Abstract
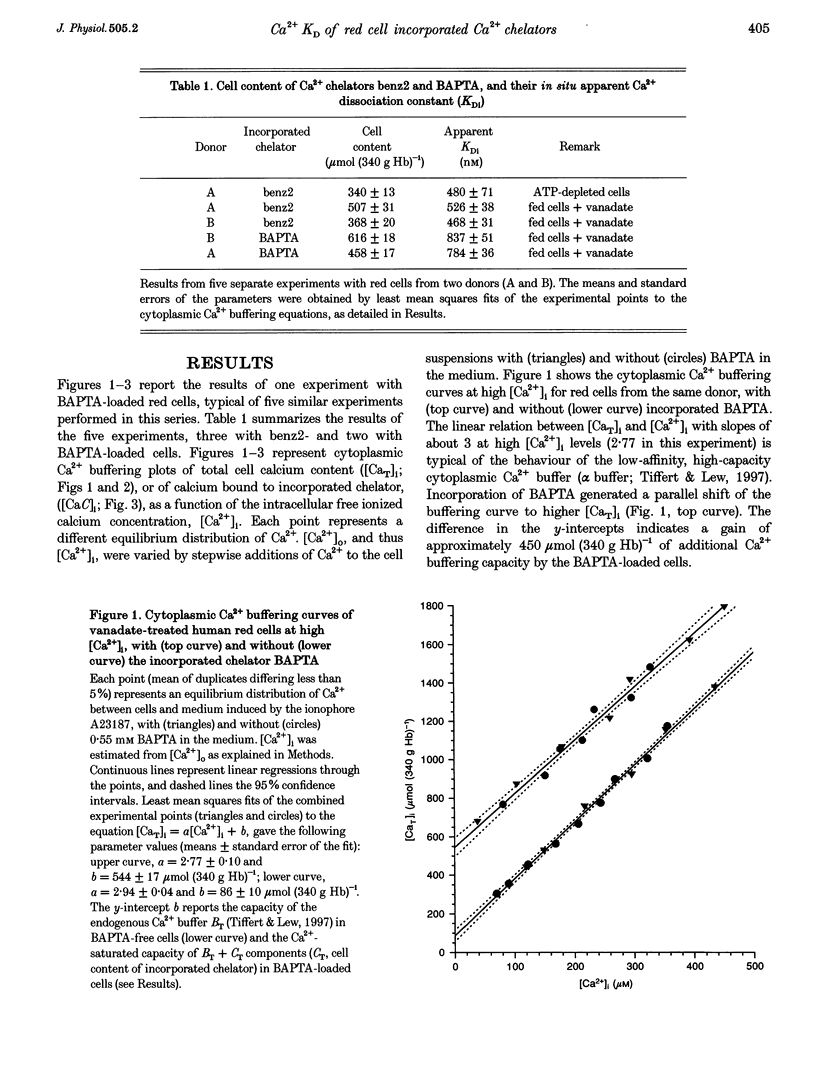
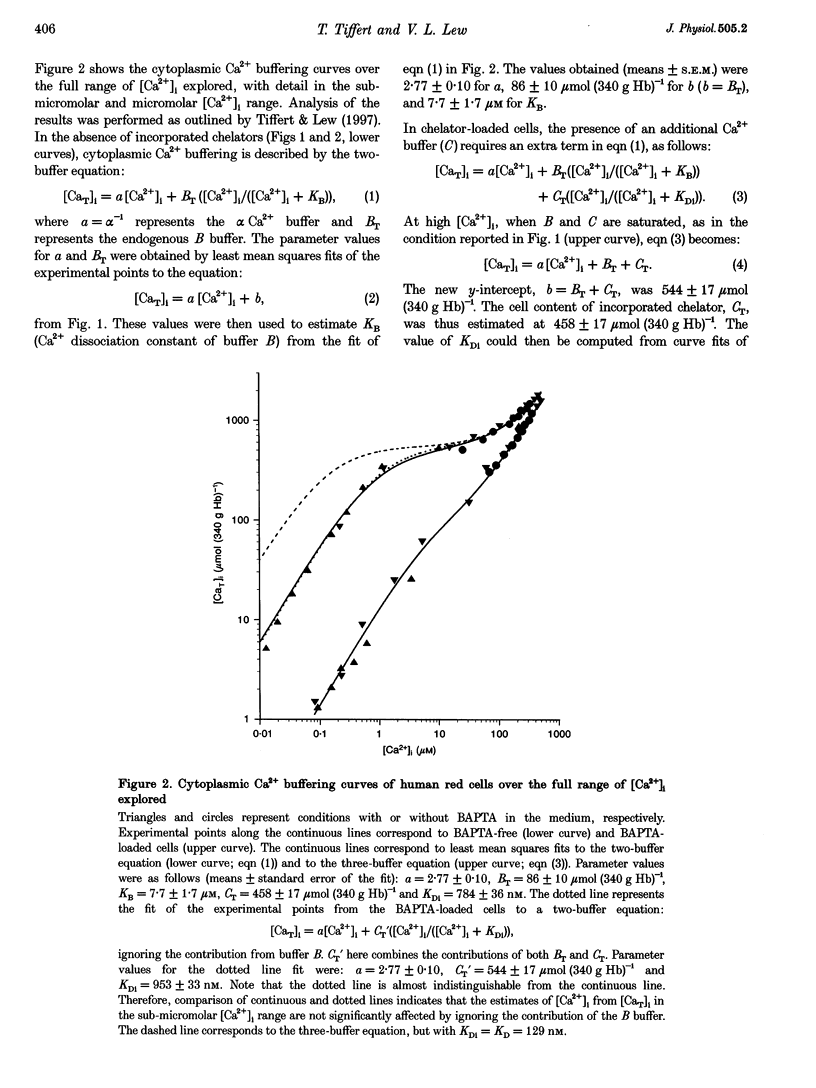
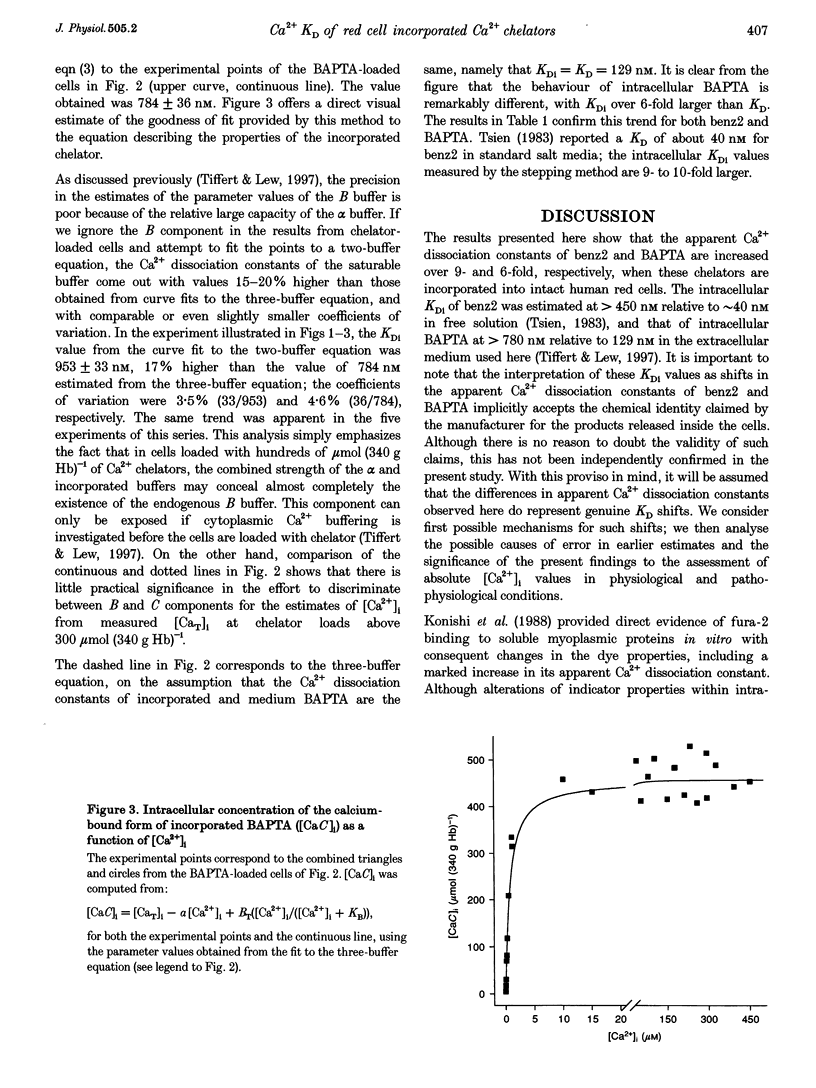
1. A recently developed method of measuring cytoplasmic Ca2+ buffering in intact red cells was applied to re-evaluate the intracellular Ca2+ binding properties of the Ca2+ chelators benz2 and BAPTA. Incorporation of the free chelators was accomplished by incubating the cells with the acetoxymethyl ester forms (benz2 AM or BAPTA AM). The divalent cation ionophore A23187 was used to induce equilibrium distribution of Ca2+ between cells and medium. 45Ca2+ was added stepwise to cell suspensions in the presence and absence of external BAPTA. To induce full Ca2+ equilibration, the plasma membrane Ca2+ pump was inhibited either by depleting the cells of ATP or by adding vanadate to the cell suspension. 2. The properties of the incorporated chelators were assessed from the difference in cytoplasmic Ca2+ buffering between chelator-free and chelator-loaded cells, over a wide range of intracellular ionized calcium concentrations ([Ca2+]i), from nanomolar to millimolar. 3. Under the experimental conditions applied, incorporation of benz2 and BAPTA into the red cells increased their Ca2+ buffering capacity by 300-600 mumol (340 g Hb)-1. The intracellular apparent Ca2+ dissociation constants (KDi) were about 500 nM for benz2 and 800 nM for BAPTA, values much higher than those reported for standard salt solutions (KD) of about 40 and 130 nM, respectively. These results suggest that, contrary to earlier observations, the intracellular red cell environment may cause large shifts in the apparent Ca2+ binding behaviour of incorporated chelators. 4. The possibility that the observed KD shifts are due to reversible binding of the chelators to haemoglobin is considered, and the implications of the present results for early estimates of physiological [Ca2+]i levels is discussed.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adovelande J., Bastide B., Délèze J., Schrével J. Cytosolic free calcium in Plasmodium falciparum-infected erythrocytes and the effect of verapamil: a cytofluorimetric study. Exp Parasitol. 1993 May;76(3):247–258. doi: 10.1006/expr.1993.1030. [DOI] [PubMed] [Google Scholar]
- Alvarez J., García-Sancho J., Herreros B. All or none cell responses of Ca2+-dependent K channels elicited by calcium or lead in human red cells can be explained by heterogeneity of agonist distribution. J Membr Biol. 1988 Sep;104(2):129–138. doi: 10.1007/BF01870925. [DOI] [PubMed] [Google Scholar]
- Barrabin H., Garrahan P. J., Rega A. F. Vanadate inhibition of the Ca2+-ATPase from human red cell membranes. Biochim Biophys Acta. 1980 Aug 14;600(3):796–804. doi: 10.1016/0005-2736(80)90482-4. [DOI] [PubMed] [Google Scholar]
- Bond G. H., Hudgins P. M. Inhibition of red cell Ca2+-ATPase by vanadate. Biochim Biophys Acta. 1980 Aug 14;600(3):781–790. doi: 10.1016/0005-2736(80)90480-0. [DOI] [PubMed] [Google Scholar]
- David-Dufilho M., Montenay-Garestier T., Devynck M. A. Fluorescence measurements of free Ca2+ concentration in human erythrocytes using the Ca2+-indicator fura-2. Cell Calcium. 1988 Aug;9(4):167–179. doi: 10.1016/0143-4160(88)90021-8. [DOI] [PubMed] [Google Scholar]
- Eilers R. J. Notification of final adoption of an international method and standard solution for hemoglobinometry specifications for preparation of standard solution. Am J Clin Pathol. 1967 Feb;47(2):212–214. doi: 10.1093/ajcp/47.2.212. [DOI] [PubMed] [Google Scholar]
- Etzion Z., Tiffert T., Bookchin R. M., Lew V. L. Effects of deoxygenation on active and passive Ca2+ transport and on the cytoplasmic Ca2+ levels of sickle cell anemia red cells. J Clin Invest. 1993 Nov;92(5):2489–2498. doi: 10.1172/JCI116857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferreira H. G., Lew V. L. Use of ionophore A23187 to measure cytoplasmic Ca buffering and activation of the Ca pump by internal Ca. Nature. 1976 Jan 1;259(5538):47–49. doi: 10.1038/259047a0. [DOI] [PubMed] [Google Scholar]
- García-Sancho J. Pyruvate prevents the ATP depletion caused by formaldehyde or calcium-chelator esters in the human red cell. Biochim Biophys Acta. 1985 Feb 28;813(1):148–150. doi: 10.1016/0005-2736(85)90357-8. [DOI] [PubMed] [Google Scholar]
- Glynn I. M., Lew V. L. Synthesis of adenosine triphosphate at the expense of downhill cation movements in intact human red cells. J Physiol. 1970 Apr;207(2):393–402. doi: 10.1113/jphysiol.1970.sp009068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konishi M., Olson A., Hollingworth S., Baylor S. M. Myoplasmic binding of fura-2 investigated by steady-state fluorescence and absorbance measurements. Biophys J. 1988 Dec;54(6):1089–1104. doi: 10.1016/S0006-3495(88)83045-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kramer R., Ginsburg H. Calcium transport and compartment analysis of free and exchangeable calcium in Plasmodium falciparum-infected red blood cells. J Protozool. 1991 Nov-Dec;38(6):594–601. [PubMed] [Google Scholar]
- Kurebayashi N., Harkins A. B., Baylor S. M. Use of fura red as an intracellular calcium indicator in frog skeletal muscle fibers. Biophys J. 1993 Jun;64(6):1934–1960. doi: 10.1016/S0006-3495(93)81564-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lew V. L., García-Sancho J. Measurement and control of intracellular calcium in intact red cells. Methods Enzymol. 1989;173:100–112. doi: 10.1016/s0076-6879(89)73008-1. [DOI] [PubMed] [Google Scholar]
- Lew V. L. On the ATP dependence of the Ca 2+ -induced increase in K + permeability observed in human red cells. Biochim Biophys Acta. 1971 Jun 1;233(3):827–830. doi: 10.1016/0005-2736(71)90185-4. [DOI] [PubMed] [Google Scholar]
- Lew V. L., Tsien R. Y., Miner C., Bookchin R. M. Physiological [Ca2+]i level and pump-leak turnover in intact red cells measured using an incorporated Ca chelator. Nature. 1982 Jul 29;298(5873):478–481. doi: 10.1038/298478a0. [DOI] [PubMed] [Google Scholar]
- Lindner A., Hinds T. R., Davidson R. C., Vincenzi F. F. Increased cytosolic free calcium in red blood cells is associated with essential hypertension in humans. Am J Hypertens. 1993 Sep;6(9):771–779. doi: 10.1093/ajh/6.9.771. [DOI] [PubMed] [Google Scholar]
- Muallem S., Miner C., Seymour C. A. The nature of the Ca2+-pump defect in the red blood cells of patients with cystic fibrosis. Biochim Biophys Acta. 1985 Sep 25;819(1):143–147. doi: 10.1016/0005-2736(85)90205-6. [DOI] [PubMed] [Google Scholar]
- Murphy E., Berkowitz L. R., Orringer E., Levy L., Gabel S. A., London R. E. Cytosolic free calcium levels in sickle red blood cells. Blood. 1987 May;69(5):1469–1474. [PubMed] [Google Scholar]
- Rhoda M. D., Galacteros F., Beuzard Y., Giraud F. Ca2+ permeability and cytosolic Ca2+ concentration are not impaired in beta-thalassemic and hemoglobin C erythrocytes. Blood. 1987 Sep;70(3):804–808. [PubMed] [Google Scholar]
- Rhoda M. D., Giraud F., Craescu C. T., Beuzard Y. Compartmentalization of Ca2+ in sickle cells. Cell Calcium. 1985 Oct;6(5):397–411. doi: 10.1016/0143-4160(85)90017-x. [DOI] [PubMed] [Google Scholar]
- Scharff O., Foder B., Skibsted U. Hysteretic activation of the Ca2+ pump revealed by calcium transients in human red cells. Biochim Biophys Acta. 1983 May 5;730(2):295–305. doi: 10.1016/0005-2736(83)90346-2. [DOI] [PubMed] [Google Scholar]
- Tiffert T., Etzion Z., Bookchin R. M., Lew V. L. Effects of deoxygenation on active and passive Ca2+ transport and cytoplasmic Ca2+ buffering in normal human red cells. J Physiol. 1993 May;464:529–544. doi: 10.1113/jphysiol.1993.sp019649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tiffert T., Garcia-Sancho J., Lew V. L. Irreversible ATP depletion caused by low concentrations of formaldehyde and of calcium-chelator esters in intact human red cells. Biochim Biophys Acta. 1984 Jun 13;773(1):143–156. doi: 10.1016/0005-2736(84)90559-5. [DOI] [PubMed] [Google Scholar]
- Tiffert T., Lew V. L. Cytoplasmic calcium buffers in intact human red cells. J Physiol. 1997 Apr 1;500(Pt 1):139–154. doi: 10.1113/jphysiol.1997.sp022005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsien R. Y. A non-disruptive technique for loading calcium buffers and indicators into cells. Nature. 1981 Apr 9;290(5806):527–528. doi: 10.1038/290527a0. [DOI] [PubMed] [Google Scholar]
- Tsien R. Y. Intracellular measurements of ion activities. Annu Rev Biophys Bioeng. 1983;12:91–116. doi: 10.1146/annurev.bb.12.060183.000515. [DOI] [PubMed] [Google Scholar]
- Tsien R. Y., Rink T. J., Poenie M. Measurement of cytosolic free Ca2+ in individual small cells using fluorescence microscopy with dual excitation wavelengths. Cell Calcium. 1985 Apr;6(1-2):145–157. doi: 10.1016/0143-4160(85)90041-7. [DOI] [PubMed] [Google Scholar]
- Varecka L., Carafoli E. Vanadate-induced movements of Ca2+ and K+ in human red blood cells. J Biol Chem. 1982 Jul 10;257(13):7414–7421. [PubMed] [Google Scholar]
- Westerblad H., Allen D. G. Intracellular calibration of the calcium indicator indo-1 in isolated fibers of Xenopus muscle. Biophys J. 1996 Aug;71(2):908–917. doi: 10.1016/S0006-3495(96)79294-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Westerblad H., Allen D. G. Methods for calibration of fluorescent calcium indicators in skeletal muscle fibers. Biophys J. 1994 Mar;66(3 Pt 1):926–928. doi: 10.1016/s0006-3495(94)80870-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams D. A., Fogarty K. E., Tsien R. Y., Fay F. S. Calcium gradients in single smooth muscle cells revealed by the digital imaging microscope using Fura-2. Nature. 1985 Dec 12;318(6046):558–561. doi: 10.1038/318558a0. [DOI] [PubMed] [Google Scholar]


