Abstract
In Drosophila, dopa decarboxylase (DDC) serves a dual role in neurotransmitter production and sclerotization of the cuticle. The Ddc gene is under complex hormonal and tissue-specific control and several sizes of Ddc RNA are observed at embryonic hatching, pupariation and adult eclosion. We present here the complete nucleotide sequence of the Drosophila dopa decarboxylase gene and the partial sequence of two corresponding Ddc cDNAs. The sequence allows us to account for the detailed structure of four of the five major Ddc RNA species observed. The cDNA sequence reveals the existence of previously undetected splicing events and provides evidence for two RNA splicing alternatives which appear to encode two protein isoforms. The structure, processing and developmental regulation of the Ddc transcripts and putative protein isoforms are discussed. Interestingly, the pyridoxal-binding peptide of porcine DDC matches the Drosophila sequence perfectly suggesting considerable selective pressure on at least portions of the sequence. This is the first available Ddc gene sequence from any organism and should serve as a basis of comparison for the related proteins of other species.
Full text
PDF




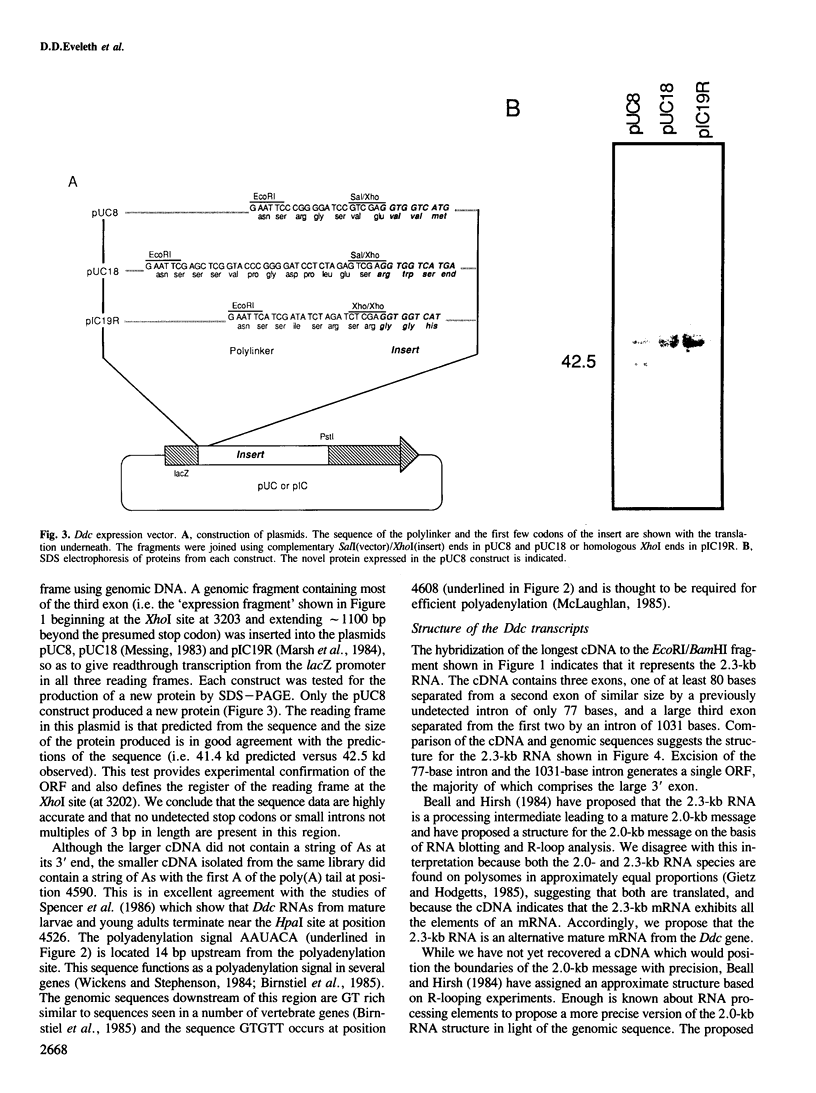




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Beall C. J., Hirsh J. High levels of intron-containing RNAs are associated with expression of the Drosophila DOPA decarboxylase gene. Mol Cell Biol. 1984 Sep;4(9):1669–1674. doi: 10.1128/mcb.4.9.1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benton W. D., Davis R. W. Screening lambdagt recombinant clones by hybridization to single plaques in situ. Science. 1977 Apr 8;196(4286):180–182. doi: 10.1126/science.322279. [DOI] [PubMed] [Google Scholar]
- Birnstiel M. L., Busslinger M., Strub K. Transcription termination and 3' processing: the end is in site! Cell. 1985 Jun;41(2):349–359. doi: 10.1016/s0092-8674(85)80007-6. [DOI] [PubMed] [Google Scholar]
- Bossa F., Martini F., Barra D., Voltattorni C. B., Minelli A., Turano C. The chymotryptic phosphopyridoxyl peptide of DOPA decarboxylase from pig kidney. Biochem Biophys Res Commun. 1977 Sep 9;78(1):177–184. doi: 10.1016/0006-291x(77)91237-2. [DOI] [PubMed] [Google Scholar]
- Breathnach R., Chambon P. Organization and expression of eucaryotic split genes coding for proteins. Annu Rev Biochem. 1981;50:349–383. doi: 10.1146/annurev.bi.50.070181.002025. [DOI] [PubMed] [Google Scholar]
- Burd J. F., Wells R. D. Synthesis and characterization of the duplex block polymer d(C15A15)-d(T15G15). J Biol Chem. 1974 Nov 25;249(22):7094–7801. [PubMed] [Google Scholar]
- Clark W. C., Doctor J., Fristrom J. W., Hodgetts R. B. Differential responses of the dopa decarboxylase gene to 20-OH-ecdysone in Drosophila melanogaster. Dev Biol. 1986 Mar;114(1):141–150. doi: 10.1016/0012-1606(86)90390-8. [DOI] [PubMed] [Google Scholar]
- Corden J., Wasylyk B., Buchwalder A., Sassone-Corsi P., Kedinger C., Chambon P. Promoter sequences of eukaryotic protein-coding genes. Science. 1980 Sep 19;209(4463):1406–1414. doi: 10.1126/science.6251548. [DOI] [PubMed] [Google Scholar]
- Dynan W. S., Tjian R. Control of eukaryotic messenger RNA synthesis by sequence-specific DNA-binding proteins. 1985 Aug 29-Sep 4Nature. 316(6031):774–778. doi: 10.1038/316774a0. [DOI] [PubMed] [Google Scholar]
- Gietz R. D., Hodgetts R. B. An analysis of dopa decarboxylase expression during embryogenesis in Drosophila melanogaster. Dev Biol. 1985 Jan;107(1):142–155. doi: 10.1016/0012-1606(85)90383-5. [DOI] [PubMed] [Google Scholar]
- Hirsh J., Davidson N. Isolation and characterization of the dopa decarboxylase gene of Drosophila melanogaster. Mol Cell Biol. 1981 Jun;1(6):475–485. doi: 10.1128/mcb.1.6.475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hiruma K., Riddiford L. M. Hormonal regulation of dopa decarboxylase during a larval molt. Dev Biol. 1985 Aug;110(2):509–513. doi: 10.1016/0012-1606(85)90109-5. [DOI] [PubMed] [Google Scholar]
- Hodgetts R. B., Clark W. C., Eveleth D. D., Jr, Gietz R. D., Spencer C. A., Marsh J. L. Hormonal aspects of the regulation of dopa decarboxylase in Drosophila melanogaster. Prog Clin Biol Res. 1986;217A:221–234. [PubMed] [Google Scholar]
- Hoopes B. C., McClure W. R. Studies on the selectivity of DNA precipitation by spermine. Nucleic Acids Res. 1981 Oct 24;9(20):5493–5504. doi: 10.1093/nar/9.20.5493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunt T. False starts in translational control of gene expression. Nature. 1985 Aug 15;316(6029):580–581. doi: 10.1038/316580a0. [DOI] [PubMed] [Google Scholar]
- Keller E. B., Noon W. A. Intron splicing: a conserved internal signal in introns of Drosophila pre-mRNAs. Nucleic Acids Res. 1985 Jul 11;13(13):4971–4981. doi: 10.1093/nar/13.13.4971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kozak M. Possible role of flanking nucleotides in recognition of the AUG initiator codon by eukaryotic ribosomes. Nucleic Acids Res. 1981 Oct 24;9(20):5233–5252. doi: 10.1093/nar/9.20.5233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraminsky G. P., Clark W. C., Estelle M. A., Gietz R. D., Sage B. A., O'Connor J. D., Hodgetts R. B. Induction of translatable mRNA for dopa decarboxylase in Drosophila: an early response to ecdysterone. Proc Natl Acad Sci U S A. 1980 Jul;77(7):4175–4179. doi: 10.1073/pnas.77.7.4175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Livingstone M. S., Tempel B. L. Genetic dissection of monoamine neurotransmitter synthesis in Drosophila. Nature. 1983 May 5;303(5912):67–70. doi: 10.1038/303067a0. [DOI] [PubMed] [Google Scholar]
- Lunan K. D., Mitchell H. K. The metabolism of tyrosine-O-phosphate in Drosophila. Arch Biochem Biophys. 1969 Jul;132(2):450–456. doi: 10.1016/0003-9861(69)90388-9. [DOI] [PubMed] [Google Scholar]
- Maneckjee R., Baylin S. B. Use of radiolabeled monofluoromethyl-Dopa to define the subunit structure of human L-Dopa decarboxylase. Biochemistry. 1983 Dec 20;22(26):6058–6063. doi: 10.1021/bi00295a003. [DOI] [PubMed] [Google Scholar]
- Maniatis T., Hardison R. C., Lacy E., Lauer J., O'Connell C., Quon D., Sim G. K., Efstratiadis A. The isolation of structural genes from libraries of eucaryotic DNA. Cell. 1978 Oct;15(2):687–701. doi: 10.1016/0092-8674(78)90036-3. [DOI] [PubMed] [Google Scholar]
- Marsh J. L., Erfle M., Wykes E. J. The pIC plasmid and phage vectors with versatile cloning sites for recombinant selection by insertional inactivation. Gene. 1984 Dec;32(3):481–485. doi: 10.1016/0378-1119(84)90022-2. [DOI] [PubMed] [Google Scholar]
- Marsh J. L., Gibbs P. D., Timmons P. M. Developmental control of transduced dopa decarboxylase genes in D. melanogaster. Mol Gen Genet. 1985;198(3):393–403. doi: 10.1007/BF00332929. [DOI] [PubMed] [Google Scholar]
- Marsh J. L., Wright T. R. Developmental relationship between dopa decarboxylase, dopamine acetyltransferase, and ecdysone in Drosophila. Dev Biol. 1980 Dec;80(2):379–387. doi: 10.1016/0012-1606(80)90412-1. [DOI] [PubMed] [Google Scholar]
- Marsh J. L., Wright T. R. Evidence for regulatory variants of the dopa decarboxylase and alpha-methyldopa hypersensitive loci in Drosophila. Genetics. 1986 Feb;112(2):249–265. doi: 10.1093/genetics/112.2.249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McLauchlan J., Gaffney D., Whitton J. L., Clements J. B. The consensus sequence YGTGTTYY located downstream from the AATAAA signal is required for efficient formation of mRNA 3' termini. Nucleic Acids Res. 1985 Feb 25;13(4):1347–1368. doi: 10.1093/nar/13.4.1347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Mount S. M. A catalogue of splice junction sequences. Nucleic Acids Res. 1982 Jan 22;10(2):459–472. doi: 10.1093/nar/10.2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulvihill E. R., LePennec J. P., Chambon P. Chicken oviduct progesterone receptor: location of specific regions of high-affinity binding in cloned DNA fragments of hormone-responsive genes. Cell. 1982 Mar;28(3):621–632. doi: 10.1016/0092-8674(82)90217-3. [DOI] [PubMed] [Google Scholar]
- Muskavitch M. A., Hogness D. S. An expandable gene that encodes a Drosophila glue protein is not expressed in variants lacking remote upstream sequences. Cell. 1982 Jul;29(3):1041–1051. doi: 10.1016/0092-8674(82)90467-6. [DOI] [PubMed] [Google Scholar]
- Möritz T., Edström J. E., Pongs O. Cloning of a gene localized and expressed at the ecdysteroid regulated puff 74EF in salivary glands of Drosophila larvae. EMBO J. 1984 Feb;3(2):289–295. doi: 10.1002/j.1460-2075.1984.tb01798.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scheidereit C., Geisse S., Westphal H. M., Beato M. The glucocorticoid receptor binds to defined nucleotide sequences near the promoter of mouse mammary tumour virus. Nature. 1983 Aug 25;304(5928):749–752. doi: 10.1038/304749a0. [DOI] [PubMed] [Google Scholar]
- Scholnick S. B., Morgan B. A., Hirsh J. The cloned dopa decarboxylase gene is developmentally regulated when reintegrated into the Drosophila genome. Cell. 1983 Aug;34(1):37–45. doi: 10.1016/0092-8674(83)90134-4. [DOI] [PubMed] [Google Scholar]
- Schwindinger W. F., Warner J. R. DNA sequence analysis on the IBM-PC. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 2):601–604. doi: 10.1093/nar/12.1part2.601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer C. A., Gietz R. D., Hodgetts R. B. Analysis of the transcription unit adjacent to the 3'-end of the dopa decarboxylase gene in Drosophila melanogaster. Dev Biol. 1986 Mar;114(1):260–264. doi: 10.1016/0012-1606(86)90402-1. [DOI] [PubMed] [Google Scholar]
- Tanase S., Kojima H., Morino Y. Pyridoxal 5'-phosphate binding site of pig heart alanine aminotransferase. Biochemistry. 1979 Jul 10;18(14):3002–3007. doi: 10.1021/bi00581a015. [DOI] [PubMed] [Google Scholar]
- Tempel B. L., Livingstone M. S., Quinn W. G. Mutations in the dopa decarboxylase gene affect learning in Drosophila. Proc Natl Acad Sci U S A. 1984 Jun;81(11):3577–3581. doi: 10.1073/pnas.81.11.3577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vogt V. M. Purification and properties of S1 nuclease from Aspergillus. Methods Enzymol. 1980;65(1):248–255. doi: 10.1016/s0076-6879(80)65034-4. [DOI] [PubMed] [Google Scholar]
- Weiher H., König M., Gruss P. Multiple point mutations affecting the simian virus 40 enhancer. Science. 1983 Feb 11;219(4585):626–631. doi: 10.1126/science.6297005. [DOI] [PubMed] [Google Scholar]
- Wickens M., Stephenson P. Role of the conserved AAUAAA sequence: four AAUAAA point mutants prevent messenger RNA 3' end formation. Science. 1984 Nov 30;226(4678):1045–1051. doi: 10.1126/science.6208611. [DOI] [PubMed] [Google Scholar]



