Abstract
In vitro data suggest that blood-borne Listeria monocytogenes organisms enter the central nervous system (CNS) by direct invasion of endothelial cells or by cell-to-cell spread from infected phagocytes to endothelial cells. However, a role for infected phagocytes in neuroinvasion and dissemination of L. monocytogenes in vivo has not been confirmed experimentally. Experiments described here tested whether L. monocytogenes-infected peripheral blood leukocytes (PBL) circulated in bacteremic mice and could establish organ infection in vivo. A mean of 30.5% of bacteria cultured from whole blood were PBL associated, and microscopy showed that 22.2% of monocytes and 1.6% of neutrophils were infected. PBL-associated bacteria spread to endothelial cells in vitro, indicating their potential for virulence in vivo. To test this possibility, mice were injected intravenously with infected PBL and CFU of bacteria in liver, spleen, and brain were quantified and compared with values for mice injected with broth-grown bacteria and in vitro-infected macrophage cell lines. An inoculum of infected macrophage cell lines led to greater numbers of bacteria in the liver than the numbers produced by a similar inoculum of broth-grown bacteria. In contrast, brain infection was best established by infected PBL. Results of intraperitoneal injection of infected peritoneal cells compared with results of injection with infected J774A.1 cells suggested that unrestricted intracellular bacterial replication within J774A.1 cells contributed to excessive liver infection in those mice. These data show dissemination of intracellular L. monocytogenes and indicate that phagocyte-facilitated invasion has a role in CNS infection in vivo. Heterogeneity with regard to bactericidal activity as well as to other phagocyte characteristics is a critical feature of this mechanism.
Blood-borne bacteria invade the central nervous system (CNS) through a variety of mechanisms in which they interact with, invade, or disrupt cells of the blood-brain barrier (reviewed in reference 42). However, less is known about how bacteria infect the CNS through phagocyte-facilitated invasion, also know as the “Trojan horse” mechanism. This form of invasion occurs when monocytes parasitized in the periphery transport intracellular microbes across the blood-brain barrier during leukocyte trafficking. This mechanism was first identified in visna virus infection of sheep and more recently implicated in CNS infection and mother-to-fetus transmission of human immunodeficiency virus type 1 (28, 31, 40). Phagocyte-facilitated invasion also has a role in bacterial infections, as has been demonstrated for Streptococcus suis meningitis in swine (45). These examples mark intracellular parasitism and the proclivity for invading protected spaces, e.g., the CNS, as two characteristics of pathogens likely to use phagocyte-facilitated invasion and suggest that facultative intracellular bacteria that naturally infect the CNS such as Brucella sp., Listeria monocytogenes, Mycobacterium bovis, and Mycobacterium tuberculosis may use this mechanism to infect protected spaces (4, 5, 44).
L. monocytogenes is a pathogen of humans and domesticated animals that causes sepsis and a variety of CNS infections, including meningitis, meningoencephalitis, and brain abscess (19, 25, 27). Experimental L. monocytogenes infection of mice and invasion of endothelial cells have been used to study neuroinvasive mechanisms of this bacterium. CNS invasion in mice follows bacteremia, presumably due to bacterial effluence from heavily infected foci in the liver and spleen (2, 33). Similarly, rhombencephalitis in a gerbil model of chronic otitis media is associated with prolonged low-grade bacteremia (3). Accumulating data suggest that one mechanism for L. monocytogenes invasion of the CNS is by infection of endothelial cells of the blood-brain barrier. In vitro studies from our laboratory and other laboratories showed that L. monocytogenes invades endothelial cells, including brain microvascular endothelial cells, in a process that is independent of internalin and InlB in the presence of serum but that is mediated by InlB in the absence of serum (16, 21, 22, 30, 46). Bacterial invasion stimulates a robust inflammatory response marked by upregulation of surface adhesion molecules on endothelial cells and neutrophil and monocyte adhesion to them (12, 23, 46). This sequence of events most likely represents a host-protective response, as numerous studies have shown that rapid neutrophil recruitment into infected tissues is essential for host defense to L. monocytogenes infection, although the exact adhesion molecules involved are not known and probably differ among organs (reviewed in reference 43).
In addition, L. monocytogenes also infects endothelial cells by cell-to-cell spread from adherent infected mononuclear phagocytes by virtue of its intracellular life cycle of phagosomal lysis, cytoplasmic replication, and F-actin-based motility (16, 22, 41). In fact, recent in vitro data show that L. monocytogenes can infect neurons by cell-to-cell spread from adherent macrophages and that this is a more efficient process than is direct invasion of neurons (11). Neuroinvasion by phagocyte-facilitated means is also suggested by histology showing L. monocytogenes-infected mononuclear phagocytes in the CNS during meningitis in the mouse model (33). In vivo, this mechanism may allow L. monocytogenes to invade protected spaces without stimulating an inflammatory response comparable in magnitude or kinetics to that elicited by naked bacteria or a response in which bacteria are sheltered from neutrophils and monocytes attracted to inflammatory foci. The experiments described here tested whether infected phagocytes have a role in dissemination of L. monocytogenes during infection of mice.
MATERIALS AND METHODS
Bacteria.
L. monocytogenes EGD, originally obtained from G. B. Mackaness, was a gift from Priscilla Campbell (National Jewish Center, Denver, Colo.) and was stored at −70°C. For experiments, 10 μl of stock culture was inoculated into 4 ml of tryptose phosphate broth (Difco, Detroit, Mich.) and then cultured overnight at 37°C with gentle shaking.
Cells.
Human umbilical vein endothelial cells were purchased from Clonetics (San Diego, Calif.) and cultured in Clonetics medium in 96-well plates in the absence of antibiotics. Prior to experimentation they were stimulated overnight with 1.0 ng of recombinant human tumor necrosis factor alpha (TNF-α; R&D Systems, Inc., Minneapolis, Minn.) per ml. The mouse macrophage-like cell line J774A.1 (BALB/c background) was purchased from the American Type Culture Collection (Manassas, Va.). The cell line H36.12J is a clonally derived macrophage precursor hybrid made by fusion of drug-selected P388D1 cells (DBA/2 background) with Percoll-separated, proteose peptone-elicited peritoneal macrophages from (C57BL/6 × DBA/2) F1 mice (6). H36.12J cells are phagocytic, but not bactericidal, for L. monocytogenes at baseline (17). Macrophage cell lines were cultured in Dulbecco’s modified Eagle’s medium (Gibco BRL, Gaithersburg, Md.) with 10% fetal calf serum in the absence of antibiotics and were infected by coculturing 106 macrophages 1:1 with bacteria in suspension for 2 h at 37°C with end-over-end rotation. The cells were washed twice with phosphate-buffered saline (PBS) by centrifugation at 300 × g for 10 min at 4°C, and then unbound bacteria were removed by centrifuging the cells through a 1-ml layer of 30% sucrose (15). The cells were resuspended to 106/ml in PBS, and then 100 μl was injected intravenously (i.v.) into each mouse via the lateral tail vein. CFU of bacteria within macrophage cell lines were measured immediately prior to injection by lysis and serial dilution in distilled H2O and plating on agar.
Mouse infection and isolation of infected phagocytes.
Female (C57BL/6 × DBA/2) F1 mice were purchased from Jackson Laboratories (Bar Harbor, Maine) and used in experiments when they were between 8 and 16 weeks of age. They were housed in microisolator cages and given food and water ad lib. Mice were infected i.v. with 0.75 × 104 to 1.5 × 104 CFU of L. monocytogenes (1 to 2 50% lethal doses) in 0.1 ml of PBS. They were anesthetized 5 to 6 days later when they were moribund and then exsanguinated by cardiac puncture. This infection protocol typically produces bacterial loads of ≥7 log10 CFU of bacteria per liver and spleen and ≥4 log10 CFU of bacteria in the brain at the time of sacrifice. Whole blood was anticoagulated by drawing it into syringes containing 200 μl of 1.5% EDTA in PBS. Erythrocytes were lysed with 4 ml of E-lyse (Cardinal Associates, Santa Fe, N. Mex.). Peripheral blood leukocytes (PBL) were washed twice with PBS containing 1.0% bovine serum albumin plus 3.5 mM EDTA by centrifugation at 300 × g for 10 min at 4°C, and then they were centrifuged through sucrose. Volumes were measured at each step, and 10-μl aliquots were removed for quantification of bacteria by serial dilution and plating. Cellular infection was quantified with cytocentrifuge preparations of Diff-Quik (Baxter Healthcare Corp., McGaw Park, Ill.)-stained PBL by counting a minimum of 100 monocytes in random oil immersion fields as well as all infected and uninfected monocytes and polymorphonuclear leukocytes (PMN) and the bacteria they contained.
Infected mouse-resident peritoneal macrophages were obtained by intraperitoneal (i.p.) injection with 2 × 107 CFU of L. monocytogenes, and then cells were harvested by peritoneal lavage with sterile PBS 60 min later. The erythrocytes were lysed, and the leukocytes were washed, centrifuged through sucrose, and resuspended to 2.5 × 106/ml in PBS. Groups of four mice were then injected i.p. with 0.3 ml of infected peritoneal cells. CFU of bacteria within peritoneal cells were quantified immediately prior to injection into recipient mice by lysis and serial dilution. By this protocol, 10 to 20% of bacteria injected into donor mice were recovered with the peritoneal cells. Microscopy of Diff-Quik-stained cytosmears showed that bacteria were found only within peritoneal macrophages, 65 to 80% of which, an occasional PMN, contained bacteria.
Coculture of infected PBL with endothelial cells.
Transfer of L. monocytogenes from infected mouse PBL to endothelial cells was performed as previously described (16). Infected PBL were suspended in 1.5 ml of RPMI 1640 (Gibco BRL) medium containing 10% fetal calf serum plus 10 μg of gentamicin per ml to kill extracellular bacteria. Next, 100 μl was added to four sets of triplicate wells, with two sets having and two sets not having TNF-α-simulated human umbilical vein endothelial cells (HUVEC). After 1 and 18 h of coculture, the cells in individual wells were harvested and CFU of L. monocytogenes were quantified by serial dilution and plating.
RESULTS
Circulating cell-associated L. monocytogenes in vivo.
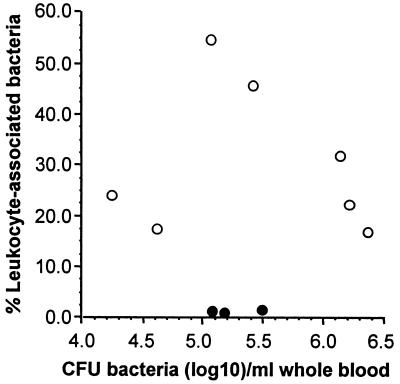
Lethal infection of mice with L. monocytogenes leads to bacteremia, presumably from heavily infected foci in the periphery (2). We used this model to test whether blood-borne L. monocytogenes could be found in association with circulating phagocytes. For this, PBL were isolated from L. monocytogenes-infected mice and CFU of bacteria in whole blood and the fraction of bacteria recovered with leukocytes were quantified. For a control, 105 CFU of L. monocytogenes were added to whole blood from uninfected mice and samples were processed in the same fashion. Figure 1 shows results from seven infected and three control mice. In infected animals, 30.5% ± 5.5% (mean ± standard error of the mean [SEM]) of CFU of L. monocytogenes present in whole blood were recovered with PBL. By comparison, only 1.4% ± 0.2% of CFU of bacteria added to uninfected blood were recovered with PBL (P = 0.01, Student’s t test). Whether the recovery rate in the control group represents bacteria that were rapidly phagocytosed or merely demonstrates the limit of detection of the washing procedure is not clear. However, the conditions under which bacteria were added to blood, i.e., low temperature, the presence of EDTA which inhibits deposition of complement on L. monocytogenes (14), and the very brief time of coincubation, probably inhibit most phagocytosis. In addition, because recovery of leukocytes will be less than 100%, whereas quantification of bacteria in whole blood is more accurate, the actual percentage of bacteria associated with cells in vivo is probably greater than we calculated.
FIG. 1.
Recovery of L. monocytogenes with PBL from mice. PBL were isolated from L. monocytogenes-infected mice (○) or from uninfected mice to whose whole blood following exsanguination 105 CFU of broth-grown bacteria were added (●). CFU of bacteria were quantified by serial dilution plating, and the percentage of leukocyte-associated bacteria was calculated as follows: (CFU of bacteria isolated with leukocytes/CFU of bacteria recovered from whole blood) × 100.
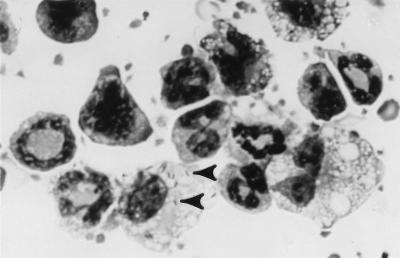
Quantitative evaluation of Diff-Quik-stained cytocentrifuge preparations of PBL from three mice showed that 21.2% ± 6.3% (mean ± SEM) of mononuclear phagocytes but that only 1.6% ± 0.5% of neutrophils were infected. In absolute numbers, infected monocytes outnumbered infected neutrophils by a mean of 9.5 ± 2.8 to 1 whereas total PMN counted outnumbered total monocytes counted by 1.8 ± 0.2 to 1. Infected monocytes contained a mean of 3.6 ± 0.5 bacteria/cell, with 9% of infected cells containing ≥10 associated bacteria. By comparison, 40% of PMN harbored ≥10 bacteria and 40% contained only 1 bacterium; however, this observation is based on small numbers of infected PMN. Microscopy also showed that the monocytes were highly vacuolated, looking more like elicited peritoneal macrophages than quiescent blood monocytes (Fig. 2). These results demonstrate that L. monocytogenes circulates in vivo cell free and in association with mononuclear phagocytes and PMN.
FIG. 2.
L. monocytogenes-infected peripheral blood mononuclear phagocytes. PBL were isolated from L. monocytogenes-infected mice. Cytocentrifuge preparations were made and stained with Diff-Quik and then evaluated by light microscopy under oil immersion. Arrowheads show bacteria.
Phagocyte-associated bacteria can invade endothelial cells in vitro and establish infection in vivo.
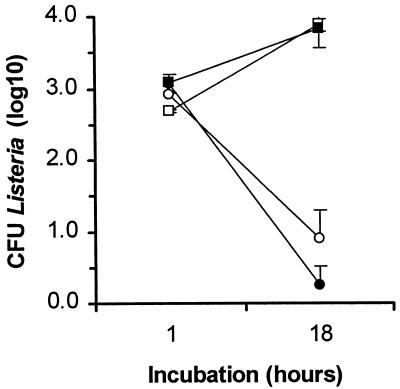
Cell-associated bacteria identified microscopically and cultured after lysis of cells and plating on agar may or may not be able to propagate an infection. Thus, we tested whether L. monocytogenes from infected PBL were capable of spreading to endothelial cells because this action depends upon coordinated expression of L. monocytogenes virulence genes resulting in phagosomal lysis and F-actin-based motility (32, 41). Infected PBL were incubated with endothelial cell monolayers in medium containing 10 μg of gentamicin per ml to kill extracellular bacteria as previously described (16). L. monocytogenes CFU remaining with the monolayer were quantified after 1 and 18 h of coculture. In two identical experiments there was net bacterial growth in wells with leukocyte-endothelial cocultures that was indicative of cell-to-cell spread from phagocytes to endothelial cells, but there was net killing of bacteria in wells without endothelial cells (Fig. 3). Wells containing only PBL were not completely sterilized, suggesting that some intracellular PBL-associated bacteria were protected from gentamicin throughout the assay. These data showed that L. monocytogenes organisms within PBL were capable of cell-to-cell spread and suggested that they could function as Trojan horses in the whole animal.
FIG. 3.
L. monocytogenes from infected PBL can spread to endothelial cells. PBL were isolated from L. monocytogenes-infected mice. They were cocultured with (□, ■) and without (○, ●) monolayers of TNF-α-stimulated endothelial cells for 1 and 18 h in the presence of 10 μg of gentamicin per ml. Results presented are the mean log10 CFU of bacteria/well (±SEM) from triplicate wells from two identical experiments (open and filled symbols indicate the separate results of the two experiments).
Phagocyte-associated bacteria can establish organ infection in vivo.
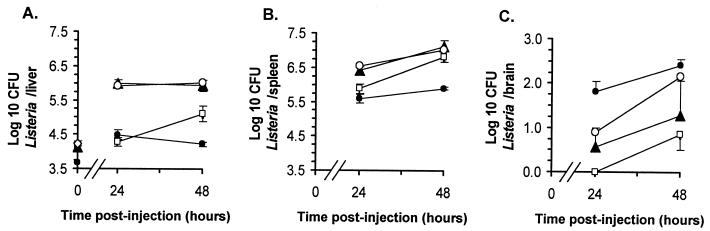
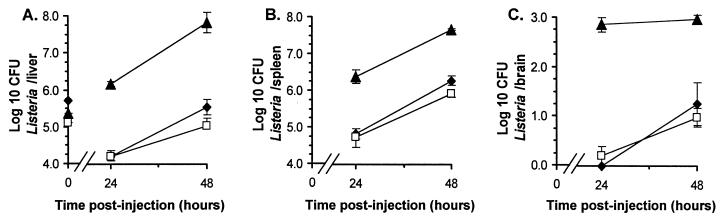
To test whether bacteria within infected phagocytes could establish organ infection in vivo, infected PBL were injected into mice i.v. and L. monocytogenes CFU in livers, spleens, and brains were quantified 24 and 48 h later. For a comparison, other mice were injected with bacteria from overnight broth culture or with the mouse macrophage cell lines J774A.1 and H36.12J infected in vitro. Figure 4 shows that infection was established in recipient mice following injection of infected PBL and infected cell lines. Bacterial loads in spleens were comparable between the four groups; however, there were notable differences in the distributions of bacteria in the liver and brain. Early infection in the liver was heaviest following injection of macrophage cell lines, which produced >1.6 log10 more bacteria/liver than were produced in mice injected with similar numbers of broth-grown bacteria. In contrast, brain infection was greatest following injection of infected PBL, from which 1.7 log10 bacteria were recovered compared with none from mice injected with a somewhat greater amount of broth-grown bacteria. By 48 h postinfection the numbers of organisms in the livers and spleens were similar in mice injected with bacteria and mice injected with the cell lines; however, there remained a striking difference in levels of brain infection between mice injected with PBL and those injected with broth-grown bacteria. Notably, the different macrophage cell lines produced very similar bacterial loads in all organs, suggesting that genetic differences between the cell lines and recipient mice were not a factor in these experiments.
FIG. 4.
Phagocyte-associated bacteria can establish systemic infection of mice. PBL were isolated from L. monocytogenes-infected mice. The J774A.1 and H36.12J macrophage cell lines were infected by 2-h incubations with L. monocytogenes. Infected PBL (●; 3.68 log10 CFU [per mouse]), J774A.1 cells (▴; 4.13 log10 CFU), and H36.12J cells (○; 4.23 log10 CFU) or broth-grown bacteria (□; 4.11 log10 CFU) were injected i.v. into mice, and CFU of bacteria in liver (A), spleen (B), and brain (C) were quantified 24 and 48 h later. CFU bacteria in each inoculum (log10/mouse) are given with the symbols described above at time zero in panel A. Results shown are the mean log10 CFU of bacteria per organ ±SEM from three or four mice. Error bars not shown are smaller than the symbol.
One characteristic that may influence the bacterial load in an organ following injection of infected phagocytes is the underlying bactericidal activity of the phagocyte. To test this, we used resident peritoneal macrophages as a model primary cell since they have a greater ability to restrict L. monocytogenes growth than the macrophage cell lines. For this, mice were infected by i.p. injection of infected peritoneal macrophages, infected J774A.1 cells, or broth-grown bacteria and CFU of bacteria were quantified in liver, spleen, and brain 24 and 48 h later (Fig. 5). Bacterial loads in the liver and spleen were much greater following injection of parasitized J774A.1 cells than following injection of infected peritoneal macrophages or broth-grown bacteria. Interestingly, organ counts following injection of plain bacteria and infected peritoneal macrophages were nearly identical despite the fact that the latter received a fourfold greater inoculum of bacteria. In addition, mice injected i.p. with infected H36.12J cells containing 6.0 log10 CFU of L. monocytogenes developed rapid and overwhelming infection in each organ, comparable to infection in mice injected with J774A.1 cells containing a similar amount of bacteria (data not shown). Somewhat surprising was the early CNS infection following i.p. injection of infected J774A.1 cells. It is possible that this early CNS infection represents more rapid trafficking of the cells to the CNS than following i.v. infection but it is more likely that it is the result of heavy parasitism of draining lymphoid tissues and of secondary spread of bacteria from these areas to the brain.
FIG. 5.
Intraperitonal injection of infected cells leads to bacterial dissemination. J774A.1 cells were infected by 2-h incubation with L. monocytogenes, and peritoneal cells were harvested by peritoneal lavage from mice 60 min following i.p. injection with 107 L. monocytogenes organisms. Infected J774A.1 cells (▴; 5.37 log10 CFU) and peritoneal cells (⧫; 5.72 log10 CFU) or broth-grown bacteria (□; 5.11 log10 CFU) were injected i.p. into mice, and CFU of bacteria in liver (A), spleen (B), and brain (C) were quantified 24 and 48 h postinjection. CFU of bacteria in each inoculum (log10 per mouse) are shown with the symbols described above at time zero in panel A. Results shown are the mean log10 CFU of bacteria/organ ± SEM from groups of four mice. Error bars not shown are smaller than the symbol.
DISCUSSION
These experiments show that cell-associated as well as cell-free L. monocytogenes organisms are present in infected mice. Circulating infected mononuclear monocytes have an appearance similar to that of activated cells as opposed to that of quiescent blood monocytes that have incidentally phagocytosed bacteria. Bacteria associated with infected PBL were virulent, as evidenced by spread to endothelial cells in vitro and the establishment of organ infection, including CNS infection, in vivo. These findings are consistent with the idea that infected phagocytes participate in dissemination and invasion of protected spaces by L. monocytogenes. Whether organ infection via parasitized phagocytes is the result of leukocyte transmigration and bacterial spread directly to parenchymal tissues or whether bacteria from adherent phagocytes first infect the endothelium and then spread to deeper tissues is not known. It is possible that both events take place in vivo depending upon the dynamics of intracellular parasitism and monocyte transmigration (26). In addition, data suggest that populations of mononuclear phagocytes differ in their abilities to function as Trojan horses and promote infection of specific organs.
Parasitized PBL were isolated from mice infected by i.v. injection of bacteria. Whether oral infection with L. monocytogenes would lead to similar findings or would also involve dissemination by cell-associated bacteria was not tested. Nevertheless, there is data suggesting that this is likely. Oral infection of rodents shows that Peyer’s patches are the initial sites of L. monocytogenes replication, and electron microscopy revealed bacteria in the cytoplasm of macrophages and PMN, indicating productive intracellular parasitism (24, 35). Bacteria then spread to the mesenteric lymph node, liver, and spleen through a poorly defined process, but a role for dissemination of intracellular bacteria within dendritic cells has been hypothesized (35). Presumably, if sufficiently high bacterial loads develop in secondarily infected foci, this would lead to a situation similar to that following i.v. infection. However, adult mice are quite resistant to very large gastrointestinal inocula of L. monocytogenes and systemic replication sufficient to produce bacteremia and circulating infected phagocytes may be found only in mice compromised by young age or from administration of immunosuppressive drugs or monoclonal antibodies (7, 10, 29).
Microscopy of infected PBL showed L. monocytogenes associated with monocytes and PMN. Theoretically, either type of cell may transmit infection, but it is more likely that infected monocytes serve this purpose. This is because inflammatory PMN are more listericidal and less heterogeneous in this function than are inflammatory macrophages (9, 18). However, the presence of cytoplasmic L. monocytogenes within PMN following oral infection and data showing that PMN can transport Yersinia enterocolitica across the endothelium suggest that trafficking of bacteria within PMN is possible (35, 38).
Differences in bactericidal activities between the cell lines and primary cells used in these experiments likely contributed to differences in bacterial loads following their injection into mice. The J774A.1 and H36.12J cell lines are permissive of intracellular growth, whereas PBL and peritoneal macrophages are less permissive cells or are less able to conceal L. monocytogenes from neutrophils, such as through a lack of internalization of bound bacteria (13). Thus, bacteria within the cell lines replicated continuously and were sheltered from recruited PMN following their injection into mice (20). In addition to bactericidal activity, other leukocyte characteristics such as expression of chemokine receptors or adhesion molecules may influence the spread of bacteria to specific organs. For example, recent data show that immature and mature dendritic cells display distinct patterns of chemokine receptors and chemokine responsiveness (reviewed in reference 39). This is hypothesized to expedite localization of immature dendritic cells to inflammatory sites and then to allow reverse migration of mature cells from inflammatory foci to lymphoid organs (36). Additionally, selective expression of endothelial cell adhesion molecules can mediate recruitment of specific leukocyte populations from blood (37). Thus, differential levels of expression of chemokine receptors and/or endothelial cell adhesion molecule counter receptors on different populations of infected phagocytes may affect phagocyte homing and, in turn, the distribution of bacteria after dissemination of infected phagocytes.
These data do not dismiss a role for neuroinvasion by cell-free L. monocytogenes. Blood-borne bacteria may be more efficient in this process than broth-grown bacteria due to differential growth characteristics, bacterial adhesins expressed only in vivo, the participation of serum proteins, or altered interactions between bacteria and the blood-brain barrier as a result of the inflammatory milieu (1, 8, 22, 34). Additionally, neuroinvasion may represent a synergistic event between cell-free and intracellular bacteria. Recent data from our laboratory show that L. monocytogenes infection of human and porcine brain microvascular endothelial cells stimulates monocyte adhesion (reference 31 and unpublished data). Thus, activation of microvascular cells during bacteremic L. monocytogenes infection may facilitate binding and/or transmigration of parasitized monocytes and consequently promote CNS infection. It seems likely that the combined actions of cell-free and cell-associated bacteria, and the potential for either to establish infection, contribute to the ability of L. monocytogenes to invade the CNS from the bloodstream.
ACKNOWLEDGMENTS
The expert technical assistance of Sandra Wilson and the critical reading of the manuscript by Ronald Greenfield are gratefully acknowledged.
REFERENCES
- 1.Badger J L, Kim K S. Environmental growth conditions influence the ability of Escherichia coli K1 to invade brain microvascular endothelial cells and confer serum resistance. Infect Immun. 1998;66:5692–5697. doi: 10.1128/iai.66.12.5692-5697.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Berche P. Bacteremia is required for invasion of the murine central nervous system by Listeria monocytogenes. Microb Pathog. 1995;18:323–326. doi: 10.1006/mpat.1995.0029. [DOI] [PubMed] [Google Scholar]
- 3.Blanot S, Joly M M, Vilde F, Jaubert F, Clement O, Frija G, Berche P. A gerbil model for rhombencephalitis due to Listeria monocytogenes. Microb Pathog. 1997;23:39–48. doi: 10.1006/mpat.1997.0131. [DOI] [PubMed] [Google Scholar]
- 4.Bolin C A, Whipple D L, Khanna K V, Risdahl J M, Peterson P K, Molitor T M. Infection of swine with Mycobacterium bovis as a model of human tuberculosis. J Infect Dis. 1997;176:1559–1566. doi: 10.1086/514155. [DOI] [PubMed] [Google Scholar]
- 5.Bouza E, Garcia de la Torre M, Parras F, Guerrero A, Rodríguez-Créixems M, Gobernado J. Brucellar meningitis. Rev Infect Dis. 1987;9:810–822. doi: 10.1093/clinids/9.4.810. [DOI] [PubMed] [Google Scholar]
- 6.Canono B P, Campbell P A. Production of mouse inflammatory macrophage hybridomas. J Tissue Cult Methods. 1992;14:3–8. [Google Scholar]
- 7.Conlan J W. Neutrophils and tumour necrosis factor-alpha are important for controlling early gastrointestinal stages of experimental murine listeriosis. J Med Microbiol. 1997;46:239–250. doi: 10.1099/00222615-46-3-239. [DOI] [PubMed] [Google Scholar]
- 8.Cundell D R, Gerard N P, Gerard C, Idanpaan-Heikkila I, Tuomanen E I. Streptococcus pneumoniae anchor to activated human endothelial cells by the receptor for platelet activating factor. Nature. 1995;377:435–438. doi: 10.1038/377435a0. [DOI] [PubMed] [Google Scholar]
- 9.Czuprynski C J, Henson P M, Campbell P A. Killing of Listeria monocytogenes by inflammatory neutrophils and mononuclear phagocytes from immune and nonimmune mice. J Leukoc Biol. 1984;35:193–208. doi: 10.1002/jlb.35.2.193. [DOI] [PubMed] [Google Scholar]
- 10.Czuprynski C J, Theisen C, Brown J F. Treatment with the antigranulocyte monoclonal antibody RB6-8C5 impairs resistance of mice to gastrointestinal infection with Listeria monocytogenes. Infect Immun. 1996;64:3946–3949. doi: 10.1128/iai.64.9.3946-3949.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Dramsi S, Lévi S, Triller A, Cossart P. Entry of Listeria monocytogenes into neurons occurs by cell-to-cell spread: an in vitro study. Infect Immun. 1998;66:4461–4468. doi: 10.1128/iai.66.9.4461-4468.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Drevets D A. Listeria monocytogenes infection of cultured endothelial cells stimulates neutrophil adhesion and adhesion molecule expression. J Immunol. 1997;158:5305–5313. [PubMed] [Google Scholar]
- 13.Drevets D A, Campbell P A. Macrophage phagocytosis: use of fluorescence microscopy to distinguish between extracellular and intracellular bacteria. J Immunol Methods. 1991;142:31–38. doi: 10.1016/0022-1759(91)90289-r. [DOI] [PubMed] [Google Scholar]
- 14.Drevets D A, Campbell P A. The roles of complement and complement receptor type 3 in phagocytosis of Listeria monocytogenes by inflammatory mouse peritoneal macrophages. Infect Immun. 1991;59:2645–2652. doi: 10.1128/iai.59.8.2645-2652.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Drevets D A, Leenen P J M, Campbell P A. Complement receptor type 3 (CD11b/CD18) involvement is essential for killing of Listeria monocytogenes by mouse macrophages. J Immunol. 1993;151:5431–5439. [PubMed] [Google Scholar]
- 16.Drevets D A, Sawyer R T, Potter T A, Campbell P A. Listeria monocytogenes infects human endothelial cells by two distinct mechanisms. Infect Immun. 1995;63:4268–4276. doi: 10.1128/iai.63.11.4268-4276.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fleming S D, Campbell P A. Macrophages have cell surface IL-10 that regulates bactericidal activity. J Immunol. 1996;156:1143–1150. [PubMed] [Google Scholar]
- 18.Fleming S D, Campbell P A. Some macrophages kill Listeria monocytogenes while others do not. Immunol Rev. 1997;158:69–77. doi: 10.1111/j.1600-065x.1997.tb00993.x. [DOI] [PubMed] [Google Scholar]
- 19.Gray M L, Killinger A H. Listeria monocytogenes and listeric infections. Bacteriol Rev. 1966;30:309–382. doi: 10.1128/br.30.2.309-382.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gregory S H, Sagnimeni A J, Wing E J. Bacteria in the bloodstream are trapped in the liver and killed by immigrating neutrophils. J Immunol. 1996;157:2514–2520. [PubMed] [Google Scholar]
- 21.Greiffenberg L, Sokolovic Z, Schnittler H J, Spory A, Böckmann R, Goebel W, Kuhn M. Listeria monocytogenes-infected human umbilical vein endothelial cells: internalin-independent invasion, intracellular growth, movement, and host response. FEMS Microbiol Lett. 1997;157:163–170. doi: 10.1111/j.1574-6968.1997.tb12768.x. [DOI] [PubMed] [Google Scholar]
- 22.Greiffenberg L, Goebel W, Kim K S, Weiglein I, Bibert A, Engelbrecht F, Stins M, Kuhns M. Interaction of Listeria monocytogenes with human brain microvascular endothelial cells: InlB-dependent invasion, long-term intracellular growth, and spread from macrophages to endothelial cells. Infect Immun. 1998;66:5260–5267. doi: 10.1128/iai.66.11.5260-5267.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Krüll M, Nöst R, Hippenstiel S, Domann E, Chakraborty T, Suttorp N. Listeria monocytogenes potently induces up-regulation of endothelial adhesion molecules and neutrophil adhesion to cultured human endothelial cells. J Immunol. 1997;159:1970–1976. [PubMed] [Google Scholar]
- 24.Marco A J, Altimira J, Prats N, López S, Dominguez L, Domingo M, Briones V. Penetration of Listeria monocytogenes in mice infected by the oral route. Microb Pathog. 1997;23:255–263. doi: 10.1006/mpat.1997.0144. [DOI] [PubMed] [Google Scholar]
- 25.McLauchlin J. Human listeriosis in Britain, 1967–85, a summary of 722 cases. 2. Listeriosis in non-pregnant individuals, a changing pattern of infection and seasonal incidence. Epidemiol Infect. 1990;104:191–201. doi: 10.1017/s0950268800059355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Muller W A, Weigl S A. Monocyte-selective transendothelial migration: dissection of the binding and transmigration phases by an in vitro assay. J Exp Med. 1992;176:819–828. doi: 10.1084/jem.176.3.819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mylonakis E, Hohmann E L, Calderwood S B. Central nervous system infection with Listeria monocytogenes. Medicine. 1998;77:313–336. doi: 10.1097/00005792-199809000-00002. [DOI] [PubMed] [Google Scholar]
- 28.Nottet H S L M, Gendelman H E. Unraveling the neuroimmune mechanisms for the HIV-1-associated cognitive/motor complex. Immunol Today. 1995;16:441–448. doi: 10.1016/0167-5699(95)80022-0. [DOI] [PubMed] [Google Scholar]
- 29.Okamoto M, Nakane A, Minagawa T. Host resistance to intragastric infection with Listeria monocytogenes in mice depends on cellular immunity and intestinal bacterial flora. Infect Immun. 1994;62:3080–3085. doi: 10.1128/iai.62.8.3080-3085.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Parida S K, Domann E, Rohde M, Müller S, Darji A, Hain T, Wehland J, Chakraborty T. Internalin B is essential for adhesion and mediates the invasion of Listeria monocytogenes into human endothelial cells. Mol Microbiol. 1998;28:81–93. doi: 10.1046/j.1365-2958.1998.00776.x. [DOI] [PubMed] [Google Scholar]
- 31.Peluso R, Haase A, Strowring L, Edwards M, Venturs P. A Trojan horse mechanism for the spread of visna virus in monocytes. Virology. 1985;147:231–236. doi: 10.1016/0042-6822(85)90246-6. [DOI] [PubMed] [Google Scholar]
- 32.Portnoy D A, Chakraborty T, Goebel W, Cossart P. Molecular determinants of Listeria monocytogenes pathogenesis. Infect Immun. 1992;60:1263–1267. doi: 10.1128/iai.60.4.1263-1267.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Prats N, Briones V, Blanco M M, Altimira J, Ramos J A, Dominguez L, Marco A. Choroiditis and meningitis in experimental murine infection with Listeria monocytogenes. Eur J Clin Microbiol Infect Dis. 1992;11:744–747. doi: 10.1007/BF01989983. [DOI] [PubMed] [Google Scholar]
- 34.Pron B, Taha M-K, Rambaud C, Fournet J-C, Pattey N, Monnet J-P, Musilek M, Beretti J-L, Nassif X. Interaction of Neisseria meningitidis with the components of the blood-brain barrier correlates with an increased expression of PilC. J Infect Dis. 1997;176:1285–1292. doi: 10.1086/514124. [DOI] [PubMed] [Google Scholar]
- 35.Pron B, Boumaila C, Jaubert F, Sarnacki S, Monnet J-P, Berche P, Gaillard J-L. Comprehensive study of the intestinal stage of listeriosis in a rat ligated ileal loop system. Infect Immun. 1998;66:747–755. doi: 10.1128/iai.66.2.747-755.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Randolph G J, Beaulieu S, Lebecque S, Steinman R M, Muller W A. Differentiation of monocytes into dendritic cells in a model of transendothelial trafficking. Science. 1998;282:480–483. doi: 10.1126/science.282.5388.480. [DOI] [PubMed] [Google Scholar]
- 37.Reinhardt P H, Kubes P. Differential leukocyte recruitment from whole blood via endothelial adhesion molecules under shear conditions. Blood. 1998;92:4691–4699. [PubMed] [Google Scholar]
- 38.Rüssmann H, Ruckdeschel K, Heesemann J. Translocation of Yersinia enterocolitica through an endothelial monolayer by polymorphonuclear leukocytes. Infect Immun. 1996;64:1016–1019. doi: 10.1128/iai.64.3.1016-1019.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sallussto F P, Lanzavecchia A. Mobilizing dendritic cells for tolerance, priming, and chronic inflammation. J Exp Med. 1999;189:611–614. doi: 10.1084/jem.189.4.611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Schwartz D H, Sharma U K, Perlman E J, Blakemore K. Adherence of human immunodeficiency virus-infected lymphocytes to fetal placental cells: a model of maternal → fetal transmission. Proc Natl Acad Sci USA. 1995;92:978–982. doi: 10.1073/pnas.92.4.978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Tilney L T, Portnoy D A. Actin filaments and the growth, movement, and spread of the intracellular bacterial parasite, Listeria monocytogenes. J Cell Biol. 1989;109:1597–1608. doi: 10.1083/jcb.109.4.1597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tuomanen E. Entry of pathogens into the central nervous system. FEMS Microbiol Rev. 1996;18:289–299. doi: 10.1111/j.1574-6976.1996.tb00245.x. [DOI] [PubMed] [Google Scholar]
- 43.Unanue E R. Inter-relationship among macrophages, natural killer cells and neutrophils in early stages of Listeria resistance. Curr Opin Immunol. 1997;9:35–43. doi: 10.1016/s0952-7915(97)80156-2. [DOI] [PubMed] [Google Scholar]
- 44.Verdun R, Chevret S, Laissy J-P, Wolff M. Tuberculous meningitis in adults: review of 48 cases. J Infect Dis. 1996;22:982–988. doi: 10.1093/clinids/22.6.982. [DOI] [PubMed] [Google Scholar]
- 45.Williams A E, Blakemore W F. Pathogenesis of meningitis caused by Streptococcus suis type 2. J Infect Dis. 1990;162:474–481. doi: 10.1093/infdis/162.2.474. [DOI] [PubMed] [Google Scholar]
- 46.Wilson S L, Drevets D A. Listeria monocytogenes infection and activation of human brain microvascular endothelial cells. J Infect Dis. 1998;178:1658–1666. doi: 10.1086/314490. [DOI] [PubMed] [Google Scholar]