Abstract
The degrees of competence of opaque and transparent colony variants of Streptococcus pneumoniae were compared. The transparent variants were transformed at 9- to 670-fold-higher rates than the opaque variants, independent of the DNA incorporated, due to decreased expression of capsular polysaccharide. Genetic transformation, therefore, tends to select for a less-encapsulated subpopulation.
Streptococcus pneumoniae, the pneumococcus, is capable of considerable interstrain heterogeneity, as evidenced by its ability to express at least 90 unique types of its major virulence determinant, the capsular polysaccharide. The virulence of the pneumococcus has been shown to be highly sensitive to relatively small differences in amounts of capsular polysaccharide between strains of the same type (7). In addition, there is marked intrastrain variation which is apparent in most isolates as differences between opaque and transparent phenotypes when colonies are viewed on translucent surfaces rather than blood agar (19, 20). Transparent variants show increased adherence to human epithelial cells and are selected for during nasopharyngeal carriage in animal models of carriage but are unable to cause sepsis (2, 4). In comparison to opaque forms, the transparent variants have greater amounts of cell wall teichoic acid (C-polysaccharide) and an altered distribution of proteins anchored to the choline moiety on the teichoic acid (14, 18). Opaque pneumococci, in contrast, express from 2.5- to 22-fold more cell-associated capsular polysaccharide, as measured by capture enzyme-linked immunosorbent assay, than do transparent variants of the same strain (5). The increased expression of capsular polysaccharide by opaque pneumococci (i) hinders cytoadherence, which may account for their inefficiency at colonization of the nasopharynx, and (ii) increases resistance to opsonophagocytosis, which may explain their greater virulence in a murine model of sepsis (4, 5, 13). An isolate of S. pneumoniae, therefore, should be considered a mixed population of phenotypes which differ in amounts of capsular polysaccharide, teichoic acid, and choline-binding proteins.
It has long been recognized that encapsulation reduces the natural competence of the pneumococcus for genetic transformation (12, 17). Many encapsulated strains have the potential to become competent when treated with sufficient amounts of competence factor, suggesting that the capsular polysaccharide acts as a barrier that prevents this factor from reaching its cellular target (21). The recent identification of quorum-sensing pheromones which act to induce competence has greatly facilitated the molecular analysis of encapsulated strains for virulence studies (3, 10, 16). Many encapsulated strains are rendered competent by either of two related 17-residue competence-stimulating peptides, CSP1 or CSP2, depending on whether the isolate carries the comC1 or comC2 allele (11). The purpose of this report was to determine the effect of variation in colony phenotype and intrastrain heterogeneity in the quantity of capsular polysaccharide on pneumococcal competence.
The degrees of competence of opaque and transparent variants of three genetically unrelated clinical isolates of types 6B, 6A, and 23F were compared (Table 1). After the colony morphology was confirmed as previously described, cells with >99% similarity to the desired phenotype were grown in C+Y medium, pH 8.0, to an optical density at 620 nm of 0.15 and were transformed by the method of Lacks (6, 20). In all transformation experiments prior to the addition of DNA, the cells were incubated for 15 min with 50 ng of synthetic CSP1 (for type 6B and mutants or variants related to D39) or CSP2 (for type 6A and 23F) per ml. The two unrelated genetic markers used in transformation experiments included (i) chromosomal DNA obtained from a spontaneous streptomycin-resistant mutant of S. pneumoniae R6 and (ii) plasmid DNA purified from Escherichia coli containing an insert with a cloned pneumococcal XbaI-like methylase gene isolated from strain P314 which was interrupted by insertion of a kanamycin resistance cassette (Ωkm-2) within the insert (9). Introduction of the plasmid, which cannot replicate in the pneumococcus, results in a chromosomal insertion event as a result of homologous recombination within the methylase gene. The frequency of transformation was determined by comparing the number of colonies in the presence (transformants) and absence (transformants plus nontransformants) of streptomycin (200 μg/ml) for chromosomal DNA or kanamycin (300 μg/ml) for plasmid DNA. In preliminary experiments, the streptomycin resistance marker was transformed from the R6 genetic background into both opaque and transparent variants of P314. Streptomycin-resistant mutants of P314 variants were then used as a source of chromosomal DNA isolated as previously described (8). There was, however, no difference in transformation frequency related to the genetic background or opacity phenotype of the chromosomal DNA used (data not shown). Genetic transformation of the encapsulated strains was rare in the absence of CSP1 or CSP2. In the presence of CSP1 or CSP2 the difference in transformation frequency between opaque and transparent variants was unaffected by the cell density at the time the DNA was added (data not shown).
TABLE 1.
Pneumococcal strains used in this study
| Strain | Characteristic(s) | Capsular polysaccharide typea | Source or reference |
|---|---|---|---|
| D39 | Clinical isolate (opaque variant) | 2 | 1 |
| R6 | D39, spontaneous unencapsulated variant | None | 1 |
| P125 | R6, opaque variant | None | 18 |
| P126 | R6, transparent variant | None | 18 |
| P138 | R6 × A66 DNA (opaque variant) | 3 | 15 |
| P314 | Clinical isolate | 6B | R. Austrian |
| P763 | P314, opaque variant | 6B | 4 |
| P765 | P314, transparent variant | 6B | 4 |
| P806 | P765, spontaneous phase variant to an opaque phenotype | 6B | 5 |
| P303 | Clinical isolate | 6A | R. Austrian |
| P376 | P303, opaque variant | 6A | 4 |
| P384 | P303, transparent variant | 6A | 4 |
| P831 | Clinical isolate | 23F | T. Cate |
| P833 | P831, transparent variant | 23F | This study |
| P861 | P831, opaque variant | 23F | This study |
The presence and type of capsular polysaccharide were confirmed by the Quellung reaction using type-specific antiserum (Statens Serum Institut, Copenhagen, Denmark).
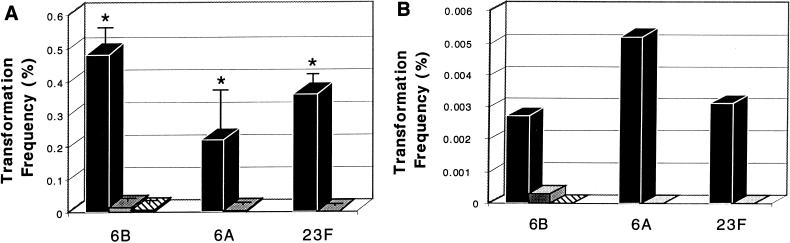
There was a strong correlation between colony phenotype and competence for genetic transformation for each of the three types tested. The transformation frequencies were an average of 34-, 54-, and 670-fold higher for transparent variants than for opaque variants of the same strain for type 6B, 6A, and 23F isolates, respectively, when chromosomal DNA was used (Fig. 1A). This difference was independent of the specific marker used or the origin of the DNA, since transformation frequencies were also higher (9- to 52-fold) for transparent variants than for opaque variants when plasmid DNA was used. Rates of transformation with plasmid DNA, however, were uniformly lower than with chromosomal DNA (Fig. 1B). P806, an opaque phase variant of the highly competent transparent P765, could no longer be transformed at a high frequency, confirming the association between colony phenotype and competence. Similar differences in transformation frequency were observed when opaque and transparent variants of strain P314 were mixed and treated together to induce competence. After addition of chromosomal DNA, 75.6% of the streptomycin-sensitive cells (nontransformants) were opaque, whereas only 1.6% of streptomycin-resistant cells (transformants) were opaque. The experiment with a mixed population showed that under identical conditions the opaque form was less competent and that no factors from transparent pneumococci could overcome the relative incompetence of opaque pneumococci derived from the same strain.
FIG. 1.
Comparison of the frequencies of genetic transformation in encapsulated variants. Isolates of the three types indicated below were divided into opaque (gray bars) and transparent (solid bars) phenotypes. After treatment to induce competence with CSP, the transformation frequency [number of transformants/(number transformants + nontransformants)] with chromosomal DNA (A) or plasmid DNA (B) was determined. For the type 6B isolate a spontaneous opaque revertant (hatched bars) of the transparent variant was also tested. Values are the means of duplicate determinations of at least two independent experiments (n) ± standard deviations (error bars) when n ≥ 3). Asterisks represent significant differences (P < 0.001; Student’s t test) in transformation frequency between opaque and transparent variants.
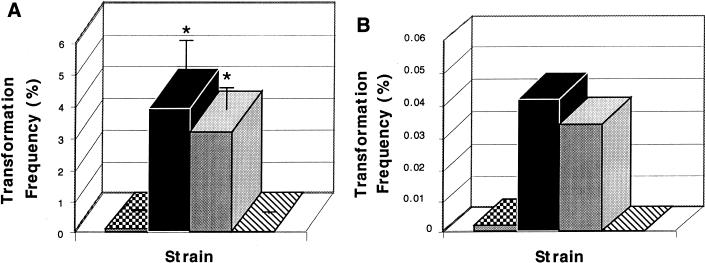
The question of which of the multiple cellular factors that vary in association with colony morphology was responsible for differences in rates of transformation was addressed by analysis of unencapsulated mutants derived from strain D39. D39 was transformed at a low frequency (0.032 and 0.0019%, with chromosomal and plasmid DNA, respectively). As expected, unencapsulated mutants P125 and P126, derived from D39, were transformed at substantially higher rates. Correction of the loss of capsule mutation to generate P138 was associated with a marked decline in competence, confirming the inhibitory effect of capsulation on transformation (Fig. 2). There was no significant difference, however, between opaque and transparent variants of the unencapsulated mutant in the rate of transformation with either the chromosomal (Fig. 2A) or plasmid (Fig. 2B) markers. Since these variants are still known to differ in amounts of cell wall teichoic acid and choline-binding proteins, this suggests that the variation in the quantity of capsular polysaccharide rather than these other factors accounts for differences in rates of transformation in encapsulated phase variants (4, 14).
FIG. 2.
The effect of capsule on transformation of the pneumococcus. After treatment to induce competence with CSP, the transformation frequencies [number of transformants/(number transformants + nontransformants)] with chromosomal DNA (A) or plasmid DNA (B) was compared in D39, an encapsulated parent strain (checkered bars); P125 and P126, opaque and transparent variants of an unencapsulated mutant of D39, respectively (gray and solid bars, respectively); and P138, a transformant of the unencapsulated mutant expressing a type 3 capsular polysaccharide (hatched bars). Values are the means of duplicate determinations of at least two independent experiments (n) ± standard deviations (error bars) (when n ≥ 3). There was no significant difference between opaque and transparent variants in the absence of capsule. Asterisks represent significant differences (P < 0.001; Student’s t test) in transformation frequency between unencapsulated and encapsulated strain variants.
This study demonstrates that genetic manipulation of the pneumococcus based on its natural competence tends to select for transparent variants with smaller amounts of cellular capsular polysaccharide regardless of the DNA incorporated. Since clinical isolates are generally mixtures of opaque and transparent variants, selection based on genetic transformation will lead to enrichment of the subpopulation with the transparent phenotype. Intrastrain variants with the transparent phenotype have been shown to be better adapted for nasopharyngeal colonization and less virulent in experimental models of pneumococcal sepsis (4, 20). Therefore, the loss of virulence in mutants generated by induction of competence may be a result of smaller amounts of capsular polysaccharide rather than the specific mutation tested. The quantity of capsular polysaccharide should be taken into consideration in virulence studies of the pneumococcus that depend on generation of mutants by natural transformation.
Acknowledgments
This work was supported by a grant from the Public Health Service (AI38436) (J.N.W.).
REFERENCES
- 1.Avery O T, MacLeod C M, McCarty M. Studies on the chemical nature of the substance inducing transformation of pneumococcal types. J Exp Med. 1944;79:137–157. doi: 10.1084/jem.79.2.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cundell D R, Weiser J N, Shen J, Young A, Tuomanen E I. Relationship between colonial morphology and adherence of Streptococcus pneumoniae. Infect Immun. 1995;63:757–761. doi: 10.1128/iai.63.3.757-761.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Håvarstein L S, Coomaraswamy G, Morrison D A. An unmodified heptadecapeptide pheromone induces competence for genetic transformation in Streptococcus pneumoniae. Proc Natl Acad Sci USA. 1995;92:11140–11144. doi: 10.1073/pnas.92.24.11140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim J, Weiser J. Association of intrastrain phase variation in quantity of capsular polysaccharide and teichoic acid with the virulence of Streptococcus pneumoniae. J Infect Dis. 1998;177:368–377. doi: 10.1086/514205. [DOI] [PubMed] [Google Scholar]
- 5.Kim J O, Romero-Steiner S, Sørensen U B S, Blom J, Carvalho M, Barnard S, Carlone G, Weiser J N. Relationship between cell surface carbohydrates and intrastrain variation on opsonophagocytosis of Streptococcus pneumoniae. Infect Immun. 1999;67:2327–2333. doi: 10.1128/iai.67.5.2327-2333.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lacks S, Hotchkiss R D. A study of the genetic material determining an enzyme activity in pneumococcus. Biochim Biophys Acta. 1960;39:508–517. doi: 10.1016/0006-3002(60)90205-5. [DOI] [PubMed] [Google Scholar]
- 7.MacLeod C M, Krauss M R. Relation of virulence of pneumococcal strains for mice to the quantity of capsular polysaccharide formed in vitro. J Exp Med. 1950;92:1–9. doi: 10.1084/jem.92.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Pearce B J, Yin Y B, Masure H R. Genetic identification of exported proteins in Streptococcus pneumoniae. Mol Microbiol. 1993;9:1037–1050. doi: 10.1111/j.1365-2958.1993.tb01233.x. [DOI] [PubMed] [Google Scholar]
- 9.Perez-Casal J, Caparon M G, Scott J R. Mry, a trans-acting positive regulator of the M protein gene of Streptococcus pyogenes with similarity to the receptor proteins of two-component regulatory systems. J Bacteriol. 1991;173:2617–2624. doi: 10.1128/jb.173.8.2617-2624.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Polissi A, Pontiggia A, Feger G, Altieri M, Mottl H, Ferrari L, Simon D. Large-scale identification of virulence genes from Streptococcus pneumoniae. Infect Immun. 1998;66:5620–5629. doi: 10.1128/iai.66.12.5620-5629.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pozzi G, Masala L, Iannelli F, Manganelli R, Håvarstein L S, Piccoli L, Simon D, Morrison D A. Competence for genetic transformation in encapsulated strains of Streptococcus pneumoniae: two allelic variants of the peptide pheromone. J Bacteriol. 1996;178:6087–6090. doi: 10.1128/jb.178.20.6087-6090.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ravin A W. Reciprocal capsular transformation of pneumococci. J Bacteriol. 1959;77:296–309. doi: 10.1128/jb.77.3.296-309.1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ring A, Weiser J N, Tuomanen E I. Pneumococcal penetration of the blood-brain barrier: molecular analysis of a novel re-entry path. J Clin Investig. 1998;102:347–360. doi: 10.1172/JCI2406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rosenow C, Ryan P, Weiser J N, Johnson S, Fontan P, Ortqvist A, Masure H R. Contribution of novel choline-binding proteins to adherence, colonization and immunogenicity of Streptococcus pneumoniae. Mol Microbiol. 1997;25:819–829. doi: 10.1111/j.1365-2958.1997.mmi494.x. [DOI] [PubMed] [Google Scholar]
- 15.Saluja S K, Weiser J N. The genetic basis of colony opacity in Streptococcus pneumoniae: evidence for the effect of box elements on the frequency of phenotypic variation. Mol Microbiol. 1995;16:215–227. doi: 10.1111/j.1365-2958.1995.tb02294.x. [DOI] [PubMed] [Google Scholar]
- 16.Spellerberg B, Cundell D R, Sandros J, Pearce B J, Idanpaan-Heikkila I, Rosenow C, Masure H R. Pyruvate oxidase, as a determinant of virulence in Streptococcus pneumoniae. Mol Microbiol. 1996;19:803–813. doi: 10.1046/j.1365-2958.1996.425954.x. [DOI] [PubMed] [Google Scholar]
- 17.Watson D A, Musher D M. Interruption of capsule production in Streptococcus pneumoniae serotype 3 by insertion of transposon Tn916. Infect Immun. 1990;58:3135–3138. doi: 10.1128/iai.58.9.3135-3138.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Weiser J, Markiewicz Z, Tuomanen E, Wani J. Relationship between phase variation in colony morphology, intrastrain variation in cell wall physiology and nasopharyngeal colonization by Streptococcus pneumoniae. Infect Immun. 1996;64:2240–2245. doi: 10.1128/iai.64.6.2240-2245.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Weiser J N. Phase variation in colony opacity. Microb Drug Resist. 1998;4:129–145. doi: 10.1089/mdr.1998.4.129. [DOI] [PubMed] [Google Scholar]
- 20.Weiser J N, Austrian R, Sreenivasan P K, Masure H R. Phase variation in pneumococcal opacity: relationship between colonial morphology and nasopharyngeal colonization. Infect Immun. 1994;62:2582–2589. doi: 10.1128/iai.62.6.2582-2589.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yother J, McDaniel L S, Briles D E. Transformation of encapsulated Streptococcus pneumoniae. J Bacteriol. 1986;168:1463–1465. doi: 10.1128/jb.168.3.1463-1465.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]