Abstract
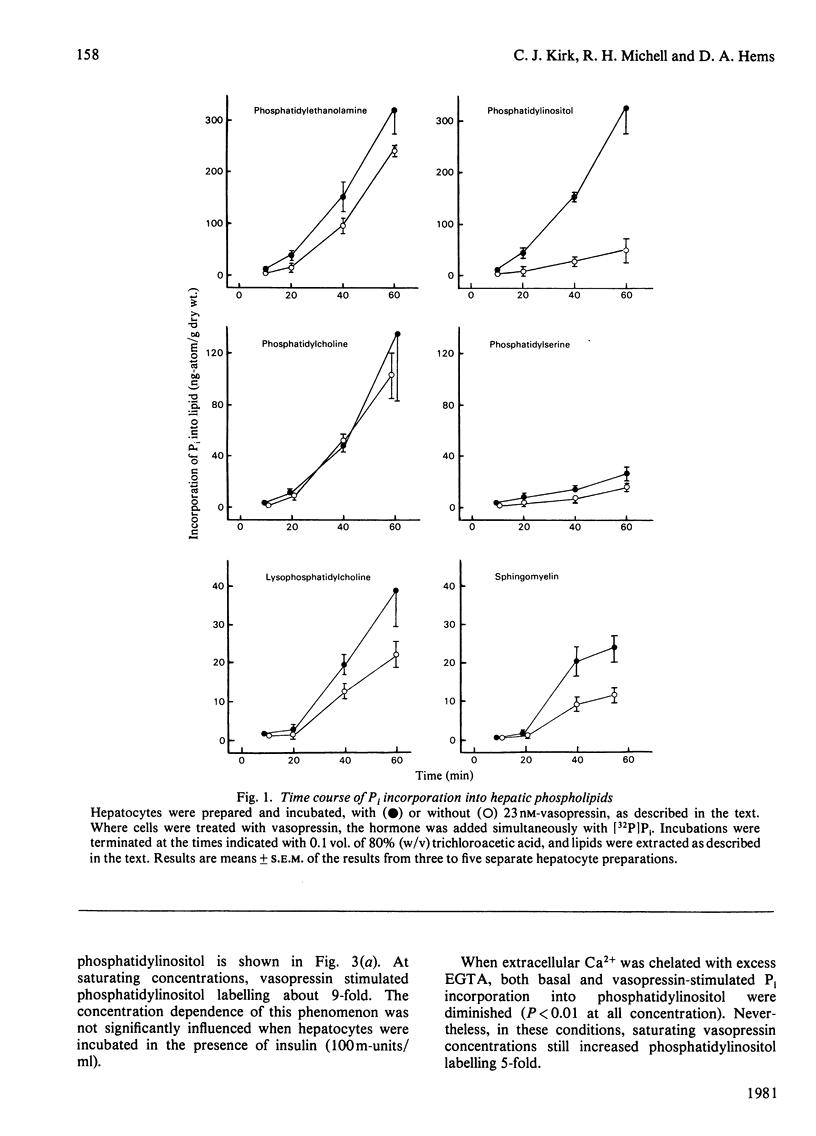
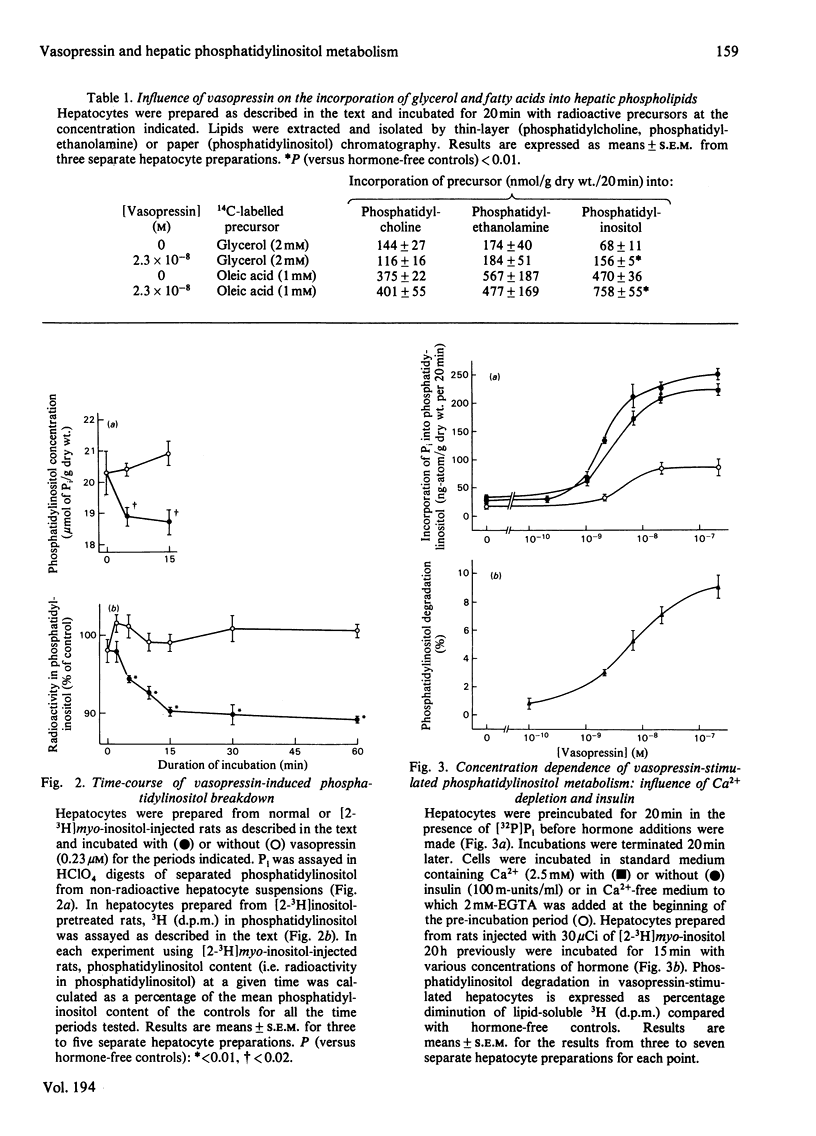
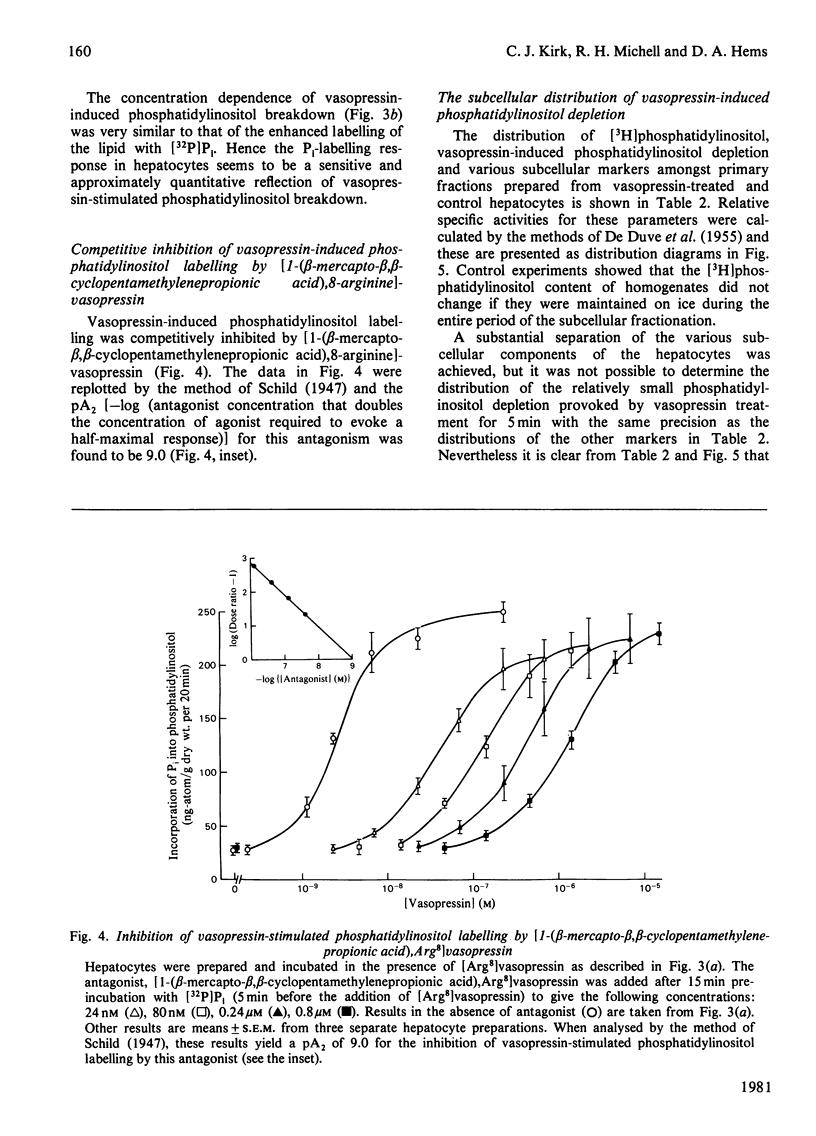
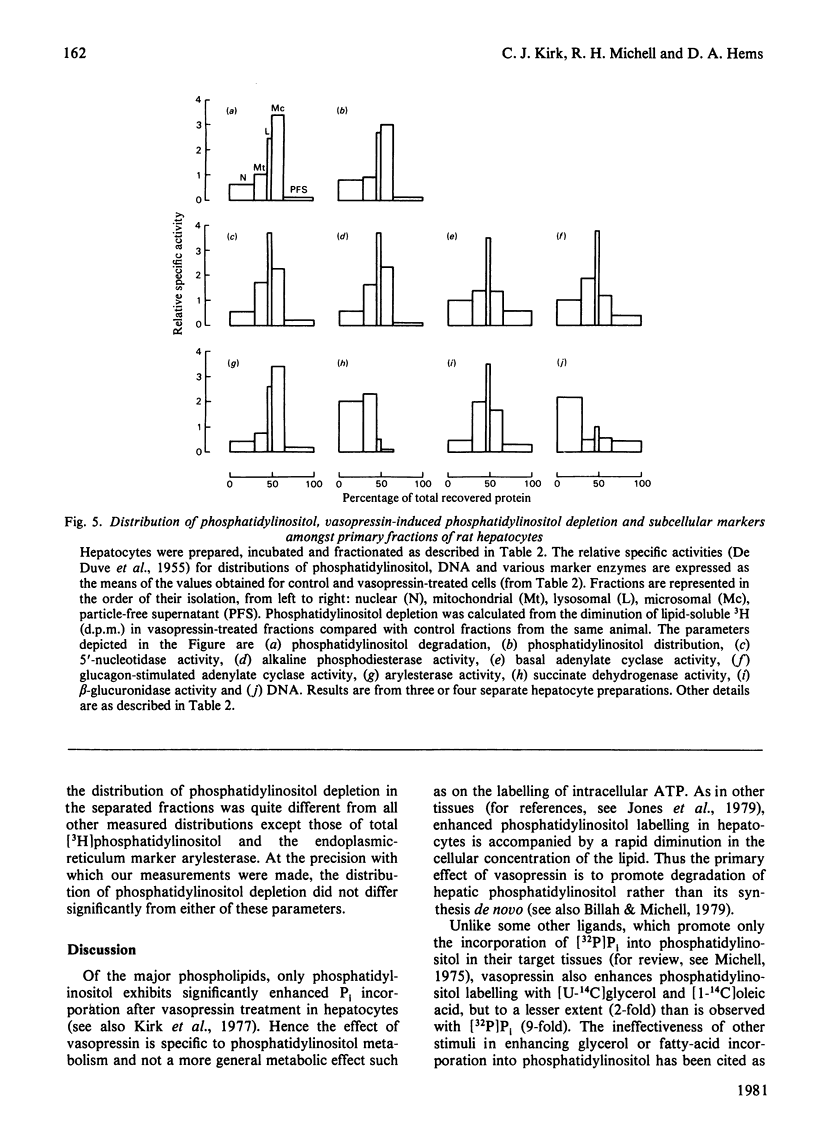
In isolated rat hepatocytes, vasopressin evoked a large increase in the incorporation of [32P]Pi into phosphatidylinositol, accompanied by smaller increases in the incorporation of [1-14C]oleate and [U-14C]glycerol. Incorporation of these precursors into the other major phospholipids was unchanged during vasopressin treatment. Vasopressin also promoted phosphatidylinositol breakdown in hepatocytes. Half-maximum effects on phosphatidylinositol breakdown and on phosphatidylinositol labelling occurred at about 5 nM-vasopressin, a concentration at which approximately half of the hepatic vasopressin receptors are occupied but which is much greater than is needed to produce half-maximal activation of glycogen phosphorylase. Insulin did not change the incorporation of [32P]Pi into the phospholipids of hepatocytes and it had no effect on the response to vasopressin. Although the incorporation of [32P]Pi into hepatocyte lipids was decreased when cells were incubated in a Ca2+-free medium, vasopressin still provoked a substantial stimulation of phosphatidylinositol labelling under these conditions. Studies with the antagonist [1-(beta-mercapto-beta, beta-cyclopentamethylenepropionic acid),8-arginine]vasopressin indicated that the hepatic vasopressin receptors that control phosphatidylinositol metabolism are similar to those that mediate the vasopressor response in vivo. When prelabelled hepatocytes were stimulated for 5 min and then subjected to subcellular fractionation. The decrease in [3H]phosphatidylinositol content in each cell fraction with approximately in proportion to its original phosphatidylinositol content. This may be a consequence of phosphatidylinositol breakdown at a single site, followed by rapid phosphatidylinositol exchange between membranes leading to re-establishment of an equilibrium distribution.
Full text
PDF






Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Assimacopoulos-Jeannet F. D., Blackmore P. F., Exton J. H. Studies on alpha-adrenergic activation of hepatic glucose output. Studies on role of calcium in alpha-adrenergic activation of phosphorylase. J Biol Chem. 1977 Apr 25;252(8):2662–2669. [PubMed] [Google Scholar]
- Baginski E. S., Foà P. P., Zak B. Microdetermination of inorganic phosphate, phospholipids, and total phosphate in biologic materials. Clin Chem. 1967 Apr;13(4):326–332. [PubMed] [Google Scholar]
- Bell R. L., Majerus P. W. Thrombin-induced hydrolysis of phosphatidylinositol in human platelets. J Biol Chem. 1980 Mar 10;255(5):1790–1792. [PubMed] [Google Scholar]
- Billah M. M., Michell R. H. Phosphatidylinositol metabolism in rat hepatocytes stimulated by glycogenolytic hormones. Effects of angiotensin, vasopressin, adrenaline, ionophore A23187 and calcium-ion deprivation. Biochem J. 1979 Sep 15;182(3):661–668. doi: 10.1042/bj1820661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Billah M. M., Michell R. H. Stimulation of the breakdown and resynthesis of of phosphatidylinositol in rat hepatocytes by angiotensin, vasopressin and adrenaline. Biochem Soc Trans. 1978;6(5):1033–1035. doi: 10.1042/bst0061033. [DOI] [PubMed] [Google Scholar]
- Blackmore P. F., Brumley F. T., Marks J. L., Exton J. H. Studies on alpha-adrenergic activation of hepatic glucose output. Relationship between alpha-adrenergic stimulation of calcium efflux and activation of phosphorylase in isolated rat liver parenchymal cells. J Biol Chem. 1978 Jul 25;253(14):4851–4858. [PubMed] [Google Scholar]
- Calderon P., Furnelle J., Christophe J. In vitro lipid metabolism in the rat pancreas. III. Effects of carbamylcholine and pancreozymin on the turnover of phosphatidylinositols, 1,2-diacylglycerols and phosphatidylcholines. Biochim Biophys Acta. 1979 Sep 28;574(3):404–422. doi: 10.1016/0005-2760(79)90236-4. [DOI] [PubMed] [Google Scholar]
- Chen J. L., Babcock D. F., Lardy H. A. Norepinephrine, vasopressin, glucagon, and A23187 induce efflux of calcium from an exchangeable pool in isolated rat hepatocytes. Proc Natl Acad Sci U S A. 1978 May;75(5):2234–2238. doi: 10.1073/pnas.75.5.2234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cockcroft S., Bennett J. P., Gomperts B. D. f-MetLeuPhe-induced phosphatidylinositol turnover in rabbit neutrophils is dependent on extracellular calcium. FEBS Lett. 1980 Jan 28;110(1):115–118. doi: 10.1016/0014-5793(80)80036-6. [DOI] [PubMed] [Google Scholar]
- DE DUVE C., PRESSMAN B. C., GIANETTO R., WATTIAUX R., APPELMANS F. Tissue fractionation studies. 6. Intracellular distribution patterns of enzymes in rat-liver tissue. Biochem J. 1955 Aug;60(4):604–617. doi: 10.1042/bj0600604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GETZ G. S., BARTLEY W., STIRPE F., NOTTON B. M., RENSHAW A. The lipid composition of rat-liver mitochondria, fluffy layer and microsomes. Biochem J. 1962 Apr;83:181–191. doi: 10.1042/bj0830181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garrison J. C., Borland M. K., Florio V. A., Twible D. A. The role of calcium ion as a mediator of the effects of angiotensin II, catecholamines, and vasopressin on the phosphorylation and activity of enzymes in isolated hepatocytes. J Biol Chem. 1979 Aug 10;254(15):7147–7156. [PubMed] [Google Scholar]
- Getz G. S., Bartley W., Lurie D., Notton B. M. The phospholipids of various sheep organs, rat liver and of their subcellular fractions. Biochim Biophys Acta. 1968 Mar 4;152(2):325–339. doi: 10.1016/0005-2760(68)90040-4. [DOI] [PubMed] [Google Scholar]
- Groener J. E., van Golde L. M. Effect of fasting and feeding a high-sucrose, fat-free diet on the synthesis of hepatic glycerolipids in vivo and in isolated hepatocytes. Biochim Biophys Acta. 1977 Apr 26;487(1):105–114. doi: 10.1016/0005-2760(77)90047-9. [DOI] [PubMed] [Google Scholar]
- Hems D. A., Davies C. J., Siddle K. Effect of hormones on content of purine nucleoside cyclic monophosphates in perfused rat liver. FEBS Lett. 1978 Mar 15;87(2):196–198. doi: 10.1016/0014-5793(78)80330-5. [DOI] [PubMed] [Google Scholar]
- Hems D. A., Rodrigues L. M., Whitton P. D. Rapid stimulation by vasopressin, oxytocin and angiotensin II of glycogen degradation in hepatocyte suspensions. Biochem J. 1978 May 15;172(2):311–317. doi: 10.1042/bj1720311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hems D. A., Whitton P. D. Stimulation by vasopressin of glycogen breakdown and gluconeogenesis in the perfused rat liver. Biochem J. 1973 Nov;136(3):705–709. doi: 10.1042/bj1360705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houslay M. D., Metcalfe J. C., Warren G. B., Hesketh T. R., Smith G. A. The glucagon receptor of rat liver plasma membrane can couple to adenylate cyclase without activating it. Biochim Biophys Acta. 1976 Jun 17;436(2):489–494. doi: 10.1016/0005-2736(76)90210-8. [DOI] [PubMed] [Google Scholar]
- Jones L. M., Cockcroft S., Michell R. H. Stimulation of phosphatidylinositol turnover in various tissues by cholinergic and adrenergic agonists, by histamine and by caerulein. Biochem J. 1979 Sep 15;182(3):669–676. doi: 10.1042/bj1820669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones L. M., Michell R. H. Breakdown of phosphatidylinositol provoked by muscarinic cholinergic stimulation of rat parotid-gland fragments. Biochem J. 1974 Sep;142(3):583–590. doi: 10.1042/bj1420583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keppens S., Vandenheede J. R., De Wulf H. On the role of calcium as second messenger in liver for the hormonally induced activation of glycogen phosphorylase. Biochim Biophys Acta. 1977 Feb 28;496(2):448–457. doi: 10.1016/0304-4165(77)90327-0. [DOI] [PubMed] [Google Scholar]
- Keppens S., de Wulf H. The nature of the hepatic receptors involved in vasopressin-induced glycogenolysis. Biochim Biophys Acta. 1979 Nov 15;588(1):63–69. doi: 10.1016/0304-4165(79)90371-4. [DOI] [PubMed] [Google Scholar]
- Kirk C. J., Hems D. A. Hepatic action of vasopressin: lack of a role for adenosine-3',5'-cyclic monophosphate. FEBS Lett. 1974 Oct 1;47(1):128–131. doi: 10.1016/0014-5793(74)80441-2. [DOI] [PubMed] [Google Scholar]
- Kirk C. J., Hems D. A. The control by vasopressin of carbohydrate and lipid metabolism in the perfused rat liver. Biochim Biophys Acta. 1979 Apr 3;583(4):474–482. doi: 10.1016/0304-4165(79)90064-3. [DOI] [PubMed] [Google Scholar]
- Kirk C. J., Rodrigues L. M., Hems D. A. The influence of vasopressin and related peptides on glycogen phosphorylase activity and phosphatidylinositol metabolism in hepatocytes. Biochem J. 1979 Feb 15;178(2):493–496. doi: 10.1042/bj1780493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kirk C. J., Verrinder T. R., Hems D. A. Rapid stimulation, by vasopressin and adrenaline, of inorganic phosphate incorporation into phosphatidyl inositol in isolated hepatocytes. FEBS Lett. 1977 Nov 15;83(2):267–271. doi: 10.1016/0014-5793(77)81020-x. [DOI] [PubMed] [Google Scholar]
- Kirk C. J., Verrinder T. R., Hems D. A. The influence of extracellular calcium concentration on the vasopressin-stimulated incorporation of inorganic phosphate into phosphatidylinositol in hepatocyte suspensions. Biochem Soc Trans. 1978;6(5):1031–1033. doi: 10.1042/bst0061031. [DOI] [PubMed] [Google Scholar]
- Kruszynski M., Lammek B., Manning M., Seto J., Haldar J., Sawyer W. H. [1-beta-Mercapto-beta,beta-cyclopentamethylenepropionic acid),2-(O-methyl)tyrosine ]argine-vasopressin and [1-beta-mercapto-beta,beta-cyclopentamethylenepropionic acid)]argine-vasopressine, two highly potent antagonists of the vasopressor response to arginine-vasopressin. J Med Chem. 1980 Apr;23(4):364–368. doi: 10.1021/jm00178a003. [DOI] [PubMed] [Google Scholar]
- Lapetina E. G., Cuatrecasas P. Stimulation of phosphatidic acid production in platelets precedes the formation of arachidonate and parallels the release of serotonin. Biochim Biophys Acta. 1979 May 25;573(2):394–402. doi: 10.1016/0005-2760(79)90072-9. [DOI] [PubMed] [Google Scholar]
- Lapetina E. G., Michell R. H. Stimulation by acetylcholine of phosphatidylinositol labelling. Subcellular distribution in rat cerebral-cortex slices. Biochem J. 1972 Mar;126(5):1141–1147. doi: 10.1042/bj1261141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lloyd-Davies K. A., Michell R. H., Coleman R. Glycerylphosphorylcholine phosphodiesterase in rat liver. Subcellular distribution and localization in plasma membranes. Biochem J. 1972 Apr;127(2):357–368. doi: 10.1042/bj1270357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ma G. Y., Hems D. A. Inhibition of fatty acid synthesis and stimulation of glycogen breakdown by vasopressin in the perfused mouse liver. Biochem J. 1975 Nov;152(2):389–392. doi: 10.1042/bj1520389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michell R. H., Hawthorne J. N. The site of diphosphoinositide synthesis in rat liver. Biochem Biophys Res Commun. 1965 Nov 22;21(4):333–338. doi: 10.1016/0006-291x(65)90198-1. [DOI] [PubMed] [Google Scholar]
- Michell R. H. Inositol phospholipids and cell surface receptor function. Biochim Biophys Acta. 1975 Mar 25;415(1):81–47. doi: 10.1016/0304-4157(75)90017-9. [DOI] [PubMed] [Google Scholar]
- Michell R. H., Jafferji S. S., Jones L. M. Receptor occupancy dose--response curve suggests that phosphatidyl-inositol breakdown may be intrinsic to the mechanism of the muscarinic cholinergic receptor. FEBS Lett. 1976 Oct 15;69(1):1–5. doi: 10.1016/0014-5793(76)80640-0. [DOI] [PubMed] [Google Scholar]
- Michell R. H., Kirk C. J., Billah M. M. Hormonal stimulation of phosphatidylinositol breakdown with particular reference to the hepatic effects of vasopressin. Biochem Soc Trans. 1979 Oct;7(5):861–865. doi: 10.1042/bst0070861. [DOI] [PubMed] [Google Scholar]
- Ontko J. A. Metabolism of free fatty acids in isolated liver cells. Factors affecting the partition between esterification and oxidation. J Biol Chem. 1972 Mar 25;247(6):1788–1800. [PubMed] [Google Scholar]
- Seglen P. O. Preparation of isolated rat liver cells. Methods Cell Biol. 1976;13:29–83. doi: 10.1016/s0091-679x(08)61797-5. [DOI] [PubMed] [Google Scholar]
- Shephard E. H., Hübscher G. Phosphatidate biosynthesis in mitochondrial subfractions of rat liver. Biochem J. 1969 Jun;113(2):429–440. doi: 10.1042/bj1130429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shohet S. B. Changes in fatty acid metabolism in human leukemic granulocytes during phagocytosis. J Lab Clin Med. 1970 Apr;75(4):659–672. [PubMed] [Google Scholar]
- Stubbs M., Kirk C. J., Hems D. A. Role of extracellular calcium in the action of vasopressin on hepatic glycogenolysis. FEBS Lett. 1976 Oct 15;69(1):199–202. doi: 10.1016/0014-5793(76)80686-2. [DOI] [PubMed] [Google Scholar]
- Takhar A. P., Kirk C. J. Stimulation of inorganic-phosphate incorporation into phosphatidylinositol in rat thoracic aorta mediated through V1-vasopressin receptors. Biochem J. 1981 Jan 15;194(1):167–172. doi: 10.1042/bj1940167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tolbert M. E., White A. C., Aspry K., Cutts J., Fain J. N. Stimulation by vasopressin and alpha-catecholamines of phosphatidylinositol formation in isolated rat liver parenchymal cells. J Biol Chem. 1980 Mar 10;255(5):1938–1944. [PubMed] [Google Scholar]
- Whitton P. D., Rodrigues L. M., Hems D. A. Influence of extracellular calcium ions on hormonal stimulation of glycogen breakdown in hepatocyte suspensions [proceedings]. Biochem Soc Trans. 1977;5(4):992–994. doi: 10.1042/bst0050992. [DOI] [PubMed] [Google Scholar]
- Williamson D. H., Ilic V., Tordoff A. F., Ellington E. V. Interactions between vasopressin and glucagon on ketogenesis and oleate metabolism in isolated hepatocytes from fed rats. Biochem J. 1980 Feb 15;186(2):621–624. doi: 10.1042/bj1860621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zborowski J. Transfer of phosphatidylinositol from microsomes to liposomes mediated by phospholipid transfer proteins: The inability of phosphatidylserine and sphingomyelin to replace phosphatidylcholine in stimulating this process. FEBS Lett. 1979 Nov 1;107(1):30–32. doi: 10.1016/0014-5793(79)80455-x. [DOI] [PubMed] [Google Scholar]
- Zborowski J., Wojtczak L. Net transfer of phosphatidylinositol from microsomes and mitochondria to liposomes catalyzed by the exchange protein from rat liver. FEBS Lett. 1975 Mar 1;51(1):317–320. doi: 10.1016/0014-5793(75)80916-1. [DOI] [PubMed] [Google Scholar]


