Abstract
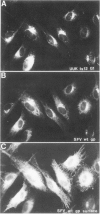
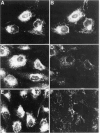
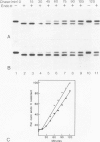
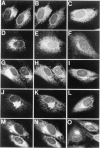
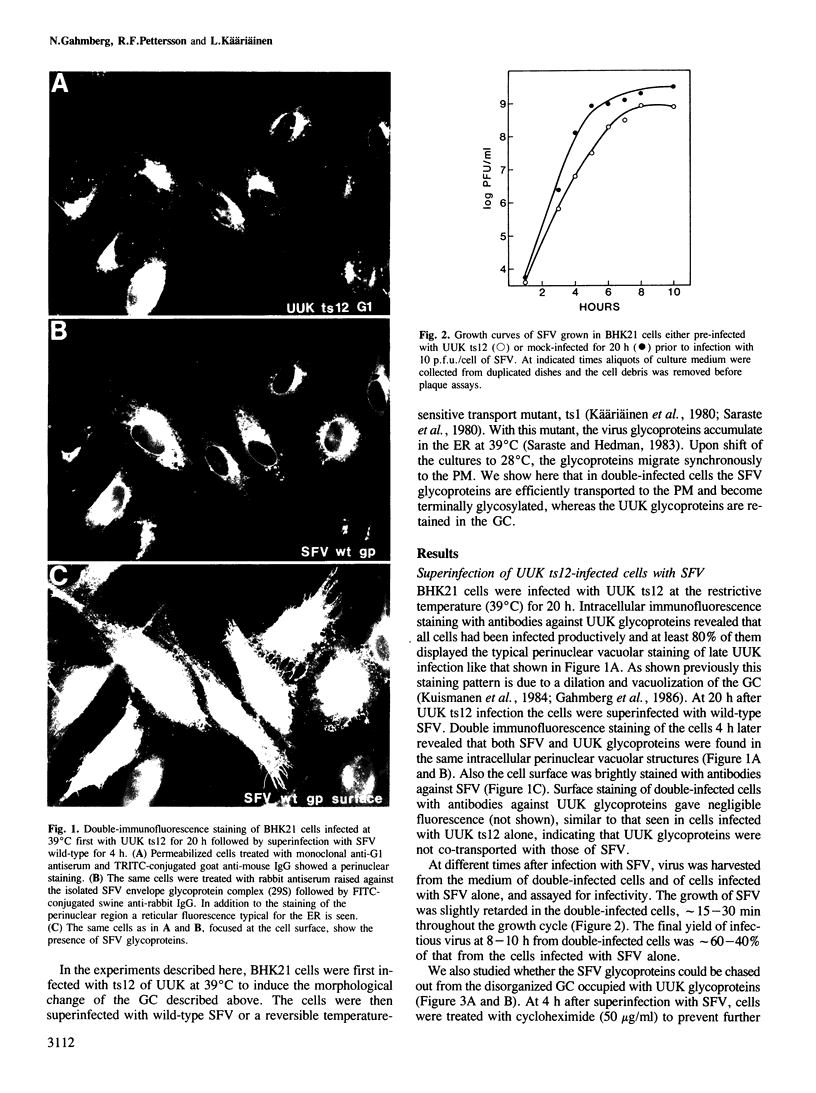
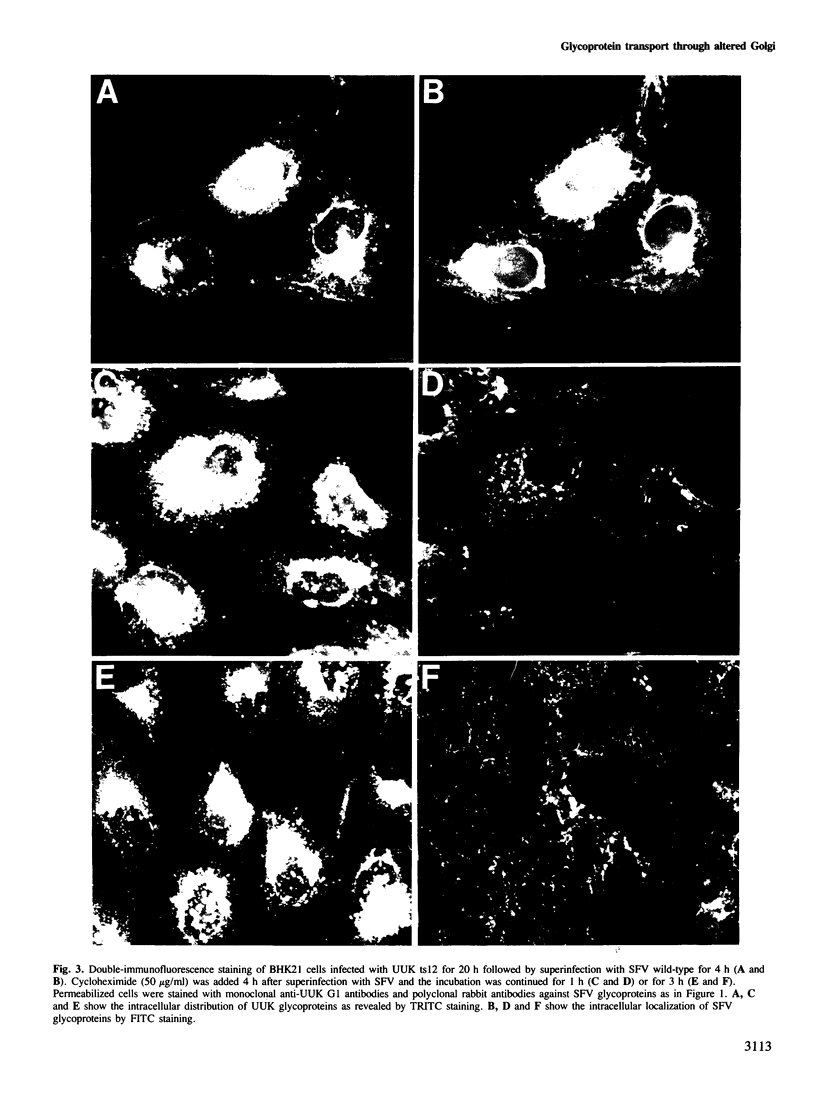
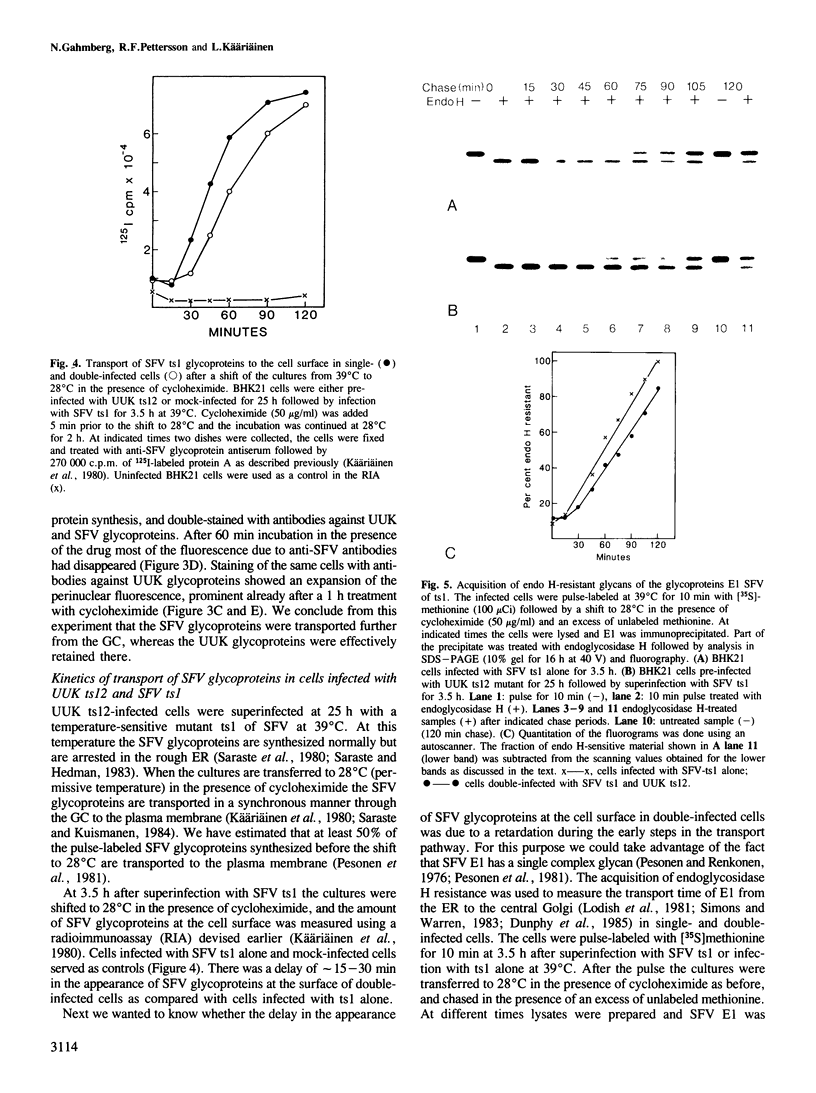
In infected BHK21 cells, the glycoproteins G1 and G2 of a temperature-sensitive mutant (ts12) of Uukuniemi virus (UUK) accumulate at 39 degrees C in the Golgi complex (GC) causing an expansion and vacuolization of this organelle. We have studied whether such an altered Golgi complex can carry out the glycosylation and transport to the plasma membrane (PM) of the Semliki Forest virus (SFV) glycoproteins in double-infected cells. Double-immunofluorescence staining showed that approximately 90% of the cells became infected with both viruses. Almost the same final yield of infectious SFV was obtained from double-infected cells as from cells infected with SFV alone. The rate of transport from the endoplasmic reticulum (ER) via the GC to the plasma membrane of the SFV glycoproteins was analysed by immunofluorescence, surface radioimmunoassay and pulse-chase labeling followed by immunoprecipitation, endoglycosidase H digestion and SDS-PAGE. The results showed that: the SFV glycoproteins were readily transported to the cell surface in double-infected cells, whereas the UUK glycoproteins were retained in the GC; the transport to the PM was retarded by approximately 20 min, due to a delay between the ER and the central Golgi; E1 of SFV appeared at the PM in a sialylated form. These results indicate that the morphologically altered GC had retained its functional integrity to glycosylate and transport plasma membrane glycoproteins.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson D. J., Mostov K. E., Blobel G. Mechanisms of integration of de novo-synthesized polypeptides into membranes: signal-recognition particle is required for integration into microsomal membranes of calcium ATPase and of lens MP26 but not of cytochrome b5. Proc Natl Acad Sci U S A. 1983 Dec;80(23):7249–7253. doi: 10.1073/pnas.80.23.7249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blobel G. Intracellular protein topogenesis. Proc Natl Acad Sci U S A. 1980 Mar;77(3):1496–1500. doi: 10.1073/pnas.77.3.1496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonner W. M., Laskey R. A. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur J Biochem. 1974 Jul 1;46(1):83–88. doi: 10.1111/j.1432-1033.1974.tb03599.x. [DOI] [PubMed] [Google Scholar]
- Dunphy W. G., Brands R., Rothman J. E. Attachment of terminal N-acetylglucosamine to asparagine-linked oligosaccharides occurs in central cisternae of the Golgi stack. Cell. 1985 Feb;40(2):463–472. doi: 10.1016/0092-8674(85)90161-8. [DOI] [PubMed] [Google Scholar]
- Dunphy W. G., Rothman J. E. Compartmental organization of the Golgi stack. Cell. 1985 Aug;42(1):13–21. doi: 10.1016/s0092-8674(85)80097-0. [DOI] [PubMed] [Google Scholar]
- Farquhar M. G. Progress in unraveling pathways of Golgi traffic. Annu Rev Cell Biol. 1985;1:447–488. doi: 10.1146/annurev.cb.01.110185.002311. [DOI] [PubMed] [Google Scholar]
- Fries E., Gustafsson L., Peterson P. A. Four secretory proteins synthesized by hepatocytes are transported from endoplasmic reticulum to Golgi complex at different rates. EMBO J. 1984 Jan;3(1):147–152. doi: 10.1002/j.1460-2075.1984.tb01775.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gahmberg C. G., Andersson L. C. Selective radioactive labeling of cell surface sialoglycoproteins by periodate-tritiated borohydride. J Biol Chem. 1977 Aug 25;252(16):5888–5894. [PubMed] [Google Scholar]
- Gahmberg N. Characterization of two recombination-complementation groups of Uukuniemi virus temperature-sensitive mutants. J Gen Virol. 1984 Jun;65(Pt 6):1079–1090. doi: 10.1099/0022-1317-65-6-1079. [DOI] [PubMed] [Google Scholar]
- Gahmberg N., Kuismanen E., Keränen S., Pettersson R. F. Uukuniemi virus glycoproteins accumulate in and cause morphological changes of the Golgi complex in the absence of virus maturation. J Virol. 1986 Mar;57(3):899–906. doi: 10.1128/jvi.57.3.899-906.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Green J., Griffiths G., Louvard D., Quinn P., Warren G. Passage of viral membrane proteins through the Golgi complex. J Mol Biol. 1981 Nov 15;152(4):663–698. doi: 10.1016/0022-2836(81)90122-4. [DOI] [PubMed] [Google Scholar]
- Hashimoto K., Erdei S., Keränen S., Saraste J., Käriäinen L. Evidence for a separate signal sequence for the carboxy-terminal envelope glycoprotein E1 of Semliki forest virus. J Virol. 1981 Apr;38(1):34–40. doi: 10.1128/jvi.38.1.34-40.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hubbard A. L., Cohn Z. A. The enzymatic iodination of the red cell membrane. J Cell Biol. 1972 Nov;55(2):390–405. doi: 10.1083/jcb.55.2.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kabcenell A. K., Atkinson P. H. Processing of the rough endoplasmic reticulum membrane glycoproteins of rotavirus SA11. J Cell Biol. 1985 Oct;101(4):1270–1280. doi: 10.1083/jcb.101.4.1270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly R. B. Pathways of protein secretion in eukaryotes. Science. 1985 Oct 4;230(4721):25–32. doi: 10.1126/science.2994224. [DOI] [PubMed] [Google Scholar]
- Keränen S., Käriäinen L. Isolation and basic characterization of temperature-sensitive mutants from Semliki Forest virus;. Acta Pathol Microbiol Scand B Microbiol Immunol. 1974 Dec;82(6):810–820. doi: 10.1111/j.1699-0463.1974.tb02378.x. [DOI] [PubMed] [Google Scholar]
- Kuismanen E., Bång B., Hurme M., Pettersson R. F. Uukuniemi virus maturation: immunofluorescence microscopy with monoclonal glycoprotein-specific antibodies. J Virol. 1984 Jul;51(1):137–146. doi: 10.1128/jvi.51.1.137-146.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuismanen E., Hedman K., Saraste J., Pettersson R. F. Uukuniemi virus maturation: accumulation of virus particles and viral antigens in the Golgi complex. Mol Cell Biol. 1982 Nov;2(11):1444–1458. doi: 10.1128/mcb.2.11.1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuismanen E. Posttranslational processing of Uukuniemi virus glycoproteins G1 and G2. J Virol. 1984 Sep;51(3):806–812. doi: 10.1128/jvi.51.3.806-812.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuismanen E., Saraste J., Pettersson R. F. Effect of monensin on the assembly of Uukuniemi virus in the Golgi complex. J Virol. 1985 Sep;55(3):813–822. doi: 10.1128/jvi.55.3.813-822.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Käriäinen L., Hashimoto K., Saraste J., Virtanen I., Penttinen K. Monensin and FCCP inhibit the intracellular transport of alphavirus membrane glycoproteins. J Cell Biol. 1980 Dec;87(3 Pt 1):783–791. doi: 10.1083/jcb.87.3.783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Käriäinen L., Virtanen I., Saraste J., Keränen S. Transport of virus membrane glycoproteins, use of temperature-sensitive mutants and organelle-specific lectins. Methods Enzymol. 1983;96:453–465. doi: 10.1016/s0076-6879(83)96040-8. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Laurence J. The immune system in AIDS. Sci Am. 1985 Dec;253(6):84–93. doi: 10.1038/scientificamerican1285-84. [DOI] [PubMed] [Google Scholar]
- Lodish H. F., Braell W. A., Schwartz A. L., Strous G. J., Zilberstein A. Synthesis and assembly of membrane and organelle proteins. Int Rev Cytol Suppl. 1981;12:247–307. doi: 10.1016/b978-0-12-364373-5.50016-0. [DOI] [PubMed] [Google Scholar]
- Lodish H. F., Kong N., Snider M., Strous G. J. Hepatoma secretory proteins migrate from rough endoplasmic reticulum to Golgi at characteristic rates. Nature. 1983 Jul 7;304(5921):80–83. doi: 10.1038/304080a0. [DOI] [PubMed] [Google Scholar]
- Pesonen M., Kuismanen E., Pettersson R. F. Monosaccharide sequence of protein-bound glycans of Uukuniemi virus. J Virol. 1982 Feb;41(2):390–400. doi: 10.1128/jvi.41.2.390-400.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pesonen M., Renkonen O. Serum glycoprotein-type sequence of monosaccharides in membrane glycoproteins of Semliki Forest virus. Biochim Biophys Acta. 1976 Dec 2;455(2):510–525. doi: 10.1016/0005-2736(76)90321-7. [DOI] [PubMed] [Google Scholar]
- Pesonen M., Saraste J., Hashimoto K., Käriäinen L. Reversible defect in the glycosylation of the membrane proteins of Semliki Forest virus ts-1 mutant. Virology. 1981 Feb;109(1):165–173. doi: 10.1016/0042-6822(81)90481-5. [DOI] [PubMed] [Google Scholar]
- Repp R., Tamura T., Boschek C. B., Wege H., Schwarz R. T., Niemann H. The effects of processing inhibitors of N-linked oligosaccharides on the intracellular migration of glycoprotein E2 of mouse hepatitis virus and the maturation of coronavirus particles. J Biol Chem. 1985 Dec 15;260(29):15873–15879. doi: 10.1016/S0021-9258(17)36339-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraste J., Hedman K. Intracellular vesicles involved in the transport of Semliki Forest virus membrane proteins to the cell surface. EMBO J. 1983;2(11):2001–2006. doi: 10.1002/j.1460-2075.1983.tb01691.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraste J., Kuismanen E. Pre- and post-Golgi vacuoles operate in the transport of Semliki Forest virus membrane glycoproteins to the cell surface. Cell. 1984 Sep;38(2):535–549. doi: 10.1016/0092-8674(84)90508-7. [DOI] [PubMed] [Google Scholar]
- Saraste J., von Bonsdorff C. H., Hashimoto K., Käriäinen L., Keränen S. Semliki forest virus mutants with temperature-sensitive transport defect of envelope proteins. Virology. 1980 Jan 30;100(2):229–245. doi: 10.1016/0042-6822(80)90516-4. [DOI] [PubMed] [Google Scholar]
- Simons K., Garoff H. The budding mechanisms of enveloped animal viruses. J Gen Virol. 1980 Sep;50(1):1–21. doi: 10.1099/0022-1317-50-1-1. [DOI] [PubMed] [Google Scholar]
- Simons K., Warren G. Semliki Forest virus: a probe for membrane traffic in the animal cell. Adv Protein Chem. 1984;36:79–132. doi: 10.1016/S0065-3233(08)60296-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strous G. J., Van Kerkhof P., Willemsen R., Geuze H. J., Berger E. G. Transport and topology of galactosyltransferase in endomembranes of HeLa cells. J Cell Biol. 1983 Sep;97(3):723–727. doi: 10.1083/jcb.97.3.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tarentino A. L., Plummer T. H., Jr, Maley F. The release of intact oligosaccharides from specific glycoproteins by endo-beta-N-acetylglucosaminidase H. J Biol Chem. 1974 Feb 10;249(3):818–824. [PubMed] [Google Scholar]
- Tooze J., Tooze S., Warren G. Replication of coronavirus MHV-A59 in sac- cells: determination of the first site of budding of progeny virions. Eur J Cell Biol. 1984 Mar;33(2):281–293. [PubMed] [Google Scholar]
- Von Bonsdorff C. H., Saikku P., Oker-Blom N. Electron microscope study on the development of Uukuniemi virus. Acta Virol. 1970 Mar;14(2):109–114. [PubMed] [Google Scholar]
- Vänänen P. The use of red cells with fused Semliki Forest virus envelope proteins in antibody determinations by hemolysis in gel. J Virol Methods. 1982 Mar;4(2):117–126. doi: 10.1016/0166-0934(82)90081-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wattenberg B. W., Balch W. E., Rothman J. E. A novel prefusion complex formed during protein transport between Golgi cisternae in a cell-free system. J Biol Chem. 1986 Feb 15;261(5):2202–2207. [PubMed] [Google Scholar]
- Williams D. B., Swiedler S. J., Hart G. W. Intracellular transport of membrane glycoproteins: two closely related histocompatibility antigens differ in their rates of transit to the cell surface. J Cell Biol. 1985 Sep;101(3):725–734. doi: 10.1083/jcb.101.3.725. [DOI] [PMC free article] [PubMed] [Google Scholar]