Abstract
Rat liver ornithine aminotransferase is found to exchange the pro-S hydrogen on the delta-carbon atom of ornithine exclusively, thus showing that the mammalian enzyme transfers the delta-amino group and not the alpha-amino group as has been demonstrated with the plant enzyme [Mestichelli, Gupta & Spenser (1979) J. Biol. Chem. 254, 640-647]. The enzyme also transfers the alpha-amino group of glutamate and the kinetics of the half reactions between the enzyme and both amino acids are compared. Rate and dissociation constants for both reactions are determined.
Full text
PDF
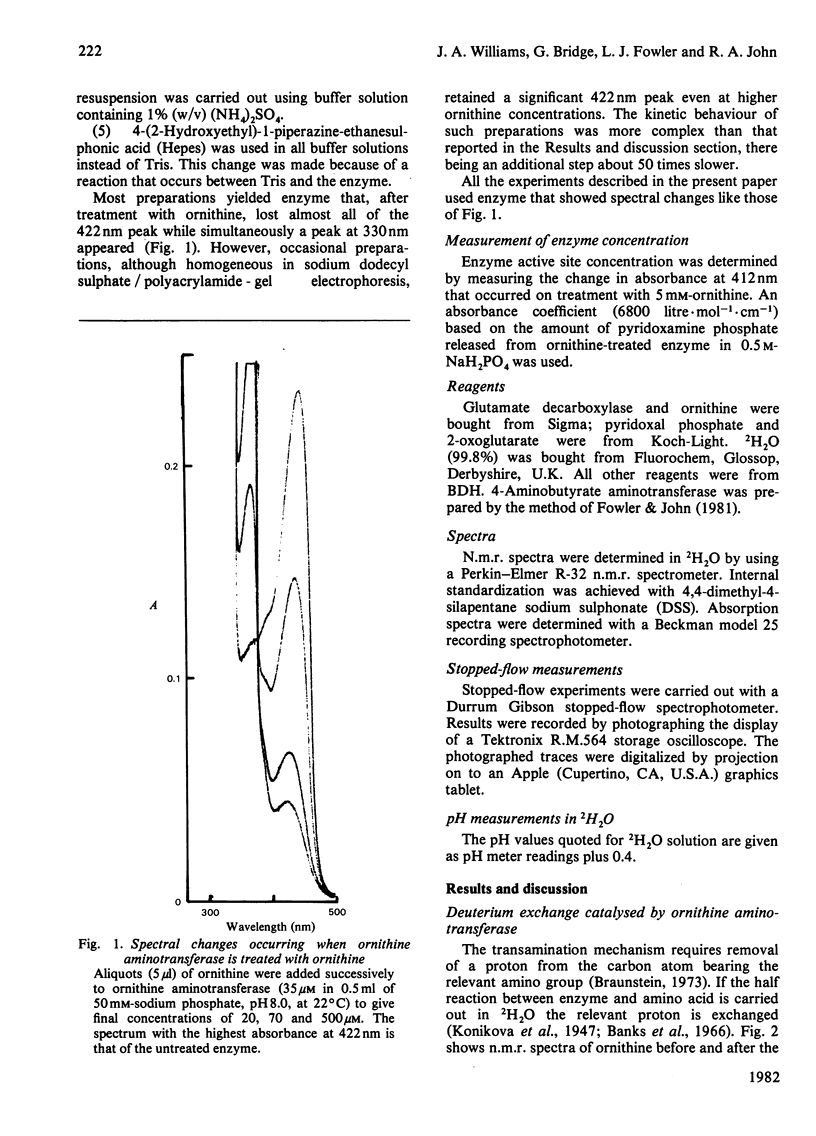
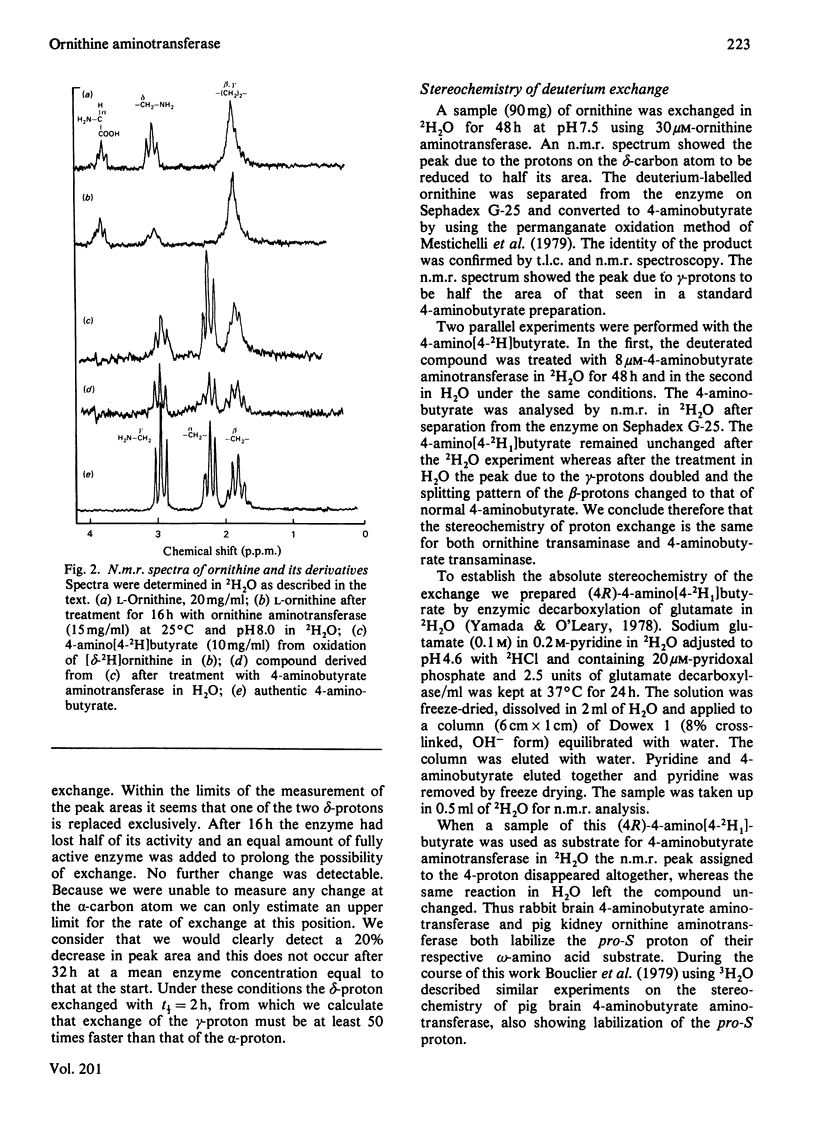
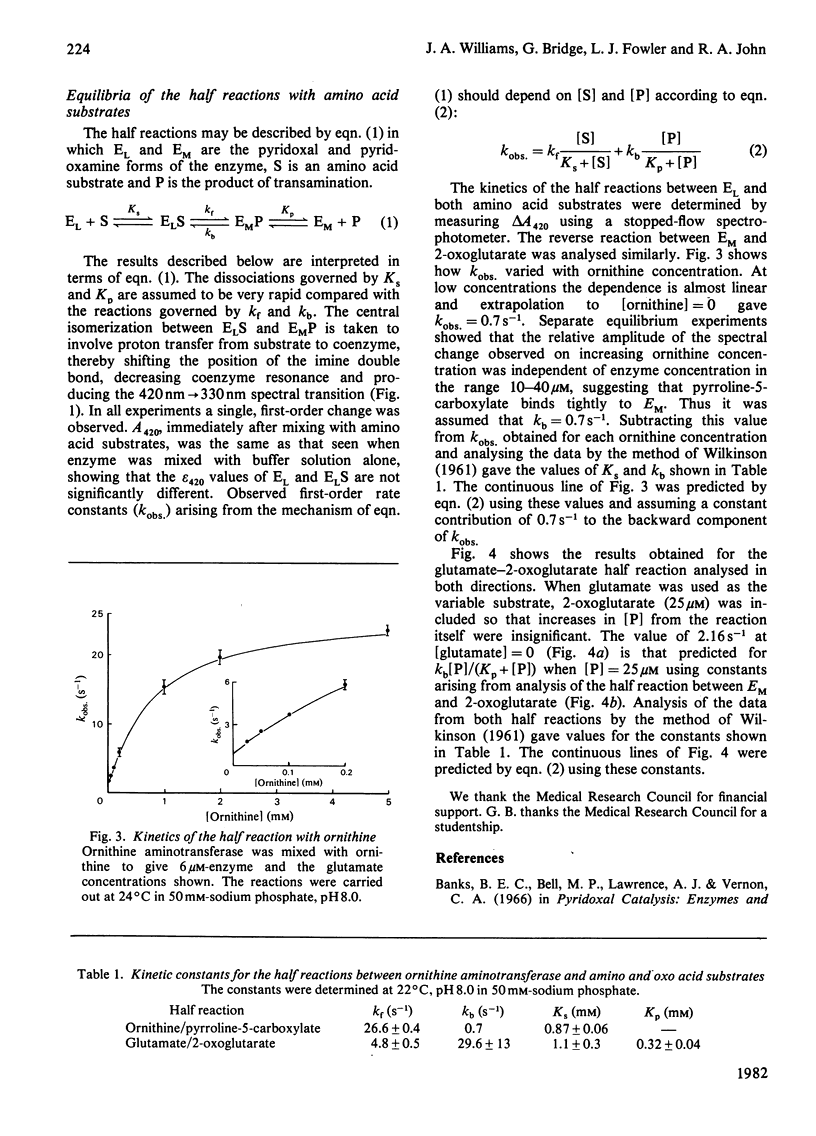
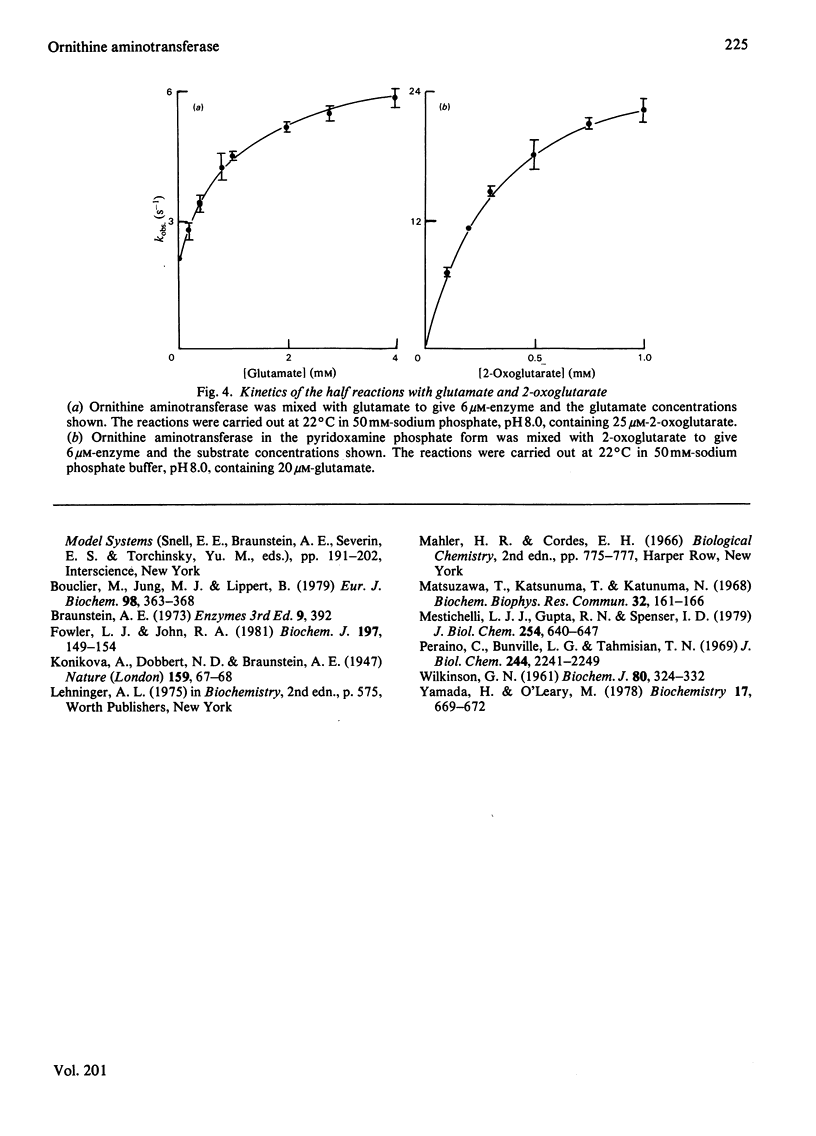
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bouclier M., Jung M. J., Lippert B. Stereochemistry of reactions catalysed by mammalian-brain L-glutamate 1-carboxy-lyase and 4-aminobutyrate: 2-oxoglutarate aminotransferase. Eur J Biochem. 1979 Aug 1;98(2):363–368. doi: 10.1111/j.1432-1033.1979.tb13195.x. [DOI] [PubMed] [Google Scholar]
- Fowler L. J., John R. A. The reaction of ethanolamine O-sulphate with 4-aminobutyrate aminotransferase. Biochem J. 1981 Jul 1;197(1):149–154. doi: 10.1042/bj1970149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuzawa T., Katsunuma T., Katunuma N. Crystallization of ornithine transaminase and its properties. Biochem Biophys Res Commun. 1968 Jul 26;32(2):161–166. doi: 10.1016/0006-291x(68)90363-x. [DOI] [PubMed] [Google Scholar]
- Mestichelli L. J., Gupta R. N., Spenser I. D. The biosynthetic route from ornithine to proline. J Biol Chem. 1979 Feb 10;254(3):640–647. [PubMed] [Google Scholar]
- Peraino C., Bunville L. G., Tahmisian T. N. Chemical, physical, and morphological properties of ornithine Aminotransferase from rat liver. J Biol Chem. 1969 May 10;244(9):2241–2249. [PubMed] [Google Scholar]
- WILKINSON G. N. Statistical estimations in enzyme kinetics. Biochem J. 1961 Aug;80:324–332. doi: 10.1042/bj0800324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamada H., O'Leary M. H. Stereochemistry of reactions catalyzed by glutamate decarboxylase. Biochemistry. 1978 Feb 21;17(4):669–672. doi: 10.1021/bi00597a017. [DOI] [PubMed] [Google Scholar]


