Abstract
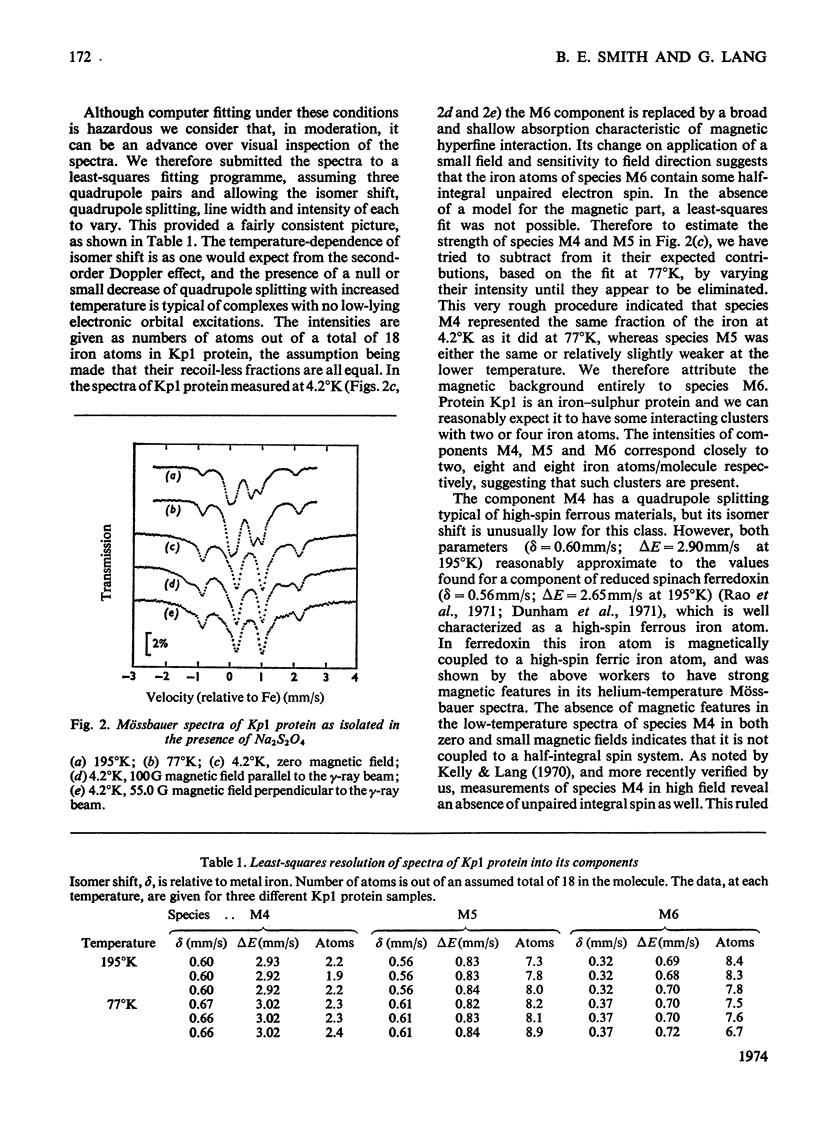
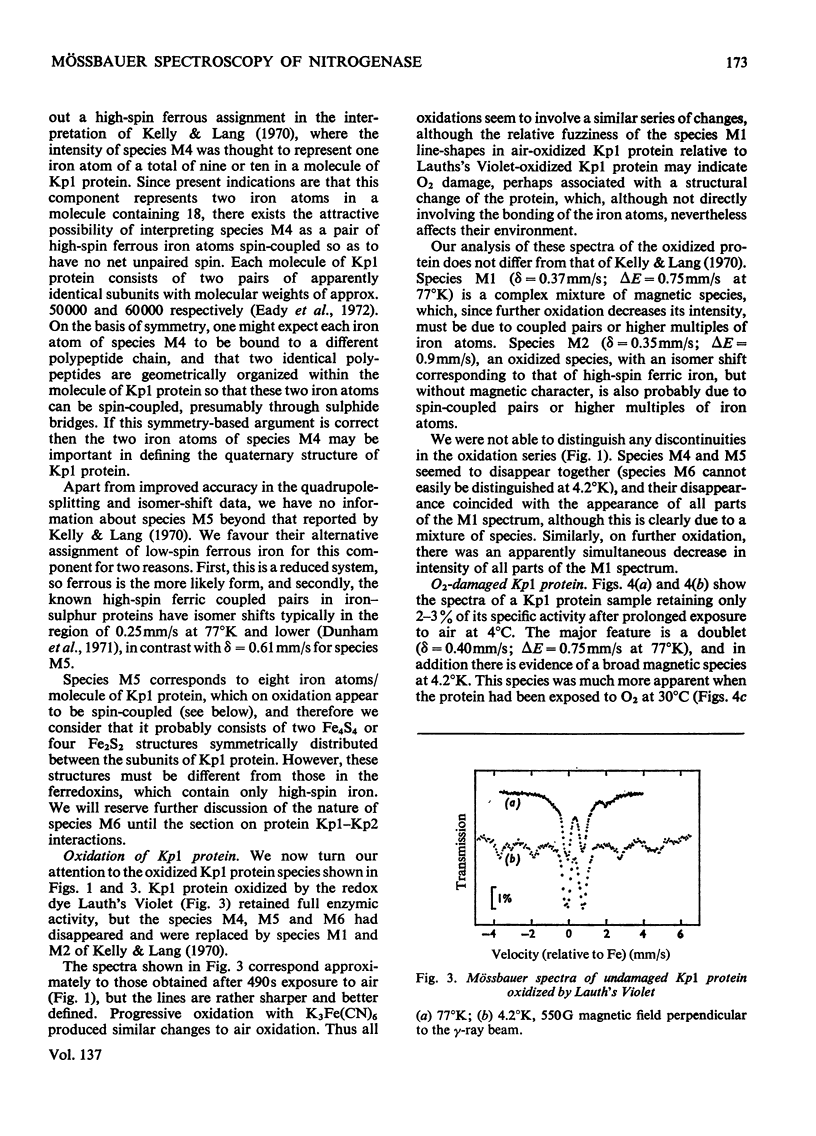
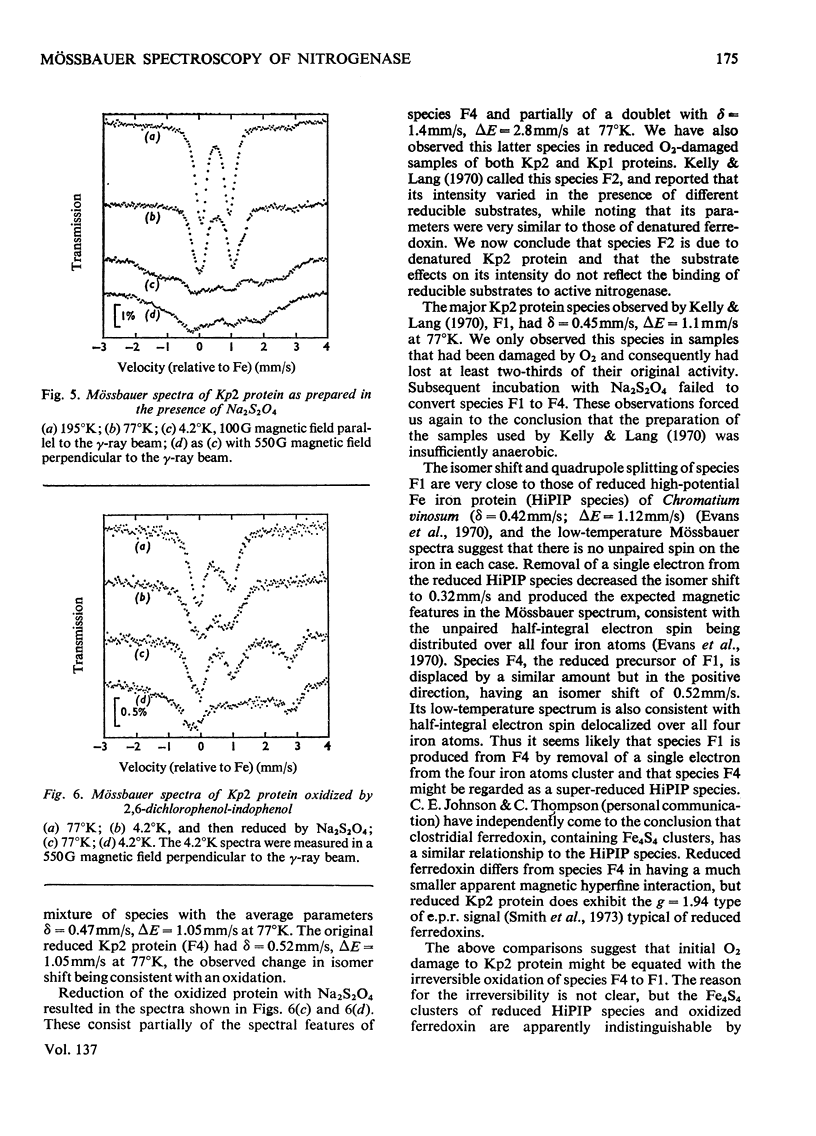
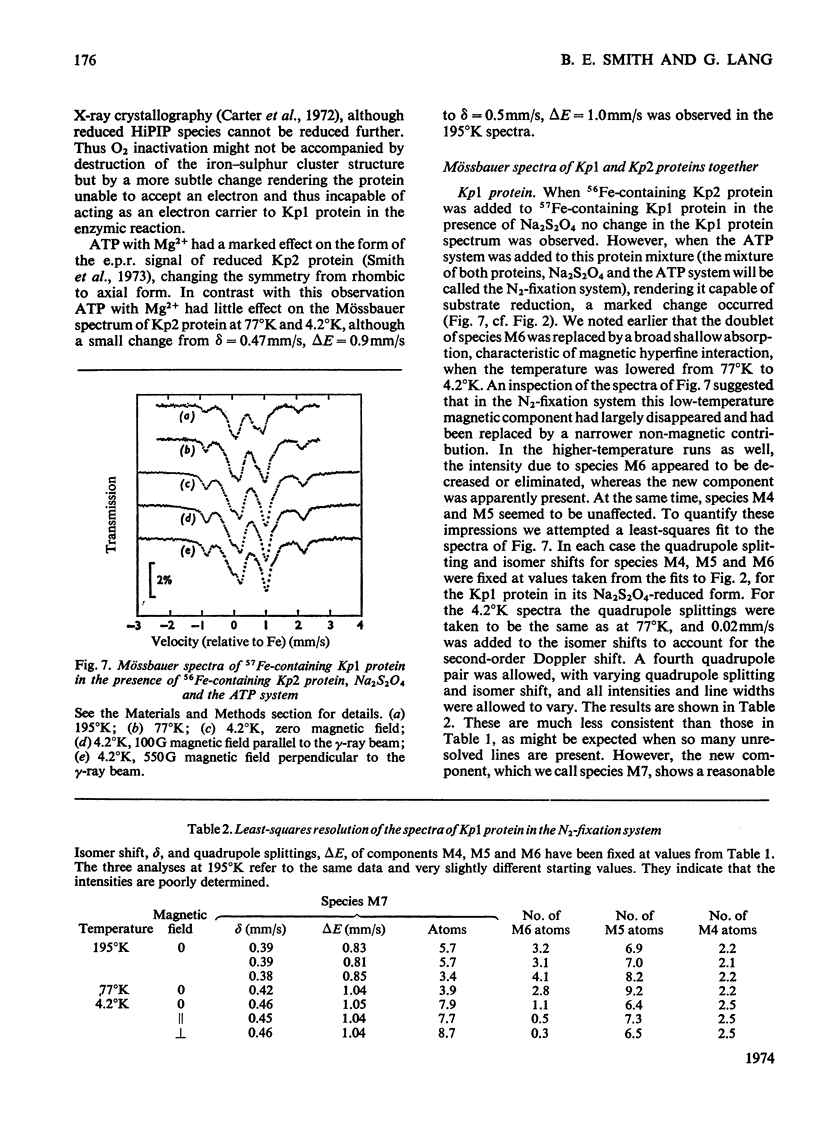
The Mo–Fe protein and the Fe protein which together constitute the nitrogenase of Klebsiella pneumoniae were prepared from bacteria grown in 57Fe-enriched medium. The Mössbauer spectrum of the Mo–Fe protein, as isolated in the presence of Na2S2O4, showed that the protein contained three iron species, called M4, M5 and M6. The area of the spectrum associated with species M4, with δ=0.65mm/s and ΔE=3.05mm/s at 4.2°K, corresponded to two iron atoms/molecule of protein and it is interpreted as being due to a high-spin ferrous, spin-coupled pair of iron atoms. The iron atoms of species M4 may be involved in the quaternary structure of the protein. Species M5, with δ=0.61mm/s and ΔE=0.83mm/s at 77°K, corresponded to eight iron atoms/molecule of protein and is interpreted as being due to Fe4S4 or Fe2S2 low-spin ferrous iron clusters. Species M6, with δ=0.37mm/s and ΔE=0.71mm/s at 77°K, also corresponded to eight iron atoms/molecule of protein and, at 4.2°K, became a broad shallow absorption, characteristic of magnetic hyperfine interaction. Oxidation of the Mo–Fe protein with the redox dye Lauth's Violet did not affect the activity of the protein but changed species M4, M5 and M6 into the species M1 (δ=0.37mm/s, ΔE=0.75mm/s at 77°K, broad magnetic component at 4.2°K) and M2 (δ=0.35mm/s, ΔE=0.9mm/s at 4.2°K). In the presence of the Fe protein, Na2S2O4, ATP and Mg2+, the M6 component of the Mo–Fe protein was replaced by species M7 with δ=0.46mm/s, ΔE=1.04mm/s at 4.2°K. The change in Mössbauer parameters associated with the M6 → M7 transformation was very similar to the change observed on reduction of the high-potential Fe protein from Chromatium vinosum. In contrast, Na2S2O4-reduced Fe protein contained only one type of iron cluster (F4). Species F4 had δ=0.50mm/s, ΔE=0.9mm/s at 195°K, and at 4.2°K broadened in a manner characteristic of a magnetic hyperfine interaction, associated with half-integral spin, equally distributed over all four atoms of the Fe protein. The Mössbauer spectra of the Mo–Fe and the Fe protein under argon were unaffected by the reducible substrates N2 and C2H2 and the inhibitor CO in the presence of ATP, Mg2+ and Na2S2O4. A number of Mössbauer spectral species associated with inactivated Mo–Fe and Fe proteins are described and discussed.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Carter C. W., Jr, Kraut J., Freer S. T., Alden R. A., Sieker L. C., Adman E., Jensen L. H. A comparison of Fe 4 S 4 clusters in high-potential iron protein and in ferredoxin. Proc Natl Acad Sci U S A. 1972 Dec;69(12):3526–3529. doi: 10.1073/pnas.69.12.3526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DONALD C., PASSEY B. I., SWABY R. J. A comparison of methods for removing trace metals from microbiological media. J Gen Microbiol. 1952 Nov;7(3-4):211–220. doi: 10.1099/00221287-7-3-4-211. [DOI] [PubMed] [Google Scholar]
- Dunham W. R., Bearden A. J., Salmeen I. T., Palmer G., Sands R. H., Orme-Johnson W. H., Beinert H. The two-iron ferredoxins in spinach, parsley, pig adrenal cortex, Azotobacter vinelandii, and Clostridium pasteurianum: studies by magnetic field Mössbauer spectroscopy. Biochim Biophys Acta. 1971 Nov 2;253(1):134–152. doi: 10.1016/0005-2728(71)90240-4. [DOI] [PubMed] [Google Scholar]
- Eady R. R., Smith B. E., Cook K. A., Postgate J. R. Nitrogenase of Klebsiella pneumoniae. Purification and properties of the component proteins. Biochem J. 1972 Jul;128(3):655–675. doi: 10.1042/bj1280655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans M. C., Hall D. O., Johnson C. E. Hyperfine structure of (57Fe) iron in the Mössbauer spectrum of the high-potential iron protein from Chromatium. Biochem J. 1970 Sep;119(2):289–291. doi: 10.1042/bj1190289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedrick J. L., Smith A. J. Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys. 1968 Jul;126(1):155–164. doi: 10.1016/0003-9861(68)90569-9. [DOI] [PubMed] [Google Scholar]
- Kelly M., Lang G. Evidence from Mossbauer spectroscopy for the role of iron in nitrogen fixation. Biochim Biophys Acta. 1970 Nov 3;223(1):86–104. doi: 10.1016/0005-2728(70)90134-9. [DOI] [PubMed] [Google Scholar]
- Mortenson L. E., Zumpft W. G., Palmer G. Electron paramagnetic resonance studies on nitrogenase. 3. Function of magnesium adenosine 5'-triphosphate and adenosine 5'-diphosphate in catalysis by nitrogenase. Biochim Biophys Acta. 1973 Feb 22;292(2):422–435. doi: 10.1016/0005-2728(73)90048-0. [DOI] [PubMed] [Google Scholar]
- Orme-Johnson W. H., Hamilton W. D., Jones T. L., Tso M. Y., Burris R. H., Shah V. K., Brill W. J. Electron paramagnetic resonance of nitrogenase and nitrogenase components from Clostridium pasteurianum W5 and Azotobacter vinelandii OP. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3142–3145. doi: 10.1073/pnas.69.11.3142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer G., Multani J. S., Cretney W. C., Zumft W. G., Mortenson L. E. Electron paramagnetic resonance studies on nitrogenase. I. The properties of molybdoferredoxin and azoferredoxin. Arch Biochem Biophys. 1972 Nov;153(1):325–332. doi: 10.1016/0003-9861(72)90452-3. [DOI] [PubMed] [Google Scholar]
- Phillips W. D., Poe M., McDonald C. C., Bartsch R. G. Proton magnetic resonance studies of Chromatium high-potential iron protein. Proc Natl Acad Sci U S A. 1970 Oct;67(2):682–687. doi: 10.1073/pnas.67.2.682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao K. K., Cammack R., Hall D. O., Johnson C. E. Mössbauer effect in Scenedesmus and spinach ferredoxins. The mechanism of electron transfer in plant-type iron-sulphur proteins. Biochem J. 1971 Apr;122(3):257–265. doi: 10.1042/bj1220257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith B. E., Lowe D. J., Bray R. C. Nitrogenase of Klebsiella pneumoniae: electron-paramagnetic-resonance studies on the catalytic mechanism. Biochem J. 1972 Nov;130(2):641–643. doi: 10.1042/bj1300641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith B. E., Lowe D. J., Bray R. C. Studies by electron paramagnetic resonance on the catalytic mechanism of nitrogenase of Klebsiella pneumoniae. Biochem J. 1973 Oct;135(2):331–341. doi: 10.1042/bj1350331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K., Osborn M. The reliability of molecular weight determinations by dodecyl sulfate-polyacrylamide gel electrophoresis. J Biol Chem. 1969 Aug 25;244(16):4406–4412. [PubMed] [Google Scholar]
- Zumft W. G., Cretney W. C., Huang T. C., Mortenson L. E., Palmer G. On the structure and function of nitrogenase from Clostridium pasteurianum W5. Biochem Biophys Res Commun. 1972 Sep 26;48(6):1525–1532. doi: 10.1016/0006-291x(72)90887-x. [DOI] [PubMed] [Google Scholar]


