Infectious diseases caused by pathogenic bacteria have been the leading cause of morbidity and mortality in human history. The discovery of the antibacterial compound from the mold Penicillium notatum by Alexander Fleming led to the development of antibiotics, which are still the main weapons for combating the deadly bacterial infections at the present time. However, over 60 years of application of antibiotics leads to the development of antibiotic resistance of many bacterial pathogens. Consequently, bacterial infections have again become the most common and deadly causes of human diseases. Two of the most lethal hospital infections are caused by methicillin-resistant Staphylococcus aureus (MRSA) and vancomycin-resistant enterococci (VRE) (10). Furthermore, Streptococcus mutans-associated tooth decay is one of the most prevalent and costly infectious diseases in the United States (4, 11; http://www.surgeongeneral.gov/library/oralhealth/).
The emergence of both “new pathogens” and resistant strains from “old pathogens” demands new antibacterial compounds to deal with this crisis. Given the fact that most commonly used antibiotics are isolated from microorganisms, it is important to search for new antibacterial compounds from new bio-resources. Chinese medicinal herbs are logical choices due to their proven ability to treat microbial infections. In traditional Chinese medicine (TCM) practice, a group of herbs has been widely used for a specific therapeutic application defined as Qing Re Jie Du (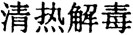 ), or “alleviating heat and relieving the symptoms caused by toxins” (2, 3). Many herbs in this category have been found to have antimicrobial activities (2, 3). Recently, we conducted a systematic screen of herbs with Qing Re Jie Du function for the inhibitory activities against S. mutans, the primary etiological agent for dental caries and other pathogens (1), and found that the extract made from Sophora flavescens contains a potent bioactivity against S. mutans, MRSA, and VRE.
), or “alleviating heat and relieving the symptoms caused by toxins” (2, 3). Many herbs in this category have been found to have antimicrobial activities (2, 3). Recently, we conducted a systematic screen of herbs with Qing Re Jie Du function for the inhibitory activities against S. mutans, the primary etiological agent for dental caries and other pathogens (1), and found that the extract made from Sophora flavescens contains a potent bioactivity against S. mutans, MRSA, and VRE.
S. flavescens is a perennial shrub found in Northeast Asia (Fig. 1A). It grows in sandy soils on mountain slopes or river valleys. In spring or autumn, the roots are collected, cleaned, sliced, and air-dried. The processed root of S. flavescens is also known as “Ku Shen,” which means “a precious medicinal root with bitter taste” (Fig. 1B). In more than 1,000 years of TCM practice, it has being used to treat pyretic and analgesic symptoms (http://www.itmonline.org/arts/sophora.htm). Although a variety of bioactive compounds have been recently isolated from S. flavescens for the treatment of inflammation, cancer, and cardiovascular disorders (5, 13), the knowledge about its antibacterial potential is limited (7).
FIG. 1.
Sophora flavescens (A) and its dried roots (Ku Shen) (B) used in TCM application.
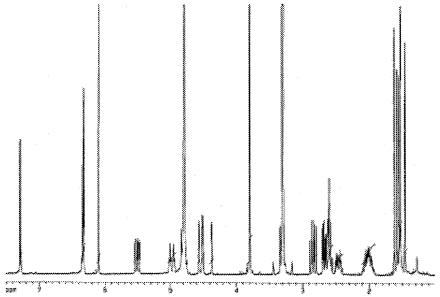
To study the active antimicrobial component(s) in S. flavescens, the following procedures were followed. First, an extract of S. flavescens was prepared according to a previously published extraction method (1), and its MIC against S. mutans was determined with a protocol recommended by the National Committee of Clinical Laboratory Standards (NCCLS) (9) (Table 1). Following the bioassay, the extract was then chromatographed over silica gel (100- to 200-mesh; Selecto, Georgia), and eluted with a hexane:ethyl gradient acetate. Equal volumes of the eluted solutions were collected, dried by evaporation, and subjected to the antimicrobial assay to track down the most active fraction(s). Following the bioassay, the chemical composition of active fractions was then analyzed by thin-layer chromatography and high-performance liquid chromatography (HPLC). The result indicated that the most active fraction contains ∼80% of the active compound. Further purification was performed on semipreparative HPLC equipment (600E system controller and 996 photodiode array detector; Waters, Milford, MA) equipped with a reverse-phase C18 column (7.8 by 300 mm). The homogeneity of the purified active compound (>95%) was established by both HPLC profiles (data not shown) and subsequent 1H nuclear magnetic resonance (NMR) spectra (500 MHz) (Fig. 2).
TABLE 1.
Susceptibility of S. mutans, MRSA, and VRE to antimicrobial compounds and kurarinone
| Drug | MICa(μg/ml) of drug for:
|
||
|---|---|---|---|
| MRSAb | VREb | S. mutansc | |
| Ampicillin | 250 | 250 | 0.15 |
| Vancomycin | 2.5 | 150 | 1 |
| Extract of S. flavescens | 16 | 16 | 16 |
| Kurarinone | 2 | 2 | 2 |
MICs were determined according to the NCCLS recommended protocol.
Clinical isolates of MRSA and VRE were provided by Dr. William H. Benjamin of University of Alabama, Birmingham.
S. mutans strain ATCC 25175 was used in the assay.
FIG. 2.
Proton-NMR spectrum of purified kurarinone.
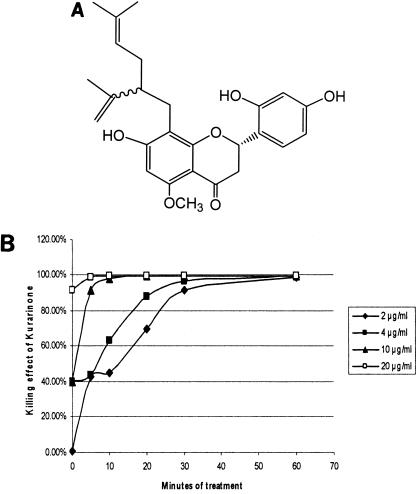
The purified compound was then analyzed by mass spectrometer and NMR spectroscopy (1H and 13C), respectively. Based on the extensive one- and two-dimensional (1D and 2D)NMR spectroscopic interpretation and mass spectrometer analysis (data not shown), we concluded that the isolated active compound is kurarinone, a known compound isolated from S. flavescens (5, 7, 8, 13). Kurarinone is a flavonoid with a lavandulyl side chain, and its chemical structure is illustrated in Fig. 3.
FIG. 3.
Kurarinone (A) and its time- and dose-dependent bactericidal effect against S. mutans (B). The chemical structure of kurarinone was elucidated on the basis of extensive 1D and 2D NMR spectroscopic interpretation. The killing effect of kurarinone was determined by counting the colony formation units on the plates after the cells were treated with the conditions illustrated in panel B.
Several bioactivities of kurarinone have been previously reported, including antifungal activities against Candida albicans and Cladosporium cucumerinum(12), antimalarial activity (6), cytotoxic activity against human tumor cells (myeloid leukemia HL-60 cells) (5), and COX-1 inhibitory activity (5). At the current stage, the molecular mechanisms of its action in both pathogens and mammalian cells are largely unknown. In this study, a strong antibacterial activity of kurarinone was detected. Its MICs against S. mutans and multidrug-resistant strains (MRSA and VRE) are at the same level (2 μg/ml) (Table 1). Time- and dose-dependent bactericidal effects of kurarinone against S. mutans (Fig. 3B), MRSA, and VRE (data not shown) were also detected. These data suggested a potential application of kurarinone for the treatment of diseases or conditions associated with S. mutans, MRSA, and VRE.
Acknowledgments
This study was supported by a grant from C3 Scientific Corporation and Biostar to W. Shi, NIH grant AT00151 to D. Heber, and a New Investigator Award from NIH to L. Chen.
We thank Dr. William H. Benjamin for providing MRSA and VRE strains used in this study. We express our appreciation and gratitude to Dr. Fengxia Qi, Dr. Jian He, and Charles Xiao Fong Zhou for their suggestions and comments on the manuscript. We also thank Dr. Fang Gu and Dr. Larry Wolinsky for their helpful discussions.
REFERENCES
- 1.Chen, L., L. Ma, N. H. Park, and W. Shi. 2001. Cariogenic actinomyces identified with a β-glucosidase-dependent green color reaction to Gardenia jasminoides extract. J. Clin. Microbiol. 39:3009-3012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen, Q. 1998. Pharmacology and clinical applications of classical TCM formulas. People's Publisher, Beijing, People's Republic of China.
- 3.Huang, T. K. 1994. A handbook of the composition and pharmacology of common Chinese drugs. China Medical Science and Technology, Beijing, People's Republic of China.
- 4.Johnson, N. W. 1991. Risk markers for oral diseases. 1. Dental Caries. Cambridge University Press, Cambridge, England.
- 5.Kang, T. H., S. J. Jeong, W. G. Ko, N. Y. Kim, B. H. Lee, M. Inagaki, T. Miyamoto, R. Higuchi, and Y. C. Kim. 2000. Cytotoxic lavandulyl flavanones from Sophora flavescens. J. Nat. Prod. 63:680-681. [DOI] [PubMed] [Google Scholar]
- 6.Kim, Y. C., H. S. Kim, Y. Wataya, D. H. Sohn, T. H. Kang, M. S. Kim, Y. M. Kim, G. M. Lee, J. D. Chang, and H. Park. 2004. Antimalarial activity of lavandulyl flavanones isolated from the roots of Sophora flavescens. Biol. Pharm. Bull. 27:748-750. [DOI] [PubMed] [Google Scholar]
- 7.Kuroyanagi, M., T. Arakawa, Y. Hirayama, and T. Hayashi. 1999. Antibacterial and antiandrogen flavonoids from Sophora flavescens. J. Nat. Prod. 62:1595-1599. [DOI] [PubMed] [Google Scholar]
- 8.Lee, H. O., N. K. Park, S. I. Jeong, Y. C. Kim, and S. H. Baek. 2001. Isolation of antimicrobial compounds from the ethyl acetate extract of Sophora flavescens. Yakhak Hoe Chi 45:588-590. [Google Scholar]
- 9.NCCLS. 2003. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard, 6th ed. NCCLS, Wanye, Pa.
- 10.Russell, A. D. 2002. Antibiotic and biocide resistance in bacteria: introduction. J. Appl. Microbiol. 92(Suppl.):1S-3S. [PubMed] [Google Scholar]
- 11.Satcher, D. 2000. Oral health in America: a report of the surgeon general-executive summary. U.S. Department of Health and Human Services, Washington, D.C.
- 12.Tan, R. X., J. L. Wolfender, L. X. Zhang, W. G. Ma, N. Fuzzati, A. Marston, and K. Hostettmann. 1996. Acyl secoiridoids and antifungal constituents from Gentiana macrophylla. Phytochemistry 42:1305-1313. [DOI] [PubMed] [Google Scholar]
- 13.Zheng, Y., J. Yao, X. Shao, and N. Muralee. 1999. Bio-active compounds from Sophora flavescens Ait. Nongyaoxue Xuebao. 1:91-93. [Google Scholar]