Abstract
Gastric dilatation and volvulus (GDV) is a life‐threatening emergency that requires urgent intervention. Radiographic features associated with 360‐GDV in dogs have not been investigated. The aim of this retrospective observational study is to describe radiographic features and clinical variables in dogs affected with 360‐GDV and to report agreement rates between different radiologists. We also report the sensitivity and specificity of radiographs to diagnose 360‐GDV in dogs. Confirmed 360‐GDV cases were retrieved, and the radiographic findings were compared with dogs presenting with gastric dilatation (GD) and 180‐GDV. Images were reviewed and graded by three blinded board‐certified radiologists. A total of 16 dogs with confirmed 360‐GDV were identified. The median age was 10 years old (2–13 years). The sensitivity for detection of 360‐GDV ranged between 43.7% and 50%, and the specificity between 84.6% and 92.1%. Interobserver agreement on final diagnosis was substantial (Kappa = 0.623; 0.487–0.760, 95% CI). The highest agreement rate was in cases of 180‐GDV (87%), followed by the GD cases (72%) and 360‐GDV (46%). Severe esophageal distension and absence of small intestinal dilation were the only radiographic features specifically associated with 360‐GDV. A similar pyloric position was found between GD and 360‐GDV. Additional radiographic variables that could help differentiate GD from 360‐GDV include the degree of gastric distension and the peritoneal serosal contrast. Two cases with 360‐GDV were misdiagnosed by the three radiologists as GD. In conclusion, radiographically, 360‐GDV cases can reassemble GD and vice versa. Radiologists and clinicians should be aware of the low sensitivity of radiographs for the detection of 360‐GDV.
Keywords: 360, dogs, gastric volvulus, radiographs
1. INTRODUCTION
Canine gastric dilatation (GD) and gastric dilatation and volvulus (GDV) are characterized by moderate to severe gas distension of the stomach. Gastric dilatation and volvulus is a life‐threatening emergency that requires urgent intervention. 1 , 2 It is a dynamic disease process that is characterized by acute torsion of the stomach along its mesenteric axis. 2 , 3 Predominant clinical features of this condition include nonproductive retching, abdominal distension, and tachycardia. 4 , 5
Diagnosis is usually based on signalment and clinical presentation with the presence of moderate to severe abdominal distension. In this case, abdominal radiography can confirm the presence of marked gastric distension. 6 , 7 Radiographs are also sensitive to detect pyloric malpositioning and can differentiate between gastric dilatation and gastric dilatation with pyloric malposition that is usually present in cases of GDV. 8 , 9 In cases of 360 degrees GDV (360‐GDV), the pyloric position remains normal, making it difficult to differentiate it from GD. 8 , 9
Following an extensive literature review, the authors concluded that studies describing the radiographic features of 360‐GDV are lacking. Given the historical assumption that 360‐GDV have very similar radiographic features when compared to GD and the fact that 360‐GDVs are life‐threatening conditions that require immediate surgical intervention, differentiating between these two conditions is of paramount importance. Additionally, the level of agreement on the pyloric position between different specialists has not been documented. The aim of this study is to describe the specific radiographic and clinical findings associated with 360‐GDV and report the sensitivity and specificity of radiographs to diagnose 360‐GDV. Additionally, we prospectively investigate the level of agreement between radiologists on the radiographic variables associated with GD, 180‐GDV, and 360‐GDV, focusing on pyloric position. We hypothesized that we would identify characteristic radiographic changes associated with 360‐GDV and that the sensitivity and specificity of radiographs would be low. Second, we hypothesized a low level of agreement between board‐certified radiologists on the prediction of pylorus position.
2. METHODS
A computerized medical record (January 1, 2000 to June 31, 2020) search was used to identify dogs admitted to four veterinary teaching hospitals in North America (Ontario Veterinary College, Guelph, ON, Canada; University of California, Davis, CA, USA; North Carolina State University, Raleigh, NC, USA, Texas A&M University, College Station, TX, USA). Additionally, a search of radiology reports from a teleradiology company (Antech Imaging Services) was performed using the keyword 360‐GDV. Dogs with an intraoperative diagnosis of 360‐GDV were included. Nonoperated dogs with <24 h postmortem necropsy exam confirming the presence of 360GDV were also included. Dogs were excluded if they were initially presented to the hospital for another ailment or had another primary lesion contributing to the abdominal crisis. Only radiographic studies containing at least two projections, including a right lateral view, were included in the study. Dogs with prior gastropexy, splenectomies, and/or additional findings during surgery (i.e., liver or splenic mass) were not included. Additionally, cases with GD and surgically confirmed 180 degrees GDV (180‐GDV) were also retrieved. The inclusion criteria for the GD cases were moderate to marked gas distension of the stomach with a normal pyloric position on radiographs that were managed conservatively. Only dogs weighing >25 kg were included in the GD and 180‐GDV groups.
Sex, age, breed, and body weight were retrieved from the medical records. The reported clinical variables include onset type (acute vs. nonacute) and duration of clinical signs (hours). The heart rate on admission (beats per minute) was also documented when available. Laboratory variables, including peripheral venous lactate on admission (mmol/L), packed cell volume (%), total solids (gr/dL), and pH in venous blood were recorded when available.
2.1. Ethics statement
This research was conducted in accordance with ethical standards and guidelines. Any potential conflicts of interest have been disclosed, and the study adhered to principles of integrity and transparency in reporting results.
2.2. Radiographic evaluation
Three blinded board‐certified radiologists evaluated all cases and recorded and graded predetermined radiographic features. These investigators were not involved in case selection, review of medical records, or medical record abstraction. Before the radiographic evaluation, one investigator (P.E.M.) anonymized all images by removing case identifiers. Sequential case numbers were assigned to each study in a randomized fashion using a free online random number generator (random.org). An Excel table was distributed indicating the grades for each of the radiographic variables. Radiologists were blinded to signalment and any clinical information of each case. Three final diagnoses were allowed in each case (GD, 180‐GDV, and 360‐GDV). No diagnostic guidelines were distributed among radiologists prior to the radiographic evaluation of the cases, and they performed the interpretations at their own pace. A total of 8 weeks were allowed to complete all the evaluations. The following radiographic variables were graded by each radiologist: esophageal gas distension (absent, mild, or marked); gastric gas (absent, mild, or marked); gastric pneumatosis (absent or present); compartmentalization (absent or present); splenic position (presence or absence of splenic malpositioning); spleen size (normal, mildly enlarged or markedly enlarged); splenic position in the ventrodorsal (or dorsoventral) projections (presence or absence of the dorsal extremity of the spleen caudal to the gastric fundus); duodenal distention (absent or present); jejunal distension (absent or present); peritoneal serosal contrast (normal or reduced); pneumoperitoneum (absent or present); size of the caudal vena cava in the lateral projection (normal or reduced size). Three variables pertaining to analyzing the position of the pylorus were evaluated: pylorus position (presence or absence of pylorus malpositioning); pylorus in the lateral projection (cranioventral, craniodorsal, caudoventral or caudodorsal); pylorus in the ventrodorsal (or dorsoventral) projections (right cranial, right caudal, left cranial or left caudal). For the latter two variables describing pyloric position, four predetermined quadrants were determined using anatomical landmarks. In the right lateral projection, the cranial to the caudal line was drawn from the ventral margin of the caudal vena cava parallel along the axis of the caudal thoracic spine. Second, the dorsal to ventral line was drawn from the T12‐13 intervertebral disc space ventrally perpendicular to the caudal thoracic spine. In the ventrodorsal or dorsoventral view (ventrodorsal 35/81, dorsoventral 14/81, non‐specified 9/81, unavailable 23/81), the spine was used as the longitudinal imaginary line separating left from right. The transverse line dividing the cranial abdomen in cranial and caudal was traced at the level of the T12‐13 intervertebral disc space. Information pertaining to the technique and equipment used for image acquisition was not available.
2.3. Sample size calculations
Sixteen confirmed 360‐GDV cases from one institution (University of California, Davis, CA, USA) were used to determine the sample size. Nine of these cases did not have radiographs available, and information was obtained from the report written by a board‐certified radiologist. The frequency of the following radiographic changes was recorded: esophageal gas distension, gastric pneumatosis, craniodorsal pylorus in the right lateral projection, and gastric compartmentalization. The frequencies of the four preselected radiographic features were compared with a similar number of GD and 180‐GDV cases from the same institution. Power analysis using the proportions of the four radiographic variables was calculated. Power was set arbitrarily at 0.8. A sample size calculation for a chi‐squared test to compare binomial proportions was performed.
2.4. Statistical analysis
The frequency of the reported variables radiographic variables for each group is reported. The sensitivity and specificity of radiographs to diagnose 360‐GDV were also calculated for each radiologist. The percentage of agreement for paired reviewers was calculated for all the radiographic variables. Interrater agreement between the radiologists was calculated using the Cohen Kappa coefficient. A weighted Cohen Kappa coefficient was calculated for variables with more than two grades (esophageal gas, gastric gas, and spleen size). Kappa coefficients were interpreted as follows: values ≤ 0 as indicating no agreement, values 0.01–0.2 as none to slight, 0.21–0.4 as fair, 0.41–0.6 as moderate, 0.61–0.8 as substantial, and 0.81–1 as almost perfect agreement. 10 Exact logistic regression was performed to evaluate the predictive value of the reported radiographic features to diagnose 360‐GDV. The median of the grades attributed by the three radiologists was used for the statistical analysis. For the nominal radiographic variables that described the position of the pylorus in the lateral and ventrodorsal (or dorsoventral) projections, the mode was selected and used for the statistical analysis. In cases of complete discrepancy between the three radiologists, the position attributed to the most experienced radiologist was selected. A receiver operating characteristic curve was designed for the variable heart rate. Odds Ratios and 95% Confidence Intervals of the selected significant variables were reported. P‐value was considered significant if <.05. Stata IC Version 13.1 was used for the statistical analysis.
3. RESULTS
3.1. Sample size
The variable “Craniodorsal pylorus” was selected to determine a specific sample size to compare GD (6%) and 360‐GDV (37%) with a final number of 28 individuals in each group. For the 180‐GDV versus 360‐GDV comparison, the variable gastric pneumatosis (6% in 180‐GDV vs. 31% in 360‐GDV) was selected, establishing a final number of required enrollments of 37 in each group. As later reported, the number of 360‐GDV with radiographic images available was sparse, and a maximum number of 16 cases were available. The calculated number of cases for the GD (28) and 180‐GDV (37) were retrieved and used for the descriptive and statistical analysis.
3.2. Animals and clinical variables
A total of 25 dogs with confirmed 360‐GDV were identified (surgically confirmed n = 18; necropsy confirmed n = 7). Sixteen of the 25 cases had radiographs available for interpretation and were used in the study. In the 360‐GDV group, 11/16 cases had a complete three‐view abdominal study for evaluation, including a ventrodorsal projection. A right lateral and ventrodorsal view were available in 3 of 16 cases. In the remaining two cases, the right and left lateral views were evaluated. Twelve of 16 of the 360‐GDV cases with radiographs available were confirmed at surgery, and the remaining (4/16) were confirmed at necropsy. The median age in the 360‐GDV group was 10 years old (2–13 years), and the mean weight was 41.5 kg (SD ± 11.3). Male dogs were significantly more frequent (P = .03) in the 360‐GDV group when compared with the other two groups (50% in the GD, 60% in the 180‐GDV, 88% in the 360‐GDV). No significant differences in age (P = .57) and body weight (P = .13) were found between the three groups. The most prevalent breed was German shepherd dog (13/81; 16%), followed by Labrador retriever (11/81; 13%), Great Danes (8/81; 10%), and poodles (7/81 8%). For the 360‐GDV group, German shepherd dogs were also the most prevalent breed (3/16; 19%), followed by Great Dane (2/16; 12.5%), Labrador retriever (2/16; 12.5%), and poodle (2/16; 12.5%). Other breeds in this group include Akita (1), Based Hound (1), Doberman (1), Newfoundland(1), and Weimaraner (1). The remaining two dogs in this group were considered mix‐breed. There was no statistical difference in the prevalence of German shepherd dogs between the three groups (P = .14).
The duration of clinical signs was recorded in 32 of 81 cases (10/16 360‐GDV; 13/37 180‐GDV; 9/28 GD). The mean duration of clinical signs for the 360‐GDV group was 5.6 h (SD ± 7.2), for the 180‐GDV was 11.13 h (SD = 19), and 5.2 h (SD = 5.75) for the GD group (Figure 1A). No significant differences in the duration of clinical signs were detected between the three groups. The heart rate on admission (beats per minute) was available in 44 of 81 cases (13/16 360‐GDV; 15/37 180‐GDV; 16/28 GD). The heart rate in cases affected with 360‐GDV was significantly higher when compared with GD (P < .001) and 180‐GDV (P = .032). The mean heart rate in cases presenting with 360‐GDV was 184 (SD ± 33.34), for 180‐GDV was 151 (SD ± 37.54), and for GD was 130 (SD ± 25.37; Figure 1B). A receiver operating characteristic curve was designed for the variable heart rate. The cut‐off value that maximizes sensitivity and specificity between 360‐GDV and GD is 150 (sensitivity 84%; specificity 80%). The cut‐off value that maximizes sensitivity and specificity between 360‐GDV and 180‐GDV is 162 (sensitivity 62%; specificity 45%). No significant differences in PCV, total solids, venous lactate, and pH were detected between the three groups.
FIGURE 1.

A, Box and whisker plot of the duration of clinical signs (hours) by disease group. Higher standard deviation was seen in the 180‐GDV group, but no statistical differences were detected between the three groups. B, Box and whisker plot of the heart rate by disease group. *Significant differences between 360‐GDV and 180‐GDV. **Significant differences between 360‐GDV and GD.
3.3. Sensitivity and specificity
The sensitivity for detection of GD cases for the three board‐certified radiologists ranged between 75% and 82.1%. The specificity ranged between 86.8% and 90.5%. The sensitivity for detection of 180‐GDV cases for the three board‐certified radiologists ranged between 83.8% and 92%. The specificity ranged between 88.6% and 91%. The sensitivity for detection of 360‐GDV cases for the three board‐certified radiologists ranged between 43.7% and 50%. The specificity ranged between 84.6% and 92.3% (Table 1).
TABLE 1.
Mean and range (%) of sensitivity and specificity of radiographs to diagnose each of the included disease groups.
| GD | 180‐GDV | 360‐GDV | |
|---|---|---|---|
| Sensitivity | 79.3 (75–82.1) | 87.4 (83.8–92) | 47.9 (43.7–50) |
| Specificity | 88.6 (86.8–90.5) | 89.4 (88.6–91) | 88.7 (84.6–92.3) |
Abbreviations: GD, gastric dilatation; GDV, gastric dilatation and volvulus.
3.4. Agreement between radiologists
Agreement rates on final diagnosis (360‐GDV, 180‐GDV, or GD) between the three radiologists ranged between 75.3 and 79%. The highest agreement rate was in cases of 180‐GDV (87%), followed by the GD cases (72%) and 360‐GDV (46%). The Kappa coefficient between radiologists on final diagnosis was considered substantial, ranging between 0.608 and 0.669. Complete agreement on the final diagnosis of 360‐GDV between the three radiologists was found in only four cases (Figure 2). Two cases with surgically confirmed 360‐GDV were graded as 180‐GDV by the three radiologists (Figure 3). Two cases with surgically confirmed 360‐GDV were graded as GD by the three radiologists (Figure 4).
FIGURE 2.

Right lateral and ventrodorsal projections in a dog with surgically confirmed 360‐GDV. In this case, the three radiologists agreed on a final diagnosis of 360‐GDV. The pylorus is gas‐distended and located in the right cranial quadrant in the ventrodorsal projection and the cranioventral abdomen in the right lateral projection (yellow arrows). The spleen is malpositioned and mildly enlarged.
FIGURE 3.
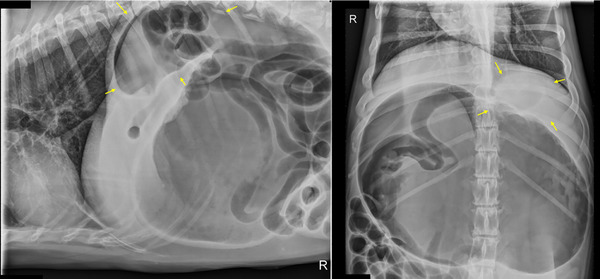
Right lateral and dorsoventral projection in a dog with a surgically confirmed 360‐GDV. In this case, the three radiologists reported a 180‐GDV as the final diagnosis. The pylorus is located in the craniodorsal quadrant in the right lateral projection and the left cranial quadrant in the dorsoventral projection (yellow arrows).
FIGURE 4.

Right lateral and ventrodorsal projection of a case with surgically confirmed 360‐GDV that was diagnosed as a GD by the three radiologists. In this case, the most commonly reported location of the pylorus in the right lateral view was cranioventral (2/3 radiologists). In the ventrodorsal projection, 2 of 3 of radiologists localized the pylorus in the right cranial quadrant (yellow arrows).
In 51 of 81 cases (63%), there was complete agreement to determine the presence (39/51) or absence (12/51) of pyloric malpositioning. The resultant Kappa coefficient was variable ranging between 0.361 and 0.751. When determining the pyloric position in the right lateral projection, in 41 observations (50%), there was full agreement between the three radiologists. Twenty‐seven of these observations (27/41; 66%) were for the craniodorsal position, which was most seen in the 180‐GDV cases (86.5% vs. 7% for the GD and 31.2% in the 360‐GDV). In eight observations (8/41; 19.5%), the agreement was for the cranioventral position, which is the considered normal position of the pylorus. The Kappa coefficients reflecting agreement by paired radiologists in determining the pyloric position in the right lateral ranged between 0.456 and 0.589. Complete agreement in pyloric position in the ventrodorsal or dorsoventral projection between the three radiologists was found in 28 of the 58 (48%) cases in which this view was available. Of these, 14 (14/28; 50%) were for the left cranial position, which was frequently reported in cases of 180‐GDV. When comparing agreement by paired radiologists in determining pyloric position in the ventrodorsal or dorsoventral projections, the Kappa coefficients ranged between 0.503 and 0.557.
3.5. Radiographic variables associated with 360‐GDV
Cases with 360‐GDV (56.2% absent; 12.5% mild; 31.25% marked) had a significantly higher degree of esophageal distension when compared to GD (82.1% absent; 10.7% mild; 7.1% marked; P = .039) and 180‐GDV (79.2% absent; 11.6% mild; 9.2% marked; P = .041). No significant differences in esophageal distension were found between GD and 180‐GDV (P = .918; Table 2). In the presence of esophageal gas distension, the odds of having 360‐GDV were 2.34 higher than a GD and 2.23 higher than a 180‐GDV.
TABLE 2.
Percentage of cases with absent, mild, and marked esophageal gas distension by disease group.
| GD | 180‐GDV | 360‐GDV | |
|---|---|---|---|
| Absent | 82.1 | 79.2 | 56.2 |
| Mild distention | 10.7 | 11.6 | 12.5 |
| Marked distension | 7.1 | 9.2 | 31.2 |
Notes: Cases with 360‐GDV had significantly higher degree of esophageal distension when compared with GD (P = .039) and 180‐GDV (P = .041). No significant differences in esophageal distension were found between GD and 180‐GDV (P = .918).
Abbreviations: GD, gastric dilatation; GDV, gastric dilatation and volvulus.
Cases presenting with 180‐GDV had significantly higher frequencies of pyloric malposition when compared to GD [91.9% vs. 17.8% (P < .001)] and 360‐GDV (91.9% vs. 56.2% [P = .005]). When evaluating the pyloric position in the right lateral projection, 180‐GDV were significantly more frequently located in the craniodorsal aspect of the abdomen (86.5% vs. 7.1% for the GD group and 31.2% for the 360‐GDV; Table 3). A relatively similar proportion of GD cases (35.7%) and 360‐GDVs (25%) had the pylorus located in the caudoventral quadrant. The majority of the GD cases (53.6%) had pylorus located in the cranioventral abdomen. Almost a third (31.3%) of the 360‐GDV cases also had the pylorus located in the normal position (cranioventral abdomen), with only 10.8% of the 180‐GDV being located in the cranioventral abdomen (Figure 5). The 180‐GDV group had significantly different pyloric distribution for each location when compared to the GD (P < .001) and 360‐GDV (P < .001) groups. No significant differences in the distribution of pyloric position were found between the GD and 360‐GDV groups (P = .071).
TABLE 3.
Percentage of cases with the pylorus located in each abdominal quadrant by disease group (pylorus located in the right lateral projection).
| GD | 180‐GDV | 360‐GDV | |
|---|---|---|---|
| Craniodorsal | 7.1 (2) | 86.49 (32) | 31.2 (5) |
| Caudodorsal | 3.57 (1) | 0 (0) | 12.5 (2) |
| Caudoventral | 35.71 (10) | 2.7 (1) | 25 (4) |
| Cranioventral | 53.57 (15) | 10.8 (4) | 31.2 (5) |
Abbreviations: GD, gastric dilatation; GDV, gastric dilatation and volvulus.
FIGURE 5.

Two‐dimensional radar chart depicting pyloric positioning by disease group. 180‐GDV cases were very frequently present in the craniodorsal quadrant. No significant differences in pyloric position were found between 360‐GDV and GD. However, 360‐GDV tends to have the pylorus more even and central distribution when compared with GD which was more frequently located in the cranioventral quadrant.
When evaluating the pyloric position in the ventrodorsal or dorsoventral projection, the majority of the GD (60.7%) and the 360‐GDV (62.5%) were located in the right cranial quadrant (Table 4). The 180‐GDV were almost exclusively located in the left cranial quadrant (91%; Figure 6). Approximately a third (32%) of the GD cases were in the right right caudal quadrant for only (6.2%) of the 360‐GDV cases. However, this difference was not statistically significant (P = .089). No statistical differences were found between 360‐GDV and GD for the remaining quadrants. The distribution of the pyloric position of the 180‐GDV in the ventrodorsal or dorsoventral projection was statistically different from GD (P < .001) and 360‐GDV in all four quadrants (P < .001).
TABLE 4.
Percentage of cases with the pylorus located in each abdominal quadrant by disease group (pylorus located in the ventrodorsal or dorsoventral projections.
| GD | 180‐GDV | 360‐GDV | |
|---|---|---|---|
| Right cranial | 60.7 (17) | 0 (0) | 62.5 (10) |
| Left cranial | 7.1 (2) | 89.1 (33) | 31.2 (5) |
| Left caudal | 0 (0) | 5.4 (2) | 0 (0) |
| Right caudal | 32.1 (9) | 5.4 (2) | 6.25 (1) |
Abbreviations: GD, gastric dilatation; GDV, gastric dilatation and volvulus.
FIGURE 6.

Two‐dimensional radar chart depicting pyloric positioning in the ventrodorsal or dorsoventral projections. The apex of the triangles is oriented towards the most frequent positions. 180‐GDV cases were very frequently present in the left cranial quadrant. No significant differences in pyloric position were found between 360‐GDV and GD. However, GD cases displayed higher frequencies in the right caudal abdomen when compared to 360‐GDV.
Gas distension of the small intestine was significantly less frequently detected in 360‐GDV (18.7%) cases when compared to GD (60.7%; P = .011) and 180‐GDV (56.76%; P = .015). No differences in the frequency of small intestinal gas distension were found between GD and 180‐GDV (P = .8). Gastric compartmentalization was present in 89.19% of the 180‐GDVs and only 37.5% of the 360‐GDVs. None of the GD cases had evidence of compartmentalization. Compartmentalization was significantly more prevalent in the 180‐GDV group when compared with the 360‐GDV (P = .031) and the GD group (P < .001). Compartmentalization was also significantly more frequently detected in cases affected with 360‐GDV when compared to GD (P = .0011). No significant differences in the degree of gastric distension were found between 180‐GDV and 360‐GDV (P = .227). 360‐GDV (P = .048) had significantly more severe gastric distension when compared with GD. No significant differences in the degree of gastric distension were found between 180‐GDV and GD (P = .472). In the presence of severe gas distension of the stomach, the odds of having a final diagnosis of 360‐GDV were 5.069 higher than a GD.
The percentage of decreased serosal contrast was significantly higher for the 360‐GDV group (62.5%) when compared to the GD group (25%; P = .035). No significant differences in the prevalence of decreased serosal contrast between the GD and 180‐GDV groups (43.2%; P = .24) nor between the 180‐GDV and the 360‐GDV (P = .19).
No significant differences in the percentage of presence of duodenal distension (GD vs. 180‐GDV (P = .31]; 180‐GDV vs. 360‐GDV [P = .33]; GD vs. 360‐GDV [P = .056]), splenic malposition (GD vs. 180‐GDV (P = .21]; GD vs. 360‐GDV [P = .184]; 180‐GDV vs. 360‐GDV [P = .534]), spleen position in the ventrodorsal or dorsoventral projections (GD vs. 180‐GDV [P = .52]; 180‐GDV vs. 360‐GDV [P = .46]; GD vs. 360‐GDV (P = .09]), splenic size (GD vs. 180‐GDV [P = .29]; 180‐GDV vs. 360‐GDV [P = .105]; GD vs. 360‐GDV [P = .45]), size of the caudal vena cava (GD vs. 180‐GDV [P = .24]; 180‐GDV vs. 360‐GDV [P = .74]; GD vs. 360‐GDV [P = .33]) were found between the three groups.
The presence of gastric pneumatosis was recorded in only two surgically confirmed 180‐GDV cases (3.7% of the cases with gastric volvulus (180 or 360‐GDV) and 2.4% of all cases). Pneumoperitoneum was detected in 4 of 81 cases. One of these cases had a surgical diagnosis of 360‐GDV. In the remaining three, the final diagnosis was 180‐GDV. The low number of cases with pneumatosis and pneumoperitoneum prevented further statistical analysis.
4. DISCUSSION
The current study reports that cases of severe gastric distension with normal pyloric position, the presence of esophageal gas distension, and the absence of small intestinal gas distension should raise concerns for 360‐GDV. This manuscript also describes additional radiographic and clinical features that could help clinicians and radiologists improve the detection of 360‐GDV. However, as hypothesized, the sensitivity of radiographs to detect 360‐GDV in dogs is low, with multiple 360‐GDV cases being commonly misinterpreted as GD. As expected, detection rates peaked in cases 180‐GDV, in which compartmentalization of the stomach was most commonly encountered. Contrary to what was initially surmised, the agreement between radiologists on pyloric position and final diagnosis was considered moderate to substantial, respectively.
As initially hypothesized, and despite the knowledge of reviewers of the purpose of the study to identify 360‐GDV cases, the reported sensitivity of radiographs to identify 360‐GDV was low (50%). Much higher sensitivity rates are reported for GD and 180‐GDV cases. The low sensitivity is explained by the high number of GD cases diagnosed by radiologists as 360‐GDV. This highlights the difficulty of using radiographs to discriminate between the two conditions. One could speculate that the detection rates of 360‐GDV would increase if history and clinical variables had been provided to the radiologists. However, all dogs had acute onset of abdominal distension, and based on our results, the duration of clinical signs did not differ between the three groups. Only the presence of tachycardia was significantly associated with the presence of 360‐GDV. Several cases with radiographic features consistent with 180‐GDV were diagnosed with 360‐GDV cases, emphasizing the dynamic nature of this condition. It is reasonable to consider that the longer the time between image acquisition and surgery, the higher the chances of further gastric rotation. Unfortunately, the time between the radiographic study and the exploratory laparotomy was not recorded.
The agreement between radiologists on the final diagnosis was considered substantial, with a Kappa coefficient ranging between 0.608 and 0.669. Similar agreement coefficients have been reported in very well‐described cardiac scores, 11 some abdominal organs, 12 or other imaging modalities such as CT 13 or scintigraphy. 14 Contrary to what was hypothesized, the agreement rates between radiologists for precise localization of the pylorus in the right lateral and ventrodorsal (or dorsoventral) projections were considered moderate, with Kappa coefficients reaching almost substantial levels of agreement (highest Kappa coefficient = 0.589). This is the first report describing interobserver agreement between radiologists on diagnosing dogs presenting with acute gastric distension.
Esophageal gas distension was significantly more severe in dogs affected with 360‐GDV (31.2% marked esophageal distension) when compared with the GD (7.1% marked esophageal gas distension) and 180‐GDV groups (8.1% marked esophageal gas distension). The higher degree of gastric torsion could cause more severe tightening and subsequent closure of the cardia, decreasing esophageal outflow and preventing the passage of fluid, gas, and ingesta. It has been suggested that in dogs affected with 360‐GDV, gastric intubation is likely not feasible due to the reported more severe narrowing at the cardia and complete sealing of the lower esophageal sphincter. 9 Esophageal gas distension has also been recently reported in a single case affected with 360‐GDV. 15 Another report of a dog with esophageal perforation diagnosed with 360‐GDV hypothesized that severe gastric torsion, as seen with 360‐GDVs, could cause esophageal necrosis and perforation. 16 Rather surprising was the lack of significant differences in esophageal distension between GD and 180‐GDV. We believe that milder gastric torsions, such as a 180‐degree torsion, might allow the passage of fluid and gas through the cardia. Clinicians and radiologists should be aware of the higher odds of having a final diagnosis of 360‐GDV when esophageal gas distension is present.
Interestingly, the 360‐GDV cases had a significantly lower prevalence of small intestinal gas distension when compared to GD. A possible explanation for this finding is that severe gastric torsion would further occlude the gastroduodenal junction and complete blockage at this level. 4 As a result, all the gas present in the stomach cannot further extend into the duodenum and remaining portions of the gastrointestinal tract. Additionally, the pathophysiology of GD without volvulus might be associated with generalized decreased gastrointestinal motility and secondary, more severe, and diffuse distension of the gastrointestinal tract.
The percentage of decreased peritoneal serosal contrast was significantly higher for the 360‐GDV group when compared to the GD group, but not significant with the 180‐GDV group. Hemoperitoneum can be secondary to tearing of the short gastric veins that course along the greater curvature of the stomach1. A higher degree of gastric torsion is likely associated with more severe tearing of the gastric vasculature, which explains the reported findings. Additionally, peritoneal inflammation with subsequent steatitis could further contribute to the difference between 360‐GDV and 180‐GDV. Mild stranding of the peritoneal fat around the torsed vasculature has been recently reported in a dog with confirmed 360‐GDV that underwent a CT examination. 15 Visceral crowding associated with higher degrees of gastric distension seen in the 180‐GDV and 360‐GDV groups relative to the GD group could also contribute to the lower serosal contrast relative to the GD group. Possible confounding associations between gastric distension and serosal contrast were not further explored in the current study.
Compartmentalization was very prevalent (89.19%) in the 180‐GDV group and present in only 37.5% of the 360‐GDVs, with none of the GD cases exhibiting evidence of compartmentalization. This finding is in agreement with the previously reported absence of compartmentalization that has been previously described in dogs affected with 360‐GDV. 8 , 9 , 15 Two surgically confirmed 360‐GDV cases displayed a well‐demarcated gastric compartment and a craniodorsal pylorus in the right lateral projection. These cases were both graded by the three radiologists as 180‐GDV (Figure 3). We believe that during the radiographic exam, the pylorus was likely located in the left craniodorsal abdomen, as seen with the 180‐GDV cases, and that further rotation of the stomach between the radiographs and the exploratory laparotomy caused the reported 360‐GDV. This emphasized that GDV in dogs is a very dynamic process and that changes in gastric torsion are to be expected between the radiographic exam and surgery.
Pyloric malposition was most frequently reported in the 180‐GDV cases when compared to GD and 360‐GDV. When comparing the pyloric position in the right lateral and ventrodorsal (or dorsoventral) view, the distribution of the pyloric position of the 180‐GDV cases significantly differs from the remaining groups. Gastric dilatation and 360‐GDV cases had a relatively high frequency of normal pyloric positioning, and no statistical differences between the two were found. This finding is in accordance with previous reports describing this condition in dogs. 8 , 15 Despite the lack of significant differences between GD and 360‐GDV, there was a trend with the pylorus in the 360‐GDV cases being more centrally located and closer to the midline in the ventrodorsal (or dorsoventral) view (Figure 6). This finding is somewhat similar to the case reported by Li et al. 15 of a 15‐year‐old spayed female with confirmed 360‐GDV. In this case, a CT exam confirmed the dorsal and medial displacement of the gastroduodenal junction.
Radiographic variables that were similar between the three groups included duodenal distension, splenic position and size, and the size of the caudal vena cava. When compared to previous studies, lower frequencies of gastric pneumatosis and pneumoperitoneum were reported in the current manuscript. 8 The difference is likely caused by the earlier presentation of the cases included in the current study.
Male dogs were significantly overrepresented in the 360‐GDV (88%), with only two being intact males. Previous reports have described a similar higher incidence of GDV in male dogs compared with females. 17 Higher activity levels and conformational differences with larger abdominal cavity and thorax in males are the most plausible explanations for this difference. The majority (14/16) of the dogs with 360‐GDV were neutered; considering that the stomach is maintained in position by several ligaments, one could speculate that increased laxity of the ligaments caused by estrogen deficiency as reported for the cranial cruciate ligament18 could predispose these patients to a higher degree of gastric rotation. The number of cases is low, and this hypothesis remains to be proven by histopathologic studies. As expected, and despite the relatively prompt presentation of most of the patients, labwork abnormalities reflecting multiple organ dysfunction syndrome were commonly found in dogs presenting with GDV. 4 In the current study, the heart rate on admission was significantly more elevated in cases affected with 360‐GDV when compared to the rest. Tachycardia could be driven by multiple pathophysiologic events occurring during GDV. Hypovolemic shock is the most likely of all these. 4 Other variables associated with shock, such as hyperlactatemia or elevated PCV, were also more severe in cases affected with 360‐GDV, but interestingly, the difference was considered not statistically significant. Prompt admission after the onset of clinical signs could explain the lack of differences in these variables.
Several limitations should be noted. There was absent documentation of dogs undergoing preoperative procedures for stabilization (trocharization, intubation) that could have modified or affected some of the reported radiographic findings. Also, several clinicians and surgeons from multiple institutions were involved in the cases. The gradings determined by radiologist for each variable were subjective and based on their previous experience. The search of medical records was not standardized between the different institutions included in the study. As a result, describing the prevalence of 360‐GDV in dogs is not possible. For the statistical analysis regarding the pyloric position, we use the mode between the three radiologists. In cases of complete discrepancy, we used the position attributed by the most experienced radiologist based on the assumption that this evaluator might be more successful at locating the pylorus. We acknowledge that this assumption might be controversial and errors might arise from it. Limitations associated with the uniplanar nature of radiographs should be noted. Also, some cases included did not undergo a complete abdominal radiographic exam with three orthogonal projections. A large proportion of these cases were in the 180‐GDV group, with most of the 360‐GDV cases (69%) and the GD cases (57%) having a complete set of abdominal radiographs. Since the difficulty relies on differentiating 360‐GDV from GD, we believe that this limitation had little impact on our results.
In conclusion, we found moderate agreement among radiologists at localizing the gastric pylorus in both the right lateral and ventrodorsal views. The agreement was substantial for the final diagnosis of GD, 180‐GDV, or 360‐GDV. The sensitivity for the detection of 360‐GDV was low, but the specificity was high. The presence of esophageal gas distension and lack of small intestinal dilation were the only radiographic features associated with 360‐GDV when compared with a group of 180‐GDV and GD. A similar pyloric position was found between GD and 360‐GDV, with a trend of 360‐GDV cases having a more central and dorsal pylorus when compared to GD. Additional radiographic variables that could help differentiate GD from 360‐GDV include the degree of gastric distension and the peritoneal serosal contrast. Radiologists and clinicians should be aware of the low sensitivity of radiographs for the detection of 360‐GDV, and clinical variables such as heart rate should always be considered for decision‐making.
LIST OF AUTHOR CONTRIBUTIONS
Category 1
-
(a)
Conception and design: Espinosa Mur, Appleby, Kathryn, Erin; zur Linden
-
(b)
Acquisition of data: Mur, Appleby, Kathryn, Lindsey, Erin, Daniaux, zur Linden
-
(c)
Analysis and interpretation of data: Mur, Appleby, zur Linden
Category 2
-
(a)
Drafting the article: Espinosa Mur, Appleby, Linden
-
(b)
Reviewing article for intellectual content: Mur, Appleby, Kathryn, Singh, Gabrielle, Lindsey, Erin, Daniaux, zur Linden
Category 3
-
(a)
Final approval of the completed article: Espinosa Mur, Appleby, Kathryn, Singh, Gabrielle, Lindsey, Erin, Daniaux, zur Linden
Category 4
-
(a)
Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved: Espinosa Mur, Appleby, Kathryn, Singh, Gabrielle, Lindsey, Erin, Daniaux, zur Linden.
CONFLICT OF INTEREST STATEMENT
The authors declare no conflict of interest.
PREVIOUS PRESENTATION DISCLOSURE
Preliminary results were presented at the 2023 annual meeting of the American College of Veterinary Radiologists, New Orleans, LA, USA.
REPORTING CHECKLISTS
The STROBE Vet checklist has been used as a guideline for manuscript preparation.
DISCLOSURE
This research is original and has not been previously submitted.
ACKNOWLEDGEMENTS
This article is dedicated to Eric and Oliver Espinosa Romero and Concepcion Romero Jimenez for their invaluable encouragement and unconditional support. The first author would like to express gratitude to Dr. Philip Kass from the University of California for assistance with power analysis.
Mur PE, Appleby R, Phillips KL, et al. Radiographic findings in dogs with 360 degrees gastric dilatation and volvulus. Vet Radiol Ultrasound. 2025;66:e13445. 10.1111/vru.13445
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Kare T, Spencer JAA, Veterinary Surgery: Small Animal. Stomach. 2nd ed. 2017;1730‐1760.
- 2. Ullmann B, Seehaus N, Hungerbühler S, Meyer‐Lindenberg A. Gastric dilatation volvulus: a retrospective study of 203 dogs with ventral midline gastropexy. J Small Anim Pract. 2016;57:18‐22. [DOI] [PubMed] [Google Scholar]
- 3. Rosselli D, Updated Information on gastric dilatation and volvulus and gastropexy in dogs. Vet Clin North Am Small Anim Pract [Internet]. 2022; 52:317‐337. Available from: https://pubmed.ncbi.nlm.nih.gov/35082096/ [DOI] [PubMed] [Google Scholar]
- 4. Sharp CR, Rozanski EA. Cardiovascular and systemic effects of gastric dilatation and volvulus in dogs. Top Companion Anim Med. 2014;29:67‐70. [DOI] [PubMed] [Google Scholar]
- 5. Broome C, Walsh V. Gastric dilatation‐volvulus in dogs. N Z Vet J. 2003;51:275‐283. [DOI] [PubMed] [Google Scholar]
- 6. Mackenzie G, Barnhart M, Kennedy S, DeHoff W, Schertel E. A retrospective study of factors influencing survival following surgery for gastric dilatation‐volvulus syndrome in 306 dogs. J Am Anim Hosp Assoc. 2010;46:97‐102. [DOI] [PubMed] [Google Scholar]
- 7. Glickman LT, Glickman NW, Schellenberg DB, Raghavan M, Lee TL. Incidence of and breed‐related risk factors for gastric dilatation‐volvulus in dogs. J Am Vet Med Assoc. 2000;216:40‐45. [DOI] [PubMed] [Google Scholar]
- 8. Fischetti AJ, Saunders HM, Drobatz KJ. Pneumatosis in canine gastric dilatation‐volvulus syndrome. Vet Radiol Ultrasound. 2004;45:205‐209. [DOI] [PubMed] [Google Scholar]
- 9. Stieger‐Vanegas SM, Stomach FrankPM, Veterinary diagnostic radiology Donald E Thrall. 2017;894‐925.
- 10. McHugh ML. Interrater reliability: the kappa statistic. Biochem Med (Zagreb). 2012;22:276‐282. [PMC free article] [PubMed] [Google Scholar]
- 11. Baisan RA, Vulpe V. Vertebral heart size and vertebral left atrial size reference ranges in healthy Maltese dogs. Vet Radiol Ultrasound. 2022 Jan 28;63:18‐22. [DOI] [PubMed] [Google Scholar]
- 12. Lobacz MA, Sullivan M, Mellor D, Hammond G, Labruyère J, Dennis R. effect of breed, age, weight and gender on radiographic renal size in the dog. Vet Radiol Ultrasound. 2012;53:437‐441. [DOI] [PubMed] [Google Scholar]
- 13. Tan YL, Marques A, Schwarz T, Mitchell J, Liuti T. Clinical and CT sialography findings in 22 dogs with surgically confirmed sialoceles. Vet Radiol Ultrasound. 2022;63:699‐710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Quiney LE, Ireland JL, Dyson SJ. Evaluation of the diagnostic accuracy of skeletal scintigraphy in lame and poorly performing sports horses. Vet Radiol Ultrasound. 2018;59:477‐489. [DOI] [PubMed] [Google Scholar]
- 15. Li W‐S, Wu R‐S, Liu P‐C, Wang H‐C, Chen K‐S. What is your diagnosis? J Am Vet Med Assoc. 2022;259:1‐4. [DOI] [PubMed] [Google Scholar]
- 16. Streeter EM, Rozanski EA, Berg J, Walker T, O'toole TE. Esophageal perforation in a dog following an acute episode of gastric dilatation with 360 degrees volvulus. J Vet Emerg Crit Care. 2004;14:125‐128. [Google Scholar]
- 17. Paris JK, Yool DA, Reed N, Ridyard AE, Chandler ML, Simpson JW. Chronic gastric instability and presumed incomplete volvulus in dogs. J Small Anim Pract. 2011;52:651‐655. [DOI] [PubMed] [Google Scholar]
- 18. Sellon DC, Marcellin‐Little DJ. Risk factors for cranial cruciate ligament rupture in dogs participating in canine agility. BMC Vet Res. 2022;18:39. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


