Abstract
Objectives
The Pituitary Tumor Registry intends to provide a platform for clinical research and basic sciences, with an emphasis on disease outcomes.
Methods
In this retrospective cohort study, all patient data, including demographics, vital signs, symptoms and signs, medical history, medications, past drug history, paraclinical data, treatment modalities, post-surgery follow-up, treatment responsiveness, and pathology reports, have been gathered from the electronic patient records of the Endocrinology and Metabolism Research Center affiliated with Tehran University of Medical Sciences.
Results
A total of 200 patients with pituitary adenoma were identified. Acromegaly was the most prevalent adenoma, accounting for 35% (n = 70) of cases, followed by clinically nonfunctional pituitary adenoma (CNFPA) at 28.5% (n = 57), prolactinoma at 23% (n = 46), and Cushing disease at 13.5% (n = 27). All of the patients with Cushing disease had surgery, with trans-sphenoidal surgery accounting for 92.59%. Prolactinoma was mostly treated with medication (82.60% of cases). Post-operative complications were reported, including cerebrospinal fluid (CSF) leakage in CNFPA cases (24.13%, n = 7) and diabetes insipidus (DI) in acromegaly (13.46%, n = 7). Unfortunately, 5 patients died: two with acromegaly, two with CNFPA and one with Cushing disease.
Conclusions
This registry permits a comprehensive management, long term follow up and treatment outcomes of patients with pituitary adenomas.
Keywords: Pituitary adenoma, Pituitary tumors, Registry, Non-functional adenoma, Prolactinoma, Acromegaly, Cushing disease
Introduction
Once considered rare, pituitary adenomas are now showing an increased prevalence according to recent studies [1]. This rise may be attributed to improved access to imaging modalities such as magnetic resonance imaging (MRI) scans and more accurate biochemical testing. Despite these advances, its rate of prevalence remains uncertain [2]. Imaging findings and postmortem diagnostic assessments have shown a prevalence of pituitary adenomas ranging from 14.4 to 22.5%. [3]. The majority of the pituitary adenomas occur sporadically, with familial cases seen in only 5% of patients, mainly in younger individuals. Gender, age, race, and genetic factors are all associated with the development of various types of pituitary adenoma [4].
Pituitary adenoma could be categorized based on tumor size as macroadenomas (greater than 1 cm) and microadenomas (less than 1 cm). Additionally, these adenomas can be grouped based on their functional characteristics [5]. There are various types of pituitary adenomas, including prolactinoma (secreting prolactin), somatotroph adenoma (secreting growth hormone), corticotrope adenoma (secreting adrenocorticotropic hormone (ACTH)), gonadotroph adenoma (secreting gonadotropin hormones), thyrotrope adenoma (secreting thyroid stimulating hormone (TSH)), and clinically nonfunctional pituitary adenoma (CNFPA) [6]. Adenomas are usually diagnosed through clinical symptoms caused by the increased pituitary hormone secretion, decreased secretion of other pituitary hormones, or symptoms caused by tumor mass effect [1]. Clinical presentations such as headaches, visual field defects, seizures, hydrocephalus, and apoplexy may be accompanied by pituitary adenomas [4].
Patients with pituitary adenomas require a comprehensive evaluation of pituitary hormones. Diagnostic approaches such as MRI imaging, stimulation and inhibition testing, and immunocytochemical techniques are all used for an accurate diagnosis [7]. Early diagnosis of these patients helps select the most suitable treatment approach. For instance, prolactinoma may be managed with dopamine agonist drugs, while other adenomas often require trans-sphenoidal surgery or radiotherapy [7, 8].
The pituitary tumor registry can be used to estimate prevalence, clinical evaluations, remission, recurrence rate, and mortality rate of these disorders. Moreover, it can also provide a platform for clinical research and basic science, focusing on disease outcomes [9].
Methods
This retrospective cohort study was conducted at Dr. Shariati Hospital, a tertiary care center in Tehran, Iran. The web-based pituitary tumor registry was established by the Endocrinology and Metabolism Research Center in Dr. Shariati hospital affiliated to Tehran University of Medical Sciences. The complete protocol for this registry has been published elsewhere [9]. The study population included individuals aged 18 and above referred to Dr. Shariati hospital with clinical symptoms of pituitary adenomas (Acromegaly, Cushing disease, Prolactinoma, and clinically nonfunctional pituitary adenoma (CNFPA)), confirmed by laboratory tests and pituitary imaging. Patients diagnosed with Rathke’s cleft cyst, Craniopharyngioma, pituitary cyst, Meningiomas, or lipomas were excluded. Moreover, those with incomplete information and loss to follow up were excluded from this analysis. Patients were included in the registry upon obtaining informed consent and permission for registry participation. All patient data, including demographics, vital signs, symptoms and signs, medical history, medications, past drug history, paraclinical data, treatment modalities, post-surgery follow-up, treatment responsiveness, and pathology reports, were documented in the electronic patient records of Dr. Shariati hospital and was compiled by the Endocrinology and Metabolism Research Center based at this hospital. Patients were regularly monitored for both clinical and biochemical changes at the intervals of three to six months.
The ethics committee of Endocrinology and Metabolism Research Institute reviewed and accepted the study protocol (ethical code: IR.TUMS.EMRI.REC.1402.019).
Definitions
Pituitary adenomas were classified into clinically nonfunctional or functional types. Functional pituitary adenomas were further divided into prolactinomas, growth hormone (GH)-secreting tumors, and ACTH-secreting tumors. Tumor size was determined by MRI, with the highest tumor diameter recorded. Microadenomas were defined as pituitary adenomas with a diameter of less than 10 mm, while macroadenomas were those with a diameter of 10 mm or more. Adenoma excision was considered complete in case of resecting at least 80% of the maximum tumor size. Central hypogonadism were identified as erectile dysfunction, decreased libido, low testosterone level, and low gonadotropin in men, and oligo-amenorrhea with low gonadotropin and low estradiol in women.
The original protocol thoroughly outlines the definition of remission for each adenoma [9]. Permanent diabetes insipidus (PDI) was identified by the need for long-term desmopressin treatment, confirmed in follow-up records at least two weeks post-surgery.
Statistical analysis
Data were presented as mean ± standard deviation (SD) or as frequency and proportion, as appropriate. All statistical analyses were performed using IBM SPSS Statistics (Version 23).
Results
Clinical characteristics of the registered patients
A total of 200 individuals diagnosed with pituitary adenoma were registered from April 2009 to March 2022. The mean age at the time of diagnosis was 42.79 ± 12.53 years in patients with acromegaly, 36.54 ± 12.13 years in patients with prolactinoma, 45.73 ± 12.94 years among those with CNFPA, and 32.25 ± 10.26 years among those with Cushing disease (Figure 1). Acromegaly patients often presented with acral enlargement and frontal bossing (both 81.43%, n = 57) as characteristic signs of GH excess, followed by headache (58.57%, n = 41). Cushing disease patients frequently reported fatigue and centripetal obesity, along with striae (stretch marks), buffalo hump, and moon face as distinctive features. Over half of Cushing disease patients had a history of hypertension (42.30%, n = 11). Prolactinoma patients primarily presented with headache (65.21%, n = 30), galactorrhea (41.30%, n = 19), and hypogonadal symptoms (41.3%, n = 19). In CNFPA cases, headache was the most prevalent symptom (48.14%, n = 26). Table 1 displays the demographic and clinical characteristics of the patients.
Fig. 1.
Age of the participant based on their tumor type
Table 1.
Demographics and clinical presentations
| Acromegaly, 70 (35%) | Cushing, 27 (13.5%) | Prolactinoma, 46 (23%) | CNFPA, 57 (28.5%) | |
|---|---|---|---|---|
| Gender (M/ F) | 27/43 (38.54/61.43) | 6/21 (22.22/77.77) | 13/33 (28.26/71.73) |
35/22 (61.40/38.59) |
| Age at diagnosis |
42.79± 12.53 |
32.25± 10.26 |
36.54± 12.13 |
45.73± 12.94 |
| Smoking | 3(4.28) | 0(0) | 1(2.17) | 5(8.77) |
| BMI | 27.40 ± 5.32 | 31.06 ± 4.89 | 26.79 ± 4.33 | 29.56 ± 4.85 |
|
Menstrual cycle abnormality Oligomenorrhea Amenorrhea |
2 (4.65) 11 (25.58) |
2(9.52) 8(38.09) |
5(15.15) 12 (36.36) |
2(9.52) 3(14.28) |
| Headache | 41 (58.57) | 8(30.76) | 30(65.21) | 26(48.14) |
| Hypogonadal symptoms | 13(18.57) | 6(24) | 19(41.3) | 13(24.52) |
| Visual field deficit | 17(24.29) | 1(4) | 4(8.88) | 5(9.43) |
| Galactorrhea | 6(8.57) | - | 19(41.30) | 6(10.90) |
| Acral enlargement | 57 (81.43) | - | - | - |
| Proximal myopathy | 12(17.14) | 14(56) | - | 1(1.88) |
| Arthralgia | 12(17.14) | - | - | 2(3.84) |
| Fatigue | 8 (11.43) | 19(76) | 3(7.31) | 10(18.84) |
| Buffalo hump | - | 21(80.76) | - | - |
| Moon face | - | 19(76) | - | - |
| Striae | - | 19(73.07) | - | - |
| Centripetal Obesity | - | 22(81.48) | - | - |
| Easy bruising | - | 8(33.33) | - | 0 |
| Frontal bossing | 57(81.43) | - | - | 0 |
| Colon cancer/ Polyp | 8(11.43) | - | - | - |
| Thyroid pathology | 10(14.29) | - | - | - |
|
PMH Diabetes Cardiac discords HTN |
14(20) 5(7.14) 12 (17.14) |
8(38.09) 0 11(42.30) |
1(2.22) 1(2.22) 2(4.44) |
1(2.17) 0 3(6.52) |
Data are presented as number (percentage)
CNFPA: Clinically nonfunctional pituitary adenoma, PMH: Past medical history, HTN: Hypertension, BMI: Body mass index, M: Male, F: Female
Distribution of tumors among registered patients
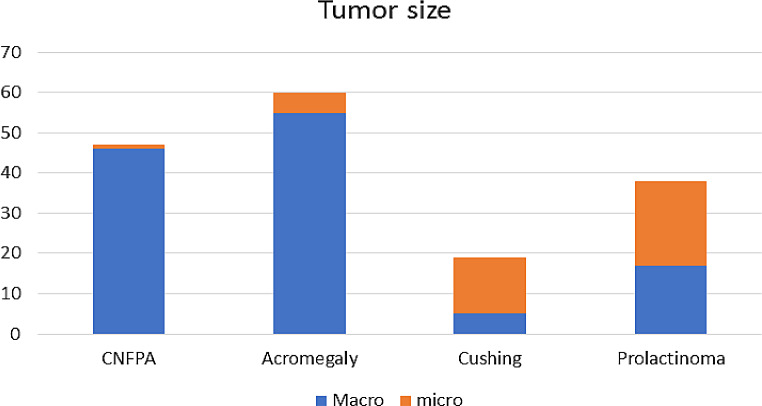
Acromegaly was the most common tumor, accounting for 35% (n = 70) of cases, followed by CNFPA at 28.5% (n = 57), prolactinoma at 23% (n = 46), and Cushing disease at 13.5% (n = 27). Acromegaly, Cushing disease, and prolactinoma were more prevalent in women (61.43%, 77.77%, and 71.73%, respectively), while CNFPA was more common in men (61.40%). Macroadenomas accounted for 97.87% of CNFPA cases, followed by acromegaly (90.91%), prolactinoma (44.73%), and Cushing disease (26.31%) (Figure 2). Additionally, optic nerve invasion was mostly observed in CNFPA cases (78.94%), followed by cavernous sinus invasion in Cushing disease and prolactinoma (66.66% and 50%, respectively).
Fig. 2.
Tumors based on their size
Therapeutic modalities and clinical outcomes
Therapeutic modalities included medication and surgery are presented in (Table 2). All of the patients with Cushing disease underwent trans-sphenoidal surgery (TSS) followed by acromegaly patients (84.12%, n = 53) and CNFPA (73.46%, n = 36). Prolactinoma was primarily treated with medication (82.60%, n = 38). Post-surgical complications were reported, including CSF leakage in CNFPA cases (24.13%, n = 7) and diabetes insipidus (DI) in acromegaly (13.46%, n = 7). Unfortunately, five patients (two with acromegaly, two with CNFPA and 1 with Cushing disease) died.
Table 2.
Therapeutic modalities and clinical outcomes
| Acromegaly | Cushing | Prolactinoma | CNFPA | |
|---|---|---|---|---|
|
Treatment modality No surgery TSS Trans-cranial |
10(15.87) 51(80.95) 2(3.17) |
0(0) 27(100) 0(0) |
38(82.60) 8(17.39) 0 |
13(26.53) 33(67.34) 3(6.12) |
|
Resection Total Partial |
17(32.08) 16(30.19) |
17 (70.83) 1(4.16) |
0 2(25) |
7(21.21) 9(27.27) |
|
Surgery complication CSF Leak Bleeding Cranial nerve injury Meningitis DI other |
4(7.69) 0 0 1(1.92) 7(13.46) 2(3.85) |
0 0 0 0 2(9.09) 0 |
0 0 0 0 1(12.5) 0 |
7(24.13) 1(3.44) 0 1(3.44) 0 0 |
|
Response Remission |
22(52.38) | 13(68.42) | 12(40) | 6(27.27) |
| Recurrence | 2(4.76) | 3(18.75) | 1(8.33) | 2(12.5) |
|
Tumor size Micro Macro |
5(9.09) 55(90.91) |
14(73.68) 5(26.31) |
21(55.26) 17(44.73) |
1(2.12) 46(97.87) |
|
Invasive Cavernous sinus Optic chiasma Sphenoid sinus |
15(46.87) 16(50) 4(12.5) |
2(66.66) 1(33.33) 0 |
3(50) 3(50) 0 |
7(36.84) 15(78.94) 6(31.57) |
| Death | 2(2.85) | 1(3.70) | 0 | 2(3.5) |
CNFPA: Clinically nonfunctional pituitary adenoma, TSS: Transsphenoidal surgery, CSF: Cerebrospinal fluid, DI: Diabetes insipidus
Of the total 70 patients diagnosed with acromegaly, 51 (80.95%) underwent TSS. Immunohistochemical (IHC) reports were available for 29 patients with acromegaly, revealing six cases of mammosomatotroph adenoma, three cases of sparsely granulated somatotroph adenoma, two cases of densely granulated somatotroph adenoma, and one case of sparsely granulated somatotroph adenoma. Six patients had a history of radiotherapy, and a total of 22 (52.38%) achieved remission. There were two recurrences (4.76%) and two deaths (2.85%).
For individuals with Cushing disease, surgical resection was conducted on 27 patients, yet only 13 of them experienced remission. Post-surgical complications included CSF leakage in one patient (3.70%). There were three recurrences (18.75%) and one death (3.70%).
For prolactinoma, surgery was not pursued for 82.60% of the patients, and remission was attained by only 12 patients. There was one recurrence (8.33%) and no deaths.
In CNFPA cases, 67.34% underwent TSS, with remission achieved in six patients (27.27%). Post-surgical complications included CSF leakage in seven patients (24.13%). There were two recurrences (12.5%) and two deaths (3.5%).
Discussion
Tumor registries serve as a valuable resource for investigating cancer epidemiology, analyzing treatment approaches, and evaluating long-term clinical results. The objective of this study was to present data on the epidemiology, clinical manifestations, and biochemical features, along with therapeutic approaches and clinical prognosis of the predominant pituitary adenoma. Using the prototype database program, we conducted an analysis focusing on the four most common types of pituitary adenomas: CNFPA, GH-secreting adenoma (acromegaly), ACTH-secreting adenomas (Cushing disease), and prolactinomas.
Overall, acromegaly emerged as the most prevalent condition among patients in this registry, followed by CNFPA, prolactinomas, and Cushing disease. This finding is notable for its deviation from previous reports in other studies. Typically, previous population-based studies identified prolactinomas as the most common tumor, followed by CNFPA, acromegaly, and Cushing disease [10–13]. However, it’s worth noting that some reports from specialized referral centers have shown similar results [14]. This discrepancy highlights the importance of registry data in capturing real-world epidemiological trends, potentially influenced by referral patterns and population characteristics.
In examining the gender factor of adenomas, there were several points worth considering. First, results from the study indicated a predominance of females among prolactinoma and Cushing disease cases which was in line with other reports [15, 16]. These findings indicate a potential association between the female gender and adenoma promotion in prolactinomas, possibly mediated by the estrogen hormone, which stimulates lactotroph proliferation and PRL gene transcription [17, 18]. However, it is likely that additional factors beyond gender likely play a role in determining tumor invasiveness. Furthermore, some studies suggest that the gender difference in Cushing disease may be caused by sexual dysmorphism [19]. Secondly, as previously mentioned [20], the most significant difference in gender distribution seemed to correspond with the peak age occurrence of each adenoma type. Thirdly, the higher female-to-male ratio observed in the acromegaly and prolactinoma microadenoma group may indicate gender-related disparities in patient healthcare-seeking behavior.
When analyzing the predominant clinical symptoms associated with each adenoma, it is evident that in acromegaly, approximately 80% of patients exhibit manifestations primarily linked to morphological alterations. Likewise, Cushing disease often exhibit symptoms associated with hormone excesses, such as myopathy, buffalo hump, moon face, striae, and central obesity, which aligns with findings from previous studies [21, 22]. Conversely, in prolactinoma and CNFPA, symptoms related to mass effect are more common, whereas having a headache is the most common clinical complaint in these adenomas [23, 24]. Headaches associated with pituitary adenomas can be attributed to various factors, including adenoma location-related aspects such as the compression of the optic chiasma, increased intrasellar pressure, and the invasion of cavernous sinus. While headaches can be induced by the functional impacts of hormones or influenced by other factors, further investigation is required to elucidate these mechanisms fully [25].
In the evaluation of treatments, surgical intervention combined with drug therapy is considered as the primary approach for acromegaly and CNFPA. A commonly reported complication post-surgery was DI, probably occurring due to injury to magnocellular neurons in the hypothalamus responsible for arginine vasopressin production [26]. Surprisingly, there was a higher prevalence of CSF leakages post-surgery compared to previous studies [27], possibly due to the complexity of these cases and the possible reason that most of these adenomas were macroadenomas. Similar to previous studies [14–16], surgical approaches were more frequently employed in the management of Cushing disease in this investigation. Regarding prolactinomas, dopamine agonists were the preferred treatment modality. Additionally, our study noted a higher prevalence of remission among patients with microadenomas.
The overall remission rate across was low, possibly due to the fact that our data collection focused mainly on a tertiary center where more complex cases are referred from other medical providers. Finally, given the significant impact of surgeons’ expertise in managing these diseases, it highlights the need for comprehensive training for neurosurgeons interested in this specific field.
The first limitation of this investigation involves its design as a retrospective observational study. Additionally, a portion of the data concerning the tumor characteristics and therapies was inaccessible. Moreover, the compilation of data solely from a singular referral center has influenced the outcomes considering the complexity of the cases and population characteristics. Finally, there is a possibility that some patients have remained unidentified due to potential inaccuracies in the system’s guidelines for diagnoses.
Conclusion
In conclusion, this pituitary tumor registry program was established to gather comprehensive information on patients with these adenomas. This registry has the potential to evaluate the effectiveness of long-term therapeutic approaches, and identify prognostic factors for recurrence. The heightened prevalence of pituitary adenomas emphasizes the critical necessity for enhanced education and awareness among all healthcare providers, alongside the proper allocation of resources.
Funding
Not applicable.
Data availability
Data will be made available on requests.
Declarations
Ethical approval
The ethics committee of Endocrinology and Metabolism Research Institute reviewed and accepted the study protocol (ethical code: IR.TUMS.EMRI.REC.1402.019).
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Shahrzad Mohseni and Niloofar Mirdamadi contributed equally to this work.
References
- 1.Aflorei ED, Korbonits M. Epidemiology and etiopathogenesis of pituitary adenomas. J Neurooncol. 2014;117(3):379–94. 10.1007/s11060-013-1354-5. [DOI] [PubMed] [Google Scholar]
- 2.Daly AF, Rixhon M, Adam C, Dempegioti A, Tichomirowa MA, Beckers A. High prevalence of Pituitary adenomas: a cross-sectional study in the Province of Liège, Belgium. J Clin Endocrinol Metabolism. 2006;91(12):4769–75. 10.1210/jc.2006-1668. [DOI] [PubMed] [Google Scholar]
- 3.Khamseh ME, Tehrani MRM, Mousavi Z, Malek M, Imani M, Tehrani NH, et al. Iran pituitary tumor registry: description of the program and initial results. Arch Iran Med. 2017;20(12):746–51. [PubMed] [Google Scholar]
- 4.Araujo-Castro M, Berrocal VR, Pascual-Corrales E. Pituitary tumors: epidemiology and clinical presentation spectrum. Hormones. 2020;19(2):145–55. 10.1007/s42000-019-00168-8. [DOI] [PubMed] [Google Scholar]
- 5.Ezzat S, Asa SL, Couldwell WT, Barr CE, Dodge WE, Vance ML, et al. The prevalence of pituitary adenomas: a systematic review. Cancer: Interdisciplinary Int J Am Cancer Soc. 2004;101(3):613–9. [DOI] [PubMed] [Google Scholar]
- 6.Arafah B, Nasrallah M. Pituitary tumors: pathophysiology, clinical manifestations and management. Endocrine-related Cancer. 2001;8(4):287–305. [DOI] [PubMed] [Google Scholar]
- 7.Chanson P, Salenave S. Diagnosis and treatment of pituitary adenomas. Minerva Endocrinol. 2004;29(4):241–75. [PubMed] [Google Scholar]
- 8.Molitch ME. Diagnosis and treatment of pituitary adenomas: a review. JAMA. 2017;317(5):516–24. [DOI] [PubMed] [Google Scholar]
- 9.Mohseni S, Asgari A-M, Saeedi S, Ostovar A, Bandarian F, Pejman Sani M, et al. Pituitary tumor registry: a multidisciplinary program protocol. J Diabetes Metabolic Disorders. 2023;22(2):1801–4. 10.1007/s40200-023-01306-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fontana E, Gaillard R. [Epidemiology of pituitary adenoma: results of the first Swiss study]. Rev Med Suisse. 2009;5(223):2172–4. [PubMed] [Google Scholar]
- 11.Daly AF, Rixhon M, Adam C, Dempegioti A, Tichomirowa MA, Beckers A. High prevalence of pituitary adenomas: a cross-sectional study in the province of Liege, Belgium. J Clin Endocrinol Metab. 2006;91(12):4769–75. 10.1210/jc.2006-1668. [DOI] [PubMed] [Google Scholar]
- 12.Fernandez A, Karavitaki N, Wass JA. Prevalence of pituitary adenomas: a community-based, cross-sectional study in Banbury (Oxfordshire, UK). Clin Endocrinol (Oxf). 2010;72(3):377–82. 10.1111/j.1365-2265.2009.03667.x. [DOI] [PubMed] [Google Scholar]
- 13.Gruppetta M, Mercieca C, Vassallo J. Prevalence and incidence of pituitary adenomas: a population based study in Malta. Pituitary. 2013;16(4):545–53. 10.1007/s11102-012-0454-0. [DOI] [PubMed] [Google Scholar]
- 14.Drange MR, Fram NR, Herman-Bonert V, Melmed S. Pituitary Tumor Registry: a Novel Clinical Resource1. J Clin Endocrinol Metabolism. 2000;85(1):168–74. 10.1210/jcem.85.1.6309. [DOI] [PubMed] [Google Scholar]
- 15.Khamseh ME, Mohajeri Tehrani MR, Mousavi Z, Malek M, Imani M, Hoshangian Tehrani N, et al. Iran Pituitary Tumor Registry: description of the program and initial results. Arch Iran Med. 2017;20(12):746–51. [PubMed] [Google Scholar]
- 16.Kwancharoen R, Deerochanawong C, Peerapatdit T, Salvatori R. Pituitary adenomas registry in Thailand. J Clin Neurosci. 2023;115:138–47. 10.1016/j.jocn.2023.07.026. [DOI] [PubMed] [Google Scholar]
- 17.Amara JF, Van Itallie C, Dannies PS. Regulation of prolactin production and cell growth by estradiol: difference in sensitivity to estradiol occurs at level of messenger ribonucleic acid accumulation. Endocrinology. 1987;120(1):264–71. 10.1210/endo-120-1-264. [DOI] [PubMed] [Google Scholar]
- 18.Lloyd HM, Meares JD, Jacobi J. Effects of oestrogen and bromocryptine on in vivo secretion and mitosis in prolactin cells. Nature. 1975;255(5508):497–8. 10.1038/255497a0. [DOI] [PubMed] [Google Scholar]
- 19.Pecori Giraldi F, Cassarino MF, Sesta A, Terreni M, Lasio G, Losa M. Sexual dimorphism in Cellular and Molecular features in human ACTH-Secreting pituitary adenomas. Cancers (Basel). 2020;12(3). 10.3390/cancers12030669. [DOI] [PMC free article] [PubMed]
- 20.Mindermann T, Wilson CB. Age-related and gender-related occurrence of pituitary adenomas. Clin Endocrinol (Oxf). 1994;41(3):359–64. 10.1111/j.1365-2265.1994.tb02557.x. [DOI] [PubMed] [Google Scholar]
- 21.Caron P, Brue T, Raverot G, Tabarin A, Cailleux A, Delemer B, et al. Signs and symptoms of acromegaly at diagnosis: the physician’s and the patient’s perspectives in the ACRO-POLIS study. Endocrine. 2019;63(1):120–9. 10.1007/s12020-018-1764-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kirk LF Jr., Hash RB, Katner HP, Jones T. Cushing’s disease: clinical manifestations and diagnostic evaluation. Am Fam Physician. 2000;62(5):1119–27. 33– 4. [PubMed] [Google Scholar]
- 23.Auriemma RS, Pirchio R, Pivonello C, Garifalos F, Colao A, Pivonello R. Approach to the patient with Prolactinoma. J Clin Endocrinol Metabolism. 2023;108(9):2400–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Iglesias P, Arcano K, Triviño V, García-Sancho P, Díez JJ, Villabona C, et al. Prevalence, clinical features, and natural history of incidental clinically non-functioning pituitary adenomas. Horm Metab Res. 2017;49(09):654–9. [DOI] [PubMed] [Google Scholar]
- 25.Gondim JA, de Almeida JP, de Albuquerque LA, Schops M, Gomes E, Ferraz T. Headache associated with pituitary tumors. J Headache Pain. 2009;10(1):15–20. 10.1007/s10194-008-0084-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schreckinger M, Szerlip N, Mittal S. Diabetes insipidus following resection of pituitary tumors. Clin Neurol Neurosurg. 2013;115(2):121–6. 10.1016/j.clineuro.2012.08.009. [DOI] [PubMed] [Google Scholar]
- 27.Slot EMH, Sabaoglu R, Voormolen EHJ, Hoving EW, van Doormaal TPC. Cerebrospinal fluid leak after transsphenoidal surgery: a systematic review and Meta-analysis. J Neurol Surg B Skull Base. 2022;83(Suppl 2):e501–13. 10.1055/s-0041-1733918. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on requests.