Abstract
Background
People living with HIV (PLHIV) demonstrate accelerated aging and immunosenescence in spite of immune-restoration following long-term antiretroviral treatment (ART). Low level inflammation leading to inflammaging plays an important role in mediating premature immunosenescence. Ongoing viral replication, antiretrovirals and subclinical infections with the common viruses like Cytomegalovirus (CMV) are known to induce inflammaging. However such data is scarce in India where persistent low level inflammation is common in general population due to various subclinical infections. Hence we conducted a study to determine the extent of immunosenescence in asymptomatic PLHIV on long term ART in comparison with their age-matched controls.
Results
The study was conducted in asymptomatic virally suppressed PLHIV on ART for more than 5 years [n = 70, M: F = 36:34] and HIV uninfected controls [n = 68, M: F = 31:37] belonging to the age-group of 40–55 years. Blood samples were collected for assessing levels of immunosenescence markers on CD4 T cells by flow cytometry and anti-CMV antibodies as well as soluble CD14 (sCD14) levels by ELISA. The levels were compared between cases and controls and correlated with the levels of anti-CMV antibody and sCD14. PLHIV had significantly lower levels of naïve T cells and higher levels of activated and immunosenescent T cells than controls as indicated by CD38, CD57, CD28 expressing CD4 and CD8 T cells. PLHIV had higher levels of anti-CMV antibodies, but lower levels of sCD14 levels and HLADR + CD8 T cells than those in controls. Immunosenescent T cells correlated positively with anti-CMV antibody levels and negatively with sCD14 levels. Duration of dolutegravir based therapy correlated negatively with sCD14 levels.
Conclusions
Thus, higher levels of immune activation and immunosenescence in the cases possibly indicate their compromised immune status predisposing PLHIV to infections and cancers. The study indicated a need for CMV treatment regimens even in asymptomatic individuals for preventing immunosenescence. The study also indicated a role of dolutegravir induced loss of sCD14 levels in predisposing PLHIV to immunosenescence.
Keywords: HIV, Immunosenescence, anti-CMV antibodies, Soluble CD14, Dolutegravir
Background
Human Immunodeficiency Virus (HIV) infection is a major public health problem in India with 2.14 million estimated people living with HIV (PLHIV) in India and 1.18 million are on treatment under the national program [1]. Anti-retroviral treatment (ART) has significantly improved the survival of HIV patients and helped to reduce Acquired immunodeficiency syndrome (AIDS)-related mortality. About 50% of all deaths among the PLHIV taking ART are not due to AIDS [2]. Many studies have reported an increase in HIV-associated non–AIDS-defining age-associated comorbidities. Higher occurrence of age associated diseases like cardiovascular diseases, kidney diseases, neurocognitive impairment, and cancer at earlier ages suggest premature aging in this population [3]. Multiple geriatric conditions have been shown to occur more frequently among PLHIV aged in their 30s and 40s compared with uninfected controls [4]. HIV infection has been suggested as a model of accelerated immunosenescence [5]. Although ART reverses the aging process partially [6], these patients continue to demonstrate premature immunosenescence possibly caused by chronic low grade immune activation. Some of the antiretroviral drugs like nucleoside reverse transcriptase inhibitors have shown to inhibit telomerase activity possibly contributing to accelerated immunosenescence [7]. Integrase inhibitors, which are recently introduced as a part of first-line ART regimen under the national program of India, have shown to have safer effect on lipid profile as well as anti-inflammatory actions including causing reduction in the levels of soluble CD14 [8–10]. However dolutegravir has been shown to exert opposing effects on the aging related morbidities [11, 12].
Low grade chronic inflammation might persist in PLHIV in spite of viral suppression by long term ART because of ongoing viral replication, incomplete immune-restoration and anti-retroviral drugs themselves [13]. Plasma levels of inflammatory markers such as high sensitivity C-reactive protein (hs-CRP), D-dimer, IL-6, Galectin-9 have been shown to be biomarkers of non-AIDS comorbidities in PLHIV, even in those on ART [14, 15]. Markers of microbial translocation such as lipopolysaccharide (LPS), bacterial DNA, and markers of monocyte activation contributing to persistent immune activation do not completely normalize under ART and strongly associate with mortality [16, 17]. Soluble CD14 (sCD14), a marker of monocyte activation, is released upon stimulation of cells of monocyte-macrophage lineage. It has been shown to possess dual activities by promoting proinflammatory responses to LPS at the site of infections on one hand and mediate anti-inflammatory actions by interfering in interaction between membrane bound CD14 and LPS on the other [18]. In addition to HIV induced immune activation, coinfections with Hepatitis B virus, Hepatitis C virus, Cytomegalovirus (CMV) and Epstein-Barr virus are known to sustain immune activation and might induce age related changes. HIV, CMV infections and older age act synergistically to cause immune senescence as determined by expression of CD57 or decline in naïve T cells [19].
Seroprevalence of CMV has been shown to increase with age and is higher in developing than developed countries [20]. Further, India has also reported a very high seroprevalence of CMV infection among normal healthy blood donors [21] indicating their risk for developing premature immunosenescence, while the risk of CMV infections is known to decrease in PLHIV with the advent of ART [22]. Hence it is important to determine if PLHIV on long-term ART are still predisposed to the higher risk of immunosenescence in comparison to their healthy uninfected peers and whether the increased risk is attributable to CMV seropositivity. We conducted a case control study to investigate the risk of immunosenescence in PLHIV on virally suppressive ART and to determine the attributes of immunosenescence in the study participants.
Methodology
Ethics Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Ethics Committee of ICMR-National Institute of Translational Virology and AIDS Research (formerly National AIDS Research Institute), protocol code: NARI-EC/2020-23.
Study population
This case control study was conducted at ICMR-National Institute of Translational Virology and AIDS Research, India after approval by the Institutional Ethics committee. Study participants were enrolled in clinics after obtaining written informed consents. Eligibility criteria for cases were PLHIV with age between 40 and 55 years, on continuous first line ART for more than 5 years and virally suppressed. Age and sex matched healthy, HIV uninfected volunteers were invited to participate in the study as controls. Both cases (n = 70) and controls (n = 68) were asymptomatic at the time of enrolment and were free from known symptomatic acute or chronic infections other than HIV. Clinical, demographic data as well as blood samples were collected from the participants at the time of enrolment. PBMCs and plasma samples were separated and cryopreserved for the subsequent analysis.
Flow cytometry
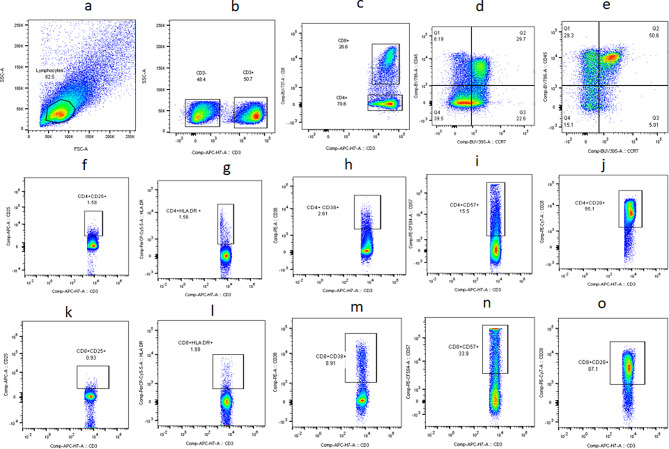
PBMCs were revived for flow cytometer analysis. The cells were stained with fluorochrome labelled antibodies against CD3, CD8, CD25, CD38, HLA-DR, CD28, and CD57 for identifying activated and immunosenescent CD4 and CD8 T cells. The cells were also stained with fluorochrome labelled antibodies against CD45RA and CCR7 for identifying memory subsets of CD4 and CD8 T cells. List of antibodies (BD biosciences, USA) used for the flow cytometry analysis is as given in Table 1. Stained cells were acquired using BD-FACS Fusion (BD biosciences, USA). Flow cytometry analysis was done using FlowJo version 8.0.3. Gating strategy used for identifying different cell populations is elaborated in Fig. 1.
Table 1.
List of antibodies used in flow cytometry assays
| Name of antibody | Fluorochrome | Catalogue No. |
|---|---|---|
| Anti-CD3 | APCH7 | 560176 |
| Anti-CD8 | BUV737 | 612754 |
| Anti-CD45 | BV786 | 563716 |
| Anti-CCR7 | BUV395 | 563977 |
| Anti-CD25 | APC | 567316 |
| Anti-HLA-DR | PerCP-Cy5.5 | 339194 |
| Anti-CD38 | PE | 555460 |
| Anti-CD57 | PECF594 | 562488 |
| Anti-CD28 | PECy7 | 560684 |
Fig. 1.
Gating strategy: Representative flow cytometry plots showing gating strategy used for analysis of the flow assays. Lymphocytes identified by forward and side scatter (a) were further gated for identification of CD3, CD4 and CD8 T cells (b, c). Maturation phenotypes of CD4 and CD8 T cells were identified based on CCR7 and CD45RA expression (d, e). All other markers were plotted on CD4 (f-j) and CD8 (k-o) T cells as shown in the plots. FMT controls which is full minus 3 were used to distinguish the population in the gating strategy. FMT controls were stained with surface markers for T cells. Gated CD3, CD4 and CD8 T cells, unstained for other markers, were further used for placing the gates
Semi quantitative estimation of anti-CMV antibodies by ELISA
Anti-CMV antibodies were estimated with a commercially available ELISA kit (BioCheck, Inc, USA). The ELISAs were performed as per the protocol given in manufacturer’s manual after diluting plasma samples to 1:40 by the sample diluent supplied with the kit. CMV IgG Index for each sample was determined by dividing optical density (OD) value of the sample by the OD value of the calibrator supplied with the kit as mentioned in the kit insert. Lower limit of detection of the analyte was 1.2 IU/ml.
Estimation of soluble CD14 (sCD14) levels by ELISA
Soluble CD14 levels were estimated using commercially available ELISA kit (R&D systems, Minneapolis, USA). The ELISA was performed as described previously [23] and according to the kit insert. sCD14 levels in the samples were determined by plotting standard curve as per the manufacturer’s instructions. Sensitivity of detection of the analyte was 125 pg/mL.
Statistical analysis
GraphPad Prism software version 9 was used for performing data analysis and plotting graphs. Unpaired data from the cases and controls enrolled in the study were compared using Mann Whitney test. Correlation analysis was performed to calculate Spearman’s rank correlation coefficient. P-values less than 0.05 were considered significant.
Results
Study participants
Demographic and clinical characteristics of cases (n = 70) and controls (n = 68) enrolled in the study are given in Table 2. Cases were virally suppressed PLHIV on ART for more than 5 years (median: 9 years; IQR:7–10 years). Age criteria for enrolling cases and controls was 40 to 55 years and there was no significant difference in their ages. 90% PLHIV enrolled in the study were shifted to dolutegravir based regimen at the time of the enrollment as per the national guidelines with the median duration of switch to the regimen of 11 months (IQR: 9–12 months).
Table 2.
Characteristics of study participants
| Controls (n = 68) | Cases (n = 70) | p value | |
|---|---|---|---|
| Age (years); Median (IQR) | 47(42–52) | 47(45–51) | NS |
| Sex; Male: Female | 31:37 | 36:34 | NS |
| CD4 Abs Count (cells/cmm); Median (IQR) | 808.5(665.5–1019) | 611.5(448-730.5) | < 0.0001 |
| CD4% Count; Median (IQR) | 41.5(37-46.75) | 30(25.75–35.25) | < 0.0001 |
| Number on DTG based therapy | - | 63 (90%) | - |
| Duration of DTG based therapy (months); Median (IQR) | - | 11 (9–12) | - |
*IQR- Interquartile ranges; NS- Not significant
Higher expression of immune activation markers on T cells in PLHIV
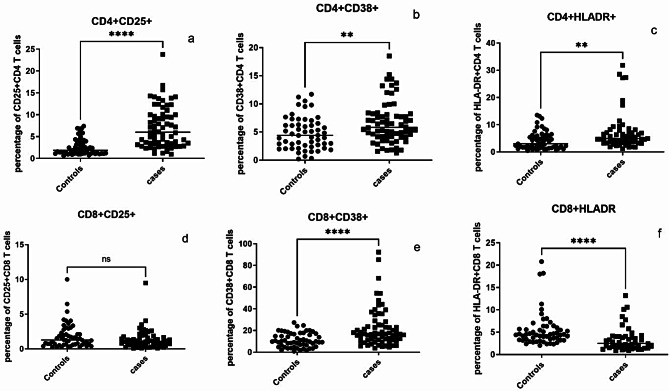
Expression of activation markers such as CD25, CD38 and HLA-DR were assessed on CD4 and CD8 T cells by flow cytometry and the results are presented in Fig. 2. PLHIV had significantly higher levels of activated CD4 and CD8 T cells than controls as indicated by CD38 expression (p = 0.0031 & <0.0001, respectively). Frequencies of CD4 T cells expressing HLA-DR and CD25 were also higher in PLHIV than the controls (p = 0.001 & <0.0001, respectively). However, surprisingly CD8 T cells expressing HLA-DR (p < 0.0001) were significantly lower in cases than the controls. Frequency of CD25 expressing CD8 T cells did not differ significantly between cases and controls.
Fig. 2.
T cells expressing activation markers: Percent CD4 and CD8 T cells expressing activation markers such as CD25 (a and d, respectively), CD38 (b and e, respectively) and HLA-DR (c and f, respectively) are shown on Y axis in controls and cases (X-axis) in the bar graphs. Horizontal lines in each data set represent medians for the values. P values of the unpaired data were calculated using Mann Whitney test and p values showing significant differences in the frequencies are indicated as ** (p < 0.01), **** (p < 0.0001). NS- not significant
Higher frequency of immunosenescent cells in PLHIV
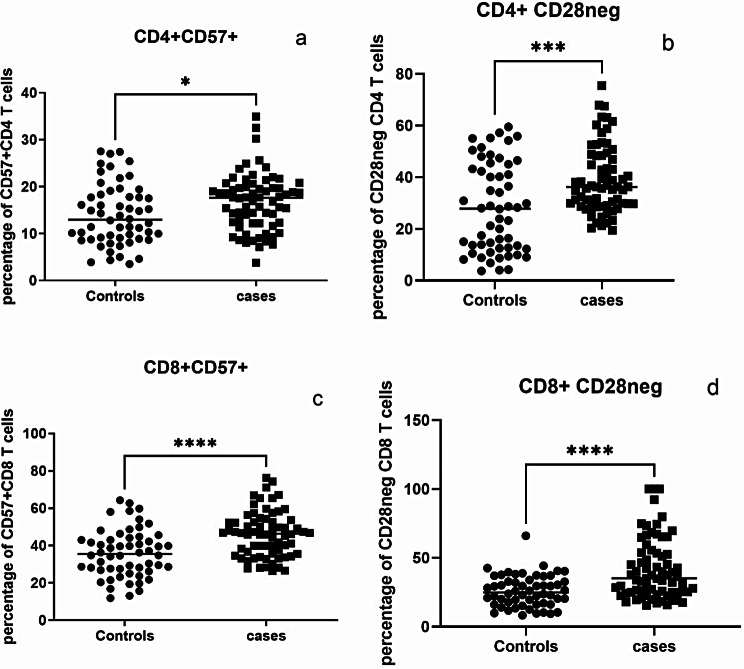
Frequency of CD28neg T cells and expression of CD57 were assessed to determine the immunosenescent status of CD4 and CD8 T cells. Frequency of CD4 and CD8 T cells expressing CD57 (p = 0.0149 & <0.0001, respectively) was higher in PLHIV than the controls as shown in Fig. 3. Similarly CD28neg CD4 and CD8 T cells were higher in the cases (p = 0.0004 & <0.0001, respectively).
Fig. 3.
Frequencies of immunosenescent T cells: Percent of immunosenescent CD4 and CD8 T cells identified as those expressing CD57 (a and c, respectively) and those not expressing CD28 (b and d, respectively) are shown on Y axis in controls and cases (X-axis). Horizontal lines in each data set represent medians for the values. P vales of the unpaired data were calculated using Mann Whitney test and p values showing significant differences in the frequencies are indicated as * (p < 0.05), *** (p < 0.001), **** (p < 0.0001)
Lower frequency of naïve T cells in PLHIV
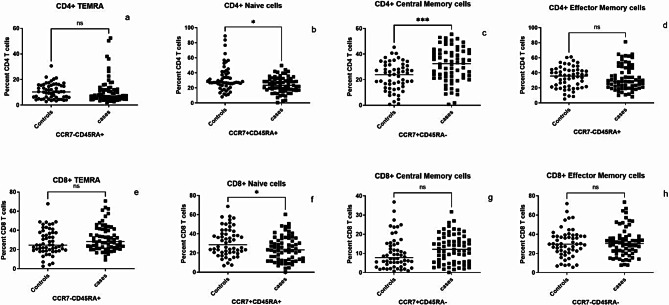
We also analyzed maturation phenotypes of CD4 and CD8 using CD45RA and CCR7 expressions. Both positive cells were labelled as naïve, CD45RA + CCR7- were terminal effector memory T cells (TEMRA cells), CD45RA-CCR7 + cells were central memory and both negative were labelled as effector memory CD4 cells. Naïve CD4 and CD8 T cells (p = 0.0114 & 0.0102, respectively) were lower in the cases as compared to controls as shown in Fig. 4. Contrarily central memory CD4 T cell population was higher in the cases than in the controls. There were no significant differences in the other cell populations.
Fig. 4.
T cells maturation phenotype: Percent Effector (a and e, respectively), Naïve (b and f, respectively), Central Memory (c and g, respectively), Effector Memory (d and h, respectively) CD4 and CD8 T cells identified based on CCR7 and CD45RA expression are shown on Y axis in controls and cases (X-axis). Horizontal lines in each data set represent medians for the values. P vales of the unpaired data were calculated using Mann Whitney test and p values showing significant differences in the frequencies are indicated as * (p < 0.05), *** (p < 0.001). NS- Not significant
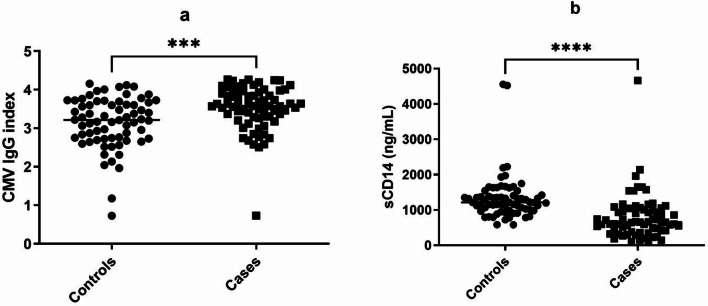
Higher CMV IgG index, but lower sCD14 levels in PLHIV
We measured CMV IgG index and sCD14 levels in plasma samples of PLHIV and uninfected controls. CMV IgG index was significantly higher in cases than the controls, while sCD14 levels were significantly lower in cases than those in the controls as shown in Fig. 5.
Fig. 5.
Anti-Cytomegalovirus (CMV) antibody and soluble CD14 (sCD14) levels: Plasma levels of Anti-CMV antibodies calculated as IgG index (a) and sCD14 (b) are plotted on Y axis in controls and cases (X-axis). Horizontal lines in each data set represent medians for the values. P value was calculated using Mann Whitney test and p values showing significant differences in the frequencies are indicated as *** (p < 0.001), **** (p < 0.0001)
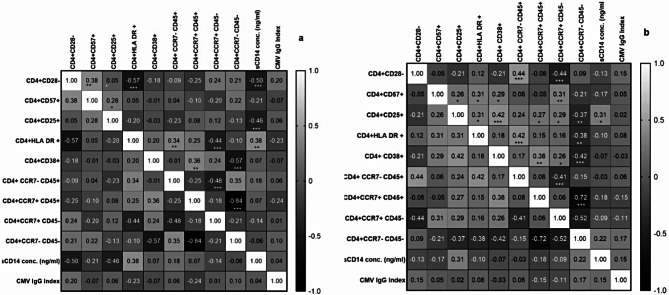
Correlation of immunosenescent CD4 T cells with immune activation markers in PLHIV and HIV uninfected controls
Correlation analysis of immunosenescent CD4 T cells with immune activation markers in PLHIV and HIV uninfected controls is shown in the form of heat-map in the Fig. 6. CD28 negative CD4 T cells in cases were found to significantly correlate positively with TEMRA and negatively with central memory CD4 T cells. While CD57 + CD4 T cells in cases correlated positively with CD4 T cells expressing activation markers such as CD25, HLA-DR and CD38 as well as with central memory CD4 T cells.
Fig. 6.
Heat map of correlation analysis of immunosenescent CD4 T cells: The heat maps show correlation analysis between different phenotypes of CD4 T cells, anti-cytomegalovirus IgG index and sCD14 levels in (a) HIV uninfected controls and (b) PLHIV on long term ART. The correlation coefficient (Spearman r) values are shown in each cell and as shades of gray as indicated in the scale shown in the figure. * (p < 0.05); ** (p < 0.01), *** (p < 0.001)
CD28 negative CD4 T cells in controls correlated positively with CD57 + CD4 T cells and negatively with and HLA-DR expression on CD4 cells as well as sCD14 levels. CD57 + CD4 cells correlated positively with CD25 + CD4 T cells.
Significant correlations observed in controls (a) with respect to CD4 T cells were as follows
CD28 neg and CD57+, CD28 neg and CD25+, CD28 neg and HLA-DR+, CD28 neg and sCD14; CD57 + and CD25; CD25 and sCD14; HLA-DR + and TEMRA; HLA-DR + and naïve cells; HLA-DR + and sCD14; CD38 + and central memory; CD38 and effector memory; TEMRA and central memory; naïve and effector memory cells.
Significant correlations observed in cases (b) with respect to CD4 T cells were as follows
CD28 neg and TEMRA; CD28 neg and central memory; CD57 + and CD25; CD57 + and HLA-DR; CD57 + and CD38; CD57 + and central memory; CD25 and HLA-DR+; CD25 and CD38; CD25 and naïve cells; CD25 and central memory; CD25 and effector memory; CD25 and sCD14; HLA-DR + and TEMRA; HLA-DR + and effector memory; CD38 + and naïve cells; CD38 + and central memory; CD38 and effector memory; TEMRA and central memory; naïve and effector memory cells.
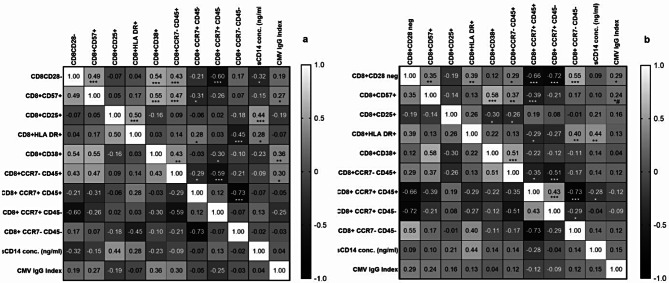
Correlation of immunosenescence markers expressed on CD8 with immune activation markers in PLHIV
Correlation analysis of immunosenescent CD8 T cells with immune activation markers in PLHIV and HIV uninfected controls is shown in the form of heat-map in the Fig. 7. CD28 negative CD8 T cells in cases were found to significantly correlate positively with CD57 + CD8 T cells, TEMRA, effector memory CD8 T cells and negatively with central memory as well as naïve CD8 T cells. CD57 + CD8 T cells in cases correlated positively with CD38 + and TEMRA CD8 T cells and negatively with naïve CD8 T cells. Both CD28 negative as well as CD57 + T cells correlated positively with CMV IgG index. HLA-DR + CD8 T cells correlated positively with effector memory CD8 T cells and sCD14 levels, but negatively with central memory CD8 T cells.
Fig. 7.
Heat map of correlation analysis of immunosenescent CD8 T cells: The heat maps show correlation analysis between different phenotypes of CD8 T cells, anti-cytomegalovirus IgG index and sCD14 levels in (a) HIV uninfected controls and (b) PLHIV on long term ART. The correlation coefficient (Spearman r) values are shown in each cell and as shades of gray as indicated in the scale shown in the figure. * (p < 0.05); ** (p < 0.01), *** (p < 0.001)
CD28 negative CD8 T cells in controls correlated positively with CD57 + and CD38 + CD8 T cells as well as TEMRA. While a negative correlation was observed between CD28 negative CD8 T cells and central memory CD8 T cells as well as sCD14 levels. Similarly CD57 + CD8 cells correlated positively with CD38 + and TEMRA CD8 T cells as well as with CMV IgG index. A negative correlation was observed between CD57 + CD8 T cells and naive CD8 T cells. A positive correlation was also observed between CD25 + CD8 T cells, HLADR + CD8 T cells and sCD14 levels. CMV IgG index correlated positively with CD38 + and TEMRA CD8 T cells.
Significant correlations observed in controls (a) with respect to CD8 T cells were as follows
CD28 neg and CD57+, CD28 neg and CD38+, CD28 neg and TEMRA; CD28 neg and effector memory; CD28 neg and sCD14; CD57 + and CD38; CD57 + and TEMRA; CD57 + and naïve cells; CD57 + and CMV IgG index; CD25 + and HLA-DR+; CD25 + and sCD14; HLA-DR + and naïve cells; HLA-DR + and effector memory; HLA-DR + and sCD14; CD38 + and TEMRA; CD38 + and central memory; CD38 and CMV IgG index; TEMRA and naïve cells; TEMRA and central memory; TEMRA and CMV IgG index; central memory and effector memory cells.
Significant correlations observed in cases (b) with respect to CD8 T cells were as follows
CD28 neg and CD57+, CD28 neg and HLA-DR; CD28 neg and TEMRA; CD28 neg and naïve cells; CD28 neg and central memory; CD28 neg and effector memory; CD28 neg and CMV IgG index; CD57 + and CD38; CD57 + and TEMRA; CD57 + and central memory; CD57 + and CMV IgG index; CD25 + and CD38; CD25 and TEMRA; HLA-DR + and naïve cells; HLA-DR + and effector memory; HLA-DR + and sCD14; CD38 + and TEMRA; TEMRA and naïve cells; TEMRA and central memory; naïve and central memory; naïve and effector memory; naïve and sCD14; central memory and effector memory cells.
Correlation of duration of dolutegravir based antiretroviral therapy with anti-CMV antibody titres and sCD14 levels
PLHIV in our study were in the phase of transition to dolutegravir based ART. We assessed correlation between duration of dolutegravir based ART in months with anti-CMV antibody titres and sCD14 levels to determine if dolutegravir had any effect on the levels. Both anti-CMV IgG index and sCD14 levels negatively correlated with the duration of dolutegravir based ART (Table 3). Although this correlation was significant for sCD14 levels, only a trend of negative correlation was observed for CMV IgG index. No significant correlations were observed between dolutegravir duration and any of the immunosenescence markers and hence that data is not shown.
Table 3.
Correlation of Dolutegravir duration with systemic immune inflammatory markers
| Correlation of Dolutegravir duration with | r value | Confidence interval | p value |
|---|---|---|---|
| sCD14 concentration (ng/ml) | -0.2177 | -0.4394 to 0.02907 | 0.0373 |
| CMV IgG Index | -0.1692 | -0.3978 to 0.07933 | 0.0840 |
Discussion
Premature inflammaging is a serious problem in HIV infection predisposing PLHIV to an enhanced risk of infections, autoimmune diseases, cancers etc. Inflammaging is mainly contributed by immune activation which is elevated in HIV infection. Long term ART has been shown to alleviate immune activation to some extent by lowering levels of many immune activation markers. However, levels of these markers may not reach the levels seen in HIV uninfected individuals. Inflammaging also occurs in elderly HIV uninfected individuals as a part of the aging process [24]. People residing in India share almost 27.5% of global burden of the infectious diseases [25] and hence are known to have higher levels of immune activation markers owing to the constant exposure to different chronic infectious diseases [26]. Hence it is important to determine levels of immune activation and immunosenescence markers in PLHIV under treatment as compared to their uninfected peers so as to understand if they are at a higher risk of age related conditions.
T cell Immune activation markers assessed cases and controls enrolled in our study were HLA-DR, CD38 and CD25. CD4 T cells expressing the markers were significantly higher in cases than the controls indicating increased CD4 T cells activation. CD38 expressing CD8 T cells were also significantly higher in cases than the controls indicating increased CD8 T cell activation. CD4 and CD8 T cell activation as assessed by CD38 and HLA-DR expression has been previously reported to be higher in PLHIV on virally suppressive ART than HIV uninfected controls [27]. Such persistently high immune activation is thought to be multifactorial being contributed by co-morbidities, chronic coinfections, residual viral expression, etc [28]. Interestingly CD8 expressing CD25 and HLA-DR markers were lower in the cases than the controls. A loss of HLA-DR expressing CD8 T cells after cryopreservation had been reported by us previously [29]. HLA-DR expressing CD8 cells are shown to expand in HIV infection as well as during the aging process [30, 31]. HLA-DR + CD8 + T lymphocytes have been shown to constitute a subset of regulatory T cells [32] and their reduced frequency might indicate an altered state of immunoregulation in PLHIV. On the flip side, these regulatory T cells might interfere in elimination of HIV reservoirs by suppressing immune responses through IL-10 dependent or independent mechanisms [33, 34]. HLA-DR + CD8 + T lymphocytes were found to be positively correlated with HIV DNA in one of the previous studies [35] indicating their possible interference in the elimination. Dolutegravir is thought to be able to reduce HIV reservoirs because of its high genetic barrier [36]. Effect of dolutegravir therapy on the size of viral reservoirs needs to be further investigated. We did not analyze HLA-DR and CD38 double positive T cells for their association with the other reported markers as they are mainly found to correlate with viremia in PLHIV [37] and were shown to be correlated differentially with the various markers reported in the study. Their expression in inflammation and morbidities associated with aging is also reported to vary on various T cell subsets [38].
We categorized CD4 and CD8 T cells as naïve, effector memory, central memory and TEMRA based on the expression of CD45RA and CCR7. Naïve CD4 and CD8 T cells were significantly lower in cases than the controls. Conversely PLHIV showed higher frequency of central memory CD4 T cells while all other subset did not differ significantly between cases and controls. Lower percentage of naïve CD4 cells and resultant increase in the percentages of memory CD4 T cells than reference ranges determined in healthy controls has been reported after long term ART in PLHIV [39]. Naïve T cells are mainly important in mounting immune responses against new antigens. Hence their decreased frequency makes these individuals susceptible to newer infections and also compromises their response to vaccine candidates.
Further the frequencies of immunosenescent CD4 and CD8 T cells identified by CD57 expression as well as negative for CD28 expression were significantly higher in PLHIV as compared to their controls. Immunosenescent T cells as identified by CD57 expression and lacking CD28 expression are shown to expand in HIV infection in spite of well controlled HIV replication [40, 41]. Chronic immune activation is thought to expand populations of TEMRA T cells having immunosenescent features, leading to premature aging of the immune system [41, 42]. Hence we looked for associations between T cell activation and immunosenescence markers. Positive correlations between immunosenescent T cells with T cells expressing either of the activation markers such as CD25, HLA-DR and CD38 in the study in both controls as well as cases, although the correlations were not consistent. CD4 + T-cell activation (i.e., percentage of CD38 + HLA-DR + cells) was strongly associated with the proportion of CD4 + CD57 + T cells in PLHIV receiving ART [27]. Immunosenescent T cells also correlated positively with TEMRA as was reported previously [41, 42].
We further performed correlation analysis to determine association of the immunosenescence markers with different inflammatory markers to understand if any of the markers could have contributed to their increased expression. We found that the CMV IgG index was higher in PLHIV than the controls and it correlated significantly with CD57 and /or CD28 negative CD8 T cells in cases as well as control. Impact of CMV infection on immunosenescence is well known. CMV infection leads to expansion of CD8 + TEMRA that re-express CD45RA [43]. We also found a positive correlation with CD8 TEMRA and CMV seropositivity in cases as well as controls. We also found higher CMV IgG index in cases than those in the controls as was also reported previously [27]. Anti CMV antibody levels have been shown to be associated with viral DNA levels or reactivation [44–46] indicating the role of CMV infection in enhancing risk of CD8 immunosenescence. Conditions like inflammation, cardiovascular disease, stroke, endothelial dysfunction are associated with the CMV sero-positivity and high antibody levels [47]. Our cases and controls were asymptomatic for CMV infection and the direct evidence of viral reactivation in the participant was lacking. However, correlation of CMV antibody levels with immunosenescence highlights the need for determining CMV viral loads for providing evidence of subclinical CMV reactivation.
We observed contrasting results with sCD14 levels. The levels were significantly lower in PLHIV than the controls. Dolutegravir is known to exert beneficial effects on inflammatory markers by reducing sCD14 levels [10]. Reductions in sCD14 levels have been reported after switch to integrase based regimen from protease as well as non-nucleoside reverse transcriptase inhibitors based regimens [10, 48]. Our cases were in transition to dolutegravir based therapy and this could be one of the reasons for lower sCD14 levels observed in our study. sCD14 levels correlate negatively with the duration of dolutegravir based therapy in the cases indicating the role of dolutergavir use in lowering sCD14 levels. Moreover sCD14 levels are also shown to be subtype dependent with lower levels associated with subtype C infections owing to reduced ability of HIV-1 C Tat in inducing inflammatory markers in monocytes [49]. This might also be a reason for the observed discrepancy in our findings in comparison to the reports from western countries with predominance of subtype B infections. We also observed a negative correlation of duration of dolutegravir based regime with CMV IgG index suggesting a need to determine the role of dolutegravir in controlling CMV infection. Since the correlation was weak and not significant, it needs to be interpreted with caution.
Negative correlation observed between immunosenescence markers with sCD14 levels was also a surprising finding. sCD14 is considered to be a monocyte activation marker and hence is thought to contribute to accelerated aging. However, one of the mechanisms by which sCD14 would act is by competing with the CD14 receptor for binding LPS reducing LPS-induced monocyte activation [18]. Soluble CD14 also acts as a negative regulator of human T cell activation [50]. The soluble form of CD14 protects against experimental inflammatory bowel disease in mice by promoting the integrity of the gut epithelium [51]. However, regulatory action of sCD14 is under-reported and needs to be further explored to understand its possible role in inducing immunosenescence. Interestingly levels of sCD14 and CD8 HLA-DR have been reported to be associated with size of integrated HIV DNA load and both these markers were significantly lower in our PLHIV indicating a need to study the size of viral reservoir in these participants [52].
Conclusion
Virally suppressed PLHIV showed higher levels of immune activation and impaired CD8 T cell mediated immune regulation despite being on long-term ART. They also had higher levels of immunosenescence markers possibly indicating their compromised immune responses against infections and cancers. Associations between anti-CMV antibody titres and immunosenescence markers indicated a need for intervention strategies for managing even subclinical CMV infection in PLHIV for countering immunosenescence in HIV infection. sCD14 levels, although considered as the marker of immune activation, were negatively associated with T cell immunosenescence questioning the protective effect of dolutegravir-induced reduction in sCD14 levels on aging related comorbidities.
Acknowledgements
We thank staff from immunology laboratory and ART centre staff for their help during the study. We especially thank the study participant for their time and participation in this study.
Abbreviations
- HIV
Human Immunodeficiency Virus
- PLHIV
People living with HIV
- ART
Antiretroviral treatment
- AIDS
Acquired immunodeficiency syndrome
- CMV
Lipopolysaccharide
- LPS
Cytomegalovirus
- sCD14
soluble CD14
- OD
Optical density
- IQR
Interquartile ranges
- NS
Not significant
- TEMRA cells
Terminal effector memory T cells
Author contributions
A.S.-Conceptualization, analysis and writing ; P.S.,A.R., M.G.-writing—review and editing, supervision, resources; N.K.,S.D.,J.S- Methodology, data analysis, writing—review and editing; R.B.,U.G.,S.K.-Methodology, writing—review and editing. All authors reviewed the manuscript.
Funding
This research was supported by Indian Council of Medical Research (ICMR) for funding.
Data availability
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.N.A.C.O.I.-N.I.o MS, India HIVE. 2017: Technical Report. New Delhi: NACO, Ministry of Health and Family Welfare, Government of India. 2018.
- 2.Maartens G, Celum C, Lewin SR. HIV infection: epidemiology, pathogenesis, treatment, and prevention. Lancet. 2014;384(9939):258–71. [DOI] [PubMed] [Google Scholar]
- 3.Smith RL, de Boer R, Brul S, Budovskaya Y, van Spek H. Premature and accelerated aging: HIV or HAART? Front Genet. 2012;3:328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rajasuriar R, Chong ML, Ahmad Bashah NS, Abdul Aziz SA, McStea M, Lee ECY, et al. Major health impact of accelerated aging in young HIV-infected individuals on antiretroviral therapy. AIDS. 2017;31(10):1393–403. [DOI] [PubMed] [Google Scholar]
- 5.Appay V, Almeida JR, Sauce D, Autran B, Papagno L. Accelerated immune senescence and HIV-1 infection. Exp Gerontol. 2007;42(5):432–7. [DOI] [PubMed] [Google Scholar]
- 6.Ianas V, Berg E, Mohler MJ, Wendel C, Klotz SA. Antiretroviral therapy protects against frailty in HIV-1 infection. J Int Assoc Provid AIDS Care. 2013;12(1):62–6. [DOI] [PubMed] [Google Scholar]
- 7.Leeansyah E, Cameron PU, Solomon A, Tennakoon S, Velayudham P, Gouillou M, et al. Inhibition of telomerase activity by human immunodeficiency virus (HIV) nucleos(t)ide reverse transcriptase inhibitors: a potential factor contributing to HIV-associated accelerated aging. J Infect Dis. 2013;207(7):1157–65. [DOI] [PubMed] [Google Scholar]
- 8.Auclair M, Guenantin AC, Fellahi S, Garcia M, Capeau J. HIV antiretroviral drugs, dolutegravir, maraviroc and ritonavir-boosted atazanavir use different pathways to affect inflammation, senescence and insulin sensitivity in human coronary endothelial cells. PLoS ONE. 2020;15(1):e0226924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Quercia R, Roberts J, Martin-Carpenter L, Zala C. Comparative changes of lipid levels in treatment-naive, HIV-1-infected adults treated with dolutegravir vs. efavirenz, raltegravir, and ritonavir-boosted darunavir-based regimens over 48 weeks. Clin Drug Investig. 2015;35(3):211–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lombardi F, Belmonti S, Borghetti A, Ciccullo A, Baldin G, Cauda R, et al. Reduced soluble CD14 levels after switching from a dual regimen with lamivudine plus boosted protease inhibitors to lamivudine plus dolutegravir in virologically suppressed HIV-infected patients. HIV Res Clin Pract. 2019;20(3):92–8. [DOI] [PubMed] [Google Scholar]
- 11.Gonzalez-Cordon A, Assoumou L, Moyle G, Waters L, Johnson M, Domingo P, et al. Switching from boosted PIs to dolutegravir decreases soluble CD14 and adiponectin in high cardiovascular risk people living with HIV. J Antimicrob Chemother. 2021;76(9):2380–93. [DOI] [PubMed] [Google Scholar]
- 12.Madzime M, Theron AJ, Anderson R, Tintinger GR, Steel HC, Meyer PWA, et al. Dolutegravir potentiates platelet activation by a calcium-dependent, ionophore-like mechanism. J Immunotoxicol. 2022;19(1):1–8. [DOI] [PubMed] [Google Scholar]
- 13.Babu H, Ambikan AT, Gabriel EE, Svensson Akusjarvi S, Palaniappan AN, Sundaraj V, et al. Systemic inflammation and the increased risk of Inflamm-Aging and Age-Associated diseases in people living with HIV on Long Term suppressive antiretroviral therapy. Front Immunol. 2019;10:1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tenorio AR, Zheng Y, Bosch RJ, Krishnan S, Rodriguez B, Hunt PW, et al. Soluble markers of inflammation and coagulation but not T-cell activation predict non-AIDS-defining morbid events during suppressive antiretroviral treatment. J Infect Dis. 2014;210(8):1248–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shete A, Wagh V, Sawant J, Shidhaye P, Sane S, Rao A et al. Antiretroviral Treatment-Induced Galectin-9 might Impact HIV Viremia in Addition to contributing to Inflammaging. Int J Mol Sci. 2023;24(15). [DOI] [PMC free article] [PubMed]
- 16.Brenchley JM, Price DA, Schacker TW, Asher TE, Silvestri G, Rao S, et al. Microbial translocation is a cause of systemic immune activation in chronic HIV infection. Nat Med. 2006;12(12):1365–71. [DOI] [PubMed] [Google Scholar]
- 17.Jiang W, Lederman MM, Hunt P, Sieg SF, Haley K, Rodriguez B, et al. Plasma levels of bacterial DNA correlate with immune activation and the magnitude of immune restoration in persons with antiretroviral-treated HIV infection. J Infect Dis. 2009;199(8):1177–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kitchens RL, Thompson PA, Viriyakosol S, O’Keefe GE, Munford RS. Plasma CD14 decreases monocyte responses to LPS by transferring cell-bound LPS to plasma lipoproteins. J Clin Invest. 2001;108(3):485–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Appay V, Fastenackels S, Katlama C, Ait-Mohand H, Schneider L, Guihot A, et al. Old age and anti-cytomegalovirus immunity are associated with altered T-cell reconstitution in HIV-1-infected patients. AIDS. 2011;25(15):1813–22. [DOI] [PubMed] [Google Scholar]
- 20.Fowler K, Mucha J, Neumann M, Lewandowski W, Kaczanowska M, Grys M, et al. A systematic literature review of the global seroprevalence of cytomegalovirus: possible implications for treatment, screening, and vaccine development. BMC Public Health. 2022;22(1):1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Das A, Munian D, Maity C, Pandey S, Paul S, Bhattacharya P. Prevalence of Cytomegalovirus infections in Blood donors and the newborn versus utility of leukocyte-reduced blood transfusion in the premature Newborn: An Observation from Eastern India. J Clin Neonatology. 2023;12(2):65–71. [Google Scholar]
- 22.Perello R, Vergara A, Monclus E, Jimenez S, Montero M, Saubi N, et al. Cytomegalovirus infection in HIV-infected patients in the era of combination antiretroviral therapy. BMC Infect Dis. 2019;19(1):1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shete A, Dhayarkar S, Dhamanage A, Kulkarni S, Ghate M, Sangle S, et al. Possible role of plasma Galectin-9 levels as a surrogate marker of viremia in HIV infected patients on antiretroviral therapy in resource-limited settings. AIDS Res Ther. 2020;17(1):43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sanada F, Taniyama Y, Muratsu J, Otsu R, Shimizu H, Rakugi H, et al. Source of chronic inflammation in aging. Front Cardiovasc Med. 2018;5:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ram B, Thakur R. Epidemiology and economic Burden of Continuing Challenge of Infectious diseases in India: analysis of Socio-demographic differentials. Front Public Health. 2022;10:901276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Batista MA, Calvo-Fortes F, Silveira-Nunes G, Camatta GC, Speziali E, Turroni S, et al. Inflammaging in endemic areas for infectious diseases. Front Immunol. 2020;11:579972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cobos Jimenez V, Wit FW, Joerink M, Maurer I, Harskamp AM, Schouten J, et al. T-Cell activation independently associates with Immune Senescence in HIV-Infected recipients of long-term antiretroviral treatment. J Infect Dis. 2016;214(2):216–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wu F, Simonetti FR. Learning from Persistent Viremia: mechanisms and implications for Clinical Care and HIV-1 cure. Curr HIV/AIDS Rep. 2023;20(6):428–39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shete A, Jayawant P, Thakar M, Kurle S, Singh DP, Paranjape RS. Differential modulation of phenotypic composition of HIV-infected and -uninfected PBMCs during cryopreservation. J Immunoass Immunochem. 2013;34(4):333–45. [DOI] [PubMed] [Google Scholar]
- 30.Imamichi H, Lempicki RA, Adelsberger JW, Hasley RB, Rosenberg A, Roby G, et al. The CD8 + HLA-DR + T cells expanded in HIV-1 infection are qualitatively identical to those from healthy controls. Eur J Immunol. 2012;42(10):2608–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lukas Yani S, Keller M, Melzer FL, Weinberger B, Pangrazzi L, Sopper S, et al. CD8(+)HLADR(+) Regulatory T cells change with aging: they increase in number, but lose checkpoint inhibitory molecules and suppressive function. Front Immunol. 2018;9:1201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Machicote A, Belen S, Baz P, Billordo LA, Fainboim L. Human CD8(+)HLA-DR(+) Regulatory T cells, similarly to classical CD4(+)Foxp3(+) cells, suppress Immune responses via PD-1/PD-L1 Axis. Front Immunol. 2018;9:2788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rocamora-Reverte L, Melzer FL, Wurzner R, Weinberger B. The Complex Role of Regulatory T Cells in immunity and aging. Front Immunol. 2020;11:616949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shete A, Suryawanshi P, Godbole S, Pawar J, Paranjape R, Thakar M. HIV-infected CD4 + T cells use T-bet-dependent pathway for production of IL-10 upon Antigen Recognition. Scand J Immunol. 2016;83(4):288–96. [DOI] [PubMed] [Google Scholar]
- 35.Zhu T, Cao W, Li T. HIV DNA positively correlates with HLA-DR + CD8 + T lymphocytes over 8-year suppressive antiretroviral therapy. AIDS. 2023;37(8):1335–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gantner P, Lee GQ, Rey D, Mesplede T, Partisani M, Cheneau C, et al. Dolutegravir reshapes the genetic diversity of HIV-1 reservoirs. J Antimicrob Chemother. 2018;73(4):1045–53. [DOI] [PubMed] [Google Scholar]
- 37.Chauhan NK, Vajpayee M, Mojumdar K, Singh R, Singh A. Study of CD4 + CD8 + double positive T-lymphocyte phenotype and function in Indian patients infected with HIV-1. J Med Virol. 2012;84(6):845–56. [DOI] [PubMed] [Google Scholar]
- 38.Bohacova P, Terekhova M, Tsurinov P, Mullins R, Husarcikova K, Shchukina I et al. Multidimensional profiling of human T cells reveals high CD38 expression, marking recent thymic emigrants and age-related naive T cell remodeling. Immunity. 2024;57(10):2362-79 e10. [DOI] [PubMed]
- 39.Lu L, Li X, Liu X, Qiu Z, Han Y, Song X, et al. The pattern and magnitude of T cell subsets reconstitution during ten years of ART with viral suppression in HIV-infected patients. Aging. 2022;14(23):9647–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Unemori P, Hunt P, Leslie K, Sinclair E, Deeks S, Martin J, et al. CD57+, a global marker of immunosenescence, is elevated in an atypical cohort of patients with Kaposi sarcoma and well-controlled HIV. Infect Agents Cancer. 2009;4(2):P43. [Google Scholar]
- 41.Fernandez S, French MA, Price P. Immunosenescent CD57 + CD4 + T-cells accumulate and contribute to interferon-gamma responses in HIV patients responding stably to ART. Dis Markers. 2011;31(6):337–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Thewissen M, Somers V, Hellings N, Fraussen J, Damoiseaux J, Stinissen P. CD4 + CD28null T cells in autoimmune disease: pathogenic features and decreased susceptibility to immunoregulation. J Immunol. 2007;179(10):6514–23. [DOI] [PubMed] [Google Scholar]
- 43.Fulop T, Larbi A, Pawelec G. Human T cell aging and the impact of persistent viral infections. Front Immunol. 2013;4:271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gianella S, Moser C, Vitomirov A, McKhann A, Layman L, Scott B, et al. Presence of asymptomatic cytomegalovirus and Epstein–Barr virus DNA in blood of persons with HIV starting antiretroviral therapy is associated with non-AIDS clinical events. AIDS. 2020;34(6):849–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hodowanec AC, Lurain NS, Krishnan S, Bosch RJ, Landay AL. Increased CMV IgG antibody titer is Associated with Non-AIDS events among Virologically suppressed HIV-Positive persons. Pathog Immun. 2019;4(1):66–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Iglesias-Escudero M, Moro-Garcia MA, Marcos-Fernandez R, Garcia-Torre A, Alvarez-Arguelles ME, Suarez-Fernandez ML, et al. Levels of anti-CMV antibodies are modulated by the frequency and intensity of virus reactivations in kidney transplant patients. PLoS ONE. 2018;13(4):e0194789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang H, Peng G, Bai J, He B, Huang K, Hu X et al. Cytomegalovirus Infection and Relative Risk of Cardiovascular Disease (Ischemic Heart Disease, Stroke, and Cardiovascular Death): A Meta-Analysis of Prospective Studies Up to 2016. J Am Heart Assoc. 2017;6(7). [DOI] [PMC free article] [PubMed]
- 48.Asundi A, Robles Y, Starr T, Landay A, Kinslow J, Ladner J, et al. Immunological and neurometabolite changes Associated With switch from Efavirenz to an integrase inhibitor. J Acquir Immune Defic Syndr. 2019;81(5):585–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.de Almeida SM, Tang B, Vaida F, Letendre S, Ellis RJ, Group H. Soluble CD14 is subtype-dependent in serum but not in cerebrospinal fluid in people with HIV. J Neuroimmunol. 2022;366:577845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Rey Nores JE, Bensussan A, Vita N, Stelter F, Arias MA, Jones M, et al. Soluble CD14 acts as a negative regulator of human T cell activation and function. Eur J Immunol. 1999;29(1):265–76. [DOI] [PubMed] [Google Scholar]
- 51.Buchheister S, Buettner M, Basic M, Noack A, Breves G, Buchen B, et al. CD14 plays a protective role in experimental inflammatory bowel disease by enhancing intestinal barrier function. Am J Pathol. 2017;187(5):1106–20. [DOI] [PubMed] [Google Scholar]
- 52.Ruggiero A, De Spiegelaere W, Cozzi-Lepri A, Kiselinova M, Pollakis G, Beloukas A, et al. During stably suppressive antiretroviral therapy Integrated HIV-1 DNA load in Peripheral Blood is Associated with the frequency of CD8 cells expressing HLA-DR/DP/DQ. EBioMedicine. 2015;2(9):1153–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.