Abstract
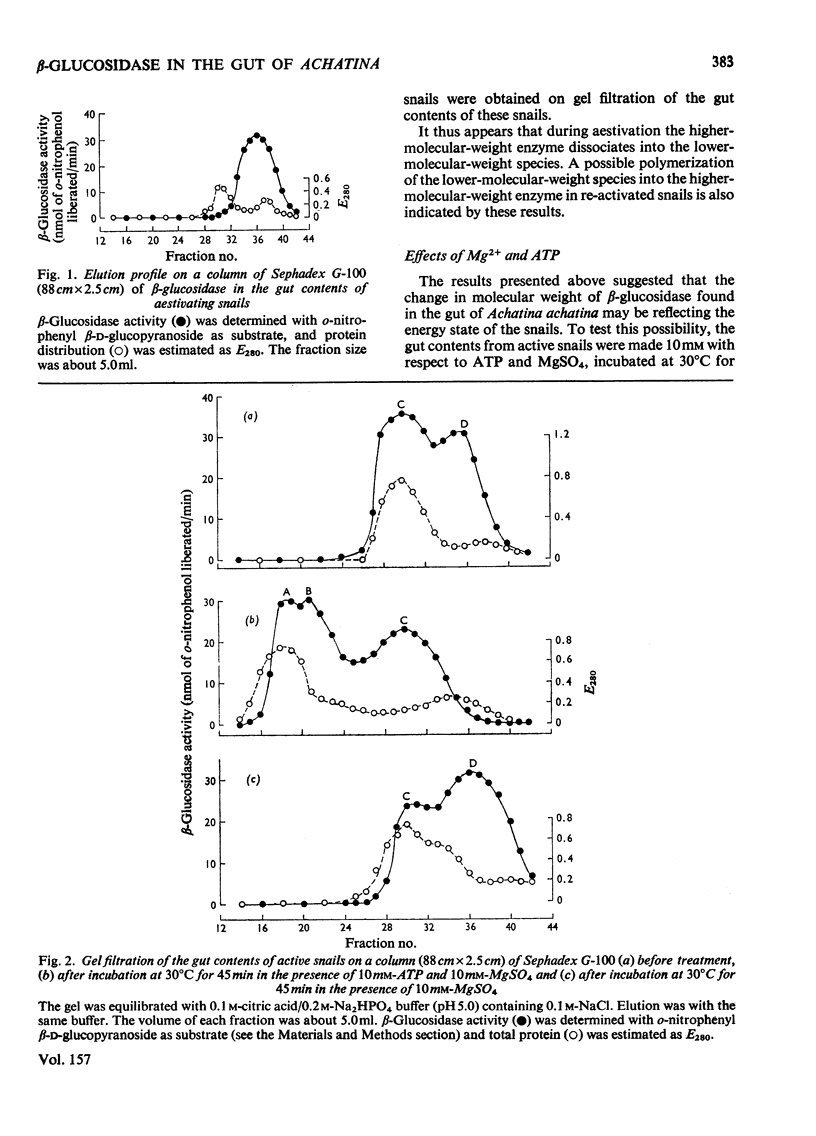
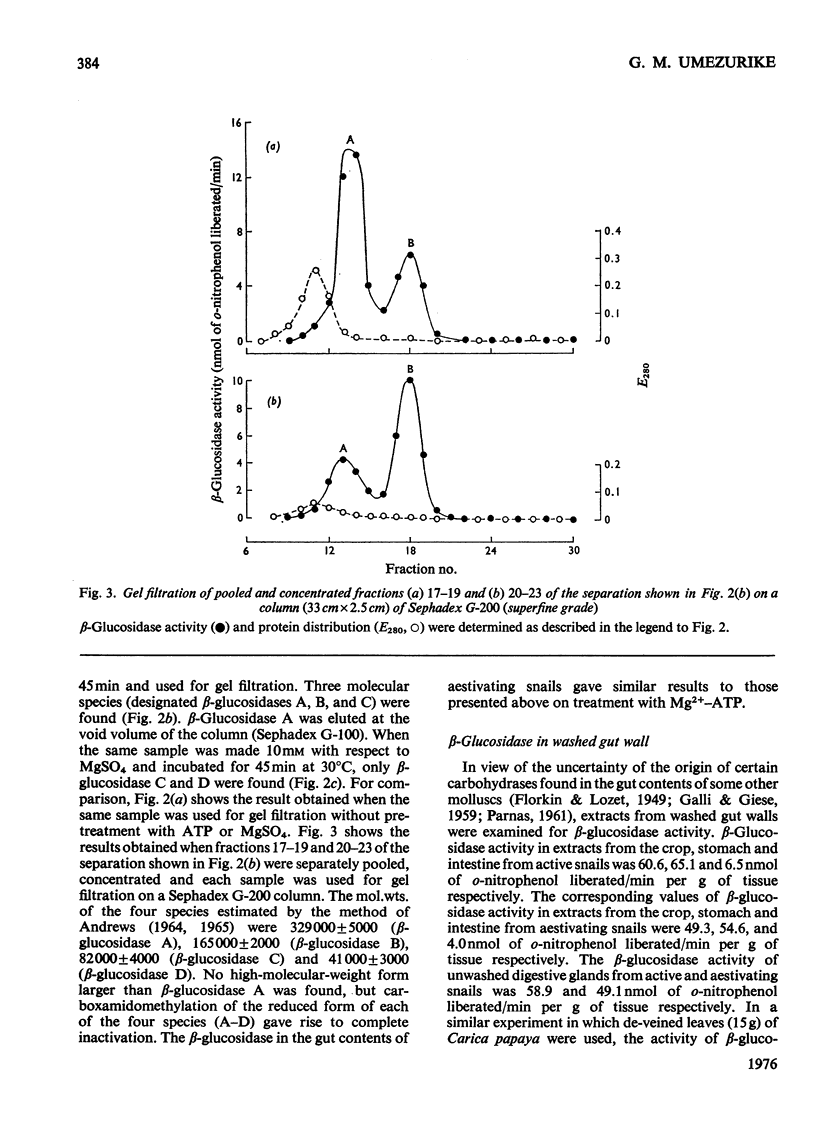
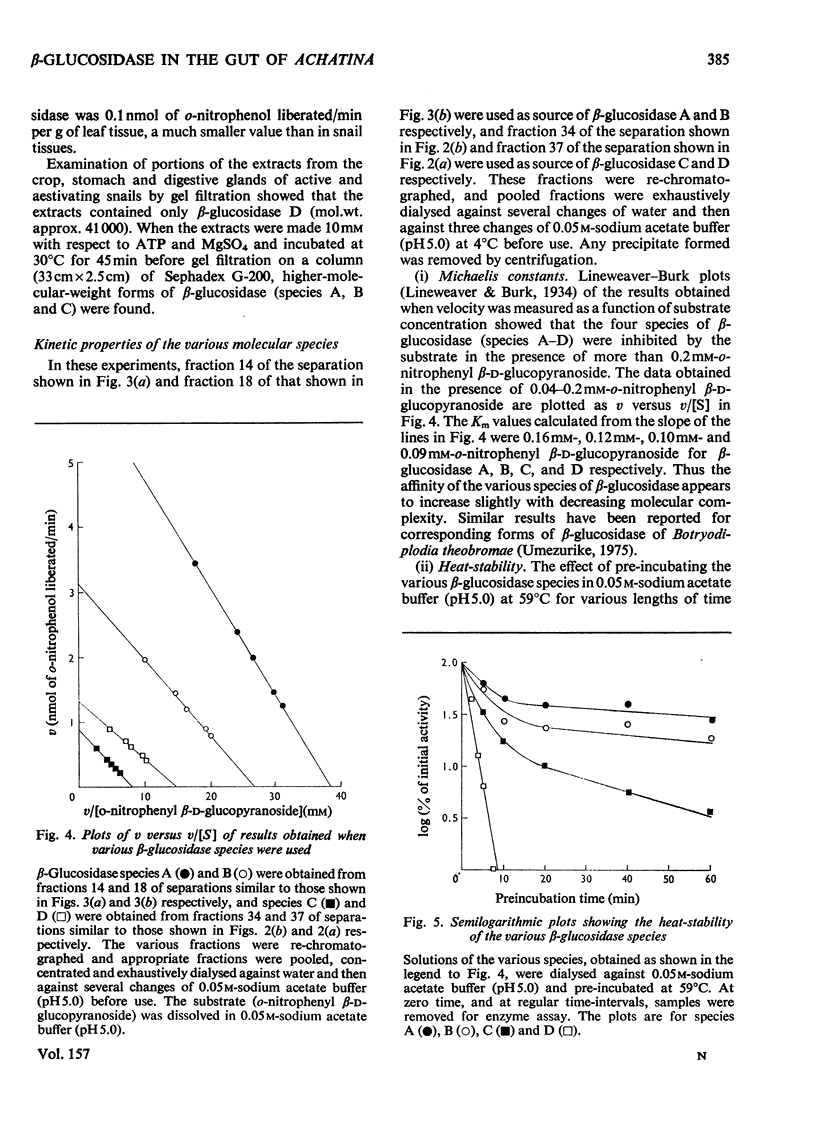
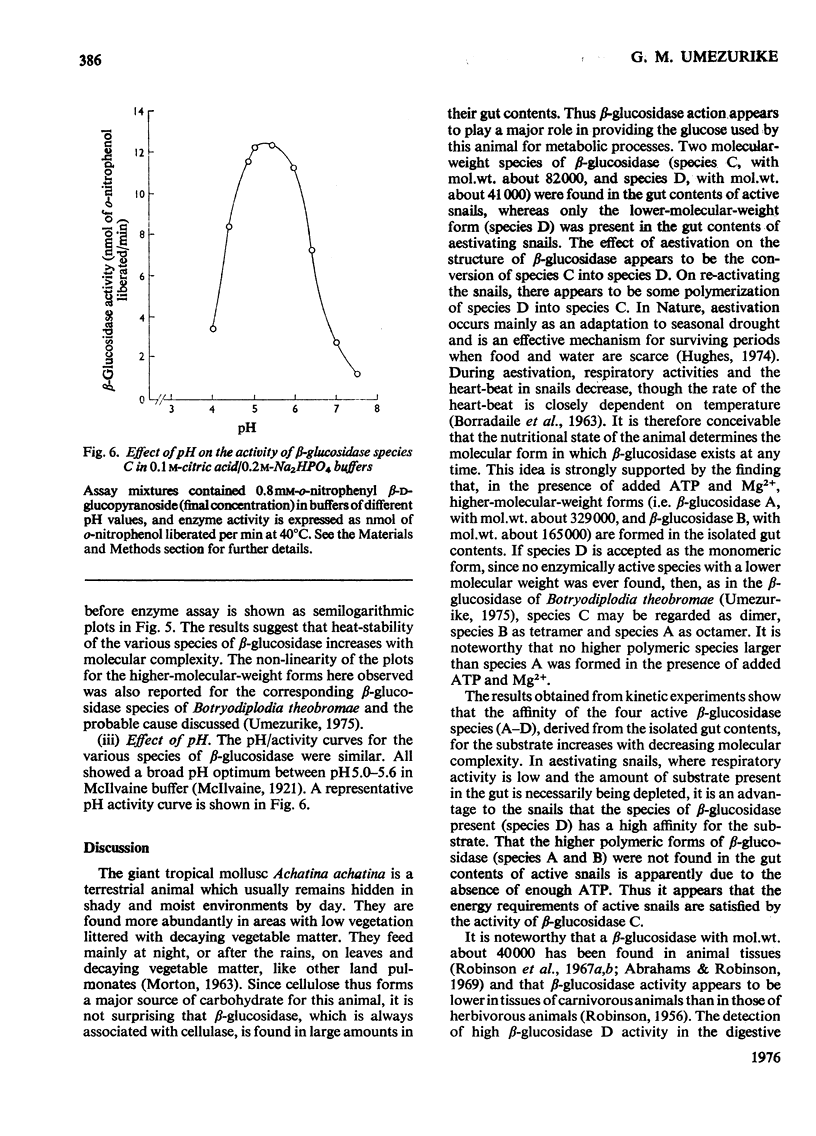
1. The enzyme beta-glucosidase (beta-D-glucoside glucohydrolase, EC 3.2.1.21) from the gut contents of active Achatina achatina exists in two molecular forms, beta-glucosidase C (mol.wt. about 82000) and D (mol.wt. about 41000). 2. Only the lower-molecular-weight species was found in the gut contents of aestivating snails or in extracts from their digestive glands and washed gut walls. 3. On re-activation of some aestivating snails, betion of ATP and Mg2+ to the isolated gut contents or to extracts from washed gut walls led to the formation of higher-molecular-weight forms of the enzyme, beta-glucosidase A (mol.wt. about 329000) and beta-glucosidase B (mol.wt. about 165000). 5. All these forms of the enzyme have similar pH optimum (pH 5.0-5.6). 6. The Michaelis constants (Km) and heat stability of the enzyme increased with increasing molecular complexity.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Abrahams H. E., Robinson D. Beta-D-glucosidases and related enzymic activities in pig kidney. Biochem J. 1969 Mar;111(5):749–755. doi: 10.1042/bj1110749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews P. Estimation of the molecular weights of proteins by Sephadex gel-filtration. Biochem J. 1964 May;91(2):222–233. doi: 10.1042/bj0910222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andrews P. The gel-filtration behaviour of proteins related to their molecular weights over a wide range. Biochem J. 1965 Sep;96(3):595–606. doi: 10.1042/bj0960595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DUERKSEN J. D., HALVORSON H. Purification and properties of an inducible beta-glucosidase of yeast. J Biol Chem. 1958 Nov;233(5):1113–1120. [PubMed] [Google Scholar]
- Fleming L. W., Duerksen J. D. Evidence for multiple molecular forms of yeast beta-glucosidase in a hybrid yeast. J Bacteriol. 1967 Jan;93(1):142–150. doi: 10.1128/jb.93.1.142-150.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fleming L. W., Duerksen J. D. Purification and characterization of yeast beta-glucosidases. J Bacteriol. 1967 Jan;93(1):135–141. doi: 10.1128/jb.93.1.135-141.1967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GALLI D. R., GIESE A. C. Carbohydrate digestion in a herbivorous snail. Tegula funebralis. J Exp Zool. 1959 Apr;140:415–440. doi: 10.1002/jez.1401400305. [DOI] [PubMed] [Google Scholar]
- HU A. S., EPSTEIN R., HALVORSON H. O., BOCK R. M. Yeast beta-glucosidase: comparison of the physical-chemical properties of purified constitutive and inducible enzyme. Arch Biochem Biophys. 1960 Dec;91:210–218. doi: 10.1016/0003-9861(60)90492-6. [DOI] [PubMed] [Google Scholar]
- Han Y. W., Srinivasan V. R. Purification and characterization of beta-glucosidase of Alcaligenes faecalis. J Bacteriol. 1969 Dec;100(3):1355–1363. doi: 10.1128/jb.100.3.1355-1363.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horikoshi K., Ikeda Y. Studies on the spore coats of aspergillus oryzae. II. Conidia coat-bound beta-glucosidase. Biochim Biophys Acta. 1965 Nov 1;101(3):352–357. doi: 10.1016/0926-6534(65)90014-x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Legler G. Labelling of the active centre of a beta-glucosidase. Biochim Biophys Acta. 1968 Mar 25;151(3):728–729. doi: 10.1016/0005-2744(68)90033-8. [DOI] [PubMed] [Google Scholar]
- Li L. H., Flora R. M., King K. W. Individual roles of cellulase components derived from Trichoderma viride. Arch Biochem Biophys. 1965 Aug;111(2):439–447. doi: 10.1016/0003-9861(65)90207-9. [DOI] [PubMed] [Google Scholar]
- MAHADEVAN P. R., EBERHART B. ARYL BETA-GLUCOSIDASE OF SOME NEUROSPORA STRAINS. Biochim Biophys Acta. 1964 Jul 15;90:214–215. doi: 10.1016/0304-4165(64)90147-3. [DOI] [PubMed] [Google Scholar]
- Marchin G. L., Duerksen J. D. Purification of beta-glucosidase from Saccharomyces lactis strain Y-123. J Bacteriol. 1968 Oct;96(4):1181–1186. doi: 10.1128/jb.96.4.1181-1186.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchin G. L., Duerksen J. D. Purification of beta-glucosidase from Saccharomyces lactis strains Y-14 and Y-1057A. J Bacteriol. 1968 Oct;96(4):1187–1190. doi: 10.1128/jb.96.4.1187-1190.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PARNAS I. The cellulolytic activity in the snail Levantina hierosolyma Boiss. J Cell Comp Physiol. 1961 Oct;58:195–201. doi: 10.1002/jcp.1030580211. [DOI] [PubMed] [Google Scholar]
- ROBINSON D. The fluorimetric determination of beta-glucosidase: its occurrence in the tissues of animals, including insects. Biochem J. 1956 May;63(1):39–44. doi: 10.1042/bj0630039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson D., Price R. G., Dance N. Rat-urine glycosidases and kidney damage. Biochem J. 1967 Feb;102(2):533–538. doi: 10.1042/bj1020533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson D., Price R. G., Dance N. Separation and properties of beta-galactosidase, beta-glucosidase, beta-glucuronidase and N-acetyl-beta-glucosaminidase from rat kidney. Biochem J. 1967 Feb;102(2):525–532. doi: 10.1042/bj1020525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umezurike G. M. The purification and properties of extracellular beta-glucosidase from Botryodiplodia theobromae Pat. Biochim Biophys Acta. 1971 Feb 10;227(2):419–428. doi: 10.1016/0005-2744(71)90073-8. [DOI] [PubMed] [Google Scholar]
- Umezurike G. M. The subunit structure of beta-glucosidase from Botryodiplodia theobromae Pat. Biochem J. 1975 Feb;145(2):361–368. doi: 10.1042/bj1450361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood T. M. The cellulase of Fusarium solani. Purification and specificity of the -(1-4)-glucanase and the -D-glucosidase components. Biochem J. 1971 Feb;121(3):353–362. doi: 10.1042/bj1210353. [DOI] [PMC free article] [PubMed] [Google Scholar]


