Abstract
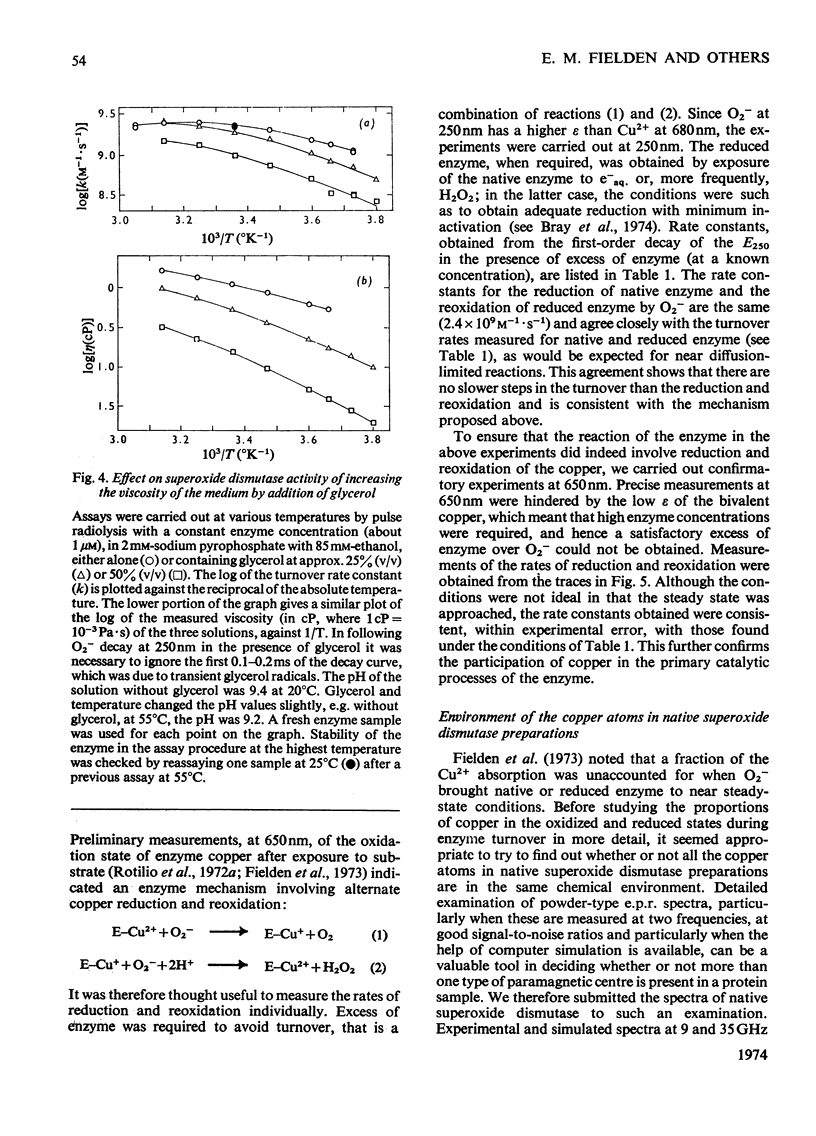
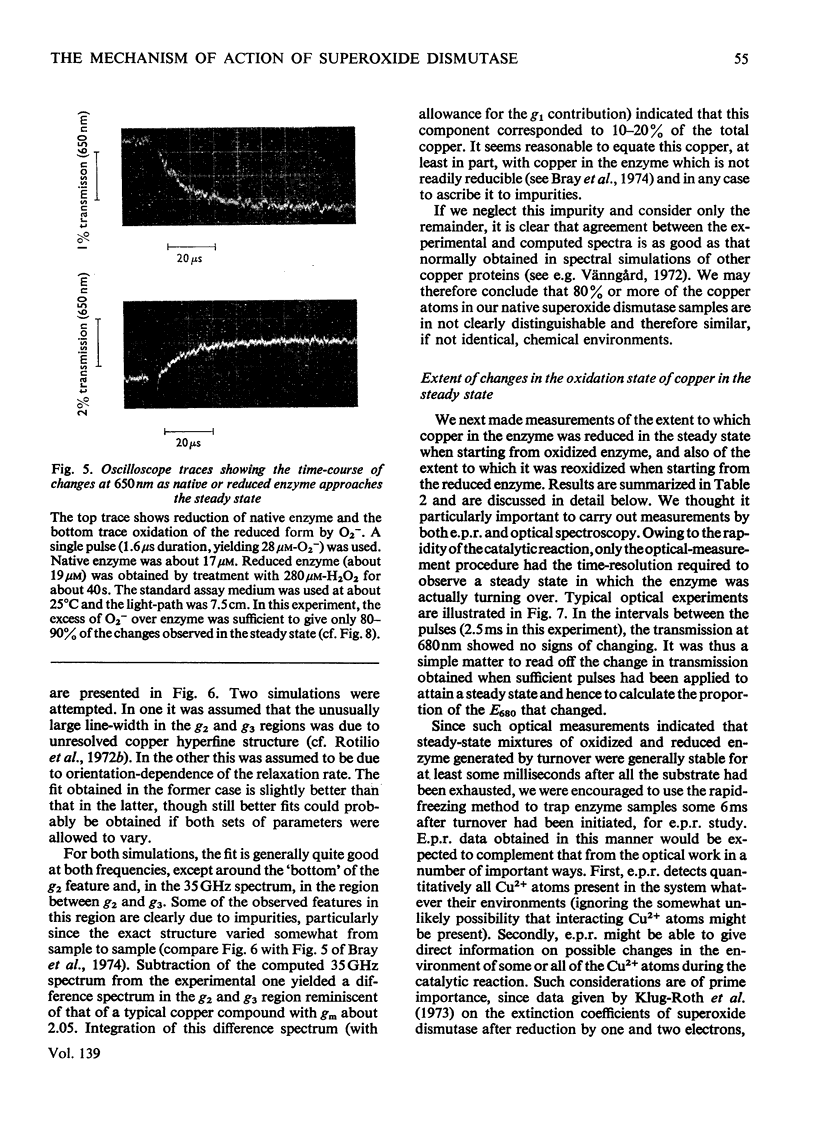
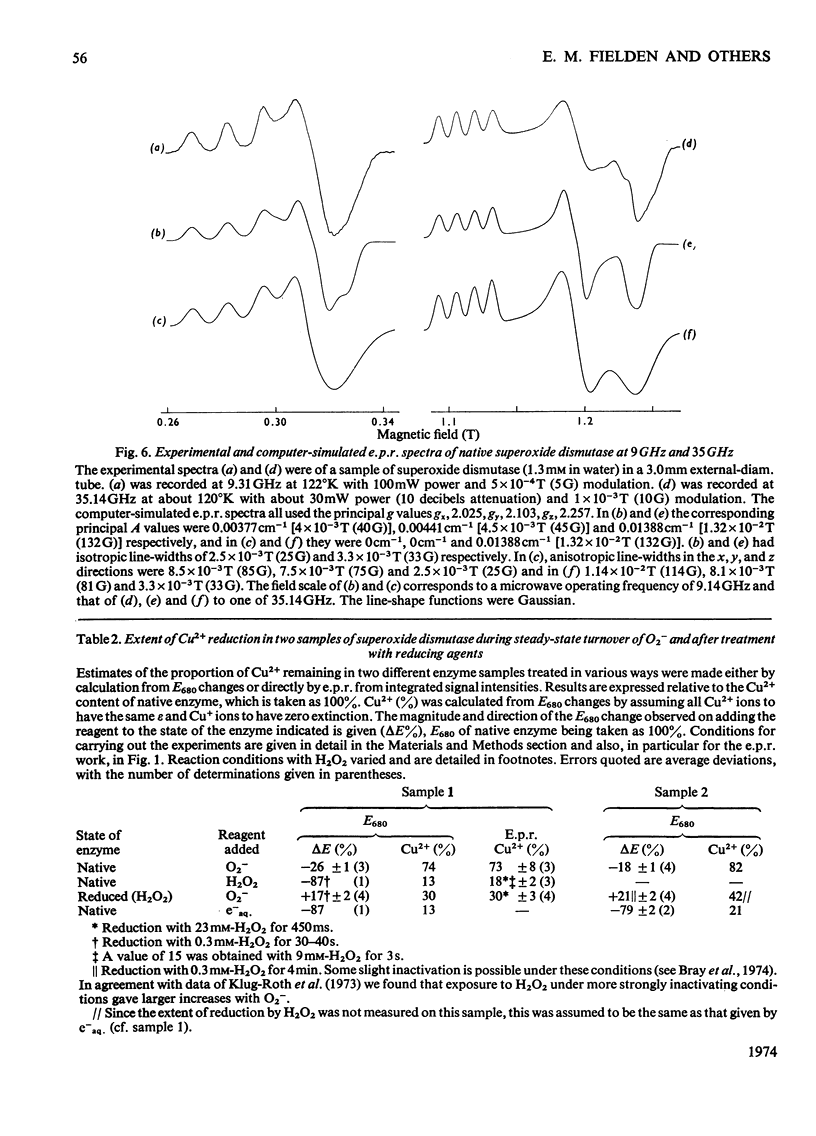
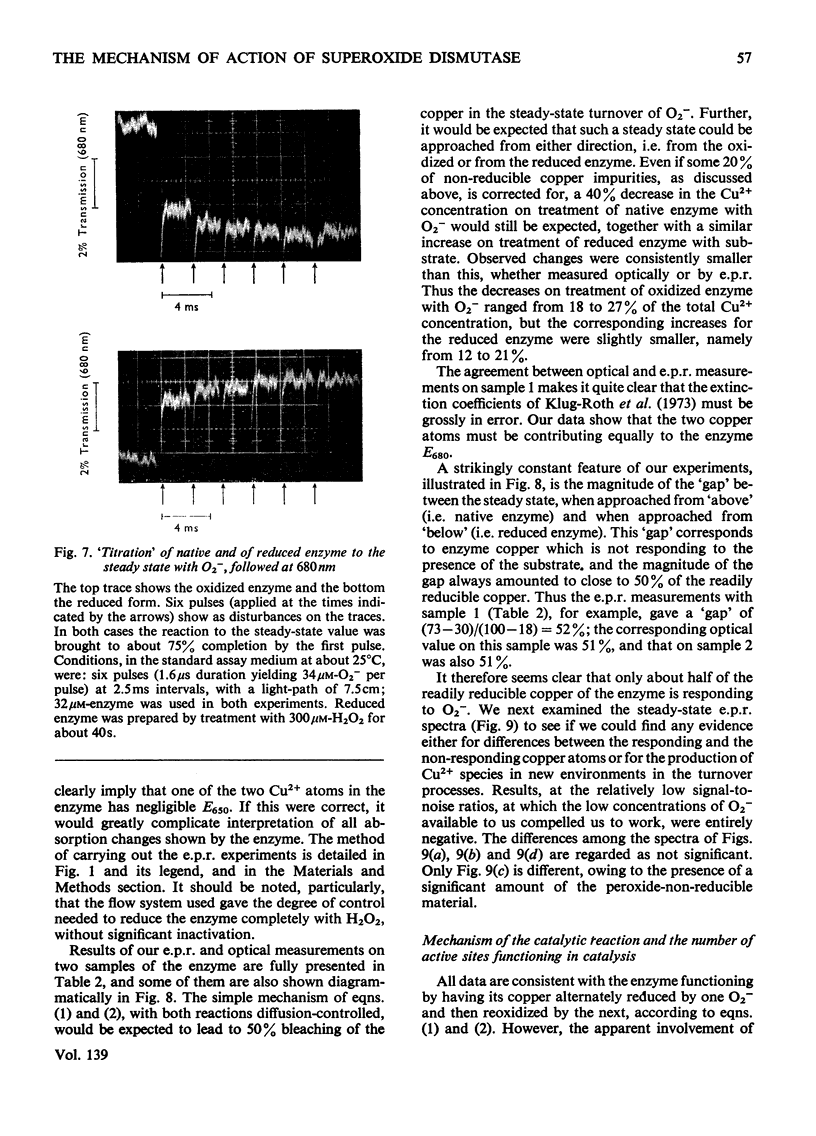
1. Detailed studies on the mechanism of the enzymic reaction of bovine superoxide dismutase were carried out by using pulse radiolysis and electron paramagnetic resonance (e.p.r.). 2. The second-order rate constant for reaction between superoxide dismutase and the superoxide ion was redetermined as (2.37±0.18)×109m−1·s−1 at 25°C. This reaction governs the turnover, and any first-order steps must have rate constants higher than about 106s−1. Turnover has a low activation energy and is slowed substantially when the viscosity is increased with glycerol, confirming that the reaction rate is near the limit for diffusion control. In water a reversible conformation change to a less active form appears to take place above about 40°C. 3. Pre-steady-state rates of reduction and reoxidation of copper in the enzyme are consistent with these processes being rate-limiting in enzyme turnover. 4. Examination, with the help of computer simulation, of the e.p.r. spectra at 9 and 35GHz of native superoxide dismutase indicated that, apart from 10–20% of impurities, only one species of Cu2+ is distinguishable. Further, the specific activity of our enzyme preparations, measured by pulse radiolysis, is at least as high as that obtained by other workers. 5. Nevertheless, measurement of the proportion of copper present as Cu2+ (determined both optically and by e.p.r. spectroscopy) in the steady states approached from both the oxidized and the reduced forms of the enzyme, indicates (after allowing for the impurities) that only half of the copper atoms participate in turnover. E.p.r. spectroscopy provided no evidence for differences between functioning and non-functioning Cu2+ atoms. 6. It is suggested that the results may be best interpreted in terms of an allosteric type of mechanism, with two initially indistinguishable copper atoms in the enzyme. Reaction of one of these with a superoxide ion then renders the other, at least transiently, unreactive.
Full text
PDF




Images in this article
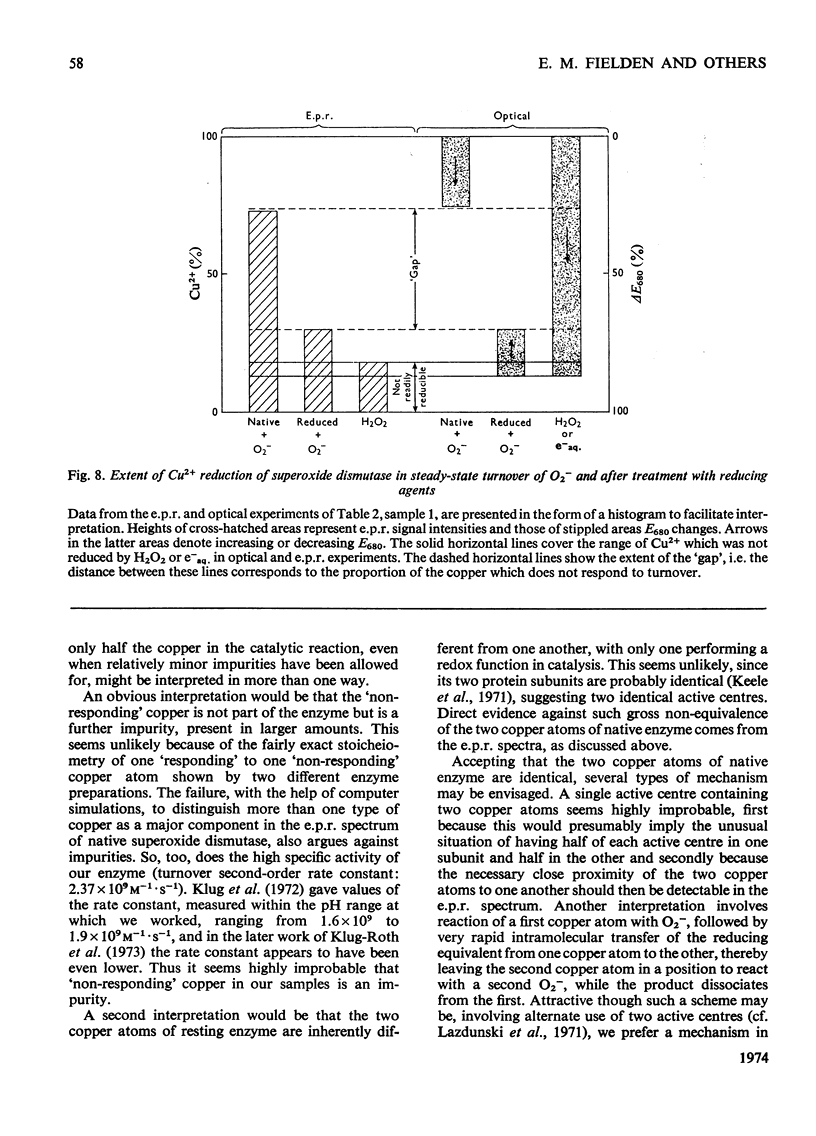
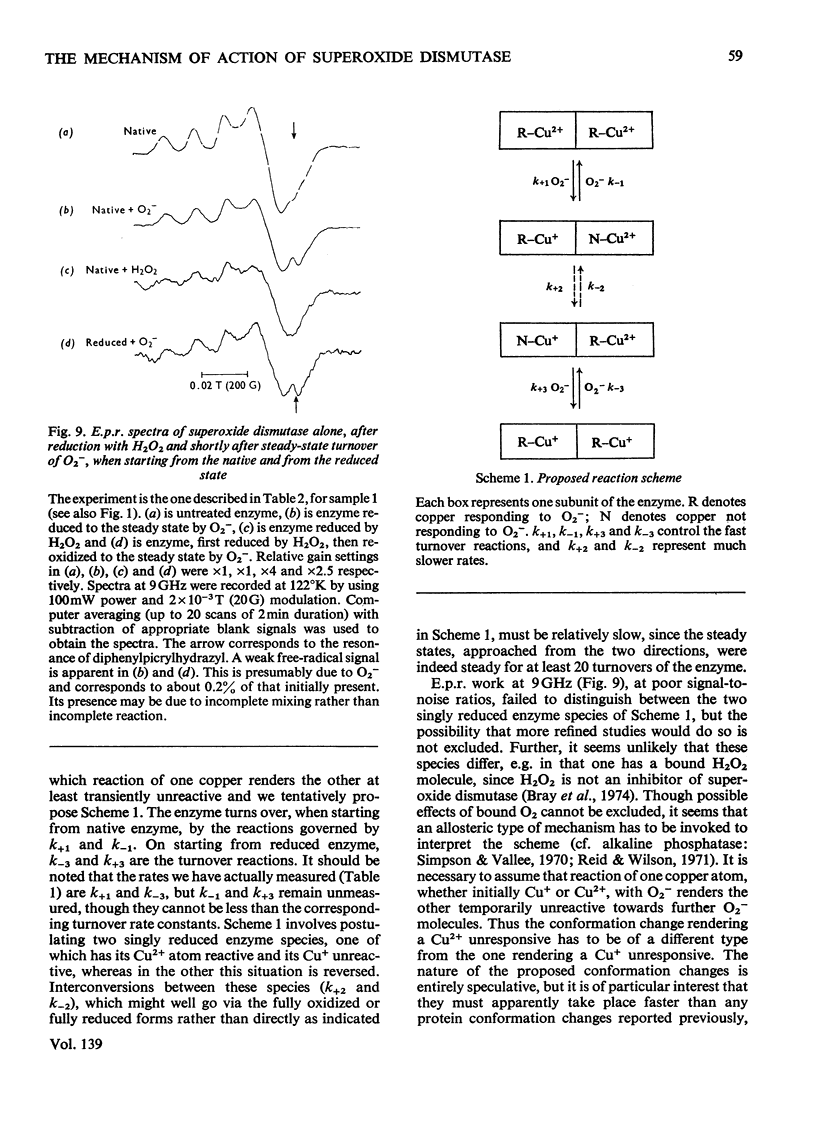
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ballou D., Palmer G., Massey V. Direct demonstration of superoxide anion production during the oxidation of reduced flavin and of its catalytic decomposition by erythrocuprein. Biochem Biophys Res Commun. 1969 Sep 10;36(6):898–904. doi: 10.1016/0006-291x(69)90288-5. [DOI] [PubMed] [Google Scholar]
- Bray G. A., Gallagher T. F., Jr Suppression of appetite by bile acids. Lancet. 1968 May 18;1(7551):1066–1067. doi: 10.1016/s0140-6736(68)91415-3. [DOI] [PubMed] [Google Scholar]
- Bray R. C., Cockle S. A., Fielden E. M., Roberts P. B., Rotilio G., Calabrese L. Reduction and inactivation of superoxide dismutase by hydrogen peroxide. Biochem J. 1974 Apr;139(1):43–48. doi: 10.1042/bj1390043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- EIGEN M., HAMMES G. G. ELEMENTARY STEPS IN ENZYME REACTIONS (AS STUDIED BY RELAXATION SPECTROMETRY). Adv Enzymol Relat Areas Mol Biol. 1963;25:1–38. doi: 10.1002/9780470122709.ch1. [DOI] [PubMed] [Google Scholar]
- Fee J. A. Studies on the reconstitution of bovine erythrocyte superoxide dismutase. I. The presence of four divalent metal-binding sites on the apo protein which are different from the native sites. Biochim Biophys Acta. 1973 Jan 25;295(1):87–95. doi: 10.1016/0005-2795(73)90076-7. [DOI] [PubMed] [Google Scholar]
- Keele B. B., Jr, McCord J. M., Fridovich I. Further characterization of bovine superoxide dismutase and its isolation from bovine heart. J Biol Chem. 1971 May 10;246(9):2875–2880. [PubMed] [Google Scholar]
- Klug-Roth D., Fridovich I., Rabani J. Pulse radiolytic investigations of superoxide catalyzed disproportionation. Mechanism for bovine superoxide dismutase. J Am Chem Soc. 1973 May 2;95(9):2786–2790. doi: 10.1021/ja00790a007. [DOI] [PubMed] [Google Scholar]
- Klug D., Rabani J., Fridovich I. A direct demonstration of the catalytic action of superoxide dismutase through the use of pulse radiolysis. J Biol Chem. 1972 Aug 10;247(15):4839–4842. [PubMed] [Google Scholar]
- Knowles P. F., Gibson J. F., Pick F. M., Bray R. C. Electron-spin-resonance evidence for enzymic reduction of oxygen to a free radical, the superoxide ion. Biochem J. 1969 Jan;111(1):53–58. doi: 10.1042/bj1110053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazdunski M., Petitclerc C., Chappelet D., Lazdunski C. Flip-flop mechanisms in enzymology. A model: the alkaline phosphatase of Escherichia coli. Eur J Biochem. 1971 May 11;20(1):124–139. doi: 10.1111/j.1432-1033.1971.tb01370.x. [DOI] [PubMed] [Google Scholar]
- McCord J. M., Fridovich I. Superoxide dismutase. An enzymic function for erythrocuprein (hemocuprein). J Biol Chem. 1969 Nov 25;244(22):6049–6055. [PubMed] [Google Scholar]
- McCord J. M., Keele B. B., Jr, Fridovich I. An enzyme-based theory of obligate anaerobiosis: the physiological function of superoxide dismutase. Proc Natl Acad Sci U S A. 1971 May;68(5):1024–1027. doi: 10.1073/pnas.68.5.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rotilio G., Bray R. C., Fielden E. M. A pulse radiolysis study of superoxide dismutase. Biochim Biophys Acta. 1972 May 12;268(2):605–609. doi: 10.1016/0005-2744(72)90359-2. [DOI] [PubMed] [Google Scholar]
- Rotilio G., Morpurgo L., Giovagnoli C., Calabrese L., Mondovì B. Studies of the metal sites of copper proteins. Symmetry of copper in bovine superoxide dismutase and its functional significance. Biochemistry. 1972 May 23;11(11):2187–2192. doi: 10.1021/bi00761a028. [DOI] [PubMed] [Google Scholar]
- Simpson R. T., Valee B. L. Negative homotropic interactions in binding of substrate to alkaline phosphatase of Escherichia coli. Biochemistry. 1970 Feb 17;9(4):953–958. doi: 10.1021/bi00806a035. [DOI] [PubMed] [Google Scholar]