Abstract
Bronchiolitis obliterans syndrome (BOS), as chronic manifestation of graft-versus-host disease (GVHD), is a debilitating complication leading to lung function deterioration in patients after allogeneic hematopoietic cell transplantation (allo-HCT). In the present study, we evaluated BOS development risk in patients after receiving myeloablative conditioning (MAC) regimens. We performed a retrospective analysis of patients undergoing allo-HCT, who received MAC with busulfan/cyclophosphamid (BuCy, n = 175) busulfan/fludarabin (FluBu4, n = 29) or thiotepa/busulfan/fludarabine (TBF MAC, n = 37). The prevalence of lung disease prior allo-HCT, smoking status, GvHD prophylaxis, HCT-CI score, EBMT risk score and GvHD incidence varied across the groups. The cumulative incidence of BOS using the NIH diagnosis consensus criteria at 2 years after allo-HCT was 8% in FluBu4, 23% in BuCy and 19% in TBF MAC (p = 0.07). In the multivariate analysis, we identified associated factors for time to BOS such as FEV1<median (99% of predicted) (HR = 2.39, p = 0.004), CMV patient serology positivity (HR = 2.11, p = 0.014), TLC < 80% of predicted (HR = 0.12, p = 0.02) and GvHD prophylaxis with in vivo T-cell depletion (HR = 0.29, p = 0.001) as predictors of BOS. In summary, we identified risk factors for BOS development in patients receiving MAC conditioning. These findings might serve to identify patients at risk, who might benefit from closely monitoring or early therapeutic interventions.
Subject terms: Translational research, Risk factors
Introduction
Chronic graft versus host disease (cGvHD) remains one of the leading causes for morbidity and mortality following allogeneic hematopoietic cell transplantation (allo-HCT). Although recent advances elucidating the basic, preclinical, and clinical biology of chronic GvHD have been made, effective preventive and treatment options are limited [1, 2]. Furthermore, among the multiple organs involved in cGvHD, pulmonary cGvHD is especially difficult to treat [3].
Although late interstitial pneumonitis (IP) and cryptogenic organizing pneumonia (COP) are often associated with cGvHD [4–6], BOS is the only manifestation considered diagnostic of cGvHD [7, 8] and results from the immune reaction in the small terminal airways, leading to fibrotic remodeling and occlusion [9]. Therapy is directed to stabilize the disease, for which currently most experts prefer regimens consisting of systemic or inhaled corticosteroids, long-acting β-2 agonists, azithromycin, and leukotriene receptor antagonists [10–13]. To harmonize the definition of BOS for comparative studies and clinical trials, the National Institutes of Health (NIH) has defined and developed consensus diagnostic criteria for BOS after allo-HCT [7, 14]. Furthermore, pretransplant obstructive lung disease have been linked to worse outcome in patients undergoing allo-HCT [15, 16] and pretransplant restrictive lung disease has been associated with early respiratory failure [17] and long-term complications [18] in the context of allo-HCT.
A combination of busulfan and cyclophosphamide (BuCy) was the first prototype of chemotherapeutic myeloablative conditioning (MAC) [19]. Later conditioning with fludarabin and busulfan (FluBu4) was introduced [20]. To further enhance the antileukemic effects of these protocols, several chemotherapeutic combinations were established. Among them, the combination of thiotepa, busulfan and fludarabin (TBF MAC) has been widely used [21–24]. In previous studies, we described and characterized clinical risk factors for BOS and investigated the impact of BOS on the outcome of patients undergoing allo-HCT with reduced toxicity/intensity conditioning (RIC) [25, 26]. In the present study, we evaluated the risk of BOS across patients conditioned with myeloablative regimens (BuCy, FluBu4, TBF MAC).
Methods
Study design
In this retrospective analysis, patients with acute myeloid leukemia (AML), myelodysplastic syndrome (MDS) and myeloproliferative syndromes (MPN) undergoing first allo-HCT at the University of Freiburg Medical Center were included. The inclusion criteria were: (1) adult (aged > 18 years) patients who received MAC with Busulfan/Cyclophosphamid (BuCy, Busulfan i.v. 3.2 mg/kg/day from day -7 to day -4, cyclophosphamide 60 mg/kg/day i.v. from day -3 to -2), Busulfan/ Fludarabin (FluBu4, Fludarabin 30 mg/m2/day from day -7 to day -4, Busulfan 3,2 mg/kg/day from day -7 to day -4) or TBF MAC (Thiotepa 5 mg/kg/day from day -7 to day -6, Fludarabin 30 mg/m2 d-5 to d-3, Busulfan 3,2 mg/kg/day from day -5 to day -3). (2) All patient with a first allo-HCT were included in this study, (3) allo-HCT from a matched sibling donor (MSD) or matched or mismatched unrelated donor (MUD 10/10 and MMUD 9/10—including HLA-A, -B, -C, or -DRB1 and DQB1 mismatches -) (4) transplantation date between January 1st, 1998 and September 30th, 2019, (5) with an unmanipulated peripheral blood stem cell graft (no in vitro T-cell depletion (TCD)). Patients undergoing haplo-identical or cord blood allo-HCT and patients with a syngeneic donor were also excluded. CR was defined as less than 5% blasts in bone marrow at the time of allo-HCT. Cyclosporine-based GvHD prophylaxis (cyclosporine 5 mg/kg body weight per day, starting day 3, targeted through serum level) was combined with methotrexate (15 mg/m2 on day +1, 10 mg/m2 on days +3 and +6), mycophenolate mofetil (2 × 1 g), antithymocyte globulin (ATLG-Grafalon (earlier Fresenius); 30 to 60 mg/kg body weight) [27, 28], prednisolone or alemtuzumab (given i.v., at day -2 and -1, ranging from 10 to 40 mg) [29]. Patients receiving BuCy underwent transplantation between January 1, 1998, and April 30, 2012, patients receiving FluBu4 or TBF MAC underwent transplantation between January 1, 2014 and September 30, 2019. Conditioning protocols FluBu4 and TBF MAC were selected by the caring physicians according to patient and disease characteristic including remission status prior allo-HCT, genetic markers and were not randomized. Post-transplant events such as hematological relapse and GvHD were defined based on standard clinical and laboratory criteria. All clinical data were prospectively collected and retrospectively analyzed. The investigators were not blinded during the outcome analysis. All data were evaluated as of December 31th 2023.
Ethics approval and consent to participate
This study was conducted in accordance with the amended Declaration of Helsinki. The institutional review board of the University of Freiburg Medical Center approved this study (study protocol Nr. 22-1490-S1-retro) and written informed consent was obtained from the subjects for data use in clinical research.
Pulmonary function tests
Patients were clinically examined weekly or every 2 weeks during the first 3 months after transplantation. PFTs were routinely performed a week before allo-HCT and after 3, 6, 12, and 24 months, then repeated on the basis of clinical suspicion of pulmonary disease. PFT parameters were evaluated and expressed as percentage of predicted normal values, calculated using published equations [30]. The diagnosis of BOS was defined by NIH criteria [7, 14, 31]. To exclude infection, thorax CT and standard culture and staining methods for bacterial, viral, and fungal pathogens were used routinely from body fluids including sputum and bronchoalveolar lavages. Each PFT from each patient was verified individually.
Statistical analysis
Outcome variables such as overall survival (OS), progression-free survival (PFS), relapse and non-relapse mortality (NRM) were defined following internal consensus guidelines [32]. Patient-, disease- and treatment-related characteristics were compared using the chi-square test for categorical data or analysis of variance (ANOVA) and Student’s t-test for continuous data. Paired comparisons were conducted with Wilcoxon’s matched-pair signed-rank test for continuous variables. Baseline characteristics were summarized using median and range for continuous data, and frequency and percentage for categorical data. OS was defined as the time from allo-HCT until death from any cause. PFS was defined as the time from allo-HCT to death from any cause, or relapse, whichever occurred first. Relapse was defined as detection of disease via cytological and histological assessment after allo-HCT; death without prior relapse was considered as a competing risk for relapse and was denoted as NRM. For cumulative incidence of BOS, acute GvHD (aGvHD) and cGvHD, death without BOS/aGvHD/cGvHD, respectively, were considered as competing events. Patients with no events were censored at the date of last follow-up. The median follow-up was calculated using the inverse Kaplan Meier method [33]. Univariate analyses were performed using Gray’s test for cumulative incidence functions as relapse, NRM, BOS, aGvHD and cGvHD [34, 35] and the log-rank test for OS and PFS. The Cox proportional-hazards model and Fine and Gray regression model for competing risks were used for multivariate regression analysis backward selection process of prognostic factors with a univariate p value < 0.1. We included the type of myeloablative conditioning (BuCy, FluBu4, TBF MAC) as hypothesis confounding variable in multivariate analysis. Results were expressed as the hazard ratios (HRs) and subdistribution hazard ratios (SHR) with 95% confidence intervals (95% CI). All tests were two sided. The Type I error was fixed at 0.1 for factors associated with time-to-event outcomes. Statistics were performed with STATA v17.0 (College Station, Texas, USA).
Results
Clinical features of patients treated with myeloablative conditioning prior allo-HCT
The clinical and transplant characteristics of the 241 patients included in this study are shown in Table 1 and Supplementary Table 1. Knowing that conditioning prior allo-HCT has a significant impact on pulmonary function and complications after allo-HCT including BOS development, we focused first on the analysis of patient characteristics of each conditioning cohort. Prior to allo-HCT, 175 patients received a conditioning with BuCy, 29 patients with FluBu4 and 37 patients with TBF MAC. The median patient age was 43 years in the BuCy, 46 years in the FluBu4 and 39 years in the TBF MAC group and the median follow-up was 65, 62 and 40.3 months, respectively. In terms of comorbidities and disease risk score, we observed a lower HCT-CI score but a higher EBMT disease risk score in the BuCy cohort, compared to the two other groups (Supplementary Table 1). Regarding GvHD-prophylaxis based on in vivo TCD, there were also significant imbalances between the three conditioning groups: alemtuzumab was only used in the BuCy (22%) cohort and ATG was more often used in FluBu4 and TBF MAC conditioning (41% vs. 59% vs 78%, respectively) (Table 1).
Table 1.
Clinical characteristics and pulmonary function tests at the time of allo-HCT.
| BuCy | FluBu4 | TBF MAC | p value | |
|---|---|---|---|---|
| N | 175 | 29 | 37 | |
| Patient sex (% of male) | 99 (57) | 16 (55) | 19 (51) | 0.84 |
| Donor sex (% of male) | 101 (58) | 20 (69) | 18 (49) | 0.25 |
| Age at allo-HCT, median (range) | 43 (18, 58) | 46 (26, 59) | 39 (19, 59) | 0.08 |
| KPS, median (range) | 90 (30, 100) | 90 (30, 100) | 90 (70, 100) | 0.77 |
| Median follow up in months (range) | 65 (3, 275) | 62 (2, 107) | 40.3 (2, 100) | 0.001 |
| Donors, n (%) | ||||
| - related | 61 (35) | 9 (31) | 10 (27) | 0.63 |
| - unrelated | 114 (65) | 20 (69) | 27 (73) | |
| GvHD prophylaxis, n (%) | ||||
| - without in vivo TCD | 65 (37) | 12 (41) | 8 (22) | 8 (22) |
| - with in vivo TCD | 110 (63) | 17 (59) | 29 (78) | 0.15 |
| Type of GvHD prophylaxis, n (%) | ||||
| - CyA/ATG | 72 (41) | 17 (59) | 29 (78) | 0.001 |
| - CyA/alemtuzumab | 38 (22) | 0 | 0 | |
| Lung disease before allo-HCT, n (%) | 74 (42) | 1 (4) | 7 (19) | 0.001 |
| - fungal pneumonia or aspergillosis | 31 | 1 | 1 | |
| - bact. pneumonia | 15 | 0 | 2 | |
| - bronchitis or upper airway infection | 11 | 0 | 0 | |
| - COPD/Asthma | 8 | 0 | 0 | |
| - atypical pneumonia | 3 | 0 | 1 | |
| - viral pneumonia | 2 | 0 | 1 | |
| - pleura effusions | 4 | 0 | 0 | |
| - pulmonary embolism | 4 | 0 | 0 | |
| - others (leukemia infiltration, pleuritis, aspiration pneumonia, interstitielle pneumonia, lung edema, restrictive lung disease) | 4 | 0 | 2 | |
| Lung disease up to 100 d after allo-HCT, n (%) | 49 (28) | 5 (17) | 7 (19) | 0.29 |
| - bact. pneumonia | 21 | 0 | 2 | |
| - fungal pneumonia or aspergillosis | 12 | 2 | 3 | |
| - obstructive lung function | 7 | 0 | 0 | |
| - viral pneumonia | 4 | 1 | 1 | |
| - bronchitis or upper airway infection | 4 | 1 | 0 | |
| - pleura effusions | 5 | 0 | 1 | |
| - others (engraftment syndrome, atypical pneumonia) | 8 | 1 | 0 | |
| Smoking current or previous (%) | 53 (30) | 3 (10) | 2 (5) | 0.001 |
| Pulmonary function tests before allo-HCT, median (range) | ||||
| - FEV1 (% predicted) | 98 (44, 142) | 99 (70, 133) | 98 (67, 127) | 0.88 |
| - FEV1/FVC ratio | 0.83 (0.55, 1.00) | 0.82 (0.66, 0.95) | 0.84 (0.67, 1.13) | 0.75 |
| - MEF50 (% predicted) | 70 (4, 159) | 81 (5, 130) | 82 (44, 147) | 0.07 |
| - MEF25 (% of predicted) | 45 (11, 122) | 57 (2, 124) | 53 (18, 242) | 0.003 |
| - DLCOc SB (% predicted) | 76 (46, 109) | 74 (52, 108) | 76 (40, 1.04) | 0.63 |
| - RV (% predicted) | 111 (42, 223) | 107 (64, 153) | 103 (55, 148) | 0.18 |
| - RV/TLC ratio | 0.33 (0.14, 1.18) | 0.34 (0.19, 0.49) | 0.30 (16, 108) | 0.18 |
| - TLC (% predicted) | 96 (51, 131) | 96 (79, 130) | 97 (65, 133) | 0.17 |
| - aCO2 (mmHg) | 37 (27, 46) | 36 (30, 44) | 37 (28, 42) | 0.15 |
| - aO2 (mmHg) | 83 (65, 103) | 84 (67, 92) | 85 (66, 106) | 0.005 |
| Time from allo-HCT to BOS diagnosis in months, median (range) | 12.5 (2.4, 111.3) | 8.91 (3.6, 14.2) | 13.6 (5.5, 30.4) | 0.75 |
| Cumulative incidence of BOS after allo-HCT, % (95% CI) at | 0.07 | |||
| - 1 year | 11.9 (7.7, 18.3) | 3.6 (0.5, 22.7) | 9.3 (3.1, 26.1) | |
| - 2 year | 23.2 (17.0, 31.1) | 8.4 (2.1, 30.1) | 19.5 (9.2, 38.6) | |
| - 3 year | 30.2 (23.1, 38.9) | 8.4 (2.1, 30.1) | 23.4 (11.0, 43.1) | |
| - 5 year | 31.2 (23.9, 39.9) | 8.4 (2.1, 20.1) | 23.4 (11.0, 43.1) | |
| BOS severity, n (%) | ||||
| - Mild (FEV1 60-79%) | 12 (26) | – | (28) | |
| - Moderate (FEV1 40-59%) | 21 (47) | 2 (100) | (57) | |
| - Severe (FEV1 ≤ 39%) | 12 (26) | – | 1 (14) | |
Allo-HCT allogeneic hematopoietic cell transplantation, KPS Karnofsky performance score, GvHD graft-versus-host disease, TCD T-cell depletion, CyA cyclosporine A, ATG antithymocyte globuline, COPD chronic obstructive pulmonary disease, bact. bacterial, BOS bronchiolitis obliterans syndrome, FEV1 forced expiratory volume in 1 s (FEV1), FVC forced vital capacity, RV residual volume, TLC total lung capacity, MEF50 mid-expiratory flow 50%, MEF25 mid-expiratory flow 25%, DLCOc SB carbon monoxide diffusion capacity corrected for hemoglobin, BuCy Busulfan/Cyclophosphamide, FluBu4 Fludarabine/Busulfan 4 days, TBF MAC Thiotepa/Busulfan/Fludarabine myeloablative conditioning.
Pulmonary characteristics and complications prior and after allo-HCT
There were significant differences in the pulmonary clinical features across the different conditioning groups (Table 1): (1) Patients in the BuCy group had the highest prevalence of lung diseases prior to allo-HCT (42% vs. 4% and 19%, respectively, p < 0.001) compared to patients in the other two groups. (2) The rate of current or previous smokers was the highest in the BuCy group (30% vs 10% and 5%, respectively, p = 0.001) compared to patients in the FluBu4 and TBF MAC group. (3) Pulmonary evaluation prior to allo-HCT differed numerically in regard to two PFT parameters: MEF25 (45% vs 57% vs 53%, p = 0.003) and arterial O2 (83 vs 84 vs 85 mmHg, p = 0.005) between groups.
BOS diagnosis and impact on outcome after allo-HCT
We analyzed the cumulative incidence of BOS using the NIH diagnosis consensus criteria [7, 14, 31] in patients conditioned with MAC regimens (Table 1, Supplementary Fig. 1). Strikingly, we observed a trend for lower BOS incidence in patients receiving FluBu4 compared to patients that received either BuCy or TBF MAC as conditioning regimen (Table 1): At 1 year, BOS incidence was 3.6% in patients treated with FluBu4, 11.9% in BuCy and 9.3% in TBF MAC and at 5 years, it was 8.4% in FluBu4 cohort compared to 31.2% in the BuCy and 23.4% in the TBF MAC cohort. Six patients developed BOS prior d + 100 and 5 patients developed cGvHD prior d + 100. Moreover, using the NIH criteria we found that most of the patients had already moderate severity of BOS at diagnosis: in the BuCy group 12 patients (26%) had severe and 21 patients (47%) moderate BOS; in the TBF MAC group 1 patient (14%) had a severe degree and 4 patients (57%) a moderate degree of BOS. In the FluBu4 group, we observed only 2 that were diagnosed with moderate severity of BOS. Pulmonary function parameters were analyzed prior allo-HCT and at BOS diagnosis only in patients, who had a suspicion of BOS (Supplementary Table 2). Almost all analyzed parameters except TLC deteriorated between pre-transplant assessment and assessment at BOS diagnosis.
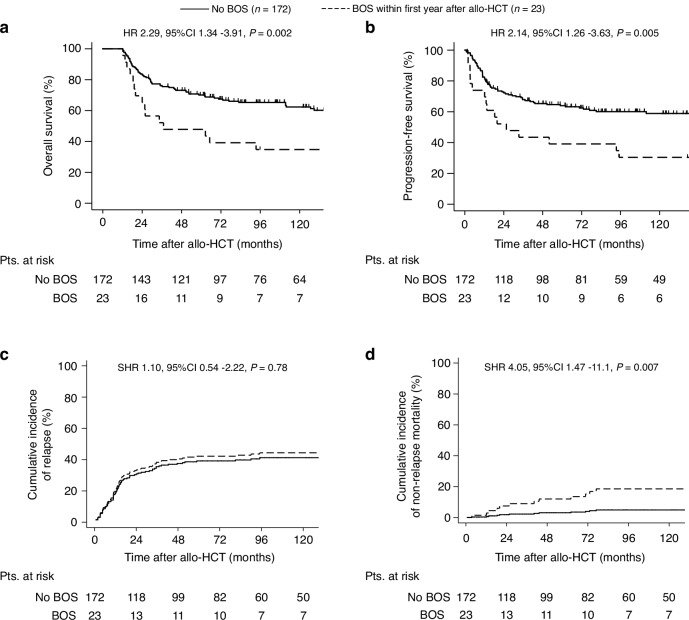
To estimate the impact of BOS on outcome variables of patients after allo-HCT, we performed a landmark analysis for patients surviving at least 1 year after allo-HCT (n = 195). We analyzed outcome variables by BOS development within the first year after allo-HCT (Fig. 1). Patients developing BOS within the first year after allo-HCT had a decreased OS (HR 2.29, 95% CI 1.34–3.91, p = 0.002) and PFS (HR 2.14, 95% CI 1.26–3.63, p = 0.005) and increased NRM (SHR 4.05, 95% CI 1.47–11.1, p = 0.007). As expected, we found no differences on relapse incidence between both groups (SHR 1.1, 95% CI 0.54–2.22, p = 0.78). Of note, we could not assess the impact of BOS severity on outcome due to the low number of patients in the subgroup analysis (n = 6 for mild, n = 12 for moderate, n = 5 for severe grade).
Fig. 1. Outcome variables by BOS development within the first year after allo-HCT.
To address the influence of BOS development on outcome after allo-HCT we performed a landmark analysis. Patients surviving 365 days after allo-HCT were included in the analysis (n = 195). Outcome variables were analyzed by BOS development within the first year after allo-HCT. Kaplan-Meier curves represent (a) overall survival and (b) progression-free survival and cumulative incidence curve represent (c) non-relapse mortality and (d) relapse incidence in patients conditioned with myeloablative conditioning regimens prior allo-HCT. Pts patients, allo-HCT allogeneic hematopoietic cell transplantation.
Univariate comparison of outcome variables and GvHD incidence after allo-HCT
We analyzed outcome variables and GvHD incidence by myeloablative conditioning in univariate analysis (Supplementary Figs. 2–3, Supplementary Table 3). Some differences were observed such as a trend for improved OS (HR 0.50, p = 0.06), PFS (HR 0.51, p = 0.05), a decreased relapse incidence (Fig. 2, SHR 0.47, p = 0.06) and a trend to lower incidence of cGvHD (Fig. 3, HR 0.52, p = 0.05) in patients treated with FluBu4 compared to patients conditioned with BuCy. No significant differences were observed in outcome variables between patients treated with TBF MAC and BuCy. These results should be interpreted with caution due to the nature of univariate analysis, lower number of patients transplanted with FluBu4 (n = 29) and unbalanced clinical features pre-allo-HCT.
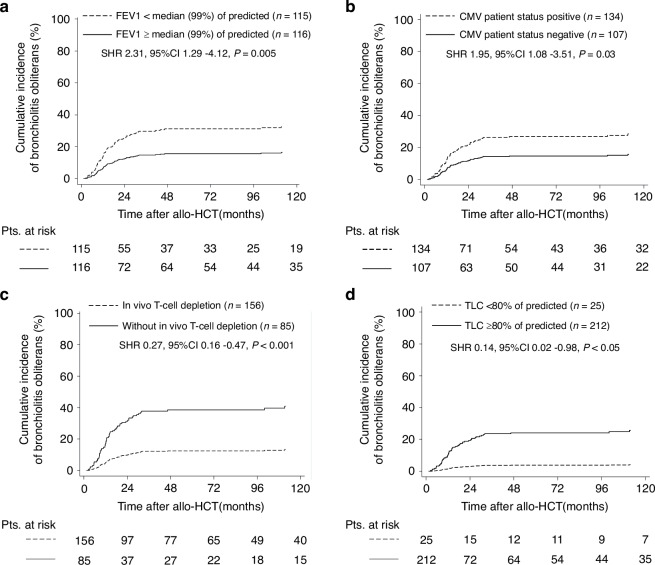
Fig. 2. Cumulative incidence of BOS by clinical factors and lung function tests associated with BOS in multivariate analysis.
Cumulative incidence curve represent bronchiolitis obliterans by (a) FEV1 ≥ or <median (99% of predicted), (b) CMV patient positivity or negativity, (c) GvHD prophylaxis with or without in vivo T-cell depletion and (d) TLC ≥ or < 80% of predicted in patients conditioned with myeloablative conditioning regimens prior allo-HCT. Statistical analysis was performed for Fine and Gray regression models in the presence of competing risks. FEV1 forced expiratory volume in 1 s., TLC total lung capacity, Pts patients, allo-HCT allogeneic hematopoietic cell transplantation, SHR subdistribution hazard ratio, CI confidence intervals.
Fig. 3. Cumulative incidence of BOS by cGvHD within the first year after allo-HCT.
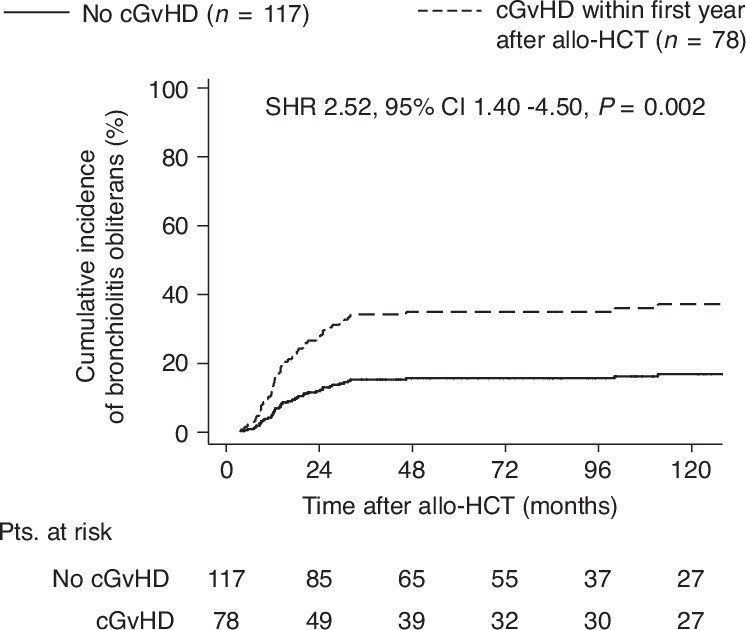
We performed a landmark analysis of cGvHD excluding BOS as risk factor for BOS development. Patients surviving 365 days after allo-HCT were included in the analysis (n = 195). Cumulative incidence of BOS was analyzed by cGvHD onset within the first year after allo-HCT. SHR subdistribution hazard risk, CI confidence interval, cGvHD chronic GvHD, allo-HCT allogeneic hematopoietic cell transplantation.
Univariate and multivariate analysis for risk factors associated with BOS
To identify clinical factors and pulmonary parameters associated with BOS in patients treated with myeloablative conditioning, we performed Fine and Gray regression model in univariate (Supplementary Table 4–5) and multivariate analysis including conditioning regimens as hypothesis confounding variable (Table 2A).
Table 2.
Cox regression multivariate analysis for lung function parameters before allo-HCT and clinical parameters for bronchiolitis obliterans syndrome and death.
| A. | ||||
|---|---|---|---|---|
| Subdistribution hazard ratio for BOS incidence | ||||
| N | p value | SHR | 95% CI | |
|
Conditioning with BuCy FluBu4 TBF MAC |
175 29 37 |
– 0.08 0.77 |
1 0.28 1.11 |
– 0.07, 1.17 0.52, 2.40 |
| TLC < 80% predicted | 25 | 0.02 | 0.12 | 0.02, 0.71 |
| FEV1 < median % of predicted (99%) | 115 | 0.004 | 2.39 | 1.31, 4.35 |
| In vivo T-cell depletiona | 110 | 0.001 | 0.29 | 0.16, 0.51 |
| CMV patient positivity | 134 | 0.014 | 2.11 | 1.16, 3.84 |
| B. | ||||
|---|---|---|---|---|
| Hazard ratio estimates for death | ||||
| n | p value | HR | 95% CI | |
|
Conditioning with BuCy FluBu4 TBF MAC |
175 29 37 |
– 0.17 0.76 |
1 0.60 0.92 |
– 0.29, 1.24 0.54, 1.55 |
| FEV1 < 80% of predicted initial | 31 | 0.005 | 2.01 | 1.23, 3.28 |
|
Disease status before allo-HCTb - early - intermediate - late |
113 15 111 |
– 0.56 0.001 |
1 0.75 2.53 |
– 0.29, 1.94 1.74, 3.68 |
Subdistribution hazard ratios (SHR) and confidence intervals (CI) for single pulmonary function test (PFT) values before allo-HCT and clinical parameters were estimated in multivariate analysis following backward selection for (A) BOS incidence using the Fine and Gray regression model in the presence of competing risks and for (B) overall survival using Cox regression proportional hazard ratio. Single PFT values and clinical parameters with a p value of 0.1 in univariate analysis were selected for the multivariate analysis. Conditioning was included as hypothesis variable. FEV1, forced vital capacity in 1 s. BuCy, Busulfan/Cyclophosphamide; FluBu4, Fludarabine/Busulfan 4 days; TBF MAC, Thiotepa/Busulfan/Fludarabine myeloablative conditioning.
aIn vivo T-cell depletion includes patients receiving alemtuzumab and antithymocyte globuline (ATG).
bDisease stage defined by EBMT score (Gratwohl et al., 2012).
In multivariate analysis, we identified FEV1<median (99% of predicted) (HR 2.39, p = 0.004) and CMV patient positivity (HR 2.11, p = 0.014) to be associated with BOS incidence. In vivo T-cell depletion with alemtuzumab and ATG was shown to be protective (SHR 0.29, p = 0.001). Unexpectedly, patients with restrictive lung disease defined as TLC < 80% showed lower incidence of BOS (SHR 0.12, p = 0.02) (Table 2A, Fig. 2). BOS incidence did not differ among the three conditioning groups in multivariate analysis (for FluBu4, SHR 0.77, p = 0.08; for TBF MAC, SHR 1.11, p = 0.28 compared to BuCy) (Table 2A, Supplementary Fig. 1).
Next, we focused on specific components of PFT for early diagnosis of BOS and their predictive capacity. Therefore, we performed a landmark analysis of patients surviving at least 100 days and with available PFTs within the first 100 days after allo-HCT. Several lung function parameters at day +100 after allo-HCT such as FEV1/FVC < 0.7 ratio, RV/TLC > 0.45 ratio, FEV1<median (99% of predicted) and changes in small airways as MEF50 < 50% predicted, MEF50 < 35% predicted, MEF25 < 35% predicted and MEF25 < 25% predicted were also associated with time to BOS in univariate analysis (Table 3). The median time from identified PFT parameters to BOS diagnosis ranged between 164 days (FEV1< median 99% of predicated) and 393 days (MEF25 < 35% of predicted).
Table 3.
Subdistribution hazard ratios for single lung function parameters after allo-HCT at day 100 for BOS incidence.
| Subsdistribution hazard ratio estimates for BOS incidence of patients surviving at least 100 days with available PFTs (n = 200/245) | Median time from selected PFT parameters d + 100 to BOS (days) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Parameter before allo-HCT | Parameter at day 100 after allo-HCT | |||||||||
| n | p value | SHR | 95% CI | n | p value | SHR | 95% CI | |||
| FEV1/FVC < 0.80 ratio | 72 | 0.35 | 1.31 | 0.75, 2.30 | 86 | 0.08 | 1.63 | 0.94, 2.82 | 268 | |
| FEV1/FVC < 0.70 ratio | 12 | 0.11 | 2.17 | 0.84, 5.73 | 18 | 0.001 | 3.47 | 1.68, 7.14 | 291 | |
| FEV1 < median (99% of predicted) | 90 | 0.002 | 2.54 | 1.41, 4.58 | 115 | 0.001 | 4.10 | 2.00, 8.43 | 164 | |
| FEV1 < 75% predicted | 8 | 0.12 | 2.20 | 0.82, 5.84 | 23 | 0.03 | 2.25 | 1.09, 4.66 | 386 | |
| RV > 120% predicted | 66 | 0.37 | 1.29 | 0.74, 2.25 | 63 | 0.97 | 0.98 | 0.54, 1.78 | 171 | |
| RV/TLC > 0.45 ratio | 13 | 0.86 | 0.90 | 0.27, 2.97 | 22 | 0.02 | 2.57 | 1.23, 5.36 | 182 | |
| TLC < 80% predicted | 15 | 0.11 | 0.21 | 0.31, 1.41 | 34 | 0.14 | 1.64 | 0.84, 3.18 | 329 | |
| MEF50 < 50% predicted | 30 | 0.12 | 1.66 | 0.87, 3.17 | 43 | 0.001 | 3.17 | 1.82, 5.51 | 379 | |
| MEF50 < 35% predicted | 6 | 0.67 | 1.30 | 0.38, 4.50 | 14 | 0.001 | 5.19 | 2.58, 10.4 | 350 | |
| MEF25 < 35% predicted | 11 | 0.41 | 1.48 | 0.57, 3.84 | 62 | 0.001 | 3.29 | 1.89, 5.72 | 393 | |
| MEF25 < 25% predicted | 6 | 0.67 | 1.30 | 0.38, 4.50 | 24 | 0.001 | 3.42 | 1.88, 6.23 | 277 | |
| DLCOc SB < 80% predicted | 97 | 0.44 | 1.28 | 0.68, 2.39 | 97 | 0.12 | 1.58 | 0.88, 2.83 | 271 | |
| DLCOc SB < 60% predicted | 20 | 0.50 | 1.35 | 0.55, 3.32 | 20 | 0.52 | 1.22 | 0.66, 2.24 | 321 | |
Patients surviving 100 days and with available pulmonary function tests were selected for the analysis (200/245, 82%). Subdistribution hazard ratios (SHR) and confidence intervals (CI) for single pulmonary function test (PFT) values at day 100 after allo-HCT were estimated for BOS in univariate analysis using the Fine and Gray regression model in the presence of competing risks. Cut-off values for PFT were chosen from the significant PFT parameters before allo-HCT and previous publications (Duque-Afonso et al. 2018). Only patients surviving 100 days after allo-HCT and with pulmonary function test (PFT) within the first 100 days after allo-HCT were included in this analysis. Median time from selected PFT parameters from d + 100 to BOS was calculated only for patients developing BOS.
BOS bronchiolitis obliterans syndrome, PFT pulmonary function tests, allo-HCT allogenic hematopoietic cell transplantation, FEV1 forced expiratory volume in 1 second (FEV1), FVC forced vital capacity, RV residual volume, TLC total lung capacity, MEF50 mid-expiratory flow 50%, MEF25 mid-expiratory flow 25%, DLCOc SB carbon monoxide diffusion capacity corrected for hemoglobin.
To address whether cGvHD excluding BOS is associated with BOS development in our cohort, we performed a landmark analysis for patients surviving at least one year and we compared the cumulative incidence of BOS by the presence of cGvHD within the first year after allo-HCT. As expected, we identified cGvHD excluding BOS as a risk factor for the development of BOS (SHR = 2.51, 95%CI 1.40–4.50, p = 0.002) (Fig. 3).
Univariate and multivariate analysis for risk factors associated with death
Regarding cause-specific hazard ratios for death, several clinical and PFT parameters were found to be associated with death in univariate analysis prior allo-HCT and at day +100 after allo-HCT (Supplementary Tables 6–8). In multivariate analysis, FEV1 < 80% of predicted (HR 2.01, p = 0.005) and advanced disease status before allo-HCT (HR 2.53, p = 0.001) were associated with increased risk of death (Table 2B).
The causes for death are described in Table 4: 58% patients have died during the follow up in the BuCy cohort, 27% in the FluBu4 and 48% in the TBF MAC cohort. The predominant reason for death was relapse or progression of the original disease (45%, 21%, 35%, in BuCy, FluBu4 and TBF MAC, respectively). A pulmonary cause of death was observed in 6% (n = 12) patients in the BuCy cohort, 10 of these patients died because of infectious pulmonary diseases, 1 because of ARDS and 1 because of bronchiolitis obliterans. In the TBF MAC group, 8% (n = 3 patients) died because of infectious pulmonary diseases, whereas in the FluBu4 cohort no deaths due to pulmonary diseases were reported.
Table 4.
Cause of death.
| BuCy | FluBu4 | TBF MAC | |
|---|---|---|---|
| N | 175 | 29 | 37 |
| Alive, n (%) | 74 (42) | 21 (72) | 19 (51) |
| Total deaths, n (%) | 101 (58) | 8 (27) | 18 (48) |
| Relapse or progression of original disease, n (%) | 78 (45) | 6 (21) | 13 (35) |
| NRM, pulmonary cause of death, n (%) |
12 (6) - bacterial Pneumonia (3x) - Aspergillus pneumonia (3x) - Viral pneumonia (COVID, parainfluenza, influenza, CMV) (4x) - bronchiolitis obliterans - ARDS |
0 |
3 (8) - bact. Pneumonia - Viral pneumonia - CMV pneumonia |
| NRM, non-pulmonary cause of death, n (%) |
9 (5) - secondary malignancy (4x) - cardiac infarction - prior malignancy (breast cancer) - cerebral infarction - cGvHD - Sepsis P. aeroginosa |
2 (6) - cGvHD - aGvHD |
2 (5) - cGvHD - Secondary malignancy |
| Unknown, n (%) | 2 (2) | 0 | 0 |
N total patient number, allo-HCT allogeneic hematopoietic cell transplantation, BuCy Busulfan/Cyclophosphamide, FluBu4 Fludarabine/Busulfan 4 days, TBF MAC Thiotepa/Busulfan/Fludarabine myeloablative conditioning.
Discussion
BOS development after allo-HCT is still an unsolved clinical problem because once established it is very difficult to treat [3, 4]. In addition, the development of risk stratification tools for BOS risk is challenging. In our previous studies, we have focused on the impact of pulmonary function prior allo-HCT on clinical outcomes such as mortality and respiratory failure. In these studies, we examined mainly older patients or with comorbidities conditioned with reduced toxicity/intensity conditioning protocols as FBM and FTM [25, 26]. Our results suggested that patients with moderate small airway disease prior to allo-HCT as depicted by MEF25 < 35% of predicted and MEF50 < 50% of predicted have a higher risk of BOS. In addition, severe small airway disease, decreased CO-diffusion capacity prior allo-HCT as well as a combined restrictive/obstructive lung disease at day +100 after allo-HCT were associated with higher risk for NRM [25].
In the present study, we included a younger patient population receiving a myeloablative conditioning. Therefore, these patients were potentially exposed to less toxic substances (chemotherapy, smoking) and thus rather preserved lung function as compared to patients receiving reduced toxicity conditioning, which are older and/or have higher a comorbidity index. As expected, clinical risk factors and PFT parameters associated with BOS and death were different between both cohorts (Table 1 and Supplementary Table 1).
These striking differences may be partly explained by the different characteristics of the treatment cohorts: First, due to institutional practice at our center, the BuCy conditioning was used up to 2012 and included 175 patients, whereas the more contemporary conditioning FluBu4 included 29 patients and the TBF MAC included 37 patients. We observed the highest prevalence of pre-existent lung disease and smoking in patients treated with BuCy. We found that the MEF25% of predicted and arterial O2 in the BuCy cohort was significant lower compared to the other conditioning cohorts. Other variables, which might have contributed to BOS incidence, are lung toxicity by the conditioning therapy itself and different GvHD prophylaxis strategies.
Regarding in vivo TCD, we found that ATG was used more frequently in patients receiving FluBu4 and TBF MAC compared to the BuCy group, in which alemtuzumab was more frequently used. In a multivariate analysis, we observed that in vivo TCD was linked to decreased incidence of BOS. Along with this, we also observed in univariate analysis that patients with an unrelated donor or with an HLA non-identical donor had also a decreased risk of developing BOS. ATG or alemtuzumab are frequently used in this setting to prevent GVHD: 149/161 (92.5%) patients with unrelated donor (p < 0.001) and 50/56 (89.2%) patients with HLA-non identical donor received ATG or alemtuzumab (p < 0.001) in our cohort. In multivariate analysis, we found a decreased risk for BOS in patients receiving in vivo T-cell depletion with ATG or alemtuzumab but no association was found for patients receiving a graft from unrelated donor or from HLA non-identical donor. These findings are in line with retrospective studies showing that ATG decreased the risk for BOS and a prospective study observing, that in vivo TCD with ATG also protected from chronic lung dysfunction [36, 37].
Moreover, through multivariate analysis, we also identified CMV patient positivity prior allo-HCT and FEV1<median (99)% of predicted prior allo-HCT as a risk factor for the development of BOS. These findings are in line with a retrospective analysis also demonstrating an association between CMV positivity and the development of BOS in multivariate analysis [38]. The same study also found an association between a reduced FEV1 at the time of transplant and the diagnosis of BOS. Interestingly, another study suggested an association between CMV pneumonitis and the development of BOS in patients undergoing lung transplantation [39]. However, so far, the association between CMV infection and the development of chronic GvHD remains controversial [40–42]. The routine use of letermovir prophylaxis for CMV positive patients questions additionally whether CMV positivity is still a risk for the development of BOS.
In multivariate analysis we also found TLC < 80% of predicted to be protective against the development of BOS. This seems to be counterintuitive at first. We hypothesize that, restriction might mask obstructive changes due to increased stiffness of lung parenchyma avoiding further remodeling of airways in this patient population [43, 44]. Interestingly, reduced MEF50 and MEF25 were not associated with time to BOS in multivariate analysis. These findings are in contrast to our study of patients receiving intermediate TCI score protocols FBM/FTM [25]. It has been demonstrated the prevalence of small airway disease, indicated by reduced MEF, increases with age [45, 46]. Therefore, our findings suggest that reduced MEF as a risk factor for the development of BOS in older patients.
Some parameters as FEV1 < 80% of predicted and advanced disease status were identified in multivariate analysis to be associated with increased risk of death. The association of FEV1 < 80% of predicted is in line with the HCT-CI score, in which FEV1 < 80% of predicted is a risk factor for dismal outcome following allo-HCT [15].
One of the strengths of our work is the relative long follow-up, which identified patients developing BOS at later time point after the allo-HCT. To address which risk factors are associated with late onset of BOS, we compared clinical characteristics and PFT parameters prior allo-HCT of patients developing BOS 2 years after allo-HCT with those developing BOS within the first 2 years after allo-HCT (Supplementary Table 9). Interestingly, we found male donor (p = 0.049) and in vivo GvHD prophylaxis with alemtuzumab (p = 0.01) with a decreased risk for late-onset of BOS.
This study has also some limitations: First, our data was analyzed retrospectively and is derived from a single-center experience, therefore sample errors cannot be excluded. Importantly, due to practice changes over time the number of patients in the different conditioning cohorts is very different with relatively small patient numbers in the FluBu4 and TBF MAC cohort. Patients transplanted between 1998 and 2012 received BuCy and from 2014 to 2019 received FluBu4 and TBF MAC. This might cause a time bias as allo-HCT might have improved over time. These practice changes also led to unbalanced clinical characteristics between the conditioning cohorts, which had different follow up periods. Hence, the BuCy cohort has longer follow up than the TBF MAC and FluBu4 cohort, which limit the conclusions for the comparison of the protocols. Lastly, we focus our work on patients with myeloid malignancies receiving MAC chemotherapy. We excluded patients with lymphoid malignancies receiving total body irradiation as part of the MAC conditioning. Future studies should address the identification of clinical and PFT risk factors in this patient cohort and should compare risk factors from chemotherapy-based with TBI-based MAC conditioning.
In conclusion, we identify and describe clinical factors and PFT parameters prior and after allo-HCT that seem to influence the incidence of BOS after allo-HCT. The identification of risk factors associated with BOS and death might serve to establish pre-emptive and early therapeutic interventions.
Supplementary information
Acknowledgements
We thank Irmgard Matt for managing the data bank of transplanted patients, the medical technicians in the Stem Cell Transplantation Unit and Pulmonary Function Test Unit of the University Medical Center Freiburg, and all the patients and staff on the Löhr ward. We thank also Elisabeth Lenartz, Stefanie Knebel and Petra Isele-Hiß for transplantation coordination.
Author contributions
JD-A, JF, KM-B designed the study, interpreted the data, and wrote the manuscript. JD-A and GI performed the statistical analysis. PR, KW, TK, BCF, DS provided and analyzed the pulmonary function test parameters. CW, RM, RW, HB, RZ, JF and KM-B provided clinical patient data. All the authors critically reviewed the final version of the manuscript.
Funding
Open Access funding enabled and organized by Projekt DEAL.
Data availability
The datasets generated during and/or analyzed during the current study are available upon reasonable request from the corresponding authors.
This article contains supplementary material.
Competing interests
The authors declare no conflicting financial interest with the submission of this article. JF received research support and speakers honoraria from Medac, Neovii and Riemser. JD-A received speaker’s honoraria from Roche, Amgen, AstraZeneca, Riemser, Sobi, Lilly, Beigene and Ipsen and travel support from Gilead, Sobi, Beigene, Alexion and Abbvie. RZ received speaker’s honoraria from Incyte, Novartis, Roche and Mallinckroth. D Stolz reports personal fees from CSL Behring, Berlin-Chemie Menarini, Novartis, GlaxoSmithKline, AstraZeneca, Vifor, Merck, Chiesi, Grifols, MSD, Merck, Sanofi, Roche, Boehringer-Ingelheim and Pfizer, and participation on advisory boards for CSL Behring, Berlin-Chemie, Menarini, Novartis, Boehringer-Ingelheim, GlaxoSmithKline, AstraZeneca, Vifor, Merck, Chiesi, Roche, Grifols, MSD, Merck and Sanofi. This project was supported by a grant of the German Federal Ministry of Education and Research (BMBF 01 EO 0803). JD-A received support from Else Kröner-Fresenius-Stiftung (Nr. 2018-A56). JD-A received research support from German Research Foundation (Deutsche Forschungsgemeinschaft, ref. DU 1287/3-1 and 1287/5-1), from the Forschungskommission of the University of Freiburg Medical School (DUQ1106/16) and Berta Ottenstein-Programm for Advanced Clinician Scientists, Faculty of Medicine, University of Freiburg. KM-B received support from Else-Kröner-Fresenius-Stiftung (2021_EKEA.131), the Berta-Ottenstein Program (Faculty of Medicine, Freiburg University), and the IMMediate Advanced Clinician Scientist-Program, Department of Medicine II, Medical Center – University of Freiburg and Faculty of Medicine, University of Freiburg, funded by the Bundesministerium für Bildung und Forschung (BMBF, Federal Ministry of Education and Research) - 01EO2103.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
The online version contains supplementary material available at 10.1038/s41409-024-02422-z.
References
- 1.MacDonald KP, Hill GR, Blazar BR. Chronic graft-versus-host disease: biological insights from preclinical and clinical studies. Blood. 2017;129:13–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zeiser R, Polverelli N, Ram R, Hashmi SK, Chakraverty R, Middeke JM, et al. Ruxolitinib for glucocorticoid-refractory chronic graft-versus-host disease. N Engl J Med. 2021;385:228–38. [DOI] [PubMed] [Google Scholar]
- 3.Bergeron A. Late-onset noninfectious pulmonary complications after allogeneic hematopoietic stem cell transplantation. Clin Chest Med. 2017;38:249–62. [DOI] [PubMed] [Google Scholar]
- 4.Williams KM. How I treat bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Blood. 2017;129:448–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hildebrandt GC, Fazekas T, Lawitschka A, Bertz H, Greinix H, Halter J, et al. Diagnosis and treatment of pulmonary chronic GVHD: report from the consensus conference on clinical practice in chronic GVHD. Bone Marrow Transpl. 2011;46:1283–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sakaida E, Nakaseko C, Harima A, Yokota A, Cho R, Saito Y, et al. Late-onset noninfectious pulmonary complications after allogeneic stem cell transplantation are significantly associated with chronic graft-versus-host disease and with the graft-versus-leukemia effect. Blood. 2003;102:4236–42. [DOI] [PubMed] [Google Scholar]
- 7.Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ, et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and staging working group report. Biol Blood Marrow Transpl. 2005;11:945–56. [DOI] [PubMed] [Google Scholar]
- 8.Pang Y, Charya AV, Keller MB, Sirajuddin A, Fu YP, Holtzman NG, et al. The ISHLT chronic lung allograft dysfunction consensus criteria are applicable to pulmonary chronic graft-versus-host disease. Blood Adv. 2022;6:4196–207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Patriarca F, Skert C, Sperotto A, Damiani D, Cerno M, Geromin A, et al. Incidence, outcome, and risk factors of late-onset noninfectious pulmonary complications after unrelated donor stem cell transplantation. Bone Marrow Transpl. 2004;33:751–8. [DOI] [PubMed] [Google Scholar]
- 10.Williams KM, Cheng GS, Pusic I, Jagasia M, Burns L, Ho VT, et al. Fluticasone, azithromycin, and montelukast treatment for new-onset bronchiolitis obliterans syndrome after hematopoietic cell transplantation. Biol Blood Marrow Transpl. 2016;22:710–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bergeron A, Chevret S, Chagnon K, Godet C, Bergot E, Peffault de Latour R, et al. Budesonide/Formoterol for bronchiolitis obliterans after hematopoietic stem cell transplantation. Am J Respir Crit Care Med. 2015;191:1242–9. [DOI] [PubMed] [Google Scholar]
- 12.Kim SW, Rhee CK, Kim YJ, Lee S, Kim HJ, Lee JW. Therapeutic effect of budesonide/formoterol, montelukast and N-acetylcysteine for bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Respir Res 2016;17:63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bergeron A, Chevret S, Granata A, Chevallier P, Vincent L, Huynh A, et al. Effect of azithromycin on airflow decline-free survival after allogeneic hematopoietic stem cell transplant: the ALLOZITHRO randomized clinical trial. JAMA. 2017;318:557–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bos S, Murray J, Marchetti M, Cheng GS, Bergeron A, Wolff D, et al. ERS/EBMT clinical practice guidelines on treatment of pulmonary chronic graft-versus-host disease in adults. Eur Respir J. 2024;63:2301727. [DOI] [PubMed]
- 15.Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Parimon T, Au DH, Martin PJ, Chien JW. A risk score for mortality after allogeneic hematopoietic cell transplantation. Ann Intern Med. 2006;144:407–14. [DOI] [PubMed] [Google Scholar]
- 17.Ramirez-Sarmiento A, Orozco-Levi M, Walter EC, Au MA, Chien JW. Influence of pretransplantation restrictive lung disease on allogeneic hematopoietic cell transplantation outcomes. Biol Blood Marrow Transpl. 2010;16:199–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Walter EC, Orozco-Levi M, Ramirez-Sarmiento A, Vigorito A, Campregher PV, Martin PJ, et al. Lung function and long-term complications after allogeneic hematopoietic cell transplant. Biol Blood Marrow Transpl. 2010;16:53–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tutschka PJ, Copelan EA, Klein JP. Bone marrow transplantation for leukemia following a new busulfan and cyclophosphamide regimen. Blood. 1987;70:1382–8. [PubMed] [Google Scholar]
- 20.Rambaldi A, Grassi A, Masciulli A, Boschini C, Micò MC, Busca A, et al. Busulfan plus cyclophosphamide versus busulfan plus fludarabine as a preparative regimen for allogeneic haemopoietic stem-cell transplantation in patients with acute myeloid leukaemia: an open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2015;16:1525–36. [DOI] [PubMed] [Google Scholar]
- 21.Sanz J, Boluda JC, Martín C, González M, Ferrá C, Serrano D, et al. Single-unit umbilical cord blood transplantation from unrelated donors in patients with hematological malignancy using busulfan, thiotepa, fludarabine and ATG as myeloablative conditioning regimen. Bone Marrow Transpl. 2012;47:1287–93. [DOI] [PubMed] [Google Scholar]
- 22.Di Bartolomeo P, Santarone S, De Angelis G, Picardi A, Cudillo L, Cerretti R, et al. Haploidentical, unmanipulated, G-CSF-primed bone marrow transplantation for patients with high-risk hematologic malignancies. Blood. 2013;121:849–57. [DOI] [PubMed] [Google Scholar]
- 23.Raiola AM, Dominietto A, Ghiso A, Di Grazia C, Lamparelli T, Gualandi F, et al. Unmanipulated haploidentical bone marrow transplantation and posttransplantation cyclophosphamide for hematologic malignancies after myeloablative conditioning. Biol Blood Marrow Transpl. 2013;19:117–22. [DOI] [PubMed] [Google Scholar]
- 24.Pagliardini T, Castagna L, Harbi S, Porta MD, Rey J, Fürst S, et al. Thiotepa, fludarabine, and busulfan conditioning regimen before T cell-replete haploidentical transplantation with post-transplant cyclophosphamide for acute myeloid leukemia: a bicentric experience of 100 patients. Biol Blood Marrow Transpl. 2019;25:1803–9. [DOI] [PubMed] [Google Scholar]
- 25.Duque-Afonso J, Ihorst G, Waterhouse M, Zeiser R, Wäsch R, Bertz H, et al. Impact of lung function on bronchiolitis obliterans syndrome and outcome after allogeneic hematopoietic cell transplantation with reduced-intensity conditioning. Biol Blood Marrow Transpl. 2018;24:2277–84. [DOI] [PubMed] [Google Scholar]
- 26.Duque-Afonso J, Ihorst G, Wäsch R, Bertz H, Müller-Quernheim J, Finke J, et al. Identification of risk factors for bronchiolitis obliterans syndrome after reduced toxicity conditioning before hematopoietic cell transplantation. Bone Marrow Transpl. 2013;48:1098–103. [DOI] [PubMed] [Google Scholar]
- 27.Finke J, Bethge WA, Schmoor C, Ottinger HD, Stelljes M, Zander AR, et al. Standard graft-versus-host disease prophylaxis with or without anti-T-cell globulin in haematopoietic cell transplantation from matched unrelated donors: a randomised, open-label, multicentre phase 3 trial. Lancet Oncol. 2009;10:855–64. [DOI] [PubMed] [Google Scholar]
- 28.Finke J, Schmoor C, Bethge WA, Ottinger H, Stelljes M, Volin L, et al. Long-term outcomes after standard graft-versus-host disease prophylaxis with or without anti-human-T-lymphocyte immunoglobulin in haemopoietic cell transplantation from matched unrelated donors: final results of a randomised controlled trial. Lancet Haematol. 2017;4:e293–e301. [DOI] [PubMed] [Google Scholar]
- 29.Bertz H, Spyridonidis A, Wäsch R, Grüllich C, Egger M, Finke J. A novel GVHD-prophylaxis with low-dose alemtuzumab in allogeneic sibling or unrelated donor hematopoetic cell transplantation: the feasibility of deescalation. Biol Blood Marrow Transpl. 2009;15:1563–70. [DOI] [PubMed] [Google Scholar]
- 30.Zaiss AW, Matthys H. A multiuser system for whole body plethysmographic measurements and interpretation. Lung. 1990;168:1185–92. [DOI] [PubMed] [Google Scholar]
- 31.Jagasia MH, Greinix HT, Arora M, Williams KM, Wolff D, Cowen EW, et al. National institutes of health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. The 2014 diagnosis and staging working group report. Biol Blood Marrow Transpl. 2015;21:389–401.e1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kanate AS, Nagler A, Savani B. Summary of scientific and statistical methods, study endpoints and definitions for observational and registry-based studies in hematopoietic cell transplantation. Clin Hematol Int. 2020;2:2–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Korn EL. Censoring distributions as a measure of follow-up in survival analysis. Stat Med. 1986;5:255–60. [DOI] [PubMed] [Google Scholar]
- 34.Dignam JJ, Kocherginsky MN. Choice and interpretation of statistical tests used when competing risks are present. J Clin Oncol. 2008;26:4027–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94:496–509. [Google Scholar]
- 36.Bacigalupo A, Lamparelli T, Barisione G, Bruzzi P, Guidi S, Alessandrino PE, et al. Thymoglobulin prevents chronic graft-versus-host disease, chronic lung dysfunction, and late transplant-related mortality: long-term follow-up of a randomized trial in patients undergoing unrelated donor transplantation. Biol Blood Marrow Transpl. 2006;12:560–5. [DOI] [PubMed] [Google Scholar]
- 37.Socié G, Schmoor C, Bethge WA, Ottinger HD, Stelljes M, Zander AR, et al. Chronic graft-versus-host disease: long-term results from a randomized trial on graft-versus-host disease prophylaxis with or without anti-T-cell globulin ATG-Fresenius. Blood. 2011;117:6375–82. [DOI] [PubMed] [Google Scholar]
- 38.Gazourian L, Rogers AJ, Ibanga R, Weinhouse GL, Pinto-Plata V, Ritz J, et al. Factors associated with bronchiolitis obliterans syndrome and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation. Am J Hematol. 2014;89:404–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Snyder LD, Finlen-Copeland CA, Turbyfill WJ, Howell D, Willner DA, Palmer SM. Cytomegalovirus pneumonitis is a risk for bronchiolitis obliterans syndrome in lung transplantation. Am J Respir Crit Care Med. 2010;181:1391–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jacobsen N, Andersen HK, Skinhøj P, Ryder LP, Platz P, Jerne D, et al. Correlation between donor cytomegalovirus immunity and chronic graft-versus-host disease after allogeneic bone marrow transplantation. Scand J Haematol. 1986;36:499–506. [DOI] [PubMed] [Google Scholar]
- 41.Ueda Oshima M, Xie H, Zamora D, Flowers ME, Hill GR, Mielcarek MB, et al. Impact of GVHD prophylaxis on CMV reactivation and disease after HLA-matched peripheral blood stem cell transplantation. Blood Adv. 2023;7:1394–403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cantoni N, Hirsch HH, Khanna N, Gerull S, Buser A, Bucher C, et al. Evidence for a bidirectional relationship between cytomegalovirus replication and acute graft-versus-host disease. Biol Blood Marrow Transpl. 2010;16:1309–14. [DOI] [PubMed] [Google Scholar]
- 43.Sheshadri A, Huang HJ, Bashoura L, Alousi AM, Alkhunaizi M, Sharifi H, et al. Graft-versus-host disease may cause pulmonary restriction, but not all restriction is graft-versus-host disease. Blood Adv. 2022;6:4984–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chien JW, Duncan S, Williams KM, Pavletic SZ. Bronchiolitis obliterans syndrome after allogeneic hematopoietic stem cell transplantation-an increasingly recognized manifestation of chronic graft-versus-host disease. Biol Blood Marrow Transpl. 2010;16:S106–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Knox-Brown B, Patel J, Potts J, Ahmed R, Aquart-Stewart A, Cherkaski HH, et al. Small airways obstruction and its risk factors in the Burden of Obstructive Lung Disease (BOLD) study: a multinational cross-sectional study. Lancet Glob Health. 2023;11:e69–e82. [DOI] [PubMed] [Google Scholar]
- 46.Xiao D, Chen Z, Wu S, Huang K, Xu J, Yang L, et al. Prevalence and risk factors of small airway dysfunction, and association with smoking, in China: findings from a national cross-sectional study. Lancet Respir Med. 2020;8:1081–93. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available upon reasonable request from the corresponding authors.
This article contains supplementary material.