Abstract
Sodium-glucose co-transporter 2 inhibitors (SGLT2i) have emerged as important agents for the treatment of type 2 diabetes mellitus (T2DM). SGLT2 inhibitors have been associated with improved cardiovascular outcomes, not only through their immediate hemodynamic effects—such as glycosuria and (at least temporary) increased natriuresis—but also due to their multifaceted impact on metabolism. Recently, studies have also focused on the effects of SGLT2 inhibitors on adipose tissue. Aside from the well-documented effects on human adiposity, SGLT2i have shown, both in vitro and in murine models, the ability to reduce fat mass, upregulate genes related to browning of white adipose tissue, influence adipocyte size and fatty acid oxidation, and improve oxidative stress and overall metabolic health. In humans, even though data are still limited, recent evidence seems to confirm that the SGLT2i effects observed in cardiovascular outcome trials could be partially explained by their impact on adipose tissue. This review aims to clarify the impact of SGLT2i on adipose tissue, highlighting their role in metabolic health and their potential to transform treatment strategies for T2DM beyond glucose metabolism.
Graphical abstract
Keywords: Diabetes, Metabolism, Adipose organ, SGLT-2i, Precision medicine, Epicardial adipose tissue
Introduction
SGLT2i are a class of medications originally employed in treatment of T2DM. Their mechanism of action and therapeutic impact extend beyond conventional glucose regulation through glycosuria, providing several benefits in cardiovascular (CV) [1–3] and kidney diseases [4]. Their immediate hemodynamic effects—resulting from glycosuria and (at least temporary) increased natriuresis—reduce intravascular volume and arterial pressure, helping to mitigate heart failure and improve kidney perfusion and function. Additionally, their cardiovascular benefits are supported by longer-term metabolic remodeling, which includes improved adipose tissue metabolism, enhanced insulin sensitivity, and reduced systemic inflammation [5, 6]. However, despite these observations, the precise mechanisms behind these benefits are not yet fully understood and are thus the focus of ongoing research with investigation into SGLT2 expression being extended beyond renal tissues. There are indications that SGLT2 are expressed (although at considerably lower levels) in other tissues such as the pancreas, liver, muscle, and adipose tissues, however, the functional relevance of SGLT2 in these non-renal tissues, remains to be fully elucidated [7].
In the DAPAHEART TRIAL, we enrolled patients with stable coronary artery disease with suboptimal glycemia, randomizing them in a 1:1 ratio to dapagliflozin or placebo for 4 weeks. We found a significant 30% increase in coronary flow reserve in the dapagliflozin group [8]. Moreover, compared to all the other visceral and subcutaneous adipose tissue (SAT) depots, we saw a reduction in the thickness of epicardial adipose tissue (EAT), associated with a reduction in glucose uptake [9]. We interpreted these results as a decrease in the pro-inflammatory state of EAT, a known risk factor for CV diseases, which justified the observed improvement in coronary flow reserve (CFR) [10]. In fact, the effects of SGLT2i on EAT inflammation has also been demonstrated in vitro and in rats, in which SGLT2i have been shown to increase the level of ketone bodies, thus reducing EAT inflammation and atrial dysfunction [11].
The role of adipose tissue in the pathogenesis of type 2 diabetes and cardiovascular diseases is well known [12] and the recent spurt in drugs that cause a significant reduction in adipose tissue together with a significant reduction in CV risk, has brought renewed attention [13–15]. Indeed, several drugs used in the management of T2DM have shown promising results in weight control [15]. In this context, SGLT2i seem to exhibit metabolic effects independently of the fluid contraction caused by glycosuria. Glycosuria reduces blood glucose levels and contributes to caloric loss [3], but there also seems to be a direct action of SGLT2i on adipose tissue metabolism, as evidenced, for example, by their impact on EAT [9].Based on these premises, this review aims to examine and clarify the effects of SGLT2i on adipose tissue, focusing on their impact on metabolic health and evaluating how they might transform treatment strategies for type 2 diabetes and metabolic diseases.
SGLT2i: class effects and unmet needs
Several SGLTi, dapagliflozin, canagliflozin, empagliflozin and ertugliflozin, are currently approved in Europe [16]. The primary mechanism of action of SGLT2i involves the inhibition of the SGLT2 protein in the proximal convoluted tubule of kidney, which is responsible for the reabsorption of approximately 90% of filtered glucose. By blocking this transporter, SGLT2i promote glucosuria, leading to a reduction in glycemia [17]. Data from randomized clinical trials suggest that the beneficial effects observed can be ascribed to class features and are not specific to the single drug [18].
Beyond their glucose-lowering effects, SGLT2i have shown significant cardiorenal benefits, extensively validated in several landmark clinical trials, demonstrating that SGLT2i reduce the incidence of major adverse cardiovascular events (MACE), to a varying extent depending on the drug and/or the enrolled population. They significantly lower the risk of cardiovascular death and regardless of T2DM status reduce the risk of hospitalization for heart failure and slow the progression of kidney failure [18, 19]. Importantly, SGLT2i have been shown to enhance heart functional capacity, as indicated by improvements in peak VO2 and performance in the 6-minute walking test (6MWT), both key metrics that reflect overall cardiovascular and metabolic health of patients. Beyond these physiological benefits, SGLT2i have also been linked to improved quality of life, reinforcing their role in comprehensive cardiometabolic care [20].
Blood pressure reduction, nephron remodeling, reduction in arterial stiffness, are all well-known mechanisms associated with the CV benefits observed with the use of SGLT2i [21] but they are not sufficient to fully explain the metabolic effects.
The impact of SGLT2i on adipose tissue, in terms of body weight loss and the shift in energy source from glucose to fat [22], has been observed in both rodents and humans. These findings suggest potential mechanisms that could explain cardiovascular (CV) benefits. However, the molecular and metabolic mechanisms underlying this interaction remain not fully understood.
We thus decided to focus on the impact of SGLT2 inhibition on adipose tissue and its direct or indirect potential effects on the cardiovascular system.
The plasticity of adipose tissue
Adipose tissue, which includes both white adipose tissue (WAT) and brown adipose tissue (BAT) forms a unitary true organ in both rodents and humans, challenging the conventional view of adipose tissues as independent functional structures [23]. Recent studies have demonstrated the continuity between various adipose depots in the body underscoring the concept of a unitary adipose organ, which exhibits significant plasticity, since, as seen in rodents, it adapts to physiological changes such as cold exposure, obesity, and lactation, due to its role as an “endocrine organ” [23, 24].
WAT is the most abundant adipose tissue in the human body, mainly functioning as an energy storage depot, accumulating lipids in the form of large unilocular fat droplets within adipocytes. The most important function of WAT is its ability to release high energy molecules (fatty acids) in the intervals between meals, allowing survival for up to 6–7 weeks of fasting [25]. WAT also has very important endocrine properties which control the main behaviors for human survival (i.e. food search and intake [26, 27]) and play crucial roles in other important aspects of metabolism, immunity and inflammation by secreting hormones known as adipokines [25].
BAT is primarily responsible for thermogenesis, a process by which energy is dissipated as heat: this is made possible by the presence of numerous small lipid droplets and a high number of big mitochondria, rich in cristae, that contain uncoupling protein 1 (UCP1). This protein is highly specific and uniquely expressed in these mitochondria [28]. UCP1 allows BAT to generate heat by uncoupling oxidative phosphorylation from ATP production, a process especially vital for thermoregulation in cold environments. BAT activity has a positive effect on glucose metabolism and also performs endocrine functions [23, 25, 29, 30].
As mentioned, the adipose organ has plastic properties and conversion of WAT into BAT, and vice versa, has been widely demonstrated both in rodents and in humans [23, 31–33]. This process, known as “browning” of WAT, is characterized by the transformation of energy-storing white adipocytes into energy-burning brown adipocytes, primarily by the enhancement of UCP1 expression and thermogenesis in adipose tissue. SGLT2i have been shown to influence this process by promoting the release of Fibroblast growth factor 21, a key regulator that drives the upregulation of UCP1 and facilitates the metabolic reprogramming of adipocytes toward a thermogenic phenotype [34]. Morphologically, this phenomenon is characterized by the presence of beige adipocytes, within the adipose tissue, which exhibit features that are intermediate between white and brown adipocytes [25, 35]. The browning of WAT increases the body’s energy expenditure and has been linked to improved metabolic health [36, 37]. Cold exposure is the major inducer of browning both in mice and humans [38], but physical exercise [39–41], nutrients [42] and even some drugs also seem to play a role [43–47].
This distinction between white and brown adipose tissue highlights the plasticity of fat cells and their varying roles in energy metabolism and thermoregulation.
Fibroblast growth factor 21 (FGF-21), a part of the fibroblast growth factor family, has been identified as a crucial regulator in the differentiation of brown adipocytes and promotion of the “browning” of adipose tissue. FGF-21 is an important metabolic regulator that also affects glucose homeostasis, insulin sensitivity and ketogenesis. Studies in rodents have shown that BAT is not only a target for FGF-21 but also a significant source of systemic FGF-21: BAT expresses and releases FGF-21 in response to thermogenic activation, for example during cold exposure, indicating that BAT plays a role in the systemic regulation of FGF-21 [48]. This discovery supports the hypothesis that FGF-21 has a physiological role in thermogenesis and the thermogenic recruitment of white adipose tissue browning through an autocrine-paracrine axis highlighting the plasticity of the adipose organ and the interplay between the WAT and BAT.
The adipose organ is thus considered a true endocrine organ involved in the metabolic system and has become a target to curb and treat diseases [38, 49]. The great plasticity of adipose tissue, effortlessly shifting between energy storage and thermogenesis, consolidates its role as a key regulator of metabolism and a powerful target for therapy in cardiovascular and metabolic diseases.
The cross-talk between adipose tissue and the cardiovascular system
The complex functions of adipose tissue, recently recognized as an organ [23], make it a crucial regulator of health and diseases. WAT and BAT are found in both subcutaneous (below the skin) and visceral depots (in close contact with viscera). Several studies have outlined the different properties of these two depots, as visceral fat accumulation is associated with adverse metabolic outcomes [50–52].
Focusing on the cardiovascular system, the endocrine and paracrine effects of adipose tissue play a core role in its physiological and pathological functions, underlying the importance of exploring the crosstalk between them [53, 54]. In fact, adipose tissue may be protective or harmful, with quality instead of quantity, as the principal variable [53, 55].
The main visceral adipose tissue involved in the cardiovascular system is the epicardial adipose tissue (EAT). It has several characteristics that render it a unique adipose tissue depot: it is in direct contact with the myocardium, which implies that the two tissues share the same microcirculation, and it contains mainly brown fat, suggesting that it is a metabolically active fat depot [56]. Physiologically, the EAT exerts protective effects: it is a source of free fatty acids [57], it secretes cardioprotective adipokines with anti-inflammatory and anti-atherogenic properties [58], it contributes to myocardial redox homeostasis through EAT-derived miRNAs [59] and last, but not least, it provides a direct source of heat to the myocardium protecting it during unfavorable hemodynamic conditions such as ischemia or hypoxia [60, 61].
In pathological conditions, such as obesity and diabetes, the EAT brown fat-like activity decreases substantially and the proportion of brown adipocytes decreases in favor of white adipocytes [35], promoting inflammation with all the well-known inflammatory consequences and deleterious metabolic effects [62–66].
On the other hand, it has been shown that pro-inflammatory and oxidative stimuli in diseased vessels and/or the myocardium can modify adipose tissue biology, suggesting a bidirectional crosstalk between the adipose tissue and the cardiovascular system [67, 68].
Other visceral adipose tissue depots are also involved in the cardiovascular system, due to their systemic, protective or harmful effects. In fact, BAT, mainly expressed in the cervical-supraclavicular (most common), perirenal/adrenal, and paravertebral regions [69, 70], exerts protective effects on cardiometabolic health [71]. In 2021, Cohen et al. reported that individuals with active BAT had lower prevalences of cardiometabolic diseases, and the presence of BAT was independently correlated with lower odds of type 2 diabetes, dyslipidemia, coronary artery disease, cerebrovascular disease, congestive heart failure and hypertension [71]. On the other hand, when adipose tissue becomes dysfunctional, as in obesity, cardiometabolic risk increases [72].
For all the above-mentioned reasons, not only the EAT, but also the other visceral adipose tissue depots have now become therapeutical targets to curb or treat cardiometabolic diseases [49, 56, 73, 74]. Since our data suggest that in just 4 weeks of treatment with the SGLT2i dapagliflozin we can obtain a reduction in EAT thickness of 19%, which probably induces a 30% increase in coronary flow reserve [8, 9, 75], we will focus our attention on the direct or indirect effects of SGLT2i on the adipose organ.
How do SGLT2i influence the adipose organ and how can they enhance wat browning? In vitro and rodent data
Several studies have described the beneficial effects of SGLT2i on the adipose organ through different mechanisms. Studies in rodents have demonstrated that SGLT2i treatment increases the synthesis of the ketone 3-hydroxybutyrate, which modifies histone H3K9 expression in adipocytes, consequently upregulating adiponectin synthesis in adipocytes [76]. This effect has also been observed in humans, as demonstrated by clinical studies that highlight the increased production of 3-hydroxybutyrate following SGLT2i treatment, further supporting its role in metabolic reprogramming [77]. Adiponectin is one of the most abundant adipokines secreted by WAT and enhances insulin sensitivity [78–80]. Indeed, adiponectin induces the activation of M2 anti-inflammatory macrophages, an important source of catecholamine for the activation of WAT browning [81].
Additionally, in a female rat model of polycystic ovary syndrome (PCOS) and insulin resistance, Pruett et al. showed that the administration of the SGLT2i empagliflozin for 4 weeks, (compared to a control group), reduced fat mass, leading to morphological changes in adipose tissue, specifically an increase in the number of small and medium-sized adipocytes and a reduction in the number of large adipocytes [82], an indirect sign of browning [25]. In fact, they found an increased expression of genes involved in mitochondrial biogenesis and fatty acid oxidation (i.e., browning induction), such as PGC1α and NRF1in WAT and the mitochondrial antioxidant enzyme superoxide dismutase (SOD2) in SAT. This suggests that SGLT2 have the potential to improve mitochondrial function and reduce oxidative stress in WAT [82]. Although the authors did not observe a significant reduction in oxidative stress after 4 weeks of treatment, other studies have described this reduction in male murine models, in which SGLT2i reduced oxidative stress and, consequently, improved mitochondrial function, thus positively influencing insulin resistance [83–85].
Studies conducted on rodents and human cardiac tissue have demonstrated that canagliflozin reduces oxidative stress by enhancing mitochondrial function and decreasing production of reactive oxygen species (ROS). This effect contributes to improved cardiac function, particularly in conditions such as heart failure and diabetic cardiomyopathy [86].
Recently, a study by Di Vincenzo et al. has investigated the impact of the SGLT2i dapagliflozin on the expression of FGF-21, one of the main inducers of WAT browning [87]. The authors assessed the gene expression of FGF-21 in myocardial tissue of obese rodents at baseline and after a 4-week treatment, and found that dapagliflozin treatment increases FGF-21 gene expression and reduces triglyceride content in myocardial tissue, indirectly suggesting that dapagliflozin promotes a browning effect [88].
Beyond these effects, it seems that dapagliflozin can promote the browning of WAT through the regulation of angiogenesis in adipose tissue. In obese rodents, Xiang et al. described an increased expression of angiogenic factors promoting the formation of new blood vessels in WAT together with a significant increase in the expression of browning genes UCP1, PGC-1α and PRDM16 (PR domain-containing 16). In this way, SGLT2i improve oxygen and nutrient delivery to the adipocytes, facilitating their metabolic shift towards a more energy-consuming phenotype thus promoting the browning process [89].
To summarize these findings, SGLT2i act on adipose tissue primarily by enhancing fat burning, increasing 3-hydroxybutyrate levels, which modify histone H3K9 methylation and acetylation in adipocytes, leading to an increase in adiponectin synthesis. The increased adiponectin induces activation of M2 anti-inflammatory macrophages, which promotes WAT browning with a consequent positive metabolic impact. In addition, in vitro and in vivo murine models have demonstrated that SGLT2i reduce fat mass, influencing adipocyte size, and upregulating genes involved in mitochondrial biogenesis and fatty acid oxidation, suggesting potential improvement in mitochondrial function and reduced oxidative stress. Lastly, SGLT2i can indirectly influence WAT metabolism by increasing the expression of myocardial FGF-21, which promotes angiogenesis and upregulates genes associated with brown adipocyte characteristics, such as UCP1, PGC-1α, and PRDM16, shifting white adipose tissue from energy storage to energy dissipation thus confirming the positive metabolic effect of this class of drugs.
Effects of SGLT2i on human adipose tissue
The positive effects of SGLT2i on the adipose organ, observed in vitro and in rodent models, also seem to be present in humans with mechanisms that go beyond simple glucose excretion. Indeed, it seems that the chronic glycosuria caused by SGLT2 inhibition leads to an adaptive increase in energy intake [22]. It has been demonstrated that the observed weight loss in patients treated with the SGLT2i empagliflozin, for example, was consistently less pronounced than expected, based on the amount of urinary glucose excretion [22]. This discrepancy suggests an adaptive increase in caloric intake as a compensatory mechanism for the calories lost through glycosuria, implying that it is necessary to combine SGLT2i with caloric restriction for major weight loss [22].
These findings were later expanded by the discovery of parallels between SGLT2i treatment and fasting-mimicking diets, suggesting that SGLT2i mimic the fasting state not only by promoting glycosuria and consequently reducing insulin concentration, a potent inhibitor of lipolysis, but also by influencing free fatty acid (FFA) metabolism by prioritizing FFA as the primary energy source for oxidation. This shift to FFA oxidation leads to increased circulating ketone bodies, resulting in more oxygen-efficient energy production, suggesting possible beneficial effects on adipose tissue morphology and physiology (i.e. browning) as observed in rodents [78–80], and also in humans [90].
The shift to FFA oxidation as a primary energy source with SGLT2i treatment, as observed in rodent models [91], could play a pivotal role in reducing fat mass. Moreover, by prioritizing FFA as the primary source of oxidation, SGLT2i signal an increased need for glucose to the cell. This leads to a higher number of GLUT4 being translocated to the cell membrane of adipocytes, improving insulin sensitivity [92, 93].
Regarding the anti-inflammatory effect seen in vitro and in rodent models, Buttice et al. recently conducted a systematic review and meta-analysis focusing on the effect of SGLT2i on inflammatory markers, adipokine profiles and insulin sensitivity, and found that SGLT2i significantly improve levels of adiponectin, interleukin-6 (IL-6), and tumor necrosis factor receptor-1 (TNF-r1). Moreover, they found an improvement in the homeostatic model assessment of insulin resistance (HOMA-IR) although there were no significant changes in other inflammatory biomarkers such as C-reactive protein (CRP), tumor necrosis factor-α (TNF-α), plasminogen activator inhibitor-1 (PAI-1), or FGF-21 levels, as seen in vitro and in murine models [94].
Further, a significant body of research has highlighted the impact of SGLT2i on adipose tissue distribution. Systematic reviews and meta-analyses of randomized controlled trials in patients with T2DM have consistently shown that SGLT2i substantially reduce not only body weight and body mass index (BMI) but also waist circumference and visceral fat area. In a systematic review and meta-analysis of randomized controlled trials, Wang X. et al., provide evidence that in patients with T2DM, SGLT-2 inhibitors significantly reduce, though to different extents, visceral adipose tissue, subcutaneous adipose tissue, and ectopic liver fat. However, the high heterogeneity of the studies included suggests caution in drawing conclusions [95]. Overall, SGLT2 inhibitors seem to have a more pronounced effect on VAT and EAT compared to SAT leading to a reduction of inflammation and oxidative stress, which is associated with better metabolic outcomes. (Table 1).
Table 1.
In humans, all the data suggest beneficial effects on the adipose organ even though direct evidence of browning of adipose tissue is still lacking.
Are SGLT2i cardiovascular benefits mediated by their effect on the adipose organ?
Several reviews and meta-analyses have highlighted the significant benefits of SGLT2i on cardiovascular outcomes in patients with diabetes, showing reduced risks of all-cause mortality, hospitalization for heart failure, CV death, and the composite of hospitalization for heart failure or CV death, regardless of baseline diabetes status.
Several mechanisms have been proposed to explain the beneficial CV effects of SGLT2i, namely reduction of blood pressure and intravascular volume, and reduction of myocardial oxygen consumption, all of which can lead to a decreased workload on the heart and improved cardiac function [86, 107]. However, recent studies suggest that CV benefits may be linked, in part, to the effects of SGLT2i on insulin resistance, a necessary condition in the pathogenesis of T2DM [108]. Thus, since adipose tissue is a fundamental part of insulin resistance, the direct action of these drugs on this tissue cannot be neglected [109, 110]. Indeed, by improving insulin sensitivity and reducing insulin resistance, SGLT2i can indirectly impact the metabolic profile, further contributing to cardiovascular protection [5]. A series of recently published studies have highlighted the significance of the effect of SGLT2i on epicardial adipose tissue (EAT) reporting that SGLT2i treatment led to a significant reduction in EAT thickness (about 19%) [111–113], the main adipose tissue depot implicated in CV risk (Table 2). It is important to note that EAT volume is a more sensitive measure than EAT thickness, with imaging modalities such as MRI or CT providing greater sensitivity compared to ultrasound. Furthermore, volumetric assessments using Simpson’s disc method are more accurate than those based on geometric assumptions. These advanced methodologies ensure a more precise evaluation of EAT, enhancing our understanding of its role in CV risk and the impact of SGLT2i therapeutic interventions [114].
Table 2.
Our group has also recently described a selective decrease in EAT glucose uptake (about 21.6%) in patients with T2DM and CAD after a 4-week treatment with dapagliflozin [9]. This could suggest that the selective reduction in EAT thickness and glucose uptake could be due to a decrease in EAT inflammation, which is a known risk factor for CV diseases [56, 73, 121–123]. We have previously described a 30% increase in myocardial flow reserve [8] and we speculate that SGLT-2i treatment, by restoring the anti-inflammatory properties of EAT, improves coronary microvascular dysfunction, leading to an amelioration of myocardial flow reserve [9]. We can also speculate that, dapagliflozin treatment longer than 4 weeks may be able to restore the brown feature of EAT [124].
The CV benefits of SGLT-2i can thus, in part, be ascribed to their effect on adipose tissue, even though further experiments are needed to confirm this.
Conclusion
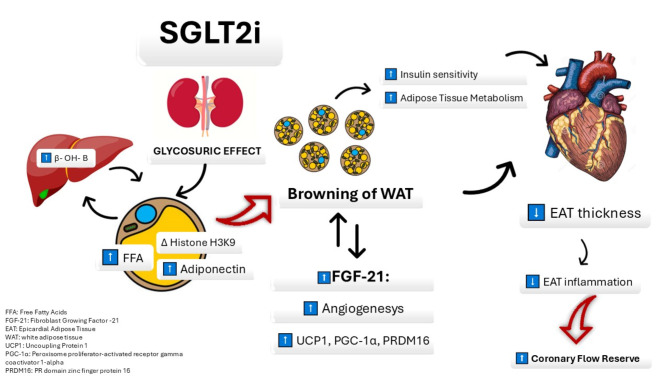
In conclusion, SGLT2i have been shown to have a fundamental role in T2DM and weight control not only by facilitating glycosuria but also by effects mediated by adipose tissue metabolism. These findings underscore the role of SGLT2i in modulating adipocyte function, promoting the browning of white adipose tissue, and enhancing insulin sensitivity (Fig. 1).
Fig. 1.
SGLT2i, by inducing glycosuria, lead to the hepatic production of the ketone β-OH-B, which stimulates FFA production in adipose tissue. This process also modifies histone H3K9, leading to increased adiponectin levels and promoting the browning of white adipose tissue (WAT). Consequently, the activation of BAT increases the secretion of FGF21, promoting brown adipose tissue (BAT) activity and browning of WAT (confirmed by the increase of UCP1, PGC1α, and PRDM16 expression). When BAT activity increases, insulin sensitivity and metabolism are enhanced. All these effects are reflected in the cardiovascular system at epicardial adipose tissue (EAT) level, where SGLT2i induce a reduction in EAT thickness which leads to a reduction of EAT inflammation, improving coronary flow reserve (CFR)
SGLT2i exhibit a direct influence on adipose tissue, characterized by alterations in adipocyte cytokine production (adipokines), which enhance insulin sensitivity and mitigate the progression of T2DM. Furthermore, these medications have been linked to the redistribution of adipose tissue, favoring a reduction in visceral fat associated with metabolic benefits. This action suggests a decrease in the pro-inflammatory state of adipose tissue, offering a new pathway for mitigating the cardiovascular and renal risks associated with metabolic syndromes.
Therefore, the clinical relevance of SGLT2i encompasses cardiovascular protection, weight management, and renal benefits. Their role in improving heart function and reducing heart failure incidence highlights the potential of these drugs to play a decisive role in the multidisciplinary treatment of T2DM patients with high cardiovascular risk. Additionally, their renal protective effects make them a cornerstone of the current therapeutic arsenal.
Given the diverse benefits of SGLT2i, there are ongoing investigations in several areas. The mechanisms underlying the different effects on various adipose tissues, including epicardial adipose tissue, and their long-term implications for cardiovascular and metabolic health are areas still ripe for exploration. Considering all the direct or indirect beneficial effects on adipose tissue metabolism, further investigations should be conducted into the use of SGLT2i in all dysfunctional adipose tissue– related diseases, such as obesity, metabolic-dysfunction associated fatty liver disease (MAFLD), insulin resistance per se and even cancer, in which they could exert bidirectional benefits, namely removing the primary energy source of tumor cells (glucose) and improving adipose tissue metabolism whose dysfunction has been strongly associated with the development of several types of cancer [125].
Future research should thus focus on further investigation of the effects of SGLT2i on the adipose organ, beyond the conventional markers of T2DM control.
Acknowledgements
The authors thank Serena Rotunno for editorial assistance in the writing of this article.
Abbreviations
- CAD
Coronary artery disease
- EAT
Epicardial adipose tissue
- HF
Heart failure
- SGLT2i
Sodium-glucose cotransporter-2 inhibitors
- T2DM
Type 2 diabetes
- CV
Cardiovascular
- MACE
Major adverse cardiovascular events
- SAT
Subcutaneous adipose tissue
- CFR
Coronary flow reserve
- WAT
White adipose tissue
- BAT
Brown adipose tissue
- UCP 1
Uncoupling protein 1
- FGF-21
Fibroblast growth factor 21
- PCOS
Polycystic ovary syndrome
- SOD2
Enzyme superoxide dismutase
- PRDM16
PR domain-containing 16
- FFAs
Free fatty acids
- IL-6
Interleukin-6
- TNF-r1
Tumor necrosis factor receptor-1
- TNF-α
Tumor necrosis factor-α
- PAI-1
Plasminogen activator inhibitor-1
Author contributions
C.M, A.G and F.C wrote the main manuscript. S.G, U.C, G.D.G., T.M., G.C., S.M., L.S., M.B., A.A., A.S. and A.P. wrote parts of manuscript. C.M. prepared the graphical abstract. C.M., A.G. and F.C. edited and formatted the manuscript. All authors reviewed the manuscript.All authors have read and agreed to the published version of the manuscript.
Funding
Not applicable.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Conflict of interest
The authors declare no competing interests.
Informed consent
Not applicable.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Zinman B, et al. Cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–28. 10.1056/NEJMoa1504720. Empagliflozin. [DOI] [PubMed] [Google Scholar]
- 2.Mahaffey KW, et al. Canagliflozin for primary and Secondary Prevention of Cardiovascular Events: results from the CANVAS Program (Canagliflozin Cardiovascular Assessment Study). Circulation. 2018;137:323–34. 10.1161/CIRCULATIONAHA.117.032038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wiviott SD, et al. Dapagliflozin and Cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347–57. 10.1056/NEJMoa1812389. [DOI] [PubMed] [Google Scholar]
- 4.Heerspink HJL, et al. Dapagliflozin in patients with chronic kidney disease. N Engl J Med. 2020;383:1436–46. 10.1056/NEJMoa2024816. [DOI] [PubMed] [Google Scholar]
- 5.Ni L, Yuan C, Chen G, Zhang C, Wu X. SGLT2i: beyond the glucose-lowering effect. Cardiovasc Diabetol. 2020;19:98. 10.1186/s12933-020-01071-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hallow KM, Helmlinger G, Greasley PJ, McMurray JJV, Boulton DW. Why do SGLT2 inhibitors reduce heart failure hospitalization? A differential volume regulation hypothesis. Diabetes Obes Metab. 2018;20:479–87. 10.1111/dom.13126. [DOI] [PubMed] [Google Scholar]
- 7.Zaawari A, Sahar AN. Sodium-glucose co-transporter-2 inhibitors (SGLT2I): a class of drugs with promising cardiorenal protective effects beyond glycemic control. Ann Med Surg (Lond). 2022;81:104536. 10.1016/j.amsu.2022.104536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Leccisotti L, et al. Dapagliflozin improves myocardial flow reserve in patients with type 2 diabetes: the DAPAHEART Trial: a preliminary report. Cardiovasc Diabetol. 2022;21:173. 10.1186/s12933-022-01607-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cinti F, et al. Dapagliflozin treatment is associated with a reduction of epicardial adipose tissue thickness and epicardial glucose uptake in human type 2 diabetes. Cardiovasc Diabetol. 2023;22:349. 10.1186/s12933-023-02091-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sorice GP, et al. Effect of Dapagliflozin on myocardial insulin sensitivity and perfusion: Rationale and Design of the DAPAHEART Trial. Diabetes Ther. 2021;12:2101–13. 10.1007/s13300-021-01083-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li L, et al. SGLT2i alleviates epicardial adipose tissue inflammation by modulating ketone body-glyceraldehyde-3-phosphate dehydrogenase malonylation pathway. J Cardiovasc Med (Hagerstown). 2023;24:232–43. 10.2459/JCM.0000000000001453. [DOI] [PubMed] [Google Scholar]
- 12.Hotamisligil GS. Inflammation, metaflammation and immunometabolic disorders. Nature. 2017;542:177–85. 10.1038/nature21363. [DOI] [PubMed] [Google Scholar]
- 13.The L. Treating obesity and diabetes: drugs alone are not enough. Lancet. 2024;403:1. 10.1016/S0140-6736(24)00003-5. [DOI] [PubMed] [Google Scholar]
- 14.Melson E, Ashraf U, Papamargaritis D, Davies MJ. What is the pipeline for future medications for obesity? Int J Obes (Lond). 2024. 10.1038/s41366-024-01473-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Caruso I, et al. Glucometabolic outcomes of GLP-1 receptor agonist-based therapies in patients with type 2 diabetes: a systematic review and network meta-analysis. EClinicalMedicine. 2023;64:102181. 10.1016/j.eclinm.2023.102181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Padda IS, Mahtani AU, Parmar M. in StatPearls (2024).
- 17.Jiang M, Steyger PS. An evaluation of US patent 2015065565 (A1) for a new class of SGLT2 inhibitors for treatment 1 of type II diabetes mellitus. Expert Opin Ther Pat. 2015;25:1349–52. 10.1517/13543776.2015.1076392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Giugliano D, et al. SGLT-2 inhibitors and cardiorenal outcomes in patients with or without type 2 diabetes: a meta-analysis of 11 CVOTs. Cardiovasc Diabetol. 2021;20:236. 10.1186/s12933-021-01430-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Davies MJ, et al. Cardiovascular outcomes trials: a paradigm shift in the current management of type 2 diabetes. Cardiovasc Diabetol. 2022;21:144. 10.1186/s12933-022-01575-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gao M, et al. SGLT2 inhibitors, functional capacity, and quality of life in patients with heart failure: a systematic review and Meta-analysis. JAMA Netw Open. 2024;7:e245135. 10.1001/jamanetworkopen.2024.5135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vallon V. State-of-the-art-review mechanisms of action of SGLT2 inhibitors and clinical implications. Am J Hypertens. 2024. 10.1093/ajh/hpae092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ferrannini G, et al. Energy Balance after Sodium-glucose cotransporter 2 inhibition. Diabetes Care. 2015;38:1730–5. 10.2337/dc15-0355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Giordano A, et al. The adipose organ is a Unitary structure in mice and humans. Biomedicines. 2022;10. 10.3390/biomedicines10092275. [DOI] [PMC free article] [PubMed]
- 24.Cinti S. Pink adipocytes. Trends Endocrinol Metab. 2018;29:651–66. 10.1016/j.tem.2018.05.007. [DOI] [PubMed] [Google Scholar]
- 25.Cinti S. Adipose Organ Development and Remodeling. Compr Physiol. 2018;8:1357–431. 10.1002/cphy.c170042. [DOI] [PubMed] [Google Scholar]
- 26.Zhang Y, et al. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994;372:425–32. 10.1038/372425a0. [DOI] [PubMed] [Google Scholar]
- 27.Duerrschmid C, et al. Asprosin is a centrally acting orexigenic hormone. Nat Med. 2017;23:1444–53. 10.1038/nm.4432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Frontini A, et al. Thymus uncoupling protein 1 is exclusive to typical brown adipocytes and is not found in thymocytes. J Histochem Cytochem. 2007;55:183–9. 10.1369/jhc.6A7013.2006. [DOI] [PubMed] [Google Scholar]
- 29.Cannon B, Nedergaard J. Brown adipose tissue: function and physiological significance. Physiol Rev. 2004;84:277–359. 10.1152/physrev.00015.2003. [DOI] [PubMed] [Google Scholar]
- 30.Giralt M, Cereijo R, Villarroya F. Adipokines and the endocrine role of adipose tissues. Handb Exp Pharmacol. 2016;233:265–82. 10.1007/164_2015_6. [DOI] [PubMed] [Google Scholar]
- 31.Barbatelli G, et al. The emergence of cold-induced brown adipocytes in mouse white fat depots is determined predominantly by white to brown adipocyte transdifferentiation. Am J Physiol Endocrinol Metab. 2010;298:E1244–1253. 10.1152/ajpendo.00600.2009. [DOI] [PubMed] [Google Scholar]
- 32.Frontini A, et al. White-to-brown transdifferentiation of omental adipocytes in patients affected by pheochromocytoma. Biochim Biophys Acta. 2013;1831:950–9. 10.1016/j.bbalip.2013.02.005. [DOI] [PubMed] [Google Scholar]
- 33.Ronkainen J, et al. Fto-Deficiency affects the gene and MicroRNA expression involved in Brown Adipogenesis and Browning of White Adipose tissue in mice. Int J Mol Sci. 2016;17. 10.3390/ijms17111851. [DOI] [PMC free article] [PubMed]
- 34.Osataphan S, et al. SGLT2 inhibition reprograms systemic metabolism via FGF21-dependent and -independent mechanisms. JCI Insight. 2019;4. 10.1172/jci.insight.123130. [DOI] [PMC free article] [PubMed]
- 35.Sacks HS, et al. Adult epicardial fat exhibits beige features. J Clin Endocrinol Metab. 2013;98:E1448–1455. 10.1210/jc.2013-1265. [DOI] [PubMed] [Google Scholar]
- 36.Rosen ED, Spiegelman BM. What we talk about when we talk about fat. Cell. 2014;156:20–44. 10.1016/j.cell.2013.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Barbera MJ, et al. Peroxisome proliferator-activated receptor alpha activates transcription of the brown fat uncoupling protein-1 gene. A link between regulation of the thermogenic and lipid oxidation pathways in the brown fat cell. J Biol Chem. 2001;276:1486–93. 10.1074/jbc.M006246200. [DOI] [PubMed] [Google Scholar]
- 38.Giordano A, Smorlesi A, Frontini A, Barbatelli G, Cinti S. White, brown and pink adipocytes: the extraordinary plasticity of the adipose organ. Eur J Endocrinol. 2014;170:R159–171. 10.1530/EJE-13-0945. [DOI] [PubMed] [Google Scholar]
- 39.De Matteis R, et al. Exercise as a new physiological stimulus for brown adipose tissue activity. Nutr Metab Cardiovasc Dis. 2013;23:582–90. 10.1016/j.numecd.2012.01.013. [DOI] [PubMed] [Google Scholar]
- 40.Bostrom P, et al. A PGC1-alpha-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8. 10.1038/nature10777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ronkainen J, et al. Fat mass- and obesity-associated gene fto affects the dietary response in mouse white adipose tissue. Sci Rep. 2015;5:9233. 10.1038/srep09233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Yoneshiro T, Matsushita M, Saito M. Translational aspects of Brown Fat activation by Food-Derived stimulants. Handb Exp Pharmacol. 2019;251:359–79. 10.1007/164_2018_159. [DOI] [PubMed] [Google Scholar]
- 43.Cypess AM, et al. Activation of human brown adipose tissue by a beta3-adrenergic receptor agonist. Cell Metab. 2015;21:33–8. 10.1016/j.cmet.2014.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gavalda-Navarro A, Villarroya J, Cereijo R, Giralt M, Villarroya F. The endocrine role of brown adipose tissue: an update on actors and actions. Rev Endocr Metab Disord. 2022;23:31–41. 10.1007/s11154-021-09640-6. [DOI] [PubMed] [Google Scholar]
- 45.Armani A, et al. Mineralocorticoid receptor antagonism induces browning of white adipose tissue through impairment of autophagy and prevents adipocyte dysfunction in high-fat-diet-fed mice. FASEB J. 2014;28:3745–57. 10.1096/fj.13-245415. [DOI] [PubMed] [Google Scholar]
- 46.Marzolla V, et al. The role of the mineralocorticoid receptor in adipocyte biology and fat metabolism. Mol Cell Endocrinol. 2012;350:281–8. 10.1016/j.mce.2011.09.011. [DOI] [PubMed] [Google Scholar]
- 47.Mammi C, et al. A novel combined glucocorticoid-mineralocorticoid receptor selective modulator markedly prevents weight gain and fat mass expansion in mice fed a high-fat diet. Int J Obes (Lond). 2016;40:964–72. 10.1038/ijo.2016.13. [DOI] [PubMed] [Google Scholar]
- 48.Ni B, Farrar JS, Vaitkus JA, Celi FS. Metabolic effects of FGF-21: Thermoregulation and Beyond. Front Endocrinol (Lausanne). 2015;6:148. 10.3389/fendo.2015.00148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Giordano A, Frontini A, Cinti S. Convertible visceral fat as a therapeutic target to curb obesity. Nat Rev Drug Discov. 2016;15:405–24. 10.1038/nrd.2016.31. [DOI] [PubMed] [Google Scholar]
- 50.Vague J. The degree of masculine differentiation of obesities: a factor determining predisposition to diabetes, atherosclerosis, gout, and uric calculous disease. Am J Clin Nutr. 1956;4:20–34. 10.1093/ajcn/4.1.20. [DOI] [PubMed] [Google Scholar]
- 51.Bowman K, et al. Central adiposity and the overweight risk paradox in aging: follow-up of 130,473 UK Biobank participants. Am J Clin Nutr. 2017;106:130–5. 10.3945/ajcn.116.147157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Guglielmi V, et al. Omental adipose tissue fibrosis and insulin resistance in severe obesity. Nutr Diabetes. 2015;5:e175. 10.1038/nutd.2015.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Oikonomou EK, Antoniades C. The role of adipose tissue in cardiovascular health and disease. Nat Rev Cardiol. 2019;16:83–99. 10.1038/s41569-018-0097-6. [DOI] [PubMed] [Google Scholar]
- 54.Badi I, et al. Editorial: exploring the Crosstalk between adipose tissue and the Cardiovascular System. Front Cell Dev Biol. 2022;10:973135. 10.3389/fcell.2022.973135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Akoumianakis I, Antoniades C. The interplay between adipose tissue and the cardiovascular system: is fat always bad? Cardiovasc Res. 2017;113:999–1008. 10.1093/cvr/cvx111. [DOI] [PubMed] [Google Scholar]
- 56.Iacobellis G. Epicardial adipose tissue in contemporary cardiology. Nat Rev Cardiol. 2022;19:593–606. 10.1038/s41569-022-00679-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Marchington JM, Mattacks CA, Pond CM. Adipose tissue in the mammalian heart and pericardium: structure, foetal development and biochemical properties. Comp Biochem Physiol B. 1989;94:225–32. 10.1016/0305-0491(89)90337-4. [DOI] [PubMed] [Google Scholar]
- 58.Iacobellis G, Bianco AC. Epicardial adipose tissue: emerging physiological, pathophysiological and clinical features. Trends Endocrinol Metab. 2011;22:450–7. 10.1016/j.tem.2011.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Carena MC, et al. Role of human epicardial adipose tissue-derived miR-92a-3p in myocardial redox state. J Am Coll Cardiol. 2023;82:317–32. 10.1016/j.jacc.2023.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Badi I, Antoniades C. Brown Adipose tissue and the take (12,13-di)HOME Message to the heart. Circulation. 2021;143:160–2. 10.1161/CIRCULATIONAHA.120.051981. [DOI] [PubMed] [Google Scholar]
- 61.Pinckard KM, et al. A novel endocrine role for the BAT-Released lipokine 12,13-diHOME to Mediate Cardiac function. Circulation. 2021;143:145–59. 10.1161/CIRCULATIONAHA.120.049813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Mazurek T, et al. Human epicardial adipose tissue is a source of inflammatory mediators. Circulation. 2003;108:2460–6. 10.1161/01.CIR.0000099542.57313.C5. [DOI] [PubMed] [Google Scholar]
- 63.Li Y, et al. Epicardial fat tissue in patients with diabetes mellitus: a systematic review and meta-analysis. Cardiovasc Diabetol. 2019;18:3. 10.1186/s12933-019-0807-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Malavazos AE, et al. The density of crown-like structures in epicardial adipose tissue could play a role in cardiovascular diseases. Eat Weight Disord. 2022;27:2905–10. 10.1007/s40519-022-01420-8. [DOI] [PubMed] [Google Scholar]
- 65.Weisberg SP, et al. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796–808. 10.1172/JCI19246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kotzbeck P, et al. Brown adipose tissue whitening leads to brown adipocyte death and adipose tissue inflammation. J Lipid Res. 2018;59:784–94. 10.1194/jlr.M079665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Valori C, Thomas M, Shillingford J. Free noradrenaline and adrenaline excretion in relation to clinical syndromes following myocardial infarction. Am J Cardiol. 1967;20:605–17. 10.1016/0002-9149(67)90001-x. [DOI] [PubMed] [Google Scholar]
- 68.Wang L, et al. Cardiac ischemia modulates white adipose tissue in a depot-specific manner. Front Physiol. 2022;13:1036945. 10.3389/fphys.2022.1036945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Cypess AM, et al. Identification and importance of brown adipose tissue in adult humans. N Engl J Med. 2009;360:1509–17. 10.1056/NEJMoa0810780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Tam CS, Lecoultre V, Ravussin E. Brown adipose tissue: mechanisms and potential therapeutic targets. Circulation. 2012;125:2782–91. 10.1161/CIRCULATIONAHA.111.042929. [DOI] [PubMed] [Google Scholar]
- 71.Becher T, et al. Brown adipose tissue is associated with cardiometabolic health. Nat Med. 2021;27:58–65. 10.1038/s41591-020-1126-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Valenzuela PL, et al. Obesity and the risk of cardiometabolic diseases. Nat Rev Cardiol. 2023;20:475–94. 10.1038/s41569-023-00847-5. [DOI] [PubMed] [Google Scholar]
- 73.Antoniades C, et al. Perivascular adipose tissue as a source of therapeutic targets and clinical biomarkers. Eur Heart J. 2023. 10.1093/eurheartj/ehad484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Iacobellis G, Baroni MG. Cardiovascular risk reduction throughout GLP-1 receptor agonist and SGLT2 inhibitor modulation of epicardial fat. J Endocrinol Invest. 2022;45:489–95. 10.1007/s40618-021-01687-1. [DOI] [PubMed] [Google Scholar]
- 75.Capece U, et al. Dapagliflozin-Induced myocardial Flow Reserve Improvement is not Associated with HDL ability to stimulate endothelial nitric oxide production. Diabetes Ther. 2023. 10.1007/s13300-023-01491-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Santos-Gallego CG, et al. Empagliflozin ameliorates adverse left ventricular remodeling in nondiabetic heart failure by enhancing myocardial energetics. J Am Coll Cardiol. 2019;73:1931–44. 10.1016/j.jacc.2019.01.056. [DOI] [PubMed] [Google Scholar]
- 77.Santos-Gallego CG, Mayr M, Badimon J. SGLT2 inhibitors in Heart failure: targeted metabolomics and energetic metabolism. Circulation. 2022;146:819–21. 10.1161/CIRCULATIONAHA.122.060805. [DOI] [PubMed] [Google Scholar]
- 78.Nishitani S, et al. Metabolomic and microarray analyses of adipose tissue of dapagliflozin-treated mice, and effects of 3-hydroxybutyrate on induction of adiponectin in adipocytes. Sci Rep. 2018;8:8805. 10.1038/s41598-018-27181-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Youssef ME, et al. Unlocking the full potential of SGLT2 inhibitors: expanding applications beyond Glycemic Control. Int J Mol Sci. 2023;24. 10.3390/ijms24076039. [DOI] [PMC free article] [PubMed]
- 80.Miyachi Y, et al. A reduced M1-like/M2-like ratio of macrophages in healthy adipose tissue expansion during SGLT2 inhibition. Sci Rep. 2018;8:16113. 10.1038/s41598-018-34305-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hui X, et al. Adiponectin enhances Cold-Induced Browning of Subcutaneous Adipose tissue via promoting M2 macrophage proliferation. Cell Metab. 2015;22:279–90. 10.1016/j.cmet.2015.06.004. [DOI] [PubMed] [Google Scholar]
- 82.Pruett JE, et al. Mitochondrial function and oxidative stress in white adipose tissue in a rat model of PCOS: effect of SGLT2 inhibition. Biol Sex Differ. 2022;13:45. 10.1186/s13293-022-00455-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Croteau D, et al. Effects of Sodium-glucose linked transporter 2 inhibition with ertugliflozin on mitochondrial function, energetics, and metabolic gene expression in the Presence and absence of diabetes Mellitus in mice. J Am Heart Assoc. 2021;10:e019995. 10.1161/JAHA.120.019995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Woods TC, et al. Canagliflozin Prevents Intrarenal Angiotensinogen Augmentation and mitigates kidney Injury and Hypertension in Mouse Model of type 2 diabetes Mellitus. Am J Nephrol. 2019;49:331–42. 10.1159/000499597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Takagi S, et al. Ipragliflozin improves mitochondrial abnormalities in renal tubules induced by a high-fat diet. J Diabetes Investig. 2018;9:1025–32. 10.1111/jdi.12802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kondo H, et al. Effects of canagliflozin on human myocardial redox signalling: clinical implications. Eur Heart J. 2021;42:4947–60. 10.1093/eurheartj/ehab420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cuevas-Ramos D, Mehta R, Aguilar-Salinas CA. Fibroblast growth factor 21 and Browning of White Adipose tissue. Front Physiol. 2019;10:37. 10.3389/fphys.2019.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Di Vincenzo A, et al. Treatment with dapagliflozin increases FGF-21 gene expression and reduces triglycerides content in myocardial tissue of genetically obese mice. J Endocrinol Invest. 2024. 10.1007/s40618-023-02273-3. [DOI] [PubMed] [Google Scholar]
- 89.Xiang L, et al. Dapagliflozin promotes white adipose tissue browning though regulating angiogenesis in high fat induced obese mice. BMC Pharmacol Toxicol. 2024;25:26. 10.1186/s40360-024-00747-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Giaccari A. Sodium-glucose co-transporter inhibitors: medications that mimic fasting for cardiovascular prevention. Diabetes Obes Metab. 2019;21:2211–8. 10.1111/dom.13814. [DOI] [PubMed] [Google Scholar]
- 91.Wallenius K, et al. The SGLT2 inhibitor dapagliflozin promotes systemic FFA mobilization, enhances hepatic beta-oxidation, and induces ketosis. J Lipid Res. 2022;63:100176. 10.1016/j.jlr.2022.100176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Diaz-Rodriguez E, et al. Effects of dapagliflozin on human epicardial adipose tissue: modulation of insulin resistance, inflammatory chemokine production, and differentiation ability. Cardiovasc Res. 2018;114:336–46. 10.1093/cvr/cvx186. [DOI] [PubMed] [Google Scholar]
- 93.Merovci A, et al. Dapagliflozin lowers plasma glucose concentration and improves beta-cell function. J Clin Endocrinol Metab. 2015;100:1927–32. 10.1210/jc.2014-3472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Buttice L, et al. The effect of sodium-glucose cotransporter-2 inhibitors on inflammatory biomarkers: a meta-analysis of randomized controlled trials. Diabetes Obes Metab. 2024;26:2706–21. 10.1111/dom.15586. [DOI] [PubMed] [Google Scholar]
- 95.Wang X, Wu N, Sun C, Jin D, Lu H. Effects of SGLT-2 inhibitors on adipose tissue distribution in patients with type 2 diabetes mellitus: a systematic review and meta-analysis of randomized controlled trials. Diabetol Metab Syndr. 2023;15:113. 10.1186/s13098-023-01085-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Bando Y, et al. The effects of ipragliflozin on the liver-to-spleen attenuation ratio as assessed by computed tomography and on alanine transaminase levels in Japanese patients with type 2 diabetes mellitus. Diabetol Int. 2017;8:218–27. 10.1007/s13340-016-0302-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Eriksson JW, et al. Effects of dapagliflozin and n-3 carboxylic acids on non-alcoholic fatty liver disease in people with type 2 diabetes: a double-blind randomised placebo-controlled study. Diabetologia. 2018;61:1923–34. 10.1007/s00125-018-4675-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ito D, et al. Comparison of Ipragliflozin and Pioglitazone effects on nonalcoholic fatty liver disease in patients with type 2 diabetes: a Randomized, 24-Week, Open-Label, active-controlled trial. Diabetes Care. 2017;40:1364–72. 10.2337/dc17-0518. [DOI] [PubMed] [Google Scholar]
- 99.Koshizaka M, et al. Comparing the effects of ipragliflozin versus metformin on visceral fat reduction and metabolic dysfunction in Japanese patients with type 2 diabetes treated with sitagliptin: a prospective, multicentre, open-label, blinded-endpoint, randomized controlled study (PRIME-V study). Diabetes Obes Metab. 2019;21:1990–5. 10.1111/dom.13750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Latva-Rasku A, et al. The SGLT2 inhibitor Dapagliflozin reduces Liver Fat but does not affect tissue insulin sensitivity: a Randomized, Double-Blind, placebo-controlled study with 8-Week treatment in type 2 diabetes patients. Diabetes Care. 2019;42:931–7. 10.2337/dc18-1569. [DOI] [PubMed] [Google Scholar]
- 101.Inoue H, et al. Ipragliflozin, a sodium-glucose cotransporter 2 inhibitor, reduces bodyweight and fat mass, but not muscle mass, in Japanese type 2 diabetes patients treated with insulin: a randomized clinical trial. J Diabetes Investig. 2019;10:1012–21. 10.1111/jdi.12985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.McCrimmon RJ, et al. Effects of once-weekly semaglutide vs once-daily canagliflozin on body composition in type 2 diabetes: a substudy of the SUSTAIN 8 randomised controlled clinical trial. Diabetologia. 2020;63:473–85. 10.1007/s00125-019-05065-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Han E, Lee YH, Lee BW, Kang ES, Cha BS. Ipragliflozin additively ameliorates non-alcoholic fatty liver disease in patients with type 2 diabetes controlled with metformin and pioglitazone: a 24-Week randomized controlled trial. J Clin Med. 2020;9. 10.3390/jcm9010259. [DOI] [PMC free article] [PubMed]
- 104.Brown AJM, et al. A randomized controlled trial of dapagliflozin on left ventricular hypertrophy in people with type two diabetes: the DAPA-LVH trial. Eur Heart J. 2020;41:3421–32. 10.1093/eurheartj/ehaa419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Kinoshita T, et al. Comparison of the effects of three kinds of glucose-lowering drugs on non-alcoholic fatty liver disease in patients with type 2 diabetes: a randomized, open-label, three-arm, active control study. J Diabetes Investig. 2020;11:1612–22. 10.1111/jdi.13279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Gaborit B, et al. Effect of empagliflozin on ectopic fat stores and myocardial energetics in type 2 diabetes: the EMPACEF study. Cardiovasc Diabetol. 2021;20:57. 10.1186/s12933-021-01237-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Lopaschuk GD, Verma S. Mechanisms of Cardiovascular benefits of Sodium glucose co-transporter 2 (SGLT2) inhibitors: a state-of-the-art review. JACC Basic Transl Sci. 2020;5:632–44. 10.1016/j.jacbts.2020.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Preda A, et al. SGLT2 inhibitors: from glucose-lowering to cardiovascular benefits. Cardiovasc Res. 2024;120:443–60. 10.1093/cvr/cvae047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.D’Marco L et al. SGLT2i and GLP-1RA in Cardiometabolic and Renal Diseases: From Glycemic Control to Adipose Tissue Inflammation and Senescence. J Diabetes Res 2021, 9032378 (2021). 10.1155/2021/9032378 [DOI] [PMC free article] [PubMed]
- 110.Ryk A, et al. Cholesin receptor signalling is active in cardiovascular system-associated adipose tissue and correlates with SGLT2i treatment in patients with diabetes. Cardiovasc Diabetol. 2024;23:211. 10.1186/s12933-024-02322-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Masson W, Lavalle-Cobo A, Nogueira JP. Effect of SGLT2-Inhibitors on Epicardial Adipose tissue: a Meta-analysis. Cells. 2021;10. 10.3390/cells10082150. [DOI] [PMC free article] [PubMed]
- 112.Iacobellis G, Gra-Menendez S. Effects of Dapagliflozin on Epicardial Fat Thickness in patients with type 2 diabetes and obesity. Obes (Silver Spring). 2020;28:1068–74. 10.1002/oby.22798. [DOI] [PubMed] [Google Scholar]
- 113.Song XT, Wei YL, Rui YF, Fan L. Echocardiographic evaluation of the effect of dapagliflozin on epicardial adipose tissue and left ventricular systolic function in type 2 diabetes mellitus. J Diabetes Complications. 2023;37:108509. 10.1016/j.jdiacomp.2023.108509. [DOI] [PubMed] [Google Scholar]
- 114.Requena-Ibanez JA, et al. Not only how much, but also how to, when measuring epicardial adipose tissue. Magn Reson Imaging. 2022;86:149–51. 10.1016/j.mri.2021.11.004. [DOI] [PubMed] [Google Scholar]
- 115.Yagi S, et al. Canagliflozin reduces epicardial fat in patients with type 2 diabetes mellitus. Diabetol Metab Syndr. 2017;9:78. 10.1186/s13098-017-0275-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Bouchi R, et al. Luseogliflozin reduces epicardial fat accumulation in patients with type 2 diabetes: a pilot study. Cardiovasc Diabetol. 2017;16:32. 10.1186/s12933-017-0516-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Sato T, et al. The effect of dapagliflozin treatment on epicardial adipose tissue volume. Cardiovasc Diabetol. 2018;17:6. 10.1186/s12933-017-0658-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Sato T, et al. The Effect of Dapagliflozin Treatment on Epicardial Adipose tissue volume and P-Wave indices: an Ad-hoc analysis of the previous Randomized Clinical Trial. J Atheroscler Thromb. 2020;27:1348–58. 10.5551/jat.48009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Requena-Ibanez JA, et al. Mechanistic insights of Empagliflozin in nondiabetic patients with HFrEF: from the EMPA-TROPISM study. JACC Heart Fail. 2021;9:578–89. 10.1016/j.jchf.2021.04.014. [DOI] [PubMed] [Google Scholar]
- 120.Hiruma S, et al. A prospective randomized study comparing effects of empagliflozin to sitagliptin on cardiac fat accumulation, cardiac function, and cardiac metabolism in patients with early-stage type 2 diabetes: the ASSET study. Cardiovasc Diabetol. 2021;20:32. 10.1186/s12933-021-01228-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Zhao J, et al. Excessive accumulation of epicardial adipose tissue promotes microvascular obstruction formation after myocardial ischemia/reperfusion through modulating macrophages polarization. Cardiovasc Diabetol. 2024;23:236. 10.1186/s12933-024-02342-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Malavazos AE, et al. Human epicardial adipose tissue expresses glucose-dependent insulinotropic polypeptide, glucagon, and glucagon-like peptide-1 receptors as potential targets of pleiotropic therapies. Eur J Prev Cardiol. 2023;30:680–93. 10.1093/eurjpc/zwad050. [DOI] [PubMed] [Google Scholar]
- 123.Kasperova BJ, et al. Sodium-glucose cotransporter 2 inhibitors induce anti-inflammatory and anti-ferroptotic shift in epicardial adipose tissue of subjects with severe heart failure. Cardiovasc Diabetol. 2024;23:223. 10.1186/s12933-024-02298-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Maliszewska K, Kretowski A. Brown Adipose tissue and its role in insulin and glucose homeostasis. Int J Mol Sci. 2021;22. 10.3390/ijms22041530. [DOI] [PMC free article] [PubMed]
- 125.Brown KA, Scherer PE. Update on adipose tissue and Cancer. Endocr Rev. 2023;44:961–74. 10.1210/endrev/bnad015. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.