Abstract
Previous studies have suggested that choroid plexus (ChP) enlargement occurs in individuals with schizophrenia-spectrum disorders (SSD) and is associated with peripheral inflammation. However, it is unclear whether such an enlargement delineates a biologically defined subgroup of SSD. Moreover, it remains elusive how ChP is linked to brain regions associated with peripheral inflammation in SSD. A cross-sectional cohort of 132 individuals with SSD and 107 age-matched healthy controls (HC) underwent cerebral magnetic resonance imaging (MRI) and clinical phenotyping to investigate the ChP and associated regions. A case-control comparison of ChP volumes was conducted, and structural variance was analyzed by employing the variability ratio (VR). K-means clustering analysis was used to identify subgroups with distinct patterns of the ventricular system, and the clusters were compared in terms of demographic, clinical, and immunological measures. The relationship between ChP volumes and brain regions, previously associated with peripheral inflammation, was investigated. We did not find a significant enlargement of the ChP in SSD compared to HC but detected an increased VR of ChP and lateral ventricle volumes. Based on these regions, we identified 3 clusters with differences in cognitive measures and possibly inflammatory markers. Larger ChP volume was associated with higher volumes of hippocampus, putamen, and thalamus in SSD but not in HC. This study suggests that ChP variability, but not mean volume, is increased in individuals with SSD, compared to HC. Larger ChP volumes in SSD were associated with higher volumes of regions previously associated with peripheral inflammation.
Subject terms: Schizophrenia, Biomarkers, Psychosis
Introduction
Schizophrenia-spectrum disorders (SSD) are severe mental illnesses with heterogeneous clinical presentation and pathophysiology, as indicated by increased brain structural variability1. This heterogeneity has largely contributed to the limited mechanistic understanding of SSD and has arguably led to a decrease in novel drug development efforts by pharmaceutical companies2. There is, therefore, an unmet need for the identification of intermediate phenotypes, which can help delineate subgroups within the schizophrenia spectrum with common pathophysiological mechanisms. Such intermediate phenotypes could aid in gaining a mechanistic understanding of defined SSD subgroups and pave the way to a more personalized treatment approach3.
Recent studies indicate that dysregulation of the immune system is a contributing factor in a subgroup of people with SSD4,5. It may affect brain structure and functioning, potentially resulting in increased risk for more pronounced symptom severity6. Furthermore, increased levels of peripheral inflammatory markers have been consistently described in SSD4. Some of those alterations have been associated with increased blood-cerebrospinal fluid (CSF) barrier (BCB) permeability7, which is a common finding in SSD8–11 and suggests concomitant neuroinflammation12. Nevertheless, there is substantial inconsistency regarding the number of proposed inflammatory subgroups in SSD and their impact on brain structure, with some studies indicating increases while others show reductions in regional (sub-)cortical volumes13,14.
In line with these findings, increased choroid plexus (ChP) volumes, altered ChP epithelia, and associated upregulation of immune genes in the ChP were described in individuals with SSD15–17. Located in each brain ventricle, the ChP produces CSF and forms the BCB, thus regulating central nervous system (CNS) homeostasis18. In recent years, its role as a unique neuroimmunological niche has been increasingly recognized19. Its ability to sense and engage signals from the periphery and CNS parenchyma and to host different immune cell populations stresses its role as a central hub of the brain-body interface19,20. ChP epithelial cells interact with peripheral leukocytes and can release cytokine effectors in the context of acute inflammation or attenuate the immune response in chronic inflammation, thus modulating peripheral and CNS immunity21.
Increased ChP volume has been consistently demonstrated in neurodegenerative and neuroinflammatory disorders22–24. A recent study indicated a volumetric increase of the ChP in people across the psychosis spectrum and linked it to worse cognition, brain atrophy, and peripheral inflammation15. Moreover, some preliminary findings suggest that the ChP volume increase was present in early but not in chronic psychosis25. However, a systematic evaluation of ChP volume variability in SSD and its relationship to peripheral inflammatory factors remains largely unexplored. This represents a critical gap in our understanding, especially considering the mounting evidence for inflammatory subgroups in SSD and the strong implication of ChP in neuroinflammatory processes. Furthermore, only one other study15 has explored the relationship between ChP volume and some, but not all, brain structures associated with peripheral inflammation in SSD13.
In the context of the described evidence gaps, we aimed to (1) replicate the findings of enlarged ChP in SSD, (2) explore the structural variance of ChP and associated ventricular regions in SSD, (3) identify distinct clusters based on the regions with increased variance and characterize them, and 4) investigate the relationship between ChP volume and volumes of subcortical and cortical regions, previously associated with peripheral inflammation in SSD.
Results
Cohort characteristics
The study cohort comprised 132 individuals with SSD (76 in- and 56 outpatients) and 107 HCs who underwent brain MRI (Table 1). There were no significant differences regarding sex ratio and age between the HC (66% male; 37.05 years) and the SSD group (77% male; 37.33 years) (p = 0.111; p = 0.721). Individuals with SSD had, on average, higher BMI values (SSD, mean ± SD = 28.09 ± 5.11; HC, mean ± SD = 23.64 ± 3.70, p < 0.001) (Table 1). A strong difference was also evident in smoking status: 50% of the individuals with SSD were active smokers, compared to 11% of the HCs (p < 0.001). The most common diagnosis was schizophrenia (72%), followed by schizoaffective disorder (22%), brief psychotic disorder (5%) and delusional disorder (1%). The mean duration of illness (DUI) was 143.68 months (SD = 115.42), and the mean duration of untreated psychosis (DUP) was 22 months (SD = 34.80), although we could only collect data on the DUP in 78 of the participants. The Positive And Negative Syndrome Scale (PANSS) total scores averaged 54.80 (SD = 15.20), indicating that patients were, on average, “mildly ill”26. Forty-seven individuals (37%) were either being treated with or reported a history of intake of clozapine in their lifetime, while 80 individuals from the SSD cohort have never received clozapine. Out of the 132 participants with SSD, nine were antipsychotic-naïve.
Table 1.
Cohort characteristics.
| SSDs | HC | ||||
|---|---|---|---|---|---|
| Mean ± SD | N | Mean ± SD | N | p | |
| Demographic characteristics | |||||
| Age, years | 37.33 ± 11.18 | 132 | 37.05 ± 11.85 | 107 | 0.721a |
| BMI | 28.09 ± 5.11 128 | 23.64 ± 3.70 103 | <0.001a | ||
| n (%) | n (%) | p | |||
| Sex, male:female | 101:31 (77%) | 71:36 (66%) | 0.111b | ||
| Current smoking, yes:no | 64:63 (50%) | 127 | 11:93 (11%) | 104 | <0.001b |
| Disease characteristics | |||||
| Duration of illness, months | 143.68 ± 115.42 | 128 | − | − | − |
| Duration of untreated psychosis, months | 22.00 ± 34.80 | 78 | − | − | − |
| PANSS positive symptoms | 12.82 ± 4.59 | 128 | 7.34 ± 0.83 | 107 | <0.001a |
| PANSS negative symptoms | 13.45 ± 5.40 | 128 | 7.41 ± 0.91 | 107 | <0.001a |
| PANSS general symptoms | 28.52 ± 7.79 | 128 | 16.92 ± 1.45 | 107 | <0.001a |
| PANSS total score | 54.80 ± 15.20 | 128 | 31.64 ± 2.53 | 107 | <0.001a |
| CPZeq, mg | 335.87 ± 254.14 | 127 | − | − | − |
| Diagnosis (DSM-5) | N (%) | N (%) | − | ||
| Schizophrenia | 95 (72%) | − | − | ||
| Schizoaffective disorder | 30 (22%) | − | − | ||
| Brief psychotic disorder | 6 (5%) | − | − | ||
| Delusional disorder | 1 (1%) | − | − | ||
BMI body mass index, CPZeq chlorpromazine equivalent doses, HC healthy control, N number of participants, p p value, PANSS Positive and Negative Syndrome Scale, SD standard deviation, SSD schizophrenia spectrum disorder, MRI magnetic resonance imaging.
aWilcoxon rank sum test.
bChi-square test.
Choroid plexus volume and asymmetry in SSD
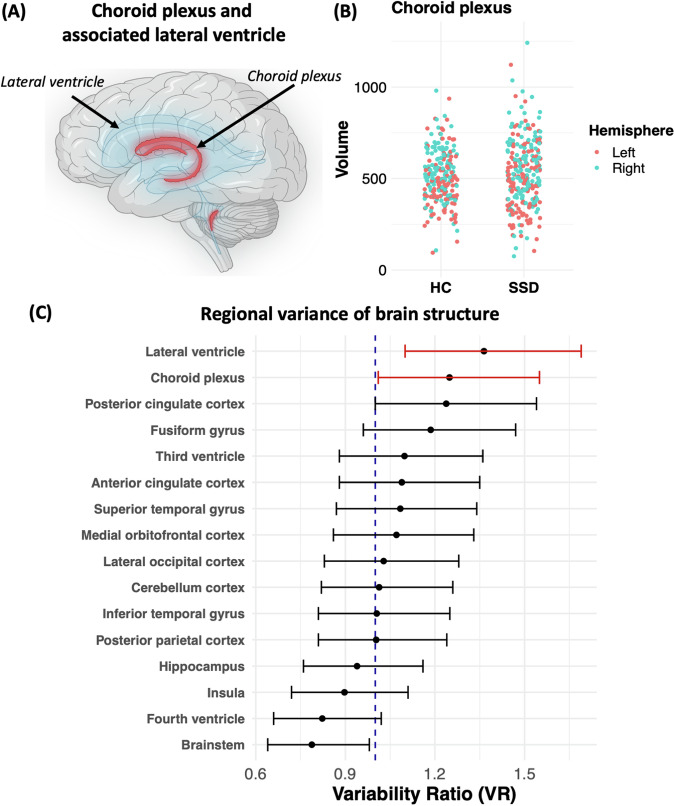
First, we aimed to replicate the recently published findings on ChP (Fig. 1A) enlargement in SSD15,25. For that reason, we compared the ChP volumes between SSD subjects and HCs (Fig. 1B; Table S1a, b). Our analysis showed no significant differences in choroid plexus volumes after controlling for age and sex as covariables (estimate [95% CI] = 19.14 mm3 [−15.37, 53.64]; p = 0.362). Furthermore, the right choroid plexus was significantly larger than the left one, independent of phenotype (estimate [95% CI] = 66.41 mm3 [43.72, 89.1]; p < 0.001). Building on the asymmetry hypothesis in schizophrenia27, we compared the choroid plexus asymmetry index (Suppl. Methods) between individuals with SSD and HCs. We did not find significant differences between both groups (Table S1c, Fig. S2).
Fig. 1. Comparison of choroid plexus volumes and structural variance between individuals with schizophrenia-spectrum disorders and healthy controls.
A Schematic illustration of the choroid plexus (red) and associated lateral ventricle (blue) of the brain. B Comparison of mean volumes of the left (red) and right (turquoise) choroid plexus between the healthy control (HC) group and the schizophrenia-spectrum disorders (SSD) group, illustrated with a jitter plot. Data points represent individual choroid plexus volumes. Groups were compared using a linear mixed-effect model, controlling for age and sex. NHC = 107, nHC = 214, NSSD = 132, nSSD = 264. C Forest plot depicting the variability ratio (VR) of choroid plexus, lateral ventricle (red) and other regions of interest, implicated in severe mental illness and schizophrenia (black). NHC = 107, NSSD = 132. N number of participants, n number of choroid plexus volumes, VR variability ratio. Created with BioRender.com.
Structural variance of choroid plexus
Driven by the observation of increased scattering of ChP volumes in the SSD group (Fig. 1B), we investigated whether volumes of the choroid plexus and other associated regions (Fig. 1C) show an increased variability (as indicated by variability ratio [VR]) in the SSD group, compared to the HC group. We calculated VR for all regions of interest outlined above and implicated in convergent transdiagnostic or schizophrenia-specific brain networks28,29 using an established method1. The variability of the ChP (estimate [95% CI] = 1.249 [1.01, 1.55]) and the associated lateral ventricle (estimate [95% CI] = 1.364 [1.1, 1.69]) was more pronounced in the SSD group compared to the variability in the HC group. Except for the posterior cingulate cortex (estimate [95% CI] = 1.238 [1.0, 1.54]), where we saw a non-significant trend towards increased variance, the variability of the volumes and the cortical thickness of all other regions was not higher in the SSD group compared to the HC group (Table S2a). The VR analyses of the unilaterally computed regions of interest mostly confirmed the results of bilaterally computed regional VRs (Table S2b).
Clusters based on regions with increased variability
Next, we conducted a K-means clustering analysis based on the choroid plexus and lateral ventricle (Lat Vent) volumes to identify subgroups within the SSD cohort with distinct patterns of the ventricular system. These regions were selected based on the empirical observation of increased variability in our data (Fig. 1C) as well as previous implications of the cerebroventricular system in immune dysregulation in SSD15,16,30,31. Employing the elbow method, we identified the ideal cluster solution to be 3 (Fig. S3). According to the scaled mean volumes of the two features (ChP and lateral ventricle) in each cluster, we deduced that cluster 1 (N = 15, scaled centers ChP/LatVent = 1.18, 1.90) was “large ChP-large lateral ventricle cluster”, cluster 2 (N = 50, scaled centers ChP/LatVent = −0.85, −0.65) was “small ChP-small lateral ventricle cluster,” and cluster 3 (N = 67, scaled centers ChP/LatVent = 0.37, 0.06) was “intermediate ChP-small lateral ventricle cluster” (Table S3a, Fig. 2A).
Fig. 2. Cluster characteristics.
A Scatter plot depicting the lateral ventricle (x-axis) and choroid plexus (y-axis) residual volumes (after correction for age and sex) of cluster 1 (red), cluster 2 (green) and cluster 3 (blue) within the SSD cohort. B Radar chart illustrating cluster mean scaled scores of age, “duration of untreated psychosis” (DUP), “duration of illness” (DUI), body-mass-index (BMI), and chlorpromazine equivalents (CPZeq). C Radar chart illustrating cluster mean z-scores of different cognitive subdomains, measured by “The Brief Assessment of Cognition in Schizophrenia” (BACS). D Comparison of mean peripheral monocyte counts between cluster 1 (N = 10), cluster 2 (N = 28), and cluster 3 (N = 38). Data points represent individual monocyte counts. *q < 0.05; ***q < 0.001. N number of participants, DUP “duration of untreated psychosis”, DUI “duration of illness”, BMI “body-mass-index”, CPZeq “chlorpromazine equivalents”, BACS “The Brief Assessment of Cognition in Schizophrenia”, Comp composite score, VM verbal memory test, VF verbal fluency test, TL tower of London test, TM token motor test, SC symbol coding test, DS digit sequence test, G/l Giga/liter.
Characteristics of clusters within the SSD group
There were no significant across-cluster differences regarding sex (χ2 = 1.47, p = 0.479), diagnosis (χ2 = 12.36, p = 0.136), treatment resistance status (χ2 = 0.91, p = 0.635), and smoking status (χ2 = 2.88, p = 0.237; Table S3a). We found no significant across-cluster differences regarding age, DUI, DUP, BMI, and CPZ equivalent (Table S3b, Fig. 2B).
There were no significant differences regarding psychopathology (operationalized by PANSS) or global functioning (measured with GAF scale) across clusters (Fig. S4A, Table S3c). Furthermore, there were significant differences regarding global cognition (operationalized by BACS composite z-score) (cluster 1, mean ± SD (z-score) = −1.41 ± 1.42; cluster 2, mean ± SD (z-score) = −0.52 ± 0.74; cluster 3, mean ± SD (z-score) = −0.52 ± 1, q = 0.04) and BACS tower of London subtest (cluster 1, mean ± SD (z-score) = −2.7 ± 3.45; cluster 2, mean ± SD = −0.47 ± 1.29, cluster 3, mean ± SD = −0.41 ± 1.14, q < 0.001; Fig. 2C, Table S3d). The Games–Howell post-hoc tests did not find significant between-cluster differences for the BACS composite and tower of London test z scores. Moreover, we observed a trend towards significant across-group differences for BACS digit sequence (q = 0.05), BACS symbol coding (q = 0.084), and BACS token motor (q = 0.084) subtests that didn’t survive FDR correction (Table S3d).
Due to the role of the choroid plexus as an immunological niche in the CNS we compared the across-cluster differences regarding peripheral leukocytes and high-sensitive C-reactive protein (hsCRP) levels in two subgroups of participants. We found significant across-cluster differences regarding absolute monocyte counts (cluster 1, mean ± SD (G/l) = 0.6 ± 0.14; cluster 2, mean ± SD = 0.55 ± 0.14, cluster 3, mean ± SD = 0.48 ± 0.13, q = 0.049; Fig. 2D) as well as hsCRP levels (cluster 1, mean ± SD (pg/ml) = 3.5 × 108 ± 3.73 × 108; cluster 2, mean ± SD = 2.56 × 108 ± 2.43 × 108, cluster 3, mean ± SD = 1.44 × 108 ± 1.49 × 108, q = 0.032; Fig. S4D). The Games–Howell post-hoc tests revealed a trend towards higher monocyte counts in cluster 1, compared to cluster 3 (p = 0.07) and higher hsCRP levels in cluster 2, compared to cluster 3 (p = 0.083), which did not reach statistical significance (Table S3e). There were no significant across-cluster differences regarding absolute neutrophil or lymphocyte counts (Figs. S4B, C).
Furthermore, we did not find significant across-cluster differences regarding the polygenic risk score for schizophrenia (SZ-PRS) after controlling for age, sex, and the first five or the first ten multidimensional scaling (MDS) components as covariates (Table S3f, Table S3g, Fig. S4E). Those results were stable across different phi thresholds.
Relationship between choroid plexus and brain structure
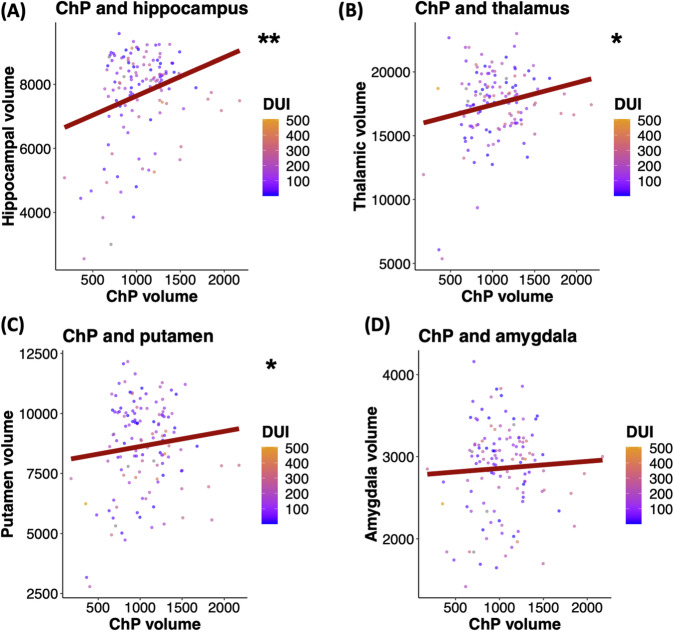
Next, we explored the relationship between choroid plexus volume and the volumes of cortical and subcortical structures previously associated with peripheral inflammation in SSD13 (Tables S4a, S4b). Among the subcortical regions of interest, we discovered a positive association between choroid plexus volume and volumes of hippocampus (estimate [95% CI] = 1.41 [0.78, 2.03]; q = 0.001), thalamus (estimate [95% CI] = 1.93 [0.67, 3.2]; q = 0.024), and putamen (estimate [95% CI] = 1.18 [0.37, 2.0]; q = 0.024), but not amygdala within the SSD cohort (Table S4a, Fig. 3). Furthermore, those associations appeared to be specific for SSD since we did not find significant associations between those structures in the HC group (Table S5, Fig. S7). Interestingly, we did not find significant associations between the choroid plexus volume and the volumes of cortical structures previously associated with peripheral inflammation (Table S4b, Fig. S5). Moreover, there was no association between the volumes of hippocampus, thalamus, putamen, and the volume of the other structure with increased variance, namely the lateral ventricle (Table S4c, Fig. S6).
Fig. 3. Association between volumes of choroid plexus and subcortical regions, previously linked to peripheral inflammation in SSD.
Regression plots illustrating relationships between choroid plexus volume, (A) hippocampus, (B), thalamus, (C) putamen, and (D) amygdala volumes in SSD. The analyses were conducted via linear regressions, controlling for age and sex. Data points represent individual volumes and are color-coded for “duration of illness” with orange color indicating higher and blue color indicating lower duration. NSSD = 132. *q < 0.05; **q < 0.01. N number of participants, DUI “duration of illness (months)”, ChP choroid plexus.
Discussion
In this cross-sectional study, we did not replicate previous findings15,25,31 for increased mean volume of the choroid plexus in SSD compared to HCs but observed a scattering of the volumes within the SSD group, which prompted us to investigate this variability further. Using a well-established technique for quantification of structural variability1, we demonstrated an increased variance of the ChP and the lateral ventricle in SSD as opposed to other regions implicated in SSD and other neuropsychiatric disorders, suggesting a measurable degree of heterogeneity. Employing a K-means clustering analysis, based on the volumes of ChP and lateral ventricle, we identified three clusters within the SSD cohort with differences in cognitive abilities, and possibly peripheral inflammatory measures. Furthermore, the ChP volume positively correlated with volumes of subcortical, but not cortical regions, previously associated with peripheral inflammation in SSD13 and this relationship appeared to be specific for the SSD group.
Some of the possible reasons for the discrepancy between our and previous findings include between-study differences in cohorts (absence of participants with affective psychosis in our study), age differences (our SSD cohort was on average older than other cohorts)25,31, and symptom severity (our cohort had on average less severe psychopathology than the cohorts of others)15,31. The high interindividual variability of the ChP volumes within the SSD group, together with a lower sample size than Lizano et al.15, might have also contributed to the lack of statistical significance in the between-group analysis. Another important factor that might have contributed to the discrepancy between our study and the one of Senay et al. is the difference in segmentation strategy25.
While there is some evidence suggesting an increased mean ChP volume, to our knowledge no study so far has investigated the variability of the ChP in any neuropsychiatric disorder. A meta-analysis including measurements from 3901 patients with first-episode schizophrenia and 4040 controls demonstrated increased VR of third ventricle, putamen and thalamus and temporal lobe, decreased VR of the anterior cingulate cortex volume, compared to HCs1. In our study, we extended this knowledge and found for our SSD sample a significant increase in VR of choroid plexus and lateral ventricle but not in other regions implicated in SSD or other neuropsychiatric disorders. The fact that the ChP and the lateral ventricle showed higher variance as opposed to the other regions suggests that alterations of the ventricular system might be seen in a SSD subgroup and could serve as an intermediate phenotype for stratification purposes. Taken together, the higher variance of the ChP suggests that it might be enlarged only in a subgroup of people with SSD.
To explore this variance, we conducted a K-means clustering analysis and identified three clusters with distinct patterns of ventricular system within the SSD group. Cluster 1 was characterized by both enlarged choroid plexus and lateral ventricle, while cluster 2 had low choroid plexus and lateral ventricle volumes. The participants from cluster 3 showed relatively low lateral ventricle and relatively high choroid plexus volumes. We aimed to investigate how the clusters differ regarding demographic and clinical features as well as peripheral inflammatory markers and genetic liability for schizophrenia. We did not observe significant across-cluster differences in demographics, psychopathology, or functioning. Interestingly, there were significant across-cluster differences regarding cognitive performance, measured by the BACS composite score and the BACS Tower of London score. Our data adds to the emerging evidence of an association between choroid plexus volume and cognitive functioning in psychosis15. Moreover, substantial evidence links larger cerebral ventricles to worse cognitive outcomes32,33. From a pathophysiological perspective, it remains unclear whether the ChP enlargement observed in some individuals with SSD is a consequence of ventricular enlargement or if it occurs independently. Given that the severity of ventricular enlargement in SSD has been consistently associated with cognitive impairment32, the observed association between ChP volume and cognitive deficits could, at least in part, be influenced by changes in ventricular volume. In our study, the clusters were identified based on both choroid plexus and lateral ventricle volumes, making it difficult to disentangle the effects of each on cognitive outcomes. Nevertheless, given the close anatomical and physiological relationship between the cerebral ventricles and the choroid plexus, both components of the cerebroventricular system34, it is possible that each could have mechanistic relevance in the observed associations.
Even though we found significant across-cluster differences regarding absolute monocyte counts as well as hsCRP levels in the main analyses, we could not detect significant pairwise differences between the clusters in the post-hoc analyses. These results suggest that while differences in levels of peripheral inflammation across the three cerebroventricular clusters might be evident, our study could have been underpowered to robustly detect specific between-cluster differences. Although our findings are concomitant with a previous study suggesting a link between peripheral inflammation and choroid plexus volume in psychosis15, they have to be interpreted carefully due to the limited sample size of our clusters. Follow-up, well-powered studies are needed to address this limitation.
Furthermore, we did not find across-cluster differences in genetic liability for schizophrenia as captured by SZ-PRS. Since SZ-PRS captures only a small proportion of the total genetic risk for schizophrenia, it remains unclear whether the increased variability of cerebroventricular measures in SSD is driven primarily by environmental factors, other genetic influences not captured by SZ-PRS, or their interaction. Additionally, given the limited power of our study, we cannot exclude the possibility that SZ-PRS itself may still partially contribute to this variability. This highlights the need for future investigations with larger sample sizes and advanced methodologies to disentangle genetic and environmental contributions to cerebroventricular variability. Tools such as a schizophrenia exposome score35, which aggregates environmental risk factors, could provide a more comprehensive assessment of environmental influences. Similarly, incorporating polygenic scores for traits related to immune function or neurodevelopmental processes might offer additional insights into genetic contributions beyond SZ-PRS. In future research, approaches like these, alongside gene-environment interaction analyses36 or multivariate modeling frameworks37, could improve statistical power, accuracy, and generalizability.
In the next step we focused on the relationship between ChP volume and (sub-)cortical regions previously associated with peripheral inflammation in SSD13. While we did not find any association between ChP volumes and volumes of any cortical region of interest, we observed a positive association between ChP measures and volumes of hippocampus, putamen, and thalamus, but not amygdala. Furthermore, those associations appeared to be specific for SSD since we did not observe them in our HC cohort. Interestingly, we did not detect any association between the lateral ventricle volumes and any of the regions previously linked to peripheral inflammation, further strengthening the notion that the associations are specific to ChP and not just a global effect. It is hard to deduce from mere associations of volumetric measures to neurobiological mechanisms behind those volumetric changes. It has been speculated that volumetric increases of regions associated with peripheral inflammation could be a result of compromised BBB and/or BCB, resulting in vasodilation, leukodiapedesis, and plasma exudation in the brain13,38. Even though we do not know why we only see an association between ChP and the subcortical, but not the cortical regions, previously associated with peripheral inflammation, one can speculate that the physical distance to the ChP might be of relevance. Since the studied subcortical structures are in direct vicinity to the ChP and the ChP forms the BCB and acts as an invasion route for peripheral leukocytes19, it could be possible that it acts as a mediator between peripheral inflammation and brain structure in SSD. Nevertheless, this hypothesis renders further verification in subsequent studies.
Some of the limitations of our study include its cross-sectional design and the fact that it is based on a monocentric cohort. This is, to our knowledge, the second-largest SSD cohort where ChP alterations have been studied. However, the increased variability ratio of this region requires larger sample sizes to provide conclusive data on its morphological changes in SSD. Even though we showed a moderate correlation between FreeSurfer segmentation and manual segmentation of ChP, some authors have questioned the reliability of FreeSurfer segmentation of ChP and have suggested other methods39,40. Furthermore, since only nine of the individuals with SSD in our cohort were antipsychotic-naïve, we cannot exclude medication effects on choroid plexus morphology17. The selection of regions of interest for the clustering analysis was partly driven by the empirical observation of increased variability in cerebroventricular regions within our cohort. While this observation holds true for our sample, this approach may limit the generalizability of our findings to other populations and warrants independent validation. The individual clusters (especially cluster 1) might have been underpowered to detect differences regarding clinical and immunological features. Even though an association between peripheral inflammation and the (sub-)cortical regions of interest (Table S4, S5) has been assumed, Lizano’s study remains, to the best of our knowledge, the only one showing such a link and renders replication. Furthermore, the complete blood count is not only a nonspecific measurement of peripheral inflammation, but it was also only assessed in acutely ill inpatients, which limits the generalizability of our findings. Also, some of the variability of the complete blood count could be explained by the fact that even though blood was collected consistently during the morning hours, it was not always done under fasting conditions.
Conclusions
In conclusion, we did not replicate the findings of previously reported choroid plexus enlargement in SSD but discovered increased structural variance of this region and the lateral ventricle. A clustering analysis based on the choroid plexus and lateral ventricle volumes identified three distinct clusters that differed in cognitive functioning and possibly peripheral inflammatory measures. Furthermore, the choroid plexus volume was positively associated with volumes of hippocampus, thalamus, and putamen, which were previously linked to peripheral inflammation in SSD. Overall, our study highlights the importance of replication efforts in external cohorts. Future investigations with large sample sizes, thorough immunological characterization, and longitudinal designs could help elucidate the role of the choroid plexus in the pathophysiology of SSD and its utility as an intermediate phenotype for patient stratification. Additionally, establishing dedicated cohorts with standardized imaging protocols and biomarker assessments could provide valuable, reusable datasets, facilitating replication and cross-validation by other researchers and fostering collaboration within the field.
Methods
Participants
This project was carried out in the context of the Clinical Deep Phenotyping (CDP) study41, an ongoing add-on study to the Munich Mental Health Biobank (project number 18-716)42 (German Clinical Trials Register, DRKS, ID: DRKS00024177) and approved by the ethics committee of the Faculty of Medicine, LMU University Hospital Munich (project numbers 20-0528 and 22-0035).
Study participants were recruited at the Department of Psychiatry and Psychotherapy, LMU University Hospital, LMU Munich, Germany, between July 14, 2021, and May 22, 2023. In- (N = 76) and outpatients (N = 56) were included. All study participants were between 18 and 65 years old and provided written informed consent. Included patients had a primary diagnosis of schizophrenia, schizoaffective disorder, delusional disorder, or brief psychotic disorder, collectively referred to as SSD throughout the manuscript. Participants with no current or past psychiatric disorder, according to the Mini International Neuropsychiatric Interview (M.I.N.I.), were defined as healthy controls (HC).
Exclusion criteria were as follows: concurrent clinically relevant central nervous system (CNS) disorders, such as multiple sclerosis and epilepsy, history of encephalitis, meningitis, stroke, traumatic brain injury or cerebral surgery; current pregnancy or lactation; rheumatic disorders, inflammatory bowel disease, active malignancy, acute or chronic infection.
The clinical assessment was carried out as previously described by our working group43. Further details are provided in the Supplementary Methods.
Magnetic resonance imaging
All participants underwent brain magnetic resonance imaging (MRI). Brain MRI recordings were performed on a 3T Siemens Magnetom Prisma scanner (Siemens Healthineers AG, Erlangen, Germany) with a 32-channel head coil. The measures of regional brain volumes (incl. choroid plexus) as well as cortical volumes were computed in mm3 using FreeSurfer (v7.3.2; https://surfer.nmr.mgh.harvard.edu/)44 and were corrected for intracranial volume45. Residuals vs fitted values plots of subcortical (Fig. S8) and cortical (Fig. S9) volumes show predominantly linear relationships between the intracranial volume and the volumes of our regions of interest, confirming the suitability of the chosen method for ICV correction in our data.
The FreeSurfer atlas was used to obtain the regional volumes for left and right brain hemispheres in the regions of interest, such as the choroid plexus (Fig. S1) (details are provided in the Supplementary Methods). Manual ChP segmentation in a subsample by two independent raters resulted in a high intraclass correlation coefficient for their ratings (0.72) and a moderate Spearman correlation (r = 0.659) with FreeSurfer volumes in line with a previous report46 (Fig. S1). The sum of the computed measures for each region of interest’s left and right brain hemispheres was used for statistical analyses. The choroid plexus volume measures for comparison between SSD and HC and the variability ratios of regions of interest were additionally computed unilaterally (Table S1, S2b). Further details are provided in the Supplementary Methods.
Statistical analyses
The R language (v4.2.1, R Core Team, 2021)47 in RStudio environment (RStudio Team, 2020) was used for all statistical analyses and visualizations. The following statistical tests were used to compare differences in demographic and clinical variables between SSD and HC: Fisher’s exact test for categorical variables, Welch’s t test for normally distributed, and Wilcoxon rank-sum test for non-normally distributed continuous variables. Shapiro–Wilk test was used to assess normality within groups48.
Linear mixed-effects models (LMM) based on restricted maximum likelihood estimates were calculated for the association between SSD and choroid plexus volume using the “lme4” package49. We included both hemispheres and adjusted for the correlation of the measurements of each participant’s hemisphere by including a random intercept for participant identification number. The choroid plexus volume was defined as the outcome variable; we incorporated a group variable (SSD vs. HC), hemisphere (left vs. right) as predictor variables, and an interaction term between the group and hemisphere as fixed factors in the model. We compared the volumes between groups and between hemispheres and investigated whether a possible group effect is mainly driven by a specific hemisphere. Sex and age were used as covariables due to their impact on choroid plexus volume15.
To quantify the variability in the volumes of the regions of interest in SSD patients and HC, we employed the established technique of Variability Ratio (VR)1. A mean VR > 1, whose 95% confidence interval did not include 1.00, was referred to as increased, as previously suggested1,50,51. Details are provided in the Supplementary Methods.
A K-means clustering analysis was performed on the SSD cohort to identify subgroups of participants with similar patterns of ventricular and choroid plexus morphology. These regions were selected based on the empirical observation of increased variability in our data (Fig. 1C) as well as previous implication of the cerebroventricular system in immune dysregulation in SSD15,16,30,31. Prior to clustering, a linear regression was used to correct for potential confounding effects of age and sex on the choroid plexus and lateral ventricle volumes. Specifically, linear models were fitted with choroid and lateral ventricle volumes as dependent variables and age and sex as independent variables. The residuals from these models, which represent the age- and sex-adjusted volumes, were extracted for subsequent analysis. The residuals were then scaled to standardize the data to ensure that each variable contributed equally to the distance calculations. The optimal cluster number (k) solution was determined by the elbow method52. Group effects across clusters were tested using analysis of covariance (ANCOVA). Due to unequal sample sizes between clusters, we employed the Games-Howell nonparametric post-hoc test. Further information regarding covariates and the clustering analysis is provided in Supplementary Methods.
To explore the effects of ChP volume on cortical and subcortical regions previously linked to peripheral inflammation13, we computed multiple linear regressions, controlling for age and sex as covariates.
The threshold for statistical significance was set at p value < 0.05. Results from the descriptive statistics and the between-cluster comparisons are shown as mean ± standard deviation (SD). We employed the false discovery rate (FDR) procedure, based on the Benjamini-Hochberg method, for correction of the p values, which were reported as q values.
Supplementary information
Acknowledgements
The study was endorsed by the Federal Ministry of Education and Research (Bundesministerium für Bildung und Forschung [BMBF]) within the initial phase of the German Center for Mental Health (DZPG) (grant: 01EE2303C to A.H. and 01EE2303A, 01EE2303F to P.F., A.S.). This research was supported by BMBF with the EraNet project GDNF UpReg (01EW2206) to V.Y. and P.F. The procurement of the Prisma 3T MRI scanner was supported by the Deutsche Forschungsgemeinschaft (DFG, INST 86/1739-1 FUGG). No funding was received by commercial or not-for-profit sectors. E.B. and M.S.K. were supported by doctoral scholarships from the Faculty of Medicine, LMU Munich, Munich, Germany. L.K., I.M., and F.J.R. were supported by the Else Kröner-Fresenius Foundation for the Residency/PhD track of the International Max Planck Research School for Translational Psychiatry (IMPRS-TP), Munich, Germany. V.Y., F.J.R., M.C., and J.M. were supported by the Förderprogramm für Forschung und Lehre (FöFoLe) of the Faculty of Medicine, LMU Munich, Munich, Germany. ECS was supported by the Munich Clinician Scientist Program (MCSP).
Author contributions
D.K., E.W., and F.J.R. designed and conceptualized the Clinical Deep Phenotyping Study. V.Y., D.K., and E.W. designed this study and wrote the protocol. V.Y., J.M., L.K., E.B., E.W., and M.S.K. recruited patients and collected study data. E.W., V.Y., and J.M. trained staff on diagnostic and clinical assessments. MRI measurements were performed by J.M. and M.S.K. under the supervision of D.K. and L.R. Genotyping and genetic analyses were performed by S.P., F.J.R., E.C.S., and V.Y. Serological analyses were performed by N.G. and E.M. Statistical analyses were performed by V.Y. Data visualization was performed by V.Y. V.Y. wrote the first draft of the manuscript. J.M., L.R., M.S.K., E.B., M.M., S.P., L.K., M.C., E.C.S., N.G., E.M., S.H., N.W., D.S., I.M., A.H., P.F., A.S., F.J.R., D.K., and E.W. provided critical review. V.Y. prepared the final manuscript version with the help of all authors.
Data availability
The documentation sheets including the de-identified data will be made available upon publication on OSF (https://osf.io/hgsq2/). All software packages used in this study are publicly available. For details, see the (supplementary) methods section.
Competing interests
The authors declare that they have no biomedical financial interests or potential conflicts of interest regarding the content of this report. A.H. received paid speakership by Janssen, Otsuka, Lundbeck, and Recordati and was member of advisory boards of these companies and Rovi. A.S. has been an honorary speaker for TAD Pharma and Roche and a member of advisory boards for Roche. P.F. received paid speakership by Boehringer-Ingelheim, Janssen, Otsuka, Lundbeck, Recordati, and Richter and was member of advisory boards of these companies and Rovi. S.P. received previous speaker fees and/or travel expenses from Novartis Pharma GmbH, Oertli AG, Bayer AG, Alcon Pharma GmbH, and Pharm-Allergan GmbH. EW was invited to advisory boards from Recordati, Teva and Boehringer Ingelheim. S.H. is supported by an Australian Research Training Program scholarship. D.S. is supported by an NHMRC Investigator Fellowship GNT 1194635. N.W. has received speaker fees from Otsuka, Lundbeck and Janssen. All other authors report no biomedical financial interests or potential conflicts of interest.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
A list of authors and their affiliations appears at the end of the paper.
These authors contributed equally: Daniel Keeser, Elias Wagner.
Contributor Information
Vladislav Yakimov, Email: V.Yakimov@med.uni-muenchen.de.
CDP Working Group:
Vladislav Yakimov, Marcel S. Kallweit, Emanuel Boudriot, Mattia Campana, Alkomiet Hasan, Andrea Schmitt, Valéria de Almeida, Stephanie Behrens, Fanny Dengl, Peter Falkai, Laura E. Fischer, Nadja Gabellini, Vanessa Gabriel, Thomas Geyer, Katharina Hanken, Genc Hasanaj, Alexandra Hirsch, Georgios Ioannou, Iris Jäger, Sylvia de Jonge, Temmuz Karali, Susanne Karch, Berkhan Karslı, Christoph Kern, Nicole Klimas, Maxim Korman, Lenka Krčmář, Isabel Lutz, Julian Mechler, Verena Meisinger, Matin Mortazavi, Joanna Moussiopoulou, Karin Neumeier, Frank Padberg, Boris Papazov, Sergi Papiol, Pauline Pingen, Oliver Pogarell, Siegfried Priglinger, Florian J. Raabe, Lukas Roell, Moritz J. Rossner, Susanne Schmölz, Enrico Schulz, Benedikt Schworm, Sven Wichert, Peter Zill, Daniel Keeser, and Elias Wagner
Supplementary information
The online version contains supplementary material available at 10.1038/s41537-024-00543-4.
References
- 1.Brugger, S. P. & Howes, O. D. Heterogeneity and homogeneity of regional brain structure in schizophrenia: a meta-analysis. JAMA Psychiatry74, 1104–1111 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hyman, S. E. Revolution stalled. Sci. Transl. Med.4, 155cm111 (2012). [DOI] [PubMed] [Google Scholar]
- 3.Owen, M. J. New approaches to psychiatric diagnostic classification. Neuron84, 564–571 (2014). [DOI] [PubMed] [Google Scholar]
- 4.Halstead, S. et al. Alteration patterns of peripheral concentrations of cytokines and associated inflammatory proteins in acute and chronic stages of schizophrenia: a systematic review and network meta-analysis. Lancet Psychiatry10, 260–271 (2023). [DOI] [PubMed] [Google Scholar]
- 5.Fillman, S. G. et al. Elevated peripheral cytokines characterize a subgroup of people with schizophrenia displaying poor verbal fluency and reduced Broca’s area volume. Mol. Psychiatry21, 1090–1098 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bishop, J. R., Zhang, L. & Lizano, P. Inflammation subtypes and translating inflammation-related genetic findings in schizophrenia and related psychoses: a perspective on pathways for treatment stratification and novel therapies. Harv. Rev. Psychiatry30, 59–70 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lizano, P., Pong, S., Santarriaga, S., Bannai, D. & Karmacharya, R. Brain microvascular endothelial cells and blood-brain barrier dysfunction in psychotic disorders. Mol. Psychiatry28, 3698–3708 (2023). [DOI] [PubMed] [Google Scholar]
- 8.Campana, M. et al. Association of symptom severity and cerebrospinal fluid alterations in recent onset psychosis in schizophrenia-spectrum disorders—an individual patient data meta-analysis. Brain Behav. Immun.119, 353–362 (2024). [DOI] [PubMed] [Google Scholar]
- 9.Warren, N. et al. Inflammatory cerebrospinal fluid markers in schizophrenia spectrum disorders: A systematic review and meta-analysis of 69 studies with 5710 participants. Schizophr. Res.266, 24–31 (2024). [DOI] [PubMed] [Google Scholar]
- 10.Yakimov, V., Moussiopoulou, J., Hasan, A. & Wagner, E. The common misconception of blood-brain barrier terminology in psychiatry and neurology. Eur. Arch. Psychiatry Clin. Neurosci.274, 1779–1781 (2023). [DOI] [PMC free article] [PubMed]
- 11.Yakimov, V. et al. Relationship between blood-cerebrospinal fluid barrier integrity, cardiometabolic and inflammatory factors in schizophrenia-spectrum disorders. medRxiv (2024).
- 12.Pollak, T. A. et al. The blood-brain barrier in psychosis. Lancet Psychiatry5, 79–92 (2018). [DOI] [PubMed] [Google Scholar]
- 13.Lizano, P. et al. Multivariate relationships between peripheral inflammatory marker subtypes and cognitive and brain structural measures in psychosis. Mol. Psychiatry26, 3430–3443 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Laskaris, L. et al. Brain morphology is differentially impacted by peripheral cytokines in schizophrenia-spectrum disorder. Brain Behav. Immun.95, 299–309 (2021). [DOI] [PubMed] [Google Scholar]
- 15.Lizano, P. et al. Association of choroid plexus enlargement with cognitive, inflammatory, and structural phenotypes across the psychosis spectrum. Am. J. Psychiatry176, 564–572 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kim, S., Hwang, Y., Lee, D. & Webster, M. J. Transcriptome sequencing of the choroid plexus in schizophrenia. Transl. Psychiatry6, e964 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Williams, M. R., Macdonald, C. M. & Turkheimer, F. E. Histological examination of choroid plexus epithelia changes in schizophrenia. Brain Behav. Immun.111, 292–297 (2023). [DOI] [PubMed] [Google Scholar]
- 18.Falcao, A. M. et al. The path from the choroid plexus to the subventricular zone: go with the flow! Front Cell Neurosci.6, 34 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Castellani, G., Croese, T., Peralta Ramos, J. M. & Schwartz, M. Transforming the understanding of brain immunity. Science380, eabo7649 (2023). [DOI] [PubMed] [Google Scholar]
- 20.Yakimov, V. et al. Continuous cuprizone intoxication allows active experimental autoimmune encephalomyelitis induction in C57BL/6 mice. Histochem Cell Biol.152, 119–131 (2019). [DOI] [PubMed] [Google Scholar]
- 21.Saunders, N. R., Dziegielewska, K. M., Fame, R. M., Lehtinen, M. K. & Liddelow, S. A. The choroid plexus: a missing link in our understanding of brain development and function. Physiol. Rev.103, 919–956 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Klistorner, S. et al. Choroid plexus volume in multiple sclerosis predicts expansion of chronic lesions and brain atrophy. Ann. Clin. Transl. Neurol.9, 1528–1537 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bergsland, N. et al. Association of choroid plexus inflammation on MRI with clinical disability progression over 5 years in patients with multiple sclerosis. Neurology100, e911–e920 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Assogna, M. et al. Association of choroid plexus volume with serum biomarkers, clinical features, and disease severity in patients with frontotemporal lobar degeneration spectrum. Neurology101, e1218–30 (2023). [DOI] [PMC free article] [PubMed]
- 25.Senay, O. et al. A preliminary choroid plexus volumetric study in individuals with psychosis. Hum. Brain Mapp.44, 2465–2478 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leucht, S. et al. Linking the PANSS, BPRS, and CGI: clinical implications. Neuropsychopharmacology31, 2318–2325 (2006). [DOI] [PubMed] [Google Scholar]
- 27.Schijven, D. et al. Large-scale analysis of structural brain asymmetries in schizophrenia via the ENIGMA consortium. Proc. Natl Acad. Sci. USA120, e2213880120 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Taylor, J. J. et al. A transdiagnostic network for psychiatric illness derived from atrophy and lesions. Nat. Hum. Behav.7, 420–429 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Opel, N. et al. Cross-disorder analysis of brain structural abnormalities in six major psychiatric disorders: a secondary analysis of mega- and meta-analytical findings from the ENIGMA Consortium. Biol. Psychiatry88, 678–686 (2020). [DOI] [PubMed] [Google Scholar]
- 30.Chen, W. et al. Inflammatory disequilibrium and lateral ventricular enlargement in treatment-resistant schizophrenia. Eur. Neuropsychopharmacol.72, 18–29 (2023). [DOI] [PubMed] [Google Scholar]
- 31.Zhou, Y. F. et al. Choroid plexus enlargement and allostatic load in schizophrenia. Schizophr. Bull.46, 722–731 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Keilp, J. G. et al. Cognitive impairment in schizophrenia: specific relations to ventricular size and negative symptomatology. Biol. Psychiatry24, 47–55 (1988). [DOI] [PubMed] [Google Scholar]
- 33.Costas-Carrera, A. et al. Difficulties during delivery, brain ventricle enlargement and cognitive impairment in first episode psychosis. Psychol. Med.54, 1339–1349 (2024). [DOI] [PubMed] [Google Scholar]
- 34.Lun, M. P., Monuki, E. S. & Lehtinen, M. K. Development and functions of the choroid plexus-cerebrospinal fluid system. Nat. Rev. Neurosci.16, 445–457 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pries, L. K. et al. Estimating exposome score for schizophrenia using predictive modeling approach in two independent samples: the results from the EUGEI study. Schizophr. Bull.45, 960–965 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhang, H., Khan, A., Kushner, S. A. & Rzhetsky, A. Dissecting schizophrenia phenotypic variation: the contribution of genetic variation, environmental exposures, and gene–environment interactions. Schizophrenia8, 51 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Boudriot, E. et al. Signature of altered retinal microstructures and electrophysiology in schizophrenia spectrum disorders is associated with disease severity and polygenic risk. Biol. Psychiatry96, 792–803 (2024). [DOI] [PubMed] [Google Scholar]
- 38.Kamintsky, L. et al. Blood-brain barrier imaging as a potential biomarker for bipolar disorder progression. Neuroimage Clin.26, 102049 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bannai, D., Cao, Y., Keshavan, M., Reuter, M. & Lizano, P. Manual segmentation of the human choroid plexus using brain MRI. J. Vis. Exp.10.3791/65341 (2023). [DOI] [PubMed]
- 40.Tadayon, E. et al. Improving choroid plexus segmentation in the healthy and diseased brain: relevance for Tau-PET imaging in dementia. J. Alzheimers Dis.74, 1057–1068 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Krcmar, L. et al. The multimodal Munich Clinical Deep Phenotyping study to bridge the translational gap in severe mental illness treatment research. Front. Psychiatry14, 1179811 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kalman, J. L. et al. Biobanking in everyday clinical practice in psychiatry—The Munich Mental Health Biobank. Front. Psychiatry13 (2022). [DOI] [PMC free article] [PubMed]
- 43.Boudriot, E. et al. Optical coherence tomography reveals retinal thinning in schizophrenia spectrum disorders. Eur. Arch. Psychiatry Clin. Neurosci.273, 575–588 (2023). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fischl, B. et al. Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron33, 341–355 (2002). [DOI] [PubMed] [Google Scholar]
- 45.Jernigan, T. L., Zatz, L. M., Moses, J. A. Jr. & Berger, P. A. Computed tomography in schizophrenics and normal volunteers. I. Fluid volume. Arch. Gen. Psychiatry39, 765–770 (1982). [DOI] [PubMed] [Google Scholar]
- 46.Li, Y. et al. Choroid plexus enlargement exacerbates white matter hyperintensity growth through glymphatic impairment. Ann. Neurol.94, 182–195 (2023). [DOI] [PubMed] [Google Scholar]
- 47.Team, R. C. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2022).
- 48.du Prel, J. B., Rohrig, B., Hommel, G. & Blettner, M. Choosing statistical tests: part 12 of a series on evaluation of scientific publications. Dtsch Arztebl Int107, 343–348 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bates, D., Mächler, M., Walker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw.67, 1–48 (2015).
- 50.Cumming, G. The new statistics: why and how. Psychol. Sci.25, 7–29 (2014). [DOI] [PubMed] [Google Scholar]
- 51.Coulson, M., Healey, M., Fidler, F. & Cumming, G. Confidence intervals permit, but do not guarantee, better inference than statistical significance testing. Front. Psychol.1, 26 (2010). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu, F. et al. Use of latent profile analysis and k-means clustering to identify student anxiety profiles. BMC Psychiatry22, 12 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The documentation sheets including the de-identified data will be made available upon publication on OSF (https://osf.io/hgsq2/). All software packages used in this study are publicly available. For details, see the (supplementary) methods section.