Abstract
Tissue engineering envisions functional substitute creation for damaged tissues. Insulin-like growth factor-1 (IGF-1) plays roles in bone marrow mesenchymal stem cell (BMSC) osteogenic differentiation (OD), and we investigated its specific mechanism. BMSCs were cultured and OD was induced. Surface antigens (CD105, CD90, CD44, CD45, CD34) were identified by flow cytometry. Adipogenic, chondrogenic, and osteogenic differentiation abilities of BMSCs were observed. BMSCs were cultured in osteogenic medium containing 80 ng/mL IGF-1 for 3 weeks. Alkaline phosphatase activity, calcification level, osteogenic factor (runt related protein 2 [RUNX2], osteocalcin [OCN], osterix [OSX]), total (t-) ERK1/2 and phosphorylated- (p-) ERK1/2 levels, and SRY-related high-mobility-group box 4 (SOX4) levels were assessed by alkaline phosphatase staining and Alizarin Red staining, Western blot, and reverse transcription-quantitative polymerase chain reaction. The mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) pathway inhibitor (PD98059) was used to inhibit the MAPK/ERK pathway in IGF-1-treated BMSCs. Small interfering-SOX4 was transfected into BMSCs to down-regulate SOX4. IGF-1 increased alkaline phosphatase activity, cell calcification, and osteogenic factor (RUNX2, OCN, OSX) levels in BMSCs, indicating that IGF-1 induced rat BMSC OD. SOX4, and p-ERK1/2 and t-ERK1/2 levels were elevated in IGF-1-induced BMSCs, which were annulled by PD98059. PD98059 partly averted IGF-1-induced rat BMSC OD. SOX4 levels, alkaline phosphatase activity, cell calcification, and osteogenic factor (RUNX2, OCN, OSX) levels were reduced after SOX4 down-regulation, showing that downregulation of SOX4 averted the effect of IGF-1 on inducing rat BMSC OD. IGF-1 induced rat BMSC OD by stimulating SOX4 via the MAPK/ERK pathway.
Keywords: Insulin-like growth factor I, Bone marrow mesenchymal stem cells, MAPK/ERK, SRY-related high-mobility-group box 4, Osteogenic differentiation
Introduction
Bone marrow mesenchymal stem cells (BMSCs) are a type of adult stem cells known for their multi-directional differentiation potential and high proliferative activity, which are widely found in mammalian bone marrow and periosteum, and have the ability to differentiate into various cells such as osteoblasts, chondroblasts, nerve cells, and adipocytes under appropriate induction conditions (1, 2). Due to the considerable plasticity of BMSCs, they are considered the optimal choice for seed cells in bone tissue engineering, and are currently applied for the repair of bone defects, and the promotion of osteogenesis and other related diseases (3, 4).
ERK1/2 pathway activation regulates osteogenic differentiation (OD) by modulating transcription factors in the nucleus, such as osterix (OSX) and runt related protein 2 (RUNX2), and osteocalcin (OCN) is a marker of late osteoblast differentiation (5). Insulin-like growth factor-1 (IGF-1) is a principal functional peptide involved in the differentiation and growth of cells, and extracellular matrix protein expression patterns, which also exerts an essential effect on bone remodeling and formation (6). Recen-tly, IGF-1 has been confirmed to stimulate OD of rat mesenchymal stem cells (MSCs) (7). IGF-I promotes proliferation and OD of BMSCs (8). Mitogen-activated protein kinase (MAPK), a class of serine/threonine protein kinases that are widely present in eukaryotic cells, is implicated in various physiological processes such as apoptosis, differentiation, and proliferation of cells (9). Resveratrol activates the extracellular signal-regulated kinase (ERK)/Wnt pathway to stimulate the OD and proliferative capacities of gingival-derived MSCs, thus mitigating periodontitis (10). Eucalyptol induces MC3T3-E1 (osteoblast cell line) OD and primary calcified osteoblasts via ERK phosphorylation (11). Platelet factor 4 represses BMSC OD via suppressing the ITGA5-FAK-ERK pathway, thus leading to bone loss (12). Moreover, IGF-1 activates the MAPK/ERK pathway (13). However, the specific mechanism of whether IGF-1 affects BMSC OD through the MAPK/ERK pathway remains unknown.
SRY-related high-mobility-group box (SOX) transcription factors exert a pivotal effect on determining cell differentiation and cell fate in the process of development (14). SOX4 controls skeletal growth and patterning and facilitates the survival of skeletal progenitor cells (15). SOX4 expression in MC3T3-E1 cells is up-regulated by the aqueous extract of Eucommia leaves, thereby promoting OD (16). Consequently, we postulated that IGF-1 induced OD of BMSCs by facilitating SOX4 expression via the MAPK/ERK pathway. However, the mechanism of IGF-1 in promoting rat BMSC OD by promoting SOX4 via the MAPK/ERK pathway has not been reported. The objective of this study was to examine the mechanism by which IGF-1 induced rat BMSC OD by facilitating SOX4 expression via the MAPK/ERK pathway. This investigation aims to offer a novel approach to enhance the therapeutic efficacy of BMSCs in bone tissue engineering.
Materials and Methods
Rat BMSC culture and identification
Rat BMSCs were purchased from MingBo Co., Ltd. and verified and tested by the short tandem repeat method. BMSCs were cultured in the Dulbecco’s modified Eagle medium (Gibco) containing 10% fetal bovine serum (Gibco) and 1% penicillin-streptomycin (Shanghai Yuanye Bio-Technology Co., Ltd.) in an incubator at 37℃ and 5% CO2, and incubated to the 4th generation and 80% confluence.
The adipogenic, chondrogenic, and osteogenic differentiation abilities of the fourth-generation BMSCs were evaluated. (a) For adipogenic differentiation, BMSCs (2×104) were seeded onto 6-well plates and sequentially fostered in adipogenic differentiation medium ADP1 (Procell) for 3 days and adipogenic differentiation medium ADP2 (Procell) for 1 day, with the operation repeated thrice. Cells were subjected to paraformaldehyde (4%) fixation and staining with Oil Red O (Sigma-Aldrich) for 30 minutes. Next, cells were observed under an inverted microscope (Olympus). (b) In chondrogenic differentiation, BMSCs (5×105) were seeded onto 6-well plates and fostered in chondrogenic differentiation medium (Procell) for 3 weeks. Cells were subjected to fixation in 4% paraformaldehyde, followed by staining with alcian blue (Procell) for 30 minutes. An Olympus inverted microscope was used to observe the cells. (c) For OD, 2×104 BMSCs were seeded onto 6-well plates. After culture in osteogenic medium (OM; Procell) for 3 weeks, cells were fixed in paraformaldehyde (4%). Subsequently, cells were stained with Alizarin Red (Procell) for 10 minutes and observed under an inverted microscope to observe mineralized nodule formation.
Cell transfection and grouping
BMSCs were allocated into the following 6 groups: blank group (BMSCs cultured in OM), IGF-1 group (cultured in OM containing 80 ng/mL IGF-1 [PeproTech] for 3 weeks) (8), IGF-1+PD98059 group (fostered with OM containing 10 μm PD98059 and 80 ng/mL IGF-1 for 3 weeks) (17), IGF-1+dimethyl sulfoxide (DMSO) group (cultured with OM containing 80 ng/mL IGF-1 and the same amount of DMSO as PD98059 for 3 weeks), IGF-1+lentiviral (LV)-small interfering (si)-SOX4 group (transfected with si-SOX4 plasmid and cultured with OM containing 80 ng/mL IGF-1 for 3 weeks), IGF-1+LV-si-negative control (NC) group (transfected with si-NC plasmid and cultured with OM containing 80 ng/mL IGF-1 for 3 weeks).
The 10 nmol/L si-SOX4 and si-NC lentivirus plasmids (GenePharma) were transfected into cells using Lipofecta-mine 2000 (Invitrogen) at a final concentration of 3.75 μL/mL (18). PD98059 (MedChemExpress) was an inhibitor of the MAPK/ERK pathway, and DMSO was used as the solvent control.
Flow cytometry
BMSC-specific cell surface marker levels were assessed by fluorescence-activated cell sorting (FACS). CD105, CD90, and CD44 are known markers of BMSCs, and CD45 and CD34 are markers of hematopoietic cells. In addition, CD34 is partially expressed in some BMSCs. Cells were incubated and separated by trypsin, washed with phosphate buffer saline (PBS), and then labeled with fluorescent antibodies (CD105-fluorescein isothiocyanate [FITC], CD90-FITC, CD44-FITC, CD45-FITC, CD34-FITC; Bio-Legend). Then, the cells incubated with antibodies were analyzed using a BD FACSCalibur flow cytometer (BD Biosciences) to analyze.
Reverse transcription-quantitative polymerase chain reaction
Total RNA was extracted from cells using the TRizol Reagent (Thermo Fisher Scientific). RNA concentration was determined using a spectrophotometer (Thermo Fisher Scientific). High-capacity cDNA reverse transcription kit (Thermo Fisher Scientific) was employed for cDNA synthesis. The ABI ViiATM 7 System (Applied Biosystems) was applied for reverse transcription-quantitative polymerase chain reaction (RT-qPCR) analysis. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as the internal reference for SOX4. Data were analyzed using the 2−ΔΔCt method. The primers are shown in Table 1.
Table 1.
Reverse transcription-quantitative polymerase chain reaction primer sequences
| Name of primer | Sequences (5’-3’) |
|---|---|
| SOX4 | F: GAAGATGACCTGCTCGACCT |
| R: GTGCAATAGTCCGGGAACTC | |
| GAPDH | F: GGCAAGTTCAACGGCACAG |
| R: CGCCAGTAGACTCCACGACAT |
SOX4: SRY-relatedhigh-mobility-group box 4, GAPDH: glyceraldehyde-3-phosphate dehydrogenase, F: forward, R: reverse.
Western blot
Total protein solution in RIPA buffer (Beyotime) was lysed and extracted. Protein concentration was determined using the bicinchoninic acidTM Protein Assay Kit (Pierce). The protein (50 μg) was separated using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (Thermo Fisher Scientific). Then, the separated protein was transferred to the polyvinylidene fluoride membranes (Millipore) and incubated with primary antibodies anti-SOX4 (PA5-72852, 1/1,000; Invitrogen), anti-RUNX2 (PA5-87299, 1/1,000; Invitrogen), anti-OCN (PA5-96529, 1/1,000; Invitrogen), anti-OSX (ab209484, 1/1,000; Abcam), anti-ERK1/2 (ab184699, 1/10,000; Abcam), anti-phosphorylated- (p-) ERK1/2 (ab201015, 1/1,000; Abcam), anti-GAPDH (ab181602, 1:10,000; Ab-cam) overnight at 4℃. After TBST (TBS+Tween) washing, the membranes were incubated with the secondary antibody goat anti-rabbit (immunoglobulin G, 1:2,000, ab205718; Abcam) coupled with horseradish peroxidase for 1 hour, and detected with enhanced chemiluminescence. With GAPDH as the internal reference, the optical value of the band was analyzed using the ImageJ software (National Institutes of Health).
Alkaline phosphatase staining and Alizarin Red staining
After 3 weeks of OD induction, cells were fixed with 4% paraformaldehyde (Beyotime) for 30 minutes and washed with PBS (Beyotime) 3 times. The alkaline phosphatase (ALP) activity of cells was measured according to the instructions of the ALP activity assay kit (Beyotime) and then observed under a microscope (Olympus). The quantity of ALP staining was calculated using the ImageJ software.
After 3 weeks of OD induction, cells were fixed with 4% paraformaldehyde for 30 minutes and washed with PBS 3 times. BMSCs were stained with Alizarin Red staining (ARS) solution (pH 4.3) (Beijing Baiao Laibo Techno-logy) at room temperature for 10 minutes and then rinsed in PBS 3 times. Calcium salt deposition was observed under a microscope. The proportion of ARS staining areas was calculated using the ImageJ software.
Statistical analysis
Prism 8.01 (GraphPad Software Inc.) was applied for data analysis and mapping. Data were expressed as mean±SD. Independent t-test was employed for comparisons between 2 groups. One-way ANOVA was employed for comparisons among groups, followed by Tukey’s multiple comparisons test. p<0.05 was indicative of a statistically significant difference.
Results
IGF-1 induced OD of rat BMSCs
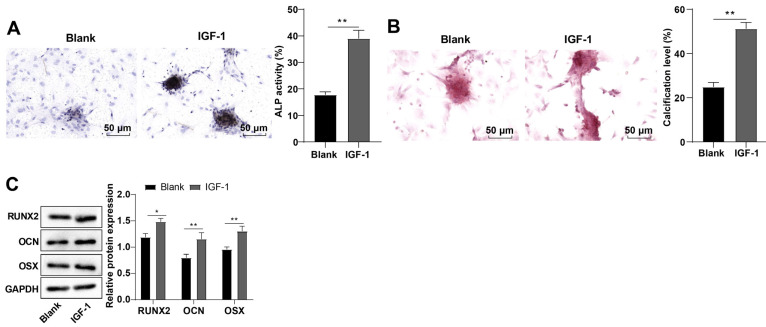
BMSCs were incubated to the 4th generation in vitro, and observed under a microscope, with results demonstrating that cells were spindle-shaped and arranged in a vortex (Supplementary Fig. S1A). BMSCs were characterized by flow cytometry to assess the expression patterns of the markers. The positive rates of BMSCs mesenchymal markers CD105, CD90, and CD44 were more than 95%, and positive rates of hematopoietic cell surface markers CD34 and CD45 were less than 2%, which were negative (Supplementary Fig. S1B). Additionally, BMSCs could differentiate into adipocytes, chondrocytes, and osteoblasts in the adipogenic, chondrogenic, and osteogenic differentiation induction culture system (Supplementary Fig. S1C). IGF-1 pitches in the proliferation and OD of BMSCs, but the exact mechanism remains elusive (8). The cells were induced by OM and treated with 80 ng/mL IGF-1. Then, ALP activity was detected by ALP staining and calcification level of cells was assessed by ARS. ALP activity of cells was enhanced (p<0.01; Fig. 1A) and calcification level was elevated (p<0.01; Fig. 1B) in the IGF-1 group vs. the blank group. Further determination by Western blot uncovered that compared with the blank group, protein levels of RUNX2, OCN, and OSX in the IGF-1 group were elevated (p<0.01; Fig. 1C). These results suggested that IGF-1 induced rat BMSC OD.
Fig. 1.
Insulin-like growth factor-1 (IGF-1) induced rat bone marrow mesenchymal stem cell (BMSC) osteogenic differentiation. (A) Alkaline phosphatase (ALP) activity of cells was assessed by ALP staining. (B) Calcification level of cells was determined by Alizarin Red staining. (C) Runt related protein 2 (RUNX2), osteocalcin (OCN), and osterix (OSX) protein levels in BMSCs were measured by Western blot. The cellular experiments were independently repeated three times. Data were expressed as mean±SD. Comparisons between the two groups were performed by independent sample t-test. GAPDH: glyceraldehyde-3-phosphate dehydrogenase. *p<0.05, **p<0.01.
IGF-1 up-regulated SOX4 expression by activating the MAPK/ERK pathway
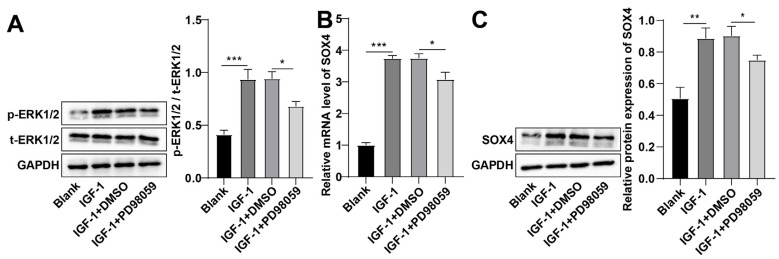
Previous results confirmed that IGF-1 induced rat BMSC OD, but its underlying mechanism was still unclear. The MAPK/ERK pathway regulates OD (11). Therefore, we speculated that IGF-1 might induce rat BMSC OD by regulating the MAPK/ERK pathway. total- (t-) ERK1/2 and p-ERK1/2 levels in BMSCs were measured by Western blot. In contrast to the blank group, p-ERK1/2 and t-ERK1/2 levels in BMSCs of the IGF-1 group were facilitated (p<0.01; Fig. 2A). SOX4 facilitates OD of MC3T3-E1 cells (16). RT-qPCR and Western blot revealed high SOX4 levels in BMSCs of the IGF-1 group than those of the blank group (all p<0.01; Fig. 2B, 2C). PD98059 was used to subdue the MAPK/ERK pathway in IGF-1-treated BMSCs. Relative to the IGF-1+DMSO group, p-ERK1/2, t-ERK1/2, and SOX4 mRNA and protein levels were abated in BMSCs in the IGF-1+PD98059 group (all p<0.05; Fig. 2). These results suggested that IGF-1 promoted SOX4 expression levels by activating the MAPK/ERK pathway.
Fig. 2.
Insulin-like growth factor-1 (IGF-1) up-regulated SRY-related high-mobility-group box 4 (SOX4) expression by activating the mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) pathway. (A) Total- (t-) ERK1/2 and phosphorylated- (p-) ERK1/2 levels in bone marrow mesenchymal stem cells (BMSCs) were assessed by Western blot. (B) SOX4 mRNA expression in BMSCs was measured by reverse transcription-quantitative polymerase chain reaction. (C) SOX4 protein expression in BMSCs was determined by Western blot. The cellular experiments were independently repeated three times. Data were expressed as mean±SD. One-way ANOVA was applied for comparisons among groups, followed by Tukey’s test. GAPDH: glyceraldehyde-3-phosphate dehydrogenase, DMSO: dimethyl sulfoxide. *p<0.05, **p<0.01, ***p<0.001.
The MAPK/ERK pathway inhibition partially abrogated the induction of IGF-1 on rat BMSC OD
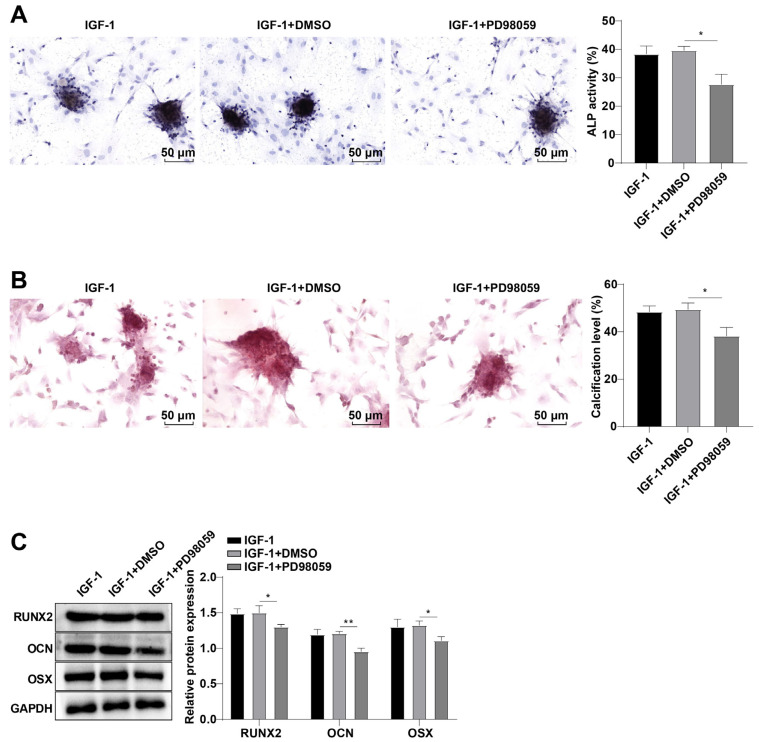
To further verify the role of suppression of the MAPK/ERK pathway in IGF-1-induced BMSC OD, the ALP activity of cells was further determined by ALP staining and the calcification level of cells was detected by ARS stai-ning. Compared with the IGF-1+DMSO group, ALP acti-vity of cells was prominently weakened (p<0.05; Fig. 3A) and calcification level was reduced (p<0.05; Fig. 3B), and RUNX2, OCN, and OSX protein levels in cells were diminished in the IGF-1+PD98059 group (all p<0.05; Fig. 3C). Altogether, the MAPK/ERK pathway repression partially reversed the induction of IGF-1 on rat BMSC OD.
Fig. 3.
The mitogen-activated protein kinase/extracellular signal-regulated kinase (MAPK/ERK) pathway inhibition partially nullified the induction of insulin-like growth factor-1 (IGF-1) on rat bone marrow mesenchymal stem cell osteogenic differentiation. (A) Alkaline phosphatase (ALP) activity of cells was detected by ALP staining. (B) Calcification level of cells was determined by Alizarin Red staining. (C) Runt related protein 2 (RUNX2), osteocalcin (OCN), and osterix (OSX) levels were measured by Western blot. The cellular experiments were independently repeated three times. Data were expressed as the mean±SD. One-way ANOVA was adopted for comparisons among groups, followed by Tukey’s test. DMSO: dimethyl sulfoxide, GAPDH: glyceraldehyde-3-phosphate dehydrogenase. *p<0.05, **p<0.01.
Suppression of SOX4 partially abrogated the induction of IGF-1 on rat BMSC OD
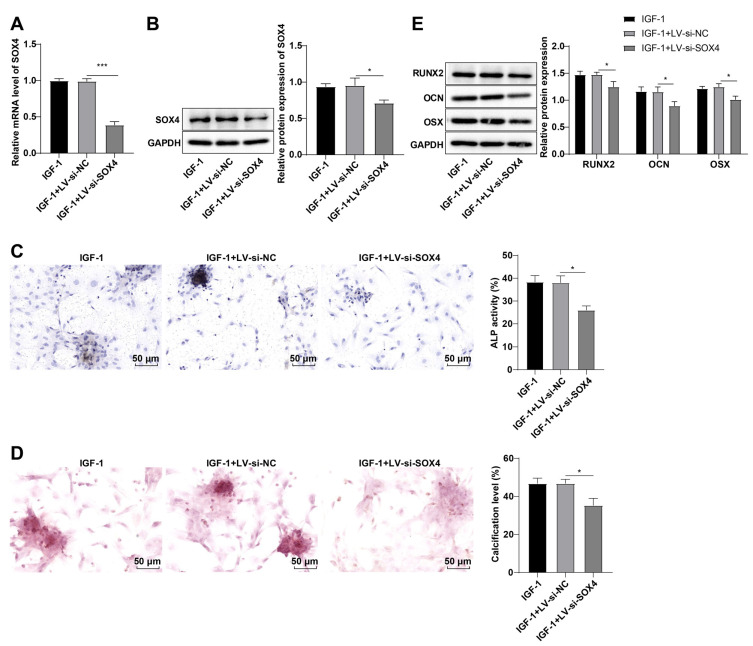
To further demonstrate whether SOX4 downregulation diminished the induction of IGF-1 on rat BMSC OD, si-SOX4 lentivirus plasmid was transfected into BMSCs to repress SOX4 expression. SOX4 mRNA and protein levels were abated (all p<0.05; Fig. 4A, 4B), ALP activity was hindered (p<0.05; Fig. 4C), calcification level was reduced (p<0.05; Fig. 4D), and RUNX2, OCN, and OSX levels were decreased in cells in the IGF-1+LV-si-SOX4 group vs. the IGF-1+LV-si-NC group (all p<0.05; Fig. 4E). Overall, SOX4 downregulation partially abrogated the induction of IGF-1 on rat BMSC OD.
Fig. 4.
Knockdown of SRY-related high-mobility-group box 4 (SOX4) partially reversed the effect of insulin-like growth factor-1 (IGF-1) on inducing rat bone marrow mesenchymal stem cell (BMSC) osteogenic differentiation. (A) SOX4 mRNA expression in BMSCs was assessed by reverse transcription-quantitative polymerase chain reaction. (B) SOX4 protein level in BMSCs was determined by Western blot. (C) Alkaline phosphatase (ALP) activity of cells was measured by ALP staining. (D) Calcification level of cells was measured by Alizarin Red staining. (E) Runt related protein 2 (RUNX2), osteocalcin (OCN), and osterix (OSX) protein levels in BMSCs were assessed by Western blot. The cellular experiments were independently repeated three times. Data were expressed as the mean±SD. One-way ANOVA was adopted for comparisons among groups, followed by Tukey’s test. GAPDH: glyceraldehyde-3-phosphate dehydrogenase, si: small interfering. *p<0.05, ***p<0.001.
Discussion
The study discovered that IGF-1 elevated the expression of SOX4 by activating the MAPK/ERK pathway, consequently promoting rat BMSC OD.
MSCs, which can differentiate directly into bone cells, attract and recruit other cells, or create a regenerative environment by producing nutritive growth factors, also play a pivotal role in the bone healing process (19). BMSCs have a principal effect on the remodeling, development, and regeneration of bones (20). Evidence has shown that IGF-1 is a vital regulator of bone metabolism and remodeling and plays an imperative role in the maintenance and achievement of bone mass throughout life (21). Preceding reference reported that miR-222-3p and miR-221-3p suppress the OD of BMSCs through the IGF-1/ERK pathway under high-glucose (22). Our results elicited that BMSCs treated with IGF-1 manifested boosted calcification level, ALP activity, and protein levels of osteogenic factors RUNX2, OCN, and OSX. Consistently, cotransfection with IGF-1 and osteogenic protein-1 stimulates ALP activity and promotes OD (23). IGF-1 promotes mineralization of bone and up-regulates RUNX2 and OCN (24). As a multifunctional peptide involved in the differentiation and prolife-ration of cells, IGF-1 induces strong OD in BMSCs (25). To conclude, IGF-1 induced the OD of rat BMSCs.
The MAPK/ERK pathway plays a vital part in manipulating OD by facilitating RUNX2 activity (26-28). miR-210-3p represses BMSC OD by constraining the MAPK pathway and targeting KRAS (29). Jin et al. (30) demonstrated that activation of the ERK pathway facilitated OD of human adipose derived stem cells. The aqueous extract of Eucommia aurantium elevates SOX4 level in osteoblast-like MC3T3-E1 cells, thereby stimulating OD (16). We spe-culated that IGF-1 might induce OD of BMSCs by modulating the MAPK/ERK pathway. Our findings elaborated that IGF-1 raised p-ERK1/2, t-ERK1/2, and SOX4 levels, which were averted by the MAPK/ERK pathway inhibitor. Likewise, IGF-1 promotes the OD and proliferation of human dental pulp stem cells via activating the MAPK pathway (31). IGF-1 promotes alveolar osteoblast proliferation through the MAPK pathway activation (32). IGF-1 facilitates OD of rat MSCs by increasing transcriptional coactivator with PDZ-binding motif expression via the MEK-ERK pathway (7). IGF-1 triggers OD and osteogenesis of STRO-1(+) periodontal ligament stem cells through JNK MAPK and ERK pathways (33). Altogether, IGF-1 facilitated SOX4 levels by activating the MAPK/ERK pathway.
To further investigate whether IGF-1 induced OD of rat BMSCs, we inhibited the MAPK/ERK pathway using its inhibitor PD98059 in BMSCs treated with IGF-1 and discovered that ALP activity and calcification level were repressed, and the protein levels of RUNX2, OCN, and OSX were diminished. Inactivation of the ERK pathway suppresses the promotion of MC3T3-E1 OD by Nell-1 (34). Blockade of the MAPK/ERK pathway inhibits p-ERK protein expression and reduces osteogenic gene expression and ALP levels of BMSCs (35). Briefly, the MAPK/ERK pathway inhibitor partially abrogated the induction of IGF-1 on OD of rat BMSCs. Furthermore, to further investigate whether downregulation of SOX4 diminished the induction of IGF-1 on OD in rat BMSCs, we transfected BMSCs with si-SOX4 lentivirus plasmid to suppress SOX4 expression. Our results manifested that SOX4 mRNA and protein expression levels, ALP activity, and calcification level were repressed, and the protein levels of RUNX2, OCN, and OSX were inhibited. It has been documented that knockdown of SOX4 in osteoblasts impedes OD and reduces proliferative ability of progenitor cells (15). Taken together, silencing of SOX4 partially counteracted the effect of IGF-1 on OD of rat BMSCs.
In summary, this study supported that IGF-1 induced the OD of rat BMSCs by promoting SOX4 via the MAPK/ERK pathway. However, we only studied the effect of IGF-1 on the OD in rat BMSCs without in vivo verification. In the future, we will further study whether IGF-1 can achieve similar effects in animals and whether IGF-1 can be used for the repair of bone defects, and the promotion of osteogenesis, and other related diseases.
Supplementary Materials
Supplementary data including one figure can be found with this article online at https://doi.org/10.15283/ijsc23165
Footnotes
Potential Conflict of Interest
There is no potential conflict of interest to declare.
Authors’ Contribution
Conceptualization: ZW. Data curation: JZ. Formal analysis: CH. Funding acquisition: ZW. Investigation: XL. Methodology: JZ. Project administration: ZW. Resources: CH. Software: JD. Supervision: ZW. Validation: JZ. Visualization: JD. Writing – original draft: QX. Writing – review and editing: JZ, ZW.
Funding
This work was supported by grants from Science and Technology Research Project of Jiangxi Provincial Depart-ment of Education (GJJ210221).
References
- 1.Zhou Y, Qiao H, Liu L, et al. miR-21 regulates osteogenic and adipogenic differentiation of BMSCs by targeting PTEN. J Musculoskelet Neuronal Interact. 2021;21:568–576. [PMC free article] [PubMed] [Google Scholar]
- 2.Shen JJ, Zhang CH, Chen ZW, et al. LncRNA HOTAIR inhibited osteogenic differentiation of BMSCs by regulating Wnt/β-catenin pathway. Eur Rev Med Pharmacol Sci. 2019;23:7232–7246. doi: 10.26355/eurrev_201909_18826. [DOI] [PubMed] [Google Scholar]
- 3.Lee YC, Chan YH, Hsieh SC, Lew WZ, Feng SW. Comparing the osteogenic potentials and bone regeneration capacities of bone marrow and dental pulp mesenchymal stem cells in a rabbit calvarial bone defect model. Int J Mol Sci. 2019;20:5015. doi: 10.3390/ijms20205015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lang E, Semon JA. Mesenchymal stem cells in the treatment of osteogenesis imperfecta. Cell Regen. 2023;12:7. doi: 10.1186/s13619-022-00146-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhuang LF, Jiang HH, Qiao SC, et al. The roles of extracellular signal-regulated kinase 1/2 pathway in regulating osteogenic differentiation of murine preosteoblasts MC3T3-E1 cells on roughened titanium surfaces. J Biomed Mater Res A. 2012;100:125–133. doi: 10.1002/jbm.a.33247. [DOI] [PubMed] [Google Scholar]
- 6.Zhou J, Wei F, Ma Y. Inhibiting PPARγ by erythropoietin while upregulating TAZ by IGF1 synergistically promote osteogenic differentiation of mesenchymal stem cells. Bio-chem Biophys Res Commun. 2016;478:349–355. doi: 10.1016/j.bbrc.2016.07.049. [DOI] [PubMed] [Google Scholar]
- 7.Xue P, Wu X, Zhou L, et al. IGF1 promotes osteogenic differentiation of mesenchymal stem cells derived from rat bone marrow by increasing TAZ expression. Biochem Biophys Res Commun. 2013;433:226–231. doi: 10.1016/j.bbrc.2013.02.088. [DOI] [PubMed] [Google Scholar]
- 8.Feng J, Meng Z. Insulin growth factor-1 promotes the proliferation and osteogenic differentiation of bone marrow mesenchymal stem cells through the Wnt/β-catenin path-way. Exp Ther Med. 2021;22:891. doi: 10.3892/etm.2021.10323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang Q, Zhang L, Qu J, et al. Insights into the regulation of MAP3K1 in hair follicle stem cells by transcriptome analysis. Anim Biotechnol. 2023;34:686–697. doi: 10.1080/10495398.2022.2145293. [DOI] [PubMed] [Google Scholar]
- 10.Jiang H, Ni J, Hu L, et al. Resveratrol may reduce the degree of periodontitis by regulating ERK pathway in gingival-derived MSCs. Int J Mol Sci. 2023;24:11294. doi: 10.3390/ijms241411294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee DW, Kim KM, Park S, An SH, Lim YJ, Jang WG. Eucalyptol induces osteoblast differentiation through ERK phosphorylation in vitro and in vivo. J Mol Med (Berl) 2023;101:1083–1095. doi: 10.1007/s00109-023-02348-x. [DOI] [PubMed] [Google Scholar]
- 12.Li W, Zhang Q, Gu R, Zeng L, Liu H. Platelet factor 4 induces bone loss by inhibiting the integrin α5-FAK-ERK pathway. Animal Model Exp Med. 2023;6:573–584. doi: 10.1002/ame2.12342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lai W, Yu L. Insulin-like growth factor 1 ameliorates pre-eclampsia by inhibiting zinc finger E-box binding homeobox 1 by up-regulation of microRNA-183. J Cell Mol Med. 2023;27:1179–1191. doi: 10.1111/jcmm.17403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Enkhmandakh B, Bayarsaihan D. Single-cell transcriptome profiling reveals distinct expression patterns among genes in the mouse incisor dental pulp. Int J Dev Biol. 2023;67:19–25. doi: 10.1387/ijdb.220173db. [DOI] [PubMed] [Google Scholar]
- 15.Lefebvre V. Roles and regulation of SOX transcription factors in skeletogenesis. Curr Top Dev Biol. 2019;133:171–193. doi: 10.1016/bs.ctdb.2019.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Guan M, Pan D, Zhang M, Leng X, Yao B. The aqueous extract of eucommia leaves promotes proliferation, differentiation, and mineralization of osteoblast-like MC3T3-E1 cells. Evid Based Complement Alternat Med. 2021;2021:3641317. doi: 10.1155/2021/3641317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Luo J, Xue D, Song F, Liu X, Li W, Wang Y. DUSP5 (dual-specificity protein phosphatase 5) suppresses BCG-induced autophagy via ERK 1/2 signaling pathway. Mol Immunol. 2020;126:101–109. doi: 10.1016/j.molimm.2020.07.019. [DOI] [PubMed] [Google Scholar]
- 18.Zietzer A, Hosen MR, Wang H, et al. The RNA-binding protein hnRNPU regulates the sorting of microRNA-30c-5p into large extracellular vesicles. J Extracell Vesicles. 2020;9:1786967. doi: 10.1080/20013078.2020.1786967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Lin H, Sohn J, Shen H, Langhans MT, Tuan RS. Bone marrow mesenchymal stem cells: aging and tissue engineering applications to enhance bone healing. Biomaterials. 2019;203:96–110. doi: 10.1016/j.biomaterials.2018.06.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pan F, Huang K, Dai H, Sha C. PHF8 promotes osteogenic differentiation of BMSCs in old rat with osteoporosis by regulating Wnt/β-catenin pathway. Open Life Sci. 2022;17:1591–1599. doi: 10.1515/biol-2022-0523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mazziotti G, Lania AG, Canalis E. Skeletal disorders associated with the growth hormone-insulin-like growth factor 1 axis. Nat Rev Endocrinol. 2022;18:353–365. doi: 10.1038/s41574-022-00649-8. [DOI] [PubMed] [Google Scholar]
- 22.Gan K, Dong GH, Wang N, Zhu JF. miR-221-3p and miR-222-3p downregulation promoted osteogenic differentiation of bone marrow mesenchyme stem cells through IGF-1/ERK pathway under high glucose condition. Diabetes Res Clin Pract. 2020;167:108121. doi: 10.1016/j.diabres.2020.108121. [DOI] [PubMed] [Google Scholar]
- 23.Yeh LC, Lee JC. Co-transfection with the osteogenic protein (OP)-1 gene and the insulin-like growth factor (IGF)-I gene enhanced osteoblastic cell differentiation. Biochim Biophys Acta. 2006;1763:57–63. doi: 10.1016/j.bbamcr.2005.11.001. [DOI] [PubMed] [Google Scholar]
- 24.Yuan Y, Duan R, Wu B, et al. Gene expression profiles and bioinformatics analysis of insulin-like growth factor-1 promotion of osteogenic differentiation. Mol Genet Genomic Med. 2019;7:e00921. doi: 10.1002/mgg3.921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wu L, Zhang G, Guo C, Pan Y. Intracellular Ca2+ signaling mediates IGF-1-induced osteogenic differentiation in bone marrow mesenchymal stem cells. Biochem Biophys Res Commun. 2020;527:200–206. doi: 10.1016/j.bbrc.2020.04.048. [DOI] [PubMed] [Google Scholar]
- 26.Zhou T, Guo S, Zhang Y, Weng Y, Wang L, Ma J. GATA4 regulates osteoblastic differentiation and bone remodeling via p38-mediated signaling. J Mol Histol. 2017;48:187–197. doi: 10.1007/s10735-017-9719-2. [DOI] [PubMed] [Google Scholar]
- 27.Wu Y, Xia L, Zhou Y, Xu Y, Jiang X. Icariin induces osteogenic differentiation of bone mesenchymal stem cells in a MAPK-dependent manner. Cell Prolif. 2015;48:375–384. doi: 10.1111/cpr.12185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Son JH, Park BS, Kim IR, et al. A novel combination treatment to stimulate bone healing and regeneration under hypoxic conditions: photobiomodulation and melatonin. Lasers Med Sci. 2017;32:533–541. doi: 10.1007/s10103-017-2145-6. [DOI] [PubMed] [Google Scholar]
- 29.Hu M, Zhu X, Yuan H, Li H, Liao H, Chen S. The function and mechanism of the miR-210-3p/KRAS axis in bone marrow-derived mesenchymal stem cell from patients with osteoporosis. J Tissue Eng Regen Med. 2021;15:699–711. doi: 10.1002/term.3215. [DOI] [PubMed] [Google Scholar]
- 30.Jin C, Shuai T, Tang Z. HSPB7 regulates osteogenic differentiation of human adipose derived stem cells via ERK signaling pathway. Stem Cell Res Ther. 2020;11:450. doi: 10.1186/s13287-020-01965-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lv T, Wu Y, Mu C, et al. Insulin-like growth factor 1 promotes the proliferation and committed differentiation of human dental pulp stem cells through MAPK pathways. Arch Oral Biol. 2016;72:116–123. doi: 10.1016/j.archoralbio.2016.08.011. [DOI] [PubMed] [Google Scholar]
- 32.Li X, Yao J, Wu J, Du X, Jing W, Liu L. Roles of PRF and IGF-1 in promoting alveolar osteoblast growth and proliferation and molecular mechanism. Int J Clin Exp Pathol. 2018;11:3294–3301. [PMC free article] [PubMed] [Google Scholar]
- 33.Yu Y, Mu J, Fan Z, et al. Insulin-like growth factor 1 enhances the proliferation and osteogenic differentiation of human periodontal ligament stem cells via ERK and JNK MAPK pathways. Histochem Cell Biol. 2012;137:513–525. doi: 10.1007/s00418-011-0908-x. [DOI] [PubMed] [Google Scholar]
- 34.Shen MJ, Wang GG, Wang YZ, Xie J, Ding X. Nell-1 enhances osteogenic differentiation of pre-osteoblasts on titanium surfaces via the MAPK-ERK signaling pathway. Cell Physiol Biochem. 2018;50:1522–1534. doi: 10.1159/000494651. [DOI] [PubMed] [Google Scholar]
- 35.Dou X, Wei X, Liu G, et al. Effect of porous tantalum on promoting the osteogenic differentiation of bone marrow mesenchymal stem cells in vitro through the MAPK/ERK signal pathway. J Orthop Translat. 2019;19:81–93. doi: 10.1016/j.jot.2019.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.