Abstract
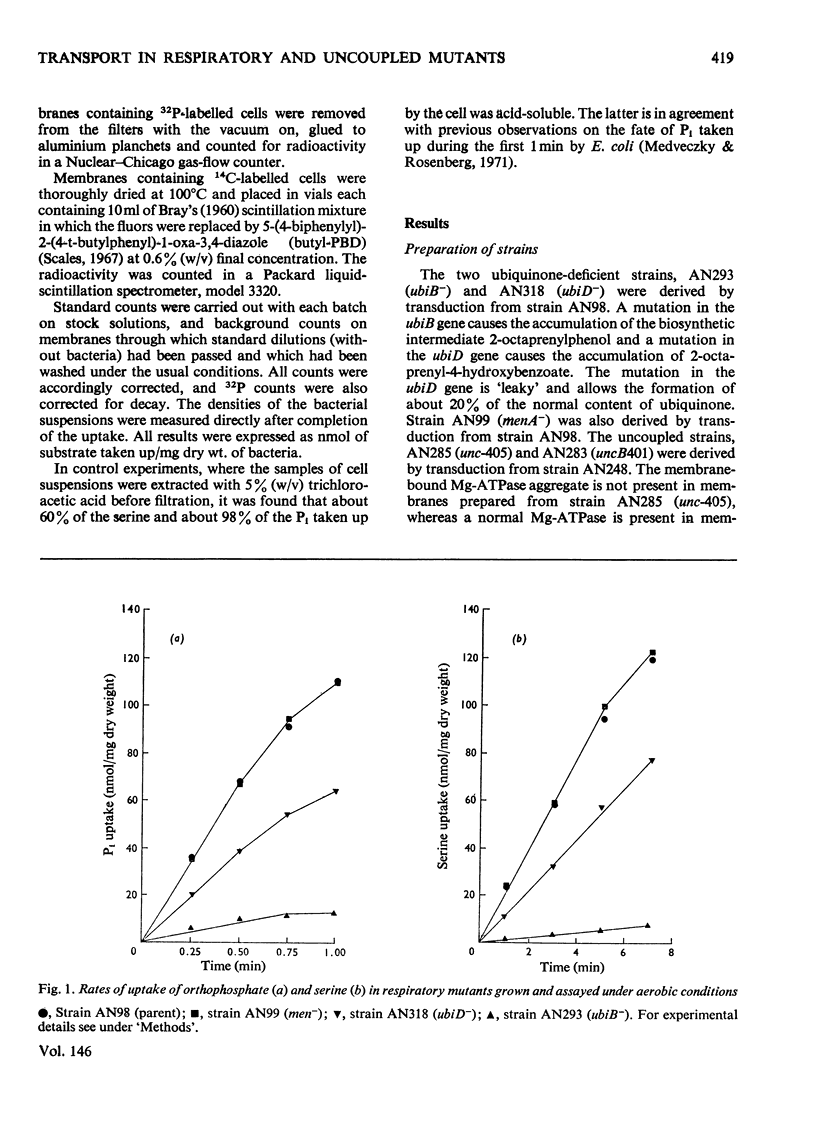
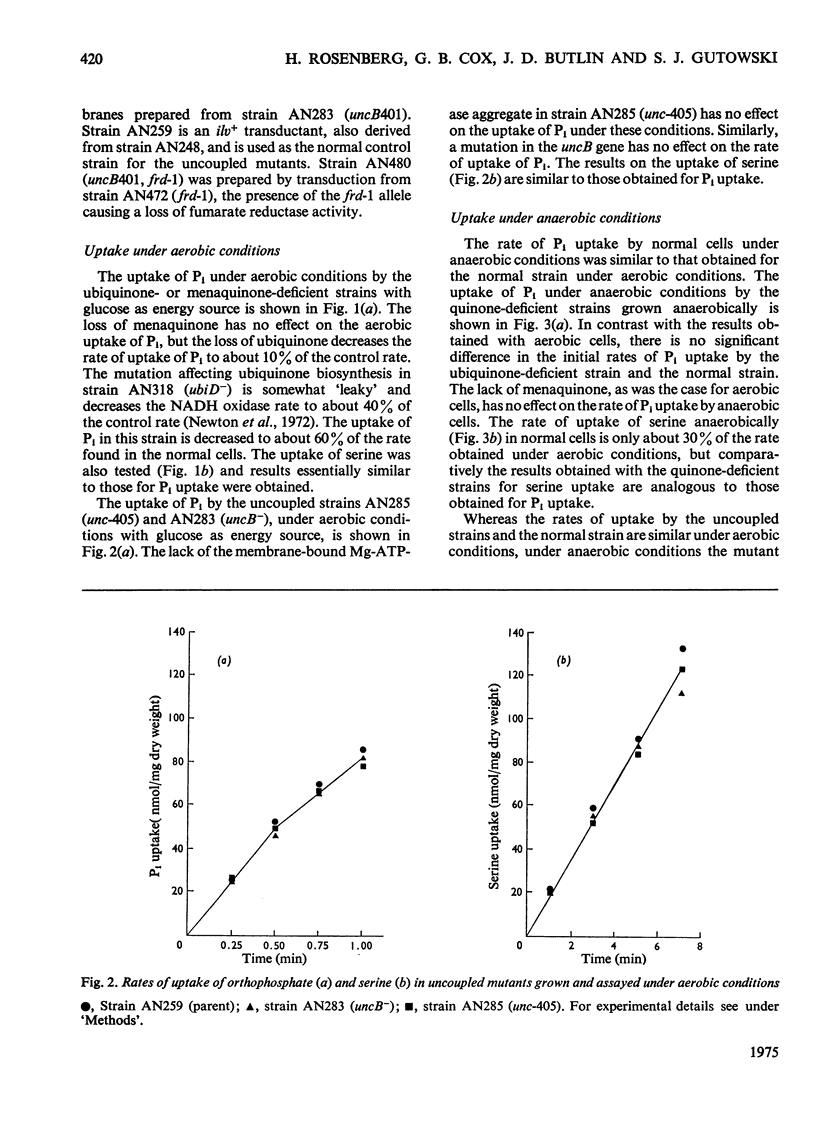
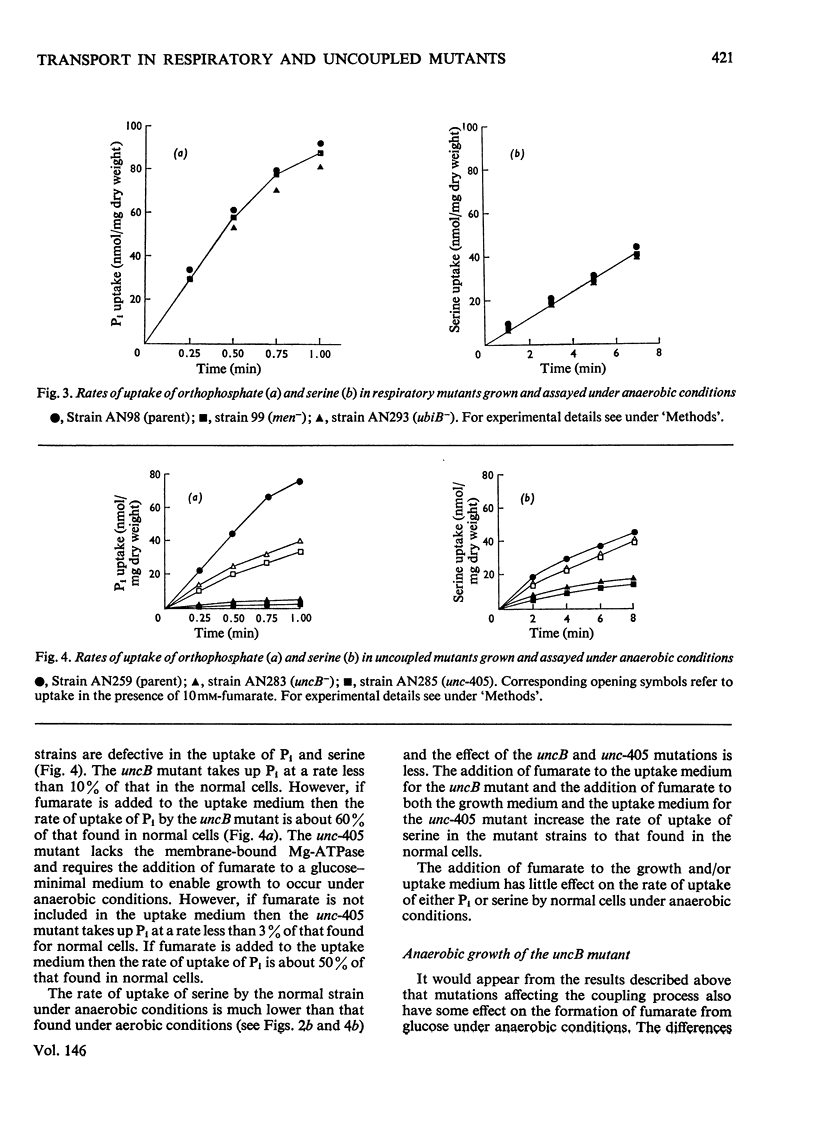
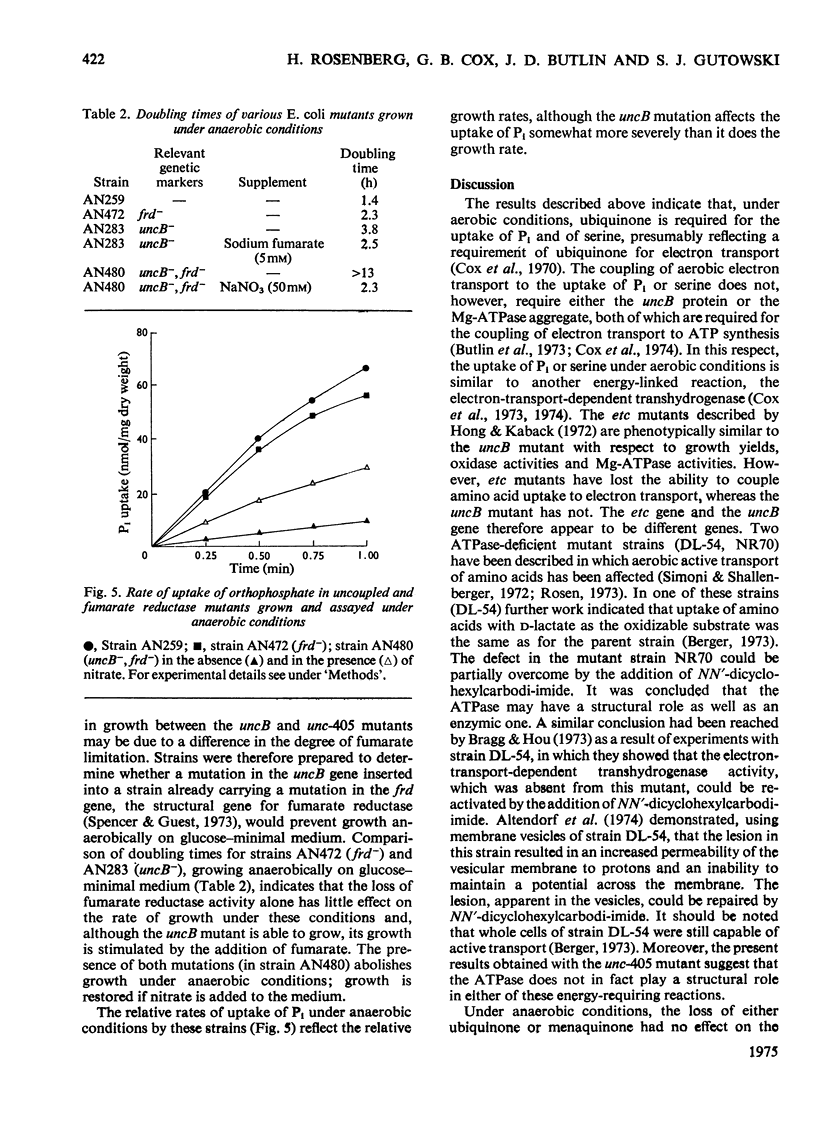
1. The uptakes of Pi and serine by whole cells of mutant strains of Escherichia coli K12, grown under both aerobic and anaerobic conditions, were studied. 2. Uptake by aerobic cells was low in a ubiquinone-less mutant but normal in two mutant strains unable to couple phosphorylation to electron transport. 3. One of these uncoupled strains, carrying the unc-405 allele, does not form a membrane-bound Mg2+-stimulated adenosine triphosphatase aggregate, and it is concluded that the Mg2+-stimulated adenosine triphosphatase does not serve a structural role in the aerobic active transport of Pi or serine. 4. The other uncoupled strain, in which aerobic uptake is unaffected, carries a mutation in the uncB gene, thus distinguishing this gene from the etc gene, previously shown to be concerned with the coupling of electron transport to active transport. 5. The uptakes of Pi and serine by anaerobic cells were normal in the ubiquinone-less mutant, but defective in both the uncoupled strains. 6. The uptake of Pi and serine by anaerobic cells of the uncB mutant could be increased by the addition of fumarate to the uptake medium. The unc-405 mutant, however, required the addition of fumarate for growth and for uptake. 7. The uncB mutant, unlike the unc-405 mutant, is able to grow anaerobically in a minimal medium with glucose as sole source of carbon. Similarly a strain carrying a mutation in the frd gene, which is the structural gene for the enzyme fumarate reductase, is able to grow anaerobically in a glucose-minimal medium. However, a mutant strain carrying mutations in both the uncB and frd genes resembles the unc-405 mutant in not being able to grow under these conditions.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Altendorf K., Harold F. M., Simoni R. D. Impairment and restoration of the energized state in membrane vesicles of a mutant of Escherichia coli lacking adenosine triphosphatase. J Biol Chem. 1974 Jul 25;249(14):4587–4593. [PubMed] [Google Scholar]
- Berger E. A. Different mechanisms of energy coupling for the active transport of proline and glutamine in Escherichia coli. Proc Natl Acad Sci U S A. 1973 May;70(5):1514–1518. doi: 10.1073/pnas.70.5.1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bragg P. D., Hou C. Reconstitution of energy-dependent transhydrogenase in ATPase-negative mutants of Escherichia coli. Biochem Biophys Res Commun. 1973 Feb 5;50(3):729–736. doi: 10.1016/0006-291x(73)91305-3. [DOI] [PubMed] [Google Scholar]
- Butlin J. D., Cox G. B., Gibson F. Oxidative phosphorylation in Escherichia coli K-12: the genetic and biochemical characterisations of a strain carrying a mutation in the uncB gene. Biochim Biophys Acta. 1973 Feb 22;292(2):366–375. doi: 10.1016/0005-2728(73)90043-1. [DOI] [PubMed] [Google Scholar]
- Cox G. B., Gibson F., McCann L. Oxidative phosphorylation in Escherichia coli K12. An uncoupled mutant with altered membrane structure. Biochem J. 1974 Feb;138(2):211–215. doi: 10.1042/bj1380211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox G. B., Gibson F., McCann L. Reconstitution of oxidative phosphorylation and the adenosine triphosphate-dependent transhydrogenase activity by a combination of membrane fractions from unCA- and uncB- mutant strains of Escherichia coli K12. Biochem J. 1973 Aug;134(4):1015–1021. doi: 10.1042/bj1341015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox G. B., Newton N. A., Gibson F., Snoswell A. M., Hamilton J. A. The function of ubiquinone in Escherichia coli. Biochem J. 1970 Apr;117(3):551–562. doi: 10.1042/bj1170551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox G. B., Young I. G., McCann L. M., Gibson F. Biosynthesis of ubiquinone in Escherichia coli K-12: location of genes affecting the metabolism of 3-octaprenyl-4-hydroxybenzoic acid and 2-octaprenylphenol. J Bacteriol. 1969 Aug;99(2):450–458. doi: 10.1128/jb.99.2.450-458.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hong J. S., Kaback H. R. Mutants of Salmonella typhimurium and Escherichia coli pleiotropically defective in active transport. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3336–3340. doi: 10.1073/pnas.69.11.3336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konings W. N., Kaback H. R. Anaerobic transport in Escherichia coli membrane vesicles. Proc Natl Acad Sci U S A. 1973 Dec;70(12):3376–3381. doi: 10.1073/pnas.70.12.3376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MONOD J., COHEN-BAZIRE G., COHN M. Sur la biosynthèse de la beta-galactosidase (lactase) chez Escherichia coli; la spécificité de l'induction. Biochim Biophys Acta. 1951 Nov;7(4):585–599. doi: 10.1016/0006-3002(51)90072-8. [DOI] [PubMed] [Google Scholar]
- Medveczky N., Rosenberg H. Phosphate transport in Escherichia coli. Biochim Biophys Acta. 1971 Aug 13;241(2):494–506. doi: 10.1016/0005-2736(71)90048-4. [DOI] [PubMed] [Google Scholar]
- Newton N. A., Cox G. B., Gibson F. Function of ubiquinone in Escherichia coli: a mutant strain forming a low level of ubiquinone. J Bacteriol. 1972 Jan;109(1):69–73. doi: 10.1128/jb.109.1.69-73.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton N. A., Cox G. B., Gibson F. The function of menaquinone (vitamin K 2 ) in Escherichia coli K-12. Biochim Biophys Acta. 1971 Jul 20;244(1):155–166. doi: 10.1016/0304-4165(71)90132-2. [DOI] [PubMed] [Google Scholar]
- Or A., Kanner B. I., Gutnick D. L. Active transport in mutants of Escherichia coli with alterations in the membrane ATPase complex. FEBS Lett. 1973 Sep 15;35(2):217–219. doi: 10.1016/0014-5793(73)80288-1. [DOI] [PubMed] [Google Scholar]
- Parnes J. R., Boos W. Energy coupling of the -methylgalactoside transport system of Escherichia coli. J Biol Chem. 1973 Jun 25;248(12):4429–4435. [PubMed] [Google Scholar]
- Prezioso G., Hong J. S., Kerwar G. K., Kaback H. R. Mechanisms of active transport in isolated bacterial membrane vesicles. XII. Active transport by a mutant of Escherichia coli uncoupled for oxidative phosphorylation. Arch Biochem Biophys. 1973 Feb;154(2):575–582. doi: 10.1016/0003-9861(73)90011-8. [DOI] [PubMed] [Google Scholar]
- ROSENBERG H., ENNOR A. H. The occurrence of free D-serine in the earthworm. Biochem J. 1961 May;79:424–428. doi: 10.1042/bj0790424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reeves J. P., Hong J. S., Kaback H. R. Reconstitution of D-lactate-dependent transport in membrane vesicles from a D-lactate dehydrogenase mutant of Escherichia coli. Proc Natl Acad Sci U S A. 1973 Jul;70(7):1917–1921. doi: 10.1073/pnas.70.7.1917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosen B. P. Beta-galactoside transport and proton movements in an adenosine triphosphatase-deficient mutant of Escherichia coli. Biochem Biophys Res Commun. 1973 Aug 21;53(4):1289–1296. doi: 10.1016/0006-291x(73)90605-0. [DOI] [PubMed] [Google Scholar]
- Scales B. A new scintillator for liquid scintillation counting. Int J Appl Radiat Isot. 1967 Jan;18(1):1–6. doi: 10.1016/0020-708x(67)90165-2. [DOI] [PubMed] [Google Scholar]
- Schairer H. U., Gruber D. Mutants of Escherichia coli K12 defective in oxidative phosphorylation. Eur J Biochem. 1973 Aug 17;37(2):282–286. doi: 10.1111/j.1432-1033.1973.tb02986.x. [DOI] [PubMed] [Google Scholar]
- Schairer H. U., Haddock B. A. -Galactoside accumulation in a Mg 2+ -,Ca 2+ -activated ATPase deficient mutant of E.coli. Biochem Biophys Res Commun. 1972 Aug 7;48(3):544–551. doi: 10.1016/0006-291x(72)90382-8. [DOI] [PubMed] [Google Scholar]
- Simoni R. D., Shallenberger M. K. Coupling of energy to active transport of amino acids in Escherichia coli. Proc Natl Acad Sci U S A. 1972 Sep;69(9):2663–2667. doi: 10.1073/pnas.69.9.2663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer M. E., Guest J. R. Isolation and properties of fumarate reductase mutants of Escherichia coli. J Bacteriol. 1973 May;114(2):563–570. doi: 10.1128/jb.114.2.563-570.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]



