Abstract
Background:
Cardiovascular disease (CVD) is one of the principal causes of mortality in the world. Various factors have been identified in the pathogenesis of CVD. Leukemia inhibitory factor (LIF) as a secretory cytokine is one of these factors. The LIF receptor is located on endothelial cells and plays a role in the expression of specific genes in these cells. Endothelial cells are the innermost cells of blood vessels, and defects in these cells cause endothelial dysfunction and eventually CVD.
Methods:
The present study is based on PubMed database information (1982–2022) using the following words: “cardiovascular disease,” “endothelial cells,” “leukemia inhibitory factor,” and “angiogenesis.”
Results:
LIF can cause arteriosclerotic plaques by activating inflammatory mechanisms in monocytes through the induction of intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 expression. LIF can also induce vascular endothelial growth factor expression by activating signaling pathways, eventually leading to angiogenesis. Additionally, it can activate the coagulation cascade by factor VII production promotion within endothelial cells.
Conclusion:
Understanding the interplay between LIF and the inflammation pathways, coagulation, and angiogenesis as key factors in CVD occurrence raises the possibility of targeting this factor as a potential strategy to mitigate CVD risk.
Keywords: Cardiovascular disease, Endothelial cells, Leukemia inhibitory factor, Angiogenesis
Introduction
Cardiovascular disease (CVD) is one of the leading causes of death worldwide. The CVD mortality rate is 31% in the United States per year.1 Coronary heart disease was the leading cause of CVD death (42.6%) in the United States in 2017.2 CVD is considered a multifactorial disease; however, its principal pathogenesis is unknown. Risk factors such as high blood pressure, high blood cholesterol, and diabetes contribute to CVD development significantly.3,4 In addition to acquired factors, genetic factors are also crucial to the incidence of CVD.5,6
While many genetic factors causing CVD remain unclear, there are several genetic mutations and abnormalities in the cell cycle of heart cells that trigger inflammatory responses, vascular endothelium abnormalities, and defects in the heart structure, finally leading to CVD.7 Furthermore, mutations in transcription factors are considered associated factors of cardiac development.8 Endothelial cells (ECs) are essential to blood flow regulation in the arteries. The disorder of these cells is commonly regarded as a CVD predictor.9
Leukemia inhibitory factor (LIF) is a secretory cytokine. It belongs to the interleukin-6 family (IL-6) and plays a critical role in numerous biological processes, including inflammatory response, nervous system development, embryonic development, and cancer progression, through its influence on various signaling factors.10, 11 The LIF receptor is located on non-blood cells, such as ECs, and the surface of blood cells.
Since LIF secretion induces EC-specific gene expression, including vascular endothelial-cadherin, fetal liver kinase-1, and cluster of differentiation-31, LIF has a functional role in ECs.12, 13 In view of the weighty role of these cells in CVD development, we studied the role of LIF in inducing various pathways such as inflammation, coagulation, and angiogenesis, which are among the central pathways in CVD development.
Association between LIF and endothelial function
The inner surface of the arteries is covered by ECs, which communicate directly with blood and its components through the luminal membrane.14 There are several types of ECs, including tip, stalk, and phalanx cells.15 These cells communicate with other ECs through intercellular connections, such as tight junctions, adhesive junctions, and gap junctions. Adhesive junction proteins are vital in intercellular binding and the maintenance of tight junction structures. In addition, tight junctions regulate cellular mechanisms, such as proliferation, differentiation, and apoptosis, within the cell.16
ECs are considered metabolically active cells. Although the best pathway for energy production in these cells is oxidative phosphorylation, the core route of energy production in these cells is the glycolysis pathway because of their small number of mitochondria.17 ECs are regularly involved in homeostasis and innate immunity processes. One of the salient functions of these cells is their role in innate immunity by expressing toll-like receptors. ECs can also be effective in the coagulation process by expressing propagation factors, such as Von Willebrand factor and tissue factor.18 Furthermore, many factors like shear stress can affect many EC genes through transcription factor activation and microRNA stabilization. Although these cells reduce vascular blood flow owing to shear stress, the stress can modify their morphology and activity.19 Therefore, defects in ECs have been suggested as a CVD risk factor.20 After defects occur, cytokines and adhesion molecules are produced, causing inflammatory conditions in the arteries. ECs are crucial to such diseases as hypertension and atherosclerosis.21 Various factors, including reactive oxygen species (ROS) and atherosclerotic plaques, can cause EC dysfunction. Indeed, atherosclerosis is a complex process and is one of the leading causes of CVD pathophysiology,22 a process where different cells like ECs and macrophages play roles.23 Defects in ECs form atherosclerotic plaques owing to the accumulation of lipids and inflammatory cells.24,25 Inflammatory stimuli and abnormal lipid metabolism can activate ECs, leading to adhesion molecule expressions. The expression of adhesion molecules causes the recruitment of immune cells, including monocytes, to the vessel wall. Monocytes are transformed into macrophages, and by absorbing atherogenic products, they turn into foam cells. This is a pathological process resulting in vascular inflammation and atherosclerosis.26
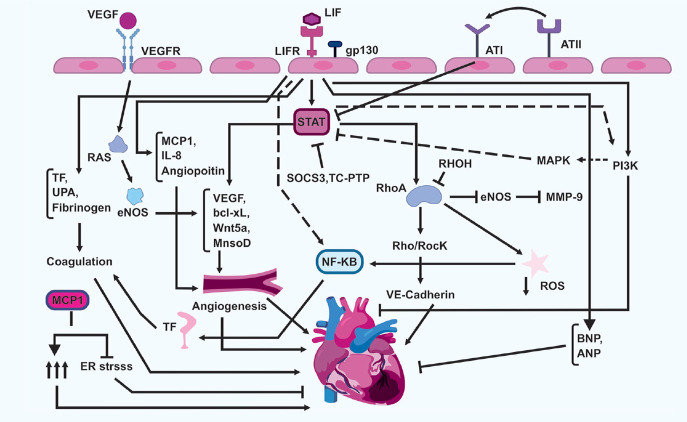
LIF can activate various signaling pathways, including Janus kinase/signal transducer and activator of transcription (JAK/STAT), mitogen-activated protein kinase (MAPK), and phosphoinositide 3-kinase (PI3K).27 IL-6 family cytokines, such as LIF, affect ECs through superfamily class I receptors. They impact ECs via the glycoprotein-130–LIF receptor complex. After binding to the complex, LIF activates JAK and heterodimerizes this complex. The heterodimerization of the glycoprotein-130–LIF receptor complex induces STAT phosphorylation.27 STAT-3 is involved in the pathogenesis of some diseases, including heart failure, hypertrophy, and atherosclerosis.28 In patients with CVD, activation of the JAK-2/STAT-3/IL-6 signaling pathway in ECs induces adhesive molecule gene expressions, such as vascular cell adhesion molecule-1 (VCAM-1) and intercellular adhesion molecule-1 (ICAM-1).29 The expression of the cited genes promotes monocyte migration and attachment to the endothelium. STAT-3 can also phosphorylate Ras homolog family member A (RhoA), an important factor for microfilament and microtubule rearrangement inhibiting the phosphorylation of endothelial nitric oxide synthase (eNOS). Subsequently, the function of matrix metalloproteinase-9 (MMP-9) is inhibited, and more mononuclear cells are recalled by VCAM-1 and ICAM-1 proteins. Eventually, the atherosclerotic plaque will be stabilized.30 STAT-3 can also activate Rho/Rho-associated protein kinase (Rock) by activating Rho, inhibiting the expression of vascular endothelial cadherin (VE-cadherin) and separating tight junctions.31 STAT-3 phosphorylation can be regulated by several mechanisms, including the inhibitory role of T-cell protein tyrosine phosphatase (TC-PTP) and suppressor of cytokine signaling-3 (SOCS-3).32 In addition to the JAK/STAT pathway, SOCS-3 can inhibit the MAPK pathway.33 Since JAK-2/STAT-5 and LIF can activate the PI3K/protein kinase B (Akt) and JAK/STAT pathways, respectively, it can be hypothesized that LIF could activate the PI3K/Akt pathway indirectly and cause cardiomyocyte proliferation. Considering the role of LIF in activating the PI3K signaling and proving the inhibitory role of the PI3K pathway in the MAPK pathway, LIF can be considered an inhibitor for the MAPK pathway.34,35 Since MAPK can on the one hand inhibit the JAK/STAT pathway and on the other hand activate the MAPK pathway, it is hypothesized that LIF can be deemed an inhibitory factor for the JAK/STAT pathway.35 Although IL-6 can activate Rho by acting on angiotensin-II and nuclear factor κ-light-chain enhancer of activated B cells (NFκB), it can also stimulate the production of ROS mediators, which in turn can generally induce the expression of VCAM-1, monocyte chemoattractant protein-1 (MCP-1), and IL-6 genes.36 MCP-1 can invoke monocytes to transform into macrophages, capable of binding to oxidized low-density lipoprotein. Therefore, macrophages can oxidize foam cells owing to oxidized low-density lipoprotein and then induce ROS production because they are the source of tumor necrosis factor-α (TNF-α) and IL-8 secretion. TNF-α is secreted in this pathway, and due to the role of this cytokine in Rho activation, this protein can be mentioned as another activator of the NFκB pathway.36 Although the NFκB pathway is effective in promoting pro-inflammatory conditions, some factors like nuclear factor of κ light polypeptide gene enhancer in B-cells inhibitor-α (IκB-α) are effective in inhibiting this factor.36 However, some studies have suggested that angiotensin-2 may inhibit LIF-mediated STAT-3 through angiotensin-1 receptor in cardiomyocytes by acting on angiotensin-1. Following Rho activation, this protein can also activate the NFκB pathway by activating Rac via a reduction in ROS levels.37, 38 The role of LIF in activating the cyclic adenosine 3′,5′-monophosphate (cAMP) signaling pathway has been demonstrated, with recent studies having suggested the inhibitory role of cAMP for NFκB. Hence, LIF can be mentioned as an NFκB pathway inhibitor.27,39 In general, the role of IL-6 differs in acute and chronic inflammation. In acute inflammation, it helps to protect the heart, while during chronic inflammation, it plays a significant role in cardiac remodeling.40 Considering the remodeling role of IL-6 in the chronic inflammation of LIF, it can stimulate the expression of brain natriuretic peptide (BNP) and atrial natriuretic peptide (ANP) genes by affecting the LIF receptor, leading to cardiac remodeling in chronic inflammation in ECs. Different pathways can activate NFκB signaling, which can be attributed to the effect of Rho. These pathways can also be inhibited by various molecules such as Ras homolog family member H (RHOH).41,42 Targeting any of these pathways can be effective in controlling inflammatory conditions, and reducing leukocyte accumulation in the inflammation site can reduce the inflammatory state in ECs (Table 1 & Figure 1).
Table 1.
A summary of the factors involved in endothelial dysfunction.
| Factor | Mechanism | Effect on the Function or Structure of ECs | Potential Therapeutic Property | Ref. |
|---|---|---|---|---|
|
|
|
|
|
|
| TNF-α | It activates the AGE product/receptor of the AGE (RAGE) and (NFκB) signaling path-way. | It induces the activation of NAD(P)H oxidase and the production of reactive oxidative species | TNF-α can induce the secretion of LIF. | (71, 72) |
| POMC | It inhibits the ET-converting enzyme in ECs. | It induces migration and tube-forming capability of ECs. | LIF can stimulate POMC expression. | (73, 74) |
| Endothelial YAP-1 | YAP expression can cause cardiomyocyte regeneration through IGF1 and Akt signaling. | It induces microRNA-152, which is effective in proliferation. | YAP-1 induces LIF. | (75, 76) |
| TGF-β | It activates the ALK-5 and Smad-2/3 pathways. | It increases the production of H2O2 and reduces cellular glutathione stores in ECs. | Transforming growth factor β1 induces leukemia inhibitory factor expression. | (77, 78) |
| SOCS-3 | SOCS-3 may protect endothelium by inhibiting IL-6/STAT-3 signaling. | It inhibits the effect of Ang-II and induces endothelial dysfunction. | SOCS-3 modulates leukemia inhibitory factor signaling. | (79, 80) |
LIF, Leukemia inhibitory factor; AGE, Advanced glycation end; AGER, Receptor of AGE; NFkB, Nuclear factor-kB; TNF-α, Tumor necrosis factorα; POMC, Proopiomelanocortin; YAP, Yes-associated protein; IGF, Insulin-like growth factor; TGF-β, Transforming growth factor-beta; SOCS-3, Suppressor of cytokine signaling-3
Figure 1.
Leukemia inhibitory factor (LIF) can induce cardiovascular disease (CVD) by binding to the LIF receptor. The attachment induces angiogenic pathways, inflammation, and coagulation. LIF can stimulate vascular endothelial (VE)-cadherin expression by activating the signal transducer and activator of transcription (STAT) pathway and inducing Ras homolog family member A (RhoA protein). It affects CVD. This factor can also be mediated if vascular endothelial growth factor (VEGF), B-cell lymphoma-extra large (Bcl-xL), Wnt family member 5A (WnT5a), manganese superoxide dismutase (Mn-SOD), monocyte chemoattractant protein-1 (MCP-1), interleukin-8 (IL-8), and angiopoietin activate the angiogenesis mechanism, inducing angiogenesis in the heart muscle. The LIF factor can also affect the system by the following proteins: tissue factor, urokinase-type plasminogen activator (UPA), and fibrinogen.
Association between LIF and angiogenesis
Angiogenesis is the process of the germination of new blood vessels from previous arteries. New blood vessel formation is necessary for many physiological processes, such as cardiovascular maturation, tissue repair, and regeneration.30 LIF upregulates genes such as vascular endothelial growth factor (VEGF), Wnt family member 5A (Wnt5a), B-cell lymphoma-extra large (Bcl-xL), and manganese superoxide dismutase (Mn-SOD) by affecting glycoprotein-130 and subsequently activating the signaling pathways STAT and extracellular signal-regulated kinases 1/2 (ERK-1/ERK-2).12 VEGF is a potent stimulant of angiogenesis because it promotes the proliferation of ECs and the formation of new blood vessels.43 Regulation of ECs in tip and stalk cells is the principal stimulus for angiogenesis.44 Tip cell differentiation is dependent on 2 primary pathways: VEGF and Notch.45 VEGF receptor is present on the surface of tip cells; after VEGF-receptor binding, this receptor complex causes Delta-like4 (DII4) expression. DII4 can inhibit VEGF receptor expression and germination in stalk cells by activating the Notch pathway. The Notch signaling pathway can also activate the Wnt pathway, involved in the tight junction process during angiogenesis.46 Although the VEGF factor can activate PI3K by the VEGF receptor pathway directly, it can activate the PI3K pathway through RAS indirectly. This pathway stimulates NO production by inducing eNOS, leading to angiogenesis. Increased NO production can increase VEGF expression via HIF-1 expression enhancement.47 In cardiac cells, prolonged phosphorylation of Akt induces the angiogenesis of the heart and maintains its contractility.48 Elevated NO production can also augment b-FGF expression, which is effective in angiogenesis.49 LIF can also stimulate the expression of pro-angiogenic genes, such as IL-8, MCP-1, and angiopoietin.50 Additionally, MCP-1 cytokine can modulate vascular formation and regulate cadherin-12 and cadherin-19 expression by inducing monocyte chemotactic protein-induced protein (MCPIP).51 Although LIF can play its role through the expression of the genes involved in the angiogenesis process, it can inhibit the angiogenesis process.52 LIF is secreted from ventricular myocytes in such pathological conditions as hypertrophied heart and failing heart and via intracellular signaling mechanisms. LIF also possesses antiangiogenic effects, defining a dual function for LIF53, 54 (Figure 1).
Association between LIF, inflammation, and coagulation
In healthy individuals, an increase in pro-inflammatory markers leads to a CVD increment.54,55 The immune system can alter the coagulation system through mechanisms like the synthesis and activation of coagulant proteins, reduction of anticoagulant synthesis, and fibrinolysis inhibition.56 The MAPK and PI3K pathways and protein kinase C can be induced by cytokines and growth factors. Moreover, they act as mediators in the tissue factor expression within ECs, muscles, and vascular cells.57 LIF can also enhance the expression level of the tissue factor by inducing glycoprotein-13058 and catalyzing the conversion of prothrombin to thrombin in the presence of factors Va and Ca. The occurrence of these events leads to fibrin formation, platelet activation, and finally, thrombus occurrence.59 In addition to the direct role of LIF in tissue factor production, VEGF can induce tissue factor production by activating the P38 and ERK pathways indirectly.60 The tissue factor is not only effective in coagulation processes but also activates immune cells and develops inflammation. Neutrophils inactivate the tissue factor pathway inhibitor by secreting elastase and protease, increasing coagulation. LIF acts as an anti-inflammatory factor, so it can reduce inflammation by targeting glycoprotein-130 and ILF receptor expression. In addition, LIF inhibits nerve growth factor and IL-1β production during inflammation.61 It exerts its anti-inflammatory properties by targeting macrophages. The infiltration of neutrophilic granulocytes is reduced in the early phase of the disease in the presence of LIF. Subsequently, this cytokine can reduce the molecular level of chemokine (C-X-C motif) ligand-1 (CXCL-1) in the later phase of the disease, causing macrophage infiltration, which occurs through C-C motif chemokine ligand-2 (CCL-2), (CCL-3), and CXCL-10 at the molecular level.62 After the formation of an acute phase reaction, LIF can be produced by active cells of the immune system during inflammation. The produced LIF can modulate the coagulation and fibrinolytic systems, which in turn leads to coagulopathy. Furthermore, LIF can affect the coagulation system by increasing the production of proteins like hepatic urokinase plasminogen activator and fibrinogen. Increased fibrinogen in arteriosclerotic plaques causes platelet aggregation. Therefore, plasma fibrinogen is considered an effective agent for coronary heart disease.63 Alpha-thrombin is another protein in the coagulation system that is activated during the coagulation cascade. It affects ECs and lymphocytes. These coagulation proteins are significant in inflammatory processes by binding leukocytes to ECs. Alpha-thrombin can also be involved in the production of IL-6 and IL-1 by macrophages and ECs. It can inhibit the STAT pathway by binding to the protease-activated receptor-1 (PAR-1) thrombin receptor using LIF.64 According to the STAT signaling role in the LIF induction in the heart and the high expression of PAR-1 on cardiomyocytes, it can be hypothesized that thrombin could be an inhibitory factor for LIF function. LIF can also produce MCP-1, which plays a major role in diapedesis, monocyte, and arteriosclerotic plaque formation.65 MCP-1 can act as a double-edged sword because it plays a prominent protective role in CVD by preventing endoplasmic reticulum (ER) stress, and prolonged exposure to this factor can cause heart failure.66 If thrombin levels increase during the coagulation process, it can activate platelets and form fibrin clots through the PAR signaling pathway. This process not only involves the invocation and activation of monocytes, neutrophils, and platelets but also contains the induction of EC-mediated leukocyte adhesion, the release of pro-inflammatory cytokines, and complement activation. Factor VIIa and tissue factor complexes produce inflammatory cytokines and chemokines through the PAR signaling pathway.67,68 Since LIF can be released from cardiomyocytes in CVD, it can activate coagulation proteins by inducing inflammatory mechanisms in addition to inflammation, followed by the induction of coagulation. These factors are generally effective in the CVD pathogenesis (Figure 1).
Association between LIF and microRNAs
MicroRNAs (miRNAs) are small noncoding RNA molecules involved in gene expression regulation. Further, miRNAs are transcribed by RNA polymerases II and III and generate precursors that eventually produce mature miRNAs after performing certain processes.69 Changes in the normal function of ECs have been proven as a result of the pathogenesis of various diseases, such as atherosclerosis, inflammatory diseases, and hypertension. Impaired vascular function disrupts several events, increasing the leukocyte count by activating ECs. Considering the roles of miRNAs and LIF in EC function and expression, identifying the relevant pathways can help prevent CVD in the future by reducing the incidence of EC dysfunction.70 CVD and its relationship with LIF are explained in Table 2 and Figure 1.
Table 2.
Involved miRNAs in endothelial dysfunction
| MiRNA | Disease | Target | Inflammation/Oxidative Stress | Mechanism | Effect on LIF | Ref |
|---|---|---|---|---|---|---|
|
|
|
|
|
|
|
|
| MicroRNA-223-3p | AMI | NLRP-3 | Anti-inflammatory | Reducing the secretion of IL-1β | Suppressing LIF expression | (70, 81) |
| MicroRNA-146a | MI | Target IRAK1 & TRAF-6 | Anti-inflammatory | Attenuating NFκB activation and inflammatory cytokine production by suppressing IRAK-1 and TRAF-6 | Inhibiting the expression of LIF | (82, 83) |
| MicroRNA-21 | MI | KBTBD-7 gene | Anti-inflammatory | Promoting the activation of p38 and NFκB signaling by interacting with MKK3/6 | LIF induces the expression of microRNA-21 (miR-21). | (84, 85) |
| MicroRNA-125a | Atherosclero-sis | ET-1 | Anti-inflammatory | Suppressing oxLDL and inducing ET-1 expression | MicroRNA 125a can induce the LIF receptor. | (86, 87) |
| MicoRNA-146a | Atherosclero-sis | NOX-4 | Anti-Inflammation | Inhibiting the activation of TLR4-dependent signaling pathway | Inhibit the expression of LIF | (82, 88) |
| MicroRNA-141 | Atherosclero-sis | PAPP-A | Inflammation | Inhibiting the vascular smooth muscle cell proliferation | LIF can induce the production of microRNA-21. | (89, 90) |
AMI, Acute myocardial infarction; NLRP3, NOD-, LRR- and pyrin domain-containing protein 3; MI, Myocardial infarction; IRAK1, Interleukin-1 receptor-associated kinase 1; TRAF-6, TNF receptor-associated factor-6; NFκB, Nuclear factor-kB; ET-1, Endothelin-1; oxLDL, Oxidized LDL; TLR-4, Toll-like receptor-4; PAPP-A, pregnancy-associated plasma protein-A
Conclusion
In recent years, due to the increase in the incidence of CVD, many studies have focused on the causes of CVD. Activation of various signaling pathways within ECs through LIF has highlighted the role of this factor in EC dysfunction. The widespread role of LIF is known in different processes, including angiogenesis, inflammation, and coagulation, which are crucial mechanisms in CVD development. Due to the role of LIF in inducing the mentioned mechanisms, targeting this factor could reduce CVD incidence. Additional studies in this area may pave the way for using this factor to mitigate CVD.
Notes:
This paper should be cited as: Rezaeeyan H, Moghimian-Boroujeni B, Abdolalian M, Javan M. The Role of Leukemia Inhibitory Factor in Cardiovascular Disease: Signaling in Inflammation, Coagulation, and Angiogenesis. J Teh Univ Heart Ctr 2024;19(1):6-13.
References
- 1.Chistiakov DA, Shkurat TP, Melnichenko AA, Grechko AV, Orekhov AN. The role of mitochondrial dysfunction in cardiovascular disease: a brief review. Ann Med 2018;50:121–127. [DOI] [PubMed] [Google Scholar]
- 2.Virani SS, Alonso A, Benjamin EJ, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Shay CM, Spartano NL, Stokes A, Tirschwell DL, VanWagner LB, Tsao CW, American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee . Heart Disease and Stroke Statistics-2020 Update: A Report From the American Heart Association. Circulation 2020;141:e139–e596. [DOI] [PubMed] [Google Scholar]
- 3.Schwartz SM, Schwartz HT, Horvath S, Schadt E, Lee SI. A systematic approach to multifactorial cardiovascular disease: causal analysis. Arterioscler Thromb Vasc Biol 2012;32:2821–2835. [DOI] [PubMed] [Google Scholar]
- 4.Zhao L, Varghese Z, Moorhead JF, Chen Y, Ruan XZ. CD36 and lipid metabolism in the evolution of atherosclerosis. Br Med Bull. 2018. Jun 1;126(1):101–112. [DOI] [PubMed] [Google Scholar]
- 5.Khera AV, Kathiresan S. Genetics of coronary artery disease: discovery, biology and clinical translation. Nat Rev Genet 2017;18:331–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sayols-Baixeras S, Lluís-Ganella C, Lucas G, Elosua R. Pathogenesis of coronary artery disease: focus on genetic risk factors and identification of genetic variants. Appl Clin Genet 2014;7:15–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bauer AJ, Martin KA. Coordinating Regulation of Gene Expression in Cardiovascular Disease: Interactions between Chromatin Modifiers and Transcription Factors. Front Cardiovasc Med 2017;4:19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McCulley DJ, Black BL. Transcription factor pathways and congenital heart disease. Curr Top Dev Biol 2012;100:253–277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Barthelmes J, Nägele MP, Ludovici V, Ruschitzka F, Sudano I, Flammer AJ. Endothelial dysfunction in cardiovascular disease and Flammer syndrome-similarities and differences. EPMA J 2017;8:99–109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yue X, Wu L, Hu W. The regulation of leukemia inhibitory factor. Cancer Cell Microenviron 2015;2:e877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jiang W, Bai W, Li J, Liu J, Zhao K, Ren L. Leukemia inhibitory factor is a novel biomarker to predict lymph node and distant metastasis in pancreatic cancer. Int J Cancer 2021;148:1006–1013. [DOI] [PubMed] [Google Scholar]
- 12.Mohri T, Fujio Y, Maeda M, Ito T, Iwakura T, Oshima Y, Uozumi Y, Segawa M, Yamamoto H, Kishimoto T, Azuma J. Leukemia inhibitory factor induces endothelial differentiation in cardiac stem cells. J Biol Chem 2006;281:6442–6447. [DOI] [PubMed] [Google Scholar]
- 13.Suman P, Malhotra SS, Gupta SK. LIF-STAT signaling and trophoblast biology. JAKSTAT 2013;2:e25155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Krüger-Genge A, Blocki A, Franke RP, Jung F. Vascular Endothelial Cell Biology: An Update. Int J Mol Sci 2019;20:4411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Eelen G, de Zeeuw P, Treps L, Harjes U, Wong BW, Carmeliet P. Endothelial Cell Metabolism. Physiol Rev 2018;98:3–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cong X, Kong W. Endothelial tight junctions and their regulatory signaling pathways in vascular homeostasis and disease. Cell Signal 2020;66:109485. [DOI] [PubMed] [Google Scholar]
- 17.Eelen G, de Zeeuw P, Simons M, Carmeliet P. Endothelial cell metabolism in normal and diseased vasculature. Circ Res 2015;116:1231–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sturtzel C. Endothelial Cells. Adv Exp Med Biol 2017;1003:71–91. [DOI] [PubMed] [Google Scholar]
- 19.Ando J, Yamamoto K. Flow detection and calcium signalling in vascular endothelial cells. Cardiovasc Res 2013;99:260–268. [DOI] [PubMed] [Google Scholar]
- 20.Bonetti PO, Lerman LO, Lerman A. Endothelial dysfunction: a marker of atherosclerotic risk. Arterioscler Thromb Vasc Biol 2003;23:168–175. [DOI] [PubMed] [Google Scholar]
- 21.Sun HJ, Wu ZY, Nie XW, Bian JS. Role of Endothelial Dysfunction in Cardiovascular Diseases: The Link Between Inflammation and Hydrogen Sulfide. Front Pharmacol 2020;10:1568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chen Q, Lv J, Yang W, Xu B, Wang Z, Yu Z, Wu J, Yang Y, Han Y. Targeted inhibition of STAT-3 as a potential treatment strategy for atherosclerosis. Theranostics 2019;9:6424–6442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lusis AJ. Atherosclerosis. Nature 2000;407:233–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Förstermann U, Xia N, Li H. Roles of Vascular Oxidative Stress and Nitric Oxide in the Pathogenesis of Atherosclerosis. Circ Res 2017;120:713–735. [DOI] [PubMed] [Google Scholar]
- 25.Menghini R, Casagrande V, Cardellini M, Ballanti M, Davato F, Cardolini I, Stoehr R, Fabrizi M, Morelli M, Anemona L, Bernges I, Schwedhelm E, Ippoliti A, Mauriello A, Böger RH, Federici M. FoxO1 regulates asymmetric dimethylarginine via downregulation of dimethylaminohydrolase 1 in human endothelial cells and subjects with atherosclerosis. Atherosclerosis 2015;242:230–235. [DOI] [PubMed] [Google Scholar]
- 26.Xu S, Ilyas I, Little PJ, Li H, Kamato D, Zheng X, Luo S, Li Z, Liu P, Han J, Harding IC, Ebong EE, Cameron SJ, Stewart AG, Weng J. Endothelial Dysfunction in Atherosclerotic Cardiovascular Diseases and Beyond: From Mechanism to Pharmacotherapies. Pharmacol Rev 2021;73:924–967. [DOI] [PubMed] [Google Scholar]
- 27.Auernhammer CJ, Melmed S. Leukemia-inhibitory factor-neuroimmune modulator of endocrine function. Endocr Rev 2000;21:313–345. [DOI] [PubMed] [Google Scholar]
- 28.Knight RA, Scarabelli TM, Stephanou A. STAT transcription in the ischemic heart. JAKSTAT 2012;1:111–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wang Z, Ni L, Wang J, Lu C, Ren M, Han W, Liu C. The protective effect of melatonin on smoke-induced vascular injury in rats and humans: a randomized controlled trial. J Pineal Res 2016;60:217–227. [DOI] [PubMed] [Google Scholar]
- 30.Wolinsky H. A proposal linking clearance of circulating lipoproteins to tissue metabolic activity as a basis for understanding atherogenesis. Circ Res 1980;47:301–311. [DOI] [PubMed] [Google Scholar]
- 31.Birukov KG. Small GTPases in mechanosensitive regulation of endothelial barrier. Microvasc Res 2009;77:46–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ng IH, Yeap YY, Ong LS, Jans DA, Bogoyevitch MA. Oxidative stress impairs multiple regulatory events to drive persistent cytokine-stimulated STAT-3 phosphorylation. Biochim Biophys Acta 2014;1843:483–494. [DOI] [PubMed] [Google Scholar]
- 33.Nicola NA, Babon JJ. Leukemia inhibitory factor (LIF). Cytokine Growth Factor Rev 2015;26:533–544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cao Z, Liao Q, Su M, Huang K, Jin J, Cao D. AKT and ERK dual inhibitors: The way forward? Cancer Lett 2019;459:30–40. [DOI] [PubMed] [Google Scholar]
- 35.Taga T, Kishimoto T. Gp130 and the interleukin-6 family of cytokines. Annu Rev Immunol 1997;15:797–819. [DOI] [PubMed] [Google Scholar]
- 36.Fiordelisi A, Iaccarino G, Morisco C, Coscioni E, Sorriento D. NFkappaB is a Key Player in the Crosstalk between Inflammation and Cardiovascular Diseases. Int J Mol Sci 2019;20:1599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sulciner DJ, Irani K, Yu ZX, Ferrans VJ, Goldschmidt-Clermont P, Finkel T. rac1 regulates a cytokine-stimulated, redox-dependent pathway necessary for NF-kappaB activation. Mol Cell Biol 1996;16:7115–7121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tone E, Kunisada K, Fujio Y, Matsui H, Negoro S, Oh H, Kishimoto T, Yamauchi-Takihara K. Angiotensin II interferes with leukemia inhibitory factor-induced STAT-3 activation in cardiac myocytes. Biochem Biophys Res Commun 1998;253:147–150. [DOI] [PubMed] [Google Scholar]
- 39.Minguet S, Huber M, Rosenkranz L, Schamel WW, Reth M, Brummer T. Adenosine and cAMP are potent inhibitors of the NF-kappa B pathway downstream of immunoreceptors. Eur J Immunol 2005;35:31–41. [DOI] [PubMed] [Google Scholar]
- 40.Gordon JW, Shaw JA, Kirshenbaum LA. Multiple facets of NF-κB in the heart: to be or not to NF-κB. Circ Res 2011;108:1122–1132. [DOI] [PubMed] [Google Scholar]
- 41.Ke Y, Zebda N, Oskolkova O, Afonyushkin T, Berdyshev E, Tian Y, Meng F, Sarich N, Bochkov VN, Wang JM, Birukova AA, Birukov KG. Anti-Inflammatory Effects of OxPAPC Involve Endothelial Cell-Mediated Generation of LXA4. Circ Res 2017;121:244–257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li X, Bu X, Lu B, Avraham H, Flavell RA, Lim B. The hematopoiesis-specific GTP-binding protein RhoH is GTPase deficient and modulates activities of other Rho GTPases by an inhibitory function. Mol Cell Biol 2002;22:1158–171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Curry JM, Eubank TD, Roberts RD, Wang Y, Pore N, Maity A, Marsh CB. M-CSF signals through the MAPK/ERK pathway via Sp1 to induce VEGF production and induces angiogenesis in vivo. PLoS One 2008;3:e3405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Eilken HM, Adams RH. Dynamics of endothelial cell behavior in sprouting angiogenesis. Curr Opin Cell Biol 2010;22:617–625. [DOI] [PubMed] [Google Scholar]
- 45.Adams RH, Alitalo K. Molecular regulation of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol 2007;8:464–478. [DOI] [PubMed] [Google Scholar]
- 46.Sewduth R, Santoro MM. “Decoding” Angiogenesis: New Facets Controlling Endothelial Cell Behavior. Front Physiol 2016;7:306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Karar J, Maity A. PI3K/AKT/mTOR Pathway in Angiogenesis. Front Mol Neurosci 2011;4:51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gogiraju R, Bochenek ML, Schäfer K. Angiogenic Endothelial Cell Signaling in Cardiac Hypertrophy and Heart Failure. Front Cardiovasc Med 2019;6:20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Yamamoto N, Oyaizu T, Enomoto M, Horie M, Yuasa M, Okawa A, Yagishita K. VEGF and bFGF induction by nitric oxide is associated with hyperbaric oxygen-induced angiogenesis and muscle regeneration. Sci Rep 2020;10:2744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Santos GC, Silva DN, Fortuna V, Silveira BM, Orge ID, de Santana TA, Sampaio GL, Paredes BD, Ribeiro-Dos-Santos R, Soares MBP. Leukemia Inhibitory Factor (LIF) Overexpression Increases the Angiogenic Potential of Bone Marrow Mesenchymal Stem/Stromal Cells. Front Cell Dev Biol 2020;8:778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Niu J, Azfer A, Zhelyabovska O, Fatma S, Kolattukudy PE. Monocyte chemotactic protein (MCP)-1 promotes angiogenesis via a novel transcription factor, MCP-1-induced protein (MCPIP). J Biol Chem 2008;283:14542–1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu CZ, Zhou HJ, Zhong JH, Tang T, Cui HJ, Zhou JH, Zhang Q, Mei ZG. Leukemia Inhibitory Factor Decreases Neurogenesis and Angiogenesis in a Rat Model of Intracerebral Hemorrhage. Curr Med Sci 2019;39:298–304. [DOI] [PubMed] [Google Scholar]
- 53.Podewski EK, Hilfiker-Kleiner D, Hilfiker A, Morawietz H, Lichtenberg A, Wollert KC, Drexler H. Alterations in Janus kinase (JAK)-signal transducers and activators of transcription (STAT) signaling in patients with end-stage dilated cardiomyopathy. Circulation 2003;107:798–802. [DOI] [PubMed] [Google Scholar]
- 54.Kurdi M, Randon J, Cerutti C, Bricca G. Increased expression of IL-6 and LIF in the hypertrophied left ventricle of TGR(mRen2)27 and SHR rats. Mol Cell Biochem 2005;269:95–101. [DOI] [PubMed] [Google Scholar]
- 55.Ridker PM, Hennekens CH, Buring JE, Rifai N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med 2000;342:836–843. [DOI] [PubMed] [Google Scholar]
- 56.Lipinski S, Bremer L, Lammers T, Thieme F, Schreiber S, Rosenstiel P. Coagulation and inflammation. Molecular insights and diagnostic implications. Hamostaseologie 2011;31:94–102, 104. [DOI] [PubMed] [Google Scholar]
- 57.Steffel J, Lüscher TF, Tanner FC. Tissue factor in cardiovascular diseases: molecular mechanisms and clinical implications. Circulation 2006;113:722–731. [DOI] [PubMed] [Google Scholar]
- 58.Meisel SR, Shimon I, Edgington TS, Melmed S, Cercek B, Shah PK. Leukaemia inhibitory factor enhances tissue factor expression in human monocyte-derived macrophages: a gp130-mediated mechanism. Br J Haematol 1999;107:747–755. [DOI] [PubMed] [Google Scholar]
- 59.Mackman N. The role of tissue factor and factor VIIa in hemostasis. Anesth Analg 2009;108:1447–1452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mechtcheriakova D, Schabbauer G, Lucerna M, Clauss M, De Martin R, Binder BR, Hofer E. Specificity, diversity, and convergence in VEGF and TNF-alpha signaling events leading to tissue factor up-regulation via EGR-1 in endothelial cells. FASEB J 2001;15:230–242. [DOI] [PubMed] [Google Scholar]
- 61.Demetz G, Ott I. The Interface between Inflammation and Coagulation in Cardiovascular Disease. Int J Inflam 2012;2012:860301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Escary JL, Perreau J, Duménil D, Ezine S, Brûlet P. Leukaemia inhibitory factor is necessary for maintenance of haematopoietic stem cells and thymocyte stimulation. Nature 1993;363:361–364. [DOI] [PubMed] [Google Scholar]
- 63.Okada H, Woodcock-Mitchell J, Fujii S. Leukaemia inhibitory factor and oncostatin M modulate expression of urokinase plasminogen activator and fibrinogen. Coron Artery Dis 1996;7:561–567. [DOI] [PubMed] [Google Scholar]
- 64.Bhat GJ, Hunt RA, Baker KM. alpha-Thrombin inhibits signal transducers and activators of transcription 3 signaling by interleukin-6, leukemia inhibitory factor, and ciliary neurotrophic factor in CCL39 cells. Arch Biochem Biophys 1998;350:307–314. [DOI] [PubMed] [Google Scholar]
- 65.Zgheib C, Zouein FA, Kurdi M, Booz GW. Chronic treatment of mice with leukemia inhibitory factor does not cause adverse cardiac remodeling but improves heart function. Eur Cytokine Netw 2012;23:191–197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Niu J, Kolattukudy PE. Role of MCP-1 in cardiovascular disease: molecular mechanisms and clinical implications. Clin Sci (Lond) 2009;117:95–109. [DOI] [PubMed] [Google Scholar]
- 67.Foley JH, Conway EM. Cross Talk Pathways Between Coagulation and Inflammation. Circ Res 2016;118:1392–408. [DOI] [PubMed] [Google Scholar]
- 68.Pawlinski R, Tencati M, Hampton CR, Shishido T, Bullard TA, Casey LM, Andrade-Gordon P, Kotzsch M, Spring D, Luther T, Abe J, Pohlman TH, Verrier ED, Blaxall BC, Mackman N. Protease-activated receptor-1 contributes to cardiac remodeling and hypertrophy. Circulation 2007;116:2298–2306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Macfarlane LA, Murphy PR. MicroRNA: Biogenesis, Function and Role in Cancer. Curr Genomics 2010;11:537–561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Fernández-Hernando C, Suárez Y. MicroRNAs in endothelial cell homeostasis and vascular disease. Curr Opin Hematol 2018;25:227–236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Lan X, Chen Q, Wang Y, Jia B, Sun L, Zheng J, Peng H. TNF-α affects human cortical neural progenitor cell differentiation through the autocrine secretion of leukemia inhibitory factor. PLoS One 2012;7:e50783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lee J, Lee S, Zhang H, Hill MA, Zhang C, Park Y. Interaction of IL-6 and TNF-α contributes to endothelial dysfunction in type 2 diabetic mouse hearts. PLoS One 2017;12:e0187189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lam HC, Kuo SM, Chuang MJ, Keng HM, Lin PR, Liu GS, Hsu CM, Howng SL, Tai MH. Blockade of endothelin-1 release contributes to the anti-angiogenic effect by pro-opiomelanocortin overexpression in endothelial cells. Exp Biol Med (Maywood) 2006;231:782–788. [PubMed] [Google Scholar]
- 74.Ray DW, Ren SG, Melmed S. Leukemia inhibitory factor (LIF) stimulates proopiomelanocortin (POMC) expression in a corticotroph cell line. Role of STAT pathway. J Clin Invest 1996;97:1852–1859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Fan M, Yang K, Wang X, Wang Y, Tu F, Ha T, Liu L, Williams DL, Li C. Endothelial cell HSPA12B and yes-associated protein cooperatively regulate angiogenesis following myocardial infarction. JCI Insight 2020;5:e139640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ai LQ, Zhu JY, Chen X, Li X, Luo LL, Hu QM, Lin S, Ye J. Endothelial Yes-Associated Protein 1 Promotes Astrocyte Proliferation and Maturation via Cytoplasmic Leukemia Inhibitory Factor Secretion in Oxygen-Induced Retinopathy. Invest Ophthalmol Vis Sci 2020;61:1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Feng W, Ying WZ, Aaron KJ, Sanders PW. Transforming growth factor-β mediates endothelial dysfunction in rats during high salt intake. Am J Physiol Renal Physiol 2015;309:F1018–1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ota K, Quint P, Weivoda MM, Ruan M, Pederson L, Westendorf JJ, Khosla S, Oursler MJ. Transforming growth factor beta 1 induces CXCL16 and leukemia inhibitory factor expression in osteoclasts to modulate migration of osteoblast progenitors. Bone 2013;57:68–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Boyle K, Robb L. The role of SOCS-3 in modulating leukaemia inhibitory factor signalling during murine placental development. J Reprod Immunol 2008;77:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Li Y, Kinzenbaw DA, Modrick ML, Pewe LL, Faraci FM. Context-dependent effects of SOCS-3 in angiotensin II-induced vascular dysfunction and hypertension in mice: mechanisms and role of bone marrow-derived cells. Am J Physiol Heart Circ Physiol 2016;311:H146–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bauernfeind F, Rieger A, Schildberg FA, Knolle PA, Schmid-Burgk JL, Hornung V. NLRP3 inflammasome activity is negatively controlled by miR-223. J Immunol. 2012. Oct 15;189(8):4175–81. [DOI] [PubMed] [Google Scholar]
- 82.Wang L, Zhang H, Lei D. microRNA-146a Promotes Growth of Acute Leukemia Cells by Downregulating Ciliary Neurotrophic Factor Receptor and Activating JAK2/STAT-3 Signaling. Yonsei Med J 2019;60:924–934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Wang X, Ha T, Liu L, Zou J, Zhang X, Kalbfleisch J, Gao X, Williams D, Li C. Increased expression of microRNA-146a decreases myocardial ischaemia/reperfusion injury. Cardiovasc Res 2013;97:432–442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yang L, Wang B, Zhou Q, Wang Y, Liu X, Liu Z, Zhan Z. MicroRNA-21 prevents excessive inflammation and cardiac dysfunction after myocardial infarction through targeting KBTBD7. Cell Death Dis 2018;9:769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Yue X, Zhao Y, Zhang C, Li J, Liu Z, Liu J, Hu W. Leukemia inhibitory factor promotes EMT through STAT-3-dependent miR-21 induction. Oncotarget 2016;7:3777–3790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Li D, Yang P, Xiong Q, Song X, Yang X, Liu L, Yuan W, Rui YC. MicroRNA-125a/b-5p inhibits endothelin-1 expression in vascular endothelial cells. J Hypertens 2010;28:1646–1654. [DOI] [PubMed] [Google Scholar]
- 87.Nandy SB, Arumugam A, Subramani R, Pedroza D, Hernandez K, Saltzstein E, Lakshmanaswamy R. MicroRNA-125a influences breast cancer stem cells by targeting leukemia inhibitory factor receptor which regulates the Hippo signaling pathway. Oncotarget 2015;6:17366–17378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Yang K, He YS, Wang XQ, Lu L, Chen QJ, Liu J, Sun Z, Shen WF. MiR-146a inhibits oxidized low-density lipoprotein-induced lipid accumulation and inflammatory response via targeting toll-like receptor 4. FEBS Lett 2011;585:854–860. [DOI] [PubMed] [Google Scholar]
- 89.Morales-Prieto DM, Schleussner E, Markert UR. Reduction in miR-141 is induced by leukemia inhibitory factor and inhibits proliferation in choriocarcinoma cell line JEG-3. Am J Reprod Immunol 2011;66 Suppl 1:57–62. [DOI] [PubMed] [Google Scholar]
- 90.Zhang Y, Chen B, Ming L, Qin H, Zheng L, Yue Z, Cheng Z, Wang Y, Zhang D, Liu C, Bin W, Hao Q, Song F, Ji B. MicroRNA-141 inhibits vascular smooth muscle cell proliferation through targeting PAPP-A. Int J Clin Exp Pathol 2015;8:14401–14408. [PMC free article] [PubMed] [Google Scholar]