Abstract
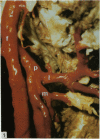
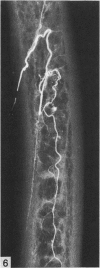
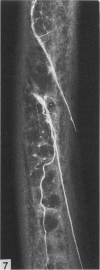
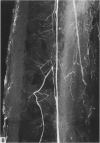
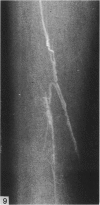
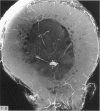
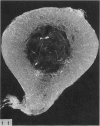
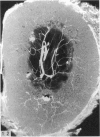
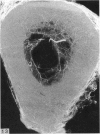
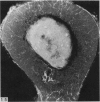
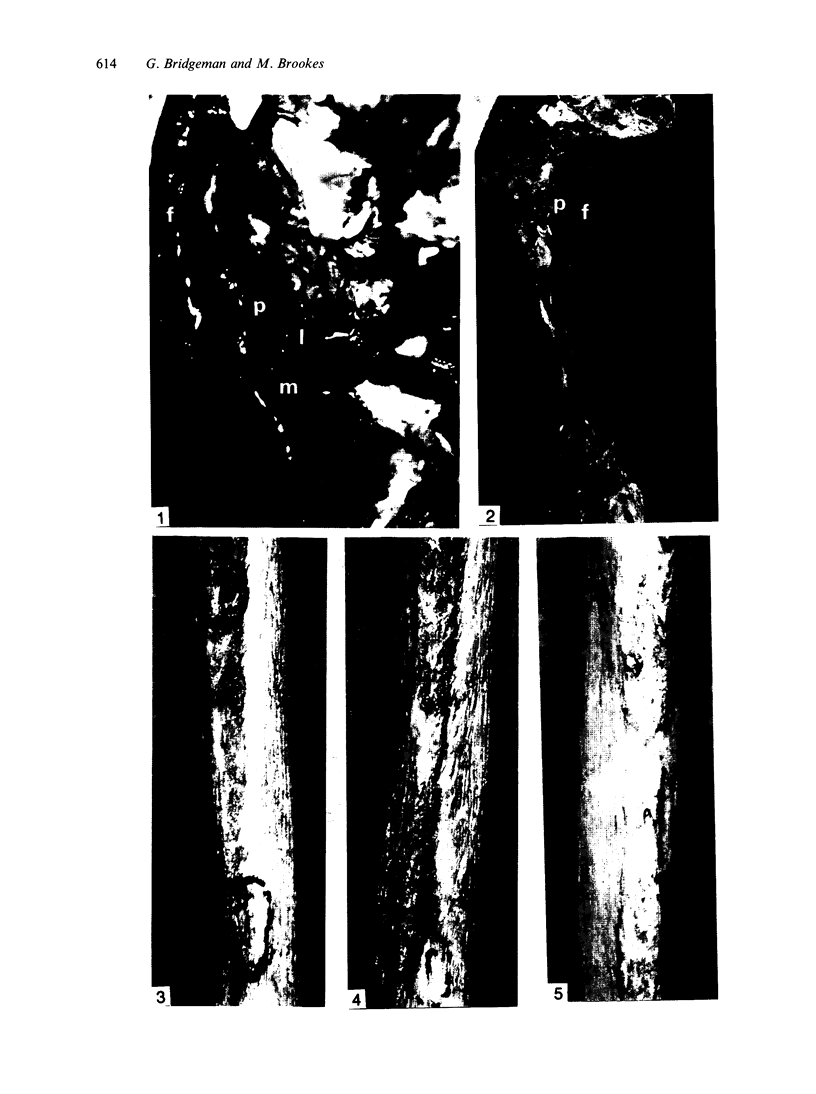
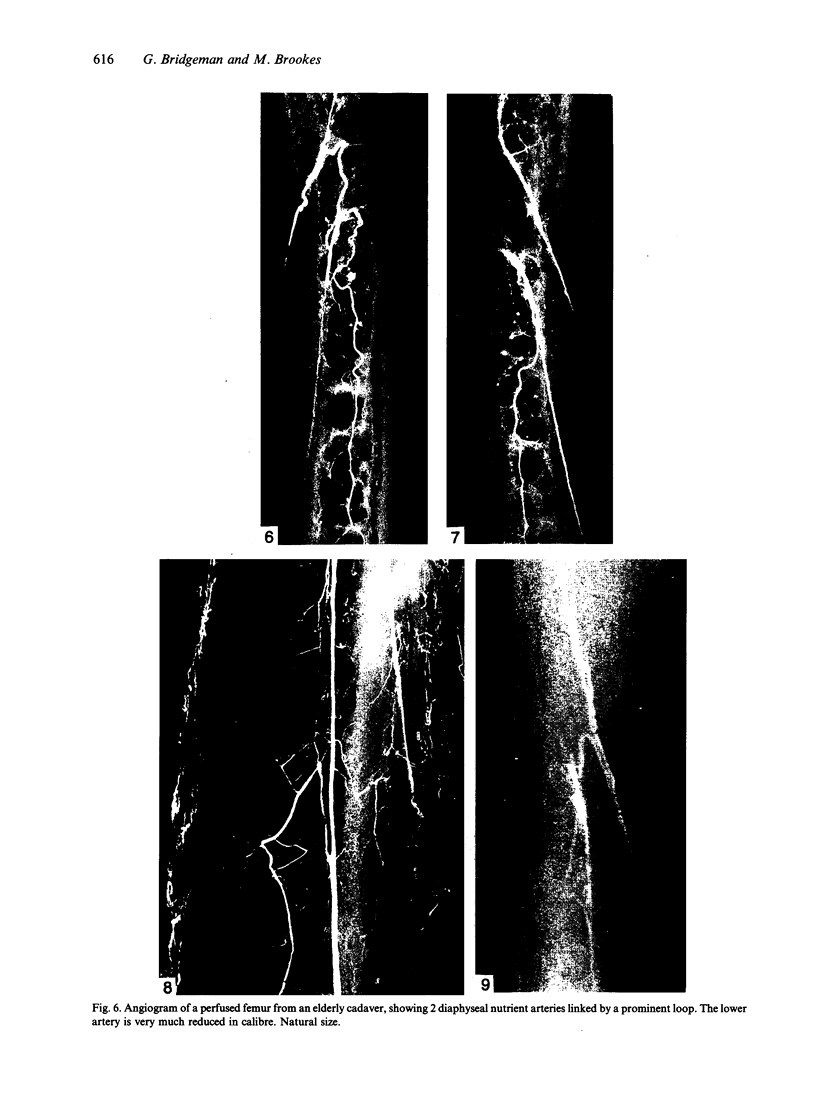
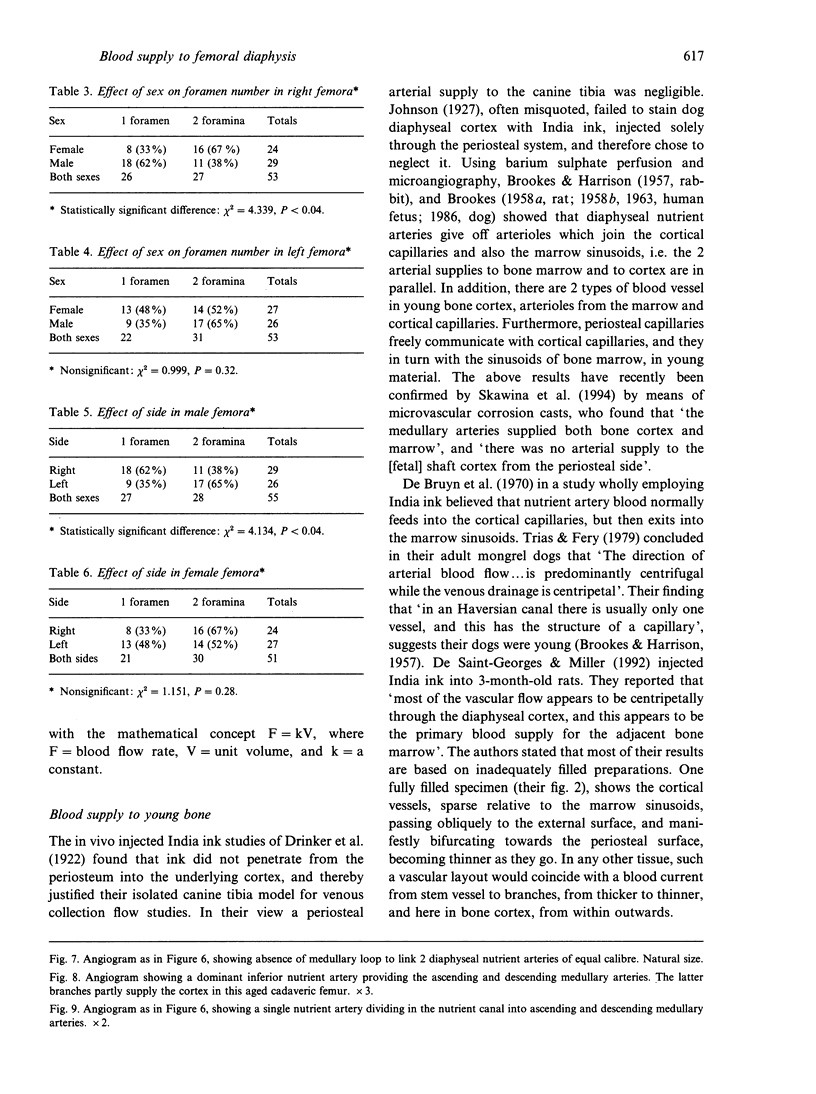
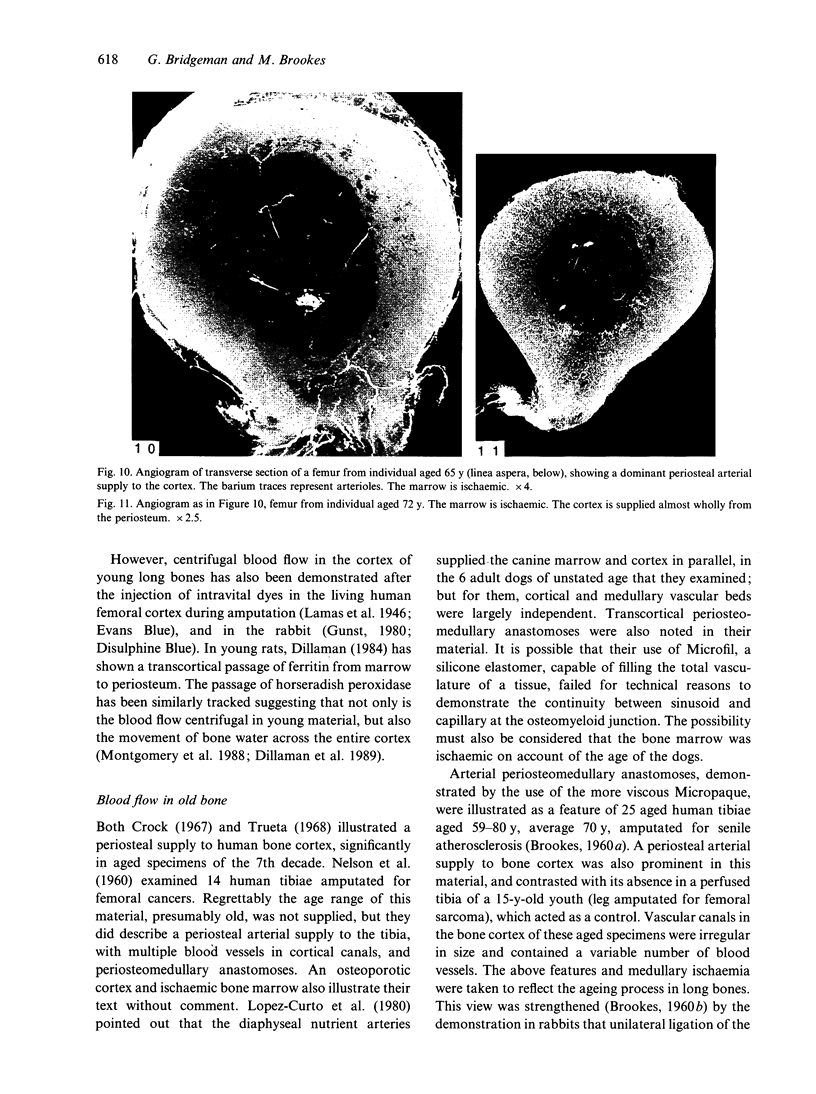
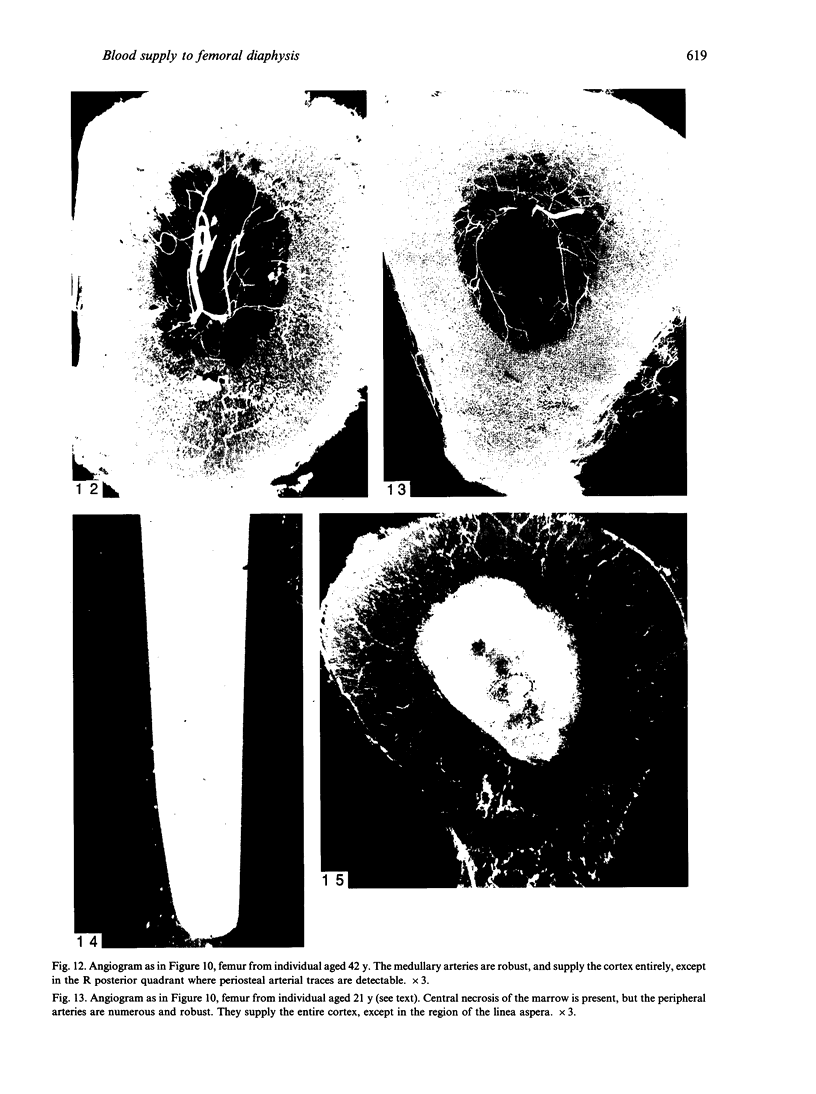
The nutrient foramina in the human femoral diaphysis were counted in 109 cleaned and dried bones, ranging in age from 38 to 98 y. Although statistical analysis for univariate associations found that foramen number was not linked to age, sex (male/female) or side (R/L), multivariate logistic analysis revealed a significant interaction between the effects of sex and side (P < 0.01). Univariate analysis for sex and for side separately revealed a significant difference in for men number between the results for men and women for R bones 62% (18/29) vs 33% (8/24) (P < 0.04), but not for L bones 35% (9/26) vs 48% (13/27) (P < 0.28). One lower limb from 15 cadavers was perfused with a barium sulphate suspension to show the blood supply of the femoral diaphysis. Twelve were in the age group 59-88 y and considered to be senescent material. In addition, 3 more femora were perfused, 1 from a young male aged 21 y, another from a 42-y-old male considered to show early senescent changes, and a third from a man aged 56 y with a hip prosthesis in situ, to provide an example of a cortex deprived entirely of its medulla. All perfused femora were fixed and decalcified, and sections were then radiographed. Cross-sections through the middiaphysis showed that aged bone cortex is supplied predominantly from the periosteum in contrast to the medullary supply in young human and animal material. The change is attributed to increasingly severe medullary ischaemia with age, brought on by atherosclerosis of the marrow vessels. An examination of the findings reported by investigators of animal bone blood supply in the past 40 y shows a large measure of agreement. Long standing controversy seems to be based on a failure to recognise that marrow ischaemia accompanies natural senescence, affecting transcortical haemodynamics, and entraining an increasing periosteal supply for bone survival in old age. The change over from a medullary to a periosteal blood supply to bone cortex is the consequence of medullary ischaemia and reduced marrow arterial pressure, brought about by medullary atherosclerosis.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnett T. R., Boyde A., Jones S. J., Taylor M. L. Effects of medium acidification by alteration of carbon dioxide or bicarbonate concentrations on the resorptive activity of rat osteoclasts. J Bone Miner Res. 1994 Mar;9(3):375–379. doi: 10.1002/jbmr.5650090312. [DOI] [PubMed] [Google Scholar]
- Arnett T. R., Dempster D. W. Protons and osteoclasts. J Bone Miner Res. 1990 Nov;5(11):1099–1103. doi: 10.1002/jbmr.5650051102. [DOI] [PubMed] [Google Scholar]
- BROOKES M. CORTICAL VASCULARIZATION AND GROWTH IN FOETAL TUBULAR BONES. J Anat. 1963 Oct;97:597–609. [PMC free article] [PubMed] [Google Scholar]
- BROOKES M., HARRISON R. G. The vascularization of the rabbit femur and tibio-fibula. J Anat. 1957 Jan;91(1):61–72. [PMC free article] [PubMed] [Google Scholar]
- BROOKES M. The vascularization of long bones in the human foetus. J Anat. 1958 Apr;92(2):261–267. [PMC free article] [PubMed] [Google Scholar]
- Brookes M. Sequelae of experimental partial ischaemia in long bones of the rabbit. J Anat. 1960 Oct;94(Pt 4):552–561. [PMC free article] [PubMed] [Google Scholar]
- De Bruyn P. P., Breen P. C., Thomas T. B. The microcirculation of the bone marrow. Anat Rec. 1970 Sep;168(1):55–68. doi: 10.1002/ar.1091680105. [DOI] [PubMed] [Google Scholar]
- Dillaman R. M. Movement of ferritin in the 2-day-old chick femur. Anat Rec. 1984 Aug;209(4):445–453. doi: 10.1002/ar.1092090404. [DOI] [PubMed] [Google Scholar]
- Dillaman R. M., Roer R. D., Gay D. M. Fluid movement in bone: theoretical and empirical. J Biomech. 1991;24 (Suppl 1):163–177. doi: 10.1016/0021-9290(91)90386-2. [DOI] [PubMed] [Google Scholar]
- Kelly P. J. Comparison of marrow and cortical bone blood flow by 125 I-labeled 4-iodoantipyrine (I-Ap) washout. J Lab Clin Med. 1973 Apr;81(4):497–505. [PubMed] [Google Scholar]
- Kita K., Kawai K., Hirohata K. Changes in bone marrow blood flow with aging. J Orthop Res. 1987;5(4):569–575. doi: 10.1002/jor.1100050412. [DOI] [PubMed] [Google Scholar]
- Lopez-Curto J. A., Bassingthwaighte J. B., Kelly P. J. Anatomy of the microvasculature of the tibial diaphysis of the adult dog. J Bone Joint Surg Am. 1980 Dec;62(8):1362–1369. [PMC free article] [PubMed] [Google Scholar]
- Montgomery R. J., Sutker B. D., Bronk J. T., Smith S. R., Kelly P. J. Interstitial fluid flow in cortical bone. Microvasc Res. 1988 May;35(3):295–307. doi: 10.1016/0026-2862(88)90084-2. [DOI] [PubMed] [Google Scholar]
- NELSON G. E., Jr, KELLY P. J., PETERSON L. F., JANES J. M. Blood supply of the human tibia. J Bone Joint Surg Am. 1960 Jun;42-A:625–636. [PubMed] [Google Scholar]
- Skawina A., Litwin J. A., Gorczyca J., Miodoński A. J. The vascular system of human fetal long bones: a scanning electron microscope study of corrosion casts. J Anat. 1994 Oct;185(Pt 2):369–376. [PMC free article] [PubMed] [Google Scholar]
- Trias A., Fery A. Cortical circulation of long bones. J Bone Joint Surg Am. 1979 Oct;61(7):1052–1059. [PubMed] [Google Scholar]
- de Saint-Georges L., Miller S. C. The microcirculation of bone and marrow in the diaphysis of the rat hemopoietic long bones. Anat Rec. 1992 Jun;233(2):169–177. doi: 10.1002/ar.1092330202. [DOI] [PubMed] [Google Scholar]