Abstract
This study aimed to investigate the bioactive metabolites produced by Streptomyces thinghirensis WAE1, an actinomycete isolated from El-Hamara Lake in Egypt. The discovery of new bioactive compounds from natural sources is crucial for the advancement of therapeutic treatments, and this study aimed to contribute to this field by exploring the potential of Streptomyces thinghirensis WAE1 as a source of such compounds. Streptomyces thinghirensis WAE1 was screened for its ability to produce antimicrobial, antioxidant, and anti-inflammatory metabolites. The results revealed that S. thinghirensis WAE1 exhibited strong antimicrobial activity against Streptococcus pneumoniae and moderate activity against Listeria monocytogenes, Staphylococcus aureus, and Candida albicans. Streptomyces thinghirensis WAE1 also displayed antioxidant activity through scavenging free radicals and chelating iron, and moderate anti-inflammatory activity as determined by its IC50 value. The isolate's demonstration of L-asparaginase activity suggests that S. thinghirensis WAE1 is a promising source of bioactive compounds with potential therapeutic uses. The high salinity and alkalinity of El-Hamara Lake, which create favorable conditions for the production of bioactive metabolites, further add to its potential as a source of actinomycetes strains with bioactive properties. These findings make both S. thinghirensis WAE1 and El-Hamara Lake valuable subjects for further exploration in the field of bioactive compounds.
Keywords: Actinobacteria, Antimicrobial, Antioxidant, Anti-inflammatory, L-asparaginase
Introduction
The emergence of multi-drug resistance in microorganisms is a significant issue that calls for creating innovative and new antibiotics [1]. The existence of free radicals has been associated with chronic illness, inflammation, and cancer. These unstable molecules can cause chemical changes in biomolecules, leading to structural and functional modifications, when there is an overabundance of free radicals and insufficient antioxidant defense [2]. Natural compounds have indeed played a key role in the discovery of drugs for the treatment of many human diseases. The number of natural compounds discovered has surpassed one million, making it a vast and continuously expanding resource for developing new and effective treatments [3]. The genus Streptomyces is well-known for producing bioactive metabolites and is considered a key microorganism in the field of drug discovery. These microorganisms are filamentous, gram-positive bacteria with similarities in appearance to fungi. They have a large genome and a high G + C content, which allows them to adapt and survive in a range of environments [4]. Scientific research has shown that the genome of streptomycetes contains more than 20 clusters of synthetic genes involved in the biosynthesis of secondary metabolites. This genetic diversity is thought to be responsible for the production of varied bioactive metabolites. The wide range of metabolites produced by these microorganisms, including enzymes, antimicrobial, anticancer, and antioxidant agents, has led to a great deal of exploration and research for various applications, making streptomycetes important subjects for study in the field of drug discovery [5]. Researchers are still searching for new streptomyces species in understudied environments such as halophilic lakes in the hope of finding new compounds or therapeutic agents for disease treatment. These extreme environments have been shown to have the potential to produce novel antioxidant and anticancer compounds, emphasizing the importance of exploring these areas [6]. Additionally, L-asparaginase, an enzyme that plays a role in cancer therapy for acute lymphoblastic leukemia, works by reducing the supply of L-asparagine to cancer cells, thus hindering their growth and survival. However, there are challenges associated with its use, such as the occurrence of hypersensitivity reactions and the development of antibodies. To address these limitations, scientists have proposed alternative forms of L-asparaginase, specifically using Streptomyces species from the actinomycetes family as a source for its production [7]. Consequently, exploring streptomycetes from halophilic environments, like the Wadi El-Natron lakes in Egypt, holds great promise in uncovering new Streptomyces species and discovering valuable bioactive molecules.
Material and Methods
Isolation of Streptomyces Strain
The collection of Streptomyces spp. was carried out from soil samples sourced from various locations in Egypt through a dilution plate method using a medium of starch nitrate agar. After incubation at 30 °C for seven days, the most dominant actinomycetes isolates were identified, purified, and stored on slants of starch nitrate agar at a temperature of 4 °C for additional analysis [8].
Antimicrobial Activity
A well diffusion assay was applied to investigate the antimicrobial effect of different bacteria (gram-positive and gram-negative) and fungi, including Listeria monocytogenes ATCC 19116, Escherichia coli ATCC 25922, Clostridium sporogenes ATCC 3584, Staphylococcus aureus ATCC 29213, Salmonella enterica ATCC 25566, Cronobacter sakazakii ATCC 29544, Pseudomonas aeruginosa ATCC 27853, Klebsiella pneumoniae ATCC 27736, and Streptococcus pneumoniae ATCC 49619 and Candida albicans ATCC 10231. The isolates were grown on starch nitrate agar for 7 days at 30 °C, and ten 5 mm agar plugs of each culture were inoculated into ISP2 broth medium in 1 L Erlenmeyer flasks, which were incubated for 14 days at 30 °C with shaking at 150 rpm. Cells were harvested by centrifugation at 5000 rpm and 4 °C. The supernatant was collected and extracted with ethyl acetate. The dried crude extract obtained after vacuum evaporation was dissolved in 90% ethanol to make a 50 mg/ml stock solution. Twenty microliters of each crude extract were transferred into a 5 mm well in nutrient agar plates seeded with the test organisms. The plates were incubated at 37 °C for one day for bacteria and 3 days for fungus and the inhibition zone around the holes was measured to determine antagonistic activity [9]. Due to the inhibitory activity observed, the crude extract was selected for further analysis of its biological activities.
Identification of Streptomyces Isolate
Morphological and Physiological Characteristics
After 14 days of incubation on starch nitrate agar at 30 °C, the strain WAE1's spore chain and surface were analyzed. The isolate was examined with a scanning electron microscope at various magnifications and evaluated for its biochemical and physiological properties using established methods [10].
Genotypic Characterization
Using genomic DNA from the chosen strain, the 16S rRNA genes were amplified, and their sequence was established by Sanger sequencing. Through the BLASTn website, the final sequence was submitted to the gene bank database. Using MEGA version 4 software, the sequence was aligned, and the taxonomy categorization was established by building a phylogenetic tree [11].
Gene Expression Analysis for PKS and NRPS
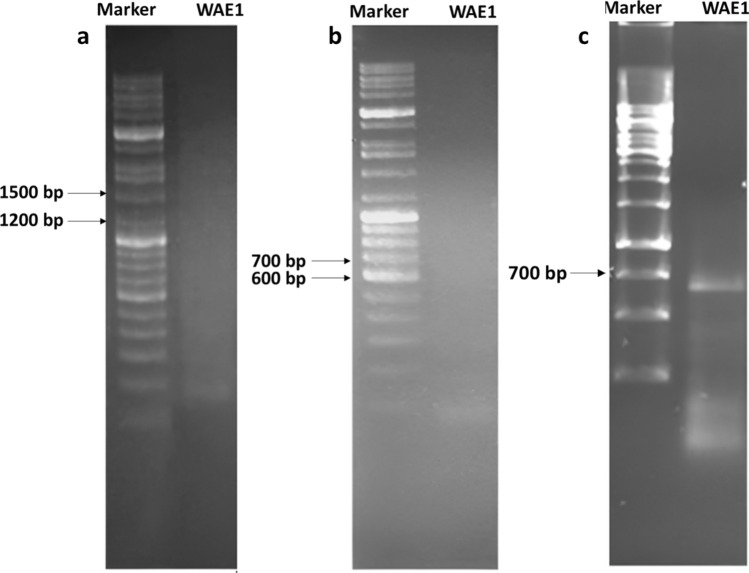
Degenerate primers were used in polymerase chain reaction analyses to detect polyketides (PKS) and non-ribosomal peptides (NRPS) genes in the actinomycete isolate. GelRed (Biotium) was used to visualize the PCR products on 1% agarose gel electrophoresis, and amplicon detecting was scored as positive if the amplicon was present and of the expected size (1200–1400 bp for PKS type I, 600–700 bp for PKS type II, and 700–800 bp for NRPS), or negative if the amplicon was absent [12].
Biological Activity
In-Vitro Antioxidant Activities
Crude extract at various concentrations (50, 100, 150, 200, and 250 µg/ml) antioxidants' ability to neutralize free radicals was measured using DPPH radical scavenging assay, ABTS radical scavenging assay, ferric-reducing antioxidant power (FRAP) assay, metal chelation assay, hydrogen peroxide radical scavenging assay, scavenging superoxide assay, nitric oxide radical scavenging assay and lipid peroxidation inhibition assay. 20 µg/ml, 40 µg/ml, 60 µg/ml, 80 µg/ml, and 100 µg/ml of ascorbic acid were used as the standard reference [13–18].
Determining the Total Amount of Phenolic and Flavonoid Content
The WAE1 strain ethyl acetate extract's total phenolic content was calculated using the Folin-Ciocalteu method, and the aluminum trichloride method was used to determine the total flavonoid compounds. A 2 mg/mL DMSO solution of the crude extract was made, and gallic acid and rutin were employed as standards for the phenolics and flavonoids, respectively [19].
In-Vitro Anti-Inflammatory Assay
RAW 264.7 murine macrophages were used in a scientific experiment to see whether an extract had anti-inflammatory properties. The cells were cultivated in a growth mixture containing fetal bovine serum and other supplements and then placed in microwell plates. The cells were exposed to the sample extract, a known anti-inflammatory compound (indomethacin), or an inflammatory inducer (LPS). After 24 h of incubation, the levels of nitric oxide (NO) produced by the cells were measured using the Griess assay. This assay involves converting nitrite, a stable byproduct of NO, into a colored compound that can be quantified by spectrophotometry at 540 nm. Using the Alamar Blue reduction test to measure cell viability, we analyzed the sample extract with the LPS-induced inflammation group and expressed the results as a percentage decrease in NO production. The experiment was performed in triplicate for each treatment group [20].
Screening for L-Asparaginase Activity
A two-step screening procedure was used to assess the enzyme L-asparaginase production by WAE1 strain. The strain's ability to produce L-asparaginase was assessed qualitatively in the first phase using modified M9 agar plates, and quantitatively in the second. The quantity of ammonia generated in the reaction mixture, which was calibrated using standard ammonium chloride solutions, was used to measure the enzyme's activity. The enzyme’s activity was measured in units, where one unit equalled the quantity of enzyme necessary to produce 1 mol of ammonia per minute under specific conditions [21].
Statistical Analysis
Three duplicates of each experiment were performed. The data's mean and standard deviation (SD) were used to express the findings by using excel software.
Results and Discussion
Isolation and Screening
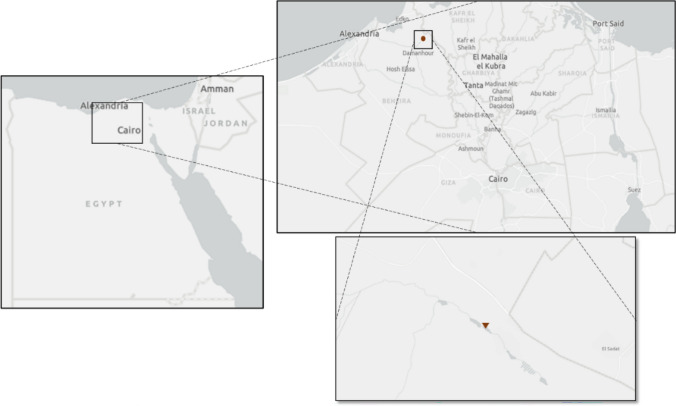
Sixty actinomycete isolates were obtained from soil samples collected from various locations across Egypt. Following purification of these isolates in culture, their ability to produce antimicrobial metabolites was evaluated using an inhibition assay. Of the sixty isolates tested, nineteen (32%) demonstrated inhibitory activity against one or more pathogenic indicator microorganisms. The isolate producing the strongest antimicrobial effect, denoted WAE1, was selected for further characterization. WAE1 was isolated from a soil sample collected near El-Hamara Lake located in Wadi El-Natron, Egypt (30° 23′ 47.86′′ N, 30° 19′ 0.46′′ E; Fig. 6). To explore its biotechnological potential, WAE1 was grown under controlled conditions to induce the biosynthesis of secondary metabolites. Both qualitative and quantitative screening of extracts from WAE1 cultures revealed potent bioactivity, warranting further investigation into its biologically active compounds. Messaoudi et al. [22] isolated 40 strains of actinobacteria from saline environments and date palm rhizospheres in Algeria, including Streptomyces, Nocardiopsis, and Saccharopolyspora. The isolates showed moderate to strong antimicrobial activities against bacteria and fungi, indicating the potential of these rare actinobacteria for bioactive compounds. Yang et al. [23] isolated 77 actinobacterial strains from soil samples of the hypersaline lake Gudzhirganskoe in Siberia. Streptomyces dominated among the isolates, and 33% of them showed inhibitory activity against multidrug-resistant pathogens like S. aureus and Acinetobacter baumannii. This suggests that unconventional environment like hypersaline lakes, could be explored for discovering novel actinobacteria with potential therapeutic applications.
Fig. 1.
shows the location of El-Hamara Lake in Wadi El-Natron, Egypt, where the potent antimicrobial-producing actinomycete isolate WAE1 was obtained from a soil sample
Bioactive Metabolites Activity
The WAE1 isolate demonstrated significant bioactive metabolite production, exhibiting antimicrobial activity against both bacterial and fungal test pathogens. Strong inhibition was observed against S. pneumoniae ATCC 49619 and L. monocytogenes ATCC 19116. Moderate activity was also seen against S. aureus ATCC 29213 and K. pneumoniae ATCC 27736, while weak activity was measured against C. albicans ATCC 10231. No inhibitory effect was found for C. sporogenes ATCC 3584, S. enterica ATCC 25566, E. coli ATCC 25922, C. sakazakii ATCC 29544, P. aeruginosa ATCC 27853. The antimicrobial metabolites produced by WAE1 showed differential activity against gram-positive and gram-negative bacterial strains as well as the fungus tested. The results are presented quantitatively in Table 1 and Fig. 1 to compare the antimicrobial potential of WAE1 against various pathogens. Al-Ansari et al. [24] studied the antibacterial activity of Streptomyces sp. AS11 against various pathogenic bacteria. The crude extract showed significant antibacterial activity, with zones of inhibition ranging from 10 to 29 mm. S. aureus was the most susceptible, with a zone of inhibition of 29 mm. Other pathogens like Proteus mirabilis and Salmonella Typhi showed zones of 20 ± 2 mm and 16 ± 2 mm, respectively. Jaroszewicz et al. [25] studied the antibacterial properties of Streptomyces strain M4_24 from the Szczelina Chochołowska cave. They found that strain showed antimicrobial activity against most tested bacteria, M4_24 produced inhibition zones ranging from 3.0 to 12.0 mm against S. aureus, S enterica, Enterococcus, E. coli, and P. aeruginosa, indicating its ability to secrete bioactive compounds.
Table 1.
Antimicrobial activity of WAE1 isolate against pathogenic strains as measured by inhibition zone diameters (mean ± standard deviation, n = 3) in agar well diffusion assays
| Microorganisms | Inhibition zone (mm) |
|---|---|
| S. pneumoniae ATCC 49619 | 33.17 ± 0.88 |
| L. monocytogenes ATCC 19116 | 28.67 ± 1.15 |
| C. sporogenes ATCC 3584 | – |
| S. aureus ATCC 29213 | 12.33 ± 0.80 |
| K. pneumoniae ATCC 27736 | 5.88 ± 1.04 |
| S. enterica ATCC 25566 | – |
| E. coli ATCC 25922 | – |
| C. sakazakii ATCC 29544 | – |
| P. aeruginosa ATCC 27853 | – |
| C. albicans ATCC 10231 | 12.63 ± 1.13 |
–, no inhibition zone
Fig. 2.
Antimicrobial activity of WAE1 isolate against various pathogenic bacterial and fungal strains as determined by agar well diffusion assays. The activities were evaluated against, a S. pneumoniae ATCC 49619, b L. monocytogenes ATCC 19116, c S. aureus ATCC 29213, d K. pneumoniae ATCC 27736, e C. albicans ATCC 10231, with no activity observed against, f C. sporogenes ATCC 3584, g S. enterica ATCC 25566, h E. coli ATCC 25922, i C. sakazakii ATCC 29544 and j P. aeruginosa ATCC 27853
Identification of WAE1 Isolate
Based on morphological, and biochemical characteristics and a polyphasic approach, indicate that the isolate is a member of the actinobacteria genus with novel characteristics (as shown in Figs. 2, 3 and Table 2). WAE1 strain was found to be closely related to Streptomyces thinghirensis, based on genomic identification (Fig. 4) which displayed typical morphological, coloration, and biochemical characteristics features of the Streptomyces genus and submitted into the GenBank database under the accession number ON584359.1 [26]. Almalki [27] isolated an actinomycete producer of polyketide antibiotics, SCA-7, from a marine sediment sample in Al-Khobar, Saudi Arabia. The isolate showed promising antimicrobial activity against Enterococcus sp. and was characterized for identification. Morphological analysis revealed filamentous growth with aerial mycelium, and biochemical characterization showed it could utilize various sugars and grow in different nitrogen sources and temperatures. Molecular identification confirmed SCA-7 belonged to the Streptomyces genus, exhibiting 99% sequence similarity to Streptomyces felleus. Maleki et al. [28] identified 12 Streptomyces isolates from soil samples in northwest Iran. Morphological analysis revealed high antibacterial activity by these isolates. Biochemical tests and 16S rDNA gene sequencing identified carbon source utilization patterns. Molecular identification showed 99% sequence similarity to Streptomyces species for all 12 isolates. RAPD-PCR clustering identified two promising isolates as S. albogriseolus and S. coelicolor. These findings confirmed the identification of Streptomyces species with potent antibacterial activity.
Fig. 3.
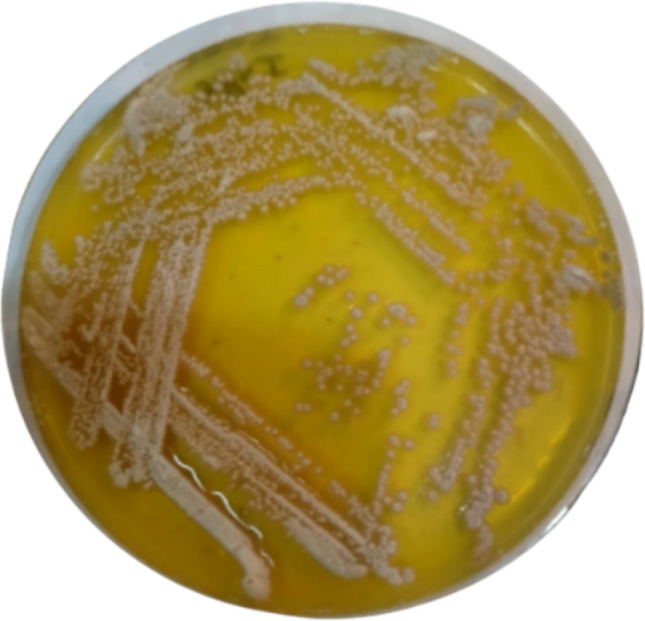
Phenotypic appearance of the actinobacterial isolate S. thinghirensis WAE1 grown on starch nitrate agar showing whitish-grey colonies exhibiting yellow-colored exopigment production
Fig. 4.

Phylogenetic tree of 16S rRNA gene sequence of WAE1 isolate and closely related type strains constructed using the neighbour-joining method, showing S. thinghirensis (NR_116901.1) as the closest type strain with 100% sequence similarity
Table 2.
Streptomyces thinghirensis WAE1's physiological and biochemical characteristics
| Test | Result | Test | Result | ||
|---|---|---|---|---|---|
| Gram stain | + | Nitrogen source | potassium nitrate | + | |
| Media | ISP 1 | Whitish grey | Tyrosine | + | |
| ISP 2 | Light grey | Cysteine | + | ||
| ISP 3 | Light grey | Tryptophane | + | ||
| ISP 4 | Grey | Arginine | + | ||
| ISP 5 | Whitish Grey | pH | 5 | – | |
| ISP 6 | Light Grey | 7 | + | ||
| ISP 7 | Light Grey | 9 | + | ||
| Carbon source | Arabinose | + | 11 | – | |
| D-Xylose | + | Temperature (oC) | 20 | + | |
| Sucrose | + | 30 | + | ||
| D-Mannitol | + | 40 | + | ||
| Galactose | – | 50 | – | ||
| Lactose | + | NaCl (%) | 0 | + | |
| Ribose | + | 2.5 | + | ||
| Rhamnose | + | 5 | + | ||
| Starch | + | 7.5 | + | ||
| D-Glucose | + | 10 | – | ||
| Fructose | + | ||||
| Mannose | + | ||||
Fig. 5.
Phylogenetic tree of S. thinghirensis WAE1 was created by neighbour-joining technique
Detection of PKS and NRPS Genes
The biosynthetic potential of the WAE1 isolate was assessed by screening for the gene sequences of non-ribosomal peptide synthetases (NRPS), polyketide synthases type I (PKS-I), and type II (PKS-II). The chromosomal DNA of the isolate was amplified using degenerate primers. This actinobacteria isolate was found to only include the adenylation domain of NRPS genes, as indicated by an amplicon size of 700–800 bp (Fig. 5). Non-ribosomal peptides genes have been documented to play a role in producing biologically active compounds such as antibiotics and antifungal metabolites, further strengthening the potential of S. thinghirensis WAE1 as a promising candidate for the discovery of new drugs [29]. Peng et al. [30] screened 42 Streptomyces isolates from rhizospheric soil of Panax notoginseng for biosynthetic genes encoding type I polyketide synthase (PKS I), type II polyketide synthase (PKS II), and non-ribosomal peptide synthetases (NRPS) using PCR with degenerate primers. They found that 41 of the 42 isolates yielded sequences related to at least one of the three gene types screened for. Specifically, they detected PKS I loci in 30 isolates, PKS II loci in 38 isolates, and NRPS loci in 7 isolates. Ghashghaei et al. [12] screened 22 actinobacterial isolates for genes involved in polyketide and non-ribosomal peptide biosynthesis. They found one isolate was positive only for the adenylation domain of NRPS genes, indicating the isolate's potential to produce secondary metabolites synthesized via NRPS gene clusters. This highlights the potential of these isolates in polyketide and peptide biosynthesis.
Fig. 6.
PCR amplification of PKS-I (a), PKS-II (b) and NRPS (c) gene sequences with an amplicon size of 1200–1400, 600–700 and 700–800 bp using primers K1F/M6R, KSαF/KSαR and A3F/A3R, respectively, on agarose gel 1%. The S. thinghirensis WAE1 isolate was found to only include the adenylation domain of NRPS genes, as indicated by an amplicon size of 700–800 bp
Biological Activity
In-Vitro Antioxidant Activities
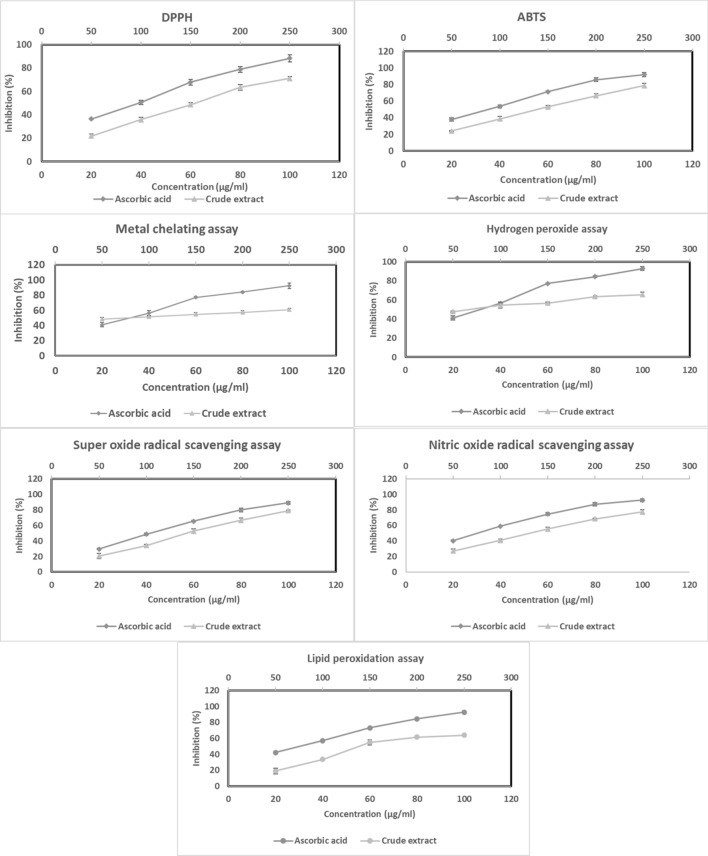
The results demonstrated that, the WAE1 extract exhibited significant dose-dependent inhibition of DPPH, ABTS, and hydrogen peroxide radical activity, with IC50 values of 157.08 ± 2.66 μg/ml, 141.39 ± 3.46 μg/ml, and 66.01 ± 1.57 μg/ml, respectively. It also demonstrated metal chelating activity with an IC50 value of 76.58 ± 3.52 μg/ml and ferric-reducing antioxidant power with a value of 0.30 ± 0.02 mM Fe2+/g. The extract showed superoxide and nitric oxide radical scavenging activity with IC50 values of 148.29 ± 3.15 μg/ml and 135.14 ± 2.81 μg/ml, respectively. Additionally, it exhibited lipid peroxidation inhibition with an IC50 value of 164.38 ± 2.18 μg/ml. For comparison, the IC50 values for vitamin C were 38.06 ± 0.67 μg/ml, 33.94 ± 0.89 μg/ml, 29.10 ± 2.67 μg/ml, 29.32 ± 1.64 μg/ml, 1.615 ± 0.7 mM Fe2+/g, 43.43 ± 2.09 μg/ml, 28.87 ± 1.67 μg/ml, and 28.98 ± 1.26 μg/ml respectively. The results are shown in (Fig. 7). These antioxidant mechanisms help counteract reactive oxygen species (ROS) that can be pathologically generated. Overproduction of ROS has been correlated with elevated risk for certain diseases associated with oxidative stress. Therefore, natural extracts from actinobacteria exhibiting antioxidant properties may offer protective effects by neutralizing ROS [31, 32].
Fig. 7.
Assessment of the antioxidant properties of the WAE1 extract and ascorbic acid used as a control by using different antioxidant assays
The extract obtained from the actinobacterium strain WAE1 was found to contain phenolic and flavonoid components that likely contribute to its antioxidant properties. The total flavonoid content of the WAE1 extract was quantified as 11.23 ± 3.15 mg rutin equivalents per 100 mg of extract, while the total phenolic content was determined to be 57.89 ± 2.67 mg gallic acid equivalents per 100 mg of extract. These results indicate that the WAE1 extract is rich in phenolic and flavonoid compounds. Previous studies have demonstrated a positive correlation between the total phenolic and flavonoid contents of microbial extracts with their antioxidant abilities, likely due to the ability of these compounds to scavenge free radicals [19].
Kemung et al. [33] studied the antioxidant potential of Streptomyces sp. MUSC 11 from mangrove soil in Malaysia. They evaluated the methanolic extract's antioxidant activity using various in-vitro assays. The results showed that the extract showed concentration-dependent radical scavenging activity, with maximum activities of 31.42 ± 1.00% and 7.27 ± 4.73% at 4 mg/mL. It also showed metal chelating activity of 21.61 ± 1.71% and ferric reducing equivalents of 3.001–3.521 ng ascorbic acid in the FRAP assay. Rammali et al. [19] studied the antioxidant properties of Streptomyces species isolated from cold soil sites in Morocco. They found that the E23-4 strain extract had the highest total phenolic content and flavonoid content. The antioxidant activities of the extracts were evaluated using DPPH and ABTS radical scavenging assays. All extracts showed lower DPPH radical scavenging activity compared to ascorbic acid, with no significant differences between extracts. In the ABTS assay, E23-4 had the highest activity at 35.79% inhibition. A significant positive correlation was found between total phenolic and flavonoid contents and antioxidant capacity, indicating that polyphenols significantly contribute to antioxidant properties. The study concluded that Streptomyces isolated from cold soil sites in Morocco produced metabolites with notable antioxidant activities, suggesting potential applications as natural antioxidants. Tangjitjaroenkun et al. [34] conducted an in-vitro study on the antioxidant properties of the ethyl acetate extract from Streptomyces achromogenes TCH4. They found that the extract had a total phenolic and flavonoid content of 107.20 ± 2.57 mg GAE/g and 44.91 ± 0.84 mg QE/g of dry extract, respectively. Phenolic and flavonoid compounds are known to contribute significantly to the antioxidant activities. The extract also demonstrated free radical scavenging activity using the DPPH and ABTS assays. In the DPPH radical scavenging assay, the extract showed concentration-dependent scavenging activity with an IC50 value of 299.64 ± 6.83 μg/mL, compared to the positive control BHT of 32.95 ± 0.26 μg/mL. In the ABTS radical scavenging assay, the extract inhibited the ABTS radical in a concentration-dependent manner with an IC50 value of 65.53 ± 0.95 μg/mL. The ferric-reducing antioxidant power (FRAP) of the extract was 822.76 ± 9.12 mM FeSO4.7H2O/g dry extract, indicating its potential in reducing oxidized species. Shobha et al. [35] studied the in-vitro antioxidant properties of a metabolite extracted from Streptomyces species KSRO-04. They assessed various antioxidant activities, including total phenolic content, flavonoids content, DPPH radical scavenging activity, metal chelating activity, total reductive capability, lipid peroxidation inhibition, superoxide anion scavenging, and nitric oxide scavenging. The metabolite showed moderate antioxidant activity in the DPPH radical scavenging assay, with an inhibition percentage of 14.39% at 125 μg/ml concentration. It also showed good metal chelating activity, inhibiting the formation of the ferrous-ferrozine complex with an inhibition percentage of 39.55% at 200 μg/ml. It also showed high nitric oxide scavenging activity of 79.25% at 125 μg/ml. Saravana et al. [36] evaluated the antioxidant properties of Streptomyces lavendulae strain SCA5 through in-vitro assays. The ethyl acetate extract (EA-SCA5) showed dose-dependent antioxidant activity in all assays, with IC50 values of 507.61 ± 0.66 μg/ml for DPPH radicals, 617.84 ± 0.57 μg/ml for hydroxyl radicals, 730.92 ± 0.81 μg/ml for nitric oxide, and 864.71 ± 1.15 μg/ml for superoxide anion radicals. This demonstrated S. lavendulae strain SCA5's ability to scavenge various free radicals and its potential as a natural antioxidant.
In-Vitro Anti-Inflammatory Assay
The LPS-induced NO generation by macrophages (RAW 264.7 cells) was significantly inhibited by WAE1 extract, with an IC50 value of 36.43 μg/ml. The IC50 value for indomethacin used as a control was 14.50 ± 0.75 μg/ml. The results are shown in (Fig. 8). This indicates that the strain has moderate anti-inflammatory activity. This is consistent with previous reports of anti-inflammatory activity from marine Streptomyces species isolated from the southeast coast of India [37]. Actinomycetes have been widely reported as a source of anti-inflammatory compounds in various studies [38, 39]. Xu et al. [40] found that a crude extract from mangrove sediment, Streptomyces sp. XY-R31, has significant anti-inflammatory effects. They used LPS-stimulated RAW264.7 macrophages as an in-vitro inflammation model and found that the R31 crude extract significantly inhibited LPS-induced nitric oxide (NO) production in a dose-dependent manner.
Fig. 8.
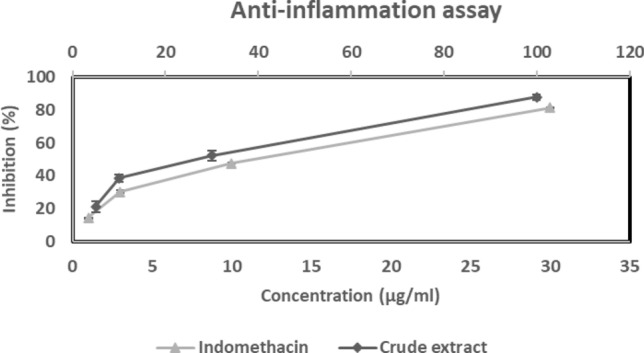
Anti-inflammatory activity of WAE1 extract and indomethacin used as a control
Screening for L-Asparaginase Activity
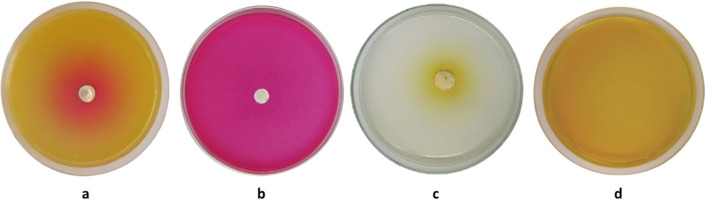
In a plate test, the L-asparaginase activity of WAE1 was determined by observing a change in color from yellow to pink around the colony (as seen in Fig. 9). L-asparaginase activity at 6 U/ml was detected in the WAE1 strain. This aligns with previous research that has shown that various actinobacteria can produce L-asparaginase, with Streptomyces sp. being the most frequently identified actinobacteria that can produce this enzyme from various sources. L-asparaginases have attracted a lot of interest because they may be used to treat acute lymphoblastic leukemia and in the food industry to prevent acrylamide production in fried and baked foods [41, 42]. El-Sabbagh et al. [43] discovered that a strain of Streptomyces halstedii from Egypt produces L-asparaginase. The production starts after 72 h of cultivation and reaches a maximum level of 3.9 U/ml after 120 h. Arévalo-Tristancho et al. [44] conducted a study on 78 actinobacterial strains from the Arauca riverbank in Colombia, focusing on their ability to produce L-asparaginase. They found that strain 112, identified as Streptomyces lacticiproducens, showed an L-asparaginase activity of 57.45 U/mL, indicating its potential as an effective producer.
Fig. 9.
Qualitative detection of extracellular L-asparaginase secretion by S. thinghirensis WAE1 cultured in modified M9 solid medium. L-asparaginase activity was visualized using a pH indicator dye phenol red, where enzymatic hydrolysis of L-asparagine resulted in localized alkalinization and consequent color change from yellow to pink surrounding colonies after (a) 3 days and (b) 7 days of incubation. Control plates showed (c) inoculated medium lacking phenol red (positive control), and (d) uniform yellow coloration for uninoculated medium containing phenol red (negative control)
Conclusion
This study isolated and characterized an actinobacteria strain, WAE1, from a hypersaline lake in Egypt. A total of sixty distinct actinomycete isolates were obtained from soil samples, of which thirty-two percent showed antimicrobial activity in preliminary screening. Isolate WAE1 was selected for further characterization based on its superior antibacterial activity against pathogenic microorganisms. Morphological, biochemical, and molecular identification characterized WAE1 as S. thinghirensis strain. WAE1 demonstrated significant bioactive metabolite activities, particularly against S. pneumoniae ATCC 49619. Extracts from WAE1 also exhibited notable antioxidant activities in several in-vitro assays, indicating their potential as natural antioxidants. The isolate was found to harbor NRPS genes for secondary metabolite production and displayed moderate anti-inflammatory activity against LPS-induced NO generation in macrophages. Additionally, WAE1 produced the enzyme L-asparaginase, which has therapeutic applications. Overall, the results indicate that S. thinghirensis WAE1 is a promising candidate for the discovery of novel bioactive compounds with various biological activities. Further investigations are warranted to purify and characterize the active metabolites produced by this isolate.
Author Contribution
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by ETM. The first draft of the manuscript was written by MEO, AAA and ETM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). The authors have no relevant financial or non-financial interests to disclose.
Data Availability
As corresponding author, I state that all data included in the manuscript are available.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Garduno A, Martín-Loeches I (2022) Efficacy and appropriateness of novel antibiotics in response to antimicrobial-resistant gram-negative bacteria in patients with sepsis in the ICU. Expert Rev Anti Infect Ther 20:513–531. 10.1080/14787210.2022.1999804 [DOI] [PubMed] [Google Scholar]
- 2.Leyane TS, Jere SW, Houreld NN (2022) Oxidative stress in ageing and chronic degenerative pathologies: molecular mechanisms involved in counteracting oxidative stress and chronic inflammation. Int J Mol Sci 23:7273. 10.3390/ijms23137273 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Law JW-F, Chan K-G, He Y-W et al (2019) Diversity of Streptomyces spp. from mangrove forest of Sarawak (Malaysia) and screening of their antioxidant and cytotoxic activities. Sci Rep 9:1–15. 10.1038/s41598-019-51622-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bahrami Y, Bouk S, Kakaei E, Taheri M (2022) Natural products from Actinobacteria as a potential source of new therapies against colorectal cancer: a review. Front Pharmacol 13:1–31. 10.3389/fphar.2022.929161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhang H, Chen Y, Li Y et al (2022) Secondary metabolites and biosynthetic gene clusters analysis of deep-sea hydrothermal vent-derived Streptomyces sp. SCSIO ZS0520. Mar Drugs 20:393. 10.3390/md20060393 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Donald L, Pipite A, Subramani R et al (2022) Streptomyces: still the biggest producer of new natural secondary metabolites, a current perspective. Microbiol Res 13:418–465. 10.3390/microbiolres13030031 [Google Scholar]
- 7.Soliman HM, El-Naggar NE-A, El-Ewasy SM (2020) Bioprocess optimization for enhanced production of L-asparaginase via two model-based experimental designs by Alkaliphilic Streptomyces fradiae NEAE-82. Curr Biotechnol 9:23–37. 10.2174/2211550109666200206100445 [Google Scholar]
- 8.Osman ME, El-Nasr AAA, Hussein HM, Hamed MM (2022) Bioactivity of metabolites from actinomycetes isolates from Red Sea, Egypt. Microbiol Biotechnol Lett 50:255–269. 10.48022/mbl.2202.02003 [Google Scholar]
- 9.Djebbah FZ, Al-Dhabi NA, Arasu MV et al (2022) Isolation and characterisation of Streptomyces sp. Strain GLD25 with antimicrobial and antioxidant effects from Gueldaman cave (GLD1), Akbou-Algeria. J King Saud Univ 34:101719. 10.1016/j.jksus.2021.101719 [Google Scholar]
- 10.El-Naggar NE-AA, Hamouda RA (2016) Antimicrobial potentialities of Streptomyces lienomycini NEAE-31 against human pathogen multidrug-resistant Pseudomonas aeruginosa. Int J Pharmacol 12:769–788. 10.3923/ijp.2016.769.788 [Google Scholar]
- 11.Swamy PS, Bhat MP, Nayaka S (2022) Amycolatopsis sp. strain MN235945 mediated biosynthesis of silver nanoparticles: characterization, antimicrobial and anticancer activity against HeLa and MCF-7 cell lines. Indian J Pharm Sci 84:1178–1188. 10.36468/pharmaceutical-sciences.1012 [Google Scholar]
- 12.Ghashghaei S, Etemadifar Z, Tavassoli M, Mofid MR (2022) Optimization of degenerate PCR conditions for reducing error rates in detection of PKS and NRPS GENE GROUPS IN Actinomycetes. Avicenna J Med Biotechnol 15:28–37. 10.18502/ajmb.v15i1.11422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mangzira Kemung H, Tan LT-H, Chan K-G et al (2020) Streptomyces sp. strain MUSC 125 from mangrove soil in Malaysia with anti-MRSA, anti-biofilm and antioxidant activities. Molecules 25:3545. 10.3390/molecules25153545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bagheri E, Hajiaghaalipour F, Nyamathulla S, Salehen N (2018) The apoptotic effects of Brucea javanica fruit extract against HT29 cells associated with p53 upregulation and inhibition of NF-κB translocation. Drug Des Devel Ther 12:657. 10.2147/DDDT.S155115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dahiru N, Paliwal R, Madungurum MA et al (2022) Study on antioxidant property of Syzygium aromaticum (Clove). J Biochem Microbiol Biotechnol 10:13–16. 10.54987/jobimb.v10i1.657 [Google Scholar]
- 16.Panneerselvi V, Shankar K, Muthukrishnan P, Prabhu A (2022) Mangifera indica resin assisted synthesis of nano silver: assessing their photocatalytic degradation of methylene blue, anticorrosive and antioxidant activity. J Clust Sci. 10.1007/s10876-020-01965-7 [Google Scholar]
- 17.Onoja SO, Nnadi CO, Udem SC, Anaga AO (2020) Potential antidiabetic and antioxidant activities of a heliangolide sesquiterpene lactone isolated from Helianthus annuus L. leaves. Acta Pharm 70:215–226. 10.2478/acph-2020-0019 [DOI] [PubMed] [Google Scholar]
- 18.Sekhara I, Benaissa O, Amrani A et al (2020) Antioxidant activity and chemical constituents of Bernh. (Apiaceae) from Algeria. Acta Sci Nat 7:59–70. 10.2478/asn-2020-0007 [Google Scholar]
- 19.Rammali S, Hilali L, Dari K et al (2022) Antimicrobial and antioxidant activities of Streptomyces species from soils of three different cold sites in the Fez-Meknes region Morocco. Sci Rep 12:17233. 10.1038/s41598-022-21644-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ghanim AM, Girgis AS, Kariuki BM et al (2022) Design and synthesis of ibuprofen-quinoline conjugates as potential anti-inflammatory and analgesic drug candidates. Bioorg Chem 119:105557. 10.1016/j.bioorg.2021.105557 [DOI] [PubMed] [Google Scholar]
- 21.Davarpanah M, Bakhtiari R, Javadi A, Eshraghi SS (2022) Manufacture of L-Asparaginase by Actinobacteria isolated from rhizosphere of plants in Tehran with strong anti-cancer activity. Egypt J Vet Sci 53:9–14. 10.21608/EJVS.2021.89728.1263 [Google Scholar]
- 22.Messaoudi O, Wink J, Bendahou M (2020) Diversity of actinobacteria isolated from date palms rhizosphere and saline environments: isolation, identification and biological activity evaluation. Microorganisms 8:1853. 10.3390/microorganisms8121853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Yang Q, Guo P, Abidueva EY et al (2023) A pilot survey on antimicrobial activity and diversity of soil-derived actinobacteria from different depths in Gudzhirganskoe Saline Lake in Eastern Siberia. J Oceanol Limnol 41:1307–1320 [Google Scholar]
- 24.Al-Ansari M, Kalaiyarasi M, Almalki MA, Vijayaraghavan P (2020) Optimization of medium components for the production of antimicrobial and anticancer secondary metabolites from Streptomyces sp. AS11 isolated from the marine environment. J King Saud Univ 32:1993–1998. 10.1016/j.jksus.2020.02.005 [Google Scholar]
- 25.Jaroszewicz W, Bielańska P, Lubomska D et al (2021) Antibacterial, antifungal and anticancer activities of compounds produced by newly isolated Streptomyces strains from the Szczelina Chochołowska cave (Tatra Mountains, Poland). Antibiotics 10:1212. 10.3390/antibiotics10101212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Loqman S, Bouizgarne B, Barka EA et al (2009) Streptomyces thinghirensis sp. nov., isolated from rhizosphere soil of Vitis vinifera. Int J Syst Evol Microbiol 59:3063–3067 [DOI] [PubMed] [Google Scholar]
- 27.Almalki MA (2020) Isolation and characterization of polyketide drug molecule from Streptomyces species with antimicrobial activity against clinical pathogens. J Infect Public Health. 10.1016/j.jiph.2019.07.002 [DOI] [PubMed] [Google Scholar]
- 28.Maleki H, Dehnad A, Hanifian S, Khani S (2013) Isolation and molecular identification of Streptomyces spp. with antibacterial activity from Northwest of Iran. BioImpacts 3:129–134. 10.5681/bi.2013.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rehan M, Gueddou A, Alharbi A, Ben Abdelmalek I (2023) In silico prediction of secondary metabolites and biosynthetic gene clusters analysis of Streptomyces thinghirensis HM3 isolated from arid soil. Fermentation 9:65. 10.3390/fermentation9010065 [Google Scholar]
- 30.Peng F, Zhang M-Y, Hou S-Y et al (2020) Insights into Streptomyces spp. isolated from the rhizospheric soil of Panax notoginseng: isolation, antimicrobial activity and biosynthetic potential for polyketides and non-ribosomal peptides. BMC Microbiol 20:1–16. 10.1186/s12866-020-01832-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Rani A, Saini KC, Bast F et al (2021) Microorganisms: a potential source of bioactive molecules for antioxidant applications. Molecules 26:1142. 10.3390/molecules26041142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kamala K, Sivaperumal P, Kumar GJJ, Ganapathy DM (2023) Antioxidant properties from novel marine actinobacteria. In: Marine antioxidants. Elsevier, pp 215–220
- 33.Kemung HM, Tan LT-H, Chan K-G et al (2019) Investigating the antioxidant potential of Streptomyces sp. MUSC 11 from mangrove soil in Malaysia. Prog Drug Discov Biomed Sci 2:1–11. 10.36877/pddbs.a0000033 [Google Scholar]
- 34.Tangjitjaroenkun J, Pluempanupat W, Tangchitcharoenkhul R et al (2021) Antibacterial, antioxidant, cytotoxic effects and GC-MS analysis of mangrove-derived Streptomyces achromogenes TCH4 extract. Arch Biol Sci 73:223–235. 10.2298/ABS210320017T [Google Scholar]
- 35.Shobha KS, Gautham SA, Onkarappa R (2012) In vitro antioxidant activity of a metabolite from Streptomyces species KSRO-04. Int J Pharma Med Sci 2:7–13. 10.5829/idosi.ijpms.2012.2.2.75135 [Google Scholar]
- 36.Saravana Kumar P, Al-Dhabi NA, Duraipandiyan V et al (2014) In vitro antimicrobial, antioxidant and cytotoxic properties of Streptomyces lavendulae strain SCA5. BMC Microbiol 14:1–12. 10.1186/s12866-014-0291-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pooja S, Aditi T, Naine SJ, Subathra Devi C (2017) Bioactive compounds from marine Streptomyces sp. VITPSA as therapeutics. Front Biol 12:280–289. 10.1007/s11515-017-1459-x [Google Scholar]
- 38.Gao M, Lee SB, Lee J-E et al (2022) Anti-inflammatory butenolides from a Marine-derived Streptomyces sp. 13G036. Appl Sci 12:4510. 10.3390/app12094510 [Google Scholar]
- 39.Shin HJ, Heo C-S, Van AC et al (2022) Streptoglycerides E-H, unsaturated polyketides from the Marine-derived Bacterium Streptomyces specialis and their anti-inflammatory activity. Mar Drugs 20:44. 10.3390/md20010044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Xu Y, Yu Y, Zheng S, Fang R (2022) A secondary metabolites produced by Streptomyces ssp. XY-R31 suppresses LPS-induced inflammatory responses through NF-κB, MAPKs, and HO-1 pathways in RAW264. 7 Cells. 10.21203/rs.3.rs-1400033/v136611804 [Google Scholar]
- 41.Qeshmi FI, Homaei A, Fernandes P, Javadpour S (2018) Marine microbial L-asparaginase: biochemistry, molecular approaches and applications in tumor therapy and in food industry. Microbiol Res 208:99–112. 10.1016/j.micres.2018.01.011 [DOI] [PubMed] [Google Scholar]
- 42.Jia R, Wan X, Geng X et al (2021) Microbial L-asparaginase for application in acrylamide mitigation from food: current research status and future perspectives. Microorganisms 9:1659. 10.3390/microorganisms9081659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.El-Sabbagh SM, El-Batanony NH, Salem TA (2013) L-Asparaginase produced by Streptomyces strain isolated from Egyptian soil: purification, characterization and evaluation of its anti-tumor. Afr J Microbiol Res 7:5677–5686. 10.5897/AJMR2013.1937 [Google Scholar]
- 44.Arévalo-Tristancho E, Díaz LE, Cortázar JE, Valero MF (2019) Production and characterization of L-asparaginases of Streptomyces isolated from the Arauca Riverbank (Colombia). Open Microbiol J 13:204–215. 10.2174/1874285801913010204 [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
As corresponding author, I state that all data included in the manuscript are available.