Abstract
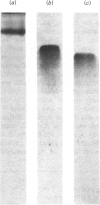
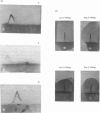
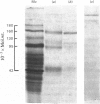
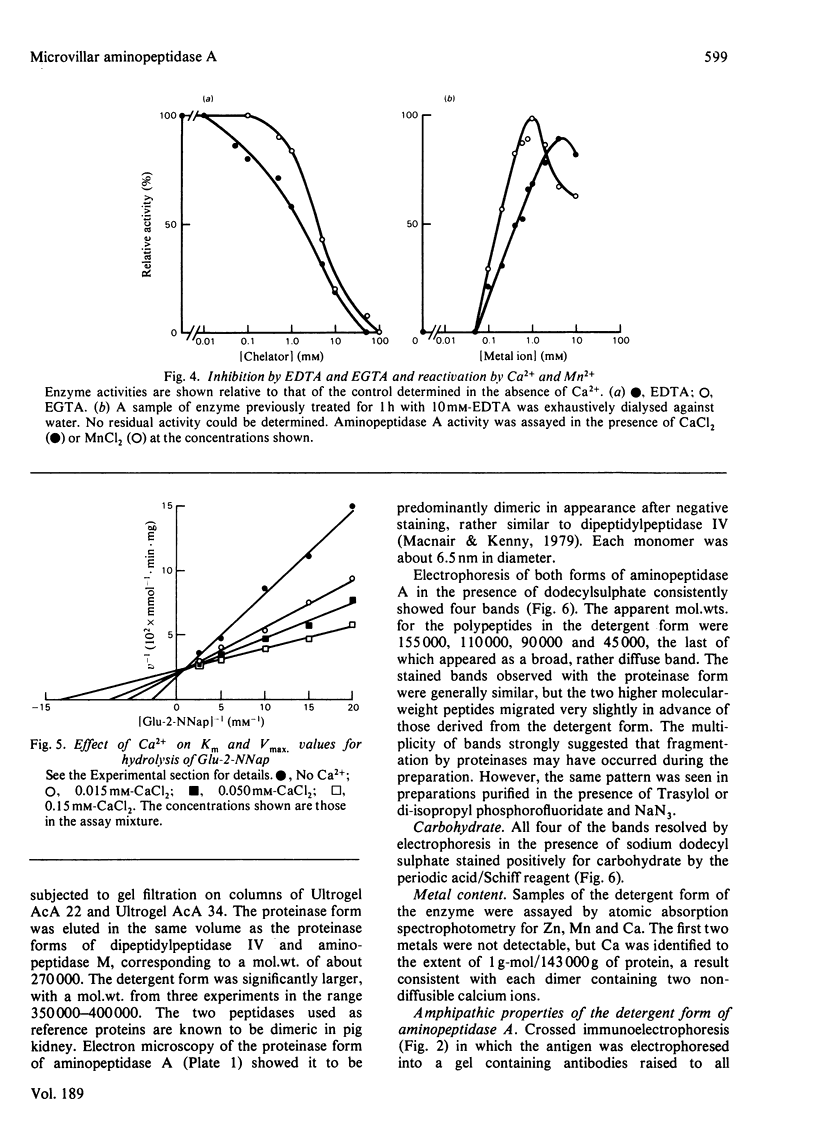
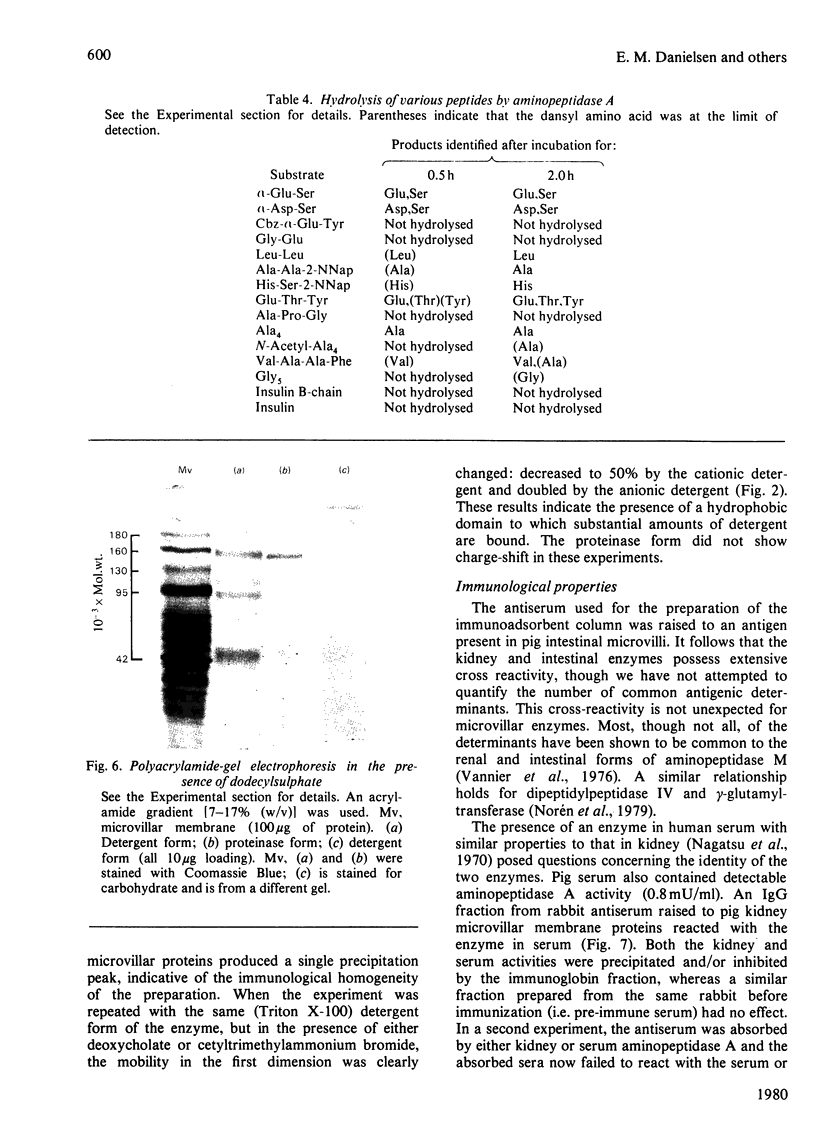
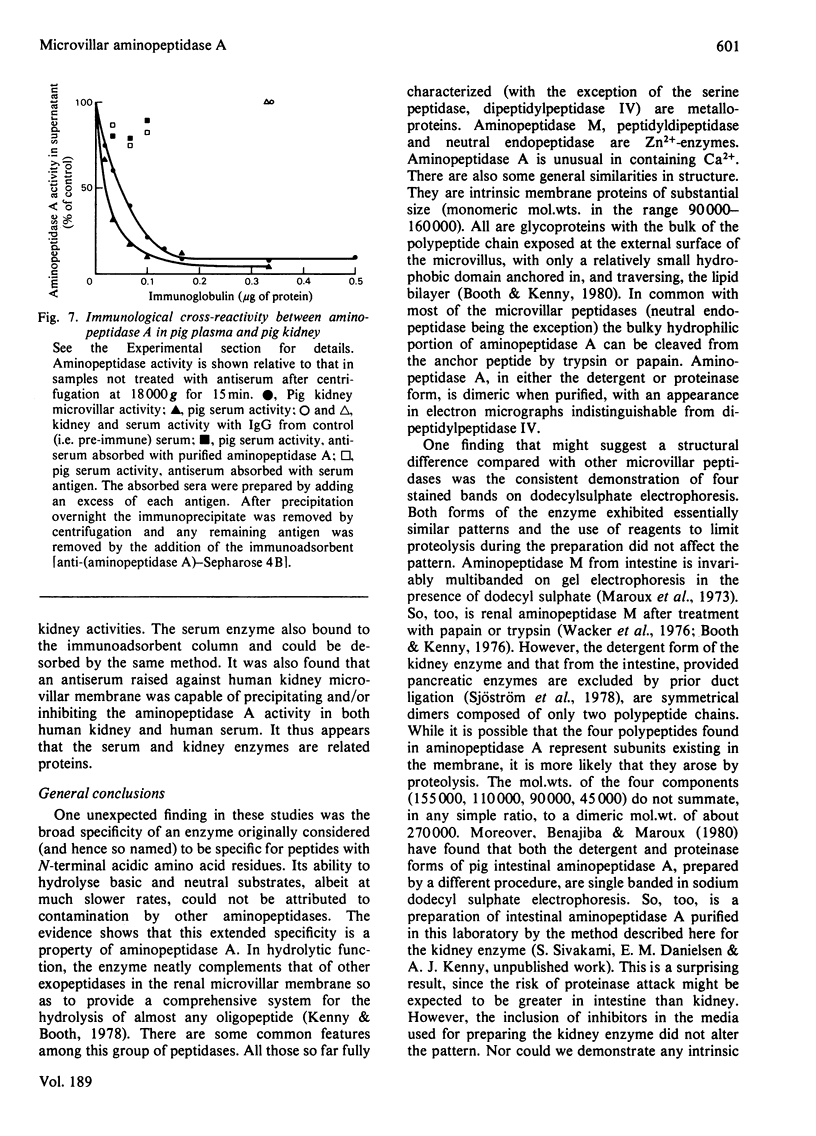
Aminopeptidase A (aspartate aminopeptidase, EC 3.4.11.7) was purified 2000-fold from pig kidney cortex. The essential step in the purification was chromatography on an immunoadsorbent column prepared from a rabbit antiserum raised against pig intestinal aminopeptidase A. Glutamyl and aspartyl substrate were attacked most rapidly and their hydrolyses were stimulated by Ca2+. The 2-naphthylamide derivatives of neutral and basic amino acids were also hydrolysed by aminopeptidase A, but at rates about two orders of magnitude lower, and Ca2+ was inhibitory. The possibility that these atypical substrates were hydrolysed by traces of aminopeptidase M (EC 3.4.11.2) contaminating the preparation could be excluded on several grounds. Aminopeptidase A was sensitive to inhibition by chelating agents and the inactive enzyme could be reactivated by Ca2+ or Mn2+. Atomic absorption spectrophotometry revealed 1 g-atom of Ca/143000 g of protein. Two forms of the enzyme were purified: an amphipathic form solubilized from the membrane by Triton X-100 (detergent form) and a hydrophilic form released by incubation with trypsin (proteinase form). The detergent form exhibited charge-shift in crossed immunoelectrophoresis when anionic or cationic detergents were present. On gel filtration, mol.wts. of 350000--400000 and 270000 were calculated for the detergent and proteinase forms. Electron microscopy after negative staining of the proteinase form revealed a dimeric structure. Electrophoresis of either form in the presence of sodium dodecyl sulphate revealed four polypeptides with mobilities corresponding to apparent mol.wts. of 155000, 110000, 90000 and 45000. All four bands stained positively for carbohydrate. Pig serum possesses weak aminopeptidase A activity; immunological experiments showed it to be a similar protein.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andria G., Marzi A., Auricchio S. Alpha-Glutamyl-beta-naphthylamide hydrolase of rabbit small intestine. Localization in the brush border and separation from other brush border peptidases. Biochim Biophys Acta. 1976 Jan 8;419(1):42–50. doi: 10.1016/0005-2736(76)90370-9. [DOI] [PubMed] [Google Scholar]
- Benajiba A., Maroux S. Purification and characterization of an aminopeptidase A from hog intestinal brush-border membrane. Eur J Biochem. 1980 Jun;107(2):381–388. doi: 10.1111/j.1432-1033.1980.tb06040.x. [DOI] [PubMed] [Google Scholar]
- Bhakdi S., Bhakdi-Lehnen B., Bjerrum O. J. Detection of amphiphilic proteins and peptides in complex mixtures. Charge-shift crossed immunoelectrophoresis and two-dimensional charge-shift electrophoresis. Biochim Biophys Acta. 1977 Oct 3;470(1):35–44. doi: 10.1016/0005-2736(77)90059-1. [DOI] [PubMed] [Google Scholar]
- Booth A. G. A novel system for the two-dimensional electrophoresis of membrane proteins. Biochem J. 1977 Apr 1;163(1):165–168. doi: 10.1042/bj1630165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booth A. G., Hubbard L. M., Kenny A. J. Proteins of the kidney microvillar membrane. Immunoelectrophoretic analysis of the membrane hydrolases: identification and resolution of the detergent- and proteinase-solubilized forms. Biochem J. 1979 May 1;179(2):397–405. doi: 10.1042/bj1790397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booth A. G., Kenny A. J. A rapid method for the preparation of microvilli from rabbit kidney. Biochem J. 1974 Sep;142(3):575–581. doi: 10.1042/bj1420575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booth A. G., Kenny A. J. Proteins of the kidney microvillar membrane. Asymmetric labelling of the membrane by lactoperoxidase-catalysed radioiodination and by photolysis of 3,5-di[125I]iodo-4-azidobenzenesulphonate. Biochem J. 1980 Apr 1;187(1):31–44. doi: 10.1042/bj1870031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booth A. G., Kenny A. J. Proteins of the kidney microvillus membrane. Identification of subunits after sodium dodecylsullphate/polyacrylamide-gel electrophoresis. Biochem J. 1976 Nov;159(2):395–407. doi: 10.1042/bj1590395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheung H. S., Cushman D. W. A soluble aspartate aminopeptidase from dog kidney. Biochim Biophys Acta. 1971 Jul 21;242(1):190–193. doi: 10.1016/0005-2744(71)90098-2. [DOI] [PubMed] [Google Scholar]
- Chulkova T. M., Orekhovich V. N. Vydelenie i svoistva aminopeptidazy A iz pochek byka. Biokhimiia. 1978 Jul;43(7):1222–1227. [PubMed] [Google Scholar]
- DAVIS B. J. DISC ELECTROPHORESIS. II. METHOD AND APPLICATION TO HUMAN SERUM PROTEINS. Ann N Y Acad Sci. 1964 Dec 28;121:404–427. doi: 10.1111/j.1749-6632.1964.tb14213.x. [DOI] [PubMed] [Google Scholar]
- Danielsen E. M., Sjöström H., Norén O., Dabelsteen E. Immunoelectrophoretic studies on pig intestinal brush border proteins. Biochim Biophys Acta. 1977 Oct 26;494(2):332–342. doi: 10.1016/0005-2795(77)90163-5. [DOI] [PubMed] [Google Scholar]
- GLENNER G. G., FOLK J. E. Glutamyl peptidases in rat and guinea pig kidney slices. Nature. 1961 Oct 28;192:338–340. doi: 10.1038/192338a0. [DOI] [PubMed] [Google Scholar]
- GLENNER G. G., McMILLAN P. J., FOLK J. E. A mammalian peptidase specific for the hydrolysis of N-terminal alpha-L-glutamyl and aspartyl residues. Nature. 1962 Jun 2;194:867–867. doi: 10.1038/194867a0. [DOI] [PubMed] [Google Scholar]
- George S. G., Kenny J. Studies on the enzymology of purified preparations of brush border from rabbit kidney. Biochem J. 1973 May;134(1):43–57. doi: 10.1042/bj1340043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Glossmann H., Neville D. M., Jr Glycoproteins of cell surfaces. A comparative study of three different cell surfaces of the rat. J Biol Chem. 1971 Oct 25;246(20):6339–6346. [PubMed] [Google Scholar]
- Hartley B. S. Strategy and tactics in protein chemistry. Biochem J. 1970 Oct;119(5):805–822. doi: 10.1042/bj1190805f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenny A. J., Booth A. G., George S. G., Ingram J., Kershaw D., Wood E. J., Young A. R. Dipeptidyl peptidase IV, a kidney brush-border serine peptidase. Biochem J. 1976 Jul 1;157(1):169–182. doi: 10.1042/bj1570169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenny A. J., Booth A. G. Microvilli: their ultrastructure, enzymology and molecular organization. Essays Biochem. 1978;14:1–44. [PubMed] [Google Scholar]
- Kessler M., Acuto O., Storelli C., Murer H., Müller M., Semenza G. A modified procedure for the rapid preparation of efficiently transporting vesicles from small intestinal brush border membranes. Their use in investigating some properties of D-glucose and choline transport systems. Biochim Biophys Acta. 1978 Jan 4;506(1):136–154. doi: 10.1016/0005-2736(78)90440-6. [DOI] [PubMed] [Google Scholar]
- Macnair D. C., Kenny A. J. Proteins of the kidney microvillar membrane. The amphipathic form of dipeptidyl peptidase IV. Biochem J. 1979 May 1;179(2):379–395. doi: 10.1042/bj1790379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maroux S., Louvard D., Baratti J. The aminopeptidase from hog intestinal brush border. Biochim Biophys Acta. 1973 Sep 15;321(1):282–295. doi: 10.1016/0005-2744(73)90083-1. [DOI] [PubMed] [Google Scholar]
- Nagatsu I., Nagatsu T., Yamamoto T., Glenner G. G., Mehl J. W. Purification of aminopeptidase A in human serum and degradation of angiotensin II by the purified enzyme. Biochim Biophys Acta. 1970 Feb 11;198(2):255–270. doi: 10.1016/0005-2744(70)90058-6. [DOI] [PubMed] [Google Scholar]
- Norén O., Sjöström H., Danielsen E. M., Staun M., Jeppesen L., Svensson B. Comparison of two pig intestinal brush border peptidases with the corresponding renal enzymes. Hoppe Seylers Z Physiol Chem. 1979 Feb;360(2):151–157. doi: 10.1515/bchm2.1979.360.1.151. [DOI] [PubMed] [Google Scholar]
- Norén O., Sjöström H. The insertion of pig microvillus aminopeptidase into the membrane as probed by [125I]iodonaphthylazide. Eur J Biochem. 1980 Feb;104(1):25–31. doi: 10.1111/j.1432-1033.1980.tb04395.x. [DOI] [PubMed] [Google Scholar]
- Sjöström H., Norén O., Jeppesen L., Staun M., Svensson B., Christiansen L. Purification of different amphiphilic forms of a microvillus aminopeptidase from pig small intestine using immunoadsorbent chromatography. Eur J Biochem. 1978 Aug 1;88(2):503–511. doi: 10.1111/j.1432-1033.1978.tb12476.x. [DOI] [PubMed] [Google Scholar]
- Vannier C., Louvard D., Maroux S., Desnuelle P. Structural and topological homology between porcine intestinal and renal brush border aminopeptidase. Biochim Biophys Acta. 1976 Nov 11;455(1):185–199. doi: 10.1016/0005-2736(76)90163-2. [DOI] [PubMed] [Google Scholar]
- Wacker H., Lehky P., Vanderhaeghe F., Stein E. A. On the subunit structure of particulate aminopeptidase from pig kidney. Biochim Biophys Acta. 1976 Apr 8;429(2):546–554. doi: 10.1016/0005-2744(76)90302-8. [DOI] [PubMed] [Google Scholar]