Abstract
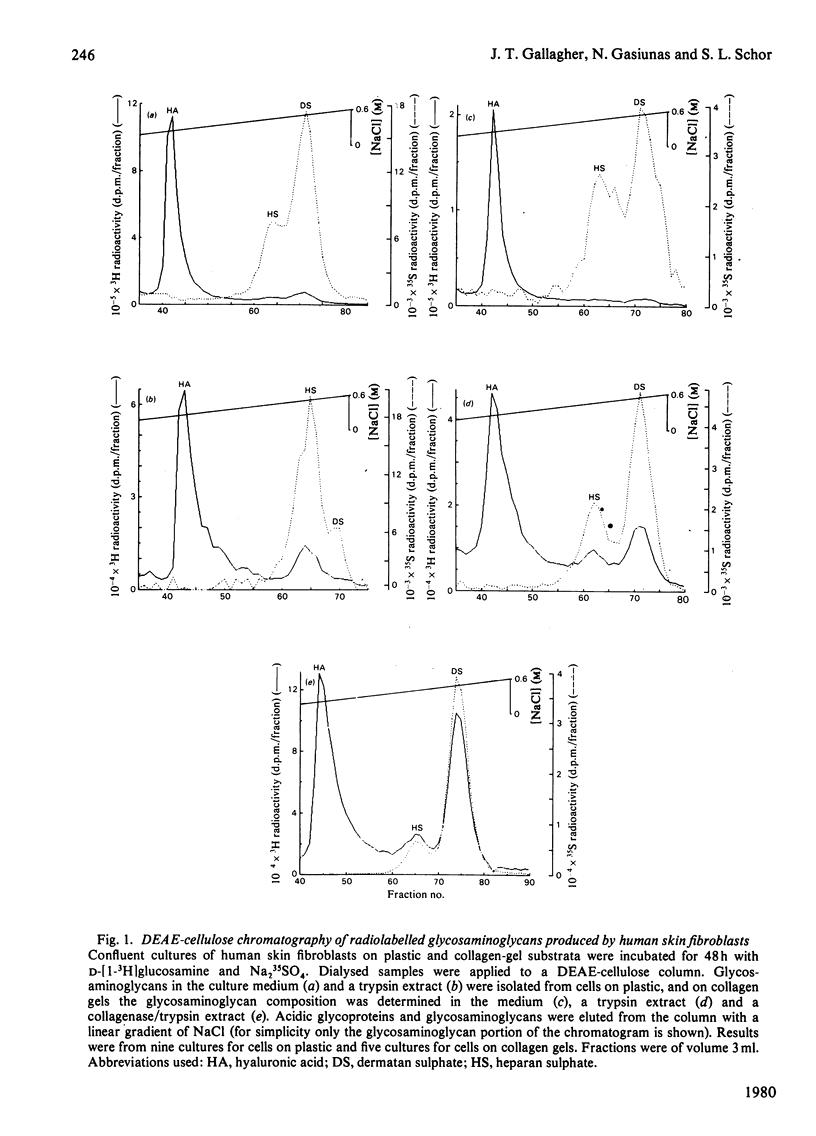
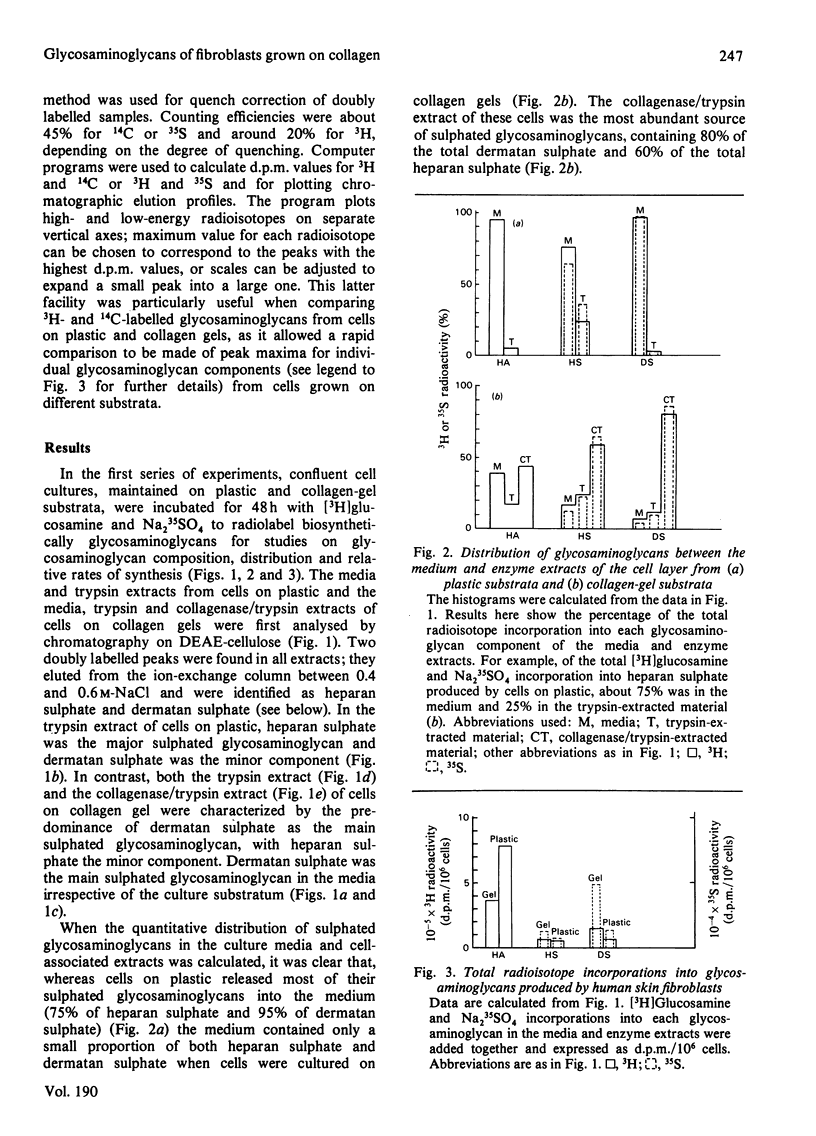
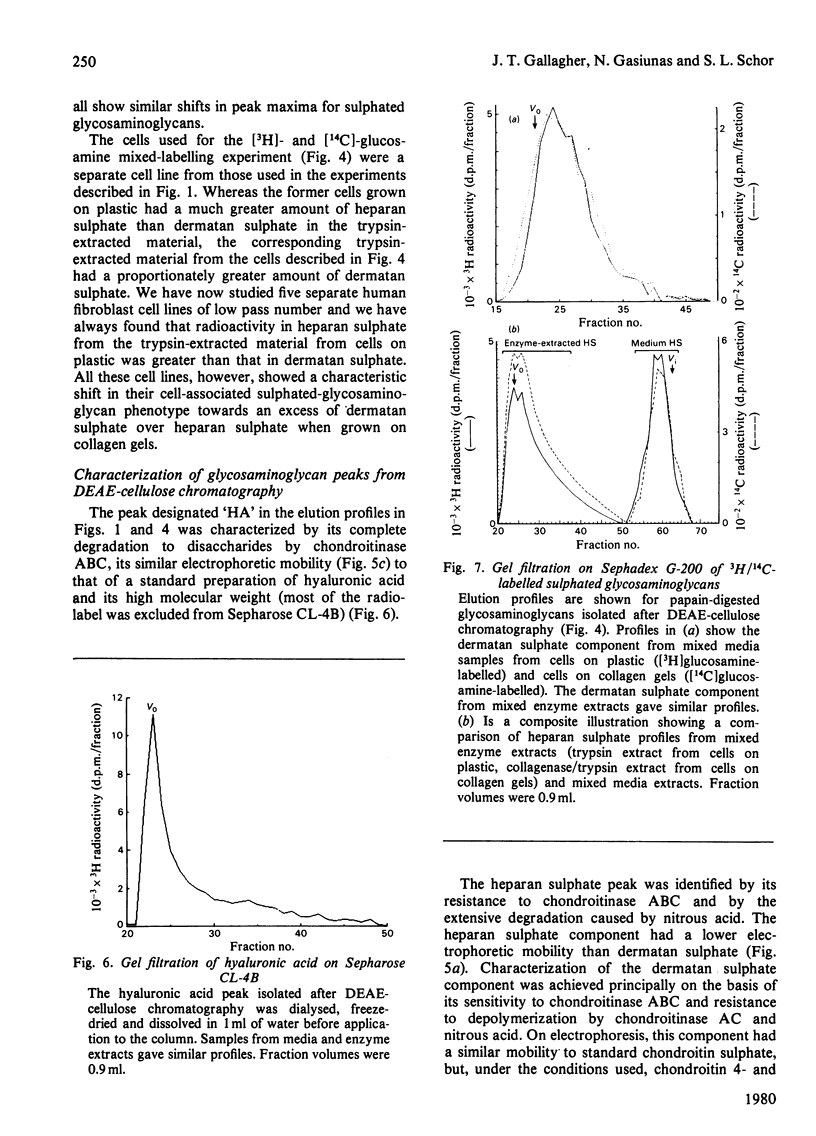
A comparison has been made of the synthesis of glycosaminoglycans by human skin fibroblasts cultured on plastic or collagen gel substrata. Confluent cultures were incubated with [3H]glucosamine and Na235SO4 for 48h. Radiolabelled glycosaminoglycans were then analysed in the spent media and trypsin extracts from cells on plastic and in the medium, trypsin and collagenase extracts from cells on collagen gels. All enzyme extracts and spent media contained hyaluronic acid, heparan sulphate and dermatan sulphate. Hyaluronic acid was the main 3H-labelled component in media and enzyme extracts from cells on both substrata, although it was distributed mainly to the media fractions. Heparan sulphate was the major [35S]sulphated glycosaminoglycan in trypsin extracts of cells on plastic, and dermatan sulphate was the minor component. In contrast, dermatan sulphate was the principal [35S]sulphated glycosaminoglycan in trypsin and collagenase extracts of cells on collagen gels. The culture substratum also influenced the amounts of [35S]sulphated glycosaminoglycans in media and enzyme extracts. With cells on plastic, the medium contained most of the heparan sulphate (75%) and dermatan sulphate (> 90%), whereas the collagenase extract was the main source of heparan sulphate (60%) and dermatan sulphate (80%) from cells on collagen gels; when cells were grown on collagen, the medium contained only 5-20% of the total [35S]sulphated glycosaminoglycans. Depletion of the medium pool was probably caused by binding of [35S]sulphated glycosaminoglycans to the network of native collagen fibres that formed the insoluble fraction of the collagen gel. Furthermore, cells on collagen showed a 3-fold increase in dermatan sulphate synthesis, which could be due to a positive-feedback mechanism activated by the accumulation of dermatan sulphate in the microenvironment of the cultured cells. For comparative structural analyses of glycosaminoglycans synthesized on different substrata labelling experiments were carried out by incubating cells on plastic with [3H]glucosamine, and cells on collagen gels with [14C]glucosamine. Co-chromatography on DEAE-cellulose of mixed media and enzyme extracts showed that heparan sulphate from cells on collagen gels eluted at a lower salt concentration than did heparan sulphate from cells on plastic, whereas with dermatan sulphate the opposite result was obtained, with dermatan sulphate from cells on collagen eluting at a higher salt concentration than dermatan sulphate from cells on plastic. These differences did not correspond to changes in the molecular size of the glycosaminoglycan chains, but they may be caused by alterations in polymer sulphation.
Full text
PDF








Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- David G., Bernfield M. R. Collagen reduces glycosaminoglycan degradation by cultured mammary epithelial cells: possible mechanism for basal lamina formation. Proc Natl Acad Sci U S A. 1979 Feb;76(2):786–790. doi: 10.1073/pnas.76.2.786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elsdale T., Bard J. Collagen substrata for studies on cell behavior. J Cell Biol. 1972 Sep;54(3):626–637. doi: 10.1083/jcb.54.3.626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fessler J. H., Fessler L. I. Biosynthesis of procollagen. Annu Rev Biochem. 1978;47:129–162. doi: 10.1146/annurev.bi.47.070178.001021. [DOI] [PubMed] [Google Scholar]
- Gallagher J. T. Concepts of metabolic pools in the metabolism of proteoglycans and hyaluronic acid. Biochem Soc Trans. 1977;5(2):402–410. doi: 10.1042/bst0050402. [DOI] [PubMed] [Google Scholar]
- Greenwald R. A., Schwartz C. E., Cantor J. O. Interaction of cartilage proteoglycans with collagen-substituted agarose gels. Biochem J. 1975 Mar;145(3):601–605. doi: 10.1042/bj1450601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hay E. D. Interaction between the cell surface and extracellular matrix in corneal development. Soc Gen Physiol Ser. 1977;32:115–137. [PubMed] [Google Scholar]
- Hopwood J. J., Dorfman A. Glycosaminoglycan synthesis by cultured human skin fibroblasts after transformation with simian virus 40. J Biol Chem. 1977 Jul 25;252(14):4777–4785. [PubMed] [Google Scholar]
- Huang D. Effect of extracellular chondroitin sulfate on cultured chondrocytes. J Cell Biol. 1974 Sep;62(3):881–886. doi: 10.1083/jcb.62.3.881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kleinman H. K., Silbert J. E., Silbert C. K. Heparan sulfate of skin fibroblasts grown in culture. Connect Tissue Res. 1975;4(1):17–23. doi: 10.3109/03008207509152193. [DOI] [PubMed] [Google Scholar]
- Kosher R. A., Church R. L. Stimulation of in vitro somite chondrogenesis by procollagen and collagen. Nature. 1975 Nov 27;258(5533):327–330. doi: 10.1038/258327a0. [DOI] [PubMed] [Google Scholar]
- LAGUNOFF D., WARREN G. Determination of 2-deoxy-2-sulfoaminohexose content of mucopolysaccharides. Arch Biochem Biophys. 1962 Dec;99:396–400. doi: 10.1016/0003-9861(62)90285-0. [DOI] [PubMed] [Google Scholar]
- Lindahl U., Hök M. Glycosaminoglycans and their binding to biological macromolecules. Annu Rev Biochem. 1978;47:385–417. doi: 10.1146/annurev.bi.47.070178.002125. [DOI] [PubMed] [Google Scholar]
- Malström A., Carlstedt I., Aberg L., Fransson L. A. The copolymeric structure of dermatan sulphate produced by cultured human fibroblasts. Different distribution of iduronic acid and glucuronic acid-containing units in soluble and cell-associated glycans. Biochem J. 1975 Dec;151(3):477–489. doi: 10.1042/bj1510477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matalon R., Dorfman A. Hurler's syndrome: biosynthesis of acid mucopolysaccharides in tissue culture. Proc Natl Acad Sci U S A. 1966 Oct;56(4):1310–1316. doi: 10.1073/pnas.56.4.1310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meier L., Hay E. D. Stimulation of corneal differentiation by interaction between cell surface and extracellular matrix. I. Morphometric analysis of transfilter "induction". J Cell Biol. 1975 Aug;66(2):275–291. doi: 10.1083/jcb.66.2.275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meier S., Hay E. D. Stimulation of extracellular matrix synthesis in the developing cornea by glycosaminoglycans. Proc Natl Acad Sci U S A. 1974 Jun;71(6):2310–2313. doi: 10.1073/pnas.71.6.2310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nevo Z., Dorfman A. Stimulation of chondromucoprotein synthesis in chondrocytes by extracellular chondromucoprotein. Proc Natl Acad Sci U S A. 1972 Aug;69(8):2069–2072. doi: 10.1073/pnas.69.8.2069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obrink B. A study of the interactions between monomeric tropocollagen and glycosaminoglycans. Eur J Biochem. 1973 Mar 1;33(2):387–400. doi: 10.1111/j.1432-1033.1973.tb02695.x. [DOI] [PubMed] [Google Scholar]
- Obrink B., Wasteson A. Nature of the interaction of chondroitin 4-sulphate and chondroitin sulphate-proteoglycan with collagen. Biochem J. 1971 Jan;121(2):227–233. doi: 10.1042/bj1210227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prinz R., Schwermann J., Buddecke E., von Figura K. Endocytosis of sulphated proteoglycans by cultured skin fibroblasts. Biochem J. 1978 Dec 15;176(3):671–676. doi: 10.1042/bj1760671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito H., Yamagata T., Suzuki S. Enzymatic methods for the determination of small quantities of isomeric chondroitin sulfates. J Biol Chem. 1968 Apr 10;243(7):1536–1542. [PubMed] [Google Scholar]
- Schor S. L., Court J. Different mechanisms in the attachment of cells to native and denatured collagen. J Cell Sci. 1979 Aug;38:267–281. doi: 10.1242/jcs.38.1.267. [DOI] [PubMed] [Google Scholar]
- Sjöberg I., Carlstedt I., Cöster L., Malmström A., Fransson L. A. Structure and metabolism of sulphated glycosaminoglycans in cultures of human fibroblasts. Structural characteristics of co-polymeric galactosaminoglycans in sequential extracts of fibroblasts during pulse-chase experiments. Biochem J. 1979 Feb 15;178(2):257–270. doi: 10.1042/bj1780257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toledo O. M., Dietrich C. P. Tissue specific distribution of sulfated mucopolysaccharides in mammals. Biochim Biophys Acta. 1977 Jun 23;498(1):114–122. doi: 10.1016/0304-4165(77)90092-7. [DOI] [PubMed] [Google Scholar]
- Toole B. P. Binding and precipitation of soluble collagens by chick embryo cartilage proteoglycan. J Biol Chem. 1976 Feb 10;251(3):895–897. [PubMed] [Google Scholar]
- Underhill C. B., Keller J. M. A transformation-dependent difference in the heparan sulfate associated with the cell surface. Biochem Biophys Res Commun. 1975 Mar 17;63(2):448–454. doi: 10.1016/0006-291x(75)90708-1. [DOI] [PubMed] [Google Scholar]
- Underhill C. B., Keller J. M. Heparan sulfates of mouse cells. Analysis of parent and transformed 3T3 cell lines. J Cell Physiol. 1977 Jan;90(1):53–59. doi: 10.1002/jcp.1040900108. [DOI] [PubMed] [Google Scholar]
- Winterbourne D. J., Mora P. T. Altered metabolism of heparan sulfate in simian virus 40 transformed cloned mouse cells. J Biol Chem. 1978 Jul 25;253(14):5109–5120. [PubMed] [Google Scholar]


