Abstract
Abstract
The global imperative to enhance crop protection while preserving the environment has increased interest in the application of biological pesticides. Bacillus thuringiensis (Bt) is a Gram-positive bacterium that can produce nematicidal proteins and accumulate them in parasporal crystals. Root-knot nematodes are obligate root plant parasitic which are distributed worldwide, causing severe damages to the infested plants and, consequently, large yield reductions. In this work, we have evaluated the toxicity of the Bt crystal proteins Cry5, Cry21, App6, and Xpp55 against two root-knot nematodes belonging to the Meloidogyne genus (M. incognita and M. javanica). The results show that all four proteins, when solubilized, were highly toxic for both nematode species. To check the potential of using Bt strains producing nematicidal crystal proteins as biopesticides to control root-knot nematodes in the field, in planta assays were conducted, using two wild Bt strains which produced Cry5 or a combination of App6 and Cry5 proteins. The tests were carried out with cucumber or with tomato plants infested with M. javanica J2, irrigated with spore + cristal mixtures of the respective strains. The results showed that the effectiveness of the nematicidal activity was plant-dependent, as Bt was able to reduce emerged J2 in tomato plants but not in cucumber plants. In addition, the toxicity observed in the in planta assays was much lower than expected, highlighting the difficulty of the proteins supplied as crystals to exert their toxicity. This emphasizes the delivery of the Bt proteins as crucial for its use to control root-knot nematodes.
Key points
• Solubilized Cry5, Cry21, App6 and Xpp55 Bt proteins are toxic to M. javanica.
• Cry21 toxicity to M. incognita is similar to that of Cry5, App6, and Xpp55 proteins.
• The Cry5 and App6 toxicities on M. javanica after Bt irrigation is crop dependent.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00253-024-13365-2.
Keywords: Bio-control, In vitro assay, In planta assay, Bioassay, Bt wild strains
Introduction
Plant parasitic nematodes (PPN) are one of the most detrimental plant parasites, causing yield losses in the agricultural sector of up to 12.3% (157 billion dollars) worldwide (Singh et al. 2015). PPNs feed on the cytoplasm of live plant cells triggering symptoms in the plant above-ground (similar to those caused by water or nutrient deficiencies in the soil) and below-ground (unhealthy roots, and root galling or swelling) (Bridge and Starr 2007). One of the most prevalent plant parasitic nematodes are root-knot nematodes (Meloidogyne spp.), which cause the majority of nematode damage to crops worldwide, mainly due to their broad host range and global distribution (Atkinson et al. 2012). They infect more than 3000 plant species including vegetables, fruit trees, cereals, and ornamental flowers (Abad et al. 2003). As an example, in the Mediterranean basin, production losses caused by Meloidogyne species in horticultural crops are estimated between 15 and 60% (Andres and Verdejo-Lucas 2011).
Currently, nearly 100 species of Meloidogyne have been identified but the four most common species are M. incognita, M. javanica, M. arenaria, and M. hapla (Bridge and Starr 2007; Jones et al. 2013), which often share habitats. They are present in almost all world regions. Indeed, the optimal temperature for the development of M. arenaria and M. javanica is 28°C, whereas for M. hapla is 24°C; although, it is able to survive at field temperatures below 0°C (Bridge and Starr 2007). M. incognita is probably the most widely distributed nematode in tropical and subtropical regions, particularly in the warmer areas (Eisenback 2022), and M. javanica is widely distributed in warm and tropical climates in the Americas, Asia, Australia, Africa, and Europe. It is often the dominant root-knot species, affecting more than 750 plant species, some of them with high economic relevance. Indeed, M. javanica is considered a major agricultural pest in many countries (CABI Head Office 2021).
Effectively combating nematodes is a formidable challenge, given their ability to persist for many years in infested agricultural soils, making their eradication extremely difficult in practice. Traditional control methods have been applied, including chemical treatments, cultural practices (such as soil resting and tillage or crop rotation), as well as the use of plant varieties with reduced susceptibility to nematode attacks (Andres and Verdejo-Lucas 2011; Khan 2023). Chemical control has been historically the prevalent method due to its rapid action and high effectiveness. However, legislative restrictions and the growing concerns related to health and environmental problems have shifted the focus towards exploring genetic, agronomic, physical interventions, and biological methods (Andres and Verdejo-Lucas 2011; Atkinson et al. 2012; Chitwood 2003; Dutta and Phani 2023; Engelbrecht et al. 2018; Khan et al. 2023; Siddique and Akker 2021).
The biopesticides predominantly used today are those derived from the Gram-positive bacterium Bacillus thuringiensis (Bt) (Olson 2015; Sanahuja et al. 2011). The Bt-based products are highly specific against a wide range of invertebrates (Jurat-Fuentes and Crickmore 2017; Palma et al. 2014) and do not pose risks to the environment or human health (Koch et al. 2015; Raymond and Federici 2017). Some Bt proteins belonging to the App6, Cry5, Cry12, Cry13, Cry14, Cry21, Cry31, and Xpp55 families have shown to be active against nematodes (Bel et al. 2022; Jouzani et al. 2017; Liang et al. 2022; Palma et al. 2014; Wei et al. 2003).
Regarding Meloidogyne spp., some in vitro studies have evidenced the toxicity of Cry5, App6, and Xpp55 against M. hapla (Guo et al. 2008; Yu et al. 2008, 2015; Zhang et al. 2012) or M. incognita (Peng et al. 2011). And when expressed in transgenic plants, Cry5 (Li et al. 2008; Wang et al. 2024) and App6 (Li et al. 2007) have shown a high protection against M. incognita infestation.
In this work, we have conducted the first assessment of Cry 21 toxicity against M. incognita and determined its toxicity parameters. In addition, we have evaluated the nematicidal activities of Cry5, Cry21, App6, and Xpp55 proteins against M. javanica. The results reveal that each solubilized protein is highly toxic for the tested nematode species pointing them as promising candidates to control these pests. However, the challenge lies in delivering effectively Bt proteins to achieve Meloydogyne control, as its toxicity decreases largely when they are supplied to the plants by traditional irrigation methods (using spore + crystal suspensions). This decline in efficacy is likely attributed to the biology and feeding behavior of the targeted nematode species.
Materials and methods
Nematode rearing
M. javanica and M. incognita were obtained from Spanish field populations, usually tomato fields, located in different geographical regions at different times. The root-knot nematode populations were adapted to laboratory conditions in the Research Koppert facilities in Almeria and maintained by infesting nematode susceptible tomato plants (Roma and Boludo varieties) in greenhouse-controlled conditions.
To amplify the nematode populations for the in vitro and in planta assays, tomato plants grown in pots containing sterile sand were inoculated with three egg masses. The plants were kept for 3–6 months. After that time, the root-knot nematode-infected roots were used to obtain second-stage juveniles (J2) for the tests, by digging up, washing, and cutting the roots into small sections. The sections were immersed in 10% NaClO and blended at high speed for two successive 10-s periods with an interval of 5 s between them. After a 5-min maceration, the suspension was passed through a 74-µm sieve to remove root debris. The dispersed eggs retained in the sieve were concentrated by using 20-µm sieves. The eggs were further purified using the sugar flotation technique, mixing the eggs with saccharose 1 M and centrifuging the suspension 10 min at 1300 rpm in 50-ml round bottom tubes. Then, the magnesium sulfate differential centrifugation method was applied, pipetting 0.9 M magnesium sulfate solution underneath the egg suspension to form a density gradient, and centrifuging the tubes at 1500 rpm for 3 min. The upper aqueous fraction containing the eggs was drawn off with a pipette, placed over a 20-µm sieve, and rinsed repeatedly with tap water to remove the residual magnesium sulfate. The egg suspension was then placed on small modified Baermann trays which were kept at 24–25 °C. The hatched J2 were collected after 3–5 days and used for the toxicity tests or plant infestations.
Bacterial strains
Two recombinant Bt strains were used to produce the Cry5 and Cry21 proteins. The recombinant Bt strain BMB0215 expressing cry5B (Guo et al. 2008) was kindly supplied by Dr. Sun (State Key Laboratory of Agricultural Microbiology, Huazhong Agricultural University, Wuhan, People’s Republic of China). The Bt strain DB27 (which contains cry21 genes) (Iatsenko et al. 2014) was kindly provided by Dr. Sommer (Max Planck Institute for Developmental Biology, Department of Evolutionary Biology, Tübingen, Germany).
To produce the nematicidal proteins App6 and Xpp55, the App6Aa2 (NCBI Acc. No. AF499736) and Xpp55Aa1 (NCBI Acc. No EU121522) sequences were chosen to design the genes to be cloned and expressed in E. coli. The synthetic gene sequences obtained after codon optimization (see Online Resource1) were synthesized by GenScript (New Jersey, USA). Plasmids containing the respective genes were cloned in BL21 E. coli cells, and the proteins were expressed in Luria–Bertani (LB) broth supplemented with kanamycin, after induction with isopropyl-β-D-thiogalactopyranoside (IPTG).
The V-CO3.3 and V-AB8.18 wild Bt strains (deposited in the Spanish Type Culture Collection, Accession numbers CECT 31068 and CECT 31067, respectively) were selected as wild-type Bt strains with potential nematicidal activity after an exhaustive screening of wild strains belonging to several Bt collections kept at the University of Valencia (Bel et al. 2023). The PCR screening to check the presence of the cry1, cry2, cry3, cry5, app6, cry12, cry13, cry14, cry21, and xpp55 genes was performed as described previously (Bel et al. 2023).
The microscopic observation of the V-CO3.3 and V-AB8.18 strains by scanning electron microscopy (SEM) was performed as reported by Bel et al. (Bel et al. 2023).
Type I β-exotoxin analyses
The ability of the wild Bt strains to produce type I β-exotoxin was determined by LC–MS/MS at the SCSIE (University of Valencia, Spain) with a mass spectrometer nanoESI Q-TOF (TripleTOFTM 5600, ABSciex) as described elsewhere (Khorramnejad et al. 2018). The Bt strain HD2, kindly supplied by I. Thiéry (Institute Pasteur, France), was used as positive control (Hernández et al. 2001), and the β-exotoxin type I toxin produced by this strain was used to generate the calibration curve. Bt HD1 strain was used as negative control (Hernández et al. 2001).
Production of the spore/crystal mixtures and obtaining the solubilized crystal proteins
The Bt crystal proteins were produced and isolated from the Bt strains following the protocol described by Estela et al. (Estela et al. 2004) with some modifications. In short, the Bt strains were grown in CCY media (Stewart et al. 1981) for 48 h. Spores and crystals were collected by centrifugation (10 min, 16,000 xg, 4 °C) and washed three times with 1 M NaCl supplemented with 10 mM ethylenediaminetetraacetic acid (EDTA) and twice with 10 mM KCl. The proteins present in the crystals were solubilized after adding 50 mM carbonate buffer pH 10.5, 10 mM dithiothreitol (DTT) to the spore + crystal mixture, and incubating the suspension o/n at 4 °C. Then, the sample was centrifuged at 24,000 xg for 15 min. The purity of the solubilized proteins in the supernatant was checked by 12% sodium dodecyl sulphate polyacrylamide gel electrophoresis (SDS-PAGE). For in vitro bioassay purposes, the solubilized proteins were dialyzed in 20 mM HEPES buffer, pH 8 (o/n, 4 °C, slight stirring). After a 5-min centrifugation at 21,130 xg, the proteins were checked by SDS-PAGE and quantified by Bradford (Bradford 1976).
For the in planta experiments, the spore + crystal samples were lyophilized after the washing steps. For quantification purposes, 1 mg of the lyophilized sample was suspended in 0.1 ml of 20 mM HEPES buffer pH 8, and the number of spores in the sample was assessed under the microscope using a Neubauer chamber. The sample was then diluted in 20 mM HEPES buffer pH 8 to the appropriate concentration for plant irrigation.
The App6Aa2 and Xpp55Aa1 proteins were obtained from the transformed E. coli cells as inclusion bodies which were isolated after cell precipitation, lysis, and six washing steps (three times with 20 mM Tris–HCl, 1 M NaCl, 1% Triton X100, pH7.5 and three times with a phosphate-buffered saline buffer (PBS, Fisher Bioreagents, Geel, Belgium)) as described elsewhere (Sayyed et al. 2005). The proteins expressed by the respective E. coli cultures were checked by 12% SDS-PAGE. The Xpp55A protein was detected in the inclusion bodies, unlike App6A protein that was detected in both inclusion bodies and supernatants. The App6 and Xpp55 proteins from inclusion bodies were solubilized and quantified as has been described above for Cry5 and Cry21 proteins.
Crystal protein identification by LC–MS/MS
The identity of the proteins present in the crystals was determined by LC–MS/MS, following the procedure described in Bel et al. (Bel et al. 2023). The protein samples were analyzed at the Proteomics Unit of Servei Central de Suport a la Investigació Experimental (SCSIE, University of Valencia, Spain). The analyses were performed with a LC–MS/MS system composed of an Ekspert nanoLC 425 (Eksigent Technologies LLC, CA, USA) and a mass spectrometer nanoESI qQTOF (6600plus TripleTOF, ABSCIEX, MA, USA) as described elsewhere (Barranco et al. 2023; Khorramnejad et al. 2020). The identification of the proteins in the samples was done by the Parangon algorithm (Shilov et al. 2007) via the Protein Pilot V 5.0 (ABSciex, Madrid, Spain) for a generation of the list of peptides and the search in the Uniprot databases. Only identification hits with a minimum of 95% confidence were considered significant. The mass spectrometry proteomics data have been deposited to the ProteomeXchange Consortium via the PRIDE (Perez-Riverol et al. 2022) partner repository with the dataset identifier PXD053413.
In vitro bioassays
The nematicidal activity of the solubilized Bt proteins was determined by performing in vitro bioassay tests in sterile conditions using 96-microwell flat-bottom sterile polystyrene plates.
The buffer 20 mM HEPES pH 8 was chosen as carrier buffer for the bioassays as it was tested as harmless for the nematodes (the Bt crystal protein solubilization buffer 50 mM carbonate buffer pH 10.5, 10 mM DTT resulted toxic for nematodes although DDT was innocuous). The solubilized Bt crystal proteins dialyzed were diluted to the different bioassay concentrations in 20 mM HEPES buffer pH 8. Buffer without Bt proteins was used as a control. In each bioassay, ten replicates per concentration were done. The controls and protein solutions were distributed in the 96-microwells in a final volume of 90 µl. Then, 10-µl aliquots of a J2 suspension containing 30–40 individuals were added to each well reaching a final volume of 100 µl. The plates were incubated at 25°C in darkness, for 24 h. The number of moving (alive) and immobilized J2 nematodes (considered dead) in each well were recorded with the help of a stereomicroscope Olympus SZ51. Each Bt protein was bioassayed three to eight times. The toxicity parameters were obtained using the POLO-PC probit analysis program (LeOra Software, Berkeley, CA, USA). The LC50 values were considered significantly different when 95% fiducial limits did not overlap.
In planta assays
The in planta assays to test the nematicidal activity of the crystal + spore suspensions of the wild Bt strains were performed with plants of cucumber (variety Jungla) or tomato (variety Roma), infested with M. javanica. The plant seeds were first deposited in a humidity chamber for 2 to 3 days at 25°C to allow germination. Then, seedlings were transplanted into 200-cc pots containing an autoclaved mixture of sand and vermiculite (2:1, v:v) and allowed to grow for 7 days in a climate chamber with controlled conditions (25°C, 75% humidity, 16:8 day:night photoperiod). At day 7, the plants were inoculated with 350 M. javanica J2 nematodes. A minimum of four plants were used in each assay, and at least three replicates of each assay were performed.
The lyophilized Bt spore + crystal productions were resuspended in 10 ml 20 mM HEPES buffer pH 8 at room temperature to a concentration of 1 × 109 spores/ml. After a vigorous stirring with vortex, the suspension was applied to the plant. The control plants consisted of plants non-inoculated with nematodes. The positive control (nematode killing control) consisted of infested plants irrigated with 0.8 cc of DuPont™ Vydate® C-LV insecticide/nematicide, whose active ingredient is oxamyl, a carbamate molecule used as nematicide on many field crops, vegetables, fruits, and ornamentals.
The treatment consisted of irrigating the plants with Bt spores + crystals twice: firstly, 3 days before the nematode infestation and then at day 7 just before the plant infestation with 350 M. javanica J2. The plants were analysed individually 8 weeks later. The parameters recorded were the number of egg masses in roots (evidenced using the vital dye erioglaucine, Sigma-Aldrich Merck KGaA, Darmstadt, Germany), the J2 that emerged from them, and the dry foliar weight (evaluated after removing the aerial part of the plant). The Grubb test (extreme studentized deviant (ESD) method) with a significance level of α = 0.05 was used to detect outliers. The one sample t-test with α = 0.5 was applied to detect differences in the means of treated plants regarding their respective controls, after setting the hypothetical value to 100.
Results
Toxicity of Cry5, App6, Cry21, and Xpp55 proteins against M. javanica and M. incognita
The Bt Cry5, Cry21, App6, and Xpp55 proteins were produced, purified, solubilized, and dialyzed in 20 mM HEPES pH 8. In vitro bioassays were performed using M. javanica and M. incognita captured from two or more different localities that had been reared for at least one generation in greenhouses before being used for the toxicity assays.
The results showed no significant variability in toxicity between bioassays from different locations, whether using M. incognita or M. javanica populations. Therefore, the toxicity data analyses were performed by pooling data from all localities in both Meloidogyne species. The toxicological parameters obtained are summarized in Table 1.
Table 1.
In vitro analysis of the susceptibility of M. javanica and M. incognita to four solubilized Bt nematicidal proteins. The toxicity parameters were obtained using POLO-PC probit analysis program (LeOra Software, Berkeley, CA, USA) and correspond to the 50% lethal concentration (LC50) with its 95% fiducial limits (FL95) and the slope of the dose-mortality curve with the error represented by the standard deviation. The toxicity curves obtained for Cry5, App6, Cry21, and Xpp55 in each of the nematode species were not parallel and therefore, the calculation of relative potencies was not applicable. The only exception occurred with Cry5 and Cry21 toxicity curves obtained with M. javanica (relative potency = 0.92, FL95 = 0.84–1.02). The relative potencies shown in the table were calculated by dividing the LC50 value of the Cry5 by that of the other proteins
| Bt protein | M. javanica | |||
| LC50 (µg/ml) | FL95 (µg/ml) | Slope | Relative potency (LC50 ratio)* | |
| Cry5 | 529 | 467–594 | 3.9 ± 0.2 | 1 |
| App6 | 234 | 176–289 | 3.0 ± 0.1 | 2.3 |
| Cry21 | 574 | 431–711 | 4.0 ± 0.1 | 0.9 |
| Xpp55 | 483 | 421–551 | 5.0 ± 0.2 | 1.1 |
| M. incognita | ||||
| Cry5 | 250 | 149–356 | 1.9 ± 0.1 | 1 |
| App6 | 181 | 144–220 | 3.9 ± 0.1 | 1.4 |
| Cry21 | 377 | 262–543 | 1.2 ± 0.1 | 0.7 |
| Xpp55 | 246 | 134–348 | 3.1 ± 0.1 | 1.0 |
*LC50 ratios in relation to Cry5 LC50 value
The results indicated that M. incognita showed a slightly higher susceptibility (lower LC50 values) to the tested Cry proteins than M. javanica (Table 1). Within each nematode species, all four Bt toxins produced similar toxicity levels, but App6 was the most effective protein for both species (Table 1). This trend is particularly evident for M. javanica, as the FL95 range did not overlap with any of the rest of the proteins studied. Cry 21 was the lowest toxic protein in both species while the toxicities levels of Cry5 and Xpp55 were very similar and close to Cry21 ones, with overlapping FL95 ranges.
The high slope values obtained from the intoxication curves indicated a quick increase in toxicity within a narrow range of concentrations. This fact is especially notable for M. javanica.
V-CO3.3 and V-AB8.18 strains’ characterization and toxicity experiments
To test the possibility of using Bt strains to control root-knot nematodes in the fields, the wild Bt strains V-CO3.3 and V-AB8.18 were selected after an exhaustive screening of 850 strains. The screening was based in the gene content (assessed by PCR screening looking for genes that code for nematicidal proteins) and in the analyses of the protein contents in their parasporal crystals (Bel et al. 2023).
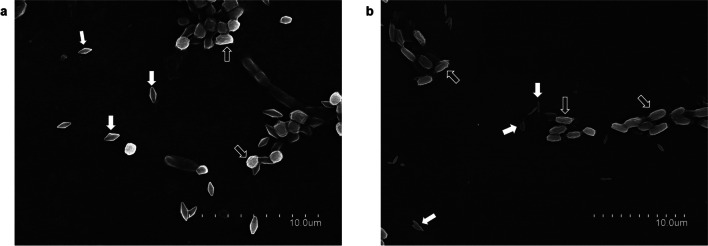
The microscopic analysis of V-CO3.3 showed that the strain produced big bipyramidal crystals (Fig. 1a) while V-AB8.18 strain produced elongated bar–shaped ones (Fig. 1b).
Fig. 1.
Scanning electron microscopy of V-CO3.3 and V-AB8.18 wild strains showing spores and parasporal crystal inclusions. a V-CO3.3 strain showing bipiramidal crystals. b V-AB8.18 strain, showing elongated bar–shaped crystals. In the images, white arrows point to crystals and empty arrows point to spores
The results of the PCR screening looking for the genes coding for the Cry1, Cry2, Cry3, Cry5, Cry12, Cry13, Cry14, Cry21, App6, and Xpp55 nematicidal proteins showed that V-CO3.3 amplified only with the specific primers designed to detect the cry5 gene while V-AB8.18 did use the specific primers for cry5 and app6 genes. The identity of the genes was confirmed by sequencing the PCR products obtained (Online Resource2). Subsequently, the gene sequences were obtained, deposited in GenBank, and their names assigned by BPPRC (Crickmore et al. 2021). As a result, the cry5 gene of V-CO3.3 codes for Cry5Ba4 (Genbank acc. No. OM424717) and the app6 gene of V-AB8.18 codes for App6Aa4 (GenBank Acc No. OM424718).
The proteomic analysis showed that the V-CO3.3 crystals were mainly composed of Cry5B protein and that V-AB8.18 crystals were made up mostly of App6A but also contained Cry5B based on the number of peptides detected and its protein coverage (Table 2). In addition, several peptides of Cry1 proteins were identified in both strains, covering a small part of the protein (probably belonging to the sequences conserved in all the Bt Cry proteins) and with a low number of unused peptides, indicating that most probably these proteins were not present in the crystal of these strains as it has been confirmed after whole-genome sequencing of the strains (Palma et al. 2024a, b).
Table 2.
Proteomic analyses of the proteins in the crystals of V-CO3.3 and V-AB8.18 strains
| Isolate | Na | Unusedb | Coverage % | Accession | Name | Peptides (95%) | Nc |
|---|---|---|---|---|---|---|---|
| V-CO3.3 | 1 | 417.93 | 90.359 | gi|862,637 | Cry5Ba | 1917 | 460 |
| 2 | 32.79 | 24.220 | gi|40,294 | Cry1Ca | 241 | 24 | |
| 3 | 19.89 | 21.259 | gi|142,765 | Cry1Aa | 170 | 19 | |
| 4 | 8.01 | 15.539 | gi|40,280 | Cry1Da | 136 | 16 | |
| 6 | 3.76 | 19.28 | gi|33,765,730 | Cry1Fa | 135 | 15 | |
| V-AB8.18 | 1 | 220.51 | 97.049 | gi|142,776 | App6Aa | 2107 | 370 |
| 2 | 87.14 | 61.53 | gi|862,637 | Cry5Ba | 187 | 57 | |
| 3 | 38 | 27.419 | gi|40,294 | Cry1Ca | 213 | 28 | |
| 4 | 18.87 | 23.549 | gi|142,765 | Cry1Aa | 327 | 37 | |
| 5 | 6.02 | 15.710 | gi|40,280 | Cry1Da | 137 | 17 |
aThe rank of the specified protein relative to all other detected proteins
bProtScore; a measure of the protein confidence for a detected protein, calculated from the peptide confidence for peptides from spectra that are not already completely used by higher scoring winning proteins
cThe number of peptides with the contribution value more than zero
The ability of V-CO3.3 and V-AB8.18 wild strains to produce type I β-exotoxin (an adenine nucleotide analog metabolite with nematicidal activity (Sánchez-Soto et al. 2015) that would prevent the possible use of the strains as bioinsecticides) was checked by LC–MS/MS, resulting negative in both of them (Online Resource3). Additionally, the non-detection of the thuE gene (the thuE gene is involved in the synthesis of type I β-exotoxin and the detection of this gene by PCR is used to predict the production of type I β-exotoxin (Sauka et al. 2014)) in none of the strains (Palma et al. 2024a, b) confirmed the LC–MS/MS results.
The toxicity experiments with the crystal proteins of V-CO3.3 and V-AB8.18 was performed in vitro (using the crystal proteins solubilized) and in planta (using spore + crystal suspensions) using M. javanica J2 juveniles. The in vitro results showed that the crystal proteins of both strains had significant nematicidal activities (Table 3). For V-CO3.3, the LC50 value found (422.5 µg/ml) was slightly lower than the one obtained for Cry5 against this pest (529.2 µg/ml, Table 1). On the other hand, the toxicity value of the V-AB8.18 crystal-solubilized proteins (102.6 µg/ml, Table 2) was about a half lower than the one found for App6 (233.5 µg/ml, Table 1), probably attributable to the simultaneous presence of Cry5 in the crystals. The high slopes observed point to a strong toxic effect of the Cry proteins, leading to nematode death within a narrow range of concentrations.
Table 3.
In vitro analysis of the susceptibility of M. javanica J2 to the solubilized crystal proteins of the V-CO 3.3 and V-AB8.18 strains
| LC50 (µg/ml) | FL95 (µg/ml) | Slope | |
|---|---|---|---|
| V-CO3.3 | 423 | 373–466 | 6.4 ± 0.6 |
| V-AB8.18 | 103 | 71–121 | 4.5 ± 0.3 |
In planta experiments
The in planta experiments were performed to test the potential of Bt strains producing nematicidal proteins in controlling Meloidogyne species in the field. Cucumber and tomato plants were watered with spore + crystal suspensions twice: the first treatment was applied on day 3 after seedling planting, and the second on day 7, immediately before plant infestation with 350 M. javanica J2. The number of egg masses in roots, the emerged J2, and the dry foliar weight were recorded 8 weeks later.
Each treatment was replicated three to seven times. In both cucumber and tomato, the control plants (not inoculated with nematodes) showed no egg masses. And there were no statistically significant differences between watering plants with water or with 20 mM HEPES buffer. Also, irrigation with the chemical oxamyl produced 100% of nematode mortality, evidenced by the absence of egg masses in the treated roots.
The first trials using cucumber and V-CO3.3 or V-AB8.18 (Fig. 2) did not produce statistically significant changes the recorded parameters regarding the respective controls. The lack of significant changes in egg masses and emerged J2 regarding controls pointed to the ineffectiveness of the Bt treatments against M. javanica in the tested conditions; although, a slight trend to decrease the egg masses was observed when plants were irrigated with either strain. In addition, no effects were detected regarding plant growth.
Fig. 2.
Impact of irrigation with V-CO3.3 or V-AB8.18 on cucumber plants infested with M. javanica J2. The parameters recorded were a egg masses per plant, b J2 per plant, and c dry foliar weight. V-CO3.3: plants treated with V-CO3.3 spore + crystal suspensions. V-AB8.18: plants treated with V-AB8.18 spore + crystal suspensions. Oxamyl: plants treated with oxamyl. Non-V-CO3.3: negative control plants (inoculated and non-treated with V-CO3.3). Non-V-AB8.18: negative control plants (inoculated and non-treated with V-AB8.18)
To check if the nematicidal effect of spore + crystal suspensions was plant-specific, the same experiments were replicated using tomato plants (Fig. 3). In tomato, both V-CO3.3 (Fig. 3a) and V-AB8.18 (Fig. 3b) decreased the number of J2 per plant. While the result was statistically significant for V-AB8.18, it was not for V-CO3.3 due to an outlier in the sample set, which, despite of differing greatly from the rest of the data, did not meet the criteria for exclusion based on Grubbs’ test. Remarkably, there was only a slight decrease in the egg masses in the Bt-treated plants regarding the controls, highlighting that the effect of the irrigation with V-CO3.3 or V-AB8.18 did not significantly impact the number of egg masses but had a negative effect on the number of J2 emerged from them. Regarding foliar weight, the results indicate that V-CO3.3 worked as a biofertilizer, significantly increasing the tomato foliar weight, whereas V-AB8.18 did not elicit a similar effect.
Fig. 3.
Impact of irrigation with V-CO3.3 or V-AB8.18 on tomato plants infested with M. javanica J2. The parameters recorded were a egg masses per plant, b J2 per plant, and c dry foliar weight. V-CO3.3: plants treated with V-CO3.3 spore + crystal suspensions. V-AB8.18: plants treated with V-AB8.18 spore + crystal suspensions. Non-V-CO3.3: negative control plants (inoculated and non-treated with V-CO3.3). Non-V-AB8.18: negative control plants (inoculated and non-treated with V-AB8.18)
Discussion
The susceptibility of some nematodes to Bt proteins is known. Regarding root-knot nematodes, while the susceptibility of some Meloidogyne species such as M. hapla or M. incognita to some Bt proteins has been studied in some extent in the past years, the susceptibility of M. javanica (a major agricultural pest in Mediterranean countries and many other temperate areas worldwide (CABI Head Office 2021)) to Bt proteins remains unknown.
Root-knot nematodes are plant-parasitic nematodes from the genus Meloidogyne, widely distributed in nature which when infecting plants impair their development or even trigger the death of young plants, causing serious economic losses to agricultural and horticultural crops. Meloidogyne spp. are very difficult to control owing to their short generation time, high fecundity, entophytic, and sedentary nature (Engelbrecht et al. 2018).
So far, the research performed to disclose the Meloidogyne susceptibility to Bt proteins has been focused on characterizing the susceptibility of M. hapla and M. incognita to Cry5, App6, and Xpp55 Bt proteins. The first studies showed LC50 values of about 7 to 24 μg/ml against M. hapla (Guo et al. 2008; Yu et al. 2008), similar to the ones obtained for the model nematode Caenorhabditis elegans intoxicated with Cry5, App6 (Geng et al. 2017b; Luo et al. 2013), or with Cry21 (Iatsenko et al. 2014). But these values were underestimated as the bioassays had been performed using proteins solubilized in carbonate buffer pH 9.5 that can result in toxicity for Meloidogyne species. Subsequent bioassays with M. hapla using the same proteins but solubilized in 20 mM HEPES pH 8 showed LC50 values higher than the previous ones (156, 71–302, and 261 µg/ml respectively Yu et al. 2015; Zhang et al. 2012)) and similar to that of M. incognita intoxicated with Cry5, App6, and Xpp55 (LC50 of 146 or approx. 350 µg/ml for Cry5, LC50 = 383 µg/ml for App6 and LC50 = 102 µg/ml for Xpp55 (Geng et al. 2017b; Peng et al. 2011)).
In this work, we have tested the susceptibility of Spanish populations of M. incognita to solubilized Cry5, App6, Xpp55, and to Cry21. The LC50 values obtained for Cry5, App6, and Xpp55 (Table 1) were similar to those previously reported (Geng et al. 2017a; Peng et al. 2011). Regarding the toxicity of Cry21, the value of LC50 = 377 μg/ml obtained was slightly higher than that of the other nematicidal Bt proteins although not significantly different from them.
The ability of Cry5, App6, Cry21, and Xpp55 to control M. javanica has been also studied, using M. javanica nematode populations obtained from naturally infested tomato plants collected in regions of southwestern Spain. The results (Table 1) showed that the four Bt proteins tested were toxic for this species, with LC50 values slightly higher (but not significantly different) to the ones obtained for M. incognita. For both M. incognita and M. javanica, the most toxic Bt protein was App6. Moreover, within each species, the LC50 values for Cry5, Cry21, and Xpp 55 were found to be similar. The results obtained in this work about the toxicity of the four Bt proteins against M incognita, M. javanica, and the published results regarding M. hapla (Yu et al. 2015; Zhang et al. 2012) point to their potential as nematicidal agents for the control Meloidogyne spp.
The mode of action of Cry proteins in nematodes remains poorly understood and has yet not been elucidated. Three-domain proteins such as Cry5 have been studied in more extent, and for them and for App6, it has been proposed that they exert their toxic effects after ingestion, being guts the primary target tissue (Griffitts et al. 2005; Peng et al. 2018; Shi et al. 2020; Wan et al. 2019). This hypothesis was supported by observations of intestinal injury in Cry-intoxicated J2 nematodes (Jouzani et al. 2008). On the other hand, PPN, including Meloidogyne species, are piercing/sucking pests that feed through a stylet structure which acts as a molecular sieve that determines their ability to ingest proteins based on size, shape, and electrostatic charge (Eves-van den Akker et al. 2014). Some authors have characterized the molecular exclusion limit of the feeding tube of some Meloidogyne species, and have confirmed that, e.g., M. hapla J2 could ingest 140 kDa solubilized proteins directly through the stylet (Zhang et al. 2012). On the other hand, the expression of Cry5B or App6 in tomato or tobacco conferred resistance to M. incognita (Li et al. 2008, 2007; Wang et al. 2024), or the expression of Cry14 in soybean protected the plant against the cys nematode Heterodera glycines (Kahn et al. 2021), highlighting that the studied proteins could be ingested by the PPNs. In this work, in vitro experiments have demonstrated that Cry5, Cry21, App6, and Xpp55 proteins once solublized are toxic to M. incognita and M. javanica, indicating that they can be ingested when are in soluble state.
Several studies have reported the nematicidal action of Bt spore + crystal mixtures. Assays using spores + crystals against M. incognita in vitro (Ramalakshmi et al. 2020) or in greenhouses with tomato plants have produced impairing egg hatching (Jouzani et al. 2008; Ramalakshmi et al. 2020) or a decrease of the number of egg masses (Mohammed et al. 2008; Ramalakshmi et al. 2020) and damage the digestive tract of hatched J2 (Jouzani et al. 2008). Also, in vitro assays with spores + crystals have shown toxicity against M. javanica (Ravari and Moghaddam 2015), as well as decreases in root galling and egg masses in Bt-treated tomatoes (Khyami-Horani and Al-Banna 2006; Osman et al. 1988) or in soybean plants (Chinheya et al. 2017); although, the nature of the toxic action has not been disclosed. Reduction of the galling index, egg masses, and a decrease in the soil nematode population has been observed in tomatoes infested with M. hapla and treated repeatedly with a Bt strain–producing App6 protein (Yu et al. 2015).
In this study, two Bt strains were selected from the collection of Bt wild strains held by UVEG to test if they were able to control Meloidogyne species in the fields. The selection was based on nematicidal gene screening supported by proteomic analyses. The strains selected were V-CO3.3 (which produced Cry5Ba) and V-AB8.18 (that produced App6Aa and Cry5Ba). The toxicities of the proteins solubilized from the crystals of these two strains were evaluated in vitro, showing that the proteins produced were toxic for M. javanica to the same extent or more than the standard purified proteins.
In planta experiments in greenhouses were performed using spore + crystal suspensions to irrigate cucumber or tomato plants infested with M. javanica J2 nematodes derived from M. javanica populations isolated from infested tomato fields. The methodology used in the experiments was similar to the one published in similar works which caused significant nematicidal activity against Meloidogyne (Berlitz et al. 2013; Chinheya et al. 2017; Khyami-Horani and Al-Banna 2006; Yu et al. 2015), regarding both the Bt concentration and the irrigation methodology (with some exceptions of constant drenching (Yu et al. 2015)). The results obtained in the present work with cucumbers showed that none of the treatments, with any of the strains, was effective in reducing significantly nor the egg masses nor the J2 recovered per plant. But with tomato plants, a trend to decrease the number of J2 per plant was obvious and statistically significant in the case of V-AB8.18, highlighting that the Bt spore + crystal suspensions impacted negatively egg hatching, in agreement with what has been described by other authors (Jouzani et al. 2008; Ramalakshmi et al. 2020).
The toxicity of the solubilized crystal proteins of V-CO3.3 and V-AB8.18 to M. javanica J2 was very high compared with the toxicity found in the in planta experiments performed with spores + crystal suspensions of the same strains. The discrepancy between these results highlights challenges in delivering Cry proteins to the nematodes within the root system or when they are free in the soil. It is likely that the structure of the crystal proteins could prevent their ingestion through the nematode stylet. The success in controlling Meloidogyne with Bt spore + crystal suspensions observed by other authors in bioassays (Jouzani et al. 2008; Mokbel and Alharbi 2014; Ravari and Moghaddam 2015) or in the in planta experiments mentioned above may be attributed either to partial solubilization of the crystal proteins in the bioassay conditions, or to the presence of secondary metabolites or strain-specific nematicidal compounds different from crystal proteins (Engelbrecht et al. 2018; Horak et al. 2019; Mohammed et al. 2008) that are produced by the strains and that could remain in the final preparations. The first hypothesis was supported by our own experiments with HEPES buffer, which can partially solubilize the crystals after incubation of short periods of time at 35°C (after 30 min, 95 to 125 µg/ml of Cry5 protein were obtained in the supernatants of the spore + crystal preparations of the strain BMB0215), and therefore can provide mortalities higher than negative controls. Overall, the results seem to indicate that Cry proteins delivered as crystals can hardly arrive to the intestine of the target root-knot nematode because of the feeding behavior of these organisms and the associated difficulty that offers their feeding tube. Most probably, based on nematode morphology and physiology, neither the bacteria nor the protein in the crystal form are ingested by these organisms.
Bt irrigations with V-CO3.3 produced a statistically significant increase in leaf weight in tomato while V-AB8.18 did not, and neither V-CO3.3 nor V-AB8.18 increased the leaf weight of cucumber. These in planta observations suggest that some Bt strains can act as biofertilizers and that this capacity is related to their specific metabolic background and their ability to interact with the plant. Indeed, some Bt strains may be plant growth–promoting bacteria, providing them fixed nitrogen, iron, soluble phosphate, and other nutrients (Raddadi et al. 2007). This effect has also been described for other nematicidal Bt strains (Yu et al. 2015) but seems specific of the strain and crop plant interaction.
Summarizing, Bt nematicidal proteins Cry5, App6, Cry21, and Xpp55, when solubilized, are excellent nematicidal agents to control Meloidogyne species, but their use in the form of spore + crystal suspensions in conventional irrigations poses significant challenges. Alternative approaches, such as incorporating these proteins into transgenic plants or using nanoparticles carrying solubilized Bt proteins (Jalali et al. 2023) (which could even allow the simultaneous delivery of two proteins with different modes of action), are necessary. Indeed, as mentioned before, Cry5, App6, and Cry14 transgenic plants have been successfully produced and tested to control PPNs. Therefore, it is crucial to continue investigating the delivery methods as well as the mode of action of these nematicidal proteins to develop effective bio-control strategies against these devastating pests.
Supplementary Information
Below is the link to the electronic supplementary material.
Author contribution
BE, YB, and MG conceived, designed, and supervised the research. YB, MG, MB-S, and MA-A conducted the experiments. YB, BE, MG, and MB-S analyzed the data. YB wrote the manuscript. YB, BE, MG, and MB-S reviewed and edited the manuscript. All authors read and approved the manuscript.
Funding
This work was supported by the European Union’s Horizon 2020 Research and Innovation program, under Grant Agreement no. 773554 (EcoStack Project), and a grant from Generalitat Valenciana, Spain (PROMETEO/2020/010 and CIPROM/2023/56). Miguel Andrés-Antón was supported by the Spanish Ministerio de Ciencia, Innovación y Universidades (Ref PEJ2018-004251-A).
Data availability
Available on reasonable request.
Declarations
Ethics approval
This work does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
All authors consented to the publication of this work.
Conflict of interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Abad P, Favery B, Rosso MN, Castagnone-Sereno P (2003) Root-knot nematode parasitism and host response: molecular basis of a sophisticated interaction. Mol Plant Pathol 4(4):217–224. 10.1046/j.1364-3703.2003.00170.x [DOI] [PubMed] [Google Scholar]
- Andres MF, Verdejo-Lucas S (2011) Enfermedades causadas por nematdos fitoparásitos en España, PHYTOMA-España and Spanish Society of Phytopathology eds, Spain
- Atkinson HJ, Lilley CJ, Urwin PE (2012) Strategies for transgenic nematode control in developed and developing world crops. Curr Opin Biotechnol 23(2):251–256. 10.1016/j.copbio.2011.09.004 [DOI] [PubMed] [Google Scholar]
- Barranco I, Sanchez-López CM, Bucci D, Alvarez-Barrientos A, Rodriguez-Martinez H, Marcilla A, Roca J (2023) The Proteome of large or small extracellular vesicles in pig seminal plasma differs, defining sources and biological functions. Mol Cell Proteomics 22(4):100514. 10.1016/j.mcpro.2023.100514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bel Y, Galeano M, Baños-Salmeron M, Escriche B (2022) The use of Bacillus thuringiensis to control plant-parasitic nematodes. J Plant Sci Phytopathol 6:2. 10.29328/journal.jpsp.1001076 [Google Scholar]
- Bel Y, Andrés-Antón M, Escriche B (2023) Abundance, distribution, and expression of nematicidal crystal protein genes in Bacillus thuringiensis strains from diverse habitats. Int Microbiol 26(2):295–308. 10.1007/s10123-022-00307-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berlitz DL, Saul DdA, Machado V, Santin RdC, Guimarães AM, Matsumura ATS, Ribeiro BM, Fiuza LM (2013) Bacillus thuringiensis: molecular characterization, ultrastructural and nematoxicity to Meloidogyne sp. J Biopest 2(2):120–128 [Google Scholar]
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254 [DOI] [PubMed] [Google Scholar]
- Bridge J, Starr J (2007) Plant nematodes of agricultural importance: A colour handbook, 1st edn. CCR Press, Boca Raton, FL, USA [Google Scholar]
- CABI Head Office W, UK (2021) Meloidogyne javanica (sugarcane eelworm) 10.1079/cabicompendium.33246. CABI Compendium
- Chinheya CC, Yobo KS, Laing MD (2017) Biological control of the rootknot nematode, Meloidogyne javanica (Chitwood) using Bacillus isolates, on soybean. Biol Control 109:37–41. 10.1016/j.biocontrol.2017.03.009 [Google Scholar]
- Chitwood DJ (2003) Research on plant-parasitic nematode biology conducted by the United States Department of Agriculture-Agricultural Research Service. Pest Manag Sci 59(6–7):748–753. 10.1002/ps.684 [DOI] [PubMed] [Google Scholar]
- Crickmore N, Berry C, Panneerselvam S, Mishra R, Connor TR, Bonning BC (2021) A structure-based nomenclature for Bacillus thuringiensis and other bacteria-derived pesticidal proteins. J Invert Pathol 186:107438. 10.1016/j.jip.2020.107438 [DOI] [PubMed] [Google Scholar]
- Dutta TK, Phani V (2023) Transgenics, application in plant nematode management. In: Khan MR (ed) Novel biological and biotechnological applications in plant nematode management. Springer Nature Singapore, Singapore, pp 203–226 [Google Scholar]
- Eisenback JD (2022) Meloidogyne incognita (root-knot nematode). Publisher. 10.1079/cabicompendium.33245Accessed26March2024 [Google Scholar]
- Engelbrecht G, Horak I, Jansen van Rensburg PJ, Claassens S (2018) Bacillus-based bionematicides: development, modes of action and commercialisation. Biocontrol Sci Technol 28(7):629–653. 10.1080/09583157.2018.1469000 [Google Scholar]
- Estela A, Escriche B, Ferré J (2004) Interaction of Bacillus thuringiensis toxins with larval midgut binding sites of Helicoverpa armigera (Lepidoptera: Noctuidae). Appl Environ Microbiol 70(3):1378–1384. 10.1128/AEM.70.3.1378-1384.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eves-van den Akker S, Lilley CJ, Ault JR, Ashcroft AE, Jones JT, Urwin PE (2014) The feeding tube of cyst nematodes: characterisation of protein exclusion. PLoS ONE 9(1):e87289. 10.1371/journal.pone.0087289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geng C, Liu Y, Li M, Tang Z, Muhammad S, Zheng J, Wan D, Peng D, Ruan L, Sun M (2017a) Dissimilar crystal proteins Cry5Ca1 and Cry5Da1 synergistically act against Meloidogyne incognita and delay Cry5Ba-based nematode resistance. Appl Environ Microbiol 83(18). 10.1128/aem.03505-16 [DOI] [PMC free article] [PubMed]
- Geng C, Liu Y, Li M, Tang Z, Muhammad S, Zheng J, Wan D, Peng D, Ruan L, Sun M (2017b) Dissimilar crystal proteins Cry5Ca1 and Cry5Da1 synergistically act against Meloidogyne incognita and delay Cry5Ba-based nematode resistance. Appl Environ Microbiol 83(18):e03505-e3516. 10.1128/AEM.03505-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffitts JS, Haslam SM, Yang T, Garczynski SF, Mulloy B, Morris H, Cremer PS, Dell A, Adang MJ, Aroian RV (2005) Glycolipids as receptors for Bacillus thuringiensis crystal toxin. Science 307(5711):922–925. 10.1126/science.1104444 [DOI] [PubMed] [Google Scholar]
- Guo S, Liu M, Peng D, Ji S, Wang P, Yu Z, Sun M (2008) New strategy for isolating novel nematicidal crystal protein genes from Bacillus thuringiensis strain YBT-1518. Appl Environ Microbiol 74. 10.1128/aem.01346-08 [DOI] [PMC free article] [PubMed]
- Hernández CS, Ferré J, Larget-Thiéry I (2001) Update on the detection of beta-exotoxin in Bacillus thuringiensis strains by HPLC analysis. J Appl Microbiol 90(4):643–647. 10.1046/j.1365-2672.2001.01288.x [DOI] [PubMed] [Google Scholar]
- Horak I, Engelbrecht G, van Rensburg PJJ, Claassens S (2019) Microbial metabolomics: essential definitions and the importance of cultivation conditions for utilizing Bacillus species as bionematicides. J Appl Microbiol 127(2):326–343. 10.1111/jam.14218 [DOI] [PubMed] [Google Scholar]
- Iatsenko I, Boichenko I, Sommer RJ (2014) Bacillus thuringiensis DB27 produces two novel protoxins, Cry21Fa1 and Cry21Ha1, which act synergistically against nematodes. Appl Environ Microbiol 80(10):3266–3275. 10.1128/AEM.00464-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jalali E, Bel Y, Maghsoudi S, Noroozian E, Escriche B (2023) Enhancing insecticidal efficacy of Bacillus thuringiensis Cry1Ab through pH-sensitive encapsulation. Appl Microbiol Biotechnol 107(20):6407–6419. 10.1007/s00253-023-12723-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones JT, Haegeman A, Danchin EG, Gaur HS, Helder J, Jones MG, Kikuchi T, Manzanilla-López R, Palomares-Rius JE, Wesemael WM, Perry RN (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14(9):946–961. 10.1111/mpp.12057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jouzani SG, Seifinejad A, Saeedizadeh A, Nazarian A, Yousefloo M, Soheilivand S, Mousivand M, Jahangiri R, Yazdani M, Amiri RM, Akbari S (2008) Molecular detection of nematicidal crystalliferous Bacillus thuringiensis strains of Iran and evaluation of their toxicity on free-living and plant-parasitic nematodes. Can J Microbiol 54(10):812–822. 10.1139/W08-074 [DOI] [PubMed] [Google Scholar]
- Jouzani GS, Valijanian E, Sharafi R (2017) Bacillus thuringiensis: a successful insecticide with new environmental features and tidings. Appl Microbiol Biotechnol 101(7):2691–2711. 10.1007/s00253-017-8175-y [DOI] [PubMed] [Google Scholar]
- Jurat-Fuentes JL, Crickmore N (2017) Specificity determinants for Cry insecticidal proteins: insights from their mode of action. J Invertebr Pathol 142:5–10. 10.1016/j.jip.2016.07.018 [DOI] [PubMed] [Google Scholar]
- Kahn TW, Duck NB, McCarville MT, Schouten LC, Schweri K, Zaitseva J, Daum J (2021) A Bacillus thuringiensis Cry protein controls soybean cyst nematode in transgenic soybean plants. Nat Commun 12(1):3380. 10.1038/s41467-021-23743-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan MR (2023) Novel biological and biotechnological applications in plant nematode management. Springer, Singapore [Google Scholar]
- Khan MR, Haque Z, Sharma RK (2023) Novel biotechnological interventions in plant nematode management technologies. In: Khan MR (ed) Novel biological and biotechnological applications in plant nematode management. Springer Nature Singapore, Singapore, pp 167–186 [Google Scholar]
- Khorramnejad A, Talaei-Hassanloui R, Hosseininaveh V, Bel Y, Escriche B (2018) Characterization of new Bacillus thuringiensis strains from Iran, based on cytocidal and insecticidal activity, proteomic analysis and gene content. Biocontrol. 10.1007/s10526-018-9901-9 [Google Scholar]
- Khorramnejad A, Gomis-Cebolla J, Talaei-Hassanlouei R, Bel Y, Escriche B (2020) Genomics and proteomics analyses revealedn candidate pesticidal proteins in a Lepidopteran-toxic Bacillus thuringiensis strain. Toxins 12(11):673. 10.3390/toxins12110673 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khyami-Horani H, Al-Banna L (2006) Efficacy of Bacillus thuringiensis jordanica against Meloidogyne javanica infecting tomato. Phytopathol Mediterr 45(2):153–157 [Google Scholar]
- Koch MS, Ward JM, Levine SL, Baum JA, Vicini JL, Hammond BG (2015) The food and environmental safety of Bt crops. Front Plant Sci 6:283. 10.3389/fpls.2015.00283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X-Q, Tan A, Voegtline M, Bekele S, Chen C-S, Aroian RV (2008) Expression of Cry5B protein from Bacillus thuringiensis in plant roots confers resistance to root-knot nematode. Biol Control 47(1):97–102. 10.1016/j.biocontrol.2008.06.007 [Google Scholar]
- Li XQ, Wei JZ, Tan A, Aroian RV (2007) Resistance to root-knot nematode in tomato roots expressing a nematicidal Bacillus thuringiensis crystal protein. Plant Biotechnol J 5. 10.1111/j.1467-7652.2007.00257.x [DOI] [PubMed]
- Liang Z, Ali Q, Wang Y, Mu G, Kan X, Ren Y, Manghwar H, Gu Q, Wu H, Gao X (2022) Toxicity of Bacillus thuringiensis strains derived from the novel crystal protein Cry31Aa with high nematicidal activity against rice parasitic nematode Aphelenchoides besseyi. Int J Mol Sci 23(15). 10.3390/ijms23158189 [DOI] [PMC free article] [PubMed]
- Luo H, Xiong J, Zhou Q, Xia L, Yu Z (2013) The effects of Bacillus thuringiensis Cry6A on the survival, growth, reproduction, locomotion, and behavioral response of Caenorhabditis elegans. Appl Microbiol Biotechnol 97(23):10135–10142. 10.1007/s00253-013-5249-3 [DOI] [PubMed] [Google Scholar]
- Mohammed SHES, Anwer M, Enan Mohamed R, Ibrahim Nasser E, Ghareeb A, Moustafa Salah A (2008) Biocontrol efficiency of Bacillus thuringiensis toxins against root-knot nematode, Meloidogyne incognita. J Cell Mol Biol 7(1):57–66 [Google Scholar]
- Mokbel AA, Alharbi AA (2014) Suppressive effect of some microbial agents on root-knot nematode, Meloidogyne javanica infected eggplant. Aust J Crop Sci 8:1428–1434 [Google Scholar]
- Olson S (2015) An analysis of the biopesticide market now and where it is going. Outlooks Pest Manag 26(5):203–206. 10.1564/v26_oct_04 [Google Scholar]
- Osman GY, Salem FM, Ghattas A (1988) Bio-efficacy of two bacterial insecticide strains of Bacillus thuringiensis as a biological control agent in comparison with a nematicide, nemacur, on certain parasitic nematoda. Anzeiger Für Schädlingskunde, Pflanzenschutz, Umweltschutz 61(2):35–37. 10.1007/BF01905720 [Google Scholar]
- Palma L, Bel Y, Escriche B (2024a) Genomic insights into Bacillus thuringiensis V-CO3.3: unveiling its genetic potential against nematodes. Data 9:97. 10.3390/data9080097
- Palma L, Bel Y, Escriche B (2024b) Draft genome sequence of Bacillus thuringiensis strain V-AB8.18, a novel isolate with potential nematicidal activity. Microbiol Resour Announc e0022724. 10.1128/mra.00227-24 [DOI] [PMC free article] [PubMed]
- Palma L, Muñoz D, Berry C, Murillo J, Caballero P (2014) Bacillus thuringiensis toxins: an overview of their biocidal activity. Toxins 6(12):3296–3325. 10.3390/toxins6123296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng D, Chai L, Wang F, Zhang F, Ruan L, Sun M (2011) Synergistic activity between Bacillus thuringiensis Cry6Aa and Cry55Aa toxins against Meloidogyne incognita. Microb Biotechnol 4(6):794–798. 10.1111/j.1751-7915.2011.00295.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peng D, Wan D, Cheng C, Ye X, Sun M (2018) Nematode-specific cadherin CDH-8 acts as a receptor for Cry5B toxin in Caenorhabditis elegans. Appl Microbiol Biotechnol 102(8):3663–3673. 10.1007/s00253-018-8868-x [DOI] [PubMed] [Google Scholar]
- Perez-Riverol Y, Bai J, Bandla C, García-Seisdedos D, Hewapathirana S, Kamatchinathan S, Kundu DJ, Prakash A, Frericks-Zipper A, Eisenacher M, Walzer M, Wang S, Brazma A, Vizcaíno JA (2022) The PRIDE database resources in 2022: a hub for mass spectrometry-based proteomics evidences. Nucleic Acids Res 50(D1):D543-d552. 10.1093/nar/gkab1038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raddadi N, Cherif A, Ouzari H, Marzorati M, Brusetti L, Boudabous A, Daffonchio D (2007) Bacillus thuringiensis beyond insect biocontrol: plant growth promotion and biosafety of polyvalent strains. Ann Microbiol 57(4):481–494. 10.1007/BF03175344 [Google Scholar]
- Ramalakshmi A, Sharmila R, Iniyakumar M, Gomathi V (2020) Nematicidal activity of native Bacillus thuringiensis against the root knot nematode, Meloidogyne incognita (Kofoid and White). Egypt J Biol Pest Control 30(1):90. 10.1186/s41938-020-00293-2 [Google Scholar]
- Ravari B, Moghaddam EM (2015) Efficacy of Bacillus thuringiensis Cry14 toxin against root knot nematode. Meloidogyne Javanica Plant Prot Sc 51(1):46–51 [Google Scholar]
- Raymond B, Federici BA (2017) In defence of Bacillus thuringiensis, the safest and most successful microbial insecticide available to humanity-a response to EFSA. FEMS Microbiol Ecol 93(7). 10.1093/femsec/fix084 [DOI] [PMC free article] [PubMed]
- Sanahuja G, Banakar R, Twyman RM, Capell T, Christou P (2011) Bacillus thuringiensis: a century of research, development and commercial applications. Plant Biotechnol J 9(3):283–300. 10.1111/j.1467-7652.2011.00595.x [DOI] [PubMed] [Google Scholar]
- Sánchez-Soto AI, Saavedra-González GI, Ibarra JE, Salcedo-Hernández R, Barboza-Corona JE, Del Rincón-Castro MC (2015) Detection of β-exotoxin synthesis in Bacillus thuringiensis using an easy bioassay with the nematode Caenorhabditis elegans. Lett Appl Microbiol 61(6):562–567. 10.1111/lam.12493 [DOI] [PubMed] [Google Scholar]
- Sauka DH, Pérez MP, López NN, Onco MI, Berretta MF, Benintende GB (2014) PCR-based prediction of type I β-exotoxin production in Bacillus thuringiensis strains. J Invertebr Pathol 122:28–31. 10.1016/j.jip.2014.08.001 [DOI] [PubMed] [Google Scholar]
- Sayyed AH, Gatsi R, Ibiza-Palacios MS, Escriche B, Wright DJ, Crickmore N (2005) Common, but complex, mode of resistance of Plutella xylostella to Bacillus thuringiensis toxins Cry1Ab and Cry1Ac. Appl aEnviron Microbiol 71(11):6863–6869. 10.1128/AEM.71.11.6863-6869.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi J, Peng D, Zhang F, Ruan L, Sun M (2020) The Caenorhabditis elegans CUB-like-domain containing protein RBT-1 functions as a receptor for Bacillus thuringiensis Cry6Aa toxin. PLOS Pathog 16(5):e1008501. 10.1371/journal.ppat.1008501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shilov IV, Seymour SL, Patel AA, Loboda A, Tang WH, Keating SP, Hunter CL, Nuwaysir LM, Schaeffer DA (2007) The Paragon Algorithm, a next generation search engine that uses sequence temperature values and feature probabilities to identify peptides from tandem mass spectra. Mol Cell Proteomics 6(9):1638–1655. 10.1074/mcp.T600050-MCP200 [DOI] [PubMed] [Google Scholar]
- Siddique S, Akker SE-vd (2021) Nematode management through genome editing. CABI 408–413. 10.1079/9781789247541.0057
- Singh S, Singh B, Singh AP (2015) Nematodes: a threat to sustainability of agriculture. Procedia Environ Sci 29:215–216. 10.1016/j.proenv.2015.07.270 [Google Scholar]
- Stewart GSAB, Johnstone K, Hagelberg E, Ellar DJ (1981) Commitment of bacterial spores to germinate. Biochem J 198:101–106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan L, Lin J, Du H, Zhang Y, Bravo A, Soberón M, Sun M, Peng D (2019) Bacillus thuringiensis targets the host intestinal epithelial junctions for successful infection of Caenorhabditis elegans. Environ Microbiol 21(3):1086–1098. 10.1111/1462-2920.14528 [DOI] [PubMed] [Google Scholar]
- Wang Y, Wang M, Zhang Y, Chen F, Sun M, Li S, Zhang J, Zhang F (2024) Resistance to both aphids and nematodes in tobacco plants expressing a Bacillus thuringiensis crystal protein. Pest Manag Sci. 10.1002/ps.8013 [DOI] [PubMed] [Google Scholar]
- Wei JZ, Hale K, Carta L, Platzer E, Wong C, Fang SC, Aroian RV (2003) Bacillus thuringiensis crystal proteins that target nematodes. Proc Natl Acad Sci U S A 100(5):2760–2765 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu Z, Bai P, Ye W, Zhang F, Ruan L, Yu Z, Sun M (2008) A novel negative regulatory factor for nematicidal Cry protein gene expression in Bacillus thuringiensis. J Microbio Biotechnol 18(6):1033–1039 [PubMed] [Google Scholar]
- Yu Z, Xiong J, Zhou Q, Luo H, Hu S, Xia L, Sun M, Li L, Yu Z (2015) The diverse nematicidal properties and biocontrol efficacy of Bacillus thuringiensis Cry6A against the root-knot nematode Meloidogyne hapla. J Invertebr Pathol 125:73–80. 10.1016/j.jip.2014.12.011 [DOI] [PubMed] [Google Scholar]
- Zhang F, Peng D, Ye X, Yu Z, Hu Z, Ruan L, Sun M (2012) In vitro uptake of 140 kDa Bacillus thuringiensis nematicidal crystal proteins by the second stage juvenile of Meloidogyne hapla. PLoS ONE 7(6):e38534. 10.1371/journal.pone.0038534 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Available on reasonable request.