 0.7844 |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 1.3357 | 0.5659 | 3.1528 | 0.66 | 0.5089 |
| Milrinone + Dobutamine | 1.4453 | 0.2550 | 8.1913 | 0.42 | 0.6773 |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Placebo | 1.0272 | 0.3950 | 2.6710 | 0.06 | 0.9561 |
Results for components (random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0820 | 0.4146 | 2.8238 | 0.16 | 0.8720 |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 1.1695 | 0.5250 | 2.6051 | 0.38 | 0.7016 |
| Milrinone | 1.3357 | 0.5659 | 3.1528 | 0.66 | 0.5089 |
| Placebo | 1.0272 | 0.3950 | 2.6710 | 0.06 | 0.9561 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 11; Number of pairwise comparisons: m = 11; Number of treatments: n = 8; Number of active components: c = 3; Number of designs: d = 7; Number of subnetworks: s = 3
Results for combinations (additive model, random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0706 | 0.5674 | 2.0204 | 0.21 | 0.8331 |
| Levosimendan | 1.1444 | 0.7553 | 1.7341 | 0.64 | 0.5246 |
| Levosimendan + Dobutamine | 1.2253 | 0.4726 | 3.1765 | 0.42 | 0.6759 |
| Levosimendan + Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Milrinone | 1.3098 | 0.8222 | 2.0867 | 1.14 | 0.2560 |
| Milrinone + Dobutamine | 1.4023 | 0.5241 | 3.7525 | 0.67 | 0.5007 |
| Milrinone + Dopamine | NA | NA | NA | NA | NA |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | ROM | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0706 | 0.5674 | 2.0204 | 0.21 | 0.8331 |
| Dopamine | NA | NA | NA | NA | NA |
| Levosimendan | 1.1444 | 0.7553 | 1.7341 | 0.64 | 0.5246 |
| Milrinone | 1.3098 | 0.8222 | 2.0867 | 1.14 | 0.2560 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; ROM: ratio of means.
Appendix 10. Supplementary material: analyses for 'number of patients requiring mechanical circulatory support'
Number of studies: k = 7; Number of pairwise comparisons: m = 7; Number of treatments: n = 6; Number of designs: d = 5; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.3333 | 0.0142 | 7.8035 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.3333 | 0.0142 | 7.8035 | ‐0.68 | 0.4947 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 3; Number of pairwise comparisons: m = 3; Number of observations: o = 90; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.3333 | 0.0147 | 7.5655 |
| Momeni 2011 | Levosimendan | Milrinone | 1.4832 | 0.1944 | 11.3145 |
| Pellicer 2013 | Levosimendan | Milrinone | 1.4832 | 0.1944 | 11.3145 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.3333 | 0.0147 | 7.5655 | ‐0.69 | 0.4904 |
| Milrinone | 0.2247 | 0.0054 | 9.3219 | ‐0.79 | 0.4322 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies, subnet 2
Results (random‐effects model): Number of studies: k = 6; Number of pairwise comparisons: m = 6; Number of observations: o = 281; Number of treatments: n = 4; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.5445 | 0.0436 | 6.7964 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9245 | 0.0191 | 44.8254 |
| Costello 2014 | Milrinone | Placebo | 0.4536 | 0.0322 | 6.3865 |
| Momeni 2011 | Levosimendan | Milrinone | 1.2005 | 0.2206 | 6.5335 |
| Pellicer 2013 | Levosimendan | Milrinone | 1.2005 | 0.2206 | 6.5335 |
| Thorlacius 2020 | Levosimendan | Milrinone | 1.2005 | 0.2206 | 6.5335 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.4193 | 0.0038 | 45.9536 | ‐0.36 | 0.7169 |
| Levosimendan | 0.5445 | 0.0436 | 6.7964 | ‐0.47 | 0.6369 |
| Milrinone | 0.4536 | 0.0322 | 6.3865 | ‐0.59 | 0.5580 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of treatments: n = 5; Number of active components: c = 3; Number of designs: d = 3; Number of subnetworks: 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.6833 | 0.2087 | 2.2378 | ‐0.63 | 0.5292 |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone | 0.7139 | 0.1526 | 3.3389 | ‐0.43 | 0.6685 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Placebo | 2.0499 | 0.2369 | 17.7385 | 0.65 | 0.5144 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.6833 | 0.2087 | 2.2378 | ‐0.63 | 0.5292 |
| Milrinone | 0.7139 | 0.1526 | 3.3389 | ‐0.43 | 0.6685 |
| Placebo | 2.0499 | 0.2369 | 17.7385 | 0.65 | 0.5144 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of treatments: n = 5; Number of active components: c = 2; Number of designs: d = 3; Number of subnetworks: 2 Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.3333 | 0.0147 | 7.5655 | ‐0.69 | 0.4904 |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone | 0.3483 | 0.0099 | 12.2320 | ‐0.58 | 0.5613 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Placebo | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.3333 | 0.0147 | 7.5655 | ‐0.69 | 0.4904 |
| Milrinone | 0.3483 | 0.0099 | 12.2320 | ‐0.58 | 0.5613 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 11. Supplementary material: analyses for 'number of patients requiring cardiac transplantation'
Number of studies: k = 7; Number of pairwise comparisons: m = 7; Number of treatments: n = 6; Number of designs: d = 5; Number of subnetworks: 2
Subnet 2
(3) Inverse variance method with increments for zero studies
Results (random‐effects model): Number of studies: k = 6; Number of pairwise comparisons: m = 6; Number of observations: o = 281; Number of treatments: n = 4; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 0.9844 | 0.0529 | 18.3300 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9245 | 0.0191 | 44.8254 |
| Costello 2014 | Milrinone | Placebo | 0.9884 | 0.0527 | 18.5311 |
| Momeni 2011 | Levosimendan | Milrinone | 0.9959 | 0.1262 | 7.8573 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9959 | 0.1262 | 7.8573 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9959 | 0.1262 | 7.8573 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.9138 | 0.0071 | 118.3373 | ‐0.04 | 0.9710 |
| Levosimendan | 0.9844 | 0.0529 | 18.3300 | ‐0.01 | 0.9916 |
| Milrinone | 0.9884 | 0.0527 | 18.5311 | ‐0.01 | 0.9938 |
| Placebo | reference | reference | reference | reference | reference |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 12. Supplementary material: analyses for 'adverse events'
Note: as we have potentially more than one event per individual, the effect measure we use here is a rate ratio, not a risk ratio (Poisson model)
Number of studies: k = 9; Number of pairwise comparisons: m = 11; Number of treatments: n = 7; Number of designs: d = 6; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | IRR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.3000 | 0.1205 | 0.7470 |
Treatment estimate:
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.3000 | 0.1205 | 0.7470 | ‐2.59 | 0.0097 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 8; Number of pairwise comparisons: m = 10; Number of observations: o = 706; Number of treatments: n = 5; Number of designs: d = 5
| Study | Treatment 1 | Treatment 2 | IRR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 1.2211 | 0.7595 | 1.9630 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9600 | 0.6511 | 1.4154 |
| Costello 2014 | Milrinone | Placebo | 1.3028 | 0.9526 | 1.7817 |
| Hoffman 2003 | Milrinone | Milrinone LD | 1.5557 | 0.8759 | 2.7629 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.8374 | 0.4681 | 1.4983 |
| Hoffman 2003 | Milrinone | Placebo | 1.3028 | 0.9526 | 1.7817 |
| Momeni 2011 | Levosimendan | Milrinone | 0.9373 | 0.6354 | 1.3825 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9373 | 0.6354 | 1.3825 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9373 | 0.6354 | 1.3825 |
| Wang 2019 | Levosimendan | Placebo | 1.2211 | 0.7595 | 1.9630 |
Treatment estimate:
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2506 | 0.7595 | 2.0593 | 0.88 | 0.3794 |
| Levosimendan | 1.2211 | 0.7595 | 1.9630 | 0.82 | 0.4096 |
| Milrinone | 1.3028 | 0.9526 | 1.7817 | 1.66 | 0.0978 |
| Milrinone LD | 0.8374 | 0.4681 | 1.4983 | ‐0.60 | 0.5500 |
| Placebo | reference | reference | reference | reference | reference |
Subnet 2, Sensitivity analysis excluding study with high RoB
Results (random‐effects model): Number of studies: k = 7; Number of pairwise comparisons: m = 7; Number of observations: o = 468; Number of treatments: n = 4; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | IRR | 95% CI lower limit | 95% CI upper limit |
| Abril‐Molina 2021 | Levosimendan | Placebo | 1.2386 | 0.7464 | 2.0554 |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9600 | 0.6511 | 1.4154 |
| Costello 2014 | Milrinone | Placebo | 1.3241 | 0.9129 | 1.9205 |
| Momeni 2011 | Levosimendan | Milrinone | 0.9355 | 0.6337 | 1.3809 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9355 | 0.6337 | 1.3809 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9355 | 0.6337 | 1.3809 |
| Wang 2019 | Levosimendan | Placebo | 1.2386 | 0.7464 | 2.0554 |
Treatment estimate:
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2711 | 0.7425 | 2.1760 | 0.87 | 0.3818 |
| Levosimendan | 1.2386 | 0.7464 | 2.0554 | 0.83 | 0.4076 |
| Milrinone | 1.3241 | 0.9129 | 1.9205 | 1.48 | 0.1390 |
| Placebo | reference | reference | reference | reference | reference |
CNMA for disconnected network
Note: Without assuming placebo as inactive, the other components are not identifiable
Placebo assumed as inactive
Number of studies: k = 9; Number of pairwise comparisons: m = 11; Number of treatments: n = 7; Number of active components: c = 4; Number of designs: d = 6; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2678 | 0.7660 | 2.0983 | 0.92 | 0.3560 |
| Levosimendan | 1.0375 | 0.6571 | 1.6381 | 0.16 | 0.8744 |
| Levosimendan + Dobutamine | 1.3153 | 0.5906 | 2.9293 | 0.67 | 0.5023 |
| Milrinone | 1.3206 | 0.9631 | 1.8107 | 1.73 | 0.0842 |
| Milrinone + Dobutamine | 1.6742 | 0.7960 | 3.5213 | 1.36 | 0.1743 |
| Milrinone LD | 0.8436 | 0.4703 | 1.5134 | ‐0.57 | 0.5684 |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | IRR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2678 | 0.7660 | 2.0983 | 0.92 | 0.3560 |
| Levosimendan | 1.0375 | 0.6571 | 1.6381 | 0.16 | 0.8744 |
| Milrinone | 1.3206 | 0.9631 | 1.8107 | 1.73 | 0.0842 |
| Milrinone LD | 0.8436 | 0.4703 | 1.5134 | ‐0.57 | 0.5684 |
Footnotes
Abbreviations CI: confidence interval; IRR: incidence rate ratio; NA: not applicable; RoB: risk of bias.
Appendix 13. Supplementary material: analyses for 'arrhythmia'
Number of studies: k = 9; Number of pairwise comparisons: m = 11; Number of treatments: n = 7; Number of designs: d = 6; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.3333 | 0.0743 | 1.4957 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.3333 | 0.0743 | 1.4957 | ‐1.43 | 0.1515 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 6; Number of pairwise comparisons: m = 8; Number of observations: o = 636; Number of treatments: n = 5; Number of designs: d = 5
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.5275 | 0.1763 | 1.5784 |
| Costello 2014 | Milrinone | Placebo | 1.7554 | 1.0976 | 2.8076 |
| Hoffman 2003 | Milrinone | Milrinone LD | 1.1766 | 0.4440 | 3.1180 |
| Hoffman 2003 | Milrinone LD | Placebo | 1.4920 | 0.5257 | 4.2341 |
| Hoffman 2003 | Milrinone | Placebo | 1.7554 | 1.0976 | 2.8076 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.6812 | 0.2675 | 1.7346 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.6812 | 0.2675 | 1.7346 |
| Wang 2019 | Levosimendan | Placebo | 1.1959 | 0.4577 | 3.1245 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.9260 | 0.2810 | 3.0511 | ‐0.13 | 0.8994 |
| Levosimendan | 1.1959 | 0.4577 | 3.1245 | 0.37 | 0.7151 |
| Milrinone | 1.7554 | 1.0976 | 2.8076 | 2.35 | 0.0188 |
| Milrinone LD | 1.4920 | 0.5257 | 4.2341 | 0.75 | 0.4521 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies, subnet 2
Results (random‐effects model): Number of studies: k = 8; Number of pairwise comparisons: m = 10; Number of observations: o = 715; Number of treatments: n = 5; Number of designs: d = 5
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.5275 | 0.1763 | 1.5784 |
| Costello 2014 | Milrinone | Placebo | 1.7481 | 1.0940 | 2.7932 |
| Hoffman 2003 | Milrinone | Milrinone LD | 1.1757 | 0.4436 | 3.1155 |
| Hoffman 2003 | Milrinone LD | Placebo | 1.4869 | 0.5241 | 4.2186 |
| Hoffman 2003 | Milrinone | Placebo | 1.7481 | 1.0940 | 2.7932 |
| Lechner 2012 | Levosimendan | Milrinone | 0.7109 | 0.2936 | 1.7217 |
| Momeni 2011 | Levosimendan | Milrinone | 0.7109 | 0.2936 | 1.7217 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.7109 | 0.2936 | 1.7217 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.7109 | 0.2936 | 1.7217 |
| Wang 2019 | Levosimendan | Placebo | 1.2428 | 0.4948 | 3.1216 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.9221 | 0.2799 | 3.0372 | ‐0.13 | 0.8939 |
| Levosimendan | 1.2428 | 0.4948 | 3.1216 | 0.46 | 0.6437 |
| Milrinone | 1.7481 | 1.0940 | 2.7932 | 2.34 | 0.0195 |
| Milrinone LD | 1.4869 | 0.5241 | 4.2186 | 0.75 | 0.4559 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 7; Number of pairwise comparisons: m = 9; Number of treatments: n = 7; Number of active components: c = 5; Number of designs: d = 6; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.7825 | 0.3112 | 1.9672 | ‐0.52 | 0.6020 |
| Levosimendan | 0.8277 | 0.4147 | 1.6517 | ‐0.54 | 0.5916 |
| Levosimendan + Dobutamine | 0.6476 | 0.2613 | 1.6051 | ‐0.94 | 0.3481 |
| Milrinone | 1.4834 | 1.0357 | 2.1247 | 2.15 | 0.0315 |
| Milrinone + Dobutamine | 1.1607 | 0.4864 | 2.7701 | 0.34 | 0.7370 |
| Milrinone LD | 1.2562 | 0.5514 | 2.8619 | 0.54 | 0.5872 |
| Placebo | 0.8286 | 0.5190 | 1.3230 | ‐0.79 | 0.4310 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.7825 | 0.3112 | 1.9672 | ‐0.52 | 0.6020 |
| Levosimendan | 0.8277 | 0.4147 | 1.6517 | ‐0.54 | 0.5916 |
| Milrinone | 1.4834 | 1.0357 | 2.1247 | 2.15 | 0.0315 |
| Milrinone LD | 1.2562 | 0.5514 | 2.8619 | 0.54 | 0.5872 |
| Placebo | 0.8286 | 0.5190 | 1.3230 | ‐0.79 | 0.4310 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 7; Number of pairwise comparisons: m = 9; Number of treatments: n = 7; Number of active components: c = 4; Number of designs: d = 6; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.9443 | 0.2869 | 3.1084 | ‐0.09 | 0.9249 |
| Levosimendan | 0.9988 | 0.4265 | 2.3389 | ‐0.00 | 0.9978 |
| Levosimendan + Dobutamine | 0.9432 | 0.1968 | 4.5209 | ‐0.07 | 0.9417 |
| Milrinone | 1.7902 | 1.1221 | 2.8560 | 2.44 | 0.0145 |
| Milrinone + Dobutamine | 1.6905 | 0.4005 | 7.1362 | 0.71 | 0.4749 |
| Milrinone LD | 1.5160 | 0.5346 | 4.2990 | 0.78 | 0.4340 |
| Placebo | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.9443 | 0.2869 | 3.1084 | ‐0.09 | 0.9249 |
| Levosimendan | 0.9988 | 0.4265 | 2.3389 | ‐0.00 | 0.9978 |
| Milrinone | 1.7902 | 1.1221 | 2.8560 | 2.44 | 0.0145 |
| Milrinone LD | 1.5160 | 0.5346 | 4.2990 | 0.78 | 0.4340 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 14. Supplementary material: analyses for 'hypotension'
Number of studies: k = 5; Number of pairwise comparisons: m = 7; Number of treatments: n = 6; Number of designs: d = 5; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.1000 | 0.0138 | 0.7239 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.1000 | 0.0138 | 0.7239 | ‐2.28 | 0.0226 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 3; Number of pairwise comparisons: m = 5; Number of observations: o = 496; Number of treatments: n = 4; Number of designs: d = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Costello 2014 | Milrinone | Placebo | 3.1789 | 0.2064 | 48.9611 |
| Hoffman 2003 | Milrinone | Milrinone LD | 6.0196 | 0.1912 | 189.4765 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.5281 | 0.0168 | 16.6259 |
| Hoffman 2003 | Milrinone | Placebo | 3.1789 | 0.2064 | 48.9611 |
| Wang 2019 | Levosimendan | Placebo | 0.9894 | 0.0199 | 49.2152 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.9894 | 0.0199 | 49.2152 | ‐0.01 | 0.9957 |
| Milrinone | 3.1789 | 0.2064 | 48.9611 | 0.83 | 0.4071 |
| Milrinone LD | 0.5281 | 0.0168 | 16.6259 | ‐0.36 | 0.7168 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies, subnet 2
Results (random‐effects model): Number of studies: k = 4; Number of pairwise comparisons: m = 6; Number of observations: o = 566; Number of treatments: n = 4; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Costello 2014 | Milrinone | Placebo | 2.1683 | 0.3570 | 13.1676 |
| Hoffman 2003 | Milrinone | Milrinone LD | 4.9712 | 0.4632 | 53.3476 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.4362 | 0.0406 | 4.6821 |
| Hoffman 2003 | Milrinone | Placebo | 2.1683 | 0.3570 | 13.1676 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.6056 | 0.0395 | 9.2950 |
| Wang 2019 | Levosimendan | Placebo | 1.3132 | 0.1103 | 15.6351 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 1.3132 | 0.1103 | 15.6351 | 0.22 | 0.8293 |
| Milrinone | 2.1683 | 0.3570 | 13.1676 | 0.84 | 0.4004 |
| Milrinone LD | 0.4362 | 0.0406 | 4.6821 | ‐0.69 | 0.4932 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 4; Number of pairwise comparisons: m = 6; Number of treatments: n = 6; Number of active components: c = 4; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5711 | 0.0956 | 3.4127 | ‐0.61 | 0.5391 |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone | 3.3500 | 0.9343 | 12.0118 | 1.86 | 0.0635 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone LD | 0.5356 | 0.0781 | 3.6749 | ‐0.64 | 0.5252 |
| Placebo | 0.9759 | 0.2729 | 3.4895 | ‐0.04 | 0.9701 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.5711 | 0.0956 | 3.4127 | ‐0.61 | 0.5391 |
| Milrinone | 3.3500 | 0.9343 | 12.0118 | 1.86 | 0.0635 |
| Milrinone LD | 0.5356 | 0.0781 | 3.6749 | ‐0.64 | 0.5252 |
| Placebo | 0.9759 | 0.2729 | 3.4895 | ‐0.04 | 0.9701 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 4; Number of pairwise comparisons: m = 6; Number of treatments: n = 6; Number of active components: c = 3; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.5852 | 0.0512 | 6.6848 | ‐0.43 | 0.6664 |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone | 3.4327 | 0.4955 | 23.7802 | 1.25 | 0.2117 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone LD | 0.5488 | 0.0391 | 7.6978 | ‐0.45 | 0.6561 |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan | 0.5852 | 0.0512 | 6.6848 | ‐0.43 | 0.6664 |
| Milrinone | 3.4327 | 0.4955 | 23.7802 | 1.25 | 0.2117 |
| Milrinone LD | 0.5488 | 0.0391 | 7.6978 | ‐0.45 | 0.6561 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 15. Supplementary material: analyses for 'hypokalaemia'
Number of studies: k = 5; Number of pairwise comparisons: m = 5; Number of treatments: n = 6; Number of designs: d = 4; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 5.0000 | 0.2524 | 99.0649 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 5.0000 | 0.2524 | 99.0649 | 1.06 | 0.2908 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 3; Number of pairwise comparisons: m = 3; Number of observations: o = 191; Number of treatments: n = 4; Number of designs: d = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9231 | 0.5874 | 1.4507 |
| Costello 2014 | Milrinone | Placebo | 1.1574 | 0.8184 | 1.6368 |
| Thorlacius 2020 | Levosimendan | Milrinone | 3.1667 | 0.9157 | 10.9513 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0684 | 0.6044 | 1.8885 | 0.23 | 0.8200 |
| Levosimendan | 3.6651 | 1.0106 | 13.2917 | 1.98 | 0.0481 |
| Milrinone | 1.1574 | 0.8184 | 1.6368 | 0.83 | 0.4084 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies, subnet 2
Results (random‐effects model): Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of observations: o = 211; Number of treatments: n = 4; Number of designs: d = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9231 | 0.5874 | 1.4507 |
| Costello 2014 | Milrinone | Placebo | 1.1574 | 0.8184 | 1.6368 |
| Pellicer 2013 | Levosimendan | Milrinone | 2.7863 | 0.8560 | 9.0697 |
| Thorlacius 2020 | Levosimendan | Milrinone | 2.7863 | 0.8560 | 9.0697 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0684 | 0.6044 | 1.8885 | 0.23 | 0.8200 |
| Levosimendan | 3.2249 | 0.9426 | 11.0338 | 1.87 | 0.0621 |
| Milrinone | 1.1574 | 0.8184 | 1.6368 | 0.83 | 0.4084 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of treatments: n = 6; Number of active components: c = 4; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.7200 | 0.4581 | 1.1318 | ‐1.42 | 0.1546 |
| Levosimendan | 2.6419 | 1.1056 | 6.3128 | 2.19 | 0.0288 |
| Levosimendan + Dobutamine | 1.9022 | 1.0032 | 3.6068 | 1.97 | 0.0489 |
| Milrinone | 0.7800 | 0.5665 | 1.0741 | ‐1.52 | 0.1280 |
| Milrinone + Dobutamine | 0.5616 | 0.2962 | 1.0649 | ‐1.77 | 0.0772 |
| Placebo | 0.6739 | 0.4504 | 1.0084 | ‐1.92 | 0.0550 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.7200 | 0.4581 | 1.1318 | ‐1.42 | 0.1546 |
| Levosimendan | 2.6419 | 1.1056 | 6.3128 | 2.19 | 0.0288 |
| Milrinone | 0.7800 | 0.5665 | 1.0741 | ‐1.52 | 0.1280 |
| Placebo | 0.6739 | 0.4504 | 1.0084 | ‐1.92 | 0.0550 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 4; Number of pairwise comparisons: m = 4; Number of treatments: n = 6; Number of active components: c = 3; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0684 | 0.6044 | 1.8885 | 0.23 | 0.8200 |
| Levosimendan | 3.9200 | 1.1841 | 12.9771 | 2.24 | 0.0253 |
| Levosimendan + Dobutamine | 4.1881 | 1.0190 | 17.2130 | 1.99 | 0.0470 |
| Milrinone | 1.1574 | 0.8184 | 1.6368 | 0.83 | 0.4084 |
| Milrinone + Dobutamine | 1.2365 | 0.5405 | 2.8289 | 0.50 | 0.6151 |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.0684 | 0.6044 | 1.8885 | 0.23 | 0.8200 |
| Levosimendan | 3.9200 | 1.1841 | 12.9771 | 2.24 | 0.0253 |
| Milrinone | 1.1574 | 0.8184 | 1.6368 | 0.83 | 0.4084 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 16. Supplementary material: analyses for 'thrombocytopaenia'
Number of studies: k = 6; Number of pairwise comparisons: m = 8; Number of treatments: n = 7; Number of designs: d = 5; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 0.1429 | 0.0078 | 2.6274 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 0.1429 | 0.0078 | 2.6274 | ‐1.31 | 0.1903 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 3; Number of pairwise comparisons: m = 5; Number of observations: o = 359; Number of treatments: n = 4; Number of designs: d = 3
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9231 | 0.7851 | 1.0854 |
| Costello 2014 | Milrinone | Placebo | 0.7332 | 0.3393 | 1.5844 |
| Hoffman 2003 | Milrinone | Milrinone LD | 0.8542 | 0.3417 | 2.1351 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.8584 | 0.3593 | 2.0507 |
| Hoffman 2003 | Milrinone | Placebo | 0.7332 | 0.3393 | 1.5844 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6768 | 0.3080 | 1.4874 | ‐0.97 | 0.3312 |
| Milrinone | 0.7332 | 0.3393 | 1.5844 | ‐0.79 | 0.4299 |
| Milrinone LD | 0.8584 | 0.3593 | 2.0507 | ‐0.34 | 0.7311 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies, subnet 2
Results (random‐effects model): Number of studies: k = 5; Number of pairwise comparisons: m = 7; Number of observations: o = 449; Number of treatments: n = 5; Number of designs: d = 4
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 0.9231 | 0.7851 | 1.0854 |
| Costello 2014 | Milrinone | Placebo | 0.7332 | 0.3393 | 1.5844 |
| Hoffman 2003 | Milrinone | Milrinone LD | 0.8542 | 0.3417 | 2.1351 |
| Hoffman 2003 | Milrinone LD | Placebo | 0.8584 | 0.3593 | 2.0507 |
| Hoffman 2003 | Milrinone | Placebo | 0.7332 | 0.3393 | 1.5844 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9861 | 0.0644 | 15.0888 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9861 | 0.0644 | 15.0888 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6768 | 0.3080 | 1.4874 | ‐0.97 | 0.3312 |
| Levosimendan | 0.7231 | 0.0425 | 12.3097 | ‐0.22 | 0.8226 |
| Milrinone | 0.7332 | 0.3393 | 1.5844 | ‐0.79 | 0.4299 |
| Milrinone LD | 0.8584 | 0.3593 | 2.0507 | ‐0.34 | 0.7311 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 4; Number of pairwise comparisons: m = 6; Number of treatments: n = 6; Number of active components: c = 5; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2606 | 0.6492 | 2.4476 | 0.68 | 0.4939 |
| Levosimendan + Dobutamine | 0.2459 | 0.0389 | 1.5542 | ‐1.49 | 0.1359 |
| Milrinone | 1.3656 | 0.7118 | 2.6200 | 0.94 | 0.3486 |
| Milrinone + Dobutamine | 1.7215 | 0.4668 | 6.3490 | 0.82 | 0.4146 |
| Milrinone LD | 1.5987 | 0.6561 | 3.8954 | 1.03 | 0.3018 |
| Placebo | 1.8625 | 0.8338 | 4.1602 | 1.52 | 0.1293 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.2606 | 0.6492 | 2.4476 | 0.68 | 0.4939 |
| Levosimendan | 0.1951 | 0.0186 | 2.0411 | ‐1.36 | 0.1725 |
| Milrinone | 1.3656 | 0.7118 | 2.6200 | 0.94 | 0.3486 |
| Milrinone LD | 1.5987 | 0.6561 | 3.8954 | 1.03 | 0.3018 |
| Placebo | 1.8625 | 0.8338 | 4.1602 | 1.52 | 0.1293 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 4; Number of pairwise comparisons: m = 6; Number of treatments: n = 6; Number of active components: c = 4; Number of designs: d = 4; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6768 | 0.3080 | 1.4874 | ‐0.97 | 0.3312 |
| Levosimendan + Dobutamine | 0.0709 | 0.0026 | 1.9193 | ‐1.57 | 0.1158 |
| Milrinone | 0.7332 | 0.3393 | 1.5844 | ‐0.79 | 0.4299 |
| Milrinone + Dobutamine | 0.4963 | 0.1054 | 2.3371 | ‐0.89 | 0.3755 |
| Milrinone LD | 0.8584 | 0.3593 | 2.0507 | ‐0.34 | 0.7311 |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 0.6768 | 0.3080 | 1.4874 | ‐0.97 | 0.3312 |
| Levosimendan | 0.1047 | 0.0052 | 2.1296 | ‐1.47 | 0.1421 |
| Milrinone | 0.7332 | 0.3393 | 1.5844 | ‐0.79 | 0.4299 |
| Milrinone LD | 0.8584 | 0.3593 | 2.0507 | ‐0.34 | 0.7311 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 17. Supplementary material: analyses for 'incidence of elevated liver enzymes'
Number of studies: k = 3; Number of pairwise comparisons: m = 3; Number of treatments: n = 5; Number of designs: d = 3; Number of subnetworks: 2
Subnet 1
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 50; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Shah 2013 | Levosimendan + Dobutamine | Milrinone + Dobutamine | 3.0000 | 0.1281 | 70.2318 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | 3.0000 | 0.1281 | 70.2318 | 0.68 | 0.4947 |
| Milrinone + Dobutamine | reference | reference | reference | reference | reference |
Subnet 2
Results (random‐effects model): Number of studies: k = 1; Number of pairwise comparisons: m = 1; Number of observations: o = 71; Number of treatments: n = 2; Number of designs: d = 1
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Costello 2014 | Milrinone | Placebo | 0.4861 | 0.0950 | 2.4869 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Milrinone | 0.4861 | 0.0950 | 2.4869 | ‐0.87 | 0.3864 |
| Placebo | reference | reference | reference | reference | reference |
Inverse variance method with increments for zero studies, subnet 2
Results (random‐effects model): Number of studies: k = 2; Number of pairwise comparisons: m = 2; Number of observations: o = 91; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Costello 2014 | Milrinone | Placebo | 0.4861 | 0.0950 | 2.4869 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.8261 | 0.0180 | 37.8461 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan | 0.4016 | 0.0063 | 25.6874 | ‐0.43 | 0.6672 |
| Milrinone | 0.4861 | 0.0950 | 2.4869 | ‐0.87 | 0.3864 |
| Placebo | reference | reference | reference | reference | reference |
Component network meta‐analysis (CNMA) for disconnected network
Number of studies: k = 2; Number of pairwise comparisons: m = 2; Number of treatments: n = 4; Number of active components: c = 3; Number of designs: d = 2; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone | 0.5452 | 0.1669 | 1.7805 | ‐1.00 | 0.3151 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Placebo | 1.1215 | 0.2470 | 5.0918 | 0.15 | 0.8819 |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan | 1.6355 | 0.1865 | 14.3446 | 0.44 | 0.6570 |
| Milrinone | 0.5452 | 0.1669 | 1.7805 | ‐1.00 | 0.3151 |
| Placebo | 1.1215 | 0.2470 | 5.0918 | 0.15 | 0.8819 |
CNMA for disconnected network, placebo assumed as inactive
Number of studies: k = 2; Number of pairwise comparisons: m = 2; Number of treatments: n = 4; Number of active components: c = 2; Number of designs: d = 2; Number of subnetworks: s = 2
Results for combinations (additive model, random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Levosimendan + Dobutamine | NA | NA | NA | NA | NA |
| Milrinone | 0.4861 | 0.0950 | 2.4869 | ‐0.87 | 0.3864 |
| Milrinone + Dobutamine | NA | NA | NA | NA | NA |
| Placebo | 1 | 1 | 1 | NA | NA |
Results for components (random‐effects model):
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | NA | NA | NA | NA | NA |
| Levosimendan | 1.4583 | 0.0419 | 50.8033 | 0.21 | 0.8350 |
| Milrinone | 0.4861 | 0.0950 | 2.4869 | ‐0.87 | 0.3864 |
Footnotes
Abbreviations CI: confidence interval; NA: not applicable; RR: relative risk.
Appendix 18. Supplementary material: analyses for 'reduced left ventricular ejection fraction (< 50%) or shortening fraction (< 28%)'
Number of studies: k = 3; Number of pairwise comparisons: m = 3; Number of treatments: n = 3; Number of designs: d = 2; Number of subnetworks: 1
Inverse variance method
Results (random‐effects model): Number of studies: k = 3; Number of pairwise comparisons: m = 3; Number of observations: o = 134; Number of treatments: n = 3; Number of designs: d = 2
| Study | Treatment 1 | Treatment 2 | RR | 95% CI lower limit | 95% CI upper limit |
| Cavigelli‐Brunner 2018 | Dobutamine | Milrinone | 1.8462 | 0.7364 | 4.6281 |
| Pellicer 2013 | Levosimendan | Milrinone | 0.9505 | 0.8439 | 1.0706 |
| Thorlacius 2020 | Levosimendan | Milrinone | 0.9505 | 0.8439 | 1.0706 |
Treatment estimate:
| Treatment | RR | 95% CI lower limit | 95% CI upper limit | z | P |
| Dobutamine | 1.8462 | 0.7364 | 4.6281 | 1.31 | 0.1910 |
| Levosimendan | 0.9505 | 0.8439 | 1.0706 | ‐0.84 | 0.4030 |
| Milrinone | NA | NA | NA | NA | NA |
Footnotes CI: confidence interval; NA: not applicable; RR = relative risk.
Data and analyses
Comparison 1. Pairwise analysis: levosimendan versus milrinone.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 All‐cause mortality within 30 days | 4 | 169 | Risk Ratio (M‐H, Random, 95% CI) | 0.59 [0.11, 3.29] |
| 1.2 Low cardiac output syndrome | 3 | 99 | Risk Ratio (M‐H, Random, 95% CI) | 0.72 [0.31, 1.70] |
| 1.3 Length of intensive care stay | 4 | 189 | Mean Difference (IV, Random, 95% CI) | 0.34 [‐1.14, 1.81] |
| 1.4 Length of hospital stay | 4 | 189 | Mean Difference (IV, Random, 95% CI) | 0.40 [‐1.42, 2.22] |
| 1.5 Duration of mechanical ventilation | 4 | 189 | Mean Difference (IV, Random, 95% CI) | 3.95 [‐14.42, 22.31] |
| 1.6 Inotrope score | 3 | 129 | Mean Difference (IV, Random, 95% CI) | ‐0.88 [‐2.42, 0.66] |
| 1.7 Number of patients requiring mechanical circulatory support | 3 | 130 | Risk Ratio (IV, Random, 95% CI) | 1.49 [0.19, 11.37] |
| 1.8 Arrhythmia | 4 | 169 | Risk Ratio (M‐H, Random, 95% CI) | 0.64 [0.20, 2.07] |
| 1.9 Hypokalaemia | 2 | 90 | Risk Ratio (M‐H, Random, 95% CI) | 3.17 [0.92, 10.95] |
| 1.10 Reduced left ventricular ejection fraction (< 50%) or shortening fraction (< 28%) | 2 | 84 | Risk Ratio (M‐H, Random, 95% CI) | 0.95 [0.84, 1.07] |
| 1.11 Renal failure requiring replacement therapy | 2 | 106 | Risk Ratio (M‐H, Random, 95% CI) | 1.17 [0.18, 7.69] |
Comparison 2. Pairwise analysis: levosimendan versus placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 All‐cause mortality within 30 days | 2 | 217 | Risk Ratio (M‐H, Random, 95% CI) | 0.57 [0.15, 2.13] |
| 2.2 Length of intensive care stay | 2 | 217 | Mean Difference (IV, Random, 95% CI) | ‐0.06 [‐1.16, 1.04] |
| 2.3 Duration of mechanical ventilation | 2 | 217 | Mean Difference (IV, Random, 95% CI) | 1.21 [‐3.50, 5.91] |
| 2.4 Inotrope score | 2 | 217 | Mean Difference (IV, Random, 95% CI) | ‐1.06 [‐2.04, ‐0.08] |
Comparison 3. Pairwise analysis: milrinone versus placebo.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 3.1 Arrhythmia | 2 | 229 | Risk Ratio (M‐H, Random, 95% CI) | 2.01 [0.83, 4.85] |
| 3.2 Hypotension | 2 | 229 | Risk Ratio (M‐H, Random, 95% CI) | 3.38 [0.16, 72.69] |
| 3.3 Thrombocytopaenia | 2 | 229 | Risk Ratio (M‐H, Random, 95% CI) | 0.73 [0.34, 1.58] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Abril‐Molina 2021.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: operating room and paediatric ICU of Virgen de las Nieves University Hospital, Granada, Spain Study start/end dates or study duration: January 2013 to November 2016 Funding sources: Andalusian Progress and Health Foundation, Spain Declarations of interest: The authors declare that there is no conflict of interest. |
|
| Participants |
Sample size: 30 patients randomised Patient age: 28 days to 13 years; mean age: levosimendan 0.6 (IQR 0.5 to 4) years, placebo 0.9 (IQR 0.6 to 8) years Patient sex: levosimendan: 9 male/6 female, placebo 10 male/5 female Diagnosis/severity: all with biventricular congenital heart disease, two of four risk factors (as listed under inclusion criteria) Inclusion criteria: at least two of the following risk factors for developing low cardiac output after surgery:
|
|
| Interventions |
Intervention 1: levosimendan (n = 15); levosimendan intravenously 0.2 μg/kg/min from 12 h before surgery until 24 h after surgery (without loading dose) Intervention 2: placebo (n = 15); continuous intravenous infusion of glucose serum at 5% from 12 h before surgery until 24 h after surgery (without loading dose) Co‐interventions: additional inotropic treatment as needed |
|
| Outcomes |
Primary outcome(s): levels of troponin I and BNP immediately, at 12 and 24 h after surgery Secondary outcome(s):
|
|
| Notes |
Intention‐to‐treat analysis: yes Trialists contacted: yes Unpublished information received: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Patients were selected consecutively among those who met the inclusion criteria. On receipt of the corresponding signed informed consent forms, the patients were assigned to a specific treatment group (levosimendan or placebo) by a simple randomisation process, in which a succession of random numbers were generated using specialised software for this purpose (MAS 2.1®, GlaxoSmithKline). |
| Allocation concealment (selection bias) | Low risk | Randomisation was performed by the hospital pharmacy service. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | “The Hospital Pharmacy Service was responsible for supplying the intervention material and the placebo as individualised daily doses through dose drug dispensing system. Identical syringes and opaque infusion systems were used in order to prevent visualisation of the drug’s colour.” Placebo was "prepared in opaque syringes identical in appearance and with the same volume as those used for the intravenous administration of levosimendan." “The intervention was masked to the investigator who assigned the treatment groups, to the patients’ parents/guardians, and to the nurse who administered the intervention (observer).” Two patients presented severely low cardiac output, and study group assignment was un‐blinded. Both patients had originally been assigned to the placebo group, then levosimendan was initiated. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | “The intervention was masked to the investigator who assigned the treatment groups, to the patients’ parents/guardians, and to the nurse who administered the intervention (observer).” |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No incomplete outcome data suspected. |
| Selective reporting (reporting bias) | Low risk | No selective reporting suspected. |
| Other bias | Low risk | No other bias suspected. |
Cavigelli‐Brunner 2018.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: operating room and cardiac ICU of University Children's Hospital, Zurich, Switzerland Study start/end dates or study duration: July 2008 to December 2011 Funding sources: grant support funding from a private donation, UBS Foundation ("the funders had no role in study design, data collection and analysis, decision to publish, or preparation of the article") Declarations of interest: otherwise authors declared to have no potential conflict of interest |
|
| Participants |
Sample size: 50 Age: range from 0.2 to 14.2 years (mean age milrinone group 0.7 years; mean age dobutamine group 1.7 years) Patient sex: 28 male, 22 female Diagnosis/severity: uni‐ or biventricular physiology Inclusion criteria: infants and children older than 6 weeks of age undergoing cardiac surgery on cardiopulmonary bypass (CPB) with a risk of postoperative LCOS based on the intracardiac anatomy and the performed surgery and in whom routine administration of postoperative inotropic support was anticipated |
|
| Interventions |
Intervention 1 (n = 24): continuous infusion of milrinone 0.75 μg/kg/min for 36 hours after cardiopulmonary bypass (started at half the dose for the first 4 hours postoperatively). Intervention 2 (n = 26): continuous infusion of dobutamine 6 μg/kg/min for 36 hours after cardiopulmonary bypass (started at half the dose for the first 4 hours postoperatively) Co‐interventions: any other drug, for example, adrenaline, noradrenaline, or sodium nitroprusside, as deemed necessary by the attending intensivist |
|
| Outcomes |
Primary outcome(s): "additional amount of vasoactive support within the first 48 hours postoperatively (number of patients who needed supplementary vasoactive support (adrenaline, noradrenaline, or sodium nitroprusside)" Secondary outcome(s):
"Data collection of invasive and noninvasive monitoring and ventilation was performed at arrival on ICU (time = 0) and every 4 hours thereafter for 48 hours. Blood tests were recorded every 8 hours." Follow‐up until hospital discharge (range 6.9 to 78 days) unless otherwise stated. |
|
| Notes |
Intention‐to‐treat: no Trialists contacted: yes Unpublished information received: study duration, randomisation, and blinding methods |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Sequence generation by independent pharmacy, algorithm not known |
| Allocation concealment (selection bias) | Low risk | Sequence generation by independent pharmacy (unpublished data) |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Study drug prepared by independent pharmacy, same infusion rates |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Envelopes with allocation code kept closed until the end of the study (unpublished data) |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No incomplete outcome data suspected. No dropouts. |
| Selective reporting (reporting bias) | Low risk | No selective reporting suspected. |
| Other bias | Low risk | No other bias suspected. |
Costello 2014.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: cardiac operating rooms and cardiac intensive care unit at Boston Children’s Hospital Study start/end dates or study duration: from October 2007 to January 2013 Funding sources: American Heart Association. "The American Heart Association had no role in study design, data collection and analysis, decision to publish, or preparation of the article." Declarations of interest: "Disclosures: None." |
|
| Participants |
Sample size: 106 Patient age: range 1.8 to 14.4 years (mean age milrinone group 2.6 years, placebo group 2.7 years) Patient sex: 19 male and 17 female in the milrinone group, 23 male and 12 female in the placebo group Diagnosis/severity: all with univentricular heart disease undergoing completion of Fontan palliation Inclusion criteria: all patients undergoing a primary Fontan operation (univentricular physiology) |
|
| Interventions |
Intervention 1: milrinone 50 μg/kg bolus followed by an infusion of 0.5 μg/kg/minute (allowed to be halved or transiently held, or to be increased up to 1 μg/kg/minute). Initiation of study drug in the operating room before rewarming from cardiopulmonary bypass, or upon admission to the ICU at the latest. "Per protocol, the study drug was discontinued in the CICU when ≥12 hours of study drug infusion had been administered and the study drug was no longer thought to be clinically indicated by the attending cardiac intensivist or surgeon or when 120 hours of study drug infusion was reached, whichever came first." Intervention 2: placebo 0.33 mL/kg bolus of 5% dextrose in water, followed by an infusion of 5% dextrose in water. Initiation of study drug in the operating room before rewarming from cardiopulmonary bypass, or upon admission to the ICU at the latest. "Per protocol, the study drug was discontinued in the CICU when ≥12 hours of study drug infusion had been administered and the study drug was no longer thought to be clinically indicated by the attending cardiac intensivist or surgeon or when 120 hours of study drug infusion was reached, whichever came first." Intervention 3: nesiritide (not eligible as a study drug for this review) Co‐interventions: volume, dopamine, "inotropic/vasopressor support" as needed in case of hypotension, "other vasodilators", "routine medical management", "postoperative management in the CICU was otherwise conducted according to routine clinical practice". |
|
| Outcomes |
Primary outcome(s): number of days alive and out of the hospital within 30 days of surgery (= combined endpoint of all‐cause mortality within 30 days and duration of hospital stay) Secondary outcome(s):
|
|
| Notes |
Intention‐to‐treat analysis: yes Trialists contacted: no Unpublished information received: no |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated numbers in a 1:1:1 allocation using block sizes of 6, stratified by whether there was a preoperative plan for additional cardiac surgical procedures at the time of Fontan operation (e.g. valvuloplasty, aortic arch reconstruction). |
| Allocation concealment (selection bias) | Low risk | “Allocation concealment was achieved using sequentially numbered opaque, sealed envelopes that were kept in a locked central pharmacy and opened by a pharmacist on the morning of surgery” “Using indistinguishable syringes and identical volumes of administered study drug” |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Patients, investigators, and treating clinicians were blinded to treatment assignment. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Patients, investigators, and treating clinicians were blinded to treatment assignment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No incomplete outcome data suspected. |
| Selective reporting (reporting bias) | Low risk | No selective reporting suspected. |
| Other bias | Low risk | No other bias suspected. |
Ebade 2013.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: Pediatrics, Cairo University Hospital Study start/end dates or study duration: from January 2011 until January 2012 Funding sources: not stated Declarations of interest: not stated |
|
| Participants |
Sample size: 50 Patient age: 7 to 38 months Patient sex: 13 males and 12 females in the dobutamine group, 16 males and 9 females in the levosimendan group Diagnosis/severity: all with biventricular heart defects: atrial or ventricular septal defects with high systolic pulmonary artery pressure exceeding 50% of systemic systolic pressure Inclusion criteria: patients with either atrial or ventricular septal defects with high systolic pulmonary artery pressure exceeding 50% of systemic systolic pressure, assigned for surgical correction of the defect with cardiopulmonary bypass |
|
| Interventions |
Intervention 1: levosimendan infusion started immediately after declamping of the aorta with an initial loading dose of 15 μg/kg given over a 10‐minute period, followed by infusion at 0.1 to 0.2 μg/kg/min. The exact rate of study drug infusion was titrated according to haemodynamic data. Postoperative infusion continued for 24 h from the time of admission to the ICU. Intervention2: dobutamine infusion started immediately after declamping of the aorta, at 4 to 10 μg/kg/min. The exact rate of study drug infusion was titrated according to haemodynamic data. Postoperative infusion continued for 24 h from the time of admission to the ICU. Co‐interventions: nitroglycerine infusion 1 to 2 μg/kg/min after induction of anaesthesia until weaning from CPB for both groups, in case of hypotension phenylephrine 1 to 2 μg/kg bolus doses |
|
| Outcomes |
Primary outcome(s):
Secondary outcome(s): duration of mechanical ventilation, duration of ICU stay |
|
| Notes | Exact rate of infusion for intervention and control drug was titrated according to haemodynamic data Intention‐to‐treat analysis: yes Trialists contacted: yes Unpublished information received: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | “Patients were randomized using sealed envelopes and allocated to two equal groups”. No further details of random sequence generation stated. |
| Allocation concealment (selection bias) | Low risk | Allocation concealment was performed using sealed envelopes. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No blinding reported, even though one of the study drugs was levosimendan, which is a yellow solution and could therefore be distinguished from other infusions by the patient and bedside personnel. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No blinding. Also, measurement of cardiac index by transoesophageal Doppler is not considered to be gold standard. Especially in the setting of an unblinded study, there is a certain risk of detection bias. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All outcomes listed in the methods section reported. |
| Selective reporting (reporting bias) | Low risk | No selective reporting suspected. |
| Other bias | Low risk | No other bias suspected. |
Hoffman 2003.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: multicentre (31 tertiary care children’s hospitals/hospitals providing paediatric congenital cardiac surgery in North America) Study start/end dates or study duration: first patient enrolled July 2000 and last patient enrolled February 2001 Funding sources: "supported by Sanofi‐Synthelabo, New York, NY, and conducted by Covance Periapproval Services, Inc, Radnor, Pa." Declarations of interest: not stated |
|
| Participants |
Sample size: 242 Patient age: placebo group 8.3 ± 14.8 months, low‐dose milrinone group 5.9 ± 10.2 months, high‐dose milrinone group 8.6 ± 16.5 months Patient sex: 36 males and 39 females in the placebo group, 48 males and 31 females in the low‐dose milrinone group, 39 males and 34 females in the high‐dose milrinone group Diagnosis/severity: all with biventricular heart disease: tetralogy of fallot, atrioventricular septal defect, d‐transposition of the great arteries, ventricular septal defect, mitral valve surgery, double outlet right ventricle, total anomalous pulmonary venous return, truncus arteriosus, Ross procedure Inclusion criteria: children between gestational age 36 weeks and 6 years without preoperative LCOS who were undergoing biventricular repair of certain cardiac lesions involving cardiopulmonary bypass |
|
| Interventions |
Intervention 1: placebo infusion at the same rate as the other study drugs. "The physicians were given the option to discontinue study drug between 24 and 36 hours for patients who appeared clinically well." Intervention 2: low‐dose milrinone bolus of 25 μg/kg over 1 hour, followed by infusion of 0.25 μg/kg/min for (23‐)35 hours, starting within 90 minutes after arrival at the ICU from the operating room. "The physicians were given the option to discontinue study drug between 24 and 36 hours for patients who appeared clinically well." Intervention 3: high‐dose milrinone bolus of 75 μg/kg over 1 hour, followed by infusion of 0.75 μg/kg/min for (23‐)35 hours, starting within 90 minutes after arrival at the ICU from the operating room. "The physicians were given the option to discontinue study drug between 24 and 36 hours for patients who appeared clinically well." Co‐interventions: "Baseline catecholamines were administered at the discretion of the physician" (inotrope score was calculated) |
|
| Outcomes |
Primary outcome(s): composite primary endpoint: “death or development of LCOS which requires mechanical support, increased need for existing pharmacologic support (at least 100% over baseline) or administration of new, open‐label positive inotropic agents or other pharmacologic interventions” “or other interventions (eg, mechanical pacing)” to treat LCOS at 36 h after randomisation Definition of LCOS: “clinical signs or symptoms (eg, tachycardia, oliguria, poor perfusion, or cardiac arrest) with or without a widened arterial‐mixed venous oxygen saturation difference or metabolic acidosis” vs. “clinical signs and symptoms of the syndrome – such as tachycardia, oliguria, cold extremities, or cardiac arrest – with or without an at least 30% difference in arterial‐mixed venous oxygen saturation or metabolic acidosis (an increase in the base deficit of >4 or an increase in the lactate of >2 mg/dl) on 2 successive blood gas measurements”. Cut‐offs for outcome variables (tachycardia, oliguria, cold extremities) were determined by the principal investigator (not reported in detail). The composite endpoint was later reviewed by an adjudication committee. Secondary outcome(s)
|
|
| Notes |
Intention‐to‐treat analysis: no. Of 242 randomised patients, 238 received the study drug. 11 were excluded "because of major protocol violations". Of the remaining 227 "per‐protocol population" (placebo n = 75; LD n = 79; HD n = 73), 13 received open‐label milrinone after 36 hours and were not included in further analyses (figure 3, p 999), and 5 were lost to follow‐up, leaving 209 patients (placebo n = 71; LD n = 74; HD n = 64). Two patients (unknown group) died on the 5th and the 13th postoperative day, respectively, leaving 207 patients for 30‐day follow‐up. Trialists contacted: yes Unpublished information received: study dates, placebo infusion rate, duration of postoperative urine collection, numbers of patients reaching outcomes |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Performed by commercial CRO, no additional information available |
| Allocation concealment (selection bias) | Unclear risk | Performed by commercial CRO, no additional information available |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Same infusion rate for all study drugs |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Blinded clinical endpoint committee |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Dropout of a total of 33 participants at different stages (treatment groups not always known), calculation of outcomes using different populations |
| Selective reporting (reporting bias) | High risk | Outcomes are only partially reported as absolute numbers or percentages. The review authors contacted the study authors and obtained additional information. However, due to the long time elapsed since the study had been conducted, many outcomes could not be comprehensively reconstructed. |
| Other bias | High risk | Study supported by a grant from the manufacturer (Sanofi Synthelabo) and reports co‐authored by manufacturer representatives |
Jothinath 2021.
| Study characteristics | ||
| Methods |
Study design: randomised, parallel arm trial Setting: Department of Cardiothoracic and Vascular Surgery at G Kuppuswamy Naidu Memorial Hospital, Coimbatore, Tamil Nadu, India Study start/end dates or study duration: not stated Funding sources: "Nil" Declarations of interest: "There are no conflicts of interest." |
|
| Participants |
Sample size: 40 Patient age: 1 to 12 months, mean age: levosimendan group 8.55 months, milrinone group 6.85 months Patient sex: not stated Diagnosis/severity: all with biventricular heart disease, Risk Adjustment for Congenital Heart Surgery‑I score < 5 Inclusion criteria: children 1 to 12 months of age undergoing corrective open heart surgery |
|
| Interventions |
Intervention 1: levosimendan starting at the time of rewarming after CPB with a loading dose of 10 μg/kg over 10 minutes, followed by infusion of 0.1 μg/kg/min for 24 hours Intervention 2: milrinone starting at the time of rewarming after CPB with a loading dose of 50 μg/kg over 10 minutes, followed by infusion at a rate of 0.5 μg/kg/min for 24 hours Co‐interventions: adrenaline in both groups to aid separation from CPB and continued in the ICU, if required |
|
| Outcomes |
Primary outcome(s)
Time points reported: induction, coming off CPB, chest closure, transfer to the ICU and subsequently every 4 hours through 24 hours Secondary outcome(s)
|
|
| Notes |
Intention‐to‐treat analysis: yes Trialists contacted: no Unpublished information received: no |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation |
| Allocation concealment (selection bias) | High risk | Not reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No blinding reported, even though one of the study drugs was levosimendan, which is a yellow solution and could therefore be distinguished from other infusions by the patient and bedside personnel. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | "Data entry was made [...] in codes" could describe the blinding of outcome assessors, if these were participant identification codes rather than software codes. Also, measurement of cardiac index by transoesophageal Doppler is not considered to be gold standard. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No incomplete outcome data suspected. |
| Selective reporting (reporting bias) | Low risk | No selective reporting suspected. |
| Other bias | High risk | Method of participant selection is unclear. |
Lechner 2012.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: paediatric cardiac ICU, Children’s and Maternity Hospital, Linz, Austria Study start/end dates or study duration: between September 2007 and July 2009 Funding sources: Department of Anaesthesia and surgical intensive care of the AKH Linz (unpublished information) Declarations of interest: "The authors have not disclosed any potential conflicts of interest." |
|
| Participants |
Sample size: 40 Patient age: milrinone group 63.9 ± 81.3 days, levosimendan group 78.4 ± 80.5 days Patient sex: 22 males, 18 females Diagnosis/severity: all with biventricular physiology, Aristotle complexity level 2‐4: ventricular septal defect, complete atrioventricular septal defect, total anomalous pulmonary venous connection, multiple ventricular septal defects, extended aortic coarctation, d‐transposition of the great arteries with or without ventricular septal defect Inclusion criteria: children younger than 1 yr of age scheduled for corrective open‐heart surgery |
|
| Interventions |
Intervention 1: milrinone infusion at 0.5 μg/kg/min for 24 hours, starting at the time of weaning from CPB Intervention 2: levosimendan infusion at 0.1 μg/kg/min for 24 hours, starting at the time of weaning from CPB Co‐interventions: additional inotropic support at the discretion of the senior consultant in charge |
|
| Outcomes |
Primary outcome(s): cardiac index assessed by transesophageal Doppler in the ventilated and sedated patient 2, 6, 9, 12, 18, 24, and 48 hours after initiation of the study drug Secondary outcome(s)
Follow‐up until hospital discharge, mortality follow‐up until 30 days |
|
| Notes |
Intention‐to‐treat analysis: yes, plus per protocol analysis Trialists contacted: yes Unpublished information received: 30‐day mortality, incidence of MCS or heart transplant, funding, duration of mechanical ventilation, duration of ICU stay, hypokalaemia, inotrope score |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random numbers |
| Allocation concealment (selection bias) | Low risk | Preparation and labelling of drugs by the local pharmacy on pharmacy premises |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Same infusion rates for both drugs, opaque black syringes and catheters. Record indicating the type of study drug was kept in a closed envelope in the patient’s chart to allow unblinding in case of emergency (never used throughout the study). All patients, parents, medical, and nursing staff members remained unaware of the study group assignments throughout the whole study period. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Data were collected continuously before unblinding at the end of the study; statistical analysis by an external statistician. However, measurement of cardiac index by transoesophageal Doppler is not considered to be gold standard. Furthermore, it was not possible to obtain reliable standard deviations for inotropic score, as the information extracted from a figure (Figure 4 in this source) deviated from information provided by personal communication with the study author. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Study authors were contacted and asked for missing values. 3 dropouts not included in per‐protocol‐analysis (1 did not receive non‐milrinone study drug due to immediate reoperation). |
| Selective reporting (reporting bias) | Low risk | Study authors were contacted and asked for missing values. |
| Other bias | Low risk | No other bias suspected. |
Momeni 2011.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: Cliniques Universitaires Saint Luc, Brussels, Belgium Study start/end dates or study duration: between June 2008 and October 2009 Funding sources: Department of Anaesthesiology, Cliniques Universitaires Saint Luc, Brussels, Belgium Declarations of interest: not stated |
|
| Participants |
Sample size: 40 (+ one randomised but did not receive the study drug (randomised to milrinone) due to a modification in the surgical plan) Patient age: 0 to 5 years allowed, but only 2 children in each group in the 1‐year‐and‐older age range Patient sex: 9 males and 9 females in the milrinone group, 6 males and 12 females in the levosimendan group Diagnosis/severity: all with biventricular physiology, for example ventricular septal defect, d‐transposition of the great arteries, truncus arteriosus communis, atrio‐ventricular septal defect, double outlet right ventricle, total anomalous pulmonary venous return, anomalous left coronary artery from pulmonary artery Inclusion criteria: all children < 5 years of age undergoing corrective surgery for congenital heart disease using CPB and who needed inotropic support |
|
| Interventions |
Intervention 1: milrinone infusion at 0.4 μg/kg/min for up to 48 hours, starting at the onset of CPB. Infusion rate "was allowed to be doubled if haemodynamic and/or echocardiographic signs of poor ventricular function” were present. “The study drug could be stopped earlier in case of favourable clinical and haemodynamic parameters.” Intervention 2: levosimendan infusion at 0.05 μg/kg/min for up to 48 hours, starting at the onset of CPB. Infusion rate "was allowed to be doubled if haemodynamic and/or echocardiographic signs of poor ventricular function” were present. “The study drug could be stopped earlier in case of favourable clinical and haemodynamic parameters.” Co‐interventions: other inotropes as needed |
|
| Outcomes |
Primary outcome(s): lactate levels 4 hours postoperatively Secondary outcome(s):
|
|
| Notes | Duration of ICU stay, hospital stay, and mechanical ventilation: analysis of 36 patients (18 per group) each, including 3 patients from the milrinone group (including 1 who did not receive milrinone for at least 4 hours in the ICU) and 2 patients from the levosimendan group who did not complete 48 hours of study drug infusion. Intention‐to‐treat analysis: yes, for the 36 patients who received the study drug and were alive and not on MCS after weaning from CPB Trialists contacted: yes Unpublished information received: allocation of dropouts, follow‐up duration for mortality, funding, blinding of outcome assessors |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated random numbers |
| Allocation concealment (selection bias) | Low risk | Concealment of the allocation code “in an envelope that was opened by the study nurse who was also in charge of preparing the medication” but who did not participate in patient care and who did not analyse the data |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Same infusion rates for both drugs (1 mL/hour); syringes and tubing system covered with aluminium foil to ensure blinding |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Separate personnel for study drug preparation versus patient care or data analysis, blinded outcome assessors ("the outcome was assessed at the end of the study": unpublished information) |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Attrition and exclusions are reported: 1 dropout in each group requiring ECMO at the end of cardiopulmonary bypass, 1 dropout in each group who "died intraoperatively because of unexpected anatomic cardiac lesions and severe myocardial dysfunction". No re‐inclusions by the review authors in analysis. |
| Selective reporting (reporting bias) | Low risk | Study authors were contacted and asked for missing values. |
| Other bias | Low risk | Departmental funding, no other bias suspected. |
Pellicer 2013.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: La Paz University Hospital, Madrid, Spain Study start/end dates or study duration: November 2009 to November 2010 Funding sources: Spanish Fondo de Investigación Sanitaria (Spanish Healthcare Research Fund), and the SAMID network, as well as the Orion Pharma Spanish Division, for the pharmacokinetic studies Declarations of interest: "The authors declared no conflict of interest." |
|
| Participants |
Sample size: 20 Patient age: 6 to 34 days Patient sex: 11 male, 9 female Diagnosis/severity: d‐transposition of the great arteries with or without ventricular septal defect, tetralogy of fallot, total anomalous pulmonary venous return, coarctation or hypoplasia of the aorta with atrial septal defect with or without ventricular septal defect Inclusion criteria: neonates undergoing cardiovascular surgery with CPB who were in stable preoperative haemodynamic condition |
|
| Interventions |
Intervention 1: milrinone infusion starting at 0.5 μg/kg/min for the duration of surgery, starting when the central lines were placed (dose 1), increase to 0.75 μg/kg/min at the time of admission to the ICU (dose 2) for 2 hours, increase to 1 μg/kg/min 2 hours after ICU admission. Infusion of milrinone for 48 hours, followed by unblinding and open‐label infusion if needed. Intervention 2: levosimendan infusion at 0.1 μg/kg/min for the duration of surgery, starting when the central lines were placed (dose 1), increase to 0.15 μg/kg/min at the time of admission to the ICU (dose 2) for 2 hours, increase to 0.2 μg/kg/min 2 h after ICU admission. Infusion of levosimendan for 48 hours. Co‐interventions: volume, other cardiovascular drugs (dopamine, epinephrine, dobutamine, urapidil, sodium nitroprusside, nitroglycerin, nitric oxide), 3 doses of methylprednisolone 8 h apart starting the night before surgery. |
|
| Outcomes |
Primary outcome(s): continuous, time‐locked, physiological, and near‐infrared spectroscopy (NIRS) (cerebral and peripheral) recordings during the first 24 h, and at 48 and 96 h postsurgery Secondary outcome(s)
|
|
| Notes |
Intention‐to‐treat analysis: yes Trialists contacted: yes Unpublished information received: blinding of outcome assessors, timing of unblinding, time to death, incidence of adverse events |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation list. "The randomisation was stratified by type of congenital heart defect and risk adjustment using the congenital heart surgery method "Jenkins KJ et al. J Thorac Cardiovasc Surg 2002;123:110‐8"", with 3 strata (20% of patients in low‐risk, 60% in moderate‐low risk, 20% in moderate‐high risk) |
| Allocation concealment (selection bias) | Low risk | “A study nurse who was not involved in the clinical care of the infants prepared the study medication and was the custodian of the allocation code.” |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Same infusion rates for both drugs, opaque black syringes and catheters. Drugs were prepared by a study nurse who was not involved in the care of the patients. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Low risk for the first 48 hours while blinded according to the study protocol (including the 48 hours measurements). Then high risk while continued as open study, follow‐up until 6 days post surgery. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Study authors were contacted and asked for additional data. |
| Selective reporting (reporting bias) | Low risk | Study authors were contacted and asked for additional data. |
| Other bias | Low risk | The manufacturer of one intervention (levosimendan, Orion Pharma Spanish Division, Espoo, Finland) supported the pharmacokinetic part of the study financially. Not deemed an important source of bias, because this financial support was restricted to pharmacokinetics instead of the RCT itself. |
Ricci 2012.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: paediatric cardiac ICU, Bambino Gesu' Children’s Hospital, Rome, Italy Study start/end dates or study duration: January 2008 to December 2010 Funding sources: not stated Declarations of interest: not stated |
|
| Participants |
Sample size: 63 Patient age: 1 to 30 days Patient sex: 20 males and 12 females in the levosimendan group, 17 males and 14 females in the non‐levosimendan group Diagnosis/severity: risk‐adjusted classification for congenital heart surgery (RACHS) 3 and 4 procedures Inclusion criteria: neonates affected by transposition of the great arteries scheduled for elective surgery |
|
| Interventions |
Intervention 1: continuous infusion of 0.1 μg/kg/min levosimendan for 72 hours added to standard inotropic support (milrinone 0.75 μg/kg/min and dopamine 5 to 10 μg/kg/min), started while weaning from CPB Intervention 2: standard post‐CPB inotrope infusion (milrinone 0.75 μg/kg/min and dopamine 5 to 10 μg/kg/min) Co‐interventions: adrenaline 0.05 to 0.3 μg/kg/min if necessary |
|
| Outcomes | Primary outcome(s):
Secondary outcome(s)
Outcomes were recorded until 72 hours postoperatively. |
|
| Notes |
Intention‐to‐treat analysis: not stated, but no dropouts Trialists contacted: yes Unpublished information received: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computerised random‐generation program |
| Allocation concealment (selection bias) | Low risk | Sealed envelopes containing the allocation group opened by a nurse in charge of preparing the infusions. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No blinding, "newborns were openly randomized to receive a 72 h intravenous infusion of levosimendan...". "No blinding was necessary for the study, due to its open label design." |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | No blinding. "No blinding was necessary for the study, due to its open label design." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | All outcomes listed in the methods section reported. No dropouts. |
| Selective reporting (reporting bias) | Low risk | No selective reporting suspected. |
| Other bias | Low risk | No other bias suspected. |
Shah 2013.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: Ahmedabad, India (type of hospital not stated) Study start/end dates or study duration: 2009 to 2013 (4 years) Funding sources: not stated Declarations of interest: not stated |
|
| Participants |
Sample size: 50 Patient age: 2 to 12 years (levosimendan group 4.1 ± 1.3 years, milrinone group 4.0 ± 1.5 years) Patient sex: 16 male and 9 female in the levosimendan group, 17 male and 8 female in the milrinone group Diagnosis/severity: biventricular and univentricular physiology with the following diagnoses: septum primum atrial septal defect, ventricular septal defect, tetrology of fallot, atrial septal defect with pulmonic stenosis, ventricular septal defect with pulmonic stenosis, unobstructed total anomalous pulmonary venous connection and partially anomalous pulmonary venous connection, atrio‐ventricular septal defect, anomalous left coronary artery from pulmonary artery, type I and II Ebstein’s anomaly, double‐chambered right ventricle, Glenn’s surgery Inclusion criteria: patients posted for on‐pump paediatric cardiac surgery, in which probable maximum aortic cross clamp time 60 to 90 minutes |
|
| Interventions |
Intervention 1: levosimendan (n = 25): intravenous bolus infusion of levosimendan 12 μg/kg over 10 minutes after cross clamp removal followed by 0.1 μg/kg/min Intervention 2: milrinone (n = 25): intravenous bolus infusion of milrinone 50 μg/kg over 10 minutes followed by 0.5 μg/kg/min Concomitant interventions: Both groups received dopamine infusion of 5 to 8 μg/kg/min after cross clamp removal. "Both study drugs along with dopamine were stopped average 24‐48 hours after shifting to SICU from operation theatre." Follow‐up period: "After the surgery, patients were monitored for the above said parameters in the post operative period at interval of 0,6,12,18 and 24 hours after shifting to SICU." |
|
| Outcomes |
Primary outcome(s)
Secondary outcome(s)
After the surgery, patients were monitored for the above parameters in the postoperative period at intervals of 0, 6, 12, 18, and 24 hours after shifting to surgical ICU.
|
|
| Notes | For the outcome hypotension, there are two sets of results (table 2, without a definition and table 3, defined as the requirement for norepinephrine). We chose the data set with definition of this outcome. Intention‐to‐treat analysis: not stated Trialists contacted: no Unpublished information received: no |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No random sequence generation described |
| Allocation concealment (selection bias) | High risk | No allocation concealment described, only "patients were randomly allocated into two groups." |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | No blinding reported (other than the label "double blind study", even though one of the study drugs was levosimendan, which is a yellow solution and could therefore be distinguished from other infusions by the patient and bedside personnel. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | No blinding of outcome assessors described |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No dropouts described, results based on complete study population |
| Selective reporting (reporting bias) | Low risk | No bias suspected |
| Other bias | Unclear risk | Funding and possible conflicts of interest not reported |
Thorlacius 2020.
| Study characteristics | ||
| Methods |
Study design: secondary analysis of randomised, controlled, parallel arm trial Setting: operating room and paediatric ICU of 2 tertiary children’s hospitals (university hospitals of Gothenburg, Sweden and Helsinki, Finland) Study start/end dates or study duration: October 2014 to April 2017 Funding sources: funds under the ALF agreement (grants from the Swedish state under the agreement between the Swedish government and county councils) Declarations of interest: authors have disclosed that they do not have any potential conflicts of interest |
|
| Participants |
Sample size: 72 patients randomised Patient age (mean ± SD): levosimendan group 5.9 ± 2.9 months, milrinone group 5.6 ± 2.7 months Patient sex: 16 male and 16 female in the levosimendan group, 18 male and 20 female in the milrinone group Diagnosis/severity: all patients had biventricular physiology. RACHS‐Score 2: levosimendan group 25/32, milrinone group 30/38. RACHS‐Score 3: levosimendan group 7/32, milrinone group 8/38 Inclusion criteria: children aged 1 to 12 months undergoing corrective surgery for non‐restrictive ventricular septal defect or complete balanced atrio‐ventricular septal defect or tetralogy of fallot |
|
| Interventions |
Intervention 1: starting in the operating room after the start of cardiopulmonary bypass: levosimendan bolus of 12 μg/kg, followed by infusion of 0.1 μg/kg/min. Infusion for 24 h plus 2 h weaning at 50% the prior infusion rate. An extra bolus dose of half the loading dose and/or increasing the infusion rate up to 0.16 μg/kg/min levosimendan was permitted. Intervention 2: starting in the operating room after start of cardiopulmonary bypass: milrinone, administered as a bolus dose of 48 μg/kg for 10 minutes, followed by infusion at a rate of 0.4 μg/kg/min. Infusion for 24 h plus 2 h weaning at 50% the prior infusion rate. An extra bolus dose of half the loading dose and/or increasing the infusion rate up to 0.67 μg/kg/min milrinone was permitted. Co‐interventions: anaesthesia with opioids, sevoflurane, and muscle relaxant. Diuretics, adrenergics, fluids at the discretion of the responsible physician. |
|
| Outcomes |
Primary outcome(s): absolute value of serum creatinine data on postoperative day 1 Secondary outcome(s):
|
|
| Notes |
Intention‐to‐treat analysis: no Trialists contacted: yes Unpublished information received: lactate levels, central venous oxygen saturation, arterial oxygen saturation, urine output, incidences of hypokalaemia, anaemia, hypocalcaemia, MCS, heart transplantation |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | "To allocate the patients to the parallel groups, the QMinim computer program was used." |
| Allocation concealment (selection bias) | Low risk | "An external randomizer informed the drug‐preparation staffs which group the patient in question was allocated to. Neither the randomizer nor the drug preparation staffs were involved in providing other care for the patient or compiling study documentation. Additionally, the medical and the study‐documentation staffs were blinded to the drug." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | "The medical and the study‐documentation staffs were blinded to the drug. The study drug [...] was prepared in a concealed syringe attached to a concealed line into the patient." |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | "An external randomizer informed the drug‐preparation staffs which group the patient in question was allocated to. Neither the randomizer nor the drug preparation staffs were involved in providing other care for the patient or compiling study documentation. Additionally, the medical and the study‐documentation staffs were blinded to the drug." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No incomplete outcome data suspected. |
| Selective reporting (reporting bias) | Low risk | All outcomes of interest listed in the methods section reported. |
| Other bias | Low risk | No other bias suspected. |
Wang 2019.
| Study characteristics | ||
| Methods |
Study design: randomised controlled trial, parallel arm Setting: single‐centre trial at a provincial hospital affiliated to Shandong University, China Study start/end dates or study duration: between July 2018 and April 2019 Funding sources: Cardiovascular Multidisciplinary Integrated Research Fund, the Natural Science Foundation of Shandong Province, the Clinical Science and Technology Innovation Program of Jinan City, and the Medicine and Health Science Technology Development of Shandong Province Declarations of interest: "The authors declare that they have no competing interests." |
|
| Participants |
Sample size: 187 Patient age: levosimendan group median 5 months (IQR 2 to 11 months), placebo group median 7 months (IQR 2 to 16 months) Patient sex: levosimendan group 42/94 females, placebo group 41/93 females Diagnosis/severity: RACHS score 2‐5 Inclusion criteria: children up to 48 months of age undergoing cardiac surgery with use of cardiopulmonary bypass |
|
| Interventions |
Intervention: continuous infusion of 0.05 μg/kg/min levosimendan, adjusting infusion rate according to adverse events. Infusion was started as quickly as possible following surgery, for 48 hours. Control: placebo with same infusion appearance, rate, and duration as in the levosimendan group Co‐interventions: any additional medications as appropriate, only concomitant nesiritide not permitted |
|
| Outcomes |
Primary outcome: LCOS incidence. "LCOS was defined as two consecutive measurements of low cardiac output (defined as a cardiac output of≤2.2L/min/m2, without associated relative hypovolaemia), one measurement of low cardiac output plus the use of two or more inotropes at or beyond 24 h after surgery, or the use of two or more inotropes at or beyond 24 hafter surgery with the indicated reason being low cardiac output." Secondary outcome(s)
Follow‐up period: 90 days |
|
| Notes |
Intention to treat analysis: yes, after randomisation Trialists contacted: yes Unpublished information received: no |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | "A computer‐generated, permuted block sequence stratified according to the trial center was used to support the study randomization" |
| Allocation concealment (selection bias) | Low risk | "Sequentially numbered opaque envelopes containing participant assignment groups were sealed" |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | All physicians, patients, outcome assessors, and research staff were blinded to patient treatment assignments, and "preparations being prepared by dedicated trial personnel such that patients and physicians were unaware of which treatment" a given patient received. Detailed description of placebo preparation provided. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | “Sequentially numbered opaque envelopes containing participant assignment groups were sealed, with all physicians, patients, outcome assessors, and research staff being blinded to the patient treatment assignments” |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No incomplete outcome data suspected. |
| Selective reporting (reporting bias) | Low risk | Drug infusion periods shorter than per protocol are reported in detail. |
| Other bias | Low risk | No other bias suspected. “Patients treated in this trial purchased the drug at full cost, and trial design, data collection, subsequent analyses, and manuscript submission were not influenced by the drug manufacturer or the funding agencies supporting this trial”. |
AET: atrial ectopic tachycardia; AKH: Allgemeines Krankenhaus der Stadt; ALF: is the abbreviation in Swedish of an agreement between central government and seven regions on physician education and clinical research; AV: atrioventricular; BNP: brain natriuretic peptide; CBP: cardiopulmonary bypass; CICU: cardiac intensive care unit; CRO: clinical research organisation; DPB: diastolic blood pressure; ECMO: extracorporeal membrane oxygenation; h: hour; HD: high dose; hr(s): heart rate; ICU: intensive care unit; IQR: interquartile range; JET: junctional ectopic tachycardia; LCOS: low cardiac output syndrome; LD: low dose; LVFS: left ventricular fraction of shortening; LVIS: levosimendan vasoactive‐inotropic score; min: minimum; MCS: mechanical circulatory support; p: page; RACHS: risk‐adjusted classification for congenital heart surgery; SBP: systolic blood pressure; SICU: surgical intensive care unit; SVT: supraventricular tachycardia; UOP: urine output; VIS: vasoactive inotropic score; vs: versus; yr: year
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Abdelbaser 2021 | Ineligible intervention (duration of drug administration not compatible with our pre‐specified study selection criteria) |
| Abd Elraouf 2019 | Ineligible comparator (study on intravenous versus inhaled levosimendan) |
| Bianchi 2015 | Ineligible outcomes of interest |
| Elbaser 2020 | Ineligible intervention (duration of drug administration not compatible with our pre‐specified study selection criteria) |
| Farah 2013 | Ineligible intervention of interest (intervention comparator is a phosphodiesterase‐5 inhibitor, not a phosphodiesterase‐3 inhibitor) and ineligible outcomes of interest (pulmonary artery pressure) |
| Hua 2020 | Ineligible study design (retrospective, not a prospective trial) |
| Innes 1994 | Ineligible outcomes of interest (none of our pre‐specified primary outcomes and none past 6 hours) |
| Kanazawa 2021 | Ineligible study design (retrospective, not a prospective trial) |
| Kwapisz 2009 | Ineligible study design (cross‐over) and ineligible outcomes |
| Laitinen 1997 | Ineligible outcomes of interest (haemodynamic data, oxygen consumption, oxygen extraction ratio, laboratory values) |
| Laitinen 1999 | Ineligible outcomes of interest (haemodynamic data, oxygen saturation, oxygen extraction ratio, laboratory values) |
| Silvetti 2022 | Ineligible study design (systematic review, not a randomised trial) |
| Soliman 2018 | Ineligible intervention (two regimens of milrinone infusion, no arm without study drug) |
| Wang 2018 | Ineligible study design (retrospective, not a prospective trial) |
| Wang 2020 | Ineligible study design (systematic review, not a randomised trial) |
Characteristics of studies awaiting classification [ordered by study ID]
Abdallah 2003.
| Methods | Randomised, prospective, parallel arm clinical trial |
| Participants | 29 paediatric patients scheduled for total correction of tetralogy of fallot |
| Interventions | Group 1: milrinone 50 μg/kg loading dose over 10 min then an infusion of 0.5 μg/kg/min Group 2: epinephrine 0.03 to 0.06 μg/kg/min plus nitroglycerine 1 to 2 μg/kg/min |
| Outcomes | Heart rate, mean arterial pressure, central venous pressure, mean pulmonary arterial pressure, mean left atrial pressure, right ventricular systolic and diastolic pressures, rate pressure index and durations of mechanical ventilation, inotropic support and ICU stay |
| Notes | No full text available; unable to contact authors. Eligibility of study therefore not classified so far. |
Gutiérrez‐Riveroll 2022.
| Methods | Randomised, prospective, parallel arm, single‐centre clinical trial |
| Participants | Children aged 4 ± 1.3 years (range 0 to 13 years) undergoing corrective surgery for tetralogy of fallot |
| Interventions |
Group 1 (9 patients): levosimendan 0.1 μg/kg/min from anaesthetic induction Group 2 (10 patients): conventional management with milrinone 0.5 μg/kg/min |
| Outcomes | Mean blood pressure, oxygen saturation by pulse oximetry, urine output, blood lactate level, central venous oxygen saturation |
| Notes |
Nag 2023.
| Methods | Prospective, randomised, controlled single‐centre trial |
| Participants | Children between 1 month and 12 years presenting with ventricular septal defect and pulmonary arterial hypertension between 2018 and 2020 |
| Interventions | Levosimendan group (66 patients): epinephrine, 0.02 μg/kg/min, and a levosimendan loading dose of 12 μg/kg over 10 minutes, followed by a maintenance dose of 0.1 to 0.15 μg/kg/min over 48 hours Milrinone group (66 patients): epinephrine, 0.02 μg/kg/min, and a milrinone loading dose of 50 μg/kg over 10 minutes, followed by a maintenance dose of 0.5 to 1 μg/kg/min over 48 hours |
| Outcomes | In‐hospital mortality, LCOS, inotrope score, pulmonary arterial pressure, myocardial performance index of both ventricles, ICU stay, duration of mechanical ventilation |
| Notes |
ICU: intensive care unit; LCOS: low cardiac output syndrome; min: minute
Characteristics of ongoing studies [ordered by study ID]
CTRI/2023/07/055845.
| Study name | The efficacy and safety of intravenous levosimendan compared with milrinone in preventing low cardiac output after corrective cardiac surgery in tetralogy of fallot |
| Methods | Randomised, single‐centre, parallel‐group, double‐blind clinical trial |
| Participants | 50 patients aged 0 to 12 years admitted to cardiac surgery intensive care after corrective cardiac surgery for tetralogy of fallot, excluding patients with tetralogy of fallot and other congenital cardiac anomalies, patients with history of anaphylactic reaction to levosimendan or milrinone, with preoperative renal or hepatic impairment |
| Interventions |
Intervention: milrinone; intravenous administration of 30 μg/kg loading dose followed by 0.5 μg/kg/min infusion for 48 hours Control: levosimendan; intravenous administration of 12 μg/kg loading dose followed by 0.1 μg/kg/min infusion for 48 hours |
| Outcomes | Aim: to study the efficacy and safety of intravenous levosimendan compared with milrinone in preventing/managing low cardiac output syndrome by evaluating the components of LCOS given below Primary outcome(s)
|
| Starting date | Not recruiting yet, ethics review approved 30 June 2023 |
| Contact information | Contact: Aspari Mahammad Azeez Email: unnikp@gmail.com |
| Notes |
Funding source: Sree Chitra Tirunal Institute for Medical Sciences and Technology, Jai Nagar W Rd, Chalakkuzhi, Thiruvananthapuram, Kerala 695011 Declarations of interest: not available |
NCT03823781.
| Study name | Prophylactic milrinone infusion for the prevention low cardiac output syndrome after corrective surgery for congenital heart disease in infants: a randomized, multi‐center, double‐blinded, placebo‐controlled study |
| Methods | Randomised, multi‐centre, double‐blind, placebo‐controlled study |
| Participants | 520 infants up to 12 months of age |
| Interventions |
Intervention: milrinone will be administered intravenously at a rate of 0.75 μg/kg/min for 35 hours Control: 0.9% normal saline will be administered intravenously for 35 hours Co‐interventions: in both groups, baseline catecholamines will be administered at the discretion of the physician |
| Outcomes |
Primary outcome(s)
Secondary outcome(s)
|
| Starting date | February 2019 |
| Contact information |
Contact 1: Jihong Huang, MD, PhD
Telephone: + 8618930830766
Email: huangjihong@scmc.com.cn Contact 2: Zhuoming Xu, MD, PhD Telephone: + 8618930830783 Email: xuzhuoming@scmc.com.cn |
| Notes |
Funding source: unknown (other than "Shanghai Children's Medical Center (Responsible Party)") Declarations of interest: not available |
AF: atrial fibrillation; CHICU: congenital heart intensive care unit; JET: junctional ectopic tachycardia; LCOS: low cardiac output syndrome; SVT: supraventricular tachycardia; VF: ventricular fibrillation; VT: ventricular tachycardia
Differences between protocol and review
Objectives
We shortened the first objective to correspond to Cochrane style recommendations, and changed "efficacy and safety" to "benefits and harms" for both objectives, because the latter is now the preferred wording.
Types of participants
We were planning to stratify the study population according to age and to circulatory physiology. If a study had included both eligible and ineligible participants, we would have contacted the study authors and asked them to provide data for the eligible patients only. If data for the eligible patient subset had not been available, we would have included a trial if at least 75% of patients had fulfilled our inclusion criteria and assessed this inclusion with a sensitivity analysis. However, there were no studies that included both eligible and ineligible participants.
Types of interventions
Combinations of drugs from the same class would have represented a separate intervention of interest and been displayed as a separate node in the network, but were not expected and did not occur. As study participants usually received many concomitant medications, we changed the term "no pharmacological treatment" to "standard care".
Types of outcome measures
While we had intended to assess the outcome "mechanical ventilation" in a time‐to‐event analysis, we assessed it as a continuous variable instead. To analyse an endpoint as time‐to‐event, one would need either individual participant data or hazards/hazard ratios. Also, censored data would require a time‐to‐event analysis. However, all primary studies reported the endpoints “intensive care unit (ICU) stay”, “hospital stay”, and “duration of mechanical ventilation” as means and standard deviations, 95% confidence intervals, or quantiles, and no censoring was reported. Therefore, we decided to analyse these endpoints as continuous endpoints using ratio of means (ROM) as the effect measure in order to account for possible skewness. We added "Requirement for renal replacement therapy" to the adverse events analysis to reflect the risk of acute kidney injury in the setting of paediatric cardiac surgery at the request of a peer reviewer.
Selection of studies
Had there been any disagreements not resolved by discussion, we would have asked a third author (BS) to arbitrate.
Data extraction and management
We entered data extracted from the included studies into RevMan Web (RevMan 2024), instead of a Review Manager 5 (RevMan 5) file (Review Manager 2014), to facilitate collaborative working because several people can work on it at the same time, and because RevMan 5 was retired in April 2023.
Measures of treatment effect
We aimed to include studies reporting quantiles in the quantitative analysis as far as possible. Therefore, we calculated means and standard deviations from medians and interquartile ranges where possible (Luo 2018), instead of narratively describing skewed data reported as medians and interquartile ranges. We calculated missing standard deviations using other data from the trial, such as confidence intervals, based on methods outlined in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019). We used R (R core team 2023) instead of the now retired software RevMan 5, mainly because R allows all analysis steps to be conducted quickly by running one script, while the results of conversions are exactly the same as in RevMan.
Relative treatment effects
For the time‐to‐event variable (time to death), if available, we would have calculated hazard ratios. If only summary statistics were available, we were going to use methods described by Tierney 2007.
Dealing with missing data
Where calculating missing standard deviations was not possible using other data from the trial, such as confidence intervals, and the missing data would have been thought to introduce serious bias, we had planned to explore the impact of excluding such studies in the overall assessment of results by a sensitivity analysis.
Assessment of heterogeneity
Pairwise comparisons
We planned to assess clinical and methodological heterogeneity within each pairwise comparison with descriptive statistics regarding study and population baseline characteristics, but did this narratively, because often the characteristics were not sufficiently detailed and studies mostly only reported means and ranges for age and ranges for severity scores. We were going to assess clinical heterogeneity (different baseline characteristics) wherever these data were available for the study populations: a) patient age, b) severity of disease (RACHS/Aristotle/STS‐EACTS score). In the case of excessive clinical heterogeneity, we would not have pooled the findings of the included studies.
Network meta‐analysis
We planned to assess the assumption of transitivity across treatment comparisons by the distribution of effect modifiers in the different pairwise comparisons. We would have assessed patient age (according to the subgroups mentioned above) and circulatory physiology (univentricular versus biventricular) as possible effect modifiers. If this distribution was substantially imbalanced (intransitivity), we would not have included those studies for indirect comparisons. However, it was not possible to separate these subgroups due to insufficient subgroup information in the included studies.
Assessment of reporting biases
Had we been able to pool more than 10 trials, we would have created and examined a funnel plot to explore possible small‐study biases for the primary outcomes or missing/unpublished studies leading to reporting bias (Higgins 2011). We planned to examine funnel plot asymmetry according to Chaimani 2013 with a comparison‐adjusted funnel plot applied to the network meta‐analysis.
Sensitivity analysis
For the outcome "all‐cause mortality within 30 days", a sensitivity analysis excluding Shah 2013 due to high risk of bias and unknown duration of follow‐up was not applicable, because this study constituted its own subnetwork.
Subgroup analysis
We had planned to carry out analyses with the following subgroup stratifications, if there had been sufficient subgroup information from the included studies:
Age groups: neonates < 1 month, infants 1 month to 1 year, and paediatric patients 1 year of age or older. In the case of insufficient patient numbers with three age subgroups that preclude drawing meaningful conclusions, subgroup analysis will involve neonates < 1 month and paediatric patients 1 month of age or older.
Cardiovascular physiology: patients with biventricular hearts, patients with univentricular hearts.
We would have used the following outcomes in analysing the subgroups: all‐cause mortality within 30 days, time to death (censored after three months), low cardiac output syndrome defined as two or more of the following: a) blood lactate > 3 mmol/L (27 mg/dL) or increase in blood lactate of at least 2 mmol/l (18 mg/dL) from baseline prior to administration of the intervention of interest; b) central venous oxygen saturation < 50% in biventricular physiology without shunts; c) increase in arterial to central venous oxygen saturation difference by at least 20% from baseline prior to administration of the intervention of interest; d) urine output < 1 m/kg/h; e) peripheral skin temperature to core body temperature difference of > 7 °C; (f) cardiac index as determined by Doppler echocardiography of < 2.2 l/min/m². We would have used the formal test for subgroup differences in the pairwise analysis in Review Manager 5 (Review Manager 2014), and based our interpretation on this.
Contributions of authors
BB: conception and design of the review, co‐ordination of the review, search and selection of studies for inclusion in the review, data collection, data assessment, assessment of the risk of bias in the included studies, calculating quantitative results, assessment of the certainty in the body of evidence, interpretation of data, writing the protocol and the review, guarantor for the review.
JH: conception and design of the review, search and selection of studies for inclusion in the review, data collection, data assessment, assessment of the risk of bias in the included studies, assessment of the certainty in the body of evidence, interpretation of data, revising the review critically for important intellectual content.
GR: conception and design of the review, providing methodological input, calculating quantitative results, data assessment, interpretation of data, revising the review critically for important intellectual content.
BS: conception and design of the review, data assessment, revising the review critically for important intellectual content.
Sources of support
Internal sources
-
None, Other
None
External sources
-
National Institute for Health Research, UK
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to Cochrane Heart until 31 March 2023. The views and opinions expressed are those of the authors and do not necessarily reflect those of the Systematic Reviews Programme, NIHR, NHS or the Department of Health.
Declarations of interest
BB: works as a health professional (paediatric cardiologist at University Children's Hospital Zürich, Switzerland). BB declares travel/accommodation/course fee from the conference organisers of the 1st Joint Meeting of the European Congenital Heart Surgeons Association (ECHSA) and the World Society for Pediatric and Congenital Heart Surgery (WSPCHS) to enable BB to give an invited lecture (19 to 21 June 2021); personal payment.
JH: works as a health professional (physician at the Department of Congenital Heart Disease/Pediatric Cardiology, Heart and Diabetes Center NRW, Ruhr‐University Bochum, Bad Oeynhausen, Germany).
GR: none known.
BS: works as a health professional (chief paediatric cardiologist at the University of Freiburg, Germany).
New
References
References to studies included in this review
Abril‐Molina 2021 {published data only (unpublished sought but not used)}
- Abril-Molina A, Gómez-Luque JM, Perin F, Esteban-Molina M, Ferreiro-Marzal A, Fernandez-Guerrero C, et al. Effect of preoperative infusion of levosimendan on biomarkers of myocardial injury and haemodynamics after paediatric cardiac surgery: a randomised controlled trial. Drugs in R&D 2021;21(1):79-89. [DOI: 10.1007/s40268-020-00332-1] [EU TRIAL REGISTRATION NUMBER: EUCTR 2012-005310-19-ES] [DOI] [PMC free article] [PubMed] [Google Scholar]
- EudraCT 2012-005310-19. Double-blind randomized clinical trial to evaluate the efficacy and safety of levosimendan as preischemic myocardial conditioner in pediatric cardiac surgery [Ensayo clínico aleatorizado doble ciego para evaluar la eficacia y seguridad de levosimendán como acondicionador miocárdico preisquémico en pacientes pediátricos de cirugía cardíaca]. www.clinicaltrialsregister.eu/ctr-search/trial/2012-005310-19/ES/ (first received 4 April 2013).
Cavigelli‐Brunner 2018 {published and unpublished data}
- Cavigelli-Brunner A, Hug MI, Dave H, Baenziger O, Buerki C, Bettex D, et al. Prevention of low cardiac output syndrome after pediatric cardiac surgery: a double-blind randomized clinical pilot study comparing dobutamine and milrinone. Pediatric Critical Care Medicine 2018;19(7):619-25. [DOI: 10.1097/PCC.0000000000001533] [DOI] [PubMed] [Google Scholar]
- Cavigelli-Brunner A, Hug MI, Dave H, Cannizzaro V, Baenziger O, Buerki C, et al. Therapy of low cardiac output syndrome after cardiac surgery in infants and children: a double blind randomized study comparing dobutamine and milrinone. Cardiology in the Young May;23(Suppl S1):S34. [ABSTRACT NUMBER: O11-3] [Google Scholar]
Costello 2014 {published data only}
- Costello JM, Dunbar-Masterson C, Allan CK, Gauvreau K, Newburger JW, McGowan FX Jr, et al. Impact of empiric nesiritide or milrinone infusion on early postoperative recovery after Fontan surgery: a randomized, double-blind, placebo-controlled trial. Circulation Heart Failure 2014;7(4):596-604. [DOI: 10.1161/CIRCHEARTFAILURE.113.001312] [DOI] [PubMed] [Google Scholar]
- Costello JM, Dunbar-Masterson C, Allan CK, Gauvreau K, Newburger JW, McGowan FX Jr, et al. Impact of empiric nesiritide or milrinone infusion on early postoperative recovery after Fontan surgery: a randomized, double-blind, placebo-controlled trial. Circulation: Heart Failure 2014;7(4):593-604. Supplemental material, Tables S1-S3, Figure S1. [DOI: 10.1161/CIRCHEARTFAILURE.113.001312] [URL: www.ahajournals.org/doi/10.1161/CIRCHEARTFAILURE.113.001312#supplementary-materials] [DOI] [PubMed]
- Costello JM, Masterson CD, Allan CK, Gauvreau K, Newburger JW, McGowan FX, et al. Impact of empiric nesiritide or milrinone infusion on early postoperative recovery following fontan surgery: a randomized, double blind, placebo-controlled clinical trial. Circulation 2013;128(Suppl 22):A12099. [DOI] [PubMed] [Google Scholar]
Ebade 2013 {published data only}
- Ebade A, Khalil M, Mohamed A. Levosimendan is superior to dobutamine as an inodilator in the treatment of pulmonary hypertension for children undergoing cardiac surgery. Journal of Anesthesia 2013;27(3):334-9. [DOI: 10.1007/s00540-012-1537-9] [DOI] [PubMed] [Google Scholar]
Hoffman 2003 {published and unpublished data}
- Hoffman TM, Wernovsky G, Atz AM, Bailey JM, Akbary A, Kocsis JF, et al. Prophylactic intravenous use of milrinoneafter cardiac operation in pediatrics (PRIMACORP) study. American Heart Journal 2002;143(1):15-21. [DOI: 10.1067/mhj.2002.120305] [DOI] [PubMed] [Google Scholar]
- Hoffman TM, Wernovsky G, Atz AM, Kulik TJ, NelsonDP, Chang AC, et al. Efficacy and safety of milrinone in preventing low cardiac output syndrome in infants and children after corrective surgery for congenital heart disease. Circulation 2003;107(7):996-1002. [DOI: 10.1161/01.cir.0000051365.81920.28] [DOI] [PubMed] [Google Scholar]
Jothinath 2021 {published data only}
- CTRI/2020/07/026449. A prospective study comparing the clinical efficacy of levosimendan with milrinone in pediatric cardiac surgery [A prospective study comparing the clinical efficacy of levosimendan with milrinone - MILE]. trialsearch.who.int/Trial2.aspx?TrialID=CTRI/2020/07/026449 (first received 9 July 2020).
- Jothinath K, Balakrishnan S, Raju V, Menon S, Osborn J. Clinical efficacy of levosimendan vs milrinone in preventing low cardiac output syndrome following pediatric cardiac surgery. Annals of Cardiac Anaesthesia 2021;24(2):217-23. [DOI: 10.4103/aca.ACA_160_19] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lechner 2012 {published and unpublished data}
- Lechner E, Hofer A, Leitner-Peneder G, Freynschlag R, Mair R, Weinzettel R, et al. Levosimendan versus milrinone in neonates and infants after corrective open-heart surgery: a pilot study. Pediatric Critical Care Medicine 2012;13(5):542-8. [DOI: 10.1097/PCC.0b013e3182455571] [DOI] [PubMed] [Google Scholar]
Momeni 2011 {published data only (unpublished sought but not used)}
- Momeni M, Rubay J, Matta A, Rennotte MT, Veyckemans F, Poncelet AJ, et al. Levosimendan in congenital cardiac surgery: a randomized, double-blind clinical trial. Journal of Cardiothoracic and Vascular Anesthesia 2011;25(3):419–24. [DOI: 10.1053/j.jvca.2010.07.004] [DOI] [PubMed] [Google Scholar]
Pellicer 2013 {published data only (unpublished sought but not used)}
- Pellicer A, Riera J, Lopez-Ortego P, Bravo MC, Madero R, Perez-Rodriguez J, et al. Phase 1 study of two inodilators in neonates undergoing cardiovascular surgery. Pediatric Research 2013;73(1):95–103. [DOI: 10.1038/pr.2012.154] [DOI] [PubMed] [Google Scholar]
Ricci 2012 {published data only}
- NCT01120106. Levosimendan administration in neonates with transposition of the great arteries. clinicaltrials.gov/study/NCT01120106 (first received 10 May 2010).
- Ricci Z, Garisto C, Favia I, Vitale V, Chiara L, Cogo P. Levosimendan infusion in newborns after corrective surgery for congenital heart disease: randomized controlled trial. Intensive Care Medicine 2012;38(7):1198–204. [DOI: 10.1007/s00134-012-2564-6] [DOI] [PubMed] [Google Scholar]
Shah 2013 {published data only}
- Shah H, Bhavsar M, Pandya M. Comparative study of levosimendan versus milrinone for paediatric cardiac surgery patients operated with cardio-pulmonary bypass. International Journal of Research in Medicine 2013;2(4):1-6. [Google Scholar]
Thorlacius 2020 {published and unpublished data}
- Thorlacius EM, Suominen PK, Wahlander H, Keski-Nisula J, Vistnes M, Ricksten S-E, et al. The effect of levosimendan versus milrinone on the occurrence rate of acute kidney injury following congenital heart surgery in infants. A randomized clinical trial. Pediatric Critical Care Medicine 2019;20(10):947-56. [DOI: 10.1097/PCC.0000000000002017] [DOI] [PubMed] [Google Scholar]
- Thorlacius EM, Vistnes M, Ojala T, Keski-Nisula J, Molin M, Romlin BS, et al. Levosimendan versus milrinone and release of myocardial biomarkers after pediatric cardiac surgery: post hoc analysis of clinical trial data. Pediatric Critical Care Medicine 2021;22(7):e402-9. [DOI: 10.1097/PCC.0000000000002712] [DOI] [PubMed] [Google Scholar]
- Thorlacius EM, Wåhlander H, Ojala T, Ylänen K, Keski-Nisula J, Synnergren M, et al. Levosimendan versus milrinone for inotropic support in pediatric cardiac surgery: results from a randomized trial. Journal of Cardiothoracic and Vascular Anesthesia 2020;34(8):2072-80. [DOI: 10.1053/j.jvca.2020.02.027] [DOI] [PubMed] [Google Scholar]
Wang 2019 {published data only (unpublished sought but not used)}
- ChiCTR1800016594. Safety evaluation and therapeutic effect of levosimendan on the low cardiac output syndrome in patients after cardiopulmonary bypass. www.chictr.org.cn/showprojEN.html?proj=28047 (first received 11 June 2018).
- Wang A, Cui C, Fan Y, Zi J, Zhang J, Wang G, et al. Prophylactic use of levosimendan in pediatric patients undergoing cardiac surgery: a prospective randomized controlled trial. Critical Care 2019;23(1):428. [DOI: 10.1186/s13054-019-2704-2] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies excluded from this review
Abdelbaser 2021 {published data only}
- Abdelbaser I, Mageed NA, Elfayoumy SI, Elgamal MF, Elmorsy MM, Taman HI. The direct comparison of inhaled versus intravenous levosimendan in children with pulmonary hypertension undergoing on-cardiopulmonary bypass cardiac surgery: a randomized, controlled, non-inferiority study. Journal of Clinical Anesthesia 2021;71:110231. [DOI: 10.1016/j.jclinane.2021.110231] [DOI] [PubMed] [Google Scholar]
Abd Elraouf 2019 {published data only}
- PACTR201911571076749. The perioperative outcome of inhaled and intravenous levosimendan in children with pulmonary hypertension undergoing on pump cardiac surgery: a comparative randomized study. pactr.samrc.ac.za/TrialDisplay.aspx?TrialID=9513 (first received 18 November 2019). [CENTRAL: CN-02070942]
Bianchi 2015 {published data only}
- Bianchi MO, Cheung PY, Phillipos E, Aranha-Netto A, Joynt C. The effect of milrinone on splanchnic and cerebral perfusion in infants with congenital heart disease prior to surgery: an observational study. Shock 2015;44(2):115-20. [DOI: 10.1097/SHK.0000000000000388] [DOI] [PubMed] [Google Scholar]
Elbaser 2020 {published data only}
- Elbaser IIA, El Aleem El Derie AA. Does inhaled milrinone facilitate weaning from cardiopulmonary bypass in children with congenital heart diseases complicated with pulmonary arterial hypertension? Turkish Journal of Anaesthesiology and Reanimation 2020;48(2):127-33. [DOI: 10.5152/TJAR.2019.91145] [DOI] [PMC free article] [PubMed] [Google Scholar]
Farah 2013 {published data only}PMC3574987
- Farah P, Ahmad-Ali A, Hanane G, Abbas E. Additive effect of phosphodiesterase inhibitors in control of pulmonary hypertension after congenital cardiac surgery in children. Iranian Journal of Pediatrics 2013;23(1):19-26. [PMID: ] [PMID: ] [PMC free article] [PubMed] [Google Scholar]
Hua 2020 {published data only}
- Hua Y, Li B, Wu K, Hu M, Liu X, Huang X. Efficacy of levosimendan on postoperative low cardiac output syndrome of infants with congenital heart disease. Chinese Journal of Applied Clinical Pediatrics 2020;35(1):32-5. [Google Scholar]
Innes 1994 {published data only (unpublished sought but not used)}
- Innes PA, Frazer RS, Booker PD, Allsop E, Kirton C, Lockie J, et al. Comparison of the haemodynamic effects of dobutamine with enoximone after open heart surgery in small children. British Journal of Anaesthesia 1994;72(1):77-81. [DOI: 10.1093/bja/72.1.77] [DOI] [PubMed] [Google Scholar]
Kanazawa 2021 {published data only}
- Kanazawa T, Shimizu K, Iwasaki T, Baba K, Otsuki S, Kotani Y, et al. Perioperative milrinone infusion improves one-year survival after the Norwood-Sano procedure. Journal of Cardiothoracic and Vascular Anesthesia 2021;35(7):2073-8. [DOI: 10.1053/j.jvca.2021.02.017] [DOI] [PubMed] [Google Scholar]
Kwapisz 2009 {published data only}
- Kwapisz MM, Neuhauser C, Scholz S, Welters ID, Lohr T, Koch T, et al. Hemodynamic effects of dobutamine and dopexamine after cardiopulmonary bypass in pediatric cardiac surgery. Paediatric Anaesthesia 2009;19(9):862-71. [DOI: 10.1111/j.1460-9592.2009.03101.x] [DOI] [PubMed] [Google Scholar]
Laitinen 1997 {published data only (unpublished sought but not used)}
- Laitinen P, Happonen JM, Sairanen H, Peltola K, Rautiainen P, Korpela R, et al. Amrinone versus dopamine-nitroglycerin after reconstructive surgery for complete atrioventricular septal defect. Journal of Cardiothoracic and Vascular Anesthesia 1997;11(7):870-4. [DOI: 10.1016/s1053-0770(97)90123-3] [DOI] [PubMed] [Google Scholar]
Laitinen 1999 {published data only (unpublished sought but not used)}
- Laitinen P, Happonen JM, Sairanen H, Peltola K, Rautiainen P. Amrinone versus dopamine and nitroglycerin in neonates after arterial switch operation for transposition of the great arteries. Journal of Cardiothoracic and Vascular Anesthesia 1999;13(2):186-90. [DOI: 10.1016/s1053-0770(99)90085-x] [DOI] [PubMed] [Google Scholar]
Silvetti 2022 {published data only}
- Silvetti S, Belletti A, Bianzina S, Momeni M. Effect of levosimendan treatment in pediatric patients with cardiac dysfunction: an update of a systematic review and meta-analysis of randomized controlled trials. Journal of Cardiothoracic and Vascular Anesthesia 2022;36(3):657-64. [DOI: 10.1053/j.jvca.2021.09.018] [DOI] [PubMed] [Google Scholar]
Soliman 2018 {published data only}
- Soliman R, Ragheb A. Assessment of the effect of two regimens of milrinone infusion in pediatric patients undergoing fontan procedure: a randomized study. Annals of Cardiac Anaesthesia 2018;21(2):134-40. [DOI: 10.4103/aca.ACA_160_17] [DOI] [PMC free article] [PubMed] [Google Scholar]
Wang 2018 {published data only}
- Wang C, Gong J, Shi S, Wang J, Gao Y, Wang S, et al. Levosimendan for pediatric anomalous left coronary artery from the pulmonary artery undergoing repair: a single-center experience. Frontiers in Pediatrics 2018;14(6):225. [DOI: 10.3389/fped.2018.00225] [DOI] [PMC free article] [PubMed] [Google Scholar]
Wang 2020 {published data only}
- Wang H, Luo Q, Li Y, Zhang L, Wu X, Yan F. Effect of prophylactic levosimendan on all-cause mortality in pediatric patients undergoing cardiac surgery-an updated systematic review and meta-analysis. Frontiers in Pediatrics 2020;8:456. [DOI: 10.3389/fped.2020.00456] [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies awaiting assessment
Abdallah 2003 {published data only (unpublished sought but not used)}
- Abdallah I, Shawky H. A randomised controlled trial comparing milrinone and epinephrine as inotropes in paediatric patients undergoing total correction of Tetralogy of Fallot. Egyptian Journal of Anaesthesia 2003;19(4):323-9. [Google Scholar]
Gutiérrez‐Riveroll 2022 {published data only}
- Gutiérrez-Riveroll KI, Mejía Picazo HJ, Dosta-Herrera JJ. Levosimendan for preventing low output syndrome in pediatric patients with correction of tetralogy of fallot [Levosimendán para prevenir el síndrome de bajo gasto en pacientes pediátricos con corrección de tetralogía de fallot]. Revista medica del Instituto Mexicano del Seguro Social 2022;60(3):304-14. [PMID: ] [PMC free article] [PubMed] [Google Scholar]
Nag 2023 {published data only}
- Nag P, Chowdhury SR, Behera SK, Das M, Narayan P. Levosimendan or milrinone for ventricular septal defect repair with pulmonary arterial hypertension. Journal of Cardiothoracic and Vascular Anesthesia 2023;37(6):972‐9. [DOI: 10.1053/j.jvca.2023.01.032] [DOI] [PubMed] [Google Scholar]
References to ongoing studies
CTRI/2023/07/055845 {published data only}
- CTRI/2023/07/055845. Comparing two medications to improve heart function after tetralogy of fallot surgery [The efficacy and safety of intravenous levosimendan compared with milrinone in preventing low cardiac output after corrective cardiac surgery in tetralogy of fallot]. trialsearch.who.int/Trial2.aspx?TrialID=CTRI/2023/07/055845 (first received 28 July 2023). [ICTRP: CTRI/2023/07/055845]
NCT03823781 {published data only}
- NCT03823781. Prophylactic use of milrinone after congenital heart surgery in infants [Prophylactic milrinone infusion for the prevention low cardiac output syndrome after corrective surgery for congenital heart disease in infants: a randomized, multi-center, double-blinded, placebo-controlled study]. clinicaltrials.gov/show/NCT03823781 (first received 30 January 2019).
Additional references
Bailey 2004
- Bailey JM, Hoffman TM, Wessel DL, Nelson DP, Atz AM, Chang AC, et al. A population pharmacokinetic analysis of milrinone in pediatric patients after cardiac surgery. Journal of Pharmacokinetics and Pharmacodynamics 2004;31(1):43-59. [DOI: 10.1023/b:jopa.0000029488.45177.48] [DOI] [PubMed] [Google Scholar]
Balduzzi 2019
- Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evidence Based Mental Health 2019;22(4):153-60. [DOI: 10.1136/ebmental-2019-300117] [DOI] [PMC free article] [PubMed] [Google Scholar]
Baysal 2010
- Baysal A, Saşmazel A, Yıldırım Aİ, Koçak T, Sunar H, Zeybek R. The effects of thyroid hormones and interleukin-8 levels on prognosis after congenital heart surgery [Doğuştan kalp cerrahisinde tiroit fonksiyonları ve interlökin-8 değerlerinin prognoz üzerine etkisi]. Turk Kardiyoloji Dernegi Arsivi 2010;38(8):537-43. [PMID: ] [PubMed] [Google Scholar]
Burkhardt 2015
- Burkhardt BE, Rücker G, Stiller B. Prophylactic milrinone for the prevention of low cardiac output syndrome and mortality in children undergoing surgery for congenital heart disease. Cochrane Database of Systematic Reviews 2015, Issue 3. Art. No: CD009515. [DOI: 10.1002/14651858.CD009515.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Chaimani 2013
- Chaimani A, Higgins JP, Mavridis D, Spyridonos P, Salanti G. Graphical tools for network meta-analysis in STATA. PLOS One 2013;8(10):e76654. [DOI: 10.1371/journal.pone.0076654] [DOI] [PMC free article] [PubMed] [Google Scholar]
Charpie 2000
- Charpie JR, Dekeon MK, Goldberg CS, Mosca RS, Bove EL, Kulik TJ. Serial blood lactate measurements predict early outcome after neonatal repair or palliation for complex congenital heart disease. Journal of Thoracic and Cardiovascular Surgery 2000;120(1):73–80. [DOI: 10.1067/mtc.2000.106838] [DOI] [PubMed] [Google Scholar]
Cheema 2023
- Cheema HA, Khan AA, Ahmad AH, Khan AA, Khalid A, Shahid A, et al. Perioperative prophylactic corticosteroids for cardiac surgery in children: a systematic review and meta-analysis. American Heart Journal 2023;266:159-67. [DOI: 10.1016/j.ahj.2023.09.006] [DOI] [PubMed] [Google Scholar]
Delmo 2010
- Delmo WE, Alexi-Meskishvili V, Huebler M, Loforte A, Stiller B, Weng Y, et al. Extracorporeal membrane oxygenation for intraoperative cardiac support in children with congenital heart disease. Interactive Cardiovascular and Thoracic Surgery 2010;10(5):753–8. [DOI: 10.1510/icvts.2009.220475] [DOI] [PubMed] [Google Scholar]
Dias 2010
- Dias S, Welton NJ, Caldwell DM, Ades AE. Checking consistency in mixed treatment comparison meta-analysis. Statistics in Medicine 2010;29(7-8):932-44. [DOI: 10.1002/sim.3767] [DOI] [PubMed] [Google Scholar]
Du 2020
- Du X, Chen H, Song X, Wang S, Hao Z, Yin L, et al. Risk factors for low cardiac output syndrome in children with congenital heart disease undergoing cardiac surgery: a retrospective cohort study. BMC Pediatrics 2020;20(1):87. [DOI: 10.1186/s12887-020-1972-y] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011
- Sterne JA, Egger M, Moher D, editor(s). Chapter 10: Addressing reporting biases. In: Higgins JP, Green S editor(s). Cochrane Handbook for Systematic Reviews of Intervention. Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.handbook.cochrane.org.
Higgins 2012
- Higgins JP, Jackson D, Barrett JK, Lu G, Ades AE, White IR. Consistency and inconsistency in network meta-analysis: concepts and models for multi-arm studies. Research Synthesis Methods 2012;3(2):90-110. [DOI: 10.1002/jrsm.1044] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2017
- Higgins JP, Altman DG, Sterne JA, editor(s). Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Churchill R, Chandler J, Cumpston MS, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017). Cochrane, 2017. Available from www.training.cochrane.org/handbook.
Higgins 2019
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (updated July 2019). Cochrane, 2019. Available from www.training.cochrane.org/handbook. [DOI] [PMC free article] [PubMed]
Higgins 2023
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.4 (updated August 2023). Cochrane, 2023. Available from www.training.cochrane.org/handbook. [DOI] [PMC free article] [PubMed]
Hummel 2017
- Hummel J, Rücker G, Stiller B. Prophylactic levosimendan for the prevention of low cardiac output syndrome and mortality in paediatric patients undergoing surgery for congenital heart disease. Cochrane Database of Systematic Reviews 2017, Issue 8. Art. No: CD011312. [DOI: 10.1002/14651858.CD011312.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Hutton 2016
- Hutton B, Catalá-López F, Moher D. The PRISMA statement extension for systematic reviews incorporating network meta-analysis: PRISMA-NMA [La extensión de la declaración PRISMA para revisiones sistemáticas que incorporan metaanálisis en red: PRISMA-NMA]. Medicina Clinica 2016;147(6):262-6. [DOI: 10.1016/j.medcli.2016.02.025] [DOI] [PubMed] [Google Scholar]
Jackson 2014
- Jackson D, Barrett JK, Rice S, White IR, Higgins JP. A design-by-treatment interaction model for network meta-analysis with random inconsistency effects. Statistics in Medicine 2014;33(21):3639-54. [DOI: 10.1002/sim.6188] [DOI] [PMC free article] [PubMed] [Google Scholar]
Jacobs 2008
- Jacobs JP. Introduction--databases and the assessment of complications associated with the treatment of patients with congenital cardiac disease. Cardiology in the Young 2008;18 Suppl 2:1-37. [DOI: 10.1017/S104795110800334X] [DOI] [PubMed] [Google Scholar]
Jentzer 2015
- Jentzer JC, Coons JC, Link CB, Schmidhofer M. Pharmacotherapy update on the use of vasopressors and inotropes in the intensive care unit. Journal of Cardiovascular Pharmacology and Therapeutics 2015;20(3):249-60. [DOI: 10.1177/1074248414559838] [DOI] [PubMed] [Google Scholar]
König 2013
- König J, Krahn U, Binder H. Visualizing the flow of evidence in network meta-analysis and characterizing mixed treatment comparisons. Statistics in Medicine 2013;32(30):5414-29. [DOI: 10.1002/sim.6001] [DOI] [PubMed] [Google Scholar]
Lapere 2022
- Lapere M, Rega F, Rex S. Levosimendan in paediatric cardiac anaesthesiology: a systematic review and meta-analysis. European Journal of Anaesthesiology 2022;39(8):646-55. [DOI: 10.1097/EJA.0000000000001711] [DOI] [PubMed] [Google Scholar]
Lefebvre 2019
- Lefebvre C, Glanville J, Briscoe S, Littlewood A, Marshall C, Metzendorf M-I, et al. Chapter 4: Searching for and selecting studies. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.0 (updated July 2019). Cochrane, 2019. Available from www.training.cochrane.org/handbook.
Liberati 2009
- Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gotzsche PC, Ioannidis JP, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. Journal of Clinical Epidemiology 2009;62(10):e1-34. [DOI: 10.1016/j.jclinepi.2009.06.006] [DOI] [PubMed] [Google Scholar]
Liu 2023
- Liu Q, Wu X, Li Y, Wang H, An R, Dou D, et al. Effect of hemoglobin and oxygen saturation on adverse outcomes in children with tetralogy of fallot: a retrospective observational study. BMC Anesthesiology 2023;23(1):346. [DOI: 10.1186/s12871-023-02290-y] [DOI] [PMC free article] [PubMed] [Google Scholar]
Loomba 2021
- Loomba RS, Culichia C, Schulz K, Vogel M, Savorgnan F, Flores S, et al. Acute effects of vasopressin arginine infusion in children with congenital heart disease: higher blood pressure does not equal improved systemic oxygen delivery. Pediatric Cardiology 2021;42(8):1792-8. [DOI: 10.1007/s00246-021-02667-1] [DOI] [PubMed] [Google Scholar]
Luo 2018
- Luo D, Wan X, Liu J, Tong T. Optimally estimating the sample mean from the sample size, median, mid-range, and/or mid-quartile range. Statistical Methods in Medical Research 2018;27(6):1785-805. [DOI: 10.1177/0962280216669183] [DOI] [PubMed] [Google Scholar]
Mills 2012
- Mills EJ, Thorlund K, Ioannidis JP. Calculating additive treatment effects from multiple randomized trials provides useful estimates of combination therapies. Journal of Clinical Epidemiology 2012;65(12):1282–8. [DOI: 10.1016/j.jclinepi.2012.07.012] [DOI] [PubMed] [Google Scholar]
Nikolakopoulou 2020
- Nikolakopoulou A, Higgins JP, Papakonstantinou T, Chaimani A, Del Giovane C, Egger M, et al. CINeMA: an approach for assessing confidence in the results of a network meta-analysis. PLOS Medicine 2020;17(4):e1003082. [DOI: 10.1371/journal.pmed.1003082] [DOI] [PMC free article] [PubMed] [Google Scholar]
Nikolakopoulou 2023 [Computer program]
- nmarank: complex hierarchy questions in network meta-analysis. Nikolakopoulou A, Schwarzer G, Papakonstantinou T, Version 0.3-0. Comprehensive R Archive Network, 2023. CRAN.R-project.org/package=nmarank.
Oh 2019
- Oh J, Song IK, Cho J, Yun TJ, Park CS, Choi JM, et al. Acute change in ventricular contractility-load coupling after corrective surgery for congenital heart defect: a retrospective cohort study. Pediatric Cardiology 2019;40(8):1618-26. [DOI: 10.1007/s00246-019-02195-z] [DOI] [PMC free article] [PubMed] [Google Scholar]
Prichard 1991
- Prichard BN, Owens CW, Smith CC, Walden RJ. Heart and catecholamines. Acta Cardiologica 1991;46(3):309-22. [PMID: ] [PubMed] [Google Scholar]
R core team 2023 [Computer program]
- A language and environment for statistical computing. R Core Team, Version 4.3.1. Vienna, Austria: Foundation for Statistical Computing, 2023. www.R-project.org/. [URL: www.R-project.org/]
Rao 1996
- Rao V, Ivanov J, Weisel RD, Ikonomidis JS, Christakis GT, David TE. Predictors of low cardiac output syndrome after coronary artery bypass. Journal of Thoracic and Cardiovascular Surgery 1996;112(1):38-51. [DOI: 10.1016/s0022-5223(96)70176-9] [DOI] [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014. Available at revman.cochrane.org.
RevMan 2024 [Computer program]
- Review Manager (RevMan). Version 2.7.0 to 7.2.0. The Cochrane Collaboration, 2021-2024. Available at revman.cochrane.org.
Roeleveld 2018
- Roeleveld PP, Klerk JCA. The perspective of the intensivist on inotropes and postoperative care following pediatric heart surgery: an international survey and systematic review of the literature. World Journal of Pediatric and Congenital Heart Surgery 2018;9(1):10-21. [DOI: 10.1177/2150135117731725] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rücker 2012
- Rücker G. Network meta-analysis, electrical networks and graph theory. Research Synthesis Methods 2012;3(4):312–24. [DOI: 10.1002/jrsm.1058] [DOI] [PubMed] [Google Scholar]
Rücker 2014
- Rücker G, Schwarzer G. Reduce dimension or reduce weights? Comparing two approaches to multi-arm studies in network meta-analysis. Statistics in Medicine 2014;33(25):4353–69. [DOI: 10.1002/sim.6236] [DOI] [PubMed] [Google Scholar]
Rücker 2015
- Rücker G, Schwarzer G. Ranking treatments in frequentist network meta-analysis works without resampling methods. BMC Medical Research Methodology 2015;15:58. [DOI: 10.1186/s12874-015-0060-8] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rücker 2019
- Rücker G, Petropoulou M, Schwarzer G. Network meta-analysis of multicomponent interventions. Biometrical Journal 2020;62(3):808-21. [DOI: 10.1002/bimj.201800167] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rücker 2021a
- Rücker G, Schmitz S, Schwarzer G. Component network meta-analysis compared to a matching method in a disconnected network: a case study. Biometrical Journal 2021;63(2):447-61. [DOI: 10.1002/bimj.201900339] [DOI] [PubMed] [Google Scholar]
Rücker 2021b [Computer program]
- netmeta: network meta-analysis using frequentist methods. Rücker G, Krahn U, König J, Efthimiou O, Davies A, Papakonstantinou T, et al, Version 2.0-2. Comprehensive R Archive Network, 2021. github.com/guido-s/netmeta. [URL: github.com/guido-s/netmeta]
Salanti 2011
- Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. Journal of Clinical Epidemiology 2011;64(2):163-71. [DOI: 10.1016/j.jclinepi.2010.03.016] [DOI] [PubMed] [Google Scholar]
Schünemann 2013
- Schünemann H, Brożek J, Guyatt G, Oxman A. Introduction to GRADE Handbook. Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach. The GRADE Working Group, Updated October 2013. [URL: https://gdt.gradepro.org/app/handbook/handbook.html] [Google Scholar]
Shi 2008
- Shi S, Zhao Z, Liu X, Shu Q, Tan L, Lin R, et al. Perioperative risk factors for prolonged mechanical ventilation following cardiac surgery in neonates and young infants. Chest 2008;134(4):768-74. [DOI: 10.1378/chest.07-2573] [DOI] [PubMed] [Google Scholar]
Shore 2001
- Shore S, Nelson DP, Pearl JM, Manning PB, Wong H, Shanley TP, et al. Usefulness of corticosteroid therapy in decreasing epinephrine requirements in critically ill infants with congenital heart disease. American Journal of Cardiology 2001;88(5):591-4. [DOI: 10.1016/s0002-9149(01)01751-9] [DOI] [PubMed] [Google Scholar]
Stocker 2006
- Stocker CF. Recent developments in the perioperative management of the paediatric cardiac patient. Current Opinion in Anaesthesiology 2006;19(4):375-81. [DOI: 10.1097/01.aco.0000236135.77733.cd] [DOI] [PubMed] [Google Scholar]
Teng 2011
- Teng S, Kaufman J, Pan Z, Czaja A, Shockley H, da Cruz E. Continuous arterial pressure waveform monitoring in pediatric cardiac transplant, cardiomyopathy and pulmonary hypertension patients. Intensive Care Medicine 2011;37(8):1297–301. [DOI: 10.1007/s00134-011-2252-y] [DOI] [PubMed] [Google Scholar]
Tierney 2007
- Tierney JF, Stewart LA, Ghersi D, Burdett S, Sydes MR. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 2007;8:16. [DOI: 10.1186/1745-6215-8-16] [DOI] [PMC free article] [PubMed] [Google Scholar]
Turanlahti 2004
- Turanlahti M, Boldt T, Palkama T, Antila S, Lehtonen L, Pesonen E. Pharmacokinetics of levosimendan in pediatric patients evaluated for cardiac surgery. Pediatric Critical Care Medicine 2004;5(5):457-62. [DOI: 10.1097/01.pcc.0000137355.01277.9c] [DOI] [PubMed] [Google Scholar]
Ulate 2017
- Ulate KP, Yanay O, Jeffries H, Baden H, Di Gennaro JL, Zimmerman J. An elevated low cardiac output syndrome score is associated with morbidity in infants after congenital heart surgery. Pediatric Critical Care Medicine 2017;18(1):26-33. [DOI: 10.1097/PCC.0000000000000979] [DOI] [PubMed] [Google Scholar]
Veroniki 2018
- Veroniki AA, Straus SE, Rücker G, Tricco AC. Is providing uncertainty intervals in treatment ranking helpful in a network meta-analysis? Journal of Clinical Epidemiology 2018;100:122-9. [DOI: 10.1016/j.jclinepi.2018.02.009] [PMID: ] [DOI] [PubMed] [Google Scholar]
Wang 2023
- Wang P, Fu C, Bai G, Cuan L, Tang X, Jin C, et al. Risk factors of postoperative low cardiac output syndrome in children with congenital heart disease: a systematic review and meta-analysis. Frontiers in Pediatrics 2023;10:954427. [DOI: 10.3389/fped.2022.954427] [DOI] [PMC free article] [PubMed] [Google Scholar]
Warnes 2001
- Warnes CA, Liberthson R, Danielson GK, Dore A, Harris L, Hoffman JI, et al. Task force 1: the changing profile of congenital heart disease in adult life. Journal of the American College of Cardiology 2001;37(5):1170-5. [DOI: 10.1016/s0735-1097(01)01272-4] [DOI] [PubMed] [Google Scholar]
Weber 2019
- Weber C, Esser M, Eghbalzadeh K, Sabashnikov A, Djordjevic I, Maier J, et al. Levosimendan reduces mortality and low cardiac output syndrome in cardiac surgery. Thoracic and Cardiovascular Surgeon 2020;68(5):401-9. [DOI: 10.1055/s-0039-3400496] [DOI] [PubMed] [Google Scholar]
Welton 2009
- Welton NJ, Caldwell DM, Adamopoulos E, Vedhara K. Mixed treatment comparison meta-analysis of complex interventions: psychological interventions in coronary heart disease. American Journal of Epidemiology 2009;169(9):1158–65. [DOI: 10.1093/aje/kwp014] [DOI] [PubMed] [Google Scholar]
Wernovsky 1995
- Wernovsky G, Wypij D, Jonas RA, Mayer JE Jr, Hanley FL, Hickey PR, et al. Postoperative course and hemodynamic profile after the arterial switch operation in neonates and infants. A comparison of low-flow cardiopulmonary bypass and circulatory arrest. Circulation 1995;92(8):2226-35. [DOI: 10.1161/01.cir.92.8.2226] [DOI] [PubMed] [Google Scholar]
Yepes‐Nuñez 2019
- Yepes-Nuñez JJ, Li SA, Guyatt G, Jack SM, Brozek JL, Beyene J, et al. Development of the summary of findings table for network meta-analysis. Journal of Clinical Epidemiology 2019;115:1-13. [DOI: 10.1016/j.jclinepi.2019.04.018] [DOI] [PubMed] [Google Scholar]
Zhang 1991
- Zhang N. Management of postoperative arrhythmia after open heart surgery in complex congenital heart disease [复杂性先天性心脏病心内直视手术后早期的心律失常及处理]. Zhonghua Xin Xue Guan Bing Za Zhi 1991;19(2):91-3, 125. [PMID: ] [PubMed] [Google Scholar]
References to other published versions of this review
Burkhardt 2020
- Burkhardt BEU, Hummel J, Rücker G, Stiller B. Inotropes for the prevention of low cardiac output syndrome and mortality for paediatric patients undergoing surgery for congenital heart disease: a network meta‐analysis. Cochrane Database of Systematic Reviews 2020, Issue 8. Art. No: CD013707. [DOI: 10.1002/14651858.CD013707] [DOI] [PMC free article] [PubMed] [Google Scholar]


