Abstract
Bladder cancer represents one of the most prevalent malignant neoplasms of the urinary tract. In the Asian context, it represents the eighth most common cancer in males. In 2022, there were approximately 613,791 individuals diagnosed with bladder cancer worldwide. Despite the availability of efficacious treatments for the two principal forms of bladder cancer, namely non-invasive and invasive bladder cancer, the high incidence of recurrence following treatment and the suboptimal outcomes observed in patients with high-grade and advanced disease represent significant concerns in the management of bladder cancer at this juncture. Nanoparticles have gained attention for their excellent properties, including stable physical properties, a porous structure that can be loaded with a variety of substances, and so on. The in-depth research on nanoparticles has led to their emergence as a new class of nanoparticles for combination therapy, due to their advantageous properties. These include the extension of the drug release window, the enhancement of drug bioavailability, the improvement of drug targeting ability, the reduction of local and systemic toxicity, and the simultaneous delivery of multiple drugs for combination therapy. As a result, nanoparticles have become a novel agent of the drug delivery system. The advent of nanoparticles has provided a new impetus for the development of non-surgical treatments for bladder cancer, including chemotherapy, immunotherapy, gene therapy and phototherapy. The unique properties of nanoparticles have facilitated the combination of diverse non-surgical therapeutic modalities, enhancing their overall efficacy. This review examines the recent advancements in the use of nanoparticles in non-surgical bladder cancer treatments, encompassing aspects such as delivery, therapeutic efficacy, and the associated toxicity of nanoparticles, as well as the challenges encountered in clinical applications.
Keywords: bladder cancer, nanoparticle, drug delivery, chemotherapy, phototherapy, toxicity
Background
Bladder cancer (BC) is a prevalent tumor of the genitourinary system. In Asia, BC represents the eighth most common cancer in men, with an estimated 613,791 cases worldwide in 2022.1 Bladder cancer consists of two main types namely, muscle invasive bladder cancer (MIBC) and non-muscle invasive bladder cancer (NMIBC). Of these, NMIBC can be responsible for up to 75% of bladder cancers.2 Transurethral resection of bladder tumor (TURBT) accompanied by postoperative perfusion therapy has become the standard treatment for NMIBC.3–5 Conversely, the primary therapeutic modality for muscle-invasive advanced bladder cancer is platinum-based chemotherapy.6
However, the treatment of bladder cancer still presents two significant challenges: a high recurrence rate after systemic treatment and an unsatisfactory therapeutic effect for advanced patients. Additionally, non-surgical treatments of bladder cancer, such as chemotherapy, immunotherapy, and gene therapy, have three main limitations: poor drug targeting, obvious side effects, and drug resistance. These factors collectively contribute to the suboptimal efficacy of non-surgical treatments. Following the advent of nanoparticles as a novel agent in drug delivery, there has been a notable enhancement in the efficacy of drug delivery. Additionally, research exploring the potential of nanoparticles in non-surgical bladder cancer treatment has yielded encouraging outcomes in recent years. (Table 1).
Table 1.
Nanoparticles in Bladder Cancer Therapy
| Therapies | Nanocarriers | Payloads | Anti-tumor Strategy | References |
|---|---|---|---|---|
| Chemotherapy | Nanodiamond | VitaminA analogs | Enhanced the penetration into tumor | [7,8] |
| Doxorubicin | Enhanced cytotoxicity | |||
| Polymeric nanostructure | Pirabucin | Efficient delivery | [9] | |
| Nanoliposome | Vincristine | Reduced side effects | [10,11] | |
| Poly(L-aspartic acid sodium salt) nanoparticle | Cisplatin | Reduced side effects | [12] | |
| Glutathione-responsive nanoparticle | Cisplatin prodrug (Pt(IV)) | Overcome drug resistance | [13] | |
| Gene Therapy | Mesoporous silica nanoparticles | siRNA | Enhanced targeting ability | [14] |
| Lipid nanoparticle | [15] | |||
| Chrysotile nanotubes | circRNA | Enhanced passive targeting ability | [16] | |
| Exosome | Enhanced active targeting ability | [17,18] | ||
| Immunotherapy | MNC-ICG-NIG@SiO2 nanoparticles | BCG | Precisely controlled of drug release in tumor | [19] |
| Macrophage-derived exosome-mimetic nanovesicles | PD-L1 inhibitor | Blocking immunosuppression | [20] | |
| Poly (propylglycolide-co-glycolide) nanoparticles | Toll-like receptor 7/8 agonist | Enhanced immune response | [21] | |
| Phototherapy | HSA-Ce6/NTZ/FCS nanoparticles | Chlorin e6 | Enhanced transmucosal delivery | [22] |
| Tumor-specific ligands | Chlorin e6 | Enhanced targeting ability | [23] | |
| Mesoporous silica nanoparticles | - | Enhanced phototoxicity | [24] | |
| Gold nanorods | MnO2 | Enhanced drug release in tumor microenvironment | [25] |
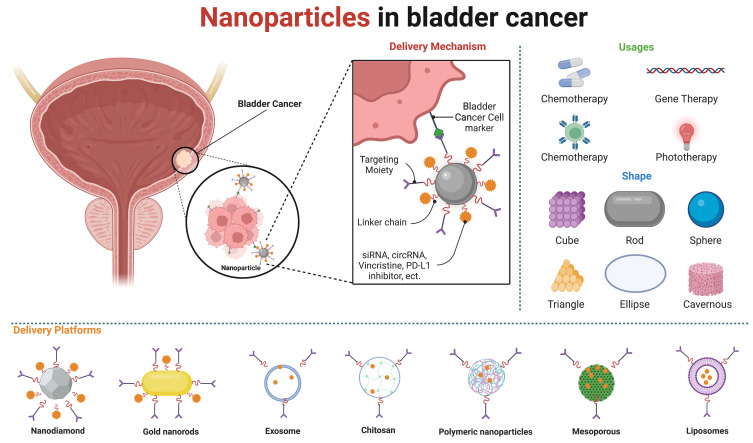
Nanoparticles have diameters between 1 and 100 nm and possess excellent stiffness, stability, porosity, and relative surface area, which render them a favored option for drug delivery systems. Nanoparticles alter the properties of drug delivery systems and improve their efficacy in cancer therapies, eg by enhancing therapeutic effects through increased bioavailability, enhanced permeability and retention (EPR) effects,26 and more targeted delivery.27–32 At this stage, the targeting strategy of nanoparticles has undergone a gradual transition from passive targeting, which relies on the enhanced permeability and retention (EPR) effect to concentrate the drug at the tumor site, to cell-specific targeting. This approach enables a more selective and efficacious internalisation of the drug into tumor cells.33,34 Previous reviews have concentrated on nanoparticle targeting strategies,26,31 while others have addressed the utilisation of nanoparticles in non-surgical bladder cancer treatment,3 and the diverse range of nanoparticles employed in bladder cancer therapy.35 Nevertheless, our analysis revealed that the majority of recent articles have not addressed the utilisation of nanoparticle delivery for the targeted delivery of specific media, including drugs, RNA, immunosuppressants, photosensitisers. This review discusses the application of different types of nanoparticles in non-surgical treatment modalities for bladder cancer, as well as the application of different types of nanoparticles in delivering specific media in various treatment modalities. It brings the perspective from the treatment modality to the delivered media, focusing on the advantages of nanoparticles in delivering different media. At the same time, it also addresses the toxic side effects of nanoparticles themselves and the barriers to their clinical application. Furthermore, this review addresses the potential toxic effects of nanoparticles and the obstacles to their clinical utilisation. (Figure 1).
Figure 1.
Nanoparticles in bladder cancer. Nanoparticles can be synthesized from a variety of materials and exhibit a range of shapes. Nanoparticle-enhanced targeted delivery greatly increases the efficiency of drug delivery, allowing nanoparticles to be used in various therapies for bladder cancer. Created in BioRender. ZHAO, X. (2024) https://BioRender.com/t06n586.
The Role of Nanoparticles as Drug Delivery Systems in Chemotherapy and the Delivery of Related Drugs
Chemotherapy plays a pivotal role in the treatment of bladder cancer.36 Intravenous injection and bladder instillation represent the predominant modes of chemotherapeutic drug delivery for bladder cancer at this stage. Nanoparticles offer a promising avenue for addressing these challenges. For instance, nanoliposomes have been extensively employed for the delivery of chemotherapy-related drugs, including doxorubicin, vincristine, and zorubicin.37 By enhancing the internalization of drugs, nanoparticles can improve drug solubility,7 increase the concentration of drugs at the tumor site,8 reduce the toxicity,38,39 and improve drug resistance.13
Nanoparticles as Drug Delivery Systems in the Delivery of Vitamin A Analogs
Nanodiamond (ND) is a carbon-based material that has great potential for medical applications due to its outstanding properties, especially its biocompatibility and surface functionalization capabilities. Its large relative surface area facilitates the attachment of various drugs, while its stability and low toxicity properties make it a new material for application in drug delivery systems.40,41 Vitamin A analogs and retinoids are thought to reduce the incidence of bladder cancer recurrence and delay the progression of bladder cancer.42,43 But the results of several clinical trials of vitamin A supplements did not meet expectations.44,45 One way to increase the bioavailability of vitamin A-like substances is to encapsulate them in various drug delivery systems.46 Nanocarriers have greater ability to penetrate mucus barriers and produce better cellular uptake.46 The superior endosomal escape capability of nanostructures is a critical factor in the delivery of therapeutics.47 Detonation nanodiamond (DND), a type of ND, embodies the potential as a non-toxic drug delivery system for urological applications.48 After retinoic acid bound to the ND enters the cell, the vitamin A-like substances adsorbed to the ND cannot simply be excreted by cytotoxicity or other extracellular exocytosis mechanisms. Instead, they are slowly desorbed from the ND within the endocytosis compartment, maintaining therapeutic concentrations.7 It is therefore possible that ND may become a viable nanoparticle for improving the efficacy of vitamin A analogs therapy for bladder cancer. Furthermore, it illustrates the potential of ND in drug delivery systems.8,49
Nanoparticles as Drug Delivery Systems in Delivering Anthracyclines
ND has also been shown to offer significant advantages in the delivery of doxorubicin (DOX). DOX was approved for clinical use by the FDA in the 1970s and has since been widely employed in the treatment of bladder cancer. DOX is topoisomerase II toxic, impairing topoisomerase II function and inducing irreversible DNA damage through the formation of an anthracycline-topoisomerase DNA ternary complex.50 The chitosan-coated doxorubicin-loaded NDs (Chi-NDX) treated with tripolyphosphate (TPP), designated as TPP-Chi-NDX, demonstrated enhanced cytotoxicity, exhibiting a high drug loading rate (>90%) and a diminished particle diameter (<150 nm). TPP-Chi-NDX also demonstrated a significantly enhanced drug release (>70%) in acidic media after 24 hour in comparison to doxorubicin-loaded NDs (NDX) (<45%), with a notable increase in the percentage of the percentage of DOX retained in the bladder wall was significantly higher after chitosan-coating and stabilization with TPP (46.3 ± 0.03%) compared to NDX (26.0 ± 0.03%) (p < 0.05).8 The use of nanoparticles facilitated a more efficacious delivery of doxorubicin, thereby enhancing the treatment of bladder cancer. Polyethylene glycol (PEG) is renowned for its exceptional structural flexibility, biocompatibility, and amphiphilicity.51 Furthermore, its application in nanoparticles enhances drug targeting and bioavailability.52 Polyethylene glycol and glutaraldehyde co-modified fluorinated chitosan as a collagen-targeted transepithelial penetration enhancer, the particle-assisted intravesical titration of the chemotherapeutic pirabucin in combination with the immune-stimulating agent interleukin-12 has been demonstrated to facilitate the passage of drugs across the mucosal barrier. This approach has the potential to trigger an effective anti-tumor chemo-immune response, which can in turn destroy the bladder tumor in situ and inhibit the recurrence of the cancer.9
A significant number of nano delivery systems currently in development are designed to deliver original forms of chemotherapeutics, with relatively little research conducted on prodrugs. A number of nanoparticles, including mucoadhesive nanoparticles based on a reactive oxygen species (ROS) - activated gambogic acid prodrug and chitosan nanoparticles coated with hyaluronic acid for the delivery of tamoxifen have demonstrated promise in the treatment of bladder cancer.53,54 The glutathione (GSH) -responsive mucosal adhesion nanocarrier, which is responsive to GSH, has the capacity to simultaneously deliver the NAD(P)H: quinone oxidoreductase 1(NQO1) substrate KP372-1 and a ROS-activatable prodrug of epirubicin. This has the potential to significantly increase the efficacy of the prodrug in an NQO1-dependent manner. Consequently, the expedient release of KP372-1 following endocytosis elevates intracellular ROS levels, thus facilitating the selective activation of epirubicin prodrugs in cancer cells while minimizing toxicity in normal cells and expanding the window of selectivity during intravesical chemotherapy.55
Nanoparticles as Drug Delivery Systems in Delivering Cisplatin
Cisplatin was the inaugural metallic pharmaceutical utilized in the treatment of neoplastic, and the administration of cisplatin-based systemic chemotherapy is the prevailing therapeutic modality for advanced bladder cancer.6,56 The mechanism of the anti-tumor activity of cisplatin is typically conceptualized as a consequence of its interaction with purine bases on DNA, resulting in the damage of DNA and the subsequent activation of several specific signaling pathways that ultimately lead to apoptosis.57 Nevertheless, there is a need to enhance the delivery efficiency of cisplatin while concomitantly reducing its neurotoxic effects.57–59 A nano-diamino-tetrac (NDAT) formulation loaded with cisplatin was administered to mice that had been inoculated with tumor xenografts derived from human bladder cancer cells. The administration of NDAT-cisplatin resulted in a five-fold increase in drug uptake into the tumor xenografts in comparison to the conventional cisplatin treatment. In the experiment, mice that received an untreated cisplatin treatment developed hindlimb spasms. In contrast, mice that received NDAT-cisplatin did not develop these symptoms. The results of this study indicate that drug delivery via the NDAT-cisplatin method may mitigate the adverse effects of cisplatin on motor nerves, namely its neurotoxicity.38
Poly(L-aspartic acid sodium salt) (PAA) nanoparticles loaded with cisplatin (CDDP) demonstrated the capacity to enhance the therapeutic efficacy of NMIBC while concurrently reducing the adverse effects of the drug. The nanoparticles were injected into mice and rats with the N-methyl-N-nitrosourea rat model of bladder cancer. It was observed that intravesical administration of a conventional CDDP solution in mice resulted in rapid systemic exposure (>2 μg/mL in plasma) in one hour. In contrast, none of the mice administered with CDDP NP had detectable amounts of CDDP in their plasma. Furthermore, the PAA-CDDP NP was observed to markedly elevate the concentration of CDDP in the bladder of rats, exhibiting a six-fold increase at the one-hour mark following intravesical administration when compared to the conventional CDDP solution (P < 0.05). At four hours post-administration of PAA-CDDP NPs, the concentration of CDDP remained at 45% of the one-hour concentration, indicating that the NPs enhanced the uptake and retention of CDDP in rats. Additionally, they may mitigate CDDP-related adverse effects when used intravesically by reducing systemic exposure and decreasing local toxicity.12
A further issue with CDDP is the emergence of substantial resistance in cancer cells. DNA damage repair represents a pivotal mechanism that underlies this resistance to CDDP.57 WEE1 is a pivotal protein in the DNA repair pathway, facilitating the repair of DNA damage through the regulation of the G2/M checkpoint and the S phase.60,61 A GSH-responsive nanoparticle was loaded with a cisplatin prodrug (Pt(IV)) and a WEE1 inhibitor (MK1775) to address the issue of cisplatin resistance. The particles ameliorate cisplatin resistance in two ways. Firstly, Pt(IV) is reduced to active CDDP, which results in DNA damage and activation of the cGAS-STING pathway. Secondly, DNA repair is inhibited by MK1775, thereby creating a synergistic antitumor effect in conjunction with CDDP.13
Nanoparticles as Drug Delivery Systems in Delivering Phytochemicals and Their Derivatives
The use of phytochemicals and their derivatives in cancer treatment represents a promising avenue of research, with preliminary evidence suggesting potential advantages in improving treatment outcomes.62,63 Phytochemicals and their derivatives, including vincristine and paclitaxel(PTX), have been extensively utilized in the treatment of bladder cancer through chemotherapy.64 Additionally, other pharmacological agents, including curcumin and tretinoin, have been demonstrated in experimental models to possess potential antineoplastic activity against bladder cancer.65–67 Nevertheless, the low water solubility, poor stability, low bioavailability, short half-life, and rapid clearance of phytochemicals have restricted their utilization in various applications.64,68 Vincristine is a naturally occurring antitumor drug derived from plants. It inhibits tumor growth by altering microtubule dynamics. Vincristine binds to tubulin, preventing its polymerization and, consequently, the inhibition of cancer cell mitosis.64,69 Nevertheless, improvements are required in the areas of drug retention time, drug distribution, and neurotoxicity associated with vincristine.70 The use of liposome nanoparticles for encapsulation represents a viable approach to addressing the aforementioned issues. A liposome is defined as a vesicle consisting of a lipid bilayer containing phospholipids and cholesterol.10 The potential of this approach has been investigated with a view to enhancing the delivery of drugs to specific sites within the body and reducing the incidence of adverse effects associated with the dosage of the medication.27,69,71 An innovative cholesterol (Chol) -modified sphingomyelin (SM) lipid bilayer structure forms SM-Chol by covalently binding cholesterol to SM. This novel lipid bilayer structure not only retains the membrane cohesive capacity of cholesterol, but also demonstrates in a systemic conformational relationship screen that SM-Chol containing disulfide bonds and longer junctions excels in blocking Chol transfer and payload leakage, and the lipid bilayer was able to increase the maximum tolerated dose of vincristine, reduce systemic toxicity, and improve drug delivery efficiency, and the lipid bilayer can significantly enhanced the antitumor efficacy in the SU-DHL-4 diffuse large B-cell lymphoma xenograft model in female mice.10 Egg sphingomyelin (ESM) was employed in the synthesis of liposomes of vincristine sulfate, utilizing cholesterol as a constituent. The resulting particle exhibited reduced toxicity in comparison to the conventional vincristine formulation and demonstrated superior antitumor efficacy against breast cancer cells.11 The aforementioned studies indicate that liposomal nanoparticles containing vincristine may have the potential to be an effective treatment for cancer. However, further experimentation is necessary to ascertain the specific effects of the nanoparticles in the context of bladder cancer.
PTX is also a type of phytochemical used in the treatment of tumors. It is a microtubule-stabilizing drug, and the anti-tumor mechanism of PTX has yet to be fully elucidated. However, there is a prevailing hypothesis that PTX exerts its anti-tumor effect by impeding the normal process of mitosis in cells.72 The antitumor effect of PTX is significantly influenced by the concentration of the drug. Consequently, increasing the concentration of the drug in a specific region represents a promising strategy to enhance the therapeutic efficacy of PTX. The use of paclitaxel/chitosan nanosupensions (PTX/CS NSs) has been explored as a means of prolonging the retention time of PTX, thereby improving the therapeutic outcome in bladder cancer. The positively charged nature of the nanosuspension facilitates its adsorption to the mucous membrane in the bladder, while simultaneously exhibiting good dispersion in water. In vitro cellular experiments demonstrated that PTX/CS NSs exhibited favorable biocompatibility and effective inhibition of bladder cancer cell proliferation.73
A further application of phytochemicals is to combine them with other drugs or nanoparticles in order to produce a more efficacious therapeutic effect. Lumbrokinase is an enzyme complex that has been employed in antitumor therapy. Experiments conducted with the Lumbrokinase/paclitaxel nanoparticle complex have demonstrated its efficacy in treating bladder cancer cells, as well as in a rat model of bladder cancer. These findings suggest that this nanoparticle mixture may serve as an effective nanocarrier to enhance anti-bladder cancer drug therapy.74 Similarly, the gold nanoparticles in combination with the Abies spectabilis extract demonstrated a greater degree of cytotoxicity against bladder T24 cancer cells than did the normal gold nanoparticles.75
The Role of Nanoparticles as Drug Delivery Systems in Gene Therapy
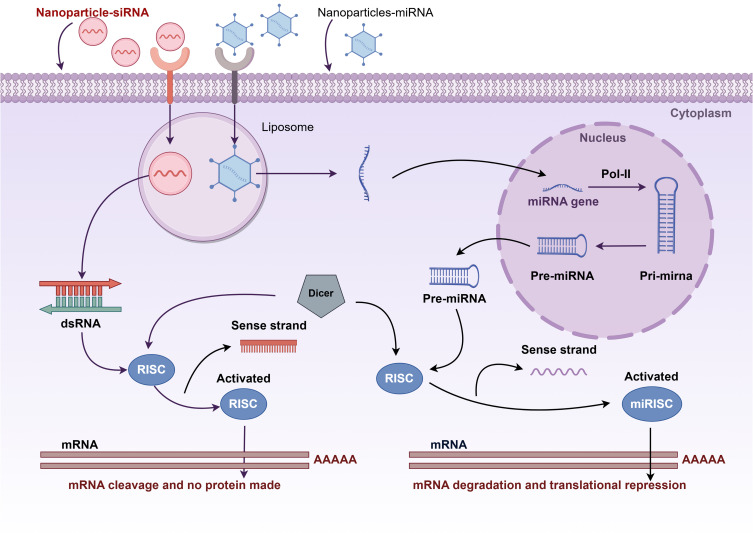
The use of gene therapy in the treatment of cancer has emerged as a prominent area of interest within the scientific community in recent years. Gene therapy is defined as the delivery of specific nucleic acids to tumor cells or modulation of the immune response. The initial gene modality entails the implementation of RNA interference (RNAi), exemplified by small interfering RNA (siRNA), with the objective of curbing gene expression levels.76 An alternative approach to gene therapy is the targeting of non-coding RNAs (ncRNAs), including microRNAs (miRNAs) and circular RNAs (circRNAs). These ncRNAs are not converted into proteins but are involved in evolutionary mechanisms and the regulation of biological events such as proliferation, migration, and differentiation.28,77 Nevertheless, the precise delivery of RNAs, including siRNAs and miRNAs, to cancer cells remains a challenge at this stage.16,78 Nucleic acid-based functional nanomaterials (NAFN) have been extensively utilized as a novel class of drug delivery nanocarriers for cancer therapy. NAFN possess the unique ability to efficiently load and protect therapeutic agents while simultaneously targeting delivery to the tumor site and triggering a responsive release.78 This paragraph focuses on the application of nanoparticles in the delivery of siRNA and circRNA (Figure 2).
Figure 2.
Nanoparticles in the delivery of siRNA and circRNA. After siRNA and miRNA are delivered by nanoparticles across a biological barrier (cell membrane), they are modified by Dicer to bind to the RNA-Induced Silencing Complex (RISC). The sense strands are excised, while the antisense strand remains bound to RISC, thereby activating it and mediating targeted mRNA degradation and translational repression. By figdraw.com.
Nanoparticles as Drug Delivery Systems in Delivering siRNAs
SiRNAs are double-stranded RNA molecules that can specifically target the messenger RNA (mRNA) of almost any pathological gene. Consequently, siRNAs have significant therapeutic potential for bladder cancer. As with all nucleic acid molecules, siRNAs can only enter cells via delivery. However, even when delivered systemically, siRNAs must traverse multiple layers of biological barriers, which results in a diminished biological effect.79,80 The utilization of nanoparticles represents a promising strategy for enhancing the delivery of siRNAs.14 The siRNA drug-loaded Ionizable lipid nanoparticles (iLNPs) modified by tumor targeting cyclic peptide (c(GRGDSPKC)) (GARP), GARP/siRNA iLNPs, are able to escape rapidly and efficiently from intracellular endosomes and lysosomes upon entry into cells. The modified GARP/siPLK1 demonstrated a more pronounced inhibitory effect on tumor growth compared to the unmodified siPLK1 siRNA-loaded lipid nanoparticles in both cell line-derived xenograft tumors and a mouse model of liver cancer patient-derived xenograft tumors. Subsequent experiments demonstrated that the nanoparticles enhanced the therapeutic effect of siRNA delivery for bladder cancer treatment.14 Programmed death-ligand 1 (PD-L1) is frequently expressed as a biomarker in human tumors. MicroRNA miR-34a is a factor that regulates tumor suppression.81 It is also involved in a number of other biological activities, including cell proliferation and apoptosis.82 Mesoporous silica nanoparticles modified by c(RGDfK) were employed for the co-delivery of miR-34 a and PD-L1 siRNA for the treatment of bladder cancer. Furthermore, the nanocarriers demonstrated favorable stability in the bloodstream and effective release of encapsulated miRNAs and siRNAs in tumor cells. Concurrent with the release of miRNAs and siRNAs, PD-L1 silencing and miR-34a overexpression led to a reduction in CD44 expression, proliferation, migration, and invasion. The results of the in vivo experiments demonstrated the efficacy of tumor growth inhibition in a bladder tumor model without significant adverse effects.15
Nanoparticles as Drug Delivery Systems in Delivering circRNAs
CircRNAs constitute a distinctive class of non-coding RNAs, characterized by a stable structure and the capacity to be reverse-spliced into loops. They play a pivotal role in numerous aspects of cancer biology, and the aberrant expression of circRNAs has been linked to the progression and development of BC.77,83 They can function as miRNA sponges, RNA-binding protein sponges, transcriptional regulators, or protein translation templates, thereby regulating biological activities.17,77,84 The synthesis of chrysotile nanotubes as nanocarriers demonstrated the capacity to prolong the half-life of si-circPRMT 5 in circulation, enhance its specific uptake by tumor cells, and maximize the silencing efficiency of circPRMT 5 when they delivered circPRMT5 siRNA. In vivo and in vitro experiments demonstrated that the nanoparticle complexes inhibited the growth of bladder cancer cells without significant toxicity, indicating the potential for their use in the treatment of bladder cancer.16
A novel type of lipid nanoparticle, designated as exosomes, has recently garnered substantial interest within the scientific community. Exosomes are derived from endosomes and typically possess a diameter of less than 100 nm.85 Natural exosomes have the capacity to transport a diverse range of signaling molecules, including double-stranded DNA, RNA, and proteins.86 Exosomes have the capacity to convey these signals to tumor cells, thereby modulating their biological activities.17,87 Exosome-mediated artificial circRNA (acircRNA) was employed as a gene therapy agent for bladder cancer. An acircRNA was constructed by ligating the cDNA sequences of transcription factors aptamers, thereby creating an acircRNA and selecting β-catenin and NF-κB as functional targets. The alterations in the expression levels of the target downstream genes following acircRNA treatment were quantified by qPCR, thereby demonstrating that the acircRNA was capable of specifically inhibiting β-catenin and NF-κB-related signaling pathways in BC cells and of impeding the activities of the target transcription factors. The acircRNA was loaded into exosomes using a CD63-HuR fusion protein. The functional experiments demonstrated that exosomal delivery of acircRNA transfected into bladder cancer cells resulted in proliferation inhibition, enhanced apoptosis, and migration inhibition.17 A bladder cancer-derived exosome circRNA_0013936 has been demonstrated to upregulate FATP 2 via the circRNA_0013936/miR-320a/JAK 2 pathway and to downregulate RIPK 3 via the circRNA_0013936/miR-301b/CREB 1 pathway. These effects significantly suppress the function of CD8+ T cells, namely the promotion of suppressive immunity. These findings suggest that circRNA_0013936 may represent a novel target for bladder cancer.18 Further studies are required to determine additional data, including cell viability, cell attachment/uptake, intracellular transport and endosomal escape, and transfection (eg, protein expression levels), with a view to developing an understanding of the effects of lipid nanoparticle delivery of RNA on cells.88
Histone lysine demethylase 6A (KDM6A) has been identified as a tumor suppressor in bladder cancer. Its absence has been observed to disrupt urothelial differentiation and induce tumor status.89 The utilization of mucoadhesive nanoparticles as drug-carrying particles to deliver messenger RNA (mRNA) in the bladder has the potential to prolong the exposure time of KDM6A-mRNA at the tumor site, thereby increasing the expression of KDM6A, which may prove beneficial in inhibiting the invasion and metastasis of bladder cancer.90
The Role of Nanoparticles as Drug Delivery Systems in Immunotherapy
Immunotherapy represents a novel therapy approach that aims to enhance the body’s immune response to malignant tumors. The human immune system is capable of suppressing tumor growth and metastasis. However, tumor cells are able to evade the immune system by establishing an immunosuppressive tumor microenvironment.91,92 The aim of immunotherapy is to reactivate anti-tumor immune cells and overcome immune escape mechanisms employed by tumors.93 The subsequent phase of immunotherapy research should concentrate on enhancing the efficacy of immunotherapeutic agents within the body, augmenting their selectivity for tumor cells, and minimizing adverse effects.94 The use of nanoparticles has been demonstrated to confer notable benefits in these domains. Moreover, a body of recent research has further substantiated the role of nanoparticles in augmenting the efficacy of immunotherapy.
Nanoparticles as a Drug Delivery System in Delivering BCG
Bacillus Calmette-Guérin (BCG) is a live attenuated strain of Mycobacterium bovis that is utilized for the immunization of humans against tuberculosis. The first report of intracystic immunization of humans with BCG was published in 1976 by Morales.94 Intravesical BCG drops represent the first immunotherapy for urothelial cancer to be approved by the FDA, having been granted approval in 1990 for use in patients with superficial bladder tumors.95 BCG is a primary agent for bladder cancer immunotherapy.3,95,96 Despite the incomplete understanding of the immune pathways induced by BCG, the resulting robust immune response has been demonstrated to elicit adaptive immunity and antitumor activity.96,97 The efficacy of intravesical instillation therapy is contingent upon the unique physiological structure of the bladder. This is due to the ease with which the drug can be diluted, the brief retention time, and the poor absorption of the drug, as evidenced in reference.98 Concurrently, BCG-induced immunotherapy may result in more significant adverse effects at the local and systemic levels.99 To ameliorate the above problems, a novel macrophage carrier system was developed, which uses a special nanoparticle namely MNC-ICG-NIG@SiO2 (MINS for short). This nanoparticle consists of CpG-conjugated magnetic nanoclusters forming the core and is encapsulated in a Se-Se bond-modified silica (SiO2) shell layer containing indocyanine green (ICG) and nigericin (NIG). This design endows MINS with the capacity to achieve precisely controlled release and immune activation within the tumor microenvironment. Following intravenous administration, BCG-mediated local inflammation in the tumor results in targeted accumulation of MINS@MΦ. MINS@MΦ accumulates in the tumor tissue and is immune-activated by laser irradiation, which results in ICG-mediated production of reactive oxygen species, Se-Se bond rupture, and subsequent release of NIGs to induce autoimmolation. The nanoparticle enhances the efficacy of BCG immunotherapy by precisely modulating cytokines, which may improve the therapeutic efficacy for bladder cancer.19 A nanovaccine based on the BCG cell wall skeleton nanoscale adjuvant (BCNA) conjugated with peptide neoantigens (M27 and M30) has been shown to have advantages in the treatment of melanoma. The nanovaccine exhibited excellent biocompatibility and antigen-presenting capacity, effectively triggering robust innate and tumor-specific immune responses, markedly enhancing the immunogenicity of neoantigens, and achieving 60% complete elimination and 25% growth prevention in a mouse melanoma model through combination with a PD-L1 antibody.100
Nanoparticles as Drug Delivery Systems in Delivering Immune Checkpoint Inhibitors
Immune checkpoints are markers that play an inhibitory regulatory role in the immune response. In normal circumstances, these immune checkpoints inhibit T cell function. Tumor cells can exploit this to evade the immune system.93 Immune checkpoint inhibitors (ICIs) have the potential to be an effective treatment for advanced bladder cancer.101,102 This may be associated with characteristics of the tumor microenvironment (TME), but the precise relationship between the two remains uncertain, and further research is required.92,103 ICI is employed in two principal modalities: as neoadjuvant therapy preceding surgical or radiotherapeutic intervention, and as a means of enhancing the patient’s complete remission rate, thereby increasing the probability of bladder preservation. The alternative approach is to administer the therapy following surgery or radiotherapy with the objective of preventing tumor recurrence or metastasis. This approach has the potential to prolong the patient’s progression-free survival and overall survival. While immunosuppressive therapy can enhance the prognosis of patients with advanced bladder cancer, several challenges remain. These include systemic immune-related adverse events (irAE), low response rates, and poor targeting due to complex tumor microenvironments (TMEs).91 PD-L1 is a transmembrane protein expressed on the surface of tumor cells. Programmed death-1 (PD-1) receptors on T cells bind to PD-L1, leading to the generation of inhibitory signals that suppress T cell function and prevent them from effectively killing tumor cells.104–106 PD-L1 overexpression is frequently observed in advanced bladder cancer.107 Macrophage-derived exosome-mimetic nanovesicles (EMVs) were utilized as nanoplatforms for the delivery of a CD73 inhibitor (AB680) and a programmed cell death ligand 1 (PD-L1) inhibitor in a mouse model of bladder cancer. The nanocomplex (AB 680@EMVs-aPDL1) demonstrated enhanced tumor targeting efficacy.20 AB 680 reduced extracellular adenosine production, and the combination treatment markedly enhanced cytotoxic T lymphocyte activation and infiltration.108 A ROS sensitive polymer (PHPM) can be employed to co-encapsulate the copper ion carriers elesclomol (ES) and copper (Cu) to form nanoparticles (NP@ESCu). The nanoparticles have been demonstrated to enhance the immune response to cancerous cells by promoting cuproptosis and combining with Anti-programmed cell death protein ligand-1 antibody (αPD-L1). The study demonstrated that NP@ESCu nanoparticles were capable of efficacious copper release and induction of copper-dependent cell death in bladder cancer cells. Additionally, NP@ESCu was observed to upregulate PD-L1 expression on tumor cells, thereby enhancing the therapeutic efficacy of αPD-L1.25
Gold nanoparticles (GNP) were bound to bacterial peptide 91–99 of listeriolysin O (LLO) to form the GNP-LLO 91–99 nanovaccine. The nanovaccine demonstrated efficacy in treating bladder cancer by blocking the immunosuppressive state, increasing the number of cytotoxic T cells and dendritic cells (DCs) within the tumor, and decreasing the number of immunosuppressive T reg cells and myeloid-derived suppressor cells. Indeed, GNP-LLO91–99 nanovaccines demonstrate an effective combination and potentiation of the action of ICI, specifically anti-CTLA-4 and anti-PD-1, thereby supporting their potential as a novel immunotherapy for BC.109
Nanoparticles as Drug Delivery Systems in Delivering Vaccine Adjuvants
The design of anti-cancer vaccines is intended to elicit a tumor-specific cytotoxic T-lymphocyte (CTL) response, which necessitates the activation of antigen-presenting cells (APCs), including DCs and macrophages, activating and harnessing the host’s immunity against cancer.21,110 The role of vaccine adjuvants in anticancer vaccines has become a subject of increasing interest as a result of the growing body of research examining the mechanisms of the immune response. Vaccine adjuvants can be classified into two main categories: immune agonists and delivery systems.111 Immune agonists promote the maturation and activation of antigen-presenting cells (APCs) by targeting Toll-like receptors and promoting the production of tumor-associated antigens (TAAs), which in turn enhance the adaptive immune response.112 Delivery systems are carrier materials that facilitate antigen presentation by enhancing the bioavailability of loaded antigens and directing antigen delivery to lymph nodes or APCs.113 Targeting the Toll-like receptor (TLR) pathway is regarded as a pivotal mechanism for immune agonists. TLR agonists have been the subject of extensive investigation as a novel class of vaccine adjuvants. Among these, TLR 7/8 agonists have been shown to improve the therapeutic effect of cancer in experiments.114–116 Nanovaccines are capable of precisely delivering tumor-specific antigens or TAAs to the immune system. Furthermore, nanovaccines can address the immune escape mechanism in the tumor microenvironment through the use of immune adjuvants, which serve to disrupt the immune-suppressed state. Additionally, the specificity of the nanoparticles allows for a notable prolongation of the circulation time of the drug, as well as an increase in the concentration of the drug at the target site.110,117 In conclusion, the use of nanovaccines based on nanoparticles represents a novel approach to enhancing the efficacy of immunotherapy. The encapsulation of a Toll-like receptor 7/8 agonist in poly(propylglycolide-co-glycolide) nanoparticles was observed to enhance the expression of DCs co-stimulatory molecules and the presentation of antigens via major histocompatibility complex (MHC) I. The nanoparticles were employed as an antigenic agent for the treatment of DCs. Upon administration via the subcutaneous route, the nanoparticles were transported to the draining lymph nodes, where they triggered the activation and expansion of DCs. This resulted in the addition of antigen-specific CD8 T cells and an enhanced cytotoxic T lymphocyte immune response, which led to a notable increase in therapeutic efficacy in a bladder cancer model and a significant reduction in systemic metastasis.21
Despite significant advancements in the field of nanoparticles and immunotherapy, current research primarily concentrates on conventional cytotoxic CD8+ T cell-mediated responses, with a limited scope for exploring the potential of nanoparticles to enhance the efficacy of cancer immunotherapy.106 It would be advantageous in the future to dedicate more research to the mechanisms of other immune cells, normal cells, and cancer cells in the immune response, as well as their role in immunotherapy.
The Role of Nanoparticles as Drug Delivery Systems in Phototherapy
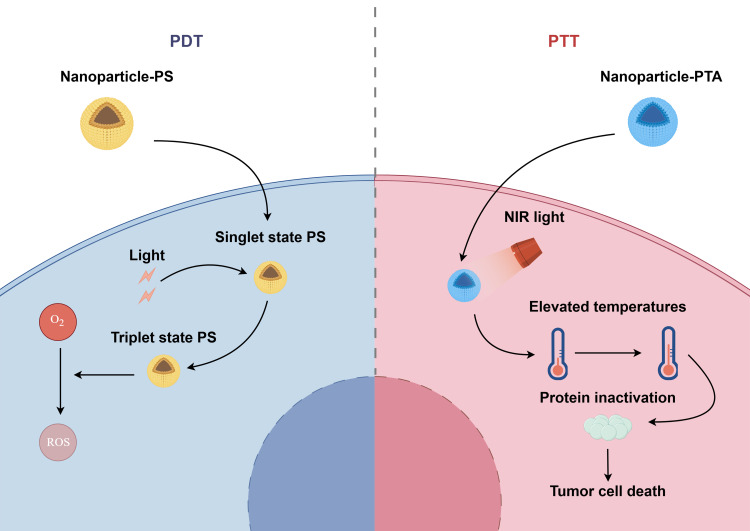
Phototherapy represents a novel non-invasive tumor treatment strategy, encompassing photodynamic therapy (PDT) and photothermal therapy (PTT).118 In comparison to conventional therapeutic modalities, such as surgical intervention and chemotherapy, phototherapy is less invasive and less toxic, and is therefore a frequently employed treatment for bladder cancer. During PDT, in the presence of suitable light, photosensitizers (PS) accumulate at the tumor site, relying on oxygen to produce cytotoxic reactive oxygen species (ROS).119 The precise mechanism involves the absorption of light by the PS, which then undergoes a transition from the ground state (singlet state) to the electronically excited state (triplet state). In this excited state, the PS reacts with oxygen to form ROS.120 PDT has been demonstrated to possess anti-tumor properties, exerting its effects through a number of mechanisms. Direct toxicity to tumor cells, leading to apoptosis and necrosis, represents one such mechanism. Additionally, PDT has been shown to destroy blood vessels in the tumor site, thereby affecting the blood supply to the tumor and ultimately blocking the supply of oxygen and nutrients. Furthermore, PDT has been observed to induce inflammatory reactions in the tumor site and activate anti-tumor immunity.121 The therapeutic efficacy of PDT is closely associated with the delivery of PS.122 Presently, numerous photosensitizers exhibit shortcomings, including low light conversion efficiency, inadequate bioavailability, and insufficient selectivity.123 However, nanoparticle delivery of PS can enhance solubility of PS in tumor regions and stability in vivo. Additionally, it can augment targeting ability of PS to tumor sites, thereby potentiating the efficacy of PDT. PTT employs photothermal agents (PTAs) to transform light heat into high heat under laser irradiation conditions, thereby modifying the permeability of the cell membrane of tumor cells. This results in the inactivation of intracellular proteins, ultimately leading to the death of tumor cells.124,125 The therapeutic efficacy of PTT is contingent upon the photothermal conversion efficiency (PTCE) and the targeting delivery efficiency of PTAs.126 The use of nanoparticles can facilitate the distribution of PTAs within the tumor area, thereby enhancing the PTCE and consequently increasing the photothermal therapeutic effect.127,128 This paragraph focuses on the application of nanoparticles in delivering PS and PTA (Figure 3).
Figure 3.
Nanoparticles in delivering PS and PTA. Once the photosensitizers (PS) and photothermal agent (PTA) have been delivered by nanoparticles to the tumor area, PS is irradiated by light, resulting in a transition from the ground state to the activated state. This process converts oxygen to reactive oxygen species (ROS). Concurrently, PTA is irradiated by near-infrared (NIR) light, whereby light energy is converted to heat, thereby increasing the temperature of the tumor area. This leads to protein inactivation and tumor cell death. By figdraw.com.
Nanoparticles as Drug Delivery Systems in Photodynamic Therapy
The initial generation of photosensitizers employed in phototherapy comprised small molecule photosensitizers, exemplified by hematoporphyrin derivatives.129–131 Chlorin e6 (Ce6) is a second-generation photosensitizer that has been approved by the FDA.132 It is a chlorophyll a derivative that has been demonstrated to generate ROS with high efficiency and to exert a broad spectrum of anticancer effects.133 The combination of the conventional antiparasitic drug nitazoxanide (NTZ) with CE6-conjugated human serum albumin (HSA) was found to self-assemble into HSA-Ce6/NTZ nanoparticles. Subsequently, the HSA-Ce6/NTZ nanoparticles were combined with synthetic fluorinated chitosan (FCS) to construct a biocompatible nanosystem, designated as HSA-Ce6/NTZ/FCS nanoparticles. This nanoparticles not only facilitates the transmucosal delivery of drugs but also enhances their stability and efficacy within biological systems. Furthermore, NTZ was observed to significantly alleviate tumor hypoxia by regulating the metabolism of tumor cells, which is crucial for enhancing the efficacy of PDT. Consequently, the efficacy of treating in situ bladder tumors can be markedly enhanced by intravesical infusion of biocompatible HSA-Ce6/NTZ/FCS nanoparticles in conjunction with cystoscopy-guided PDT treatment.22
Meanwhile, an innovative nano drug delivery system has been developed that employs tumor-specific ligands to facilitate the precise delivery of the photosensitizer Ce6 to bladder tumor cells. Furthermore, the therapeutic effect of PDT is enhanced by the addition of catalase to the drug complex, which increases the oxygen concentration in the tumor tissue. In comparison to using Ce6 alone, the aptamer-guided DNA nanotrain (NT) carrying Ce6 and catalase (NT-Catalase-Ce6) demonstrated enhanced efficacy in recognizing and targeting bladder cancer cells. This complex has been demonstrated to increase oxygen production at the local level, while also triggering the generation of ROS in tumor cells. This, in turn, has been shown to trigger mitochondrial apoptosis. In an in situ model study of bladder cancer in mice, it was demonstrated that NT-Catalase-Ce6, when administered via intravesical instillation, exhibited enhanced drug uptake in tumor tissues and prolonged drug retention, thereby exhibiting notable superiority over normal urinary epithelium. Furthermore, PDT utilizing nanoparticles demonstrated efficacy in impeding tumor growth and exhibited a markedly reduced incidence of adverse effects (eg, cystitis) in comparison to conventional therapeutic modalities.23
Mesoporous silica nanoparticles represent an excellent vehicle for drug delivery, offering versatility for a range of therapeutic modalities.134 The objective of this study was to investigate the influence of the physicochemical properties of mesoporous silica nanoparticles on the effect of PDT. For this purpose, mesoporous silica nanoparticles (MSNPs) and mesoporous silica nanorods (MSNRs) of different shapes were prepared and their properties were experimentally demonstrated as new third-generation PS for PDT against two BC cell lines, HT-1376 and UMUC-3.The findings revealed that MSNPs not only generated a greater quantity of singlet oxygen but also demonstrated heightened phototoxicity in both cancer cell lines relative to MSNRs. Moreover, spherical MSNPs exhibited enhanced potential as nanocarriers in PDT.24
Nanoparticles as Drug Delivery Systems in Photothermal Therapy
Gold nanorods (GNRs) possess a relatively ideal size, excellent near-infrared (NIR) light absorption,135 photothermal conversion,136,137 and strong tunable extinction in the NIR region (600–1100 nm), which is also known as the biological window.138 These characteristics render them suitable for use as a PTA for PTT therapy.
The second near-infrared window (NIR-II) has a wavelength range of approximately 1000 nm to 1700 nm.139 It is postulated that this window has greater tissue penetration than the first near-infrared (NIR-I) window, and thus the development of photothermal agents targeting this region has been the focus of recent research.140,141 Hollow gold nanorods (AuHNRs) exhibit an intense, strong localized surface plasmon resonance peak in the second near-infrared (NIR-II) window, which endows the nanoparticles with a robust photothermal conversion capability. A hollow AuHNR surface coated with MnO2 and chitosan(CS) nanoparticles, AuHNR@MnO2@CS (AuMC), was observed to trigger the degradation of the MnO2 layer. This was found to be due to the overexpression of GSH in the tumor microenvironment (TME), which resulted in the release of Mn2+, thereby increasing the PTCE and consequently the PTT effect.25
A gold-silicon oxide-gold nanoparticle (Au@SiO2@Au) was modified by the attachment of hyaluronic acid (HA) to the surface of the nanoparticle, forming HA-Au@SiO2@Au. This was utilized to target the CD44 receptor, which is overexpressed on the surface of tumor cells. The attachment of the nanoparticle to the receptor was achieved through the use of 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride, which enhanced the accumulation of the nanoparticle in the tumor tissue. The nanoparticles demonstrated high photothermal conversion efficiency (47.6%) and excellent photostability. In the in vivo experiments, tumor-bearing mice were randomly assigned to one of four groups and subjected to different treatments. The results demonstrated that mice treated with HA-Au@SiO2@Au nanoparticles and combined with near-infrared laser irradiation exhibited a gradual reduction in tumor volume and complete regression of prostate tumors and bladder tumors, with no recurrence at 15 and 18 days post-treatment, respectively. The aforementioned research evidence indicates that HA-Au@SiO2@Au nanoparticles may be employed as an efficacious PTA for the minimally invasive treatment of genitourinary tumors.137
Gold nanoparticles have the potential to be used in conjunction with PDT and PTT, and they offer advantages in improving the imaging efficiency of surface-enhanced Raman spectroscopy (SERS).142 SERS is a highly sensitive analytical technique that significantly enhances the Raman scattering signal of a target molecule by adsorbing the molecule to be tested, such as urine, on the surface of gold or silver nanoparticles. This technique not only markedly enhances the signal-to-noise ratio, but also enables the detection of exceedingly low concentrations of biomolecules due to its high sensitivity and specificity.143,144 As a novel phototherapeutic agent, methylene blue-loaded mesoporous silica-coated gold nanorods on graphene oxide (MB-GNR@mSiO2-GO) demonstrated favorable biocompatibility and high efficiency of photothermal conversion in in vitro experiments. The PTCE of MB-GNR@mSiO2-GO was 48.93%, and the combination of PTT and PDT demonstrated a notable synergistic effect. The particles demonstrated the capacity to promote apoptosis and damage mitochondria and nuclei when irradiated with a 785 nm laser, indicating that they possess robust ROS generation and effective SERS imaging abilities.145 While mesoporous silica-layered gold nanorod core@silver shell nanostructures loaded with methylene blue (GNR@Ag@mSiO2-MB) also possess high stability, a wide NIR absorption range, and excellent photothermal stability, the nanoparticles also have high stability, a wide NIR absorption range, and excellent photothermal stability, and can effectively kill cancer cells through the synergistic effect of PTT and PDT. Additionally, the nanoparticles demonstrate remarkable SERS activity under 785 nm NIR laser excitation, rendering them an effective contrast agent for rapid SERS imaging of cancer cells.136
Other nanoparticles are also employed in combined PDT and PTT therapy, including a triphenylphosphine-functionalized gold nanorod/zinc oxide core-shell nanocomposite (CTPP-GNR@ZnO). The nanomaterial demonstrated exceptional photothermal properties, ROS generation efficiency, extensive NIR absorption, stability, and mitochondrial targeting efficiency. In the absence of 780 nm laser irradiation, the CTPP-GNR@ZnO core-shell nanocomposite exhibited minimal cytotoxicity against CT-26 cells and demonstrated excellent biocompatibility. The CTPP-GNR@ZnO core-shell nanocomposite demonstrated effective cell ablation (95%) following 50 minutes of 780 nm NIR laser irradiation, indicating that the elevated temperature could potentially eradicate cancer cells entirely. Additionally, the CTPP-GNR@ZnO core-shell nanocomposite induced a notable accumulation of intracellular ROS after 780 nm laser irradiation, suggesting that the nanocomposite could specifically target the mitochondria of CT-26 cells. The GNR@ZnO core-shell nanocomposite has been demonstrated to possess the ability to target the mitochondria of CT-26 cells with a high degree of specificity.146
In addition to the aforementioned factors, the distribution of GNR also influences the efficacy of PTT for bladder cancer. In particular, when GNR is distributed at the edge or surface of the tumor, more uniform heating can be achieved, which reduces damage to surrounding healthy tissues. Furthermore, it is imperative that the laser power be properly adjusted in order to achieve complete tumor ablation.147 It is important to note that during PTT, thermal induction from the flow of urine may potentially impact the therapeutic effect. A study was conducted to simulate the effects of natural convection inside the bladder and on the skin surface on GNR-assisted PTT in different body positions (supine, lateral, and prone) by constructing a 3D mouse bladder model. The findings revealed that natural convection plays a complex role in PTT, assisting in the dispersion of heat from the treatment area to surrounding tissues. This process, to some extent, can reduce the unwanted thermal damage to healthy tissues, demonstrating its positive effects. However, this heat dissipation process may also diminish the thermal effect on the tumor tissue, thus reducing the therapeutic effect and demonstrating its potential negative effects.135
The Role of Nanoparticles as Drug Delivery Systems in Combination Therapy
At present, a single treatment strategy is frequently inadequate to achieve an optimal therapeutic effect in the clinical management of bladder cancer. A combination therapy approach, utilising multiple treatment modalities, has been shown to yield superior therapeutic outcomes in comparison to traditional therapeutic modalities, as evidenced by recent studies.3 At this juncture, the majority of researchers are employing nanoparticles to integrate phototherapy with other therapeutic modalities, and the results thus far have been promising, substantiating the potential of nanoparticles in the combined treatment of bladder cancer.
Nanoparticles as Drug Delivery Systems in Phototherapy in Combination with Other Therapies
Magnetic nanoparticles represent a new type of nanoparticle that can be used in combination therapy. They can be created by encapsulating superparamagnetic iron oxide nanoparticles (SPIONs) in reduced poly(β-amino ester)s-PEG amphiphilic copolymer (rPAE), which can then be used as a nanoplatform. This process is illustrated by the example of magnetic nanoparticles (rPAE@SPION). The platform exhibits a multitude of therapeutic functions. First, the platform is capable of achieving photothermal conversion when exposed to NIR, which results in mild heating of the tumor region. This local heating facilitates the release of drugs and enhances the cytotoxicity of chemotherapeutic agents on cancer cells. Secondly, it induces iron-mediated apoptosis in tumor cells. Thirdly, it stimulates the conversion of macrophages to the M1 phenotype with antitumor activity, which also promotes iron-mediated apoptosis in tumor cells. Finally, it is capable of loading DOX and can be used in combination of chemotherapy with other therapy.148 Another core-shell structured superparamagnetic HA-modified magnetic nanoparticle capable of targeting bladder tumor tissues is core-shell Zn-CoFe 2 O 4 @Zn-MnFe 2 O 4 nanoparticles. The nanoparticles could significantly inhibit bladder tumor cells by inhibiting the expression of HSP 70 through mild magnetic hyperthermia (MHT), which in turn inhibited the downstream expression of cyclin D1 and Bcl-2. The combination of MHT and immunotherapy was realized and showed significant therapeutic effects.149
Nanoporphyrins can be used in PDT and PTT.150,151 Nanoporphyrin particles have the potential to enhance the effects of PDT while simultaneously combining PDT with PTT. A J-aggregated nanoporphyrin (MTE) was constructed based on the self-assembly of a demagnesium chlorophyllate methyl ester derivative, MPa-TEG (MT), and a natural polyphenolic compound, epigallocatechin gallate (EGCG). MTE effectively eliminates excess ROS, thereby reducing phototoxic side effects that may occur during PS transport. Furthermore, it enhances the effect of PDT through two mechanisms: First, MTE enhances the effect of PDT by decreasing the expression of the anti-apoptotic protein Survivin. Second, MTE enhances the effect of PDT by inhibiting the overexpression of heat shock protein 70 (HSP 70) and heat shock protein 90 (HSP 90), which reduces the tumor’s resistance to heat, thus assisting in the realization of mild-temperature laser interstitial thermal therapy (LITT).152
Nanoporphyrins also show promise as a potential treatment for photoimmunotherapy.151 PLZ4-nanoporphyrin (PNP) is a porphyrin-based nanoscale micelle modified with PLZ4 on its surface, rendering it capable of cancer-specific targeting. The combination of this nanoparticle with anti-programmed death protein-1 (anti-PD-1) antibodies demonstrated a synergistic effect in the treatment of locally advanced and metastatic bladder cancer when used in photodynamic therapy. This resulted in an intensification of the anti-tumor effect and an improvement in survival. This enhancement was achieved by inducing immunogenic cell death, promoting dendritic cell maturation, attracting immune cell infiltration, and improving the tumor immune microenvironment.153 Similarly, a porphyrin nanomaterial (P18-APBA-HA) constructed from the photosensitizer purpurin 18 (P18), hyaluronic acid (HA), and 4-(aminomethyl)phenylboronic acid (APBA) has the potential to be employed in photoimmunotherapy. The nanomaterial was inserted into a phospholipid membrane, and the indoleamine-2,3-dioxygenase (IDO) inhibitor epacadostat (EPA) was simultaneously loaded into the inner phase, thereby constructing a dual drug delivery system (Lip\EPA\P18-APBA-HA). Lip\EPA\P18-APBA-HA releases EPA upon exposure to thermal radiation, which inhibits IDO activity in the tumor microenvironment and promotes activation of the immune response. Intravenous injection of Lip\EPA\P18-APBA-HA effectively induces anti-tumor immunity by promoting dendritic cell maturation, cytotoxic T-cell activation and regulatory T-cell suppression, as well as regulating cytokine secretion, thereby inhibiting melanoma proliferation.154 Although the experiment did not use a bladder cancer model as the subject, the results nonetheless demonstrate that nanoporphyrin particles can enhance the therapeutic effect in cancer photoimmunotherapy. Furthermore, the results highlight the necessity for further research on nanoporphyrin particles in bladder cancer photoimmunotherapy.
Nanoporphyrins have demonstrated potential in a range of applications, including photoimmunotherapy and the combination of photodynamic therapy and chemotherapy. Lin, T.Y. et al employed PLZ4-nanoporphyrin (PNP) to construct a nanoplatform that integrates phototherapy, photothermal therapy, and targeted chemotherapy. They tested this platform in a mouse xenograft (PDX) model of bladder cancer and found that the PNPs were considerably more effective than 5-aminolevulinic acid for PDT, eliminating the need for intravesical treatment in the bladder of the in situ PDX bladder cancer. The combination of image-guided PDT and PTT with doxorubicin -targeted chemotherapy resulted in a notable prolongation of overall survival in mice with bladder cancer xenografts. This was accompanied by a slower release of the drugs, a longer body circulation time, and a greater reduction in tumor cell viability when DOX was loaded onto the nanoparticles.155
Phytochemicals also have applications in combination therapy. Cluster-structured nanoparticles (CNP) denoted Fe₃O₄@Chl/Fe CNP have been assembled from Fe₃O₄ and iron chlorophyll (Chl/Fe) photosensitizers after 4-carboxyphenylboronic acid modification. These nanoparticles have been shown to target glycoproteins on the surface of bladder cancer cells, thereby enhancing the internalization into bladder cancer cells. Th nanoparticles eradicated bladder cancer through a combination of three therapeutic modalities: PDT-induced singlet oxygen, photochemotherapy-initiated ferroptosis and immunostimulation. This resulted in a significant enhancement of the therapeutic efficacy of bladder cancer treatment, as evidenced by an increase in the survival rate of in situ MB 49 tumor-bearing mice from 0% to 91.7%.156
Nanoparticles as Drug Delivery Systems in Combination of Chemotherapy and Immunotherapy
A co-delivered chitosan (CS) carrier system, LRO-BCG/CS with OXA prodrug liposomes and BCG, has the potential to be employed in combination chemotherapy and immunotherapy.157 This nanosystem incorporates GSH-responsive lipophilic oxaliplatin (OXA) prodrug (octadecyl-OXA-carboxylic acid, OOC) into cationic liposomes (denoted as LRO), which contain a stearyl cell-penetrating peptide C18-R8H3. CS has been demonstrated to promote the retention of LRO and BCG in the bladder for periods exceeding 24 hours, while simultaneously enhancing the permeability of LRO. LRO has been observed to respond to reducing agents present within tumor cells, resulting in the release of OXA and the subsequent triggering of immunogenic cell death. The combination of BCG has been shown to efficiently promote the induction of immunogenic cell death. LRO-BCG/CS has been demonstrated to reverse the immunosuppressed tumor microenvironment, thereby activating the tumor-specific immune response against tumor cell proliferation. Consequently, the combination of LRO-BCG/CS demonstrated a favorable inhibitory effect on in situ bladder cancer in mice, thereby substantiating its potential clinical application value.157 At this juncture, nanomedicines for combination therapy are predominantly utilized in conjunction with phototherapy and immunotherapy, whereas research on alternative combination modalities remains limited. The potential of nanoparticles as an optimal drug delivery system in combination therapy is a promising avenue for further investigation, particularly in the context of exploring the integration of additional therapeutic modalities.
The Toxicity of Nanoparticles and the Challenges They Present for Clinical Applications
Despite the advantages demonstrated by nanoparticles as drug delivery systems, the translation of these systems into clinical applications still presents considerable challenges. Indeed, only a small number of nano-formulations have entered clinical trials (Table 2). At this juncture, research has concentrated on the capacity of nanoparticles to mitigate the toxicity of the drugs they are encapsulated with. However, there has been a paucity of attention directed towards the toxicity of the nanoparticles themselves, particularly with regard to local or systemic toxicity following treatment with nanoparticles. For instance, cationic nanoparticles of gold and polystyrene have been demonstrated to induce haemolysis and blood coagulation, while certain concentrations of anionic nanoparticles and cationic nanoparticles have been identified as toxic to the blood-brain barrier,158 The toxicity of silica nanoparticles has been demonstrated to affect a number of biological systems, including the respiratory, nervous, digestive and circulatory systems.159
Table 2.
Clinical Trials of Nano Drug Delivery Systems for Bladder Cancer
| Nanoparticles | Drugs | Conditions | Sponsor | Status | NCT Number |
|---|---|---|---|---|---|
| Nanoparticle Cisplatin | Gemcitabine | BC | NanoCarrier Co., Ltd. | Completed | NCT02240238 |
| Nanoparticle albumin | Rapamycin | Advanced Malignant Neoplasm | Mayo Clinic | Completed | NCT02646319 |
| Paclitaxel Albumin-Stabilized Nanoparticle | Paclitaxel | High-Risk BC | Mayo Clinic | Withdrawn | NCT02718742 |
| PLZ4-coated paclitaxel-loaded micelle | Paclitaxel | Recurrent or Refractory NMIBC | Mamta Parikh, University of California, Davis | Recruiting | NCT06173349 |
| Albumin-bound Rapamycin Nanoparticles | Rapamycin | NMIBC | Aadi Bioscience, Inc. | Completed | NCT02009332 |
| Albumin-Bound Nanoparticles | Paclitaxel | BC | Columbia University | Unknown status | NCT00583349 |
| Nanoparticles & Metallic Compounds | – | BC | Carmel Medical Center | Completed | NCT02811952 |
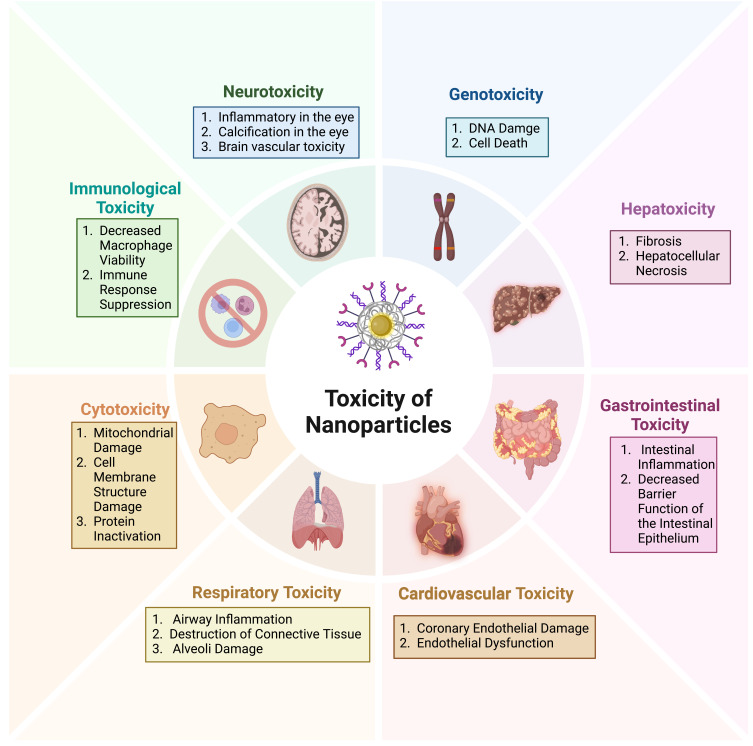
The enhanced reactivity of nanoparticles, attributable to their diminutive size and extensive surface area, not only augments drug delivery efficiency but also intensifies the interaction of nanoparticles with cells, thereby elevating the toxicity of nanoparticles.160 The physical properties, concentration, and in vivo biodistribution and clearance of nanoparticles exert a modulating influence on cellular endocytosis and nanoparticle toxicity.161,162 The solubility of nanoparticles is a pivotal property that influences the acute toxicity of nanoparticles. The free ions released from the intracellular solubility of specific metal oxide nanoparticles (eg, zinc oxide and iron oxide) are linked to their toxicity. Furthermore, given that the utilisation of nanoparticles for therapeutic purposes is a prolonged process, the chronic toxicity of nanoparticles also warrants consideration. Prolonged exposure to nanoparticles has been demonstrated to induce genotoxicity, as evidenced by the promotion of breast cancer metastasis following prolonged exposure to multi-walled carbon nanotubes,163 and the increase in the number of ROS cells resulting in DNA damage observed in the presence of titanium dioxide nanoparticles.164 Nevertheless, the chronic toxicity of nanoparticles remains poorly understood, and further experimentation is required to validate their long-term toxicity before they can be safely applied in clinical settings. Nanoparticles have also been demonstrated to possess cytotoxic properties. In particular, single-walled and multi-walled carbon nanotubes have been shown to exhibit cytotoxicity at elevated concentrations. Similarly, quantum dots have been observed to elicit a cytotoxic response when not surface-coated, resulting in the release of toxic ions within the body.165 Nanoparticles have also been demonstrated to induce immunotoxicity. For example, carbon nanotubes have been shown to reduce macrophage viability, while gold nanoparticles have been observed to inhibit the immune response.166(Figure 4).
Figure 4.
The toxicity of nanoparticles represents a significant challenge in the field of nanoparticle research. The potential for nanoparticles to cause adverse effects on various body systems, including gastrointestinal, hepatotoxic, cardiovascular, neurotoxic, and more serious forms of cytotoxicity, immunotoxicity, and genotoxicity, hinders the development of nanoparticles for clinical applications. Created in BioRender. ZHAO, X. (2024) https://BioRender.com/d91p004.
It would be remiss not to consider the potential of nanoparticles in the treatment of tumor metastases. At this juncture, studies on nanoparticles in the treatment of bladder cancer have largely been confined to the primary tumor. While some studies have employed cell line-derived xenograft (CDX) tumors and patient-derived xenograft (PDX) models, these do not fully reflect the characteristics of tumor metastases observed in clinical cases. Additionally, the location of tumor metastases is not fixed in clinical cases, whereas the method of tumor implantation in studies is generally rather homogeneous.167,168 Translational research represents a pivotal stage in the process of translating fundamental research findings into clinical practice. This is particularly relevant in the context of nanoparticles, yet the translational research process is inherently lengthy and contingent upon numerous factors. Consequently, only a small proportion of basic research findings ultimately result in the development of clinical applications. First in human (FIH) trials represent a crucial stage in the translational research process. However, in a field as specialized as nanoparticles, where different nanomaterials exhibit distinct mechanisms and target sites, and where the funding and ethical approvals required for human trials are substantial, it is unlikely that the initial experimental process for nanoparticles will benefit the subjects, making the implementation of FIH trials challenging. The implementation of FIH trials is a challenging process.169 Concurrently, in order to facilitate the utilisation of nanoparticles in clinical contexts, the preparation of nanoparticles necessitates the adherence to pre-established standards of quality, controls and good manufacturing practice.168 The financial implications of manufacturing nanoparticles cannot be overlooked. The probability of successfully translating a small molecule drug from the preclinical proof-of-concept stage to a commercially marketed product is approximately 6%,170 and even when the risks of development are not taken into account, the complex formulations of nanoparticles and the lengthy process of their preparation are costly to produce. Therefore, it is essential to consider the cost-effectiveness of nanoparticles in relation to cheaper drug therapies.169,171
Conclusions
In recent years, nanoparticles have been the subject of extensive research as an emerging material, with a particular focus on their potential applications in drug delivery systems. Despite the existence of well-established therapeutic modalities for low-grade bladder cancer, non-surgical therapies for patients with high-grade and advanced bladder cancer have not demonstrated comparable efficacy. Nanoparticles represent a novel approach to enhancing the efficacy of non-surgical treatments for bladder cancer. Nanoparticles enhance the efficacy of drug delivery through their intrinsic high porosity, facilitate targeted delivery, and mitigate the adverse effects associated with pharmaceutical agents. Nanoparticles represent a novel material that can facilitate the delivery of a range of therapeutic agents, including chemotherapeutic drugs such as CDDP, DOX, and PTX in chemotherapy; siRNA and circRNA in gene therapy; BCG, immune checkpoint inhibitors, and vaccine adjuvants in immunotherapy; and PS and PTA, in phototherapy.
Despite the encouraging outcomes that have been attained, the challenge of translating nanoparticles into clinical applications persists. Nanoparticles are manufactured using a diverse range of materials, and the toxicity of nanoparticles, drug clearance after delivery, and the genotoxicity, immunotoxicity, and cytotoxicity of nanoparticles after treatment require further experimental verification. In addition to the inherent disadvantages of nanoparticles, the preparation of nanoparticles for clinical treatment in standard, large quantities and at low cost represents a significant challenge to their application in the clinic. Notwithstanding the aforementioned challenges, it is irrefutable that nanoparticles as drug delivery systems markedly enhance the therapeutic efficacy of non-surgical treatment modalities for bladder cancer, while also offering a novel avenue for research into non-surgical treatments for bladder cancer.
Funding Statement
Liaoning Provincial Education Department (No. JYTMS20230577);Dalian Life and Health Guidance Program Project.
Author Contributions
All authors have made major contributions to this work; contributed to drafting, revising, and critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Disclosure
The authors declare no competing interests in this work.
References
- 1.Bray F, Laversanne M, Sung H, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2024;74(3):229–263. doi: 10.3322/caac.21834 [DOI] [PubMed] [Google Scholar]
- 2.Babjuk M, Burger M, Capoun O, et al. European Association of urology guidelines on non-muscle-invasive bladder cancer (Ta, T1, and Carcinoma in Situ). Eur Urol. 2022;81(1):75–94. doi: 10.1016/j.eururo.2021.08.010 [DOI] [PubMed] [Google Scholar]
- 3.Zhang C, Zhao J, Wang W, et al. Current advances in the application of nanomedicine in bladder cancer. Biomed Pharmacother. 2023;157:114062. doi: 10.1016/j.biopha.2022.114062 [DOI] [PubMed] [Google Scholar]
- 4.Song FX, Xu X, Ding H, et al. Recent progress in nanomaterial-based biosensors and theranostic nanomedicine for bladder cancer. Biosensors. 2023;13(1):106. doi: 10.3390/bios13010106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jin YH, Zeng X-T, Liu T-Z, et al. Treatment and surveillance for non-muscle-invasive bladder cancer: a clinical practice guideline (2021 edition). Mil Med Res. 2022;9(1):44. doi: 10.1186/s40779-022-00406-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Patel VG, Oh WK, Galsky MD. Treatment of muscle-invasive and advanced bladder cancer in 2020. CA Cancer J Clin. 2020;70(5):404–423. doi: 10.3322/caac.21631 [DOI] [PubMed] [Google Scholar]
- 7.Zupancic D, Veranic P. Nanodiamonds as possible tools for improved management of bladder cancer and bacterial cystitis. Int J Mol Sci. 2022;23(15):8183. doi: 10.3390/ijms23158183 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ali MS, Metwally AA, Fahmy RH, et al. Chitosan-coated nanodiamonds: mucoadhesive platform for intravesical delivery of doxorubicin. Carbohydr Polym. 2020;245:116528. doi: 10.1016/j.carbpol.2020.116528 [DOI] [PubMed] [Google Scholar]
- 9.Li G, Tao T, Deng D, et al. Collagen-targeted tumor-specific transepithelial penetration enhancer mediated intravesical chemoimmunotherapy for non-muscle-invasive bladder cancer. Biomaterials. 2022;283:121422. [DOI] [PubMed] [Google Scholar]
- 10.Wang Z, Li W, Jiang Y, et al. Cholesterol-modified sphingomyelin chimeric lipid bilayer for improved therapeutic delivery. Nat Commun. 2024;15(1):2073. doi: 10.1038/s41467-024-46331-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu Z, Liu Y, Wu Z, et al. Research on the loading and release kinetics of the vincristine sulfate liposomes and its anti-breast cancer activity. Int J Pharm X. 2024;7:100258. doi: 10.1016/j.ijpx.2024.100258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kates M, Date A, Yoshida T, et al. Preclinical evaluation of intravesical cisplatin nanoparticles for non–muscle-invasive bladder cancer. Clin Cancer Res. 2017;23(21):6592–6601. doi: 10.1158/1078-0432.CCR-17-1082 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang W, Yang F, Zhang L, et al. Targeting DNA damage and repair machinery via delivering WEE1 Inhibitor and Platinum (IV) prodrugs to stimulate STING pathway for maximizing chemo-immunotherapy in bladder cancer. Adv Mater. 2024;36(1):e2308762. doi: 10.1002/adma.202308762 [DOI] [PubMed] [Google Scholar]
- 14.Guo S, Li K, Hu B, et al. Membrane-destabilizing ionizable lipid empowered imaging-guided siRNA delivery and cancer treatment. Exploration. 2021;1(1):35–49. doi: 10.1002/EXP.20210008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shahidi M, Abazari O, Dayati P, et al. Multicomponent siRNA/miRNA-loaded modified mesoporous silica nanoparticles targeted bladder cancer for a highly effective combination therapy. Front Bioeng Biotechnol. 2022;10:949704. doi: 10.3389/fbioe.2022.949704 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 16.Yu C, Zhang Y, Wang N, et al. Treatment of bladder cancer by geoinspired synthetic chrysotile nanocarrier-delivered circPRMT5 siRNA. Biomater Res. 2022;26(1):6. doi: 10.1186/s40824-022-00251-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zhou Q, Fang L, Tang Y, et al. Exosome-mediated delivery of artificial circular RNAs for gene therapy of bladder cancer. J Cancer. 2024;15(6):1770–1778. doi: 10.7150/jca.90620 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shi X, Pang S, Zhou J, et al. Bladder-cancer-derived exosomal circRNA_0013936 promotes suppressive immunity by up-regulating fatty acid transporter protein 2 and down-regulating receptor-interacting protein kinase 3 in PMN-MDSCs. Mol Cancer. 2024;23(1):52. doi: 10.1186/s12943-024-01968-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guo P, Dai P, Yang S, et al. Engineered macrophages tune intratumoral cytokines through precisely controlled self-pyroptosis to enhance bladder cancer immunotherapy. Small. 2024;20(13):e2306699. doi: 10.1002/smll.202306699 [DOI] [PubMed] [Google Scholar]
- 20.Zhou Q, Ding W, Qian Z, et al. Immunotherapy strategy targeting programmed cell death Ligand 1 and CD73 with macrophage-derived mimetic nanovesicles to treat bladder cancer. Mol Pharm. 2021;18(11):4015–4028. doi: 10.1021/acs.molpharmaceut.1c00448 [DOI] [PubMed] [Google Scholar]
- 21.Kim H, Niu L, Larson P, et al. Polymeric nanoparticles encapsulating novel TLR7/8 agonists as immunostimulatory adjuvants for enhanced cancer immunotherapy. Biomaterials. 2018;164:38–53. doi: 10.1016/j.biomaterials.2018.02.034 [DOI] [PubMed] [Google Scholar]
- 22.Wang S, Jin S, Li G, et al. Transmucosal delivery of self-assembling photosensitizer-nitazoxanide nanocomplexes with fluorinated chitosan for instillation-based photodynamic therapy of orthotopic bladder tumors. ACS Biomater Sci Eng. 2021;7(4):1485–1495. doi: 10.1021/acsbiomaterials.0c01786 [DOI] [PubMed] [Google Scholar]
- 23.Zhang Y, Jia R, Wang X, et al. Targeted delivery of catalase and photosensitizer Ce6 by a tumor-specific aptamer is effective against bladder cancer in vivo. Mol Pharm. 2024;21(4):1705–1718. doi: 10.1021/acs.molpharmaceut.3c01047 [DOI] [PubMed] [Google Scholar]
- 24.Borzęcka W, Pereira PMR, Fernandes R, et al. Spherical and rod shaped mesoporous silica nanoparticles for cancer-targeted and photosensitizer delivery in photodynamic therapy. J Mater Chem B. 2022;10(17):3248–3259. doi: 10.1039/D1TB02299G [DOI] [PubMed] [Google Scholar]
- 25.Guo B, Yang F, Zhang L, et al. Cuproptosis induced by ROS responsive nanoparticles with elesclomol and copper combined with alphaPD-L1 for enhanced cancer immunotherapy. Adv Mater. 2023;35(22):e2212267. doi: 10.1002/adma.202212267 [DOI] [PubMed] [Google Scholar]
- 26.Golombek SK, May J-N, Theek B, et al. Tumor targeting via EPR: strategies to enhance patient responses. Adv Drug Deliv Rev. 2018;130:17–38. doi: 10.1016/j.addr.2018.07.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Handa M, Beg S, Shukla R, et al. Recent advances in lipid-engineered multifunctional nanophytomedicines for cancer targeting. J Control Release. 2021;340:48–59. doi: 10.1016/j.jconrel.2021.10.025 [DOI] [PubMed] [Google Scholar]
- 28.Ashrafizadeh M, Zarrabi A, Karimi‐Maleh H, et al. (Nano)platforms in bladder cancer therapy: challenges and opportunities. Bioeng Transl Med. 2023;8(1):e10353. doi: 10.1002/btm2.10353 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Amreddy N, Babu A, Muralidharan R, et al. Recent advances in nanoparticle-based cancer drug and gene delivery. Adv Cancer Res. 2018;137:115–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tomlinson B, Lin T-Y, Dall’Era M, et al. Nanotechnology in bladder cancer: current state of development and clinical practice. Nanomedicine. 2015;10(7):1189–1201. doi: 10.2217/nnm.14.212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fu S, Li G, Zang W, et al. Pure drug nano-assemblies: a facile carrier-free nanoplatform for efficient cancer therapy. Acta Pharm Sin B. 2022;12(1):92–106. doi: 10.1016/j.apsb.2021.08.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Buss JH, Begnini KR, Bender CB, et al. Nano-BCG: a promising delivery system for treatment of human bladder cancer. Front Pharmacol. 2017;8:977. doi: 10.3389/fphar.2017.00977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Fan D, Cao Y, Cao M, et al. Nanomedicine in cancer therapy. Signal Transduct Target Ther. 2023;8(1):293. doi: 10.1038/s41392-023-01536-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bertrand N, Wu J, Xu X, et al. Cancer nanotechnology: the impact of passive and active targeting in the era of modern cancer biology. Adv Drug Deliv Rev. 2014;66:2–25. doi: 10.1016/j.addr.2013.11.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jain P, Kathuria H, Momin M. Clinical therapies and nano drug delivery systems for urinary bladder cancer. Pharmacol Ther. 2021;226:107871. doi: 10.1016/j.pharmthera.2021.107871 [DOI] [PubMed] [Google Scholar]
- 36.Ismaili N, Amzerin M, Flechon A. Chemotherapy in advanced bladder cancer: current status and future. J Hematol Oncol. 2011;4:35. doi: 10.1186/1756-8722-4-35 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wang J, Zhu M, Nie G. Biomembrane-based nanostructures for cancer targeting and therapy: from synthetic liposomes to natural biomembranes and membrane-vesicles. Adv Drug Deliv Rev. 2021;178:113974. doi: 10.1016/j.addr.2021.113974 [DOI] [PubMed] [Google Scholar]
- 38.Sudha T, Bharali DJ, Yalcin M, et al. Targeted delivery of cisplatin to tumor xenografts via the nanoparticle component of nano-diamino-tetrac. Nanomedicine. 2017;12(3):195–205. doi: 10.2217/nnm-2016-0315 [DOI] [PubMed] [Google Scholar]
- 39.Lammers T, Kiessling F, Ashford M, et al. Cancer nanomedicine: is targeting our target? Nat Rev Mater. 2016;1(9). doi: 10.1038/natrevmats.2016.69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mochalin VN, Shenderova O, Ho D, et al. The properties and applications of nanodiamonds. Nat Nanotechnol. 2011;7(1):11–23. doi: 10.1038/nnano.2011.209 [DOI] [PubMed] [Google Scholar]
- 41.Schrand AM, Huang H, Carlson C, et al. Are diamond nanoparticles cytotoxic? J Phys Chem B. 2007;111(1):2–7. doi: 10.1021/jp066387v [DOI] [PubMed] [Google Scholar]
- 42.Luo P, Zheng L, Zou J, et al. Insights into vitamin A in bladder cancer, lack of attention to gut microbiota? Front Immunol. 2023;14:1252616. doi: 10.3389/fimmu.2023.1252616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Tratnjek L, Jeruc J, Romih R, et al. Vitamin A and retinoids in bladder cancer chemoprevention and treatment: a narrative review of current evidence, challenges and future prospects. Int J Mol Sci. 2021;22(7):3510. doi: 10.3390/ijms22073510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hotaling JM, Wright JL, Pocobelli G, et al. Long-term use of supplemental vitamins and minerals does not reduce the risk of urothelial cell carcinoma of the bladder in the VITamins And Lifestyle study. J Urol. 2011;185(4):1210–1215. doi: 10.1016/j.juro.2010.11.081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Holick CN, De Vivo I, Feskanich D, et al. Intake of fruits and vegetables, carotenoids, folate, and vitamins A, C, E and risk of bladder cancer among women (United States). Cancer Causes Control. 2005;16(10):1135–1145. doi: 10.1007/s10552-005-0337-z [DOI] [PubMed] [Google Scholar]
- 46.Ferreira R, Napoli J, Enver T, et al. Advances and challenges in retinoid delivery systems in regenerative and therapeutic medicine. Nat Commun. 2020;11(1):4265. doi: 10.1038/s41467-020-18042-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Yang J, Liu B, Wang Q, et al. Carboxylated mesoporous silica nanoparticle-nucleic acid chimera conjugate-assisted delivery of siRNA and doxorubicin effectively treat drug-resistant bladder cancer. Biomed Pharmacother. 2024;178:117185. doi: 10.1016/j.biopha.2024.117185 [DOI] [PubMed] [Google Scholar]
- 48.Zupancic D, Kreft ME, Grdadolnik M, et al. Detonation nanodiamonds are promising nontoxic delivery system for urothelial cells. Protoplasma. 2018;255(1):419–423. doi: 10.1007/s00709-017-1146-4 [DOI] [PubMed] [Google Scholar]
- 49.Patil S, Mishra VS, Yadav N, et al. Dendrimer-functionalized nanodiamonds as safe and efficient drug carriers for cancer therapy: nucleus penetrating nanoparticles. ACS Appl Bio Mater. 2022;5(7):3438–3451. doi: 10.1021/acsabm.2c00373 [DOI] [PubMed] [Google Scholar]
- 50.Mattioli R, Ilari A, Colotti B, et al. Doxorubicin and other anthracyclines in cancers: activity, chemoresistance and its overcoming. Mol Aspect Med. 2023;93. doi: 10.1016/j.mam.2023.101205 [DOI] [PubMed] [Google Scholar]
- 51.Harris JM, Chess RB. Effect of pegylation on pharmaceuticals. Nat Rev Drug Discov. 2003;2(3):214–221. doi: 10.1038/nrd1033 [DOI] [PubMed] [Google Scholar]
- 52.D’souza AA, Shegokar R. Polyethylene glycol (PEG): a versatile polymer for pharmaceutical applications. Expert Opin Drug Delivery. 2016;13(9):1257–1275. doi: 10.1080/17425247.2016.1182485 [DOI] [PubMed] [Google Scholar]
- 53.Pereira FM, Melo MN, Santos ÁKM, et al. Hyaluronic acid-coated chitosan nanoparticles as carrier for the enzyme/prodrug complex based on horseradish peroxidase/indole-3-acetic acid: characterization and potential therapeutic for bladder cancer cells. Enzyme Microb Technol. 2021;150:109889. doi: 10.1016/j.enzmictec.2021.109889 [DOI] [PubMed] [Google Scholar]
- 54.Xu X, Liu K, Jiao B, et al. Mucoadhesive nanoparticles based on ROS activated gambogic acid prodrug for safe and efficient intravesical instillation chemotherapy of bladder cancer. J Control Release. 2020;324:493–504. doi: 10.1016/j.jconrel.2020.03.028 [DOI] [PubMed] [Google Scholar]
- 55.Jiao B, Liu K, Gong H, et al. Bladder cancer selective chemotherapy with potent NQO1 substrate co-loaded prodrug nanoparticles. J Control Release. 2022;347:632–648. doi: 10.1016/j.jconrel.2022.05.031 [DOI] [PubMed] [Google Scholar]
- 56.Kamat AM, Hahn NM, Efstathiou JA, et al. Bladder cancer. Lancet. 2016;388(10061):2796–2810. doi: 10.1016/S0140-6736(16)30512-8 [DOI] [PubMed] [Google Scholar]
- 57.Ghosh S. Cisplatin: the first metal based anticancer drug. Bioorg Chem. 2019;88:102925. doi: 10.1016/j.bioorg.2019.102925 [DOI] [PubMed] [Google Scholar]
- 58.Trendowski MR, El Charif O, Dinh PC, et al. Genetic and modifiable risk factors contributing to cisplatin-induced toxicities. Clin Cancer Res. 2019;25(4):1147–1155. doi: 10.1158/1078-0432.CCR-18-2244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Qi L, Luo Q, Zhang Y, et al. Advances in toxicological research of the anticancer drug cisplatin. Chem Res Toxicol. 2019;32(8):1469–1486. doi: 10.1021/acs.chemrestox.9b00204 [DOI] [PubMed] [Google Scholar]
- 60.Ghelli Luserna Di Rorà A, Cerchione C, Martinelli G, et al. A WEE1 family business: regulation of mitosis, cancer progression, and therapeutic target. J hematol oncol. 2020;13(1). doi: 10.1186/s13045-020-00959-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lu Z, Yeh T-K, Tsai M, et al. Paclitaxel-loaded gelatin nanoparticles for intravesical bladder cancer therapy. Clin Cancer Res. 2004;10(22):7677–7684. doi: 10.1158/1078-0432.CCR-04-1443 [DOI] [PubMed] [Google Scholar]
- 62.Wu Q, Wong JPC, Kwok HF. Putting the brakes on tumorigenesis with natural products of plant origin: insights into the molecular mechanisms of actions and immune targets for bladder cancer treatment. Cells. 2020;9(5):1213. doi: 10.3390/cells9051213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Barani M, Hosseinikhah SM, Rahdar A, et al. Nanotechnology in bladder cancer: diagnosis and treatment. Cancers. 2021;13(9):2214. doi: 10.3390/cancers13092214 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Seca A, Pinto D. Plant secondary metabolites as anticancer agents: successes in clinical trials and therapeutic application. Int J Mol Sci. 2018;19(1):263. doi: 10.3390/ijms19010263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Xia Y, Chen R, Lu G, et al. Natural phytochemicals in bladder cancer prevention and therapy. Front Oncol. 2021;11. doi: 10.3389/fonc.2021.652033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wroński P, Wroński S, Kurant M, et al. Curcumin may prevent basement membrane disassembly by matrix metalloproteinases and progression of the bladder cancer. Nutrients. 2021;14(1):32. doi: 10.3390/nu14010032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Piwowarczyk L, Kucinska M, Tomczak S, et al. Liposomal nanoformulation as a carrier for curcumin and pEGCG-Study on stability and anticancer potential. Nanomaterials. 2022;12(8):1274. doi: 10.3390/nano12081274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Kashyap D, Tuli HS, Yerer MB, et al. Natural product-based nanoformulations for cancer therapy: opportunities and challenges. Semi Cancer Biol. 2021;69:5–23. doi: 10.1016/j.semcancer.2019.08.014 [DOI] [PubMed] [Google Scholar]
- 69.Shukla R, Singh A, Singh KK. Vincristine-based nanoformulations: a preclinical and clinical studies overview. Drug Delivery Transl Res. 2023;14(1):1–16. doi: 10.1007/s13346-023-01389-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Douer D. Efficacy and safety of vincristine sulfate liposome injection in the treatment of adult acute lymphocytic leukemia. Oncologist. 2016;21(7):840–847. doi: 10.1634/theoncologist.2015-0391 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Muller RH, Keck CM. Challenges and solutions for the delivery of biotech drugs – a review of drug nanocrystal technology and lipid nanoparticles. J Biotechnol. 2004;113(1–3):151–170. doi: 10.1016/j.jbiotec.2004.06.007 [DOI] [PubMed] [Google Scholar]
- 72.Weaver BA. How Taxol/paclitaxel kills cancer cells. Mol Biol Cell. 2014;25(18):2677–2681. doi: 10.1091/mbc.e14-04-0916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Liu Y, Wang R, Hou J, et al. Paclitaxel/Chitosan nanosupensions provide enhanced intravesical bladder cancer therapy with sustained and prolonged delivery of paclitaxel. ACS Appl Bio Mater. 2018;1(6):1992–2001. doi: 10.1021/acsabm.8b00501 [DOI] [PubMed] [Google Scholar]
- 74.Hu B, Yan Y, Tong F, et al. Lumbrokinase/paclitaxel nanoparticle complex: potential therapeutic applications in bladder cancer. Int J Nanomed. 2018;13:3625–3640. doi: 10.2147/IJN.S166438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wu T, Duan X, Hu C, et al. Synthesis and characterization of gold nanoparticles from Abies spectabilis extract and its anticancer activity on bladder cancer T24 cells. Artif Cells Nanomed Biotechnol. 2019;47(1):512–523. doi: 10.1080/21691401.2018.1560305 [DOI] [PubMed] [Google Scholar]
- 76.Kara G, Calin GA, Ozpolat B. RNAi-based therapeutics and tumor targeted delivery in cancer. Adv Drug Deliv Rev. 2022;182:114113. doi: 10.1016/j.addr.2022.114113 [DOI] [PubMed] [Google Scholar]
- 77.Zhang ZH, Wang Y, Zhang Y, et al. The function and mechanisms of action of circular RNAs in urologic cancer. Mol Cancer. 2023;22(1):61. doi: 10.1186/s12943-023-01766-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yuan Y, Gu Z, Yao C, et al. Nucleic acid-based functional nanomaterials as advanced cancer therapeutics. Small. 2019;15(26):e1900172. doi: 10.1002/smll.201900172 [DOI] [PubMed] [Google Scholar]
- 79.Halib N, Pavan N, Trombetta C, et al. An overview of siRNA delivery strategies for urological cancers. Pharmaceutics. 2022;14(4):718. doi: 10.3390/pharmaceutics14040718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.El Moukhtari SH, Garbayo E, Amundarain A, et al. Lipid nanoparticles for siRNA delivery in cancer treatment. J Control Release. 2023;361:130–146. doi: 10.1016/j.jconrel.2023.07.054 [DOI] [PubMed] [Google Scholar]
- 81.Slabáková E, Culig Z, Remšík J, et al. Alternative mechanisms of miR-34a regulation in cancer. Cell Death Dis. 2017;8(10):e3100. doi: 10.1038/cddis.2017.495 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yu G, Yao W, Xiao W, et al. MicroRNA-34a functions as an anti-metastatic microRNA and suppresses angiogenesis in bladder cancer by directly targeting CD44. J Exp Clin Cancer Res. 2014;33(1):779. doi: 10.1186/s13046-014-0115-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Chen L, Shan G. CircRNA in cancer: fundamental mechanism and clinical potential. Cancer Lett. 2021;505:49–57. doi: 10.1016/j.canlet.2021.02.004 [DOI] [PubMed] [Google Scholar]
- 84.Dong W, Bi J, Liu H, et al. Circular RNA ACVR2A suppresses bladder cancer cells proliferation and metastasis through miR-626/EYA4 axis. Mol Cancer. 2019;18(1):95. doi: 10.1186/s12943-019-1025-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Pap E, Pállinger É, Pásztói M, et al. Highlights of a new type of intercellular communication: microvesicle-based information transfer. Inflamm Res. 2009;58(1):1–8. doi: 10.1007/s00011-008-8210-7 [DOI] [PubMed] [Google Scholar]
- 86.Li J, Li Y, Li P, et al. Exosome detection via surface-enhanced Raman spectroscopy for cancer diagnosis. Acta Biomater. 2022;144:1–14. doi: 10.1016/j.actbio.2022.03.036 [DOI] [PubMed] [Google Scholar]
- 87.Yang Y, Miao L, Lu Y, et al. Exosome, the glass slipper for Cinderella of cancer-bladder cancer? J Nanobiotechnology. 2023;21(1):368. doi: 10.1186/s12951-023-02130-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Ma Y, Li S, Lin X, et al. A perspective of lipid nanoparticles for RNA delivery. Exploration. 2024. doi: 10.1002/EXP.20230147 [DOI] [Google Scholar]
- 89.Qiu H, Makarov V, Bolzenius JK, et al. KDM6A loss triggers an epigenetic switch that disrupts urothelial differentiation and drives cell proliferation in bladder cancer. Cancer Res. 2023;83(6):814–829. doi: 10.1158/0008-5472.CAN-22-1444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Kong N, Zhang R, Wu G, et al. Intravesical delivery of KDM6A -mRNA via mucoadhesive nanoparticles inhibits the metastasis of bladder cancer. Proc Natl Acad Sci U S A. 2022;119(7). doi: 10.1073/pnas.2112696119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Tian Y, Liu Z, Wang J, et al. Nanomedicine for combination urologic cancer immunotherapy. Pharmaceutics. 2023;15(2):546. doi: 10.3390/pharmaceutics15020546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.van Dorp J, van der Heijden MS. The bladder cancer immune micro-environment in the context of response to immune checkpoint inhibition. Front Immunol. 2023;14:1235884. doi: 10.3389/fimmu.2023.1235884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Liu J, Gao Y, Song C, et al. Immunotherapeutic prospects and progress in bladder cancer. J Cell Mol Med. 2024;28(5):e18101. doi: 10.1111/jcmm.18101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Morales A, Eidinger D, Bruce AW. Intracavitary Bacillus Calmette-Guerin in the treatment of superficial bladder tumors. J Urol. 2017;197(2s):S142–s145. doi: 10.1016/j.juro.2016.10.101 [DOI] [PubMed] [Google Scholar]
- 95.Rhea LP, Mendez-Marti S, Kim D, et al. Role of immunotherapy in bladder cancer. Cancer Treat Res Commun. 2021;26:100296. doi: 10.1016/j.ctarc.2020.100296 [DOI] [PubMed] [Google Scholar]
- 96.Redelman-Sidi G, Glickman MS, Bochner BH. The mechanism of action of BCG therapy for bladder cancer--A current perspective. Nat Rev Urol. 2014;11(3):153–162. doi: 10.1038/nrurol.2014.15 [DOI] [PubMed] [Google Scholar]
- 97.Mukherjee N, Julián E, Torrelles JB, et al. Effects of Mycobacterium bovis Calmette et Guérin (BCG) in oncotherapy: bladder cancer and beyond. Vaccine. 2021;39(50):7332–7340. doi: 10.1016/j.vaccine.2021.09.053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Merrill L, Gonzalez EJ, Girard BM, et al. Receptors, channels, and signalling in the urothelial sensory system in the bladder. Nat Rev Urol. 2016;13(4):193–204. doi: 10.1038/nrurol.2016.13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Zeng S, Xing S, Zhang Y, et al. Nano-Bacillus Calmette-Guérin immunotherapies for improved bladder cancer treatment. J Zhejiang Univ Sci B. 2024;25(7):557–567. doi: 10.1631/jzus.B2300392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Liu K, Peng J, Guo Y, et al. Expanding the potential of neoantigen vaccines: harnessing Bacille Calmette-Guérin cell-wall-based nanoscale adjuvants for enhanced cancer immunotherapy. ACS Nano. 2024;18(18):11910–11920. doi: 10.1021/acsnano.4c01691 [DOI] [PubMed] [Google Scholar]
- 101.Zheng X, Chen J, Deng M, et al. G3BP1 and SLU7 jointly promote immune evasion by downregulating MHC-I via PI3K/Akt activation in bladder cancer. Adv Sci. 2024;11(7):e2305922. doi: 10.1002/advs.202305922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Maiorano BA, Di Maio M, Cerbone L, et al. Significance of PD-L1 in metastatic urothelial carcinoma treated with immune checkpoint inhibitors: a systematic review and meta-analysis. JAMA Network Open. 2024;7(3):e241215. doi: 10.1001/jamanetworkopen.2024.1215 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gil-Jimenez A, van Dijk N, Vos JL, et al. Spatial relationships in the urothelial and head and neck tumor microenvironment predict response to combination immune checkpoint inhibitors. Nat Commun. 2024;15(1):2538. doi: 10.1038/s41467-024-46450-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Salmaninejad A, Valilou SF, Shabgah AG, et al. PD-1/PD-L1 pathway: basic biology and role in cancer immunotherapy. J Cell Physiol. 2019;234(10):16824–16837. doi: 10.1002/jcp.28358 [DOI] [PubMed] [Google Scholar]
- 105.Ohaegbulam KC, Assal A, Lazar-Molnar E, et al. Human cancer immunotherapy with antibodies to the PD-1 and PD-L1 pathway. Trends Mol Med. 2015;21(1):24–33. doi: 10.1016/j.molmed.2014.10.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Oh DY, Kwek SS, Raju SS, et al. Intratumoral CD4(+) T Cells mediate anti-tumor cytotoxicity in human bladder cancer. Cell. 2020;181(7):1612–1625.e13. doi: 10.1016/j.cell.2020.05.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Germanà E, Pepe L, Pizzimenti C, et al. Programmed cell death Ligand 1 (PD-L1) immunohistochemical expression in advanced urothelial bladder carcinoma: an updated review with clinical and pathological implications. Int J Mol Sci. 2024;25(12):6750. doi: 10.3390/ijms25126750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Piovesan D, Tan JBL, Becker A, et al. Targeting CD73 with AB680 (Quemliclustat), a novel and potent small-molecule CD73 inhibitor, restores immune functionality and facilitates antitumor immunity. Mol Cancer Ther. 2022;21(6):948–959. doi: 10.1158/1535-7163.MCT-21-0802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Terán-Navarro H, Zeoli A, Salines-Cuevas D, et al. Gold glyconanoparticles combined with 91-99 peptide of the bacterial toxin, Listeriolysin O, are efficient immunotherapies in experimental bladder tumors. Cancers. 2022;14(10):2413. doi: 10.3390/cancers14102413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Feng C, Tan P, Nie G, et al. Biomimetic and bioinspired nano-platforms for cancer vaccine development. Exploration. 2023;3(3):20210263. doi: 10.1002/EXP.20210263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Zhao T, Cai Y, Jiang Y, et al. Vaccine adjuvants: mechanisms and platforms. Signal Transduct Target Ther. 2023;8(1):283. doi: 10.1038/s41392-023-01557-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Kaur A, Baldwin J, Brar D, et al. Toll-like receptor (TLR) agonists as a driving force behind next-generation vaccine adjuvants and cancer therapeutics. Curr Opin Chem Biol. 2022;70:102172. doi: 10.1016/j.cbpa.2022.102172 [DOI] [PubMed] [Google Scholar]
- 113.Paston SJ, Brentville VA, Symonds P, et al. Cancer vaccines, adjuvants, and delivery systems. Front Immunol. 2021;12:627932. doi: 10.3389/fimmu.2021.627932 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kim H, Griffith TS, Panyam J. Poly(d,l-lactide-co-glycolide) nanoparticles as delivery platforms for TLR7/8 agonist-based cancer vaccine. J Pharmacol Exp Ther. 2019;370(3):715–724. doi: 10.1124/jpet.118.254953 [DOI] [PubMed] [Google Scholar]
- 115.Kim H, Khanna V, Kucaba TA, et al. TLR7/8 agonist-loaded nanoparticles augment NK cell-mediated antibody-based cancer immunotherapy. Mol Pharm. 2020;17(6):2109–2124. doi: 10.1021/acs.molpharmaceut.0c00271 [DOI] [PubMed] [Google Scholar]
- 116.Bhagchandani S, Johnson JA, Irvine DJ. Evolution of toll-like receptor 7/8 agonist therapeutics and their delivery approaches: from antiviral formulations to vaccine adjuvants. Adv Drug Deliv Rev. 2021;175:113803. doi: 10.1016/j.addr.2021.05.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Desai N, Chavda V, Singh TRR, et al. Cancer nanovaccines: nanomaterials and clinical perspectives. Small. 2024;20:e2401631. doi: 10.1002/smll.202401631 [DOI] [PubMed] [Google Scholar]
- 118.Overchuk M, Weersink RA, Wilson BC, et al. Photodynamic and photothermal therapies: synergy opportunities for nanomedicine. ACS Nano. 2023;17(9):7979–8003. doi: 10.1021/acsnano.3c00891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Sun Y, Zhao D, Wang G, et al. Recent progress of hypoxia-modulated multifunctional nanomedicines to enhance photodynamic therapy: opportunities, challenges, and future development. Acta Pharm Sin B. 2020;10(8):1382–1396. doi: 10.1016/j.apsb.2020.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Hu X, Zhang Y-S, Liu Y-C, et al. Emerging photodynamic/sonodynamic therapies for urological cancers: progress and challenges. J Nanobiotechnology. 2022;20(1):437. doi: 10.1186/s12951-022-01637-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Donohoe C, Senge MO, Arnaut LG, et al. Cell death in photodynamic therapy: from oxidative stress to anti-tumor immunity. Biochim Biophys Acta Rev Cancer. 2019;1872(2):188308. doi: 10.1016/j.bbcan.2019.07.003 [DOI] [PubMed] [Google Scholar]
- 122.Zhou Z, Zhang L, Zhang Z, et al. Advances in photosensitizer-related design for photodynamic therapy. Asian J Pharm Sci. 2021;16(6):668–686. doi: 10.1016/j.ajps.2020.12.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ballut S, Makky A, Loock B, et al.New strategy for targeting of photosensitizers. Synthesis of glycodendrimeric phenylporphyrins, incorporation into a liposome membrane and interaction with a specific lectin. Chem Commun. 2009;(2):224–226. doi: 10.1039/B816128C [DOI] [PubMed] [Google Scholar]
- 124.Pattani VP, Shah J, Atalis A, et al. Role of apoptosis and necrosis in cell death induced by nanoparticle-mediated photothermal therapy. J Nanopart Res. 2015;17(1):20. doi: 10.1007/s11051-014-2822-3 [DOI] [Google Scholar]
- 125.Hussein EA, Zagho M, Nasrallah G, et al. Recent advances in functional nanostructures as cancer photothermal therapy. Int J Nanomed. 2018;13:2897–2906. doi: 10.2147/IJN.S161031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Duan S, Hu Y, Zhao Y, et al. Nanomaterials for photothermal cancer therapy. RSC Adv. 2023;13(21):14443–14460. doi: 10.1039/D3RA02620E [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Liu Y, Bhattarai P, Dai Z, et al. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem Soc Rev. 2019;48(7):2053–2108. doi: 10.1039/c8cs00618k [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Granja A, Pinheiro M, Sousa CT, et al. Gold nanostructures as mediators of hyperthermia therapies in breast cancer. Biochem Pharmacol. 2021;190:114639. doi: 10.1016/j.bcp.2021.114639 [DOI] [PubMed] [Google Scholar]
- 129.Schuitmaker JJ, Baas P, Van leengoed HLLM, et al. Photodynamic therapy: a promising new modality for the treatment of cancer. J Photochem Photobiol B. 1996;34(1):3–12. doi: 10.1016/1011-1344(96)07342-3 [DOI] [PubMed] [Google Scholar]
- 130.Zhao X, Liu J, Fan J, et al. Recent progress in photosensitizers for overcoming the challenges of photodynamic therapy: from molecular design to application. Chem Soc Rev. 2021;50(6):4185–4219. doi: 10.1039/D0CS00173B [DOI] [PubMed] [Google Scholar]
- 131.Nguyen VN, Zhao Z, Tang BZ, et al. Organic photosensitizers for antimicrobial phototherapy. Chem Soc Rev. 2022;51(9):3324–3340. doi: 10.1039/D1CS00647A [DOI] [PubMed] [Google Scholar]
- 132.Hak A, Ali MS, Sankaranarayanan SA, et al. Chlorin e6: a promising photosensitizer in photo-based cancer nanomedicine. ACS Appl Bio Mater. 2023;6(2):349–364. doi: 10.1021/acsabm.2c00891 [DOI] [PubMed] [Google Scholar]
- 133.Liao S, Cai M, Zhu R, et al. Antitumor effect of photodynamic therapy/sonodynamic therapy/sono-photodynamic therapy of Chlorin e6 and other applications. Mol Pharm. 2023;20(2):875–885. doi: 10.1021/acs.molpharmaceut.2c00824 [DOI] [PubMed] [Google Scholar]
- 134.Theivendran S, Lazarev S, Yu C. Mesoporous silica/organosilica nanoparticles for cancer immunotherapy. Exploration. 2023;3(3):20220086. doi: 10.1002/EXP.20220086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Ooi EH, Popov V, Alfano M, et al. Influence of natural convection on gold nanorods-assisted photothermal treatment of bladder cancer in mice. Int J Hyperthermia. 2020;37(1):634–650. doi: 10.1080/02656736.2020.1771437 [DOI] [PubMed] [Google Scholar]
- 136.Seo SH, Joe A, Han H-W, et al. Mesoporous Silica-layered gold nanorod Core@Silver shell nanostructures for intracellular SERS imaging and phototherapy. Pharmaceutics. 2024;16(1):137. doi: 10.3390/pharmaceutics16010137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Wang R, Du N, Jin L, et al. Hyaluronic acid modified Au@SiO(2)@Au nanoparticles for photothermal therapy of genitourinary tumors. Polymers. 2022;14(21):4772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Huang X, Neretina S, El-Sayed MA. Gold nanorods: from synthesis and properties to biological and biomedical applications. Adv Mater. 2009;21(48):4880–4910. doi: 10.1002/adma.200802789 [DOI] [PubMed] [Google Scholar]
- 139.Huang J, Pu K. Near-infrared fluorescent molecular probes for imaging and diagnosis of nephro-urological diseases. Chem Sci. 2020;12(10):3379–3392. doi: 10.1039/D0SC02925D [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Ren S, Dai R, Zheng Z, et al. A novel bidirectional perfusion-like administered system for NIR-II fluorescence imaging precision diagnosis of bladder cancer. Nanomedicine. 2023;49:102661. doi: 10.1016/j.nano.2023.102661 [DOI] [PubMed] [Google Scholar]
- 141.Yu Z, Chan WK, Zhang Y, Tan TT. Near-infrared-II activated inorganic photothermal nanomedicines. Biomaterials. 2021;269:120459. [DOI] [PubMed] [Google Scholar]
- 142.Liu B, Qi Z, Chao J. Framework nucleic acids directed assembly of Au nanostructures for biomedical applications. Interdiscip Med. 2023;1(1). doi: 10.1002/INMD.20220009 [DOI] [Google Scholar]
- 143.Lee S, Jue M, Lee K, et al. Early-stage diagnosis of bladder cancer using surface-enhanced Raman spectroscopy combined with machine learning algorithms in a rat model. Biosens Bioelectron. 2024;246:115915. doi: 10.1016/j.bios.2023.115915 [DOI] [PubMed] [Google Scholar]
- 144.Xu Q, Li T, Lin J, et al. Label-free screening of common urinary system tumors from blood plasma based on surface-enhanced Raman spectroscopy. Photodiagnosis Photodyn Ther. 2024;45:103900. doi: 10.1016/j.pdpdt.2023.103900 [DOI] [PubMed] [Google Scholar]
- 145.Seo SH, Joe A, Han H-W, et al. Methylene blue-loaded mesoporous Silica-coated gold nanorods on graphene oxide for synergistic photothermal and photodynamic therapy. Pharmaceutics. 2022;14(10):2242. doi: 10.3390/pharmaceutics14102242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Joe A, Han H-W, Lim Y-R, et al. Triphenylphosphonium-functionalized gold nanorod/zinc oxide core-shell nanocomposites for mitochondrial-targeted phototherapy. Pharmaceutics. 2024;16(2):284. doi: 10.3390/pharmaceutics16020284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Cheong JK, Ooi EH, Chiew YS, et al. Gold nanorods assisted photothermal therapy of bladder cancer in mice: a computational study on the effects of gold nanorods distribution at the centre, periphery, and surface of bladder cancer. Comput Methods Programs Biomed. 2023;230:107363. doi: 10.1016/j.cmpb.2023.107363 [DOI] [PubMed] [Google Scholar]
- 148.Cai X, Ruan L, Wang D, et al. Boosting chemotherapy of bladder cancer cells by ferroptosis using intelligent magnetic targeting nanoparticles. Colloids Surf B Biointerfaces. 2024;234:113664. doi: 10.1016/j.colsurfb.2023.113664 [DOI] [PubMed] [Google Scholar]
- 149.Qi F, Bao Q, Hu P, et al. Mild magnetic hyperthermia-activated immuno-responses for primary bladder cancer therapy. Biomaterials. 2024;307:122514. doi: 10.1016/j.biomaterials.2024.122514 [DOI] [PubMed] [Google Scholar]
- 150.Li Y, Lin T-Y, Luo Y, et al. A smart and versatile theranostic nanomedicine platform based on nanoporphyrin. Nat Commun. 2014;5:4712. doi: 10.1038/ncomms5712 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Sun Y, Han R, Wang J, et al. A single-beam of light priming the immune responses and boosting cancer photoimmunotherapy. J Control Release. 2022;350:734–747. doi: 10.1016/j.jconrel.2022.08.057 [DOI] [PubMed] [Google Scholar]
- 152.Yang M, Li X, Kim G, et al. A J-aggregated nanoporphyrin overcoming phototoxic side effects in superior phototherapy with two-pronged effects. Chem Sci. 2022;13(43):12738–12746. doi: 10.1039/D2SC04873F [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Zhu Z, Ma A-H, Zhang H, et al. Phototherapy with cancer-specific nanoporphyrin potentiates immunotherapy in bladder cancer. Clin Cancer Res. 2022;28(21):4820–4831. doi: 10.1158/1078-0432.CCR-22-1362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Chen Y, Xu S, Ren S, et al. Design of a targeted dual drug delivery system for boosting the efficacy of photoimmunotherapy against melanoma proliferation and metastasis. J Adv Res. 2024. [DOI] [PubMed] [Google Scholar]
- 155.Lin TY, Li Y, Liu Q, et al. Novel theranostic nanoporphyrins for photodynamic diagnosis and trimodal therapy for bladder cancer. Biomaterials. 2016;104:339–351. doi: 10.1016/j.biomaterials.2016.07.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Chin YC, Yang L-X, Hsu F-T, et al. Iron oxide@chlorophyll clustered nanoparticles eliminate bladder cancer by photodynamic immunotherapy-initiated ferroptosis and immunostimulation. J Nanobiotechnology. 2022;20(1):373. doi: 10.1186/s12951-022-01575-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Ma C, Zhong X, Liu R, et al. Co-delivery of oxaliplatin prodrug liposomes with Bacillus Calmette-Guérin for chemo-immunotherapy of orthotopic bladder cancer. J Control Release. 2024;365:640–653. doi: 10.1016/j.jconrel.2023.11.050 [DOI] [PubMed] [Google Scholar]
- 158.De Jong WH, Borm PJ. Drug delivery and nanoparticles: applications and hazards. Int J Nanomed. 2008;3(2):133–149. doi: 10.2147/IJN.S596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Huang Y, Li P, Zhao R, et al. Silica nanoparticles: biomedical applications and toxicity. Biomed Pharmacother. 2022;151:113053. doi: 10.1016/j.biopha.2022.113053 [DOI] [PubMed] [Google Scholar]
- 160.Arora S, Rajwade JM, Paknikar KM. Nanotoxicology and in vitro studies: the need of the hour. Toxicol Appl Pharmacol. 2012;258(2):151–165. doi: 10.1016/j.taap.2011.11.010 [DOI] [PubMed] [Google Scholar]
- 161.Najahi-Missaoui W, Arnold RD, Cummings BS. Safe nanoparticles: are we there yet? Int J Mol Sci. 2020;22(1):385. doi: 10.3390/ijms22010385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Ali M. What function of nanoparticles is the primary factor for their hyper-toxicity? Adv Colloid Interface Sci. 2023;314:102881. doi: 10.1016/j.cis.2023.102881 [DOI] [PubMed] [Google Scholar]
- 163.Neetika N, Sharma M, Thakur P, et al. Cancer treatment and toxicity outlook of nanoparticles. Environ Res. 2023;237(Pt 1):116870. doi: 10.1016/j.envres.2023.116870 [DOI] [PubMed] [Google Scholar]
- 164.Shi J, Han S, Zhang J, et al. Advances in genotoxicity of titanium dioxide nanoparticles in vivo and in vitro. NanoImpact. 2022;25:100377. doi: 10.1016/j.impact.2021.100377 [DOI] [PubMed] [Google Scholar]
- 165.Lewinski N, Colvin V, Drezek R. Cytotoxicity of nanoparticles. Small. 2008;4(1):26–49. doi: 10.1002/smll.200700595 [DOI] [PubMed] [Google Scholar]
- 166.Pandey RK, Prajapati VK. Molecular and immunological toxic effects of nanoparticles. Int J Biol Macromol. 2018;107(Pt A):1278–1293. doi: 10.1016/j.ijbiomac.2017.09.110 [DOI] [PubMed] [Google Scholar]
- 167.Lammers T. Nanomedicine tumor targeting. Adv Mater. 2024;36(26):e2312169. doi: 10.1002/adma.202312169 [DOI] [PubMed] [Google Scholar]
- 168.Gonzalez-Valdivieso J, Girotti A, Schneider J, et al. Advanced nanomedicine and cancer: challenges and opportunities in clinical translation. Int J Pharm. 2021;599:120438. doi: 10.1016/j.ijpharm.2021.120438 [DOI] [PubMed] [Google Scholar]
- 169.Satalkar P, Elger BS, Hunziker P, et al. Challenges of clinical translation in nanomedicine: a qualitative study. Nanomedicine. 2016;12(4):893–900. doi: 10.1016/j.nano.2015.12.376 [DOI] [PubMed] [Google Scholar]
- 170.Cook D, Brown D, Alexander R, et al. Lessons learned from the fate of AstraZeneca’s drug pipeline: a five-dimensional framework. Nat Rev Drug Discov. 2014;13(6):419–431. doi: 10.1038/nrd4309 [DOI] [PubMed] [Google Scholar]
- 171.Metselaar JM, Lammers T. Challenges in nanomedicine clinical translation. Drug Deliv Transl Res. 2020;10(3):721–725. doi: 10.1007/s13346-020-00740-5 [DOI] [PMC free article] [PubMed] [Google Scholar]