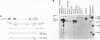Abstract
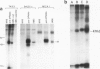
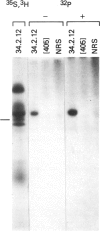
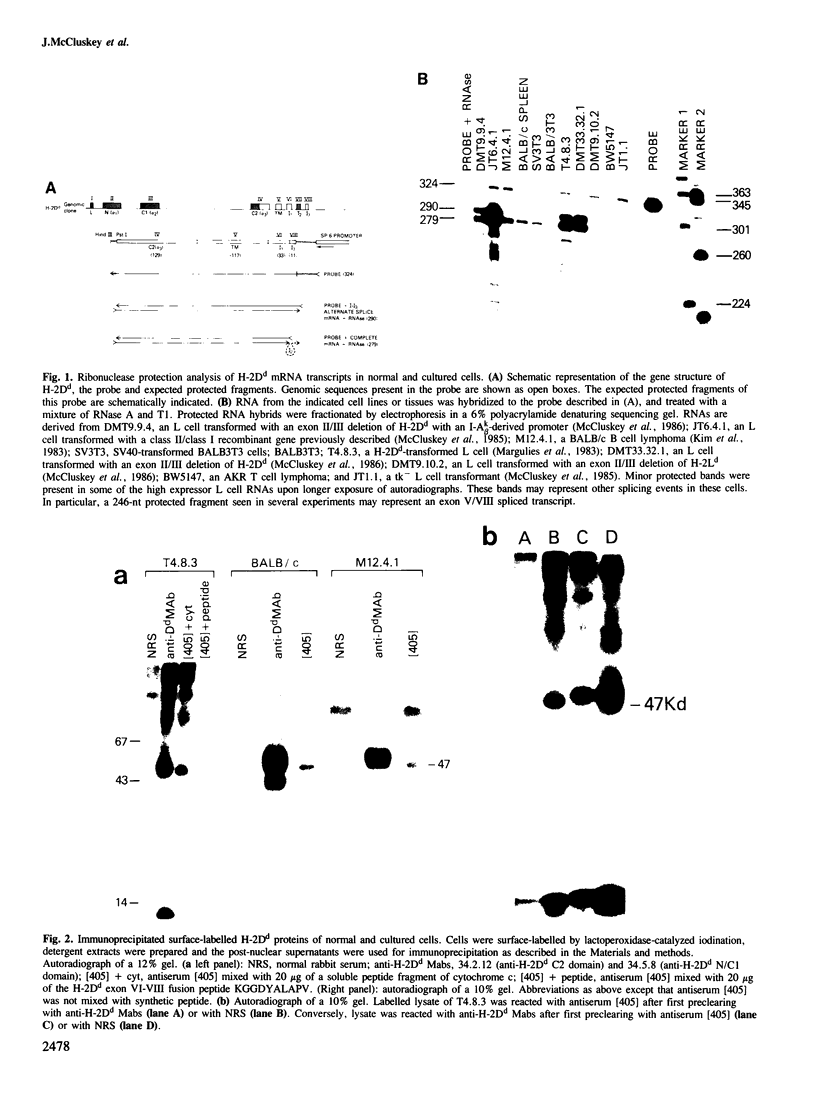
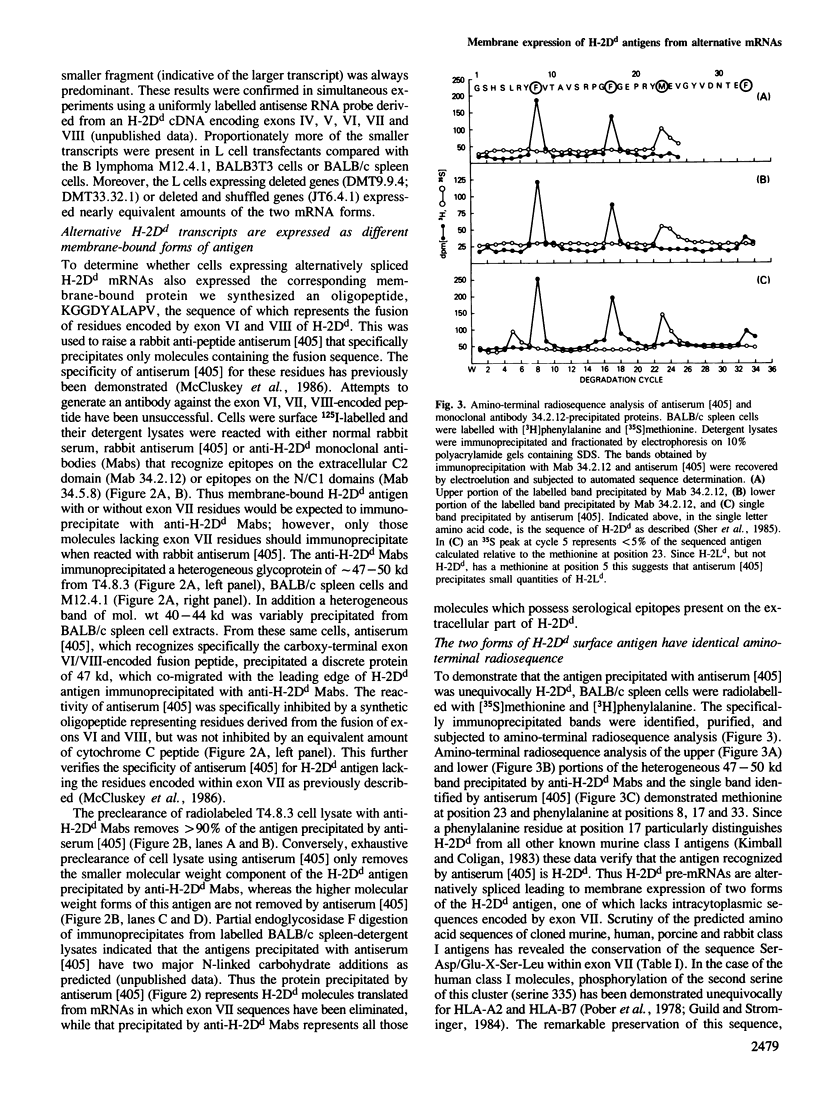
Two distinct mRNA species encoding the mouse major histocompatibility antigen H-2Dd have been identified in BALB/c spleen cells as well as in cultured cell lines expressing this cell surface glycoprotein. The alternate transcripts of H-2Dd arise from either removal or inclusion of exon VII (encoding I2) during pre-mRNA processing. The relative levels of each kind of H-2Dd transcript varied considerably between different cell types, and in all cells examined both forms of alloantigen were expressed on the cell membrane. Antigen derived from both types of transcript reacted with H-2Dd-specific monoclonal antibodies, whereas only protein lacking the 13 amino acids of I2 reacted with a specific antiserum raised against a predicted exon VI/VIII fusion peptide. Those H-2Dd proteins translated from full length, but not smaller, transcripts were phosphorylated in resting and phorbol myristate acetate-stimulated BALB/c spleen cells, suggesting that the major site of in vivo phosphorylation is within the highly conserved sequence encoded by exon VII. Thus alternative splicing of pre-mRNA transcripts is a mechanism which leads to membrane expression of two forms of H-2Dd, one of which lacks a major site of phosphorylation.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Archibald A. L., Thompson N. A., Kvist S. A single nucleotide difference at the 3' end of an intron causes differential splicing of two histocompatibility genes. EMBO J. 1986 May;5(5):957–965. doi: 10.1002/j.1460-2075.1986.tb04309.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arnold B., Burgert H. G., Archibald A. L., Kvist S. Complete nucleotide sequence of the murine H-2Kk gene. Comparison of three H-2K locus alleles. Nucleic Acids Res. 1984 Dec 21;12(24):9473–9487. doi: 10.1093/nar/12.24.9473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Breitbart R. E., Nguyen H. T., Medford R. M., Destree A. T., Mahdavi V., Nadal-Ginard B. Intricate combinatorial patterns of exon splicing generate multiple regulated troponin T isoforms from a single gene. Cell. 1985 May;41(1):67–82. doi: 10.1016/0092-8674(85)90062-5. [DOI] [PubMed] [Google Scholar]
- Brickell P. M., Latchman D. S., Murphy D., Willison K., Rigby P. W. Activation of a Qa/Tla class I major histocompatibility antigen gene is a general feature of oncogenesis in the mouse. Nature. 1983 Dec 22;306(5945):756–760. doi: 10.1038/306756a0. [DOI] [PubMed] [Google Scholar]
- Brickell P. M., Latchman D. S., Murphy D., Willison K., Rigby P. W. The class I major histocompatibility antigen gene activated in a line of SV40-transformed mouse cells is H-2Dd, not Qa/Tla. Nature. 1985 Jul 11;316(6024):162–163. doi: 10.1038/316162a0. [DOI] [PubMed] [Google Scholar]
- Chen Y. T., Obata Y., Stockert E., Old L. J. Thymus-leukemia (TL) antigens of the mouse. Analysis of TL mRNA and TL cDNA TL+ and TL- strains. J Exp Med. 1985 Oct 1;162(4):1134–1148. doi: 10.1084/jem.162.4.1134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Cohen S., Carpenter G., King L., Jr Epidermal growth factor-receptor-protein kinase interactions. Co-purification of receptor and epidermal growth factor-enhanced phosphorylation activity. J Biol Chem. 1980 May 25;255(10):4834–4842. [PubMed] [Google Scholar]
- Crabtree G. R., Kant J. A. Organization of the rat gamma-fibrinogen gene: alternative mRNA splice patterns produce the gamma A and gamma B (gamma ') chains of fibrinogen. Cell. 1982 Nov;31(1):159–166. doi: 10.1016/0092-8674(82)90415-9. [DOI] [PubMed] [Google Scholar]
- Early P., Rogers J., Davis M., Calame K., Bond M., Wall R., Hood L. Two mRNAs can be produced from a single immunoglobulin mu gene by alternative RNA processing pathways. Cell. 1980 Jun;20(2):313–319. doi: 10.1016/0092-8674(80)90617-0. [DOI] [PubMed] [Google Scholar]
- Ebina Y., Ellis L., Jarnagin K., Edery M., Graf L., Clauser E., Ou J. H., Masiarz F., Kan Y. W., Goldfine I. D. The human insulin receptor cDNA: the structural basis for hormone-activated transmembrane signalling. Cell. 1985 Apr;40(4):747–758. doi: 10.1016/0092-8674(85)90334-4. [DOI] [PubMed] [Google Scholar]
- Fehlmann M., Peyron J. F., Samson M., Van Obberghen E., Brandenburg D., Brossette N. Molecular association between major histocompatibility complex class I antigens and insulin receptors in mouse liver membranes. Proc Natl Acad Sci U S A. 1985 Dec;82(24):8634–8637. doi: 10.1073/pnas.82.24.8634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher D. A., Hunt S. W., 3rd, Hood L. Structure of a gene encoding a murine thymus leukemia antigen, and organization of Tla genes in the BALB/c mouse. J Exp Med. 1985 Aug 1;162(2):528–545. doi: 10.1084/jem.162.2.528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guild B. C., Strominger J. L. Human and murine class I MHC antigens share conserved serine 335, the site of HLA phosphorylation in vivo. J Biol Chem. 1984 Jul 25;259(14):9235–9240. [PubMed] [Google Scholar]
- Haustein D. Effective radioiodination by lactoperoxidase and solubilisation of cell-surface proteins of cultured murine T lymphoma cells. J Immunol Methods. 1975 Apr;7(1):25–38. doi: 10.1016/0022-1759(75)90127-1. [DOI] [PubMed] [Google Scholar]
- Heldin C. H., Ek B., Rönnstrand L. Characterization of the receptor for platelet-derived growth factor on human fibroblasts. Demonstration of an intimate relationship with a 185,000-Dalton substrate for the platelet-derived growth factor-stimulated kinase. J Biol Chem. 1983 Aug 25;258(16):10054–10061. [PubMed] [Google Scholar]
- Jacobs S., Kull F. C., Jr, Earp H. S., Svoboda M. E., Van Wyk J. J., Cuatrecasas P. Somatomedin-C stimulates the phosphorylation of the beta-subunit of its own receptor. J Biol Chem. 1983 Aug 25;258(16):9581–9584. [PubMed] [Google Scholar]
- Kim K. J., Kanellopoulos-Langevin C., Merwin R. M., Sachs D. H., Asofsky R. Establishment and characterization of BALB/c lymphoma lines with B cell properties. J Immunol. 1979 Feb;122(2):549–554. [PubMed] [Google Scholar]
- Kimball E. S., Coligan J. E. Structure of class I major histocompatibility antigens. Contemp Top Mol Immunol. 1983;9:1–63. doi: 10.1007/978-1-4684-4517-6_1. [DOI] [PubMed] [Google Scholar]
- King C. R., Piatigorsky J. Alternative RNA splicing of the murine alpha A-crystallin gene: protein-coding information within an intron. Cell. 1983 Mar;32(3):707–712. doi: 10.1016/0092-8674(83)90056-9. [DOI] [PubMed] [Google Scholar]
- Koller B. H., Orr H. T. Cloning and complete sequence of an HLA-A2 gene: analysis of two HLA-A alleles at the nucleotide level. J Immunol. 1985 Apr;134(4):2727–2733. [PubMed] [Google Scholar]
- Kress M., Glaros D., Khoury G., Jay G. Alternative RNA splicing in expression of the H-2K gene. Nature. 1983 Dec 8;306(5943):602–604. doi: 10.1038/306602a0. [DOI] [PubMed] [Google Scholar]
- Kvist S., Roberts L., Dobberstein B. Mouse histocompatibility genes: structure and organisation of a Kd gene. EMBO J. 1983;2(2):245–254. doi: 10.1002/j.1460-2075.1983.tb01413.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lalanne J. L., Cochet M., Kummer A. M., Gachelin G., Kourilsky P. Different exon-intron organization at the 5' part of a mouse class I gene is used to generate a novel H-2Kd-related mRNA. Proc Natl Acad Sci U S A. 1983 Dec;80(24):7561–7565. doi: 10.1073/pnas.80.24.7561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lalanne J. L., Transy C., Guerin S., Darche S., Meulien P., Kourilsky P. Expression of class I genes in the major histocompatibility complex: identification of eight distinct mRNAs in DBA/2 mouse liver. Cell. 1985 Jun;41(2):469–478. doi: 10.1016/s0092-8674(85)80020-9. [DOI] [PubMed] [Google Scholar]
- Laskey R. A., Mills A. D. Quantitative film detection of 3H and 14C in polyacrylamide gels by fluorography. Eur J Biochem. 1975 Aug 15;56(2):335–341. doi: 10.1111/j.1432-1033.1975.tb02238.x. [DOI] [PubMed] [Google Scholar]
- Laski F. A., Rio D. C., Rubin G. M. Tissue specificity of Drosophila P element transposition is regulated at the level of mRNA splicing. Cell. 1986 Jan 17;44(1):7–19. doi: 10.1016/0092-8674(86)90480-0. [DOI] [PubMed] [Google Scholar]
- Lew A. M., Lillehoj E. P., Cowan E. P., Maloy W. L., van Schravendijk M. R., Coligan J. E. Class I genes and molecules: an update. Immunology. 1986 Jan;57(1):3–18. [PMC free article] [PubMed] [Google Scholar]
- Lew A. M., Margulies D. H., Maloy W. L., Lillehoj E. P., McCluskey J., Coligan J. E. Alternative protein products with different carboxyl termini from a single class I gene, H-2Kb. Proc Natl Acad Sci U S A. 1986 Aug;83(16):6084–6088. doi: 10.1073/pnas.83.16.6084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malissen M., Malissen B., Jordan B. R. Exon/intron organization and complete nucleotide sequence of an HLA gene. Proc Natl Acad Sci U S A. 1982 Feb;79(3):893–897. doi: 10.1073/pnas.79.3.893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marche P. N., Tykocinski M. L., Max E. E., Kindt T. J. Structure of a functional rabbit class I MHC gene: similarity to human class I genes. Immunogenetics. 1985;21(1):71–82. doi: 10.1007/BF00372243. [DOI] [PubMed] [Google Scholar]
- Margulies D. H., Evans G. A., Ozato K., Camerini-Otero R. D., Tanaka K., Appella E., Seidman J. G. Expression of H-2Dd and H-2Ld mouse major histocompatibility antigen genes in L cells after DNA-mediated gene transfer. J Immunol. 1983 Jan;130(1):463–470. [PubMed] [Google Scholar]
- Margulies D. H., Evans G. A., Ozato K., Camerini-Otero R. D., Tanaka K., Appella E., Seidman J. G. Partial nucleotide sequence of H-2Dd major histocompatibility antigen gene. J Immunol. 1985 Aug;135(2):1537–1537. [PubMed] [Google Scholar]
- McCluskey J., Bluestone J. A., Coligan J. E., Maloy W. L., Margulies D. H. Serologic and T cell recognition of truncated transplantation antigens encoded by in vitro deleted class I major histocompatibility genes. J Immunol. 1986 Feb 15;136(4):1472–1481. [PubMed] [Google Scholar]
- McCluskey J., Germain R. N., Margulies D. H. Cell surface expression of an in vitro recombinant class II/class I major histocompatibility complex gene product. Cell. 1985 Feb;40(2):247–257. doi: 10.1016/0092-8674(85)90139-4. [DOI] [PubMed] [Google Scholar]
- Melton D. A., Krieg P. A., Rebagliati M. R., Maniatis T., Zinn K., Green M. R. Efficient in vitro synthesis of biologically active RNA and RNA hybridization probes from plasmids containing a bacteriophage SP6 promoter. Nucleic Acids Res. 1984 Sep 25;12(18):7035–7056. doi: 10.1093/nar/12.18.7035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moore K. W., Sher B. T., Sun Y. H., Eakle K. A., Hood L. DNA sequence of a gene encoding a BALB/c mouse Ld transplantation antigen. Science. 1982 Feb 5;215(4533):679–682. doi: 10.1126/science.7058332. [DOI] [PubMed] [Google Scholar]
- Morita T., Delarbre C., Kress M., Kourilsky P., Gachelin G. An H-2K gene of the tw32 mutant at the T/t complex is a close parent of an H-2Kq gene. Immunogenetics. 1985;21(4):367–383. doi: 10.1007/BF00430802. [DOI] [PubMed] [Google Scholar]
- Murre C., Reiss C. S., Bernabeu C., Chen L. B., Burakoff S. J., Seidman J. G. Construction, expression and recognition of an H-2 molecule lacking its carboxyl terminus. Nature. 1984 Feb 2;307(5950):432–436. doi: 10.1038/307432a0. [DOI] [PubMed] [Google Scholar]
- Mushinski J. F., Blattner F. R., Owens J. D., Finkelman F. D., Kessler S. W., Fitzmaurice L., Potter M., Tucker P. W. Mouse immunoglobulin D: construction and characterization of a cloned delta chain cDNA. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7405–7409. doi: 10.1073/pnas.77.12.7405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- N'Guyen C., Sodoyer R., Trucy J., Strachan T., Jordan B. R. The HLA-AW24 gene: sequence, surroundings and comparison with the HLA-A2 and HLA-A3 genes. Immunogenetics. 1985;21(5):479–489. doi: 10.1007/BF00430931. [DOI] [PubMed] [Google Scholar]
- Nevins J. R., Wilson M. C. Regulation of adenovirus-2 gene expression at the level of transcriptional termination and RNA processing. Nature. 1981 Mar 12;290(5802):113–118. doi: 10.1038/290113a0. [DOI] [PubMed] [Google Scholar]
- Obata Y., Chen Y. T., Stockert E., Old L. J. Structural analysis of TL genes of the mouse. Proc Natl Acad Sci U S A. 1985 Aug;82(16):5475–5479. doi: 10.1073/pnas.82.16.5475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Padgett R. A., Konarska M. M., Grabowski P. J., Hardy S. F., Sharp P. A. Lariat RNA's as intermediates and products in the splicing of messenger RNA precursors. Science. 1984 Aug 31;225(4665):898–903. doi: 10.1126/science.6206566. [DOI] [PubMed] [Google Scholar]
- Pober J. S., Guild B. C., Strominger J. L. Phosphorylation in vivo and in vitro of human histocompatibility antigens (HLA-A and HLA-B) in the carboxy-terminal intracellular domain. Proc Natl Acad Sci U S A. 1978 Dec;75(12):6002–6006. doi: 10.1073/pnas.75.12.6002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reyes A. A., Schöld M., Wallace R. B. The complete amino acid sequence of the murine transplantation antigen H-2Db as deduced by molecular cloning. Immunogenetics. 1982;16(1):1–9. doi: 10.1007/BF00364437. [DOI] [PubMed] [Google Scholar]
- Ruskin B., Krainer A. R., Maniatis T., Green M. R. Excision of an intact intron as a novel lariat structure during pre-mRNA splicing in vitro. Cell. 1984 Aug;38(1):317–331. doi: 10.1016/0092-8674(84)90553-1. [DOI] [PubMed] [Google Scholar]
- Satz M. L., Wang L. C., Singer D. S., Rudikoff S. Structure and expression of two porcine genomic clones encoding class I MHC antigens. J Immunol. 1985 Sep;135(3):2167–2175. [PubMed] [Google Scholar]
- Sher B. T., Nairn R., Coligan J. E., Hood L. E. DNA sequence of the mouse H-2Dd transplantation antigen gene. Proc Natl Acad Sci U S A. 1985 Feb;82(4):1175–1179. doi: 10.1073/pnas.82.4.1175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sodoyer R., Damotte M., Delovitch T. L., Trucy J., Jordan B. R., Strachan T. Complete nucleotide sequence of a gene encoding a functional human class I histocompatibility antigen (HLA-CW3). EMBO J. 1984 Apr;3(4):879–885. doi: 10.1002/j.1460-2075.1984.tb01900.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sood A. K., Pan J., Biro P. A., Pereira D., Srivastava R., Reddy V. B., Duceman B. W., Weissman S. M. Structure and polymorphism of class I MHC antigen mRNA. Immunogenetics. 1985;22(2):101–121. doi: 10.1007/BF00563508. [DOI] [PubMed] [Google Scholar]
- Steinmetz M., Hood L. Genes of the major histocompatibility complex in mouse and man. Science. 1983 Nov 18;222(4625):727–733. doi: 10.1126/science.6356354. [DOI] [PubMed] [Google Scholar]
- Steinmetz M., Moore K. W., Frelinger J. G., Sher B. T., Shen F. W., Boyse E. A., Hood L. A pseudogene homologous to mouse transplantation antigens: transplantation antigens are encoded by eight exons that correlate with protein domains. Cell. 1981 Sep;25(3):683–692. doi: 10.1016/0092-8674(81)90175-6. [DOI] [PubMed] [Google Scholar]
- Takahashi N., Roach A., Teplow D. B., Prusiner S. B., Hood L. Cloning and characterization of the myelin basic protein gene from mouse: one gene can encode both 14 kd and 18.5 kd MBPs by alternate use of exons. Cell. 1985 Aug;42(1):139–148. doi: 10.1016/s0092-8674(85)80109-4. [DOI] [PubMed] [Google Scholar]
- Weiss E., Golden L., Zakut R., Mellor A., Fahrner K., Kvist S., Flavell R. A. The DNA sequence of the H-2kb gene: evidence for gene conversion as a mechanism for the generation of polymorphism in histocompatibilty antigens. EMBO J. 1983;2(3):453–462. doi: 10.1002/j.1460-2075.1983.tb01444.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zamoyska R., Vollmer A. C., Sizer K. C., Liaw C. W., Parnes J. R. Two Lyt-2 polypeptides arise from a single gene by alternative splicing patterns of mRNA. Cell. 1985 Nov;43(1):153–163. doi: 10.1016/0092-8674(85)90020-0. [DOI] [PubMed] [Google Scholar]
- Zuniga M. C., Hood L. E. Clonal variation in cell surface display of an H-2 protein lacking a cytoplasmic tail. J Cell Biol. 1986 Jan;102(1):1–10. doi: 10.1083/jcb.102.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuniga M. C., Malissen B., McMillan M., Brayton P. R., Clark S. S., Forman J., Hood L. Expression and function of transplantation antigens with altered or deleted cytoplasmic domains. Cell. 1983 Sep;34(2):535–544. doi: 10.1016/0092-8674(83)90386-0. [DOI] [PubMed] [Google Scholar]
- de Ferra F., Engh H., Hudson L., Kamholz J., Puckett C., Molineaux S., Lazzarini R. A. Alternative splicing accounts for the four forms of myelin basic protein. Cell. 1985 Dec;43(3 Pt 2):721–727. doi: 10.1016/0092-8674(85)90245-4. [DOI] [PubMed] [Google Scholar]